Abstract
Drug-induced liver injury in children (cDILI) accounts for about 1% of all reported adverse drug reactions throughout all age groups, less than 10% of all clinical DILI cases, and around 20% of all acute liver failure cases in children. The overall DILI susceptibility in children has been assumed to be lower than in adults. Nevertheless, controversial evidence is emerging about children’s sensitivity to DILI, with children’s relative susceptibility to DILI appearing to be highly drug-specific. The culprit drugs in cDILI are similar but not identical to DILI in adults (aDILI). This is demonstrated by recent findings that a drug frequently associated with aDILI (amoxicillin/clavulanate) was rarely associated with cDILI and that the drug basiliximab caused only cDILI but not aDILI. The fatality in reported cDILI studies ranged from 4% to 31%. According to the US Food and Drug Administration–approved drugs labels, valproic acid, dactinomycin, and ampicillin appear more likely to cause cDILI. In contrast, deferasirox, isoniazid, dantrolene, and levofloxacin appear more likely to cause aDILI. Animal models have been explored to mimic children’s increased susceptibility to valproic acid hepatotoxicity or decreased susceptibility to acetaminophen or halothane hepatotoxicity. However, for most drugs, animal models are not readily available, and the underlying mechanisms for the differential reactions to DILI between children and adults remain highly hypothetical. Diagnosis tools for cDILI are not yet available. A critical need exists to fill the knowledge gaps in cDILI. This review article provides an overview of cDILI and specific drugs associated with cDILI.
Introduction
Drug-induced liver injury (DILI) accounts for about 10% of all reported adverse drug reactions (ADRs). 1 Seemingly a small portion of ADRs, DILI has nevertheless become one of the leading causes of postmarketing drug withdrawal and clinical acute liver failure. 2 –6 Because of its low incidence (∼1 in 10,000 patients), currently used standard testing, including the extensive preclinical (animal) and clinical studies that usually involve only hundreds to thousands of test participants, is insufficient to fully predict DILI. It is for this reason that DILI is often recognized solely during the postmarketing surveillance of ADRs. The rarity of DILI poses a significant challenge to understand its mechanisms and risk factors.
It is generally believed that DILI is mostly idiosyncratic in nature, being highly host dependent. The host factors include the genetic makeup, nutritional conditions, concomitant medications, underlying diseases, and others, among which age is an outstanding one. It has long been assumed that older age presents a significant risk factor for DILI. This assumption is so well accepted that age ≥55 is assigned 1 additional causality point in the Council for International Organizations of Medical Scientists' Roussel Uclaf Causality Assessment Method scale, a standard diagnostic tool to establish the culprit drug in clinical DILI cases 7 (http://www.livertox.nih.gov/rucam.html; date last accessed: April 4, 2016). This may be a strong contributing factor in the paucity of studies related to DILI in children (cDILI). To our knowledge, this review represents the first effort to summarize available cDILI information from various resources including scientific literature reports and regulatory documents.
This article define children as age ≤18. From conception through development, birth and maturation, a human progresses through several developmental phases and life stages. Various factors in this process may make the developing human more, less, or equally susceptible to DILI when compared to adults.
Clinical Features of Overall cDILI
In PubMed, we found only 4 studies focusing on the clinical features of overall cDILI. The major findings are summarized in Table 1. Other clinical cDILI studies are anecdotal in nature and therefore only briefly mentioned.
Clinical Features of cDILI in 3 Major Studies.

Abbreviations: cDILI, drug-induced liver injury in children; DILIN, Drug-Induced Liver Injury Network; F, female; M, male; NA, not available; WHO, World Health Organization.
aWithout the causality data.
Drug-Induced Liver Injury in Children Accounts for <10% of All DILI Cases and ∼20% of Acute Liver Failure in Children
In a prospective study of the Drug-Induced Liver Injury Network (DILIN), cDILI was found to represent only a small portion of all the DILI victims, that is, only 30 cDILI cases were identified among >300 patients with DILI. 8,9 This small percentage (<10%) of cDILI in all DILI cases is surprisingly similar to a study from India in which cDILI accounted for 8.7% (39 of 450) of all DILI cases. 10 In the World Health Organization database VigiBase, possible cDILI records were found to be 1% of all the reported ADRs. 11 As DILI accounts for about 10% of all ADRs, 1 this study 11 again supports the idea that cDILI represents <10% of all DILI cases. Of note, another study showed that children of ages 10 to 19 accounted for 4% of all the 603 DILI cases during a 13-year study period, though the number of cDILI cases younger than the age of 10 is unknown. 7 As for acute liver failure in children, a multicenter study showed that cDILI accounts for 20% of all the cases. 12 In contrast, with no regard to age, DILI accounts for 52% of all acute liver failure cases. 13 All these data clearly show that cDILI is a very small subset of all the DILI cases. Due to the low number of cDILI cases, it is extremely difficult to collect a sufficient number of qualified participants from a single research center. Collaborative efforts like the DILIN 9 and data mining approach using adverse event reporting system (AERS) 14 and other ADR databases or medical records 15 may prove particularly useful in future studies.
Children’s Overall Susceptibility to DILI
It has long been held that children are less susceptible to DILI than adults (https://www.uptodate.com/contents/drug-induced-liver-injury). However, this could partly be due to a failure to diagnose and report drug hepatotoxicity in children. Other factors playing a role in children’s susceptibility include taking relatively fewer drugs commonly associated with hepatotoxicity (eg, cardiovascular, antihypertensive, antidepressant), having a lean body mass and fewer concomitant diseases, and lacking other common predisposing factors such as ethanol consumption and cigarette smoking. However, a recent study suggested that children may be at greater risk of developing DILI associated with antituberculous drugs isoniazid (INH) and rifampin (6.9% in children vs 2.7% in adults). 10 Due to the lack of drug consumption data, it is difficult to calculate the true incidence of cDILI, thereby obviating a convincing conclusion.
Culprit Drugs in cDILI
Many culprit drugs in DILI appear similar between adults and children. The major drugs in cDILI are summarized in Tables 2 and 3. However, exceptions do exist. For example, a surprising finding in the DILIN study is that none of the cDILI cases were caused by amoxicillin/clavulanate, a drug commonly implicated in adult DILI (aDILI) patients. Interestingly, in a study from India, of the 39 cDILI cases, only 1 (3%) was attributable to amoxicillin/clavulanate. In the VigiBase, only 38 among the 6,595 (0.5%) cDILI cases were associated with amoxicillin/clavulanate. In a Chinese study, amoxicillin was responsible for only 3 (4%) of 69 cDILI cases. 16 These data show that amoxicillin/clavulanate is not a major cause of cDILI, although it is a highly implicated drug for aDILI. Similarly, herbal and dietary supplements, some of which are used as drugs in Asian countries, seem to be of less significance in cDILI than in aDILI, though severe cDILI cases due to these supplements were occasionally reported. 17 Interestingly, in a recent study examining cDILI in 69 children hospitalized for DILI in a Chinese hospital, it was found that traditional Chinese herbal treatments were associated with more severe cDILI when compared to Western drugs. 16 In this same study, it was also found that antibiotic treatment had the highest incidence of cDILI, with cephalosporins having the highest incidence among the different types of antibiotics. On the other hand, the drug basiliximab was identified to be a causative agent of cDILI in the VigiBase, though it has never been associated with aDILI. 11 However, it should be noted that the number of children treated with these drugs was not known, and therefore, the above observations are not based on the normalized incidence data. It should also be pointed out that DILIN does not collect the DILI cases associated with acetaminophen overdose. 8
Selected Drugs Associated With cDILI.

Abbreviations: cDILI, drug-induced liver injury in children; NA, not available.
Age Effects on DILI Susceptibility as Stated in FDA-Approved Drug Labels.
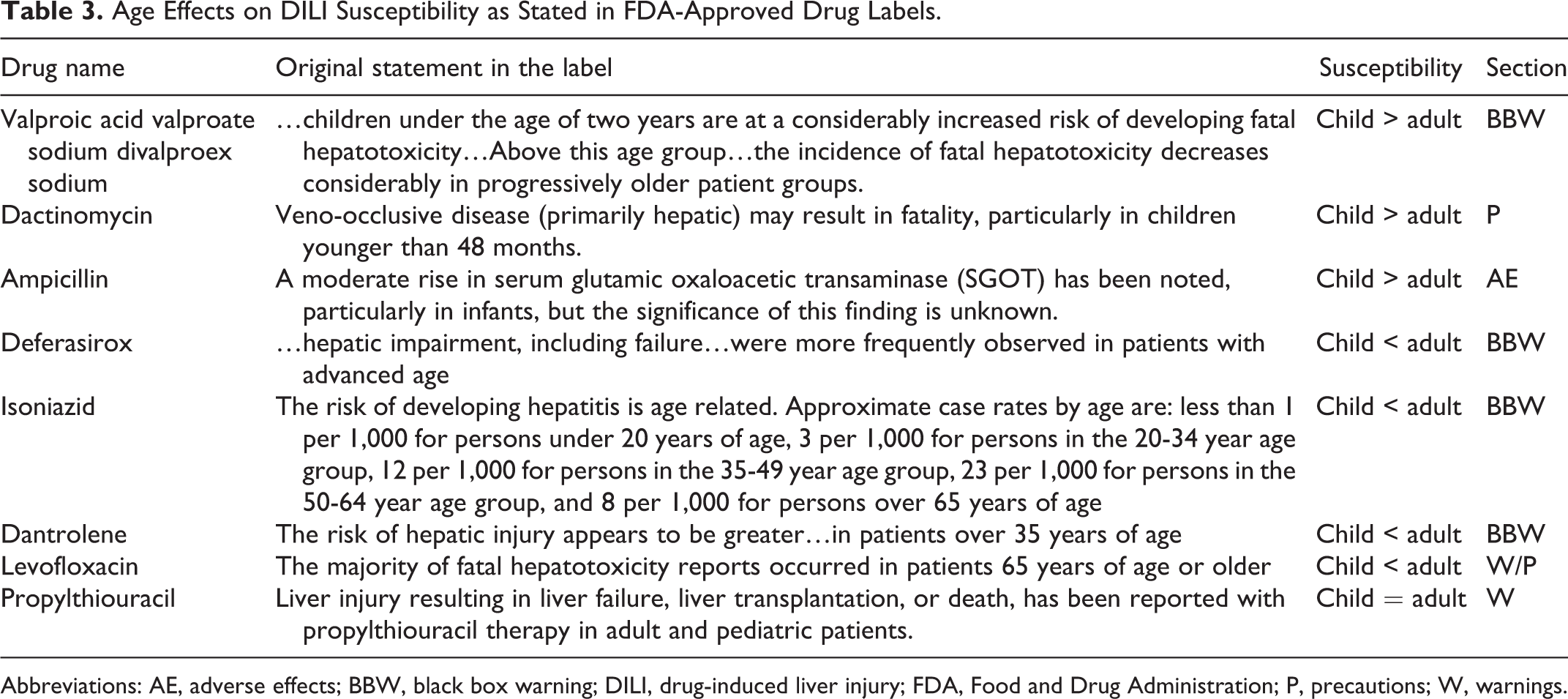
Abbreviations: AE, adverse effects; BBW, black box warning; DILI, drug-induced liver injury; FDA, Food and Drug Administration; P, precautions; W, warnings.
Liver Injury Pattern in cDILI
Clinically, DILI has been classified into hepatocellular, cholestatic, or mixed (hepatocellular/cholestatic) according to the biochemical characteristics. Hepatocellular DILI often involves direct damage of the hepatocytes and is associated with elevated serum transaminase levels (ie, alanine and aspartate aminotransferases) due to leakage from damaged hepatocytes. Cholestatic DILI often involves injury to some part of the bile excretion apparatus resulting in impaired bile excretion and is associated with elevated serum levels of analytes such as γ-glutamyl peptidase, alkaline phosphatase, and bilirubin that denote bile duct injury. Mixed injury presents with a mixture of both types of effects. The hepatocellular liver injury pattern was predominant in cDILI and fibrosis was common in biopsy, though other patterns were also observed. 9,16 A notable finding is that none of the reported cDILI cases had cirrhosis, which is a common finding in aDILI. 10,16,18 However, cDILI does share many histopathological characteristics with aDILI, such as the broad spectrum of manifestations, including inflammation, cholestasis, and steatosis. The immune system also seems to be critically involved in the pathogenesis of cDILI as in aDILI, 9,10 but cDILI cases with hypersensitivity characteristics showed a better outcome. 10,16
Outcomes of cDILI
The prognosis of cDILI appears controversial. In the United States, the recovery rate of cDILI is high and fatality is only 4%, 9 but a surprisingly high fatality rate of 31% was observed in India. 10 The reason for such discrepancy is unknown, and more data are needed to establish the true outcomes of cDILI.
Regulatory Understandings and Policies Related to cDILI
The US Food and Drug Administration
It was only recently that Food and Drug Administration (FDA) was required by law to specifically consider the pediatric safety of drugs under the Pediatric Research Equity Act (Public Law 108-155) of 2003. 19 Prior to this, drug registrants were not required to assess the safety of drugs in the pediatric population, which has resulted in a paucity of data concerning cDILI. Although the FDA recently issued a new “guidance for industry” to cover many aspects of studying DILI, cDILI is not specifically mentioned, 20 possibly due to the lack of data. The FDA-endorsed biomarkers for DILI are intended for use in the general population without specific considerations of children. 21 However, the recent FDA “guidance for industry” does recommend that “the contribution of…age”, as well as other factors, “should be explored” in patients who are suspected to be suffering from DILI. 20
For preclinical studies, as required by the FDA, a typical toxicology study package for a new drug includes an assessment of the drug on the developing organism through weaning. These studies are often divided into developmental phases and focus on gross developmental defects and the reproductive capacity of the animals. However, the specific effect of the drug on the liver is rarely assessed. Therefore, if a drug causes liver toxicity in the developing animal, it would likely be missed since the current study designs are not specifically assessing DILI. Juvenile toxicity studies are conducted for some drugs, and these present a better opportunity for assessing the potential for cDILI.
To obtain some cDILI-specific insights from a regulatory perspective, we reviewed all of the FDA-approved drug labels in DailyMed, which provides a comprehensive and up-to-date resource of US drug labels. Of the ∼1,700 single active ingredient human prescription drugs, we found only 7 carry cDILI-related information in their labels. Details are presented in Table 3. Three drugs including valproic acid (VPA), dactinomycin, and ampicillin sodium are believed to cause DILI more often in children. In contrast, 4 drugs including deferasirox, INH, dantrolene sodium, and levofloxacin appear to cause DILI more often in adults. Despite many inherent limitations of the information presented in the drug labels, these findings may provide useful clues for further cDILI studies. 22 On the other hand, the fact that only an extremely limited number of drugs carry cDILI information on their labels highlights a significant knowledge gap in regulatory decision-making.
It should be pointed out that the American College of Gastroenterology (ACG) Clinical Guideline states that children are more susceptible to propylthiouracil-induced DILI than adults, 23 but this has not been included in FDA-approved drug label (Table 3). The ACG Clinical Guideline provides detailed criteria for the diagnosis of DILI in general but only briefly mentioned that children’s susceptibility to DILI is drug specific. 23 Although this guideline lists “age” among the “suggested minimum data required for the diagnosis of DILI,” exactly how the age information needs to be used for DILI diagnosis is not available. No regulatory documents provide the diagnosis criteria of DILI.
Non-FDA Regulatory Agencies
Some non-FDA regulatory bodies, such as the US Environmental Protection Agency (EPA) under the Food Quality Protection Act of 1996 (FQPA), assume that children are significantly more sensitive than adults to chemical exposures unless there are clear data to the contrary. 24 In risk assessments for products such as pesticides, the EPA uses an additional 10-fold margin of safety to protect infants and children from potential pre- and postnatal toxicity. The 10-fold FQPA safety factor can be modified only if reliable data demonstrate that the resulting level of exposure would be safe for infants and children. The EPA’s approach is a mixture of science (children may or may not be more susceptible depending on the chemical and type of exposure) and public health policy (assuming by default that children are always more sensitive than adults). However, cDILI-specific information is not available from these regulatory policies.
Possible Contributing Factors to the Differences Between cDILI and aDILI
The human liver goes through many stages of development as a child matures into an adult. Depending on the various physiologic, ontogenic, environmental, and genetic factors, a child may be more, equally, or less susceptible to a drug when compared to an adult.
Factors Related to Pharmacokinetics and Pharmacodynamics
Phase I drug metabolism is mainly carried out by hepatic cytochrome P450 (CYP) enzymes, which undergo developmental changes in expression and activity levels after birth, which can lead to differential drug sensitivity. For example, CYP3A4 is present at high levels in adult human liver and is responsible for the metabolism of a wide variety of drugs. In contrast, in the neonatal liver, CYP3A7 is the predominant CYP. CYP3A4 expression increases during the first year of life reaching approximately 50% of adult levels and continuing to increase with age. 25 –28 Although many of the drugs metabolized by CYP3A4 are also metabolized by CYP3A7, there are some differences that could lead to altered metabolic profiles for a given drug and in turn differential sensitivity to DILI. This differential pattern of CYP expression and activity also applies to many other CYPs. In general, there are 3 patterns of CYP developmental expression: (1) CYPs that are expressed in the fetal liver and decline with age (eg, CYP3A7), (2) CYPs that begin expression in the early neonatal period within hours after birth (eg, CYP2D6 and CYP2E1), and (3) CYPs expressed later in neonatal development (eg, CYP1A2 and CYP3A4). 29,30
Although phase I metabolism is typically considered a detoxifying reaction, many drugs are metabolized to reactive intermediates that can cause cellular injury. Acetaminophen metabolism by CYP1A2, CYP2E1, and CYP3A4 to the reactive metabolite N-acetyl-p-benzoquinoneimine (NAPQI) that binds cellular macromolecules, such as free sulfhydryl groups on proteins, is a classic example. 31 –33 Therefore, depending on the role phase I metabolism plays in activating or detoxifying a drug, 34,35 the fetus or pediatric patient may be more or less susceptible to a given drug depending on the interplay of the rate of metabolism and the toxicity of the drug or metabolite.
Most phase II conjugation reactions involve adding a hydrophilic cofactor to the drug or metabolite to greatly expedite elimination of the drug via the kidneys or the liver. Although phase II reactions often attach cofactors to those functional groups added or exposed during phase I reactions, phase II reactions can occur directly on functional groups on the parent drug (eg, on a free hydroxyl group). Phase II conjugation reactions include glucuronidation, sulfation, methylation, acetylation, and amino acid and glutathione conjugation. Most phase II biotransforming enzymes are located in the cytosol, with some exceptions such as uridine 5′-diphospho-glucuronosyltransferases located in the microsomes. Phase II reactions often proceed at a relatively fast rate, making phase I reactions the rate-limiting step. It is already known that cofactors used in phase II reactions vary with age. 36 These changes are likely to impact the metabolic pathways a drug goes through as a child ages.
The metabolism of acetaminophen provides a great example of the interplay of the phases I and II metabolic pathways. At therapeutic doses, acetaminophen is primarily glucuronidated and sulfated. At supratherapeutic doses, phase II cofactors are depleted, and a larger portion of acetaminophen is metabolized by CYP enzymes to the reactive NAPQI. N-acetyl-p-benzoquinoneimine is detoxified by glutathione; however, there is a finite supply and once that becomes depleted, NAPQI can bind and disrupt cellular macromolecules. In the young, glucuronidation is low, whereas sulfation is similar to adult levels. Although acetaminophen is extensively glucuronidated in adults, the low activity in children results in the sulfation pathway being the predominant route of metabolism. 37,38 The complicated interplay of phase I and phase II pathways is active for many drugs and the influence of age adds another layer of complexity to predicting the metabolism in pediatric versus adult patients. Two examples of neonatal sensitivity due to deficient metabolism are the Grey Baby syndrome observed in the 1950s due to reduced glucuronidation of the antibiotic chloramphenicol and gasping syndrome identified in the 1980s due to benzyl alcohol used as an antibacterial agent in intravenous solutions. 29,39 –41 Both of these produced very clear clinical signs of toxicity due to poor metabolism of the compounds in children. It is likely that similar types of metabolic factors can lead to differential sensitivity to cDILI.
Drug transporters are often referred to as phase III metabolism, although they do not alter the chemical structure of a drug or metabolite. Drug transporters are responsible for transporting (influx and efflux) drugs across various biological surfaces such as the gastrointestinal tract, blood–brain barrier, placenta, hepatocytes, and renal tubular cells. The multidrug resistance protein (also called P-glycoprotein) is the most notable of the drug transporters and is a member of the adenosine triphosphate–binding cassette family. Many of the transporters prevent the accumulation of drugs and aid their elimination from the body. Inhibition of drug transporters by compounds such as grapefruit juice can result in serious drug interactions due to the accumulation of toxic levels of the drug. Unfortunately, very little data exist on the ontogeny of phase III transporters; however, it is likely that there are developmental changes in the levels of these transporters that impact the exposure to drugs and in turn their potential to cause DILI. 42 –44
The developing child undergoes various physiological changes that can impact the absorption, distribution, metabolism, and elimination of drugs, which could in turn lead to differential sensitivity to DILI. In general, the rate of absorption of most drugs is slower in neonates and infants and increases with age. There are also developmental differences in the activity of intestinal flora, intestinal drug-metabolizing enzymes, and phase III transporters that can affect drug bioavailability. For example, immature intestinal CYP3A4 metabolism of midazolam reduces clearance in preterm infants. 45 Once absorbed, in general, the overall clearance of drugs appears to be reduced during the first year of life. 37 Metabolic clearance then increases and reaches a maximum between 2 and 10 years of age, declining thereafter with age. This higher clearance in young children often requires relatively higher weight-based doses. Clearance is the result of various factors, such as liver metabolism, biliary excretion, and kidney elimination. The higher clearance in children is believed to be partly due to the increased liver volume normalized to body weight.
Physiological factors, such as body weight and organ volume, can also affect drug clearance significantly. As compared to adults, children have higher amount of body water content and body surface area to mass index and lower body fat and plasma protein-binding capacity of drugs. These factors may have profound impacts on the pharmacokinetics of many drugs. Allometric models, which take into account the difference in body weight among age groups, have been adopted to predict drug clearance in children. 46
Another key factor in determining child sensitivity to DILI is pharmacodynamics. It represents the interaction of a drug with the target, such as a receptor on a cell, and the subsequent response. Although it is likely that there are developmental differences in pharmacodynamics, very little data are available about changes with age. 47 –51 Combining age-specific pharmacokinetic/metabolism changes with potential pharmacodynamic changes results in a complex paradigm of drug sensitivity that changes over age, and this is likely to vary for each type of drug.
Factors Related to Defense Mechanisms
It is well known that oxidized proteins accumulate with age, providing an indirect indication that defense mechanisms may be high in the young and decrease with age. 52 –54 Glutathione is a sulfhydryl-containing compound that acts as an antioxidant by scavenging reactive metabolites. Glutathione either can directly bind reactive metabolites or can be conjugated to reactive metabolites through an enzymatic process by glutathione S-transferases. Glutathione levels in human blood were reported to be inversely correlated with age, with the exception of infants <1 year of age, supporting a role of high levels of defense mechanisms in children. 55 However, even if certain defense mechanisms are elevated in the young, they might also be more overwhelmed with normal physiological insults and less able to deal with exogenous insults from drugs. For example, Tsukahara reported that urinary biomarkers of oxidative stress were highest in young children and decreased with age to reach constant levels by early adolescence. 56 It was concluded that the markers of oxidative stress are probably due to the high metabolic rate of growing children compared to adults. Obesity may also play a role since higher levels of oxidative stress have been associated with excessive adipose tissue in both children and adults. 57 Addition of a drug that causes oxidative stress on top of the high metabolic rate or prooxidant environment could overwhelm endogenous defense mechanisms and lead to DILI.
Environmental Factors
An often overlooked factor in drug toxicity is concomitant environmental exposures that may have an additive or a synergistic effect with the drug. The growing child is particularly vulnerable to these concomitant exposures since they are starting to probe their environment without knowledge of the potential risks. A good example is hand-to-mouth activity of young children. A child might pick up a toy off a floor that was recently treated with a household pesticide. The pesticide is transferred to the child’s hand and subsequently to the child’s mouth. Assuming a sufficient quantity of pesticide or other chemical is ingested, it might interact with a concomitantly administered drug resulting in DILI. An even more problematic time is adolescence where young adults may experiment with alcohol and illicit drugs. The drugs themselves might cause liver injury (eg, alcohol, ecstasy, and cocaine) or the illicit drug could interact with concomitantly administered drugs resulting in DILI. 58,59 An example is where alcohol consumption can potentiate the hepatotoxicity of acetaminophen. 60,61
Specific Drugs Associated With cDILI
Drug-induced liver injury in children is possibly best assessed on a drug-by-drug basis. The following sections provide an overview of drugs that have been associated with cDILI. A comprehensive review of every drug that has been associated with cDILI is beyond the scope of this review. Instead, the commonly implicated drugs in cDILI are briefly summarized. Whenever available, the mechanistic data from animal models are also included. The representative drugs are summarized in Table 2.
Acetaminophen
Acetaminophen is an over-the-counter analgesic and antipyretic that is widely used by both children and adults. Although acetaminophen is used safely by millions of people each year, it remains a leading cause of DILI in the United States and is by far the leading cause of acute liver failure in adults. 62 –65 In children, acetaminophen is the second most frequent cause of acute liver failure only behind indeterminate causes. 12,66,67 Due to the high incidence of hepatotoxicity, an FDA advisory committee recommended lowering the daily therapeutic dose of acetaminophen (http://www.fda.gov/AdvisoryCommittees/Calendar/ucm143083.htm; date last accessed: April 4, 2016). The committee concluded that in addition to people unknowingly consuming multiple products containing acetaminophen, some individuals may be especially sensitive to liver injury from acetaminophen and that more research is needed to understand whether age, among other factors, might have a role in making some individuals more sensitive. In response to the committee’s recommendations, the FDA subsequently worked with industry to label acetaminophen-containing products in a manner that sufficiently warns about the hepatotoxicity risk of acetaminophen-containing over-the-counter and prescription drugs, including combination drug products (http://www.fda.gov/Drugs/DrugSafety/ucm394916.htm and http://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm310477.pdf; date last accessed: April 4, 2016).
At therapeutic doses, acetaminophen is metabolized by phase II metabolic pathways of glucuronidation and sulfation, with the glucuronidation pathway accounting for ∼60% of the metabolism after a therapeutic dose in adults. 68 At higher doses, phase II cofactors are depleted and a larger portion of acetaminophen is metabolized by CYP enzymes to the reactive metabolite NAPQI. N-acetyl-p-benzoquinoneimine is detoxified by glutathione. However, once glutathione levels become depleted, NAPQI can bind and disrupt cellular macromolecules resulting in cellular injury. In the young, the sulfation pathway is the predominant route of metabolism until about 12 years of age since glucuronidation activity is low compared to adults. 37,38
Although in most cases acetaminophen hepatotoxicity is due to intentional overdose in suicide, a child might be mistakenly given a large amount of acetaminophen by careless caregivers or health-care providers causing liver injury. 69,70
In general, children appear to be more resistant to acetaminophen-induced hepatotoxicity compared to adults. 11,70 –72 This is backed up by a query of the US FDA AERS database where children (ages 0-16 years) have observed reporting incidences of hepatotoxicity due to acetaminophen that were similar to or less than adults (≥17 years). The reason for children’s general decreased sensitivity is likely due to a combination of factors such as differential metabolic pathways, higher capacity for producing glutathione, and fewer risk factors, such as alcohol consumption.
Acetaminophen overdose is a medical emergency. It is important to note that criteria used for assessing acetaminophen overdose in adults may not be entirely applicable for children, such as the King’s College Hospital criteria. 72 In adults, the clinical course of an overdose starts with gastric distress, such as anorexia, nausea, vomiting, and abdominal pain, over the first couple of days. Unfortunately, these early signs do not foretell the underlying seriousness of the injury and this may delay intervention. Serum transaminase levels become elevated within 12 to 36 hours after ingestion with peak levels within 3 to 4 days. Clinical signs of hepatotoxicity (right subcostal pain, tender hepatomegaly, coagulopathy, and jaundice) occur within 2 to 4 days. Poor prognosis is often associated with worsening coagulopathy, onset of hepatic encephalopathy, or renal failure. Biopsy reveals centrilobular necrosis with none to minimal damage to the periportal regions. If the injury is not fatal, the centrilobular necrosis is reversible with remodeling of the injured areas over the course of several weeks or months. 68
Animal models have been used to understand the mechanism of why children seem less susceptible to acetaminophen hepatotoxicity than adults. In both mice 74 –76 and rats, 77 –81 younger animals, but not older ones, have been shown to be resistant to acetaminophen hepatotoxicity, although 1 study in mice reported no significant age-related differences in sensitivity 82 and another study in rats reported that young Fischer 344 rats were more susceptible to acetaminophen hepatotoxicity than older rats. 83 Various mechanisms contributing to such differences have been proposed and are summarized in Table 2. It appears that one important factor is related to the metabolic activation mediated by specific CYP isoenzymes, subsequent conjugation reaction with glutathione, and covalent binding to hepatic proteins. 74,75 Although these are the relatively well-established mechanisms for acetaminophen hepatotoxicity, they cannot fully explain the overall toxicity process and likely represent only the first step of toxicity. Newer mechanisms such as protein nitration and mitochondrial membrane permeability transition have been proposed to be critical in the second stage of acetaminophen hepatotoxicity, but they have not been examined with respect to the effect of age. This may represent an interesting new direction for future cDILI studies focusing on acetaminophen.
It should be noted that significant species differences exist for cDILI study. The abovementioned mechanisms for cDILI have only been established in mice but not rats. Factors other than metabolic activation are responsible for the decreased susceptibility observed in young rats. 77,78,80 Even for the same species, differential susceptibility was observed in different strains of animals. For example, Fischer 344 rats did not display age-dependent sensitivity to acetaminophen hepatotoxicity. 84 These studies show that the differential susceptibility of children and adults to acetaminophen hepatotoxicity can be reproduced in some rodent models.
Halothane and Halogenated Inhalation Anesthetics
Halothane is an inhaled anesthetic that produces mild liver injury in 1 in 5 patients 85 and a more severe immune-mediated ADR, or “HAL hepatitis,” in 1 in 6,000 to 30,000 patients exposed to the drug. 86,87 Halothane is metabolized by CYP 2E1 to form trifluoroacetyl (TFA) chloride, which binds covalently to proteins and lipids, forming TFA adducts. Studies, in which halothane hepatotoxicity in guinea pigs was ameliorated by SKF-525A, a broad-based P450 inhibitor, and exacerbated by 4-methylpyrazole, a CYP2E1 inducer, illustrated the requirement for metabolism in the development of hepatotoxicity. 88,89 Furthermore, susceptibility to HAL hepatotoxicity in a guinea pig model correlated with the formation of liver TFA protein adducts. 90,91
Children have been suggested to be relatively resistant to halothane hepatotoxicity as compared to adults. 91 In a mouse model, younger animals were found to be less susceptible to halothane-induced liver injury. However, this was only observed in BALB/cJ mice but not C57BL/6 mice, suggesting that halothane hepatotoxicity also depends on the genetic makeup of recipients. 92 In the guinea pigs, halothane also induced more severe liver injury in adults than in the young, but the mechanisms for such differences are difficult to explain, because similar pharmacokinetics were observed in old and young animals. 93
Due to halothane-induced hepatitis, other halogenated inhalation anesthetics are typically used, such as isoflurane, sevoflurane, enflurane, and desflurane, since they appear to have a lower propensity to cause liver injury. However, the drug labels of each of these anesthetics warn against potential hepatotoxicity, and there have been clinical cases of hepatotoxicity reported for some of these drugs. 87,94 –96 Due to the similarity in the structures and mechanisms of action, it is expected that the possible differential sensitivity of children to halothane DILI would also occur for these various halogenated inhalation anesthetics.
Valproic Acid
The VPA exposure is generally well tolerated but can cause hepatotoxicity. The VPA-induced hepatotoxicity manifests as a variety of different adverse effects, including transient elevations in liver enzymes, hepatic dysfunction, hyperammonemia, and a more rare progressive liver failure that resembles Reye syndrome. In 1996,
Many studies have suggested that VPA-induced hepatotoxicity is much more frequent in children compared with adults and the risk of hepatotoxicity is estimated up to 1 in 600 for children less than 2 years of age and decreases thereafter. 11,99 –101 In vitro testing has shown 4-ene-VPA to be toxic to human HepG2 cells. 102 The activity of CYP2C9, which is responsible for the production of 4-ene-VPA, is highest in young children and then declines with age. Concurrent with the changing activity of CYP2C9, the level of 4-ene-VPA decreases with age, 103 suggesting that CYP2C9 and 4-ene-VPA are responsible for the differential sensitivity of young children to VPA.
In a rat model, VPA induced hepatic steatogenesis in older animals but not in younger ones. 104 This finding was later confirmed by another group. 105 However, in precision-cut rat liver slices, VPA toxicity was stronger in younger animals than in older ones. 106 The mechanisms for such differential reactions are not clear but have been proposed to be related to the hepatic metabolism of VPA. 106
Propylthiouracil
Propylthiouracil was approved by the FDA in 1947 for the treatment of hyperthyroidism in both children and adults. It interferes with the incorporation of iodine into thyroglobulin, thereby inhibiting thyroid hormone production. 107 Although hepatitis is rare, it is potentially dangerous and is the third most common cause of drug-induced acute liver failure in children (<18 years of age). 108 Recently, the FDA identified 34 severe DILI cases attributable to propylthiouracil, among which 32% are children. The FDA therefore issued a box warning, the strongest regulatory warning for current prescription drugs, of liver injury to the drug label of propylthiouracil.
Children appear to be more sensitive to propylthiouracil-induced hepatotoxicity than adults. 109,110 In a systematic review of adverse drug event reports submitted to the US FDA AERS, propylthiouracil had a higher adjusted reporting ratio for severe liver injury in children (<17 years of age) compared to adults. 14 In addition, propylthiouracil had a higher adjusted reporting ratio when compared to methimazole, another antithyroid medication, further supporting that children exhibit a unique sensitivity to propylthiouracil. However, the exact mechanism for such differential sensitivity is unknown. It is not even known whether propylthiouracil hepatotoxicity could be reproduced in animal models, although metabolomics profiling showed promise at predicting propylthiouracil hepatotoxicity in rats. 111
Antituberculosis Drugs
Isoniazid
As a cornerstone drug commonly used for treating tuberculosis (TB), INH is well known to cause liver failure. 112,113 In children with latent TB infection, INH therapy remains the treatment of choice, effectively preventing disease progression. Serum transaminase elevation is the most frequent abnormality, and it occurs in approximately 7% to 14% of children receiving INH monotherapy. 114,115 More severe INH-induced hepatitis has been considered to be more common in adults than in children with a frequency of 0.1% in children versus 1.3% in adults. 116 However, the incidence of INH -associated liver failure in children is estimated at 3.2 per 100 000 patients, approaching the 4.2 to 14 per 100 000 rate reported in adults. 112 Clinical symptoms consist of jaundice, nausea, fatigue, loss of appetite, abdominal pain, and vomiting. The histological findings resemble acute viral hepatitis, and the symptoms of hepatotoxicity typically develop 2 to 3 months after the start of treatment. Current guidelines recommend immediate withdrawal of INH and frequent measurement of serum transaminase levels at the first symptoms suggestive of hepatitis 117 (http://www.cdc.gov/mmwr/preview/mmwrhtml/rr5211a1.htm; date last accessed: April 4, 2016). In most cases, children recover spontaneously with INH withdrawal. 118
It has been speculated that INH -associated hepatic injury is attributed to the INH metabolites hydrazine or acetylhydrazine. Children receiving simultaneous treatment with CYP enzyme inducers such as rifampicin, phenytoin, or phenobarbital seem to be at greater risk of hepatotoxicity. In human hepatocytes, preexposure to rifampicin, which is a potent CYP inducer and a drug commonly used together with INH for TB treatment, increased the severity of cytotoxicity. 119 In addition, there are a few liver injury cases reported due to an interaction of INH and carbamazepine. 120,121 The INH itself has been reported to induce CYP2E1 expression, and 1 study reported that CYP2E1 polymorphisms correlated with increased hepatotoxicity in pediatric patients. 122 Plasma hydrazine concentrations are also dependent on whether the patient is a fast, intermediate, or slow acetylator, determined by the NAT2 genotype, which differs between ethnic groups and ages. Slow acetylators, who have a higher plasma concentration of hydrazine, have been associated with greater risk of INH-induced hepatotoxicity. 123 –126 Although children are slow acetylators until approximately 4 years of age, phenotypic studies in children failed to demonstrate a significant role of the slow acetylator phenotype in the development of hepatotoxicity. 122 Other risk factors associated with INH hepatotoxicity are severe or meningeal TB, viral hepatitis, pregnancy, and ingestion of acetaminophen or ethanol. 127
Rifampicin plus INH
At therapeutic doses, rifampicin is well tolerated for the treatment of TB. There are few data on rifampicin-associated hepatotoxicity in children when using rifampicin alone. However, in patients who received INH and rifampicin, the incidence of hepatic dysfunction is 5% to 8% more frequent and present earlier than with either medication alone. 115,128 In most cases, children seem as vulnerable as adults to the combination of INH and rifampicin treatment. O’Brien et al 129 performed a retrospective review of hepatotoxicity rates in anti-TB therapy and reported a 3.3% incidence of drug-induced hepatotoxicity in 430 children receiving INH and rifampicin. The histological appearance is characteristically patchy, and there is less periportal inflammation than is seen with INH alone. In a rat model, rifampicin plus INH induced apparent liver toxicity, but the effect of age on toxicity was not investigated. 130
Pyrazinamide
Pyrazinamide is used more commonly in combination with other agents for active TB disease than as a monotherapy. There are reports of pyrazinamide-induced hepatotoxicity in patients, 128,131 with some studies showing a correlation with dosage and duration of treatment. 125 A recent epidemiology study reports pyrazinamide is idiosyncratic because high-dose therapy did not significantly increase hepatotoxicity compared to the low dose currently used. 132 Some evidence suggests that age younger than 5 years was a factor that contributed to the development of severe hepatotoxicity in Japanese pediatric patients receiving pyrazinamide in combination with INH and rifampicin. 133 Because pyrazinamide in combination with rifampicin has a high incidence of hepatotoxicity (7.7% severe hepatotoxicity), it is no longer recommended as latent TB treatment. 134 Pyrazinamide was recently shown to cause mild liver injury in a rat model, but the effect of age on hepatotoxicity was not examined. 135 In addition, another recent study showed that transcriptomic and proteomic analyses of liver tissue from rats treated with pyrazinamide exhibited molecular changes representative of DILI. 136
Ketoconazole
Ketoconazole is associated with a high risk of hepatotoxicity, and its use is prohibited in some countries or tightly controlled in others where it is still approved for use. 137,138 The incidence of ketoconazole-induced hepatotoxicity is estimated at 13.4 per 10 000 prescriptions. 139 Older women appear to be the most sensitive to ketoconazole DILI. 71 Serum transaminase elevations occur in 2% to 17% of patients, with onset typically occurring after weeks of therapy. Hepatocellular injury is the dominant pattern; however, cholestatic or mixed patterns are also observed. Acute liver failure in the pediatric and adult populations has been observed with the use of ketoconazole. In primary rat hepatocytes, the toxicity of ketoconazole has been quantified by the lactate dehydrogenase release, a marker commonly associated with hepatocyte necrosis. 140 No animal models have been established to test the differential sensitivity to DILI between children and adults.
Sulfonamides
Multiple sulfonamide-containing medications (eg, sulfanilamide, trimethoprim–sulfamethoxazole, and pyrimethamine–sulfadoxine) are routinely used for pediatric infections and have been reported to cause idiosyncratic hepatotoxicity. 141 Acute liver failure is rare in children, and most patients recover within 3 months after the discontinuation of the drug; however, instances of liver failure leading to the need for liver transplantation and/or death have been reported. 142 Symptoms of hepatotoxicity occur after a latency period of days to a month after therapy has begun. Most reported cases describe a centrilobular cholestasis with portal infiltration, but mixed hepatic cholestatic and hepatocellular necrosis have also been observed. The mechanism of sulfonamide-induced hepatotoxicity has been suggested to be a hypersensitivity reaction, supported by symptoms of fever, rash, and peripheral eosinophilia.
Phenytoin
Phenytoin-induced hepatotoxicity occurs in children, with 10% to 38% of those cases progressing with hepatic necrosis and liver failure. 143 The interval between the initiation of phenytoin therapy and the onset of clinical abnormalities ranges from 1 to 6 weeks in the vast majority of patients. 144 The most common symptoms are fever, rash, and lymphadenopathy, with jaundice and hepatosplenomegaly being found as well.50 Biochemical features are variable but generally include abnormal serum bilirubin, transaminases, and alkaline phosphatase levels, as well as eosinophilia and leukocytosis. The morphologic and pathologic abnormalities are nonspecific, including, but not limited to, primary hepatocellular degeneration and necrosis. Hepatic injury with phenytoin is most likely secondary to a hypersensitivity reaction rather than a direct hepatotoxic effect. Traditional animal models of phenytoin hepatotoxicity are not available; however, a recent study demonstrated that treating mice with phenytoin in combination with buthionine sulfoximine, an inhibitor of glutathione synthesis, could produce hepatotoxicity. 145 In addition, metabolomics profiling of phenytoin-treated rats showed promise in predicting phenytoin hepatotoxicity based on metabolite pattern changes. 111,146
Carbamazepine/Oxcarbazepine
A transient and asymptomatic elevation of liver enzymes has been associated with carbamazepine, which occurs in 25% to 61% of patients 144 The onset of symptoms usually occurs within 3 to 4 weeks after the initiation of therapy and is independent of serum carbamazepine levels. In adults, 2 forms of hepatic injury have been reported: the predominant type is granulomatous hepatitis presenting with fever and right upper quadrant pain, and the other is presented with small portal bile duct paucity and hepatocellular necrosis attributed to chronic toxicity from the drug. In children, the usual clinical picture is hepatitis, sometimes associated with a drug hypersensitivity syndrome similar to that of phenytoin. Serious adverse reactions may proceed even after early intervention and discontinuation of the drug, and liver failure may occur. Because carbamazepine causes autoinduction of hepatic enzymes, a higher dose may be required to ensure adequate serum drug concentrations. 147 Oxcarbazepine is a derivative of carbamazepine; however, it does not induce hepatic enzymes. Drug–drug interactions may play a role in susceptibility to DILI. For example, phenytoin, felbamate, lamotrigine, and valproate have been shown to increase the serum concentration of the active metabolite of carbamazepine, carbamazepine-10,11 epoxide, thus causing potential toxicity. 148,149
Lamotrigine
Lamotrigine has been increasingly used in the pediatric population because it is an effective treatment both for focal and generalized epilepsies. Hepatotoxicity is uncommon, but hepatic failure and multi-organ failure has been described in adult and pediatric patients taking lamotrigine. 150,151 The dose of the drug needs to be reduced in patients with liver disease, who may have an altered metabolic clearance.
Approaches and Challenges in cDILI Study
Different approaches have been used to characterize cDILI. Mining the worldwide and national spontaneous reports of ADRs 11,14 and systematically reviewing the accumulated cDILI cases 9,10 have proven useful in finding the culprit drugs and defining the relative risks for children as compared to adults. Animal models have provided some mechanistic insights into why children respond differently to a hepatotoxic drug compared to adults. 75,92,106 However, integrating the data from different approaches will be a formidable challenge. The major problems will be the lack of data, the difficulties involved in establishing animal models for cDILI, and the uncertainties in animal-to-human extrapolation. It should also be stressed that age is not an independent factor. It often interacts with other factors such as the sex of the child making them different from adults in terms of DILI. 7,80
Emerging technologies, such as genome-wide association study and omics (genomics, proteomics, and metabolomics), provide promising approaches to help understand the mechanisms and risk factors for DILI. 111,146,152,153 These novel approaches have not been extensively introduced into cDILI study, but it might represent a future direction. It is also anticipated that the regulatory agencies will play a more active role in cDILI study as more data are accumulated.
Currently, there are no diagnostic biomarkers for DILI. 23 Blood and urinary microRNAs, blood bile acids, and serum NAPQI (acetaminophen metabolite) adducts were recently found to be able to reflect acetaminophen-induced liver injury in children. 154,155 However, these findings might be acetaminophen-specific and need to be confirmed in large cohorts of patients. The majority of DILI biomarker studies focused only on aDILI, and future investigations should take cDILI into considerations.
Conclusions
Drug-induced liver injury in children is a small subset (<10%) of all DILI cases but accounts for ∼20% of acute liver failures in children. 156 Depending on the implicated drugs, children may have higher or lower susceptibility to DILI than adults, and such differential susceptibility has been reproduced in animal models for a few drugs. However, for most drugs, it remains unknown whether children differ significantly from adults with respect to DILI. Available mechanistic data are predominantly for aDILI but not cDILI. There is no regulatory guidance in place to deal with cDILI, possibly due to the lack of data. More studies are needed to better predict the potential of cDILI and identify the risk factors for cDILI, so that potentially hepatotoxic drugs can be used safely in children. Drug-induced liver injury in children, as DILI in general, is very often of low incidence and clinical trials usually do not include enough number of patients for the identification of risk factors. More vigorous postmarketing surveillance, such as mandatory report of cDILI cases after initial marketing approval, and multicenter collaborative efforts at the international level, such as the Pediatric Acute Liver Failure Study (https://www.palfstudy.org), are needed to better understand the risk factors for cDILI.
Footnotes
Authors’ Note
This article is not an official guidance or policy statement of the US Food and Drug Administration (FDA). No official support or endorsement by the US FDA is intended or should be inferred.
Author Contributions
William F. Salminen contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. Alec T. Salminen contributed to design, contributed to acquisition, analysis, and interpretation, and critically revised the manuscript. Gary M. Russotti contributed to design, contributed to interpretation, and critically revised the manuscript. Qiang Shi contributed to conception, contributed to acquisition and analysis, and drafted the manuscript. Xi Yang contributed to conception, contributed to acquisition and analysis, and drafted the manuscript. James J. Greenhaw contributed to design, contributed to acquisition, and drafted the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the US FDA’s Office of Women’s Health.
