Abstract
This is the first utilization of advanced analytical electron microscopy methods, including high-resolution transmission electron microscopy, high-angle annular dark field scanning transmission electron microscopy, electron energy loss spectroscopy, and energy-dispersive X-ray spectroscopy mapping to characterize the organ-specific bioprocessing of a relatively inert nanomaterial (nanoceria). Liver and spleen samples from rats given a single intravenous infusion of nanoceria were obtained after prolonged (90 days)
The long-term fate of nanomaterials is not well understood. Concerns about adverse human health impacts due to a single or repeated exposure may deter some nanotechnology developments from reaching commercial implementation. A greater understanding of the roles of nanoparticle biodistribution and fate, interactions with tissues, and
Nanomaterials can be bioprocessed/biodegraded
Nanoceria has current applications and there is great interest in its potential as a therapeutic agent. It has industrial applications that capitalize on its abrasiveness and autocatalytic oxidation/reduction, mediated by reversible oxygen binding at its surface, interconverting reduced cerium (Ce) (III) and oxidized Ce(IV). It is used in chemical mechanical polishing/planarization (e.g., integrated circuit manufacture) and as a catalyst (in diesel fuel additives, solid oxide fuel cells, and other electrochemical devices; Reed et al. 2014). Capitalizing on its autocatalytic redox, antioxidant/anti-inflammatory, and enzyme-mimetic (nanozyme) activity, nanoceria has been assessed and shown, mostly in
Nanoceria was reported to be insoluble in water, physiological saline, cell culture medium, phagolysosomal simulant fluid, and artificial phagolysosomal fluid at pH 4.5 and 5.5, and only 0.02% solubilized in 0.1N HCl after 28 days (He et al. 2010; Cho et al. 2012; Geraets et al. 2012; Wohlleben et al. 2013; Keller et al. 2014). It persists
Exactly how
Materials and Methods
Nanomaterial
Synthesis and characterization of the 5.2% citrate-stabilized cubic nanoceria sterile aqueous dispersion used for iv administration of a single high dose to rats was described (Yokel et al. 2012; Yokel et al. 2013). The synthesized nanoceria was crystalline. It had Miller indices of (100), (010), (200), and lesser amounts of (311), and a measured

High-resolution transmission electron microscopy image of a cubic ∼30-nm nanoceria particle from the dispersion given by iv infusion to rats and corresponding diffraction pattern. The dominant face is (100).
Animals
The animals (male Sprague-Dawley rats) and their housing, diet, and monitoring; nanoceria administration; samples collected at termination; and tissue preparation and histological evaluation procedures were described (Yokel et al. 2012). Nanoceria-treated rats received a 1hr iv infusion of 85 mg/kg nanoceria. This single high-dose nanoceria exposure was utilized following the toxicology/experimental pathology approach of first determining whether a novel substance produces effects (hazard identification) to be followed by lower exposures to determine the dose–response determination. The high dose was also used to maximize the possibility of quantifying Ce by ICP-MS and detecting nanoceria by microscopy up to 90 days later. Given the biopersistence of nanoceria, tissue levels achieved after the single high dose might be reached after repeated smaller doses. Initial studies showed the dose to be well tolerated (Yokel et al. 2012). Control rats received vehicle infusion. Three nanoceria and 3 control rats were terminated each of 1, 7, and 30 days after completion of the infusion. Six control and 7 nanoceria-treated rats were terminated 90 days after the infusion. They were anesthetized to lack of response to stimulus with 80 mg/kg ketamine and 10 mg/kg xylazine ip prior to termination. Tissues were harvested from the liver and spleen and were fixed in 10% neutral-buffered formalin and processed for histopathology analysis. Animal work was approved by the University of Kentucky Institutional Animal Care and Use Committee. The research was conducted in accordance with the Guiding Principles in the Use of Animals in Toxicology.
Microscopy for Histological Examination
Samples of liver and spleen were cut into 3 mm3 pieces, dehydrated, and embedded in Araldite 502. The blocks collected for histology were sectioned to 1 μm thickness and stained with toluidine blue for light microscopy screening. Selected blocks were further sectioned, mounted on Formvar/carbon-coated copper grids (200 mesh, Ted Pella Inc., Redding, CA), and examined with a Phillips CM-10 low-resolution electron microscope equipped with a LaB6 cathode (Phillips Electronic Instruments Co., Eindhoven, the Netherlands) operated at 60 kV. Nanoceria-containing Kupffer cells were detected by their enlarged size and sinusoidal location in the liver, while Ce-phosphate nanoneedles in the white pulp of the spleen were recognized at low resolution as dense agglomerates around lymphatic nodules.
HRTEM/STEM, EELS, and EDS Methods
For HRTEM tissue, sample blocks were cut to ∼50- to 70-nm thick sections and collected on 200 mesh Formvar/carbon-coated copper grids without staining. Instruments included a JEOL 2100F field emission TEM/STEM operated at 200 kV with an analytic pole piece. Images were recorded with a Gatan Ultrascan 4k × 4k CCD camera, and data analysis and processing used Gatan Digital Micrograph software. High Resolution Scanning Transmission Electron Microscopy (HRSTEM) imaging, EELS, and EDS were performed with a Gatan high-angle annular dark field (HAADF) detector, Digiscan II, and Gatan 2000 Image Filter. An Oxford Aztec EDS system was used for select elemental mapping. All HRSTEM images were acquired using an analytical probe with 0.17-nm resolution. The EELS measurements and EELS trace lines were obtained using a 1-nm probe, α of 12 mrad and β of 6 mrad. Select elemental intensity maps were obtained from core edge intensity obtained after background subtraction using an integration window ranging from 10 to 30 eV depending on the edge of interest (Ce, O, and P edges). A FEI Talos transmission and scanning electron microscope was used for fast EDS mapping with a high degree of sensitivity due to the wrap-around style EDS detector mounted on the objective lens. Maps generally took 1 to 2 min to acquire with a sensitivity great enough to detect elemental concentrations in 4-nm size particles. EDS provides the means to determine the relationship between elemental accumulation and tissue regions, particularly in a situation where dynamic processes may be in play such as in bioprocessing. This study focused on a high dose to obtain excellent EDS signal strength in different organ tissue, but low dose exposure of nanoparticles can also be traced using HRSTEM applications (Graham, Jacobs, et al. 2017).
Results
Rats intravenously infused with a single high dose (85 mg/kg) of ∼30-nm ceria displayed particle retention in the liver and spleen up to 90 days post infusion. Hepatic and splenic granulomas were dispersed throughout the liver as reported previously (Tseng et al. 2012). Agglomerates of densely packed nanoceria were found in engorged Kupffer cells (Figure 2A). Spleen cellular agglomerates are shown in Figure 2B and were comprised of intracellular retention of nanoparticles which were particularly abundant in the white pulp. Light microscopic examination also revealed that the spleen showed a much greater extent of granuloma formation at 90 days compared to the liver. Ce concentration differences in liver versus spleen for various time end points up to 90 days postinfusion are detailed elsewhere (Yokel et al. 2012). Ninety days postinfusion, the spleen had a much higher concentration of Ce (2,800 µg/g) than the liver (300 µg/g). At that time, histology of the liver revealed intracellular agglomerations up to micron size diameter made up of nests of closely packed particles (nanoceria and/or Ce-containing degradation products). The agglomerations could be linked to adverse effects in the liver, including the formation of granuloma seen in light microscopy (Figure 2A). The presence of Kupffer cells provided a “marker” for the identification of hepatic granuloma, as described in Tseng et al. (2012) and Tseng et al. (2014). Large cytoplasmic nanoceria-containing agglomerates were observed in macrophages throughout the red and white pulp splenic parenchyma, more abundantly found in the white pulp (Figure 2B). In light microscopy, we saw dense particles but could not determine whether the primary nanoceria particles displayed any transformation inside the organs.
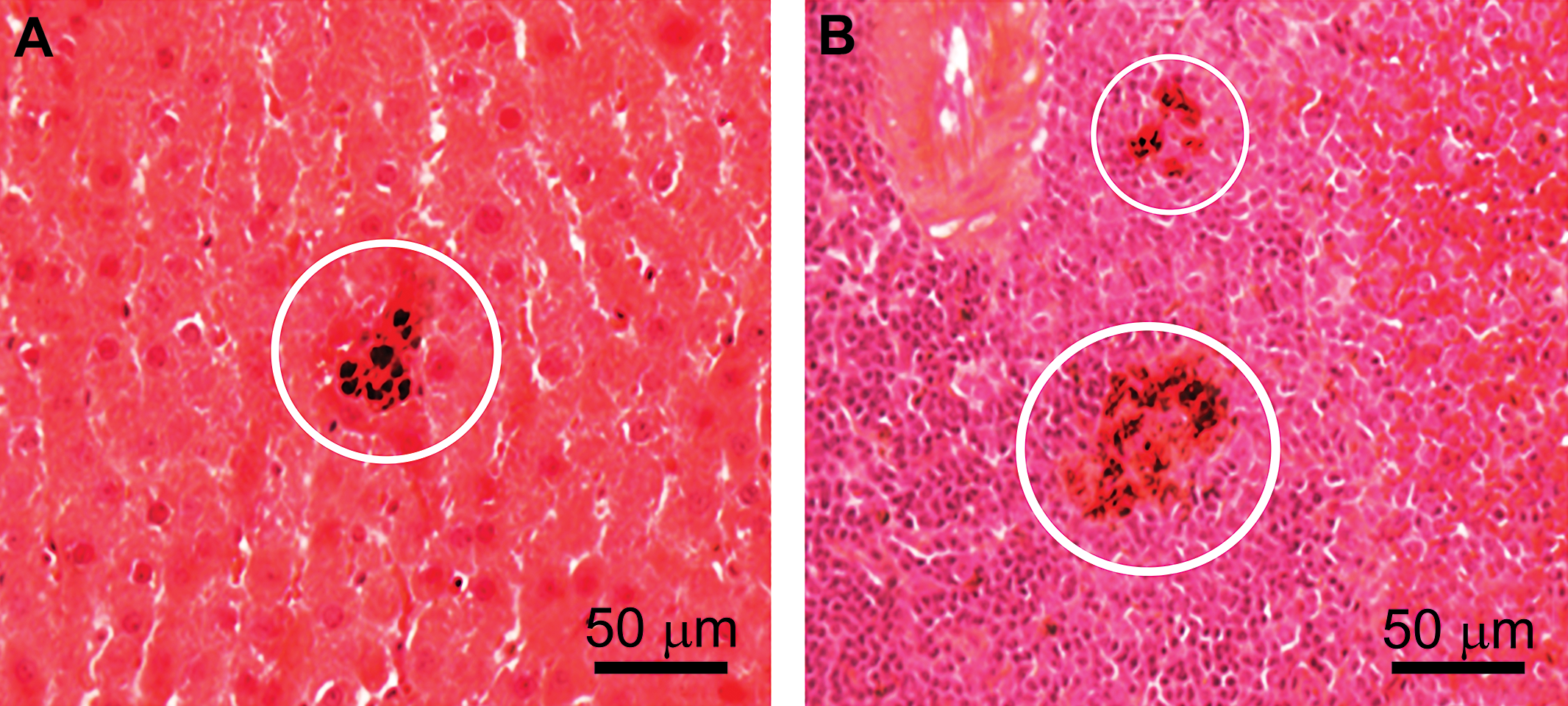
Hepatic and splenic granulomas. (A) Agglomerates of densely packed nanoceria are shown in engorged Kupffer cells in the liver (circled area). (B) Cellular agglomerates with intracellular retention of nanoparticles are shown to occur abundantly in the spleen white pulp (circled areas).
HRTEM revealed structural evidence that nanoceria in the liver was not inert to bioprocessing but was degrading over 90 days (Figure 3A and B). This was evidenced by loss of some of the original structure of the as-dosed cubic nanoparticles that exhibited well-rounded corners after 90 days (Figure 3B) and formation of second-generation Ce-containing ultrafine (<3 nm) crystalline particles in the immediate vicinity of the partially degraded as-dosed nanoparticles (Figure 3B). The second-generation particles were significantly smaller than the as-dosed nanoparticles and had a different chemical composition (more reduced Ce(III) as determined by EELS) than the as-dosed nanoceria (Figure 3D compared to Figure 3C). In the EELS spectrum, the M4 peak is strongly influenced by Ce(III), while the M5 peak height is a relative measure of the presence of Ce(IV). The high M5/M4 ratio (Figures 3D) can be linked to high Ce(III) and to antioxidant properties, as was previously shown for nanoceria bioprocessing in the liver (Graham et al. 2014). The morphological and chemical differences between the as-dosed (Figure 1) and second-generation particles (Figure 3B shows morphology; C and D show composition) indicate a dissolution-reprecipitation process occurred. The possibility that nanoceria may also undergo bioprocessing in organs other than the liver prompted us to examine events in the spleen which contained the highest Ce concentration, and third greatest amount of Ce (after liver and bone marrow) that was sequestered after iv infusion (Yokel et al. 2012). Nanoceria translocated and accumulated in the spleen (Figure 3E). Initial scanning of spleen tissue sections in TEM and STEM modes using a high-resolution probe (0.2 nm) revealed localized areas with agglomerated, crystalline nanoceria which match the original morphologies and size range of the as-dosed particles (Figure 1), but displayed partially missing corners due to dissolution and bioprocessing (Figure 3E and F), which were seen with dark field STEM imaging. The degree of bioprocessing occurred to a lesser extent compared to nanoceria sequestered in the liver (Figure 3E and F compared to Figure 3B). Localized second-generation plumes of ultrafine (0.5–2.5 nm) nanoparticles formed close by, collecting as ultrafine particles next to the partially dissolving primary nanoceria (Figure 3F). Corresponding EELS analyses (small probe size [0.2 nm] to minimize beam/tissue interactions) were used to compare the redox state of the as-dosed primary nanoceria and
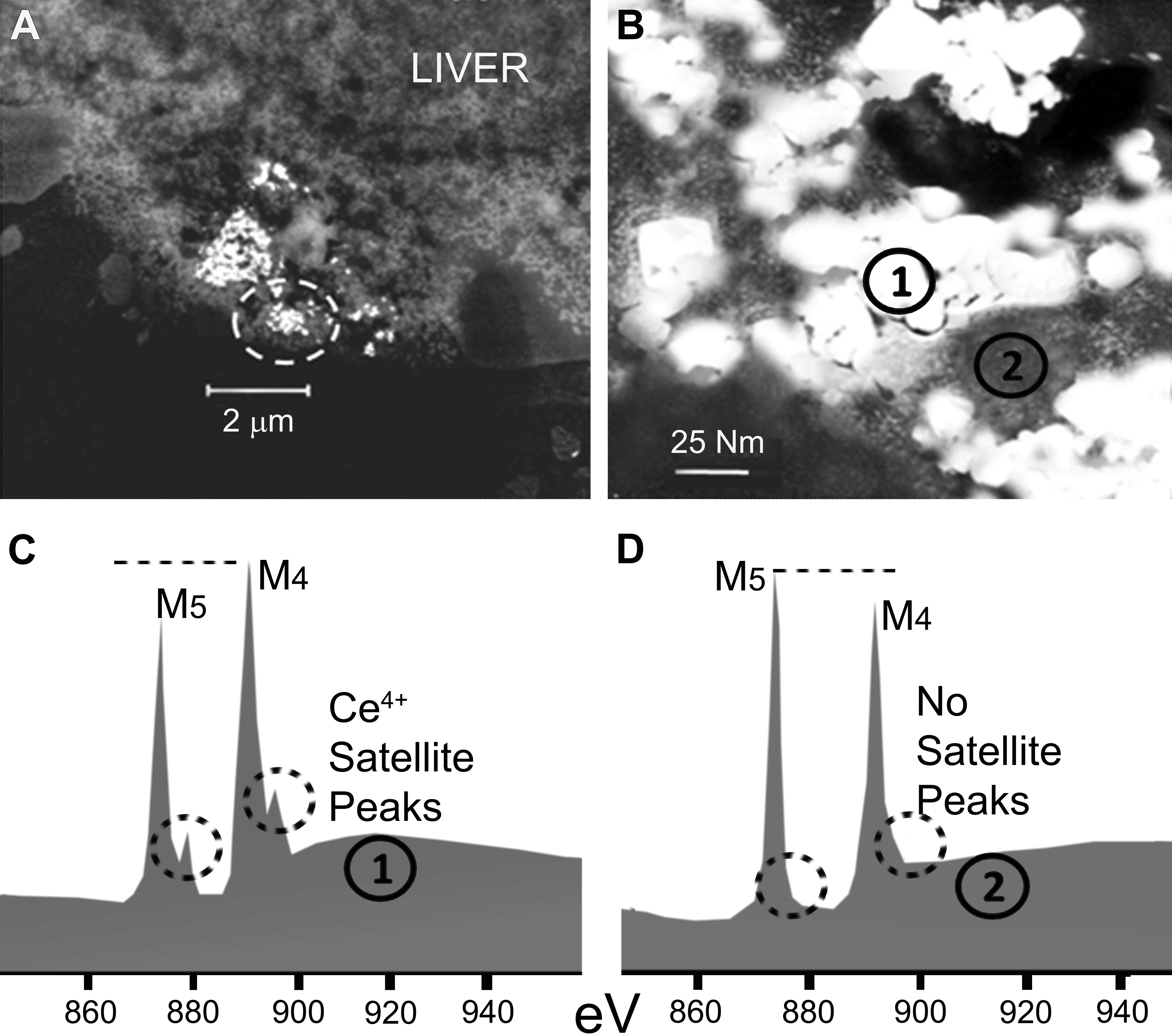
Bioprocessing of nanoceria in liver and spleen. Figure (A) shows transmission electron microscopy and (B) shows HRSTEM of nanoceria in the liver. Figure (B) illustrates high degree of nanoceria rounding (circled area 1) and clouds of ultrafine particles (circled area 2). Figures (C) and (D) show electron energy loss spectroscopy (EELS) measurements with peak intensities for M4 and M5 at locations 1 and 2 in Figure (B), respectively.
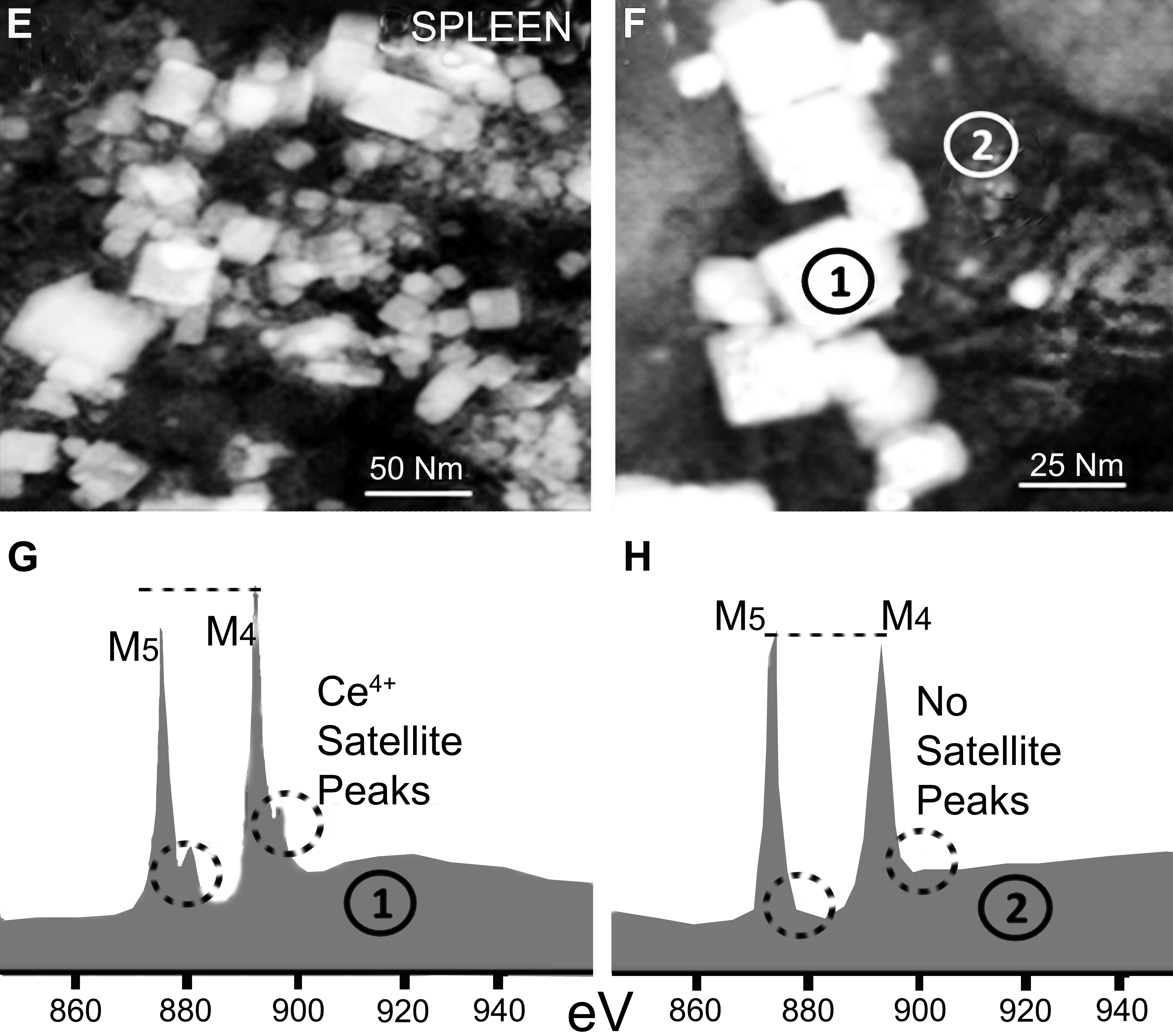
Figures (E) and (F) show HRSTEM of nanoceria in the spleen with lesser degree of rounding in circled area 1 than seen in the liver (B); clouds of ultrafine particles are shown in circled area 2. Figures (G) and (H) show EELS measurements with peak intensities for M4 and M5 at locations 1 and 2 in Figure (F), respectively.
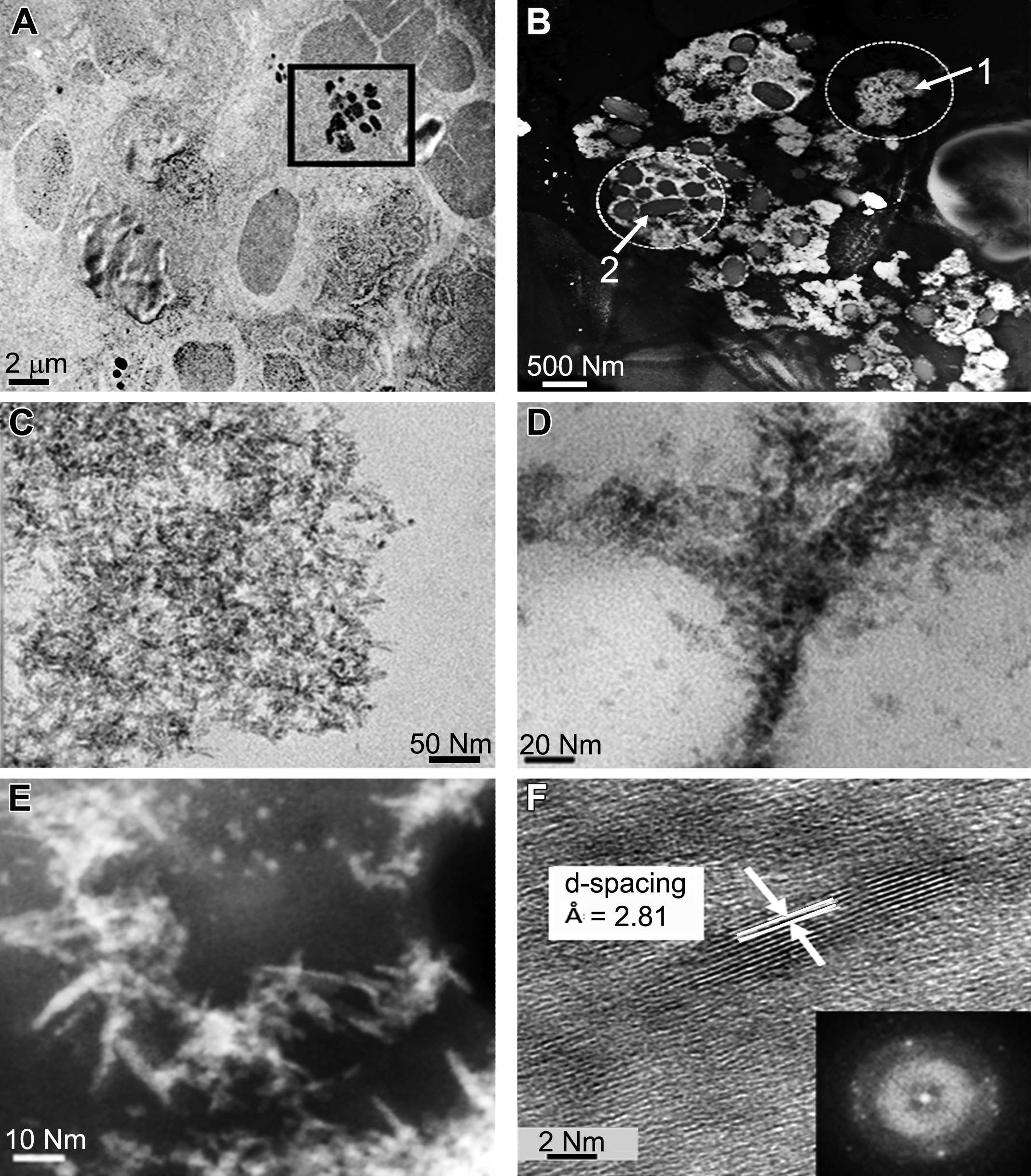
High-resolution transmission electron microscopy (HRTEM)/STEM images show retention of nanoceria and precipitation of Ce-phosphate nanoneedles in the spleen. (A) Transmission electron microscopy shows general location of nanoceria in spleen tissue. (B) HRSTEM illustrates formation of ultrafine precipitates as free networks in location 1 and as surface coatings around semispherical splenic structures in location 2. (C) HRTEM shows a network of ultrafine nanoneedles from location 1 in Figure (B). (D) HRTEM illustrates selective location of nanoneedles and single crystallites on splenic structures from location 2 in Figure (B). (E) HRSTEM shows select regions on splenic structures with larger nanoneedles. (F) HRTEM shows atomic resolution of nanoneedles with corresponding
A Talos F200 STEM with an ultrahigh resolution analytical beam at 200 kV energy was used to obtain better insight into the elemental components of the nanoneedle structures and the surrounding host cellular components in the spleen. A relatively larger area containing a network of nanoneedles spanning ∼1 micron in 3-D was used for EDS mapping (Figure 5). The high-angle annular dark field scanning transmission electron microscopy (HAADF-STEM) image (Figure 5A) illustrates the nanoneedle agglomerate. Corresponding EDS maps indicate a match for O, P, and Ce for each nanoneedle (Figure 5A), which corroborates the earlier findings that the nanoneedles represent Ce phosphate. Nanoneedles are very thin, giving a greatly reduced EELS signal. The mass of a single partially formed nanoneedle (Figure 5B) provided an EELS signal with a sufficient signal to noise ratio to assess the M5/M4 ratio. The M5 peak being even with the M4 peak is indicative of the presence of Ce(III) and a more reduced state of Ce (Graham, Jacobs, et al. 2017). Further work is required to assess the beam dose sensitivity of the nanoneedles, so that methods can be developed to further access the oxidation state of the needles in greater detail.

Talos—energy-dispersive X-ray spectroscopy (EDS) mapping. (A) EDS maps of spleen regions containing nanoneedles: high-angle annular dark field scanning transmission electron microscopy (HAADF-STEM) image shows nanoneedle networks with corresponding maps for O, P, and Ce in blue, green, and yellow, respectively. (B) HAADF-STEM image including a single nanoneedle in the spleen with corresponding electron energy loss spectroscopy spectrum of the nanoneedle.
A high-resolution elemental EDS map was also constructed for individual nanoneedles (Figure 5C). Individual crystallites that had self-aligned to form the nanoneedles can be seen in the higher-resolution EDS map (Figure 5C), which shows perfect overlap for Ce and P in each individual aligned crystallite, where the nanoneedles formed in the spleen. Oxygen was also identified but is not shown in the EDS maps. Iron, corresponding to a ferritin particle (biomineralized iron ∼5 nm), occurs in the immediate vicinity of the Ce-phosphate nanoneedles (Figure 5C). The importance of ferritin nanoparticle formation in regions next to invader nanoparticles was described by Graham, Jacobs, et al. (2017). The elemental spectra shown in Figure 5D for 2 select regions, spectrum 1 (green) taken directly from a selected nanoneedle from box 1 in Figure 5C and spectrum 2 (red) taken from the ferritin nanoparticle from box 2, clearly indicate that the nanoneedles are composed of Ce, P, and O and do not represent nanoceria that were iv infused into the rats; the ferritin is composed of only Fe and O and represents biomineralized iron. The coexistence of P and nanoneedles constitutes a clear difference between liver and spleen since Ce phosphate is characteristic for the spleen and plays a lesser role in the liver.
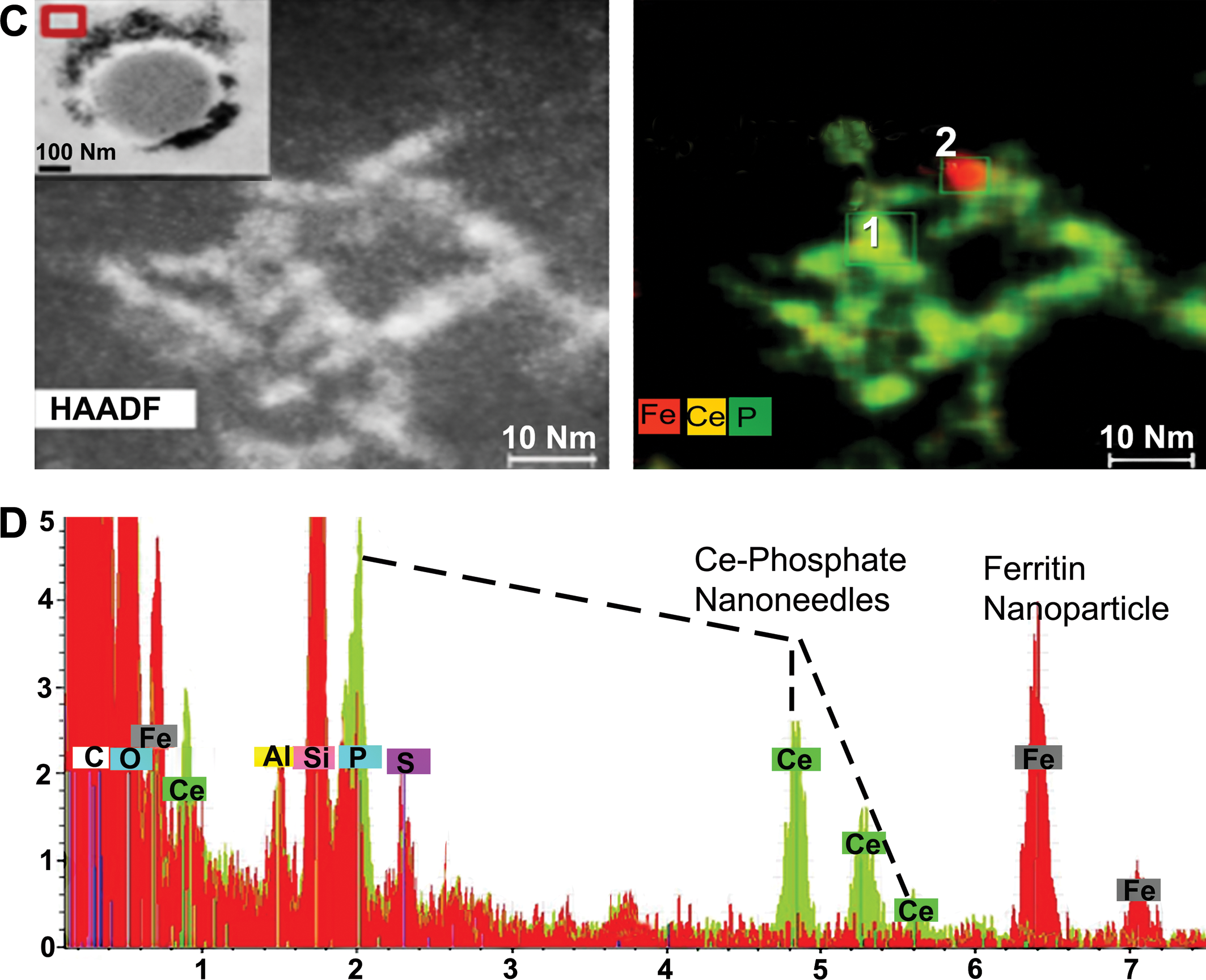
Talos—energy-dispersive X-ray spectroscopy (EDS) mapping. (C) HAADF-STEM image (with transmission electron microscopy [TEM] inset in left upper corner) showing accumulations of nanoneedles around splenic structure; red-lined box in TEM inset marks location where HAADF-STEM image was derived, which included only a few isolated nanoneedles. Corresponding EDS map for the nanoneedles shows Fe (red), Ce (yellow), and P (green). Red area corresponds to a 4-nm ferritin nanoparticle. Overlapping yellow and green areas correspond to nanoneedles. (D) Two select EDS spectra obtained for a nanoneedle at location 1 in Figure (C) and a ferritin nanoparticle at location 2 in Figure (C).
Discussion
Nanoceria persistence in both the liver and spleen has been shown previously to contribute to the observed toxicity in these organs (Tseng et al. 2012). Here, we report that the spleen shows even more granuloma formation than the liver 90 days after a single nanoceria infusion. The observed granulomas in both liver and spleen are the result of the high iv exposures and would not be expected in humans. The high dose was chosen to observe differences during microscopic analysis. The white pulp contained numerous foci where clusters of cells with nanoceria inclusion were observed (Figure 2). The splenic microanatomy can be analyzed through its vasculature. Once the lineal artery enters the spleen through the hilus, it branches into progressively smaller caliber arteries before entering the interior fibrous trabeculae. At that point, they become known as the central arteries surrounded by sheaths of enlarging lymphatic tissue to form splenic follicles known as the white pulp. The central arteries exiting from the white pulp branch to form the penicillar arteries before entering the red pulp of the spleen. While the splenic white pulp is essentially made of lymphatic tissue, the red pulp contains the sinuses and the splenic cords (Steiniger, Bette, and Schwarzbach 2011). Within the splenic cord cavities, mixtures of red and white cells are abundantly present in the red pulp. This is especially relevant for experimental treatment entities including nanoparticles. Through these structural features of the splenic microcirculation, invader nanoparticles may permeate throughout the splenic parenchyma. It has been reported that more than 90% of the splenic blood flow bypasses the red pulp in favor of the white pulp (Schmidt et al. 1993). It highlights the prevalent role of the spleen as a lymphoid organ and suggests that blood-borne entities, including nanoparticles (nanoceria), could preferentially affect the white pulp upon entering the spleen, as observed in the current study. In contrast, nanoceria uptake in the liver is mainly conducted by Kupffer cells and T cells leading to the formation of granuloma (Tseng et al. 2012) but not as pronounced as in the spleen.
Instability of nanoparticles after exposure, cellular uptake,
Biodegradation of iron-containing nanoparticles has been described. Degradation of superparamagnetic iron oxide nanoparticles (SPIONs) in rat liver was suggested by reduction of the dark T2* Magnetic Resonance Imaging (MRI) signal intensity produced by these particles and a reduction in Prussian blue staining over 4 weeks (P.-W. Lee et al. 2010). Using inductively coupled plasma optical emission spectrometry (ICP-OES) to quantify total iron and ferromagnetic resonance and a superconducting quantum interference device magnetometer to measure magnetization (intact SPIONs), Levy et al. (2011) found a decrease in Superparamagnetic Iron Oxide Nanoparticles (SPION) iron in mouse liver, and to a lesser extent the spleen, and increase in non-SPION iron in the spleen over 90 days. These results suggest SPION degradation in the liver and release of iron-containing degradation products that accumulate in the spleen. TEM showed a reduction in electron-dense particle size with less-defined contours after 30 days compared to the dosed SPIONs and a lack of nanoparticles at 90 days. Using a magnetophoresis technique to measure magnetization in tissue composed of human mesenchymal cells, Mazuel et al. (2016) observed a decrease in citrate-coated iron oxide nanoparticles over 27 days. Their results suggested dissolution of some particles, which was confirmed by TEM.
The present study applied sophisticated electron microscopy methods to the study of nanomaterial bioprocessing. This is the first utilization of advanced, analytical electron microscopy methods, HRTEM, HAADF-STEM, EELS, and EDS mapping, to characterize the effects of bioprocessing on the physicochemical properties of nanomaterials in different organs. Specifically, we compare bioprocessing effects of intravenously administered nanoceria in liver and spleen samples after a 90-day exposure. These methods were applied to elucidate the organ-specific cellular and subcellular fate of nanoceria after its uptake. As noted by Mazuel et al. (2016, pg. 4), “Transmission Electron Microscopy (TEM) is the method of choice for in situ nanoscale location of nanoparticles within the model tissue and its component cells, and for monitoring their individual fate as a function of tissue-maturation.” We demonstrated that the
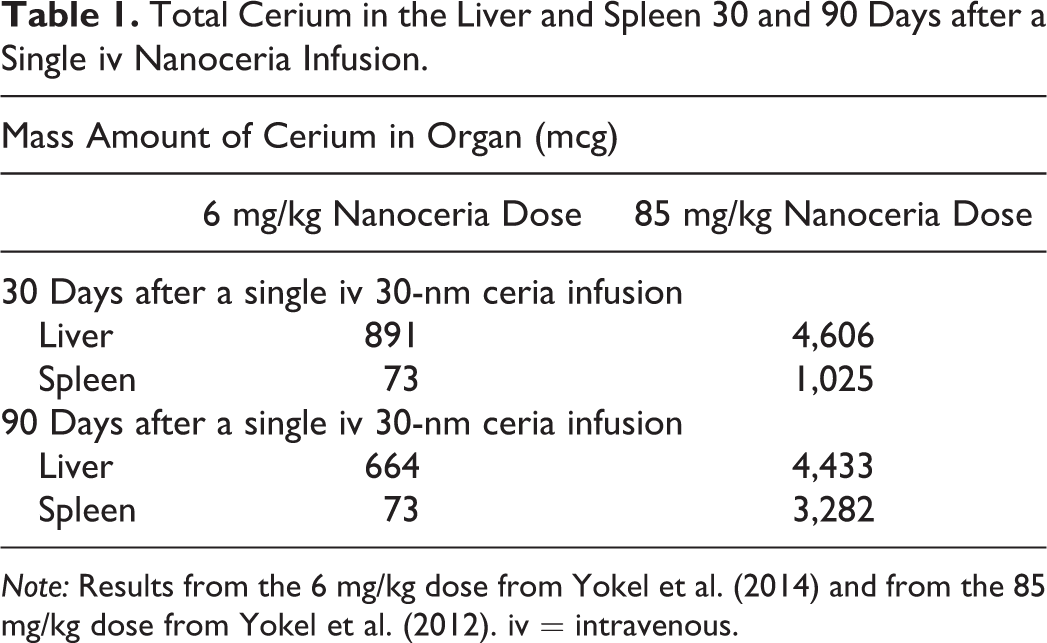
The spleen, unlike the liver, has a very high concentration of Ce-phosphate nanoneedles, as shown in Figure 4. Sisler et al. (2016) reported that La2O3 nanoparticle conversion to La-phosphate nanoparticles (LaPO4) was associated with chronic pulmonary inflammation and mild fibrosis in mice. The formation of second-generation Ce- and P-containing nanoneedles in large 3-D networks in the current study is indicative of a precipitation mechanism involving Ce ion containing supersaturated solution and ion transport. We also observed nearly spherical structures in the spleen (Figure 4B) that are surrounded/coated by Ce-phosphate nanoneedles, which further indicates the nanoneedles formed when Ce ions interacted/reacted with a P-containing surface or membrane. This also requires a Ce-ion transport mechanism as indicated in the bioprocessing model in Figure 6. The source for the abundant Ce-phosphate nanoneedles in the spleen would require that a large fraction of the primary nanoceria was bioprocessed (dissolved) providing abundant Ce ions. However, in the spleen, the primary nanoceria shows little evidence for dissolution. Therefore, the Ce-phosphate nanoneedles in the spleen (Figures 4 and 5) must have formed from Ce ions transported to the spleen rather than via bioprocessing of the primary nanoceria that were initially cleared by the spleen after iv infusion. Major bioprocessing of nanoceria in the liver via dissolution processes and a lack of accumulation of Ce phosphate in the liver indicates that some transport of Ce ions from liver to the spleen occurs, as schematically illustrated in Figure 6. The decrease of Ce in the liver from 30 to 90 days after a single iv nanoceria infusion paired with the concomitant lack of decrease, or increase, in the spleen (Table 1) is consistent with Ce-ion transport from liver to spleen. Availability of Ce “ions” for complexation with regionally accessible phosphate groups in phosphate-rich or depleted regions may govern the amount of Ce-phosphate nanoneedles that can form. Some metal complexing without nanoparticle precipitation (ion adsorption) may take place even before (yellow zone in Figure 6) the Ce ions arrive at phosphate-rich regions (green zone in Figure 6). Therefore, local ion transport rates will have some control on the rate of formation of Ce nanoneedles. Ion transport rates will be most likely affected by the local environment. This may explain why we observe second-generation nanoparticles in both liver and spleen but with very different amounts. This, in part, is due to the translocation of Ce from liver to spleen (Figure 6). Our findings are consistent with previous data that show a decrease of Ce in the liver for 2 doses of nanoceria (6 mg/kg, Yokel et al. 2014; 85 mg/kg, Yokel et al. 2012) 30 and 90 days after iv administration that was accompanied by an increase in Ce in the spleen. A considerable increase in Ce was seen in the spleen by Yokel et al. (2012) after the greater dose (85 mg/kg) which is the same dose used in this study (Table 1).
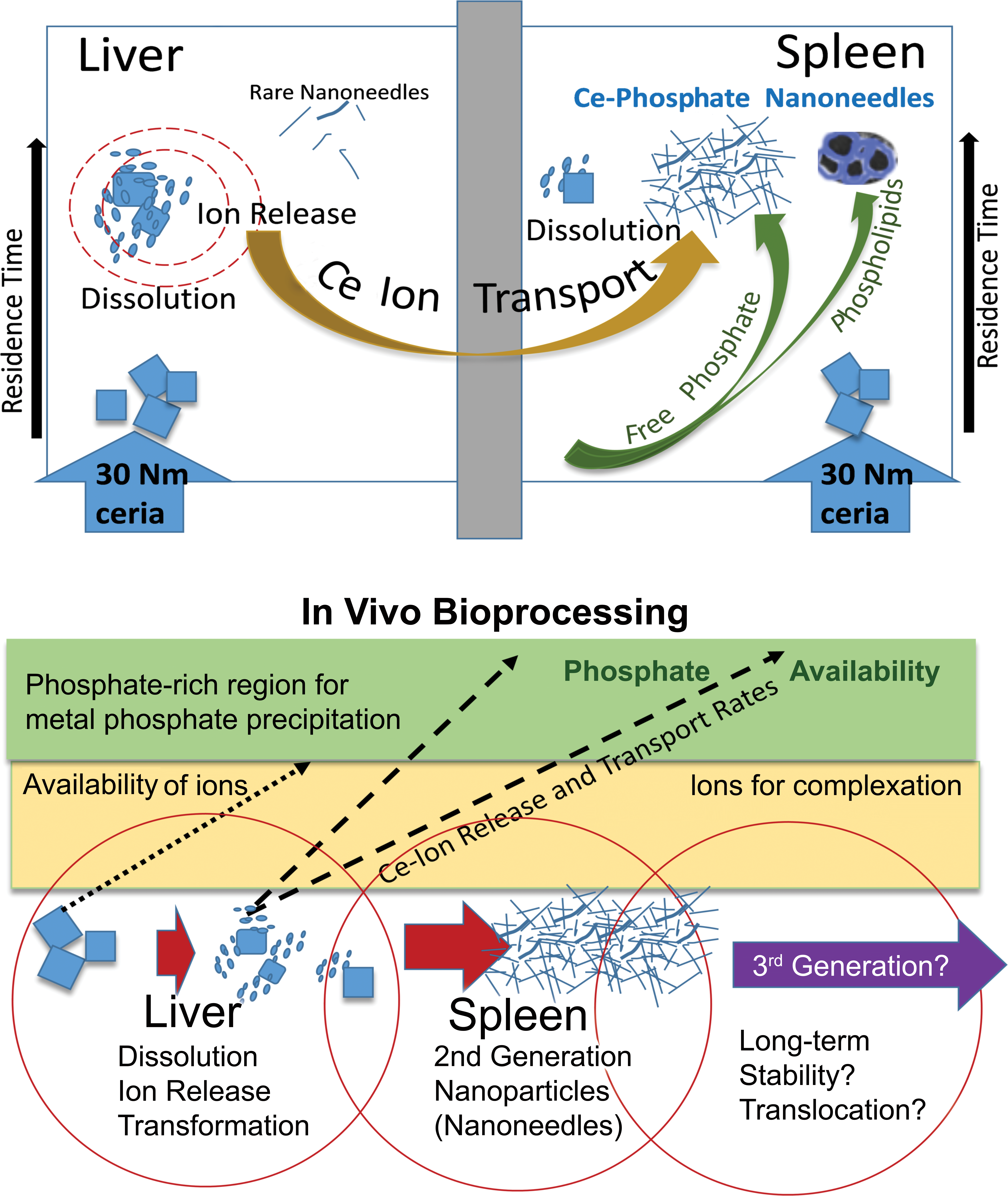
Schematic comparison of nanoceria bioprocessing in liver and spleen.
Total Cerium in the Liver and Spleen 30 and 90 Days after a Single iv Nanoceria Infusion.
Comparing the toxicological response of nanoceria to its ion form, it was demonstrated using Ce-ion instillation into lungs that ion uptake induced a greater initial toxicity than the metal nanoparticles and resulted in a high retention of Ce in the lung (Molina et al. 2014). The high retention of Ce (75–92% at 28 days) in the lung after Ce-ion instillation was unexpected. Follow-up HRTEM and EELS revealed that
Biopersistence of nanoparticles typically refers to the elemental concentrations retained in tissues rather than the actual nanoparticles, their concentration, structural and morphological integrity, chemical composition, redox characteristics, size, or reactive surface area. Elemental concentrations cannot distinguish between solids (nanoparticle concentration) or released ions which can be readily transported, absorbed, or precipitated after dissolution of the primary particles (Figure 6). This limitation/shortfall highlights the importance of analytical HRTEM which can determine the dynamic physiochemical changes of nanoparticles in tissue sections as a function of residence time and for different organs.
Footnotes
Authors’ Note
The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or National Institute for Occupational Safety and Health (NIOSH). Mention of any company name or product does not constitute endorsement by NIOSH.
Author Contribution
Authors contributed to conception or design (UG, RY, AD, LD, KM, MT, MB, JF); data acquisition, analysis, or interpretation (UG, RY, AD, LD, KM, MT, MB, JF); drafting the manuscript (UG, RY); and critically revising the manuscript (UG, RY, AD, LD, KM, MT, MB, JF). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by U.S. Environmental Protection Agency Science to Achieve Results (grant number RD-833772) and the National Institute of General Medical Sciences of the National Institutes of Health under Award Number R01GM109195. Although the research described in this article has been funded wholly or in part by the United States Environmental Protection Agency through STAR Grant RD-833772, it has not been subjected to the Agency’s required peer and policy review and therefore does not necessarily reflect the views of the agency, and no official endorsement should be inferred.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
