Abstract
An imbalance of matrix metalloproteinases (MMPs) and their inhibitors is thought to play a major role in the pathophysiology of joint diseases. The aim of this study is to provide additional insights into the relevance of MMP levels in arthroplasty patients in relation to inflammation and thrombosis. Deidentified plasma samples from 100 patients undergoing total hip arthroplasty or total knee arthroplasty were collected preoperatively, on postoperative day 1, and on postoperative day 3. Tissue inhibitor of MMP 4, tumor necrosis factor α (TNF-α), pro-MMP1, MMP3, MMP9, MMP13, and
Introduction
Due to the aging populations, the number of total hip arthroplasty (THA) and total knee arthroplasty (TKA) procedures performed is rapidly growing.
1,2
Although THA and TKA procedures have high success rates, complications are still present
1
and can include deep vein thrombosis (DVT), pulmonary embolism (PE),
3
and inflammation.
4
Many times, it is difficult or impossible to predict DVT/PE complications, as 50% of patients who have a postoperative DVT do not have an identifiable clinical risk factor.
2,3
Matrix metalloproteinases (MMPs) are zinc-dependent endopeptidases and are the main proteases that degrade collagen. 6 –8 Matrix metalloproteinases are transcribed in pro-MMP form and are inactive until they are cleaved to form active MMPs. 6,9 This process can be mediated through other MMPs, 6 notably MMP3 10 and MMP9. 11 Tissue inhibitors of MMPs (TIMPs) bind to the active site on MMPs rendering them inactive. 6,10 An imbalance of MMPs with their respective TIMPs is thought to play an important role in the progression of arthritis. 6,9 –16
Matrix metalloproteinase 1, MMP3, MMP9, and MMP13 are of particular interest in the pathophysiology of arthritis. 6 –17 Matrix metalloproteinase 1 and MMP13 are collagenases that degrade triple helical collagen. 7,13 Type II collagen is the most common type of collagen found in articular joints and is degraded by MMP13, with more specificity than the other collagenase MMPs. 7,8,13,15 –17 In addition, MMP13 also plays a large role in activating other MMPs, notably MMP1, MMP9, and MMP13. 7 Matrix metalloproteinase 3 is a stromelyesin that degrades extracellular matrix tissue and can activate other MMPs. 8,10,13,14 Matrix metalloproteinase 9 is a gelatinase but also has activity against collagen types I, III, IV, and V. 10 Expression of MMP is known to be upregulated by several proinflammatory factors, notably IL-1β and TNF-α. 1,7 –9,11,13,17 The upregulation of MMPs contributes significantly to the local synovial dysregulation of proteases. The analysis of these enzymes in the synovial fluid collected from patients undergoing joint arthroplasty may provide useful information on the progression of the joint diseases. In addition to MMPs, the role of the renin–angiotensin system and angiotensins in mediating inflammation in local synovium has been reported. 18 However, a study comparing the MMPs to the renin–angiotensin system and its components is not available.
The aim of this study was to evaluate the effect of arthroplasty on the expression of MMPs and the markers of inflammatory and thrombotic expression. Additionally, we sought to determine whether there was a correlation between MMP expression and markers of inflammatory or thrombotic activation in these patients.
Methods
Deidentified citrated plasma samples from 100 patients undergoing THA or TKA procedures were taken for routine Prothrombin Time/ International Normalized Ratio (PT/INR) testing prior to the operation on the first postoperative day and on the third postoperative day if the patient was not discharged. After the necessary clinical testing was completed, these deidentified plasma samples were collected from the Core Lab. When possible, blood samples were also collected from patients on postoperative day 3 (n = 10). The plasma samples were aliquoted into separate tubes to reduce the number of freeze-thaw cycles and stored at −80°C. Citrated plasma samples from 15 male and 15 female participants (ages 18-35, healthy, and nonsmokers) were obtained from George King Biomedical, Inc (Overland Park, Kansas) to serve as controls.
Commercially available sandwich enzyme-linked immunosorbent assay (ELISA) kits were used to measure the concentrations of the stated biomarkers on the collected plasma samples. Human pro-MMP1, human total MMP3, MMP9, TNF-α, and TIMP4 were measured using ELISA obtained from R&D Systems (Minneapolis, Minnesota). An ELISA kit manufactured by Abcam (Cambridge, England) was used for the analysis of MMP13.
Statistical Analysis
GraphPad Prism program version 6 (GraphPad Software, Inc La Jolla, California) was used for statistical analysis. Nonparametric tests were used for all analysis, and significance was determined by P < .05 (two-sided). Analysis of variance tests with post hoc Mann-Whitney U tests were run to identify significant differences between control and experimental groups. Wilcoxon matched paired t tests were then run to determine whether there were significant changes in biomarkers on postoperative day 1 and postoperative day 3. Spearman correlation tests were used to determine correlations between 2 variables. Median and interquartile ranges were also used to stratify data.
Results
Frequency distributions indicated the data collected had a nonparametric distribution. Preoperative TNF-α concentrations in arthroplasty patients were found to be significantly elevated compared to healthy controls (median [interquartile range, IQR]: 16.81 [0.91-44.09] vs 0.29 [0.12-0.84] pg/mL, P < .0001; Mann-Whitney). Postoperative day 1 and postoperative day 3 levels did not change compared to the preoperative levels (Wilcoxon matched paired t test) but were still elevated compared to normal (P < .0001; Mann–Whitney U test; Figure 1).
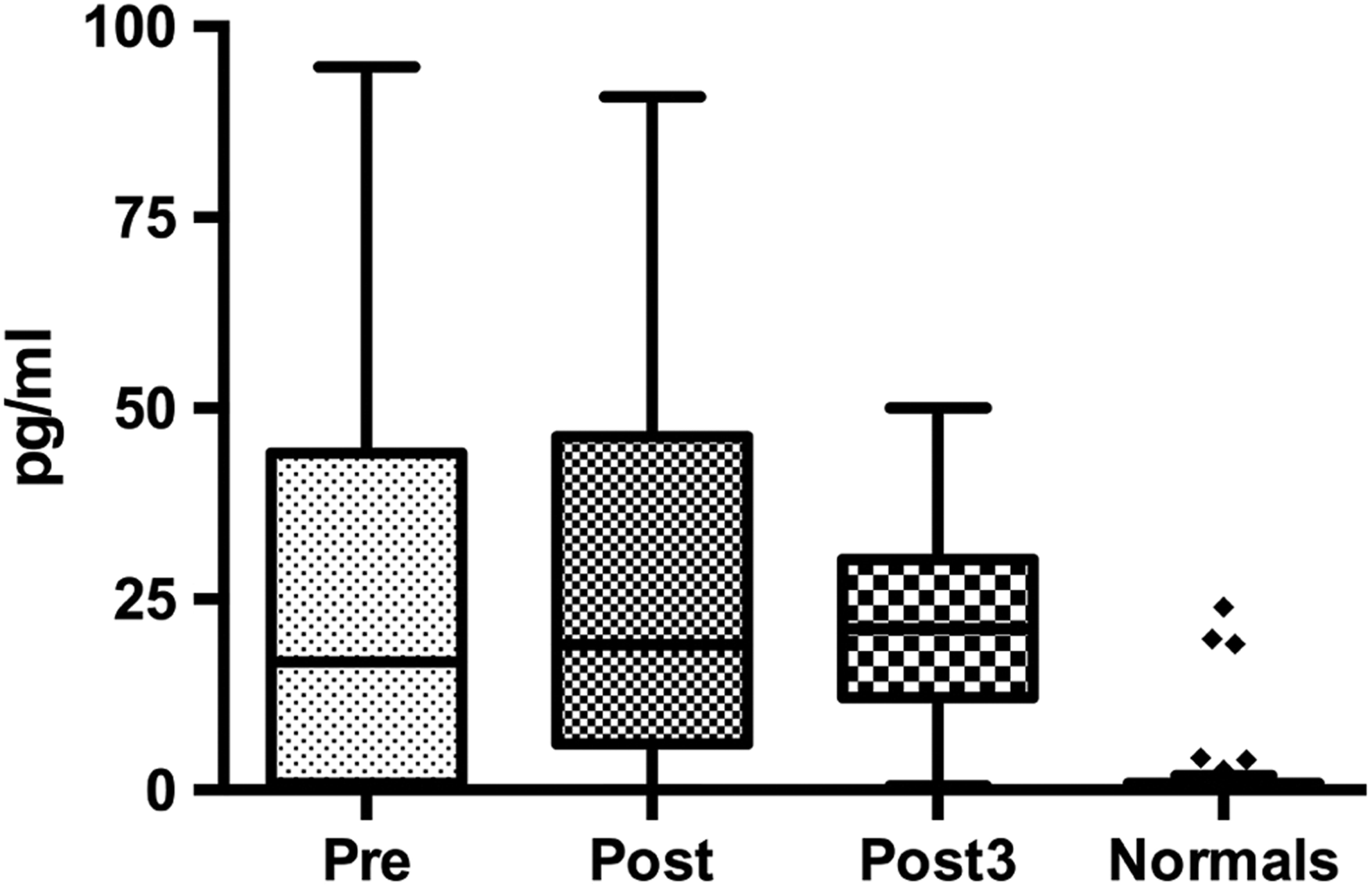
Tukey boxplot for TNF-α levels in pg/mL in pre-, post-, and postoperative day 3 compared to healthy controls. Pre-, post-, and post3 TNF-α levels were significantly elevated compared to the normals. TNF-α indicates tumor necrosis factor α.
Preoperative
Tukey boxplot for
Preoperative MMP1 concentrations in arthroplasty patients were found to be significantly elevated compared to healthy controls (median [IQR]: 0.99 [0.64-1.69] vs 0.34 [0.20-0.53] ng/mL, P < .0001; Mann-Whitney; Figure 3). Postoperative day 3 levels were significantly elevated over preoperative levels (median [IQR]: 1.62 [0.94-2.96], P < .0195; Wilcoxon matched paired t test), but levels of MMP1 on postoperative day 1 were not significantly different compared to preoperative or postoperative day 3 samples.
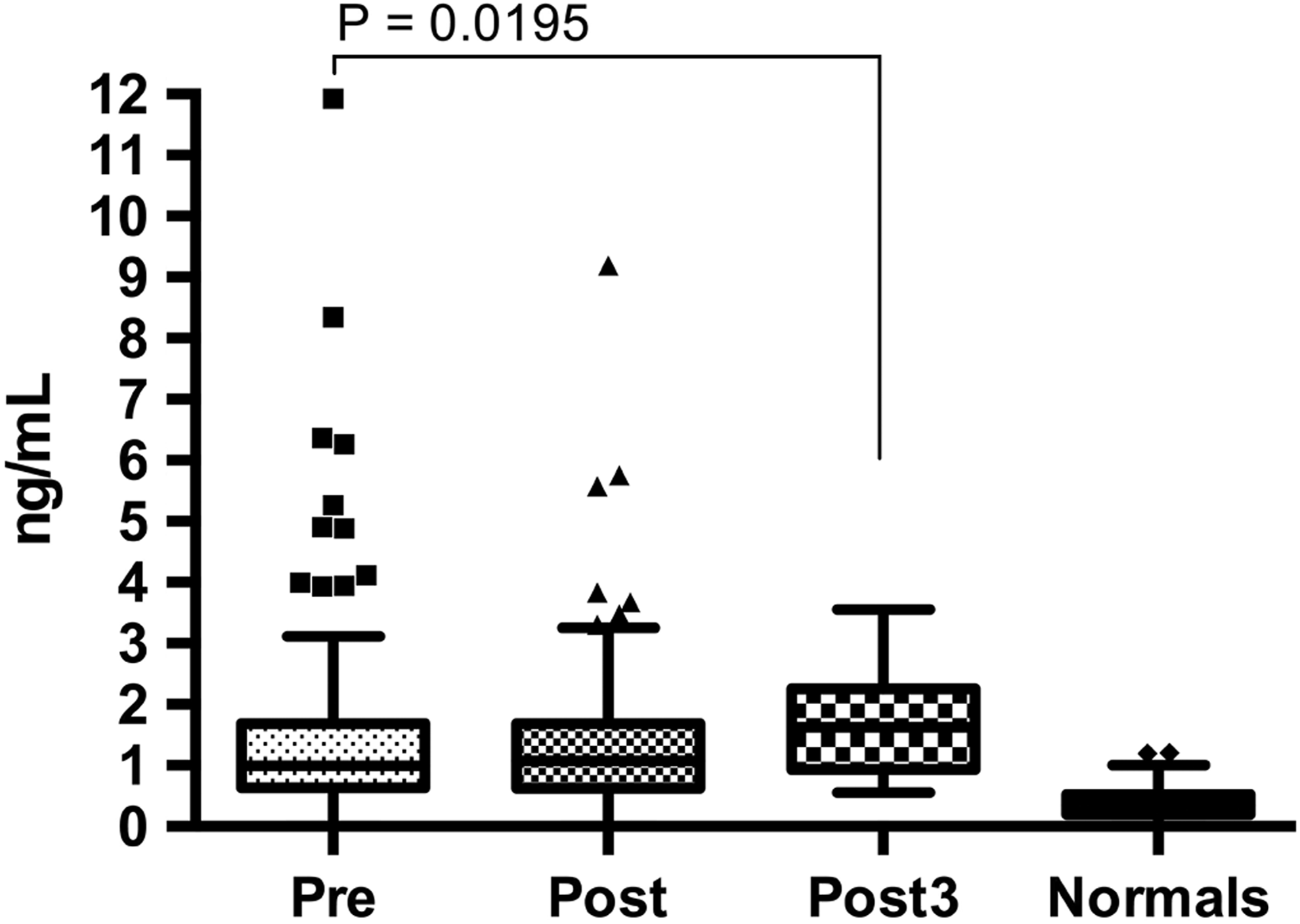
Tukey boxplot for MMP1 levels in ng/mL in pre-, post-, and postoperative day 3 compared to healthy controls. Pre-, post-, and post3 MMP1 levels were significantly elevated compared to the normals. MMP1 indicates matrix metalloproteinase 1.
Concentrations of MMP3 in preoperative, postoperative day 1, and postoperative day 3 samples were not found to be significantly different than in samples from normal individuals (median [IQR]—pre: 7.71 [4.86-13.07]; post: 6.48 [3.60-10.83]; post3: 10.93 [8.45-12.86]; normals- 6.71 [4.31-9.43]; P > .9999; Mann–Whitney U test).
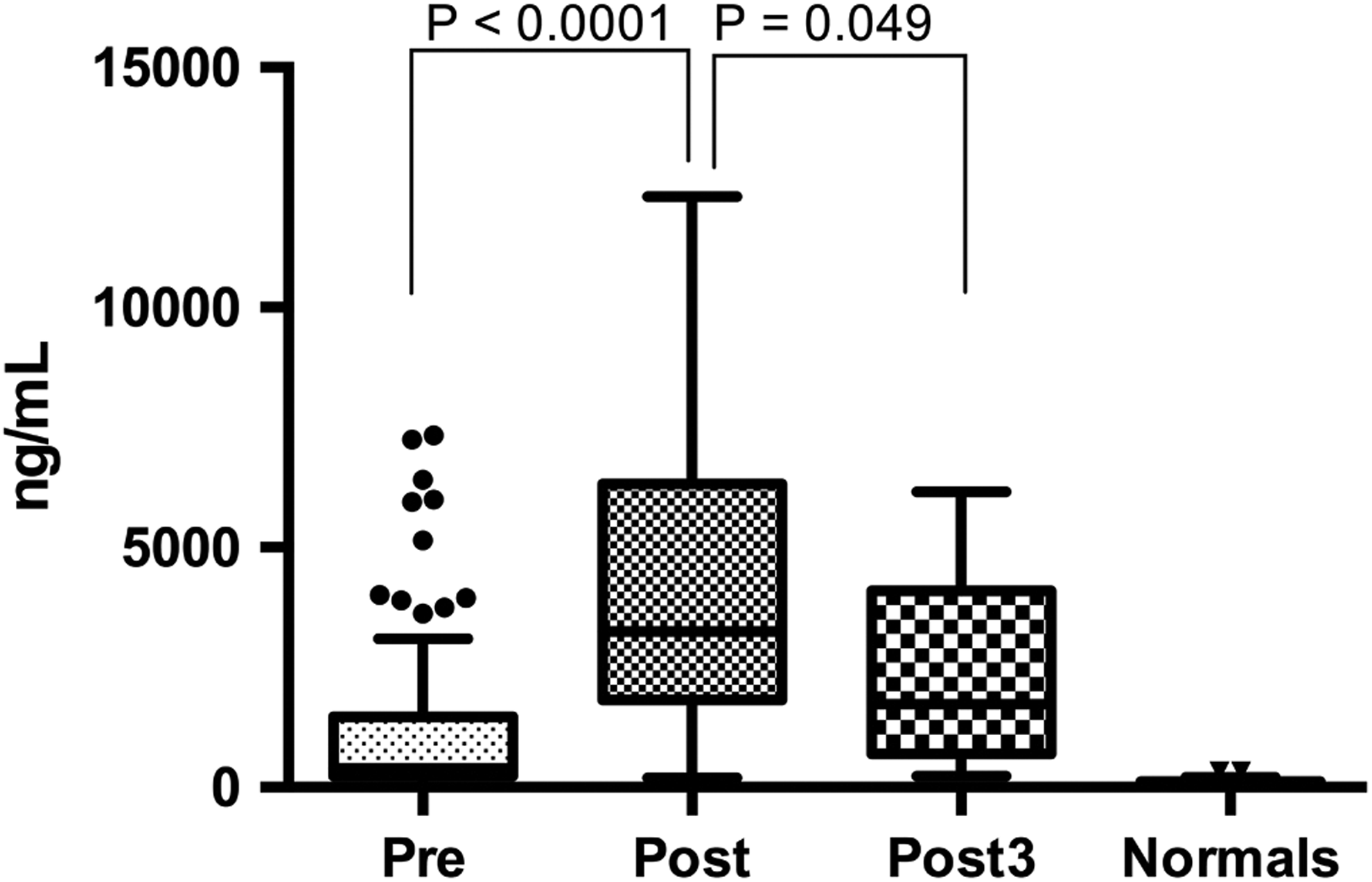
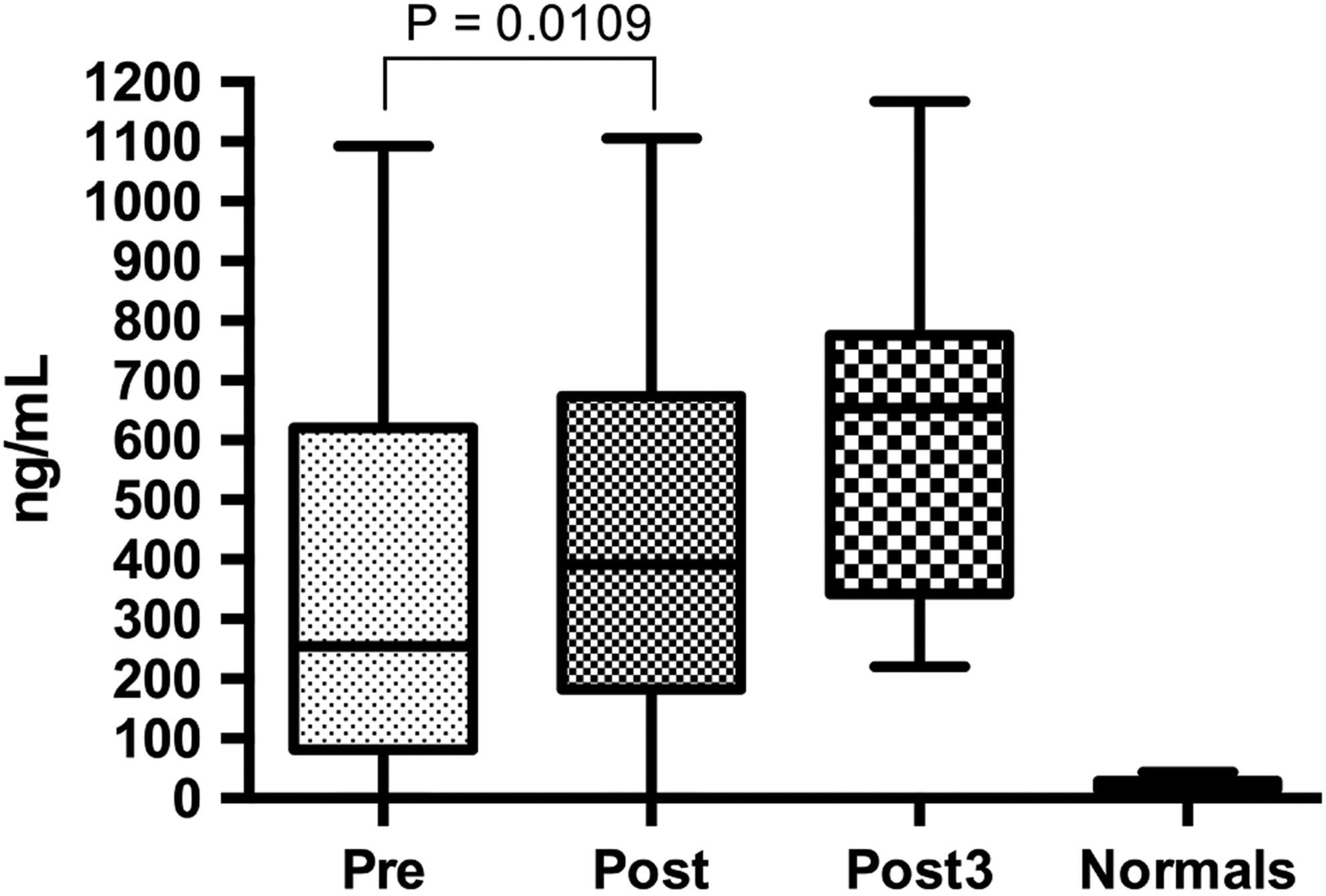
The concentrations of preoperative MMP9 in arthroplasty patients were found to be significantly elevated compared to healthy controls (median [IQR]: 254.9 [81.3-620.6] vs 21.5 [14.9-28.7] ng/mL, P < .0001; Mann-Whitney; Figure 4). Postoperative day 1 levels were significantly elevated compared to levels in preoperative samples (median [IQR]: 391.3 [182.7-673.7], P < .0109; Wilcoxon matched paired t test), but the median level of MMP9 on postoperative day 3 (median [IQR]: 653.6 [342.3-775.6]) was higher than levels measured in preoperative and postoperative day 1 samples; statistical significance could not be shown due to the small sample size.
Tukey boxplot for MMP9 levels in ng/mL in pre- post- and postoperative day 3 compared to healthy controls. Pre-, post-, and post3 MMP9 levels were significantly elevated compared to the normals. MMP9 indicates matrix metalloproteinase 9.
Preoperative MMP13 concentrations in arthroplasty patients were found to be significantly elevated compared to healthy controls (median [IQR]: 206.8 [138.0-200.3] vs 59.5 [13.0-82.4] ng/mL, P < .0001; Mann-Whitney). Postoperative day 1 and postoperative day 3 levels did not change compared to preoperative levels (Wilcoxon matched paired t test) but were still elevated compared to normal (P < .0001; Mann-Whitney). Data not shown.
Preoperative TIMP4 concentrations in arthroplasty patients were found to be significantly elevated compared to healthy controls (median [IQR]: 0.53 [0.45-0.83] vs 0.28 [0.25-0.31], P < .0001; Mann-Whitney; note: units in terms of optical density). Postoperative day 1 and postoperative day 3 levels did not change compared to preoperative levels (Wilcoxon matched paired t test) but were still elevated compared to normal (P < .0001; Mann-Whitney U test). Data not shown.
Spearman correlation analysis was also performed comparing MMPs with levels of
Summary of Spearman r values for preoperative samples.
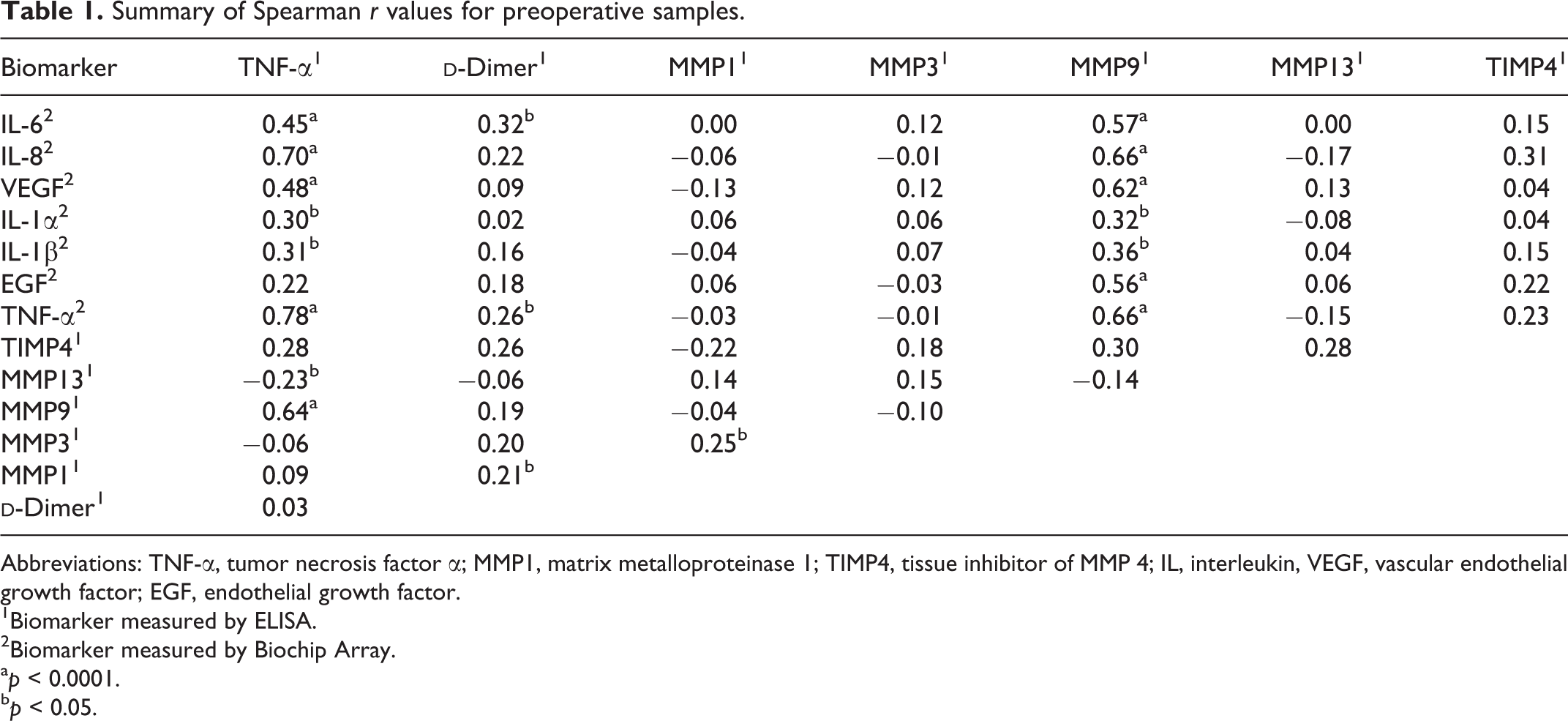
Abbreviations: TNF-α, tumor necrosis factor α; MMP1, matrix metalloproteinase 1; TIMP4, tissue inhibitor of MMP 4; IL, interleukin, VEGF, vascular endothelial growth factor; EGF, endothelial growth factor.
1Biomarker measured by ELISA.
2Biomarker measured by Biochip Array.
a p < 0.0001.
b p < 0.05.
In postoperative samples, TNF-α was found to have significant correlations with IL-8, VEGF, IL-1α, IL-1β, EGF, and MMP9.
Summary of Spearman r values for postoperative samples.
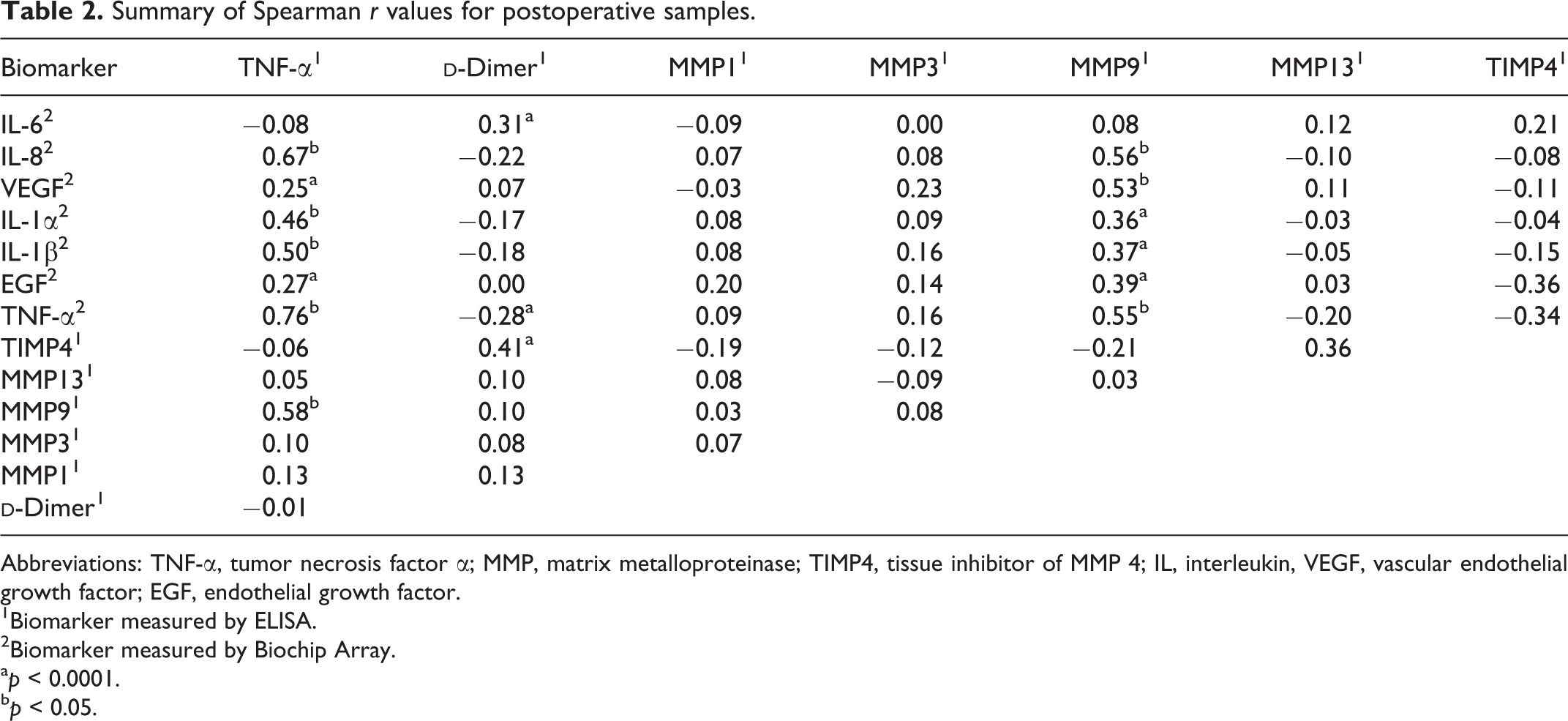
Abbreviations: TNF-α, tumor necrosis factor α; MMP, matrix metalloproteinase; TIMP4, tissue inhibitor of MMP 4; IL, interleukin, VEGF, vascular endothelial growth factor; EGF, endothelial growth factor.
1Biomarker measured by ELISA.
2Biomarker measured by Biochip Array.
a p < 0.0001.
b p < 0.05.
Discussion
High levels of TNF-α in preoperative, postoperative day 1, and postoperative day 3 samples compared to the control group indicate increased levels of inflammation in arthroplasty patients compared to healthy controls. The main indication for arthroplasty procedures is severe arthritis, notably osteoarthritis. Although osteoarthritis was traditionally thought of as a noninflammatory process, the discovery of MMP upregulation by several inflammatory cytokines has shown that osteoarthritis does have an inflammatory component to its pathogenesis. 19
Arthroplasty patients also showed increased coagulation activity preoperatively.
Matrix metalloproteinase 1 is a collagenase that degrades triple helical collagen. 7,13 It showed a significant elevation in preoperative, postoperative day 1, and postoperative day 3 arthroplasty samples, supporting the theory that it plays a role in the progression of arthritis. Furthermore, MMP1 levels were elevated on postoperative day 3 compared to preoperative samples. This could indicate stress on the body, such as surgery, may play a role in upregulation of MMP1. However, this upregulation may be delayed, as postoperative day 1 samples were not significantly higher than preoperative samples. Further studies trending MMP1 levels past postoperative day 3 may provide further insight into this interaction.
Total MMP3 did not show any significant change in concentration between the arthroplasty patients and the controls. Previous literature displayed elevated MMP3 concentrations in patients with arthritis. 8,10,13,14 One possible reason for this discrepancy may be due to the different methods of measurement. The assay used in this experiment measured total immunologic MMP3 and not MMP3 activity. Although total MMP3 in arthroplasty patients was not elevated compared to the controls, it is possible that MMP3 activity may be higher in this population.
Matrix metalloproteinase 9 showed the largest increase in concentration in arthroplasty patients compared to healthy controls. Like MMP1, MMP9 concentrations were higher in postoperative samples compared to preoperative samples. This indicates surgery may be related to MMP9 upregulation. The elevated MMP9 levels were not found to be statistically significant in postoperative day 3 samples, but this could be due to a low samples size (n = 10) in the postoperative day 3 group. Like MMP1, further studies, with more participants, are needed to define the trend in MMP9 levels past postoperative day 3.
Matrix metalloproteinase 13 also showed a significant increase in arthroplasty samples for pre, post, and post3 samples over the normal. Matrix metalloproteinase 13 is thought to play a pivotal role in arthritic progression, 7,8,13,15 –17 and this is supported by the results obtained in this study. Matrix metalloproteinase 13 primarily degrades collagen type II which is the most common type of collagen found in articular joints. 7,13
Tissue inhibitor of MMP 4 also showed elevation in the arthroplasty patients when compared to healthy controls. An imbalance in MMP–TIMP4 ratio is thought to play a major role in the progression of arthritis. 6,9 –16 However, without having data on MMP activity relative to TIMP4 activity, it is difficult to draw definitive conclusions on the MMP–TIMP interactions.
Positive correlations were found between MMP9 and biomarkers for inflammation, angiogenesis, and cellular growth/differentiation. It is well documented that various MMPs are upregulated by TNF-α and IL-1, and the results of our study confirm this correlation. Previous studies have also shown MMP9 upregulation by IL-6, IL-8, EGF, and VEGF in various cancer cell lines. 20 –23 Similar positive correlations found in patients undergoing arthroplasty procedures indicate MMP9 activation by IL-6, IL-8, EGF, and VEGF may also be present in the pathophysiology of arthritis. Our results indicate that surgery does not significantly affect these correlations, as they are present in both pre- and postoperative samples.
One limitation of this study is that the patients in the control group were not aged matched. It is difficult to find older patients with no comorbidities that would affect expression of the biomarkers studied; as a result, the control group is likely composed of younger individuals compared to the experimental group. In this study, MMPs and other biomarkers were measured using antigenic determination methods. Measuring MMPs in terms of their activity may yield more information regarding their role in inflammatory and thrombotic activation. Furthermore, most patients in this study were discharged on hospital day 2, leading to low numbers of patients who were studied on postoperative day 3. Future research evaluating these biomarkers beyond the third postoperative day, with larger numbers of patients, would give more insight into the postsurgical trends of the studied biomarkers.
The current study was carried out on plasma samples collected from arthroplasty patients. As reported by other investigators, the local synovial dysregulation of proteases may contribute to the pathogenesis of inflammation in these patients. Renin- and angiotensin-converting enzymes have been reported to be elevated in synovial fluid in patients with rheumatoid arthritis. Elevated levels of angiotensin II contribute to the upregulation of various inflammatory biomarkers such as IL-1β and TNF-α. 18 Since these markers are also found to be increased in association with MMPs, there may be a common link in the regulation of MMPs and renin–angiotensin system. It would have been of interest to measure the component of renin–angiotensin system in the plasma samples included in this study. Moreover, the inclusion of the local synovial fluid samples for such studies will also provide additional insight into the local pathogenesis of joint diseases in patients undergoing arthroplasty.
In conclusion, this study confirms that levels of MMPs, notably MMP1, MMP9, and MMP13, are elevated in arthroplasty patients. This is consistent with the existing literature and suggests they may play a pivotal role in the arthritic disease progression.
6,9
–16
Furthermore,
Footnotes
Acknowledgements
The authors gratefully acknowledge the technical support from the staff of the hemostasis and thrombosis laboratories for facilitating the logistics of the blood collection, storage, and analysis of the samples included in this study. This research was a part of Students Training in Approaches to Research (STAR) program for medical students. We are thankful to Dr Gail Hecht and Dean Brubaker for their support and encouragement during this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by internal research funds from the Department of Pathology.
