Abstract
We previously reported on a histological classification of cynomolgus monkey testis into six grades (1, immature; 2, prepuberty; 3, onset of puberty; 4, puberty; 5, early adult; 6, adult) based on spermatogenesis development. In this investigation, the accessory reproductive organs from the same animals underwent histomorphometric examination, in addition to being examined histologically and weighed, to evaluate relationships between these parameters and the six grades. Seminiferous tubule diameter increased corresponding to the testicular maturity grade and was notably increased at grade 6. Beginning from grade 3, increases in the areas of the ductus epididymis were noted, and reserved sperm was visible in the lumen. In the prostate, the glandular lumen area per unit area showed an increase beginning from grade 3 but no clear differences between grades 4 and 6; advanced development of epithelial height was observed at grade 6. In the seminal vesicle, development of the epithelial cell layer was markedly increased at grade 6. It was concluded that development of the male accessory reproductive organs began after reserved sperm was observed in the lumen of the ductus epididymis (grade 3) and that these organs were developed notably when the testis reached sexual maturity (grade 6).
Introduction
Cynomolgus monkeys are used frequently in single- and repeated-dose toxicity studies as a nonrodent species. However, those used in these studies are predominantly young cynomolgus monkeys, in which testicular toxicity cannot be appropriately assessed. According to an International Conference on Harmonization M3(R2) document (“Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals,” available at http://www.ich.org), an evaluation of male reproductive organs is performed in repeat-dose toxicity studies before inclusion of men in clinical trials. A clear correlation between the degree of testicular maturity based on histology, when classified into six grades, and testis weight was noted in the results of an investigation with cynomolgus monkeys (Haruyama et al. in press). Moreover, testicular volume has been reported as a useful indicator of sexual maturity in humans and monkeys (Haruyama et al. in press; Ku et al. 2010; Bhehre, Nashan, and Nieschlag 1989; Fuse et al. 1990; Hamm and Fobbe 1995; Korte et al. 1995; Chipkevitch et al. 1996; Diamond et al. 2000; Schiff, Li, and Goldstein 2004; Meyer et al. 2006). From histomorphometrical examinations of the testis and prostate (Arima et al. 2009; Zhengwei et al. 1997; Marshall and Plant 1996; Simorangkir et al. 2005; Simorangkir, Marshall, and Plant 2009; Chandolia et al. 2006; Neubauer et al. 2004) with image analysis measurements of the upper epithelial layer and lumen, an investigation of the relationship between the degree of development of these accessory reproductive organs and testicular maturity was considered feasible.
In this investigation, the epididymis, seminal vesicle, and prostate were examined histomorphometrically with image analysis, in addition to being examined histologically and weighed, to determine relationships of these parameters with the degree of testicular maturity.
Materials and Methods
One hundred thirty-six male cynomolgus monkeys (purpose-bred; source: China) were used in this study. These animals had been used as controls in single- or repeated-dose toxicity studies conducted at Shin Nippon Biomedical Laboratories Ltd., Kagoshima, Japan, and were euthanized between September 2008 and August 2009. Their age ranged from 38 to 83 months, and weight ranged from 3.03 to 6.89 kg. All animals had been housed individually in an environment with controlled temperature (23°C to 29°C) and humidity (35% to 75%) and provided with 108 g of solid food (HF Primate 5K91 12G 5K9 J; Purina Mills, Gray Summit, MO) daily. The above-mentioned studies had been reviewed and approved by the Institutional Animal Care and Use Committee of Shin Nippon Biomedical Laboratories Ltd.
The testes were weighed individually, fixed in Bouin’s solution, and then refixed in 10% neutral buffered formalin. The prostate, epididymis, and seminal vesicle were weighed individually and fixed in 10% neutral buffered formalin. In accordance with routine histological methods, these organs were embedded in paraffin, sectioned, and stained with hematoxylin-eosin. The testis and epididymis were examined under a light microscope, focusing on development. The testes were classified into six grades according to the histological findings (Haruyama et al. in press). In addition, the seminal vesicles and prostate were examined histologically.
Histomorphometry
The testis, epididymis, seminal vesicle, and prostate from 36 of 136 cynomolgus monkeys (6 animals at each grade) were examined histomorphometrically using image analysis (analysis; Olympus Soft Imaging Solutions GmbH, Münster, Germany) to investigate morphometry’s relationship to the degree of testicular maturity. Sections were examined under the light microscope. For the testis, the minor diameters of the seminiferous tubules (20 seminiferous tubules/monkey) were measured. For the epididymis, areas of the ductus epididymis, lumen of the ductus epididymis, epithelial cell layer in the ductus epididymis, epithelial height, and the area of the reserved sperm in the lumen (20 epididymal ducts/monkey) were measured. For the prostate, the glandular lumen area and glandular area per unit area (1 mm2) and epithelial height of the glands were measured. For the seminal vesicle, the areas of the gland, epithelial cell layer, and glandular lumen in each slide specimen were measured, and the ratios of the areas of the epithelial cell layer and glandular lumen to the area of the gland were determined.
Statistical Analysis
Each organ’s histomorphometric parameters and weights were analyzed by pairwise comparison between grades using the Tukey-Kramer HSD test. A correlation chart was constructed to examine mutual correlation between organ weights, and the correlation coefficient was determined.
Results
The organ weights at each grade are shown in Table 1 and Figure 1. The mean testicular weights (right) at grades 1, 2, 3, 4, 5, and 6 were 1.26, 2.80, 5.61, 9.68, 13.84, and 24.79 g, respectively, and no significant increase was noted between grades 1 and 2, but significant increases between grades were noted beginning from grade 2 and above. The mean epididymal weights (right) at grades 1, 2, 3, 4, 5, and 6 were 0.61, 0.71, 1.07, 1.61, 1.97, and 3.18 g, respectively, and no significant increase in epididymal weight was noted between grades 1 and 2 or 2 and 3, but significant increases between grades were noted beginning from grade 3 and above. The mean prostate weights at grades 1, 2, 3, 4, 5, and 6 were 0.49, 0.76, 0.93, 1.19, 1.53, and 2.14 g, respectively, and no significant increase was noted between grades 1 and 2, 2 and 3, or 3 and 4, but significant increases between grades were noted beginning from grade 4 and above. The mean seminal vesicle weights at grades 1, 2, 3, 4, 5, and 6 were 1.42, 2.06, 3.52, 5.43, 8.37, and 17.90 g, respectively, and no significant increase was noted between grades 1 and 3, 2 and 4, or 4 and 5, but a significant increase between grades 5 and 6 was noted. No significant increase in the weight of any organ was noted between grades 1 and 2; however, significant increases were noted beginning from grade 3 and above. A high correlation with each organ (
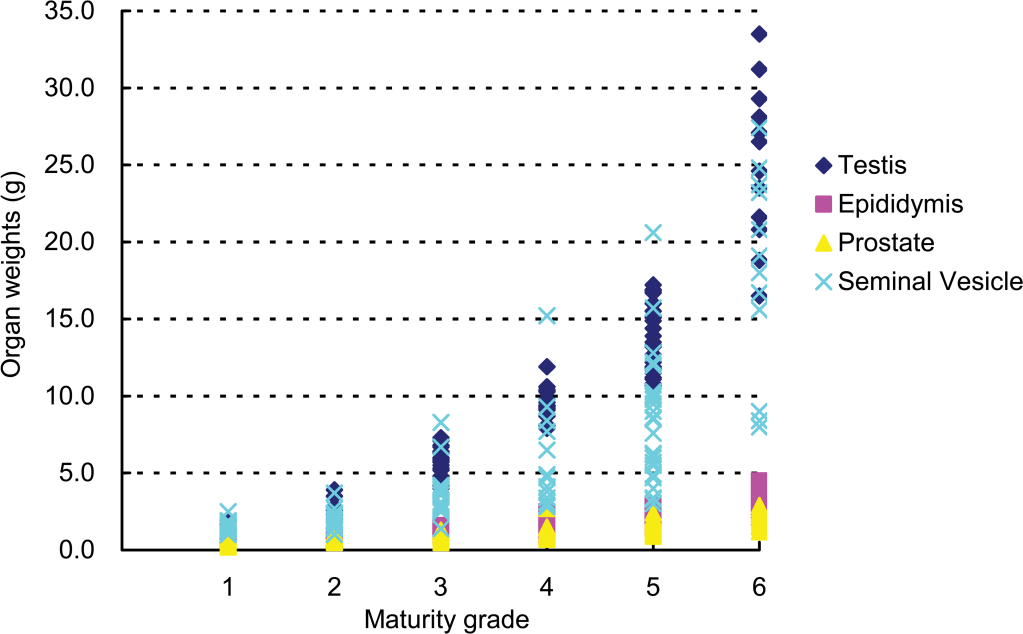
Relationship between testicular maturity grade and organ weights.
Organ weights (g, mean ±
A to E: Different symbols indicate a significant difference (
Results of testicular histomorphometory showed that the diameters of the seminiferous tubules generally increased corresponding to testicular maturity grade. No significant increase was noted between grades 1 and 2, but a significant difference was noted between Grades 2 and 3. Results were constant between grades 3 and 5, but a significant increase was noted at grade 6 (Figure 2; Table 2). In the epididymis, no significant difference in the area of the ductus epididymis was noted between grades 1 and 2, but a significant increase was noted beginning from grade 3 and above. No significant difference in the area of the epithelial cell layer of the ductus epididymis was noted between grades 1 and 3, but a significant increase was noted beginning from grade 4 and above. No significant difference in epithelial cell height was noted between grades 1 and 6. Almost no reserved sperm in the lumen of the ductus epididymis was observed at grade 1 or 2, and although sperm was noted beginning from grade 3, no significant increase was noted at up to grade 4. A significant increase in sperm was noted beginning from grade 5 (Figure 3; Table 3). In the cynomolgus monkey prostate, glandular and glandular lumen areas per unit area (1 mm2) and epithelial height were measured. Although a very slight tendency toward an increase in glandular area corresponding to testicular maturity grade was observed, this increase was not significant at any grade. No significant difference in glandular lumen area was noted between grades 1 and 2. A significant increase was noted beginning from grade 3; however, there were large individual differences. No significant increase was noted between grades 4 and 6. Epithelial height could not be determined at grade 1 or 2 because the lumen was not fully formed; therefore, these grades were excluded from this analysis. At grades 3, 4, and 5, mean epithelial heights were 13.5, 14.8, and 13.4 µm, respectively, and showed no difference between grades, but a mean height of 20.2 µm at grade 6 indicated a significant increase (Figure 4; Table 4). Histological evaluation of prostate showed that lumen formation began at grade 2, but there was no marked difference between grades 1 and 2. At grade 3, the lumen was fully formed, with the mean area showing a broad range of 46,672 to 376,939 µm2 (data not shown), and large individual differences were observed. No difference was noted between grades 4 and 6. The mean lumen areas at these grades were 144,038 to 287,995 µm2 (grade 4), 262,601 to 368,823 µm2 (grade 5), and 170,573 to 282,075 µm2 (grade 6; data not shown), and it is considered that prostate maturity was reached by grade 4 based on the lumen area. However, no increase in epithelial height was observed at up to grade 5, but increased development of epithelial height was observed at grade 6 (Figure 5). In the seminal vesicle, no significant difference between grades was noted in the glandular lumen area to glandular area ratio. The epithelial cell layer area to glandular area ratio increased corresponding to grade. No significant increase in this ratio was noted between grades 1 and 3, but a significant increase in the ratio was noted beginning from grade 4 (Figure 6; Table 5). Histology showed a similarly narrow lumen at grades 1 and 2 with flattened epithelial cell and simple folding of the mucosal epithelium, which at grade 3 began to become more complex. Beginning from grade 4, an apparent increase in accumulation of secretions in the lumen was observed, and further development of the epithelial cell layer was noted at grade 6 (Figure 7).
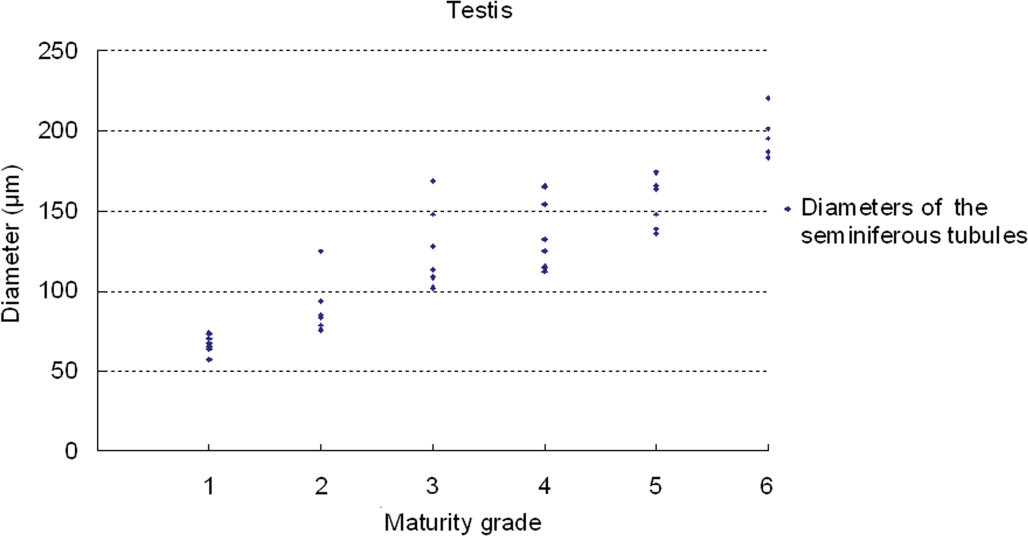
Relationship between testicular maturity grade and diameters of the seminiferous tubules.
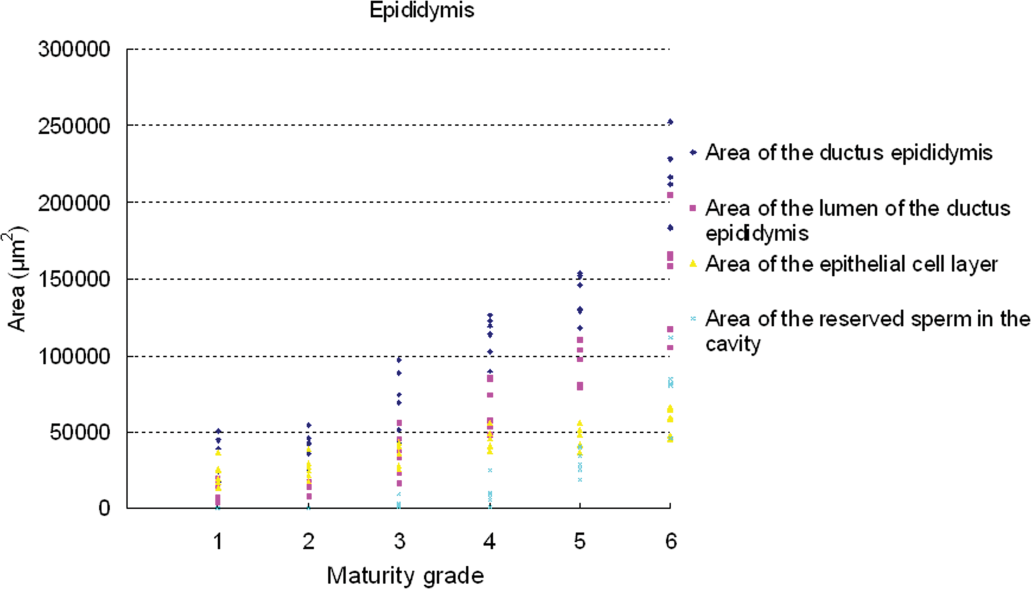
Relationship between testicular maturity grade and epididymis histomorphometric values.
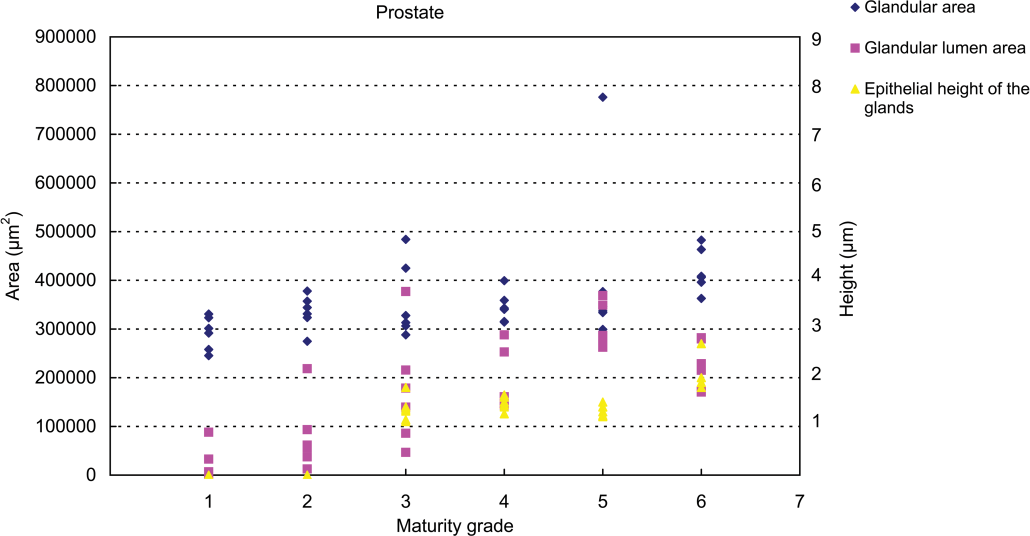
Relationship between testicular maturity grade and prostate histomorphometric values.
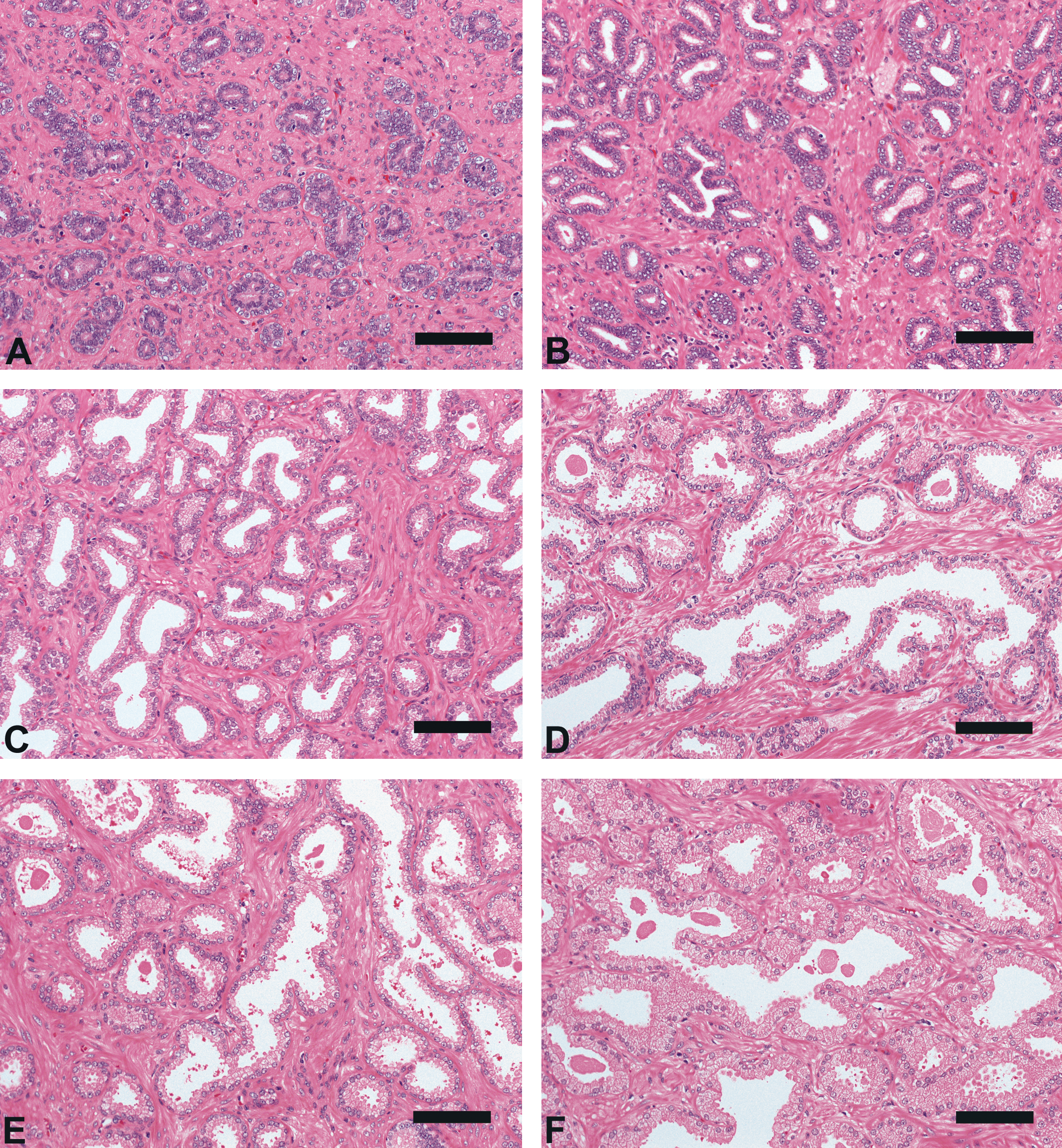
Prostate morphologic characteristics (hematoxylin-eosin, bar = 100 µm). (A), Grade 1 (Immature); (B), Grade 2 (Pre-Puberty); (C), Grade 3 (Onset of Puberty); (D), Grade 4 (Puberty); (E), Grade 5 (Early Adult); (F), Grade 6 (Adult): The grades were determined according to the examination of the testis and epididymis (Haruyama et al. in press).
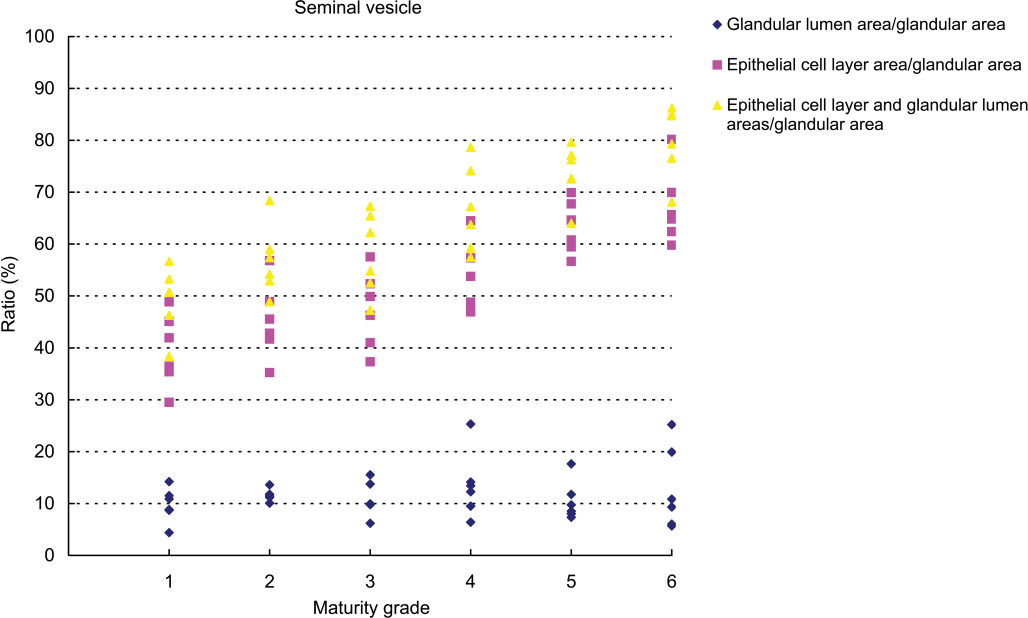
Relationship between testicular maturity grade and seminal vesicle histomorphometric values.
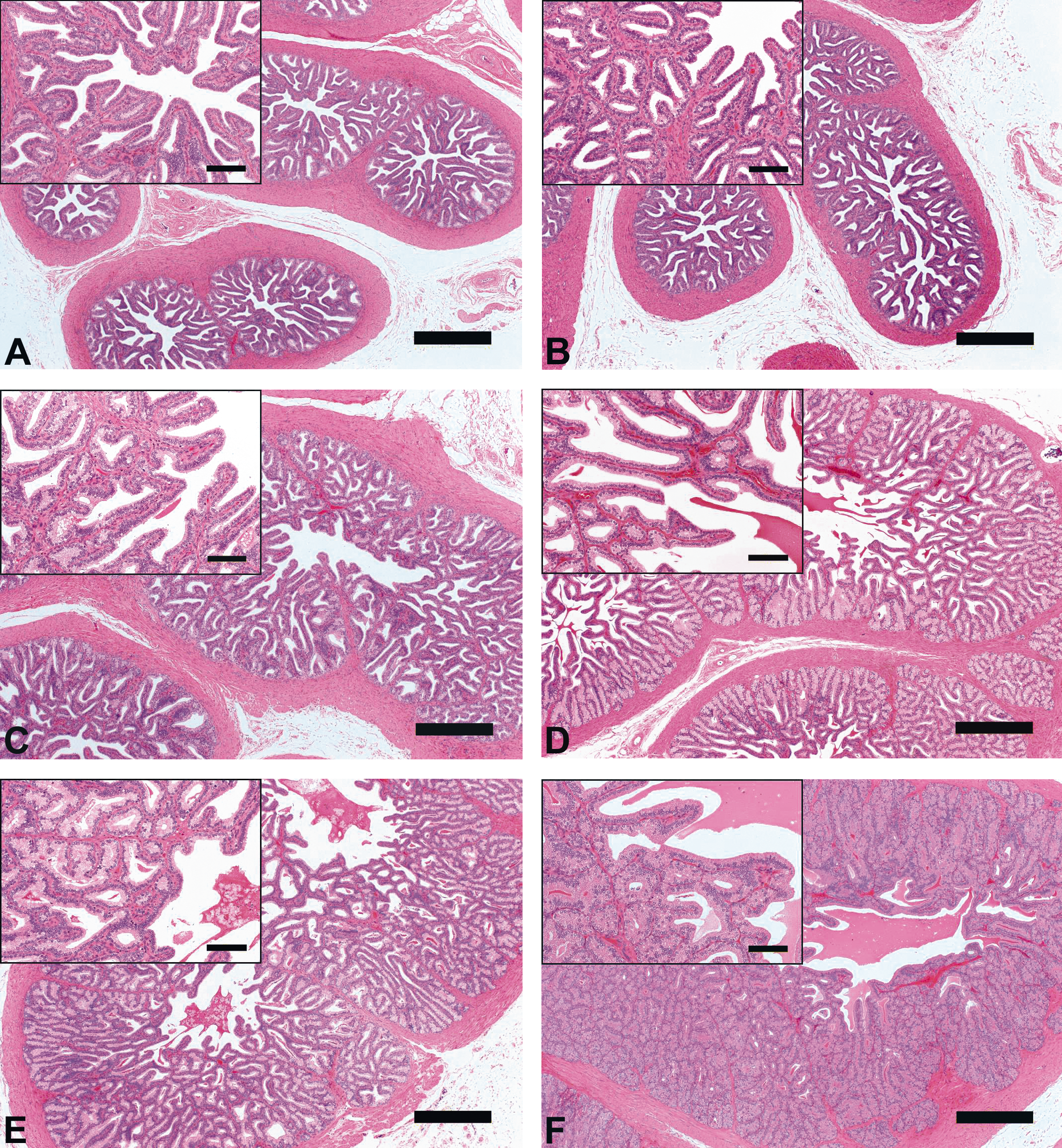
Seminal vesicle morphologic characteristics [hematoxylin-eosin, Bar = 100 µm (magnification) or 500 µm]. (A), Grade 1 (Immature); (B), Grade 2 (Pre-Puberty); (C), Grade 3 (Onset of Puberty); (D), Grade 4 (Puberty); (E), Grade 5 (Early Adult); (F), Grade 6 (Adult): The grades were determined according to the examination of the testis and epididymis (Haruyama et al. in press).
Histomorphometry data in the testis (mean ±
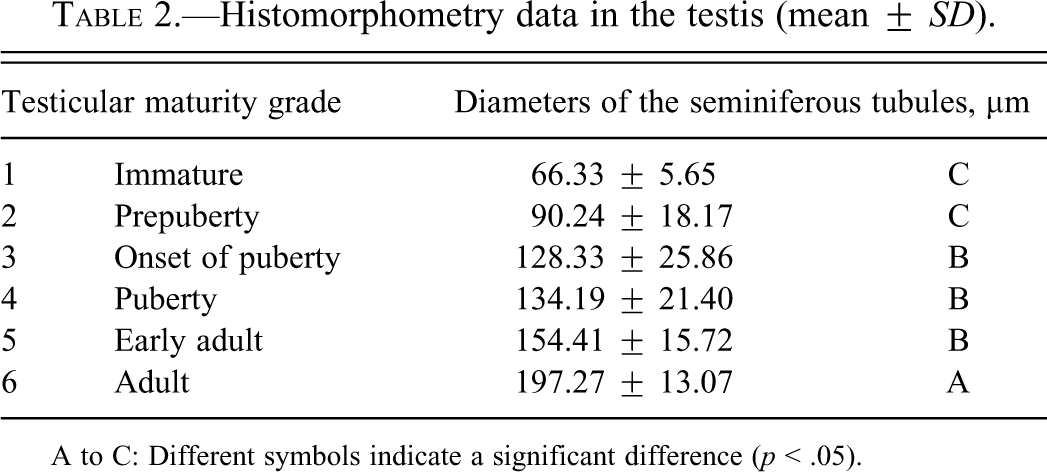
A to C: Different symbols indicate a significant difference (
Histomorphometry data in the epididymis (mean ±
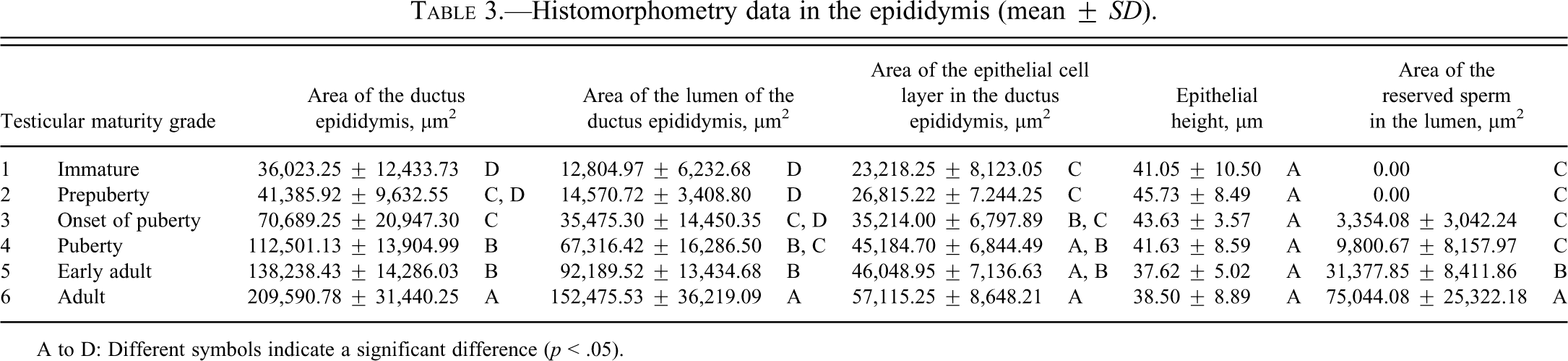
A to D: Different symbols indicate a significant difference (
Histomorphometry data in the prostate (mean ±
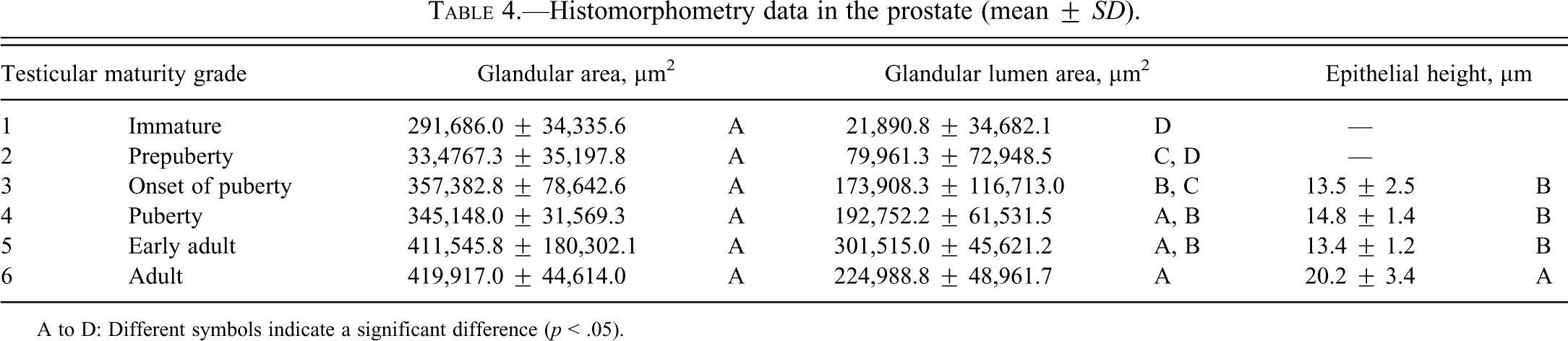
A to D: Different symbols indicate a significant difference (
Histomorphometry data in the seminal vesicle (mean ±
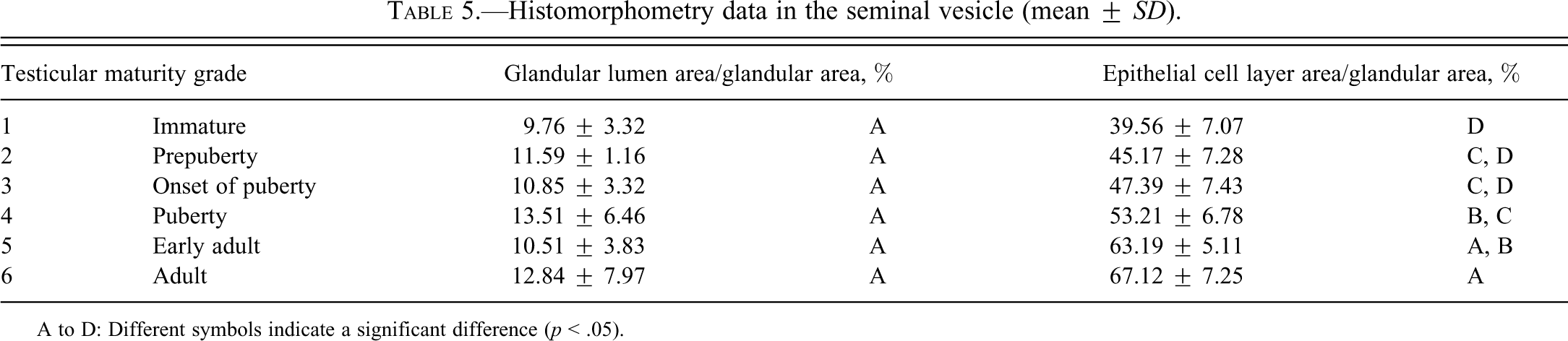
A to D: Different symbols indicate a significant difference (
Discussion
Nonhuman primates are frequently used for general or reproductive toxicity studies for biopharmaceuticals because they commonly exhibit pharmacological activities that resemble those in humans (Chellman et al. 2009). Macaque monkeys such as cynomolgus and rhesus monkeys are used in such studies; however, the selection of sexually mature males is important when evaluating testicular toxicity. Rhesus monkeys have a seasonal breeding cycle, with reduced spermatogenesis outside of this season. Therefore, it is difficult to evaluate testicular toxicity in rhesus monkeys. On the other hand, with cynomolgus monkeys, mature males are capable of sperm production throughout the year, and this species provides a suitable model for evaluation of testicular toxicity. In a previous investigation using the testis of control group cynomolgus monkeys (136 males) from repeated-dose toxicity studies performed between September 2008 and August 2009 at this facility, a clear correlation between degree of testicular maturity and testis weight was observed (Haruyama et al. in press). In the current investigation, a further evaluation was performed investigating the relationship between testis and accessory reproductive organ weights and testicular maturity grade. In addition, the testis, epididymis, seminal vesicle, and prostate from 36 of 136 cynomolgus monkeys (6 animals/grade) were examined morphometrically using image analysis to investigate relationships of histomorphometric parameters to the degree of testicular maturity.
At grades 1 and 2 (immature and prepubertal), no significant increase was noted in any organ weight or histomorphometric parameters. At these grades, no sign of spermatogenesis had occurred in the testis, and no sperm was observed in the epididymis (Haruyama et al. in press). In the prostate, the glandular lumen was not visible or barely visible. In the seminal vesicle, narrow lumen, flattened epithelial cells, and simple folding of the mucosal epithelium were observed. At grade 3 (onset of puberty), testis weight was significantly increased compared with grades 1 and 2, and prostate weight was significantly increased compared with grade 1 (no significant difference from grade 2); however, no significant increase in seminal vesicle weight was noted between grades 1 and 3. Significant increases in the diameter of the seminiferous tubule and areas of the ductus epididymis and glandular lumen in the prostate were noted between grades 2 and 3. Histological examination showed that the glandular lumen in the prostate was fully formed at grade 3 with a markedly broad area, and large individual differences in area were observed to a similar degree at grades 1, 2, and 4 to 6. Although folding of the mucosal epithelium in the seminal vesicle had begun to become complex, hardly any likely accumulation of secretions in the lumen was observed at grade 1 or 2. Also at grade 3, all classes of germ cells, including step 14 spermatids, were present in the seminiferous tubules; however, desquamation of germ cells was observed, and the morphological stage of spermatogenesis was unclear. Moreover, moderate debris in the epididymis was also observed to a greater extent than at other grades. These findings in the testis and epididymis suggested the onset of spermatogenesis and immature Sertoli cell function (Haruyama et al. in press). Organ weight and histomorphometric parameters in accessory reproductive organs showed significant increases beginning from grade 3. Based on histological assessment, spermatogenic wave in cynomolgus monkeys was considered to begin at grade 3 (Haruyama et al. in press), and maturation of accessory reproductive organs is thought to have begun at grade 3 based on data obtained in this study.
Between grades 4 and 6 (puberty, early adult, and adult), significant increases were noted in testicular, epididymal, and prostate weights and in seminal vesicle weight between grades 5 and 6. Significant increases in the areas of the ductus epididymis, epithelial cell layer of the ductus epididymis, and epithelial cell layer ratio in the seminal vesicle were noted between grades and in the area of the reserved sperm in the lumen of the ductus epididymis from Grade 5. Beginning from grade 4, all classes of germ cell in the seminiferous tubules and sperm in the epididymis were observed. Determination of spermatogenic stages in the seminiferous tubules and histopathological evaluation of consideration of stage-specific changes were possible. These males were considered to be suitable for evaluation of testicular toxicity. No significant increase in the area of the glandular lumen in the prostate was noted between grades 4 and 6, and histological images of the prostate at grade 4 were similar to those previously reported at grade 5 (adult; Ku et al. 2010), and it was considered that the prostate reached maturity at grade 4. However, no change in epithelial height was noted up to grade 5, and advanced epithelial height was noted only at grade 6. Effects of toxicant on the epithelial cell layer had previously been reported (Arima et al. 2009), from which it was concluded that in studies to evaluate toxicity to prostatic epithelium, grade 6 animals should be used because epithelial height reaches its peak at this grade. No significant difference in the diameter of the seminiferous tubules was noted between grades 3 and 5, but a marked increase was noted at grade 6. Also at grade 6, males showed a large amount of sperm in the epididymis, and sperm analysis using ejaculated semen collected by electroejaculation stimulation (Meyer et al. 2006) would have been possible. The evaluation of both testicular sperm production capability and epididymal sperm maturity would have been possible with repeated sperm analysis from the pretreatment period to the end of the treatment period for these males. In the seminal vesicle, histologic images showed that simple folding of the mucosal epithelium began to become more complex from grade 3, the grade at which spermatogenesis begins, and accumulation of secretions in the lumen increased at grade 4, the grade at which increased reserved sperm in the lumen of the ductus epididymis was also observed. At grade 6, at which point sperm analysis using ejaculated semen collected by electroejaculation stimulation was possible, development of the epithelial cell layer including glandular secretions was noted in the seminal vesicle. These secretions produce most of the seminal fluid that affects sperm capacitation and motility, and toxicity on the fluid or fluid production may have an effect on male fertility (Pin et al. 2008; Coroner et al. 1992; de Lamirande et al. 2001; Huang, Chu, and Chen 2000; Kawano and Yoshida 2007). Therefore, considering the notable development of the epithelial layer in the prostate and seminal vesicle from grade 5 and through grade 6, as well as that of the accessory reproductive organs, thorough evaluation of male reproductive toxicity was considered feasible at grade 6. In mice, most of the branching in the seminal vesicle is complete at approximately 2 weeks after birth, and at this stage, epithelial cell differentiation begins and cells assume a highly secretory function. Through hypertrophy, the gland continues to grow into adulthood (Pin et al. 2008). Brewster (1985) reported that in humans, at 18 years of age, seminal vesicles showed extensive epithelial folding around the circumference of the lumen, and at 45 years of age, there was a great increase in the extent of the epithelial folding in the seminal vesicle when compared with that in the specimen from the 18-year-old. Seminal vesicle weights in cynomolgus monkeys at grade 6 ranged from 8 to 27.4 g, and individual differences were large. These results suggested that the seminal vesicle continues to develop after testicular maturity is reached. Overall results of our analyses showed increases in organ weights corresponding to increasing testicular maturation grade; these increases tended to start in the order of testis, epididymis, prostate, and seminal vesicle, and histomorphometric parameters showed a similar tendency. These trends were considered to be associated with individual organ function. Moreover, a high correlation (
In conclusion, development of the male accessory reproductive organs began after reserved sperm was observed in the lumen of the ductus epididymis (grade 3), and these organs were developed notably when the testis reached sexual maturity (grade 6).
Footnotes
Acknowledgment
The authors gratefully acknowledge Dr. Kok Wah Hew for his critical review of the article.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
