Abstract
International regulatory and pharmaceutical industry scientists are discussing revision of the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) S1 guidance on rodent carcinogenicity assessment of small molecule pharmaceuticals. A weight-of-evidence approach is proposed to determine the need for rodent carcinogenicity studies. For compounds with high human cancer risk, the product may be labeled appropriately without conducting rodent carcinogenicity studies. For compounds with minimal cancer risk, only a 6-month transgenic mouse study (rasH2 mouse or p53+/− mouse) or a 2-year mouse study would be needed. If rodent carcinogenicity testing may add significant value to cancer risk assessment, a 2-year rat study and either a 6-month transgenic mouse or a 2-year mouse study is appropriate. In many cases, therefore, one rodent carcinogenicity study could be sufficient. The rasH2 model predicts neoplastic findings relevant to human cancer risk assessment as well as 2-year rodent models, produces fewer irrelevant neoplastic outcomes, and often will be preferable to a 2-year rodent study. Before revising ICH S1 guidance, a prospective evaluation will be conducted to test the proposed weight-of-evidence approach. This evaluation offers an opportunity for a secondary analysis comparing the value of alternative mouse models and 2-year rodent studies in the proposed ICH S1 weight-of-evidence approach for human cancer risk assessment.
Introduction
The International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) is discussing changes to the ICH S1 regulatory guidance for rodent carcinogenicity testing to support the registration of small molecule pharmaceuticals. The goal of the ICH discussions is to revise the carcinogenicity assessment paradigm by defining criteria that would justify waivers of 2-year rat carcinogenicity testing, without compromising human safety, for some pharmaceutical candidates that currently would require such studies. These references have been moved to the end of the next sentence. Regulatory notices have been issued proposing the considerations to be evaluated during these discussions (European Medicines Agency [EMA] 2012; Federal Register 2013; Japanese Ministry of Health, Labor and Welfare, Pharmaceuticals and Medical Devices Agency [PMDA] 2012; ICH 1996). These considerations include mechanisms of on-target and off-target pharmacology, genotoxicity, microscopic (histopathologic) risk factors for carcinogenicity identified in rat toxicity studies up to 6 months in duration, exposure margins in rat toxicity studies, hormonal activity, immunosuppression, results of non-rodent chronic toxicity studies, and results of medium-term alternative mouse studies if available. A prospective evaluation of the weight-of evidence approach is proposed to guide future changes to the final regulatory guidance (EMA 2012; Federal Register 2013; PMDA 2012). According to the current proposal, pharmaceutical industry sponsors will be requested to submit voluntary Carcinogenicity Assessment Documents for each investigational small molecule pharmaceutical subject to rodent carcinogenicity testing prior to completion of the 2-year rat carcinogenicity study. Carcinogenicity Assessment Documents will be shared among all ICH Drug Regulatory Agencies (the U.S. Food and Drug Administration [FDA], the EMA, and the PMDA). The Carcinogenicity Assessment Documents will summarize the overall carcinogenic risk of each compound, predict the outcome of the rat carcinogenicity study, and propose whether or not a rat carcinogenicity study would add value for human risk assessment. Neoplastic outcomes in the 2-year rat studies and the impact of the 2-year rat studies on human cancer risk assessment will be assessed as the information becomes available.
Since 1997, alternative, medium-term, genetically modified mouse models have been acknowledged as suitable substitutes for 1 of the 2 required 2-year rodent carcinogenicity studies (ICH 1997). The FDA now considers the p53+/− hemizygous knockout mouse model suitable for compounds with evidence or residual concerns of genotoxicity (Boverhof et al. 2011; Jacobs and Hatfield 2013; MacDonald et al. 2004). The Tg.AC transgenic mouse model may be acceptable for carcinogenicity assessment of compounds administered topically and intended for topical use in humans, but this model is no longer recommended by the FDA (Boverhof et al. 2011). The rasH2 transgenic mouse model is more broadly accepted to replace a 2-year mouse carcinogenicity study to support human cancer risk assessment for both genotoxic and nongenotoxic pharmaceuticals given orally or parenterally. The rasH2 mouse model has significant advantages compared to 2-year mouse studies and should contribute more prominently to future cancer risk assessment strategies under the new proposed ICH S1 weight-of-evidence–based paradigm.
Liabilities of the 2-year Rodent Model
The 2-year rat and mouse models for carcinogenicity assessment have demonstrated drug-related neoplastic findings for approximately 56% of marketed drugs tested (Alden et al. 2011; Friedrich and Olejniczak 2011; Van Oosterhout et al. 1997). Approximately 11 to 15% of marketed drugs tested have prominent label warnings regarding human cancer risk, indicating that approximately 75% of positive neoplastic outcomes in rodents are not considered important or predictive for human cancer risk assessment (Alden et al. 2011; Friedrich and Olejniczak 2011). In a retrospective review of 182 pharmaceutical candidates, 14 compounds (approximately 8%) produced positive neoplastic outcomes in 2-year rat carcinogenicity studies that were not predicted by positive genotoxicity findings, hormonal activity, or microscopic risk factors for neoplasia in 6-month and/or 12-month rat toxicity studies (Sistare et al. 2011). In all 14 of these cases, the neoplastic findings in the rat were of questionable human relevance. Ten of these compounds are marketed, 2 were still in development at the time of publication despite positive rat carcinogenicity findings, and 2 were discontinued for reasons unrelated to the rat carcinogenicity findings. Many positive rodent neoplastic responses to pharmaceutical administration have minimal relevance for human risk assessment (Alden et al. 2011; International Agency for Research on Cancer 1999). Reasons that neoplastic findings in rodents have been discounted for human risk assessment include (1) rodent-specific mechanisms of carcinogenicity; (2) high margins between exposures at the no observed adverse effect level in rodents and expected clinical exposures in humans; (3) a small neoplastic effect size based on historical control data; (4) absence of a dose–response relationship; (5) the types of neoplasms that were increased after drug administration were commonly observed in concurrent or historical controls; (6) scientific literature and/or clinical data indicated lack of relevance; and (7) differences in metabolism or local drug concentrations between rodents and humans (Friedrich and Olejniczak 2011). These irrelevant positive findings confound interpretation and may shorten life span in rodent carcinogenicity studies. In addition, 2-year rodent carcinogenicity studies failed to detect cancer risk for about 15% of drugs that have carcinogenicity warnings in their labels (Alden et al. 2011). Therefore, 2-year rodent carcinogenicity studies suffer from many “false positives,” some “false negatives,” and poor prediction for human cancer risk.
The rasH2 Mouse Model
The rasH2 mouse (CByB6F1-Tg(HRAS)2Jic mouse, Taconic Farms, Germantown, New York, and Central Institute for Experimental Animals, Kawasaki, Japan) contains several copies of the human ras oncogene with its own promoter, making this mouse susceptible to a wide range of chemically induced neoplasms within a 6-month dosing period (Mitsumori et al. 1998; Yamamoto et al. 1998). The rasH2 model is susceptible to neoplasms caused by genotoxic and nongenotoxic chemicals (Mitsumori et al. 1998; Morton et al. 2002; Nambiar and Morton 2013; Usui et al. 2001). In studies conducted under the direction of the Alternatives to Carcinogenicity Testing committee sponsored by the International Life Sciences Institute, Health and Environmental Sciences Institute (ILSI-HESI), the rasH2 mouse model was comparable to conventional 2-year rodent studies in identifying known human carcinogens, but produced fewer neoplastic outcomes in response to rodent-specific carcinogens (believed to be not carcinogenic in humans) than 2-year rodent studies (Morton et al. 2002; Usui et al. 2001). Examples of compounds with positive 2-year rodent neoplastic findings that are not relevant to human risk assessment, and that are negative in the rasH2 model, include phenobarbital, methapyrilene, reserpine, dieldrin, chloroform, chlorpromazine, haloperidol, metaproterenol, and sulfamethoxazole (Usui et al. 2001).
Alternative medium-term mouse models have been accepted since 1997 as substitutes for one of the two 2-year rodent carcinogenicity studies required for pharmaceutical registration (ICH 1997). The pharmaceutical industry initially was slow to embrace these models, but use of the rasH2 model has increased recently (Boverhof et al. 2011; Friedrich and Olejniczak 2011; Jacobs and Hatfield 2013). As of 2011, 20 rasH2 mouse studies had been submitted to the FDA in support of pharmaceutical registrations (Boverhof et al. 2011). Of these 20 compounds, 3 (15%) were positive in the rasH2 model. The 15% of drugs positive in the rasH2 model correspond well with the 11 to 15% of drugs that pose cancer risk of humans as indicated in label warnings. The results from 21 rasH2 mouse carcinogenicity studies conducted to support pharmaceutical development are now publicly available (Table 1). Of these 21 compounds, 19 have been assessed both in the rasH2 model and in the 2-year rat model. Two of these compounds (sunitinib and bazedoxifene) were positive both in the rasH2 model and in the 2-year rat studies. Sunitinib produced gastroduodenal (Brunner’s gland) neoplasms in both rasH2 mice and rats and hemangiosarcomas in rasH2 mice. Bazedoxifene produced an increase in benign ovarian granulosa cell neoplasms in the rasH2 mouse study and in the 2-year rat study, consistent with its expected hormonal activity. Male rats administered bazedoxifene also developed renal tubular neoplasms that were attributed to rodent-specific increased incidence and severity of chronic progressive nephropathy (Nambiar and Morton 2013; Perry et al. 2013). None of the other 17 compounds tested in both models were positive in the rasH2 model. In this limited group of 19 compounds with results for both rasH2 studies and 2-year rat studies, the rasH2 model and the 2-year rat studies produced similar neoplasms of possible relevance for human cancer risk assessment for two compounds (11%), while 9 compounds (47%) were negative in the rasH2 mouse model and positive in the 2-year rat studies. Eight compounds (42%) were negative in both rasH2 mouse and 2-year rat studies. Most rat tumor findings were not relevant for human risk assessment. Neoplastic findings in rasH2 mice or rats did not result in any label warnings for human cancer risk, and two compounds (maraviroc and tofacitinib) with warnings for human cancer risk had immunosuppressive modes of action. Drug-related neoplasms with low relevance to human risk assessment in the 9 rat studies included thyroid follicular neoplasms associated with hepatic enzyme induction, gastric carcinoid associated with gastric parietal cell atrophy and foveal hyperplasia, renal tubular neoplasms associated with increased chronic progressive nephropathy, pheochromocytomas, angiomas of mesenteric lymph nodes, interstitial cell neoplasms of the testis, and ovarian leiomyomas associated with β2 adrenergic agonism (Alison, Capen, and Prentice 1994; Cook et al. 1999; Greaves 2012; Greim et al. 2009; Hard, Betz, and Seeley 2012; Hard and Khan 2004; Nambiar and Morton 2013; Prentice and Meikle 1995; Radi and Morton 2012). Therefore, using the rasH2 mouse model instead of the 2-year rat model would have provided similar identification of the 15% of compounds with potentially significant concern for human cancer while eliminating most rodent tumor outcomes that have little or no relevant contribution to human risk assessment.
Comparisons of neoplastic responses in 6-month rasH2 mouse studies and 2-year rat carcinogenicity studies and FDA label warnings for cancer risk. All compounds were negative for genotoxic findings.
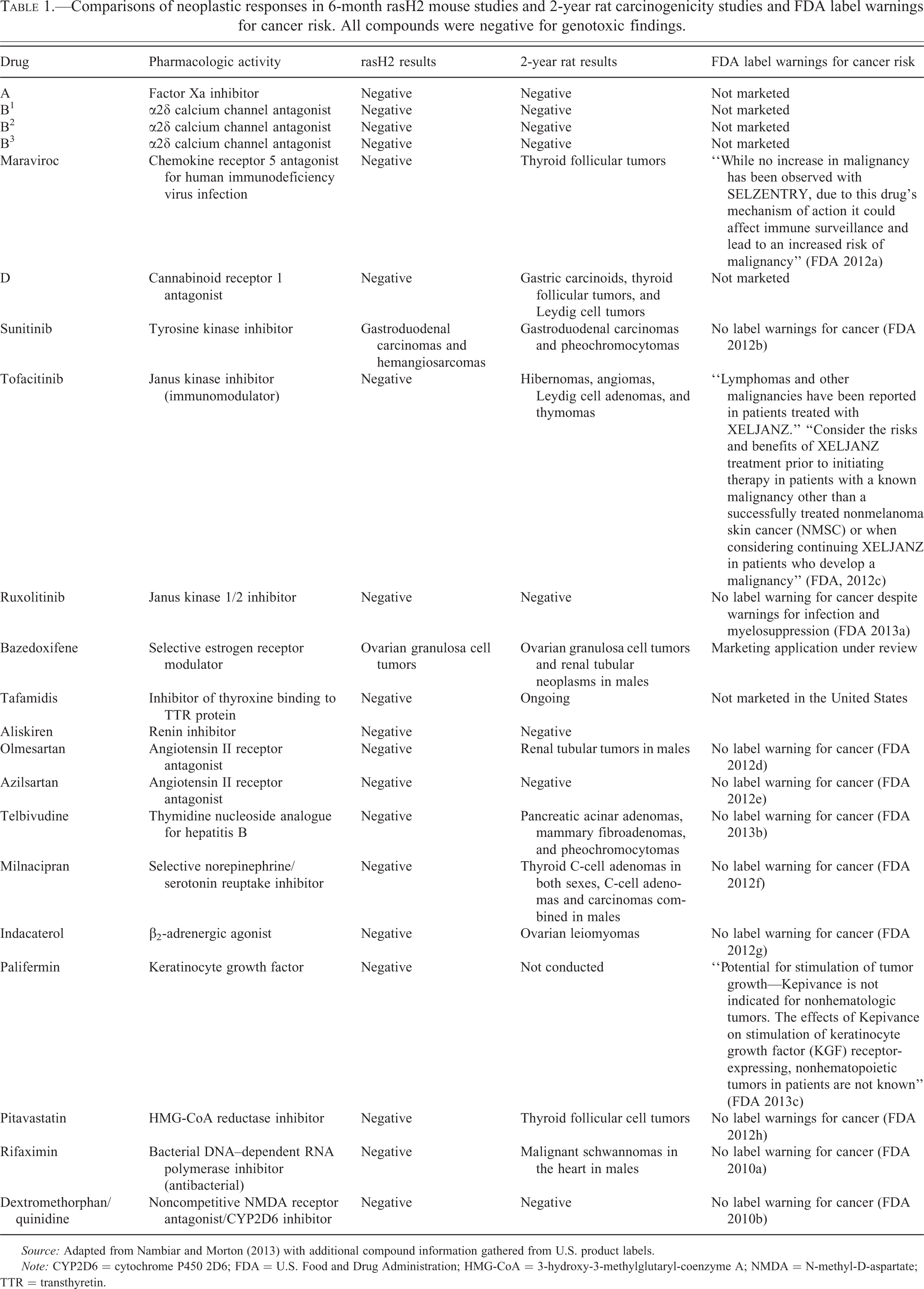
Source: Adapted from Nambiar and Morton (2013) with additional compound information gathered from U.S. product labels.
Note: CYP2D6 = cytochrome P450 2D6; FDA = U.S. Food and Drug Administration; HMG-CoA = 3-hydroxy-3-methylglutaryl-coenzyme A; NMDA = N-methyl-D-aspartate; TTR = transthyretin.
For compounds that require a rodent carcinogenicity study, the advantages and considerations for conducting an alternative transgenic mouse study instead of a standard 2-year mouse study have been described (Nambiar and Morton 2013; Storer et al. 2010). The rasH2 mouse model often will be the preferred model for multiple reasons. First, the rasH2 model predicts genotoxic and nongenotoxic chemicals of known concern for human cancer risk as well as 2-year rodent studies (Usui et al. 2001; Morton et al. 2002; Nambiar and Morton 2013). Second, the relatively low incidence of spontaneous neoplasms in rasH2 mice makes findings in this model more specific for chemically mediated carcinogenic effects than the 2-year rodent models (Usui et al. 2001; Nambiar, Turnquist, and Morton 2012; Paranjpe et al. 2013). Third, compared with the 2-year rodent models, the rasH2 model produces far fewer positive tumor outcomes that are either rodent-specific or otherwise irrelevant for human cancer risk assessment (Nambiar and Morton 2013). Use of the rasH2 model is expected to improve the prediction of relevant human cancer risk without producing as many confounding tumor outcomes that are common with the 2-year rodent models. Fourth, 6-month carcinogenicity studies in the rasH2 model are less likely to have spontaneous age-related morbidity, mortality, and neoplastic outcomes that may shorten life span in 2-year rodent studies and confound interpretation. Fifth, the ability to complete the in vivo portion of the carcinogenicity study within a 6-month period greatly enhances rapid execution when results are needed quickly or permits the delay of the study until additional animal and human data support continued development and registration. Finally, low mortality and fewer spontaneous neoplastic findings allow rasH2 and p53+/− carcinogenicity studies to be conducted using only 25 animals/sex/group (MacDonald et al. 2004). This greatly reduces animal use in accordance with the 3Rs (reduce/refine/replace) principles and lowers costs compared with a 2-year rodent study. The p53+/− mouse model may be the single preferred model for compounds with evidence or residual concerns of genotoxicity; however, the rasH2 model is the only alternative model that is acceptable for compounds with positive, equivocal, or negative genotoxicity findings (Boverhof et al. 2011; MacDonald et al. 2004).
Considerations and Recommendations
Within the ICH, a revision of cancer risk assessment paradigms based on weight of evidence is being considered. Options to eliminate or replace 2-year rodent studies based on weight of evidence are being addressed to improve human risk assessment while reducing, refining, and replacing animal testing. The potential future role of alternative mouse models for carcinogenicity testing can be visualized clearly only by considering the entire cancer risk assessment strategy. Sponsors and regulatory agencies would need to consider the weight of evidence for human cancer risk early in development of a novel pharmaceutical, well in advance of rodent carcinogenicity studies, to determine if any rodent carcinogenicity study is necessary. Options for carcinogenicity assessment of some categories of compounds proposed in the regulatory notice may include opportunities to eliminate the need to conduct any rodent carcinogenicity study in certain situations.
As proposed, one scenario could warrant a waiver for all rodent carcinogenicity studies. In this scenario (proposed category 1 in the ICH regulatory notice), a compound that is likely to be a human cancer risk at concentrations relevant to human clinical exposure may be approved based on benefit–risk considerations, with an appropriate warning in the label, and a carcinogenicity study would not add value and would not be required. Criteria that may contribute to this type of waiver of rodent carcinogenicity studies include mechanisms of action that have been established to contribute to human cancer, mutagenicity, clastogenicity without an appropriate margin of safety, potent immunosuppression, or hormonal activity relevant to human cancer risk. Registration would be expected if the benefit of treatment outweighs the overall risks. Alternatively, the sponsor may choose to voluntarily conduct one or more carcinogenicity studies and/or mechanistic studies to better characterize and/or mitigate the perceived human cancer risk, and results of these studies would contribute to the weight of evidence during the cancer risk assessment. In such cases, a sponsor may choose to conduct only a medium-term alternative mouse study, a 2-year rat study, or both, but these would not be required.
Two additional scenarios could warrant a waiver of 2-year rat carcinogenicity studies while maintaining the expectation to conduct a mouse carcinogenicity study. In the first of these two scenarios (proposed category 3b), compounds may merit a waiver for rat carcinogenicity studies because they present minimal risks of human cancer based on weight of evidence and are highly unlikely to cause neoplasia in rats. A waiver of the 2-year rat study usually should be granted if, at adequate margins to clinically relevant drug exposure, a compound has minimal on-target and off-target mechanistic concern for human carcinogenicity, no evidence of genotoxicity, no hormonal activity suggestive of increased human cancer risk, minimal or no immunosuppressive effects, and no proliferative microscopic (histopathologic) risk factors for neoplasia relevant to human risk assessment in rodent and non-rodent toxicity studies. Absence of microscopic risk factors for tumor outcomes in 6-month rat studies is a good predictor of negative tumor outcomes in 2-year rat studies and should contribute to the weight of evidence that a rat carcinogenicity study may not add value (Sistare et al. 2011). In such cases, a sponsor may voluntarily elect to conduct a medium-term alternative mouse study early in development to bolster the weight-of-evidence proposal for a waiver of the rat carcinogenicity study (not described in Figure 1), but whether or not this early study would add value could be considered on a case-by-case basis.
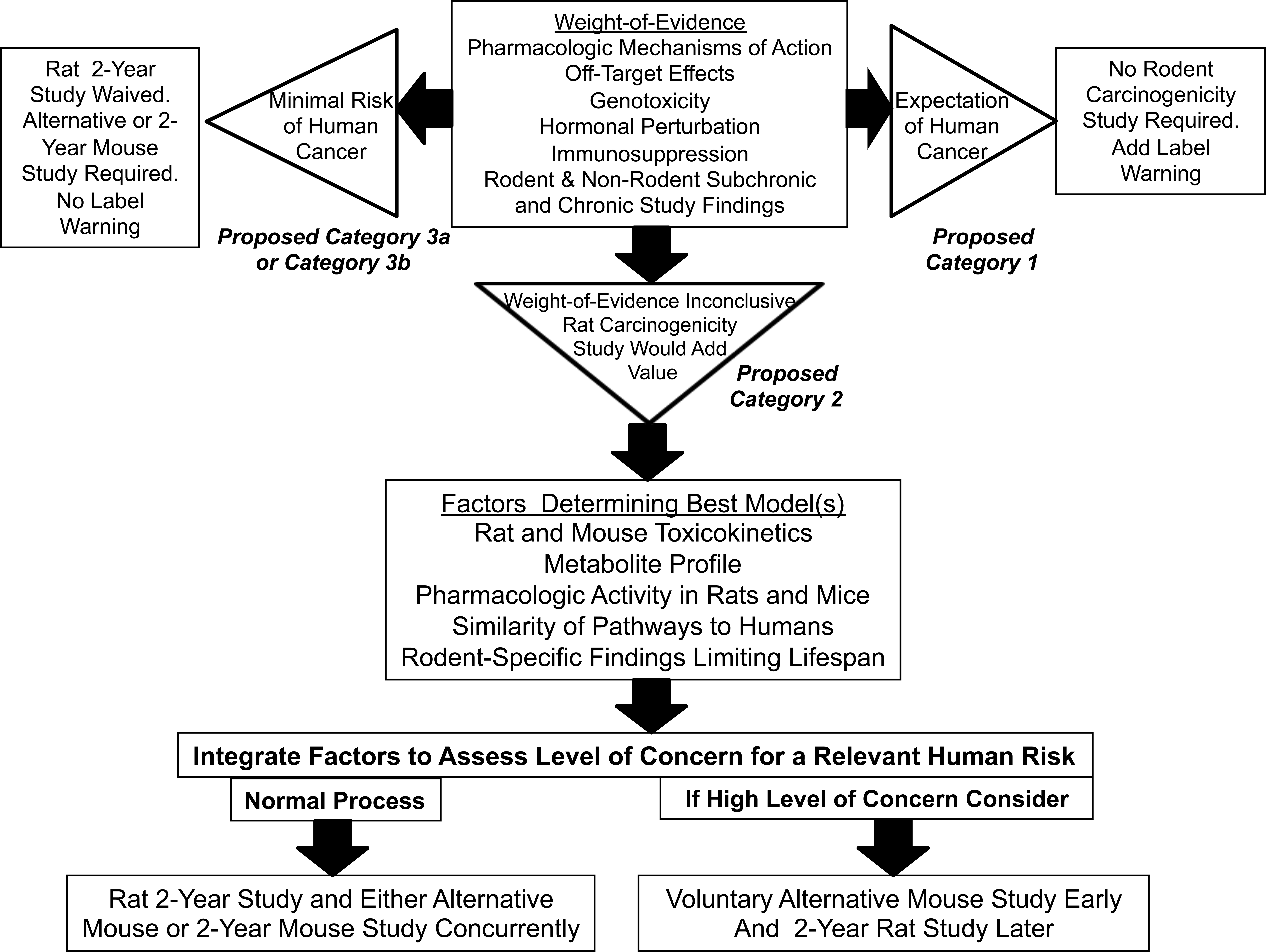
The suggested weight-of-evidence decision process guiding the conduct of rodent carcinogenicity studies includes options for a single rodent carcinogenicity study (alternative mouse or 2-year mouse study) when human cancer risk is low, and no rodent carcinogenicity study with an appropriate label warning when a compound is expected to cause human cancer. When human cancer risk is uncertain and rodent carcinogenicity studies will contribute to risk assessment, two rodent carcinogenicity studies will be required. These will include a 2-year rat study plus either an alternative 6-month mouse study or a 2-year mouse study.
The second of the two scenarios (proposed category 3a) that may justify waivers of rat carcinogenicity studies includes compounds that present minimal cancer risks of patients, but are expected to produce neoplasms in rats that are unlikely to be relevant for human cancer risk assessment. In this scenario, a rat carcinogenicity study would not be expected to add value if microscopic risk factors in nonclinical toxicity studies are all compatible with rat-specific carcinogenic mechanisms or occur only at high multiples of human exposure. Positive microscopic risk factors for rat neoplasia in chronic rat studies should not automatically trigger the need for a rat carcinogenicity study because many proliferative changes in chronic rat studies do not predict for positive rat carcinogenicity study outcomes (Sistare et al. 2011) and because neoplastic findings in rodents usually do not predict human cancer risk (Alden et al. 2011; Friedrich and Olejniczak 2011). The possible mechanisms producing microscopic risk factors should be considered in the context of the total weight of evidence. While the ICH regulatory notice mentions rat-specific neoplasms related to microsomal enzyme induction as an example (EMA 2012; Federal Register 2013; PMDA 2012), other rodent-specific mechanisms are equally important in determining if a rat carcinogenicity study should be performed. In addition to hepatocellular and thyroid follicular neoplasms associated with microsomal enzyme induction (Graham and Lake 2008; Holsapple et al. 2006; International Agency for Research on Cancer 1999; McClain 1992; Wu and Farrelly 2006), other examples of rodent-specific mechanisms not relevant for human risk assessment include hepatocellular neoplasms secondary to peroxisome proliferation (Gonzales, Peters, and Cattley 1998; Greaves 2012); renal tubular neoplasms associated with increased severity of chronic nephropathy (Hard, Betz, and Seeley 2012; Hard and Khan 2004) or α2u-globulin nephropathy (Alison, Capen, and Prentice 1994; International Agency for Research on Cancer 1999); urothelial neoplasia secondary to crystalluria in rodents that is not expected in patients (Cohen 2002; Cohen, Arnold, and Suzuki 2010; Cohen and Lawson 1995); mesovarian leiomyomas in rats associated with β2-receptor agonism (Alison, Capen, and Prentice 1994); gastric carcinoids secondary to decreased acid secretion (Alison, Capen, and Prentice 1994); adrenal pheochromocytomas (Greim et al. 2009); and some cases of testicular interstitial (Leydig) cell neoplasms (Prentice and Meikle 1995; Cook et al. 1999). Here again, a sponsor may elect to conduct a medium-term alternative mouse study earlier in development to bolster the weight-of-evidence proposal when specific circumstances indicate these data would add value (not described in Figure 1).
A fourth scenario (proposed category 2) occurs when the weight of evidence does not present a clear case either for or against significant human cancer risk of a given compound, and rodent carcinogenicity studies would be expected to add value for human cancer risk assessment. A 2-year rat study and either a 6-month study in an alternative mouse model (currently the rasH2 mouse or the p53+/− mouse) or a 2-year mouse study would be appropriate in such cases. The most appropriate testing strategy should be determined based on all available scientific evidence and the level of concern (Figure 1).
Although current regulatory guidance generally requires only that carcinogenicity studies be completed at the time a marketing application is submitted (ICH 2009); in some cases, a sponsor may voluntarily decide to conduct a medium-term alternative mouse study early in development. For compounds with novel mechanisms of action, ubiquitous target distribution, or very complex or poorly understood pharmacology, the early conduct of a carcinogenicity study in a rasH2 or p53+/− mouse model may benefit the sponsor’s decision making. Results can be available and decisions can be made within 1 year of study initiation. Negative results in such a carcinogenicity study will enhance the sponsor’s confidence in clinical safety, can be included in the Carcinogenicity Assessment Document provided to regulatory agencies, and can contribute to weight of evidence that a 2-year rodent study would not add further value for cancer risk assessment. If drug-related neoplasms are demonstrated in an alternative mouse model conducted early in development, costly investments in clinical trials may be halted if evidence of human cancer risk is sufficient to terminate development, or development and registration can continue with enhanced knowledge of cancer hazards and the need for appropriate product labeling.
The regulatory notice describing the proposed process for modifying ICH S1 regulatory guidance includes a prospective evaluation process to gather additional information on assessment of human carcinogenicity risks using weight of evidence, prediction of carcinogenicity findings in the 2-year rat study, and the value of the 2-year rat study to human risk assessment (EMA 2012; Federal Register 2013; PMDA 2012). While not a clearly stated objective, the prospective evaluation process could secondarily present an opportunity to compare the relative values of the alternative mouse and 2-year rodent models. Sponsors could be encouraged to utilize alternative models (particularly the rasH2 mouse model for nongenotoxic compounds) during the evaluation period. Explanation of the most appropriate approach (alternative mouse model, 2-year rodent model, or both) for human cancer risk assessment of each compound could be addressed in the Carcinogenicity Assessment Document submitted to Drug Regulatory Authorities when carcinogenicity assessment in rodents seems justified. Sponsors will be submitting rodent carcinogenicity study results to ICH Drug Regulatory Authorities during the prospective evaluation period. A critical comparison of the relative value of the alternative models to the 2-year rodent studies for human cancer risk assessment could provide value toward further improving carcinogenicity assessment strategies for small molecule pharmaceuticals.
Footnotes
*This commentary was created by members of the limited duration key issues team that supports the representatives of the Pharmaceutical Research and Manufacturers Association on the ICH S1 Expert Working Group. This is an opinion article submitted to the Regulatory Forum and does not constitute an official position of the Society of Toxicologic Pathology or the journal Toxicologic Pathology. The views expressed in this article are those of the authors and do not necessarily represent the policies, positions, or opinions of their respective organizations. The Regulatory Forum is designed to stimulate broad discussion of topics relevant to regulatory issues in toxicologic pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to regulatoryforum@toxpath.org.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
