Abstract
We conducted a retrospective analysis of publicly available preclinical toxicology studies with erythropoiesis-stimulating agents (ESAs) to examine common adverse events in rats, Beagle dogs, and cynomolgus monkeys. Mortality and/or thrombotic events were reported sporadically in a subset of studies and attributed to the high hematocrit (HCT) achieved in the animals. However, similarly high HCT was achieved in both high-dose and low-dose groups, but there were no reported adverse events in the low-dose group suggesting HCT was not the sole contributing factor leading to toxicity. Our analysis indicated that increased dose, dose frequency, and dosing duration in addition to high HCT contributed to mortality and thrombosis. To further evaluate this relationship, the incidence of toxicities was compared in rats administered an experimental hyperglycosylated analog of recombinant human erythropoietin (AMG 114) at varying dosing schedules in 1-month toxicity studies. The incidence of mortality and thrombotic events increased in higher dose groups and when dosed more frequently, despite a similarly high HCT in all animals. The results from the investigative study and retrospective analysis demonstrate that ESA-related toxicities in preclinical species are associated with dose level, dose frequency, and dosing duration, and not solely dependent upon a high HCT.
Introduction
Erythropoiesis is under the central control of erythropoietin (EPO), a glycoprotein hormone that demonstrates a high degree of DNA sequence homology across mammalian species (Wen et al. 1993; Galson et al. 1993; Nagao et al. 1992). Human EPO is biologically active when administered to animals (Bugelski et al. 2008; Woo and Jusko 2007) and exogenous replacement of EPO has reversed EPO-deficient anemia in nephrectomized animal models (Kawamura et al. 1990; Oishi et al. 1995). In 1989, epoetin alfa (Epogen®, Amgen Inc., Thousand Oaks, CA) became the first recombinant human EPO (rHu-EPO) marketed for the treatment of anemia related to chronic kidney disease (CKD; Lin et al. 1985; Jacobs et al. 1985). Treatment with epoetin alfa effectively increased hematocrit (HCT)/hemoglobin (Hb) in CKD patients by promoting erythropoiesis within the patient’s own bone marrow, significantly decreasing or eliminating the need for red blood cell transfusions (Eschbach et al. 1987; Boran et al. 1993; Mohini 1989).
Following the approval of epoetin alfa, other structurally similar erythropoiesis-stimulating agents (ESAs) were developed that also increased erythropoiesis (Elliott et al. 2008). In some, drug half-life was extended through altered glycosylation or by the addition of a propylene glycol (PEG) to the amino acid backbone of rHu-EPO (Macdougall 2002; Elliott, Pham, and Macdougall 2004; Kiss et al. 2010; Tillmann et al. 2006). A synthetically derived PEGylated EPO-mimetic peptide that directly activates the EPO receptor is currently being developed (Fan et al. 2006).
A subset of preclinical toxicology studies with structurally different ESAs was identified that reported sporadic occurrences of mortality and/or thrombotic events in rats, Beagle dogs, and cynomolgus monkeys, after a high HCT/Hb had been achieved (Table 1). In the study reports, the high HCT resulting from repeated dosing with the ESA was cited as the underlying pathogenesis for the toxicities. These findings were not considered readily translatable to humans since treatment was intended to improve the clinical state of anemia and not increase HCT above normal reference values. The results from recent clinical trials that dosed to a higher Hb target raised concerns of an increased risk of death or adverse outcomes that included, among others, thromboembolic events and stroke (Besarab et al. 1998; Singh 2010; Phrommintikul et al. 2007; Regidor et al. 2011; Safety Alerts for Human Medicinal Products—United States Federal Drug and Safety Administration [USFDA], 2007; European Public Assessment Report [EPAR]—European Medicines Agency [EMA], 2008a, 2008b, 2008c, 2008d ). This article provides a retrospective analysis of toxicology data generated from rat and non-rodent species administered structurally different ESAs. We describe common toxicities that occurred in preclinical species administered ESAs, determine whether any toxicities were unique or prevalent to any particular form of an ESA, and evaluate the relationship of these toxicities to HCT. We also present findings from a rat toxicology study using a hyperglycosylated analog of rHu-EPO, in which an increased dose frequency at a high-dose level increased the incidence of thrombotic toxicities compared to animals dosed less frequently and in lower dose groups, despite a similarly high HCT across all groups. The results from this retrospective analysis and investigative study indicate that ESA toxicities in preclinical species cannot solely be explained by high HCT.
Erythropoiesis stimulating agents with preclinical toxicology studies that reported mortality and/or thrombotic events.
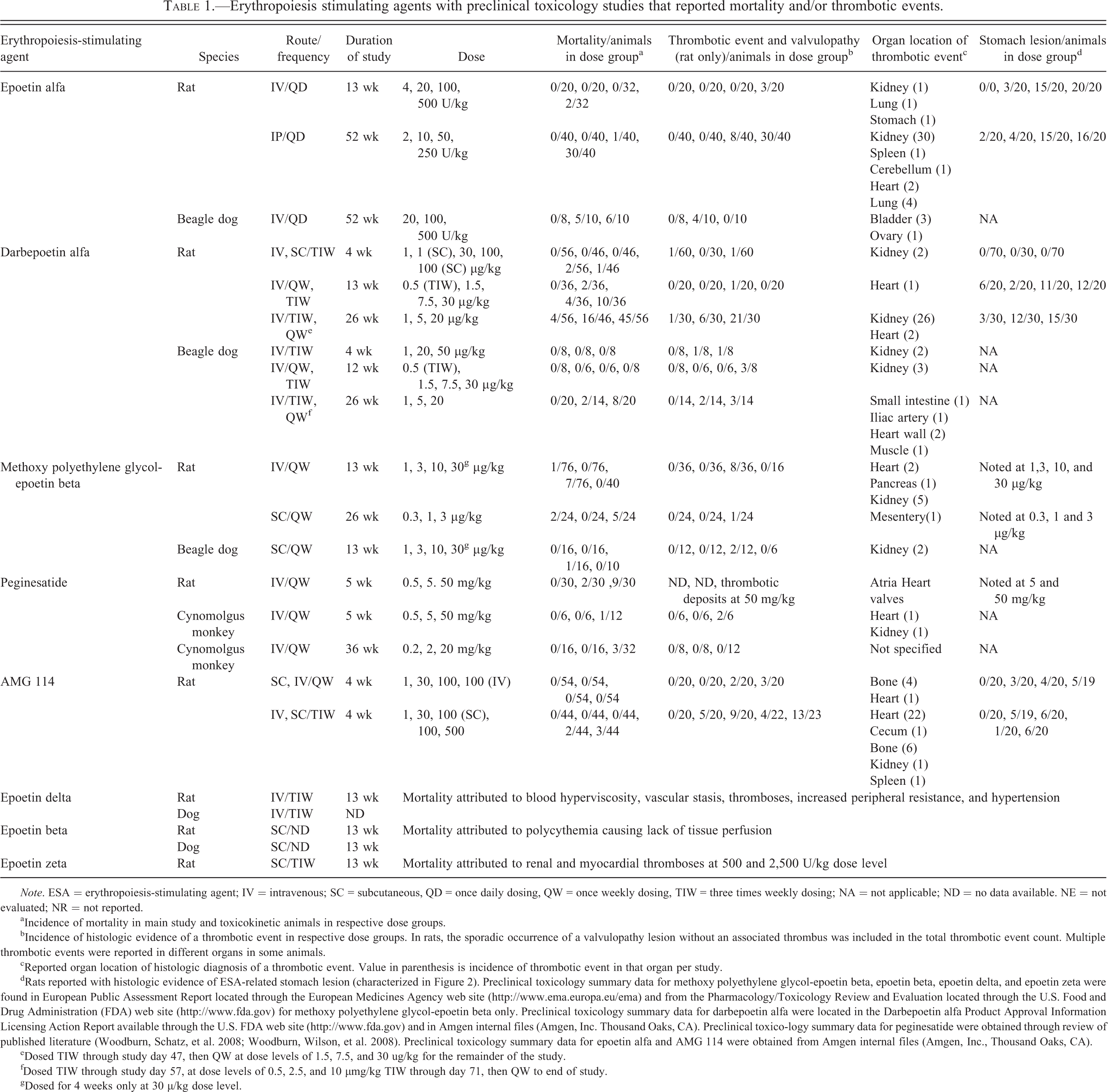
Note. ESA = erythropoiesis-stimulating agent; IV = intravenous; SC = subcutaneous, QD = once daily dosing, QW = once weekly dosing, TIW = three times weekly dosing; NA = not applicable; ND = no data available. NE = not evaluated; NR = not reported.
aIncidence of mortality in main study and toxicokinetic animals in respective dose groups.
bIncidence of histologic evidence of a thrombotic event in respective dose groups. In rats, the sporadic occurrence of a valvulopathy lesion without an associated thrombus was included in the total thrombotic event count. Multiple thrombotic events were reported in different organs in some animals.
cReported organ location of histologic diagnosis of a thrombotic event. Value in parenthesis is incidence of thrombotic event in that organ per study.
dRats reported with histologic evidence of ESA-related stomach lesion (characterized in Figure 2). Preclinical toxicology summary data for methoxy polyethylene glycol-epoetin beta, epoetin beta, epoetin delta, and epoetin zeta were found in European Public Assessment Report located through the European Medicines Agency web site (http://www.ema.europa.eu/ema) and from the Pharmacology/Toxicology Review and Evaluation located through the U.S. Food and Drug Administration (FDA) web site (http://www.fda.gov) for methoxy polyethylene glycol-epoetin beta only. Preclinical toxicology summary data for darbepoetin alfa were located in the Darbepoetin alfa Product Approval Information Licensing Action Report available through the U.S. FDA web site (http://www.fda.gov) and in Amgen internal files (Amgen, Inc. Thousand Oaks, CA). Preclinical toxico-logy summary data for peginesatide were obtained through review of published literature (Woodburn, Schatz, et al. 2008; Woodburn, Wilson, et al. 2008). Preclinical toxicology summary data for epoetin alfa and AMG 114 were obtained from Amgen internal files (Amgen, Inc., Thousand Oaks, CA).
eDosed TIW through study day 47, then QW at dose levels of 1.5, 7.5, and 30 ug/kg for the remainder of the study.
fDosed TIW through study day 57, at dose levels of 0.5, 2.5, and 10 µmg/kg TIW through day 71, then QW to end of study.
gDosed for 4 weeks only at 30 µ/kg dose level.
Material and Method
Search Strategy for Retrospective Analysis of ESA Toxicities in Preclinical Species
A list of ESAs with preclinical toxicology study summaries that reported mortality and/or thrombotic events is given in Table 1. The main sources for identifying this information were the PharmaPendium database from Elsevier (PharmaPendium; Elsevier, New York, 2008; https://www.pharmapendium.com/; accessed 2011) which contains FDA/Center for Drug Evaluation and Research (CDER)/Freedom Of Information (FOI) archived FDA drug approval review documentation and EMA EPAR documents, the EMA web site (http://www.ema.europa.eu/ema) and Pubmed (http://www.ncbi.nlm.nih.gov/pubm-ed), which were searched by drug class, drug names, treatment of anemia, and preclinical species. The corresponding finalized toxicology study reports with epoetin alfa (Amgen internal files) represent a subset of those submitted in support of epoetin alfa’s market approval. No summary approval documents exist; this regulatory practice took form many years after epoetin alfa’s approval. The finalized toxicology study reports with darbepoetin alfa (Amgen internal files) were included to supplement the toxicology study summaries found in the Darbepoetin alfa Product Approval Information—Licensing Action Report (USFDA 2001). In addition, the finalized repeat dose toxicology study reports with AMG 114, a hyperglycosylated analog of rHu-EPO, conducted in rats and Beagle dogs were included for review (Amgen internal files). The ESA regulatory documents, designated toxicology study reports, and publications were individually reviewed and a subset identified that reported mortality, the microscopic finding of a thrombus, and/or indirect evidence of a thrombotic event described by organ infarct and/or necrosis/degeneration in any main study or satellite toxicokinetic (TK) animals. When available, the dose group and incidence where mortality and/or an identified thrombotic event occurred, the organ involved, adverse clinical observations, and cause of early death were also recorded for the individual studies. The preclinical toxicology studies with ESAs listed in Table 1 with available detailed study data that included dose levels, dose frequency, duration of dosing, group mean HCT values, and individual animal pathology diagnoses were included in our analysis examining the relationship between HCT and mortality or ESA-related toxicities. Adequately detailed study data from toxicology studies with non-Amgen ESAs were limited to publications of toxicology studies with rats and cynomolgus monkeys with peginesatide (Woodburn, Schatz, et al. 2008; Woodburn, Wilson, et al. 2008). All clearly nontreatment-related deaths (including found dead control animals) were excluded from analysis. All treated animals found dead in these studies were included in the analysis with the exception of 2 rats in the 52-week epoetin alfa study with a microscopically confirmed infection and 2 rats in the 4-week darbepoetin alfa study found dead on day 9 before a high HCT was achieved. The exclusion of these animals did not impact the interpretation. Formation of antidrug antibodies was seen only in a minority of animals and was not considered a component of the toxicity (data not shown).
Investigative Study with AMG 114 in Rats
We conducted a 1-month investigative study in rats to explore the relationship between increased dose and dose frequency and the incidence of ESA-related toxicities in animals with a similar maximum HCT across all dose groups. The results of the investigative study were compared to those in a prior 1-month rat study where AMG 114 was dosed once weekly.
Test Article
AMG 114 was stored at −70°C. Erythropoietic activity was verified by cell-based in vitro assays and by increased HCT in Sprague-Dawley rats administered a single dose of AMG 114 in dose range finding studies (data not shown). The vehicle was 20 mM sodium phosphate, 140 mM sodium chloride, and 0.005% polysorbate 80, pH 6.2. Test and control article vials were stored at 2°C to 8°C and used within 1 week of opening. Dose preparations were prepared on each day of dosing, stored at 2°C to 8°C and allowed to equilibrate to approximate room temperature before dosing.
Test Animals
Male and female Crl:CD(SD) rats were cared for in accordance to the Guide for the Care and Use of Laboratory Animals, 8th ed. (National Research Council, 2011) at an Association for Assessment and Accreditation of Laboratory Animal Care International, internationally accredited facility and determined to be specific pathogen free for Kilham Rat Virus, Rat Coronavirus/Sialodacryoadenitis Virus, Rat Parvovirus, Rat Minute Virus, Helicobacter species, pinworms, and fur mites. All research protocols were approved by the Charles River Laboratories (Portage, Michigan) Institutional Animal Care and Use Committee. At initiation of treatment, the animals were 73 to 79 days old, and their body weights ranged from 306 to 376 g for males and 218 to 301 g for females. Animals had ad libitum access to pelleted feed (Certified Rodent Diet #2016C, Harlan Teklad) and purified municipal tap water via automatic watering system. Animals were maintained on a 12:12 hr light–dark cycle in rooms at 18° to 26°C and 30 to 70% relative humidity. Animals were provided with cage enrichment devices.
Experimental Design
Peer review of pathology sections and TK analysis were conducted at Amgen. Animals were randomized into groups based on body weight. Rats (toxicity, n = 10/sex/group; TK, n = 12/sex/group) were assigned to groups receiving 0 (vehicle), 1.0, 30, 100, and 500 IV or 100 SC µg/kg/dose AMG 114, 3 times weekly for 1 month (12 doses). The low dose was anticipated to be the minimal dose required to produce a maximal HCT. The high dose, 500 µg/kg, was 5-fold above the highest dose level used in the prior 1-month rat study (100 µg/kg) with AMG 114. The low dose and mid dose were to establish a dose response. The IV route of administration was selected to maximize exposure and minimize antidrug antibody response. The single SC dose group was chosen to compare effects directly to the same dose group in a prior 1-month toxicology study with AMG 114 dosed once weekly. The TK animals were included for blood sampling only and were evaluated histologically only if they were found dead or were euthanized at an unscheduled interval. Each animal was observed regularly for mortality, abnormalities, and signs of pain or distress, including 2 hr after each dose. Body weights were taken at least once during the predose phase, before dosing on study day 1 and weekly thereafter.
Pathology
Animals were fasted overnight prior to the scheduled necropsy. For hematology parameters, blood was collected by syringe from the jugular vein of all toxicity animals after 4 weeks of treatment on the day of scheduled necropsy (day 30) and transferred into tubes with potassium ethylenediaminetetraacetic acid as anticoagulant. Blood was analyzed by the Advia 2120 Hematology Analyzer (Siemens Healthcare Diagnostics, Deerfield, Illinois) and a peripheral blood smear was prepared for each animal. Toxicity animals were euthanized via exsanguination from the abdominal aorta/vena cava under deep isoflurane anesthesia and necropsied. A standard full set of tissues including heart valves was obtained from all toxicity animals, preserved in 10% neutral-buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E). All tissues from any control, toxicity, or TK animal that was found dead or euthanized at an unscheduled interval were also examined microscopically.
Histologic Characterization of ESA-related Toxicities in Preclinical Species
To develop standardized morphologic descriptors for common ESA-related toxicities in rats, Beagle dogs, and cynomolgus monkeys identified in the retrospective analysis of toxicology study reports for the ESAs listed in Table 1, and to provide a basis for comparison to evaluate the incidence of ESA-related toxicities in the investigative study with AMG 114, the H&E-stained tissue slides of control and treated animals from target organs identified as ESA-related (heart/heart valve, stomach, kidney, spleen and bone/bone marrow from femur, and sternum) from a prior 1-month toxicology study with AMG 114 were retrieved from the Amgen internal archives. From this study, target tissues were peer reviewed microscopically for the presence of a thrombus or indirect evidence of a thrombotic event such as an infarct and/or combinations of findings such as focal necrosis, fibrosis, inflammation, and hemorrhage. All standard tissues from the investigative study with AMG 114 were reviewed microscopically and compared to findings in the 1-month AMG 114 toxicity study dosed once weekly.
Statistical Analysis
The sporadic, incomplete, and nonuniform data and variable study designs prohibited statistical analysis across ESA toxicology studies gathered for the retrospective analysis. Statistical analyses performed on individual preclinical toxicology study data sets were not shown.
Results
Characterization of ESA-related Valvulopathy and Gastric Lesion in Rats
A valvulopathy and gastric lesion were identified as ESA-related toxicities unique to the rat during the retrospective analysis of ESA preclinical toxicology studies listed in Table 1 and further characterized in the AMG 114–dosed rats. The number and type of valves present in each section of heart was recorded and a grading system developed to characterize the lesion (Table 2). The severity of the microscopic heart valve lesion was based predominantly on the magnitude of cellular proliferation that distorted the size and shape of the valve (Figure 1). The degree and type of inflammatory infiltrates (typically a combination of mononuclear cells and neutrophils), presence of hemorrhage or thrombi, and deposition of stroma by the proliferative cells were also considered when grading the lesion. The stomach lesion ranged from mucosal necrosis, interpreted to be incipient erosions (identified by glandular epithelial cell necrosis that was typically horizontal, linear, and associated with acute congestion/hemorrhage) to frank erosions and rarely to small ulcerations (Figure 2). The mucosa adjacent to the erosions and ulcerations was typically also congested, and mucosal and/or submucosal edema, inflammation, and hemorrhage (particularly subjacent to the ulcerations) were sometimes observed.
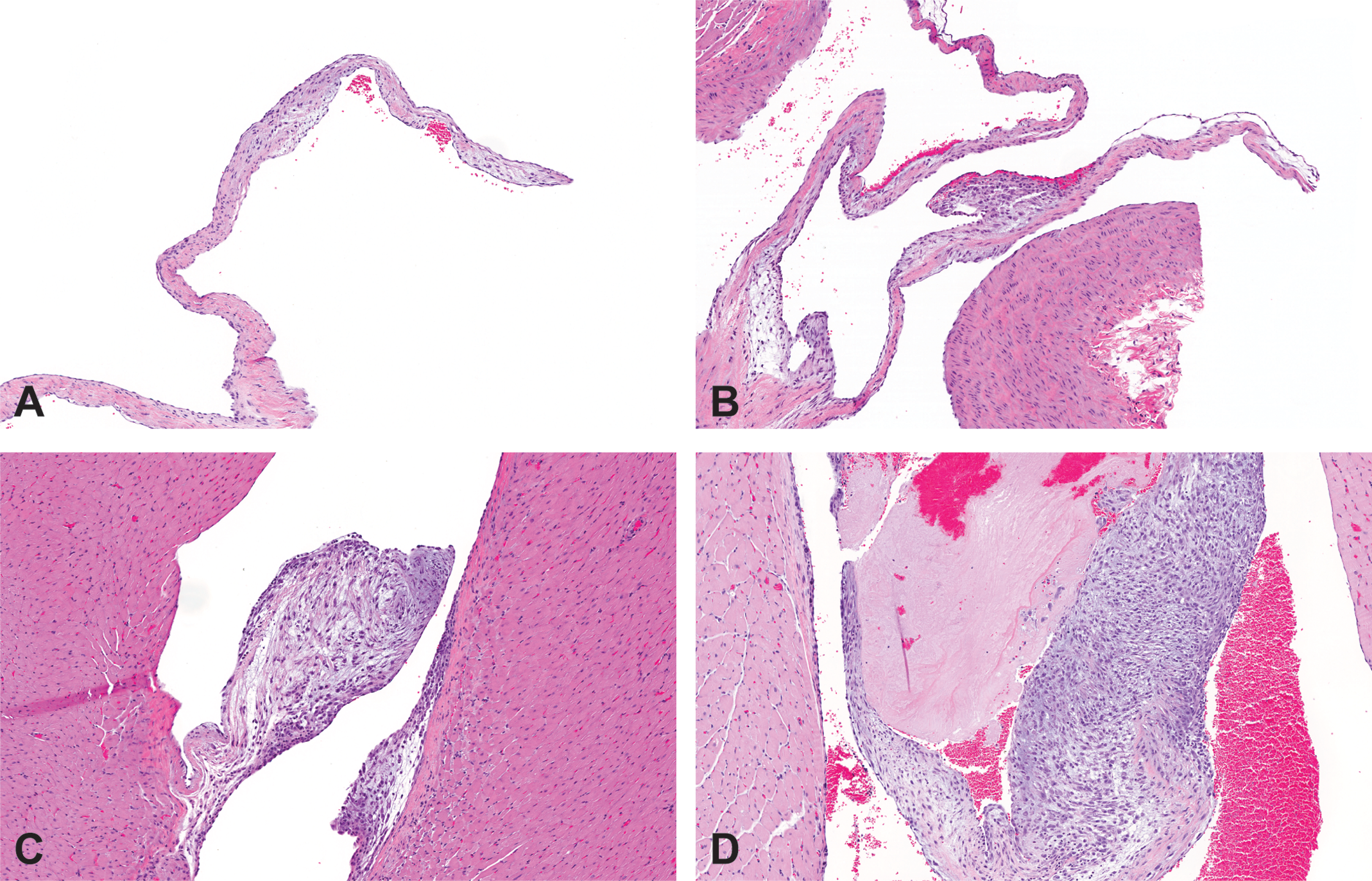
Valvulopathy in Sprague-Dawley rats dosed for 1 month 3 times weekly with a hyperglycosylated analog of recombinant human erythropoietin, AMG 114. (A) Left atrioventricular valve, grade 0 (absent), vehicle control, IV. (B) Aortic valve, grade 1 (minimal), 30 µg/kg/dose, IV. (C) Left atrioventricular valve, grade 2 (mild), 100 µg/kg/dose SC. (D) Left atrioventricular valve, grade 3 (moderate/severe) with associated thrombus, 30 µg/kg/dose, IV. Hematoxylin and eosin stain; 10×. See Table 3 for detailed microscopic grading classification. IV = intravenous; SC = subcutaneous.
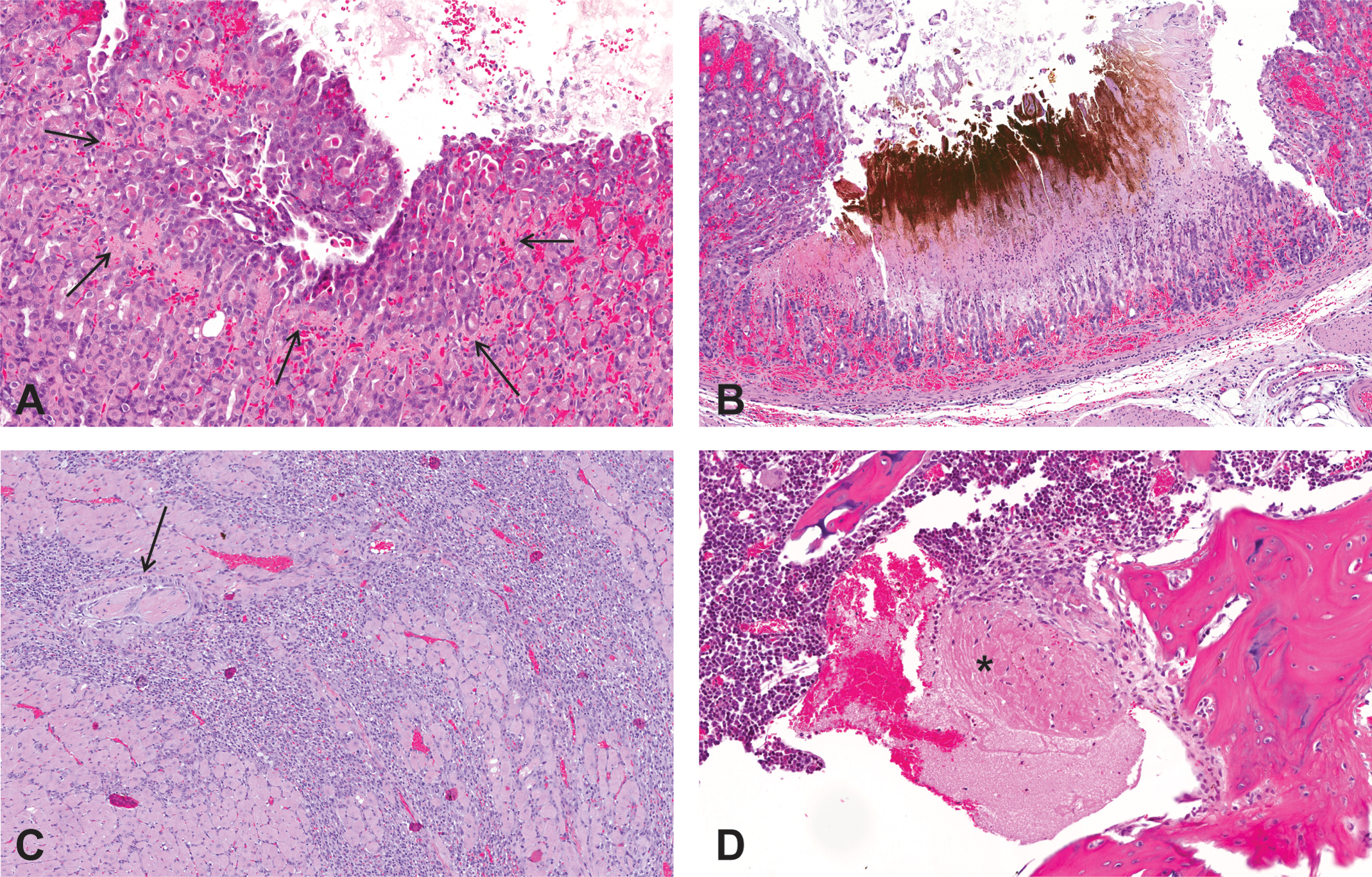
Thrombotic events in Sprague-Dawley rats dosed for 1 month with a hyperglycosylated analog of recombinant human erythropoietin, AMG 114. (A) Glandular necrosis/incipient erosion in the glandular stomach of a rat dosed at 100 µg/kg/dose, IV, once weekly; 20×. The line of necrosis is indicated by the arrows. (B) Deep erosion with mucosal/submucosal congestion and hemorrhage in the glandular stomach, 30 µg/kg/dose, IV, 3 times weekly; 10×. The black/brown material is acid hematin from the staining procedure. (C) Arterial thrombosis (arrow) with cardiac myocyte necrosis/loss and inflammation in the left ventricular free wall, 30 µg/kg/dose, IV, 3 times weekly; 10×. (D) An organizing thrombus (*) in the diaphyseal marrow cavity in the femur, 100 µg/kg/dose, IV, 3 times weekly; 20×. Hematoxylin and eosin stain. IV = intravenous.
Microscopic grading classification for valvulopathy observed in Sprague-Dawley rats in repeat dose toxicology studies with a hyperglycosylated analog of recombinant human erythropoietin, AMG 114.

High HCT and Common Toxicities in Preclinical Species Administered Structurally Different ESAs
Pharmacodynamic Response
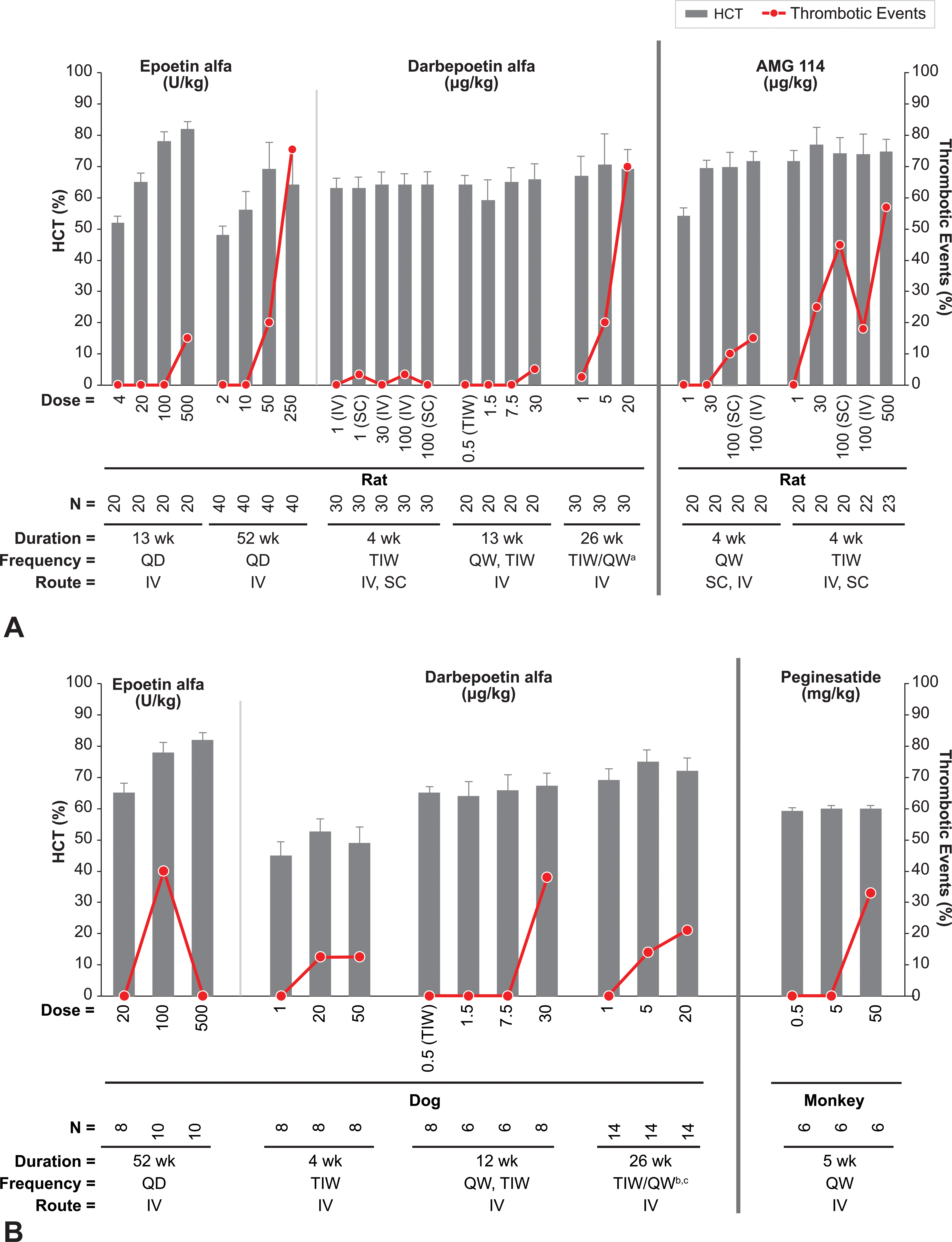
Maximal HCT was achieved in two or more dose groups in the ESA toxicology studies with epoetin alfa, darbepoetin alfa, peginesatide, or AMG 114 (Figure 3). In many of these studies, all dose groups achieved a similarly high HCT. The maximum HCT values in the toxicology studies with epoetin alfa, darbepoetin alfa, and peginesatide ranged from 63 to 82% in rats, 64 to 75% in Beagle dogs, and 59 to 69% in cynomolgus monkeys and the mean highest HCT values for the different species was 68%, 67%, and 63%, respectively. In the AMG 114 toxicology studies, all dosed groups of animals achieved a similarly high HCT that ranged from 69 to 77% except for the animals dosed once weekly with 1.0 µg/kg/dose AMG 114 that had an average HCT of 54%.
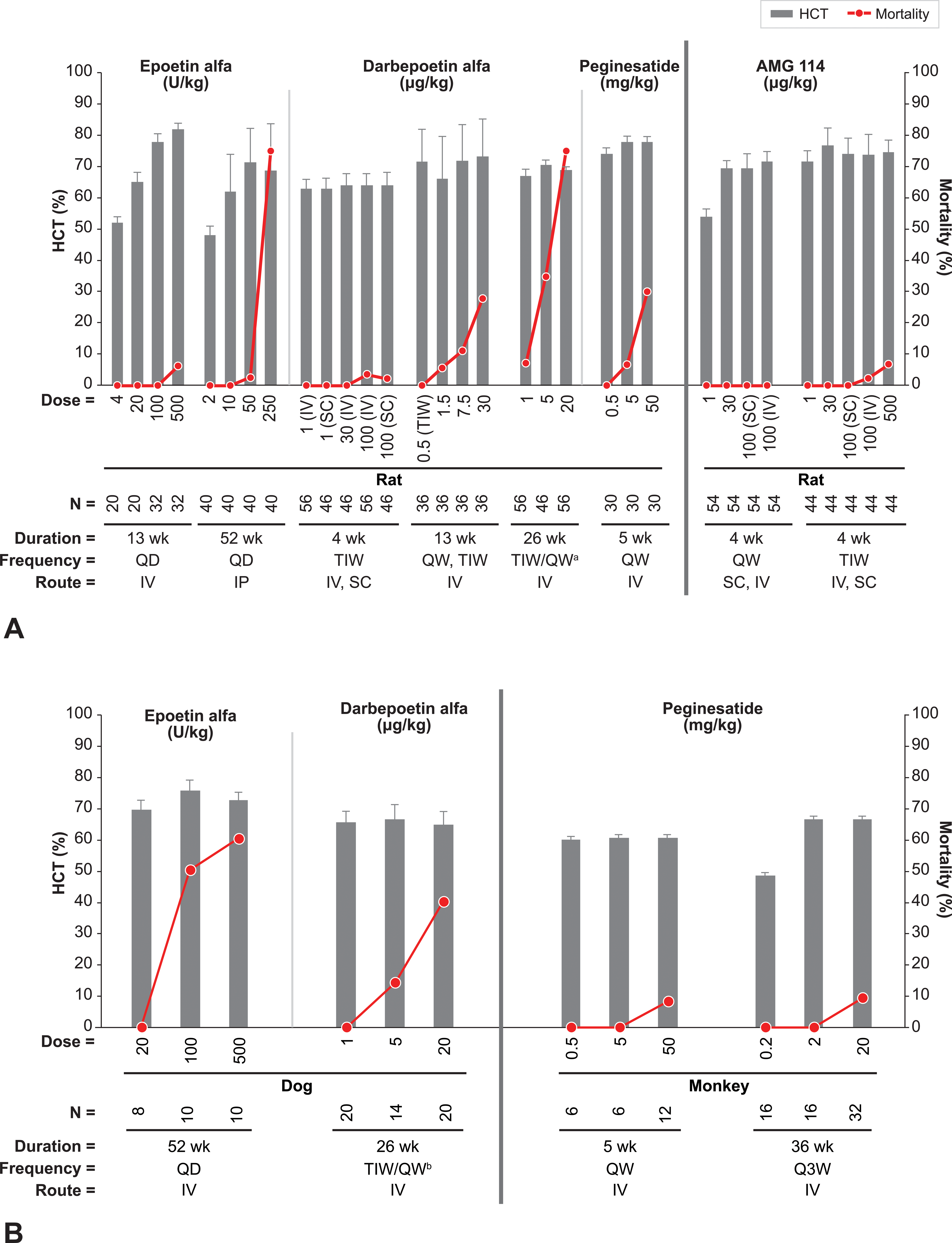
The incidence of mortality in rats (A), and Beagle dogs or cynomolgus monkeys (B) by dose as a function of hematocrit (HCT) in toxicology studies with epoetin alfa, darbepoetin alfa, peginesatide, and a hyperglycosylated analog of recombinant human erythropoietin, AMG
Mortality
In general, the incidence of mortality in the epoetin alfa, darbepoetin alfa, peginesatide, and AMG 114 toxicology studies in rats, Beagle dogs, or cynomolgus monkeys was dose related despite a similarly high HCT achieved in most or all dose groups (Figure 3). Increased length of dosing also increased mortality for the same ESA (Figure 3).
No treatment-related mortality occurred when AMG 114 was dosed once weekly, but 5 unscheduled deaths occurred when AMG 114 was dosed 3 times weekly (3 times total dose per week). Mortality occurred only in the highest dose groups (100 or 500 µg/kg/dose) despite a similar HCT in the similar and/or lower dose groups of animals in both studies (Figure 3). All unscheduled deaths occurred in satellite animals used for TK analysis (1 male, 4 females) following a scheduled venipuncture at the end of the study. In 4 of these 5 animals, no prior adverse clinical observations or prodromal signs were observed. At necropsy, the found dead animals all had enlarged spleens and microscopic findings of valvulopathy, thrombus, and/or cardiomyocyte degeneration. The remaining animal was euthanized due to adverse clinical observations of swollen and limited use of the hind end that correlated histologically with osteolysis/necrosis in the femur and tarsal joint. This animal also had an enlarged spleen and valvulopathy.
Thrombotic Events
The same ESA toxicology studies included in the analysis of HCT and mortality all reported thrombotic events, but not necessarily in the same animals that died or were euthanatized early. Data from these same studies, with the addition of data from 2 dog toxicology studies with darbepoetin alfa that reported thrombotic events but no mortality (4 and 12 weeks), were used to examine the relationship of HCT and thrombotic events.
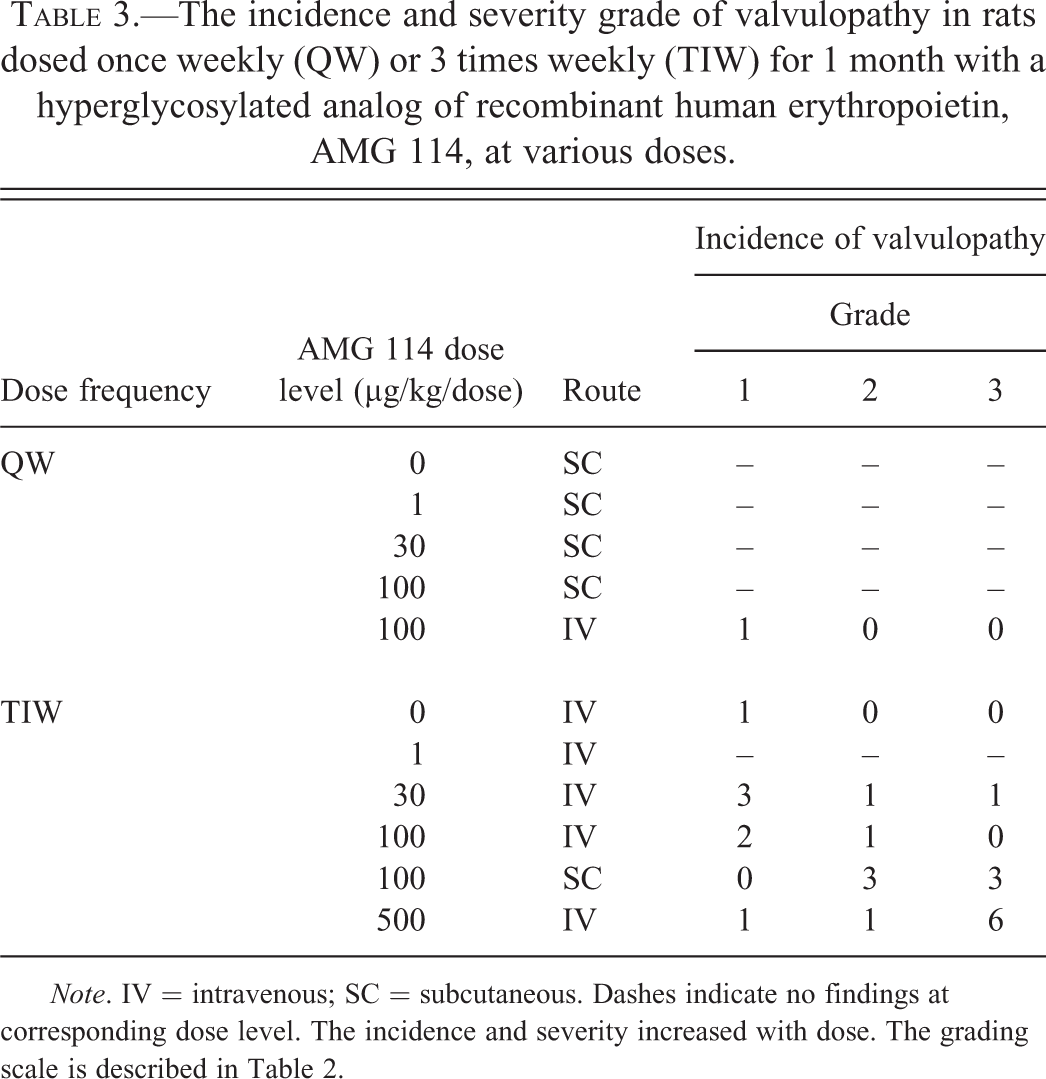
In general, the incidence of thrombotic events in toxicology studies with epoetin alfa, darbepoetin alfa, peginesatide, or AMG 114 was dose related despite a similarly high HCT achieved in the majority of dose groups (Figure 4). In rats and Beagle dogs, increased length of dosing generally increased the incidence of thrombotic events for the same ESA (Figure 4). In the rat toxicology studies with AMG 114, total thrombotic events increased ∼5-fold, in animals dosed 3 times weekly (31/105) compared to once weekly dosing (5/80) and the highest incidence of thrombotic events occurred in the highest dose group overall (500 µg/kg), despite a similarly high HCT achieved across most dose groups in both studies. The incidence of the valvulopathy lesion alone was ∼20-fold higher in the rats in the AMG 114 study dosed 3 times weekly (22/105) compared to rats dosed once weekly (1/80). The incidence and severity of the valvulopathy was increased in the higher dose groups (Table 3). In the study when animals were dosed 3 times weekly, the valvulopathy was identified predominantly in the left atrioventricular valve (12/22 total valve lesions) and with slightly less frequency in the aortic valve (10/22). No lesions were identified in the right atrioventricular valve. The pulmonic valve was underrepresented in heart sections and could not be accurately assessed. A valvulopathy of grade 1 severity was observed in 1 male control rat in the AMG 114 study dosed 3 times weekly and in 3/194 (2 males; 1 female) control animals analyzed from nonrelated studies. Therefore, the grade 1 valvulopathy lesion identified in 1 high dose (100 µg/kg) animal in the AMG 114 study dosed once weekly could be a background lesion.
The incidence of total thrombotic events and valvulopathy in rats (A), and thrombotic events in Beagle dogs or cynomolgus monkeys (B) by dose as a function of hematocrit (HCT) in toxicology studies with epoetin alfa, darbepoetin alfa, peginesatide, and a hyperglycosylated
The incidence and severity grade of valvulopathy in rats dosed once weekly (QW) or 3 times weekly (TIW) for 1 month with a hyperglycosylated analog of recombinant human erythropoietin, AMG 114, at various doses.
Note. IV = intravenous; SC = subcutaneous. Dashes indicate no findings at corresponding dose level. The incidence and severity increased with dose. The grading scale is described in Table 2.
The kidney was the most common location identified in reported ESA-related thrombotic events in rats (64/81) or Beagle dogs (7/16) and thrombotic events were reported with much less frequency (2–5 events) in the renal artery, lung, heart wall, or heart valves in rats and in the bladder in Beagle dogs. Single thrombotic events were identified in the stomach, spleen, and cerebellum in rats and in the ovary, small intestine, liver, heart wall, and skeletal muscle in Beagle dogs. Infarcts in the heart and kidneys were identified in 1 cynomolgus monkey dosed with peginesatide (Woodburn, Schatz, et al. 2008). In the AMG 114-dosed rats, the most frequently identified location for a thrombotic event was the heart valve (14), followed by infarcts in the bone (10), and heart wall (3). A single thrombotic lesion was identified in the cecum, kidney, or spleen in animals from the AMG 114 study dosed 3 times weekly. Thrombi in the heart wall were located in the coronary artery or attached to the wall of the aorta.
The ESA toxicology study reports consistently described a necrotic lesion in the glandular portion of the stomach in rats (Figure 2) except in one 4-week study with darbepoetin alfa. The incidence of the stomach lesion was generally dose dependent (Figure 5). However, unlike the other ESA-related toxicities, an increased incidence was not associated with longer study duration for the same ESA (Figure 5).
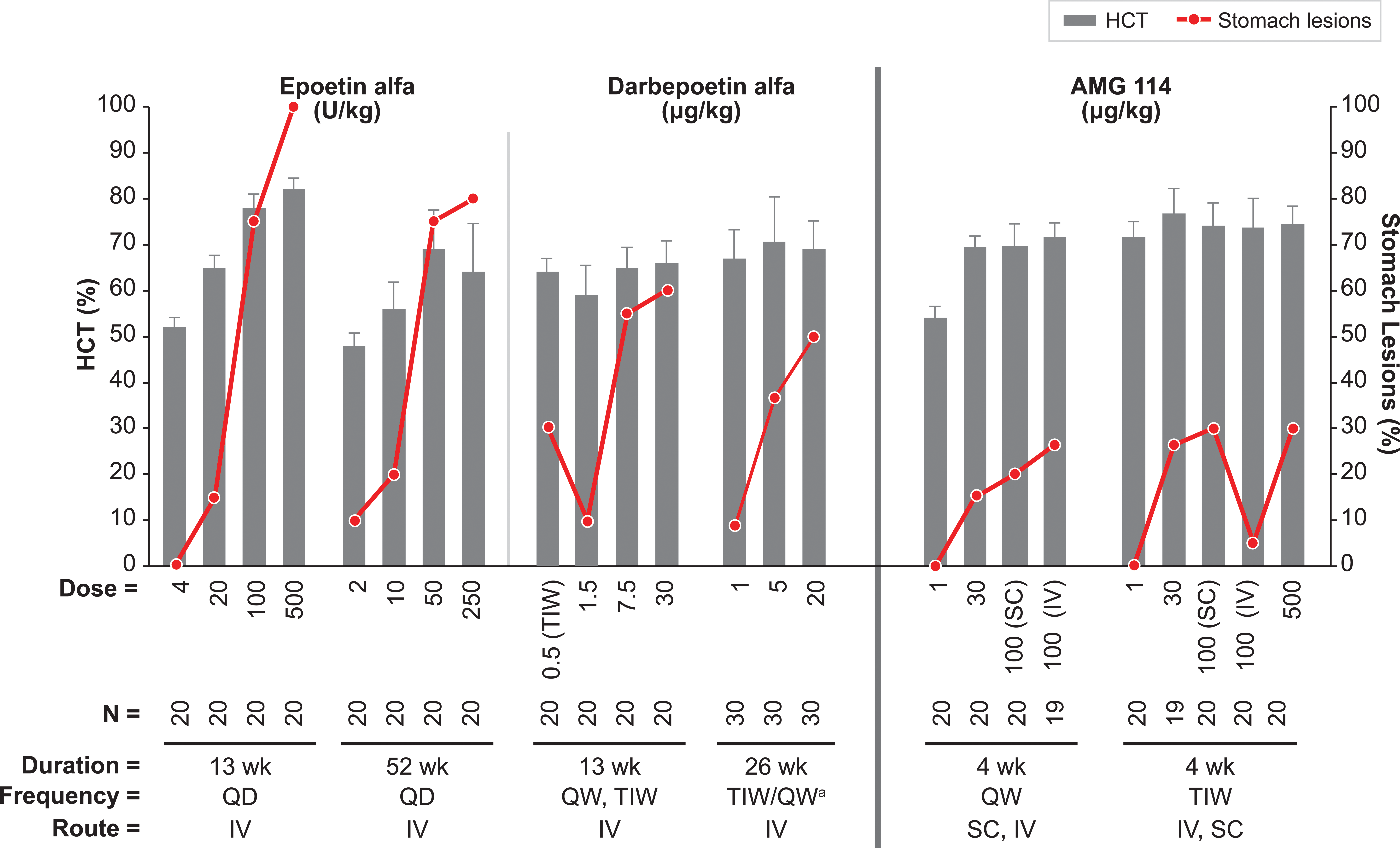
The incidence of stomach lesions in rats by dose as a function of hematocrit (HCT) in toxicology studies with epoetin alfa, darbepoetin alfa, and a hyperglycosylated analog of recombinant human erythropoietin (AMG 114). The incidence of stomach lesions increased with dose, while similar HCT was achieved in 2 or more dosed groups. HCT is displayed as the group M ± SD. QD = once daily; QW = once weekly; TIW = three times weekly. a Dosed TIW through study day 47, then once weekly at dose levels of 1.5, 7.5, and 30 µg/kg to the end of study.
Discussion
To better understand the relationship between HCT and toxicities in animals repeatedly dosed with ESAs, we conducted a retrospective analysis of study data extracted from internal Amgen and publicly available ESA preclinical toxicology study reports. A consistent set of toxicities were reported regardless of the ESA administered, and only in the presence of a high HCT. The toxicities included mortality and/or thrombotic events in rats, Beagle dogs, and cynomolgus monkeys and a valvulopathy and stomach lesion identified only in rats. The cause for mortality was frequently undetermined according to the study reports. Dose levels and/or dose frequencies above those producing a maximal HCT and extended study durations resulted in an increased incidence of ESA-related toxicities without further increases in HCT. This suggested that a high HCT was not the sole pathogenesis underlying the development of toxicities in ESA preclinical toxicology studies. We tested this hypothesis by administering AMG 114, a hyperglycosylated analog of rHu-EPO to Sprague-Dawley rats more frequently (3 times weekly) and at a higher dose level (500 µg/kg/dose) than was administered in a previous 1-month AMG 114 rat study (100 µg/kg/dose, dosed once weekly). The incidence of mortality and/or thrombotic toxicities increased in the animals dosed 3 times weekly compared to once weekly, and was highest in the 500-µg/kg dose level despite a similarly high HCT in all groups of animals. The results from the retrospective analysis and investigative study indicate that mortality and/or ESA-related thrombotic toxicities in preclinical species are also a function of increased dose, dose frequency, and dosing duration, and not solely dependent on a high HCT.
Multiple hypothetical mechanisms and/or contributing factors have been explored in vitro and in vivo in an attempt to reveal the link between ESA administration and thrombosis, but no definitive conclusions have been made (Goodkin 2009; Christensson, Danielson, and Lethagen 2001; Rios et al. 2011; Lindenblatt et al. 2007; Pawlak, Pawlak, and Mysliwiec 2007; Kirkeby et al. 2008). All of the animals with ESA-related toxicities had an extremely high HCT due to repeat dosing with an ESA for at least 1 month which was considered a predisposing factor in development of the toxicities. However, a comparably high HCT was also achieved in animals with no toxicities and therefore not considered solely causal. A high HCT is considered the greatest factor contributing to increased viscosity of the blood, whose other principal determinants include RBC deformability and the viscosity of plasma (Jeong et al. 2010). Increased viscosity alters the hemodynamic properties of blood and is considered a risk factor for thrombosis (Ogunshola et al. 2006; Kwaan and Bongu 1999; Kwaan and Wang 2003; Barshtein, Ben-Ami, and Yedgar 2007). This rheological relationship implies that polycythemia, regardless of origin, always imparts an increased risk of thrombosis which is a clinically inconsistent association (Fedde et al. 1996). There are several examples of humans that maintain an extremely high HCT of 75 to 91% as an adaptive measure to living and/or working in extreme altitudes with no apparent adverse sequelae (Santolaya et al. 1989). A high Hb has not been shown as a predominant risk factor for thrombosis in patient populations with polycythemia vera (PV) due to a naturally occurring JAK2 mutation (Di Nisio et al. 2007; Barbui et al. 2009). A similar disconnect between high HCT and thrombosis has been observed in animal models. Transgenic mice that overexpress EPO have extremely high HCT (often >85%), but no thromboembolic complications have been reported (Shibata et al. 2003). Mouse models of PV or Chuvash polycythemia also have severe polycythemia due to the introduction of a JAK2 V617F or vHL mutation, respectively, but no evidence of thrombosis (Zaleskas et al. 2006; Hickey et al. 2010). Thrombotic events have not been documented in nontransgenic animal models of polycythemia including normal mice that were transplanted with bone marrow cells transfected with a retroviral vector carrying EPO cDNA (Villeval, Metcalf, and Johnson 1992).
The lack of thrombotic events in animal models of severe polycythemia has also been investigated. Despite their high HCT, blood pressure and cardiac output in transgenic mice overexpressing EPO is normal (Ruschitzka et al. 2000). Adaptive mechanisms that likely mitigate the thrombotic risk in these animals include increased nitric oxide–mediated vasodilation and increased erythrocyte deformability to decrease blood pressure and regulate blood viscosity (Ruschitzka et al. 2000; Vogel et al. 2003; Shibata et al. 2003). Similar adaptive mechanisms were evoked in adult animal models with rapidly induced polycythemia (Fedde et al. 1996; Vogel et al. 2003). No reports of increased blood pressure were identified in the retrospective review of the rare repeat dose ESA toxicology studies that measured this parameter. Other antithrombotic mechanisms are likely important because increased HCT alone under high shear stress in an ex vivo primate model did not promote thrombus growth (Cadroy and Hanson 1990). Conversely, phlebotomy alone does not consistently mitigate the risk of a thrombotic event (Crisà et al. 2010). Correlations between HCT and the risk of thrombosis is also quite variable between different disease states further supporting a multifactorial pathogenesis (Fullmer and Miller 2009; Szczech et al. 2008; Christensson Danielson, and Lethagen 2001; Di Nisio et al. 2007).
The rate of HCT/Hb rise after ESA administration is an additional factor postulated to potentiate thrombosis, although this association has not been conclusively demonstrated (Palmer et al. 2010; Singh 2010). In publicly available dog and rat preclinical toxicology studies with darbepoetin alfa, epoetin alfa, or AMG 114, the rate of Hb rise was not a predictor of mortality or toxicities (data not shown).
The results of the retrospective analysis identified the kidney as the most prevalent site for ESA-related thrombotic events, but additional thrombotic events were identified in a wide variety of other organs in the same studies. Endothelial heterogeneity in different vascular beds can influence thrombosis and account for the predisposition for the kidney identified in the retrospective analysis (Kwaan and Samama 2010). The wide distribution of thrombotic events in many different organ systems suggests that polycythemia, which is common to all ESA-dosed animals, is a predisposing factor that potentiates thrombosis in general, but unrecognized prothrombotic factors are necessary to elicit the lesion at a specific site in these animals. The pathogenesis of the ESA-related stomach lesion in rats is likely ischemic, originating from (undetected) microthrombi that lodge in small capillaries of the gastric mucosa causing necrosis, erosion, or ulceration. While this lesion is likely the manifestation of a thrombotic tendency in ESA-dosed animals, it occurs with high frequency and in the lowest dose groups of animals, unlike the other ESA-related toxicities. This may be due in part to increased sensitivity of the rat gastric mucosa to hypoxic injury (Wang et al. 2011), compounded by the predisposition conferred by chronic polycythemia.
The ESA-related valvulopathy was a sporadic lesion that occurred only in the rat suggesting its development was not solely due to the added work of chronic polycythemia (Butcher et al. 2006). The morphology of the ESA-related valvulopathy was unlike that described for drug-induced heart valve lesions in rats as a result of 5-hydroxytryptamine-2B receptor activation (Elangbam 2010; Elangbam et al. 2008) or recently, due to ALK5 inhibition (Anderton et al. 2011). Any morphologic similarities are likely due to the relatively few available avenues for heart valves to react to stimuli (Elangbam 2010; Donnelly 2008). In our results, we described a proliferative valvulopathy as a low incidence background lesion in young Sprague-Dawley rats. The morphology was distinctly different from the fibromyxoid change described with relatively high incidence in aged Harlan Sprague-Dawley rats (Elangbam et al. 2002; Droogmans et al. 2009). From the literature, descriptions of a similar minor proliferative heart valve lesion were identified as a background lesion in young Sprague-Dawley rats (Oka et al. 1966; Angrist et al. 1960). It is unknown if a preexisting proliferative background valve lesion could be a predecessor of a more severe, valvulopathy exacerbated by AMG 114 administration. The AMG 114–related valvulopathy in rats was commonly accompanied by an attached thrombus representing a potential source for emboli (De Castro et al. 1997). Embolic events may have been the cause for sudden death in some animals, but were not consistently described in the ESA dosed rats nor linked to mortality on any study.
The results from this retrospective analysis associate toxicities including mortality, thrombotic events, valvulopathy, and stomach lesions with high and frequent, repeat doses of ESAs in preclinical toxicology studies at least 1 month in duration. Extremely high HCTs achieved in these animals as a result of repeat dosing with ESAs are a predisposing factor in development of these toxicities, but comparably high HCTs in lower dose groups with no toxicities suggest that HCT as the sole causal factor is unlikely. Unidentified factors associated with high and frequent doses of ESAs contribute to the development of toxicities in preclinical species.
Footnotes
Acknowledgments
The authors offer special thanks to Amgen colleagues Audrey Mancini and Kathy Quon for guidance and technical support with the internal ESA preclinical toxicology studies, to Eric Tien and Mark Fielden for their expertise with PharmaPendium, to pathologists Jim Turk and Jon Werner for sharing their knowledge of pathobiology, and to Rogely Boyce for her invaluable scientific contributions and constructive guidance with this project. We additionally thank Heather Hartley-Thorne (SurfMedia Entertainment) for her talents in preparation of the figures, as well as the toxicologists, animal research scientists, and pathologists at Covance Laboratories Inc., Madison, Wisconsin, involved in the investigative study for their collaboration and expertise.
The authors are all employees of Amgen, Inc.
Amgen, Inc. sponsored the retrospective analysis and preclinical investigative studies conducted with AMG 114, epoetin alfa and darbepoietin alfa reported in this manuscript. Amgen, Inc. manufactures and markets epoetin alfa and darbepoietin alfa and manufactured AMG 114 for investigative purposes only.
