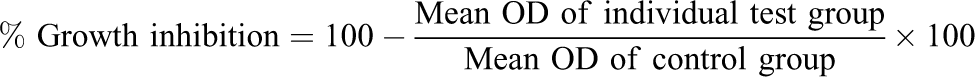Abstract
As a bone mineral component, hydroxyapatite (HA) has been an attractive bioceramic for the reconstruction of hard tissues. However, its poor mechanical properties, including low fracture toughness and tensile strength, have been a substantial challenge to the application of HA for the replacement of load-bearing and/or large bone defects. In this study, HA is reinforced with high-purity and well-functionalized multiwalled carbon nanotubes (MWCNTs; >99 wt%) having an average diameter of 15 nm and length from 10 to 20 μm. The cellular response of these functionalized CNTs and its composites were examined in human osteoblast sarcoma cell lines. Calcium nitrate tetrahydrate (Ca(NO3)2·4H2O) and diammonium hydrogen phosphate ((NH4)2HPO4) were used to synthesize HA in situ. MWCNTs were functionalized by heating at 100°C in 3:1 ratio of sulfuric acid and nitric acid for 60 min with stirring and dispersed in sodium dodecyl benzene sulfonate by sonication. HA particles were produced in MWCNTs solution by adding Ca(NO3)2·4H2O and (NH4)2HPO4 under vigorously stirring conditions. The composite was dried and washed in distilled water followed by heat treatment at 250°C to obtain CNT-HA powder. Physiochemical characterization of the composite material was carried out using Fourier transform infrared spectroscopy, field-emission scanning electron microscopy, energy-dispersive X-ray spectrometer, and X-ray diffractometer. Furthermore, this study investigates the cytotoxic effects of functionalized-MWCNTs (f-MWCNTs) and its composites with HA in human osteoblast sarcoma cell lines. Human osteoblast cells were exposed with different concentrations of f-MWCNTs and its composite with HA. The interactions of f-MWCNT and MWCNT-HA composites were analyzed by 3-(4,5–dimethyl thiazol-2-yl)-2,5-diphenyl tetrazolium bromide assay. The results indicate no detrimental effect on survival or mitochondrial activity of the osteoblast cells. Cell viability decreased with an increase in CNT concentration indicating that MWCNTs and its composite can be cytotoxic at higher dosages. This result provides further evidence that the bionano interface can be developed for CNT-reinforced HA composites for load-bearing bone implants, drug delivery, and tissue engineering.
Keywords
Introduction
Since the discovery of carbon nanotubes (CNTs) in 1991, 1 they have become the synonymous of nanotechnology because of their unique structural, mechanical, and electronic properties. 2 Several promising biomedical applications have also been anticipated, such as drug design, drug delivery, 3 –5 tumor therapy, 6,7 tissue engineering, 8 DNA recognition, 9,10 and biosensor design. 11
But issue of toxicity of CNTs is still controversial. Inhibition of human embryonic kidney (HEK 293) cell proliferation after exposure to single-walled CNTs (SWCNTs) was reported by Cui et al. 12 Ding et al. observed that multiwalled CNTs (MWCNTs) induce cell cycle arrest and increase apoptosis/necrosis of human skin fibroblasts. 13 However, neither of these studies used functionalized CNTs. Apoptosis of T lymphocytes induced by oxidized MWCNTs were observed by Bottini et al.. 14 However, simple oxidation is not enough to render CNTs soluble and stable in saline and cell media and thus does not represent a biocompatible functionalization. Sayes et al. further reported that the toxicity of CNTs depends on the density of functionalization, for example, phenyl-SO3 X groups that were heavily functionalized with high density had the minimal toxicity. 15 These results infer that CNTs without proper functionalization have a highly hydrophobic surface, thereby they aggregate in the cell culture and interact with cells by binding to various biological species, including proteins, via hydrophobic interactions and induce certain cell responses such as cell toxicity.
Other factors may also contribute to the observed toxicity of CNT samples in vitro. Surfactants, present in excess in the CNT suspensions, are known to be highly toxic to cells. 16 The metal catalyst content in CNTs should also be considered when the toxicity of CNTs is investigated. 17 Moreover, proper assays must be employed in toxicity tests to avoid interference of CNTs with the assay reagents. 18,19 For these reasons, in vitro toxicity assays of CNTs should be carefully designed and performed with well-prepared and characterized materials as well as suitable assay methods.
Methods
Materials
The MWCNTs samples were purchased from Nanoshel LLC (Wilmington, Delaware, USA). The nanotube has a specified diameter of 20–30 nm. Purity is greater than 98%. Analytical grade of calcium nitrate tetrahydrate (Ca(NO3)2·4H2O) and diammonium hydrogen phosphate ((NH4)2HPO4) were purchased from HiMedia Laboratories (Mumbai, Maharashtra, India) with a molecular weight of 164.15 and 132.06, respectively. Ammonia solution (25%) was purchased from Merck. 3-(4,5-dimethyl thiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT), fetal bovine serum (FBS), phosphate-buffered saline (PBS), Dulbecco’s modified Eagle’s medium (DMEM), and trypsin were obtained from Sigma Aldrich Co. (St Louis, Missouri, USA). Ethylenediaminetetraacetic acid (EDTA), glucose, and antibiotics were obtained from HiMedia Laboratories Ltd. Dimethyl sulfoxide (DMSO) and propanol were purchased from Merck Ltd (Mumbai, Maharashtra, India).
Functionalization of MWCNTs
According to our previously published protocol, 20 the MWCNTs (100 mg/60 ml) were heated at 100°C in 3:1 ratio of 20% sulfuric acid (H2SO4) and 20% nitric acid (HNO3) for 60 min with stirring. These treated MWCNTs were washed until neutral pH and dried at 60°C for further use.
Biosynthesis of HA
The HA were synthesized in situ by sol–gel method. First, 5 ml of 0.5 M Ca(NO3)2·4H2O is dissolved in distilled water, and pH were adjusted to ≥10; then, 5 ml of absolute ethanol is added to the solution. Second, 10 ml of 0.5 M (NH4)2HPO4 is prepared in distilled water, and the pH was adjusted to ≥10 and rapidly added to the first prepared 0.5 M Ca(NO3)2·4H2O solution under vigorously stirring conditions. The pH of the mixed solutions was kept ≥10 by adding ammonia solution. Stirring was continued for 3 h, followed by aging for 24 h at room temperature. The gel was dried at 60°C overnight and repeatedly washed with distilled water followed by heating at 250°C in a muffled furnace for 30 min to obtain HA powder.
Inclusion of MWCNTs in HA matrix
Functionalized MWCNTs (f-MWCNTs) were dispersed in sodium dodecyl benzene sulfonate (SDBS) by sonication. Hydroxyapatite (HA) particles were produced in the above MWCNTs solution by adding 0.5 M Ca(NO3)2·4H2O and 0.5 M (NH4)2HPO4 under vigorous stirring conditions. 21 The pH of the solution was kept ≥10 by adding ammonia solution. Stirring was continued for 3 h followed by aging for 24 h at room temperature. The composite was dried at 60°C followed by washing in distilled water for 3 to 4 times. Heat treatment at 250°C was carried out for 30 min to obtain CNT-HA powder.
Physiochemical characterization of MWCNT and MWCNT-HA composites
FTIR spectroscopy
Fourier transform infrared (FTIR) spectra of specimen powders were recorded on a Shimadzu Prestige IR spectrometer (Shimadzu Prestige 21 series, Kyoto, Japan). Prior to analysis potassium bromide (KBr) pellets were prepared by mixing in the ratio of 1:10 of sample/KBr (w/w), followed by uniaxially pressing the powder under vacuum. All spectra were obtained between 4000 and 400 cm−1 at a resolution of 2 cm−1.
Surface topography characterization
The surface morphology and atomic structure of the HA scaffolds and HA/MWCNT scaffolds were examined under the field-emission scanning electron microscopy (FESEM; Hitachi SU6600, Japan) and energy-dispersive spectroscopy (EDS; Horiba- EMAX, Japan). Prior to the microstructure analysis, specimens were coated with gold using an ion sputter coating instrument (Hitachi E-1010, Japan) with a current set at 15 mA for a coating time of 15 s.
XRD analysis
X-Ray diffractometer (XRD; MiniFlex II; Rigaku Corporation, Tokyo, Japan) utilizing copper Cu K α radiation was used to investigate the crystallinity and phase content of the MWCNT and MWCNT-HA scaffolds on powder. Data were collected from 10° to 50° 2θ values, with a step size of 0.02° and a counting time of 2 s per step.
Cell preparation
Saos (human osteoblast sarcoma cell line) was procured from National Centre for Cell Sciences (NCCS), Pune, Maharashtra, India. Stock cells were cultured in DMEM supplemented with 10% inactivated FBS, penicillin (100 IU ml−1), streptomycin (100 μg ml−1), and amphotericin B (5 μg ml−1) in a humidified atmosphere of 5% carbon dioxide (CO2) at 37°C until confluent. The cells were dissociated with trypsin phosphate versene glucose solution (0.2% trypsin, 0.02% EDTA, and 0.05% glucose in PBS). The stock cultures were grown in 25 cm 2 culture flasks, and all experiments were carried out in 96 microtiter plates (Tarsons India Pvt. Ltd., Kolkata, West Bengal, India).
Preparation of test solutions
For cytotoxicity studies, each weighed f-MWCNT and MWCNT-HA composites were separately dispersed in distilled DMSO, and the volume was made up with DMEM supplemented with 2% inactivated FBS to obtain a stock solution of 1 mg ml−1 concentration and sterilized by filtration. Serial twofold dilutions were prepared from this for carrying out cytotoxic studies.
MTT assay
The monolayer cell culture was trypsinized, and the cell count was adjusted to 1.0 × 10
5
cells ml−1 using DMEM containing 10% FBS. To each well of the 96-well microtiter plate, 0.1 ml of the diluted cell suspension (approximately 10,000 cells) was added. After 24 h, when a partial monolayer was formed, the supernatant was flicked off, washed the monolayer once with medium, and 100 μl of different test concentrations of f-MWCNT and MWCNT-HA composites were added on to the partial monolayer in microtiter plates. The plates were then incubated at 37°C for 3 days in 5% CO2 atmosphere, and microscopic examination was carried out and observations were noted every 24 h interval. After 72 h, the compound solutions in the wells were discarded and 50 μl of MTT in PBS was added to each well. The plates were gently shaken and incubated for 3 h at 37°C in 5% CO2 atmosphere. The supernatant was removed, then 100 μl of propanol was added, and the plates were gently shaken to solubilize the formed formazan. The absorbance was measured using a microplate reader at a wavelength of 540 nm. The percentage growth inhibition was calculated using the following formula, and the concentration of test drug needed to inhibit cell growth by 50% (CTC50) values is generated from the dose–response curves for each cell line.
Statistical analysis
Results were compared using one-way analysis of variance (ANOVA) testing using all pairwise multiple comparison procedures. Values were reported as standard error of the mean. The values of p < 0.001 were considered to be statistically significant.
Results
FTIR analysis
FTIR spectroscopy was used to characterize and understand the molecular vibration/structure of the MWCNT-HA composites; Figure 1 shows the FTIR spectra MWCNT-HA powder. The FTIR spectra of the MWCNT-HA composites were detected with intense absorption bands at 590 and 550 cm−1 and 950–1090 cm−1, which are assigned to phosphate groups with the frequency of vibration ν4 and ν3, respectively. The bands observed at 3400–3430 cm−1 corresponds to the stretching mode of –OH group, which is the characteristic of calcium phosphates such as HA.
22
A weak band of

FTIR patterns showing the formation of HA calcined at 250°C of (a) pure HA; (b) in situ-synthesized HA; (c) MWCNT-HA composites; and (d) f-MWCNT. FTIR: Fourier transform infrared; HA: hydroxyapatite; MWCNT: multiwalled carbon nanotubes; f-MWCNT: functionalized multiwalled carbon nanotubes.
Surface topography characterization
According to Coulomb’s law, the electrostatic forces are inversely proportional to the dielectric constant of the medium. The electrostatic forces between the ions in HA crystals are higher when organic solvents are used for synthesis. Longer interaction between the ions in organic solvents leads to faster reaction and nucleation rate. Hence, the smaller particles with regular morphology are synthesized Figure 2(c) and (f). These HA nanoparticles are densely decorated on MWCNTs successfully by a simple and effective in situ synthetic method Figure 2(b) and (e). Figure 2(a) and (d) shows the f-MWCNT at low and high magnification. The EDS results confirmed the peak of the elements such as calcium (Ca), phosphorus (P), and carbon (C) in the respective samples, though some impurities like iron (Fe) also exist in f-MWCNTs sample (introduced as catalyst during the synthesis of CNTs). The EDS elemental microanalysis (wt%) is listed in Table 1. From the data, we could see the MWCNT-HA sample has C as the major element with 96.97 wt%, while Ca and P are present in 2.23 and 0.80 wt%, respectively.

SEM image of (a, d) f-MWCNTs; (b, e) MWCNT-HA composites; and (c, f ) in situ-synthesized HA powder. Images (a, b, and c) are taken at low magnification (×60). Images (d and e) are taken at higher magnification (×50k), while image (f ) at ×5k. Comparison of (d) with (e) shows that the sidewall of the MWCNT was rougher after sintering. The scale bar of (a, b, and c) is 500 μm; (d, e) is 1 μm; and (f ) is 10 μm. SEM: scanning electron microscopy; HA: hydroxyapatite; MWCNT: multiwalled carbon nanotubes; f-MWCNT: functionalized multiwalled carbon nanotubes.
EDS elemental microanalysis (wt%) of f-MWCNT, HA, and MWCNT-HA composites.
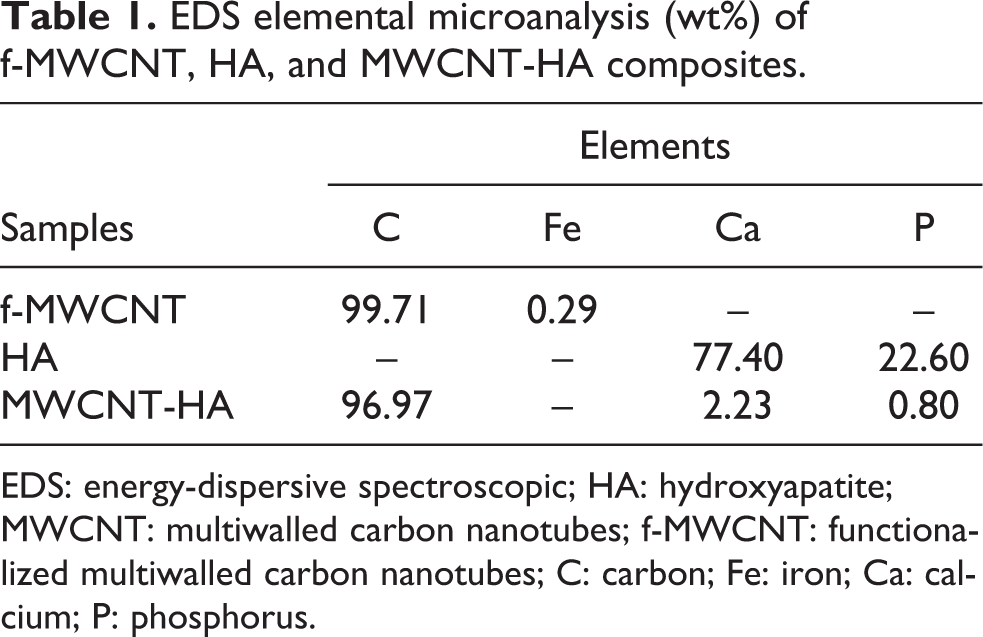
EDS: energy-dispersive spectroscopic; HA: hydroxyapatite; MWCNT: multiwalled carbon nanotubes; f-MWCNT: functionalized multiwalled carbon nanotubes; C: carbon; Fe: iron; Ca: calcium; P: phosphorus.
XRD analysis
The XRD was used to investigate the crystallinity and phase content of inorganic layer of the HA-MWCNT scaffolds. Figure 3 shows the XRD pattern of the composite (on powder) after sintering at 250°C for 30 min. The XRD patterns of the synthesized MWNT-HA (Figure 4) were obtained by powder XRD. No other crystalline peak is observed except for the diffraction peaks attributed to the MWCNTs and HA in the synthesized composite.

X-Ray diffraction spectrum of f-MWCNT, in situ-synthesized HA, and MWCNT-HA composite depicting the HA peak. HA: hydroxyapatite; MWCNT: multiwalled carbon nanotubes; f-MWCNT: functionalized multiwalled carbon nanotubes.

Dose–response relationship of covalently functionalized MWCNT and MWCNT-HA composites. Results are presented from exposure of the composites with different concentrations to Saos cell lines. Individual test groups are significantly different from the control group (by ANOVA p < 0.001 followed by Tukey’s HSD test). HA: hydroxyapatite; MWCNT: multiwalled carbon nanotubes; ANOVA: analysis of variance; HSD: honestly significant difference.
MTT assay
The ability of the cells to survive a toxic insult has been the basis of most cytotoxicity assays. This assay is based on the assumption that dead cells or their products do not reduce tetrazolium. The assay depends both on the number of cells present and on the mitochondrial activity per cell. The principle involved is the cleavage of tetrazolium salt MTT into a blue-colored product (formazan) by mitochondrial enzyme succinate dehydrogenase. The number of cells was found to be proportional to the extent of formazan production by the cells used. 26 Figure 4 shows the dose–response relationship of covalently f-MWCNTs samples as well as the MWCNT-HA composites with CTC50 value of >1000 µg ml−1.
Discussion
Determining the toxicity of CNTs has been one of the most persistent questions in nanotechnology. Results from various scientific tests on cells have so far proven confusing, with some results indicating it to be highly toxic and others showing no signs of toxicity.
Several toxicity mechanisms have been proposed for CNTs including interruption of trans membrane electron transfer, disruption/penetration of the cell envelope, oxidation of cell components, and production of secondary products such as dissolved heavy metal ions or reactive oxygen species. 27 –29 Toxicity of a CNT sample is dependent on its composition along with its geometry and surface functionalization. Several studies have suggested that well-functionalized CNTs are safe to animal cells, while raw CNTs or CNTs without functionalization show severe toxicity to animal or human cells at even moderate dosage. 30,31
Many groups have successfully used well-functionalized nanotubes for in vitro cellular uptake experiments without observing apparent toxicity. 32 –41 Cells exposed to SWCNTs, PEGylated by various photoluminescence–polyethylene glycol amphiphiles, exhibited neither enhanced apoptosis/neurosis nor reduced proliferation of various cell lines in vitro. 34,35,42 CNTs covalently functionalized by 1,3-dipolar cycloaddition developed by Prato and coworkers also appeared to be safe to the tested cell lines, including primary immune cells. 36,37 CNTs with a biomimetic coating engineered by Bertozzi and coworkers were also nontoxic to cells. 43,44 Several other independent groups also reported that CNTs coated by DNA, amphiphilic helical peptides, and serum proteins were nontoxic to cells. 41,45,46 Recently, it is discovered that SWNTs taken up by cells via endocytosis exited cells through exocytosis without affecting the viability of cells. 47,48 It appears that raw CNTs and CNTs without functionalization show toxicity to cells at moderate dosage, while functionalized CNTs and coated CNTs show less toxicity even at high dosages.
In this study, Saos cells were exposed to increasing concentration (62.5–1000 µg ml−1) of the compound showing relative increase in cytotoxicity. The rationale of testing these concentrations is that the composite can be used in the replacement of long bone and scaffold for tissue engineering. Cell death (% cytotoxicity) was measured by mean optical density (OD) of individual test group divided by the mean OD of control group. The statistical result analysis from the ANOVA test of control and individual test group revealed that the f-MWCNT and MWCNT-HA composites are nontoxic to Saos cells with CTC50 value of >1000 µg ml−1; those results are statistically significant at p < 0.001, F(0.0143, 0.0001) = 116.94.
The covalently functionalized CNTs become water dispersible but suffer the disadvantages of chemical modification of the original aromatic nanotube structure. Due to this reason, mild oxidation of CNTs were done with 20% of H2SO4 and HNO3. The SEM and FTIR revealed the nanotube structure were intact as well as the tubes were appended with functional groups which makes them dispersible in water/culture media without/least damage. The rationale is functionalized CNTs with the functional group like =CO, –OH, and –COOH that allow them to disperse well in the aqueous solution. By improving the aqueous dispersion, the aggregation of the tubes will reduce in the solution. 43 On the other hand, mild acid treatment of MWCNT leads to oxidation and purification of the CNTs, as well as it retains the original structure, which is obvious from the EDS data presented in Table 1. The f-MWCNTs are >99 wt% pure, while the impurities like Fe is present in very minute (0.29 wt%) concentration. The MWCNT-HA composites were prepared in MWCNTs solution by adding 0.5 M Ca(NO3)2·4H2O and 0.5 M (NH4)2HPO4 under vigorously stirring conditions. Dispersing nanotubes in SDBS (an anionic surfactant) solution impart a negative charge on MWCNTs. The Ca2+ ions then can be adsorbed onto the surface of MWCNTs due to the electrostatic attraction and in situ reaction with PO3− via electrovalent bond to form HA-MWCNT precipitates. The SEM/EDS of the composites revealed the sintering of HA over the MWCNT surface (Figure 2(e)). FTIR results confirm the attachment of CO3 and PO4 groups (Figure 1). XRD results confirm a highly crystalline structure of the composites (Figure 3), which improve the biocompatibility of the nanocomposites with cells in culture. Synthesis of HA particles in the CNT solution, which was prepared in advance, leads to an excellent dispersion of CNTs in HA matrix. On the other hand, presence of CNT in composite of HA-CNT has caused the faster crystallization at lower temperatures compared to pure HA due to heterogeneous nucleation and creation of more diffusion pathways.
As the dispersed composite were added directly on to the confluent layer of Saos cell lines in vitro, the exposure time and the extent of contacts of the material could be the limitations of the presented cytotoxicity study. Hence, further studies on biocompatibility of these materials can be extended to animal model where prolonged contact between MWCNT-HA scaffold and bone cells will occur. However, studies suggest that CNT behavior in vivo also depends on their length, functionalization, and degree of agglutination. 49,50 –52 Up to now, several studies reported low or undetected liver and systemic toxicity in mice, although CNT presence has been shown in the liver, lung, and feces after intravenous injection. 49,51,53
Conclusions
The toxicity of f-MWCNTs and MWCNT-HA composites to the Saos cell lines was measured with MTT salt viability assay in which the metabolically active cells reduces the tetrazolium salt MTT into a blue-colored product (formazan) in the presence of mitochondrial enzyme succinate dehydrogenase. The cells were treated with varying concentrations of f-MWCNTs and MWCNT-HA composites. Both the compounds showed the dose-dependent toxicity to the Saos cell lines (p < 0.001). We conclude with the remark that properly functionalized CNTs with a purity of >99 wt% and the CNT-reinforced HA composites are nontoxic to Saos cell lines. The interaction result shows no detrimental effect on cell survival or mitochondrial activity (CTC50 > 1000 µg ml−1). This result provides further evidence that the bionano interface can be developed for CNT-reinforced HA composites for load-bearing bone implants, drug delivery, and diagnostic applications.
Footnotes
Acknowledgment
The authors thank the Science and Technology Unit, King Abdulaziz University, Jeddah, Kingdom of Saudi Arabia, for providing technical support.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This project was supported by the National Science, Technology and Innovation Plan (NSTIP) strategic technologies program in the Kingdom of Saudi Arabia—Project No. 11-NAN1544-03.