Abstract
The detection of drug-induced hepatotoxicity remains an important safety issue in drug development. A liver-specific microRNA species, microRNA-122 (miR-122), has recently shown potential for predicting liver injury in addition to the standard hepatic injury biomarkers. The objective of this study was to measure miR-122 together with several other liver markers in distinct settings of acute liver toxicity in rats to determine the value of miR-122 as a biomarker for liver injury in this species. Rats were exposed to 3 well-established liver toxicants (acetaminophen, allyl alcohol, and α-naphthyl isothiocyanate), a liver-enzyme inducer (phenobarbital), or a cardiotoxicant (doxorubicin). There was a clear increase in plasma miR-122 following administration of acetaminophen, allyl alcohol, and α-naphthyl isothiocyanate. The response of miR-122 paralleled that of other markers and was consistent with liver injury as indicated by histopathological evaluation. Furthermore, the changes in miR-122 were detected earlier than standard liver injury markers and exhibited a wide dynamic range. In contrast, miR-122 responses to phenobarbital and doxorubicin were low. Based on these findings, miR-122 shows significant promise and may provide added value for assessing liver toxicity in drug development.
Introduction
The detection of drug-induced hepatotoxicity remains an important safety issue and laboratory investigations for monitoring drug-induced liver injury include measuring a combination of hepatocellular and cholestatic markers because both injuries may occur simultaneously. Alanine aminotransferase (ALT) and bilirubin are the principal preclinical and clinical biomarkers driving the diagnosis of drug-induced liver injury by Hy’s law (Ozer et al. 2008, 2010; Ramaiah 2007). Although ALT elevation is a sensitive and relatively specific biomarker for liver injury, current preclinical testing regimens do not always successfully correlate with adverse events in the human liver (Assis and Navarro 2009; Chang and Schiano 2007). In several instances during preclinical studies, ALT elevations occur in the absence of any observed histopathology, making interpretation challenging. Clearly, new biomarkers are needed to improve the general as well as mechanism-specific assessment of hepatotoxicity.
Specific circulating microRNAs (miRNAs or miRs) have recently emerged as sensitive and specific biomarkers for tissue injury (Laterza et al. 2009; Wetmore et al. 2010). miRNAs are short, noncoding RNA molecules of approximately 18 to 23 nucleotides in length. They regulate gene expression either by translational repression or affecting messenger RNA (mRNA) stability. Circulating miRNAs are potentially attractive biomarkers for many reasons; miRNAs are evolutionarily conserved, can be readily obtained from preclinical species and patients, are extremely stable in blood (Mitchell et al. 2008), and can be easily and quantitatively measured via simple and sensitive assays, such as polymerase chain reaction (PCR). Some miRNAs are differentially expressed and may be highly enriched in certain tissues or demonstrate limited tissue distribution (Kerr, Korenblat, and Davidson 2011; Lewis and Jopling 2010). In particular, miR-122 is highly expressed in the liver and accounts for 70% of the total liver miRNA (Wang et al. 2009; Li et al. 2011).
miR-122 has been shown to parallel increased serum aminotransferase levels in a mouse model of acetaminophen overdose-induced liver injury (Wang et al. 2009). Another study reported changes in urinary miRNA profiles after acute administration of acetaminophen in rats (Yang et al. 2012). Recently, evidence was also provided for the potential use of miRNAs as biomarkers of acute liver injury in humans (Zhang et al. 2010; Lewis et al. 2011). Serum miR122 was substantially higher in patients with acetaminophen-induced acute liver injury, compared to healthy controls.
The purpose of this work was to address the potential utility of miR-122 as a marker of liver injury in the rat, the main preclinical species for safety assessment, and to further assess whether the cellular release of miR-122 is associated with specific types of liver injury. For this purpose, Sprague-Dawley rats were exposed to four well-characterized compounds associated with different types and mechanisms of liver toxicity. Allyl alcohol and acetaminophen are associated with periportal (Atzori, Dore, and Congiu 1989) and centrilobular (Amar and Schiff 2007) hepatocellular necrosis, respectively, α-naphthyl isothiocyanate (ANIT) with bile duct damage and intrahepatic cholestasis (Orsler et al. 1999), and phenobarbital with hepatocellular hypertrophy (Crampton et al. 1977). In addition, the cardiotoxicant doxorubicin was used to assess specificity (Robert 2007). Time points of blood sampling were incorporated to monitor early and overt toxicity. Circulating miR-122 was analyzed in plasma and compared to standard markers such as ALT, histopathology, and additional markers like aspartate aminotransferase (AST), glutamate dehydrogenase (GLDH), sorbitol dehydrogenase (SDH), alpha-glutathione-S-transferase (α-GST), liver-fatty acid–binding protein (L-FABP), alkaline phosphatase (ALP), total bilirubin, conjugated bilirubin, and γ-glutamyl transferase (GGT). Several markers included in this study have been used previously for specific injury assessments such as liver structure and/or function. α-GST was included since a new and robust assay is now available and the marker is being reconsidered.
Materials and Methods
Statement of Ethical Approval
All animal experiments were performed in accordance with the European (ETS No. 123 2007) and Belgian (Belgian Law 1991) guidelines, and with the principles of euthanasia as stated in the “AVMA Guidelines on Euthanasia” (AVMA 2007). The experiment was approved by the Janssen Research & Development Ethical Committee and conducted in an AAALAC-approved facility.
Animals
Forty-two healthy, approximately 12-week-old, male Sprague-Dawley rats were supplied by Charles River Laboratories (Germany) and acclimatized for 1 week before the start of the experiment. Animals were group housed (four or six rats/cage) in polysulfone cages with a wire mesh roof under routine conditions of temperature, relative humidity, ventilation, and illumination. A pelleted maintenance diet (Ssniff, Germany) was available ad libitum, with free and continuous access to drinking water. Prior to blood sampling and terminal kill, animals had access to water but were fasted (20 hr ± 30 min).
Study Design
Prior to the start of treatment, the rats were randomly allocated to the vehicle (three groups of four rats each) or compound-dosed groups (five groups of six rats each). Dose levels were selected based on published literature. Animals were administered a single dose of either 35 mg/kg allyl alcohol (Sigma-Aldrich, Germany) in saline intraperitoneally (IP; Atzori, Dore, and Congiu 1989), 1,000 mg/kg acetaminophen (Sigma) in 0.5% Methocel (f4m premium hydroxypropyl methylcellulose) orally (Amar and Schiff 2007), 100 mg/kg ANIT (Sigma) in olive oil orally (Orsler et al. 1999) or 20 mg/kg doxorubicin in saline IP (Sigma, Sigma-Aldrich, Germany; Robert 2007); four doses of 100 mg/kg phenobarbital (Sigma) in saline IP on 4 consecutive days (Crampton et al. 1977); or respective vehicles according to the details in Table 1. Injection volume for each treatment or vehicle control was 10 ml/kg body weight. Clinical observations were performed daily and body weight was recorded prior to dosing and sacrifice. Blood was collected from the sublingual veins after anesthesia with isoflurane 6 hr after the (first) dose and immediately before necropsy. Necropsy was performed 24 hr after single dosing, except for doxorubicin-treated animals that were sacrificed 72 hr after single dosing, and phenobarbital-treated rats that were euthanized 96 hr after the first dose.
Study design.
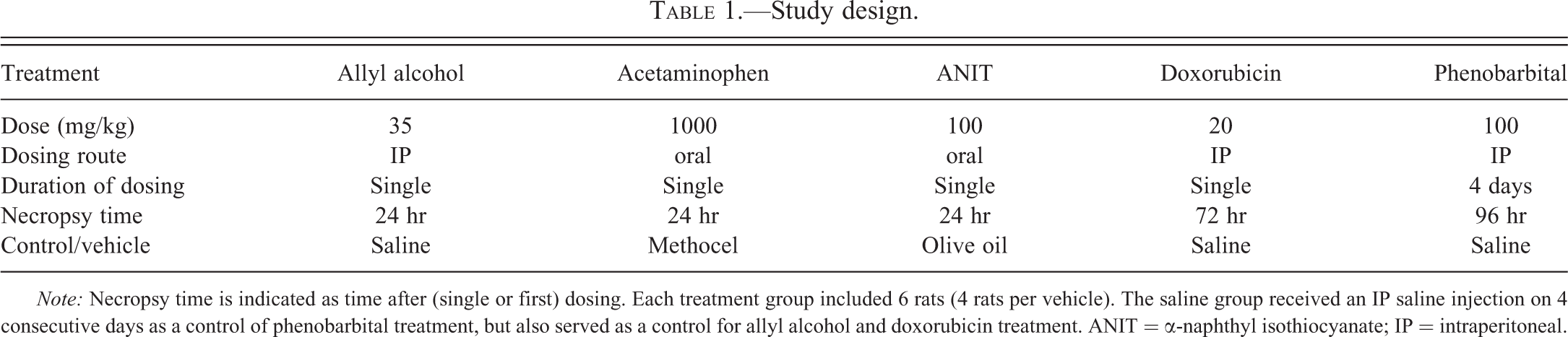
Note : Necropsy time is indicated as time after (single or first) dosing. Each treatment group included 6 rats (4 rats per vehicle). The saline group received an IP saline injection on 4 consecutive days as a control of phenobarbital treatment, but also served as a control for allyl alcohol and doxorubicin treatment. ANIT = α-naphthyl isothiocyanate; IP = intraperitoneal.
Serum Biomarkers
Serum ALT, AST, ALP, GGT, GLDH, and bilirubin (conjugated and unconjugated) were analyzed on a Hitachi Modular automated clinical chemistry analyser (Roche Diagnostics, Germany) using Roche reagent kits. SDH activity was measured on the same instrument using an assay from Diagnostics Chemicals Limited (DCL, Canada). A rat-specific enzyme immunoassay was used for the detection of L-FABP (Hycult Biotech, PA, USA) and α-GST was measured using an electrochemiluminescence assay with the Argutus AKI Test® on a SECTOR™ Imager 6000 (Meso Scale Diagnostics, LLC, Meso Scale Discovery, MD, USA).
miRNA Isolation from Plasma
miRNA from plasma was isolated using the miRNeasy kit (Qiagen, Germany) together with the RNeasy MinElute Cleanup kit (Qiagen). Briefly, 700 µl of Qiazol reagent was added to 200 µl plasma, the sample was vortexed thoroughly before 3 µl of a spiked-in Arabidopsis thaliana (ath) miRNA control (Integrated DNA technologies sequence 5’-rUrUrU rGrGrA rUrUrG rArArG rGrGrA rGrCrU rCrUrA-3) at a concentration of 1 nM, and 140 µl chloroform were added. After thorough vortexing, the sample was centrifuged at 12,000g for 15 min. The upper, aqueous phase was transferred to a new collection tube and 1 volume of 70% ethanol was added. The sample was applied to an RNeasy mini spin column and the supernatant collected. Then, 0.65 volumes of 100% ethanol were added and the sample applied to an RNeasy MinElute spin column. The miRNA was bound and cleaned using the manufacturer’s protocol and buffers. The miRNA was eluted with RNase-free water and the quantity and quality of miRNA was evaluated using the small RNA kit on the Agilent 2100 Bioanalyzer (Agilent 2100 Bioanalyzer, CA, USA). The efficiency of the miRNA isolation was monitored by the amount of recovered spiked-in miRNA using reverse transcription–polymerase chain reaction (RT-PCR).
Real-time Quantitative RT-PCR Analysis of miR-122
The expression levels of miRNA were confirmed using Applied Biosystem’s TaqMan® MicroRNA Reverse Transcription Kit using 5 µl miRNA. Complementary DNA (cDNA) was generated using individual miRNA-specific primers (Applied Biosystems). The level of specific miRNA was then monitored with the 7900HT real-time PCR system from Applied Biosystems. Ct-values were determined using the miR-122 specific and the ath spiked-in sequence-specific probe (ath-miR159a). For every sample within every exposed group, the fold change to the median vehicle sample was calculated using the comparative Ct method (Livak and Schmittgen 2001).
Pathology
Necropsy was performed on all animals with macroscopic evaluation of the thoracic and abdominal cavity and tissues. After weighing, samples of liver and heart were snap frozen in liquid nitrogen and stored at −80°C until further processing. Tissue samples for histology were fixed in 10% phosphate-buffered formalin and embedded within 48 hr in paraffin. Sections were stained with hematoxylin–eosin (H&E). Histological examination was performed on liver and on a subset of hearts (1/group; hearts of other rats were used for method development—not further specified) by an experienced pathologist.
Statistical Analysis
Biomarker data were expressed as means ± standard error (SE). The significance of differences between the compound-dosed groups (six rats per treatment) and their respective vehicle groups (four rats per vehicle) was assessed by the Mann-Whitney U test for pairwise comparison, according to Siegel (two-tailed with correction for ties). Fold changes of biomarkers’ results were expressed versus the respective controls. Biomarkers were graphically analyzed using log-transformed values. Liver weights were analyzed by Dunn’s test (for comparison of more than 2 groups) or Wilcoxon’s test (for comparison of 2 groups). Values significantly different from vehicle are indicated as *p < .05 and **p < .01. Biomarker changes were graded as not observed (no change), minimal, mild, moderate, or marked based on the fold change of the biomarker versus the concurrent control (except for GGT). Based on in-house historical control data, biological variation, and/or statistical significance, the criteria used for grading were set as follows: miR-122 (<4 = not observed; 4–19.9 = minimal; 20–99.9 = mild; 100–249 = moderate; and >250 = marked); ALT, AST, a-GST, GLDH, SDH, and L-FABP (<1.3 = not observed; 1.3–1.49 = minimal; 1.50–2.99 = mild; 3–8.99 = moderate; and >9 = marked), ALP (<1.5 = not observed; 1.50–1.99 = minimal; 2–3.99 = mild; 4–10.99 = moderate; and >11 = marked), and total bilirubin (<1.40 = not observed; 1.40–1.99 = minimal; 2–5.99 = mild; 6–22.99 = moderate; and >23 = marked). Interpretation of GGT data was based on its absolute values (<3.0 = not observed; 3.0–3.9 = minimal; 4.0–7.99 = mild; 8.0–11.9 = moderate; and >12 = marked).
Results
Circulating miR-122 Increases after Exposure to Allyl Alcohol, Acetaminophen, and ANIT and Correlates to the Severity of Hepatocyte Necrosis
Rats were treated with a single dose of allyl alcohol (35 mg/kg IP), acetaminophen (1,000 mg/kg orally [PO]), ANIT (100 mg/kg PO), or their respective vehicles and sacrificed 24 hr later. The levels of circulating miR-122 and other biomarkers were analyzed 6 hr post-dosing and just prior to sacrifice. Typical photomicrographs of the liver histopathology sections are presented in Figure 1. Animals receiving allyl alcohol showed an increased mean liver weight (+43.4%, p < .01) associated with moderate-to-severe hepatocellular necrosis (Figure 1A). The distribution of necrosis was mainly periportal and subcapsular, with bridging and areas of diffuse necrosis in some animals. In comparison to the vehicle group, circulating miR-122 levels were substantially higher in allyl alcohol–treated rats with a mean fold change value of 363 ± 146 (p < .05) at necropsy. Acetaminophen-treated rats showed slight-to-moderate hepatocellular centrilobular single-cell necrosis and/or apoptosis, with one rat exhibiting larger areas of centrilobular necrosis (Figure 1B–C) and mean liver weight was slightly increased (11.1%). At necropsy, levels of circulating miR-122 in acetaminophen-treated rats were 147 ± 49 fold higher (p < .05) in comparison to the vehicle group. ANIT administration resulted in a 9.9% increase in the mean liver weight, and all animals showed moderate-to-marked bile duct necrosis (Figure 1D), characterized by multifocal partial to complete necrosis of the bile duct epithelium. The mean fold change values of plasma miR-122 by 24 hr after single exposure to ANIT was 52 ± 9 (p < .05).
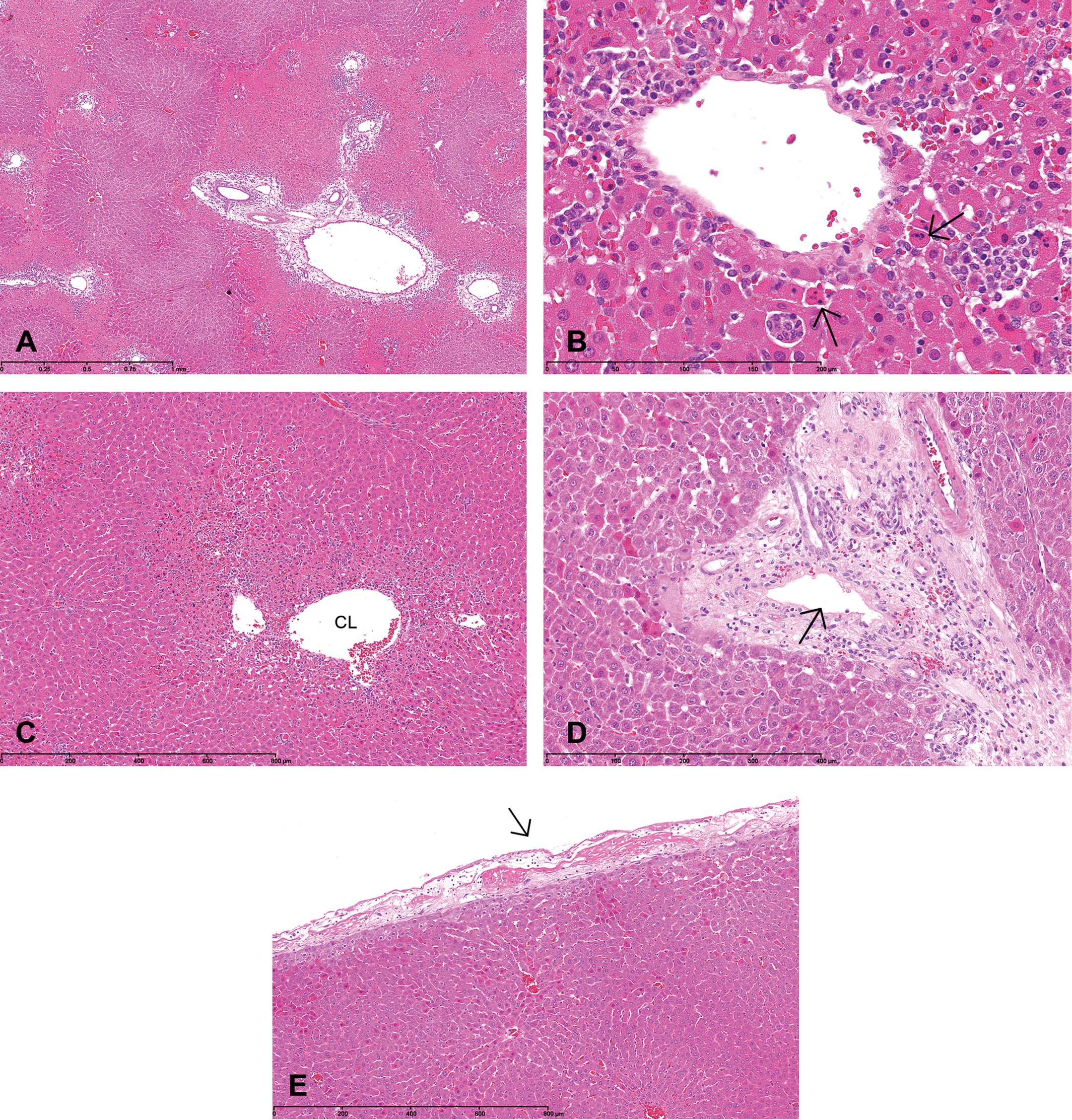
Histological features of liver sections stained with hematoxylin eosin (H&E). A, Allyl alcohol (single intraperitoneal [IP] dose at 35 mg/kg); periportal hepatocellular bridging necrosis. Original magnification: 5×. B, C, Acetaminophen (single oral dose at 1000 mg/kg); hepatocellular apoptosis (B, arrow), and more extensive centrilobular necrosis (C), associated with slight subacute inflammation. CL = centrilobular vein. Original magnifications B and C: 40× and 10×, respectively. D, α-naphthyl isothiocyanate (ANIT, single oral dose at 100 mg/kg); bile duct necrosis resulted in loss of bile duct epithelium (arrow); this was associated with acute portal inflammation. Original magnification: 20×. E, Intraperitoneal injection of doxorubicin (single IP dose at 20 mg/kg) resulted in peritonitis (arrow). Original magnification: 10×.
Administration of phenobarbital at 100 mg/kg IP for 4 consecutive days resulted in adaptive liver changes characterized by a mean liver weight increase of 35.5% (p < .05), which correlated with slight centrilobular hepatocellular hypertrophy observed at histology. The levels of plasma miR-122 in phenobarbital-treated rats were not different from their vehicle control group (mean fold change value was 1.44 ± 0.79 prior to necropsy). All but one rat that received a single IP dose of 20 mg/kg of doxorubicin showed peritonitis and necrosis of the abdominal fat (Figure 1E), considered the result of the IP administration of the formulation. This possibly resulted in a mean fold change value of plasma miR-122 of 2.5 ± 0.9 at necropsy, although a fold change of 16.4 ± 3.7 was measured at 6 hr after doxorubicin dosing. There was no evidence of liver injury based on histopathology. It is possible that the interim increase in miR-122 in these animals was due to a transient change in the liver resulting from the peritonitis and necrosis observed in the abdominal fat. There were no relevant heart findings in the examined hearts (exposure not determined; might be related to short exposure time or to lower exposure than anticipated). Individual results of circulating miR-122 levels following treatment with different compounds are shown in Figure 2.
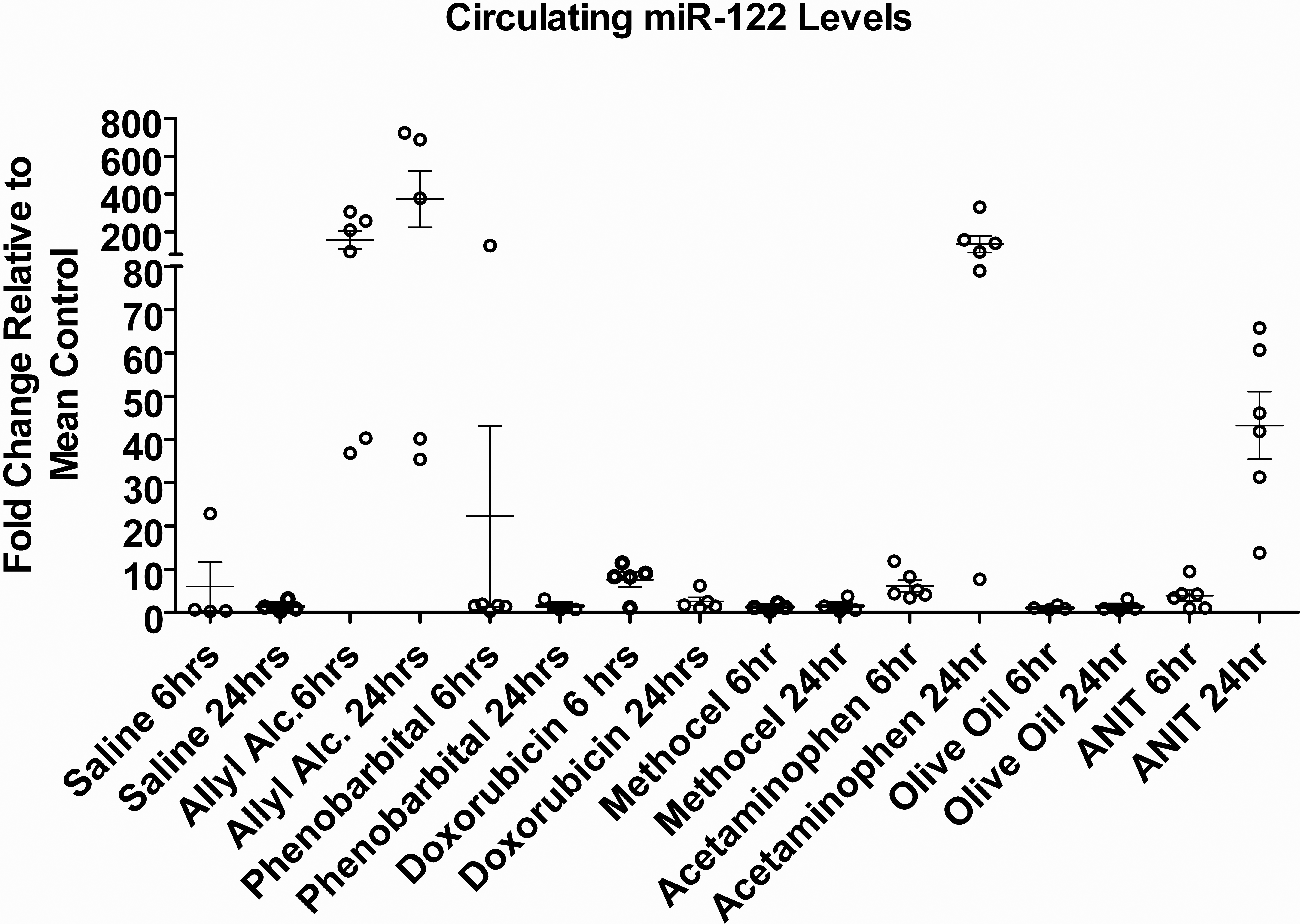
Circulating microRNA-122 (miR-122) levels following treatment with different compounds. Individual results are presented with indication of mean and standard error bar. Each treatment group included 6 rats (4 rats per vehicle).
Circulating miR-122 Parallels ALT, AST, GLDH, SDH, and α-GST
Circulating miR-122 was measured along with ALT, AST, GLDH, SDH, α-GST, L-FABP, ALP, bilirubin, and GGT. Several of these biomarkers have been described previously as biomarkers of liver toxicity (Giffen et al. 2002; O’Brien et al. 2002; Ozer et al. 2008; Pelsers et al. 2002, Pelsers, Hermens, and Glatz 2005). Historically, α-GST measurements by enzyme-linked immunosorbent assay (ELISA) have been unreliable due to limitations of the assays in use. However, the α-GST assay used in this study is different and was qualified prior to use, yielding reliable data.
Allyl alcohol resulted in moderate to marked increases in the majority of hepatocellular markers in all rats (ALT × 153.3 ± 49.0, AST × 60.6 ± 20.4, GLDH × 152.2 ± 18.5, SDH × 146.7 ± 37.4, α-GST × 17.9 ± 5.0, L-FABP × 18.6 ± 5.8; Table 2) and also hepatobiliary markers were largely affected given the extent of damage (bilirubin × 5.9 ± 2.5; GGT 6.7 ± 2.5 U/L as compared to 0.0 ± 0.0 U/L in the vehicle group; Table 2). The lower-grade hepatocellular injury present in the acetaminophen-treated animals was consistent with a moderate increase in miR-122, also reflected in other liver biomarkers. Whereas miR-122 was clearly increased in all animals, ALT and AST were normal (2/6 rats), minimally (3/6 rats; 2-fold), or moderately (1/6 rats; >3-fold) increased (Figure 3). Interestingly, fold changes in miR-122 (individual fold changes of 8 and 86) were lower in animals with an ALT in the normal range and higher in rats with a minimal (individual fold change of 152, 103, and 172) or moderate (individual fold change of 361) increase in ALT. Although histological evidence of necrosis was present in all acetaminophen-treated animals, the animal exhibiting larger areas of centrilobular necrosis also presented the largest increase in miR-122 and ALT. GLDH and SDH were clearly increased in 5 out of 6 rats (Figure 3), whereas L-FABP (× 8.3 ± 1.2) and α-GST (× 15.3 ± 2.6) were elevated in all rats (Table 2, Figure 3). In the ANIT group, the increases in miR-122 in all rats paralleled increases in ALT (× 2.7 ± 0.3), α-GST (× 9.0 ± 1.8), GLDH (× 23.4 ± 4.5), total bilirubin (× 19.4 ± 5.5), and L-FABP (× 7.0 ± 0.9) in all but one treated rat (Table 2, Figure 3). As expected, there were no relevant changes in serum parameters of the rats in the phenobarbital group. In the doxorubicin group, all but one rat showed changes in ALT (× 2.3 ± 0.3) which were considered to be related to the peritonitis as described earlier (Table 2, Figure 3).
Clinical chemistry changes by category of injury.
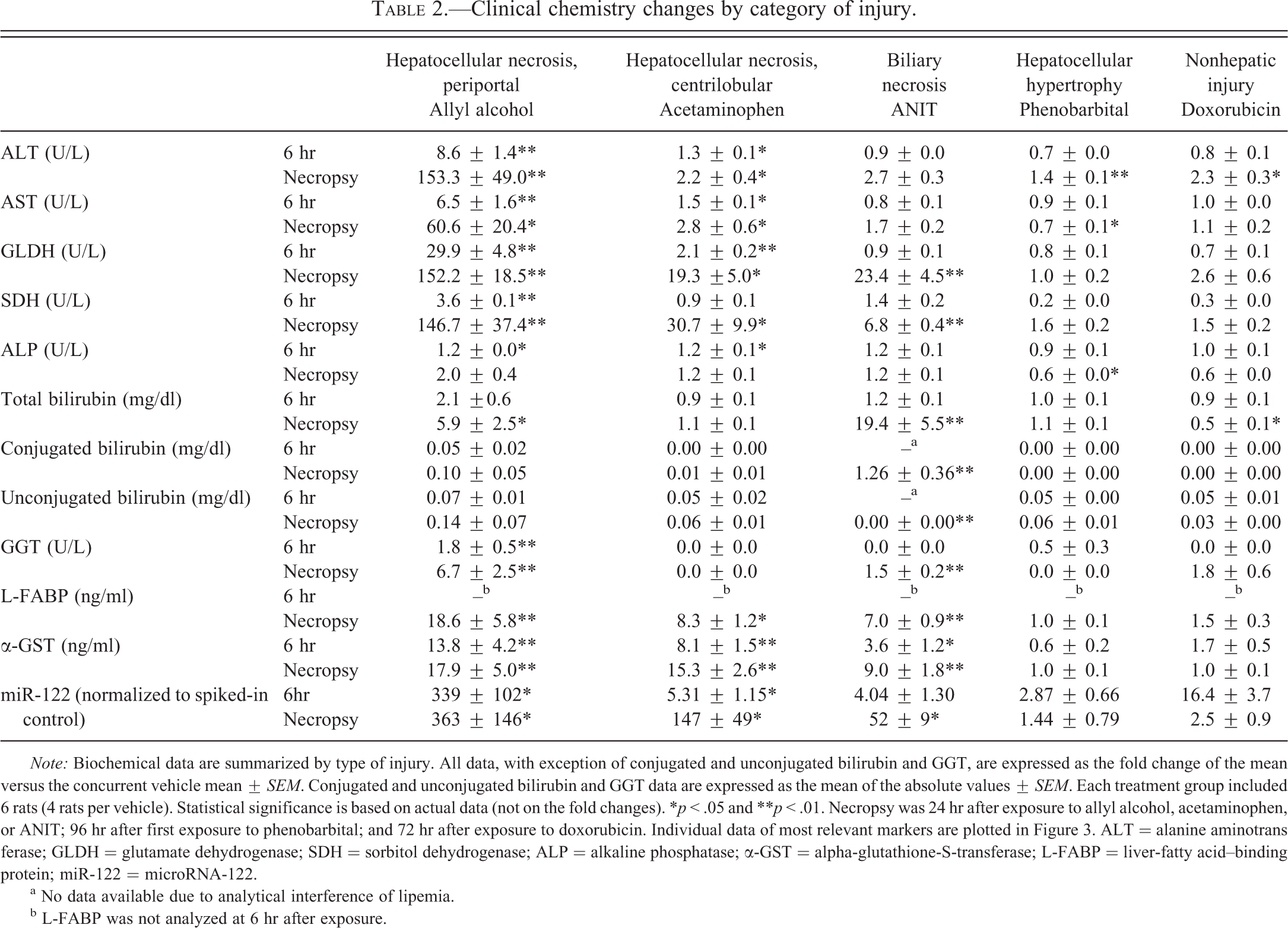
Note: Biochemical data are summarized by type of injury. All data, with exception of conjugated and unconjugated bilirubin and GGT, are expressed as the fold change of the mean versus the concurrent vehicle mean ± SEM. Conjugated and unconjugated bilirubin and GGT data are expressed as the mean of the absolute values ± SEM. Each treatment group included 6 rats (4 rats per vehicle). Statistical significance is based on actual data (not on the fold changes). *p < .05 and **p < .01. Necropsy was 24 hr after exposure to allyl alcohol, acetaminophen, or ANIT; 96 hr after first exposure to phenobarbital; and 72 hr after exposure to doxorubicin. Individual data of most relevant markers are plotted in Figure 3. ALT = alanine aminotransferase; GLDH = glutamate dehydrogenase; SDH = sorbitol dehydrogenase; ALP = alkaline phosphatase; α-GST = alpha-glutathione-S-transferase; L-FABP = liver-fatty acid–binding protein; miR-122 = microRNA-122.
a No data available due to analytical interference of lipemia.
b L-FABP was not analyzed at 6 hr after exposure.
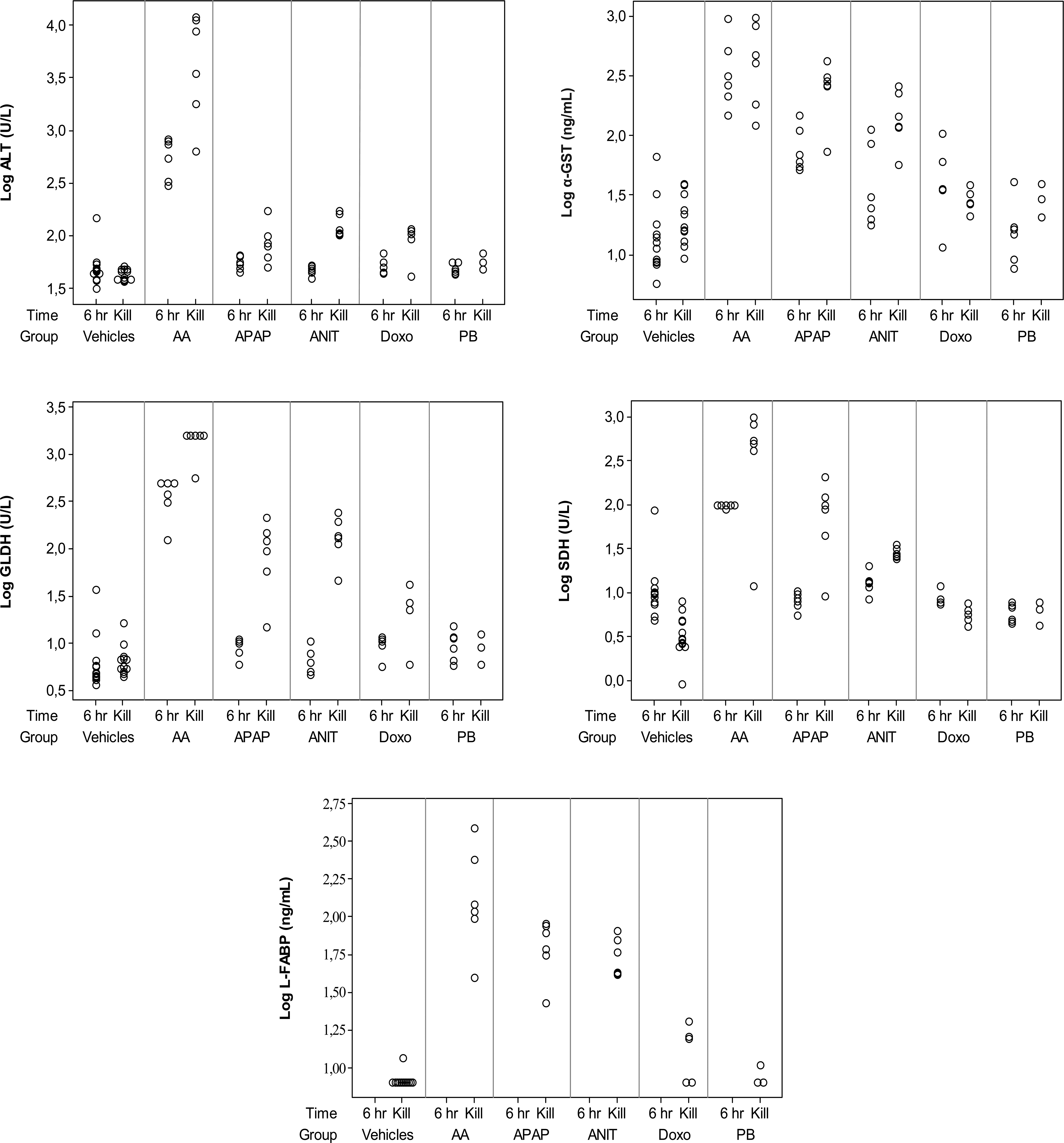
Individual value plots of ALT, GLDH, SDH, α-GST, and L-FABP. L-FABP was not analyzed at 6 hr after exposure. Log-transformed values are represented. Sacrifice is 24 hr after exposure to allyl alcohol (AA), acetaminophen (APAP), or ANIT; 96 hr after first exposure to phenobarbital (PB); and 72 hr after exposure to doxorubicin (Doxo). Each treatment group included 6 rats. Vehicle data were pooled in this figure (3 × 4 rats). ALT = alanine aminotransferase; GLDH = glutamate dehydrogenase; SDH = sorbitol dehydrogenase; α-GST = alpha-glutathione-S-transferase; L-FABP = liver-fatty acid–binding protein.
Circulating miR-122 Is an Early Marker of Liver Injury
Early changes in biomarkers were assessed by analyzing the plasma samples collected at 6 hr post-dosing. In allyl alcohol–treated rats, all measured hepatocellular biomarkers, in addition to miR-122, were already elevated at 6 hr indicating the induction of toxicity (Table 2, Figure 3). In acetaminophen and ANIT-treated rats, the 6-hr time point changes coincided with minimal to moderate increases in miR-122 and α-GST only. miR-122 was increased by 5.31 ± 1.15 (p < .05) and α-GST levels were approximately 8-fold higher (p < .01) at 6 hr in the acetaminophen group (Table 2). At 6 hr after exposure to ANIT, miR-122 increased by 4.04 ± 1.30-fold (p > .05) and α-GST increased by 3.6- ± 1.2-fold (p < .05). There were no changes in ALT, AST, GLDH or SDH at 6 hr in acetaminophen- or ANIT-treated animals (L-FABP was not analyzed at 6 hr after exposure). Early biomarker changes were absent after 6 hr exposure to phenobarbital or doxorubicin, with the exception of a transient increase in miR-122 after IP administration of doxorubicin. The data suggest that circulating miR-122 and α-GST are possibly early markers of liver injury. Figure 4 summarizes the multiple end points in a heat map representation.
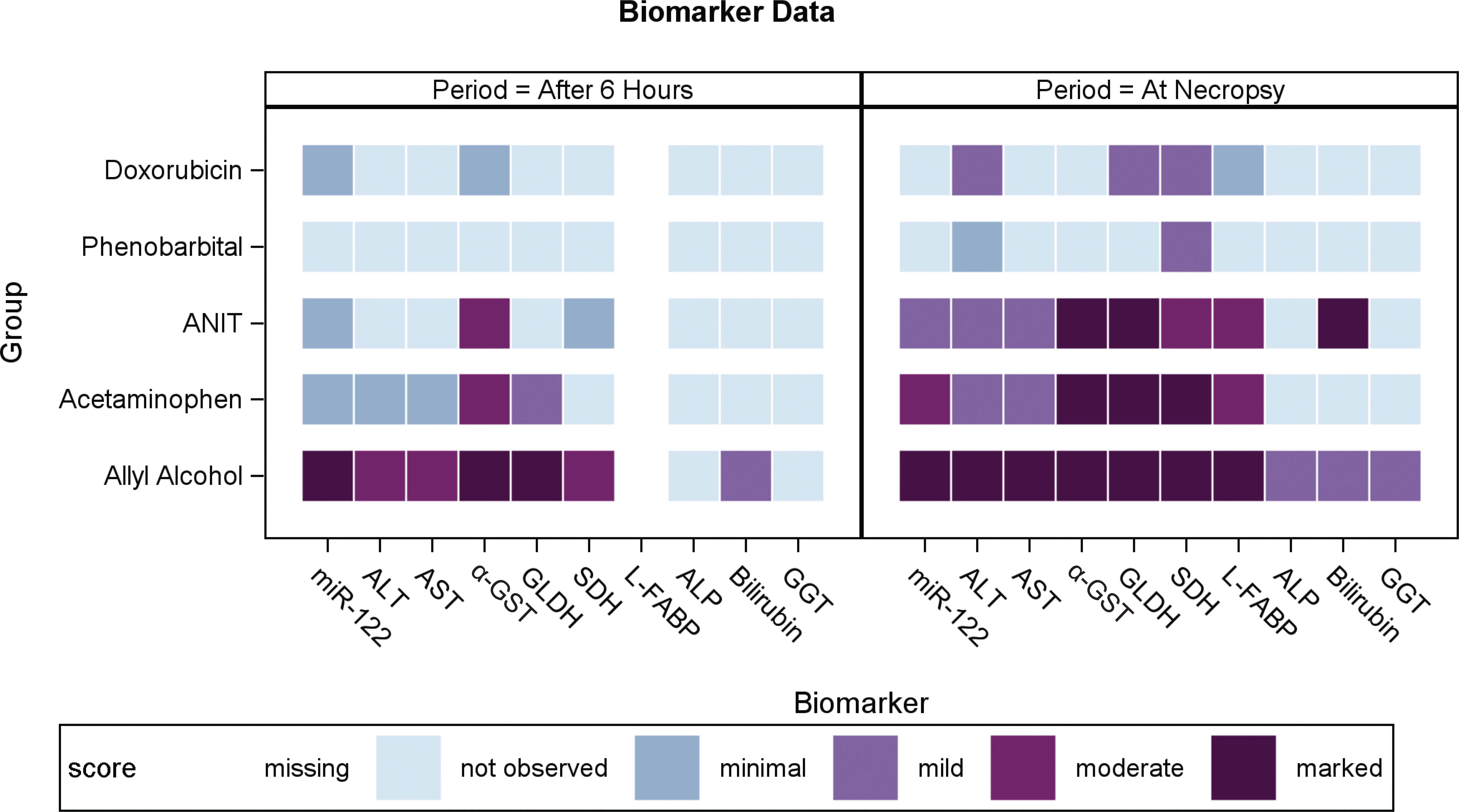
Visualization of biomarker changes according to their toxicological relevance. Biomarker changes were graded as not observed (no change), minimal, mild, moderate, or marked based on the fold change of the biomarker versus the concurrent control (except for GGT). Grading criteria are described in the Materials and Methods section. Sacrifice is 24 hr after exposure to allyl alcohol, acetaminophen or ANIT; 96 hr after first exposure to phenobarbital; and 72 hr after exposure to doxorubicin. miR-122 = microRNA-122; ALT = alanine aminotransferase; AST = aspartate aminotransferase; GLDH = glutamate dehydrogenase; SDH = sorbitol dehydrogenase; a-GST = alpha-glutathione-S-transferase; L-FABP �ver-fatty acid -binding protein; ALP = alkaline phosphatase; GGT = gamma-glutamyl transferase.
Discussion
miR-122 is an miRNA species that is highly expressed in the liver. It has been reported that circulating levels of miR-122 are significantly increased in mice after treatment with acetaminophen (Laterza et al. 2009),
Histologically, the compound treatments resulted in the expected liver changes, namely periportal to diffuse necrosis, centrilobular apoptosis and necrosis, bile duct necrosis and centrilobular hepatocellular hypertrophy for allyl alcohol, acetaminophen, ANIT, and phenobarbital, respectively, and confirmed the validity of this experiment. Plasma miR-122 exhibited a statistically significant increase upon administration of allyl alcohol, acetaminophen, and ANIT. This was due to cellular damage in the liver causing the release of cellular miR-122 into circulation. miR-122 increase was easily and rapidly measurable in plasma and the increase in plasma showed a large dynamic range. Moreover, the increase in circulating miR-122 in rats treated with these liver toxins correlated with liver injury. The magnitude of the change in miR-122 was greater with the high-grade hepatocellular injury by allyl alcohol than with the lower-grade hepatocellular injury by acetaminophen. The correlation of miR-122 with specific liver areas is not established and was not further clarified from this study as miR-122 does not differentiate between periportal or centrilobular injury.
Unambiguous elevation of miR-122 levels has been reported in the plasma as early as 1 hr after exposure to 150 mg/kg of acetaminophen in mice (Wang et al. 2009). In addition, in the author’s experience, miR-122 increase preceded the detection of ALT. Despite histological evidence of necrosis after acetaminophen challenge, ALT was not or only minimally or moderately increased, while a far larger increase was evident in miR-122, as well as in α-GST, SDH, GLDH, or L-FABP. Therefore, miR-122 in addition to the other markers may provide increased sensitivity and specificity when ALT changes are subtle or unequivocal.
Although a significant increase in miR-122 was observed with ANIT-induced biliary injury, there were no indications of hepatocellular damage by histology. In addition to plasma miR-122, ALT and other biomarkers of hepatocellular membrane integrity were elevated upon ANIT treatment, possibly indicating early stages of necrosis that would have progressed and become evident by histopathology. These increases possibly represent the first signs of hepatocellular membrane leakage. It has been described that ANIT treatment, in addition to the biliary injury, might be accompanied by hepatocellular necrosis (Kossor et al. 1993).
Interestingly, miR-122 responses to phenobarbital were negligible and minimal ALT elevations were observed after 4 days. These observations again suggest that miR-122 may evolve as a biomarker that brings clearer interpretation to the ALT signal.
We acknowledge that the presented study is preliminary and that further experiments with larger animal group sizes including male and female animals and with additional compounds with other modes of hepatotoxicity are warranted for complete characterization of miR-122 in terms of sensitivity, tissue specificity, and persistence in the circulation following liver injury. Our understanding for application of miR-122 as a biomarker of liver injury is still limited to date. Definition of baseline levels of miR-122 in normal and healthy rats is vital. Chronic studies addressing histopathology assessments corresponding to miR-122 collection time points correlating to injury, reversibility, and adaptation of the liver are important. Measuring miR-122 as part of a panel of biomarkers, as represented in Figure 4, will be the future of liver toxicity assessment. It is unlikely that any single biomarker will adequately indicate specific liver injury in the near future.
In conclusion, this report demonstrates that circulating miR-122 is elevated with hepatocellular injury but not with hepatocellular hypertrophy resulting from different hepatotoxicants. Since miR-122 elevations were detectable earlier, this biomarker shows significant promise to complement the existing biomarkers for assessing liver toxicity in drug development.
Footnotes
Acknowledgments
We are grateful to the biotechnicians and the laboratory staff of the Drug Safety Sciences Department for excellent technical assistance in this labor-intensive study. We also wish to thank the Hepatotoxicity Expert Group and the Liver Integrated Safety Assessment (ISA) team within Janssen Research and Development for their support received in this study. Sincere thanks also to Helena Geys, Xiang Yao, Erik Hansen, and Graham Bailey for their helpful contributions, suggestions, and discussions.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
