Abstract
Silicone oil droplets have been reported in the eyes of human patients following intravitreous (IVT) injections with several marketed biotherapeutic products. Intravitreous administration of a novel biotherapeutic in a 14-week cynomolgus monkey study using insulin syringes was associated with 2, non-test-article-related phenomena: “vitreous floater/clear sphere” on indirect ophthalmoscopy and intrascleral “foreign material near injection track” on histopathology. Retrospective analysis of 81 other preclinical studies of IVT administration of novel biotherapeutics found a greater frequency of clear spheres in monkey IVT studies using insulin syringes and formulations containing polysorbate. We were able to correlate microscopic findings of clear circular to oval areas in the sclera near the injection track with an energy-dispersive X-ray spectroscopy (EDS) signal for silicon at the same location in the sclera. These observations provide further evidence that silicone lubricant in insulin syringes/needles is the source of clear spheres noted in the vitreous and foreign material noted near the injection track in the sclera. Although considered inert and toxicologically insignificant, silicone deposition within the eye should form part of the risk–benefit equation in a clinical setting.
Keywords
Introduction
Silicone oil droplets have been reported in the eyes of patients following intravitreal (IVT) injections with several marketed anti–vascular endothelial growth factor (VEGF) biotherapeutic products since 2006. 1 The ultrasonographic characteristics of the droplets in patient eyes were consistent with silicone. 1 –4 Human patients often describe these droplets as mobile vitreous floaters with a translucent/iridescent or dust/dot-like appearance. 5 These symptoms are typically transient in nature, not vision threatening, and are trivial—in comparison to the symptoms associated with the disorder for which they are being treated. A very small percentage of patients undergo surgical vitrectomy, with its attendant risks, in an effort to alleviate the floater symptoms. 6 It is important to keep in mind, however, that the amount of silicone oil unintentionally introduced by IVT injection is many orders of magnitude less than that used to treat retinal detachment (approximately 5 mL). Additionally, there are no reports of differences in the degree of inflammation, intraocular pressure, or corneal clarity between eyes with and without small amounts of intravitreous silicone secondary to IVT. 7 This strongly suggests that the clinical risk to patients is minimal and likely limited to the symptoms caused by the vitreous floaters.
Silicone oil droplets observed post IVT administration of anti-VEGF biotherapeutics have been hypothesized to originate from the syringe barrel lubricant in certain batches of syringes used for IVT injection. 2,8 In insulin syringes, silicone lubricant is applied between the plunger and the adjacent barrel. Referred to as “stack-on” or “stacked-in” syringes, the needle is fixed to the barrel and the barrel is siliconized by the manufacturer using a “spray-on” process that uses more silicone oil than for noninsulin luer-lock syringes. 9 In contrast, noninsulin syringes are typically siliconized using a “baked-on” process, which allows for a much thinner layer of silicone oil. 9 Olea et al, 10 using micro-flow imaging, confirmed in vitro that oil droplet–like particles were observed every time a siliconized syringe was used, while no silicone oil droplet–like particles were observed with a silicone-free syringe. Melo et al 11 confirmed in vitro the release of the oil droplets from insulin and noninsulin types of syringes used for IVT injection and went on to demonstrate the silicone oil (polysiloxane) nature of the droplets using Fourier transformation spectroscopy. The same authors also found that the droplet release was triggered by the “flicking” used to eliminate air bubbles during the filling and priming of the syringes. 12
The purpose of this investigation was to determine whether silicone oil droplets are also present in preclinical studies (that is, do animal studies translate to humans), to better characterize some of the parameters associated with the presence of silicone droplets within the vitreous, and to explore the histopathologic and toxicologic significance of these droplets.
In this investigation, the chemical element silicon (Si) was assayed in an effort to detect silicone in the form of polysiloxane. Polysiloxane polymers consist of siloxane, an inorganic Si–oxygen backbone chain with 2 organic groups attached to each elemental Si center. Most common silicone oils consist of linear polydimethylsiloxanes.
Materials and Methods
Association for Assessment and Accreditation of Laboratory Animal Care Accreditation and Animal Welfare Compliance Statement
Covance Laboratories Inc is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. The procedures in animal studies were in compliance with applicable animal welfare acts and were approved by the local Institutional Animal Care and Use Committee.
Monkey Study Outline
A 14-week Good Laboratory Practices study in cynomolgus monkeys (Macaca fascicularis) was performed to assess the safety of a novel biotherapeutic. Group 1 (3 males, 3 females) received 2 injections of 50 μL/dose/eye (100 µL total volume) of vehicle control, whereas group 2 (3 males, 3 females) received a single injection of 50 μL/dose/eye of the test article, constituting the low-dose group. Group 3 (5 males, 5 females) received 2 injections of 50 μL/dose/eye (100 µL total volume) of the same test article, constituting the high-dose group. All animals were dosed every other week for a total of 8 doses. The dosing apparatus (Becton-Dickinson, 3/10-mL insulin syringe with ½-in ultra-fine 30-gauge needle, REF 328431) used to administer the doses was equivalent to those used in human clinical settings. Ophthalmic examinations using slit-lamp biomicroscopy and indirect ophthalmoscopy were performed at frequent intervals including prior to administration of each dose, 2 days and 1 week after each dose, and weekly for a 4-week recovery period after the last dose.
At necropsy, the right eye was collected and immersed in modified Davidson’s fixative for 48 to 96 hours and subsequently stored in 10% neutral-buffered formalin. These eyes were then processed into 3 paraffin blocks with a standard horizontal central section containing the optic disc, optic nerve, macula, and fovea plus a superior and inferior calotte. Three hematoxylin and eosin–stained (H&E) sections, 200 µm apart, were prepared from each block, creating 9 slides per eye for examination.
Retrospective Review of IVT Studies
To further characterize this finding, a retrospective review was done of IVT studies conducted at Covance Laboratories Inc (Covance) over approximately 5 years. Search parameters of draft or finalized reports included species, dosing apparatus, formulation components, presence or absence of “vitreous floater/clear sphere,” and presence or absence of “foreign material near injection track.”
In Vitro Study for Release of Silicone Oil Droplet
An in vitro evaluation was conducted to determine the presence or absence of oil droplets in material expressed from noninsulin and insulin syringes. Balanced salt solution (BSS, formulation A), 0.04% (wt/vol) Tween 80 in BSS (formulation B), and 0.01% (wt/vol) silicone oil in BSS (formulation C, positive control) were the 3 formulations tested. Formulations were vortexed to ensure a homogenous state prior to syringe filling and were held at ambient conditions until used to fill the syringes. Formulations A, B, and C were drawn through a Becton Dickinson 18-gauge needle attached to a 1-mL syringe with a locking luer (noninsulin syringe). The drawing needle was discarded and replaced with a sterile, 30-gauge × ½-in needle for testing. Formulations A and B were drawn through a 0.3-mL insulin syringe with an attached 30-gauge × ½-in needle. Five syringes of each dosing apparatus/formulation were used for testing. Each syringe was primed to allow 50 µL to be expressed and were held under ambient conditions for at least 4 hours. Formulations from 5 syringes in each group were expressed onto separate glass slides (one slide for each syringe) and covered with a coverslip in an attempt to avoid the introduction of air bubbles. Each slide was evaluated for the presence/absence of clear spheres or material that appeared different from the main portion of expressed material.
Energy-Dispersive X-Ray Spectroscopy Methods
For each paraffin tissue block, 3 serial 5 µm formalin-fixed paraffin-embedded (FFPE) sections were generated, sampling ∼15 µm of tissue in total. The first 5 µm section was stained with H&E for histological analysis. The second 5 µm section was mounted on a 25-mm-diameter spectrographically pure carbon tab (Electron Microscopy Sciences 77825-25), deparaffinized in Histo-Clear (National Diagnostics), dehydrated through an upgraded alcohol series, and coated with ∼50 angstrom of spectrographically pure carbon (Denton Desk IV, Denton Vacuum). Analysis was performed with a FEI/Thermo Quanta 650FEG with Bruker X-Flash energy-dispersive X-ray spectroscopy (EDS) detector. Analysis conditions were 15 kV accelerating voltage, ∼15 mm working distance, high vacuum, 0 tilt, secondary electron, and EDS detection simultaneously. Energy-dispersive X-ray spectroscopy was performed on all paraffin tissue blocks regardless of the histological analysis. The last 5 µm section was reserved for future analysis. Histo-Clear was selected as the deparaffinizing agent because of the miscibility of silicone oil in xylene, and Histo-Clear, being a citrus-based agent, was less likely to cause washout of the silicone oil during sample processing.
Results
In the 14-week study conducted in the cynomolgus monkey, 2 non-test article-related phenomena were observed. These were “multifocal fine white vitreous floaters/clear spheres” on ophthalmic examination and “foreign material” in the sclera/episclera on histopathologic examination.
Multiple, variably sized, small clear spheres (droplets) were frequently observed within clusters of smaller multifocal fine white vitreous floaters (Figure 1) at a number of intervals, starting with the ocular exam on day 45 following the fourth IVT dose, in both vehicle- and test article-treated groups in the 14-week study (Table 1), indicating that they were not test article related. However, there was a higher incidence of clear spheres in the highest dose group (Table 1). Their location at the dosing site and appearance as multifocal clear spheres or smaller fine white vitreous floaters were consistent with silicone oil droplets reported in clinical settings in humans. 4,5,7,13,14 The spheres and floaters were not associated with an appreciable inflammatory response on ophthalmic examination, and their multifocal nature suggested that they represented emulsified silicone oil.
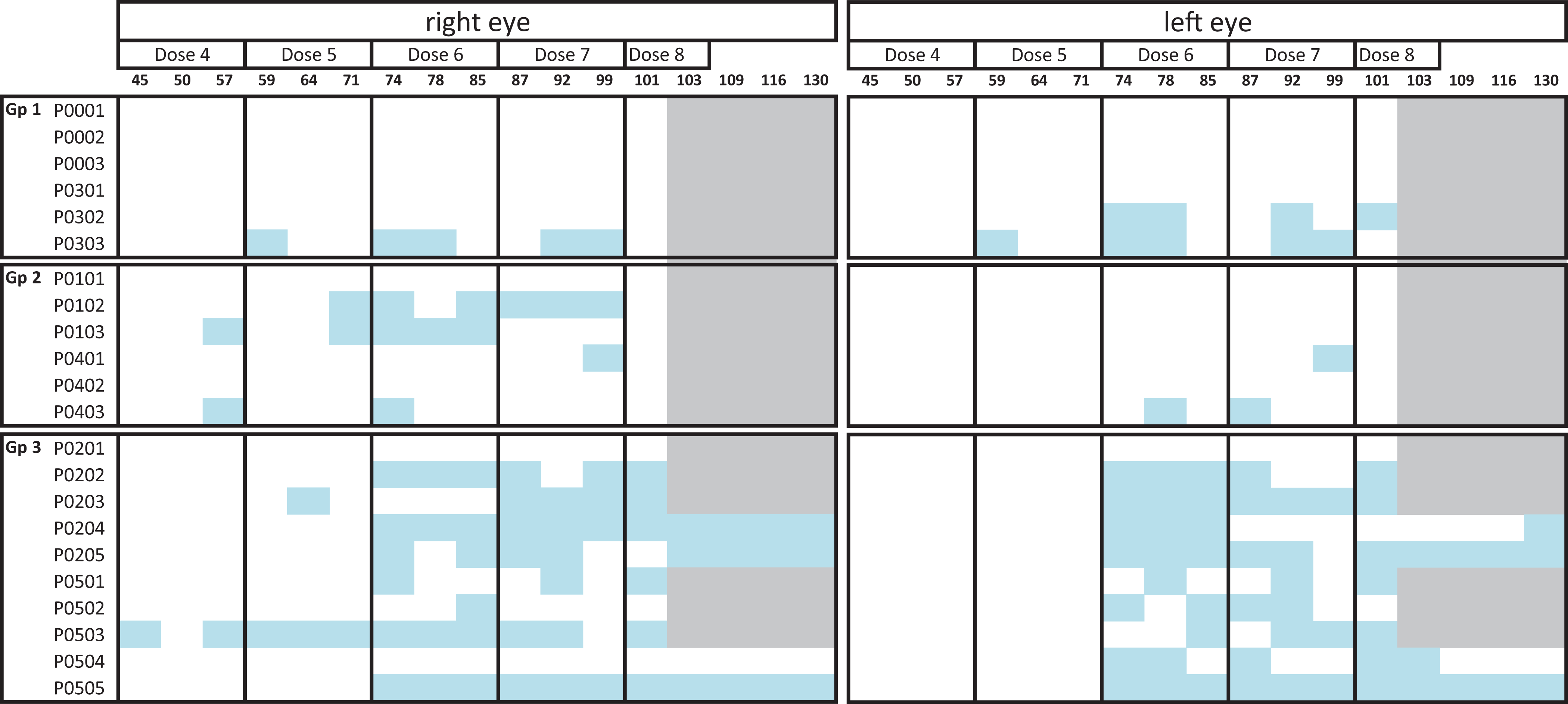
Incidence of “Vitreous Floater/Clear Sphere” by Slit-Lamp Biomicroscopy and Indirect Ophthalmoscopy in a 14-Week Plus 4-Week Recovery IVT GLP Cynomolgus Monkey Study.a
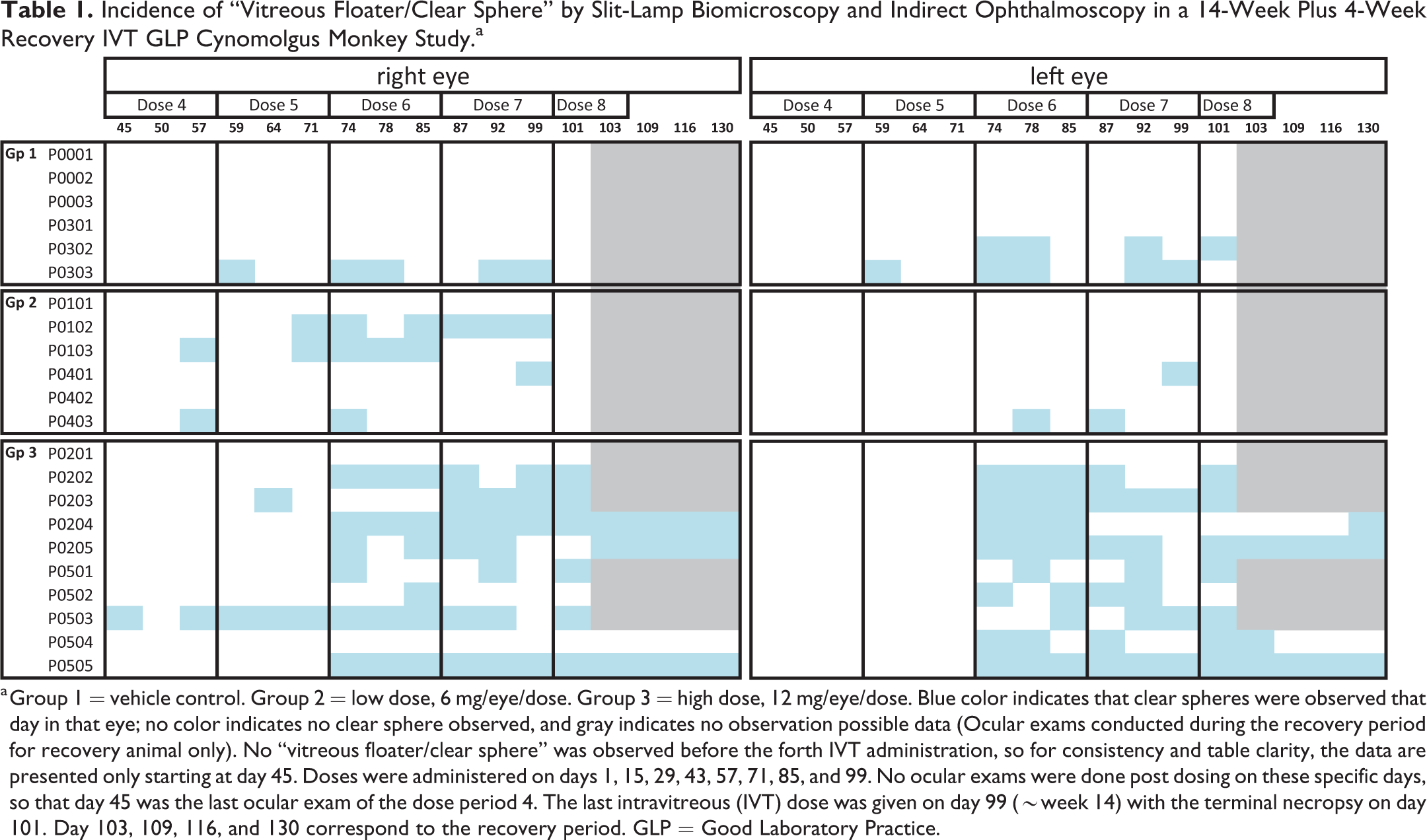
a Group 1 = vehicle control. Group 2 = low dose, 6 mg/eye/dose. Group 3 = high dose, 12 mg/eye/dose. Blue color indicates that clear spheres were observed that day in that eye; no color indicates no clear sphere observed, and gray indicates no observation possible data (Ocular exams conducted during the recovery period for recovery animal only). No “vitreous floater/clear sphere” was observed before the forth IVT administration, so for consistency and table clarity, the data are presented only starting at day 45. Doses were administered on days 1, 15, 29, 43, 57, 71, 85, and 99. No ocular exams were done post dosing on these specific days, so that day 45 was the last ocular exam of the dose period 4. The last intravitreous (IVT) dose was given on day 99 (∼week 14) with the terminal necropsy on day 101. Day 103, 109, 116, and 130 correspond to the recovery period. GLP = Good Laboratory Practice.
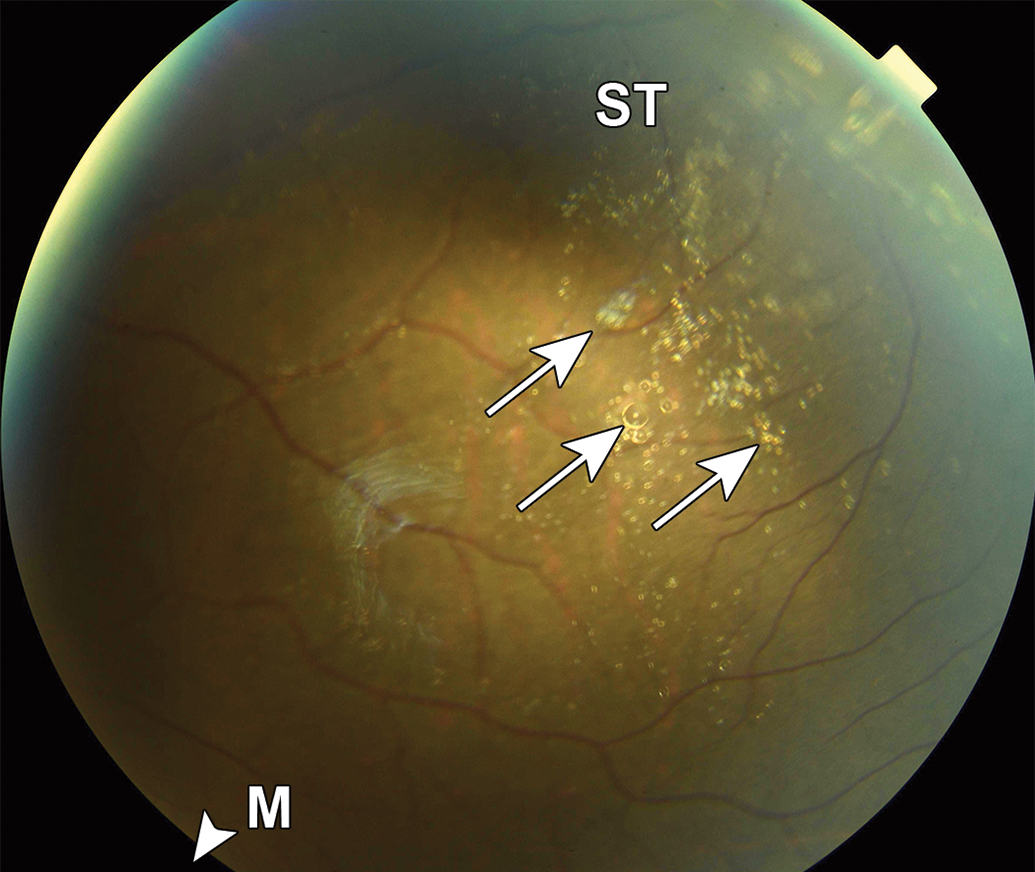
Silicone oil droplets in monkey vitreous from the 14-week study. Representative fundus photograph of a cynomolgus monkey with numerous clear droplets (white arrows) in the superior–temporal vitreous. The macula (M) is outside of the photographic field in the direction of the arrowhead. ST indicates superotemporal.
To get insight into the etiology of these white vitreous floaters, we carried out a retrospective analysis of 81 preclinical IVT studies conducted at Covance—in the past 5 years. In the vast majority of these studies, novel biotherapeutics were administered, noninsulin syringes were utilized, and “clear spheres” were not reported. However, 16 of the 81 IVT studies used insulin syringes (see Table 2). In 1 rabbit study, 2 dog studies, and 1 monkey study using insulin syringes, no “clear spheres” were observed. In the monkey studies where clear spheres were observed, the presence of clear sphere was sometimes made sooner after dosing than in the 14-week monkey study, and the incidence in the control and test article–treated animals was generally equivalent, again indicating that they were not test article related. In many studies, the clear spheres were not visualized at every interval in every eye, likely because they were often located in the far peripheral temporal fundus (near the dosing site) and this area is difficult to thoroughly evaluate in common laboratory species including cynomolgus monkeys due to their deep set eyes and a relatively prominent nose. Clinically, it was believed that the multifocal fine white vitreous floaters represented very small diameter silicone spheres in which the greater reflective radiance from the spheres’ surface prevented visualization of a clear interior. With the larger “floaters,” it was possible to determine that the interior was clear, allowing them to be classified as “clear spheres” in the vitreous. These structures had thicker walls and, as in the 14-week study, appeared identical to those described as silicone oil droplets in humans dosed with a variety of biologics in a clinical setting. 4,5,7,13,14 The affected animals in the historical studies also exhibited no appreciable impact on vision associated with the floaters/spheres (although our abilities to detect such changes in animals is limited), and there was also no evidence that the material was associated with a greater inflammatory response or alteration in intraocular pressure.
Retrospective review of IVT studies conducted at Covance Madison over approximately 5 years.a

aWithin 81 studies, 16 used insulin syringes for dose administration. The study search parameters included animal models, ophthalmic observation parameters (clear sphere, white vitreous floaters), insulin syringe/needle, polysorbate in formulation, and foreign material on histopathologic examinations, and the parameters are presented in rows. “X” indicates that the observation was made and “-” that the observation was not, NA indicates that histopathology was not conducted, “X*” that the observation was uncertain because of concurrent ocular inflammation, and “?” indicates that the complete formulation was not provided.
In all of these historical studies, we also reviewed the incidence of the histopathological finding of “foreign material” (Table 2). However, as histopathological processing was not always done or the histopathological sectioning did not always include the injection track, the observation was considered a proxy for the site of injection and infrequently recorded as a distinct finding; therefore, no definitive conclusions could be drawn with regard to whether such material was exclusively present in studies that used insulin syringes.
Interestingly, formulations containing a polysorbate emulsifier were associated with a higher incidence of intravitreous silicone droplets, although an exact number could not be definitively determined as detailed formulation data were not disclosed for all historical studies.
To test the hypothesis that the polysorbate component of the vehicle may emulsify the silicone oil within the syringe causing very small amounts of silicone to become available for injection into the vitreous, we attempted to determine in vitro if oil droplets could be expressed from noninsulin (tuberculin) and insulin syringes after filling them with BSS or Tween 80 in BSS. In this in vitro study, the material expressed from the majority of syringes (insulin and tuberculin, with or without Tween 80) were observed microscopically to contain lipid droplets and air bubbles, and we could not definitively differentiate between them.
In the 14-week monkey study, histopathologic examination revealed “minimal foreign material near the injection track” in the sclera of the eyes that received either vehicle or the test article (Figures 2 –4), and like the “clear spheres” seen ophthalmologically in the vitreous, the presence of this material was not considered a test article–related finding. No histopathologic findings were noted in the vitreous. The material in the sclera typically occupied a singular or several closely associated circular to oval areas in the sclera, displacing normal connective tissue, with a predilection for the episclera, near episcleral vessels. These areas measured up to approximately 100 µm at maximum diameter were refractile, lacked any clear staining, and had the appearance of “broken glass.” The material did not exhibit any birefringence under polarized light. In some instances, the material was clearly associated with an injection track as evidenced by distortion of tissue along a line within the sclera and ciliary body pars plana, with the extension of connective tissue into the vitreous (Figure 2). It was also noted that there was typically very limited to no microscopic evidence of inflammation associated with the “foreign material” suggesting minimal to no safety concern. If there was inflammation, it was considered most likely a response to needle trauma and not the foreign material per se. The material was also seen in the sclera of recovery animals, at the same site and of similar appearance, suggesting that the material did not “recover” under the conditions of this study.
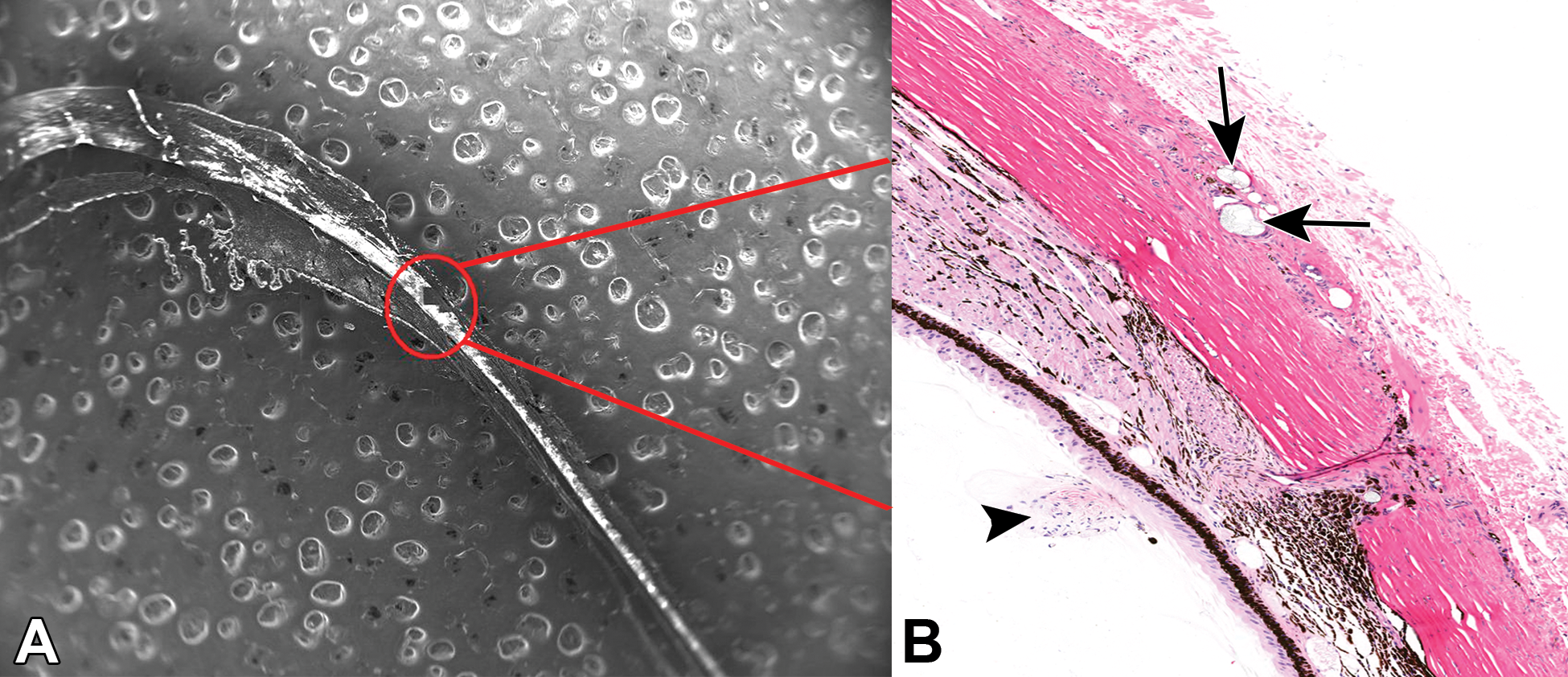
Representative energy-dispersive X-ray spectroscopy (EDS) image (A, original magnification ×18) and corresponding hematoxylin and eosin–stained photomicrograph (B, original objective ×20) from a recovery male animal. The EDS was focused at the IVT injection site (A and B: red circle) within the superotemporal quadrant of the globe, through the sclera and ciliary body pars plana, into the vitreous. A, White “bubble” effect around the red circle and the ocular tissue represent the tissue-mounting block (tab). B, Black arrow indicates injection track with extension of fibrous connective tissue into the vitreous; black arrows indicate the location of foreign material within the sclera/episclera, along the injection track.
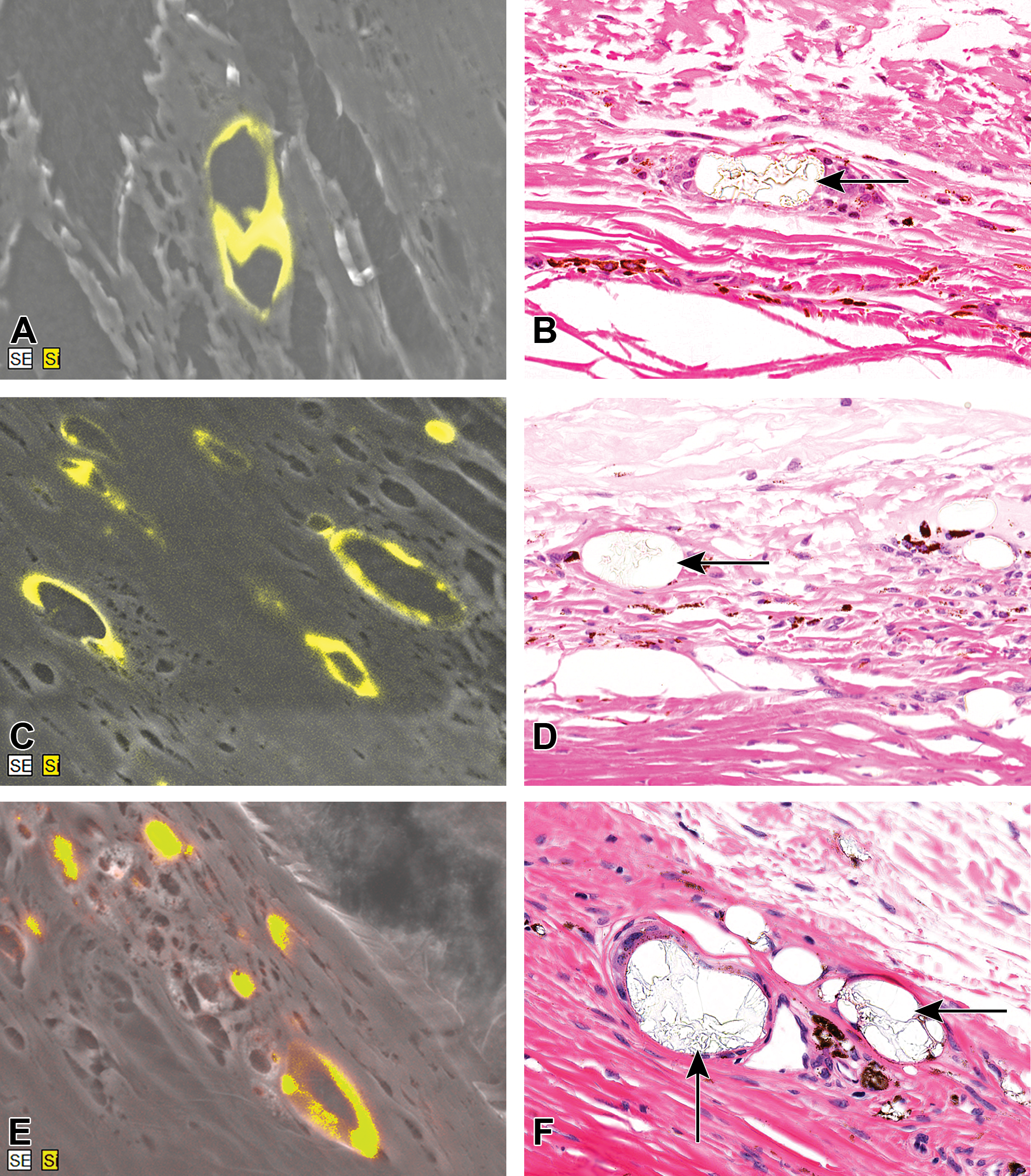
Representative energy-dispersive X-ray spectroscopy (EDS) images. A, Low-dose male, original magnification ∼×2100. C, High-dose female, original magnification ∼×3100. E, High-dose recovery male, original magnification ∼×3500. Corresponding hematoxylin and eosin–stained photomicrographs (B, D, and F, original objective ×40) at the sclera/episclera. The yellow signal within (A), (C), and (E) indicates the position of elemental silicon (Si), typically seen at the circumference of the roughly oval area. The black arrows within (B), (D), and (F) indicate the corresponding location of the clear, refractile scleral/episcleral foreign material, which irregularly spans the roughly oval areas with a refractile, “broken glass”-type appearance. There is no overt cellular/tissue reaction to the material.
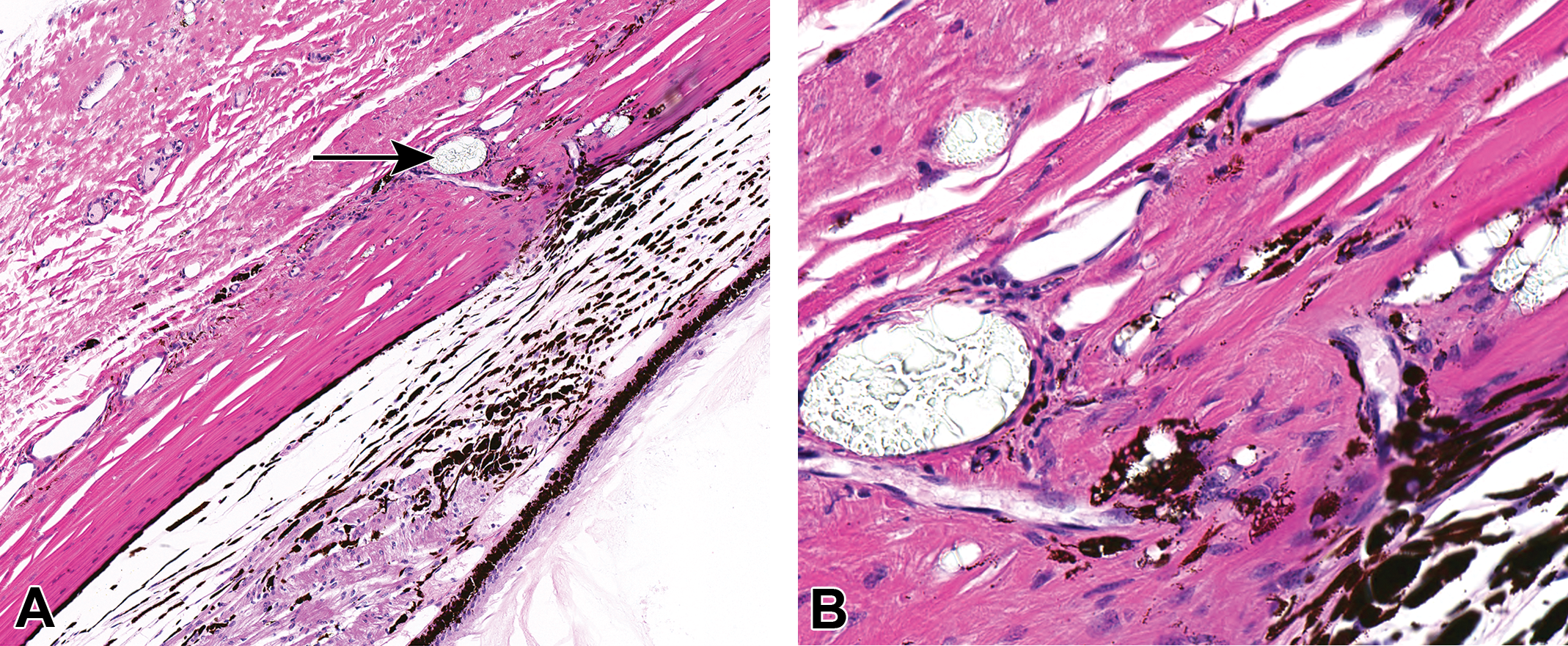
Representative hematoxylin and eosin–stained photomicrographs (A: original objective ×10, B: original objective ×40) from one additional intravitreal cynomolgus monkey study. The higher magnification (B) highlights the clear, refractile, “broken glass” scleral/episcleral foreign material indicated by the black arrow in (A)—control female monkey.
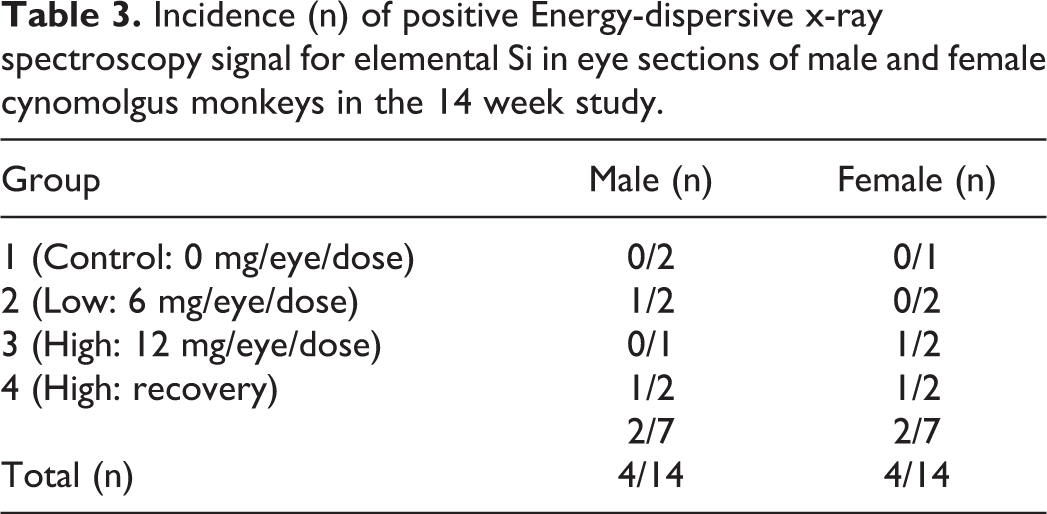
We further investigated the elemental content of the “foreign material” that we saw in the FFPE tissue using an EDS approach. The EDS samples from multiple positions around the globe were taken and we observed a clear signal of elemental Si in the same region as the “foreign material,” only in the area of the sclera where it was noted in the adjacent H&E section. Based on proximity (5 µm between the 2 sections), the EDS signal appeared to be colocalized with the foreign material observed in the H&E sections. A lack of signal in the control group sections (Table 3) was hypothesized to be due to an incidental lack of silicone oil in the particular sections analyzed with EDS and not due to a lack of silicone oil in the “foreign material” observed microscopically in the control group. With the disparate nature of the findings using EDS, multiple sets of serial sections would likely be needed to reliably capture the disposition of “foreign material” using this technique. The Si signal was typically localized circumferentially and was absent from the center of the area in contrast to the refractive light microscopic material which spanned the oval area where it was observed (Figure 3). We hypothesized that the apparent difference in the distribution of silicone oil from the H&E to the EDS was a result of processing artifact. With most silicone-based compounds being miscible in xylene, it is likely that the silicone oil migrated into the surrounding tissues during the creation of the FFPE tissue blocks. The standard FFPE process in the laboratory uses vacuum-assisted embedding (VIP; Sakura, Inc) to ensure complete sample infiltration and thus has a large amount of xylene flowing through the tissue; under this process, there was a likely spread of silicone oil into the edge of the focus and surrounding tissues. The exact chemical nature of the refractile ‘broken glass” material in the center of the foci is uncertain.
Incidence (n) of positive Energy-dispersive x-ray spectroscopy signal for elemental Si in eye sections of male and female cynomolgus monkeys in the 14 week study.
Discussion
The most likely explanation of our observations is that the silicone oil/Si originates from siliconized insulin syringes used for the IVT administration. We hypothesize that the silicone oil was emulsified and released into the vitreous with this syringe type, perhaps with the aid of polysorbate 80. Silicone oil was also possibly locally deposited as “foreign material” in the needle injection track through the sclera, as evidenced by the detection of elemental Si using EDS.
The IVT studies are routinely conducted in the cynomolgus monkey and accepted by regulatory agencies because the anatomy of the monkey eye is very similar to that of the human eye. The location of the silicone oil droplets at the dosing site and appearance as multifocal clear spheres or smaller fine white vitreous floaters is consistent with silicone droplets reported in the human clinical studies 4,5,7,13,14 indicating that this preclinical finding translates from cynomolgus monkeys to humans and that the monkey model can be used to further investigate this phenomenon. Investigation into previous preclinical studies revealed 2 studies conducted in dog and 1 in rabbit that did not support our hypothesis that this phenomenon would translate for all species. However, the number of studies in these species was low and it is difficult to arrive at any definitive conclusion for dogs and rabbits. It is also possible that the dog may not be a good model for investigation of silicone oil droplets given the length of its nose (which makes it difficult to observe the superior temporal peripheral fundus) and the brightly reflective tapetum which reduces the contrast between a sphere and its background.
Historically, we observed identical appearing spheres in several other studies of IVT biotherapeutic agents in cynomolgus monkeys, with the frequencies of clear spheres being comparable in eyes administered vehicle control or a variety of test articles. Interestingly, formulations containing the emulsifier polysorbate (Tween 80) were associated with a higher frequency of intravitreous silicone droplets. We hypothesize that this component of the vehicle may emulsify the silicone oil used to lubricate the syringe barrel, resulting in very small amounts of oil becoming available for injection into the vitreous, especially when using insulin syringes which are more siliconized than the noninsulin syringes. 9 We could not further test this hypothesis in vitro nor could we test whether some biotherapeutic proteins are contributing to the emulsification that could explain a higher frequency of silicone oil droplets with a higher dose of test article. Melo et al have resolved this similar in vitro scoring issue with “blind testers” and picture scoring and conclude that “flicking” the syringe to release the air bubbles from the syringes before the IVT procedure is critical to generate silicone oil droplets. 15 “Flicking” is a common practice during the syringe-filling process used prior to dosing preclinical studies.
One of the most common complications of large amounts of silicone oil in the vitreous in retinal detachment repair is emulsification. Biological solutes such as lipoproteins, cholesterol, and retinol act as intraocular emulsifiers which decrease the surface tension on the spheres facilitating their emulsification. 16 Movement of the eye is also thought to serve as a mild form of agitation further enhancing emulsification. 16 The size of the resulting emulsified oil droplets can be extremely variable. In one study, 65% were 1 to 2 µm in size, with a smaller percentage of droplets being present in the 7 to 30 µm range, which can be appreciated on slit-lamp biomicroscopy or indirect ophthalmoscopy under optimal viewing conditions. 17 This observation, and relative sizes of the droplets, is consistent with the observations in both the 14-week monkey study and other historical animal studies in which multifocal, very fine, white vitreous floaters (presumably spheres so small that their internal structure could not be appreciated on indirect ophthalmoscopy) and larger clear spheres of variable size were observed.
We found elemental Si in the foreign material of the formalin-fixed eye tissue. The location (sclera/episclera) correlated with the injection site, with no similar material seen at any other sites in the globe. Histopathological silicone “bubbles,” very similar to what we report here, have been observed in human ocular tissues at multiple sites after large IVT volumes have been injected in clinical cases of retinal detachtment. 18 The elemental Si is thus most likely coming from silicone oil used to pretreat the injection syringe, but how precisely this silicone oil became embedded in the tissue is still an unresolved question. Funke et al 9 indicated that insulin syringes are siliconized by manufacturers using a “spray-on” process and overall have a higher content of silicone oil than noninsulin syringes. 9 Silicone oil has also been found coated on needles, 6 which is consistent with a silicone coating applied by the manufacturer (videos of coating the needles are available on the internet 19 ) to smooth the movement of the needle insertion into the ocular tissue. In general, silicone oils come with different molecular composition and polymer sizes and the variable release of these compounds based on each process (“sprayed-on” in barrel or “sprayed-on” on needles or “baked-on” in barrel) seems to be possible. Thus, the silicone oil embedded in the sclera could be coming from many different sources with a differing chemical composition based on the syringe manufacturing processing. One cannot exclude that the foreign material in the sclera/episclera and that within the vitreous droplets may slightly have different compositions and/or are derived from different sources. For example, different chemical or polymer compositions of silicone oil may be derived from the syringe barrel or needle. We attempted to use tissue imaging mass spectroscopy to analyze the chemical nature and the molecular signature of “foreign material/silicone material” of the FFPE tissue. While we were able to detect silicone oil reference compounds representative of silicone oils used in both the syringe and barrel, at the expected concentrations of these compounds in these thin tissue sections, the analysis was not successful. To our knowledge, the “broken glass” microscopic morphology was also not indicative of a difference between silicone oils (spray-on vs baked-on) so we could not attribute the exact provenance of the foreign material. However, distinguishing the origin of released silicone oil from either the needle or the syringe barrel would be interesting if the choice of dosing apparatus needs to be modified for a “silicone oil-free” treatment plan. Overall, our clear elemental Si EDS signal strongly suggested that silicone oil can become embedded in ocular tissue after IVT administration in cynomolgus monkeys with no apparent toxicity as previously noted.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320966543 - Observation of Silicone Oil Within the Vitreous and Sclera Following Intravitreal Administration of Biotherapeutics Using Insulin Syringes in Cynomolgus Monkeys
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320966543 for Observation of Silicone Oil Within the Vitreous and Sclera Following Intravitreal Administration of Biotherapeutics Using Insulin Syringes in Cynomolgus Monkeys by François Huet, Jackie Miller, Paul E. Miller, Chris Hayden, James J. Glick, Adam Bentley, Chris De Benedetto, Leslie E. McPherson and Oliver C. Turner in Toxicologic Pathology
Footnotes
Acknowledgments
The authors wish to acknowledge Niraj Tripathi for his work on the in vitro assessment and Evan Streng for his support and coordination of dosing apparatus and syringe-filling techniques.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.