Abstract
Advances in chemistry and engineering have created a new technology, nanotechnology, involving the tiniest known manufactured products. These products have a rapidly increasing market share and appear poised to revolutionize engineering, cosmetics, and medicine. Unfortunately, nanotoxicology, the study of nanoparticulate health effects, lags behind advances in nanotechnology. Over the past decade, existing literature on ultrafine particles and respirable durable fibers has been supplemented by studies of first-generation nanotechnology products. These studies suggest that nanosizing increases the toxicity of many particulates. First, as size decreases, surface area increases, thereby speeding up dissolution of soluble particulates and exposing more of the reactive surface of durable but reactive particulates. Second, nanosizing facilitates movement of particulates across cellular and intracellular barriers. Third, nanosizing allows particulates to interact with, and sometimes even hybridize with, subcellular structures, including in some cases microtubules and DNA. Finally, nanosizing of some particulates, increases pathologic and physiologic responses, including inflammation, fibrosis, allergic responses, genotoxicity, and carcinogenicity, and may alter cardiovascular and lymphatic function. Knowing how the size and physiochemical properties of nanoparticulates affect bioactivity is important in assuring that the exciting new products of nanotechnology are used safely. This review provides an introduction to the pathology and toxicology of nanoparticulates.
Introduction
The recent interest in nanotoxicology and nanoscale particulates (NPs) results from scientific advancements that have improved the capability to synthesize specific particulates in a size range from 1 to 100 nm. The toxicology of some NPs has been studied for a considerable time period as part of the study of ultrafine particles (Oberdörster, Oberdörster, and Oberdörster 2005). Ultrafine particles include NPs that are components of emissions from combustion and dust-generating industrial processes. However, the newly engineered NPs that are products of nanotechnology have changed the abundance, chemical composition, and physical characteristics of very small particulates in potential workplace and environmental exposures.
Nanotechnology is the manipulation of matter on the atomic scale to create structures that can be developed into new products for use in engineering, science, and medicine. Nanotechnology has been revolutionized by improved techniques for synthesis, rapid advances in chemistry and physics at an atomic level, and improved understanding of intracellular structures at the molecular level (Iijima 1991; National Research Council 2006). Within the U.S. government, nanotechnology investments reached an estimated $1.1 billion in 2006 (National Research Council 2006). The estimated market value of nanotechnology products for 2009 is $254 billion, with anticipated growth to $2.5 trillion by 2015 (Bradley, Nordan, and Tassinari 2009). Although some question precise predictions, rapidly increasing patent numbers support the increasing economic impact of nanotechnology (National Research Council 2006). The new engineered NPs include particulates that have never been studied and other particulates that have previously been studied only as components of mixtures. This review is an introduction to nanotoxicology and toxicologic pathology of the first-generation products of nanotechnology.
An important consideration for toxicologic pathologists accustomed to working in the pharmaceutical industry is that regulation of many NPs is very different from regulation of pharmaceuticals. With the exception of some mineral fibers, most nonpharmaceutical particulates are regulated by their chemical composition, not by their size and shape. In workplaces, airborne particulates with a chemical composition not specifically noted in regulations are regulated by the Occupational Safety and Health Administration (OSHA) as particulates not otherwise regulated (PNOR). The OSHA permissible exposure limit (PEL) for an 8-hour time-weight average total PNOR concentration is 15 mg/m3. However, the fraction of those PNOR in the respirable range is less than for total PNOR, so that there is another PEL for PNOR with an aerodynamic diameter of 5 µm or less; that PEL is 5 mg/m3 (Hubbs et al. 2005; OSHA 2006). Thus, the current regulations permit the commercial production and use of most NPs without additional safety testing, using standards developed for larger respirable particulates of the same chemical composition or as PNOR (Murashov et al. 2009). In many cases, toxicologic pathologists will evaluate the pathologic changes caused by NPs in current use in research laboratories or even NPs in widespread industrial use. Ongoing NP exposures in students and workers increase the urgency of the studies.
One concern is that surface area to mass ratio increases as the size of particulates decreases, and the toxicity of particulates often, but not always, correlates with surface area more than mass (Bonner 2007; Brown et al. 2001; Dankovic, Kuempel, and Wheeler 2007; Gonzalez, Smith, and Goodman 1996; Lison et al. 1997; Monteiller et al. 2007; Oberdörster, 1996; Oberdörster, Oberdörster, and Oberdörster 2005; Sager and Castranova 2009; Sager, Kommineni, and Castranova 2008; Tran et al. 2000). As will be described later, some NPs can cross epithelial barriers in the skin and lung, can penetrate flexed skin, can cause lymphangiectasia, may be transported in sensory nerves, and can interact with mitotic spindles (Hubbs et al. 2009, 2008; Oberdörster, Oberdörster, and Oberdörster 2005; D. Porter et al. 2010, 2009; Sargent et al. 2009; Tinkle et al. 2003). Pathologists played a critical role in identifying these changes, but identifying changes caused by NPs requires a thorough and often high-magnification evaluation. The old saying “high power, low brain” is inappropriate when evaluating morphologic alterations caused by particulates with dimensions < 100 nm.
The terminology is relatively easy to understand but not well standardized. The prefix
History and Properties of Nanoscale Particulates
The element carbon is the major component of biological organisms and many familiar items such as coal, graphite, carbon black, and diamonds. Surprisingly, a new structural arrangement of carbon was identified in 1985, where the carbon was organized into pentagons and hexagons to form a hollow sphere comprised of 60 carbon atoms (C60), a structure originally known as buckmisterfullerene (Kroto et al. 1985).
Less than a decade after the discovery of C60, carbon sheets arranged into nanoscale tubes were synthesized (Ebbesen and Ajayan 1992; Iijima 1991). These hollow tubes, known as nanotubes, were formed from one or more walls comprised solely of carbon arranged into multiple hexagons (Bethune et al. 1993; Ebbesen and Ajayan 1992; Iijima 1991; Iijima and Ichihashi 1993). Multi-walled carbon nanotubes (MWCNTs; Figure 1A) have multiple walls, while single-walled carbon nanotubes (SWCNTs; Figure 1B) have one wall (Ajayan, Charlier, and Rinzler 1999). Additional members of this new family of carbon structures, known as fullerenes, were subsequently discovered. A common feature of the fullerenes was pi-bonding of carbon atoms causing a planar organization into carbon balls, capsules, and tubes (Adams et al. 1992). Each carbon atom was attached to three other carbon atoms using a combination of single and double bonds, but a difference between fullerenes and most organic compounds was the absence of hydrogen or other chemical groups (Nielsen et al. 2008; Taylor and Walton 1993). A variety of methods for synthesizing carbon nanotubes have been developed, and the different techniques, such as the use of metal catalysts, can affect the composition of the final product, including the presence or absence of metals in the nanotubes (Bonner 2010).
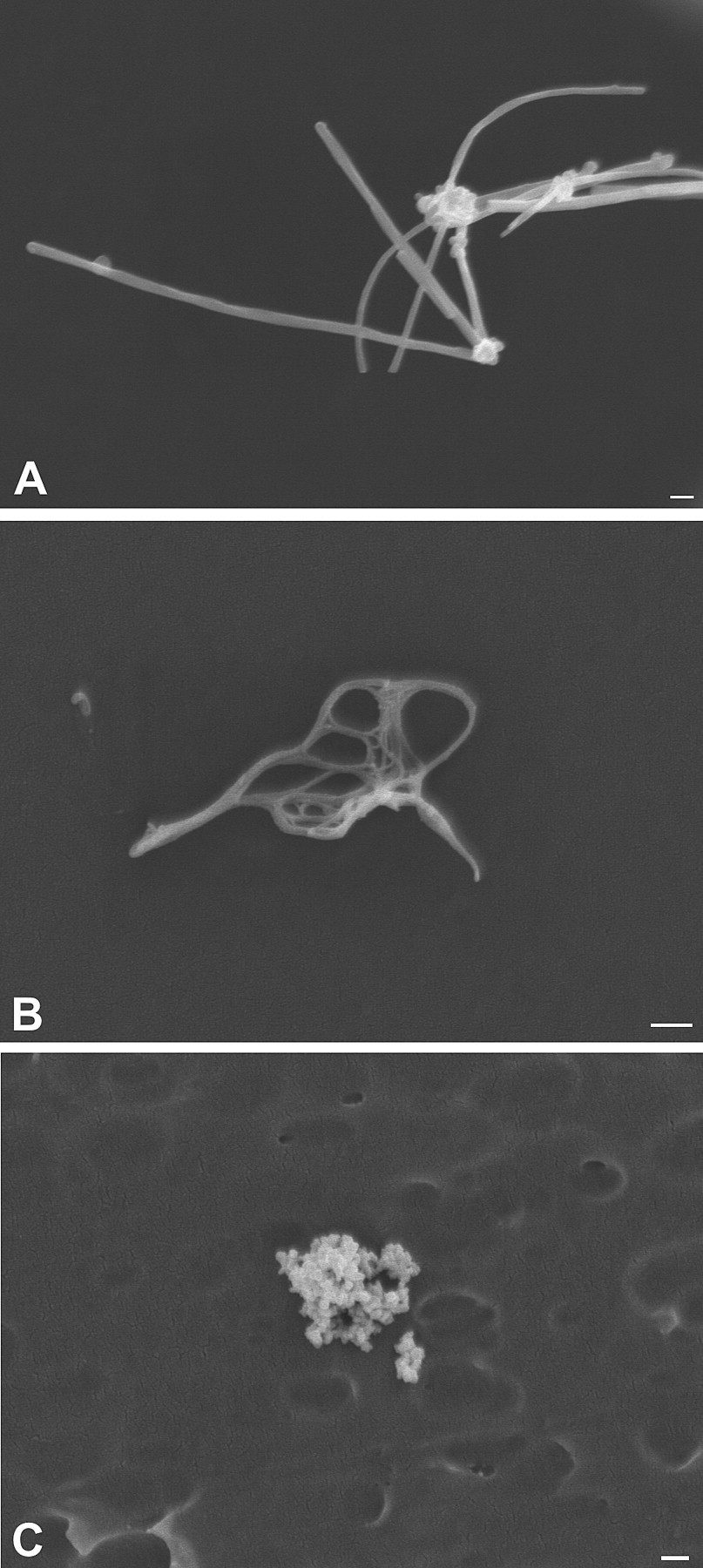
Nanoparticulates as seen by scanning electron microscopy. (A) Multi-walled carbon nanotubes. (B) Single-walled carbon nanotubes. (C) Nanoscale titanium dioxide is also known as ultrafine titanium dioxide and here illustrates that nanoparticulates often agglomerate to form larger particles, making issues of measuring particle number, size, and surface area particularly difficult. Bar = 200 nm.
Carbon nanotubes have many potential applications because of their controlled composition, electrical conductivity, and great tensile strength (Ajayan, Charlier, and Rinzler 1999; Yu et al. 2000). It is the ability to control the shape and composition of nano-objects first revealed by carbon nanotubes that makes nanoscale engineering possible. Of particular importance, carbon nanotubes revolutionized the interest in controlled synthesis of nanoparticulates, which rapidly evolved to include elements other than carbon. Controlled manufacturing of nanoscale products usually involves the engineered growth of particulates (Sayes and Warheit 2009). The chemical composition is determined by the intended use of the nanotechnology product. For example, these products may now be comprised of fullerene carbon, minerals, metals, light emitting structures known as quantum dots, or anything else that can be engineered in nanoscale dimensions (Aitken et al. 2006; Curl and Smalley 1988; Iijima 1991; Kroto 1988). Today, a variety of different elements can be arranged into specific and highly controlled shapes with nanoscale dimensions. Examples include nanotubes and sheets made of boron (J. Wang, Liu, and Li 2009), zinc oxide nanowires (F. Wang et al. 2010), ultrathin zinc oxide nanorods (Cao, Wang, and Wang 2010), gold nanotubes and nanorods (Niidome et al. 2003; Wirtz, Yu, and Martin 2002), and an ever-increasing number of more complex structures and mixtures (Devika et al. 2010; Diaz and Cagin 2010; W. Kim, Choi, et al. 2009; Koppinen et al. 2010; Paulose, Varghese, and Grimes 2003).
A general concern for the NPs is that decreasing particulate size increases the surface area per unit volume or mass. For reactive particles, this increases the surface area for reaction; and for soluble particles, this leads to more rapid dissolution (Watari et al. 2009).
Of particular importance to toxicologic pathologists is an expanding group of nanotechnology products with medical applications, including nanoparticulates for medical imaging and targeted drug delivery (Cirstoiu-Hapca et al. 2009, 2010; Cormode et al. 2009; Kateb et al. forthcoming; Lee et al. 2010; Partha and Conyers 2009; Tian et al. 2010). Understanding how smaller dimensions can alter biologic responses should improve safety assessment for these new products. However, the small size of NPs also means that subcellular interactions can be critical. In short, for pathologists who have discarded their slide holder, they may want to buy a new one.
Unfortunately, studies of NP toxicology lag well behind the development of new engineered NPs. However, several reviews of the toxicology of NPs underscore the importance, as well as the unique difficulties, of nanotoxicology studies (Bonner 2010; Boverhof and David 2010; De Jong and Borm 2008; Doak et al. 2009; Duffin, Mills, and Donaldson 2007; Fischer and Chan 2007; Hoet et al. 2009; Landsiedel et al. 2009; Linkov, Satterstrom, and Corey 2008; Oberdörster, Oberdörster, and Oberdörster 2005; Oberdörster, Stone, and Donaldson 2007; Tsuji et al. 2006). Nevertheless, a recent PubMed search using the search term “nanotechnology” recovered in excess of 20,000 scientific publications in the past 10 years. A PubMed search using the term “nanomedicine” recovered 1,417 references, and there is indeed now a journal with that name. Yet a PubMed search using the term “nanotoxicology” recovered only 77 references, while a search of the terms “nano” and “toxicology” recovered 82 references. Surprisingly, a PubMed search using the term “nanopathology” recovered only 1 reference (Gatti et al. 2009), while a search of the terms “nano” and “pathology” recovered 430 references. No reviews of the toxicologic pathology of nanoparticulates were found. With a whole spectrum of new nanotechnology products and an increasing interest in nanomedicine, this seems to be a very good time for toxicologists, pathologists, and toxicologic pathologists to join the nanotechnology revolution.
Exposures
Few studies have looked at NP exposures. As has recently been noted, the number of newly emerging engineered NPs seems almost countless (Bonner 2010). Another result of the explosion in nanotechnology research is that it translates into the development and use of diverse new NPs in research laboratories at universities (D. Johnson et al. 2010; Tsai et al. 2009). There, students are presumably exposed during graduate and potentially undergraduate education. Such exposures are largely unregulated, and no research papers were identified that investigated or discussed exposures in this population.
The principal routes of occupational and environmental NP exposures are through the skin, gastrointestinal tract, and respiratory tract (Aitken et al. 2006; Cormode et al. 2009; Han et al. 2008; Y. Kim et al. 2008; Maynard and Kuempel 2005; Tinkle et al. 2003). Many current NP exposures are skin exposures to consumer products. Conversely, many NPs intended for medical imaging and/or therapeutics are still in an investigational stage, with intravascular and other forms of parenteral exposure being major exposure routes (Cormode et al. 2009; Y. Kim et al. 2008; Murashov 2009). Importantly, nanomedical products can expose workers as well as patients, so these are not entirely distinct kinds of exposures (Murashov 2009).
Unfortunately, currently available techniques for measuring airborne particulates were not developed to measure workplace exposures to particulates with nanoscale dimensions (Maynard and Kuempel 2005). As has recently been reviewed, to measure aerosol concentrations of NPs, it may be necessary to measure the number of particles, their surface area, and/or mass particle concentration (Maynard and Aitken 2007). As noted by the National Institute for Occupational Safety and Health (NIOSH; 2009), current data suggest that surface area, particle size, shape, and surface chemistry of NPs may be more important measures of exposure than particle mass. An additional issue is whether particle number represents the number of agglomerates or the number of NPs that make up that agglomerate (Figure 1C). Current instrumentation can make some of these exposure measurements, but practical instruments for routinely measuring personal exposures in the breathing zones of workers are not yet available (Maynard and Aitken 2007; Methner, Hodson, and Geraci 2010; Murashov et al. 2009). Specific occupational exposure limits do not yet exist for most NPs (Maynard and Aitken 2007; Murashov et al. 2009; NIOSH 2009).
Within research laboratories, NPs can be released into the air in very high concentrations. Thus, in one study the task of weighing and transferring raw multi-walled carbon nanotubes (MWCNTs) of approximately 1 µm diameter caused an airborne particle release of 4,514 particles/L; while the task of weighing and transferring MWCNTs of approximately 300 nm diameter released in excess of 123,403 particles/L air (D. Johnson et al. 2010). Sonicating raw MWCNTs in water containing natural organic matter generated an aerosol containing 42,796 particles/L (D. Johnson et al. 2010). Under simulated production conditions, limited data indicate that aerosols of single-walled carbon nanotubes (SWCNTs) may be generated, but concentrations are generally low on a mass basis (<53 µg/m3) (Maynard et al. 2004). However, higher exposures may occur during specific manufacturing processes in the absence of appropriate engineering controls (Methner, Hodson, Dames, et al. 2010). Carbon nanotubes can also be produced from high temperature burning of hydrocarbon fuels (Bang et al. 2004; Lam et al. 2006; Murr 2008). While the generation of MWCNTs from burning of hydrocarbons has received the most study, it was the thinner SWCNTs that were detected in four of the seven samples of dust from the World Trade Center site and in lung tissues from three of four World Trade Center responders with extensive interstitial/parenchymal lung disease (M. Wu et al. 2010). The SWCNTs from the World Trade Center site are believed to be the products of carbon and metal combustion (M. Wu et al. 2010). Since identification of SWCNTs in World Trade Center dust is very recent, the frequency of SWCNT exposures during and following structural fires remains unknown.
Dermal exposures to carbon nanotubes are important in workers. As much as 6 mg of SWCNTs may contaminate each glove of workers at a SWCNT production facility (Maynard et al. 2004). Contamination of gloves raises the possibility of transfer to unprotected skin and also potential migration of skin-deposited NPs into flexed or damaged skin of workers (Tinkle et al. 2003).
Dermal exposure to NPs also occurs with consumer products. Current NP exposures in consumer products include inorganic NPs. Consumers are exposed to TiO2 and ZnO NPs in sunscreens (Nohynek et al. 2010). In addition, wound dressings have been reported to contain nanocrystalline silver as an antimicrobial agent (Bhattacharyya and Bradley 2006, 2008). Particles less than 1 µm in diameter can penetrate flexed skin in motion, reaching at least the epidermis and potentially the dermis (Tinkle et al. 2003). After dermabrasion, polyethylene glycol coated cadmium selenide quantum dot NPs can penetrate the skin, with 2% of the cutaneously applied cadmium actually reaching the liver (Gopee et al. 2009). However, in a recent study in minipigs, application of TiO2 NPs in sunscreens to intact skin did not increase TiO2 concentrations in the lymph nodes or liver, suggesting that they are not systemically absorbed (Sadrieh et al. 2010). However, TiO2 NPs reach distant organs after topical application in hairless mice (J. Wu et al. 2009). Skin penetration of NPs may be influenced by other components in topical formulations. Oily formulations appear to enhance skin penetration of TiO2 NPs, while aqueous formulations decrease skin penetration (Bennat and Muller-Goymann 2000).
Thus, NPs are currently in topical formulations of cosmetics and wound dressings. In intact skin, NPs appear to reach the epidermis but in most studies do not go further. However, within the epidermis of the intact skin, NPs could potentially interact with antigen presenting cells (Nestle et al. 2009). In addition, NPs appear to be capable of traversing through damaged or wounded skin. Finally, from an occupational standpoint, these skin products are in current production, but data on exposures to workers are not yet available. An overarching issue is that modern technology is influencing the nature and extent of NP exposures for workers, consumers, and patients.
Demonstrating NPs in Tissue
Factors That Limit the Ability to Identify NPs in Tissue Sections
Given the high number of NPs present in even a microgram of NP material, visualization of NPs might appear to be a routine microscopic exercise. For instance, a 50 µg lung burden of well-dispersed MWCNTs in the mouse lung could easily distribute into 10 billion or more nanotubes throughout the lungs. Given that the mouse lung has on the order of 4 million alveoli (Mercer, Russell, and Crapo 1994), this would, on average, yield approximately 10,000 or more nanotubes per alveolus.
Numerous factors work in concert to significantly reduce the visibility of this apparently vast number of particles. These include the following:
Light micrographs of particulates in tissue sections. (A) Light microscopic image of asbestos fibers (arrow) penetrating an alveolar macrophage. Individual fibers, such as asbestos, are easily seen in sections, as single fibers effectively block light over a significant fraction of the field of view. In addition, the fibers are “in- focus” throughout the section even when imaged with higher power objectives that have limited depth of field. (B) Sirius Red—Hematoxylin stained section from mouse lung 1 month after aspiration dose of 2 mg/kg SWCNTs. The Sirius Red stain highlights the extensive collagen fibers, which are interspersed within the large, gray to black, masses of SWCNTs. The SWCNTs “visible” in this image vary from a light gray (right arrow, few fibers in the light path) to nearly back (left arrow, large, dense grouping of fibers in the light path). (C) Silver-enhanced demonstration of colloidal gold-labeled SWCNTs in the mouse lung. This micrograph illustrates the utility of using a labeled nanomaterial to demonstrate the distribution of material that would not otherwise be visible in sections (arrows).
Depending on the specific characteristics, these factors can limit the ability to detect and identify NPs in conventional histology slides and require specialized techniques.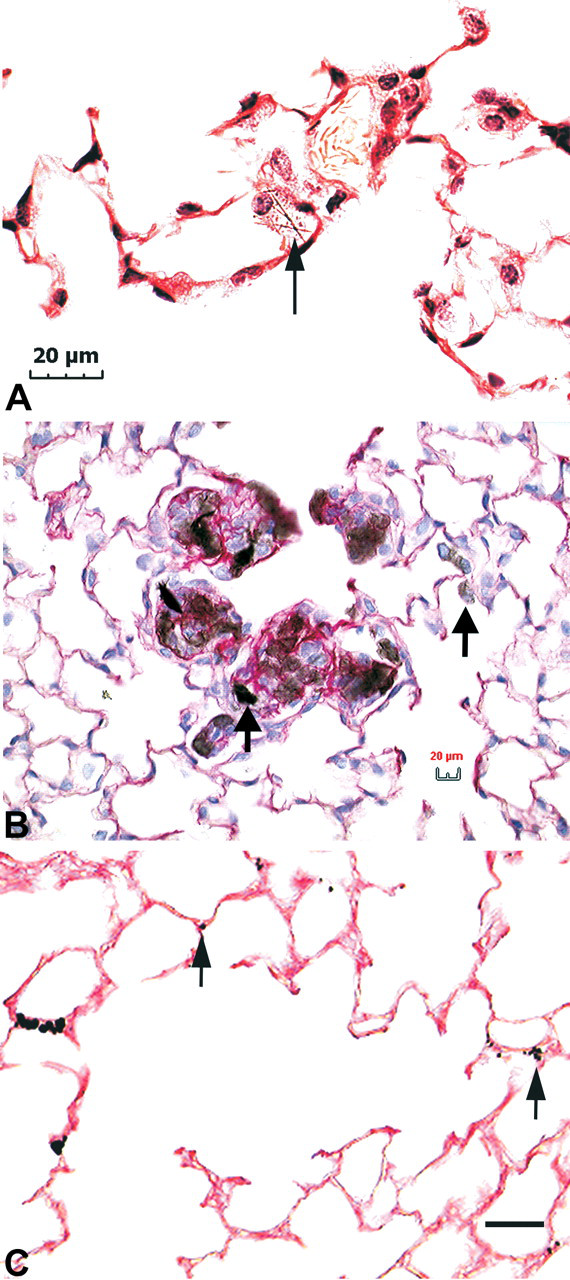
Labeled NPs
Labeling of NPs with a fluorescent indicator or some conveniently detected particle such as colloidal gold is one possible solution to make NPs easily visible in sections. Functionalization of the carbon-carbon bonds in carbon nanotubes has been done to enhance the carbon nanotubes for commercial use (Sinnott 2002). Functionalization such as the oxidation of the carbon-carbon bonds may be used to label the carbon nanotubes with colloidal gold. Labeling with colloidal gold allows the application of a variety of established techniques which have already been developed for immunohistochemistry and other fields.
These techniques can be used to allow detection in microscopic sections by silver enhancement and to aid in identification in TEM/FESEM (transmission electron microscope/field emission scanning electron microscope) observation (Mercer et al. 2008). Figure 2C shows an example from that study in which silver enhancement of the gold labeled SWCNTs was used to detect a highly dispersed preparation of SWCNTs that would not otherwise have been visible. This section is from the lungs of a mouse given a 10 µg preparation of highly dispersed and size limited SWCNTs, which were 1.1 µm or less. In conventional sections, these highly dispersed SWCNTs are rarely if at all detectable, but by using the silver enhancement to enlarge the gold label on the SWCNTs, the widespread distribution could be detected
High-Resolution FESEM
Conventional scanning electron microscopes (SEM) that are used for biologic specimens have been applied with great success to imaging of many of the micrometer-dimensioned inhaled particles that were studied prior to the advent of nanomaterials. Figure 3A shows a backscatter electron SEM image from a section of a lung exposed to silica particles that has been placed on a carbon planchet. The high-level backscatter emissions from the silica particles are easily identified in the alveolar macrophages of the section.
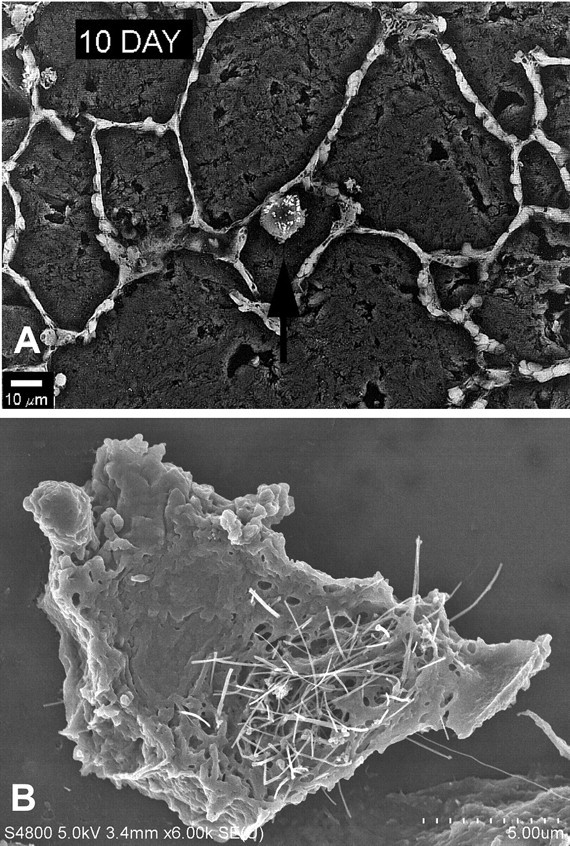
Scanning electron micrographs of particulates in tissue section. (A) Backscatter SEM image of lung section 10 days after silica inhalation. An alveolar macrophage loaded with fine-sized silica particles is identified by the arrow. Imaging of traditional particles such as silica and asbestos can be accomplished at low resolution due to the large particle size and the heightened electron emissions associated with the elemental composition of the particles. (B) FESEM of alveolar macrophage loaded with MWCNTs. Image shows the typical appearance of mouse lung 28 days after exposure to an 80 µg dose of MWCNTs. Bar = 30 µm.
For NPs, the conventional SEM does not have the sub-micrometer resolution necessary to resolve or identify NPs. Difficulties in imaging NPs with an SEM are further complicated by the fact that many NPs, such as carbon nanotubes, have no significant difference in secondary electron or backscatter emissions from the organic carbon in which they are immersed.
Introduction of the field emission scanning electron microscope (FESEM) has significantly improved the resolution and applicability of the SEM to examination of NPs in tissue. The unique “cold” cathode design of the FESEM produces high-quality, low-voltage images with negligible electrical charging that can be used to identify NPs in tissues at levels of resolution not previously available with the conventional SEM. The high-resolution capability of the FESEM greatly facilitates the imaging of MWCNT interactions with cells and tissues of the lung. The FESEM image in Figure 3B shows the penetrating nature of MWCNTs in an alveolar macrophage 28 days after exposure.
Histopathologic Changes in NP-Exposed Tissues
The explosion in nanotechnology research means that the histopathologic changes following exposure to most nanotechnology products are uninvestigated. The first indications of the potential risks from the nanoscale forms of compounds with known toxicity profiles came from studies of ultrafine particulates and from studies of carbon nanotubes (Bonner 2010; Donaldson et al. 2006; Oberdörster 2010; Oberdörster et al. 2005; Oberdörster, Oberdörster, and Oberdörster 2005; Oberdörster, Stone, and Donaldson 2007).
The prototypical engineered NPs, SWCNTs, cause pulmonary inflammation (Shvedova et al. 2008, 2005; Warheit et al. 2004). However, the significance of the SWCNT-induced inflammation has been questioned by some scientists. In the first study of intratracheally instilled SWCNTs in rats, discrete granulomas that were not dose-responsive and an absence of signs of inflammation in bronchoalveolar lavage, suggested the possibility that large agglomerates of SWCNTs caused the granulomas (Warheit et al. 2004). A second study in rats, using SWCNT aspiration, also noted little change in the differentials of bronchoalveolar lavage and a relative paucity of histopathologic evidence of inflammation (Mangum et al. 2006). However, bronchoalveolar lavage fluid analysis is an insensitive indicator of granulomatous inflammation in the lung (D. Porter et al. 2007), providing a possible explanation for the paucity of bronchoalveolar lavage changes in SWCNT-exposed rats. Studies in mice demonstrated significant inflammation, confirmed that SWCNT-induced inflammation was often granulomatous, and, most importantly, demonstrated that inflammation was present whether the SWCNTs were inhaled or aspirated (Lam et al. 2004; Shvedova et al. 2008, 2005). In mice, the number of polymorphonuclear leukocytes and macrophages in bronchoalveolar lavage fluid increased after inhalation exposure and declined after 7 days (Shvedova et al. 2008). Inflammation was confirmed by histopathology and by elevated inflammatory cytokines (Shvedova et al. 2008). Thus, at least some SWCNTs were inflammatory in mice and that inflammation was not due to agglomeration (Shvedova et al. 2008).
Fibrosis has been demonstrated
Like SWCNT exposure, MWCNT exposure causes pulmonary inflammation (Muller et al. 2005; D. Porter et al. 2010). The first inhalation study conducted on MWCNTs turned out to be a study of the effects of a mixture of MWCNTs and nanofibers (Lison and Muller 2008; McDonald and Mitchell 2008). In that study, exposures to concentrations of 5 mg/m3 or less for 14 days produced little evidence of pulmonary inflammation but suppressed T-cell dependent immune functions (Mitchell et al. 2007). An intratracheal instillation study did not demonstrate inflammation or fibrosis but used MWCNTs 0.5–2 µm in length, shorter than the fiber dimensions of greatest toxicologic concern (Elgrabli et al. 2008). Recent MWCNT studies generally used more completely characterized exposure materials than did early studies. Inhalation of MWCNTs for up to 13 weeks caused granulomatous inflammation and pleural thickening at exposure concentrations greater than 6 mg/m3, while bronchoalveolar junction hypercellularity and interstitial fibrosis were demonstrated at exposures above 0.4 mg/m3 (Pauluhn 2010). After a single 6 hr inhalation exposure to 30 mg/m3 MWCNTs, mononuclear cell aggregates were seen on the pleural surface after 1 day, with subpleural fibrosis demonstrated at 2 and 6 weeks postexposure (Ryman-Rasmussen et al. 2009). MWCNTs were seen in subpleural tissue (Ryman-Rasmussen et al. 2009).
A recent study in mice exposed to MWCNTs by aspiration identified that MWCNTs, like SWCNTs, cause granulomatous inflammation and very early fibrosis at 7 days postexposure (Figure 4 ). This study also provides additional information on the histopathologic changes contributing to bronchoalveolar junction hypercellularity, pleural inflammation, and pleural thickening (D. Porter et al. 2010). In MWCNT-exposed mice, the inflammation was often bronchiolocentric with abundant macrophages and lesser numbers of multi-nucleated giant cells, neutrophils, and eosinophils. With time postexposure, lymphocytes and plasma cells were often seen in the highest exposure groups. Inflammation frequently extended to the pleura. Podoplanin immunofluorescence demonstrated peribronchiolar and subpleural lymphangiectasia (Figure 5 ). In addition, rare MWCNTs penetrated through the pleura to reach the mesothelium, the classical target for fiber-induced carcinogenesis (D. Porter et al. 2010). The dilated subpleural and peribronchiolar lymphatics may facilitate delivery of MWCNTs to the delicate bronchioloalveolar junction and to the pleura because the lymphatic endothelium provides homing signals to direct alveolar macrophages into the pulmonary lymphatics to facilitate particle clearance to the tracheobronchial lymph nodes (Harmsen et al. 1987, 1985; L. Johnson et al. 2006; L. Johnson and Jackson 2008) and because macrophages phagocytize MWCNTs (D. Porter et al. 2010). Quantitative data, currently published only in abstract form, confirms that MWCNTs penetrate the pleura after a single 20-80 µg aspiration exposure. At the 80 µg exposure, 1 in 400 lung-deposited MWCNTs reach either the pleural surface or the subpleural alveolar epithelium (Mercer et al. 2010).
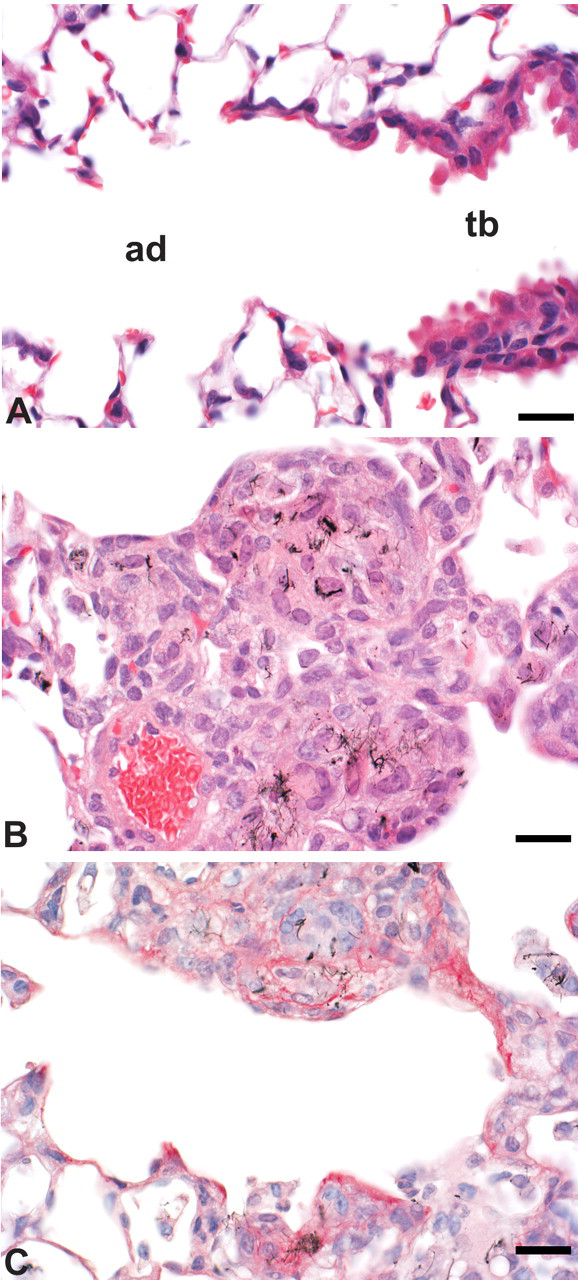
Peribronchiolar granulomatous inflammation and rapid fibrosis following MWCNT aspiration in the mouse. (A) Normal terminal bronchiole (tb) and alveolar duct (ad) in a control mouse. (B) Peribronchiolar granulomatous inflammation in a mouse 7 days after aspirating 80 µg MWCNTs. (C) Fibrosis is demonstrated by red staining in this Sirius Red stained section from a mouse 7 days after aspirating 80 µg MWCNTs. Bar = 20 µm.
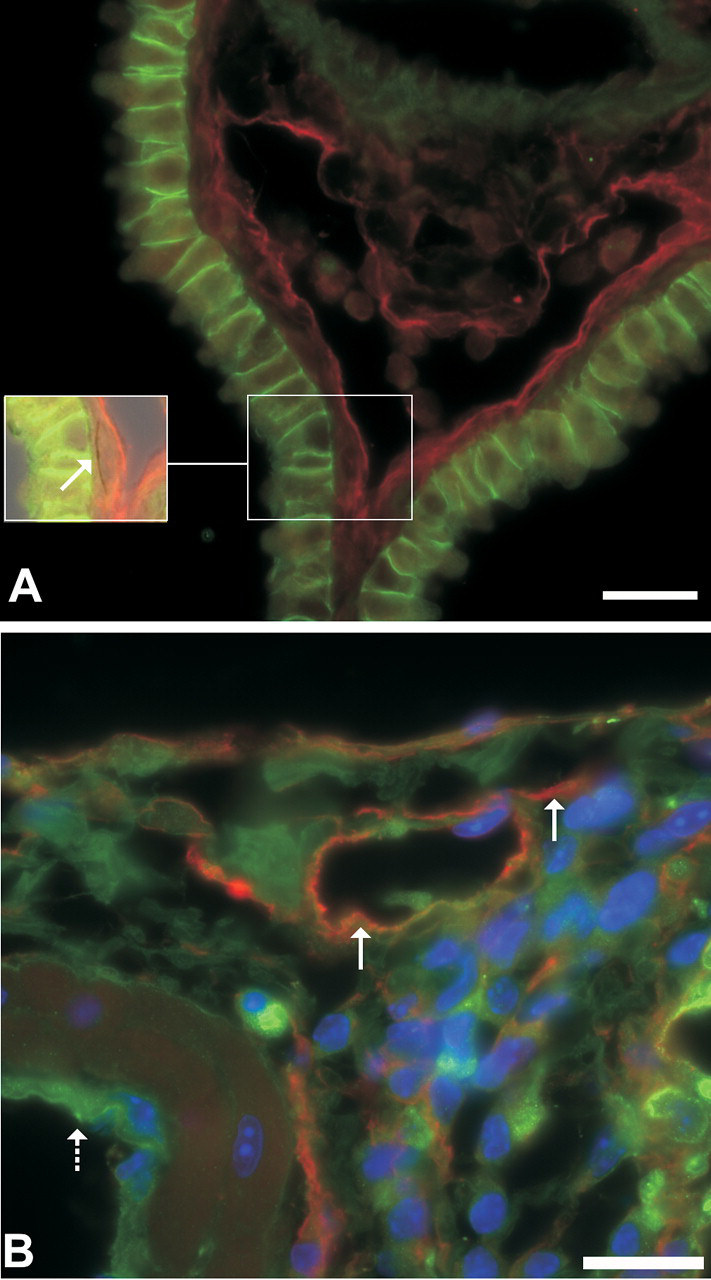
Podoplanin and e-cadherin double-label immunofluorescent images of MWCNTs in tissue section illustrating changes in the lymphatics. (A) Lymphatic endothelium stains red using podoplanin indirect immunofluorescence and e-cadherin oulines the intercellular junctions of bronchiolar epithelial cells. The inset is a portion of the field that has been further illuminated by low levels of transmitted light to demonstrate a MWCNT (arrow) within the wall. Bar = 20 µm. (B) Red staining of lymphatic endothelium demonstrates dilation of subpleural lymphatics (solid arrows). The vascular endothelium does not contain podoplanin and is unstained (dashed arrow). Bar = 20 µm.
The similarities in physical shape and biological persistence between MWCNTs and amphibole asbestos fibers have been reviewed and have caused several research groups to hypothesize that MWCNTs, like asbestos, could cause mesothelioma (Donaldson et al. 2010; Jaurand, Renier, and Daubriac 2009; Pacurari, Castranova, and Vallyathan 2010; Takagi et al. 2008). Indeed, 87.5% of p53 heterozygous mice developed mesothelioma after intraperitoneal injection of 3 mg MWCNTs/mouse (Takagi et al. 2008). However, the 3 mg exposure is more than 10- to 100-fold greater than typical doses used for pulmonary exposures. The particle characteristics in the study also increased the chance of identifying the carcinogenicity of MWCNTs. The MWCNTs had a mean width of ~100 nm, 27.5% of the nanotubes exceeded 5 µm in length, and the iron content was ~0.35%, dimensions similar to the dimensions of carcinogenic asbestos fibers and an iron content sufficient to generate reactive oxygen species through the Fenton reaction (Takagi et al. 2008). Additional support for the carcinogenicity of these same long MWCNTs in mesothelial cells came from the subsequent demonstration of mesotheliomas in 86% of male rats receiving intrascrotal injections of 240 µg MWCNTs/rat (1 mg/kg) (Sakamoto et al. 2009).
A subsequent 2-year bioassay of intraperitoneally injected MWCNTs averaging < 1 µm in length, with and without structural defects, failed to produce statistically significant numbers of mesotheliomas or other peritoneal tumors in Wistar rats. However, the comparisons were between any of the three MWCNT treated groups and the control group (where one rat developed a peritoneal mesothelioma) or with historical control numbers for mesotheliomas in rats (Muller et al. 2009). This negative finding may need to be viewed with caution since the MWCNTs used in the bioassay were short; there were 5 peritoneal mesotheliomas in 150 rats exposed to any MWCNT; and the finding of a primary peritoneal mesothelioma in a control Wistar rat is unexpected since mesothelioma is an uncommon spontaneous primary peritoneal tumor in this rat strain (Muller et al. 2009; Poteracki and Walsh 1998; Walsh and Poteracki 1994). An additional study that was negative for mesothelioma used gelatin capsules containing MWCNTs and SWCNTs implanted into the omentum of rats. However, the tendency for carbon nanotubes to agglomerate would be exacerbated by this exposure procedure and the experimental duration of 12 months would miss late-developing mesotheliomas (Varga and Szendi 2010). Thus, the potential for NPs with fiber-like dimensions to cause mesothelioma needs further study/investigation. A related issue is the potential for the NPs to cause subcellular injury and, in particular, to cause mitotic spindle changes reminiscent of amphibole asbestos and other hazardous fibers; these issues are addressed in the next two sections of this review.
For the nonfibrous NPs, the existing data on respirable particles provides important insights into how particle number, mass, surface area, surface properties, and composition affect particle toxicity and how nanoscaling may influence particle-induced histopathologic alterations. For soluble particles, toxicity is usually determined by the toxicity of the components. In addition, for metals and some other elements, which may have low solubility but high cytotoxicity, the composition may also have major effects on toxicity. For metals, such as hexavalent chromium, nickel, and beryllium, even low dissolution of metal ions can largely determine the toxicity (Cohen et al. 2007; Wise et al. 2004, 2006). Thus, “insoluble” nickel particles undergo dissolution in the acidic environment of cytoplasmic vacuoles, releasing nickel ion (Abbracchio, Simmons-Hansen, and Costa 1982; Costa et al. 2005). Since the composition of highly toxic, immunogenic, and/or soluble particles largely determines the toxicity, the composition also largely determines the histopathologic changes. For example, beryllium particles persist in the lung after exposure (Finch, Nikula, and Hoover 1998), but if the host MHC class II protein is a polymorph able to present beryllium to the immune system, that combination of an immunogenic exposure and host susceptibility correlates with chronic beryllium disease (Falta et al. 2010; Fontenot and Amicosante 2008; McCleskey et al. 2009; Snyder et al. 2008). For the example above, inhaled beryllium will cause a lymphocytic to lymphogranulomatous response that is highly influenced by genotype (Fontenot and Amicosante 2008; Freiman and Hardy 1970; McCleskey et al. 2009; Nikula et al. 1997; Tarantino-Hutchison et al. 2009). Because the composition of soluble and/or acutely cytotoxic particles drives the toxicity, the mass of these particles deposited in the lung can be a major determinant of lung toxicity (Cassee et al. 2002). However, for these particulates, a major effect of decreased particle size is that it can increase pulmonary deposition (Cassee et al. 2002).
For poorly soluble particles of low acute cytotoxicity, histopathologic alterations in the exposed lung generally develop with time and are generally characterized by dose-dependent chronic inflammation, fibrosis, varying degrees of alveolar epithelial cell hypertrophy and hyperplasia, and, with some species and particles, lung tumors (Baan 2007; Bermudez et al. 2002, 2004; Muhle et al. 1995; D. Porter et al. 2001; Roller 2009). However, composition clearly matters, since exposure cessation causes regression of histopathologic alterations in TiO2 exposed rats, but not in rats exposed to crystalline silica (Baggs, Ferin, and Oberdörster 1997; D. Porter et al. 2004). In addition, the alveolar epithelial proliferative response and lipoproteinosis are particularly prominent following crystalline silica exposure (Miller et al. 1987; Miller, Dethloff, and Hook 1986; Miller and Hook 1990; D. Porter et al. 2001). The characteristics of the particle surface may also influence particle toxicity. Particles may have nonhomogeneous composition that affects the particle surface and alters cytotoxicity (Wallace et al. 1990, 1985). When compared with stored silica or coated silica, freshly fractured silica particles, for example, produce more free radicals from their surface, increase the respiratory burst when phagocytized, and cause more pulmonary inflammation (Vallyathan et al. 1995, 1991, 1988). Some NPs appear to generate more hydroxyl radicals from their surface than larger, but still respirable, particulates (Donaldson, Beswick, and Gilmour 1996). Consistent with these earlier studies, surface reactivity (radical generation) of TiO2 NPs can influence toxicity (Warheit, Reed, and Sayes 2009).
Although surface free radical generation and coatings clearly influence NP toxicity, there are general trends for NPs that cannot be explained by their surface chemistry and composition. When compared with larger respirable particles on a mass basis, NPs cause increased pulmonary inflammation (Oberdörster, Ferin, and Lehnert 1994; Renwick et al. 2004). Where particle composition and surface properties are held constant, particle toxicity then correlates well with surface area (Oberdörster, Ferin, and Lehnert 1994; Sager and Castranova 2009; Sager, Kommineni, and Castranova 2008). Since the surface area of NPs is much higher than for larger respirable particles, this may explain some of the enhanced toxicity of NPs.
While particle toxicity has historically been studied in the lung, recent studies suggest that NPs can be incorporated into gastrointestinal epithelial cells (Gaiser et al. 2009). Indeed, it has been suggested that particles may also play a role in gastrointestinal disorders, including Crohn’s disease (Lomer, Thompson, and Powell 2002). Additional information on the toxicologic pathology associated with particles and ultrafine particles can be found in several excellent reviews (Baan 2007; BeruBe et al. 2007; Donaldson et al. 2002; Maynard and Kuempel 2005; Nikula 2000; Oberdörster 1996, 2002; Oberdörster et al. 2000; Oberdörster, Oberdörster, and Oberdörster, 2005; Schmid et al. 2009; Stone, Johnston, and Clift 2007).
Nonfibrous NPs can also have toxic effects that are not seen with larger particles. Among the current products using engineered NPs, silver NPs appear to be the most common, and the toxicity of silver NPs has recently been reviewed (Johnston et al. 2010). When silver NPs are inhaled, silver is detectable in multiple tissues (Ji et al. 2007; W. Kim, Kim, et al. 2009; Sung et al. 2009; Takenaka et al. 2001). After a 90-day exposure, pulmonary inflammation is demonstrable, while biliary hyperplasia in the liver suggests hepatic effects of the redistributed silver (Sung et al. 2009). Additional recent studies on silver NPs suggest that similar to fibrous NPs, cell membranes may be ineffective in containing the nonfibrous NPs. When injected subcutaneously, silver NPs, unlike larger silver microparticles, distributed widely to many tissues (Tang et al. 2009). This demonstrates the principle that NPs can pass cell membranes and emphasizes the need to consider the potential for NPs to reach distant target tissues. In addition, the antimicrobial properties of silver NPs and their use in open wounds combined with converging lines of evidence suggesting potential neurotoxicity and vascular effects of silver NPs indicate a need for additional research (Bhattacharyya and Bradley 2006, 2008; Kalishwaralal et al. 2009; Powers et al., 2010; Rahman et al. 2009; Rosas-Hernandez et al. 2009; Sharma et al., 2010; Tang et al. 2009; Y. Wu et al. 2010). While current data on silver NPs suggest that at least some of the vascular and neurotoxic effects are associated with direct effects of NPs after migration, systemic effects may or may not involve translocation of the NPs (see section on systemic effects). Indeed, a recent
Studies of how NPs affect mixed exposures are just beginning. MWCNT inhalation causes airway fibrosis and elevates IL-5 mRNA in ovalbumin-sensitized mice, changes not seen in mice exposed to ovalbumin or MWCNTs alone (Ryman-Rasmussen et al. 2009). TiO2 NPs act as adjuvants when mice are intraperitoneally sensitized with ovalbumin and then challenged by ovalbumin aerosol. Either TiO2 NPs or the classic adjuvant, Al(OH)3, enhance ovalbumin sensitization with increases in ovalbumin-specific IgE and IgG1 in serum and increases in eosinophils, neutrophils, and lymphocytes in bronchoalveolar lavage fluid (Larsen et al. 2010). Similarly, in traditionally ovalbumin-sensitized mice, inhaled carbon black NPs enhance IL-4, IL-5, IL-13, and cellular infiltration upon subsequent ovalbumin challenge (Alessandrini et al. 2006). Thus, the NPs investigated to date appear to enhance sensitization and inflammation, and in some scenarios, the NP effect may be more than additive. Since environmental and occupational exposures to NPs are likely to be part of mixed exposures, the potential interactions of NPs in mixed exposures will undoubtedly receive additional future investigation.
Subcellular Morphologic Changes in the NP-exposed Cells
Inhalation is a major route of exposure to engineered NPs (D. Johnson et al. 2010). The first-generation products of nanotechnology include nanotubes, nanofibers, and nanowires. For these NPs, the aspect ratio, the ratio of length to width, can be quite high (D. Porter et al. 2010; Takagi et al. 2008). The existing literature on natural fibers is useful for guiding investigation of the newer engineered NPs and the potential for NPs with high aspect ratio to injure intracellular organelles.
For the classic carcinogenic durable fibers, amosite and crocidolite asbestos, many of these fibers have nanoscale diameters and higher aspect ratios that are associated with the carcinogenicity of inhaled asbestos (Churg 1988; Murai et al. 1995; Stanton et al. 1981). In his final posthumously published paper, Mearl Stanton along with several colleagues noted that the probability of experimentally induced pleural sarcomas was highest when asbestos fibers were 250 nm or less in diameter and 8 µm or greater in length (Stanton et al. 1981). Importantly, some carcinogenic asbestos fibers have diameters < 100 nm and are natural mineral NPs (Stanton et al. 1981). Negligible phagocytosis of the long, thin asbestos fibers was suggested as a possible cause of the relationship between asbestos fiber dimensions and carcinogenesis (Stanton et al. 1981). Incomplete, rather than negligible, phagocytosis of long asbestos fibers was an observation of Beck and coworkers (Beck, Holt, and Manojlovic 1972).
Generation of free radicals during incomplete phagocytosis of long fibers has long been suggested as a potential factor in asbestosis and asbestos carcinogenicity (Beck, Holt, and Manojlovic 1972; Kamp et al. 1992). However, asbestos fibers also inhibit cytokinesis and can cause the formation of cells joined by fiber-containing intercellular bridges or cause bi- and tri-nucleated cells (Jensen and Watson 1999). In addition, asbestos fibers cause multinucleated giant cells to form via cell fusion (Jensen and Watson 1999). Whether from failure of cytokinesis or from cell fusion, the very large multinucleated giant cells seen in asbestos exposure should have sufficient cytoplasm to complete the phagocytic process, raising some questions regarding whether incomplete phagocytosis is the result of fibers too long for phagocytosis or a fiber so thin that it cannot be retained within a lipid bilayer. Indeed, in Mearl Stanton’s final posthumous paper, there is a suggestion that the thin fibers, by permitting cell survival, might be particularly important in asbestos carcinogenesis. In the final paragraph, the paper considers the mechanism of carcinogenesis from long, thin fibers and notes: “A provocative explanation relates to the ability of fine, long fibers to penetrate cells without killing them” (Stanton et al. 1981). Consistent with that hypothesis, a subsequent study indicates that
Among the first-generation NPs, SWCNTs, like long thin asbestos fibers, have a high aspect ratio, are extraordinarily thin, and, unless they are functionalized, can have very low levels of
There are additional similarities between asbestos and SWCNT effects. As with asbestos exposure, failure of cytokinesis with formation of intercellular bridges has been reported in SWCNT-exposed macrophages (Mangum et al. 2006). In addition, SWCNTs cause somatic cell mutations in the lung in addition to the
In contrast to SWCNTs, many MWCNTs are avidly phagocytized by macrophages (Cheng et al. 2009; D. Porter et al. 2010). In addition to phagocytosis, MWCNTs can penetrate the cytoplasm of macrophages and epithelial cells by apparent direct penetration of the cytoplasm (Cheng et al. 2009; D. Porter et al. 2010). Within macrophages, MWCNTs frequently extend from the cytoplasmic margins and penetrate subcellular structures such as nuclei (Cheng et al. 2009). In some cases, extension of MWCNTs through the cytoplasmic membrane may represent incomplete phagocytosis; but in other cases, MWCNTs appear to mechanically penetrate the cytoplasm (Cheng et al. 2009; D. Porter et al. 2010). Indeed, in mobile cells such as macrophages, and in tissues such as lung and heart that repeatedly expand and contract, it seems unreasonable to expect cell membranes comprised of lipid bilayers to effectively contain fibrous NPs of only a few nm diameter. Thus, one can expect migration of fibrous NPs within mobile cells and tissues with resulting direct damage to cell membranes and intracellular organelles. Pathologists and toxicologists should be aware of this potential when evaluating
Mitotic Spindle Interactions
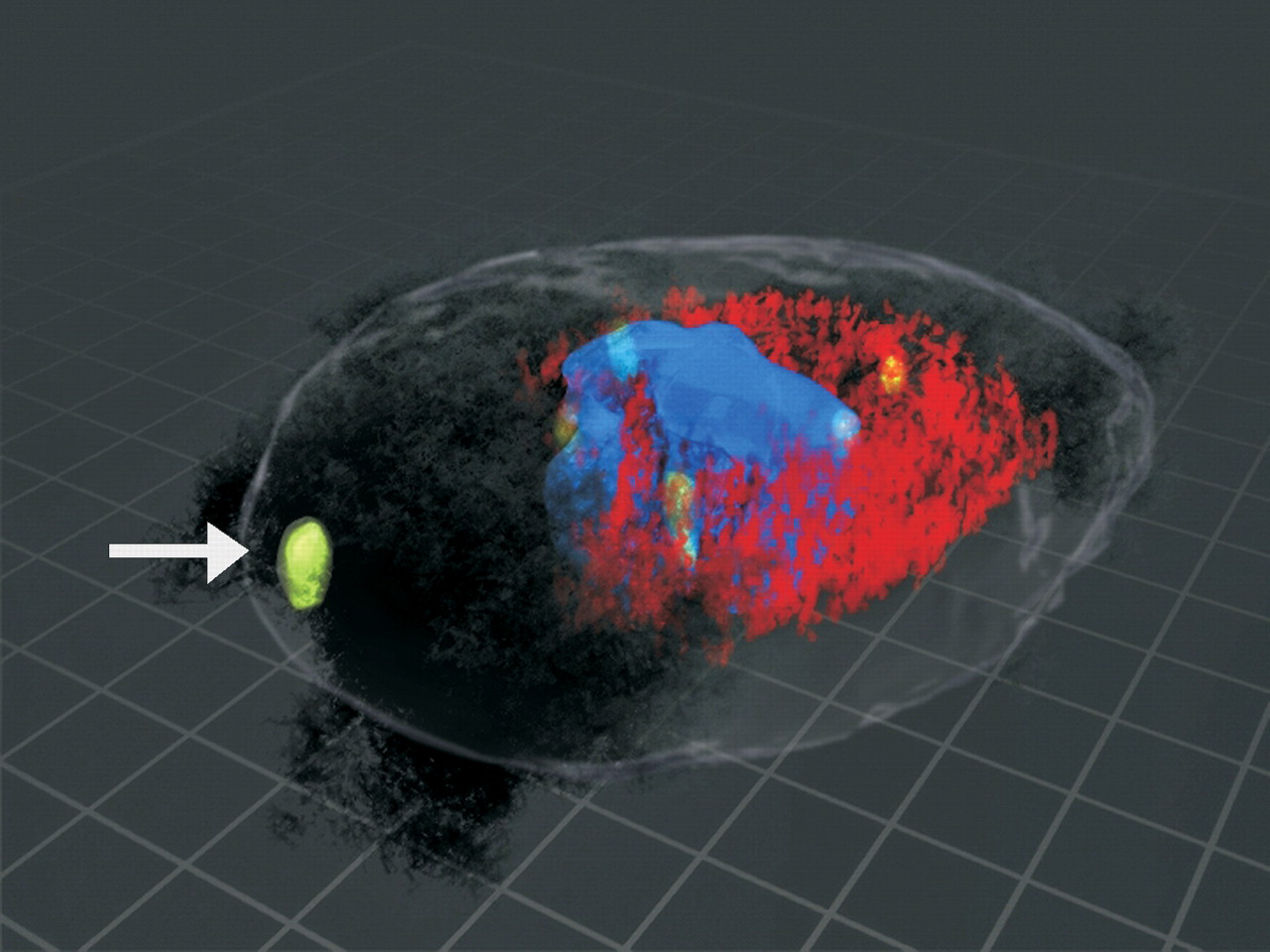
3D reconstruction showing the interaction of the centrosome, the DNA, and the microtubules with SWCNTs. BEAS-2B cells were grown in culture to 70% confluence and then treated with SWCNTs at 24, 48, or 96 µg/cm2 cell plate surface area. The tubulin was stained red using Spectrum red and indirect immunofluorescence. The centrosomes are stained green using Alexa 488 and indirect immunofluorescence. The DNA was detected by DAPI and was blue. The nanotubes were imaged using differential interference contrast. A minimum of 100 cells per sample and a total of 300 cells of good centrosome and good mitotic spindle morphology were analyzed for each treatment dose. To localize carbon nanoropes to the microtubules of the mitotic spindle and the centrosome, serial 0.1 µm optical slices were obtained to create a Z-stack (Sargent et al. 2009). The Z-stack images were used to construct a 3-dimensional image using Light-Wave software (Haas and Fischer 1997). The mitosis is multipolar and has fragmented centrosomes. The 3-dimensional image shows nanotubes in association with the centrosomes, the microtubules, and the DNA. The white arrow indicates a fragmented centrosome that was associated with SWCNTs. The nanotubes form an aberrant spindle-like structure that is pulling the DNA toward the nanotubes.
The diameter of SWCNT nanoropes can be comparable to the microtubules that form the mitotic spindle (Figure 7 ) (Lu 1997; Pampaloni and Florin 2008). In addition, carbon nanotubes have many physical properties in common with microtubules including bending properties, high resiliency, stiffness, low density, and high strength (Pampaloni and Florin 2008). Recent laboratory studies indicate that carbon nanotubes can form functional nanotube/tubulin hybrids (Dinu et al. 2009). The similar size, shape, and physical properties make it possible for carbon nanotubes to displace critical cellular targets including the mitotic spindle.
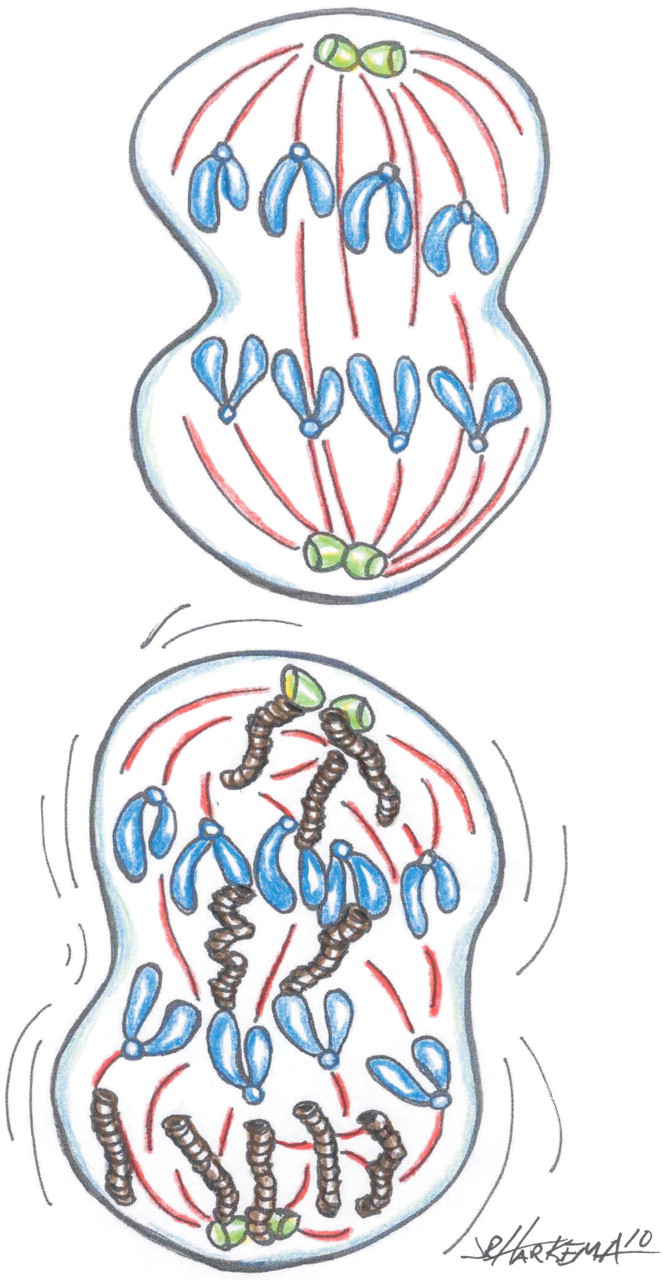
Potential interactions between NPs and the dividing cell. In normal mitosis (top), the centrosomes (green) and microtubules (red) of the mitotic spindle assure that each daughter cell receives the same number of chromosomes. NPs can disrupt the orderly segregation of chromosomes into daughter cells (bottom). SWCNTs (brown) form nanoropes that may vary in diameter but can be similar to the diameter of the microtubules of the mitotic spindle. SWCNT nanoropes interact with centrosomes, microtubules, and DNA, disrupting chromosomal segregation and cause potential aneuploidy.
Systemic Effects
Cardiovascular Effects
A consistent association between levels of inhaled particulates in ambient air and a significant elevation in cardiovascular mortality and morbidity has been reported in a number of epidemiological studies (Abbey et al. 1999; Dockery et al. 1993; Pope et al. 1995; Samet et al. 2000). However, the mechanisms by which inhaled particulates could induce cardiovascular effects remains to be fully elucidated.
Recent studies have reported that pulmonary exposure to engineered NPs also results in a variety of cardiovascular effects. Z. Li et al. (2007) exposed mice to purified single-walled carbon nanotubes (10–40 µg/mouse) by pharyngeal aspiration and monitored oxidant stress in aortic and cardiac tissue at 1, 7, and 28 days postexposure. Such an exposure caused rapid but transient pulmonary inflammation (Shvedova et al. 2005). No evidence of oxidant stress to cardiovascular tissue was noted at 1 day postexposure. However, at 7 days postexposure, significant increases in oxidant damage to aortic mitochondrial DNA and in heme oxygenase 1 activity in aortic and cardiac tissue were reported. The heme oxygenase 1, a marker of cardiovascular oxidant stress, returned to control levels by 28 days postexposure, indicating that a single aspiration of SWCNTs can cause transient cardiovascular effects. Z. Li et al. (2007) also reported data supporting subchronic effects resulting from multiple exposures of ApoE-/-mice (genetically susceptible to atherosclerosis) to purified SWCNTs (aspiration of 20 µg/mouse, every 2 weeks, for 2 months). Extended aspiration of SWCNTs increased aortic plaque formation by 65% and brachiocephalic artery plaque area by 113%. Since plaque formation elevates peripheral resistance and is a risk factor for cardiovascular disease, these data suggest that cardiovascular complications may follow long-term exposure of workers to airborne carbon nanotubes.
Nurkiewicz et al. (2008) reported that inhalation of titanium dioxide NPs, at lung burdens that did not elevate bronchoalveolar lavage markers of pulmonary inflammation or damage, decreased the ability of arterioles in systemic skeletal muscle to respond to intravascular infusion of dilators 24 hours postexposure. On an equal mass lung burden basis, titanium dioxide NPs were 7 times more potent than fine-sized titanium dioxide particles. Recently, LeBlanc et al. (2009) showed similar impairment of endothelium-dependent vasodilation in coronary arterioles 24 hours after inhalation of low lung burdens of titanium dioxide NPs. Such systemic microvascular dysfunction would result in an inability to alter peripheral resistance and increase coronary blood flow during exercise. This would result in an increased workload and decreased oxygen delivery to the heart. In the case of a person with preexisting cardiovascular disease, the failure to modify coronary hemodynamics in response to exercise may precipitate a cardiovascular event.
Possible Mechanisms Explaining Cardiovascular Effects
1. Translocation of NPs to Systemic Tissue
It has been proposed that inhaled NPs may cause adverse systemic effects by escaping normal pulmonary clearance by alveolar macrophage phagocytosis, entering the pulmonary capillaries, and translocating to systemic organs (Oberdörster, Oberdörster, and Oberdörster 2005). Such translocation of NPs versus fine-sized particles would be favored because of their ultrafine size and ability to cross membrane barriers. Oberdörster, et al. (2002) reported that inhaled radiolabeled carbon NPs appeared in the liver of rats 24 hours after exposure. In addition, Nemmar et al. (2002) observed NPs in the blood of human volunteers after inhalation of technetium-labeled carbon NPs. However, these results have been questioned for the following reasons: (1) The Oberdörster, et al. (2002) study involved whole-body inhalation. Thus, oral ingestion during grooming may have resulted in NPs in the liver due to ingestion rather than pulmonary translocation. (2) It is possible that the label dissociated from the NPs in the Nemmar et al. (2002) study. Indeed, inhalation exposure of rats to radiolabeled iridium NPs via a tracheal tube resulted in low translocation (<1%) of NPs from the lung to systemic organs over a 7 day postexposure period (Kreyling et al. 2002). Following pharyngeal aspiration of colloidal gold-labeled SWCNTs in the mouse, no measurable levels of labeled SWCNTs were found in brain, heart, and kidney by neutron activation analysis for gold label (Mercer et al. 2009). Roberts et al. (2009) monitored the translocation of quantum dots from the lung of rats after intratracheal instillation. They reported that translocation to systemic tissue was low at pulmonary doses that did not cause a breakdown in air/blood barrier integrity.
However, morphologic studies support translocation of particulates from the lung to the cardiovascular tissue in necropsy specimens from Mexico City dogs (Calderón-Garcidueñas et al. 2001). In addition, MWCNTs enter the lymphatics, some reach the tracheobronchial lymph nodes, and are even seen within macrophages in the medullary sinuses, suggesting that a percentage may eventually reach the vasculature via the thoracic duct (Hubbs et al. 2009; D. Porter et al. 2010).
Considering the dilution of translocating NPs by the blood volume, concentrations of NPs delivered directly to the cardiovascular tissue are likely to be low. However, the chronic effects of particle accumulation may be important (Calderón-Garcidueñas et al. 2001). In the absence of particle translocation, gradual dissolution of NPs over time, particularly of lung-deposited metal and metal-based NPs, can result in extrapulmonary distribution of the soluble elemental component(s) of the NPs. When this occurs, toxicity may be mediated by the soluble toxic metal. Therefore, the direct action of NPs in systemic organs and the slow dissolution of soluble components may be important in the chronic effects of NPs but is unlikely to explain the rapid, acute, cardiovascular effects following pulmonary exposure described above. This suggests that the mediators for the acute cardiovascular effects may be delivered via the vasculature.
2. Effect of Blood Cytokines on Systemic Tissue
Cytokines can have broad effects on multiple tissues, including both the cardiovascular and nervous system. Blood levels of inflammatory markers (IL-5, IL-6, KC/GROα, and CCL22) were significantly elevated 4 hours after aspiration of MWCNTs in mice (Erdely et al. 2009). These findings suggested that these inflammatory mediators could be carried by the blood to systemic tissue to induce adverse effects, such as those reported by Z. Li et al. (2007). In this study, pulmonary exposure levels of MWCNTs were high enough to cause an 83-fold increase in neutrophils harvested by bronchoalveolar lavage and a 3-fold increase in systemic neutrophils. However, microvascular dysfunction of systemic and coronary arterioles was reported following pulmonary exposure to levels of titanium dioxide NPs that did not increase lavagable or peripheral blood neutrophils (LeBlanc et al. 2010; Nurkiewicz et al. 2008). Therefore, an elevation of blood-borne inflammatory mediators does not appear to completely explain all cardiovascular effects reported following pulmonary exposure to NPs.
3. Activation of Peripheral Neutrophils
Nurkiewicz et al. have shown that pulmonary exposure to particles enhanced adhesion and rolling of polymorphonuclear leukocytes to the systemic microvessels and increases the generation of reactive oxygen species at the vessel wall (Figure 8
) (Nurkiewicz et al. 2006, 2008). Furthermore, treatment of microvascular tissue with reactive oxygen species scavengers or an inhibitor of myeloperoxidase partially reversed the inhibitory effect of pulmonary NPs exposure on microvascular nitric oxide production and resultant vasodilation in response to endothelial stimulation. This may be due to the location of myeloperoxidase between the localized alveolar inflammation at the site of particle deposition, potentiation of neutrophils as they flow through adjacent pulmonary capillaries, adherence of neutrophils to systemic microvascular walls, hemoprotein deposition in the microvascular wall, generation of reactive oxidant species at the microvessel wall, scavenging of endothelial-derived nitric oxide, decreased nitric oxide production, and impairment of nitric oxide-dependent microvascular dilation (Nurkiewicz et al. 2008).

TiO2 NP inhalation causes oxidative stress in coronary arterioles. (A). Representative coronary arteriole from a sham-control rat. (B). Representative coronary arteriole from a rat exposed to 10 µg TiO2 NP (measured lung deposition). Arterioles were incubated with dihydroethidium (DHE, 10–4 M, 20 minutes). Superoxide oxidizes DHE to form ethidium bromide. Ethidium bromide is intercalated into nuclear DNA, and is fluorescent at 480 nm. Note the increased density of fluorescent nuclei in the microvascular wall of the NP exposed rat in B. Differences in dye loading are resolved by measuring background DHE fluorescence at 330 nm and subtracting this image from the ethidium bromide image. The remaining fluorescent light intensity is quantified with image analysis software. Measurements are made 24 hours after NP inhalation. Bar = 50 µm.
4. Activation of Neuronal Input to Systemic Tissue
It is possible that sensory neurons are activated at the site of pulmonary deposition of NPs and that a neuronal signal could be sent to the brain and then to the cardiovascular tissue to alter microvascular function. Evidence to support such a mechanism is from data demonstrating transport of particles from airway sensory nerves (Hunter and Dey 1998; Hunter et al. 2000; Hunter and Undem 1999; Z. Wu et al. 2008). Preliminary data from our laboratories indicate the blockade of neural input to systemic microvascular smooth muscle with tetrodotoxin partially reversed dilatory inhibition in response to pulmonary exposure to NPs (Nurkiewicz et al. 2009). Further experiments are ongoing to elucidate this mechanism.
Central Nervous System Effects
The central nervous system (CNS) is also a potential target for NP toxicity. Following pulmonary deposition, NPs can permeate the lung-blood barrier by endocytosis, transcytosis, or stochastic transport and enter the systemic circulation (Oberdörster, Oberdörster, and Oberdörster 2005). NPs deposited in the lungs have been reported to translocate to the circulatory system and eventually to organs like liver, kidney, and brain (Nemmar et al. 2001; Shimada et al. 2006). Similarly, NPs inhaled through the nose can translocate to the brain via the olfactory neurons (Elder et al. 2006; Hunter and Undem 1999; Oberdörster et al. 2004). In addition, retrograde translocation of fluorescent beads from sensory nerve fibers that project into the airway epithelium of the nose, trachea, and intrapulmonary airways has been documented in experimental animals (Figure 9 ) (Hunter and Dey 1998; Hunter and Undem 1999; Z. Wu et al. 2008). This suggests multiple potential pathways for movement of NPs from the lung to extrapulmonary tissues (Figure 10 ). Calderón-Garcidueñas and coworkers have documented the accumulation of particulates in olfactory nerves, the olfactory bulb of the brain, Kupffer cells of the liver, and also demonstrated NPs in the erythrocytes and endothelial cells in lung capillaries of autopsy specimens from Mexico City residents (Calderón-Garcidueñas et al. 2008). Translocation of NPs from systemic circulation or neuronal transport to the brain can have far reaching consequences by causing irreversible damage to the CNS. Importantly, the limited regenerative capability of the CNS, its cellular heterogeneity and high lipid content may render it more vulnerable to toxic insults.
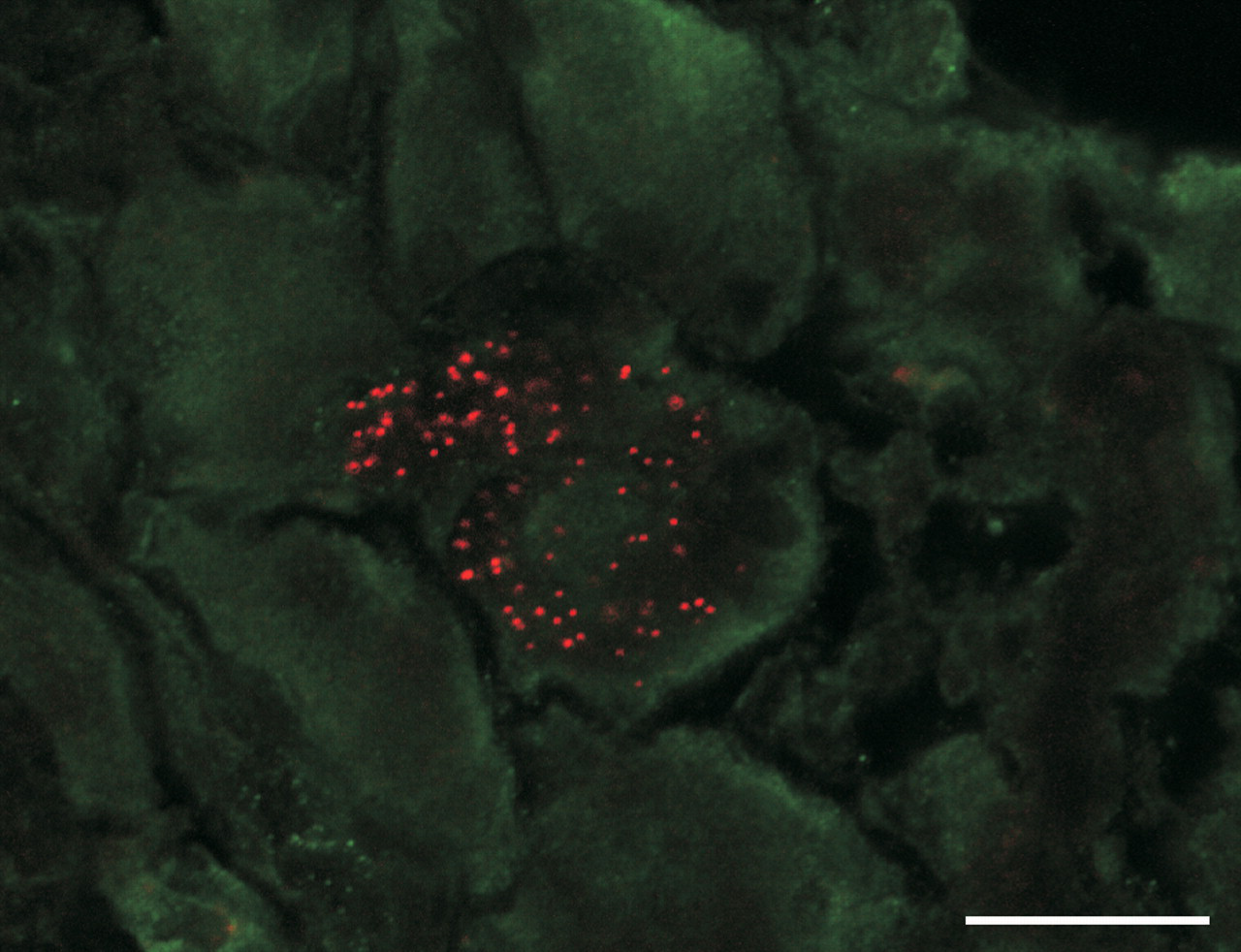
Red rhodamine beads in a neuron cell body of the jugular ganglion demonstrate neuronal transport of particles from the trachea of a rat after intratracheal instillation of fluorescent beads. Bar = 20 µm.
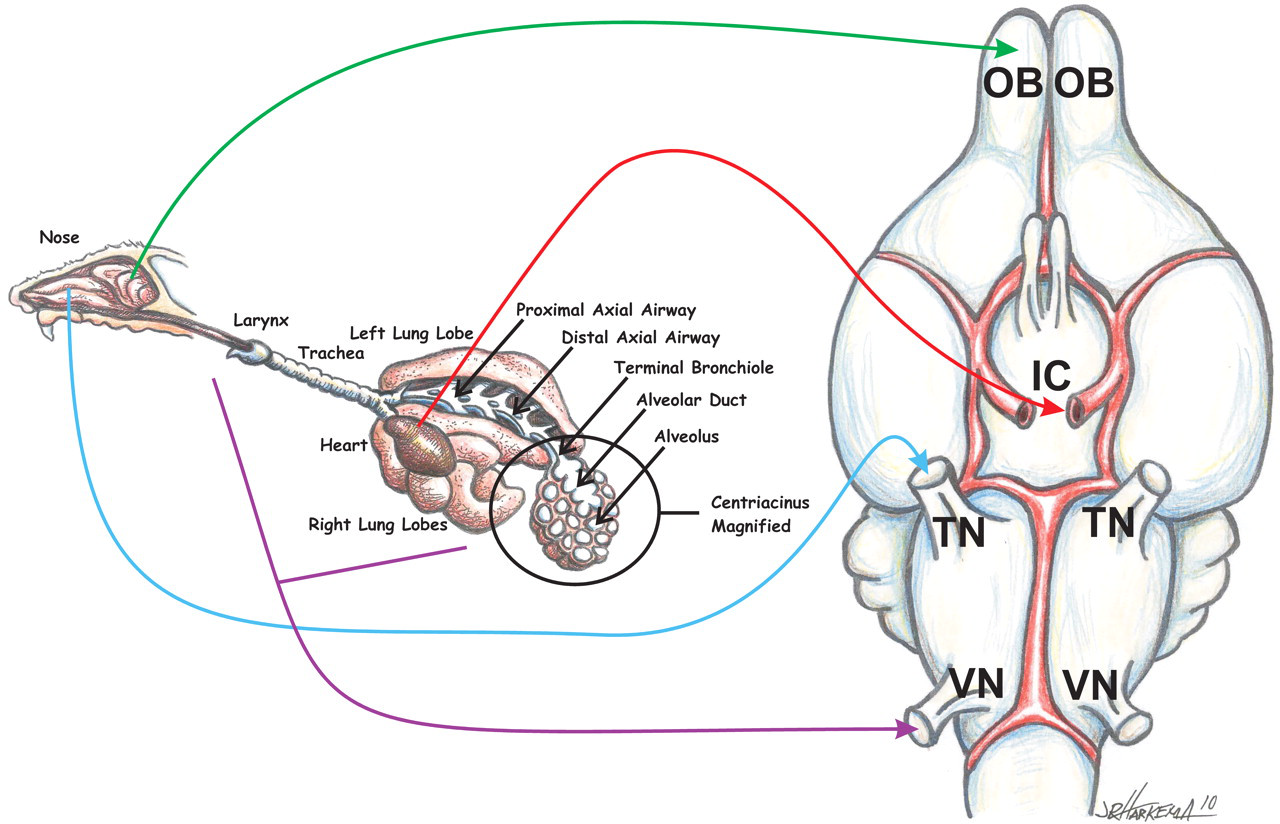
The major potential pathways for inhaled NP transport into the brain. NPs can be carried to the brain via the vasculature, neuronal transport from the olfactory neurons in the olfactory epithelium of the nose and then through the cribiform plate to the olfactory bulb, neuronal transport from sensory nerves of the nasal passageways to the trigeminal nerve, or neuronal transport from sensory nerves of the trachea and intrapulmonary airway to the vagus nerve. ICA = internal carotid artery; TN = trigeminal nerve; OB = olfactory bulb; VN = vagal nerve.
Particle size and/or shape can be a key determinant in the biodistribution of NPs. Several studies have employed model NPs as tools to understand the influence of particle size on kinetics of
The surface chemistry of the NP can also influence its translocation and/or permeability across the BBB. Indeed, NP surface charge has been shown to affect the integrity and permeability of the barrier (Lockman et al. 2004). Utilizing emulsified wax NPs with neutral, anionic, or cationic surface charge, Lockman et al. (2004) evaluated BBB integrity and brain permeability by
Intraperitoneal administration of 25 nm Ag NPs caused alterations in the expression of genes associated with oxidative stress in specific brain areas that resulted in apoptosis and neurotoxicity (Rahman et al. 2009). Subcutaneous administration of Ag NPs was shown to cause BBB changes, neuronal injury, and astrocytic swelling (Tang et al. 2009). Administration of Ag and Cu NPs (50–60 nm) via intravenous, intraperitoneal, or intracerebral routes caused disruption of BBB function and induced brain edema in rats (Sharma et al. 2010). Inhalation of ultrafine manganese oxide resulted in translocation of these particles to the CNS (Elder et al. 2006). Nano-sized TiO2 (Degussa P25) has been shown to trigger oxidative stress response and interfere with mitochondrial function in brain BV2 microglial cells (Long et al. 2006). Administration of 80 nm TiO2 NPs to mice produced subtle brain lesions and vacuolation in hippocampal neurons (J. Wang et al. 2007). Neuroinflammation in discrete brain areas was observed 24 hours following pharyngeal aspiration of titanium dioxide nanowires but not titanium dioxide nanospheres (D. Porter et al. 2008).
Like metal or metal-oxide-based NPs, carbon-based NPs can also potentially elicit neurotoxicity. A few studies have investigated the neurotoxic effects of carbon-based NPs, indicating the involvement of neuroinflammatory and oxidative stress events. Intranasal instillation of ultrafine carbon black induced the expression of proinflammatory cytokines and chemokines in the olfactory bulb (Tin Tin Win et al. 2006). When administered orally, water-miscible 14C60-fullerene has been shown to penetrate the blood-brain barrier (Yamago et al. 1995). C60-fullerenes have also been shown to induce oxidative stress in the brains of largemouth bass (Oberdörster et al. 2004). These findings suggest that NPs can pose a risk for targets sensitive to oxidative stress, like the brain, because of their high iron and lipid content. Thus, carbon-based nanomaterials can pose significant neurological risk following exposure. Indeed, we have observed brain-region-specific mRNA expression of several proinflammatory chemokines, cytokines, selectins, and markers of cellular stress in mice exposed to a single dose of MWCNTs (10–80 µg) by pharyngeal aspiration. Furthermore, MWCNTs decreased the expression of certain BBB-related markers suggestive of altered BBB integrity (Sriram et al. 2009, 2007). Further investigation is required to determine if persistent adverse CNS changes could result from chronic exposure to NPs.
Conclusions
Advances in nanotechnology have produced an explosion of new products, including consumer products, medical devices, and engineered pharmaceuticals. The existing nanotoxicology studies have generally lagged behind the advances in nanotechnology. However, the existing nanotoxicology studies show a general trend for increased toxicity as particulate sizes decrease. The enhanced toxicity is associated with many factors, including increased surface area, ability to pass through cellular barriers, interaction with subcellular structures, and increased ability to activate neutrophils and stimulate the release of inflammatory mediators. Systemic effects of NP exposures are increasingly recognized, particularly in the cardiovascular and neurologic systems. However, enormous opportunities come along with these discoveries and the challenges they present for toxicologic pathologists. Because many NPs are engineered, they can be tools that allow toxicologists and pathologists to explore long-standing hypotheses. Thus, the early fibrosis induced in the lung of SWCNT- and MWCNT-exposed rodents suggests the need to consider new mechanisms for fibrosis. The ability to add labels to NPs presents the opportunity to follow the distribution of some NPs within different tissues and subcellular structures within those tissues. The ability to engineer NPs within specific dimensions allows the detection of critical dimensions for producing interaction with, and penetration of, cellular structures. Finally, merging advances in molecular pathology with an understanding of nanotechnology allows detection of surprising effects sometimes seen in NP studies, including disruption of mitosis, lymphangiectasia, changes in sensory nerves, and disruption of the blood-brain barrier. Toxicologic pathologists can play an important role in recognizing the hazards of NPs so that health risks are reduced as the opportunities of nanotechnology are realized.
Footnotes
Acknowledgment
We thank Richard D. Dey, PhD, for advice on the figures.
