Abstract
If nonhuman primates represent the only relevant species for nonclinical safety evaluation of biotechnology-derived products, male and female fertility effects can be assessed in repeat dose toxicity studies given that sexually mature monkeys are used. This opinion piece provides recommendations for determining sexual maturity and when/how fertility assessments should be conducted in the cynomolgus monkey. Male sexual maturity should be proven by presence of sperm in a semen sample, female sexual maturity by at least two consecutive menstrual bleedings. As per regulatory guidance, default parameters for an indirect assessment of fertility in both sexes are reproductive organ weight and histopathology. Beyond default parameters, daily vaginal swabs are recommended for females, and for males, it is recommended to include blood collections (for potential analysis of reproductive hormones), testis volume sonography, and collection of frozen testis samples at necropsy. Only if there is a cause for concern, blood collection for potential reproductive hormone analysis should be conducted in females and semen analysis in males. In principle, adverse reproductive effects can be detected within 4 weeks of test article administration, depending on study design and reproductive end point chosen. Therefore, there are options for addressing reproductive toxicity aspects with studies of less than 3 months dosing duration.
*This is an opinion article submitted to the Toxicologic Pathology Forum. It represents the views of the authors. It does not constitute an official position of the Society of Toxicologic Pathology, British Society of Toxicological Pathology, or European Society of Toxicologic Pathology, and the views expressed might not reflect the best practices recommended by these Societies. This article should not be construed to represent the policies, positions, or opinions of their respective organizations, employers, or regulatory agencies.
Keywords
For the safety of volunteers participating in clinical trials for new medicinal products, the trials need to be supported by appropriate nonclinical safety studies. Timing and scope of these nonclinical studies are outlined in the guidance document M3(R2) issued by the International Conference on Harmonization (ICH) of technical requirements for registration of pharmaceuticals for human use. The general goal of the nonclinical safety evaluation, which is usually conducted in two different animal species, is the identification and characterization of hazards; their relationship to exposure; and, if appropriate, their potential reversibility. One important aspect of nonclinical safety evaluation is the detection of any adverse effect on reproduction. This includes developmental toxicity in the offspring as well as adverse effects on sexual function and fertility in adult males and females. The general strategy on how to approach reproductive toxicity assessment is outlined in the ICH guidance S5(R2) from November 2000, which is currently under revision (adoption scheduled for November 2019), and in the ICH guidance S6(R1) from June 2011 with specific recommendations for testing biopharmaceuticals. For most conventional (small molecule) medicinal products, the conduct of three different studies is recommended, addressing the followings: (1) fertility and early embryonic development, (2) pre- and postnatal development (PPND) including maternal function, and (3) embryo-fetal development (EFD). These studies are typically performed in rodents and rabbits. However, for many biopharmaceuticals, the conventional testing strategy with three studies in rodents and rabbits is not appropriate if neither rodents nor rabbits show a pharmacological activity that is somehow comparable to that observed in humans. For many of these products, the nonhuman primate (NHP) is the only relevant model. In such cases, ICH S6(R1) allows restriction to a single animal species for safety assessment including developmental and reproductive toxicity testing (single-species testing strategy).
In terms of PPND toxicity assessment, specific study designs are available for NHPs (Weinbauer et al. 2011; Weinbauer, Luft, and Fuchs 2013), which have now become widely accepted and fulfill the regulatory expectations outlined in ICH S6(R1). In contrast, addressing potential adverse effects on sexual function and fertility in male and female NHPs is less well-defined. The addendum to ICH S6(R1) recognizes that mating studies are not practical in NHPs, and it specifically states that: When the NHP is the only relevant species, the potential for effects on male and female fertility can be assessed by evaluation of the reproductive tract (organ weights and histopathological evaluation) in repeat dose toxicity studies of at least 3 months duration using sexually mature NHPs. If there is a specific cause for concern based on pharmacological activity or previous findings, specialized assessments such as menstrual cyclicity, sperm count, sperm morphology/motility, and male or female reproductive hormone levels can be evaluated in a repeat dose toxicity study.
In rodents, fertility is typically investigated by test article administration to male and female animals during a premating period and throughout mating. Test article–dosed male animals are mated with untreated females and vice versa. In contrast, mating studies for assessing fertility are not recommended in long-tailed macaques because fertility rates are comparatively low. Pregnancy rates are in the range of 25% to 45% per ovarian cycle and 60% per animal. Preimplantation losses are around 25% (Binkerd, Tarantal, and Hendrickx 1988), and prenatal losses can be as high as 40% (Chellman et al. 2009; Jarvis et al. 2010). Another limitation is that—unlike in rodents, rabbits, dogs, and minipigs—females give birth to one offspring only. Twin pregnancies/births are extremely rare in macaques, with an overall incidence of twin live births around 0.1% (Hendrie et al. 1996; Jarvis et al. 2010). It should be mentioned that physical mating studies could be feasible and justified in NHP models under special circumstances and concerns, for example, if the test article is suspected to interfere directly with mating behavior, fertilization, or implantation. Overall, however, specific male and female reproductive end points are typically incorporated into repeat dose NHP general toxicity studies for an indirect assessment of fertility.
The actual need for fertility testing depends on the product, the clinical indication, and the intended patient population, since nonclinical findings of toxicity regarding male or female fertility are generally used to determine whether a specific clinical assessment of fertility is recommended (Food and Drug Administration 2015a) and to support appropriate product labeling (Food and Drug Administration 2015b). The guideline ICH S6(R1) specifically mentions that public information might be available, which obviates the need for formal reproductive toxicity studies. Concerning this matter, the FDA draft guidance on testicular toxicity (Food and Drug Administration 2015a) lists some conditions, under which additional evaluations of fertility may not be warranted. Specifically, these are (1) class of drug (e.g., chemotherapeutics), (2) drug indication (e.g., treatment of severe or life-threatening conditions), and (3) intended patient population (e.g., females beyond menopause or patients with life-threatening illness). These circumstances may justify that male or female fertility not be addressed in nonclinical studies. Additional guidance on the strategy for reproductive toxicity assessment is given in the draft (currently step 3) guideline ICH S5(R3) from August 2017.
According to the addendum to ICH S6(R1), assessment of organ weights and histology of reproductive organs in repeat dose NHP toxicity studies are generally considered sufficient to address male and female fertility, and a specialized assessment is only needed if there is a specific cause for concern based on pharmacological activity or previous findings. There is no published definition for this “cause of concern.” Nevertheless, the FDA draft guidance on testicular toxicity (Food and Drug Administration 2015a) lists some general aspects that might raise concern for a more detailed fertility assessment: any pathology that may suggest impaired reproductive function, any pathology that correlates with changes in organ weight, findings that occur in multiple species, findings that persist after drug withdrawal, findings that do not provide a reassuring safety margin compared to clinical exposure, and findings suggestive of perturbations of the endocrine system.
While most of these aspects mentioned in the FDA draft guidance (Food and Drug Administration 2015a) apply to all sorts of adverse findings rather than only adverse effects on fertility, they represent an oversimplification and cannot serve as a comprehensive guidance on when to implement additional fertility end points. Specifically, accepted definitions of adversity are lacking for findings in male and female reproductive organs.
Since histopathology of reproductive organs is an important parameter for an indirect assessment of fertility, the study pathologist is often in a good position to help correlate across end points. This opinion piece is intended to make study pathologists aware of the nonpathology end points that are typically integrated in NHP studies with an indirect assessment of fertility, and it shall help in data interpretation in scope of the risk assessment process.
When to Assess Male and Female Fertility in Long-tailed Macaques
In cases where male and female fertility need to be investigated in long-tailed macaques, this is usually done in the scope of repeat dose general toxicity studies. Hence the question arises, to which specific study an indirect assessment of male and female fertility should be added? There is no simple answer to this question, but generally these end points should be addressed as early as possible in order to safeguard male and female patients participating in clinical trials (see ICH M3[R2] for further guidance on the appropriate timing of reproduction toxicity studies). Yet, both the selection of end points to indirectly evaluate fertility and the duration of test article exposure are of great importance. Consequently, first-in-human clinical trials are often supported by a risk assessment based on literature data rather than dedicated investigations in long-tailed macaques. As will be explained in this article, there are options for indirectly addressing fertility aspects with studies of less than 3 months duration (referring to continuous test article exposure) and thus early in the drug development program.
Once the decision has been made to which study an indirect assessment of male and female fertility will be added, it is important to assure that long-tailed macaques enrolled in this study are sexually mature, since adverse effects on male or female reproduction can only be determined in animals that have reached sexual maturity. It is the authors’ opinion that it is sufficient to indirectly evaluate male and female fertility in one well-designed study, rather than addressing this aspect in every long-term toxicity study.
How to Prove Sexual Maturity in Long-tailed Macaques
In studies, where the age of animals indicates that they are certainly immature, there is limited use of a microscopic analysis of reproductive organs in order to address male/female fertility.
In cases where sexual maturity is likely but unknown, it is recommended to conduct a thorough histopathological analysis of male and female reproductive organs. Histopathology in combination with testicular weight is able to determine the maturity of the testis (Haruyama et al. 2012a), and there is a likelihood that testicular toxicity can be determined if a sufficient number of sexually mature animals is present in the study. In females, microscopy of the ovary and uterus may allow determination of sexual maturity, but differentiation from puberty can be difficult in animals with nonovulatory cycles.
Wherever possible, sexual maturity should be proven before animals are enrolled in a study that aims to address male/female fertility end points.
Male Animals
Male fertility comprises the development, maturation and release of gametes, sperm composition, mating behavior, and finally fertilization of ova and, therefore, can only be investigated in animals that are sexually mature. The immature testis, apart from having incomplete germ cell development, has an approximately 10-fold lower number of Leydig cells (Verhagen et al. 2014) and Sertoli cells (Marshall and Plant 1996) than an adult testis, lacks the G protein–coupled estrogen receptor in peritubular cells (Sandner et al. 2014), and possesses Sertoli cells with limited responsiveness to follicle stimulating hormone (FSH; Bhattacharya et al. 2015). Another important difference between immature and mature testis is the formation of the blood–testis barrier (also known as the Sertoli cell barrier), formed by tight junctions, adherens junctions and gap junctions between Sertoli cells. This barrier allows controlling the adluminal environment. Spermatogonia are located outside of this barrier; hence they could be more easily exposed to toxic substances opposed to spermatocytes and spermatids.
Testicular maturation during puberty is not a single event but a gradual change in testicular cell composition and function (Simorangkir et al. 2012). Histological studies on the pubertal establishment of spermatogenesis in macaques concluded that there is no privileged region for spermatogenic activity and that spermatogenesis occurs homogenously, irrespective of the part of testis examined or the arterial blood supply (Dang and Meusy-Dessolle 1984). Based upon serial testicular biopsies, it was concluded that in humans it takes 2 to 5 months from the occurrence of meiosis (i.e., presence of spermatocytes) to the appearance of elongated spermatids, that is, testicular spermatozoa (Dang and Meusy-Dessolle 1984). The corresponding duration in cynomolgus macaques is around 1.2 months, but full sperm counts in macaques are only achieved around 5 to 6 months after first occurrence of spermatocytes (Luetjens and Weinbauer 2012).
Sexual maturity of male macaques can be defined by completion of spermatogenesis, which typically occurs before epiphyseal closure. Completion of spermatogenesis can be proven by the presence of sperm in a semen sample, and in fact it is the recommended functional parameter of sexual maturity in male monkeys (Chellman et al. 2009; Luetjens and Weinbauer 2012). The ability to produce an ejaculate and the presence of sperm in a semen sample provide sufficient evidence of complete maturation of the entire male reproductive tract.
The onset of sexual maturity in cynomolgus macaques is difficult to predict and is subject to high interindividual variability. Generally, cynomolgus macaques from Mauritius seem to mature earlier than those from Asian origin (Luetjens and Weinbauer, 2012). To bypass the need for obtaining semen samples, several surrogate parameters for sexual maturity have been suggested, such as age, body weight, or testis volume. However, since the sexual maturation in NHPs shows high interindividual variability, none of these parameters is reliably predictive of testicular maturity. An age of 5 years and 5 months combined with a body weight of at least 5.3 kg is supposed to be associated with a 90% probability of testicular maturity (Smedley et al. 2002), but some uncertainty remains (Lawrence and Saladino 2009). A testicular volume of more than 10 ml may also predict sexual maturity (Ku et al. 2010). However, the methodology used to determine testicular volume is important. It has been shown that caliper measurements underestimate testicular volume, while ultrasound measurements more accurately reflect testicular volume (Ramaswamy and Weinbauer 2015). In addition, a testis weight above 20 g is a good indicator of testicular maturity at necropsy (Haruyama et al. 2012a). Maturation of the male accessory reproductive organs correlates with testicular maturation and these organs are fully developed when the testes are fully functional, that is, when they produce sperm and testosterone (Haruyama et al. 2012b). Histologically, a testis is fully mature (stage 6 of development), if complete spermatogenesis is observed in the seminiferous tubules and a moderate or large number of sperm are found in the epididymis (Haruyama et al. 2012a). The single measurement of hormones such as luteinizing hormone (LH), FSH, testosterone, or inhibin B is not a reliable biomarker for sexual maturity, since the secretion of key hormones (e.g., LH and testosterone) is highly pulsatile with short half-lives and consequently large intra- and interindividual variability (Chapin and Creasy 2012). Nevertheless, testosterone levels above 20 nmol/L usually reflect sexual maturity in cynomolgus monkeys (Dreef, Van Esch, and De Rijk 2007).
The authors recommend that sexual maturity in male long-tailed macaques is proven by the presence of sperm in a single semen sample, irrespective of ejaculate volume and number of sperm. Animals that do not give an ejaculate and those with no sperm should be deselected and not included in a study that has the objective to determine potential adverse effects on male fertility. Considering the high interindividual variability in ejaculate volume and sperm count (see subsequent sections on male fertility assessment), the authors do not recommend using these parameters as selection criteria for inclusion into a study.
Female Animals
Female fertility comprises the development, maturation and release of gametes, regular ovarian cycling, mating behavior, fertilization, tubal transport, preimplantation development, and finally implantation. Sexual maturity is reached most commonly in long-tailed macaques that are at least 4 years of age and weigh at least 2.5 kg (Bussiere et al. 2013), but like in males some uncertainty remains. Ovarian cycling in primates is quite different from nonprimate species, particularly with regard to a comparatively long life span of the corpus luteum, irrespective of conception (Weinbauer et al. 2008). The first day of an ovarian cycle is defined as the first day of menstrual bleeding. Occurrence of bleeding varies from one to eight days with an average of 4 (±1) days (Shaikh, Naqvi, and Shaikh 1978). Estradiol levels rise and concentrations peak around day 12 of the ovarian cycle, followed by a peak in LH and FSH, which triggers ovulation. Ovulation occurs around day 12 to 14 of the ovarian cycle with a periovulatory interval of about 3 days. Ovulation is associated with the production of progesterone. Hence, progesterone concentrations rise and peak around day 22 of the ovarian cycle. This luteal phase lasts 14 to 16 days and will again result in menstruation. The entire duration of the ovarian cycle in long-tailed macaques is 30 (±5) days (Weinbauer et al. 2008; Bussiere et al. 2013).
It is important to consider that only about 50% of long-tailed macaques show a regular ovarian cycle when housed individually. Interestingly, up to 80% of animals show regular menstrual cycles when they are group housed (Luetjens and Weinbauer 2013; Niehoff, Bergmann, and Weinbauer 2010; Mitchell et al. 2014). The remaining animals will show variable cycle length within a range of 19 to 69 days (Weinbauer et al. 2008). Duration and regularity of ovarian cycling can be influenced by a large number of confounding factors (Bussiere et al. 2013; Weinbauer et al. 2008). Transportation stress is a well-known factor in this regard and animals should be allowed a three months acclimation time, before their ovarian cycle is assessed. Social housing of long-tailed macaques can markedly affect sexual function, up to a cessation/prolongation of ovarian cycles for several months (Weinbauer et al. 2008; Bussiere et al. 2013). Again, a period of at least three months should be included in studies to allow animals to get acquainted with each other. It is also important to note that female long-tailed macaques do not synchronize their ovarian cycles when housed together (Weinbauer et al. 2008).
The most reliable way of confirming sexual maturity in female macaques is by assessing menstrual bleeding. This can be proven by collecting daily vaginal swabs and by identifying at least two days of bleeding on at least two occasions that are at least 20 days apart (Weinbauer et al. 2008). Vaginal bleeding needs to be assessed by grading the amount of bleeding from the swab (Weinbauer et al. 2013), while mere clinical observation for bleeding is considered feasible but less precise and reliable (
The authors recommend that sexual maturity in female long-tailed macaques is proven by collecting daily vaginal swabs and by identifying at least two days of bleeding on at least two occasions that are at least 20 days apart. Animals with a cycle range of 30 ± 5 days should preferably be included in a study that has the objective to determine potential adverse effects on female fertility (Luetjens and Weinbauer 2013; Mitchell et al. 2014), whereas animals with a cycle duration outside of this range should be deselected for such a study (the authors are aware that availability of sexually mature animals may not always allow deselection of animals with a cycle range outside of 30 ± 5 days, but it is recommended that this limitation is clearly stated in the study report).
How to Assess Male Fertility in Long-tailed Macaques
Once sexual maturity is proven, animals will be repeatedly administered the test article in order to achieve systemic exposure over several weeks or months. A summary on the experimental approach that is recommended by the authors of this opinion piece to indirectly address male fertility in cynomolgus monkeys is provided in Figure 1.
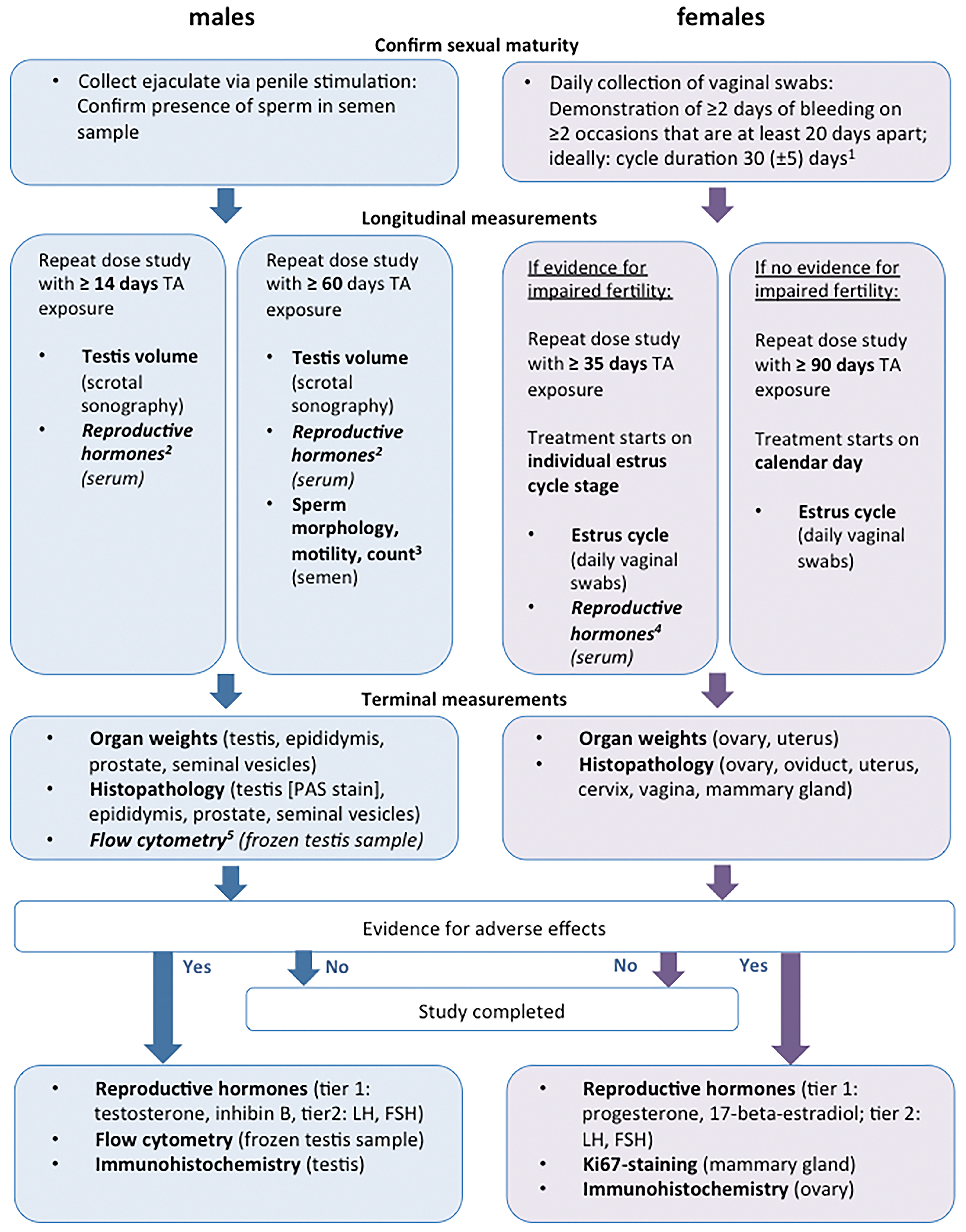
Algorithm how to assess male and female fertility in sexually mature long-tailed macaques. FSH = follicle stimulating hormone; LH = luteinizing hormone; PAS = periodic acid-Schiff; TA = test article; 1 = pending availability of animals, it might not always be possible to exclude animals with a cycle duration outside of 30 ± 5 days; 2 = analysis of hormones only if study reveals evidence for adverse effects on fertility; 3 = sperm count has very low power and needs to be interpreted with caution; 4 = high frequency of blood collection required, analysis only if study reveals evidence for adverse effects on fertility; 5 = archiving of sample, analysis only if study reveals evidence for adverse effects on fertility.
Study Duration
For male reproductive parameters, the duration of dosing (i.e., continuous test article exposure) can vary depending on the end point(s) selected for detection of effects on male reproduction. Histopathology of the testis and epididymis can determine development, maturation, and release of gametes and according to ICH S5(R2) is considered the most sensitive end point for detecting adverse effects on the spermatogenic process. The germinal epithelium of the testicle is composed of Sertoli cells and spermatogonia. The latter are morphologically divided into type A cells with dark nuclei (reserve spermatogonial stem cells which do not divide) and type A cells with pale nuclei; these are the spermatogonial stem cells that undergo mitosis and produce type B spermatogonia which have round nuclei and heterochromatin attached to the nuclear envelope and the center of the nucleolus. Type B spermatogonia move to the adluminal compartment and become primary spermatocytes (Figure 2). The primary spermatocyte undergoes reductional division (meiosis I), which reduces the ploidy from diploid to haploid. Meiosis I and II are divided into prophase, metaphase, anaphase, and telophase stages. Morphologically, the prophase of meiosis I can further be divided into distinct stages (leptotene, zygotene, pachytene, diplotene, diakinesis), and consequently, primary spermatocytes as they appear in the seminiferous tubules are morphologically divided into preleptotene spermatocytes, leptotene spermatocytes, zygotene spermatocytes, and pachytene spermatocytes (Figure 2). Meiosis I of primary (diploid) spermatocytes results in secondary (haploid) spermatocytes, which undergo meiosis II, which is an equational division, resulting in two spermatids. Spermatids begin to form a tail by forming microtubules, which are known as the axoneme. The anterior part of the tail is called the midpiece. It thickens because mitochondria are arranged around the axoneme. In parallel, the nucleus condenses and the Golgi apparatus becomes the acrosome. As spermatids elongate, the excess cytoplasm is phagocytosed by surrounding Sertoli cells. Mature but immotile spermatozoa are released into the lumen of the seminiferous tubule. They acquire motility while in the epididymis.
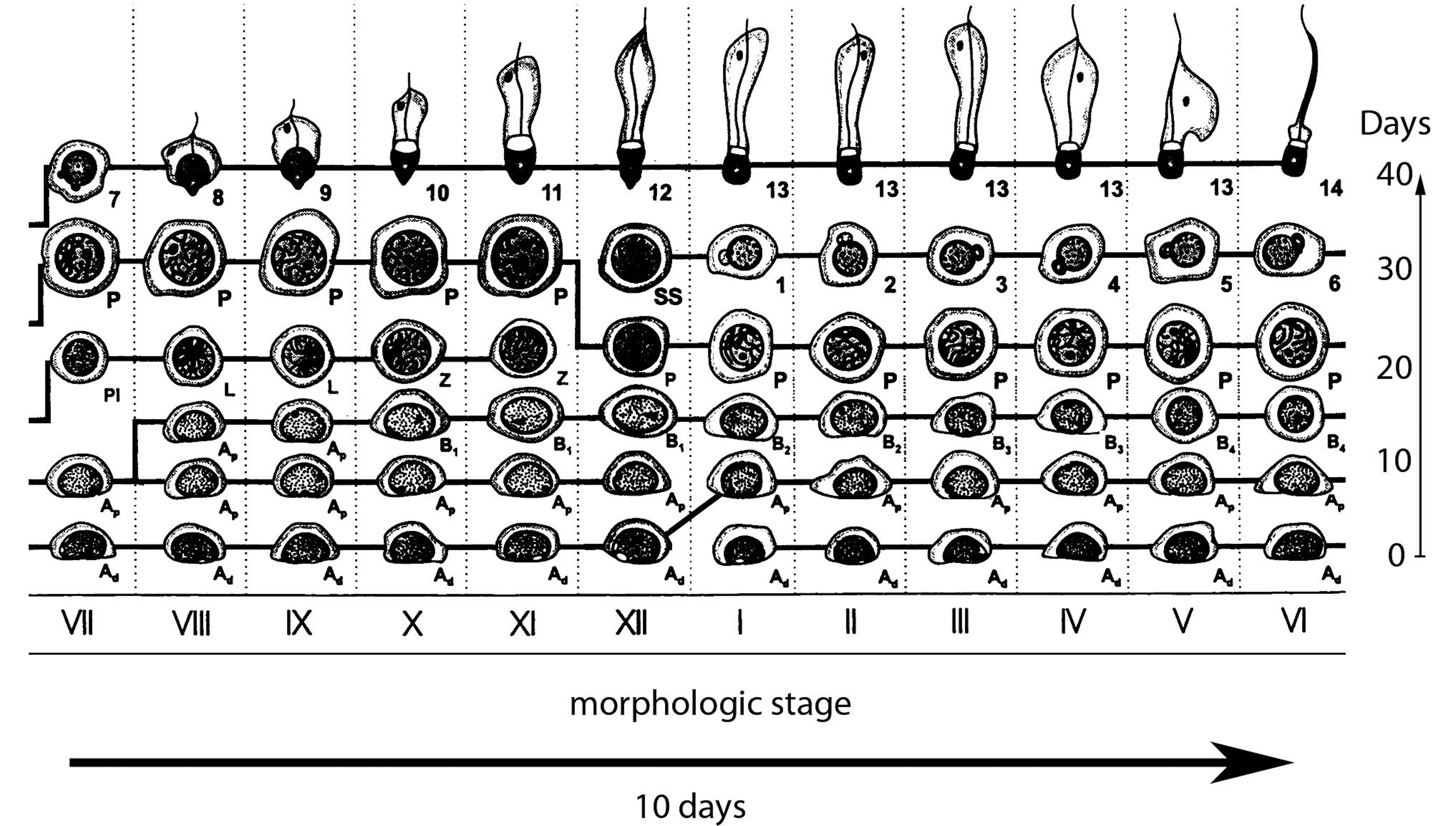
Graphic representation of the 12 spermatogenic stages in the cynomolgus monkey. Ad = dark type A spermatogonium; Ap = pale type A spermatogonium; B = type B spermatogonium; L = leptotene spermatocyte; P = pachytene spermatocyte; PL = preleptotene spermatocyte; Z = zygotene spermatocyte; 1 to 14 = type 1 to 14 spermatids.
Spermatogenesis in the long-tailed macaque can morphologically be divided into 12 stages (Dreef et al. 2007). One cycle of the seminiferous epithelium/spermatogenic cycle takes approximately 10 days (Aslam et al. 1999; Zhengwei et al. 1998), and 4 cycles, that is, 40 days, are needed from spermatogonial division to spermiation, that is, the release of mature spermatozoa (Aslam et al. 1999; Luetjens, Weinbauer, and Wistuba 2005; Figure 2). Hence, for test articles acting directly on the testis and for test articles that acutely suppress gonadotropic hormone secretion, altered histopathology can be evident within 14 days of dosing initiation (Ramaswamy and Weinbauer 2015; Weinbauer, Hoffmann, and Luetjens 2016, Figure 2); although morphological changes may be difficult to detect and require a very thorough examination of testicular tissue with awareness of the spermatogenic cycle (see below).
As the conversion of proliferating spermatogonia into fully developed testicular spermatozoa requires approximately 40 days, and since another five days are required for epididymidal maturation/transit of sperm (Amann et al. 1976; Wen and Yang 2000), it can take up to 45 days until defects can be recognized in epididymal sperm or ejaculated sperm. It has been recommended that for detection of adverse effects on sperm in semen or the cauda epididymidis, the dosing period should be at least equal to six times the duration of one cycle of the seminiferous epithelium (Amann 1986). For macaques, this would correspond to at least 60 days of dosing.
In summary, a dosing phase of 14 days may suffice to detect adverse effects on spermatogenesis in the long-tailed macaque based upon detailed histopathology, while a dosing period of at least 60 days will be required to detect adverse effects on spermatogenesis based upon sperm analysis in semen samples. As per ICH S6(R1), test article exposure (dosing) of at least three months is recommended in order to assess potential effects of male fertility based upon histopathology and reproductive organ weights. It is the opinion of the authors, however, that well-designed studies with considerably shorter treatment duration will suffice to assess potential adverse effects on male fertility when based upon testicular histology and organ weights. Studies of less than 3 months duration have the advantage that they can histologically reveal the actual target cell of toxicity in the testis, opposed to longer duration studies which may only show end-stage morphological findings such as seminiferous tubule atrophy.
If reversibility of effects on the male reproductive system is to be investigated, the duration of the recovery phase should take into consideration the clearance of the test article and the duration of the spermatogenic cycle. Duration of recovery should also consider the sensitivity of the end point that is chosen to indirectly examine male fertility. While it may require two to three full spermatogenic cycles until complete recovery of sperm numbers can be demonstrated in semen (if at all keeping the variability of sperm numbers in mind, see below), one to two spermatogenic cycles can be sufficient to demonstrate reversibility by histopathology and testis size, depending at which germ cell level the spermatogenic process had been disrupted and how consistent this disruption is across the entire testis. For example, in cases where spermatogenesis is inhibited at the spermatogonial level, testis size completely recovers within 10 weeks upon cessation of treatment, while recovery of full sperm numbers is seen by 18 weeks upon cessation of treatment (Ramaswamy and Weinbauer 2015). This difference of approximately eight weeks between recovery of testis size and sperm number in semen is compatible with the reported duration of approximately 40 days for the development from differentiated spermatogonia into testicular spermatids.
Group Size
A group size of at least 5 animals (typically separated into 3 animals that will be euthanized directly after test article exposure and an additional 2 animals that are used to investigate recovery) should be employed in long-tailed macaque studies indirectly investigating male fertility. This means a group size of 3 for terminal parameters and a group size of 5 for in-life parameters during the drug exposure phase. The group size of 3 to 5 animals yields low statistical power for some male reproductive end points, but it is acceptable for a number of parameters (Table 1). Statistical power is 10% to 35% for reproductive organ weights, below 10% for ejaculate volume and sperm number, but over 70% for sperm morphology and motility. It is below 20% for LH and testosterone concentrations in serum, but over 50% for concentrations of FSH and inhibin B (Cappon et al. 2013). Therefore, in most cases, interpretation needs to be based on a weight-of-evidence approach rather than statistical significance. Power for testicular/epididymal histopathology has not been calculated but is considered to be fairly high (ICH S5[R2]).
Statistical Power Estimates for Various in-life and Terminal Parameters to Detect a 50% Change in Male and Female Long-tailed Macaques, Considering a Group Size of 3 Animals for Terminal Parameters and a Group Size of 5 Animals for In-life Parameters.

aParameters derived from Cappon et al. (2013).
bComparable results were obtained when using data from different publications (Bussiere et al. 2013; Luetjens and Weinbauer 2013; Weinbauer et al. 2008).
cFlow cytometry yields five testicular cell populations (HC/1C/2C/S-ph/4C) with close to or acceptable statistical power of 80% except for HC cell population (power is 20%) which contributes <10% of cells.
Routine Assessment of Male Fertility
As stated above, fertility in long-tailed macaques is only assessed indirectly. The standard parameters as per ICH S6(R1) are reproductive organ weights (specifically testicular weight, epididymal weight, weight of the seminal vesicles, and prostate weight) and histopathology. Due to difficulties in its preparation, the prostate weight usually shows high interindividual variability and poor statistical power (Table 1), but it is still considered a useful parameter in conjunction with the other organ weights and histopathology. Reduction of seminal vesicle weight is a reliable indicator of a reduced androgenic stimulus (Chapin and Creasy 2012).
Histopathology should be conducted on testes, epididymides, prostate gland, and seminal vesicles. As emphasized by ICH S5(R2), proper fixation of the testis is important (Latendresse et al, 2002). A recommendation about trimming and wax embedding of male reproductive organs, which allows discrimination between right and left organs, is shown in Figure 3. Histopathology of the testes and epididymides is a reliable method to confirm sexual maturity and to detect adverse effects on spermatogenesis. If not already proven by the presence of sperm in a semen sample (which is the recommended approach), the histopathological assessment of the testis should begin with an assessment of testicular maturity. Based on the number and desquamation of spermatids in association with testicular weight, six distinct developmental stages from an immature testis to an adult testis have been described for cynomolgus monkeys (Haruyama et al. 2012a). The pathology report should mention whether the examined testis was mature (stage six as described by Haruyama et al. 2012a), was in puberty (stages 2 to 5 as described by Haruyama et al. [2012a]), or was immature (stage 1 as described by Haruyama et al. [2012a]). There is an ongoing debate as to whether a stand-alone pseudo-organ (e.g., sexual maturity) should be included in the pathology report for this purpose or if the status of maturity is recorded under testis.
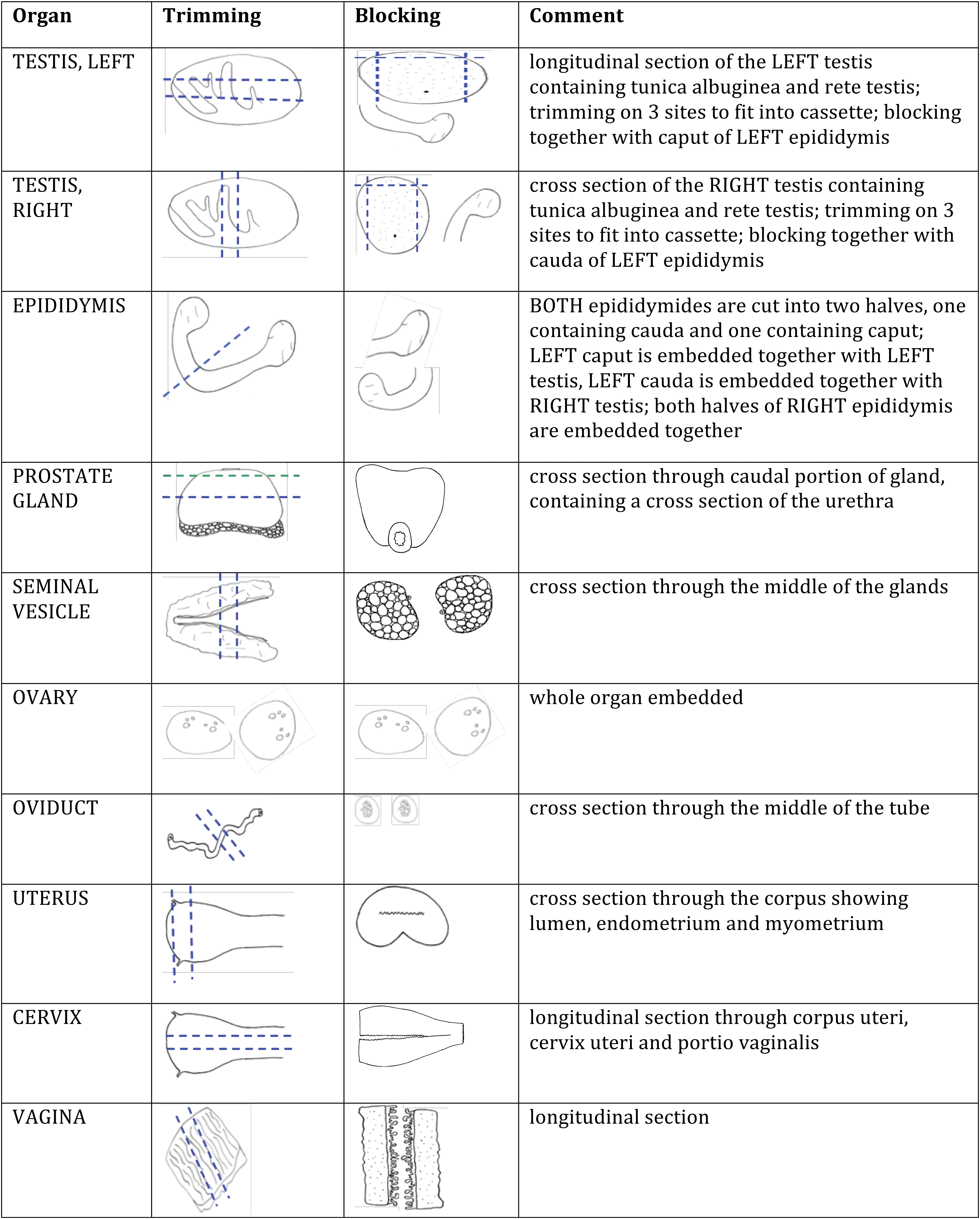
Recommendation for the trimming of male and female reproductive organs from long-tailed macaques.
Where possible, the terminology that is used to describe morphological changes in the testis and epididymis should follow the recommendations made by the International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice (INHAND) project (Creasy et al. 2012), which was developed for rodents but can be used for NHPs as well (specific INHAND terminology for NHP is in process). Morphological terms should be allocated to specific cell populations within the testis (e.g., germ cells, Sertoli cells, Leydig cells) or specific anatomical regions of the excurrent duct system (e.g., rete testis, efferent ducts, caput epididymis, cauda epididymis, vas deferens). Histopathological evaluation of testicular seminiferous tubules should be conducted qualitatively with a thorough understanding of the spermatogenic cycle (Foley 2001; Dreef et al. 2007). The system suggested by Dreef et al. (2007) differentiates 12 stages
Morphological changes induced by a test article need to be differentiated from spontaneous background findings. Common testicular findings in control cynomolgus monkeys are tubular degeneration and tubular atrophy, which represent a continuum of germ cell degeneration (Chamaza et al. 2010). Incidence rates of such findings vary across laboratories. It is recommended that historical reference data is only derived from animals with proven sexual maturity. Most common morphological findings in sexually mature male cynomolgus monkeys according to the experience of these authors are minimal to moderate seminiferous tubule atrophy, and mononuclear inflammatory cell infiltrates in testis, epididymis, or prostate gland. Testicular fibrosis (synonym: increased stromal collagen) is an uncommon observation in sexually mature long-tailed macaques, but when present is often associated with cystic tubular atrophy of the seminiferous epithelium (Pereira Bacares, Vemireddi, and Creasy 2017). Histopathology should also be conducted on the prostate gland and seminal vesicles. Hyperplasia of the prostate gland and squamous metaplasia are well-known consequences of a chronic estrogenic stimulus (Heywood and Wadsworth 1980). Histopathology of the male mammary gland could also be considered, since glandular lobules respond to endocrine stimuli (Tavassoli, Casey, and Norris 1988).
Testis volume is another useful parameter that is recommended to be included in long-tailed macaque toxicity studies that indirectly investigate male fertility. Testis volume is easily measured serially using ultrasonography or calipers. The use of ultrasonography is preferred since this approach closer reflects actual testicular weight compared to caliper measurements, which tend to underestimate testicular weight (Ramaswamy and Weinbauer 2015). A measurement should be conducted once before treatment and at least twice during/after treatment. In case of alterations in testicular weight and histology, the ultrasonography data can add valuable information with regard to the occurrence and development of this finding over time.
Specialized Assessment of Male Fertility
The guideline ICH S6(R1) recommends that a specialized assessment of sperm count, sperm morphology/motility, and reproductive hormone levels should be conducted, if there is a specific cause for concern. In such cases, the following end points can be considered on a case-by-case basis for an indirect specialized assessment of male fertility: (1) collection and analysis of semen, (2) blood collection and analysis for male reproductive hormones, (3) quantitative evaluation of testicular cell suspensions by flow cytometry, (4) immunohistochemistry on testicular tissue, and (5) prostate size measured via transrectal ultrasonography.
Semen is typically collected via electrostimulation—either rectal probe stimulation or penile stimulation, with the latter being clearly recommended over the rectal approach for animal welfare reasons and because of higher semen volumes and sperm numbers (Lanzendorf et al. 1990); sperm fertilizing ability is comparable between the two methods (Gould and Mann 1988). A potential shortfall of penile stimulation to be mentioned is that some animals—unlike with rectal probe stimulation—do not respond to penile stimulation and will not provide an ejaculate. For each animal, semen collection should be conducted at least once before dosing with the test article and then every 4 to 8 weeks throughout the treatment period (Enright et al. 2012). Macaque semen solidifies and the resulting coagulum is digested with a trypsin solution in order to release sperm for enumeration. Following digestion, sperm are usually decapitated and immotile. Hence, for analysis of sperm motility and morphology—either manually or using automated systems—only the liquid portion (exudate) of ejaculated semen is available. End points for evaluation include semen volume, sperm count, sperm motility, and sperm morphology (Weinbauer et al. 1988; Hurtt et al. 1992; Enright et al. 2012). Results from sperm analysis, however, show very high intra- and interindividual variability (Cappon et al. 2013). For a group size of 5 animals, the statistical power is >90% for sperm motility and >70% for sperm morphology, but it is less than 10% for sperm count (Table 1). Therefore, a group size of 5 animals can only reveal major changes in sperm numbers, that is, changes that would also be evident in testicular and epididymal histology. This is in concordance with sperm analysis in humans, where a group size of 100 subjects in order to detect a 50% change is recommended by the FDA draft guidance on testicular toxicity (Food and Drug Administration 2015a). Therefore, semen evaluations are not routinely needed in toxicity studies indirectly addressing male fertility, unless a strong scientific rationale is evident.
Male reproductive hormones that could be analyzed throughout a repeat dose toxicity study are testosterone, FSH, LH, and inhibin B (Weinbauer et al. 2001). Testosterone is produced by testicular Leydig cells upon stimulation by pituitary-derived LH. In macaques, LH secretion is closely followed by a surge of testosterone production. However, there is distinct pulsatility of hormone secretion rendering LH/testosterone levels highly variable. In addition, there are significant circadian differences in hormone secretion, with higher values obtained during the dark phases (Chapin and Creasy 2012). Pituitary-derived FSH stimulates Sertoli cells and secondarily spermatogenesis. Inhibin B is produced by Sertoli cells and provides a negative feedback for FSH secretion from the pituitary. Endocrine factors are considered indicators of testicular damage but with variable sensitivity. Alterations in LH and testosterone levels do not necessarily imply an impact on spermatogenesis. Selective elevation of serum FSH concentrations has been associated with altered spermatogenesis. Serum inhibin B concentrations have been proposed as a biomarker for testicular toxicity (Stewart and Turner 2005). It has been suggested that a pronounced reduction in circulating inhibin B levels signifies severe and potentially irreversible alterations in Sertoli cells and spermatogenesis (Pfaff et al. 2013), and inhibin B serum levels have been shown to decline rapidly after testicular irradiation (Foppiani et al. 1999; Kamischke et al. 2003). However, systematic investigations about the suitability of inhibin B as a biomarker in long-tailed macaques are lacking, and data from humans and rodents indicate that serum levels of inhibin B may not always be suitable as peripheral markers for monitoring testicular toxicity (Meachem, Nieschlag, and Simoni 2001; Chapin et al. 2013). Serum levels of anti-Müllerian hormone have been proposed as a useful biomarker of testicular toxicity in patients based upon correlation with increased FSH and testosterone and decreased inhibin B levels (Levi et al. 2015), but again systematic investigations in nonclinical studies have not been performed so far.
The authors recommend that blood samples are collected from each long-tailed macaque toxicity study that aims at determining potential adverse effects on male fertility. To capture pulsatility and circadian hormone patterns is technically feasible but practically extremely challenging due to the necessity of frequent blood sampling throughout a 24-hr period. For practical purposes and in our experience, morning samples (e.g., 08:00–10:00 a.m.) are considered sufficient and it is important that the sampling times are kept constant within animals and throughout a study. Blood samples should be collected at least once before dosing and twice throughout dosing. Analysis of male reproductive hormones, however, should only be conducted retrospectively, if other parameters (organ weights, histopathology, and testis volume) have revealed adverse effects which could be associated with hormonal disturbances. In this case, access to appropriately validated species-specific assays is required, because gonadotropic hormones, unlike steroid hormones, are highly species specific. It is recommended to analyze testosterone and inhibin B first and to proceed with determination of gonadotropic hormones only if testosterone/inhibin B levels are altered.
Due to the pulsatility of LH and testosterone secretion, statistical power is low for these hormones while power is above 50% and 90% for inhibin B and FSH, respectively (Table 1). This variability can be better managed by repeated sampling and an intraindividual analysis rather than making comparison across dose groups. LH and testosterone levels should only be evaluated in conjunction with other androgen-dependent end points (e.g., body weight, organ weight, and histology of seminal vesicles and prostate). It is important to recognize that changes in testicular histology usually do not reflect androgen deficiency in primates, where spermatogenesis—unlike in rodents—particularly depends on FSH (Ramaswamy and Weinbauer 2015).
Further methods on analyzing testicular cell composition and function in testicular tissue are still in an experimental stage, but they may emerge as valuable add-on tools in cases where further mechanistic insight into testicular toxicity is needed. The quantitative evaluation of testicular cell suspensions by flow cytometry is one such technique that has shown good correlation to histopathology (Weinbauer et al. 2016). Briefly, suspended cell DNA is stained with propidium iodide, and five specific cell populations are recorded: elongated spermatids (HC), round and elongating spermatids (1C), spermatogonia and somatic cells (2C), cells in DNA synthesis phase (S-ph), and spermatocytes and G2 spermatogonia (4C); cellular debris is also recorded (Aslam et al. 2002). Interestingly flow cytometry data achieves considerable statistical power even with a group size of 3 animals, with power values of 74% to 99% (Figure 4, Table 1). For HC, cells power is around 20%, but this cell population only represents less than 10% of all testicular cells (including somatic cells). While this method is still in the early stages of development, the authors recommend that a fragment of testicular tissue is frozen in liquid nitrogen from every repeat dose toxicity study whose objective is to assess potential effects on male fertility for potential subsequent analysis by flow cytometry.
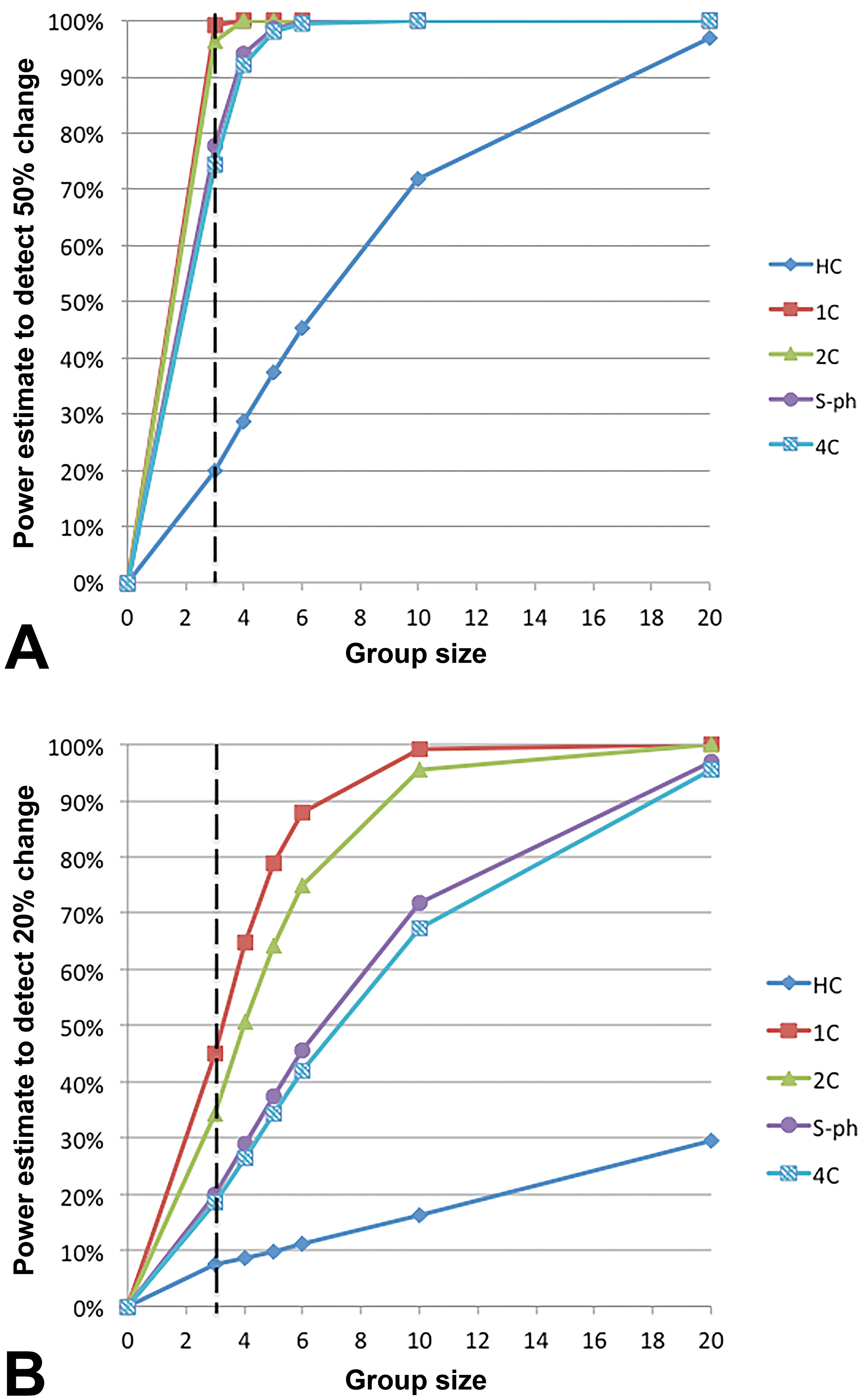
Statistical power estimates for detecting a 50% change (A) or a 20% change (B) in testicular cell populations determined by flow cytometry. The power estimate assumes that analysis is done with a two-sided two-sample
Transmission electron microscopy or immunohistochemistry may be useful for determining the target cell of toxicity in the testis. Leydig cells can be labeled by staining for the enzyme 3β-hydroxysteroid dehydrogenase (Verhagen et al. 2014), and Sertoli cells are typically labeled with GATA-4 (Ketola et al. 2000), a member of the GATA transcription factors, characterized by their ability to bind to the DNA sequence “GATA.” Reserve germ cells are characterized by the nuclear expression of a protein known as deleted in azoospermia-like (Levi et al. 2016). The functionality of the blood–testis barrier can be examined by using hypertonic tissue fixation (Pfaff et al. 2013).
Prostate gland size can be measured serially via transrectal ultrasonography (Kamischke et al., 1997). Implementing repeated sonographic measurements of the prostate size can be considered for test articles that are known to influence androgen levels.
How to Assess Female Fertility in Long-tailed Macaques
Once female cynomolgus monkeys have been acclimated to housing conditions for at least three months and once ovarian cyclicity has been proven, animals will be repeatedly administered the test article in order to achieve systemic exposure over several weeks or months. There are basically two options for a study design that includes an indirect assessment of female fertility (Figure 1). One option is to dose all female animals on the same calendar day, which is typically the case for repeat dose toxicity studies. Alternatively, females could be dosed on the same day of their ovarian cycle. This is a less common approach but is the preferred approach for studies specifically investigating female fertility (Weinbauer et al. 2008; Chellman et al. 2009). The advantage of this approach is that all ovarian cycle parameters can be normalized to the day of ovulation thus reducing the interindividual variability. This approach allows a shorter duration of treatment to reveal effects on cyclicity; the disadvantage of this approach is that each animal has a specific study time line with inherent logistical and operational challenges. Therefore, it is difficult to implement this design in a standard general toxicity study. However, it is important that scientists are aware of this concept and that they consider dedicated satellite groups of sexually mature females in short-term studies, if effects on ovarian cycling are to be expected.
Study Duration
For female reproductive parameters, the duration of dosing is essentially related to the duration of the ovarian cycle. Typically, an entire cycle duration (on average 30 days in the long-tailed macaque) is required in order to demonstrate adverse effects. It is important to note that ovarian cycles in the long-tailed macaque are not synchronized (Weinbauer et al. 2008).
As outlined above, animals can either be dosed on the same calendar day or dosing is initiated on the same day of every animal’s ovarian cycle. For practical purposes, dosing on the same calendar day is most often used in general toxicity studies. In this case, however, it is the author´s opinion that treatment duration should at least cover two ovarian cycles, and at least one cycle should be followed after discontinuation of treatment; this will allow for the detection of prolonged ovarian cycles. In terms of study duration (referring to continuous test article exposure), this approach would require at least 90 days of study duration to follow at least 2 complete cycles in all animals (considering that animals are not synchronized) and at least 45 days of recovery.
If dosing is initiated on the same day of every animal’s ovarian cycle, it is considered sufficient to follow just one ovarian cycle, that is, treatment duration can be reduced to 1 month. For example, in case of a NK-3 (tachykinin) receptor antagonist known to interfere with LH synthesis/secretion, dosing started on day 2 of the individual ovarian cycle, lasted for 35 days and was sufficient to reveal effects on cycling (G. L. Fraser et al. 2015). For studying the effects of interferons, animals were dosed for one complete cycle starting at the first day of menstrual bleeding (followed by two treatment-free cycle periods) and the study revealed a delayed mid-cycle hormone peak and prolonged cycle duration (Enright et al. 2012). In the case of antigonadotropic factors, a 30-day study duration with a synchronized dosing start enabled detection of cycle disruption (unpublished data) and detection of related endocrine effects (H. M. Fraser et al. 1997). In conclusion, for indirect assessment of female fertility, the study duration may vary from 1 to 3 months, depending on whether animals are dosed relative to their individual cycle or by calendar day.
If reversibility of effects on the female reproductive system is to be investigated, the duration of the recovery phase should take into consideration the clearance of the test article and that it may require several ovarian cycles until regular cyclicity is resumed.
Group Size
A group size of at least 5 animals (typically separated into 3 animals that will be euthanized directly after test article exposure and an additional 2 animals that are used to investigate recovery) should be employed in NHP studies indirectly investigating female fertility. This means a group size of 3 for terminal parameters and a group size of 5 for in-life parameters. The group size of 3 to 5 animals yields very low statistical power, around 17% and 20% for ovarian and uterus organ weights, respectively (Table 1). Equally low statistical power was shown for female reproductive hormones. In contrast, ovarian cycle duration has high statistical power. It has previously been reported that a group size of 6 would allow detecting a 1.5-fold difference in menstrual cycle length compared to controls with an 80% statistical power (at the significance level of 0.05 and a coefficient of variation of 20–25%; Bussiere et al. 2013). Our own data indicate that a group size of 3 to 5 is sufficient for detecting a 50% change at a power of >80% (Figure 5, Table 1). Comparable power estimates were obtained when retrospectively analyzing data from Weinbauer et al. (2008), Luetjens and Weinbauer (2013), and Bussiere et al. (2013). It should be mentioned that in specific cases, a group size of 4 animals was sufficient for detection of altered ovarian cyclicity (G. L. Fraser et al. 2015).
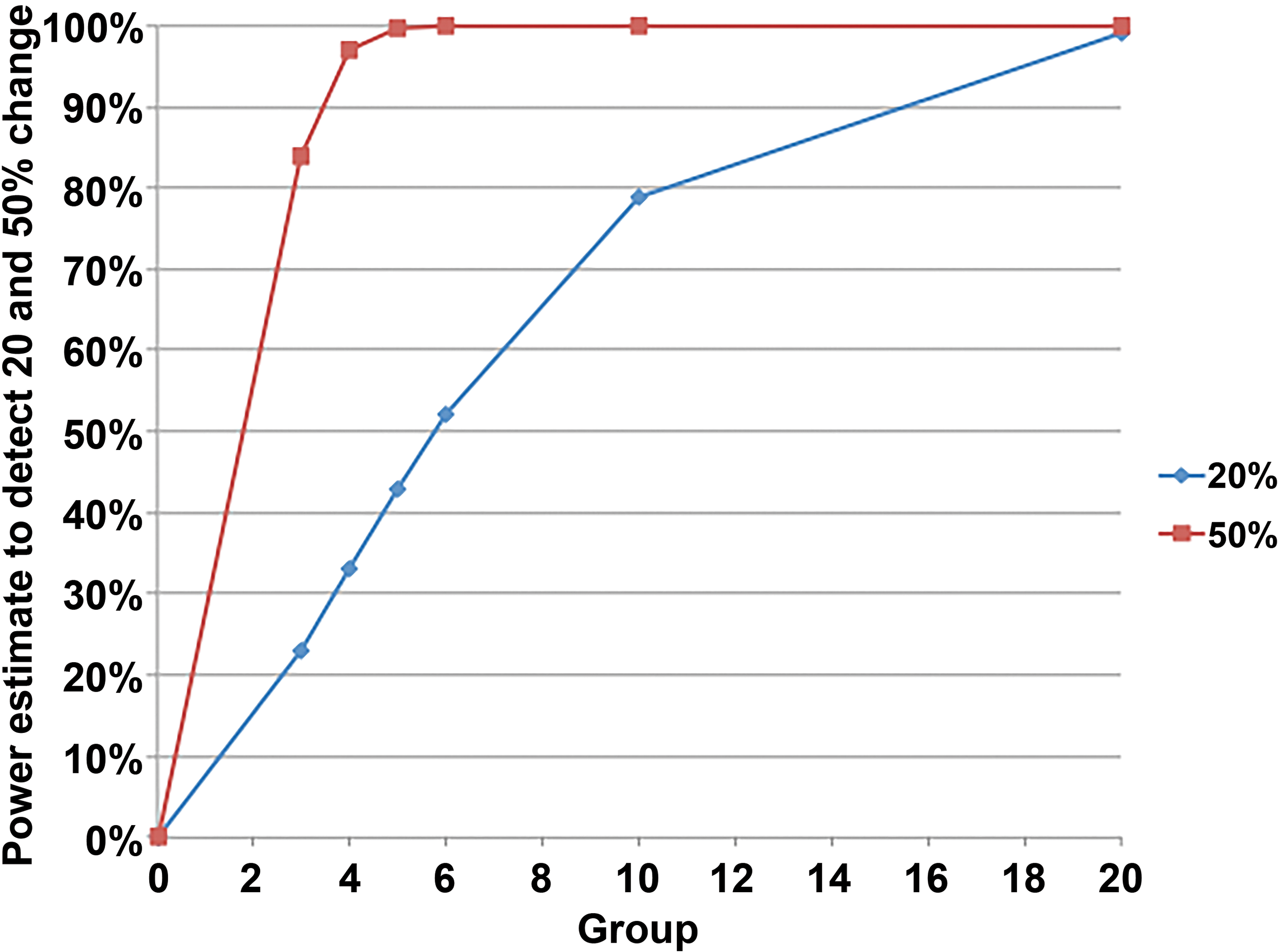
Statistical power estimates for detecting a 20% or 50% change in ovarian cycle duration determined by analysis of daily vaginal swabs in long-tailed macaques. The statistical power estimate assumes that analysis is done with a two-sided two-sample
Routine Assessment of Female Fertility
As stated above, fertility in long-tailed macaques can only be assessed indirectly. Although the standard assessment of female fertility as recommended by ICH S6(R1) is limited to organ weights and histopathological evaluation of reproductive organs, these parameters have low statistical power. In contrast, the statistical power of ovarian cycle duration is above 99% with a group size of 5 animals. Therefore, it is the opinion of the authors that an assessment of ovarian cyclicity should be included in every repeat dose toxicity study that has the objective to determine potential adverse effects on female fertility. Ovarian cycle duration is an easily obtained, reliable, and statistically meaningful parameter (Bussiere et al. 2013). Irregularities in the cynomolgus macaque ovarian cycle can predict impaired fertility in humans (Kolstad et al. 1999; Jensen et al. 1999; Small et al. 2006; Brodin et al. 2008). Test article–related prolongation of the ovarian cycle is not uncommon and is known for particular classes of test articles such as alpha2-interferons or inhibitors of epidermal growth factor signaling (Enright et al. 2009; Bussiere et al. 2013). The ovarian cycle duration might be secondarily affected by general toxicity of a test article, but systematic analyses in this direction have not been performed so far (Williams et al. 2001; Williams, Berga, and Cameron 2007).
Animals are easily trained to tolerate sampling by vaginal swabs (Weinbauer et al. 2013). Vaginal swabs are collected daily over at least 2 ovarian cycles before dosing, and the procedure is continued throughout dosing, monitoring at least two menstrual cycles. The mean/median cycle duration should be determined for each study phase (predose vs. dosing vs. recovery) and for each individual animal. This facilitates interpretation and manages interanimal variability.
Besides the assessment of ovarian cyclicity, standard histopathological evaluation of reproductive organs is recommended as per ICH S6(R1). Details about the histopathological evaluation are not given in the guideline, but the authors of this opinion piece recommend microscopic evaluation of ovary, oviduct, uterus, cervix, and vagina and determination of ovarian weight and uterus weight (despite their low statistical power, see above). A recommendation about trimming and wax embedding of female reproductive organs from long-tailed macaques is shown in Figure 3. As outlined in this figure, it is recommended that the cervix be evaluated in a longitudinal section. Microscopic evaluation of the ovary comprises a thorough qualitative evaluation of all major components, that is, follicles, corpora lutea, stroma, interstitium, and vasculature. Particular emphasis should be placed on primordial and primary follicles in order to detect increased atresia as a test article–related effect (Regan et al. 2005; Appt et al. 2006). The presence and characteristics of corpora lutea are also important. Detailed descriptions of the histomorphology of the ovary throughout the ovarian cycle have been published with a luteinized regressive corpus luteum in the early follicular phase, a dominant large antral follicle in the late follicular phase and various stages of small, growing, antral, and atretic follicles and an old corpus luteum in the menstrual phase (Buse, Zöller, and van Esch 2008; Taketa et al. 2018). Since corpora lutea may be missed on a histological section through the ovary, it is recommended to generate gross photographs of the ovaries before they are embedded in paraffin wax.
When evaluating the reproductive organs, the study pathologist should be aware of the ovarian cycle data obtained from vaginal swabs and should confirm the stage of the ovarian cycle at the time of necropsy (follicular phase, luteal phase, menstrual phase) based on a microscopic evaluation of ovary and uterus. For purposes of documentation, it is recommended to include a separate organ named “ovarian cycle.” This organ would also be the most appropriate place to document sexual immaturity (either immature or peripubertal stage) or any dyssynchrony between ovarian and endometrial stage. The microscopic assessment of reproductive organs from an individual animal should be conducted with consideration of all ancillary data such as organ weights and ovarian cyclicity.
Where possible, the terminology that is used to describe morphological changes in the ovary and remaining female reproductive tract should follow the recommendations made by the INHAND project (Dixon et al. 2014), which was developed for rodents but can be used for NHPs as well (specific INHAND terminology for NHP is in progress). Morphological changes induced by a test article need to be differentiated from spontaneous background findings. Incidence rates of microscopic findings vary across laboratories. It is recommended that historical reference data are only derived from animals with proven sexual maturity. Most common morphological findings in sexually mature female cynomolgus monkeys according to the experience of these authors are parovarian or follicular cysts in the ovary and squamous metaplasia in the cervix (the latter is a common finding in pubertal animals but also occurs in sexually mature females). Rare lesions known to occur in the sexually mature uterus of long-tailed macaques are epithelial plaques, decidual reactions, and dyssynchronous endometrium (characterized by features of both the proliferative and the secretory phase); further microscopic observations are described in Cline et al. (2008).
The female mammary gland is routinely investigated in repeat dose toxicity studies. It undergoes changes that are associated with the ovarian cycle and could therefore also indicate changes in ovarian cyclicity. Proliferation (as detected by staining for the Ki67 antigen) is highest in the luteal phase, while progesterone receptor expression in ducts is lowest in this phase (Buse et al. 2008; Stute et al. 2004).
Specialized Assessment of Female Fertility
The guideline ICH S6(R1) and its addendum recommend that a specialized assessment of ovarian cyclicity and reproductive hormone levels should be assessed if there is a specific cause for concern. The authors of this opinion piece recommend that ovarian cyclicity is included in the routine assessment of female fertility (see above). A specialized (still indirect) assessment of female fertility can comprise the following additional end points: (1) blood collection and analysis for female reproductive hormones, (2) transabdominal ultrasonography for measuring endometrial thickness, and (3) immunohistochemical staining of the ovary.
Female reproductive hormones that can be assessed within a study are progesterone, 17-beta-estradiol, LH, and FSH. As mentioned before, frequent/regular blood sampling is needed and should follow one complete ovarian cycle before dosing as well as one or two ovarian cycles throughout dosing (and one in recovery, if applicable). Many studies include the collection of frequent blood samples, for example, on cycle days 1, 4, 7, 10, 11, 12, 13, 14, 15, 16, 18, 20, 24, and 27 (Enright et al. 2012; Weinbauer et al. 2008). Analysis of hormones is typically only included if vaginal swabs have indicated endocrine abnormalities. However, the practical limitation of this frequent sampling approach is the demand on blood volume and, hence, only a limited number of cycles can be monitored. Collection of blood samples every two to three days for more than one ovarian cycle seems to be a viable alternative, but the use of episodic or weekly sampling is not recommended, as this usually does not allow a sufficiently precise judgment of possible endocrine effects (Weinbauer et al. 2008; Perigard et al. 2016). The statistical power of a single time point female reproductive hormone value is rather low (Table 1). In consideration of these limitations, implementing blood sampling for hormone analysis is not recommended for routine studies indirectly investigating female fertility in NHPs and should only be considered in studies specifically investigating endocrinologic effects. Moreover, hormonal disturbances in female animals are usually secondary and are sufficiently detected by recording ovarian cyclicity and histopathology.
Transabdominal ultrasonography can be used for measuring endometrial thickness (Foster, Stals, and McMahon 1992) or following follicular growth during the ovarian cycle (Bishop et al. 2009), but so far, this technique is not routinely used in regulatory NHP toxicity studies.
In case of suspected test article–related effects on follicles, immunohistochemical staining for beta2-adrenergic receptor or immunostaining for various cytochrome P450 enzymes may be considered for a more detailed analysis of follicles (Merz et al. 2015; Ethun et al. 2012). A quantitative assessment of follicles as suggested for rodents by several guidelines (Regan et al. 2005) has not been established in NHPs and is currently not recommended.
Conclusion
The authors’ opinion and experience has shown that guidance on fertility assessment in NHP is subject to interpretation, particularly with regard to the definition of sexual maturity, when an assessment was needed, and how such an assessment should be conducted.
If nonhuman primates represent the only relevant species for nonclinical safety evaluation, male and female fertility can be assessed indirectly in repeat dose toxicity studies given that sexually mature monkeys are used. Male sexual maturity should be proven by presence of sperm in a semen sample and female sexual maturity by at least two consecutive menstrual bleedings.
As per regulatory guidance, default parameters for both sexes are reproductive organ weight and histopathology. Beyond default parameters, daily vaginal swabs are recommended for females, and for males, blood collections (for potential analysis of reproductive hormones), testis volume sonography, and collection of frozen testis samples at necropsy. Only if there is a cause for concern, blood collection for potential reproductive hormone analysis should be conducted in females and semen analysis should be conducted in males.
While the addendum to ICH S6(R1) recommends that male and female fertility be assessed in repeat dose toxicity studies of at least 3 months duration, adverse effects on spermatogenesis or the ovarian cycle can be detected within 2 to 4 weeks of test article administration, depending on study design and reproductive end point chosen. Therefore, in the authors’ opinion, there are options for addressing fertility aspects with studies of less than 3 months dosing duration, potentially eliminating the need for fertility end points in longer-term toxicity studies. Studies with a treatment duration of 2 to 4 weeks can help to identify specific effects on reproduction and can assist in characterizing the pathogenesis of effects.
Footnotes
Authors’ Note
The authors are employees of Covance Preclinical Services GmbH, Münster, Germany, which is a contract research organization conducting studies that address male and female fertility in NHP.
Author Contributions
Authors contributed to conception or design (LM, CML, GW); data acquisition, analysis, or interpretation (LM, CML, GW); drafting the manuscript (LM); and critically revising the manuscript (LM, CML, GW). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
