Abstract
We previously reported an increased incidence of thrombotic toxicities in Sprague-Dawley rats administered the highest dose level of a hyperglycosylated analog of recombinant human erythropoietin (AMG 114) for 1 month as not solely dependent on high hematocrit (HCT). Thereafter, we identified increased erythropoiesis as a prothrombotic risk factor increased in the AMG 114 high-dose group with thrombotic toxicities, compared to a low-dose group with no toxicities but similar HCT. Here, we identified pleiotropic cytokines as prothrombotic factors associated with AMG 114 dose level. Before a high HCT was achieved, rats in the AMG 114 high, but not the low-dose group, had imbalanced hemostasis (increased von Willebrand factor and prothrombin time, decreased antithrombin III) coexistent with cytokines implicated in thrombosis: monocyte chemotactic protein 1 (MCP-1), MCP-3, tissue inhibitor of metalloproteinases 1, macrophage inhibitory protein-2, oncostatin M, T-cell-specific protein, stem cell factor, vascular endothelial growth factor, and interleukin-11. While no unique pathway to erythropoiesis stimulating agent-related thrombosis was identified, cytokines associated with increased erythropoiesis contributed to a prothrombotic intravascular environment in the AMG 114 high-dose group, but not in lower dose groups with a similar high HCT.
Introduction
Thrombosis is a multifactorial process. Virchow’s triad, postulated over 150 years ago, described changes in blood coagulability, the vessel wall, and blood flow as required participants for thrombosis formation. This hypothesis remains today the foundation for described risk factors that contribute to thrombosis (Chung and Lip 2003; Lowe 2003). A wide range of experimental studies in animals and meta-analysis of published clinical trial data has accumulated to identify factors/mechanisms that link at-risk patient populations to a thrombotic event (Barbui et al. 2010). However, due to the multitude of interrelated factors known to influence hemostasis, no biomarker/biomarkers have proven sufficiently robust to confidently predict thrombotic complications in many patients (Fox and Kahn 2005).
Identifying the underlying pathogenesis of thrombosis in healthy animals assigned to preclinical toxicity studies is equally elusive (Ramot and Nyska 2007). Treatment-related thrombotic events in animals are rare, unpredictable events with a large list of potentially interrelated contributing factors. For this reason, experimental models of thrombosis have evolved to reliably initiate thrombosis through direct or indirect injury to the vessel wall in vivo (Bodary and Eitzman 2009; Vilahur, Padro, and Badimon 2011). Genetically modified mouse models of thrombosis have also been created to investigate genetic diseases (Denis et al. 1998) and to fulfill an intended purpose such as evaluating efficacy of novel antithrombotic therapeutics (Magallon et al. 2011).
We recently reported results from a retrospective study demonstrating an increased incidence of thrombotic events in preclinical species administered an erythropoiesis stimulating agent (ESA), as not solely related to a high hematocrit (HCT). Thrombotic toxicities reported in a subset of preclinical toxicology studies conducted with all different classes of ESAs (Elliott, Pham, and Macdougall 2008) were related to ESA dose level, dose frequency, and study duration despite a similarly high HCT across all dosed groups (Andrews, Pyrah, et al. 2013). We confirmed this observation by eliciting thrombotic toxicities in the highest dose groups of Sprague-Dawley rats dosed 3 times weekly for 1 month with a novel, hyperglycosylated analog of recombinant human erythropoietin (rHu-EPO), AMG 114, but not in the lowest dosed groups or when the higher dose levels were administered once weekly despite a similarly high HCT across all dose groups and experimental conditions (Andrews, Boren, et al. 2013). The pathogenesis of ESA-related thrombotic toxicities in the highest dose groups was examined by conducting additional investigative studies that compared biologic differences between rats administered AMG 114 at high- or low-dose levels prior to (day 9) and after (day 30) a maximum HCT was achieved, on a 3 times weekly dosing schedule. In these studies, we identified an increased magnitude of erythropoiesis and related sequelae as prothrombotic risk factor/factors present in the AMG 114 high, compared to the AMG 114 low-dose group despite a similar HCT at both dose levels (Andrews, Boren, et al. 2013). While an extremely high HCT was generally accepted as a prothrombotic risk factor, these studies suggested it was a requisite cofactor in ESA-related thrombosis since no histologic evidence of thrombosis was found in rats in the 9-day study.
The goals of the current investigation were to (1) identify prothrombotic mechanisms unique to the AMG 114 high-dose group with thrombotic toxicities compared to the low-dose group with no thrombotic toxicities despite a similar HCT at both dose levels and (2) define the pathogenesis of thrombosis in the AMG 114-dosed animals with histologic evidence of thrombotic toxicities.
Materials and Method
Investigative Studies with AMG 114 in Rats
Two investigative studies were conducted in Crl:CD(SD) male rats (approximately 10 weeks old) cared for in accordance to the Guide for the Care and Use of Laboratory Animals, 8th ed. (National Research Council 2011) at the Association for Assessment and Accreditation of Laboratory Animal Care internationally accredited facilities. Detailed explanation of the test article, study conditions, and experimental conduct were previously described (Andrews, Boren, et al. 2013; Andrews, Pyrah, et al. 2013). Briefly, rats were administered vehicle, or AMG 114, IV, 3 times weekly at a low (maximal pharmacologic, 1.0 µg/kg) or high (suprapharmacologic, 500 µg/kg) dose level for up to 9 days or 1 month. Dose levels and dose frequency were chosen for investigative purposes as both achieved a similar maximum HCT in rats despite differing magnitudes of increased erythropoiesis (represented by increased reticulocytes; Figure 1), but thrombotic toxicities only occurred at the high-dose level (Andrews, Boren, et al. 2013).
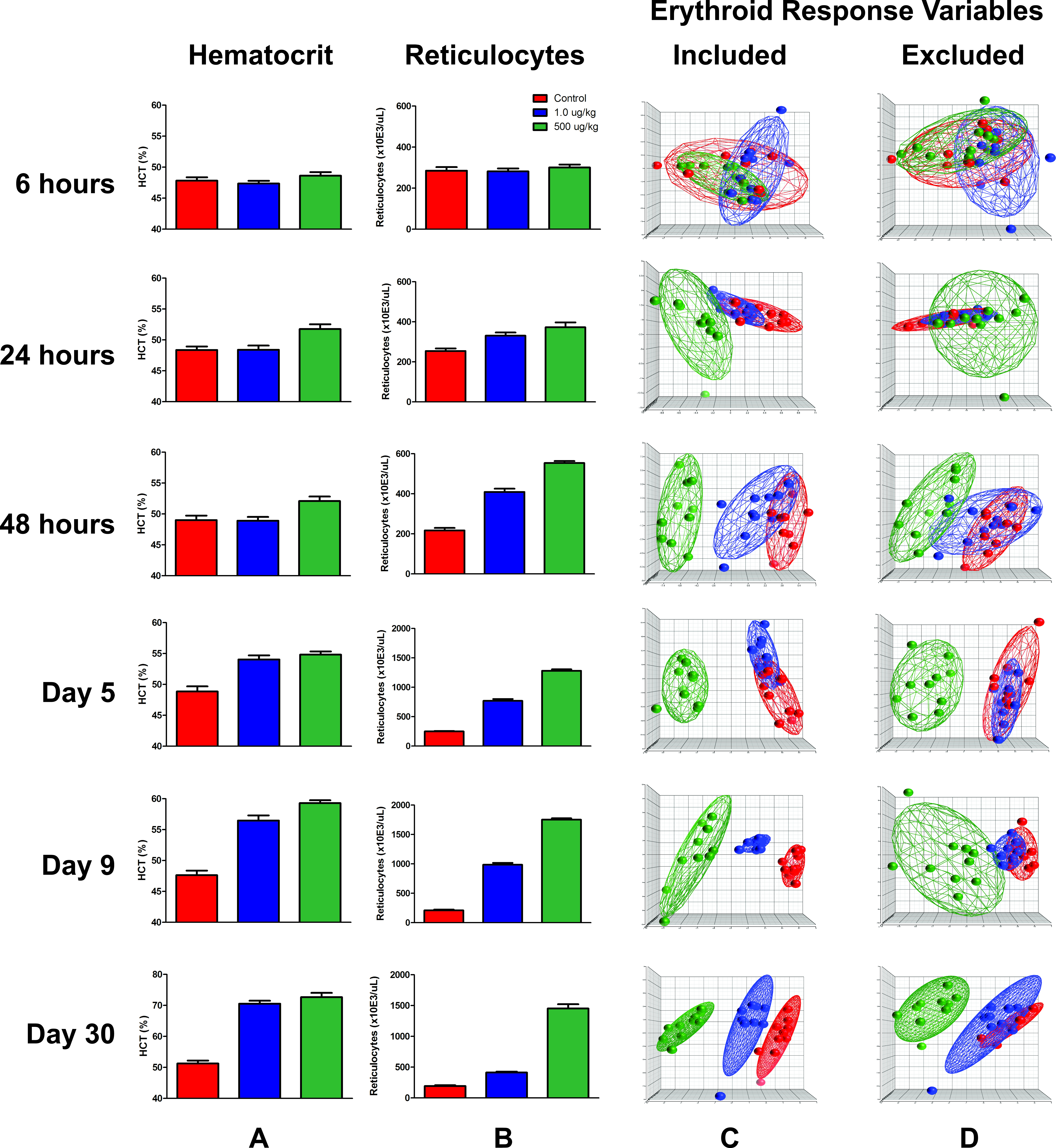
Principle component analysis (PCA) of study data obtained from Sprague-Dawley rats administered a low- (1.0 µg/kg) or high (500 µg/kg)-dose level of a hyperglycosylated analog of recombinant human erythropoietin, AMG 114, three times weekly for up to 1 month. Results of the PCA that included (C) or excluded (D) end points that measured increased erythropoiesis (erythroid response variables) are shown mapped by a scatter plot of individual animals color coded according to dose level as red (vehicle control), blue (low AMG 114 dose group), or green (high AMG 114 dose group). Ellipsoids represent the distribution of study end points for individual animals for each dose group within ± 2 SD of the mean. Rats administered the high-dose level of AMG 114 were discriminated from those in the low-dose group despite a similar high HCT (A) and differing magnitudes of increased erythropoiesis [represented by increased reticulocytes (B)] at 24 and 48 hr after a single dose of AMG 114. The PCA of study data that excluded erythroid response variables at day 5 and day 9 did not influence the separation of the AMG 114 high from the low dose and vehicle control groups. The AMG 114 low-dose group behaved more like the vehicle control group at all time points except for day 9 and day 30 when erythroid response variables were included in the PCA.
Serum and Plasma Measurements
Blood was collected by syringe from the jugular vein of 10 animals per group and placed into tubes containing ethylenediaminetetraacetic acid (EDTA) or sodium citrate as anticoagulants or placed into tubes without anticoagulant. Time points for blood collection in the 9-day study included 6 (day 1), 24 (day 2), or 48 (day 3) hr post the first dose, 48 hr after the second dose (day 5), or 48 hr after the fourth dose (day 9). In the 1-month study, rats had blood drawn 24 hr after the 13th dose (day 30) prior to necropsy. All study data included for analysis are listed in Table 1 and consisted of hematology end points generated from EDTA-anticoagulated whole blood by the Advia 2120 Hematology Analyzer (Siemens Healthcare Diagnostics, Deerfield, IL), clinical chemistry end points analyzed from available serum by the Roche Modular Analytics System (Hoffmann-La Roche, Basal, Switzerland), and coagulation end points (prothrombin time [PT], activated partial thromboplastin time [APTT], and fibrinogen) from sodium citrate anticoagulated blood. In the 1-month study, reported values for PT and APTT were excluded from analysis due to high concentration of sodium citrate anticoagulant in the decreased plasma volume (due to the high HCT) erroneously prolonging clotting times. When sufficient serum volume remained, animals were assayed for serum iron and unbound iron-binding capacity (UIBC) on the Roche Modular Analytics System (Hoffman-La Roche, Basal, Switzerland). Total iron-binding capacity (TIBC) and percentage of transferrin saturation (TSAT) were calculated from measured values when available. Assay for ferritin was performed at Antech Diagnostics (Morrisville, NC) on an aliquot of serum stored at −80°C and shipped on dry ice. Multiplexed protein immunoassays were applied to rat sodium citrate anticoagulated plasma and analyzed with the RodentMAP Version 2 multiplex immunoassay platform developed and optimized at Myriad Rules Based Medicine (http://www.myriadrbm.com; Austin, TX). The antigen panel consisted of 57 proteins that were assayed via bead-based immunoassays and read out on a Luminex 200 system (Luminex, Austin, TX). Analytes previously determined with poor cross reactivity to rat in this panel that included interferon gamma (IFN-γ), interleukin-1 alpha (IL-1α), interleukin-1 beta (IL-1β), IL-6, monocyte chemotactic protein-1 (MCP-1), and tumor necrosis factor alpha (TNF-α) were assayed by Myriad Rules Based Medicine using separate bead-based immunoassay panels specifically optimized for rat antigens. Sample values of analytes reported below the limit of detection in multiplexed protein immunoassays were assigned a numeric value one-half the least detectable dose and included for statistical analysis as a means to overcome the inherent insensitivity, yet maximize the benefit of multiplexed discovery-based assay platforms (Liu M et al. 2005; Uh et al. 2008). Specialized coagulation end points including thrombin antithrombin complexes (TAT), plasminogen activator inhibitor-1 (PAI-1), and von Willebrand factor (vWF) were assayed by enzyme-linked immunosorbent assay from commercially available kits at the Cornell Comparative Coagulation Laboratory (Ithaca, NY) on an aliquot of citrated plasma stored at −80°C and shipped on dry ice for analysis. Activated factor VII (FVIIa) was also assayed at Cornell using the STA clot VIIa-rTF assay kit (Diagnostica STAGO).
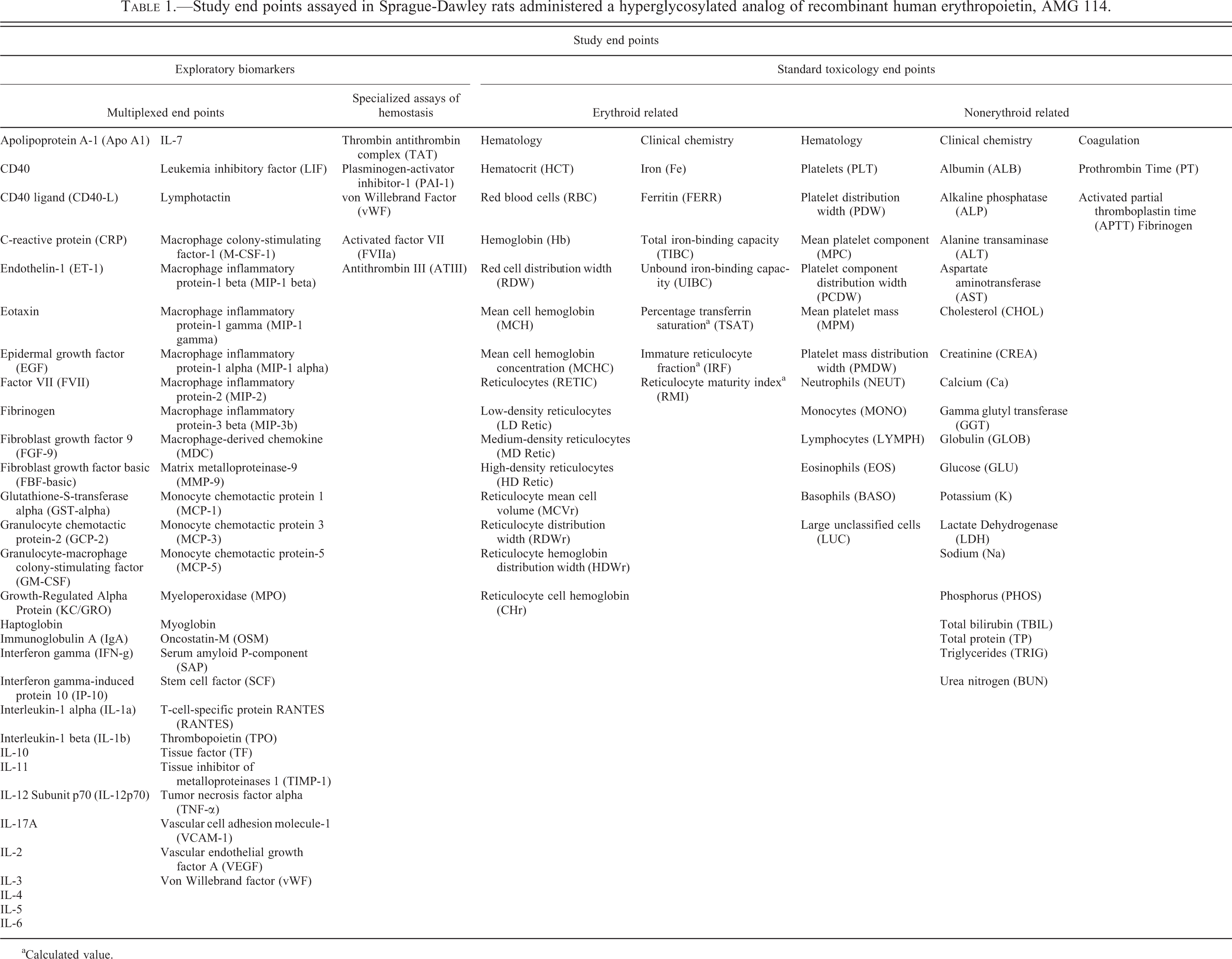
Study end points assayed in Sprague-Dawley rats administered a hyperglycosylated analog of recombinant human erythropoietin, AMG 114.
aCalculated value.
Anatomic Pathology
Necropsy schedule and tissue handling/routine staining procedures for animals in the 9-day and 1-month study were previously described (Andrews, Pyrah, et al. 2013). Briefly, histology end points included in the current analyses were target organs previously identified with microscopic evidence of thrombus or indirect evidence of a thrombotic event (such as an infarct and/or combinations of findings such as focal necrosis, fibrosis, inflammation, and hemorrhage) in ESA-dosed animals (Andrews, Pyrah, et al. 2013). These included heart/heart valve, stomach, kidney, spleen, bone/bone marrow from femur and sternum, liver, lung with large bronchi, duodenum, jejunum, ileum, and mesenteric vessels. Additionally, characteristic ESA-related toxicities that included valvulopathy and/or stomach erosions described previously were included in the analysis (Andrews, Pyrah, et al. 2013).
Historical Data
Due to the low incidence of thrombotic toxicities in animals belonging to the AMG 114 high-dose group in the study described above, study end points from 20 Crl:CD(SD) rats (10/sex/group) at least 10 weeks of age dosed at 30 or 100 µg/kg/dose AMG 114 and 10 female Crl:CD(SD) rats at least 10 weeks of age dosed at 500 µg/kg AMG 114, 3 times weekly for 1 month, were retrieved from the Amgen internal archives and included in the analysis of biomarkers altered in animals with histologic evidence of an ESA-related thrombotic toxicity. Irrelevant of dose, including these animals in the analysis increased the number of AMG 114-dosed animals with thrombotic toxicities, increasing our ability to identify biomarkers that correlated with ESA-related thrombosis.
Statistical Analysis
Statistical analyses and visualization tools commonly used to interpret high-density data sets (i.e., DNA microarrays) were applied to study data in order to uncover dose-related biologic patterns and associations amidst known similarities associated with treatment-related increased erythropoiesis (measured by increased reticulocytes) and HCT. These techniques have proved successful to approach interpretation of preclinical toxicity studies with high-density data sets that include histology and clinical pathology end points (Hamadeh et al. 2002; Lobenhofer et al. 2006; Heinloth et al. 2007).
For clarity, the end points that comprised the study data are listed in Table 1. “Exploratory biomarkers” refers to the study data set obtained from multiplexed assays (Rules Based Medicine) and the specialized coagulation end points assayed at the Cornell University Comparative Coagulation Laboratory. The term end point(s) refers to any measured variable in the cumulative data set of exploratory biomarker, hematology, clinical chemistry, coagulation, and organ weight end points. End points designated “erythroid-related” describe increased red cell mass (HCT, red blood cells [RBC], hemoglobin [Hb], red cell distribution width [RDW], mean cell hemoglobin [MCH], mean cell hemoglobin concentration [MCHC]) and/or increased erythropoiesis (reticulocytes, reticulocyte parameters, and iron parameters). The remaining end points are designated “non-erythroid related.”
Principal component analysis (PCA) and hierarchical clustering was used to explore the natural groupings of study data using Partek Genomic Suite statistical software package (Partek, Inc, St. Louis, MO). Prior to analysis, end points were transformed into Z-scores to account for differing dynamic ranges of the measured end points and to satisfy assumptions for normality and variance.
Univariate and multivariate statistical analyses were applied on the study data to discover end points present solely in the AMG 114 high-dose group compared to the AMG 114 low- and vehicle control dose groups, and those present both in the AMG 114 high- and low-dose groups compared to vehicle controls (generally treatment related). Thirty-one statistical tests were performed on each nontransformed response variable that included pairwise t-tests between dose levels consisting of 0 µg/kg versus 1.0 µg/kg, 0 µg/kg versus 500 µg/kg, 1 µg/kg versus 500 µg/kg (n = 15) at each time point, and between time points consisting of 6 hr versus 24 hr, 6 hr versus 48 hr, 6 hr versus 96 hr, and 6 hr versus 192 hr (n = 12) for each dose level in a 1-way multigroup analysis of variance (ANOVA) by dose (n = 3) and 2-way ANOVA by dose and time (n = 1). For each analysis, p values were adjusted for multiple comparisons using false discovery rate. Data from at least 5 of the 10 animals were required in each dose groups for inclusion in analyses; a factor that limited comparison of several end points between the 9-day and 1-month study due to insufficient plasma/serum volume available from high HCT animals for sample analysis after 1 month of repeat dosing with AMG 114. A cluster analysis (k-means clustering) was then applied to Z-transformed data of end points (92/124) that reached statistical significance (p < .01) in any statistical test across time or dose (k = 10) in order to colocalize analytes and identify trends in analytes across the study time points. The Z-scores were clustered by k-means (n = 10) and visualized through translation of numerical values into a heat map. A Pearson’s correlation coefficient analysis was used to identify correlative relationships between altered hematologic, clinical chemistry, coagulation, and exploratory biomarker end points relative to HCT and reticulocytes using data up to and including day 9 and at day 30. Cutoff values for inclusion in analysis were r ≥ .60 and p ≤ .001. Study end points unique to the AMG 114 high- or low-dose group at day 9 or day 30 were determined by construction of Venn Diagrams generated by the Partek Genomic Suite statistical software package (Partek, Inc, St. Louis, MO).
Biologic interpretation of the altered protein biomarkers unique to the AMG 114 high-dose group was guided by pathway analysis tools that included IPA (Ingenuity Systems, www.ingenuity.com) and the GeneGo Metacore database (GeneGo, Inc. www.genego.com). Proteins amenable to analysis were those with a corresponding Entrez Gene identifier that matched them to the corresponding canonical signaling pathways, cellular and/or physiologic functions contained within the pathway analysis tools.
Results
Discrimination of AMG 114 Dose Groups Using Multidimensional Analysis Tools Applied to High-density End Points
PCA of all study end points (n = 127) collected at each time point sampled, initially separated away the AMG 114 high, from the AMG 114 low and vehicle control-dosed groups at 24 hr after a single dose (Figure 1). The separation was distinct at 48 hr and each subsequent time point through day 30. Separation of the AMG 114 high-dose group was not sensitive to exclusion of erythroid end points at day 5, day 9 or day 30, indicating a strong influence of nonerythroid end points that distinguished the AMG 114 high-dose group at these time points. Unsupervised hierarchical clustering patterns also organized the AMG 114 high-dose group in a similar manner (data not shown). From day 5 onward, results of both analyses indicate the AMG 114 low-dose group behaved more like the vehicle control group than those treated with high-dose AMG 114.
Statistical Results for Comparison of the AMG 114 High- and Low-dose Group Study End Points
To identify end points responsible for dose-level separation identified in the multivariate analyses, a series of univariate and multivariate statistical tests (see Materials and Method section) were applied to the study data to identify end points that reached statistical significance in the AMG 114 high- and low-dose groups compared to vehicle controls (generally treatment related) and in the AMG 114 high, but not the AMG 114 low and vehicle control dose groups (unique to the AMG 114 high-dose group) at each time point. The p values of all 31 statistical tests were used to construct a heat map of Z-transformed data that were clustered into significance patterns and organized by AMG 114 dose level and time point (Figure 2). End points of interest visualized as unique to the AMG 114 high-dose group were generally classified into bins of exploratory biomarkers that included a subset of chemokines, cytokines, and growth factors, white blood cells, and platelets (PLTs)/PLT parameters. Altered end points that moved similarly in both AMG 114 dose groups compared to controls were comprised predominantly of end points associated with increased erythropoiesis, particularly those that measured reticulocytes. Individual study end points were further analyzed by review of box and whisker plots constructed for each end point on an individual animal basis over time (data not shown). The nonerythroid-related end points considered altered in a biologically relevant manner relative to AMG 114 administration (including those with, or approaching, statistical significance) are listed in Table 2 and Figure 3.
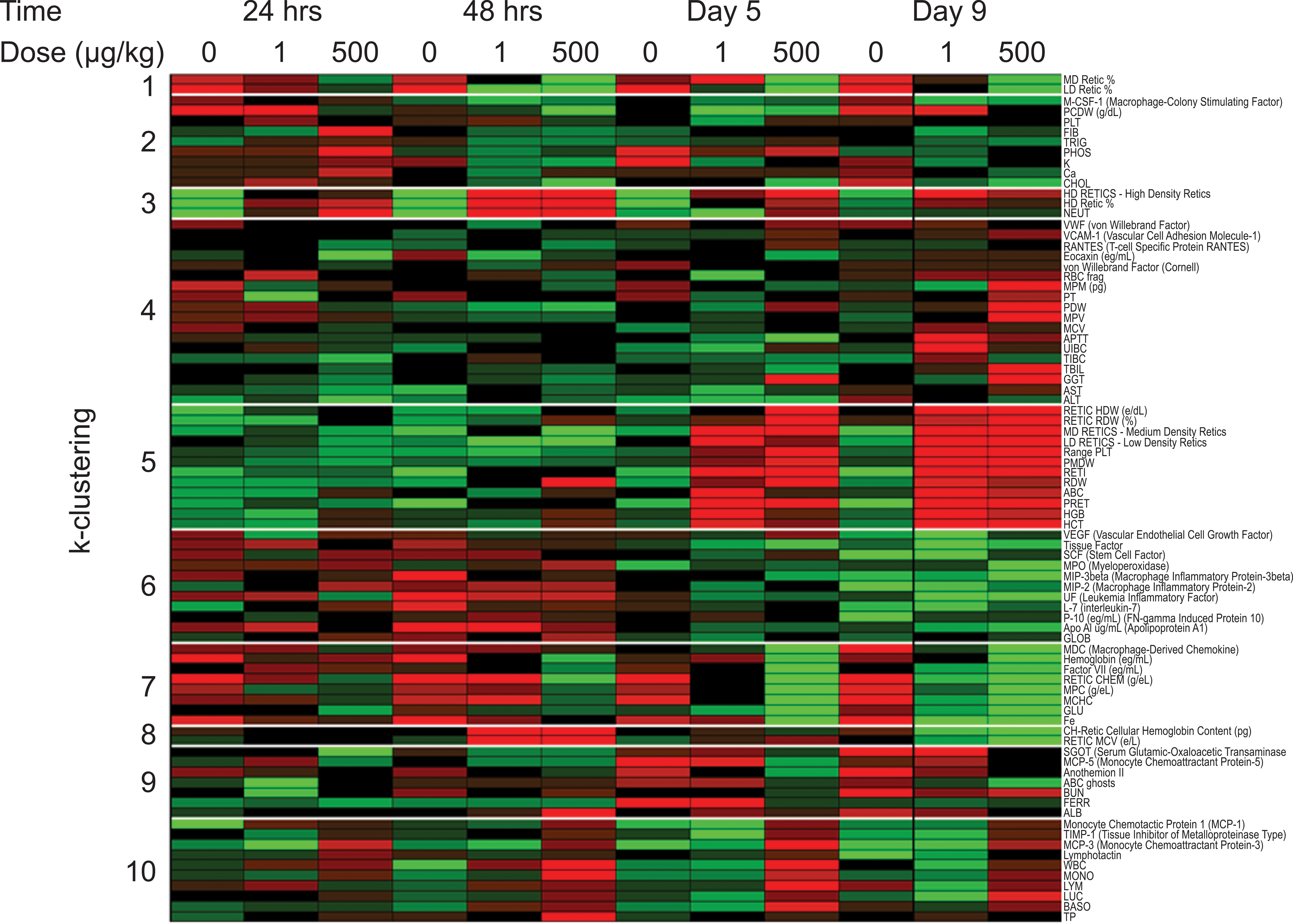
Heatmap organized by significance pattern of Z-transformed study data from rats dosed with a hyperglycosylated analog of recombinant human erythropoietin, AMG 114, at a high (500 µg/kg) or low (1.0 µg/kg) dose level in a 9-day study. End points included for analysis reached statistical significance (p < .5) in one or more statistical tests that compared all study data from rats dosed with AMG 114 across dose levels and time points (see Materials and Method). Analyte data (y-axis) was clustered using k-means (k = 10) whereas treatments (averaged across replicate animals) were organized on the x-axis according to user preference. Color indicated whether values were above (red), below (green), or at the mean value (black) for respective analytes across the study. Study data obtained at 6 hr was excluded since Z-scores reflected changes in baseline attributed to diurnal variation. Clusters along the y-axis grouped together analytes that demonstrated similar behavior across the entirety of the study. The cluster analysis revealed several patterns of analyte behavior that had different combinations of dose response and time course dependencies. Cluster 5, for example, contained analytes that were increased by both dose levels of AMG 114, whereas cluster 10 contains analytes that were increased only at the high-dose level. The cluster diagram is also useful for highlighting the time dependency of the changes and identifying the thresholds for triggering specific changes in the animals. Analytes grouped in cluster 5 seem to be increased at the 96-hr threshold whereas changes associated with cluster 10 start at 24 hr.
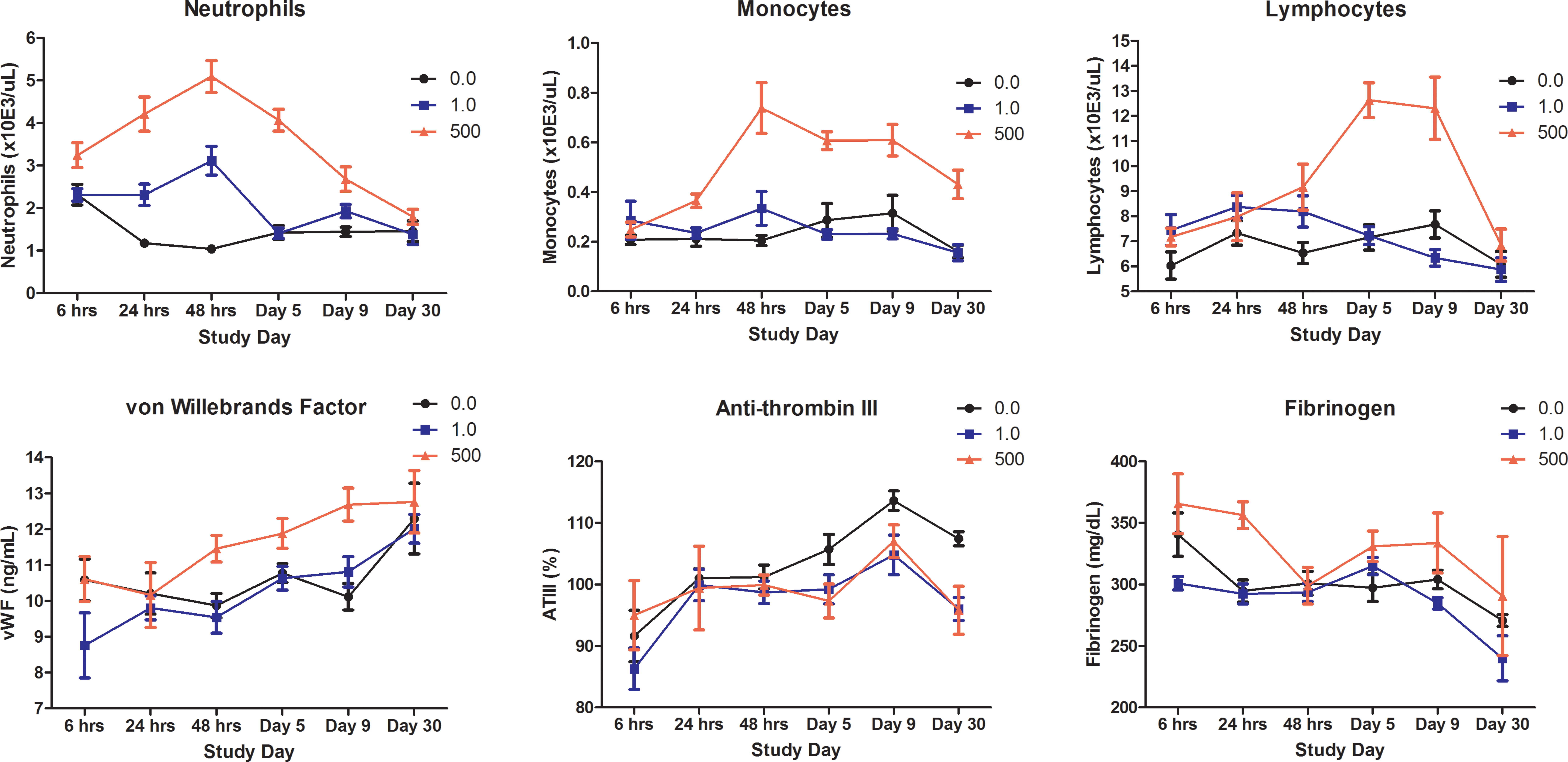
A subset of quantitative study end points including neutrophils, monocytes, lymphocytes, vWF, ATIII, and fibrinogen graphed over time by dose level in rats administered vehicle control (black), a low (1.0 µg/kg, blue)- or high (500 µg/kg, red)-dose level of a hyperglycosylated analog of recombinant human erythropoietin, AMG 114, three times weekly for up to 1 month.
Biologically relevant nonerythroid-related study end points altered in Sprague-Dawley rats dosed with AMG 114 at a low (1.0 µg/kg)- or high (500 µg/kg)-dose level, 3 times weekly in a 9-day or 1-month investigative study.
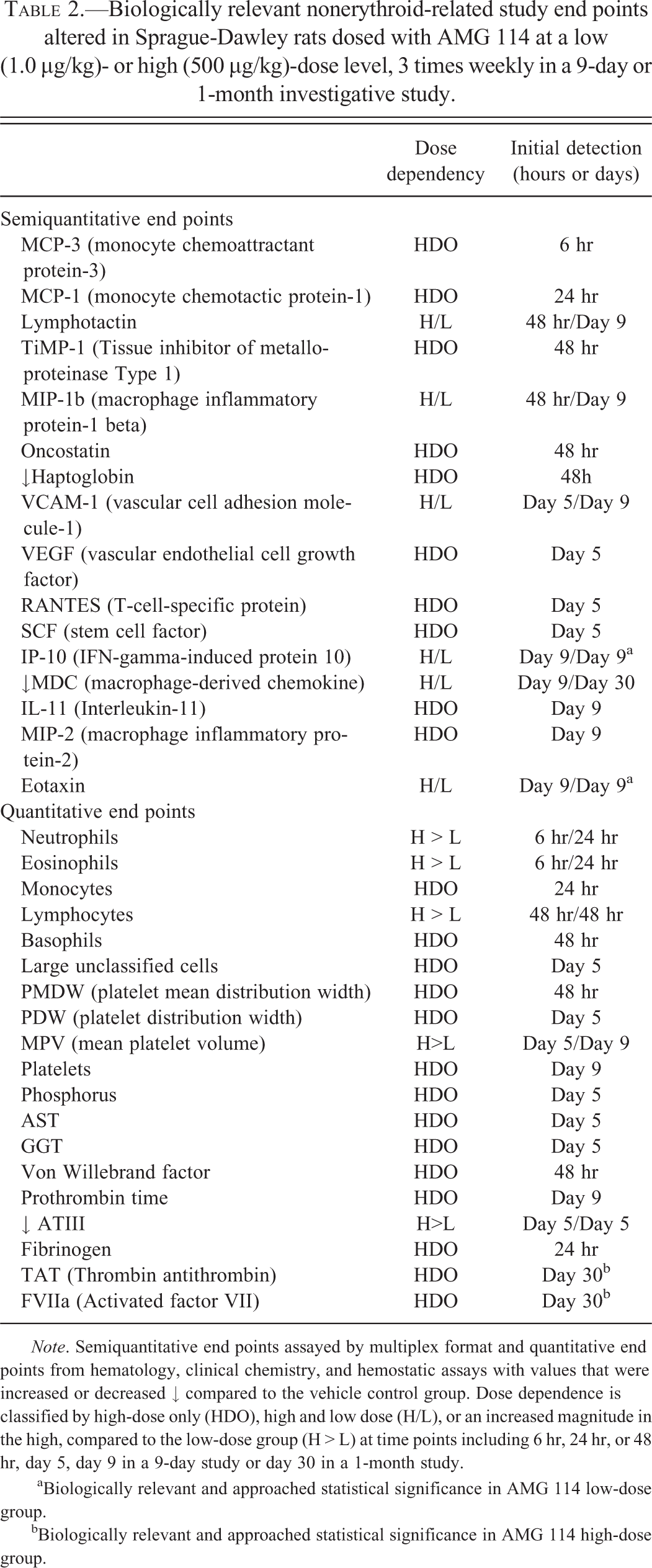
Note. Semiquantitative end points assayed by multiplex format and quantitative end points from hematology, clinical chemistry, and hemostatic assays with values that were increased or decreased ↓ compared to the vehicle control group. Dose dependence is classified by high-dose only (HDO), high and low dose (H/L), or an increased magnitude in the high, compared to the low-dose group (H > L) at time points including 6 hr, 24 hr, or 48 hr, day 5, day 9 in a 9-day study or day 30 in a 1-month study.
aBiologically relevant and approached statistical significance in AMG 114 low-dose group.
bBiologically relevant and approached statistical significance in AMG 114 high-dose group.
An initial assessment of biologic relevance related to altered end points unique in AMG 114 high-dose animals was guided by pathway analysis tools. Modulated end points included for analysis were limited to those with corresponding Entrez Gene identifiers and included MCP-3, MCP-1, tissue inhibitor of metalloproteinases 1 (TIMP-1), oncostatin M (OSM), vascular endothelial growth factor (VEGF), T-cell specific protein (RANTES), stem cell factor (SCF), IL-11, and macrophage inhibitory protein 2 (MIP-2). Results from the pathway analysis software generally identified the most relevant biological functions associated with hematological system development and function, and immune modulation. Subordinate cellular functions common to both processes were leukocyte trafficking, cell-to-cell signaling and interaction, cellular growth and proliferation, and cell death. Results from analyses that included all altered protein biomarkers in AMG 114-dosed animals (including lymphotactin, IFN-γ-induced protein 10 [IP-10], macrophage inhibitory protein 1 beta [MIP-1β], and eotaxin) generally promoted the more ubiquitous cellular functions listed above to a position of greater statistical significance than the more specific identifiers of hematopoiesis or immune modulation.
Cooperative End Points with Hematocrit or Reticulocytes in Blood of AMG 114-dosed Rats
We previously recognized increased numbers of immature reticulocytes as a prothrombotic risk factor increased in the AMG 114 high, compared to the low-dose group, before a high HCT was achieved (Andrews, Pyrah, et al. 2013). We also determined high HCT as a requisite cofactor, but not solely causal, in ESA-related thrombosis that occurred in the highest dose groups of rats only (Andrews, Boren, et al. 2013; Andrews, Pyrah, et al. 2013). To further explore the biologic consequences of increased reticulocytes or high HCT to thrombosis in AMG 114-dosed rats, a correlative analysis was conducted between all study end points and reticulocytes or HCT prior to (9-day study) or after a high HCT was achieved (1-month study).
Results from the correlation analysis (Figure 4) identified unique associations between reticulocytes and increased MCP-3, MCP-1, TIMP-1, and mean platelet volume (MPV) in the AMG 114 high-dose, but not the low-dose group, prior to a high HCT (9-day study). No unique nonerythroid-related biomarkers correlated to reticulocytes at day 30 (serum iron only), an expected consequence of higher numbers of RBCs (as a component of high HCT) relative to reticulocytes in circulation at later time points. No unique correlations of end points to HCT were identified at day 9. At day 30, rare end points that correlated to HCT (MCP-3, large unclassified cells [LUC], monocytes) did not provide a mechanistic link to thrombosis conferred by high HCT.

Correlation analysis of study data to hematocrit or reticulocytes in Sprague-Dawley rats administered a high (500 ug/kg)- compared to a low (1.0 ug/kg)-dose level of a hyperglycosylated analog of recombinant human erythropoietin, AMG 114, three times weekly at time points including 6, 24, and 48 hr, day 5 and day 9 in a 9-day study and at day 30 in a 1-month repeat dose study. Venn Diagrams intersect biologically relevant end points (r ≥ .60 and p value < .001) that correlated to (A) hematocrit or (B) reticulocytes in a Pearsons correlation analysis segregated by AMG 114-dose level and time. Abbreviations of end points are found in Table 1.
Biomarker Profiles in AMG 114 High-dose Animals with Thrombotic Toxicities
The AMG 114 high-dose group animals shared similar prothrombotic risk factors through day 9, and in a 1-month repeat dose toxicology studies. However, only rare individuals have histologic evidence of thrombosis following 30 days dosing with AMG 114. To determine whether these animals could be identified early in the study based on predisposing factors prior to an achieved high HCT, individual animals that separated from their group mates by PCA were examined for subclinical pathologies. Two animals in the AMG 114 high dose were visualized in a unique space in the PCA that excluded erythroid end points at day 9 (Figure 1D). An analysis of their study end points identified different profiles of altered analytes, which may have predisposed them to thrombosis if dosed for the remainder of the 1-month study. One animal was discriminated due to altered hemostatic end points associated with a consumptive coagulopathy (decreased fibrinogen, increased TAT, prolonged PT, and APTT), large PLT size (increased MPV, platelet distribution width [PDW], mean platelet mass [MPM]), and increased endothelin-1, eotaxin, and glutathione-s-transferase alpha (GST-α). The other animal had increased higher blood concentrations of inflammatory end points that included IL-4, lymphotactin, MIP-2, MCP-3, SCF, RANTES, TIMP-1, vascular cell adhesion molecule 1 (VCAM-1), VEGF, MCP-1, and ferritin as well as large platelet size (MPV, PDW, PLT) increased gamma glutyl transferase (GGT) and total bilirubin.
All study end points from all AMG 114–dosed animals with histologic evidence of thrombosis in 1-month repeat dose toxicology studies were evaluated for common subclinical pathologies that selected individual animals for thrombosis. The results of this analysis did not identify any underlying subclinical pathologies or common biomarker patterns in AMG 114–dosed rats (n = 120) with histologic evidence of an AMG 114–related pathology (n = 25), and/or subsets of pathologies that included infarct/thrombosis (n = 17), valvulopathy (n = 15), or gastric erosion/ulceration (n = 18) when evaluated by hierarchical clustering analysis of study end points anchored to a respective pathology (data not shown). Two animals with an infarct/thrombus in the heart wall or heart valve had multiple end points altered of sufficient magnitude to be considered uniquely higher compared to the remaining AMG 114–dosed animals (± thrombosis). Altered end points in these animals were prothrombotic biomarkers (markedly decreased Antithrombin III [ATIII], increased FVIIa, or increased TAT) and end points associated with acute (increased TNF-α, IL-1a, IL-1b) and chronic inflammation (increased IL-2, IL-7, IP-10, lymphotactin, MCP-1, MCP-3, OSM, RANTES, SCF, and TIMP-1). Sporadic AMG 114–dosed animals with no histologic evidence of thrombosis had one, or occasionally more, of the same end points altered to a similar magnitude. Of the 18 animals with an ESA-related gastric erosion/necrosis/ulceration, 3 had high outlier blood concentrations of TAT complexes (increased ∼15-fold above mean value of animals with no thrombosis) and one or more increased inflammatory end points that included IL-1a, IL-6, TNF-α, TIMP-1, IP-10, RANTES, SCF, FGF-9, and/or IL-3. Hierarchical clustering analysis identified 1 additional animal with a biomarker profile similar to animals with the gastric lesion, but no histologic evidence was found. Sex-related differences in exploratory biomarkers were observed but did not impact the incidence of thrombotic events which were similar between male and female animals.
Discussion
We previously reported results that increased dose level, dose frequency, and dosing duration, along with a high HCT, were associated with an increased incidence of thrombotic toxicities in ESA preclinical toxicology studies conducted in Sprague-Dawley rats, Beagle dogs, and cynomolgus monkeys (Andrews, Boren, et al. 2013). Following on investigative studies we conducted in rats administered a hyperglycosylated analog of rHu-EPO (AMG 114) confirmed our original observation that increased ESA dose and dose frequency was associated with an increased incidence of thrombotic toxicities, and not solely related to a high HCT. Correlations between HCT and thrombotic risk are also quite variable in preclinical species and patients with diverse disease states supporting a multifactorial pathogenesis for this observation (Andrews, Boren, et al. 2013; Andrews, Pyrah, et al. 2013). Subsequently, we indentified an increased magnitude of ESA-stimulated erythropoiesis as a prothrombotic risk factor increased in the AMG 114 high, compared to the low-dose group, despite a similarly high HCT at both dose levels (Andrews, Pyrah, et al. 2013). These risk factors were observed in the AMG 114 high-dose group 48 hr after a single dose of drug and increased at subsequent time points in a 9-day study in the absence of a maximum high HCT. The lack of histologic evidence of thrombotic toxicities in rats in the AMG 114 high-dose group from the same study suggested that the sustained high erythropoietic rate and high HCT achieved by repeated dosing in the 1-month study was a requisite cofactor for thrombosis. However, the role of high HCT as a predisposing factor was inferred since thrombotic toxicities were only identified in animals with a high HCT. Several rodent models of polycythemia or anemia utilized to investigate AMG 114–mediated biologic effects dissociated from high HCT failed due to multiple confounding factors that impacted data interpretation and/or for technical reasons. Therefore, AMG 114 administration could not be dissociated from erythropoiesis in our investigative studies.
To further define the pathway to thrombosis in the AMG 114 high-dose group, we applied a discovery-based approach to analysis of data sets collected from rats administered AMG 114 at a high (500 µg/kg) or low (1.0 µg/kg) dose level at multiple time points before and after a high HCT was achieved. The measured end points that accounted for differences in the AMG 114 high-dose, compared to AMG 114 low-dose group and vehicle controls, were then evaluated for their mechanistic links to thrombosis independent of, or associated with, study end points that measured increased erythropoiesis. An additional analysis was conducted to correlate biomarker change in AMG 114–dosed rats with thrombosis, to those without, in the 1-month study.
AMG 114 high- and low-dose groups were differentiated after a single dose of drug by PCA. At early time points (24 and 48 hr), erythroid-related end points associated with increased erythropoiesis contributed significantly to separation of the AMG 114 high, from the AMG 114 low-dose group and vehicle controls, since their exclusion in parallel analyses diminished the dose level separation. However, individual AMG 114 high-dose animals were distinguishable from the AMG 114 low-dose rats in these same visualizations, suggesting nonerythroid-related end points were significantly modulated in those rats. At later time points, the AMG 114 high-dose group was discriminated from the AMG 114 low-dose group and vehicle controls in the presence or absence of erythroid end points. Dose level separation was expected based on an increased magnitude of erythropoiesis in the AMG 114 high-dose group that we previously described (Andrews, Pyrah, et al. 2013). However, the persistent separation in analyses that excluded erythroid-related end points indicated a fundamental difference existed between AMG 114 dose levels apart from increased erythropoiesis. Excluding the erythroid-related end points caused the AMG 114 low-dose group to generally overlap the vehicle control groups suggesting that, apart from increased erythropoiesis, the AMG 114 low-dose group was similar to vehicle controls throughout 1 month of repeated dosing.
In order to assess the biologic relevance of modulated study end points to thrombosis in AMG 114–dosed rats, a panel of circulating hemostatic biomarkers (ATIII, TAT, vWF, aFVII, and PAI-1) was assayed to recognize correlative imbalances in the hemostatic environment that developed before or after a high HCT was achieved. PT and APTT were assayed only in the 9-day study, since overcitrated plasma obtained from severely polycythemic animals in the 1-month study artificially prolongs clotting times (Kurata et al. 1998). Increased circulating vWF was identified in the AMG 114 high-dose group only, present 48 hr after a single dose of drug and at each subsequent time point through day 9. vWF is a well-described prothrombotic mediator of endothelial cell and PLTs in clot formation (van Galen et al. 2012) and plays a critical role in formation of microvascular thrombosis in animal models of vascular injury (Ogunshola et al. 2006; Patel et al. 2008; Denis et al. 1998). Increased circulating vWF from activated endothelial cells and/or PLTs (Rondaij et al. 2006) has been documented in diverse patient populations with thrombotic complications (Tousoulis et al. 2007; Adachi et al. 2006; Mina, Favaloro, and Koutts 2007) and correlated with clinical deterioration of acute stroke patients (Barber et al. 2004). While increased expression of vWF has been detected in vitro on endothelial cells and PLTs incubated with rHu-EPO (Fuste et al. 2002), increased circulating vWF was not detected in chronic renal failure patients (Pawlak, Pawlak, and Mysliwiec 2007; Christensson, Danielson, and Lethagen 2001) or healthy human volunteers (Heinisch et al. 2012) administered chronic or an acute dose of rHu-EPO, respectively. This suggests the early increased vWF in AMG 114 high-dose rats was most likely indirectly related to AMG 114 administration and not due to direct activation of endothelial cells and/or PLTs. Additional biomarkers of imbalanced hemostasis identified in the 9-day study included decreased ATIII (procoagulant; day 5 and day 9) in both AMG 114–dose groups and prolonged PT (hypocoagulant; day 9) in the AMG 114 high-dose group only. The accompanying PT prolongation in the AMG 114 high-dose group was minor (<3 sec) and likely of no clinical consequence, but in conjunction with decreased ATIII and increased vWF is further evidence of activated coagulation. Despite these findings, the AMG 114 high-dose animals had no thrombotic toxicities identified in the 9-day study. Therefore, the panel of hemostatic end points served to characterize imbalanced hemostasis in the AMG 114 high-dose group, although none performed consistently or with sufficient sensitivity to predict individual animals at increased risk for thrombosis after 1 month of repeat dosing with AMG 114.
The modulated chemokines, cytokines, and growth factors (hereafter termed cytokines) measured in blood from the AMG 114–dosed rats are promiscuous and pleiotropic ligands with roles in a variety of physiologic and pathologic processes. One unifying function of increased cytokines associated with AMG 114 administration is the mobilization, trafficking, stimulation, and proliferation of hematopoietic and mesenchymal stem cells required for effective intramedullary and extramedullary hematopoiesis (EMH; Janowska-Wieczorek et al. 2001). Literature regarding specific contributions of these cytokines in blood during stress erythropoiesis are much less numerous than those examining paracrine and autocrine functions in the erythroid niche (Chasis and Mohandas 2008; Johns and Christopher 2012). Several members of macrophage-related chemokine family members (MCP-1, MCP-3, MIP-2, MIP1b, and RANTES) were increased in the AMG 114 high-, but not the low-dose group at most time points assayed in the 9-day study. These chemokines have redundant roles in recruitment/migration of monocytes from blood to the vessel wall (Deshmane et al. 2009; Uguccioni et al. 1995) facilitating establishment of macrophages in erythropoietic islands required for effective stress erythropoiesis (Perry, Harandi, and Paulson 2007; Chasis and Mohandas 2008; Sadahira et al. 2000). An additional role reported for MIP-2 (Pelus and Fukuda 2006), as well as VCAM-1, and SCF, is the mobilization and trafficking of progenitor cells between bone marrow and blood (Dosquet et al. 2001; Levesque et al. 2001; Duits et al. 2002). This mechanism is activated during stress erythropoiesis in rodents to facilitate increased erythropoiesis in the spleen (Hara and Ogawa 1976). Expansion of hematopoietic stem cells and erythroid progenitors has also been reported for SCF, OSM, TIMP-1, VEGF, and RANTES in vitro (Lambert et al. 2003; Muta et al. 1995; Gerber and Ferrara 2003; Ergen, Boles, and Goodell 2012; Broudy 1997; Tanaka et al. 2003). IL-11 was increased in the AMG 114 high-dose group animals at day 9 and reported to stimulate multiple phases of erythropoiesis and thrombopoiesis in vitro (Nandurkar, Robb, and Begley 1998) and correlated with increased PLTs in the AMG 114 high-dose group. Most of the cytokines increased in the AMG 114 high-dose group correlated strongly with one another (data not shown) and have identified synergistic activities stimulating hematopoiesis in vivo. SCF, erythropoietin (EPO), and OSM combined were superior to the influence of a single cytokine in promoting stress-induced erythropoiesis in mice (Menon et al. 2006). Some cytokines increased in the AMG 114 high-dosed rats, most notably MCP-1, have inhibitory effects on hematopoiesis in vitro (Cashman et al. 1998). However, in vivo administration of MCP-1 (or family member MIP-1α) to animal models or patient populations with CML did not provide consistent evidence of an antiproliferative effect in the bone marrow (Cashman et al. 1999). Cytokine concentrations were presumably balanced in the erythroid niche to maximize increased erythropoiesis in the AMG 114 high-dose group. The majority of these cytokines were not detected in blood in the AMG 114 low-dose group despite increased erythropoiesis sufficient to achieve a similar maximum HCT as the AMG 114 high-dose group. This may be due to sensitivity limitations of our current assays. The dose-dependent behavior of increased cytokines in AMG 114–dosed rats may generally reflect the increased magnitude of erythropoiesis in the AMG 114 high-, compared to the low-dose group.
These same cytokines have well-studied proinflammatory and profibrotic roles in acute and chronic inflammation (Deshmane et al. 2009; Tarrant 2010). The immune-modulating effects of EPO have been evaluated in a wide variety of animal models. Results from these studies are conflicting and appear dependent on variables in experimental conditions such as the inflammatory process under investigation (sepsis, infectious, ischemia–reperfusion), EPO dose level, and timing of EPO delivery. Anti-inflammatory (Cuzzocrea et al. 2006; Aoshiba et al. 2009), proinflammatory (Wu et al. 2010), and a lack of immune-modulating effects (Solling et al. 2011) have been reported. The increased neutrophils (AMG 114 high- and low-dose groups) and fibrinogen (high-dose group only) at 6 and/or 24 hr in the AMG 114 high- and low-dose groups, respectively, are features of an acute inflammatory response. Absence of detectable blood concentrations of other cytokines classically associated with an acute phase response (i.e., TNF, IL-1, IL-6) in these same animals may be a consequence of missed peak blood concentrations that generally occur within 90 min after an experimental inflammatory stimulus or from lack of sensitivity in current assays used in our investigative studies (Tarrant 2010). This acute inflammatory response in AMG 114–dosed animals most likely originates from activation of the innate immune response secondary to IV injection of a foreign protein into healthy rats and not suggestive of a direct immune-modulating effect of AMG 114 administration (Nairz et al. 2012). The dose–response relationship to increased neutrophils in AMG 114–dosed rats is likely a result of higher concentrations of foreign protein delivered to the high-dose group. The neutrophils are a logical source for the rapidly increased serum MIPs/MCPs in AMG 114 high-dose group rats (Kumar and Sharma 2010) that may have downstream effects activating other effector cells that in turn elaborate additional cytokines that were identified at later time points in the study. Formation of antidrug antibodies (ADAs) is not likely a significant contributor to the enriched cytokine profile since ADA formation in IV dosed rats reviewed in historical AMG 114 preclinical toxicology studies was negligible (data not shown; Amgen internal files). Therefore, the rapid increased neutrophils and serum cytokines observed in AMG 114–dosed rats may in part originate from a mild inflammatory response to intravenous injection of a foreign protein and represent an additional prothrombotic risk factor to the AMG 114 high-dose group animals.
Proinflammatory cytokines have been linked to thrombosis as inflammation and coagulation have proved intimately intertwined (Delvaeye and Conway 2009; Esmon 2009). The pleiotropic cytokines increased in AMG 114–dosed rats have also been demonstrated integral to formation and resolution of a thrombus during hemostasis (Henke and Wakefield 2009; Saha et al. 2011; Myers and Wakefield 2005; Charo and Taubman 2004), and directly associated with pathologic arterial and venous thrombosis in a variety of disease states (Cai and Harrison 2000; Fox and Kahn 2005). The procoagulant features of increased inflammatory cytokines lie in their ability to regulate monocyte–endothelial cell interactions; both key effector cells in inflammation-induced thrombosis. The monocyte–endothelial cell axis could be activated, independent of an increased HCT, by MCPs/MIPs released from activated neutrophils from a single high dose of a foreign protein (AMG 114). These cytokines favor an evolving prothrombotic environment by recruiting monocytes to the vasculature where they subsequently become activated and attach to endothelial cells that stimulates the elaboration of endothelial derived cytokines, chemoattractants, and adhesion molecules that activate and recruit additional monocytes, endothelial cells, T cells, eosinophils, and/or NK cells (Lei and Takahama 2012; Goebeler et al. 2001; Dorner et al. 2002; Schall et al. 1990; Tokuhira et al. 2000; Damas et al. 2003; Jinquan et al. 2000; Bless et al. 2000). This process correlates to the increased white blood cells and cytokines including lymphotactin, RANTES, IP-10, and VCAM-1 observed at later time points in the study. RANTES and/or VCAM-1 have also been demonstrated to increase monocyte adherence to endothelial cells in animal models and patients (Baggiolini and Dahinden 1994; Tokuhira et al. 2000). Additional prothrombotic features of the elaborated cytokines in AMG 114 high-dose group animals include induction of tissue factor (TF) expression on the monocyte and endothelial cells surface, which activates the coagulation cascade resulting in thrombin generation and down regulation of anticoagulant proteins thrombomodulin and protein C pathway (ten Cate et al. 1997; Napoleone, Di Santo, and Lorenzet 1997; Charo and Taubman 2004; van der Poll et al. 1990). The pleiotropic cytokines identified in AMG 114 high-dose rats represent an additional prothrombotic risk factor independent of increased HCT that contribute to a prothrombotic circulatory environment prior to increased HCT (Napoleone, Di Santo, and Lorenzet 1997; Charo and Taubman 2004; van der Poll et al. 1990).
The results from our retrospective analysis of study data from ESA preclinical toxicology studies and investigative studies in rats administered a hyperglycosylated analog of rHu-EPO, AMG 114, have identified several prothrombotic risk factors in the highest ESA-dose groups that likely contributed to the increased incidence of thrombotic toxicities. The serum cytokines identified either uniquely, or in increased magnitude in the AMG 114 high-, compared to the low-dose groups in these investigative studies, represent additional prothrombotic risk factors. A potential pathway to ESA-related thrombosis that depicts the constellation of prothrombotic risk factors identified in preclinical species administered an ESA at a high-dose level and dose frequency is depicted in Figure 5. We did not identify individual animal variables that selected individual rats for thrombotic toxicities above those identified in all AMG 114 high-dose group animals. This task may prove as difficult as predicting thrombosis in humans (De Groot and Derksen, 2005). In conclusion, multiple prothrombotic risk factors were identified with increased magnitude, or uniquely in, the AMG 114 high-dose group, prior to an increased HCT that likely contributed to an increased incidence of ESA-related thrombotic toxicities in the highest dose groups. While a high HCT appears to be a requisite cofactor in development of ESA-related thrombotic toxicities, our data indicate it is not the sole causality of thrombosis in ESA-dosed preclinical species.
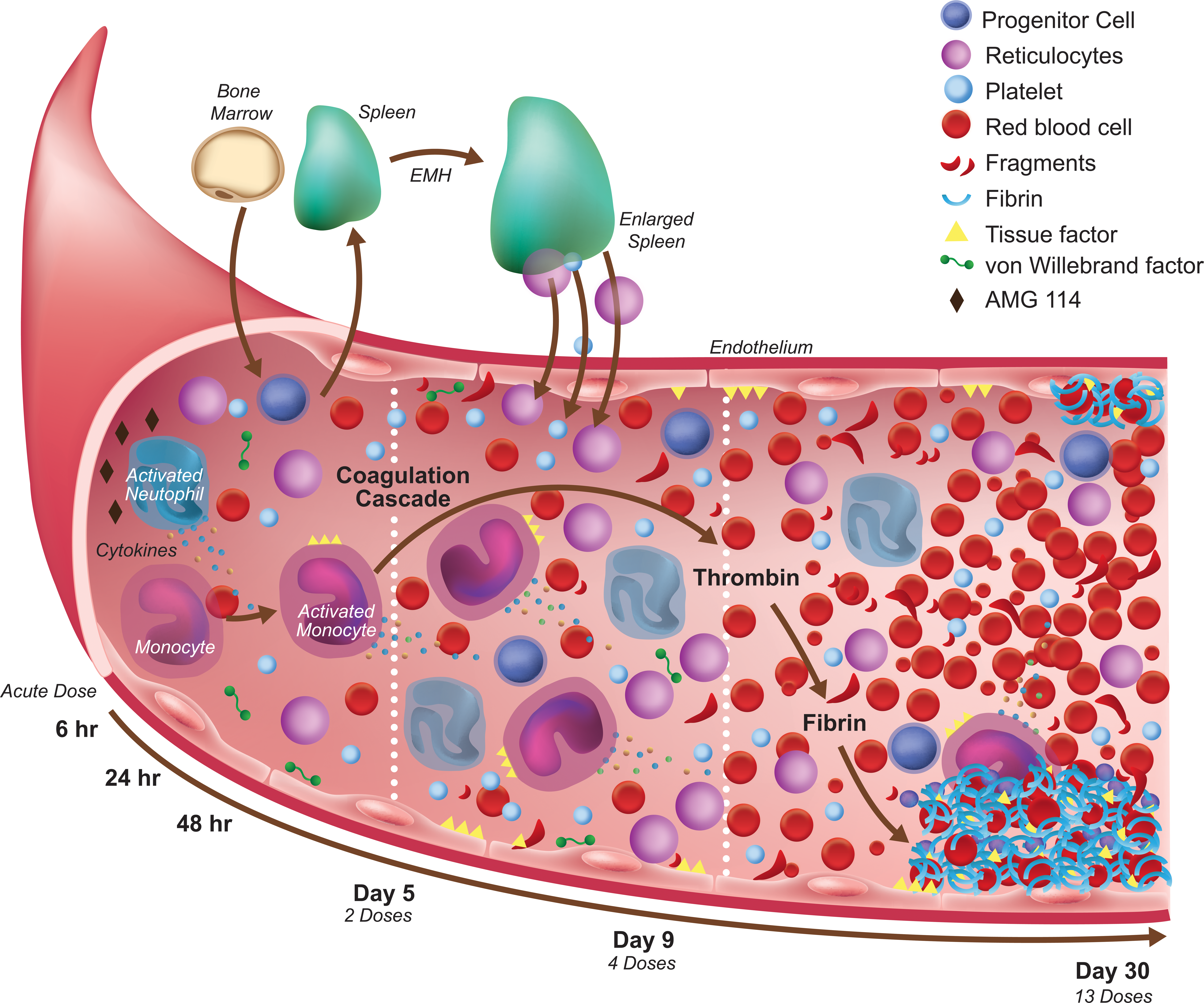
A subset of prothrombotic risk factors identified in Sprague-Dawley rats administered a hyperglycosylated analog of recombinant human erythropoietin, AMG 114, at a high-dose level (500 µg/kg) and increased dose frequency (3 times weekly) in a 1-month repeat dose toxicology study are depicted in this diagram. After a single dose of AMG 114, IV, increased cytokines/chemokines (i.e., MCP-1 and MCP-3), potentially derived in part from neutrophils activated in response to injection of a foreign protein promote increased erythropoiesis and an inflammatory intravascular environment. Increased numbers of neutrophils and their elaborated cytokines/chemokines activate effector cells in the vasculature such as monocytes and/or endothelial cells that elaborate additional cytokines that recruit/activate additional leukocytes, induce surface expression of tissue factor (TF), and stimulate release of von Willebrands Factor (vWF) from Weibel-Palade bodies of endothelial cells (and possibly platelets). The early hematopoietic response to AMG 114 includes redistribution of hematopoietic precursors from the bone marrow to the spleen and increased numbers of immature (high density [HD]) reticulocytes which introduce a generally larger, less deformable and more adhesive red cell population into circulation. With repeated dosing, sequelae from an increased magnitude of increased erythropoiesis represent additional prothrombotic risk factors that include higher numbers of increasingly immature reticulocytes and nucleated RBCs in circulation and severe functional iron deficiency that leads to increased intravascular destruction of iron deficient reticulocyte/RBCs (RBC fragments) and increased platelets that increase platelet-activated endothelial cell interactions. Increasing concentrations of cytokines/chemokines from increased leukocytes upregulate monocyte–endothelial cell interactions leading to increased TF expression on effector cells and activation of the coagulation cascade. The resulting increased thrombin generation decreases circulating antithrombin III (ATIII) leading to excess fibrin available for deposition in a developing subclinical thrombus. Increased viscosity of blood from polycythemia contributes to a prothrombotic circulatory environment at later time points in the 1-month study.
Footnotes
Acknowledgments
The authors offer special thanks to Amgen colleagues Graham Molineux (Oncology Research) for his invaluable scientific contributions and Christine Karbowski (Discovery Toxicology) for her expertise and guidance regarding appropriate execution of the pathway analysis tools. We additionally thank James Catalfamo, the director of the Comparative Coagulation Laboratory at Cornell University, for his scientific and technical expertise regarding nonstandard hemostatic biomarkers in preclinical species, as well as the toxicologists, animal research scientists, and pathologists at Covance Laboratories involved in the investigative study for their collaboration and expertise.
All authors are employees of Amgen, Inc.
Amgen, Inc. sponsored the retrospective analysis and preclinical investigative studies conducted with AMG 114 reported in this manuscript. Amgen, Inc. manufactures and markets epoetin alfa and darbepoietin alfa and manufactured AMG 114 for investigative purposes only.
