Abstract
Dose selection for the 6-month rasH2 mouse carcinogenicity studies depends heavily on the maximum tolerated dose (MTD) obtained from 1-month range-finding studies. A retrospective evaluation of range-finding studies and pivotal 6 month rasH2 mouse studies for 11 compounds demonstrated that the MTD based on at least a 10% decrease in body weight gain, mortality, and target organ toxicity in range-finding studies appropriately identified high doses for pivotal studies for 8 of 11 compounds. Two of the selected high doses were based on decreased body weight gain alone, while 7 were based on mortality at higher doses in shorter duration range-finding studies. High-dose selection was based on the maximum feasible dose for one study. The Center for Drug Evaluation and Research, U.S. Food and Drug Administration Executive Carcinogenicity Assessment Committee often suggested different doses than those proposed by the sponsor. High mortality was observed in only one pivotal study and the high dose was lowered during the course of that study.
Dose selection for carcinogenicity studies is a complicated mix of science, experience, and guesswork. Information used to determine the high dose in 6-month rasH2 mouse studies may include factors identified in the range-finding studies that would decrease survival in a pivotal 6-month study. These factors include decreases in body weight or body weight gain, increased mortality, and clinical or anatomic pathology findings (Table 1; International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use [ICH] 2008). The maximum feasible dose may also be used to set the high dose in a 6-month rasH2 mouse study. The article “Retrospective Evaluation of Doses in the 26 Week Tg.rasH2 Mice Carcinogenicity Studies: Recommendation to Eliminate High Doses at Maximum Tolerated Dose (MTD) in Future Studies” by Paranjpe et al. (2015, see this issue) illustrates legitimate concerns about selection of the dose levels for 6-month studies using rasH2 mice (CByB6F1-Tg(HRAS)2Jic, Taconic Farms, Germantown, NY). An important issue highlighted by Paranjpe et al. is one where the occurrence of significant toxicity, moderate to severe weight loss, and increased mortality in the high-dose group in the pivotal 6-month studies may result in lower incidences of neoplasms in the high-dose groups relative to the controls, thereby diminishing or negating the value of the high-dose group.
Dose selection criteria for pivotal rodent carcinogenicity studies.
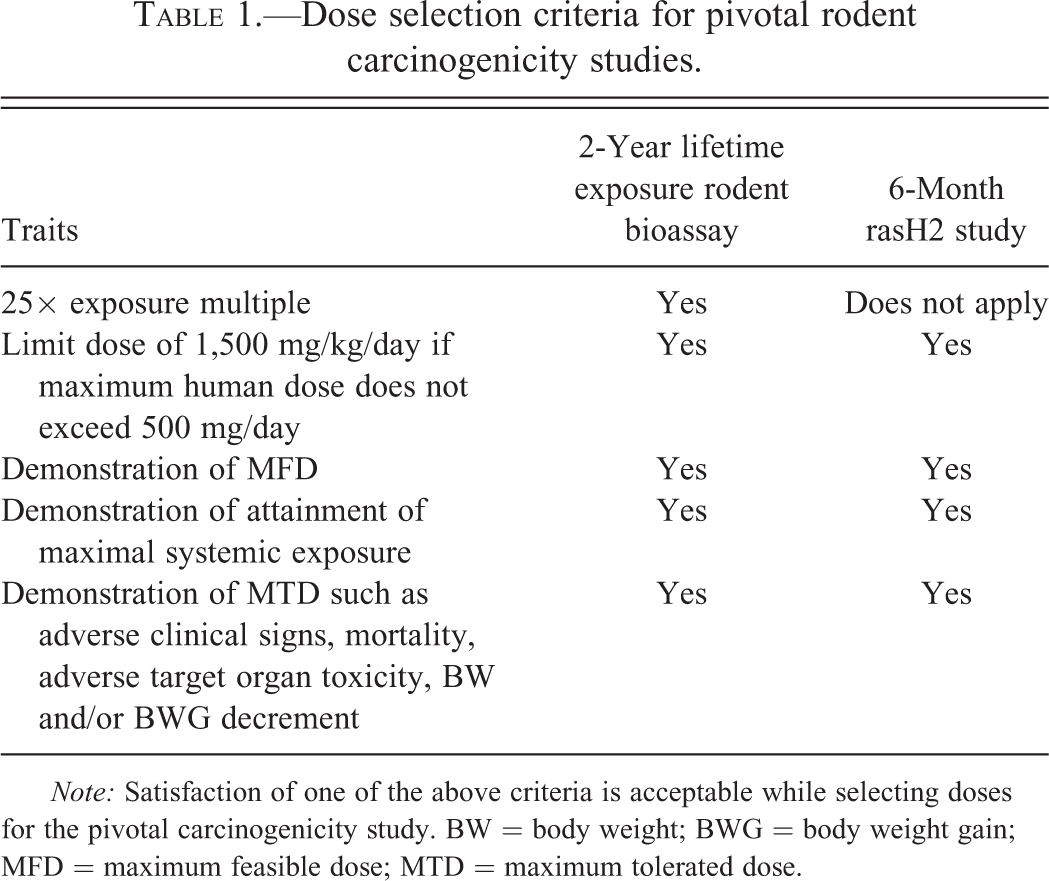
Note: Satisfaction of one of the above criteria is acceptable while selecting doses for the pivotal carcinogenicity study. BW = body weight; BWG = body weight gain; MFD = maximum feasible dose; MTD = maximum tolerated dose.
In the series of studies presented by Paranjpe et al., the high doses decreased body weight gain, increased mortality and/or lowered tumor incidences in males in 75% of 6-month rasH2 mouse studies. Unfortunately, Paranjpe et al. did not sort out the different factors determining the maximum tolerated dose (MTD) in the 1-month range-finding studies to help identify variables that were most suitable for high-dose selection in the pivotal studies. This makes it difficult for readers to determine if decreased body weight gain or other factors determined the MTD in these studies. This is important because dose selection for the pivotal 6-month studies based on the MTD might be approached differently depending on the findings at the MTD and at the next higher dose (if the MTD was exceeded). For example, if the MTD in a range-finding study is set based on a slight (<10%) decrement in body weight gain with greater body weight changes at higher doses, there may be some concern that there would be more dramatic decrease in body weight gain in the 6-month study. If the MTD is based on mortality at a considerably higher dose and there is no evidence of toxicity at the MTD, then it may be appropriate to set the high dose in the 6-month study at the MTD or perhaps even above the MTD observed in the range-finding study.
To independently assess the recommendations of Paranjpe et al. (2015), a retrospective evaluation was conducted using studies not included in the article by Paranjpe et al. In this report, data from 11 six-month rasH2 mouse studies are summarized, with a special focus on the high-dose selection strategy using data from the range-finding studies. Table 2 highlights the high-dose selection rationale for each compound.
Summary of dose selection and results for 6-month rasH2 mouse studies.
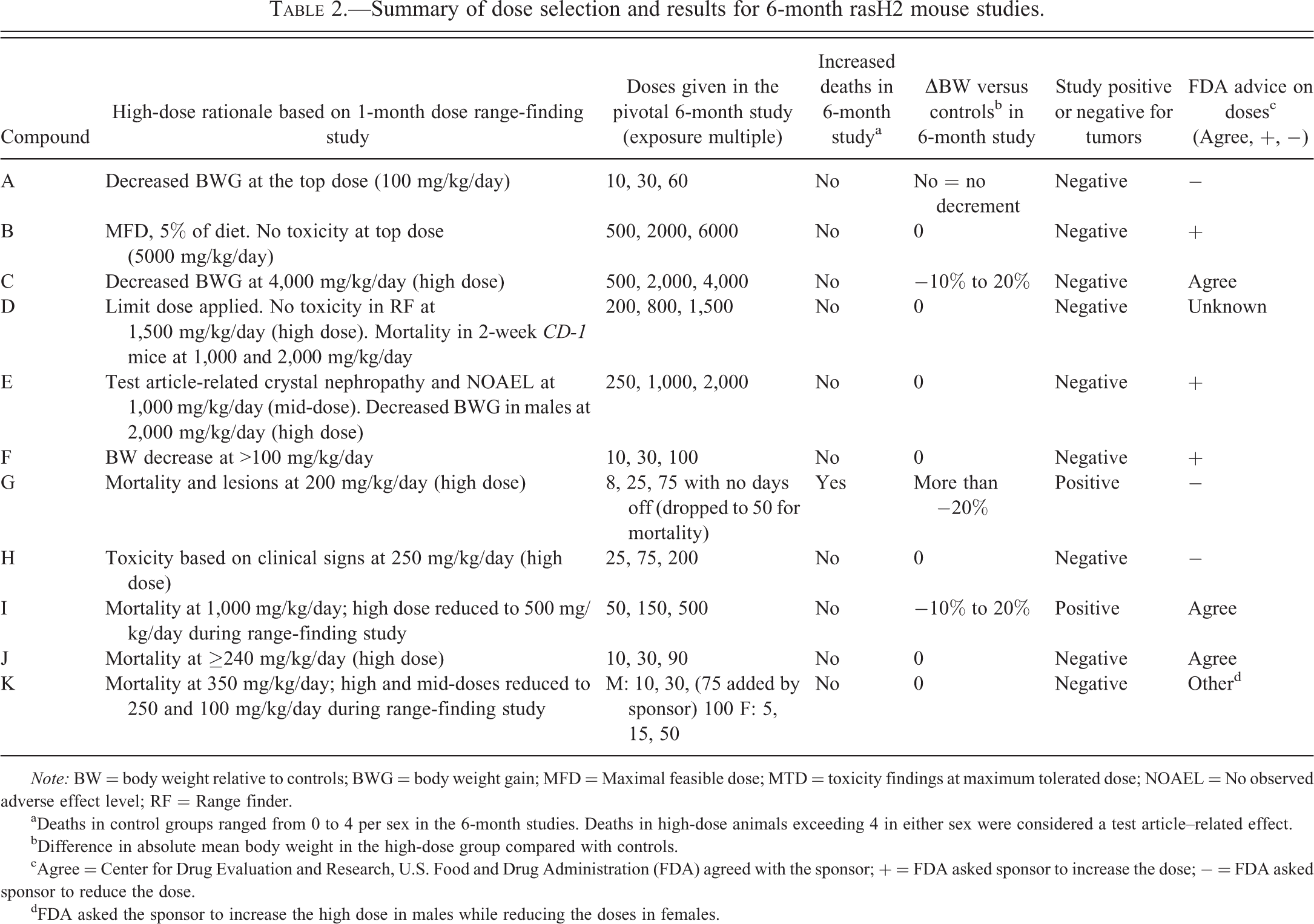
Note: BW = body weight relative to controls; BWG = body weight gain; MFD = Maximal feasible dose; MTD = toxicity findings at maximum tolerated dose; NOAEL = No observed adverse effect level; RF = Range finder.
aDeaths in control groups ranged from 0 to 4 per sex in the 6-month studies. Deaths in high-dose animals exceeding 4 in either sex were considered a test article–related effect.
bDifference in absolute mean body weight in the high-dose group compared with controls.
cAgree = Center for Drug Evaluation and Research, U.S. Food and Drug Administration (FDA) agreed with the sponsor; + = FDA asked sponsor to increase the dose; − = FDA asked sponsor to reduce the dose.
dFDA asked the sponsor to increase the high dose in males while reducing the doses in females.
The outcomes of high-dose selection for the pivotal 6-month studies in Table 2 differ from results reported by Paranjpe et al. (2015). Paranjpe et al. found that the MTD was exceeded in 75% of the 6-month rasH2 mouse studies. In the present series of studies, albeit a small set, unacceptable mortality or significant body weight changes was observed only in 1 (9%) out of 11 six-month rasH2 mouse studies, while the MTDs established in the range-finding studies were used to set the high dose in 8 of these studies. The criterion used by Paranjpe et al. to quantify the number of pivotal 6-month rasH2 studies that actually exceeded the MTD was based solely on >10% decrease in body weight gain relative to controls. In our experience, a 10% decrease in body weight gain in the 6-month studies may not impact tumor formation, morbidity, mortality, or study interpretation. Different criteria, such as a significant increase in mortality, considerably more than 10% decreased body weight gain, body weight loss, and a significant decrease in incidences of neoplasms, would be more appropriate. Thus, the utility of MTDs from the range-finding studies to set appropriate high doses for the 6-month studies as defined by Paranjpe et al. cannot be easily evaluated by the reader.
In the present report, the rationale for dose selection was based on the 1-month range-finding studies and utilized all standard parameters such as body weight loss, body weight gain, adverse target organ toxicity, adverse clinical signs, mortality, and maximal feasible dose (MFD). In 8 out of 11 compounds profiled in this report, the MTD was the rationale for selecting the high dose in the pivotal 6-month study. Specifically, out of these 8 compounds, 7 had MTDs defined by mortality at a higher dose and 1 had lower body weight gain in the high-dose cohort than in controls in the range-finding study. Among the remaining 3 compounds, 1 used the MFD as a rationale for selecting the high dose, 1 used a limit dose rationale, and 1 high dose was based on a dose in the range-finding study that produced body weight gain decrements greater than 20% relative to controls. The high doses used in the pivotal 6-month studies were tolerated in all but 1 case.
The high doses in 2 out of 11 pivotal studies were based solely on decreased body weight gain in range-finding studies without any clinical signs, severe toxicity, and mortality. High doses selected for the pivotal 6-month studies in 7 out of 11 cases were established by mortality at higher doses in short duration range-finding studies, followed by establishment/refinement of MTD via target organ toxicity (n = 1), or as a fraction of the lethal dose (n = 4), or equivalent to a non-tolerated dose (n = 2). In studies where the top dose was established as a fraction of the lethal dose, the fraction applied was typically 40 to 50% of the lethal dose. This is consistent with the Center for Drug Evaluation and Research, U.S. Food and Drug Administration (FDA) communication to the sponsor that an appropriate high dose in a 6-month rasH2 mouse study can be 33% (one-third) to 50% (one-half) of the established lethal dose in a range-finding study if other findings do not justify lower doses.
The present series of studies suggests that if a clear non-tolerated upper dose limit is established in range-finding studies along with a lower dose that is tolerated with decreased body weight gain of ≤10% relative to the control group, the probability of developing non-tolerated toxicities that can impact the 6-month study interpretation is less than 10%. In other words, when decreased body weight gain in 1-month range-finding studies was used to set the high dose in the 6-month rasH2 mouse studies, the high-dose groups in the 6-month studies did not suffer significant increase in toxicity or mortality, and study interpretation was not affected. In this context, it is important to note that 2 of 11 test articles in the present series did produce test article–related tumors in the pivotal 6-month rasH2 mouse study despite decrements in body weight change. With respect to mortality in the 6-month studies, mortality in control groups ranged from 0 (most common) to 4 in a single sex (females in one study). Since 20 animals per sex per group are sufficient for statistical analyses of a rasH2 mouse carcinogenicity study (Morton et al. 2002), increased mortality was not considered test article related and did not impair study interpretation unless there were 5 or more deaths in either sex in the high-dose group. In the few programs with 5 or more deaths in a low- or intermediate-dose group, but fewer deaths in the high-dose group (no dose response), the increase in deaths was not considered test article related. Taken together, loss of up to 5 animals of one sex in one group and mean body weight no less than 80% of mean control body weight were considered acceptable.
The Executive Carcinogenicity Assessment Committee of the FDA provided feedback on proposed doses for 10 studies in the current series and suggested different doses than those recommended by the sponsor for 7 out of 10 studies. The FDA asked the sponsor (Pfizer) to increase the proposed dosages in 3 instances. In one case, the high dose was increased to the MFD because of the absence of any toxicity in the range-finding study (227× human area under the drug concentration curve [AUC] exposure multiple in the 1-month study). In the second case, the sponsor had proposed dosages in the pivotal 6-month study that were lower than in the range-finding study because of test article–related non-adverse crystal nephropathy/urinary bladder hyperplasia and slight body weight gain decrement (between 0.8× and 0.9× control). The FDA disagreed and asked the sponsor to conduct the studies using the same high and mid-doses as the range-finding study. In another case, the FDA asked the sponsor to increase the low dose alone to make it one-third the mid-dose such that the exposure multiple would be 1 to 2× human clinical exposure. In one study, the sponsor recommended the same doses for males and females, while the FDA suggested a broader range of doses (lower low dose and higher high dose) for males and lower doses for females.
The FDA asked the sponsor to reduce the proposed dosages in 3 instances. In the first case, the FDA asked the sponsor to lower the top dose despite the absence of adverse toxicity in the range-finding study. This may have been triggered by a concern associated with pharmacology-related effects observed in chronic rat studies and observations of non-adverse increased coagulation times in the 1-month range-finding mouse studies. In the second example for an oncology indication, the sponsor suggested 5 cycles of 10, 30, and 100 mg/kg/day for 4 weeks, followed by a week off; the FDA requested doses of 8, 15, and 75 mg/kg/day every day for 6 months. In this third example, the high dose recommended by the FDA produced early mortality, and the high dose was dropped to 50 mg/kg/day during the study. In all of the cases, the sponsor accepted the FDA’s dose recommendations. For one compound with an MTD in the range-finding study based on mortality at higher dose, the FDA commented that body weight and differences in body weight gain were not appropriate for high-dose selection for drugs intended to lower body weight in patients.
Paranjpe et al. (2015) proposed that a pivotal 6-month rasH2 mouse study should contain only a control group and 2 dose levels. A positive control (methyl nitrosourea or urethane) likely will also be required for the foreseeable future. Their simple and straightforward method for dose selection sets the high dose at one-half the MTD for males and two-thirds the MTD for females. The low dose would be half the high dose (one-fourth the MTD for males and one-third the MTD for females). Regulatory agencies currently expect 3 dose groups and appropriate controls, and it will take considerable time and discussion to reach broad agreement that the 2 dose groups might be acceptable. Paranjpe et al. (2015) indirectly acknowledged this by stating that if 3 dose groups are needed, they should be one-half, one-fourth, and one-eighth the MTD for males and two-third, one-third, and one-sixth the MTD for females. These recommendations are made without consideration of the factors defining the MTD in range-finding studies, the maximal feasible dose, exposures in mice at the proposed doses in each sex, or exposure margins compared to humans. All of these factors can be important when selecting the dose levels for rasH2 mouse carcinogenicity studies. For some compounds (e.g., those with microsomal enzyme induction and extensive metabolism), there may be no clear differentiation of exposure between one-half MTD and one-fourth MTD, and these doses would not demonstrate a clear dose response. It is often appropriate to set the low dose to produce clinically relevant exposures, while the mid-dose may also be influenced by exposure margins. For example, if the high dose is based on an estimated MTD that generates very high exposure margins exceeding 100×, it may be reasonable to set the mid-dose to produce exposure margins of 10× to 25× the AUC at the human efficacious exposure. Taken together, it is clear that multiple factors should be considered during the dose selection, and a generalized paradigm based only on fractions of MTD might not be suitable in many cases.
In the studies presented in this article, the MTD in the range-finding studies was effective in determining the high-dose levels for the 6-month studies, as there were no increases in mortality or toxicity sufficient to complicate the interpretation of data from the high-dose group in 10 of 11 studies. Based on the present small series of studies, traditional use of the MTD appears reasonable for high-dose selection in the 6-month rasH2 mouse studies. This contradicts the conclusions of Paranjpe et al. (2015). Until additional studies can be evaluated and regulatory authorities can reach a new consensus on high-dose selection, all available data including the traditional use of the MTD should be considered when determining the high doses for the 6-month rasH2 mouse studies.
We support the proposal to allow the high dose to be the limit dose of 1,500 mg/kg/day if the same criteria applied to the limit dose for other rodent carcinogenicity studies are also applied to rasH2 mouse studies (Food and Drug Administration 2002; ICH 2008). It seems reasonable to use the limit dose as the high dose if the human dose does not exceed 500 mg/day and if the limit dose produces exposures of at least 10× human AUC at the maximum recommended human dose. If these criteria are not met, then a high dose exceeding the limit dose may be appropriate for rasH2 mice. Others agree with this approach (Jacobs and Brown 2015).
In a previous publication, Paranjpe, Denton, and Elbekai (2014) suggested microscopic examination of all protocol tissues and necropsy observations only in high-dose and control animals, while examining only target tissues in low- and intermediate-dose groups in the pivotal 6-month studies. If the high doses in 6-month studies reduce body weight, increase mortality, and reduce incidences of neoplasia in males in 75% of studies and in females in 45% of studies as indicated by Paranjpe et al. (2015), then failing to examine all tissues in the low- and mid-dose groups may result in missing test article–related neoplastic findings in some studies. Therefore, examining only target tissues should be considered only if survival and body weights are comparable in control and high-dose groups. Moreover, the trend test cannot be used to detect low incidence, test article–related, dose-dependent neoplastic effects if all tissues in all groups are not examined. Microscopic examination of all protocol tissues in all groups administered the test article provides the most complete, robust assessment of neoplastic potential of a compound in the rasH2 mouse model and eliminates possible delays and costs incurred if a regulatory agency would request examination of all tissues in low- and intermediate-dose groups when this was not done initially.
In summary, two different surveys that reviewed the use of the MTD to effectively select high doses in rasH2 mouse carcinogenicity studies reached different conclusions. Paranjpe et al. (2015) determined that high doses based on MTDs estimated in range-finding studies usually were too high, producing significant toxicity, increased mortality, and decreased incidences of neoplasia in the 6-month studies. In a separate series of studies presented in this article, the MTDs estimated in the range-finding studies appeared to be appropriate for high-dose selection in the 6-month studies without negative impact on toxicity, mortality, or study interpretation. The standard study design with 3 dose groups, 1 vehicle control group, and 1 positive control group appeared reasonable and appropriate based on the second survey. If sponsors adopt the approaches recommended by Paranjpe et al. (Paranjpe, Denton, and Elbekai 2014; Paranjpe et al. 2015), sponsors should consider reaching prior agreement on study design and dose selection with all major regulatory regions (United States, Europe, and Japan) that will be asked to evaluate and register the compound so that the final study will be accepted globally.
Footnotes
Author Contribution
All authors (PRN, DM) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
*This is an opinion article submitted to the Regulatory Forum and does not constitute an official position of the Society of Toxicologic Pathology or the journal Toxicologic Pathology. The views expressed in this article are those of the authors and do not necessarily represent the policies, positions, or opinions of their respective organizations. The Regulatory Forum is designed to stimulate broad discussion of topics relevant to regulatory issues in toxicologic pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to
