Abstract
The amnesic shellfish toxin, domoic acid, interferes with glutamatergic pathways leading to neuronal damage, most notably causing memory loss and seizures. In this study, the authors utilized a recently developed rat model for domoic acid–induced epilepsy, an emerging disease appearing in California sea lions weeks to months after poisoning, to identify structural damage that may lead to a permanent epileptic state. Sprague Dawley rats were kindled with several low hourly intraperitoneal doses of domoic acid until a state of status epilepticus (SE) appears. This kindling approach has previously been shown to induce a permanent state of epileptic disease in 96% animals within 6 months. Three animals were selected for neurohistology a week after the initial SE. An amino cupric silver staining method using neutral red counterstain was used on every eighth 40 µm coronal section from each brain to highlight neural degeneration from the olfactory bulb through the brain stem. The most extensive damage was found in the olfactory bulb and related olfactory pathways, including the anterior/medial olfactory cortices, endopiriform nucleus, and entorhinal cortex. These findings indicate that damage to olfactory pathways is prominent in a rat model for domoic acid–induced chronic recurrent spontaneous seizures and aggressive behavior.
Introduction
Domoic acid is an environmental neurotoxin that causes seizures and anterograde memory loss in humans (Perl et al. 1990; Teitelbaum et al. 1990). As an analog of glutamate, the toxin prolongs activation of glutamatergic receptors resulting in a neuroexcitation potentiated by increased glutamate release (Novelli et al. 1992) and ultimately neuronal damage (Peng et al. 1994). Previous studies in mice (Peng et al. 1994), rats (Appel, Rapoport, and O' Callaghan 1997; Tryphonas and Iverson 1990), and primates (Scallet et al. 1993; Schmued et al. 1995) have shown the hippocampus to be a primary target for domoic acid toxicity. Damage to the brain in experimental animals can be extensive, especially throughout projections to the limbic region. When more rostral regions of the brain are analyzed, damage is also reported in neurons of the olfactory regions and septum (Colman et al. 2005; Peng et al. 1994; Schmued et al. 1995; Tryphonas and Iverson 1990).
The most thorough investigation to date into domoic acid–induced neuronal damage comes from the Colman et al. (2005) mapping of neurodegeneration in a mouse brain. The mouse received a single LD50 dose of 4 mg/kg domoic acid, and the brain was analyzed using histochemistry with a modified cupric silver method. This stain identifies damage at the level of dendritic terminals, cell bodies, and axons. Cupric silver histochemistry can be observed at a wide range of magnification allowing for a global view of damaged neuronal pathways to identify neuronal damage throughout the brain (De Olmos, Beltramino, and De Olmos De Lorenzo 1994; Switzer 2000). Colman et al. found extensive damage in the olfactory regions and hippocampus pointing to key areas of domoic acid poisoning.
Domoic acid poisoning is also responsible for several thousand sea lions stranding with seizures on California beaches between 1998 and 2005 (Gulland 2000). Over this 8-year period, analysis of stranding sea lions with seizures revealed the emergence of an epilepsy-like syndrome occurring weeks to months after poisoning in approximately 20% of the study group (Goldstein et al. 2008). This syndrome in sea lions showed close similarities to the experimental kainic acid model in rats for temporal lobe epilepsy (Ramsdell and Stafstrom 2009) and promoted interest in laboratory-based investigation into longer term consequences of domoic acid poisoning (Ramsdell 2010). The direct connection between domoic acid poisoning and epileptic disease could only be theorized due to the difficulty in confirming prior exposure in free-ranging populations. Creating a laboratory model for a potential disease condition was complicated, as a single experimental exposure to domoic acid, at levels sufficient to induce a seizing state of status epilepticus (SE), is characteristically lethal. To combat lethality, doses were lowered or administered in the presence of agents to control seizures, but failed to lead to long-term epileptic behavior (Perez-Mendes et al. 2005). Muha and Ramsdell (2011) used a modified dosing triturated protocol, similar to an established kindling protocol with the related experimental drug kainic acid, in rats to model aspects of human temporal lobe epilepsy (Hellier and Dudek 2005). This model accurately replicated a domoic acid–nduced SE that successfully leads to a chronic state of recurrent spontaneous seizures weeks to months later (Muha and Ramsdell 2011) and atypical aggressive behavior (Fuquay et al. 2012). Each of these behaviors has been observed as part of a domoic acid–associated chronic neurological disease in California sea lions (Goldstein et al. 2008).
As described above, domoic acid has been reported to cause widespread neurodegeneration or “lesions” and multiple studies have been reviewed by Pulido (2008). The purpose of this study was to identify the structural damage caused by domoic acid sufficient to induce SE in a model that reproducibly leads to a chronic neurological symptoms with striking similarity to domoic acid-associated neurological disease of the California sea lion. As a precursor to an epileptic-like disease, there is understanding that some brain regions typically damaged by domoic acid poisoning may not be essential to lead to the epilepsy and severe behavioral changes observed in the sea lions. Our previous study indicated that a single bout of SE was sufficient after a latent period of weeks to months to induce recurrent spontaneous seizures in 96% of rats (Muha and Ramsdell 2011). This suggests that SE is an early biological effect of domoic acid poisoning that can promote epileptic disease. In this study, we sought to identify structural damage specific to the minimal extent of SE according to this model that reproducibly leads to chronic recurrent spontaneous seizures. Hence the toxin exposure protocol used here is different from previous experimental studies that were used to evaluate poisoning without reference to the minimal damage necessary to progress to neurological disease. In this study, we examined 4 rat brains, 1 from a control and 3 from treated animals, taken a week after domoic acid-induced SE. Using cupric silver–stained sections from every eighth 40-µ coronal section of the entire brain from each animal, we have identified the sites of damage in the rat brain evident 7 days after a domoic acid treatment protocol that caused SE and was previously shown to lead to epileptic disease.
Materials and Methods
Laboratory Animals
Twelve male Sprague Dawley rats weighing 150 to 200g (Charles River, Wilmington, MA) were used for this study. Animals were housed in small groups in polycarbonate micro-isolator cages (Allentown Inc., Allentown, NJ) with controlled temperature, humidity, and 12 hr light cycles. Rodent chow (PMI Nutrition, Brentwood, MO) and water were allowed ad libitum and animals were given a 72-hr acclimation period prior to any experimental manipulation. All animal experiments were conducted in accordance with the National Institutes of Health (NIH) Guide for the care and use of laboratory animals, and all animal procedures were approved by the Center for Coastal Environmental Health and Biomolecular Research Institutional Animal Care and Use Committee.
Experimental Procedures
Two groups of 6 late juvenile (7 week) male rats (5 treated with domoic acid and one with PBS vehicle) were subject to an established protocol developed for kainic acid (Hellier and Dudek 2005) and modified for domoic acid (Muha and Ramsdell 2011) to induce a nonlethal SE that develops over weeks to chronic spontaneous recurrent seizures. Male rats were chosen because the estrous cycle has been reported to add variance in the kainic acid dosing protocol to induce a nonlethal SE (Hellier and Dudek 2005). Rats aged between 6 weeks and 6 months are designated for this protocol because although kainic acid induces SE in immature rats, they do not develop spontaneous recurrent seizures, and aged rats have a high rate of mortality to the kainic acid dosing protocol (Hellier and Dudek 2005). A domoic acid (B1380, Doe and Ingalls of NC, Inc., Durham, NC) dosing solution (0.35 mg/ml) in phosphate buffered saline (PBS) buffered to a final pH 7.2 to 7.4 was prepared. The rats received an initial sub-lethal dose of 1.0 mg/kg intraperitoneal that characteristically leads to stereotypic scratching. A second 1.0 mg/kg dose was given an hour later. Doses after that time were tailored to achieve a minimal dose to lead to SE and assure survival. A half dose was given if the rat showed early signs of seizures such as foaming, staring, or atypical posturing. Hourly dosing was stopped when an animal exhibited its first motor seizure determined by forelimb clonus, a stage III convulsion adapted from the modified Racine scale (Hellier et al. 1998), within the hour following the last dose. SE was determined to be the point at which the animals exhibited one or more stage IV (forelimb clonus with rearing) or V (forelimb clonus with rearing and loss of balance) seizures per hour for 3 consecutive hours. The control rat was given 5 injections of an equivalent volume of PBS over the course of 5 hr. During recovery, treated rats received a subcutaneous bolus between the scapulae of lactated Ringer’s solution (20 ml/kg at 37°C) for rehydration.
All animals were allowed to recover from the dosing for 1 week. Typically, recovery times of 2 or 7 days are used to examine the acute effects of neurotoxicants using cupric silver stains (Switzer 2000). Selection of a 7-day recovery period may miss some damage to terminals and cell bodies but was deemed most likely to identify major areas of damage before animals begin to express recurrent spontaneous seizures. Previous studies using silver histochemistry techniques in rats and monkeys have used a 7-day recovery period or shorter periods depending on survivability (Appel, Rapoport, and O'Callaghan 1997; Scallet et al. 1993; Schmued et al. 1995). Although the studies differed in species, dose, and brain regions examined, overall, there are no clear-cut differences in regional staining between animals that demonstrated typical seizure symptoms based upon recovery periods of 7 days and less. Three animals expressing a nonlethal SE and one vehicle control, ages matched on post natal day (PND) 42 for domoic acid treatment, were selected for histochemistry on PND 49. Significant studies have been conducted on the persistent effects of low-dose domoic exposure to early postnatal rats as a model for early life development of human epilepsy (reviewed in Ramsdell 2010); however, our selection of the 6-week postnatal rat is significant because it approximates the brain developmental stage for the late prenatal California sea lion in which neurodevelopment advances on a more precocious timeline than humans (Ramsdell and Zabka 2008). The end of gestation of the California sea lion appears to be a “perfect storm” convergence of pharmacokinetic and neurodevelopment susceptibility and seasonality of domoic acid blooms to understand the predominance of acute domoic poisoning in pregnant sea lions and appearance of chronic epilepsy in young animals (Ramsdell 2010). The animals chosen for histochemistry were from one group of 6 matched rats, which include three rats that developed SE, two rats that did not survive the dosing, and the sixth which was asymptomatic receiving only PBS treatments. The second group of 6 rats did not provide enough successful SE animals as 3 of the domoic acid–treated animals did not survive. The data collected for the rats used for the histological analysis is presented in Table 1. Each rat was deeply anesthetized using isoflurane and then transcardially perfused with a buffered sucrose solution containing sodium cacodylate (1222SK, Electron Microscopy Sciences, Hatfield, PA) at a rate of 1 L/kg B.W./hr. To minimize artifactual damage, heads were placed into a fixative solution containing 4% paraformaldehyde, 4% sucrose, 1.4% sodium cacodylate, and 0.02% sodium azide (1223SK, Electron Microscopy Sciences, Hatfield, PA) left to fix in situ for 24 hr prior to brain removal and then shipped in solution to Neuroscience Associates (Lexington, KY) who performed the following sectioning and degenerative staining method adapted from De Olmos, Beltramino, and De Olmos De Lorenzo (1994).
Dosage and seizure response of rats used for histological analysis.
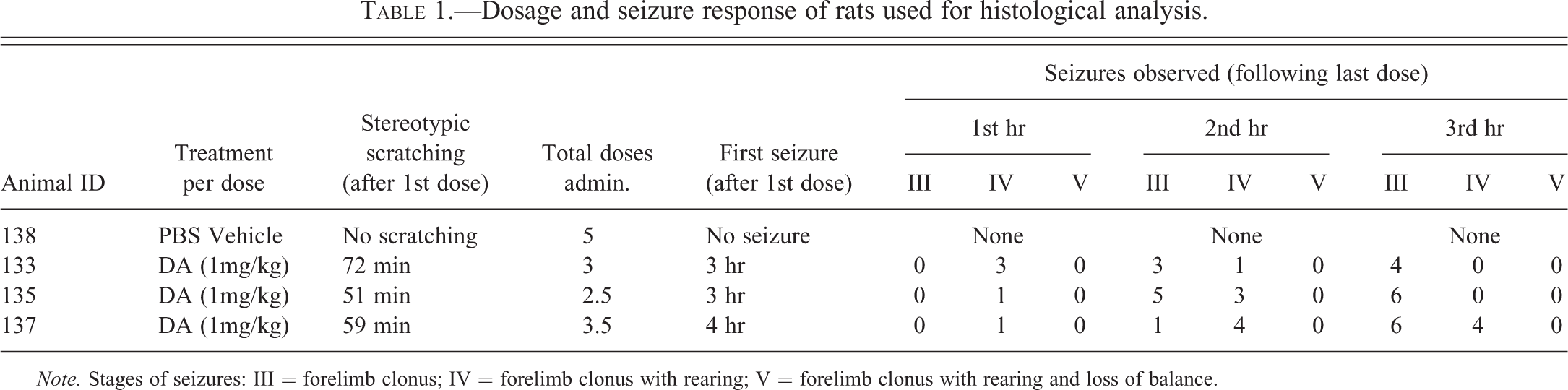
Note. Stages of seizures: III = forelimb clonus; IV = forelimb clonus with rearing; V = forelimb clonus with rearing and loss of balance.
Histochemical Procedures and Analysis
Coronal brain sections (40 µm) were blocked using MultiBrain® (Neuroscience Associates, Lexington, KY) technique and cut using a rotary microtome. Every eighth section was separated for staining and placed into a container filled with buffered ethylene glycol. These sections were rinsed with deionized water, placed into a solution containing cupric nitrate, silver nitrate, cadmium nitrate, lanthanum nitrate, neutral red, alpha-amino butyric acid, alanine, pyridine, triethanolamine, isopropanol, and deionized water, heated to 45 to 50°C and left to incubate at room temperature for 24 hr. Sections were then rinsed in deionized water, rinsed again in acetone, placed into a solution containing silver nitrate, 100% ethanol, acetone, lithium hydroxide, ammonium hydroxide, and deionized water, and incubated at room temperature for 50 min. Sections were then placed into a solution containing 100% ethanol, formalin, citric acid, and deionized water and incubated at 32 to 35°C for 25 min. Unreduced silver was removed by rapidly bleaching the tissue in a series of three solutions: potassium chlorate with lactic acid, potassium permanganate with sulfuric acid, and sodium thiosulfate. Sections received a final rinse with deionized water, then fixed to slides, and counterstained with neutral red to reveal normal cell bodies.
A total of 75 slides, of coronal sections from the front of the olfactory bulb to the back of the cerebellum (approximately −14.6 Bregma) for each animal, were examined using an Olympus CX31 microscope (Center Valley, PA) using 4×, 10×, and 40× objectives. Damage revealed by the stain was categorized by type (cell bodies, terminals, or axons) and relative density at a minimum of 100× magnification. Terminals were defined by staining of synaptic ends identified by punctuate staining and distinguished from the linear staining of axons and cytoplasmic staining of cell bodies. Location of the staining was assigned to corresponding nuclei of the brain determined using The Rat Brain in Stereotaxic Coordinates (Paxinos and Watson 1998). The entire coronal field of each brain was examined for every eighth 40-µ section and summarized. Staining was ranked according to the area of nucleus covered and quantity of a type (cell body, terminal, or axon) for each slide section on each animal. Those values were then averaged over the range of the sections to determine the values represented in Table 2 and to summarize the entire nucleus. Stained areas observed in the domoic acid-treated animals were compared to the control rat brain to confirm that the damage was specific, and not an artifact of manipulation. Micrographs of damaged areas were taken using an Olympus DD71 camera on an Olympus BX51 microscope using 2×, 20×, and 40× objectives, (20×, 200×, and 400× magnifications).
Type of damage in different nuclei of the brain between three rats with domoic acid–induced SE.
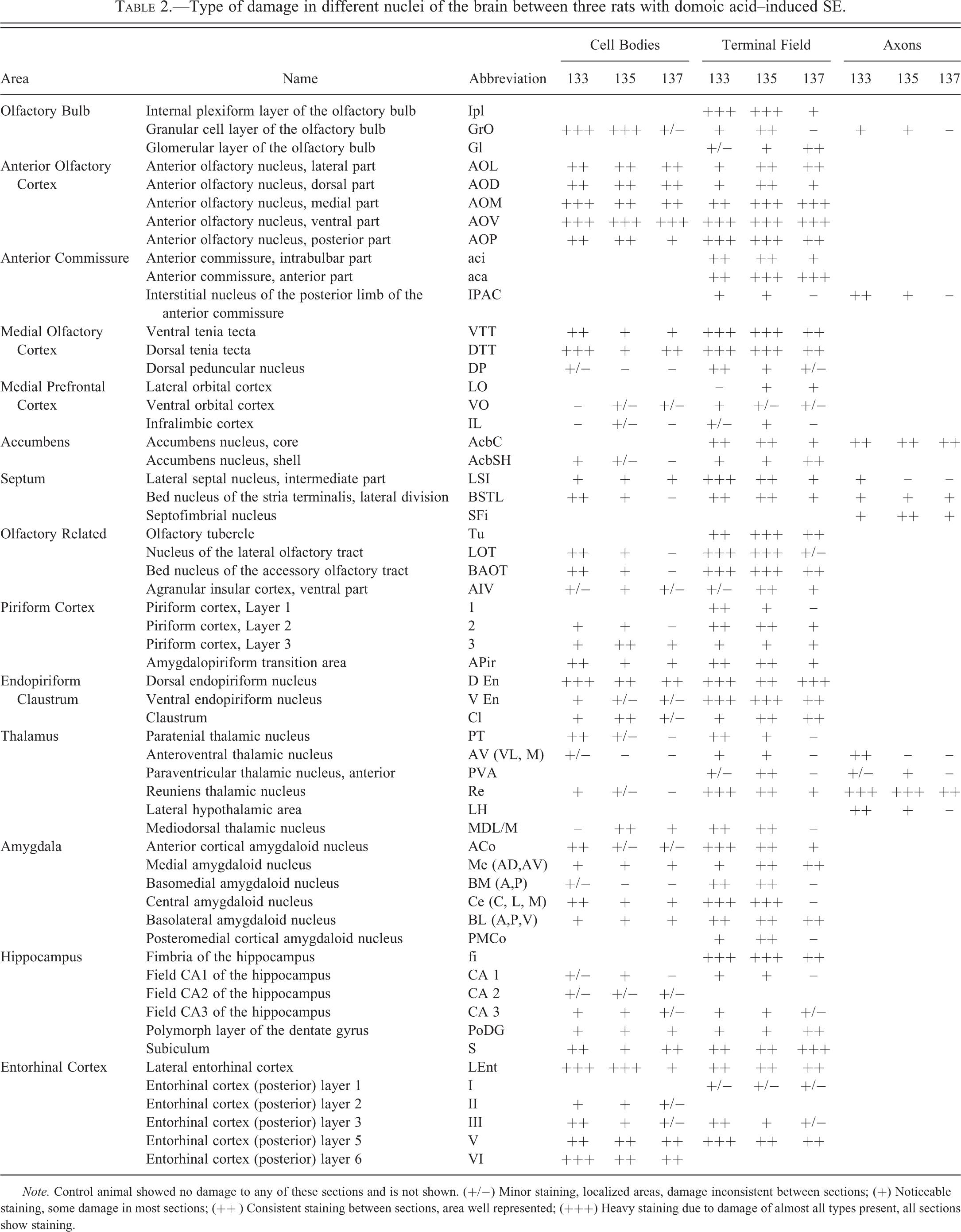
Note. Control animal showed no damage to any of these sections and is not shown. (+/−) Minor staining, localized areas, damage inconsistent between sections; (+) Noticeable staining, some damage in most sections; (++ ) Consistent staining between sections, area well represented; (+++) Heavy staining due to damage of almost all types present, all sections show staining.
Results
Global Damage to Brain Structures
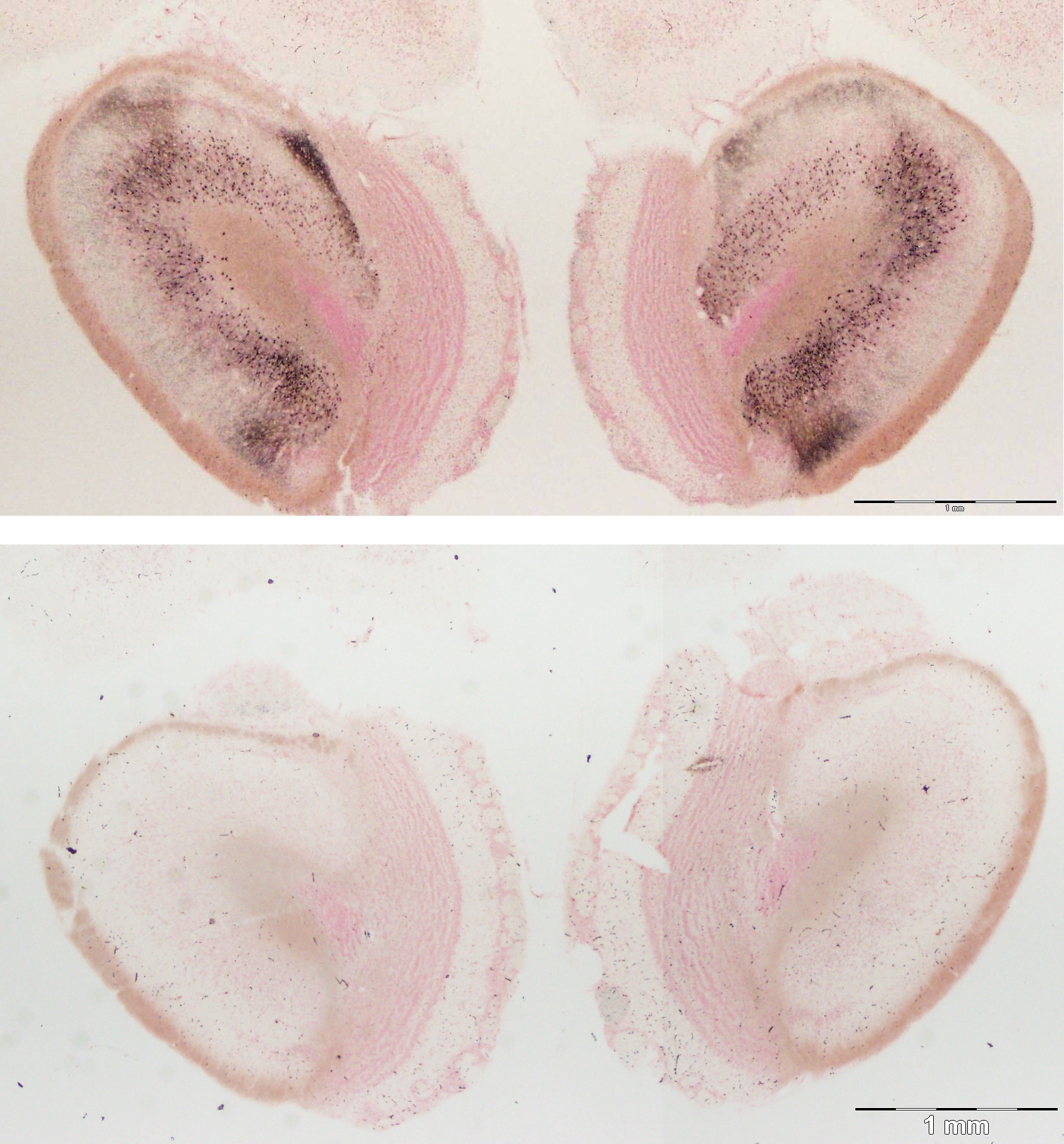
Serial coronal brain sections of rats 7 days after domoic acid–induced SE show intense and widespread silver reaction product in the olfactory bulb (Figure 1A). A region typically subject to intense degenerative damage in animals treated with a single maximal dose of domoic acid (as documented with silver stain by Peng et al. [1994]) the hippocampus, showed minor or no evident damage (Figure 1B) in the Ammon’s horn region. Staining was predominant in olfactory regions of the rhinencephalon (Figure 2) whereas no staining was evident in the cerebral cortex, midbrain, hindbrain, and cerebellum (data not shown). The staining of cell bodies, terminals, and axons varied throughout the different brain regions and in prevalence between animals (Table 2), while the affected regions remained consistent and are described below in greater detail. The control brain showed no specific staining on any sections (matched for all figures) and was used to distinguish the damage brought about by the domoic acid treatment and seizures.
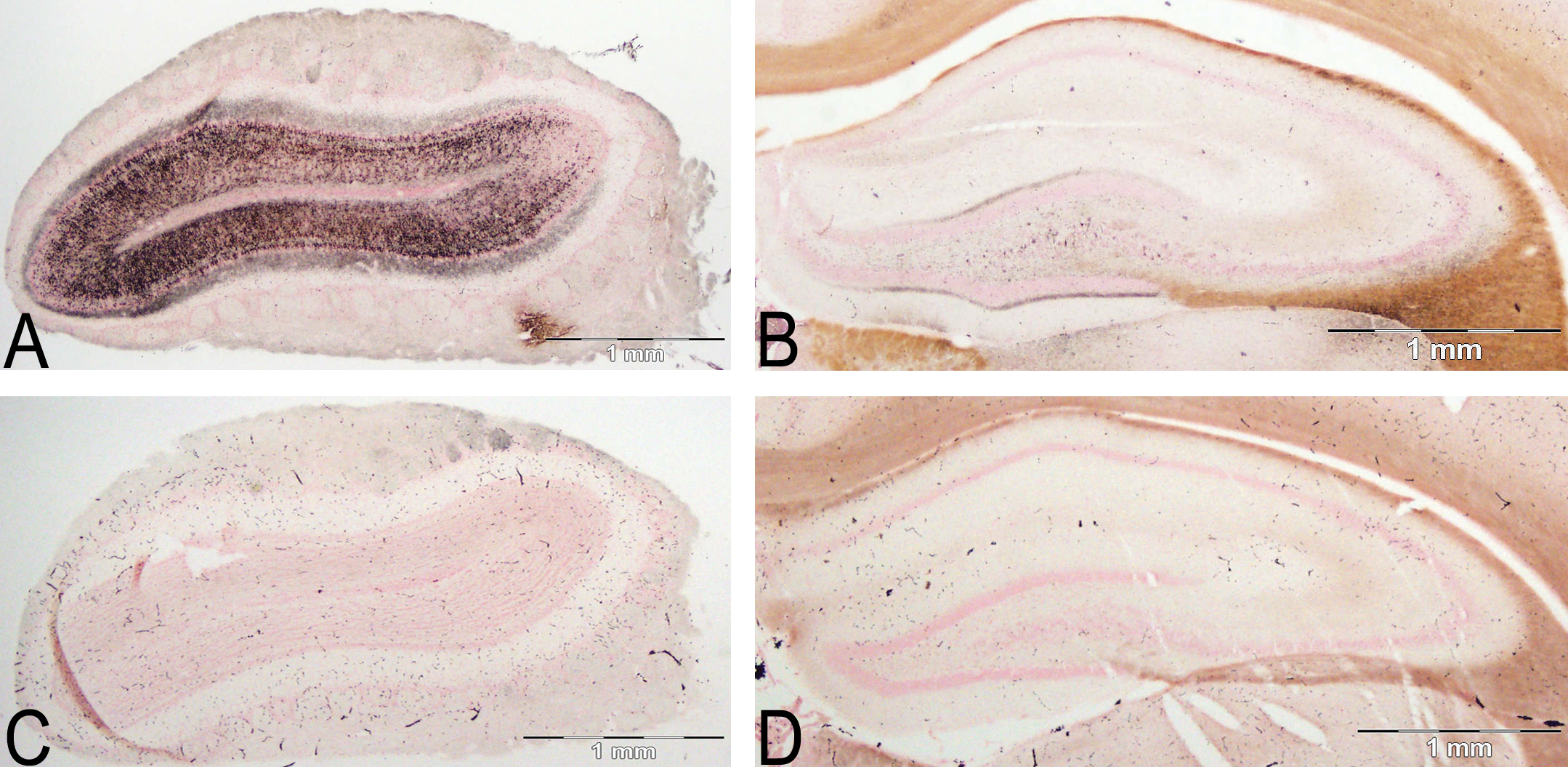
Intense (Table 2 +++) silver stain deposition in the olfactory bulb (A) compared to light silver stain (Table 2 +) present in the hippocampus (B), highlighted in the dentate molecular layer. Control olfactory bulb (C), also oriented sideways, and control hippocampus (D) are shown to highlight the specific staining present in the treated sections (A, B). Amino cupric silver stained 40 µm section, bregma 7.3 mm, and −3.3 mm, respectively, taken from rat 7 days after domoic acid induced SE; neutral red counterstaining; magnification 20×.
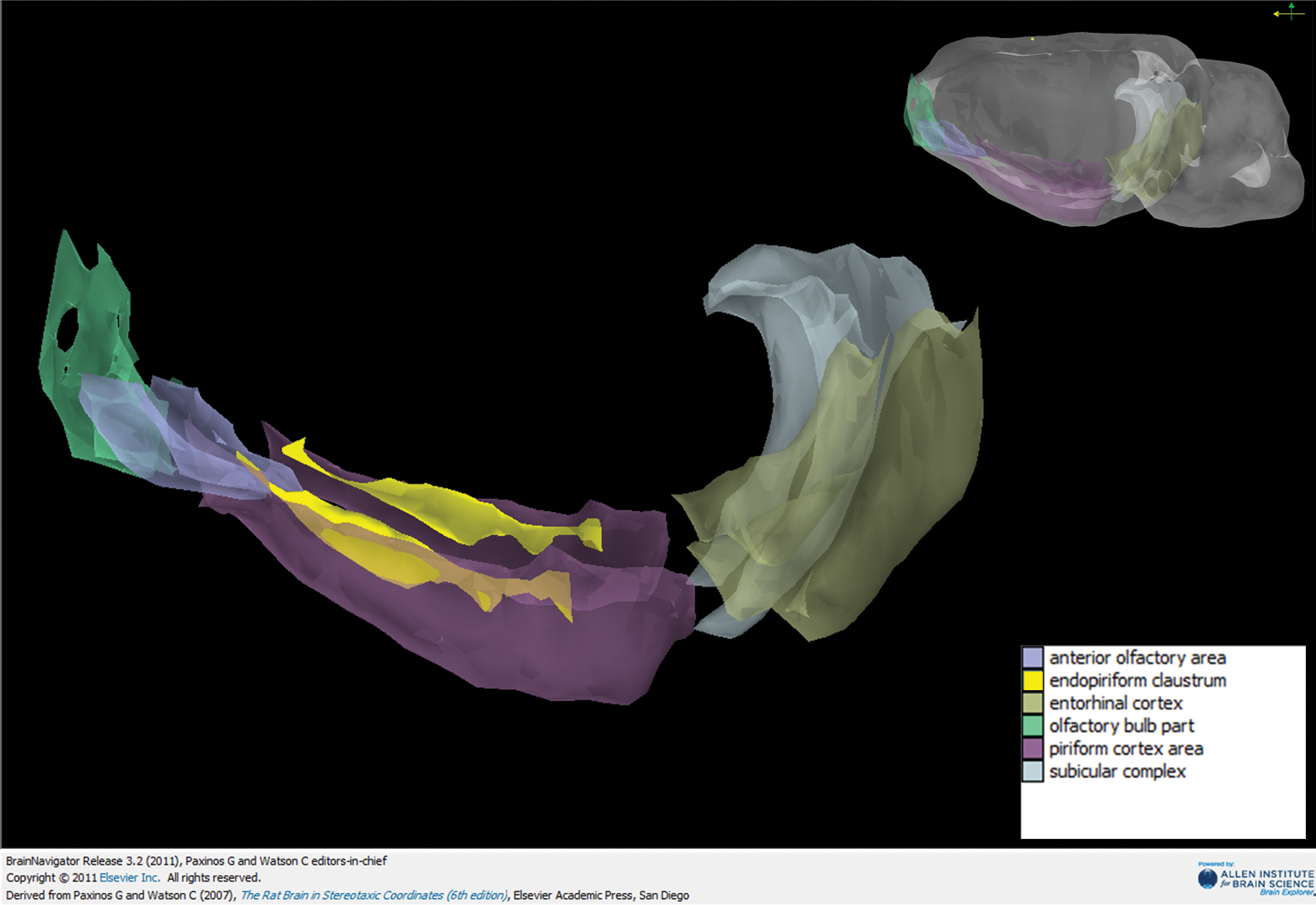
A representative model depicting the olfactory areas which showed the most cell body damage. Olfactory bulb, anterior olfactory cortex, piriform cortex, endopiriform claustrum (highlighted for contrast), subiculum, and entorhinal cortex depicted from left to right. Upper right represents the orientation of the brain, color-based legend lower right. Image created using BrainNavigator 3D.
Intense Damage in the Olfactory Bulb
Dense silver deposition was evident in the olfactory bulbs, in which cupric silver staining of the neurons were strongly regionalized in the granular cell layer (Figure 3). Most of the granule layer soma was impregnated with reaction product. No reaction product was observed on mitral cells, whereas the second population of granule cells located between in the mitral cell layer and internal plexiform layer also shows extensive stain. It is worth noting that mitral cells are the primary glutamatergic olfactory neurons to receive input from the sensory afferents from the olfactory epithelium and provide a major component of the lateral olfactory tract projecting to olfactory cortical regions (Figure 2). The external plexiform layer, superficial to the mitral cell layer, had intense punctuate reaction product, the result of extensive damage to the spiny dendritic arborization of the granule cells. The absence of reaction product in cell bodies of the external plexiform layer indicated that tufted cells, the second population of olfactory neurons to receive input from the olfactory sensory afferents, were not damaged as result of domoic acid–induced SE. Light, punctuated, silver staining of synaptic terminals is also observed in the glomerular layer, which is otherwise devoid of silver deposition. The accessory olfactory bulb also remained void of silver deposition (photomicrograph not shown).
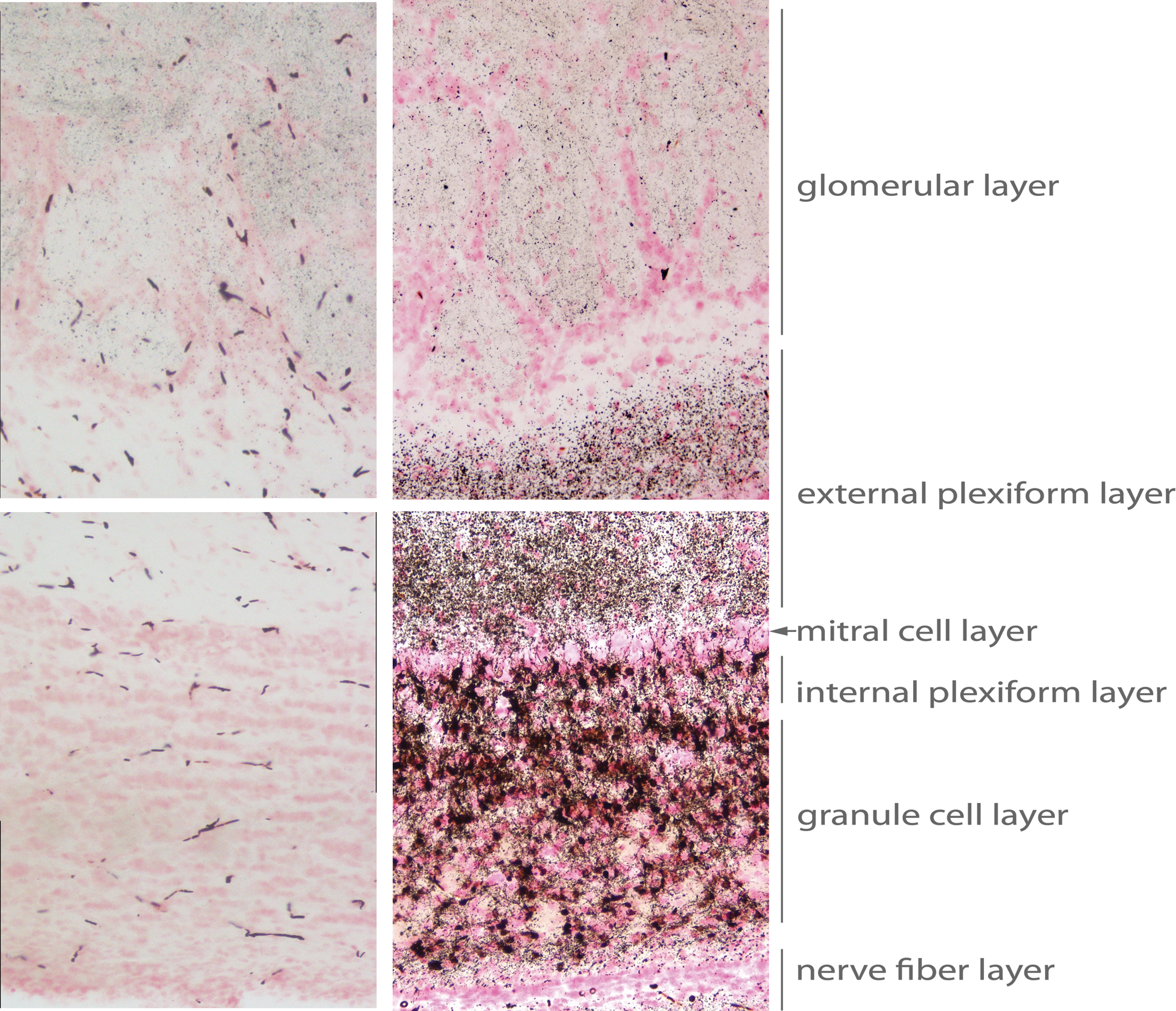
Damage to layers of the olfactory bulb, showing superficial (top) to deep (bottom) layers: glomerular cell layer, external plexiform layer, mitral cell layer, internal plexiform layer, granule cell layer, and nerve fiber layer. Matched images from control animal are on the left. Amino cupric silver stained 40 µm section, bregma 7.3 mm, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 200×.
Widespread Damage in the Olfactory Cortex and Associated Areas
Damaged cell bodies are numerous at the appearance of the olfactory tubercle in the anterior olfactory nucleus (herein referred to as anterior olfactory cortex [Haberly 2001]). Intense (++ or higher) impregnation of silver was found in the inner cell zone of the pars principalis and dendritic contacts with projections from the olfactory bulb via the lateral olfactory tract in the outer plexiform layer (Figure 4). All zones of the pars principalis showed stained cell bodies with larger numbers of cells stained in the pars externa. Many pyramidal cells with reaction product were oriented in sections to highlight long axons projecting deep toward the anterior commissure. Intense silver reaction product was also evident in the synaptic terminals of these pyramidal cells within both the inner cellular zone and the outer plexiform layer.
Damage to the anterior olfactory cortex (top), control (bottom). Degenerated pyramidal cells and terminals are present in the pars principalis and pars externa of the anterior olfactory cortex. Amino cupric silver stained 40 µm section, bregma 5.2 mm, taken from rat 7d after domoic acid–induced SE; neutral red counterstaining; magnification 20×.
Silver-impregnated pyramidal cells continue posteriorly along the cortical layer of the tubercle toward the piriform cortex. At the transition of the anterior olfactory cortex to the piriform cortex, intense silver disposition was evident in the pars posterior division of the anterior olfactory cortex and in the medial olfactory cortex, comprising ventral and dorsal divisions of the tenia tecta (Figure 5). The rostral extent of the anterior piriform cortex shows a small number of pyramidal cells stained with silver in layer II. Stained neurons were also prominent in the endopiriform nucleus. Intense staining of neurons was evident in more caudal sections in the dorsal tenia tecta and the dorsal peduncular cortex either side of the rhinal incisura (Figure 6). Uniform reaction product in neurons of the endopiriform nucleus and bands of reaction product in the piriform cortex begin to intensify. Silver-impregnated fields of axons and cell bodies sweep dorsal to the medial prefrontal cortex to align on the medial edge of the minor forceps of the corpus callosum.
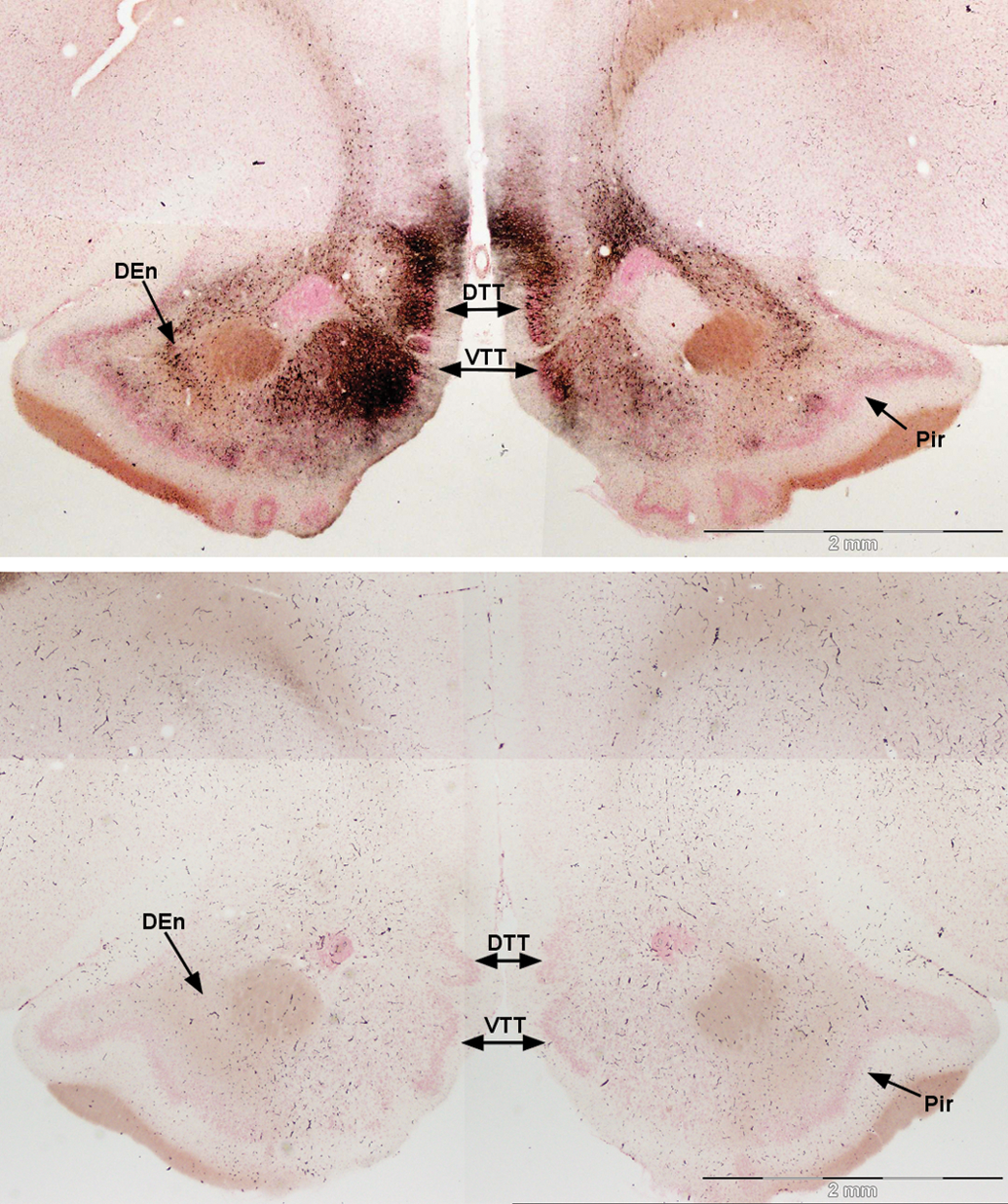
Damage to the anterior regions of the medial olfactory cortex (top), control (bottom). Dense stain is present in the medial olfactory cortex, including the ventral (VTT) and dorsal tenia tecta (DTT). Stained cells are also evident medial and lateral to the capsule of the nucleus accumbens in the anterior olfactory cortex pars posterior and endopirform nucleus (DEn), respectively. Some heavier precipitated stain is evident in the left section of the pars posterior. Amino cupric silver stained 40 µm section, bregma 3.0 mm, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×.
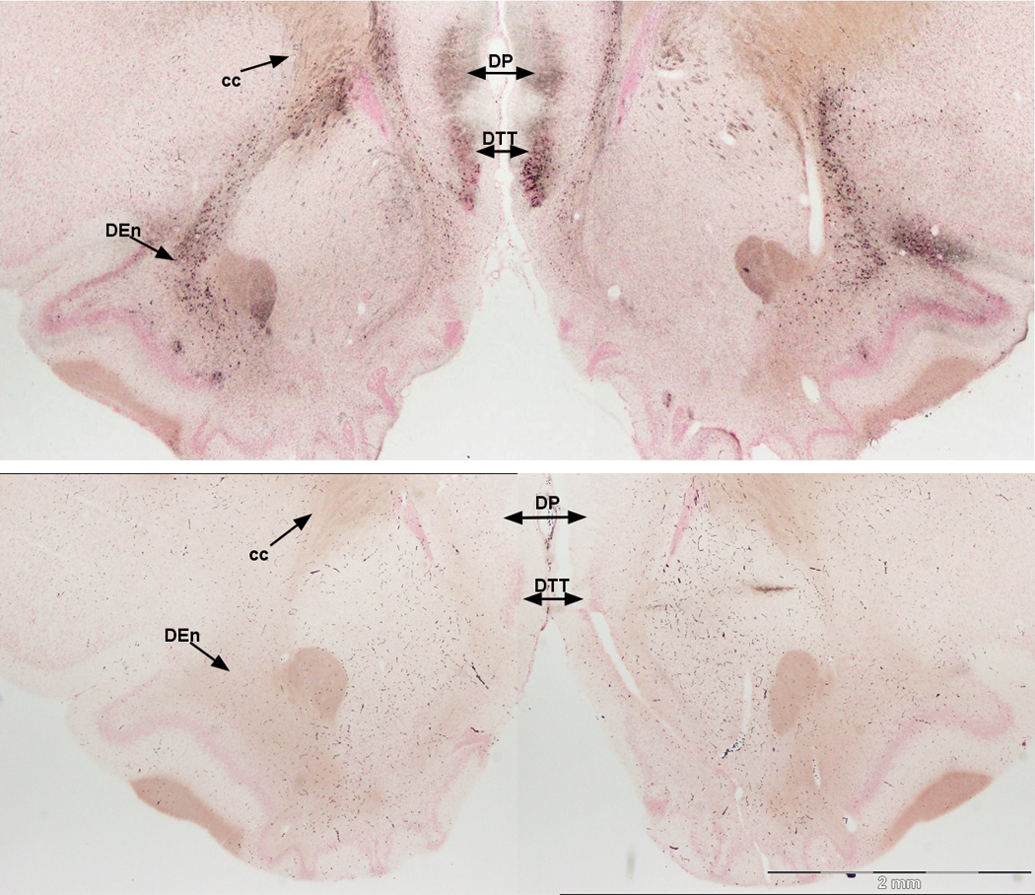
Damage to the posterior regions of the medial olfactory cortex (top), control (bottom). Silver deposition still marks damage in the dorsal tenia tecta (DTT) and highlights the dorsal peduncular nucleus (DP) and axons from the dorsal endopiriform nucleus (DEn) sweeping dorsal toward the corpus callosum (cc). Amino cupric silver stained 40 µm section, bregma 2.4, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×.
Silver staining of cells was also evident in the midline just posterior to the medial prefrontal cortex in the lateral septum (Figure 7). The lateral septal nucleus had silver deposits in the cell bodies and terminals in the dorsal, intermediate, and ventral divisions. Further caudal, silver reaction product was apparent throughout the lambdoid septal zone and paralambdoid septal nucleus (photomicrographs not shown).
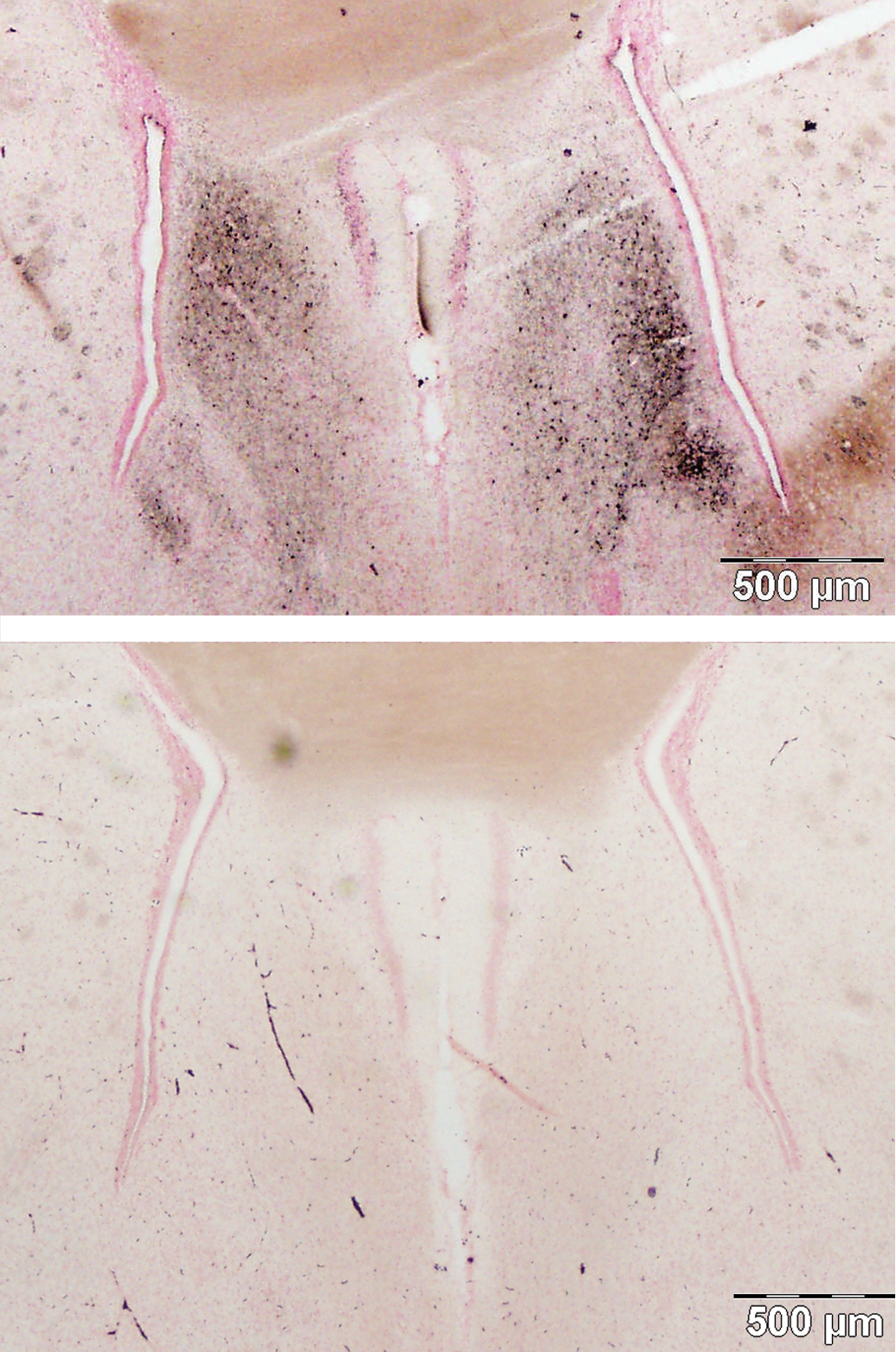
Damage to the septal region (top), control (bottom). The lateral septal nucleus shows intense staining of cell bodies and terminals. Amino cupric silver stained 40 µm section, bregma 0.7 mm, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×.
Transitioning caudally, just medial to the olfactory tract, the cupric silver staining continued in pyramidal cells of the anterior piriform cortex. Prominent staining of pyramidal cells was also seen deep to the anterior piriform cortex and into the endopiriform nucleus. The staining of the endopiriform nucleus also extends dorsal to the claustrum and then lateral to multiple lamellae of the insular cortices (Figure 8A). Silver-impregnated cells continued throughout the extent of the piriform cortex and endopiriform nucleus and insular cortex (Figure 8B).
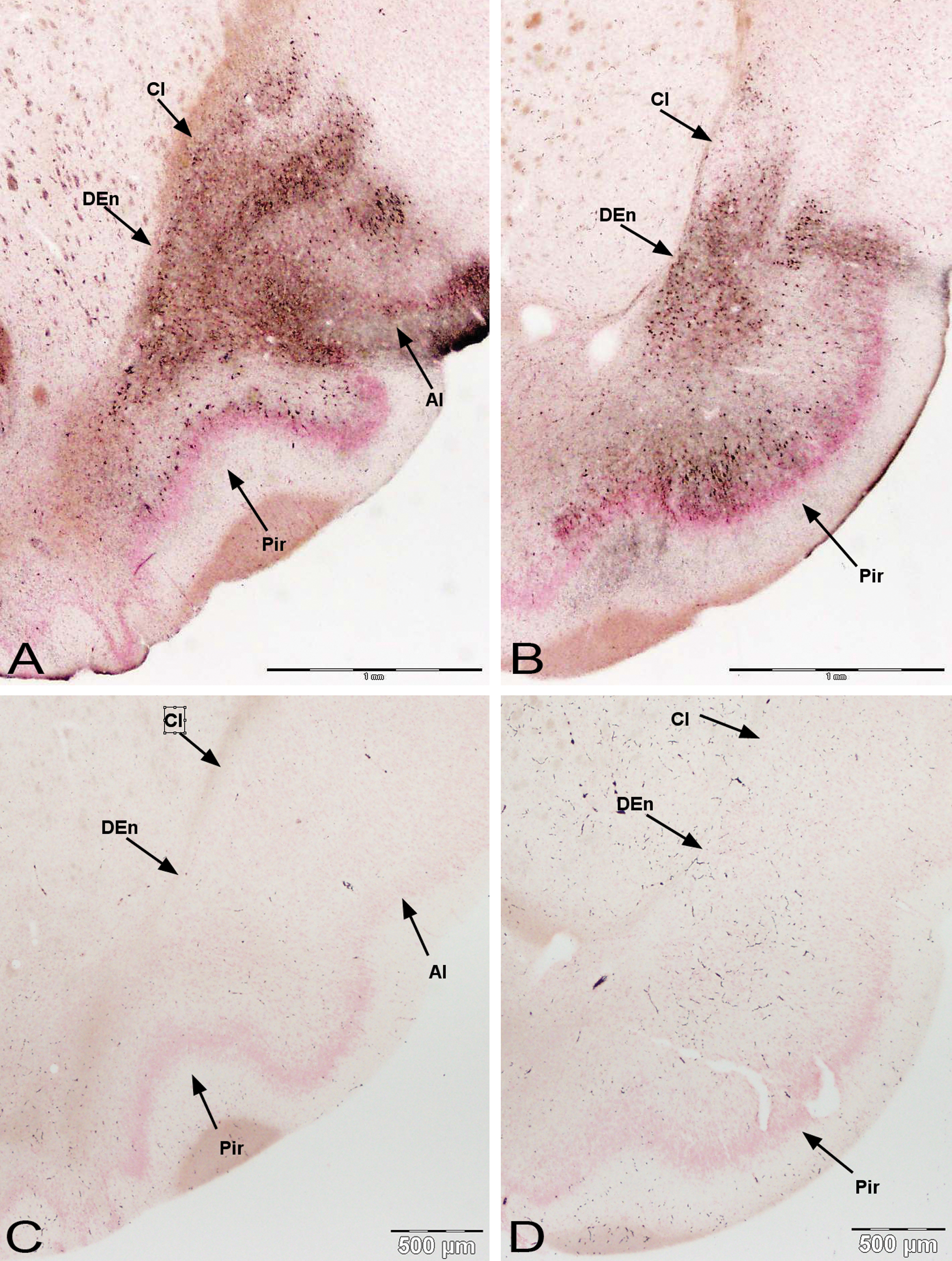
Damage to anterior and posterior regions of the olfactory cortex along the lateral olfactory tract. Silver deposition is prominent in the endopiriform nucleus (DEn) and extends dorsal to the claustrum (Cl) and lateral to the insular cortices (AI) (A). Damage of piriform cortex (Pir) continues through more caudal sections with occasional heavy bands of pyramidal cell staining (B). Control medial olfactory cortex (C, D) for respective sections below. Amino cupric silver stained 40 µm section, bregma 1.6 mm and −0.3 mm, respectively, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×.
At the caudal terminus of the anterior piriform cortex, a dense aggregate of silver stained cells was evident, just lateral to the optic chiasm and supraoptic nucleus, in the nucleus of the lateral tract (Figure 9). Staining of the piriform cortex occurred in clusters or bands of cells as shown adjacent to the nucleus of the lateral olfactory tract. The uniform staining of cell bodies of the endopiriform nucleus extends its full length from the anterior olfactory cortex to the entorhinal cortex.
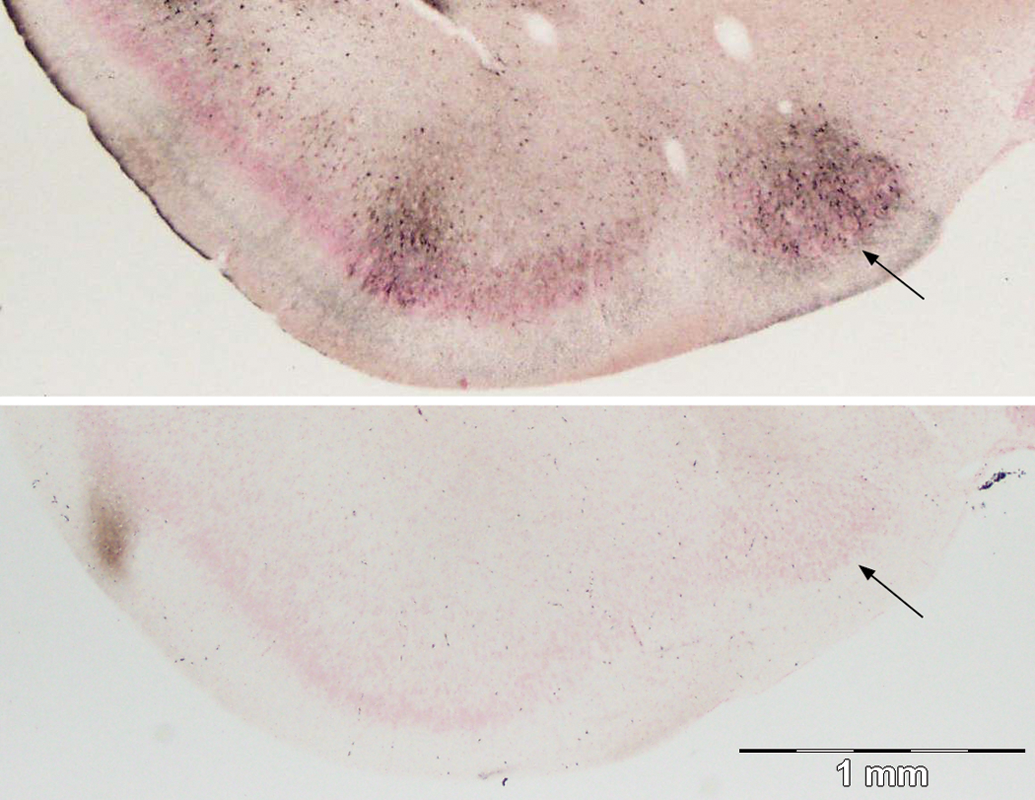
Damage to the olfactory cortex near the end of the lateral olfactory tract (top), control (bottom). A band of cells and terminals are stained in the piriform cortex as well as a medial cluster of silver impregnated cells in the nucleus of the lateral olfactory tract (arrow). Amino cupric silver stained 40 µm section, bregma −1.3 mm, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×.
Damage to the Midline Thalamus
The thalamus showed dense reaction product on axon fibers coursing from the piriform lobe to the midline thalamus. The nucleus reuniens was heavily stained with reaction product with many cells impregnated with silver (Figure 10). The staining of the reuniens cells extends lateral to the ventral reuniens nucleus. Intense silver impregnated axons extend dorsolaterally from the main reuniens nucleus and laterally from the ventral reuniens nucleus. More dorsal on the midline, the paraventricular and paratenial thalamic nuclei also showed reaction product in the soma and fibers (Figure 11, bottom left corner).
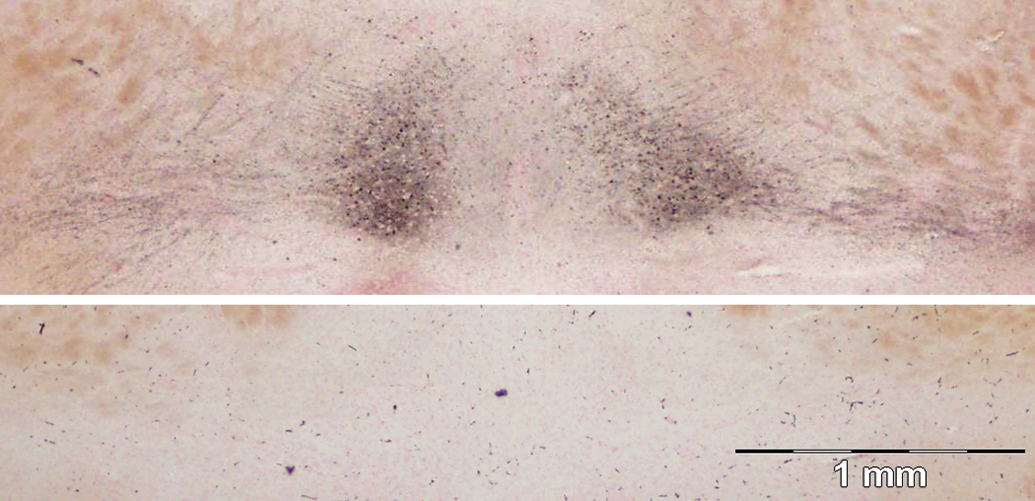
Damage to the reuniens nucleus of the midline thalamus (top), control (bottom). Reuniens nucleus connections selectively stained highlights the directional axon and staining of the cell bodies. Amino cupric silver stained 40 µm section, bregma −1.8 mm, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×.
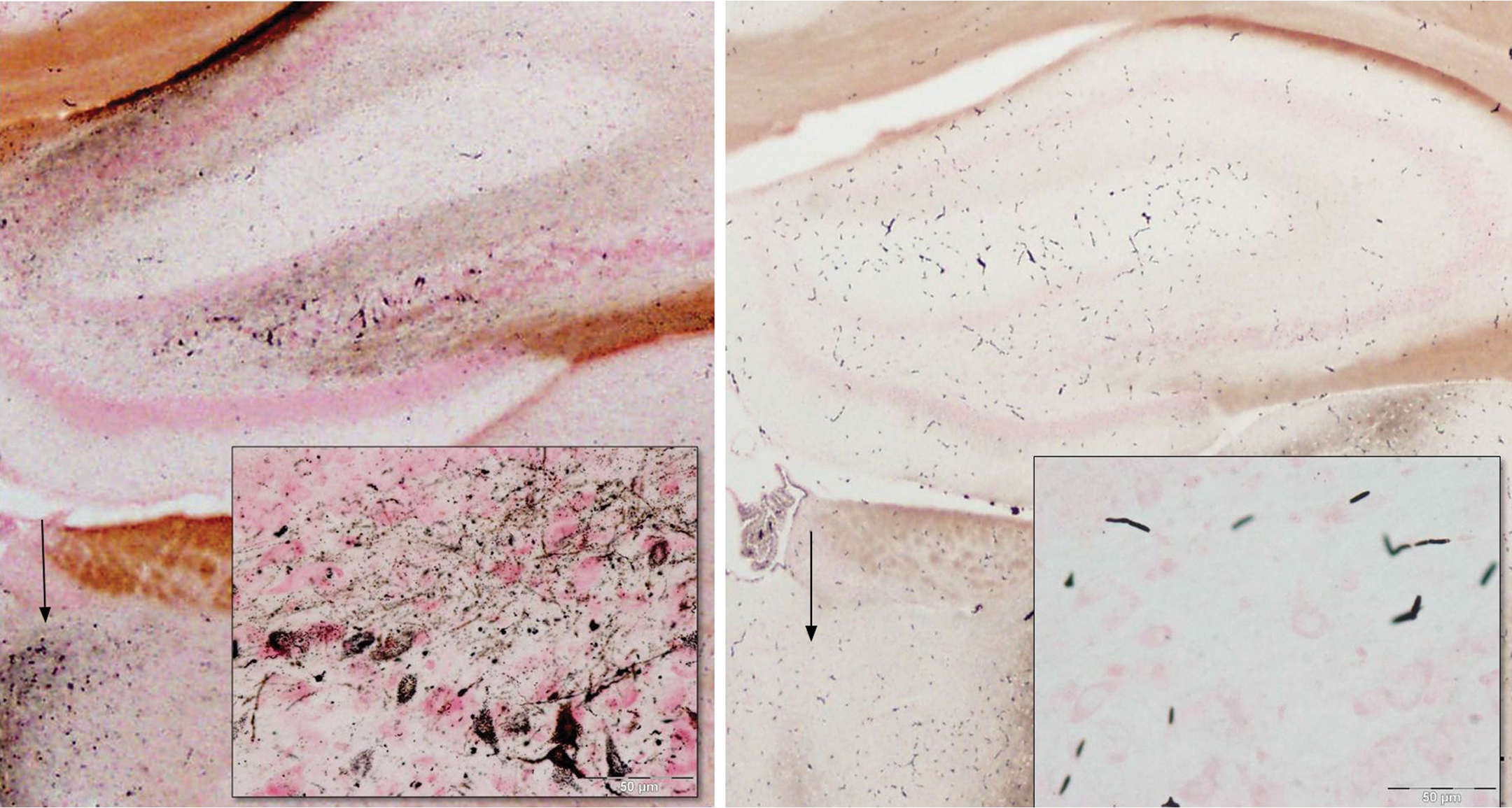
Damage to the dorsal hippocampus and adjacent dorsal midline thalamus. Consistent stain is present in axons, terminals, and hilar cells (inset) in the dentate gyrus, while the granule cell layer remains unstained. Staining of cells in the periventricular thalamic nucleus (arrow) is evident on the bottom left of the main figure. Matched images from control animal are on the right. Amino cupric silver stained 40 µm section, bregma −2.1 mm, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×, inset magnification 400×.
Limited Damage to the Amygdala and Hippocampus
Select nuclear groups of the amygdaloid complex showed low-level staining of cell bodies with higher intensity staining terminal fields in domoic acid–kindled SE rats. The amygdalopiriform transition area was the most notably stained (Table 1). Limited silver staining was evident in select regions of the hippocampus, but was more prominent in the rostral sections, close to the subiculum. A light-to-moderate stained band of terminals can be observed in the molecular layer of the dentate gyrus (Figure 1B). No stain was found in the dentate granule cell layer; however, staining of terminal fields and hilar cells is commonly evident (Figure 11). Higher magnification views of dentate hilar cells (Figure 11, inset) indicate there were still many viable cells covered with punctuate silver terminals.
Sparsely stained pyramidal cells and dendrites were found in the CA1 and the CA3 fields; however, staining was not consistently found in these sections of the Ammon’s horn. By contrast, the subiculum consistently accumulated silver reaction product in pyramidal cells and terminals in both its dorsal and its ventral subfields (Figure 12, top and lower left panels, respectively). Staining of cell bodies and axons could also be found occasionally to continue from the subiculum to the retrosplenal cortex (photomicrographs not shown).
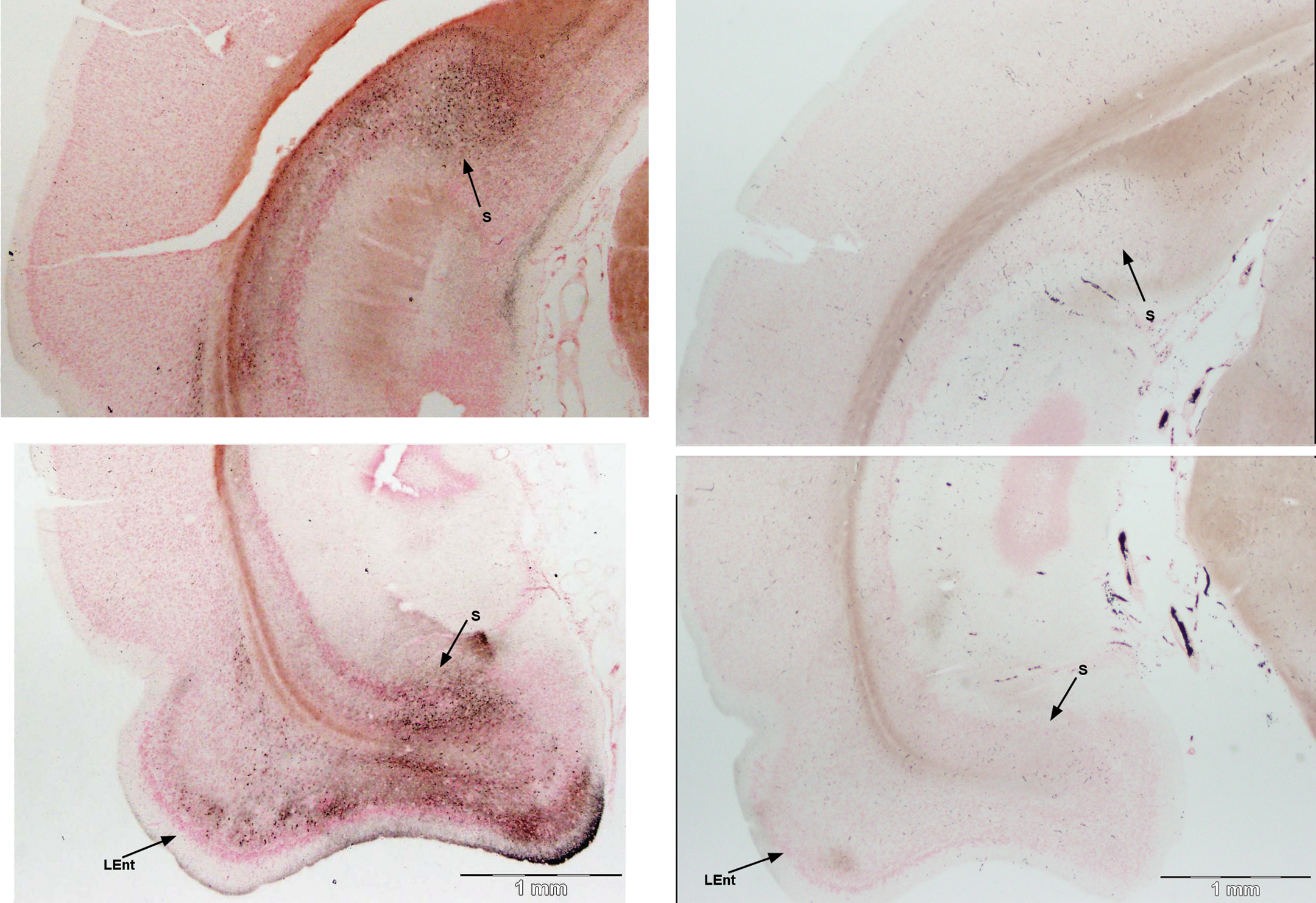
Damage to the dorsal (top) and ventral (bottom) body of the hippocampus and anterior extent of the entorhinal cortex. The subiculum (S) consistently showed silver stain in cell bodies and terminals in both dorsal (top) and ventral (bottom) regions of the same section. The entorhinal cortex (LEnt) shows staining, as it transitions from the piriform cortex (bottom). Matched images from control animal are on the right. Amino cupric silver stained 40 µm section, bregma −6.3 mm, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×.
Damage to the Entorhinal Cortex
At the transition of the posterior piriform cortex and the entorhinal cortex, silver staining was evident in the lateral part of the entorhinal cortex (Figure 12, lower left panel). Moderate staining of pyramidal cells and dendritic fields was found both in the superficial and in the deep layers of the lateral entorhinal cortex. By contrast, no staining was evident in the medial part of the entorhinal cortex. Silver staining was more prominent in the caudal terminus of the entorhinal cortex. Staining of cells in layer III was extensive and the deeper layers V and VI also had substantial numbers of cell soma impregnated with silver (Figure 13; Table 2).
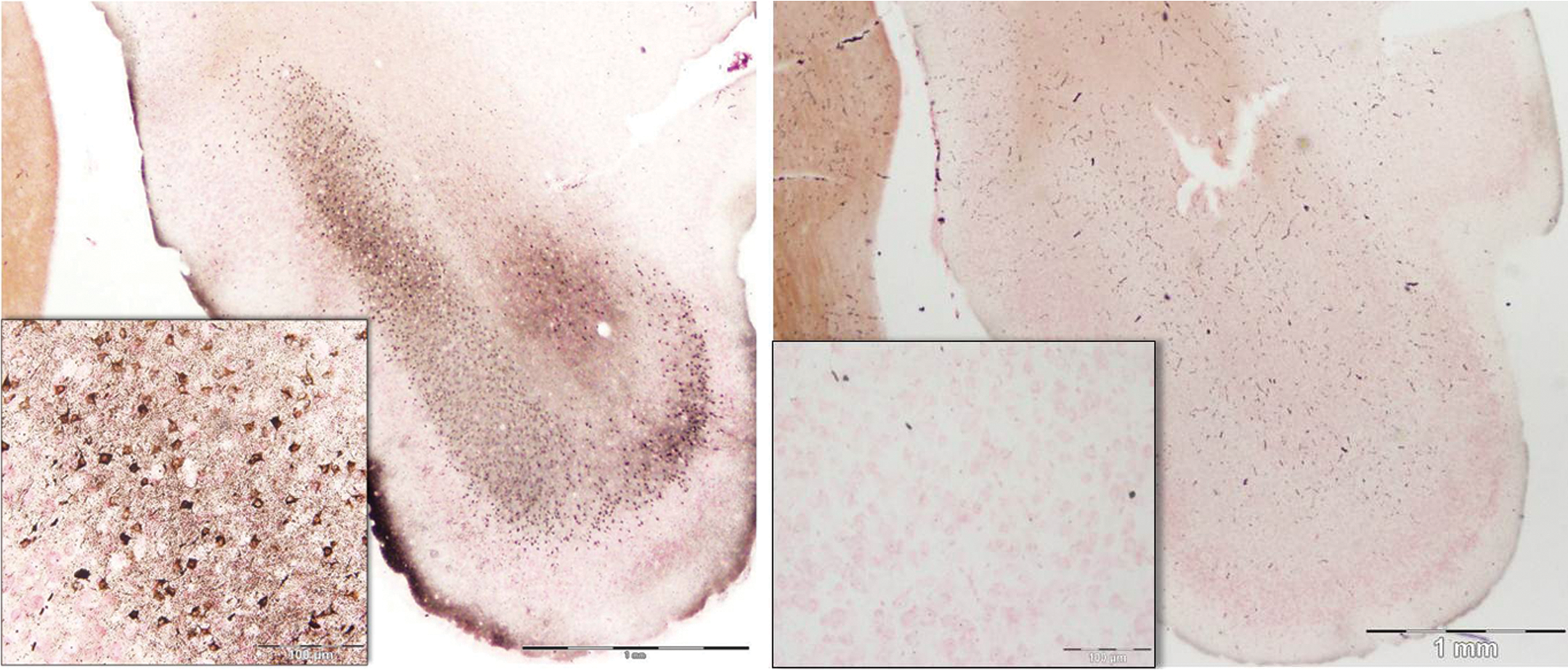
Damage to the posterior extent of the entorhinal cortex. Caudal areas of the lateral entorhinal cortex show staining in distinct layers, with heavy cell body and terminal staining evident in layer III (inset). Matched images from control animal are on the right. Amino cupric silver stained 40 µm section, bregma −8.0 mm, taken from rat 7 days after domoic acid–induced SE; neutral red counterstaining; magnification 20×, inset magnification 200×.
Discussion
Extensive Damage to the Olfactory Bulb and Its Granule Cells
Domoic acid–induced SE resulted in widespread damage to the olfactory bulb. Punctuated silver deposition stained nearly half of the external plexiform layer, and silver impregnated the majority of cell bodies deep in the granule cell layer. The external plexiform layer of the olfactory bulb has dense dendrodendritic connections comprised of secondary dendrites of mitral cells and long dendritic spines of granule cells (Price and Powell 1970). These dendrodendritic connections yield reciprocal inputs with glutamatergic stimulation of granule cells and GAB Aergic (gamma-aminobutyric acid) inhibition of mitral cells (Wellis and Kauer 1993). The glutamatergic action is under tonic inhibition by magnesium block of N-Methyl-D-aspartate (NMDA) receptors (Jahr and Nicoll 1982) and this tonic inhibition is a likely target for domoic acid disruption. Domoic acid acts as a partial agonist at several subtypes of non-NMDA ionotropic glutamatergic receptors and leads to a partially opened ion-conducting pore that fails to deactivate (Nanao et al. 2005; Swanson et al. 1997). This sustained depolarization of the neuron can enhance presynaptic release of glutamate and extinguish the magnesium block of NMDA receptors at dendritic spines of granule cells, resulting in excitotoxic damage (Ramsdell 2007).
Extensive silver impregnation of granule cell bodies was also evident in our sections after domoic acid–induced SE. Granule cells receive additional excitatory input from axon collaterals of mitral cells that traverse deep into the granule cell layer. In addition, granule cells receive extensive centrifugal input from excitatory pathways of the olfactory cortex that leads to a reduction of magnesium block and NMDA-mediated activation (Balu, Pressler, and Strowbridge 2007). This positive feedback loop, at the level of the granule cell layer, could increase susceptibility to excitotoxic damage from domoic acid.
Damage Follows the Lateral Olfactory Tract
Structural damage associated with domoic acid–induced SE spreads from the main olfactory bulb through regions of the primary olfactory cortex. The circuitry of the primary olfactory cortex represents a dominant forward flow of excitation from the olfactory bulb including back-projection loops. These loops have been proposed to function in a design similar to the hippocampal formation (Haberly 2001). Extensive damage found in the anterior olfactory cortex, anterior and posterior piriform cortex, nucleus of the lateral olfactory tract, and reaching caudally to entorhinal cortex are all part of the forward flow of olfactory excitation by direct connection of mitral and tufted cell axons via the lateral olfactory tract (Shipley and Adamek 1984).
Vomeronasal System Intact
It should be noted that although prolific silver deposition was present in the olfactory bulb, the accessory olfactory bulb was unstained in rats kindled to SE. Some staining of granule cells of the accessory olfactory bulb was observed in the analysis of Colman et al. This may be due to differences in dose, species difference, or a shorter recovery window. The accessory olfactory bulb, or vomeronasal bulb, is responsible for processing chemical-communication signals or species-specific pheromones, which influence behaviors such as aggression and mating. Unlike the olfactory bulb, the accessory olfactory bulb bypasses the olfactory cortex and projects directly to the amygdala and hypothalamus, which are closely linked to behavior. It has been suggested that the vomeronasal and olfactory systems work in conjunction with each other and that having only one system operational could have a detrimental effect on behavior (Kudjakova, Sarycheva, and Kamenskii 2007). These results suggest that domoic acid–induced SE may disrupt olfactory processing on several levels in the rat, but leaves the vomeronasal system structurally intact for behavior-driven scent cues.
Damage to the Anterior and Medial Olfactory Cortex
Pyramidal cells of the anterior olfactory cortex receive direct excitatory input from mitral/tufted cell axons traversing the lateral olfactory tract and provide extensive back excitatory projections to the granule cells of the olfactory bulb. Hence, damage to the olfactory granule cells by domoic acid could be expected to reciprocate damage, via mitral/tufted cells to the pyramidal cells of the anterior olfactory cortex where extensive cellular damage was evident. Damage is also evident in pyramidal cells of the tenia tecta and dorsal peduncular cortex, which are recognized to comprise a medial olfactory cortex (Haberly 2001; Haberly and Price 1978a). These medial olfactory cortical divisions differ from the anterior olfactory cortex in that they have little to no direct connection to the olfactory bulb. However, they do have a strong unilateral projection to the anterior olfactory cortex and heavy reciprocal connections to more caudal olfactory cortical areas including the piriform cortex and entorhinal cortex (Brunjes, Illig, and Meyer 2005; Haberly and Price 1978b). Hence, it is possible that domoic acid may exacerbate strong reciprocal excitation from the posterior piriform and entorhinal cortex to promote cellular damage in the medial olfactory cortex.
Damage to the Piriform Cortex and Endopiriform Nucleus
The piriform cortex receives extensive input from the olfactory bulb via the lateral olfactory tract and showed pyramidal cell damage in its deeper lamellae. However, more extensive silver impregnation was observed deep into the piriform cortex concentrated in the endopiriform nucleus. The endopiriform nucleus has strong reciprocal connections with the piriform cortex and has been found to trigger seizures in explants of the deep piriform region (Hoffman and Haberly 1996).
Dorsal to the endopiriform nucleus, the claustrum and the adjacent insular cortices show extensive soma and terminal damage after domoic acid-induced SE. Although the endopiriform nucleus and claustrum are closely associated, only minimal direct neuronal connections have been observed (Behan and Haberly 1999). Despite the lack of connections, epileptiform discharges have been found to originate at the interface of the endopiriform nucleus and claustrum in slice preparations of kindled rats (Hoffman and Haberly 1996). The claustrum is noted to have dendrodendritic synapses (Crick and Koch 2005), which may be particularly sensitive to excitotoxic damage by domoic acid. By contrast, the insular cortex receives dense projections from the endopiriform nucleus and is observed to show synchronous seizure activity in slice preparations (Demir, Haberly, and Jackson 1998). In this study, the damage to the endopiriform nucleus ranked high among the brain regions with damage comparable to the granule cell layer of the olfactory bulb and the pars principalis of the anterior olfactory cortex. The latter two groups of neurons receive direct excitatory input from the mitral cells that are subject to domoic acid–induced excitotoxicity. The endopiriform nucleus, on the other hand, receives only indirect input from mitral cells, largely through the piriform cortex. Nevertheless, its role to entrain and propagate seizure activity relies on extensive intrinsic excitatory connections (Behan and Haberly 1999), which could be susceptible to domoic acid excitotoxicity during SE.
Damage to Thalamic Relays
Domoic acid–induced SE leads to neuronal damage to several midline thalamic nuclei. Most conspicuously, cell bodies and laterally projecting axon fibers in both the main and the ventral divisions of the nucleus reuniens showed extensive damage. The nucleus reuniens has a strong projection to the CA1 and subiculum causing a powerful excitatory stimulus on the CA1 region (Bertram and Zhang 1999). It receives afferents from widespread limbic areas including the lateral septum, tenia tecta, and claustrum, and it is the major source of thalamic projection to the hippocampus and prefrontal cortex (McKenna and Vertes 2004; Vertes et al. 2006). Additional midline thalamic regions that showed damage after domoic acid–induced SE were the paratenial and paraventricular nuclei located in the dorsal thalamus adjacent to the third ventricle. Like the nucleus reuniens, these nuclei share output to the medial prefrontal cortex and subiculum (Moga, Weis, and Moore 1995). The capacity of these thalamic relays to connect widespread limbic input to the hippocampus is consistent with their role to intensify seizure behavior during SE (Bertram and Zhang 1999; Bertram, Zhang, and Williamson 2008).
Damage to the Entorhinal Cortex
The most caudal brain region to receive direct input from the olfactory tract is the entorhinal cortex, mainly in its lateral division. The entorhinal cortex has silver deposition on pyramidal cells of the superficial and deep layers, comparable to the adjacent subiculum, and silver staining intensifies caudally showing prominent damage in layer III. Pyramidal cells of layer III project directly to the subiculum and CA1 field and are not part of the primary perforant path to the dentate gyrus with output via the CA3 to CA1 to subiculum (Canto, Wouterlood, and Witter 2008). Taken together, this indicates that a substantial component of posterior entorhinal regions damaged in this study may project “backward” into the hippocampus by way of the subiculum and CA1 pyramidal cells.
Limited Damage to Commonly Implicated Hippocampal Formation
It is of great interest that the hippocampus showed only modest damage by cupric silver histochemistry after domoic acid–induced SE. The hippocampus, particularly the pyramidal cells of the Ammon’s horn, is the most noted of targets for structural damage by domoic acid across laboratory species (Appel, Rapoport, and O'Callaghan 1997; Peng et al. 1994; Scallet et al. 1993; Schmued et al. 1995; Tryphonas and Iverson 1990). Hippocampal damage is also present following environmental poisoning of humans (Teitelbaum et al. 1990) and marine wildlife, notably the California sea lion (Silvagni et al. 2005). A full brain mapping of domoic acid–induced degeneration, as assessed by cupric silver neurohistochemistry of the mouse brain after a single maximal tolerable dose of domoic acid, reported profuse staining of hippocampal soma, terminal fields, and axons (Colman et al. 2005). The author noted a maximal density of staining of cell bodies in the pyramidal cell layer of the hippocampus and a maximal density of silver impregnated terminals was also found in the Ammon’s horn, including the CA1, CA2, and CA3 fields as well as oriens and lacunosum molecular layers (Colman et al. 2005).
By contrast, in the current study, the hippocampus was not the primary target for structural damage. Cupric silver staining of the pyramidal cells of the Ammon’s horn and associated terminal fields was minimal, unlike the silver impregnation of the entire pyramidal cell and fiber tracts seen in the single dose mouse study. Perhaps the most notable damage seen in our study of the hippocampus was found deep in the dentate gyrus, with punctuate silver staining of what appears to be synaptic terminals on dentate hilar cells. This area is associated with the perforant pathway from the superficial layer of the entorhinal cortex (Kudjakova, Sarycheva, and Kamenskii 2007) and one of the most sensitive regions to damage during epileptogenesis.
Potential Role of the Olfactory Bulb as a Proximal Site of Action for Domoic Acid
The dendrodentric connections between mitral cells and granule cells in the external plexiform layer of the olfactory bulb provide an optimal target for domoic acid–induced neurotoxicity as an initial site of action. Domoic acid induces c-fos, a marker for neuroexcitation in olfactory bulb granule cells (Peng et al. 1994). A study by Scallet et al. (2004) demonstrated that domoic acid, administered at a dose below a behavioral response threshold, induced c-fos in the anterior olfactory cortex. By contrast, c-fos was not induced in the hippocampus at this dose but required a higher dose, sufficient to induce convulsions, before c-fos induction was observable in the hippocampus. An excitotoxic action of domoic acid in the olfactory bulb at dendritic GABAergic spines of the olfactory granule cells would release inhibition of mitral cells and enhance monosynaptic glutamatergic activation of downstream targets via the lateral olfactory tract through the anterior olfactory cortex, piriform cortex, and entorhinal cortex. Neuroexcitation in these areas and their downstream targets would likewise amplify excitotoxicity in the presence of domoic acid. Excitotoxic damage leading to nerve cell degeneration is likely exasperated by reverberation of the downstream and contralateral excitatory connections of the olfactory cortex, which can entrain other regions of the limbic brain, especially the hippocampus. Indeed, a study by Gary et al. (2002) found that olfactory bulbectomy results in a 3-fold reduction of damage to hippocampal pyramidal cells following application of kainic acid directly to the hippocampus.
Clearly many studies have established that domoic acid induces substantial excitotoxic damage to the hippocampus, which has become a dominant focus of experimental and environmental studies. However, using a protocol designed to administer hourly domoic acid doses just enough to kindle SE, the damage to the olfactory pathways is more extensive than that found in the hippocampus. We propose that the domoic acid–induced damage to dendritic spines of olfactory granule cells may be a proximal site of domoic acid action that promotes excitability of mitral cells and increases susceptibility to downstream pathways of the olfactory cortex. In this manner, olfactory pathway damage may be sufficient to progress to a chronic state of recurrent spontaneous seizures and aggressive behavior observed long after the domoic acid is eliminated; the hallmark of chronic neurological disease in the California sea lion.
Footnotes
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This publication does not constitute an endorsement of any commercial product or intend to be an opinion beyond scientific or other results obtained by the National Oceanic and Atmospheric Administration (NOAA). No reference shall be made to NOAA, or this publication furnished by NOAA, to any advertising or sales promotion which would indicate or imply that NOAA recommends or endorses any proprietary product mentioned herein, or which has as its purpose an interest to cause the advertised product to be used or purchased because of this publication.
The authors received no financial support for the research, authorship, and/or publication of this article.
