Abstract
Previous work indicates that silver nanoparticles (AgNPs) given IP to mice alter the regulation of inflammation- and oxidative stress–related genes in brain. Here we assessed the distribution and toxic potential of AgNP following intranasal (IN) exposure. Adult male C57BL/6J mice received 25-nm AgNP (100 or 500 mg/kg) once IN. After 1 or 7 days, histopathology of selected organs was performed, and tissue reduced glutathione (GSH) levels were measured as an indicator of oxidative stress. Aggregated AgNP were found in spleen, lung, kidney, and nasal airway by routine light microscopy. Splenic AgNP accumulation was greatest in red pulp and occurred with modestly reduced cellularity and elevated hemosiderin deposition. Aggregated AgNP were not associated with microscopic changes in other tissues except for nasal mucosal erosions. Autometallography revealed AgNP in olfactory bulb and the lateral brain ventricles. Neither inflammatory cell infiltrates nor activated microglia were detected in brains of AgNP-treated mice. Elevated tissue GSH levels was observed in nasal epithelia (both doses at 1 day, 500 mg/kg at 7 days) and blood (500 mg/kg at 7 days). Therefore, IN administration of AgNP permits systemic distribution, produces reversible oxidative stress in the nose and in blood, and mildly enhances macrophage-mediated erythrocyte destruction in the spleen.
Introduction
Nanoparticles (NPs) are particles with at least one dimension between 1 and 100 nm. By 2014, nanomaterials (engineered NPs) are predicted to be incorporated into 15% of global manufacturing products, representing an economic impact of $2.6 trillion (National Institute for Occupational Safety and Health [NIOSH] 2009). The small size and high surface area/mass ratio of NPs give them different properties than those found in bulk materials of the same mass (Quadros and Marr 2010). NPs can cross cell membranes and interact with intracellular structures (Oberdörster et al. 2007). Engineered silver NPs (AgNPs) have antibacterial activity and are used as coatings for hospital beds, medical implants, and textiles. Nanosilver is also used in wound and burn dressings, as a chemical for water purification, in intragenital contraceptives, and as a disinfectant room spray (Chen and Schluesener 2008). Although nanosilver has many uses in consumer products, the health effects of this material have not been well characterized, even though the toxicity of nanosilver has been demonstrated in in vitro models (Navarro et al. 2008; Lanone et al. 2009; Powers et al. 2010).
Human exposure to nanomaterials during manufacturing, which is currently the most common venue for NP contact, is most likely to occur via the respiratory tract. For example, airborne AgNPs are produced during electrorefining of silver (Miller et al. 2010). The potential concern for this situation is that the respiratory tract has many structural traits (e.g., tortuous air passages, moist mucus covering, minimal intervening tissue between air and blood) which are likely to permit extensive systemic uptake and distribution of NPs in exposed workers.
Much of the work understanding the effects of NPs on human health comes from the study of ultrafine particles (UFPs) found in air pollution. The hypothesized mechanism for the cellular changes following UFP exposure is an increase in oxidative stress due to direct action of NPs on living tissue (Peters et al. 2006; Calderon-Garciduenas et al. 2008b). Previous work has shown that mice given an IP injection of 25-nm diameter AgNPs had altered expression of oxidative stress– and inflammation-related genes in the brain (Rahman et al. 2009), strongly suggesting that AgNP can penetrate the blood–brain barrier (BBB) after systemic delivery. The size of AgNP and other NPs would predict that they would be deposited throughout the respiratory tract if inhaled. Furthermore, translocation of NPs from the respiratory system to extrapulmonary sites (brain, liver, spleen, kidneys, etc.) has been documented in rats, dogs, nonhuman primates, and humans (Kreyling et al. 2002; Oberdörster et al. 2002, 2007; Elder et al. 2006; Calderon-Garciduenas et al. 2008a). NPs can reach the brain either indirectly by nasal absorption followed by hematological distribution or directly by olfactory epithelial uptake and subsequent transfer along the olfactory nerves (Oberdörster et al. 2004).
The dual purposes of this study were to determine the distribution and potential impact of intranasally (IN) administered AgNPs on organ histology (i.e., to identify target tissues for cytotoxicity and inflammation) and to measure the tissue concentrations of reduced glutathione (GSH) as an indicator of oxidative stress. The study was not designed to mimic a relevant level of human occupational exposure but rather to define the impact of a previously used AgNP test article (Rahman et al. 2009) when given by the most likely exposure route (nasal cavity) to be encountered in the occupational setting; the comparison between the current experiment and the older study was strengthened by using identical doses of nanosilver and animals of the same species strain, sex, and age. Our hypothesis was that IN-administered AgNPs would be distributed widely to many organs, particularly to the brain, thereby inducing inflammatory cell infiltration and aggregation as well as oxidative stress. Our present data have revealed several sites of AgNP accumulation following IN exposure and begun exploring the impact of such deposits on target organ function.
Methods
Animal Welfare Assurance
This study was conducted in accordance with federal animal care guidelines, in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility, and was approved in advance by the University of Cincinnati Institutional Animal Care and Use Committee.
AgNPs
AgNPs (25 nm diameter) were identical to those used in a previous study (Rahman et al. 2009) and were obtained from NovaCentrix (Austin, TX). The particles were dispersed by vortexing and sonicating (Kontes micro-ultrasonic cell disrupter, Kimble Chase, Vineland, NJ; ∼10 sec) in ultrapure water. Previous analysis of these particles by transmission electron microscopy revealed a size distribution of 29 ± 12.5 nm (when dispersed in deionized water). Dynamic light scattering showed agglomeration of the 25 nm NPs into aggregates of 118 nm in diameter, or four times their primary particle size (Rahman et al. 2009).
Source and Treatment of Mice
Male C57BL/6J mice (Jackson Laboratory, Bar Harbor, ME; 21–24 g body weight) were divided into groups of 6 mice each and acclimated for at least 1 week prior to use. Animals were housed two to four per cage, maintained under constant environmental conditions (22 ± 1°C, relative humidity 50%, 12-hr:12-hr light–dark cycle), and given tap water and pelleted chow (LM-485; Harlan/Teklad, Indianapolis, IN) ad libitum. Acclimated mice were treated with AgNPs by IN instillation (single treatment, 100 mg/kg or 500 mg/kg, delivered in a volume of 10 µl in sterile water). Mice were lightly anesthetized with isoflurane, and NPs were delivered into one nostril using a sterile pipette tip (inserted to a depth of approximately 3 mm). Particles were administered within 10 min of sonication. Mice were watched for several hours after treatment, and then daily after exposure to monitor for outward indicators of toxicity. Control mice received a single IN instillation of 10 µl of sterile water. Mice were necropsied 1 or 7 days after treatment.
Tissue Preparation for Biochemical Analyses
At necropsy, whole blood was collected in a heparinized tube by intracardiac puncture. Small pieces (20–100 mg) of brain (divided at Bregma into forebrain and hindbrain [including both gray and white matter]), kidney (half of one kidney/mouse, divided along the longitudinal axis), liver, and nasal epithelia (naso turbinates and ethmoid turbinates plus nasal septum) as well as whole blood (
Hemoglobin and Methemoglobin
Concentrations were measured in heparinized blood as previously described (Winterbourn 1985).
GSH Assay
GSH concentration was determined using a previously described fluorometric assay (Senft, Dalton, and Shertzer 2000). GSH concentrations were expressed per mg of tissue, and in blood per micromole of hemoglobin.
Pathology
The following tissues were evaluated and collected at necropsy and then fixed in neutral buffered 10% formalin for histological evaluation (
Immunohistochemistry (IHC)
In order to visualize macrophages in the spleen, a rat anti-mouse Mac-3 antibody (BD-Pharmingen, Franklin Lakes, NJ) was used. Briefly, primary antibody was applied at a 1:100 dilution for 2 hr at room temperature (RT), followed by a horseradish peroxidase–conjugated anti-rat immunoglobulin (Ig)G secondary antibody (Santa Cruz Biotechnology, Santa Cruz, CA) at 1:100 for 30 min at RT. Antibody binding was visualized using diaminobenzidine (Novacastra, Vector Laboratories, Burlingame, CA) as the substrate. Slides were counterstained with hematoxylin and coverslipped using permanent mounting medium. In order to visualize the activated microglia in the brain, a sensitive marker of neuroinflammation (Thompson, Karpus, and Van Eldik 2008), immunohistochemistry was similarly performed with a rat anti-mouse CD45 antibody (Santa Cruz Biotechnology). This primary antibody was applied at a 1:50 dilution for 2 hr at RT. The subsequent IHC steps and reagents were identical to those used for the anti-Mac-3 antibody.
Hemosiderin Localization
Serial sections of spleen were also stained with Perls Prussian Blue to visualize hemosiderin, a marker for erythrocyte degradation (http://library.med.utah.edu/WebPath/HISTHTML/MANUALS/IRON.PDF). Briefly, sections were microwaved in a mixture of 5% potassium ferrocyanide and 5% hydrochloric acid (1 min), rinsed in distilled water, counterstained with nuclear fast red (1 g nuclear fast red, 1 mL glacial acetic acid, 99 ml deionized water), rinsed, dehydrated, and coverslipped with permanent mounting medium.
Autometallography (AMG)
This technique is a tool for metal enhancement and as such provides a potent histochemical means for detecting tissue deposition of nanocrystals of various metals including silver, mercury, bismuth, or zinc. We used a previously described cellulose silver lactate protocol (Danscher and Stoltenberg 2006) to determine whether nonaggregated silver could be detected in various brain regions of mice treated with AgNP by IN instillation. Briefly, brain sections were deparaffinized and rehydrated (into water) using standard methods. In a clean glass Coplin jar, the following reagents were mixed and heated to 30°C in a water bath positioned on an orbital shaker: 30 mL of 2% carboxymethylcellulose (Sigma-Aldrich, St. Louis, MO); 5 mL of citrate buffer solution (1.2 M citric acid + 0.8 M sodium citrate, pH 3.7); 0.43 g of hydroquinone dissolved (by heating in near boiling water) in 7.5 mL of distilled water; and 60 mg of silver lactate (Acros Chemicals, Brunswick, NJ) dissolved by vigorous vortexing in 7.5 mL of distilled water. Slides were incubated in this mixture in a 30°C water bath (covered with foil to shield the silver mixture from light) for 90 min. After washing slides in distilled water, sections were incubated in 5% sodium thiosulfate in water at RT for 5 min. Sections were washed in distilled water, counterstained with nuclear fast red, rinsed, dehydrated, and coverslipped with permanent mounting medium.
Statistical Analyses
Hemoglobin, methemoglobin, and GSH levels were presented as means ± SEM. Data were analyzed using the unpaired
Results
Clinical Presentation
No differences in body weight, physical appearance, or general behavior were observed between control mice and those in any of the AgNP treatment groups.
Hemoglobin and Methemoglobin Concentrations
Methemoglobin levels ranged from 0% to 2.58%, with no dose- or treatment-related trend (data not shown). There was no difference in hemoglobin concentration in the blood among any of the treatment groups (Table 1).
Hemoglobin (Hgb) content in core blood (μM) following intranasal nanosilver treatment.
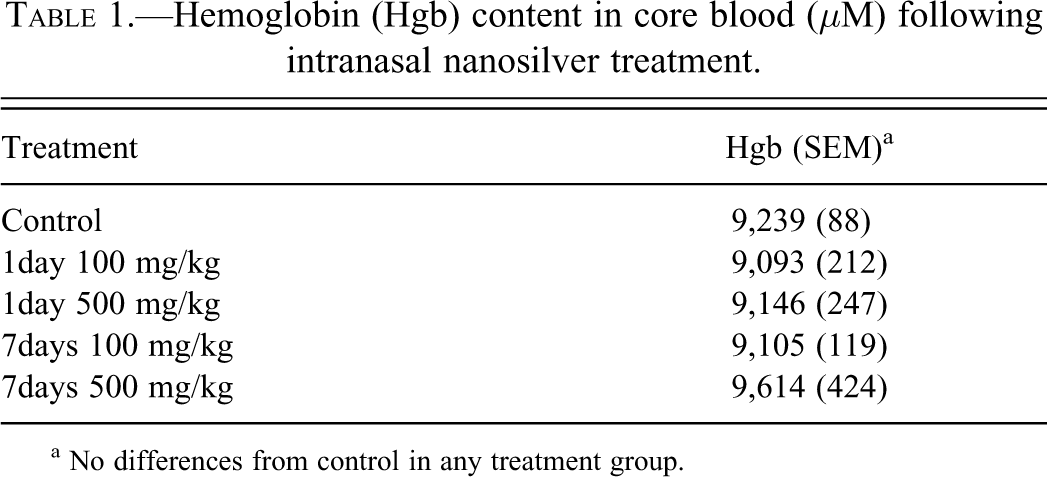
a No differences from control in any treatment group.
GSH Concentrations
GSH concentrations in forebrain, hindbrain, kidney, liver, and lung were not different from vehicle control mice in any of the treatment groups (data not shown). An increase in GSH was found in blood (only in the high-dose mice 7 days after treatment; Table 2). The only tissue in which GSH levels were affected by IN administration of AgNP was the nasal epithelium itself, which was subjected to direct contact with the nanosilver (Table 2).
Reduced glutathione (GSH) concentrations in core blood and nasal mucosa following intranasal nanosilver treatment.
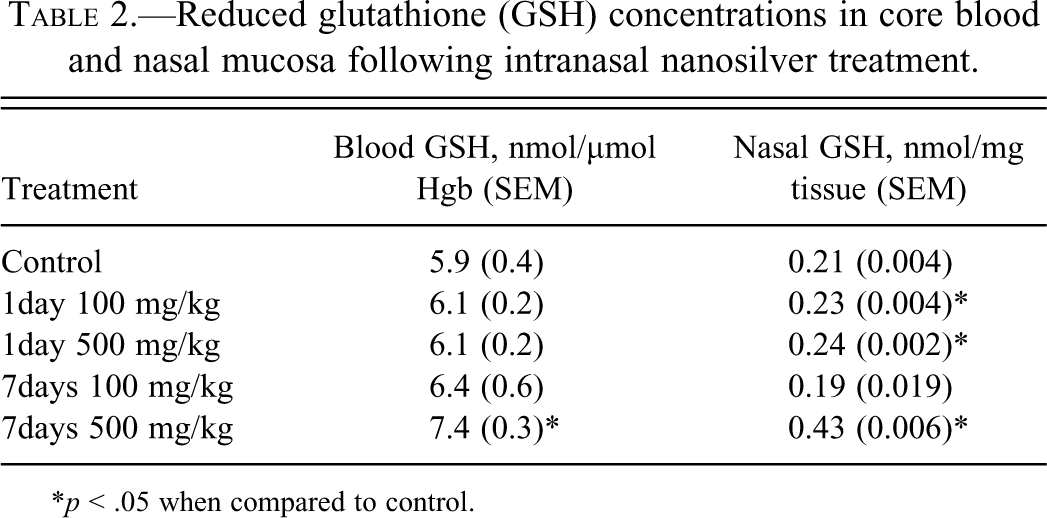
*
Pathology
Gross observations at necropsy indicated that all organs of AgNP-exposed mice exhibited the anatomic features (e.g., characteristics of color, consistency, shape, and size) to be expected based on their appearance in control animals. H&E-stained brain sections from all animals (an average of twenty sections through the brain from rostral [anterior] to caudal [posterior] were examined) were scored as within normal limits. Specifically, cytoarchitectural alterations in neural cells (neurons, glia, choroid plexus epithelium and stroma, and ependyma) were not observed. No inflammatory cell infiltrates or CD45 immunoreactivity (i.e., activated microglia) were observed at either AgNP dose or time point. While aggregated AgNPs were not visible in H&E-stained brain sections of any of the Ag-treated mice by routine light microscopy, AMG demonstrated the widespread presence of AgNP in multiple brain regions, including most prominently the olfactory bulb (perivascular localization) and the choroid plexus and ependyma of the lateral ventricles (Figure 1).
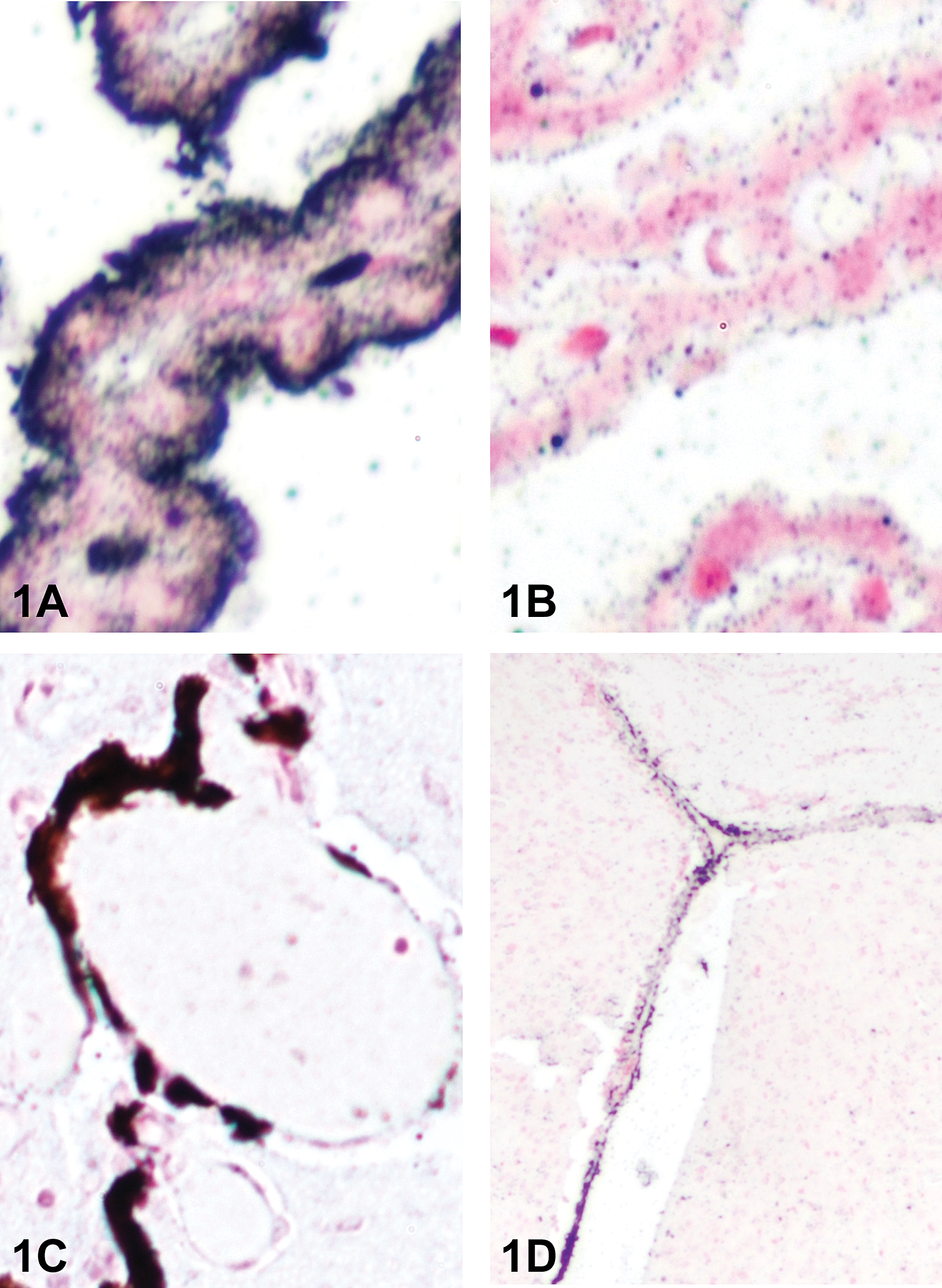
Detection of silver in mouse brain by autometallography (AMG) following intranasal (IN) administration of 25 nm silver nanoparticles (AgNPs). (A), Choroid plexus from the lateral ventricle of a mouse treated IN with 500 mg/kg of AgNPs and necropsied 1 day later. The granular black reaction product indicates extensive localization of silver deposition in the tissue, particularly along the apical surface. 400× magnification. (B), No AgNPs are apparent in the choroid plexus of a vehicle-treated control mouse run concurrently through the AMG protocol. (C), Silver was associated with blood vessels of the olfactory bulb (at approximately 2.58 Bregma) in a mouse given 500 mg/kg of AgNPs and necropsied 7 days later. 200× magnification. (D), Silver particles were distributed to the ependymal lining of the lateral ventricle in a mouse treated with 100 mg/kg of AgNPs and necropsied 7 days later. 40× magnification. All sections were counterstained with neutral red.
Aggregated AgNP accumulation was observed in the spleen (Figure 2B), nasal cavity, renal medulla, and occasionally in lung. Nanomaterial accumulation in the spleen was primarily observed in the red pulp and was associated with a modest loss of cellularity in this region; the reduced cellularity was most evident at the 1-day time point with both doses of AgNP but had not been completely reversed by 7 days. Red pulp depletion appeared to represent a mild decrease in the normal complement of extramedullary hematopoietic elements rather than damage to the splenic stroma. Structural changes were not seen in the white pulp or in splenic blood vessels (arteries or venous sinuses), nor in intravascular erythocytes in any tissue. AgNP accumulation in the spleen colocalized with macrophages, as indicated by Mac-3 immunohistochemistry (Figure 2C, D). Hemosiderin was also prominent in the red pulp of the spleen of Ag-treated mice, in contrast to control mice (Figure 2E, F), likely a consequence of red blood cell destruction. Nanomaterial accumulation in the nose sometimes was associated with epithelial erosion, the presence of neutrophils in the airway, and proteinaceous exudates (Figure 3A, C). This change was localized, of mild degree, and affected the rostral mucosa (near the junction of the respiratory and olfactory epithelia). A single mouse (500 mg/kg, 1 day after treatment) exhibited perivasular expansion and aggregated NPs in the lung (Figure 4). In contrast to the spleen, none of the other tissues had structural changes associated with the presence of AgNP.
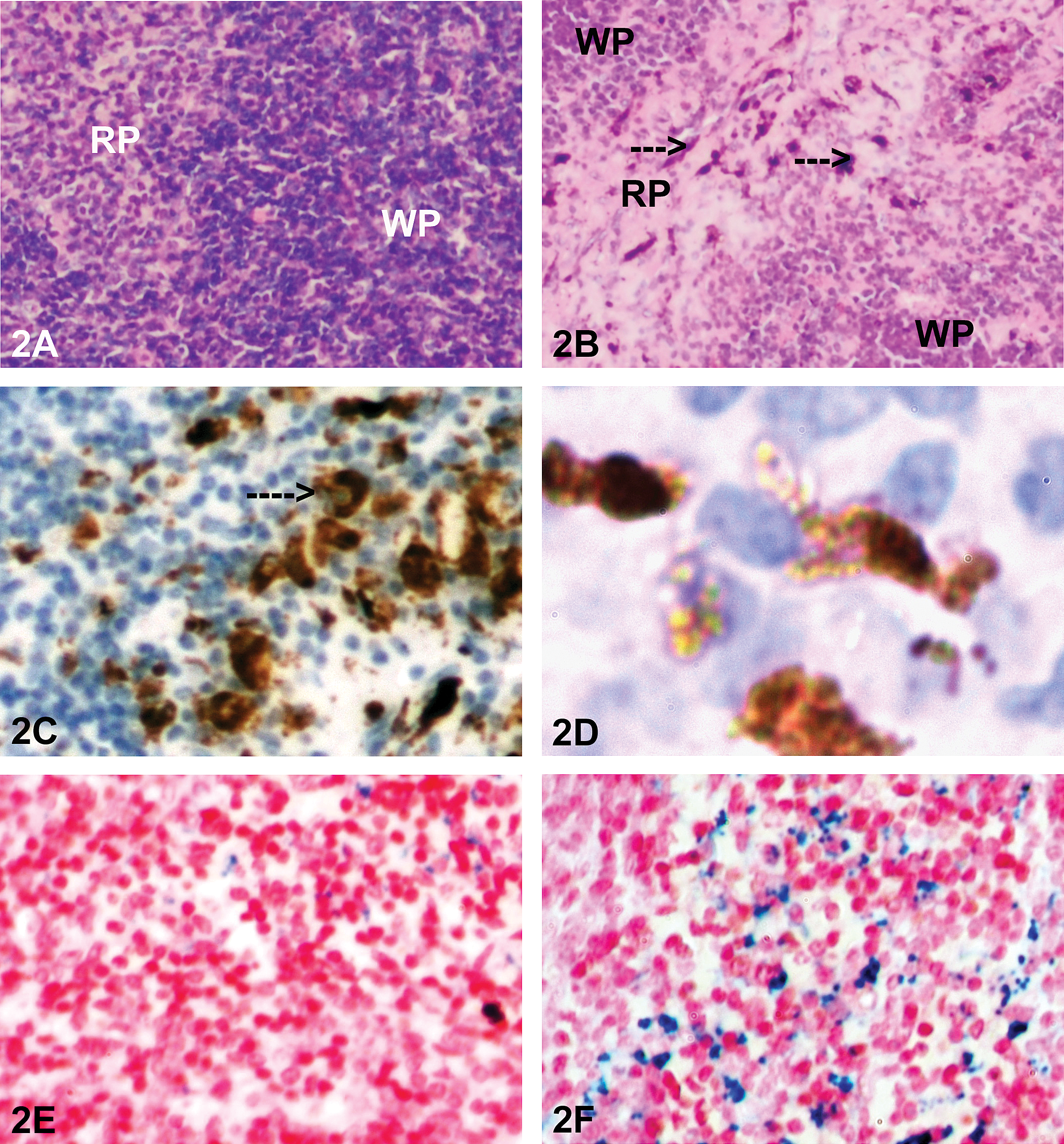
Key histological and immunohistochemical findings in rats administered 25 nm silver nanoparticles (AgNPs) by intranasal (IN) instillation. (A), Spleen from a control mouse, showing red pulp (RP) and white pulp (WP) with normal architecture and cell density. 100×; H&E. (B), Spleen from a mouse treated with AgNPs (500 mg/kg) and necropsied 24 hr later. Note the prominent reduction in cellularity in the red pulp (RP) and the presence of aggregated nanoparticles (arrows) in the depleted regions. WP indicates white pulp. 100×; H&E. (C), In a serial section to B, the presence of macrophages is demonstrated in an area of the spleen in which the aggregated nanoparticles were readily observed by positive immunostaining with anti-Mac-3 antibody (brown product). 200×; hematoxylin counterstain. (D), Higher power photomicrograph of Mac-3 positive cells in the red pulp. 1,000×. (E), Perl’s Prussian blue staining (for recognition of ferric iron ions, indicative of hemoglobin degradation) indicates the presence of minimal hemosiderin (blue staining) in the red pulp of the spleen of a control mouse. 200×; neutral red counterstain. (F), Extensive hemosiderin (blue staining) in the red pulp of a mouse treated with AgNPs (500 mg/kg) and necropsied 7 days later. 200×; neutral red counterstain.
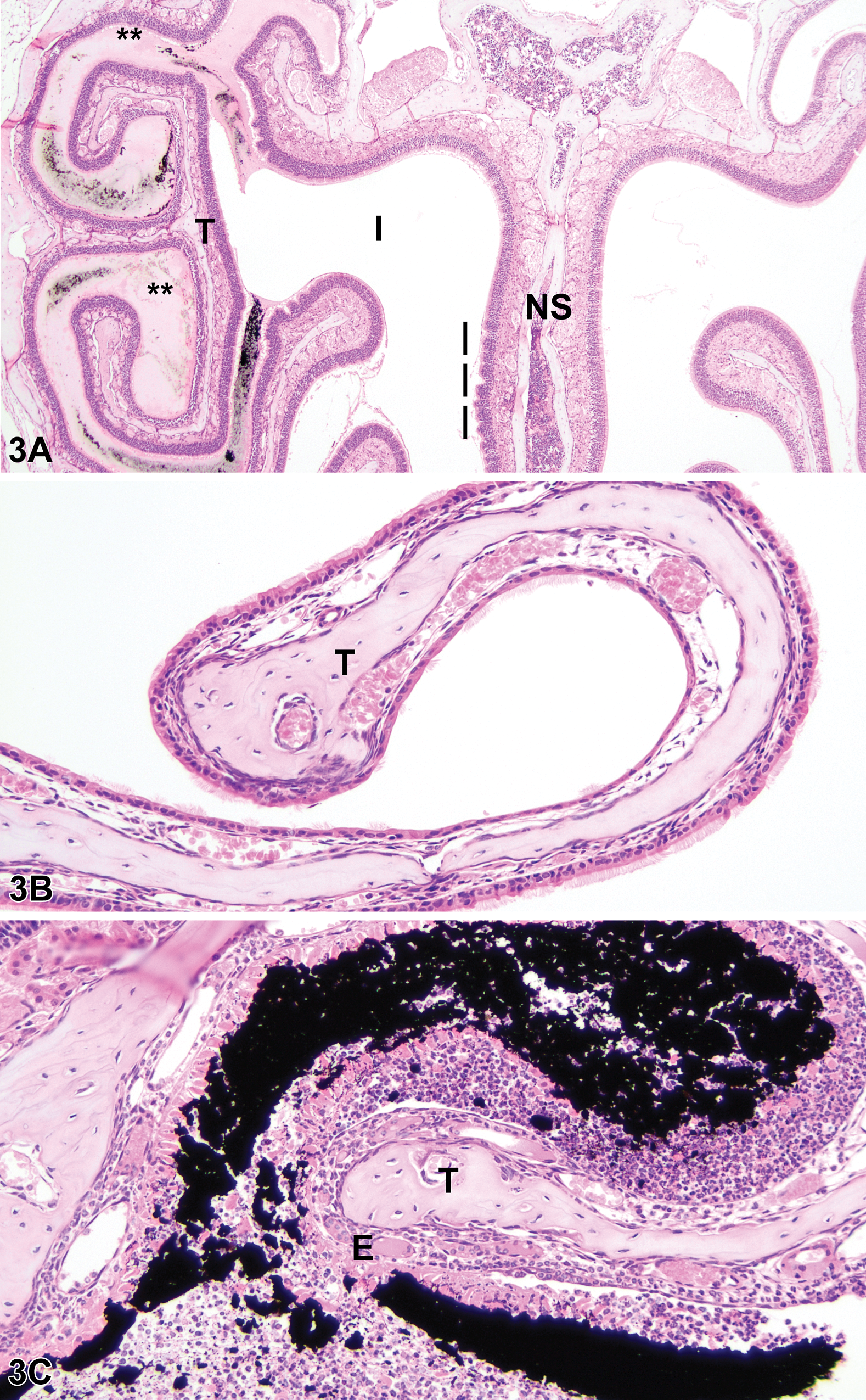
Airway changes associated with intranasal exposure to 25 nm silver nanoparticles (AgNPs). (A), Frontal section of a mouse treated with AgNPs (500 mg/kg) 1 day earlier, showing the presence of aggregated nanoparticles (black material in airway) and proteinaceous exudates (**) surrounding a turbinate (T) in the instilled (I) side of the nasal cavity. Vertical bars are adjacent to ruffling of the olfactory epithelium of the nasal septum (NS), a feature that often precedes sloughing of the epithelium. 40×; H&E. (B), Turbinate (T) from the untreated side of another mouse (500 mg/kg, necropsied 1 day later) showing normal epithelium and clear airways. 200×; H&E. (C), Instilled side of same mouse shown in (B) showing the corresponding turbinate (T) with eroded epithelium (E) amidst an inflammatory cell infiltrate and aggregated nanoparticles (black material). 200×; H&E.
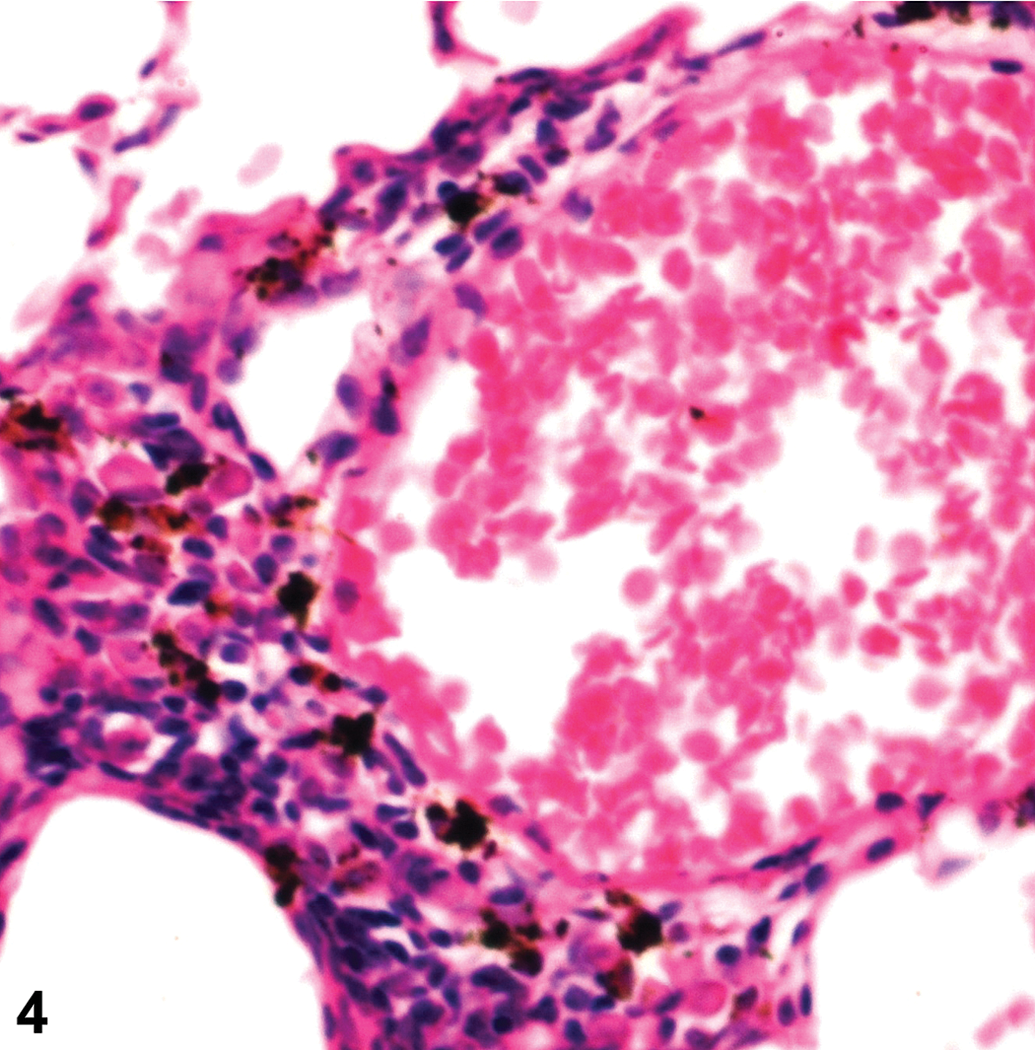
Lung of a mouse treated with silver nanoparticles (AgNPs; 500 mg/kg) and necropsied 1 day later, demonstrating perivascular expansion and aggregated AgNPs in the alveolar interstitum. 400×; H&E.
Discussion
Given the increasing use of nanomaterials across industry sectors, and particularly nanosilver materials in applications ranging from medical settings to surface and water disinfection, an understanding of their potential adverse effects in intact animals following exposure by a relevant route of administration is critical. Other investigators have previously demonstrated that IP-administered AgNP alters the expression of multiple genes in the mouse brain, including some associated with inflammation and oxidative stress (Rahman et al. 2009), but the IP route is not a route of exposure in either the medical or the occupational setting. Therefore, we undertook the present additional study with these NPs to evaluate their distribution and target tissue toxicity upon IN instillation. This route was used to mimic potential human exposures via the respiratory tract, which are possible in manufacturing operations where the nanosilver is actually prepared in bulk.
Two striking findings leapt out as we evaluated the tissues from the current experiment. The first was the accumulation of aggregated AgNPs in the spleen, accompanied by a mild loss of cellularity that was most pronounced in the red pulp. Red blood cells and their precursors are normally the major cell population in this zone of the spleen, and the cells of the erythroid lineage appeared to be the ones that were depleted. Furthermore, the histochemical evidence demonstrating increased hemosiderin deposition in the splenic red pulp of the AgNP-treated mice, compared to its nearly complete absence in the control mice, in the absence of inflammatory cell infiltrates in any organ suggests that locally enhanced destruction of erythrocytes within the spleen might be a possible explanation for erythrocyte scarcity. Mac-3-positive macrophages were clearly present in the red pulp (Figure 2), and these cells would have participated in erythrocyte turnover. Enhanced oxidative stress can promote premature senescence and removal of erythrocytes. Our current data support a dose-dependent change in oxidative state of the erythron over time, as the GSH levels in whole blood were higher 7 days after AgNP exposure—and significantly so at the high dose (Table 2). As GSH represents a reserve to restore oxidized molecules to their reduced state, a likely explanation for this increase in GSH is a physiological response to counteract the toxic effects of an oxidizing agent. Destruction of red blood cells in the spleen was not accompanied by a systemic decrease in hemoglobin levels (Table 1). This finding is reasonable, given the mild extent of the erythrocytic reduction in the splenic red pulp and the absence of red blood cell lesions in intravascular erythrocytes in all tissues. Additional work including bone marrow evaluation and blood smear cytology will be required in future studies to determine whether repeated AgNP exposure will result in an acute hemolytic episode or persist as an indolent, subclinical source of chronic blood loss. Furthermore, future experiments should also include additional end points to evaluate the extent of proinflammatory signaling, as local and circulating concentrations of cytokines may begin rising in advance of histologically demonstrable inflammatory cell infiltrates. Finally, an intriguing unanswered question to be investigated in coming studies is the manner in which “inhaled” NPs reached the spleen.
The second striking finding of the present study was the clear demonstration that nanosilver in the respiratory tract readily reaches the brain. While we did not identify aggregated Ag nanomaterial in the brain using conventional light microscopy, the presence of AgNP throughout the brain was clearly demonstrated using AMG as a special staining technique to enhance the visualization of nanosilver deposits (Figure 1). Accumulation was particularly prominent in the olfactory bulb and in association with cells lining the lateral ventricle, locations which are relatively close to the IN site of AgNP introduction. This association suggests that the particles reached the brain via the nasal cavity. Our current data cannot confirm this supposition, but the interpretation is reasonable. Rodents are obligate nose breathers, so the nearly complete absence of visible AgNP distribution within the lung (especially at the 1-day time point) suggests that few NPs reached the lung where they could have crossed the thin alveolar walls to reach the brain by a more indirect route.
We saw no morphologic evidence of inflammatory cell infiltrates in any organ, suggesting that AgNP do not induce a florid inflammatory response following IN administration. In particular, neuroinflammation was not appreciated either by routine light microscopic evaluation or by using CD45 immunohistochemistry as a marker of microglial activation (Thompson, Karpus, and Van Eldik 2008). Likewise, cytoarchitectural changes indicative of degeneration or death were not found in neurons or cells accumulating AgNP (e.g., choroid plexus epithelium, ependyma). Taken together, these observations suggest that acute, high-level, IN exposures to AgNPs will not incite an immediate inflammatory process in the brain. That said, the current experimental conditions do not exclude the possibility that neural inflammation (as indicated by subtle changes in gene expression, delayed microglial activation, etc.) could develop as a sequel to more chronic exposures. This potential points to the need for tests of neurological function in nanosilver-exposed animals. Evaluation of potential neurotoxic effects following pre- and/or perinatal AgNP exposure is also warranted, given that sub-cytotoxic concentrations of AgNPs have been shown to prevent neuronal differentiation in vitro of PC12 cells (a rat adrenal medulla cell line; Powers et al. 2010).
Aggregated NPs were readily observable in other tissues by routine light microscopy, including those of the respiratory tract (lung and nasal cavity) and the renal medulla. The airways would be the expected sites of AgNP deposition, since materials introduced IN have a natural route to the lower respiratory tract. Inhaled fine particles accumulate over time in the lung (Elder et al. 2006). The pulmonary parenchyma was unaffected in our present acute study, indicating that the single acute, high-level, IN exposure administered in this experiment did not induce substantial damage in the lower respiratory tract. The particles in the kidney were located within the tissue, not the lumens of nephrons, indicating that this organ was accumulating rather than eliminating AgNP. Changes were not evident in the excretory epithelium, interstitial stroma, and blood vessels, suggesting that the kidney also is not affected to a major degree by an acute, high-level, IN exposure to nanosilver. As stated above, the present experimental conditions do not imply that the integrity of these organs would remain unaffected in the face of a more chronic exposure, even one resulting from low-level IN exposure.
The prominent accumulation of nanosilver in the spleen in our mice suggests the theoretical potential to impact the function of the immune system following respiratory tract exposure to AgNP. The reasons for this hypothesis are that the AgNP appeared to be sequestered in splenic macrophages and that such cells may secrete a variety of cytokines that modulate immune cell activities. Nanomaterials can be engineered to avoid immune system recognition, but they can inhibit or stimulate immune system responses (Zolnik et al. 2010). For example, neutrophil function is inhibited in fathead minnows by hydroxylated fullerenes (Jovanović et al. 2011). In contrast, intratracheal instillation or inhalation of carbon black exacerbates lipopolysaccharide-induced lung inflammation in mice (Inoue 2011). AgNPs (20–80 nm) have been shown to increase BBB permeability and proinflammatory cytokine induction in cultured rat brain microvessel endothelial cells, with the smaller NPs having the most pronounced effect (Trickler et al. 2010). NPs can also induce allergic contact dermatitis (e.g., palladium) or autoimmunity (e.g., diesel exhaust particles; Di Gioacchino et al. 2011). We did not test immune function in the current study, so the potential effect of nanosilver on immune system responses remains to be assessed. In particular, the present findings warrant further study of the distribution and potential toxicity in other lymphoid organs, such as lymph nodes, thymus, mucosa-associated lymphoid tissues (e.g., associated with bronchi and the intestinal tract), and bone marrow. The absence of histopathological lesions in the splenic white pulp suggests that acute, high-level, IN exposure to AgNP did not impair acquired immunity (i.e., the lymphocyte-mediated arm). However, as noted above for the brain, this inference should not be extended on the basis of the current data to the chronic, low-level exposure scenario that would be expected in the occupational setting.
Conclusions
Our findings add to the body of knowledge indicating that the route of exposure is pivotal in defining the target organs to which nanosized particles will distribute and cause toxicity. First, the current mouse study indicates that AgNP readily enters the brain following IN instillation. Furthermore, our data indicate that AgNP will reach potential target sites that are not directly linked to the respiratory tract (e.g., kidney, spleen), which suggests the possibility that nanosilver can be taken up from the respiratory mucosa following IN exposure and then redistributed internally (perhaps inside circulating phagocytic cells). We anticipate that such properties will have major implications for NP risk assessment.
Second, our results also indicate the need to undertake a more comprehensive evaluation of AgNP toxicity going forward. The current experiment (and also prior studies) employed a narrow range of end points as a preliminary screen for potential target organs of toxicity and possible mechanisms. Our experience indicates that future studies should include gross and histopathological assessment of a complete tissue battery, including especially bone marrow and other lymphoid organs; cytological evaluation of blood smears; and functional assays for biomarkers that might prove to be useful indices for currently suspected mechanisms of AgNP-induced tissue injury (e.g., upregulated production of messenger RNA [mRNA] transcripts or translated proteins for molecules in proinflammatory or antioxidant pathways).
Finally, our results demonstrate the utility of a relatively simple histochemical technique, AMG, in localizing the deposition of AgNPs. When we did not observe aggregated AgNPs in the brain using conventional light microscopy, as was observed in spleen and lung, we initially concluded that nanosilver was not distributed into the brain upon IN instillation. However, our post hoc AMG analysis clearly demonstrated that silver could be detected in specific brain regions after IN instillation. Furthermore, their proximity to the nasal cavity permits the inference that the NPs penetrated the rostral brain by translocation across the nasal mucosa, while the presence of nanosilver in the lateral ventricles suggests that the material might be spread more widely over time. Obviously, the validity of these interpretations will require verification. Nonetheless, we predict that AMG will become an important ancillary method for exploring nanomaterial uptake and distribution during future hazard identification and risk assessment efforts for ultrafine metals.
Footnotes
Acknowledgments
We acknowledge the University of Cincinnati Center for Environmental Genetics (P30-ES006096), NIOSH ERC T42-OH008432, Molecular Epidemiology in Children’s Environmental Health T32-ES10957, and the University of Cincinnati Dean’s Bridge Fund for financial support. These sponsors had no other input other than financial support. We thank Dr. Michael Borchers for assistance with inflation fixation of mouse lungs. We acknowledge the contribution of Dr. Saber M. Hussain for his previous characterization of the AgNPs used in this study. The authors thank the reviewers, and especially reviewer No. 2, for the many helpful comments that have served to greatly improve the final form of this article.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This research was supported through the University of Cincinnati Center for Environmental Genetics P30-ES006096, NIOSH ERC T42-OH008432, Molecular Epidemiology in Children's Environmental Health T32-ES10957, and the University of Cincinnati Dean's Bridge Fund.
