Abstract
More than 80,000 chemicals are in commercial use worldwide. Hepatic metabolism to toxic intermediates is often a key mechanism leading to tissue damage and organ dysfunction. Effective treatment requires prompt detection of hepatotoxicity, ideally with rapid, minimally invasive diagnostic assays. In this study, archetypal histologic features of chemically induced hepatic injury were compared with clinical chemistries (including liver enzymes) and serum concentrations of microRNA-122 (miR-122, the processed form miR-122-5p), a biomarker of liver injury. The hepatotoxicants 4,4’-methylenedianiline (4,4’-MDA), allyl alcohol (AA), or carbon tetrachloride (CCl4) were orally administered to male Sprague-Dawley rats for 1, 5, 14, or 28 days to induce liver damage. Formalin-fixed, paraffin-embedded liver sections were evaluated histologically for inflammation, fibrosis, necrosis, and lipid accumulation. Liver enzymes were measured in serum, and serum miR-122 concentrations were assessed by quantitative polymerase chain reaction (qPCR). Histologic features of hepatic injury dose-dependently increased in both severity and frequency. Increases in liver enzymes and bilirubin were more pronounced in response to AA or 4,4’-MDA than to CCl4 at early time points. Elevated serum miR-122 levels in animals administered CCl4, AA, or 4,4’-MDA were more strongly associated with degree of hepatic histopathology than with dosage. Given this sensitive expression pattern postexposure, liver-specific miR-122 may improve the diagnostic accuracy of early hepatic injury.
The liver is a primary site of xenobiotic metabolism, and exposure to environmental toxicants and pollutants can induce hepatotoxicity directly or following conversion to toxic metabolites (Casarett and Klaassen 2008). In response to toxicant exposure, hepatocytes may accumulate lipid, leading to functional deterioration, necrosis, and replacement with fibrosis (Bataller and Brenner 2005). Initial changes in hepatocellular architecture are often reversible, but continued exposure may overwhelm detoxification mechanisms and lead to chronic inflammation, fibrosis, cirrhosis, and, occasionally, neoplasia (Arias 1982). Development of a rapid means to assess hepatic injury after chemical exposure but prior to the development of clinical symptoms is a key strategic goal for biomarker discovery and diagnostic assay development according to U.S. Guidance for Industry (Burkhardt et al. 2011).
Recent literature has identified small noncoding RNA molecules, termed microRNAs (miRNAs or miRs), that contain a single hairpin loop when transcribed. miRs are processed to form short molecules (22–23 nucleotide in length), which are complementary to messenger RNA (mRNA). miRs affect protein abundance primarily by regulating mRNA stability and/or translation (Valinezhad Orang, Safaralizadeh, and Kazemzadeh-Bavili 2014). Like posttranslational modifications, miRNAs have been shown to modulate gene and protein expression in a highly specific manner. Increased tissue expression of miRs correlates with decreased expression of the corresponding mRNA, leading to functional repression. miR expression is often highly tissue-specific, suggesting that miRs may be candidates for more specific and sensitive biomarkers. miRs specific for cardiac, liver, and renal endpoints hold promise for establishing noninvasive, early indicators of toxic organ injury (Cardenas-Gonzalez et al. 2017; Cheng et al. 2012; Wang et al. 2009). A significant increase in serum concentrations of miR-122 above background or control levels has been shown to be an especially specific and sensitive indicator of liver injury (Starckx et al. 2013). For the most part, the presence of miRNA-122 in serum correlates with general hepatocellular injury but does not denote a specific pathology or pathogenesis. The time course of miR concentration in serum depends on the severity and duration of injury (Tan et al. 2014). In this study, serum miR-122 levels were evaluated in rats by assessing histopathological changes observed after exposure to hepatotoxic industrial chemicals over enough time to permit development of archetypal injury. Escalating doses of the hepatotoxicants such as 4,4’-methylenedianiline (4,4’-MDA), allyl alcohol (AA), or carbon tetrachloride (CCl4) were orally administered to male Sprague-Dawley rats aged 8 weeks for 1, 5, 14, or 28 consecutive days. miR-122 levels were phenotypically anchored to histopathology scores and demonstrate that miR-122 levels were more strongly associated with histopathologic severity and serum liver enzyme levels than with toxic dose throughout the study.
Method
Chemicals
AA (CAS #107-18-6), 4,4’-MDA (CAS #101-77-9), and CCl4 (CAS #56-23-5) were purchased from Sigma Chemicals (St. Louis, MO). Corn oil (CAS #8001-30-7) was from MD Biomedicals, LLC (Salem, OH).
Animals and Dosing
Experimental Design.
Male Sprague-Dawley rats (CD IGS [CRL: CD (SD)]) were purchased from Charles River Laboratories (Raleigh, NC) at six weeks of age. Rats were dually housed in temperature- and humidity-controlled rooms on a 12-hr light/dark cycle. Animals were allowed
Range-finding studies were conducted at ILS to determine tolerable dosages of test chemicals that resulted in hepatic injury after 1, 5, 14, or 28 days of oral gavage. Groups of five male, eight-week-old Sprague-Dawley rats were administered AA at doses of 4.5 mg/kg, 9.7 mg/kg, 20.9 mg/kg, or 45.0 mg/kg for one or five days. Five additional rats were administered 45.0 mg/kg for 14 days. Then 4,4’-MDA was administered at 8.1 mg/kg, 22.0 mg/kg, 59.7 mg/kg, or 162.0 mg/kg for one or five days, and CCl4 was administered at doses of 20.0 mg/kg, 43.1 mg/kg, 92.8 mg/kg, or 200.0 mg/kg for five days, or 200.0 mg/kg for 14 days. Vehicle control animals were administered corn oil. The one- and five-day range-finding AA, CCL4, and 4,4’-MDA experiments were performed by ILS as previously described (Ippolito et al. 2016).
An additional range-finding study and definitive studies with AA and CCl4 were performed at USACEHR. A 12-hr light/dark cycle was maintained in animal rooms following a normal diurnal pattern. Rats were housed either two or three per cage in accordance with space recommendations outlined in
Histopathology
Formalin-fixed, paraffin-embedded liver specimens were sectioned to 4 to 5 µm thickness. For studies conducted at ILS, ILS histology technicians processed the tissues and performed histochemical staining. For studies conducted at USACEHR, histology technicians at the U.S. Army Public Health Center Division of Toxicologic Pathology processed and stained the tissues. Both laboratories stained sections with hematoxylin and eosin, Oil Red O, or Masson’s trichrome. Oil Red O staining was performed on flash-frozen sections. All tissues were evaluated by board-certified anatomic veterinary pathologists (at ILS, M.H.B.; at USACEHR, E.E.C.). The pathologists were blinded to the biomarker data but not to the treatment groups, as recommended by the Standards for Reporting of Diagnostic Accuracy initiative (Burkhardt et al. 2011). Microscopic evaluations were the result of the combinatorial average of two assessment criteria: (1) overall severity of the affected tissue as subjectively evaluated by the pathologist, including the complexity or context of the morphologic change and (2) the amount of tissue affected on the slide when considering gross observations, including the distribution of the pathology as described in Thoolen et al. (2010). A generic histopathologic grading scheme was used similar to the criteria specified in Schafer et al. (2018): none (0), minimal (1), mild (2), moderate (3), or marked/severe (4). Pilot experiments that used identical doses of toxicant were performed to verify that the two laboratories’ scoring systems were comparably assessing severity of tissue injury.
Clinical Chemistries
Clinical chemistry values were determined using the Vetscan Mammalian Liver Profile reagent rotor on the Abaxis Vetscan 2 per the manufacturer’s instructions (Abaxis, Union City, CA). Blood was collected by cardiac puncture 24 hr after the last day of toxicant administration and was analyzed for liver enzyme activity. The activity of alanine aminotransferase (ALT) and alkaline phosphatase (ALP) was measured in serum from rats in all range-finding and definitive studies, with the exception of AA doses ≥27 mg/kg for 14 days. Given the lack of pathology at the lower doses in the definitive study at 14 days, liver enzymes were only evaluated for the high dose of AA (20.9 mg/kg) relative to control. Data were analyzed by either dose or histological severity score as the dependent variable.
miR-122 Isolation from Serum
Total RNA was isolated from rat serum using Qiagen’s miRNeasy Serum/Plasma kit (Cat No 217184) following the manufacturer’s instructions with 200 µl serum input and a final elution volume of 14 µl. The optional miRNeasy Serum/Plasma Spike-In Control (Cat No 219610) and corresponding Ce_miR-39_1 miScript PrimerAssay were also incorporated following manufacturer’s instructions. Samples were quantitated using 1 µl eluate on a NanoDrop 8000 (ThermoFisher, Waltham, MA).
Real-time Quantitative Reverse Transcription Polymerase Chain Reaction (RT-PCR) Analysis of miR-122
In order to quantify miR-122 in the serum samples, the qScript™ microRNA cDNA Synthesis Kit (QuantaBio, Beverly, MA) was utilized to convert miRNA to cDNA from 50 ng total RNA input following the manufacturer’s instructions. The full hour was utilized for the PolyA tailing reaction rather than the optional 20 min. At the completion of the reaction, 20 µl of RNase free water was added to dilute the cDNA reaction.
The miR-122 (primer sequence: CAACGCCATTATCACACTAAATAAA) and U87 (primer sequence: CCAGCTGAGGGTTTCTTTGA; reference control for normalizing) targets were quantified using SYBR Green quantitative reverse transcription polymerase chain reaction (qRT-PCR) amplification reactions with PerfeCTa microRNA Assays (QuantaBio) for each. Real-time qRT-PCR amplification was performed on an ABI 7500 (Applied Biosystems, Foster City, CA) following the manufacturer’s instructions using 9 µl diluted cDNA in a 25 µl reaction with a three-step cycling protocol (95°C for 2 min and 40 cycles of 95°C for 5 s, 60°C for 15 s, and 70°C for 15 s).
Statistical Analysis
GraphPad Prism (version 7.03) was used to determine statistical significance at an α of 0.05. A Bartlett’s
Results
All doses of 4,4’-MDA and CCl4 were sublethal; animals showed no signs of systemic clinical toxicity. All animals administered AA at 45.0 mg/kg for 14 days exhibited clinical signs of toxicity characterized by body weight loss and lethargy (Online Supplemental Tables 1 and 2). One animal experienced body weight loss that exceeded the threshold for euthanasia (i.e., >25%). It was found moribund and euthanized before study termination (Online Supplemental Tables 1 and 2; Online Supplemental Figures 1 and 2). Weight loss recorded for the remaining four animals approached the threshold for wasting syndrome (20%) by the end of the 14-day dosing period.
Histological Evidence of Hepatotoxicity
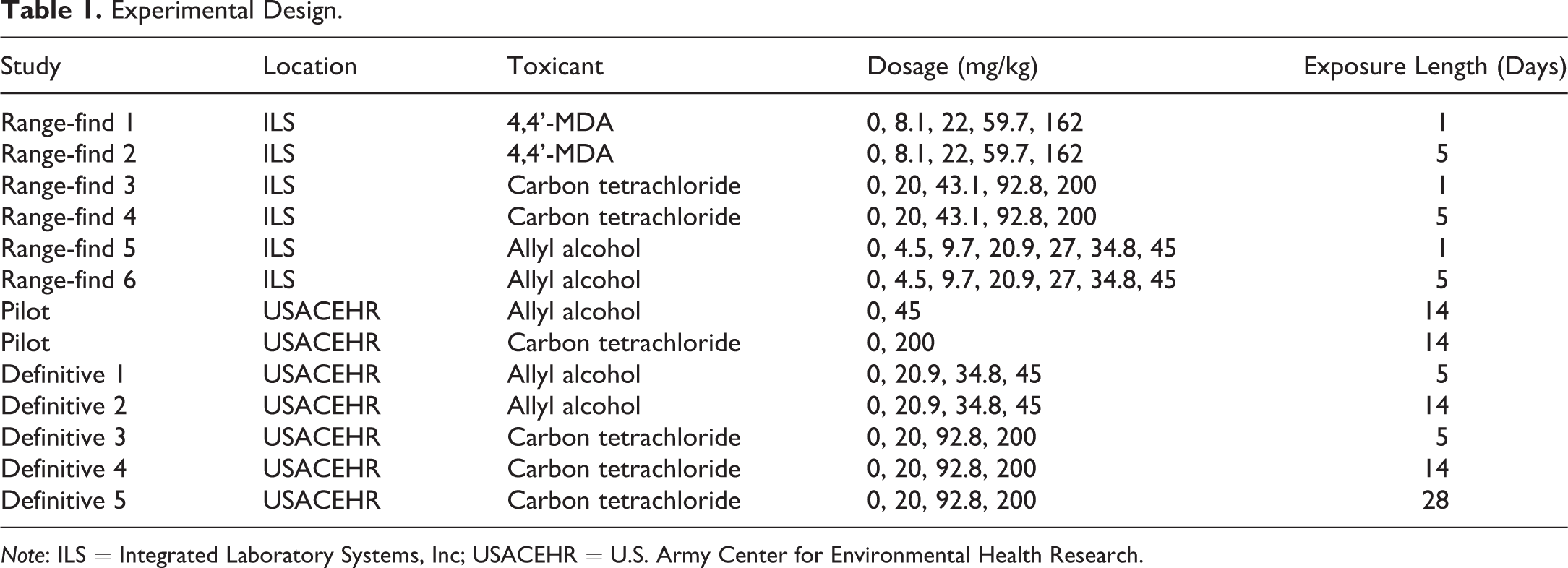
Histological evidence of hepatotoxicity was evident in all chemicals tested in a dose- and time-dependent manner (see Online Supplemental Tables 3 and 4). All four surviving animals administered 45 mg/kg AA for 14 days exhibited proliferation of fibroblasts (Figure 1). Based on these data, doses of 4.5, 9.7, and 20.9 mg/kg AA were chosen for subsequent studies at 5 or 14 days of administration. Animals administered 4.5 or 9.7 mg/kg AA for five days lacked visible proliferation of fibroblasts, but degeneration and necrosis were apparent (Figure 1C and D). Animals administered AA for 14 days (Figure 1E and F) developed fibrosis in a dose-dependent manner (verified by Masson’s trichrome staining). After 14 days of AA administration, fibrosis was often associated with minimal lymphoplasmacytic or neutrophilic portal infiltrates.
Representative rat liver sections showing biliary hyperplasia, proliferation of fibroblasts, cystic degeneration, and necrosis after AA administration (45 mg/kg, 5 or 14 days). (A) Vehicle control, day 5. (B) Vehicle control, day 14. AA was administered to male Sprague-Dawley rats for 5 days (C, D) or 14 days (E, F) by oral gavage. At day 5, a mixed mononuclear and granulocytic infiltrate (inflammation) surrounded several portal tracts (C). At higher magnification, hepatocytes were pale, swollen with variably vacuolated cytoplasm (degenerate) or hypereosinophilic, shrunken, and pyknotic (necrotic) (D). By day 14, marked portal bridging (arrows) (E) comprised of biliary hyperplasia with inflammation was present (F). Hematoxylin and eosin stained. AA = allyl alcohol.
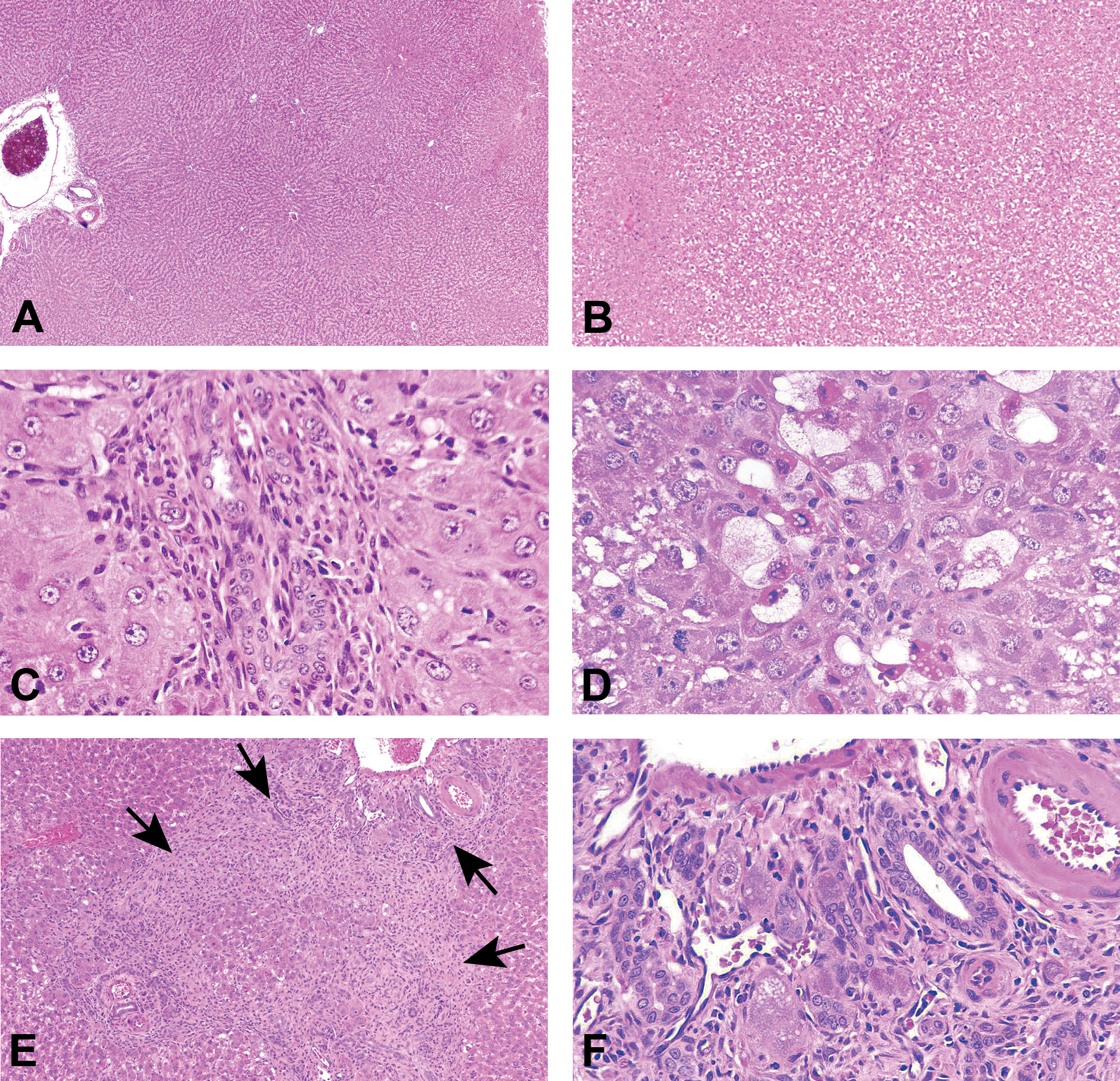
Fibrosis was evident in a dose-dependent manner after five days of 4,4’-MDA administration (Figure 2). Inflammatory infiltrates accompanied fibrosis in all affected animals.
Hepatobiliary fibrosis after 4,4’-MDA administration (5 days, 162 mg/kg) (A, B). Liver from male Sprague-Dawley rats administered 4,4’-MDA by oral gavage for one day. Hepatocellular cytoplasmic alteration, observed in day 1 animals, was characterized by increased granularity and eosinophilia in periportal areas. Subacute inflammation and mitotic figures are denoted by arrows in panels A and B, respectively. (C, D) Oval cell hyperplasia, subacute inflammation, hepatocellular necrosis, and bile duct hyperplasia and fibrosis were present at day 5. Hematoxylin and eosin stained. 4,4’-MDA = 4,4’-methylenedianiline.
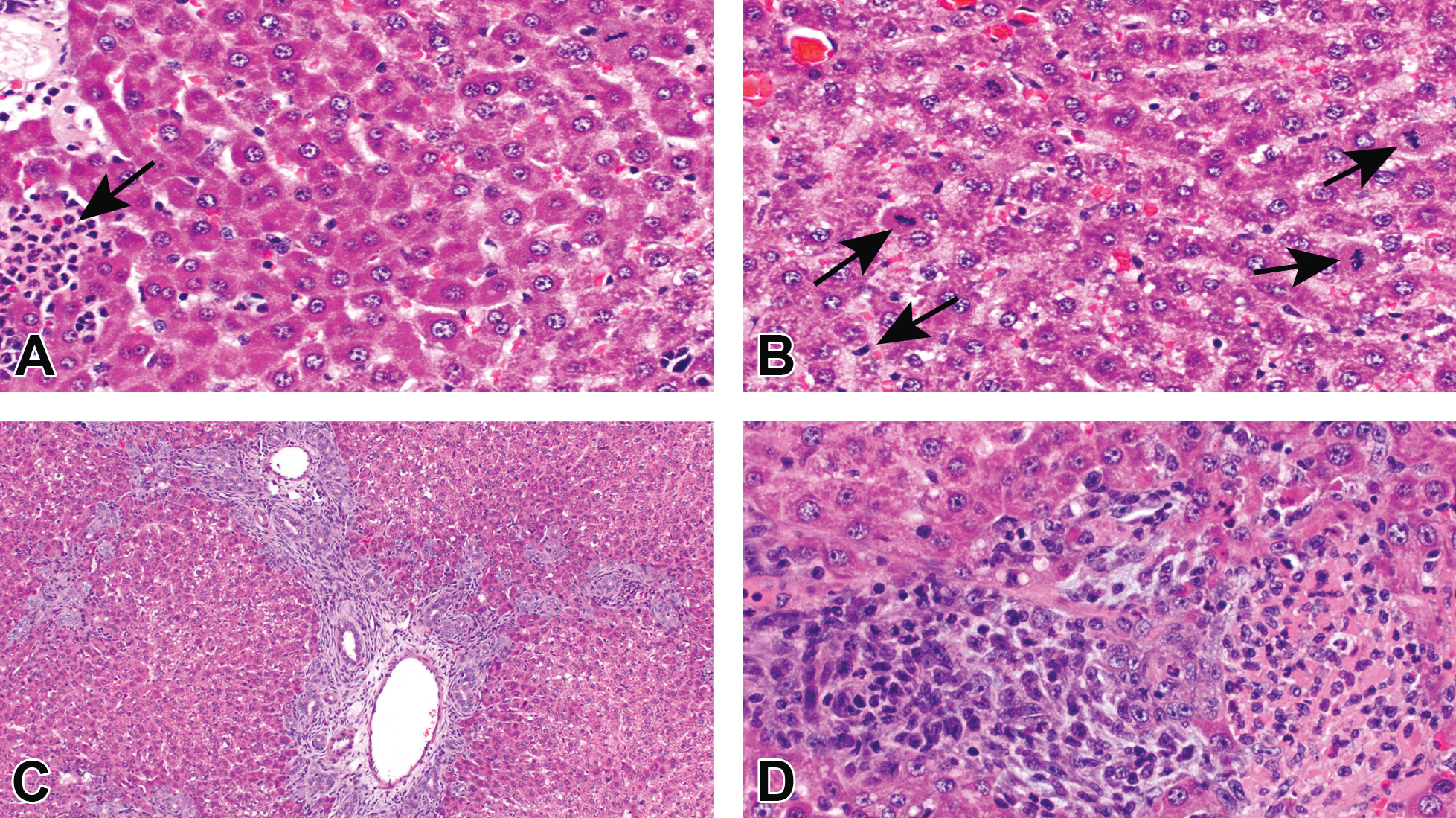
CCl4 administration (20.0, 43.1, 92.8, and 200.0 mg/kg/day for 1, 5, 14, or 28 days) resulted in a dose- and time-dependent increase in hepatocellular vacuolation (Figure 3). These vacuoles were confirmed to contain lipid by Oil Red O histochemical staining. CCl4 administration for five days resulted in increased Masson’s trichrome staining (reflecting collagen accumulation) and exhibited fibroblast proliferation that surrounded centrilobular veins in a dose-dependent manner. After 14 days at 200 mg/kg CCl4, all five animals had minimal increases in centrilobular collagen (fibrosis) and minimal to marked micro- and macrovesicular hepatocellular vacuolation (cystic degeneration). By 28 days, fibrosis was evident in the majority of the animals administered CCl4 (Figure 3E and F), and all high-dose animals had minimal to moderate hepatocellular cystic degeneration and single-cell hepatocellular necrosis. Proliferation of oval cells and biliary hyperplasia were also evident; bridging fibroblast proliferation occurred only in the medium- and high-dose groups. Masson’s trichrome staining for fibrosis was positive in centrilobular areas in one animal at 20.0 mg/kg, one at 92.8 mg/kg, and five at 200.0 mg/kg. At 28 days, single-cell necrosis and mitotic figures were present in a dose-dependent manner.
CCl4 oral administration (200 mg/kg) for 28 days is associated with portal and centrilobular fibrosis. CCl4 was administered to male Sprague-Dawley rats for 5 days (A, B), 14 days (C, D), or 28 days (E, F) by oral gavage. Macro- and microvesicular hepatocellular vacuolation occurred early in a centrilobular to mid-zonal pattern with later development of portal and centrilobular fibrosis. Fibrosis was confirmed by Masson’s Trichrome histochemical staining (F). CCl4 = carbon tetrachloride.
Liver Enzymes Were Associated with High Doses, Long Exposures, and Microscopic Evidence of Liver Injury
AA elicited the greatest variability in serum levels of liver enzymes (ALT and ALP) among individual animals in both studies (Figure 4). AA administration resulted in statistically significantly elevated ALT and ALP levels only after five days of 45.0 mg/kg (Figure 4A and B). There were no statistically significant changes at 14 days at 20.9 mg/kg relative to control. When liver enzyme activities were analyzed with histology score as the independent variable (Figure 4C), ALT and ALP were positively associated with score. Similarly, CCl4 was associated with statistically significant increases in liver enzyme activity only at the highest dosages and longer time points (Figure 5A and B). When enzyme activity was analyzed with histology score as the independent variable, statistically significant elevations in ALT and ALP were observed in animals with severe pathology (scores 3–4, Figure 5C). The highest doses of 4,4’-MDA resulted in elevated ALT and ALP liver enzymes on day 5 (Figure 6A and B). Increases in liver enzyme activity were more closely associated with histology score than with dose (Figure 6C).
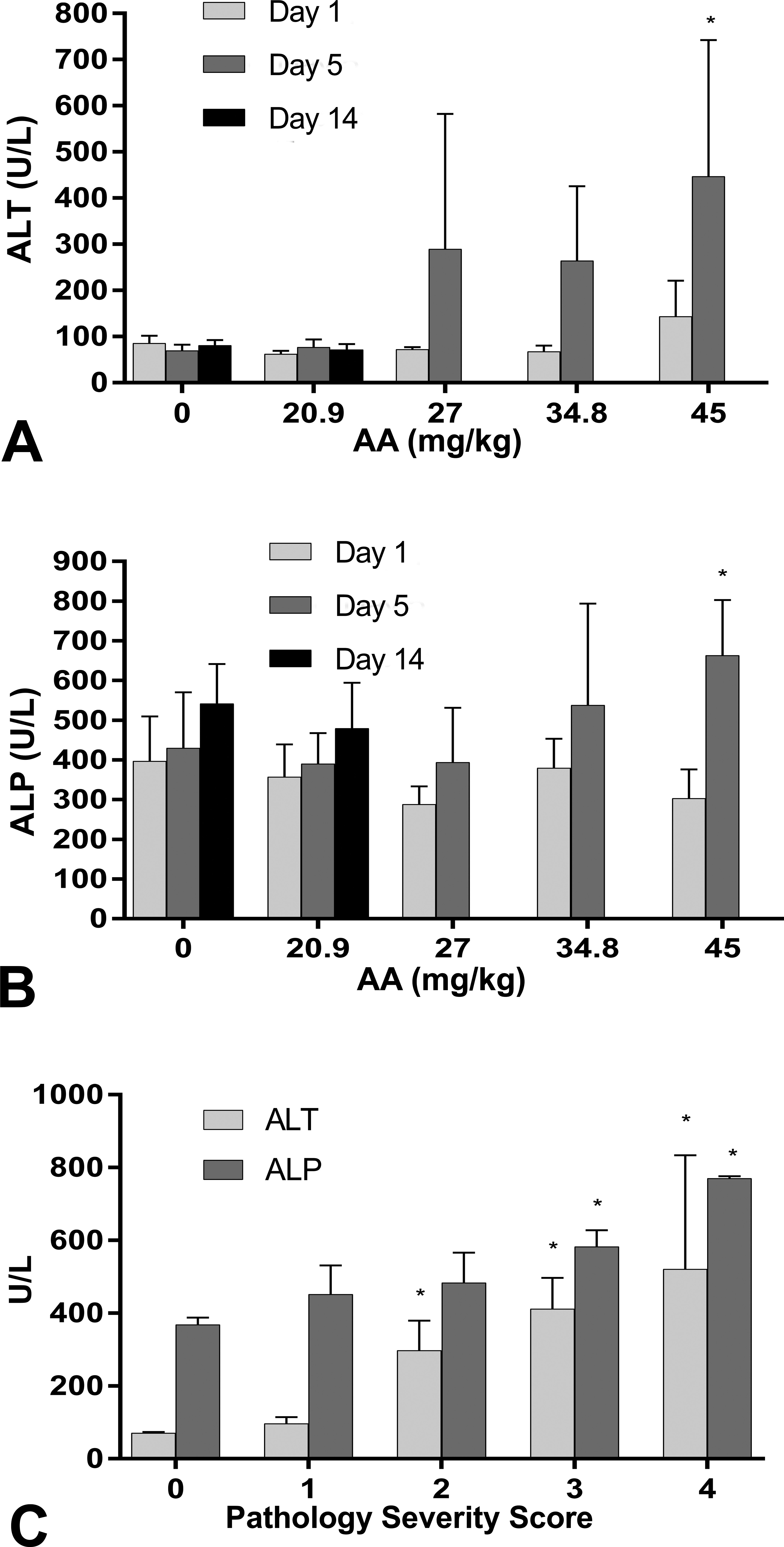
Liver enzymes are elevated after AA administration. (A) Mean serum ALT and (B) serum ALP levels are shown for rats receiving AA for 1 day, 5 days, or 14 days by oral gavage. (C) Mean serum ALT and ALP activities and severity scores are shown. The association between enzyme levels and severity score was tested using Kruskal–Wallis ANOVA with post hoc Dunnett’s multiple comparisons test (*
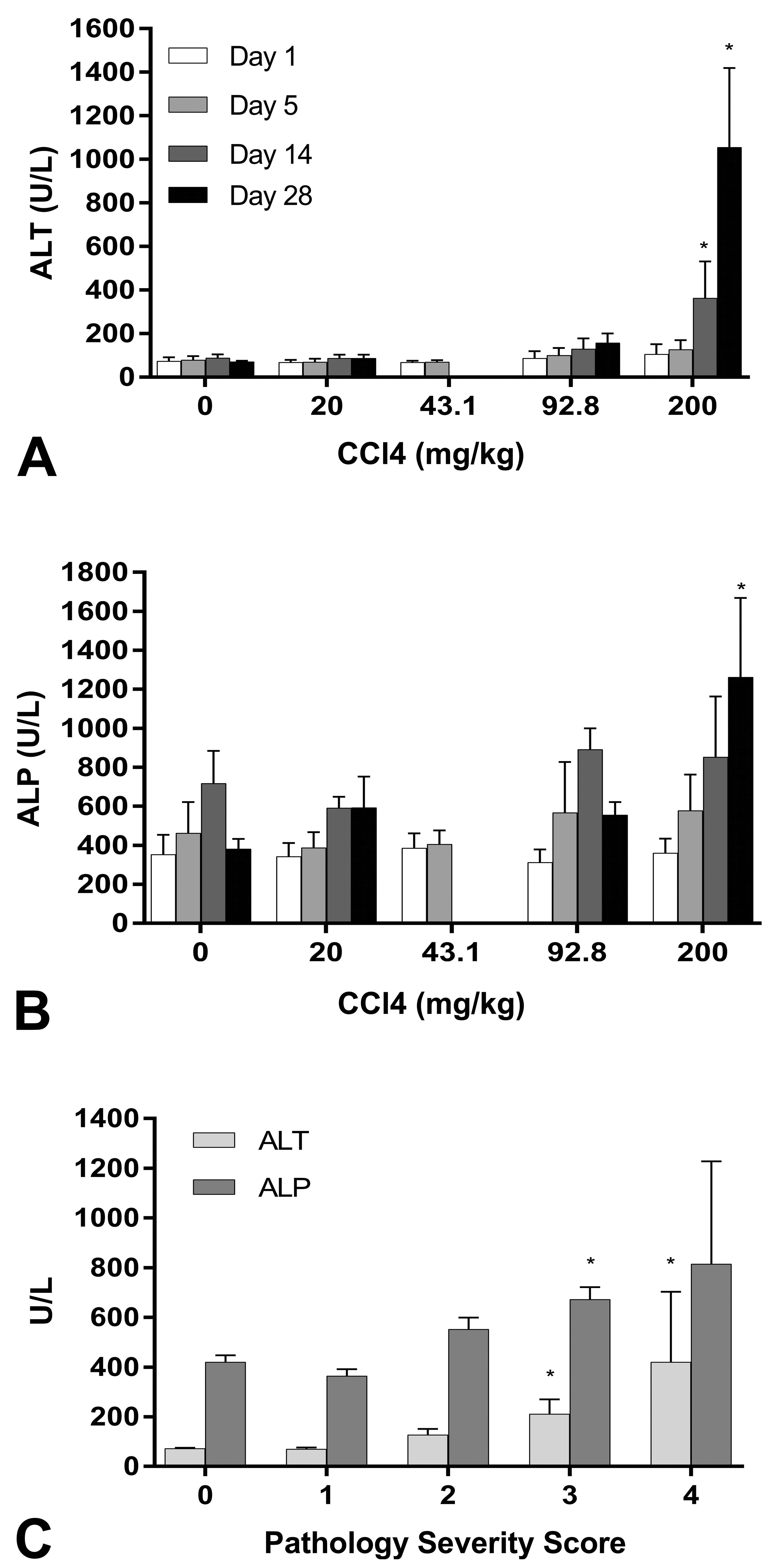
Liver enzymes are elevated after CCl4 administration. (A) Serum ALT and (B) serum ALP were measured after CCl4 was administered to male Sprague-Dawley rats for 1 day, 5 days, 14 days, or 28 days by oral gavage. (C) Serum ALT and ALP activities were plotted with histopathological score as the independent variable (*
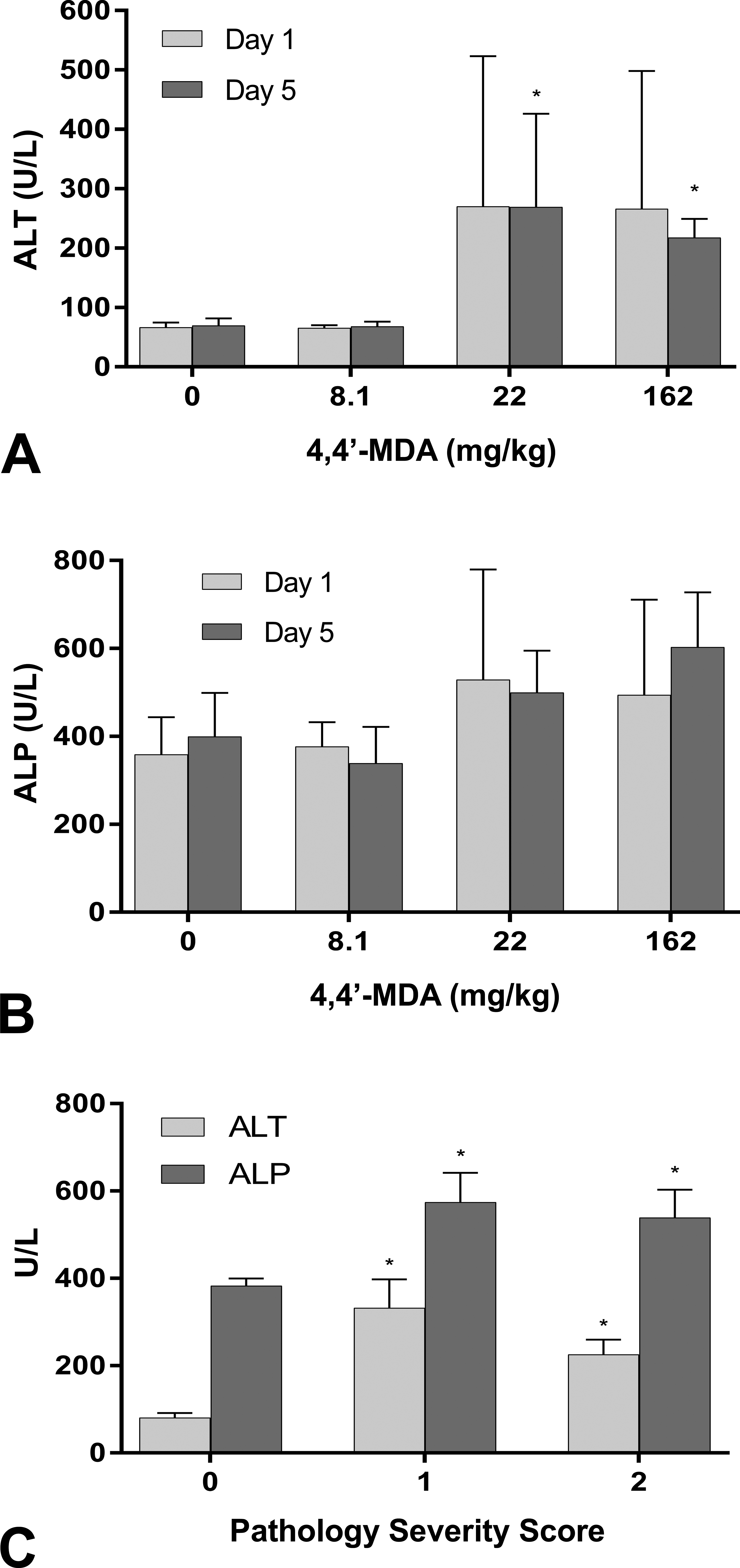
Liver enzymes are elevated after 4,4’-MDA administration. (A) Serum ALT and (B) serum ALP activities were measured after 4,4’-MDA was administered to male Sprague-Dawley rats for 1 day or 5 days by oral gavage. (C) Serum ALT and ALP activities were plotted with histopathological score as the independent variable. *
miR-122 Is an Early Indicator of Hepatic Injury Under High Dose, Acute Exposure Conditions
Serum miR-122 concentration was a better predictor of histopathology than dose in all exposure groups. Serum miR-122 levels after AA administration showed greater interindividual variability when plotted as a function of dose (Figure 7A) than when plotted as a function of pathology severity score, either by fibrosis end points alone (Figure 7B) or all histologic scores (Figure 7C). After CCl4 administration, miR-122 levels were significantly higher than controls as early as one day after exposure (Figure 8A). Serum miR-122 levels were less closely associated with fibrosis scores (Figure 8B) than with composite histopathology scores (Figure 8C). Similarly, 4,4’-MDA administration was associated with elevated serum miR-122 as early as one day after administration of 22.0 mg/kg (Figure 9A). Serum miR-122 levels were comparably elevated at all histopathology scores >0 after 4,4’-MDA administration, either compared to fibrosis alone (Figure 9B) or to the total histopathology score (Figure 9C). When data from all three chemical exposures were combined and plotted as a function of pathology severity score (Figure 10A) or exposure duration (Figure 10B), serum levels of ALT, ALP, and miR-122 were positively associated with pathology severity score and exposure duration.
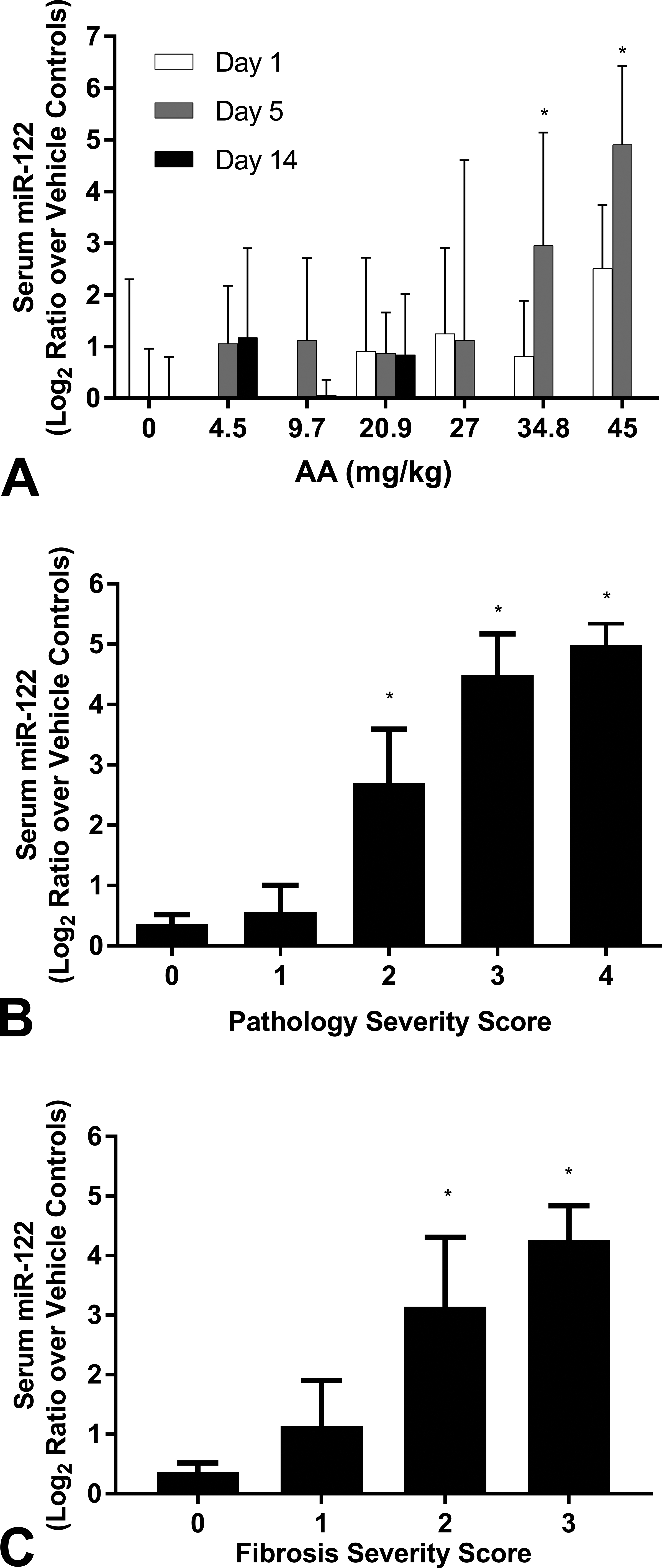
Serum miR-122 levels are elevated after AA administration. (A) Serum miR-122 levels were plotted with (A) dose, (B) histopathological score (fibrosis-specific), or (C) histopathological score (all histopathologies) as the independent variable after AA administration. See Supplemental Figure 1 for segregation by score and day. *
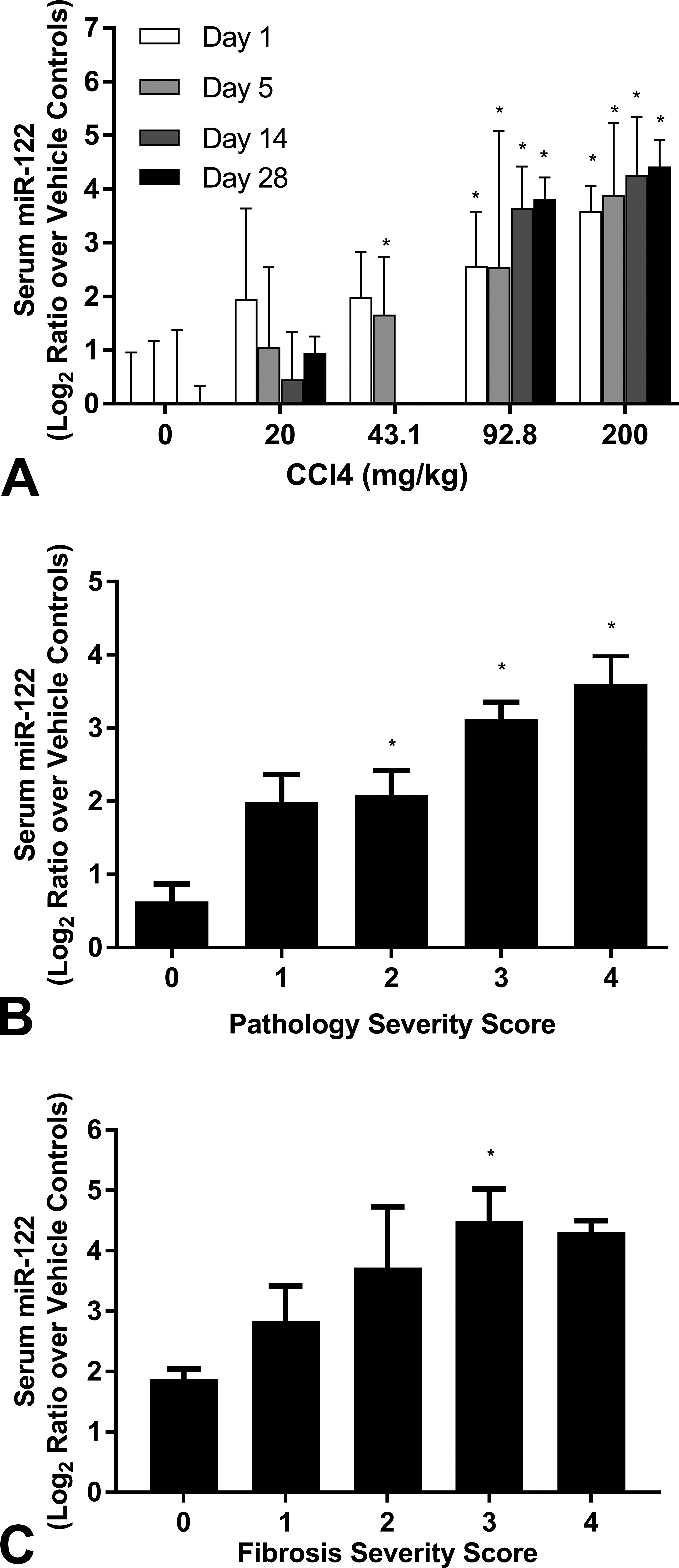
Serum miR-122 levels are elevated after CCl4 administration. (A) Serum miR-122 levels were plotted with (A) dose, (B) histopathological score (fibrosis-specific), or (C) histopathological score (all histopathologies) as the independent variable after CCl4 administration. See Supplemental Figure 2 for segregation by score and day. *
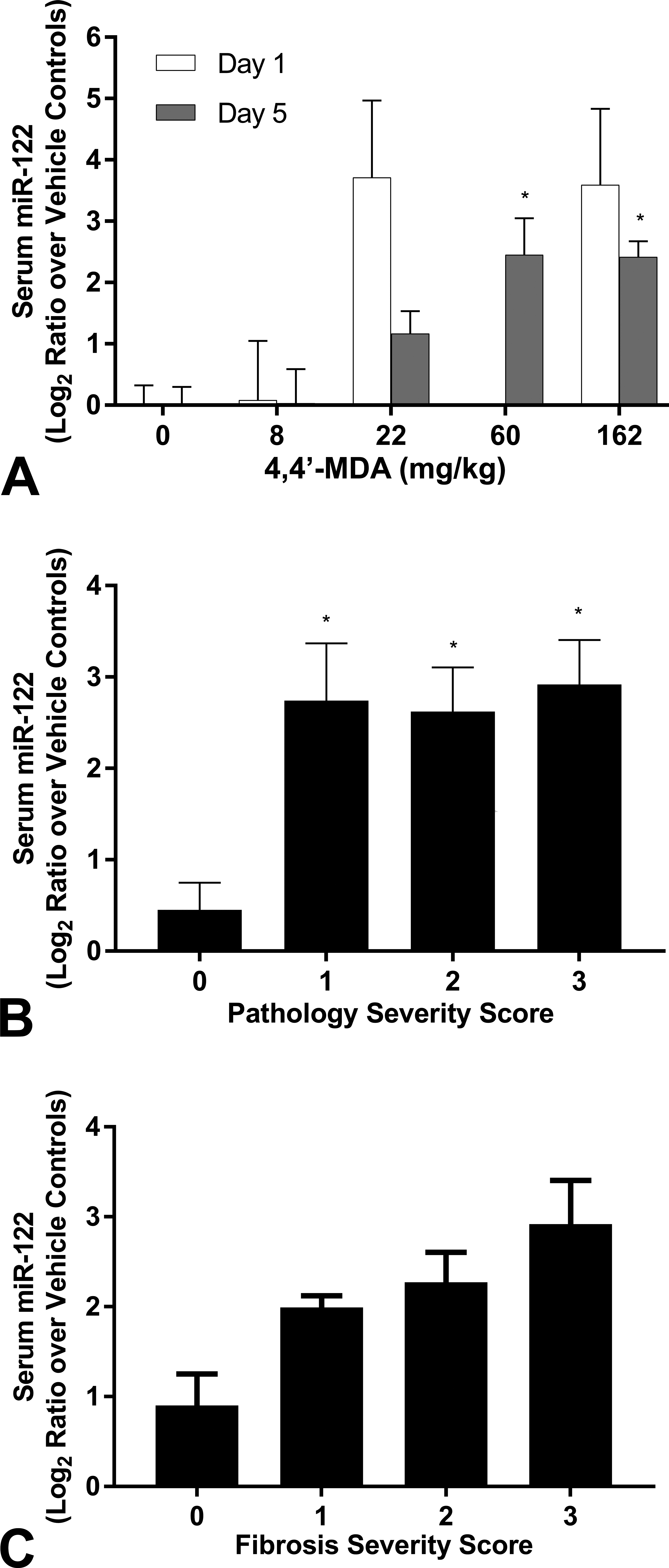
Serum miR-122 levels are elevated after 4,4’-MDA administration. (A) Serum miR-122 levels were plotted with (A) dose, (B) histopathological score (fibrosis-specific), or (C) histopathological score (all histopathologies) as the independent variable after 4,4’-MDA administration. See Supplemental Figure 3 for segregation by score and day. *
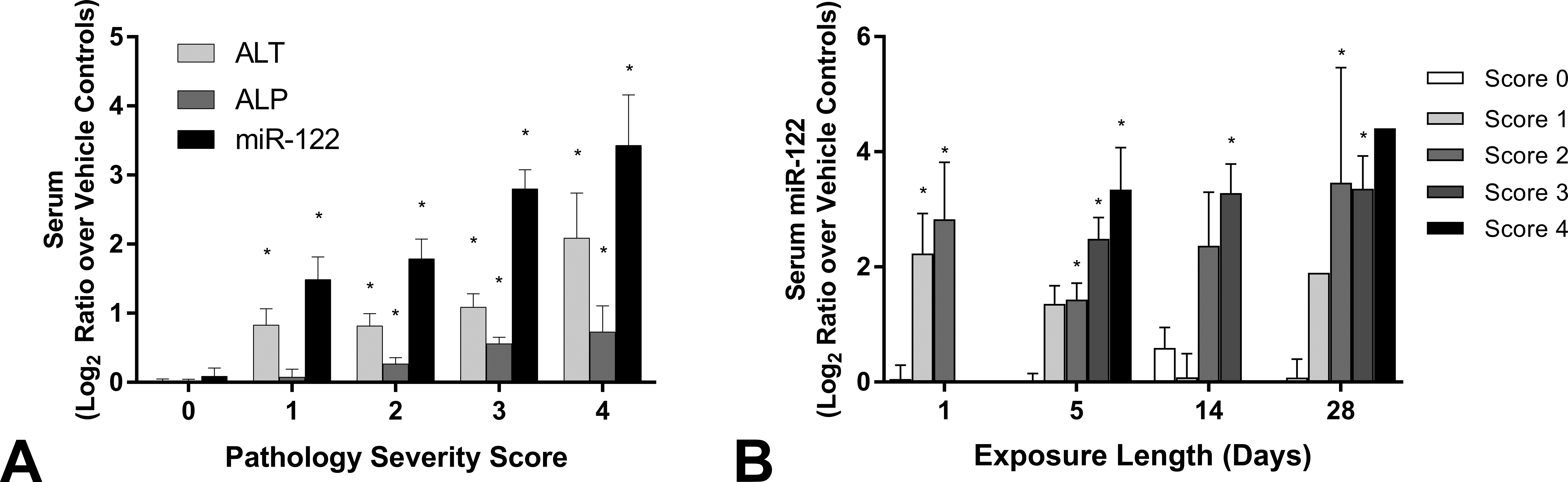
Serum miR-122 levels are a sensitive indicator of liver injury resulting from diverse chemical exposures. Serum miR-122, ALT, and ALP activities were normalized to U87 internal standards or miR-16 and plotted as a percentage of the average vehicle control. (A) The association of serum ALT, ALP, and miR-122 log2 difference levels with pathology severity scores was tested using Kruskal–Wallis ANOVA with a Dunnett’s post hoc multiple comparison correction (*
Discussion
This study demonstrates that serum miR-122 concentrations are associated with hepatic histopathology and increases of ALT and ALP enzyme activity after acute to subchronic exposure to archetypal hepatotoxicants. Increases in miR-122 associated better with histopathologic severity than with dose, especially after AA administration.
Hepatic fibrosis was the most deleterious microscopic change observed in this study. All three toxicants used in this study have been shown to cause hepatic fibrosis in prior reports (Ippolito et al. 2016). Three primary mechanisms have been proposed for the development of hepatic fibrosis: chronic activation of the wound-healing process, oxidative stress, and derangement of epithelial–mesenchymal interactions (especially in cholangiopathies; Ismail and Pinzani 2009). The most common mechanism is chronic hepatic injury with repeated attempts to replace lost cells. This process is characterized by necrosis and/or apoptosis, inflammation, and the activation of cells capable of producing extracellular matrix proteins, such as hepatic stellate cells, liver progenitor cells, and portal myofibroblasts. Although necrosis was not a prominent feature observed in the definitive study, it generally accompanied prominent lipid-like vacuolation that was confirmed to be lipid by Oil Red O histochemical staining. Necrosis and inflammation were key features in animals administered 45.0 mg/kg AA for 14 days, suggesting that these histological features may have stimulated the fibrosis response observed in this dose group. Inflammation was inconsistently present in liver tissue from animals in the definitive study, with the exception of animals administered 200.0 mg/kg CCl4. Interestingly, miR-122 levels were more closely associated with histopathologic changes than with changes in liver enzyme activities after CCl4 treatment, a compound that causes fibrosis only after weeks of dosing. In earlier studies, gene signatures of fibrogenesis were phenotypically anchored to the histopathology (Ippolito et al. 2016). In this study, too, CCl4 administration was associated with weak Masson’s trichrome staining after 14 days, but the Masson’s trichrome staining was more pronounced after higher doses of 4,4’-MDA and AA at five days (Ippolito et al. 2016). Fibrosis was clearly evident at 28 days, supporting a slower fibrogenic process in the case of CCl4 (Lee et al. 2005). CCl4’s mechanism of fibrogenesis is known to result from cytochrome P450-induced activation of reactive oxygen species (ROS) and the generation of CCl3• radicals, principally in centrilobular hepatocytes. Depletion of glutathione results in unopposed ROS generation and contributes to fibrogenesis as a result of hepatocellular necrosis and extracellular matrix deposition around the central veins (Ismail and Pinzani 2009). Subchronic exposure to CCl3• radicals leads to prolonged activation of repair mechanisms, including proliferation of fibroblasts and oval cells leading to fibrosis and biliary hyperplasia, respectively, as evidenced by the histologic changes observed in the 28-day CCl4 exposure group.
AA and 4,4’-MDA exposures were both associated with biliary hyperplasia and bridging portal fibrosis. AA metabolizes to acrolein by alcohol dehydrogenase in periportal hepatocytes, resulting in glutathione depletion and periportal fibrosis (Jaeschke, Kleinwaechter, and Wendel 1987; Moustafa 2001). Similarly, 4,4’-MDA converts to toxic metabolites in the periportal region of the liver, resulting in the portal damage observed in this study and other reports (Zhang et al. 2006). Both chemicals stimulate myofibroblast proliferation and the secretion of cytokines and other molecular inducers of extracellular membrane formation (Zhang et al. 2006). The immune response affects hepatic fibrogenesis. The Th1 immune response tends to be anti-fibrogenic, while the Th2 response tends to be pro-fibrogenic. Th2 immunity activates interleukin (IL)-13, which stimulates fibrogenesis. In addition, the Th2 response stimulates secretion of IL-4, IL-10, granulocyte–macrophage colony-stimulating factor, and matrix metalloproteinase 9 (O’Garra 1998). These cytokines in turn activate macrophages that secrete TGF-β, l-arginine, and l-ornithine, which converts to proline and stimulates proliferation of fibroblasts and collagen production (Durante et al. 2001).
miR-122 is highly liver-specific. Better understanding of changes in miR-122 levels over time when associated with different liver conditions will improve its diagnostic potential. Concordant with recent literature (Starckx et al. 2013; Vliegenthart et al. 2015; Sharapova et al. 2016), our study in rats found that miR-122 elevation occurred soon after hepatotoxicant exposure. In rats, miR-122 levels associated strongly with liver enzymes (i.e., ALT, aspartate aminotransferase, and glutamate dehydrogenase) after exposure to 20 compounds of diverse chemical properties including CCl4 and 4,4-MDA (Sharapova et al. 2016), supporting combining liver enzyme analysis with miR-122 quantification for improved diagnostic accuracy. In mice, miR-122 elevation preceded histological evidence of injury and preceded increased activity of enzymes that are considered to be the clinical “gold standard” of hepatic injury (Starckx et al. 2013). In prior reports, early elevation of miR-122 in response to hepatic injury has been followed by a return to baseline as other miRs increase (e.g., miR-885-5p; Vliegenthart et al. 2015). In this study, miR-122 levels also increased earlier and with greater fold changes than the liver-associated enzymes ALT and ALP in AA-exposed animals, suggesting that miR-122 could provide an earlier and more sensitive indication of hepatic injury than ALT or ALP alone. Further, the observation that miR-122 was more highly associated with histopathology severity score suggests that miR-122 may be a better biomarker of biological effect than chemical exposure alone.
Interindividual variability is inherent in the response to administration of highly volatile compounds (e.g., AA). Response to AA exposure has previously been shown in animal studies to have a poorly characterized dose–response curve (Ippolito et al. 2016). Concordantly, this study found great variation in the AA-induced dose–response curve of miR-122 and liver enzymes compared to that of other chemicals. miR-122 levels associated more closely with histological evidence of injury than with dosage, especially in AA-exposed animals. These features support the utility of miR-122 as a phenotypic biomarker of disease throughout disease progression, especially in the early, preclinical stages of disease. After a single hepatotoxic administration of fibrogenic chemicals such as α-naphthyl isothiocyanate, miR-122 levels returned to baseline shortly after exposure, suggesting the utility of miR-122 as a biomarker in the early stages of fibrotic injury (Church et al. 2016). This study illustrates that oral administration of the toxicants AA, CCl4, and 4,4’-MDA resulted in dose- and time-dependent elevations in serum miR-122 levels after acute and subacute exposures. Thus, miR-122 in combination with other biomarkers may have the potential to concomitantly evaluate exposure and disease progression in cases of toxic liver injury.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623318795435 - Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats
Supplemental Material, DS1_TPX_10.1177_0192623318795435 for Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats by Erica E. Carroll, Danielle L. Ippolito, Matthew G. Permenter, B. Claire McDyre, Christine E. Baer, David M. Kumsher, Molly H. Boyle, Valerie T. DiVito, John A. Lewis, and Jason M. Koontz in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS2_TPX_10.1177_0192623318795435 - Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats
Supplemental Material, DS2_TPX_10.1177_0192623318795435 for Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats by Erica E. Carroll, Danielle L. Ippolito, Matthew G. Permenter, B. Claire McDyre, Christine E. Baer, David M. Kumsher, Molly H. Boyle, Valerie T. DiVito, John A. Lewis, and Jason M. Koontz in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS1_TPX_10.1177_0192623318795435 - Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats
Supplemental Material, TS1_TPX_10.1177_0192623318795435 for Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats by Erica E. Carroll, Danielle L. Ippolito, Matthew G. Permenter, B. Claire McDyre, Christine E. Baer, David M. Kumsher, Molly H. Boyle, Valerie T. DiVito, John A. Lewis, and Jason M. Koontz in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS2_TPX_10.1177_0192623318795435 - Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats
Supplemental Material, TS2_TPX_10.1177_0192623318795435 for Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats by Erica E. Carroll, Danielle L. Ippolito, Matthew G. Permenter, B. Claire McDyre, Christine E. Baer, David M. Kumsher, Molly H. Boyle, Valerie T. DiVito, John A. Lewis, and Jason M. Koontz in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS3_TPX_10.1177_0192623318795435 - Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats
Supplemental Material, TS3_TPX_10.1177_0192623318795435 for Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats by Erica E. Carroll, Danielle L. Ippolito, Matthew G. Permenter, B. Claire McDyre, Christine E. Baer, David M. Kumsher, Molly H. Boyle, Valerie T. DiVito, John A. Lewis, and Jason M. Koontz in Toxicologic Pathology
Supplemental Material
Supplemental Material, TS4_TPX_10.1177_0192623318795435 - Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats
Supplemental Material, TS4_TPX_10.1177_0192623318795435 for Utility of Serum miR-122, Liver Enzymes, and Hepatic Histopathology in Response to Hepatotoxicants in Sprague-Dawley Rats by Erica E. Carroll, Danielle L. Ippolito, Matthew G. Permenter, B. Claire McDyre, Christine E. Baer, David M. Kumsher, Molly H. Boyle, Valerie T. DiVito, John A. Lewis, and Jason M. Koontz in Toxicologic Pathology
Footnotes
Authors’ Note
The views, opinions, and/or findings contained in this report are those of the author(s) and should not be construed as official Department of the Army position, policy, or decision, unless so designated by other official documentation. Citations of commercial organizations or trade names in this report do not constitute an official Department of the Army endorsement or approval of the products or services of these organizations. This research complied with the Animal Welfare Act and implementing Animal Welfare Regulations, the Public Health Service Policy on Humane Care and Use of Laboratory Animals and adhered to the principles noted in
Acknowledgments
The authors would like to thank LTC Vazquez for program management oversight. The authors would like to thank Integrated Laboratory Systems, Inc., for conducting a portion of the
Author Contributions
Authors contributed to conception or design (EC, DI, MP, BM, CB, DK, MB, VD, JL, JK); data acquisition, analysis, or interpretation (EC, DI, MP, BM, CB, DK, MB, VD, JL, JK); drafting the manuscript (EC, DI, MP, BM, JK); and critically revising the manuscript (EC, DI, JK, DK, KJ, HJ, RH, NC). Authors contributed equally to this work (EC, DI). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
