Abstract
The Scientific and Regulatory Policy Committee of the Society of Toxicologic Pathology (STP) appointed a working group to address risk assessment for increases in alveolar macrophages following inhalation of pharmaceutical materials. This position paper provides recommendations for inhalation study–specific terminology and interpretation based on literature and information from marketed inhaled drugs. Based on a weight-of-the-evidence approach, and with appropriate consideration of the physical and pharmacological characteristics of the compound, uncomplicated increases in the size or number of alveolar macrophages in nonclinical species are interpreted as nonadverse.
Keywords
Introduction
Pulmonary alveolar macrophages, which are normal resident cells, respond to changes in the local micro environment. Responses by alveolar macrophages may represent normal tissue adaptations that maintain normal lung function or may contribute to pathophysiological processes with potentially adverse sequelae. Currently, there are no generally accepted criteria by which to distinguish nonadverse versus adverse findings that include increases in alveolar macrophages when observed in inhalation studies of pharmaceutical materials. The absence of established or standardized morphological nomenclature, or inconsistent application of nomenclature, further contributes to lack of clarity in interpreting alveolar macrophage responses for human risk assessment. The lack of accepted criteria and clarity fosters a conservative approach to risk assessment of increased alveolar macrophages in inhalation studies. An overly conservative approach to identifying the no-adverse-effect-level (NOAEL) with regard to macrophage increases can have an undue impact on the ability to dose escalate to efficacious levels in the clinic, thereby affecting overall inhalation drug development success or timelines. Therefore, it is imperative to establish which findings represent normal adaptive responses and to distinguish adaptive responses from those having potentially adverse consequences.
A working group was established to review currently available data regarding alveolar macrophage responses to inhaled pharmaceuticals in toxicity studies and to evaluate parameters used for risk assessment. The working group had five objectives: Recommend standardized nomenclature to characterize increases in pulmonary alveolar macrophages and any associated histomorphological findings in inhalation preclinical safety studies. Integrate current understanding of alveolar macrophage responses to inhaled particles, including inert, insoluble materials, and marketed inhaled drugs, in the interpretation of pathologic findings and determination of a relevant NOAEL. Evaluate the potential for clinical pathology in preclinical studies to contribute to risk assessment for increased alveolar macrophages and provide recommendations, where appropriate, for clinical pathology end points in inhalation studies. Address the functional implications of the alveolar macrophage response to inhaled materials. Does this response have an effect on pulmonary function, bacterial or viral clearance, or other physiological parameters? Recommend points to consider in determining the NOAEL when increased alveolar macrophages are observed in rodent inhalation studies.
This article, which addresses these five objectives, focuses on alveolar macrophage responses in rats. The implications of the various types of macrophage responses have been studied in more depth for rats than other species, and rats tend to be as sensitive to the effects of inhaled particles as other rodent and nonrodent species, although in some cases, rats are more sensitive (Bermudez et al. 2002; Hext 1994). For example, rats have a greater propensity to develop chronic inflammation and an increased susceptibility to lung tumor formation than hamsters or mice when exposed to large amounts of poorly soluble particles such as titanium dioxide (TiO2; Bermudez et al. 2002). Rats chronically exposed to coal dust or diesel exhaust (DE) have a greater tendency than nonhuman primates to accumulate macrophages in alveolar lumens and, at high exposure concentrations, to develop alveolar epithelial hyperplasia, fibrosis, and alveolar proteinosis (Nikula et al. 1997). The preponderance of rat data is attributed to technical capabilities that allow more efficient and cost-effective exposure of multiple rats for extended periods as compared to nonrodent inhalation procedures. Additionally, greater doses or dose ranges are typically achieved in rodent than nonrodent studies because dose is largely determined by time (duration) of exposure for any given exposure concentration of respirable material (Pauluhn 2008). Nevertheless, similar considerations may be applicable to terminology and interpretation for nonrodent studies. Additionally, although this article focuses on increases in alveolar macrophages observed in preclinical studies of inhaled pharmaceutical compounds, inhaled chemical substances with similar physiochemical characteristics may induce similar effects and the proposed terminology and points to consider in risk assessment may be applicable to nonpharmaceutical materials.
Objective 1: Recommended Terminology
The INHAND Project (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice) published nomenclature for classifying proliferative and nonproliferative changes in the respiratory tract of laboratory mice and rats (Renne et al. 2009). The INHAND terminology proposed the term alveolar macrophage aggregation and further stated that alveolar histiocytosis and alveolar phospholipidosis represent synonyms. However, as acknowledged by Renne et al. (2009), alveolar histiocytosis has been used to describe foci of alveolar macrophages as well as mixtures of inflammatory cells in which macrophages predominate, macrophage accumulations located within predominately fibrotic foci, or foci of macrophages in alveoli with accompanying type II cell hyperplasia (Dungworth et al. 1992; Boorman and Eustis 1990). In preclinical studies of pharmaceuticals, the term alveolar phospholipidosis is reserved for accumulations of phospholipid-containing macrophages observed after treatment with cationic amphiphilic drugs regardless of the route of administration (Halliwell 1997; Hook 1991). The INHAND terminology further described alveolar macrophage aggregates as occurring after inhalation of cytotoxic materials and toxicants that were associated with necrosis of pulmonary parenchyma.
The recommendations in this article are suggested as a refinement to the INHAND nomenclature because the INHAND nomenclature did not directly address terminology applicable to uncomplicated increases in the numbers of alveolar macrophages in response to inhaled materials nor did the nomenclature specifically address inflammation that might accompany macrophage responses. The terminology proposed herein provides specific recommendations for use in inhalation studies of pharmaceuticals and serves to distinguish findings of increased numbers of alveolar macrophages lacking evidence of cytotoxicity or inflammation from findings complicated by inflammation or associated with other changes in the pulmonary parenchyma. These recommendations are based on the assumption that the preferred terminology would allow the pathologist to draw distinctions within data summary tables between nonadverse, adaptive increases in alveolar macrophages and other changes deemed potentially adverse.
Macrophages
Recommended Term
“Increased alveolar macrophages” is the suggested term, followed by designation of the predominant location (e.g., centriacinar, subpleural, etc.). Further details can be supplied in the report narrative as necessary. Where the severity is minimal, and the predominant subanatomic location is less obvious, the locator may simply be omitted. It is suggested to reserve the terms focal/multifocal for changes that are isolated and occur in apparently random locations, as opposed to changes that occur at a consistent subanatomic location. If the macrophages from treated animals have cytoplasmic changes (pigment, vacuolation, foreign material, etc.) that are distinct from features in controls (historical or current), these changes should be part of the diagnostic term. For example, increased alveolar macrophages; centriacinar, pigmented, mild. Other descriptors, for example, foamy/granular cytoplasm or presence of bi- to multinucleated cells may be entered as “free text” and would generally be addressed in the report narrative. The term increased alveolar macrophages also pertains to any increases in interstitial macrophages that might accompany “increased alveolar macrophages.” The affected compartments would be addressed in the report narrative. Increased interstitial macrophages would only be diagnosed separately if the pulmonary interstitium represented the predominant location.
Thresholds
The decision whether to apply a “threshold” for recording alveolar macrophages during evaluations on an inhalation study ultimately resides with the study pathologist. The recommendation here is that the finding of increased alveolar macrophages would apply to all instances where macrophage accumulation is over and above the expected, randomly scattered individual macrophages within the alveolar spaces. Increases in alveolar macrophages, as perceived by the experienced pathologist relative to the expectation for low numbers of scattered individual macrophages in young, “normal” animals, should be recorded, whether observed in a naive or vehicle control group or in a group exposed to the test article.
The rationale for avoiding use of a threshold in these circumstances includes the following: When present in controls (commonly the case, particularly at greater study duration and/or in older animals), the recording of increased alveolar macrophages documents that this change is part of a normal adaptive process and (in the absence of secondary changes) would not be considered adverse. If the control range for a particular study is used to set a threshold above which changes in treated animals are recorded, subsequent use of historical data may be limited. In other words, one may not be able to place proper context for subsequent studies if the control database contained very few findings. The lack of historical data would become important for small studies where the controls have no recordable changes, but a treated group does have findings. A higher incidence of increased alveolar macrophages in treated animals as compared to controls, but with a severity below the threshold for recording, cannot be captured objectively if a threshold is used. Avoiding a threshold level may be helpful in circumstances where pathology data will be reviewed by a third party such as a pathology working group. Discrepancies between the original data and that of the reviewing pathologists would be less likely.
Accompanying Changes
It is preferable to use “increased alveolar macrophages” as a stand-alone diagnosis and not as an umbrella term that includes other inflammatory, degenerative, or proliferative responses. This will allow a nonadverse level of findings to be readily recognizable within tabulated data such as summary incidence tables. For example, there may be cases where increased alveolar macrophages is the only finding at lower doses, but other changes in addition to increased alveolar macrophages are present at higher doses. The other changes would be entered separately and an association to increased macrophages, if any, could be made by the pathologist in their report narrative. Such changes might include one or more of the following terminologies: Inflammation (i.e., presence of inflammatory cells); Debris (free in alveoli, origin not specified); Edema; Alveolar proteinosis; Type II pneumocyte hyperplasia (or hypertrophy); Necrosis of bronchiolar epithelium; Bronchiolar epithelial cell hyperplasia/hypertrophy at the bronchiolar–alveolar junction; Squamous metaplasia (alveolar or bronchiolar epithelium); Fibrosis; Lymphoid hyperplasia; Perivascular infiltrates; Pleural hyperplasia/hypertrophy.
Whether one or several of the above terms are used (i.e., “lumping” vs. “splitting”) should be at the discretion of the pathologist. Factors to consider include the following: If one diagnostic term is used, ensuring that the distribution and/or the level of tissue change is sufficiently consistent across the various findings to allow determination that they constitute a single process. Where this is not the case, separate terminology should be applied. Similarly, if the presence or absence of certain features will be a determinant of adverse versus nonadverse changes, recordings should be separated into groups or categories that allow at least this distinction to be made in the data summary tables.
When increases in alveolar macrophages are consistently accompanied by other changes such as inflammation and type II pneumocyte hyperplasia, it is often most appropriate to use a single term, such as chronic inflammation, and not to enter increased alveolar macrophages as a separate finding. In this situation, inflammation, not increased alveolar macrophages, would be the umbrella term. However, in situations where the inflammation is not clearly colocated with the increase in alveolar macrophages, it may be more appropriate to enter findings of increased alveolar macrophages and have a separate recording for inflammation.
Other Incidental Lung Changes That May Occur in Inhalation Studies
Terminology used to record incidental lung changes may make data tables difficult to interpret in the context of nonadverse increased alveolar macrophages alone versus a situation such as adverse increased alveolar macrophages complicated by inflammation or additional changes. For example, a common incidental change in rodents is small focal areas of alveoli containing some or all of the following: hemorrhage, crystalline material, macrophages, neutrophils, pyknotic debris, and type II pneumocyte hyperplasia. These features are often, but not exclusively, encountered in animals subjected to inhalation of anesthesia during the course of the study. Acute hemorrhage may occur during anesthesia immediately prior to death. These changes are discussed here because incidental findings should not be confused with some of the changes listed above in association with increased alveolar macrophages. These incidental findings are usually distinguishable based on their random distribution, and the fact that macrophages typically do not exhibit foamy cytoplasm and are not seen in large numbers or as the predominant component of the change. Other incidental findings include histiocytic inflammation or granulomas secondary to aspiration of feed or bedding materials. The pathologist must be alert to these differentiating features and use alternate terminology as appropriate. Unless appropriate terminology is used to distinguish these nontest article–related findings, data entries could be confused with secondary degenerative changes or inflammation related to test article–induced increased macrophages and lead to an inappropriate interpretation of recordings. Specific practices differ among pathologists with regard to terminologies used for incidental findings. Terminologies must allow one to distinguish test article effects, including nonadverse or adverse responses, from incidental changes. The pathologist should make every attempt to clarify these differences within the narrative of the pathology report and provide logic for the interpretations.
Objective 2: Current Understanding of Responses to Inhaled Particles and Marketed Inhaled Drugs
Sources of Increased Alveolar Macrophages in Response to Inhaled Particles
A critical function of the alveolar macrophage is to maintain alveolar regions of the lung free of inhaled particles and foreign matter. Macrophages, in order to be effective, need to respond rapidly to an increased burden of particulate matter, whether this increased burden is pronounced and sudden or is introduced gradually over a protracted period. In general, there is a direct relationship between the particulate load reaching the alveoli and the accumulation of alveolar macrophages within terminal air spaces (Bowden 1984; Brain 1971).
It has generally been thought that alveolar macrophages are ultimately derived from monocyte precursors in the bone marrow. Some monocytes were thought to migrate directly from the circulation into the alveolar lumen, while others were thought to take up residence in the alveolar interstitium with a small proportion of the cells in the interstitium continuing to divide. Several studies suggested that under basal conditions, the majority of alveolar macrophages came from direct influx of circulating monocytes from venules, which occurred fairly rapidly, while only a minor component came from the resident interstitial cells that migrated into alveoli (Bowden 1984; Pérez-Arellano, Alcázar-Montero, and Jiménez-López 1990; Laskin, Weinberger, and Laskin 2001). More recent investigations suggest that under steady state conditions, lung macrophages proliferate locally in adult animals with minimal contribution from circulating monocytes (Hashimoto et al. 2013; Yona et al. 2013; Holt et al. 2008).
Under conditions of a pronounced continuing and sudden particle burden (intratracheal instillation), alveolar macrophages originate both from circulating monocyte pools and from interstitial mononuclear cell pools, which leads to a biphasic response by macrophages (Bowden 1984). High particle loads can induce substantive increases (as much as 10-fold) of alveolar macrophages within 24 hr, prior to any increase in division of interstitial macrophages, due to newly migrating monocytes from the circulation. Later, as the stimulus for macrophages continues, the mitotic activity of mononuclear cells in the interstitium increases. Migration of these proliferating interstitial cells into the alveolar lumen is responsible for maintaining numbers of alveolar macrophages over extended time periods. It should also be noted that free macrophages in the alveolar lumen can divide, although such events are relatively rare (Bowden 1984; Lohmann-Matthes, Steinmüller, and Franke-Ullmann 1994). Both the earlier and more recent studies show that the macrophage population is not a finite pool but, instead, can be increased or replenished when challenged or depleted (Bowden 1984; Hashimoto et al. 2013; Yona et al. 2013; Holt et al. 2008).
Alveolar Macrophage Increases as a Nonspecific Effect of Poorly Soluble Drugs: Adverse versus Nonadverse Responses Based on Published Inert Particle Data
When inert, “nontoxic,” insoluble particles of respirable size reach the alveoli, these particles are removed from the alveolar spaces by macrophages, which then translocate either to local lymph nodes or to airways where mucociliary action ultimately eliminates these materials from the lung. These mechanisms normally remove inhaled environmental dusts, and depending on the particle burden, the macrophage response may be noted histologically. Clusters of macrophages, presumably performing this function, are occasionally observed in control animals, including rats, dogs, and nonhuman primates, when the lungs are examined histologically in toxicology studies, and such findings are not associated with notable tissue destruction or adverse effects (Sato, Doi, Kanno, et al. 2012; Sato, Doi, Wako, et al. 2012; Chamaza et al. 2010). However, if present in sufficient quantity, undissolved material may evoke responses by alveolar macrophages that are associated with secondary inflammatory responses and/or tissue damage. 1,2
In a similar manner, when inhaled particulate pharmaceuticals are dosed at high levels in toxicology studies, large amounts of particulate material reach the alveolar spaces. Historically, inhaled drugs tended to be relatively soluble, and accumulation of insoluble material in the lungs of preclinical species was minimal. Consequently, increases in alveolar macrophages related to lung particle burdens were generally not observed. More recently, attempts to minimize systemic exposures to inhaled pharmaceuticals and maximize efficacy in the lung have yielded newer drug candidates with poor solubility and greater propensity to accumulate in lungs when dosed at high concentrations in toxicology studies. The macrophage response in the lungs under these circumstances has typically been interpreted by sponsors and regulators alike to be independent of any chemically or pharmacologically mediated effects of the drug. Similar responses are typically not observed in vehicle control animals because the vehicles used (such as lactose) are readily soluble and do not persist in particulate form within the alveolar spaces. The challenge for regulators and for those engaged in pharmaceutical development is to appropriately interpret nonspecific macrophage responses to poorly soluble drugs and arrive at meaningful risk assessment for conduct of clinical studies. The pathologist must also consider the possibility of a pharmacologically mediated macrophage response when knowledge of the target or solubility and predicted lung burden data do not support a nonspecific effect related to particle burden.
Fortunately, numerous published (Lee et al. 1986; Driscoll et al. 1996; Warheit et al. 1997; Elder et al. 2005) rat inhalation studies using inert, insoluble particles demonstrate alveolar macrophage responses similar to those noted with inhaled pharmaceuticals. These studies can be used as models that show the development, progression, and reversibility of changes in alveolar macrophage distribution and/or number. These studies were usually conducted to elucidate mechanisms of lung “overload” or carcinogenesis in rats receiving very high doses of particles. However, some studies also used lower doses that provide useful data to demonstrate nonadverse adaptations, which consist of increased alveolar macrophages in the absence of inflammation or other adverse changes, even after lifetime (2-year) dosing. In many cases, the alveolar macrophage increases seen in pharmaceutical inhalation toxicology studies will have an identical pathogenesis (i.e., caused by the particle burden of poorly soluble drugs). Regardless of the drug or chemical causing the response, the data show that development and outcome of the alveolar macrophage increases are well documented and understood. When present at low levels of severity, it is reasonable to interpret them as nonadverse, based on their small magnitude, origin in a physiological adaptation process, presence of qualitatively similar changes in occasional control animals, lack of clinical effects, lack of evidence of accompanying inflammation or tissue destruction, lack of progression (even in 2-year studies), and their reversibility.
An important difference between the situation with inhaled pharmaceuticals and models using insoluble material is that even following extended “recovery periods,” at the higher doses used in the models using insoluble material, essentially all the inhaled material is still present and exerting its effects in the lung (e.g., Driscoll et al. 1996). This is because the only removal mechanism is via alveolar macrophage activity, which is relatively slow. Even for small amounts that do not overload macrophages, the half-life is approximately 100 days in the rat lung (Oberdorster, Ferin, and Morrow 1992). In contrast, for drugs, even those that are poorly soluble, the solubility is such that dissolution is still the predominant means of particle removal from the lung at the relatively low amounts administered (Jones and Neef 2012). The reversibility data from inert particle studies is therefore only relevant at the lowest doses, where there is appreciable removal of particulate material over the course of the recovery period. Even in these cases, this would be a worst-case scenario for reversibility when applied to an inhaled pharmaceutical, since the macrophage stimulus would persist for much longer in the case of insoluble particles (Jones and Neef 2012). For these reasons, it is important not to assume that reversibility times for changes resulting from inhaled pharmaceuticals would parallel those for completely insoluble particles.
The studies listed below describe the full range of histopathological responses of rat lung to administration of insoluble materials considered to have low inherent toxicity: TiO2, carbonyl iron (CI), or carbon black. In all cases, the particle size was adequate to reach the alveolar spaces when inhaled.
Warheit et al. (1997) showed that rats exposed to inhalation of low concentrations of TiO2 and CI (5 milligrams per cubic meter [mg/m3]) for 4 weeks had a minimal diffuse increase in alveolar macrophages that was fully reversible within 1 week after the end of the exposure. Higher concentrations (50 and 250 mg/m3) produced severe macrophage increases, including dense aggregates of particle-laden macrophages in the alveoli and alveolar ducts, hyperplasia/hypertrophy of epithelial cells in the alveoli and alveolar ducts in association with macrophage aggregates, and neutrophils in lavage fluids. These changes at the higher concentrations were not reversed following a 6-month recovery period.
Elder et al. (2005) described similar findings for administration of carbon black particles by inhalation to rats for 13 weeks (1, 7, and 50 mg/m3), with evaluations at the end of the treatment period and after 3- and 11-month recovery periods. There were essentially no findings (other than the visible presence of the inhaled material within macrophages) at the lowest dose. At the mid dose, there was accumulation of hypertrophic, vacuolated macrophages containing carbon black in the alveolar ducts and adjacent alveoli, with occasional cell debris, minimal inflammation, and local epithelial hyperplasia. The inflammation and epithelial changes were reversible by 3 months postexposure (although particle-containing macrophages remained). At the highest dose, there was more extensive macrophage accumulation, degeneration, and necrosis, local neutrophil inflammation, cell debris, hyperplasia/hypertrophy of type II pneumocytes, and alveolar proteinosis. Interstitial and alveolar fibrosis developed during the recovery period in the high-dose group. Small foci of epithelial metaplasia were associated with fibrotic areas. The material persisted in the lung and the changes were still present at the end of the recovery period. Body weights were not significantly affected at any dose; lung weights were significantly increased at the highest dose only.
Driscoll et al. (1996) exposed rats to carbon black by inhalation for 13 weeks (1.1, 7.1, and 52.8 mg/m3) with recovery periods of 3 and 8 months. At the lowest dose, accumulation of particle-containing macrophages around the alveolar ducts and adjacent alveoli was the only change observed. The mid dose produced the same change, accompanied by neutrophils and macrophages within alveolar spaces, local epithelial hyperplasia, and mild interstitial fibrosis (the latter apparent on trichrome-stained sections). The highest dose produced similar, but more severe changes, often associated with alveolar ducts. There was an increase in fibrosis at the end of the recovery period. There was no reduction in the lung burden of particles over the recovery period for the mid- and high-dose animals, and no reversibility of the changes.
Finally, Lee et al. (1986) administered TiO2 particles to rats by inhalation for 2 years (10, 50, and 250 mg/m3). At the lowest dose, exposure resulted in a small increase in clusters of alveolar macrophages adjacent to the alveolar ducts, with slight local hyperplasia of type II pneumocytes and increased macrophages in local lymphoid tissue. Over the 2-year period, there were no changes in body weight, clinical signs, morbidity, or mortality compared with untreated animals, and no evidence of any adverse effect on the respiratory system. Thus, a dose sufficient to induce slight type II pneumocyte hyperplasia in association with accumulation of alveolar macrophages over the lifetime of the animal, including in old age when susceptibility and intercurrent disease might be expected to magnify any compromise related to the lung changes, did not compromise health and did not cause adverse sequelae in the lungs of treated rats.
The changes described in the above studies at the lower doses of inert, insoluble material are typical of findings commonly seen at the highest doses in inhalation toxicology studies of poorly soluble pharmaceuticals in rats. It is therefore reasonable to conclude that changes in macrophage distribution, appearance, and/or number, centered on the alveolar ducts but with minimal or no changes in the adjacent epithelium and without appreciable inflammation, 3 represent the same nonspecific adaptive response as observed with inhalation of inert particles, and would be expected to have the same benign course, even if present for the lifetime of the animal. Inhalation toxicology data for 4 poorly soluble inhaled pharmaceutical development candidates were recently reviewed (Jones and Neef 2012). The lung particulate burden achieved for each molecule was estimated. The results indicated that regardless of the duration of the study, estimated lung burdens greater than approximately 1 mg/g lung were associated with adverse changes in the lung. Lung burdens of approximately 0.5 mg/g (estimates of 0.1–1.0 mg/g) appeared to represent the point of transition between adaptive and adverse changes in the lung. This estimate is similar to the lung burdens of insoluble particles associated with adverse changes (Lee et al. 1986; Driscoll et al. 1996; Warheit et al. 1997; Elder et al. 2005) supporting the conclusion that the pathogenesis of the observed changes was related to low solubility and high lung particle burdens.
Summary of Alveolar Macrophage Increases in Preclinical Studies with Pharmaceuticals
A search of the Physician’s Desk Reference (PDR®3D™ Digital Drug Database 2012) and public domain data on the Food and Drug Administration’s (FDA) website (Drugs@FDA: FDA Approved Drug Products) revealed that the finding of increased alveolar macrophages was documented in inhalation toxicology studies supporting the registration of Advair (fluticasone/salmeterol), Alvesco (ciclesonide), Arcapta (indacaterol maleate), Asmanex (mometasone), Dulera (mometasone/formoterol), Foradil (formoterol), Pulmicort (budesonide), Serevent (salmeterol), Tilade (nedocromil sodium), Tobi Nebulizer (tobramycin), Tudorza (aclidinium bromide), Tyvaso (treprostonil sodium), and Relenza (zanamivir; Table 1). Most of these products have been used in humans for many years. These products are used in subjects with preexisting lung diseases, that is, potentially “susceptible populations,” and none of these products have been removed from the market due to adverse effects, thus supporting the position that observation of uncomplicated increases in macrophages in preclinical studies does not predict unacceptable risk in humans.
U.S. marketed or recently discontinueda inhalation drugs and occurrence of drug-related alveolar macrophages.
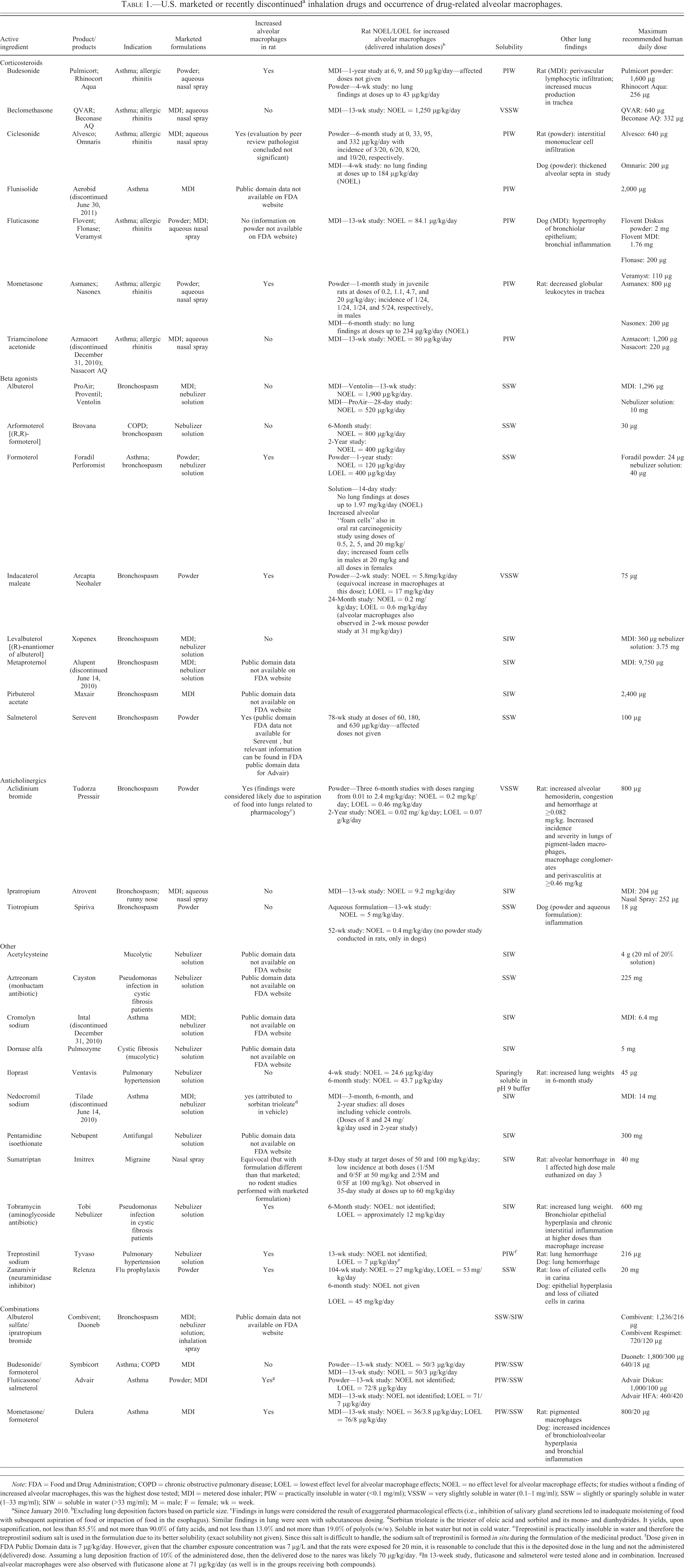
Note: FDA = Food and Drug Administration; COPD = chronic obstructive pulmonary disease; LOEL = lowest effect level for alveolar macrophage effects; NOEL = no effect level for alveolar macrophage effects; for studies without a finding of increased alveolar macrophages, this was the highest dose tested; MDI = metered dose inhaler; PIW = practically insoluble in water (<0.1 mg/ml); VSSW = very slightly soluble in water (0.1–1 mg/ml); SSW = slightly or sparingly soluble in water (1–33 mg/ml); SIW = soluble in water (>33 mg/ml); M = male; F = female; wk = week.
aSince January 2010. bExcluding lung deposition factors based on particle size. cFindings in lungs were considered the result of exaggerated pharmacological effects (i.e., inhibition of salivary gland secretions led to inadequate moistening of food with subsequent aspiration of food or impaction of food in the esophagus). Similar findings in lung were seen with subcutaneous dosing. dSorbitan trioleate is the triester of oleic acid and sorbitol and its mono- and dianhydrides. It yields, upon saponification, not less than 85.5% and not more than 90.0% of fatty acids, and not less than 13.0% and not more than 19.0% of polyols (w/w). Soluble in hot water but not in cold water. eTreprostinil is practically insoluble in water and therefore the treprostinil sodium salt is used in the formulation due to its better solubility (exact solubility not given). Since this salt is difficult to handle, the sodium salt of treprostinil is formed in situ during the formulation of the medicinal product. fDose given in FDA Public Domain data is 7 µg/kg/day. However, given that the chamber exposure concentration was 7 µg/L and that the rats were exposed for 20 min, it is reasonable to conclude that this is the deposited dose in the lung and not the administered (delivered) dose. Assuming a lung deposition fraction of 10% of the administered dose, then the delivered dose to the nares was likely 70 µg/kg/day. gIn 13-week study, fluticasone and salmeterol were tested alone and in combination. Increased alveolar macrophages were also observed with fluticasone alone at 71 μg/kg/day (as well is in the groups receiving both compounds).
For Tilade, the increased alveolar macrophages were attributed to sorbitan trioleate (tri-ester of oleic acid) in the vehicle and not to the active ingredient, nedocromil sodium. For the remaining drugs, all but one (tobramycin), showed poor solubility in water (i.e., “practically insoluble in water” to “sparingly soluble in water”). Tobramycin (Tobi nebulizer solution), like other aminoglycoside antibiotics, has been associated with phospholipidosis (Josepovitz et al. 1986) and this may explain the increase in alveolar macrophages observed with this compound. Although alveolar macrophages were observed with aclidinium bromide (Tudorza Pressair), the macrophages were accompanied by hemosiderin, congestion, hemorrhage, and perivascular infiltrates that together were attributed to aspiration as a result of exaggerated pharmacological effects (i.e., inhibition of salivary gland secretions led to inadequate moistening of food with subsequent aspiration of food or impaction of food in the esophagus). These data provide evidence that the pathogenesis of the increased alveolar macrophages for the compounds in Table 1, except aclidinium, nedocromil sodium, and tobaramycin (for the reasons described), is likely a nonspecific effect related to the burden of undissolved particles in the lung.
Objective 3: Contribution of Clinical Pathology
Review of Current Clinical Pathology Measurements in Standard Inhalation Studies
Inhalation toxicology studies typically evaluate standard parameters for hematology, coagulation, clinical chemistry, and urinalysis, similar to evaluations conducted on general toxicology studies. Time points for evaluation may include pretest collection in large animal studies, during or at end of the dosing phase, and during the recovery phase. Among marketed inhaled compounds known to elicit increased alveolar macrophages in preclinical toxicity studies in rodents, and for compounds that occasionally elicit changes in nonrodent species (isolated reports of bronchioalveolar cell hyperplasia rather than increased alveolar macrophages), no compound was identified that was associated with meaningful alterations or effects on any standard clinical pathology parameter during the inhalation study (Heyder et al. 2009). Similarly, there were no changes in standard clinical pathology parameters indicative of altered respiratory function, or related to pulmonary injury, or suggestive of an inflammatory process when increased alveolar macrophages were reported histologically in the absence of additional tissue changes in the lung.
Comments Regarding Other Parameters Evaluating Cellular Responses and Mediators in the Lung
Given the current standard of practice for conduct of inhalation toxicology studies, it is unlikely that nonroutine clinical pathology parameters would provide information beyond that provided by histopathology that could be relied upon to determine adverse versus nonadverse effects and assess risk. Nonetheless, in circumstances where satellite groups of animals are available or for short-term studies conducted to pursue mechanistic end points related to effects of the test article, alternative evaluations might be considered. The examples mentioned below, including evaluations of bronchioalveolar lavage (BAL) for cell counts, protein profiles, or targeted mRNA analyses, may provide additional information with regard to time course of tissue responses or associated cellular responses in the lungs. However, these are not expected to affect the determination of adverse versus nonadverse based on anatomic pathology criteria.
The analysis of BAL fluid in rats following administration of insoluble materials such as TiO2, CI, and carbon black (Warheit et al. 1997; Driscoll et al. 1996) was indicative of an inflammatory cell response at doses where inflammation was observed histopathologically. BAL analytes indicative of inflammation were not present at the lowest doses where uncomplicated increases in macrophages were observed microscopically. In a recent inhalation study of carbon nanofibers (DeLorme et al. 2012), rats were exposed to 0, 0.50, 2.5, or 25 mg/m3 of carbon nanofibers for 90 days. Minimal inflammation of the terminal bronchioles and alveolar ducts was observed microscopically at the 2.5 mg/m3 level, but there were no changes in BAL total cells, differential cell counts, or in BAL chemistry analytes. Only at 25 mg/m3, where microscopic changes increased to slight inflammation of terminal bronchioles and alveolar ducts with hypertrophy/hyperplasia of type II cells, BAL differential cell counts and chemical analyte levels were different from controls. In view of the studies cited above (Warheit et al. 1997; Driscoll et al. 1996; DeLorme et al. 2012), it is doubtful that analysis of BAL fluid in inhalation studies with uncomplicated accumulation of alveolar macrophages would add additional information beyond that provided by histopathological evaluation of the lungs (Henderson 2005). However, when designing more targeted inhalation studies to investigate pathological findings, analyses of BAL collected by standardized experimental procedures may be considered when an inflammatory response or local tissue response in the lungs is anticipated. Parameters that could be assessed in the BAL fluid include following: BAL total cell count and differential: determination of total and differential cell count to provide an assessment of the cell population obtainable by lavage. Cytological evaluation of a stained preparation (typically cytospin preparation) provides a differential count and excellent morphologic detail of macrophages. Macrophages containing phagocytized material often can be identified and the percentage of affected macrophages can be determined. Note that the majority of the cells in a typical BAL sample are macrophages, so increased percentage of alveolar macrophages is not reported per se. Instead, the total cell count in the BAL preparation provides information on overall cellularity of the nucleated cell population that can be lavaged from the bronchoalveolar space so that an increase in the overall number of macrophages can be appreciated. BAL preparations can detect the percentage and absolute numbers of other cell types such as neutrophils and eosinophils. Because of the increased interest for macrophage differentiation, additional characterization such as detection of cellular markers by flow cytometry and analysis of mRNA expression could be considered to help further characterize the inflammatory cell types from BAL. BAL total protein: determination of total protein could provide information on the degree of any inflammatory response in the bronchoalveolar space. An increase in BAL total protein would typically be observed with an inflammatory process or altered permeability of microvasculature in alveolar walls. BAL protein electrophoresis: protein electrophoresis to determine the protein fractions (α, β, and γ). This characterization of lavage protein may be useful when the total protein is increased. BAL biomarkers: analytes such as acute phase proteins, interleukins, and interferons could be measured in the BAL fluid to further characterize lung responses. Measurement of these biomarkers could provide support for an inflammatory process within the lung, and in combination with the evaluation of serum biomarkers and comparison of the immune–inflammatory profile, these could characterize the type of response observed in the lung.
Currently, to the best of the authors’ knowledge, there are no specific or validated biomarkers from analysis of BAL that identify early physiopathological changes associated with increases in alveolar macrophages prior to the occurrence of morphological changes in the lung and that could be relied upon to determine an adverse versus nonadverse designation.
Objective 4: Functional Implications of Increased Alveolar Macrophages
Assessment of Lung Functional Parameters
Pulmonary function parameters are routinely measured during preclinical inhalation studies. However, there is a paucity of published pulmonary function test (PFT) data from preclinical evaluations of pharmaceutical formulations. Of the products listed in Table 1 that had a preclinical alveolar macrophage response, PFTs were reported for one 13-week rat study conducted with Dulera (or its components) and for studies with Tyvasco. Slight reductions in minute and tidal volume were observed in animals that received mometasone furoate alone or in combination with other components. The overall effect was attributed to body weight loss in those exposure groups. In the Tyvaso studies, a decrease in minute volume was observed at 6 and 13 weeks. Decreased minute volume was not mentioned as adverse in the FDA’s summary basis of approval, perhaps because it was observed in the supporting acute dose safety pharmacology study. These studies do not provide evidence that an uncomplicated macrophages response would have an effect on minute volume because in addition to potential pharmacologic effects on minute volume, the pulmonary macrophage response in rats that inhaled Tyvasco was accompanied by minimal to slight pulmonary hemorrhage and findings in the nasal cavity.
Open literature publications of PFT assessments in concert with the alveolar macrophage response are primarily limited to inhalation studies of environmental materials in rodents (see below). However, pulmonary function was assessed and reported, in combination with an increased alveolar macrophage response, during evaluations of a biologic, anti-αvβ-6 integrin. Although anti-αvβ-6 integrin was administered by subcutaneous injection in cynomolgus macaques and mice, the lung findings were nearly indistinguishable from those observed in rats after inhalation of some pharmaceuticals (Table 1) or environmental particles. In pilot preclinical studies, no changes were observed in diffusing capacity (DLCO) or vital capacity after weekly treatments of anti-αvβ-6 integrin up to 10 mg/kg in mice or 20 mg/kg in monkeys (Olivier et al. 2006). Baseline, predosing assessments were similar to end of treatment and recovery (8-week, mice only) assessments in all treatment groups.
Although DE is a complex mixture containing hundreds of gaseous, semivolatile, and particulate components, it remains one of the most well-studied particulate-based atmospheres. By far, the most work describing PFT assessments after exposure to particulate-containing atmospheres has been with DE (Mauderly 1994, 1996). In rats exposed to DE at concentrations of up to 7 mg/m3, 7 hours per day (hr/d), 5 days per week (d/wk) for up to 12 months, the predominant exposure-related findings were minimally increased alveolar macrophage size and number and, associated particle accumulation (Mauderly 1994, 1996). Similar observations were noted after 30 months of DE exposure at 0.35 mg/m3 for 7 hr/d, 5 d/wk (Mauderly 1994). In neither case were pulmonary function parameters affected (Mauderly 1994, 1996).
Exposures to high concentrations of DE (3–7 mg/m3, 7 hr/d, 5 d/wk) extended to 24 months or longer, exceeded particle clearance mechanisms and resulted in lung overload. Increased alveolar macrophages were observed along with inflammation and additional morphological changes in the lung. These rats with significant inflammatory and structural lung changes had dose-related changes in pulmonary function, for example, restrictive changes including decreased lung volume/compliance, altered alveolar gas exchange, and nonuniform gas distribution (Mauderly 1994, 1996).
There are no published assessments of the potential for an increased alveolar macrophage response to exacerbate preexisting pulmonary disease in preclinical studies with pharmaceutical materials. However, rats with preexisting elastase-induced emphysema exposed to 3.5 mg/m3 DE for 5 d/wk/7 hr/d had no differences in respiratory function parameters as compared to air controls with preexisting emphysema after 6, 12, 18, and 24 months of exposure (Mauderly et al. 1990). Individual macrophages containing soot and aggregates of soot-laden alveolar macrophages, as well as additional morphologic changes at the 24-month sacrifice, were present in the DE-exposed rats. These findings suggest that even in susceptible populations, there is no evidence for any functional, adverse effect due to increased alveolar macrophages.
Measurement of pulmonary function (e.g., flow, volumes, diffusion capacity) might be used to investigate consequences of macrophage responses in preclinical species in an attempt to evaluate potential progression of changes in the lungs during extended dosing paradigms. However, PFTs would likely not demonstrate differences in lung dynamics in studies where increased alveolar macrophages were observed in absence of additional tissue changes in the lung. Although data for effects on PFT following inhalation of pharmaceutical materials are sparse, results from various studies cited above strongly suggest that an alveolar macrophage response alone will have no effect on pulmonary function of rodents.
Resistance to Infection
Alveolar macrophages have critical roles as a first line of defense against inhaled viruses and bacteria as well as in pathogen recognition. Concerns have been raised that a treatment-related alveolar macrophage response may impact pulmonary innate immune function and resistance to bacterial or viral infection. We were unable to identify examples among inhaled pharmaceutical products (Table 1) or in the open literature that isolate the primary end point (increases in alveolar macrophage size and number) with a functional infection-based outcome.
Numerous in vitro and in vivo environmental studies have been performed, whereby particles alone or particles plus environmental copollutant exposures have been used to assess macrophage responses and infectious disease end points (reviewed in U.S. Environmental Protection Agency, 1982, 1989, 1996, 2004). Many of these studies correlated exposure–time relationships relative to alveolar macrophage responses and subjected the test animals or isolated cells to viral or bacterial challenges. Results of these studies were largely dependent on the pathogen, experimental design, exposure atmosphere or mixture, and specific exposure parameters.
Objective 5: Points to Consider in Determining the NOAEL When Increased Alveolar Macrophages Are Observed in Inhalation Studies
The available literature from inhalation toxicology studies (Lee et al. 1986; Driscoll et al. 1996; Warheit et al. 1997; Elder et al. 2005; Mauderly 1994, 1996) and clinical experience with marketed, inhaled drugs provides a basis to determine whether an increase in alveolar macrophages in rodent inhalation studies represents an adverse outcome or potentially impacts the risk assessments for conduct of clinical studies. Consideration of the following points will guide risk assessment for effects of a given test article when increased alveolar macrophages are noted in preclinical studies. In the absence of other, adverse lung changes, increases in alveolar macrophage number and/or size are interpreted as nonadverse. Data from studies of inhaled, poorly soluble, and low-toxicity particles and from nonclinical studies of marketed drugs indicate that findings of increased numbers of macrophages or increased numbers and sizes of macrophages, whether dispersed throughout the pulmonary structures, aggregated at the bronchiolar–alveolar duct junction, or aggregated at the periphery of the lung, are not adverse when encountered in absence of other lung findings. Conversely, presence of additional responses, such as inflammation, hyperplastic epithelial responses, or other alterations to the pulmonary architecture, that are associated with increased alveolar macrophages indicate potentially adverse processes and should be taken into consideration when determining the NOAEL. Similarly, development of inflammatory, degenerative, or hyperplastic epithelial responses above background with increased study duration at a given exposure concentration, for example, when a 2-week study is extended to 4 weeks or 6 months, suggests that the adaptive, steady state response of increased macrophages to inhaled particles has been exceeded. Findings similar to those observed in studies conducted with approved products at a similar exposure multiple are not interpreted as adverse. FDA public domain data (Table 1) could be cited to support an overall risk assessment. Resolution of a finding of increased alveolar macrophages during the recovery phase supports the interpretation that increased alveolar macrophages did not initiate a pathway leading to adverse findings.
Although reversibility per se does not represent evidence that a finding is nonadverse, confirming reversibility supports the argument that the test article was cleared from the lung without adverse sequelae and that the alveolar macrophage response failed to initiate irreversible pathologic processes. Conversely, development of morphologic findings such as inflammation, tissue damage, and fibrosis during the recovery phase indicates that homeostasis was exceeded and that an adverse response occurred.
Conclusion
Mandated by Society of Toxicologic Pathology (STP), a dedicated working group has provided guidance to distinguish those responses in the lungs that represent adaptive, nonadverse changes from those deemed to represent adverse processes following inhalation of pharmaceutical agents intended for pulmonary delivery. The presence of increased numbers or size of alveolar macrophages in terminal air spaces as the sole observation is considered nonadverse when encountered in lungs of nonclinical species having preservation of normal tissue architecture. On the other hand, tissue responses in the lung characterized by a combination of increased alveolar macrophages and other processes such as frank inflammation, degeneration, or alterations to normal structure of terminal air spaces or airways are more likely to represent adverse effects.
A weight of evidence approach is advocated when distinguishing adverse from nonadverse accumulations of macrophages in the lung. Consideration of adequate characterization of exposure conditions to ensure delivery of the test article into the deep lung, pharmacokinetics and pharmacology of the test article, the nature of findings at each dose level, and reversibility of any findings in the lungs, are critical to establishing an appropriate risk assessment.
Further, the occurrence of increased alveolar macrophages in preclinical inhalation studies is well documented for currently marketed, orally inhaled products that have demonstrated efficacy and safety in humans. It is imperative to avoid an overinterpretation regarding accumulation of alveolar macrophages in lungs of rats, as the presence of alveolar macrophages in the lungs of rats has not been associated with compromise of pulmonary function nor associated with compromise of innate immunity, particularly given the range of functional reserve among macrophage populations and given the plasticity of monocyte and macrophage populations in rodents.
The working group advocates consistent, appropriate application of terminology by the study pathologist as presented herein during evaluations of inhalation studies in preclinical species. Having incidence tables that distinctly delineate nonadverse findings in the lungs of preclinical species, and providing appropriate interpretations in report narratives, are incumbent upon study pathologists.
Footnotes
Notes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
