Abstract
Acyl-coenzyme A: cholesterol O-Acyltransferase (ACAT) and Acyl-coenzyme A: diacylglycerol O-acyltransferase (DGAT) enzymes play important roles in synthesizing neutral lipids, and inhibitors of these enzymes have been investigated as potential treatments for diabetes and other metabolic diseases. Administration of a Acyl-coenzyme A: diacylglycerol O-acyltransferase 1 (DGAT1) inhibitor with very limited cellular selectivity over ACAT resulted in significant adrenocortical degenerative changes in dogs. These changes included macrosteatotic vacuolation associated with adrenocyte cell death in the zonae glomerulosa and fasciculata and minimal to substantial mixed inflammatory cell infiltration and were similar to those described previously for some ACAT inhibitors in dogs. In the mouse, similar but only transient adrenocortical degenerative changes were seen as well as a distinctive nondegenerative reduction in cortical fine vacuolation. In the marmoset, only the distinctive nondegenerative reduction in cortical fine vacuolation was observed, suggesting that the dog, followed by the mouse, is the most sensitive species for cortical degeneration. Biochemical analysis of adrenal cholesterol and cholesteryl ester indicated that the distinctive reduction in cortical fine vacuolation correlated with a significant reduction in cholesteryl ester in the mouse and marmoset, whereas no significant reduction in cholestryl ester, but an increase in free cholesterol was observed in dogs. Administration of a DGAT1 inhibitor with markedly improved selectivity over ACAT to the marmoset and the mouse resulted in no adrenal pathology at exposures sufficient to cause substantial DGAT1 but not ACAT inhibition, thereby implicating ACAT rather than DGAT1 inhibition as the probable cause of the observed adrenal changes. Recognizing that the distinctive nondegenerative reduction in cortical fine vacuolation in the mouse could be used as a histopathological biomarker for an in vivo model of the more severe changes observed in dogs, the mouse has subsequently been used as a model to select DGAT1 inhibitors free of adrenocortical toxicity.
Keywords
Introduction
Acyl-coenzyme A: diacylglycerol O-acyltransferase 1 (DGAT1), a key enzyme that catalyzes triglyceride synthesis from monoglycerides and diglycerides, has a potential role as a pharmacological target in the treatment of type 2 diabetes and obesity (Yen et al. 2005, 2008; Zammit et al. 2008; Birch, Buckett, and Turnbull 2010). Administration of a drug candidate, AZD3988, to dogs caused adrenal toxicities which prompted concern regarding dosing to humans. Similar adrenocortical degenerative changes have previously been described following administration of several ACAT (1 and/or 2) inhibitors to laboratory animals and had been considered to be associated with an excess of free unesterified cholesterol.
Cholesterol is a lipid present in the membranes of mammalian cells and is essential for their viability. ACAT catalyzes intracellular esterification of cholesterol and formation of cholesteryl ester (CE) in many mammalian tissues (Cases et al. 1998; Chang et al. 2009; Farese 2006; Leon, Hill, and Kishor 2005). Esterification of cholesterol by ACAT limits its solubility in the cell membrane and promotes accumulation of cholesteryl ester in cytoplasmic fat droplets. ACAT activity prevents the accumulation of potentially toxic free cholesterol in various cell membranes with excess cholesterol being stored as inactive cholesteryl ester. In steroidogenic tissues, such as the adrenal glands, cholesteryl esters serve as the cholesterol reservoir for producing steroid hormones. Using cellular systems, it has been demonstrated that ACAT1 inhibitors can cause undesirable accumulation of cellular free cholesterol leading to apoptosis (Feng et al. 2003; Warner et al. 1995). Furthermore, a number of ACAT inhibitors, some of which are nonspecific inhibitors of ACAT1/ACAT2, have induced adrenal toxicities while others, such as Avasimibe, which has since failed in clinical development, have been described as lacking significant adrenal toxicity (Reindel, Dominick, and Krause 1992; Reindel et al. 1994; Wolfgang et al. 1995; Riddell et al. 1996; Wilde et al. 1996; Tanaka et al. 1998; Sliskovic et al. 1998; Sliskovic, Picard, and Krause 2002; Robertson, Breider, and Milad 2001). It is noteworthy that to date, no ACAT inhibitor has successfully emerged as a safe and efficacious therapy from clinical research where studies in humans showed that several drugs lacked efficacy for lowering cholesterol (Farese 2006). It is not clear whether toxicities in animals, which have been described for at least some of these drugs, resulted in the restriction of doses to levels which would not result in substantial ACAT inhibition in humans.
In this communication, we show that the DGAT1 inhibitor AZD3988 with minimal cellular selectivity over ACAT caused the same adrenocortical degenerative pathological changes in dogs as reported in the development of ACAT inhibitors. Following administration of AZD3988 to the marmoset and the mouse, we demonstrated a species-dependent difference in response to this DGAT1 inhibitor–mediated adrenal toxicity. Thus, unlike the degenerative changes observed in dogs, a distinctive nondegenerative reduction in cortical fine vacuolation was observed following administration of AZD3988 to the marmoset and mouse. This change was clearly associated with a reduction in adrenal cholesteryl ester in these species. However, in the mouse transient degenerative changes as well as the nondegenerative reduction in cortical fine vacuolation were observed, indicating that the dog, followed by the mouse, is the most sensitive species for the detection of the degenerative adrenocortical consequences of AZD3988 administration.
Finally, the use of the substantially more selective DGAT1 inhibitor AZ4, which did not induce any adrenocortical changes, provided strong evidence that DGAT1 inhibition alone does not result in adrenocortical pathology, thus firmly implicating only ACAT in the observed adrenal changes. This distinctive reduction in cortical fine vacuolation appeared to be a meaningful histological marker of ACAT inhibition in the mouse and marmoset prompting our consideration of the mouse as a predictive model of ACAT-mediated adrenal changes. This mouse model of ACAT-mediated adrenocortical toxicity has subsequently been used in the discovery of DGAT1 inhibitors that are free of adrenocortical toxicity in dogs.
Materials and Methods
Test Compounds
AZD3988 is a trans-4-{4-[({5-[(3,4-difluorophenyl)amino]-1,3,4-oxadiazol-2-yl}carbonyl)amino]phenyl}cyclohexylacetic acid with dual DGAT1 inhibitory activity synthesized by AstraZeneca R&D, Alderley Park (McCoull et al. 2012). In an in vitro recombinant mouse, dog, and human DGAT1 enzyme assay, this compound has an IC50 of approximately 1, 2, and 0.6 nM, respectively, and an IC50 of 86 nM against the human ACAT1 and 600 nM against the human ACAT2 enzyme. This compound displays tight enzyme–binding characteristics and thus in vivo potency for both enzymes might differ. No activity against DGAT2 was detected. The IC50 for cellular triacylglycerol (TAG) was 4 nM and the IC50 for ACAT-mediated cellular cholesteryl ester synthesis was 7.7 nM.
AZ4 ({4-[4-(4-amino-7,7-dimethyl-7H-pyrimido[4,5-b][1,4]-oxazin-6-yl)phenyl]bicycle [2.2.2] oct-1-yl}acetic acid) was synthesized by AstraZeneca R&D, Alderley Park, United Kingdom, and has selective DGAT1 inhibitory activity (Birch et al. 2009). In an in vitro recombinant mouse, dog, and human DGAT1 enzyme assay, AZ4 has an IC50 of approximately 5 nM, 12 nM, and 16 nM, respectively, and an IC50 of 5,300 nM and >35,000 nM against human ACAT1 and ACAT2 enzymes. No activity against DGAT2 was detected. The IC50 for cellular TAG was 10 nM and the IC50 for ACAT-mediated cellular cholesteryl ester synthesis was 5,240 nM.
Measurement of cellular potency was carried out according to the method described in Birch et al. (2009), but for the separation of cholesteryl ester and triglyceride, the high-performance liquid chromatography methodology was altered to an initial mixture of isohexane:acetic acid 99.9:0.1 changing to 90% isohexane:acetic acid 99.9:0.1, 10% isohexane-propan-2-ol:acetic acid 85:15:1 over the first 6 min. The flow rate was increased to 3 ml/min and the solvent changed to 75% isohexane:acetic acid 99:1, 25% isohexane-propan-2-ol:acetic acid 85:15:1, following a gradient to 75% isohexane-propan-2-ol:acetic acid 85:15:1 by 4.5 min.
For the in vivo experiments, the test compounds were formulated as 50 mg/ml (AZD3988) or 6 mg/ml (AZ4) nanosuspension in 0.67% w/v PVP (polyvinylpyrrolidone, Sigma-Aldrich, United Kingdom), 0.033% w/v AOT (Aerosol OT, Sigma-Aldrich, United Kingdom), and 99.297% w/v water and diluted with vehicle to produce the final formulations required. The vehicle (0.67% w/v PVP/0.033% w/v AOT/99.297% w/v water) served as the control. Generally, compounds and vehicles were formulated on the day prior to study start and then remade every 7 days where necessary. The suspensions were stirred continuously from preparation throughout use and protected from light.
Animals
Four female mice (C57BL/6Jax) per treatment group were obtained from the AstraZeneca breeding unit. The animals, which were 7 to 8 weeks of age, were allowed to acclimatize for at least 1 week, and were multiply housed and given ad libitum access to water and standard rodent diet.
Two female beagle dogs per treatment group were obtained from the AstraZeneca breeding unit. The animals, which were 12 to 24 months of age, were allowed to acclimatize for at least 1 week, were multiply housed, and were given ad libitum access to water and standard dog diet.
Two to three female marmosets (Callithrix jacchus) per treatment group were obtained from the University of Manchester, United Kingdom. The animals, which were 12 to 24 months of age, were allowed to acclimatize for at least 2 weeks, were housed in pairs, and were given ad libitum access to water and standard primate diet. Additional food items, usually including fresh fruit, were offered daily.
Standard procedures and conditions were applied for animal care, feeding, housing, and environment, and studies were conducted in strict adherence to the U.K. Home Office regulations for animal welfare.
Experimental Design
The frequency of dosing for AZ3988 and AZ4 doses was decided following consideration of species-specific tolerability and pharmacokinetic parameters. Doses predicted to result in exposures which were likely to cause maximum DGAT1 inhibition without resulting in intolerability were chosen. For AZ4, doses were also limited to ensure that exposures remained below levels which would result in significant ACAT inhibition. Animals were randomized into groups according to their body weight and dosed twice daily (bid) with the test compounds orally by gavage. Toxicokinetic (TK) sampling on the last dosing day prior to necropsy was employed to assess the exposure profiles of animals treated with the test compounds.
Experimental Procedures
All animals were observed daily for clinical signs of toxicity. Body weight, food, and water intake measurements were measured daily prior to dosing.
AZD3988 was administered to dogs at 0.1, 1, and 4 mg/kg bid, 12 hr apart, for up to 7 days. An additional group was dosed at 100 mg/kg bid, 12 hr apart, for a single day, but was terminated after 3 days to allow any potential adrenal pathological changes to develop without causing unnecessary stress to the animal. Marmosets were given 30 mg/kg AZD3988 or 30 mg/kg AZ4 bid, 12 hr apart, for 7 days. AZD3988 or AZ4 were administered to mice at 30 and 100 mg/kg, respectively, bid, 12 hr apart, for up to 14 days.
Representative samples of both adrenal glands were taken at necropsy and either freeze clamped and stored in liquid nitrogen before being transferred to a −80°C freezer until analysis for lipid content, or were placed in 10% buffered formalin and processed for histopathological assessment. Conventional processing into wax and H&E staining of sections preceded their light microscopic examination.
Adrenal cholesteryl ester and free cholesterol were determined following homogenization of adrenal tissue samples in isopropyl alcohol, lipid extraction from the samples, and content measurement using a modification of the enzymatic, colerimetric assay of Allain et al. (1974).
For pharmacokinetic analysis, blood samples were collected into lithium heparin tubes from animals on either the first or penultimate day of the study. Plasma was separated by centrifugation at 4°C and stored for analysis at −20°C. Plasma concentrations of test compounds were determined by liquid chromatography–mass spectrometry. Pharmacokinetic parameters of maximum plasma concentration (C max) and concentration at dose interval (Clast) were calculated.
Results
Effects of Non-ACAT-selective Adrenal DGAT1 Inhibition in the Dog
The non-ACAT-selective DGAT1 inhibitor AZD3988 was administered to dogs, orally, bid at 12-hr intervals for 7 days, at doses predicted to result in exposures which were likely to cause substantial DGAT1 inhibition down to exposures which would deliver only minimal DGAT1 inhibition in this species. Toxicokinetic analysis revealed that, using inhibition of cellular TAG synthesis as an indicator of DGAT1 activity, plasma drug concentrations were above the DGAT1 IC50 at doses of 1 mg/kg and above (Figure 1). This resulted in plasma drug concentrations above the DGAT1 IC50 for at least part of the dose interval (approximately 5 hr and 10 hr for the 1 mg/kg and 4 mg/kg doses, respectively) and for the full dose interval for the 100 mg/kg dose. The plasma drug concentration at 0.1 mg/kg was below the DGAT1 IC50 for the whole dose interval. Similarly, using inhibition of cellular cholesteryl ester synthesis as an indicator of ACAT activity, only a reduction (approximately 2-fold) difference in levels of DGAT1 and ACAT inhibition was seen and thus, at a dose of 1 mg/kg, the plasma compound concentration was just below the IC50 for cellular TAG and cholesteryl ester synthesis for part of the dose interval (5 hr). Indeed, extensive inhibition was likely to occur for both DGAT1 and ACAT at doses of 4 mg/kg and above.
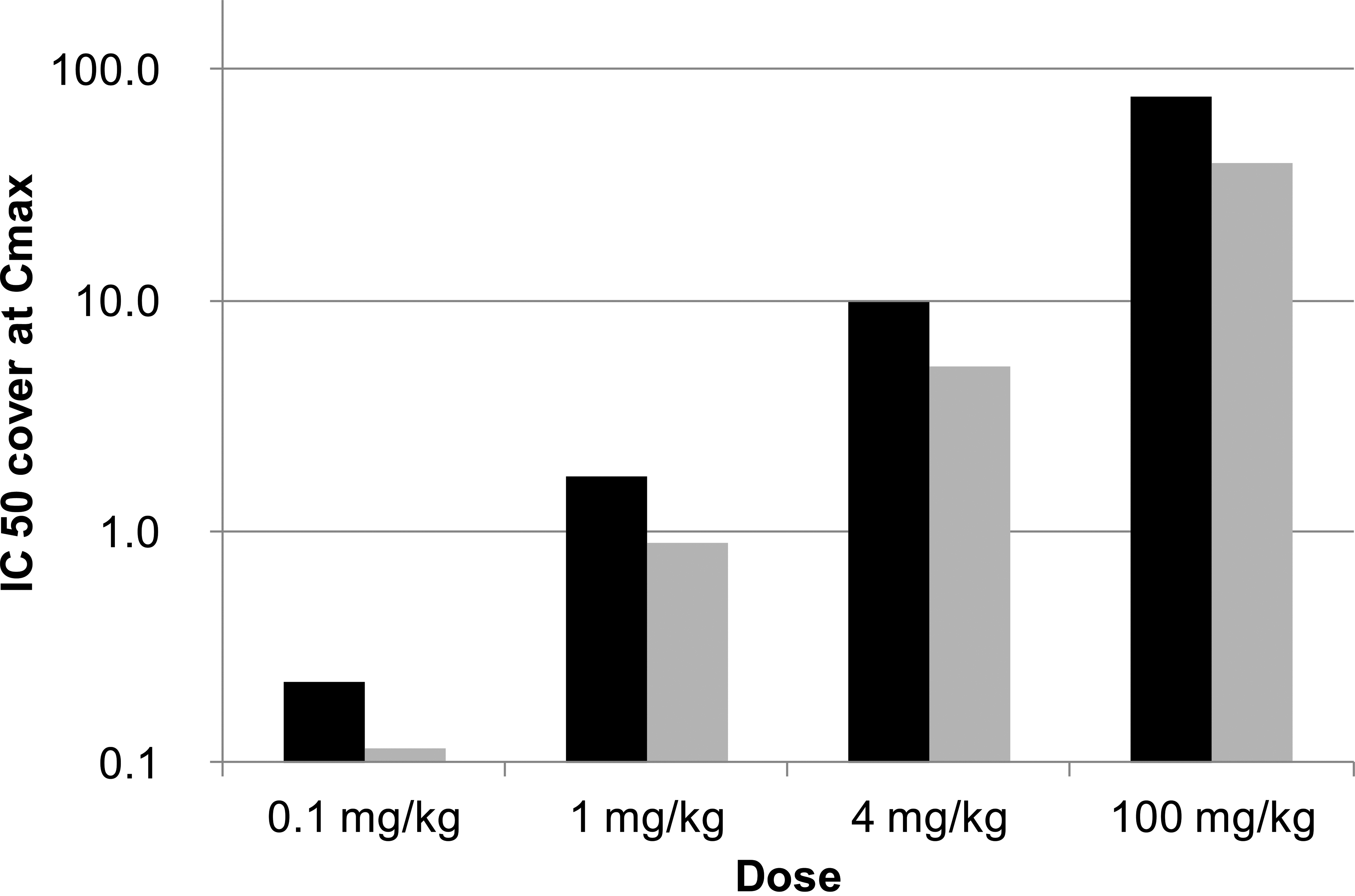
Exposures in dogs given AZD3988 at 0.1, 1, 4, and 100 mg/kg (twice daily [bid]; n = 2 per group) on the last day of dosing. The level of acyl-coenzyme A: diacylglycerol O-acyltransferase 1 (DGAT1) and acyl-coenzyme A: cholesterol O-acyltransferase (ACAT) inhibitions expressed as C max relative to the IC50 for DGAT1-mediated cellular triacylglycerol (TAG) synthesis (black column) and ACAT-mediated cellular TAG synthesis (gray column). Exposures were insufficient to significantly inhibit both, DGAT1 and ACAT1, at 0.1 mg/kg, but increased to levels at the IC50 level for both enzymes at 1mg/kg and significantly exceeded IC50 levels at the 4 and 100 mg/kg doses.
AZD3988 administration at the doses chosen had no significant effects on body weight or any other clinical parameter. However, AZD3988 administration induced cellular vacuolation in the adrenal cortex at 1 or 4 mg/kg AZD3988 for 7 days or 100 mg/kg for 1 day. Specifically, large vacuolation of macrosteatotic appearance associated with cortical adrenocyte cell death in the zonae glomerulosa and fasciculata were noted. These findings were also associated with minimal to substantial mixed inflammatory cell infiltration which included lymphocytes, predominantly monocytes/macrophages and some neutrophils (Figure 2). Cholesterol clefts were also present at higher doses. The severity and incidence of these lesions were clearly dose-related where administration of AZD3988 for 7 days at 0.1 mg/kg revealed no significant effects, while animals given 100 mg/kg for 1 day and terminated after 3 days showed severe adrenocortical vacuolation and hemorrhage.
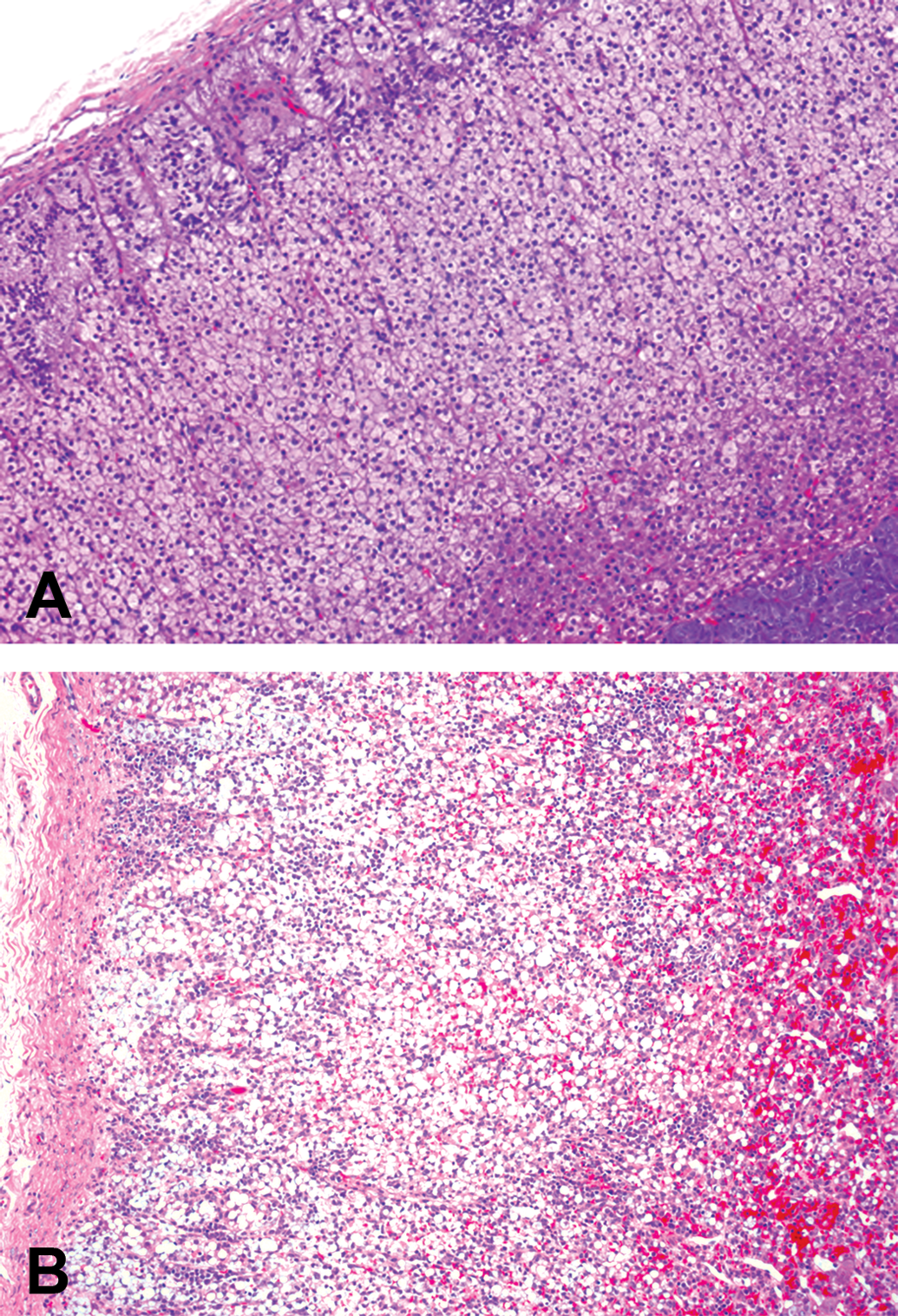
Photomicrographs of adrenal of (A) a control and (B) a dog dosed with 4 mg/kg/day (twice daily [bid]) AZD3988 for 7 days. Note in (A) the slight variation in cortical vacuolation from the outer zona glomerulosa through to the zona fasciculate to the zona reticularis with no pathological changes evident. In (B) cortical macrosteatotic vacuolation is present with substantial inflammatory cell infiltration and minimal hemorrhage (H&E. Original objective lens magnification ×10).
Adrenal glands of dogs treated with AZD3988 were analyzed for cholesterol and cholesteryl ester, and a slight decrease in mean levels of cholesteryl ester was observed with increasing dose (Figure 3). An average of 48 µg cholesteryl ester was detected per mg of adrenal gland protein in dogs treated with 0.1 mg/kg, which decreased to 34 µg per mg in dogs treated with 100 mg/kg. Increases in free cholesterol levels from 2.1 µg free cholesterol per mg of adrenal gland protein at 0.1 and 1 mg/kg to 3.4 µg and 11 µg per mg were detected in dogs treated with 4 mg/kg and 100 mg/kg, respectively. Thus, there were no clear changes in levels of cholesteryl ester in the adrenals of dogs across the dose range, although a reduction and apparent trend in the reduction in cholesteryl ester synthesis in the adrenals of dogs was noticed. An increase in free cholesterol was seen in dogs given doses of 4 mg/kg and was substantial at 100 mg/kg.
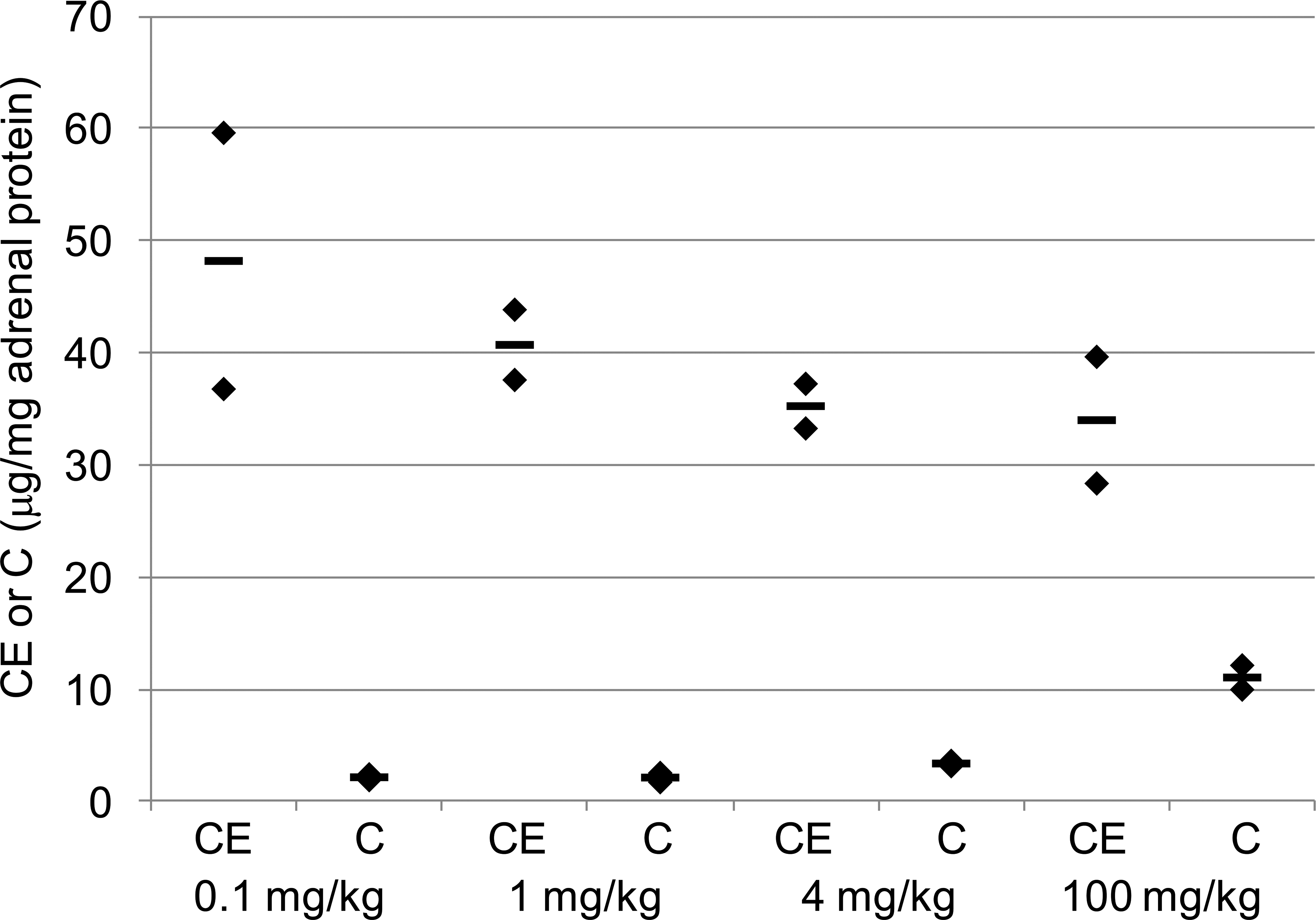
Adrenal cholesteryl ester (CE) and free cholesterol (C) levels in dogs given 0.1, 1, 4, or 100 mg/kg (twice daily [bid]) of AZD3988 (n = 2 per dose group) showing a dose-related decrease in CE and dose-related increase in C.
Effects of Non-ACAT-selective and ACAT-selective Adrenal DGAT1 Inhibition in the Marmoset
The non-ACAT-selective DGAT1 inhibitor AZD3988 as well as the ACAT-selective DGAT1 inhibitor AZ4 were administered to marmosets, orally, bid at 12-hr intervals for 7 days at doses that were predicted to result in maximum DGAT1 inhibition, while being well tolerated and, in the case of the selective DGAT1 inhibitor AZ4, remaining below levels that would result in significant ACAT inhibition in this species. Indeed, plasma concentrations for both drugs were in large excess of concentrations required to inhibit DGAT1-mediated TAG synthesis (Figure 4). AZD3988 exposures covered the IC50 for DGAT1-mediated cellular TAG synthesis by approximately 11-fold at C max and remained above the IC 50 for approximately 8 hr of the 12-hr dose interval. AZ4 exposures reached close to 120-fold the DGAT1 IC50 at C max and were still 9-fold above the IC50 at the dose interval. While exposures of AZD3988 at C max were 6-fold at the IC50 for ACAT-mediated cellular cholesteryl ester synthesis and remained above the IC50 for approximately 6 hr of the 12-hr dose interval, AZ4 exposures were insufficient to cover the IC50 for ACAT-mediated cellular cholesteryl ester synthesis at any time during the dose interval. Thus, while the plasma exposure of AZD3988 in the marmoset was sufficient to substantially inhibit DGAT1 as well as ACAT, exposures of AZ4 were sufficient to substantially inhibit DGAT1 only.
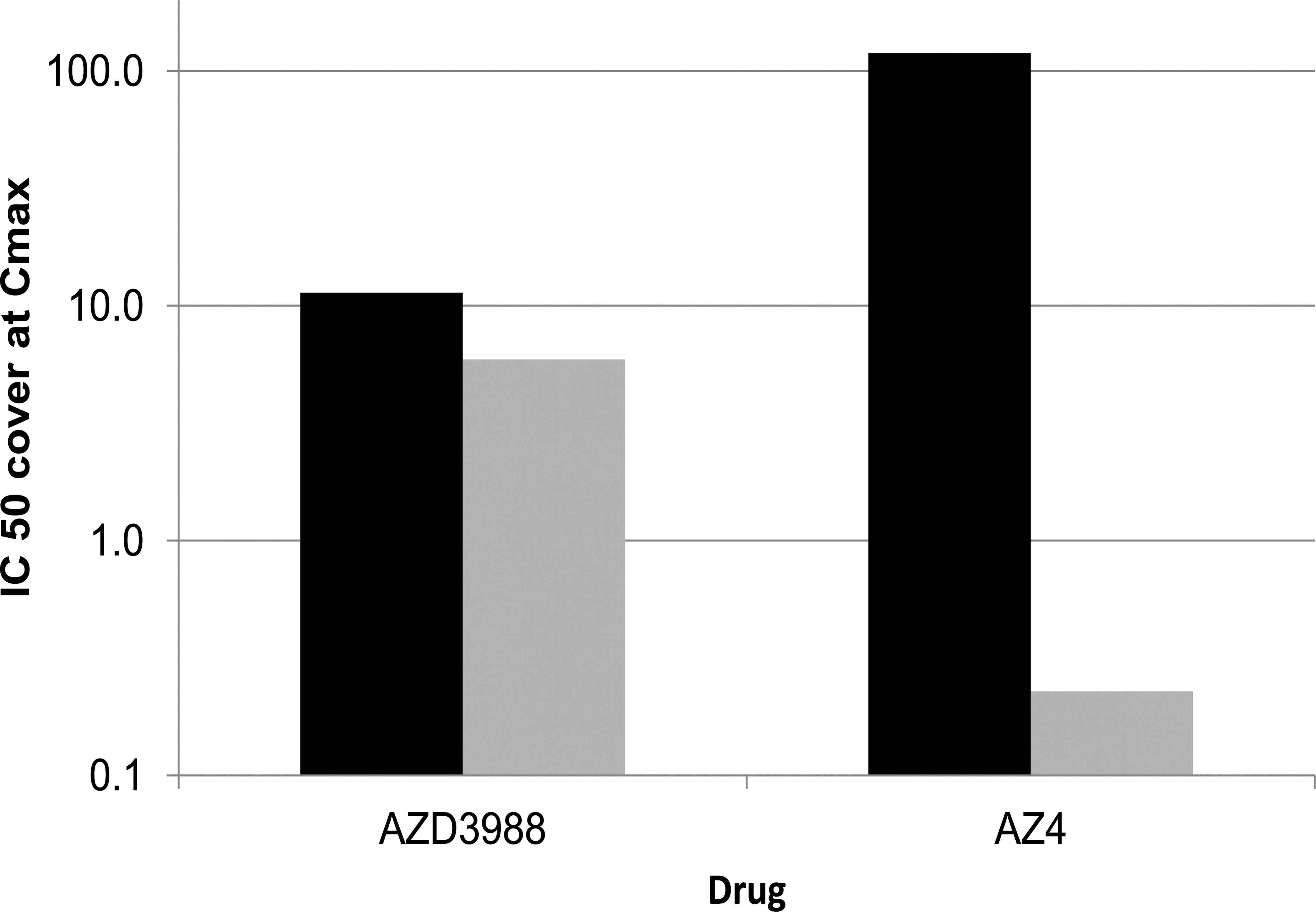
Exposures in marmosets given 30 mg/kg AZD3988 (twice daily [bid]; n = 2) or 30 mg/kg AZ4 (bid; n = 3) on day 7. The level of acyl-coenzyme A: diacylglycerol O-acyltransferase 1 (DGAT1) and acyl-coenzyme A: cholesterol O-acyltransferase (ACAT) inhibition is expressed as C max relative to the IC50 for DGAT1-mediated cellular triacylglycerol (TAG) synthesis (black column) and ACAT-mediated cellular TAG synthesis (gray column). Exposures of AZD3988 were sufficient to significantly inhibit both DGAT1 and ACAT1, while exposures of AZ4 were only sufficient to inhibit DGAT1 but not ACAT1.
Following exposure of marmosets to both drugs, no significant effects of drug administration on clinical observations, body weight, or food consumption were observed.
Histopathological examination of the adrenal glands revealed moderate to severe reduced cortical fine vacuolation in animals given 30 mg/kg AZD3988 (Figure 5) with no degenerative or inflammatory features. Importantly, there were no salient histopathological findings in the adrenals of animals dosed with AZ4.
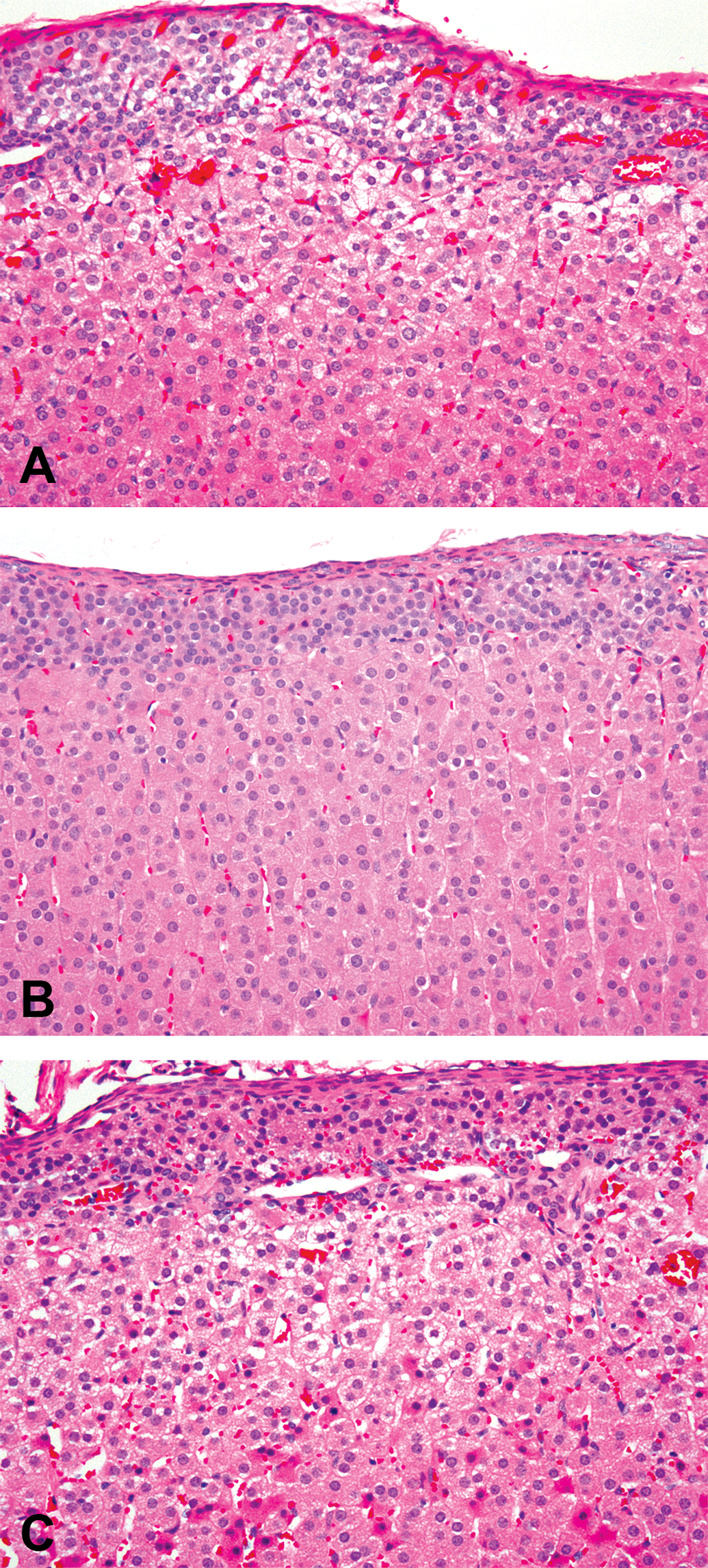
Photomicrographs of marmoset adrenal cortices from (A) a control animal showing minimal variation in adrenocyte vacuolation with no pathological changes present; (B) an animal given 30 mg/kg (bid) AZD3988 for 7 days indicating substantial reduction in zona glomerulosa and zona fasciculata vacuolation; (C) an animal given 30 mg/kg (bid) AZ4 for 7 days showing no effects on cellular vacuolation. (Haematoxylin and eosin. Original objective lens magnification × 40).
Adrenal biochemical analyses revealed cholesteryl ester synthesis was inhibited by AZD3988 but not AZ4. In contrast to the relatively reduction changes in cholesteryl ester seen in dogs, AZD3988 treatment in the marmoset completely inhibited formation of cholesteryl ester without an associated increase in free cholesterol levels (Figure 6). Thus, while cholesteryl ester levels were substantially reduced from control levels of 63 µg per mg of adrenal gland protein to 2 µg per mg following administration of AZD3988, cholesteryl ester levels remained high at 77 µg per mg following treatment with the ACAT-selective DGAT1 inhibitor AZ4. Following administration of AZD3988 and AZ4, free cholesterol levels of 9.5 and 12 µg per mg, respectively, were relatively unchanged against control levels of 14 µg per mg of adrenal gland protein.
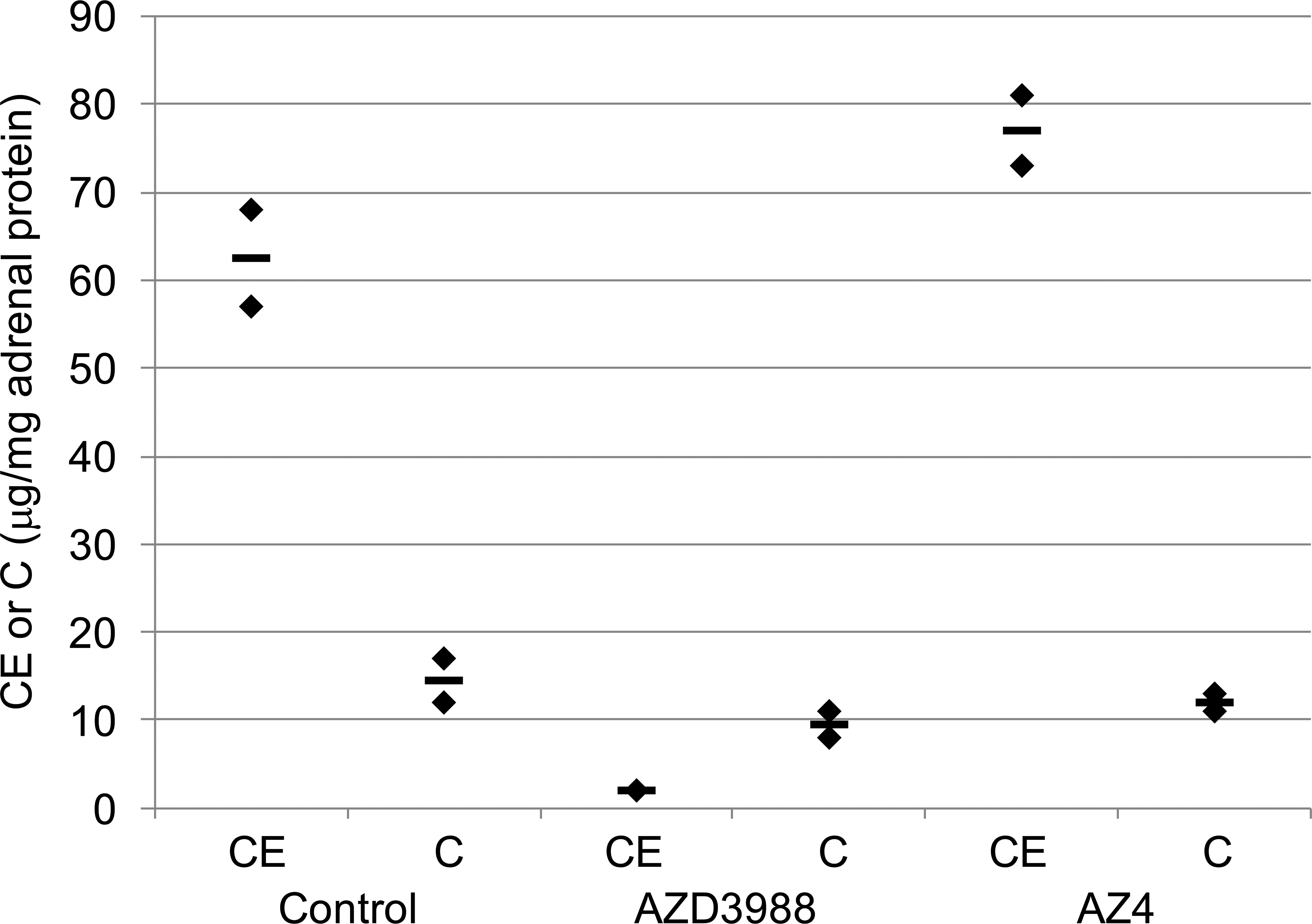
Adrenal cholesteryl ester (CE) and free cholesterol (C) levels in marmosets given 30 mg/kg AZD3988 or 30 mg/kg AZ4 for 7 days (twice daily [bid]; n = 2 per group) showing a marked reduction in CE but only a minimal change in C following administration of AZD3988, while administration of AZ4 did not affect CE or C.
Effects of Non-ACAT1-selective and ACAT1-selective Adrenal DGAT1 Inhibition in the Mouse
The non-ACAT-selective DGAT1 inhibitor AZD3988 as well as the ACAT-selective DGAT1 inhibitor AZ4 were administered to mice, orally, bid at 12-hr intervals for up to 14 days at doses which were predicted to result in maximum DGAT1 inhibition, while being well tolerated and, in the case of the selective DGAT1 inhibitor AZ4, remaining below levels that would result in significant ACAT inhibition in this species. Toxicokinetic analysis revealed that plasma concentrations for both drugs were in large excess of concentrations required to inhibit DGAT1-mediated TAG synthesis (Figure 7). At C max AZD3988 and AZ4 exposures covered the IC50 for DGAT1-mediated cellular TAG synthesis approximately 46-fold and 265-fold, respectively, and remained above the IC50 for the duration of the dose interval (37- and 200-fold at the dose interval, respectively). While exposures with AZD3988 at C max were 24-fold at the IC50 for ACAT-mediated cellular cholesteryl ester synthesis and were still 19-fold above the IC50 at the dose interval, AZ4 exposures at C max were insufficient to cover the IC50 for ACAT-mediated cellular cholesteryl ester synthesis at any time during the dose interval. Thus, while the plasma exposure of AZD3988 in the mouse was sufficient to substantially inhibit DGAT1 as well as ACAT1, exposures of AZ4 were sufficient to substantially inhibit DGAT1 only.
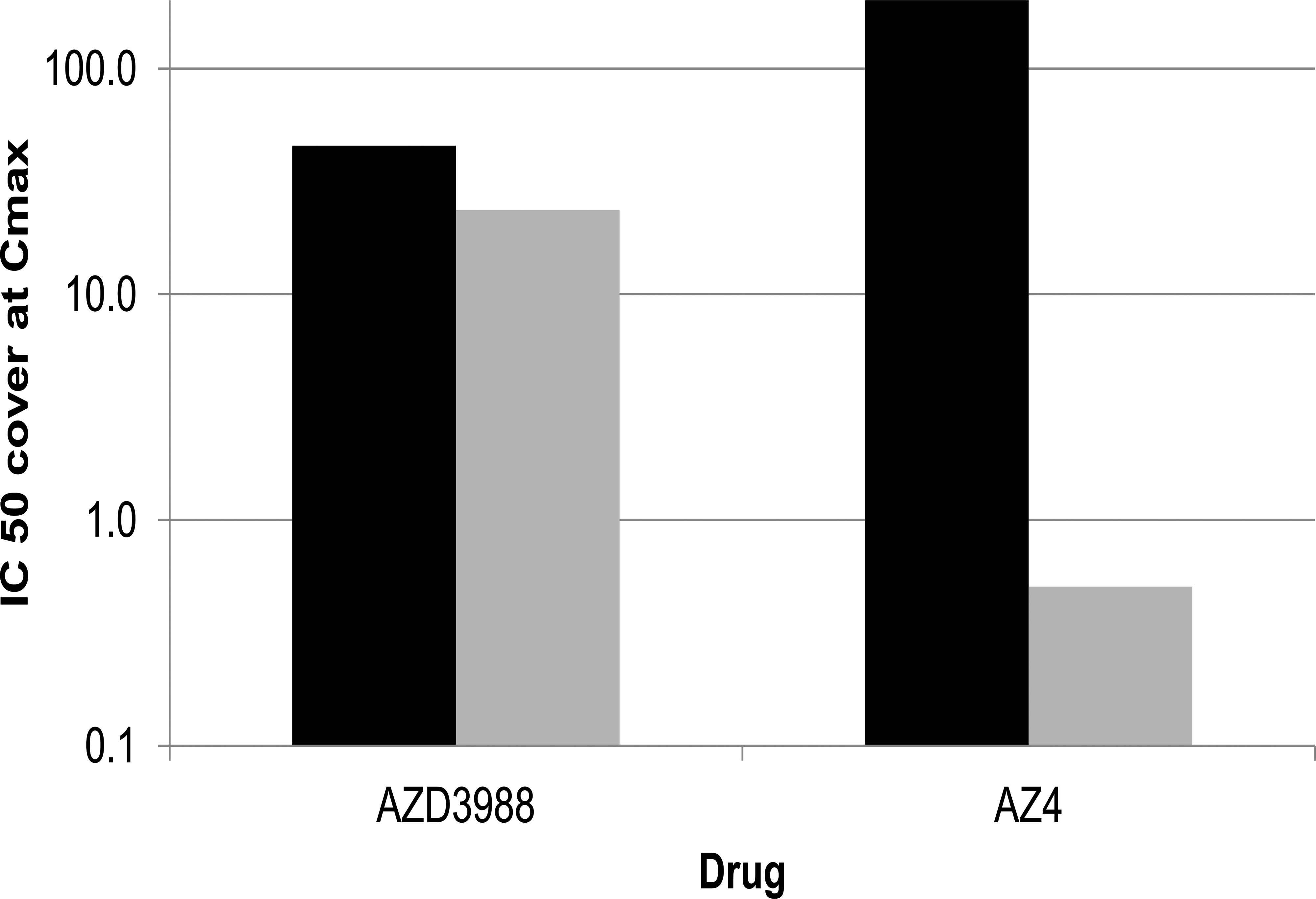
Exposures in mice given 100 mg/kg AZD3988 or 30 mg/kg AZ4 (twice daily [bid]; n = 2 per group) on day 14. The level of acyl-coenzyme A: diacylglycerol O-acyltransferase 1 (DGAT1) and acyl-coenzyme A: cholesterol O-acyltransferase (ACAT) inhibition is expressed as C max relative to the IC50 for DGAT1-mediated cellular triacylglycerol (TAG) synthesis (black column) and ACAT-mediated cellular TAG synthesis (gray column). Exposures of AZD3988 were sufficient to significantly inhibit both, DGAT1 and ACAT1, while exposures of AZ4 were only sufficient to inhibit DGAT1 but not ACAT1.
Following administration of AZD3988 or the more selective AZ4 to mice twice daily for up to 14 days, no significant effects of drug exposure on clinical observations, body weight, or food consumption were observed.
Histopathological examination of adrenal glands revealed a severe reduction in cortical adrenocyte fine vacuolation in all animals treated with AZD3988 for up to 14 days (Figure 8). Additionally, a mild to moderate cortical fatty degeneration characterized by a distinct midcortical band of degeneration/necrosis with substantial cellular vacuolation and mononuclear cell infiltration was also observed in adrenals of animals treated with AZD3988 for 7 days. Following 14 days of dosing, this finding was present in about half of the animals and was reduced to minimal severity. Most AZD3988-treated animals displayed mild cortical atrophy on day 14 and it is likely that this was a consequence of the earlier more severe cortical fatty degeneration and necrosis seen on day 7. Following treatment beyond 14 days, the histopathological appearance was generally restricted to the same severe reductions in cortical fine vacuolation (data not shown).
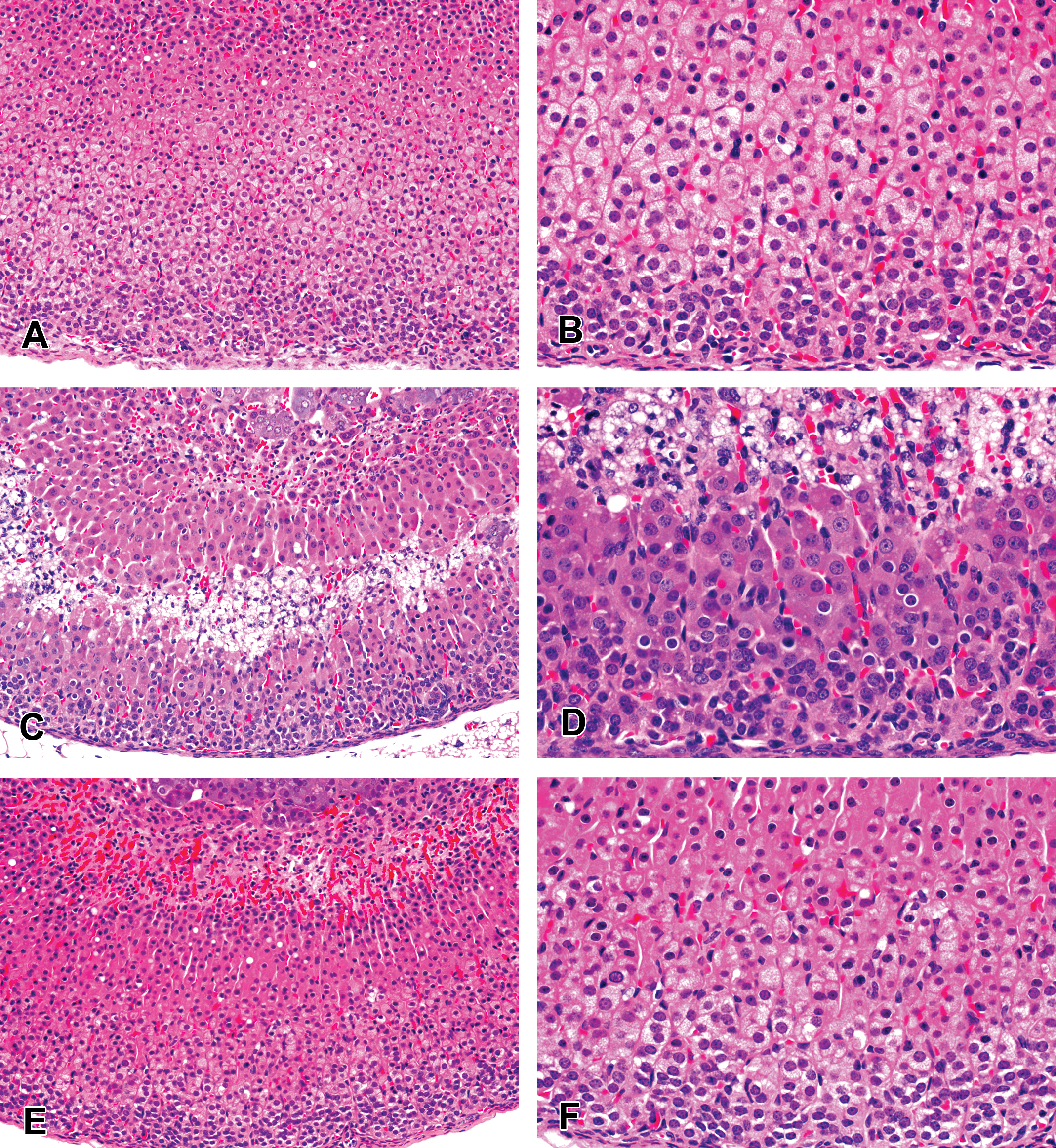
Photomicrographs of adrenal cortices from (A, B) control mice showing normal appearance; (C, D) mice given 100 mg/kg (twice daily [bid]) of AZD3988 for 7 days illustrating substantial reduction of cortical vacuolation with a central cortical band of cells undergoing fatty degeneration/ necrosis with associated inflammatory cell infiltration; (E, F) normal appearance of cortex from mice given 30 mg/kg (bid) AZ4 for 14 days. (H&E. Original objective lens magnifications A,C,E = ×20 and B,D,F = ×40).
No changes were seen in adrenals of animals treated with the DGAT1 selective AZ4. Despite significant variations in the amount of cholesteryl ester content in control mice, a marked decrease in mean cholesteryl ester levels was seen in AZD3988-treated mice (Figure 9). A time course investigation indicated that, relative to a control value of 0.8 µg cholesteryl ester per mg adrenal protein, a substantial reduction to 0.15 µg per mg had already occurred on day 3 with further, but smaller, decreases to 0.04 and 0.01 µg per mg on day 7 and 14, respectively. By contrast, cholesteryl ester levels were reduced to 0.4 µg per mg in animals treated with AZ4 for 14 days, with individual values at or below control values.
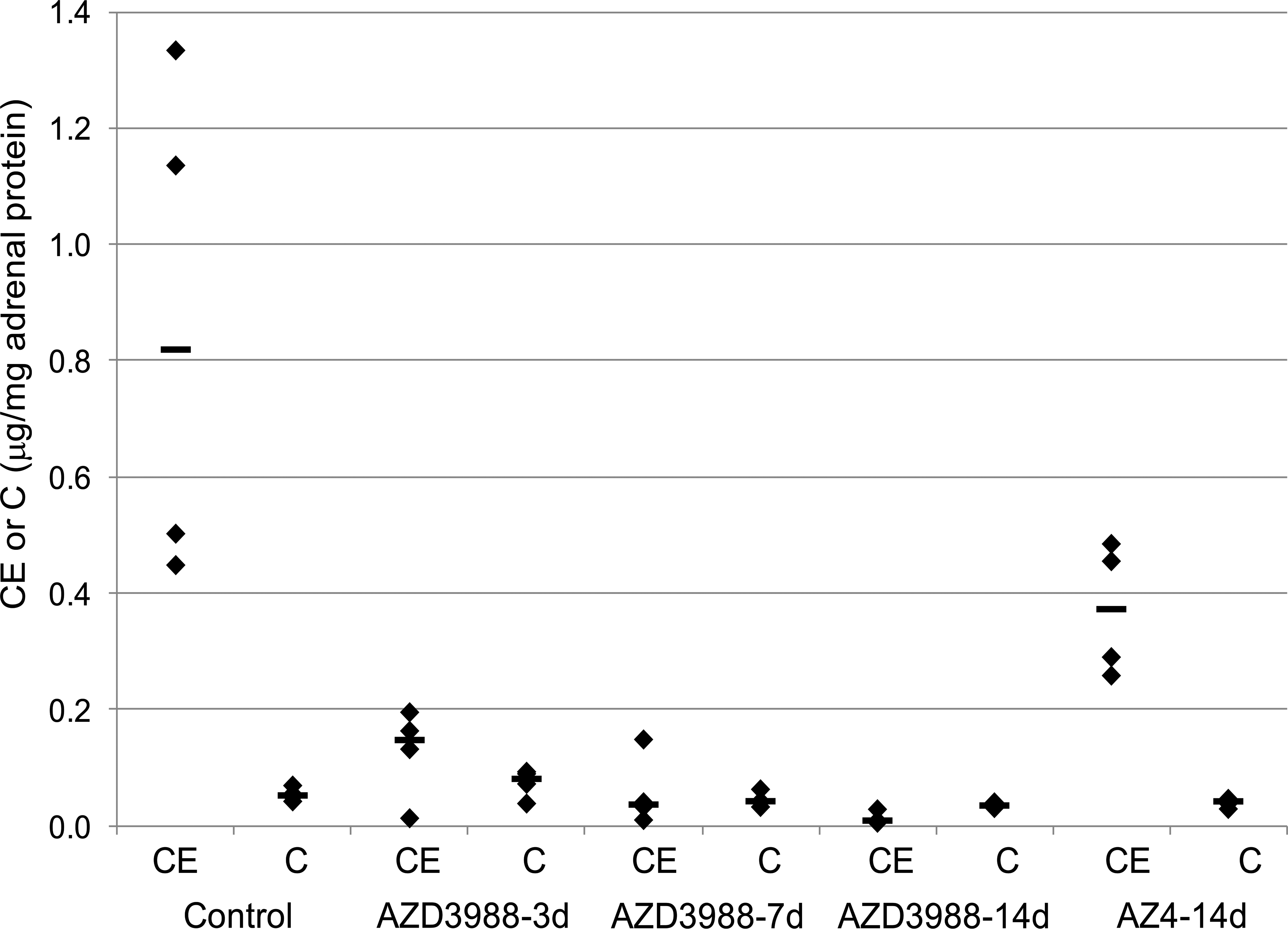
Adrenal cholesteryl ester (CE) and free cholesterol (C) levels in mice treated with 100 mg/kg AZD3988 or 30 mg/kg AZ4 for 3, 7, or 14 days (twice daily [bid]; n = 4 per group) showing a marked reduction in CE but only a minimal change in C following administration of AZD3988 after 3 days of treatment, with further slight reduction in CE at the 14 day time point. Administration of AZ4 did result in individual CE levels at or below those seen in controls.
Neither treatment with AZD3988 nor AZ4 had substantial effects on the low levels of free cholesterol levels.
Discussion
In pharmaceutical development, DGAT1 inhibitors are considered as potential treatments for type 2 diabetes and obesity (Yen et al. 2005, 2008; Zammit et al. 2008; Birch, Buckett, and Turnbull 2010). The DGAT1 inhibitor AZD3988 caused adrenocortical vacuolation of macrosteatotic appearance and degeneration with inflammation during toxicity testing in dogs. Interestingly, this lesion was similar to that induced by ACAT inhibitors in this species (Dominick et al. 1993; Wolfgang et al. 1995) suggesting little or no in vivo selectivity of this DGAT1 inhibitor over ACAT. In vitro enzyme assays for this drug indicated approximately 40-fold selectivity over the closest homologue ACAT1, but the in vitro cellular assays of TAG and cholesteryl ester synthesis, as cellular indicators of DGAT1 and ACAT activity, respectively, were supportive of a lack of ACAT selectivity of this drug. Due to a lack of bioavailability of the ACAT-selective DGAT1 inhibitor AZ4 in dogs, it was not possible to use this drug to deliver further evidence that ACAT, but not DGAT1 inhibition, is responsible for the observed adverse adrenal changes. However, in accord with similar toxicities having previously been observed following administration of ACAT inhibitors to dogs, it is likely that the observed adrenocortical toxicity related directly to pharmacological inactivation of ACAT. Since inhibition of ACAT1 as the main ACAT enzyme in adrenal glands (Lee et al. 2000; Meiner et al. 1997; Parini et al. 2004; Leon, Hill, and Kishor 2005) has been associated with adrenocortical toxicities, a lack of selectivity over ACAT1, rather than ACAT2, was the most probable cause of the observed toxicity, although it needs to be noted that the potencies for ACAT1 and ACAT2 inhibition of the drugs used in the experiments described here are not known for the different species tested. Biochemical analysis of adrenal cholesteryl ester and free cholesterol content demonstrated an AZD3988 dose-related reduction in cholesteryl ester concentrations in the adrenals of dogs. Although this reduction was not complete, it was indicative of in vivo inhibition of cholesteryl ester synthesis. An AZD3988 dose-related increase in free cholesterol was seen in dogs, indicating a reduced capability to store free cholesterol as inert cholesteryl ester at dose levels where ACAT inhibition would be significant. While it cannot be excluded that levels of cholesteryl ester and free cholesterol may have been impacted by the presence of degenerative and inflammatory changes in the dog adrenals, reduced cholesteryl ester and increased free cholesterol levels were clear biochemical indicators of ACAT inhibition.
In the marmoset, AZD3988 administered at a dose sufficient to cause significant DGAT1 and ACAT1 inhibition caused a reduction in cortical fine vacuolation, a substantial but nondegenerative change in adrenal morphology which was dissimilar to the adrenocortical vacuolation of macrosteatotic appearance associated with chelsterol clefts, degeneration, and inflammation seen in dogs. Importantly, following administration of the ACAT-selective DGAT1 inhibitor AZ4 at doses sufficient to significantly inhibit DGAT1, but not inhibit ACAT1, no pathological changes in the adrenals were seen in this species. This observation indicates that in the marmoset, DGAT1 inhibition alone does not result in adrenocortical changes and that the observed adrenal changes were most likely due to ACAT inhibition. This is supported by the biochemical evidence, where in the marmoset AZD3988 inhibited the formation of adrenal cholesteryl ester. Interestingly, there was no significant increase in free cholesterol level. The ACAT-selective DGAT1 inhibitor AZ4 did not affect cholesteryl ester or free cholesterol levels. It is probable that the reduced adrenal cortical fine vacuolation observed in the marmoset is indicative of a lack of adrenal cholesteryl ester as caused by the pharmacological inactivation of ACAT and thus represents a meaningful histological marker of ACAT inhibition in this species.
In the mouse, administration of AZD3988 resulted in the same distinctive decrease in adrenal cortical fine vacuolation which was observed in the marmoset, and had also been previously described for ACAT(−/−) mice, a change that has been described as indicative of inhibition of ACAT-mediated cholesteryl ester synthesis (Meiner et al. 1996). However, in somewhat similar manner to the adverse adrenocortical changes observed in dogs, AZD3988 also caused cortical fatty degeneration and necrosis in the mouse, but in contrast, these changes appeared to be transient and less severe. Biochemical analysis of adrenal glands from mice treated with AZD3988 at doses resulting in significant DGAT1 and ACAT inhibition, but not from mice treated with AZ4 at doses resulting in DGAT1 inhibition only, confirmed a marked reduction of cholesteryl ester, lending further support to the hypothesis that the observed reduction in adrenal fine vacuolation was linked to a reduction in cholesteryl ester synthesis and thus to the pharmacology of ACAT inhibition. A time course investigation indicated that most of this effect had already occurred within 3 days of AZD3988 administration and remained at significant levels throughout the 14-day duration of the experiment. Interestingly and as already observed in the marmoset following administration of AZD3988 or AZ4, there was no significant increase in free cholesterol level in the mouse at any of the time points measured.
A comparison between the 3 species revealed significant differences in adrenal histopathological appearances resulting from non-ACAT-selective DGAT1 inhibition in the mouse and marmoset by comparison to the dog. While the histopathological changes in dogs were the most marked, the only adrenal histopathological change observed in the marmoset was that of decreased cortical fine vacuolation, indicating that the dog is the most sensitive species for the detection of ACAT inhibition–mediated degenerative adrenal changes, followed by the mouse which showed only transient cortical degenerative changes as well as the nondegenerative reduction in cortical fine vacuolation. A similar finding of adrenocortical cells containing little lipid in the zona fasiculata following ACAT inhibition has been reported previously in the rat (Kaneko et al. 2001), but the detection of changes in the rat appeared difficult since they were only seen in a fraction of animals and only at doses that were not well tolerated, an observation that reflects our experience. It is clear that an accurate assessment of adrenal toxicity during the development of non-ACAT-selective drugs in rat and marmoset would be insufficient: evaluation in the mouse and dog would deliver the greatest confidence that such potential drugs do not pose a significant risk following administration to human. As long as the underlying reason for the differences in pathological responses to ACAT inhibition across the species tested—and thus its relevance to man—is unknown, safety margins in the most sensitive species (i.e., dogs) will need to be considered during the clinical development of ACAT inhibitors. In drug development programs, where the marmoset is chosen as the nonrodent toxicity species, the investigators need to assume that a nondegenerative reduction in cortical fine vacuolation has the potential to translate into a more severe and degenerative adrenal toxicity in the dogs and potentially humans. Alternative explanations for the observed ACAT inhibition–mediated changes and cross-species differences, such as species differences in potency or bioavailability of the drugs evaluated in our experiments, are unlikely since we have not seen any evidence supportive of such hypotheses. A plausible explanation for the observed interspecies differences lies in adrenal cholesterol metabolism, and histopathology is a difference in control or activity of cholesterol biosynthesis (Matsuo et al. 1996) and/or differences in regulation in more general lipid synthetic pathways (Dove et al. 2005). Such differences could permit the mouse, rat, and monkey to shunt excess nonesterified cholesterol, which could accumulate following the inhibition of cholesterol esterification by ACAT, into other pathways, thereby limiting the accumulation of toxic free unesterified cholesterol. By contrast, dogs may be less efficient in the elimination of unesterified cholesterol when ACAT is inhibited. This would explain the increased free cholesterol and the adrenocortical toxicity, which may be a consequence of excessive accumulation of free cholesterol. The relative to mouse and marmoset slight reduction in adrenal cholesteryl ester concentrations in the dog might also be a consequence of the inability to effectively eliminate unesterified cholesterol during ACAT, all of which would contribute to the adrenocortical vacuolation of macrosteatotic appearance observed in this species.
It is important to note that, using a significantly more selective DGAT1 inhibitor, we demonstrated that in the marmoset and the mouse, substantial inhibition of DGAT1 alone did not cause any adrenocortical changes. Clearly, if this also translates to dog or human, high selectivity over ACAT is essential in the design of DGAT1-inhibiting drugs. It was not possible to achieve significant exposures of the ACAT-selective DGAT1 inhibitor AZ4 in the dog to provide further evidence for this view. However, ACAT-selective DGAT1 inhibitors with no adrenocortical toxicopathology in dogs have subsequently been developed in our laboratory using the mouse as a predictive in vivo model to mitigate the risk of ACAT-mediated adrenal toxicities.
In conclusion, significant evidence indicated that ACAT but not DGAT1 inhibition causes significant adverse adrenocortical changes in dogs (i.e., adrenocortical vacuolation of macrosteatotic appearance and degeneration with inflammation), the same adrenocortical degenerative pathological changes that have been reported in the development of ACAT inhibitors. A cross-species comparison indicated that in the marmoset inhibition of ACAT, but not DGAT1, resulted in a distinctive nondegenerative reduction in cortical fine vacuolation, but not the adverse degenerative changes seen in dogs. In the mouse, ACAT inhibition–mediated transient degenerative changes as well as the nondegenerative reduction in cortical fine vacuolation were observed, making the mouse an ideal in vivo model to assess the risk of ACAT-mediated adrenal toxicities at early stages in drug development, where compound availability is limited.
Footnotes
Acknowledgments
The authors would like to acknowledge all their colleagues within AstraZeneca who have contributed to this research.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
