Abstract
Because of the size of the nanoparticles, their detection and exact anatomical localization in tissue samples are very difficult. Consequently, suitable methods are needed to prove their presence, especially co-localized to histological lesions. Therefore, the aim of this study was to investigate whether nanoparticles were detectable in specimens after reprocessing samples from glass slides using the pop-off technique. Tissue slides containing agglomerates of titanium dioxide nanoparticles already visible on a light microscopic level and amorphous silicium dioxide (SiO2) particles not observable in tissue slides were reprocessed. Furthermore, cytospots of bronchoalveolar lavage acquired from rats that previously inhaled carbon nanotubes were used. After reprocessing the samples, they were investigated using transmission electron microscopy. In all the reprocessed samples, the respective nanoparticles were detectable. Even the light microscopically invisible amorphous SiO2 particles were observed as electron dense structures. Titanium and silicium were additionally confirmed in the respective nanoparticles by energy-dispersive X-ray spectroscopy (EDX). In summary, the pop-off technique represents a fast and easy way to detect nanoparticles in histological sections. This enables further characterization of these particles by additional techniques such as EDX, and their direct correlation with light microscopic lesions at exactly the same location is investigated.
Keywords
Introduction
Nanoparticles are widely used in industries and their amount is increasing every year (Oberdörster, Oberdörster, and Oberdörster 2005; Oberdörster, Stone, and Donaldson 2007; Rivera Gil et al. 2010). Because of the minute size of nanoparticles, their detection and exact anatomical localization in tissue samples lead to certain difficulties, especially because the amount of translocated nanoparticles may be quite low. Analytical methods may detect the element but an exact anatomical localization within the tissue or even the cell compartment is not possible (Wang et al. 2008). Within light microscopic samples, only agglomerated nanoparticles are visible; single particles are not detected using this method (Van Ravenzwaay et al. 2009). Even single particles can be seen ultrastructurally, but investigating the sample amount is tremendously laborious and expensive because often only small amounts of nanoparticles could be observed (Geiser et al. 2005). However, methods already exist to enable reevaluation of formalin-fixed, paraffin-embedded tissue (Lighezan et al. 2009; Widéhn and Kindblom 1988). In addition, hematoxylin and eosin (H&E)-stained slides have been reprocessed successfully for electron microscopy using a special method called pop-off technique (Bergmann and Kinder 1987; Ngai et al. 1985). This pop-off technique was used to identify the intralesional infectious organisms like virus particles or protozoal structures (Bergmann 1991; Bretschneider, Burns, and Morrison 1981; Kleeman 2002; Rossi, Luginbühl, and Probst 1970; Wohlsein et al. 2011). Furthermore, the investigation of ultrastructural phenotype of cells allows identifying the cell origin, for example, to further characterize tumor cells (Peters et al. 2003).
This study is aimed to test the applicability of the pop-off technique on glass slides to demonstrate the presence of nanoparticles intralesionally in areas where they are not detectable by light microscopy. In addition, the possibility of further characterizing these particles morphologically or using energy-dispersive X-ray spectroscopy (EDX) needs to be addressed.
Materials and Methods
Three different specimens were used for the present investigation. The first specimens served as positive controls and consisted of H&E-stained sections from the lung of rats that have previously inhaled titanium dioxide (TiO2) nanoparticles (JRC Nanomaterials Repository NM-105; Evonik Degussa GmbH, Essen, Germany) and clearly showed nanoparticle agglomerates on the light microscopy level (Eydner et al. 2012). The second specimens were acquired from the larynx of the same animals. In these tissues, the nanoparticles were only suspected in the epithelial lesions. Furthermore, also sections of rats that previously inhaled amorphous silicium dioxide (SiO2; JRC Nanomaterials Repository NM-200) were used. The latter consisted of cytospots from 3 bronchoalveolar lavages (BALs) of rats previously treated with carbon nanotubes to further test whether the pop-off technique can also be used on BALs. All animal experiments were conducted in compliance with the law of animal welfare of Lower-Saxony, Germany (#33.9-42502-04-07/1340).
The formalin-fixed and paraffin-embedded tissue samples were prepared for light microscopic examination. Slices of 5 µm thickness were cut and stained with H&E. Areas of interest were marked with a xyline-resistant pen on the backside of the slide. Similarly, the BALs were May-Grünwald stained and areas of interest were marked accordingly.
Electron Microscopy, Pop-Off Technique
Initially, the cover slips of the H&E-stained slides were removed by incubating in xylene for 1 day or up to 7 days, depending on the time postcovering. Thereafter, all slides including the BALs were rinsed twice with 100% ethanol for 5 min each followed by incubation for 2 min in propylene oxide and 100% ethanol (1:1). Afterward, samples were coated with pure propylene oxide and immediately covered with propylene oxide and epoxy resin (1:1) for 20 min. A gelatin capsule measuring 0.56 cm in diameter, filled with epoxy resin was placed upside down on top of the marked area (Figure 1). The epoxy resin was polymerized for 1 hr at 35°C followed by 1 hr at 45°C and overnight at 85°C.
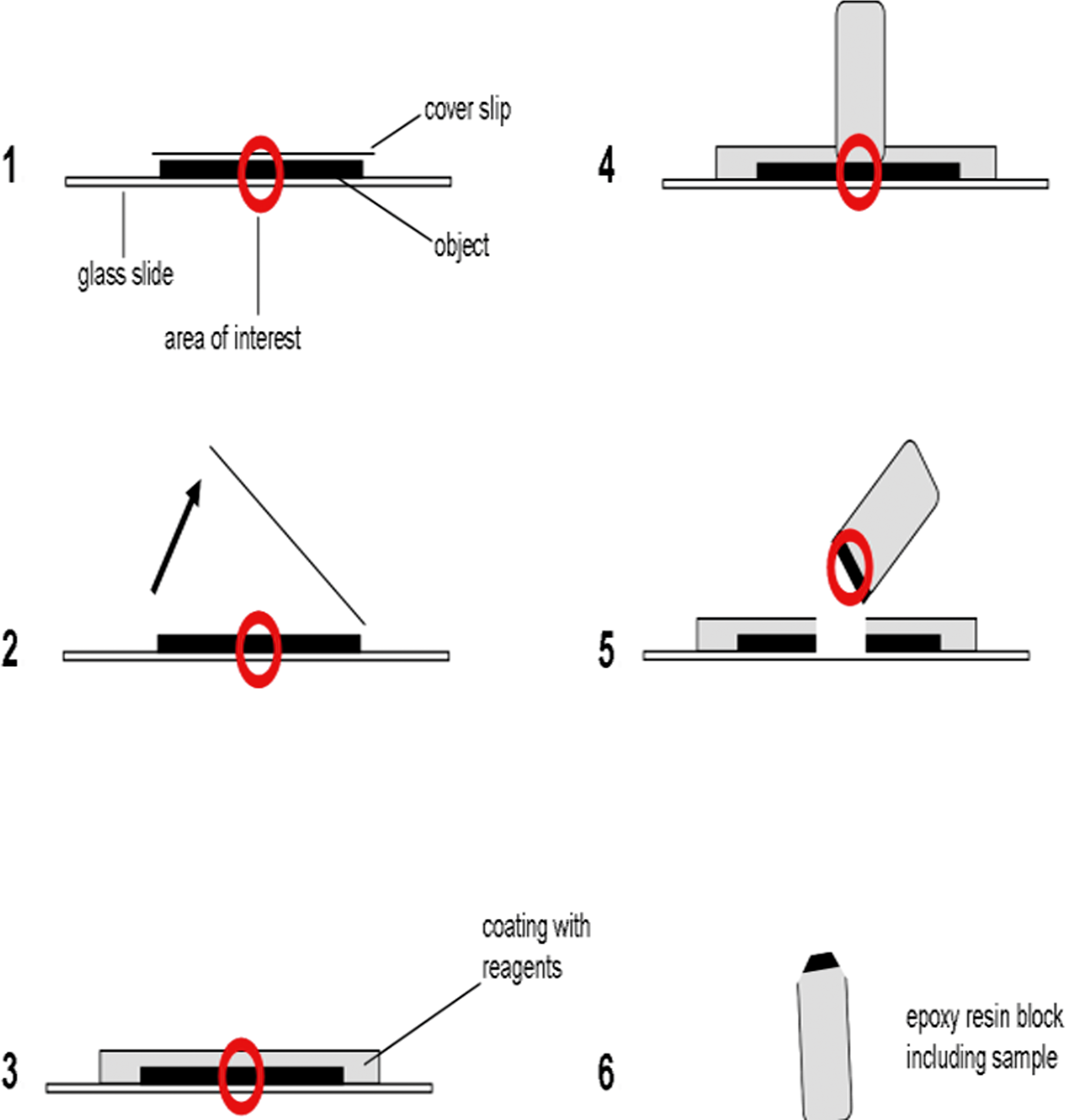
Schematic illustration of the pop-off technique steps. (1) Marking the area of interest. (2) Removal of cover slip. (3) Coating of the slide with a sequence of different reagents. (4) Placing the galantine capsule filled with epoxy resin on the marked area. (5) Removing the Epon block after polymerization using liquid nitrogen. (6) Trimming of the area of interest for the preparation of ultrathin sections. Red circle: area of interest.
After polymerization, the epoxy resin blocks were removed from the glass slide by dipping in liquid nitrogen. Thereafter, the areas of interest in the blocks were trimmed under visible control for ultrasectioning. The 70-nm thick ultrathin sections were prepared and mounted on 200 mesh copper grids. Afterward, samples were investigated using a transmission electron microscopy (LEO 910; Zeiss, Oberkochen, Germany).
Furthermore, electron dense structures in the specimens of animals previously treated with TiO2 and SiO2, respectively, were additionally investigated using an energy-dispersive X-ray spectroscope (Ametek, Wiesbaden, Germany) to further proof the intralesional presence of nanoparticles.
Results
Light Microscopy
The H&E-stained sections of the first specimens consisting of lung tissue of rats that previously inhaled nanosized TiO2 showed fine granular, brown to golden intracytoplasmatic particles within the intra-alveolar macrophages under the light microscope (Figure 2A). In addition, these particles were found to be freely distributed within the alveoli and also adjacent to the alveolar epithelium.
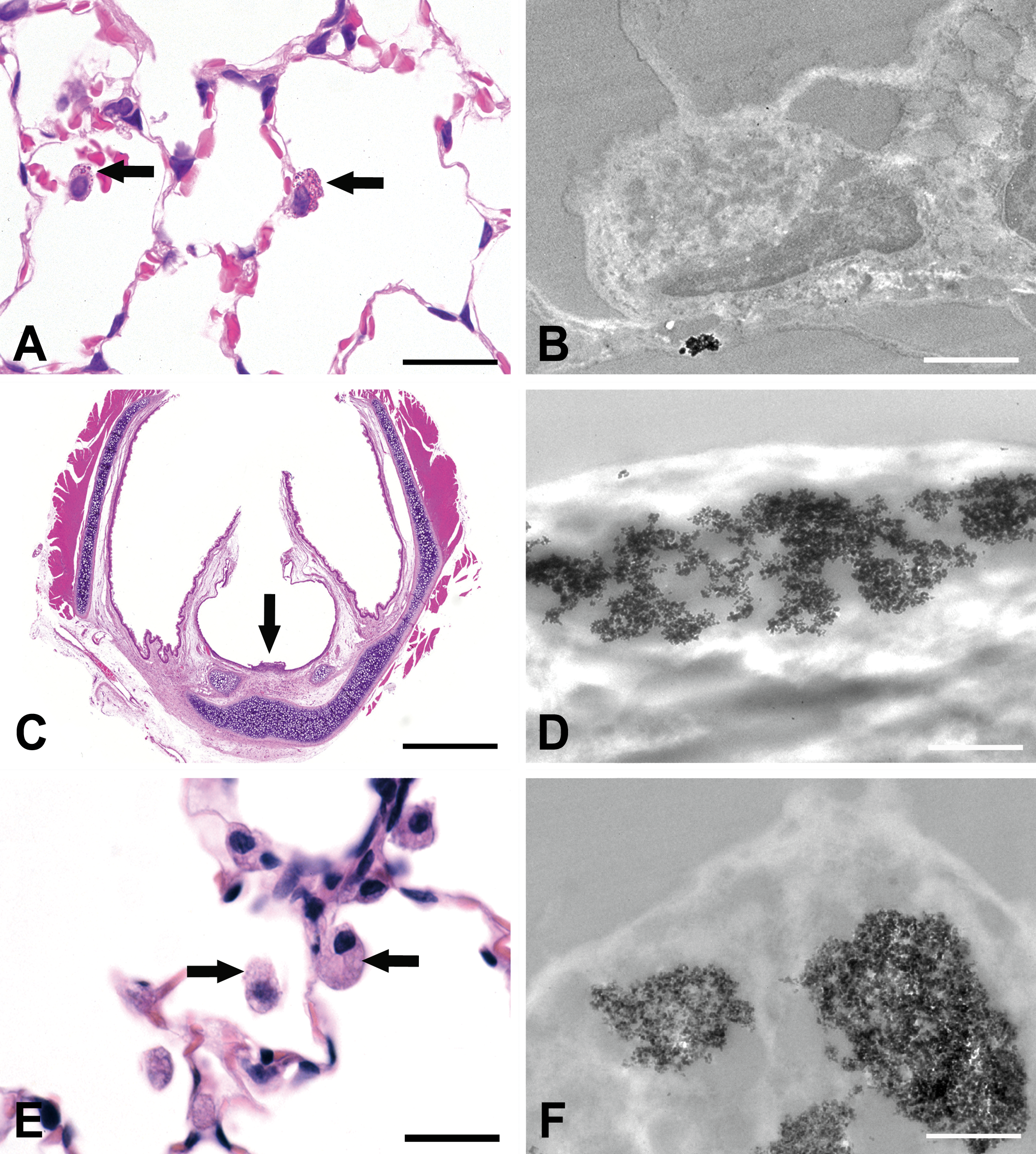
The left column shows the light microscopic images (hematoxylin and eosin staining) and the right column the electron microscopic findings. (A) Intrahistiocytic, golden-appearing TiO2 nanoparticles are visible (arrows), bar = 25 µm. (B) Showing the same particles on ultrastructural level as moderately electron-dense, and agglomerated particles, bar = 1.5 µm. (C) Larynx of a rat after inhalation of TiO2 nanoparticles displays a focal ulceration (arrow) and no visible nanoparticles, bar = 250 µm. (D) Intralesional-agglomerated TiO2 nanoparticles within the laryngeal ulceration, bar = 1 µm. (E) No particles are visible under the light microscope in lungs after inhalation of SiO2 nanoparticles (arrows), bar = 25 µm. (F) Intrahistiocytic-agglomerated, moderately electron dense SiO2 nanoparticles can be seen within alveolar macrophages, bar = 1 µm. TiO2 = titanium dioxide; SiO2 = silicium dioxide.
The larynx of the same animals contained epithelial ulcerations and epithelial alterations located at the ventral part at the transition zone from the squamous to the respiratory epithelium. Within the necrotic tissue areas, in addition to multinucleated giant cells, macrophages, and few neutrophils, structures with similar morphologic features of a fine granular, golden shining particles similar to that found in the lungs were identified (Figure 2C).
The samples of lungs after inhalation of SiO2 only showed an increased amount of alveolar macrophages with a finely granular, slightly eosinophilic to foamy or vacuolated cytoplasm, without clear identifiable nanoparticles (Figure 2E).
Light microscopic examination of the BAL samples revealed macrophages with large foamy to vacuolated cytoplasm containing black, curled carbon nanotubes (Figure 3A).
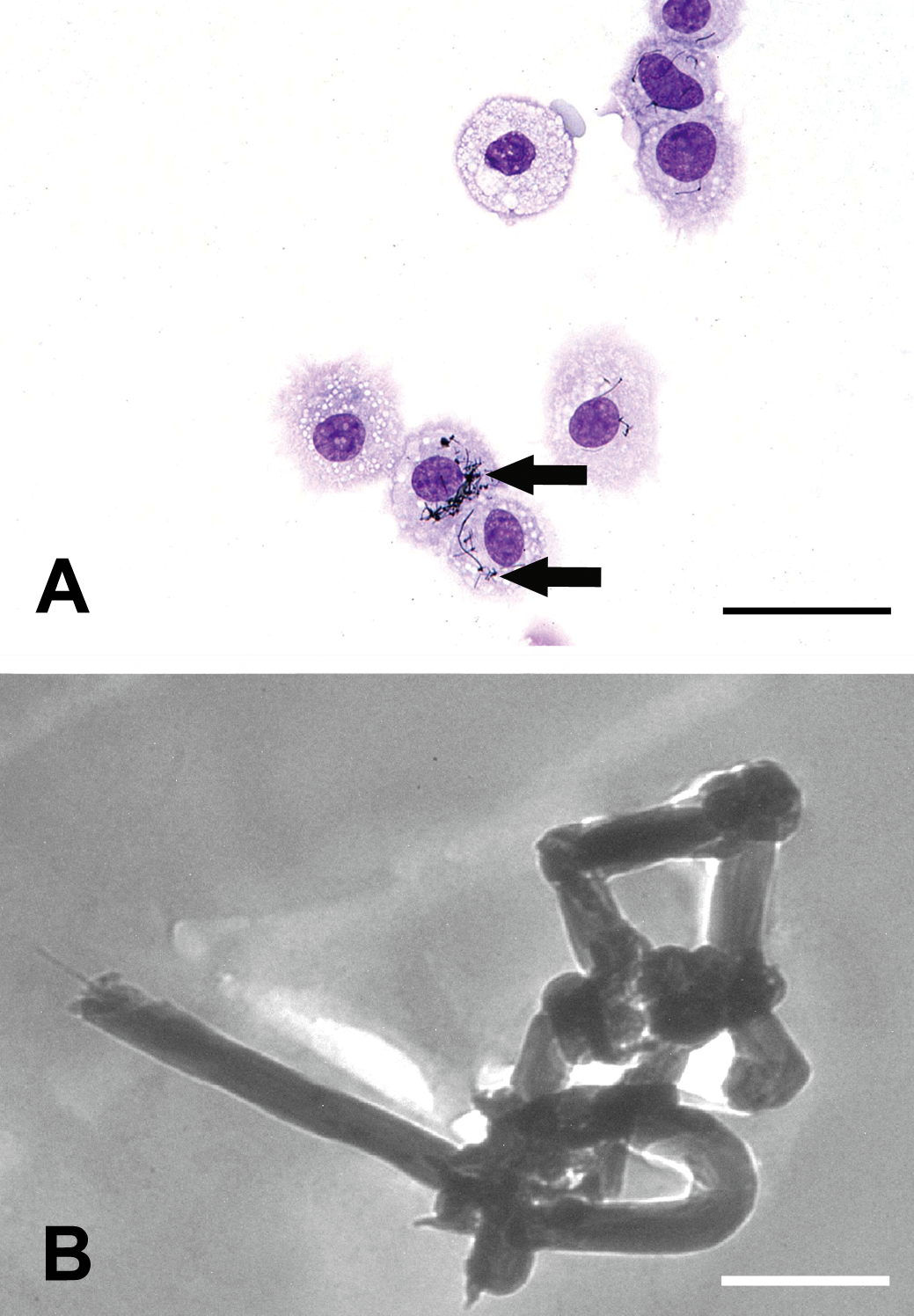
(A) May-Grünwald staining. A sample of a bronchioalveolar lavage after inhalation of carbon nanotubes (arrows) displays cytoplasm of alveolar macrophages, bar = 25 µm. (B) Electron microscopic findings, the carbon nanotubes are observed at the ultrastructural level showing curled structure and their electron density, bar = 500 nm.
Electron Microscopy
Within selected areas of paraffin-embedded, H&E-stained sections, cytospots of BAL fluid nanoparticles and nanofibers were detected intracellularly within macrophages and extracellularly within necrotic tissue. In the lung samples, TiO2 nanoparticles were present within the cytoplasm of alveolar macrophages as it was already observed using light microscopy. The nanoparticles within the cytoplasm of alveolar macrophages appeared as moderately electron dense, irregularly shaped, and round to spheroidal. The primary particles measured about 22 nm in diameter and agglomerated up to 1 µm in diameter (Figure 2B). TiO2 nanoparticles were detected within the center of the ulceration (Figure 2D) and occasionally within the cytoplasm of macrophages within the larynx. They had the same morphologic appearance as that found in the lungs.
The alveolar macrophages with a foamy to vacuolated appearance as observed after the inhalation of SiO2 nanoparticles with no visible nanoparticles on the light microscopic level revealed intracytoplasmic, moderately electron-dense, irregular shapes, agglomerated nanoparticles by electron microscopy. The diameters of the primary particles were about 20 nm, and the agglomerates measured up to 1 µm in diameter (Figure 2F).
Carbon nanotubes were seen within the cytoplasm of cells within the BAL fluid, which were suspected to be macrophages because of their cell morphology. They appeared as moderately electron dense, spherical, and curled structures measuring up to 100 nm in width and a few micrometers in length (Figure 3B).
Nanoparticles were easily detected when no contrast enhancement such as uranyl acetate and lead citrate was applied. However, cellular structures in ultrathin sections were easily recognized with contrast enhancement.
Energy-Dispersive X-Ray Spectroscopy
To verify the detection of the mentioned TiO2 and SiO2 particles, an EDX was used. Electron-dense structures showed a characteristic peak at 4.51 and 4.93 keV (Figure 4A) as well as at 1.7 keV (Figure 4B), correlating with the K-α and K-β shells of titanium and K-α shell of silicon, respectively. Thus, the detection of titanium and silicon was confirmed.
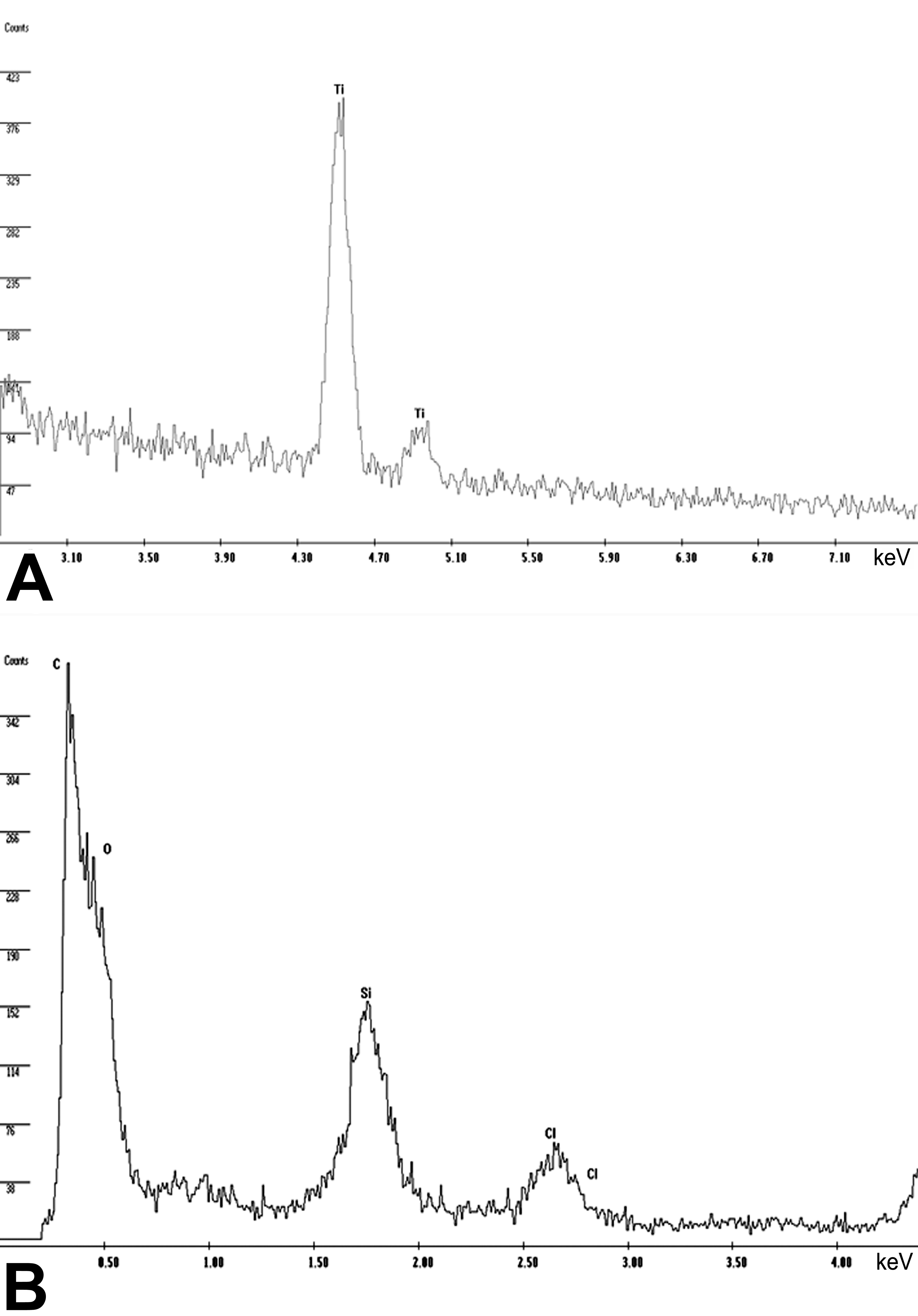
Two spectra of the samples investigated acquired by energy-dispersive X-ray spectroscopy. (A) The spectrum for titanium (Ti) at 4.51 and 4.93 keV within the lung sample displayed in Figure 2B. (B) The spectrum for silicium (Si) at 1.7 keV in the lung tissue shown in Figure 2F as well as other biological background elements such as carbon (C), oxygen (O), and chlorine (Cl).
Discussion
Nanoparticles are of great concern because of their increasing use, therefore, increasing the risk of human exposure. In the past decades, nanoparticles have been associated with respiratory or cardiovascular diseases (Andujar et al. 2011; Mills et al. 2009), and neurodegenerative disorders are also discussed (EPA 2004; Rückerl et al. 2007; Song et al. 2011; Wichmann et al. 2000). As these particles are so small in size, their detection in biological samples is difficult (Geiser et al. 2005; Wang et al. 2008), thereby requiring suitable methods to detect their presence or absence within the tissue samples. Chemical tissue analysis such as mass spectrometry only demonstrates the elements’ presence intralesionally and not the presence of nanoparticles as such (Wang et al. 2008). The pop-off technique for transmission electron microscopy, which was formerly used to investigate the presence of intralesional infectious agents like protozoa or viruses (Bergmann 1991; Kleeman 2002), seems to provide a suitable and rapid method for the identification of intralesional nanoparticles. Our results show that nanoparticles can be easily detected within different biological samples such as larynx or lung tissue as well as within cytospot preparations of BAL fluid. Lung-agglomerated nanoparticles in alveolar macrophages could readily be observed using the light microscope, and they were also observed under the transmission electron microscope. Also within the laryngeal tissue, agglomerated TiO2 nanoparticles were observed, which were only a suspicion under a light microscope. After inhalation of amorphous nano SiO2, no nanoparticles were detectable within the lungs under the light microscope. Only an increased amount of alveolar macrophages with a foamy to vacuolated cytoplasm was visible. The cytospot preparations of the BAL fluid revealed the presence of intracellular carbon nanotubes that were also observed under electron microscope. So the direct pop-off technique using transmission electron microscopy represents a suitable method to detect the nanoparticles in light microscopic preparations. Certain factors limit the ability to identify nanoparticles in tissue sections using light microscopy. Many nanoparticles were developed as structural materials and as such they are relatively unreactive to conventional biologic stains (Hubbs et al. 2011). Furthermore, the dimensions of the nanoparticles are frequently less than the visible wavelengths of light, which further hampers the detection of nanoparticles (Hubbs et al. 2011). The presented technique can surmount these difficulties. Moreover, it is possible to correlate the detection of nanomaterial with previously observed histological lesions. In addition, the particles can further be characterized by EDX methods. The disadvantage of this technique is a loss of morphologic quality due to the delayed fixation in formalin. Buffered formalin leads to better morphologic results than nonbuffered fixations (Kleeman 2002). Ngai et al. (1985) also demonstrated the formation of numerous clefts within the nuclei and cytoplasm due to the heat damage caused by the use of warm water floating bath. To overcome this problem, they invented a free-floating method. In the present study, we did not observe the clefts, in spite of using the warm water floating bath. Nanoparticles are not affected by delayed fixation. Although an exact localization of the particles in cellular subcompartments might be difficult, it is still possible to detect the particles as such.
In summary, the pop-off technique represents a fast and easy way to detect nanoparticles in histological sections and allows their further characterization by additional techniques such as EDX and by directly correlating light microscopic and transmission electron microscopic findings at exactly the same location.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
