Abstract
With the increasing use of and interest in nanoparticles in medicine and technology, the tissue and cell-specific localization of the particles are important considerations when the nanomaterials find their way into biological systems. This brief communication shows the utility of autometallography in determining the location of metal deposition at the light microscopic level. Although primarily focusing on studies of the toxicity and deposition of silver nanoparticles, use of autometallography to localize zinc and other metals at the tissue and subcellular localization is also recognized.
Autometallography (AMG) is an established light microscopic technique used to localize the deposition of certain metals in tissues using either light or transmission electron microscopy. Originally described in 1984 by Gorm Danscher, the method amplifies metals deposited in tissues by the principle that a few atoms, for example, of gold “can initiate a reduction of silver ions to metallic silver when an electron donator, a reducing molecule like hydroquinone, is present. Gold, and later the adhering silver, acts as an electrode that conducts electrons from the reducing molecules to the surface bound silver ions.” 1(p331) The use of AMG is likely to grow dramatically as evaluation of the tissue deposition and toxicity of various nanomaterials is evaluated due to the fact that it allows for cellular localization without the need for electron microscopy. The purpose of this brief communication is to share some of our experiences with the technique to help other investigators maximize their results.
Autometallography methods have been refined over the years for the detection of gold, silver, mercury, and bismuth in various tissues. A specific procedure is required to detect each of the metals. 2 –5 Various investigators used this method to localize silver nanoparticles deposition in rodent tissues after intranasal administration. 6 –9
The general protocol for AMG localization of silver nanoparticles is summarized here. Given the sensitivity of the technique, it is important to adhere to specific cleaning procedures for glassware used in the process. Danscher and Stoltenberg 5 recommend cleaning glassware with “Farmer solution” (9 parts 10% sodium thiosulfate and 1 part 10% potassium ferricyanide). Studies conducted in our laboratory were successful using glass Coplin jars treated for 48 hours prior to each staining run with Farmer solution, followed by extensive rinsing in ultrapure water. Although gelatin-coated slides were recommended, 5 our laboratory has found that slides designed to enhance tissue adherence (eg Fisherbrand Colorfrost Plus precleaned microscope slides (Thermo Scientific, Hanover Park, IL) work well. To stain sections for localization of exogenously administered silver, deparaffinized, rehydrated sections were placed into clean (as mentioned previously) Coplin jars, and AMG developer was added (see Recipes). The jars containing the slides and developer were wrapped with foil and placed into a gently shaking 26°C water bath. The entire water bath was similarly covered with foil to decrease light penetration. After 45 to 90 minutes of incubation, the developing solution was replaced with a 5% sodium thiosulfate solution for 10 minutes, followed by gentle and repeated washing. Test slides can be sequentially removed from the solution in order to determine the optimal incubation time for a particular treatment regimen and tissue. Slides can then be counterstained (eg, with hematoxylin or nuclear fast red) and coverslipped. Autometallography for localization of silver nanoparticles can also be accomplished using commercially available kits. 7,8
Recipes
Cellulose Silver Lactate Developer
The following reagents are combined to prepare the silver lactate developer solution: Carboxymethylcellulose (30 mL of a 2% solution; Sigma-Aldrich catalog C-4888), 5 mL sodium citrate buffer (recipe follows), 0.42 g hydroquinone dissolved in 7.5 mL deionized water (heating and vortexing are necessary for solubilization; mix immediately prior to addition to the solution), and 0.06 g silver lactate dissolved in 7.5 mL deionized water. 5 Add all ingredients to a clean sterile 50-mL tube and mix well prior to adding to slides and developer solution into the Coplin jars. All ingredients are added to a clean sterile 50-ml tube and mixed well prior to adding the developer solution to slides in clean Coplin jars (VWR, Radnor, PA).
Citrate Buffer, pH 3.8
Dissolve 25.5 g citric acid (monohydrate) and 23.5 g trisodium citrate (dehydrate) in distilled water to make 100 mL. Adjust to pH 3.8 with citric acid. Buffer forms an insoluble precipitate after standing for about a week at room temperature or at 4°C, so preparation of large batches is not recommended.
Applications
Rat Silver Nanoparticle Inhalation Study
Autometallography was performed on slides from nanosilver-treated rats (treated by inhalation with various concentrations of silver nanoparticles, with concurrent assessment of pulmonary function and subsequent histological evaluation
9,10
). The treatment groups were as follows:
fresh air controls, low dose (0.6 × 106 particle/cm3, 49 μg/m3), middle dose (1.4 × 106 particle/cm3, 133 μg/m3), and high dose (3.0 × 106 particle/cm3, 515 μg/m3).
The rats were exposed to silver nanoparticles (average diameter 18-19 nm) for 6 h/d, 5 d/wk, for 13 weeks in a whole-body inhalation chamber. Sections from lung, brain, and kidney were stained by AMG to show silver nanoparticle localization. Figure 1 shows the results of AMG staining of the lungs, and Figure 2 shows the results of AMG staining of the kidneys from rats in this study. The lung sections showed progressively more intense staining as the exposure concentration increased, with staining along the conducting airways and gas exchange spaces, as well as in pulmonary macrophages and blood vessel walls (Figure 1). Although significant silver localization in glomeruli was not observed in the kidneys of the rats exposed to the highest concentration of silver nanoparticles, the walls of the small blood vessels around the glomeruli in the renal cortex were the site of the most significant AMG staining (Figure 2). In contrast to mice treated with silver nanoparticles by intranasal instillation (described subsequently), the brains of the rats treated by inhalation did not have detectable silver nanoparticles deposition, following AMG analysis (not shown).
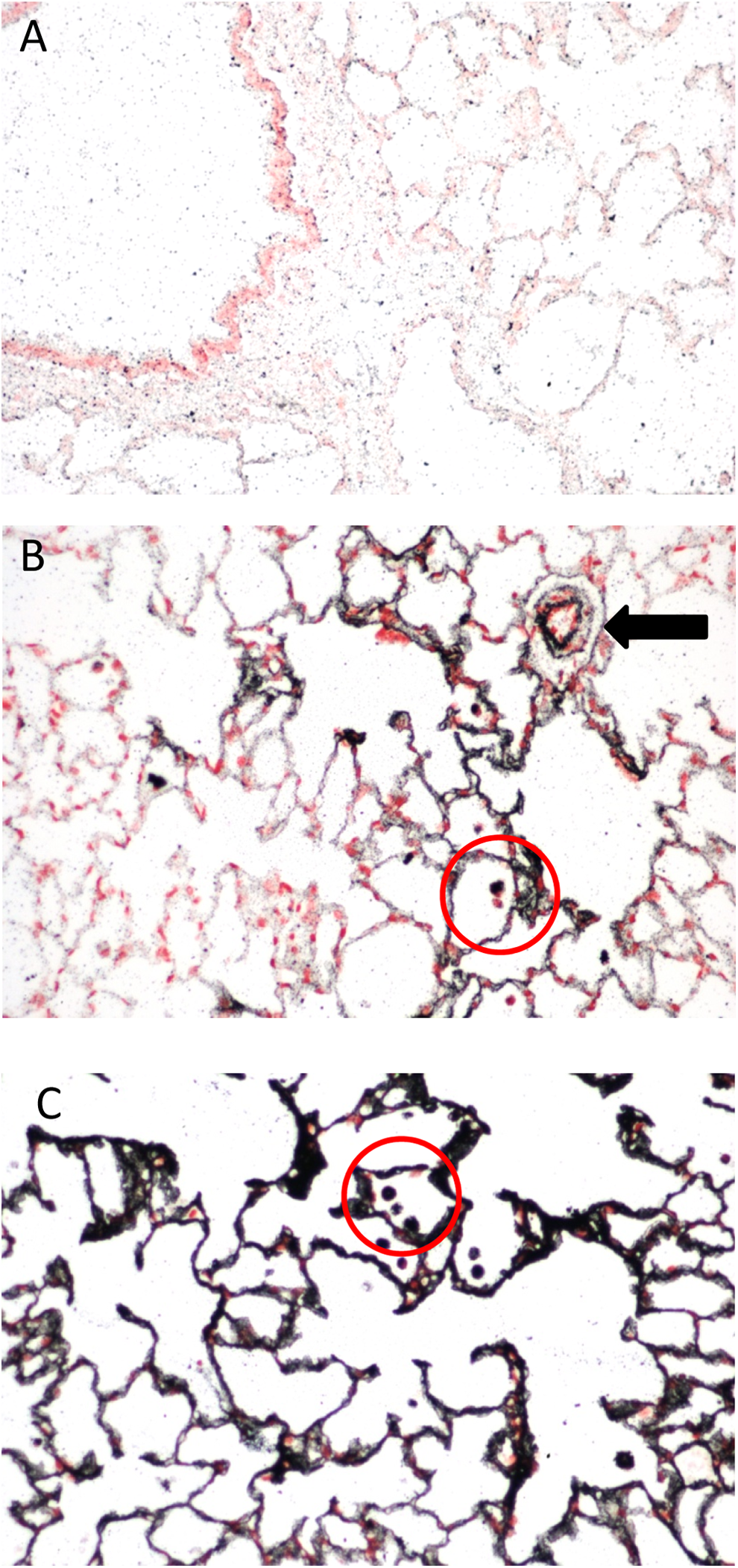
Lung sections stained using the AMG protocol described here. A, Control lung. B, Lung of a male rat following 13 weeks of exposure to silver nanoparticles (average diameter 18-19 nm; 0.6 × 106 particle/cm3, 49 µg/m3). Note AMG reactivity primarily in the alveolar regions, in alveolar macrophages (circle), and in perivascular spaces (arrow). C, Lung of a male rat following 13 weeks of exposure to silver nanoparticles (average diameter 18-19 nm; 3.0 × 106 particle/cm3, 515 µg/m3). Note more intense AMG reactivity throughout the lung and in alveolar macrophages (circle). Sections were counterstained with nuclear fast red and photographed at ×200. AMG indicates autometallography.
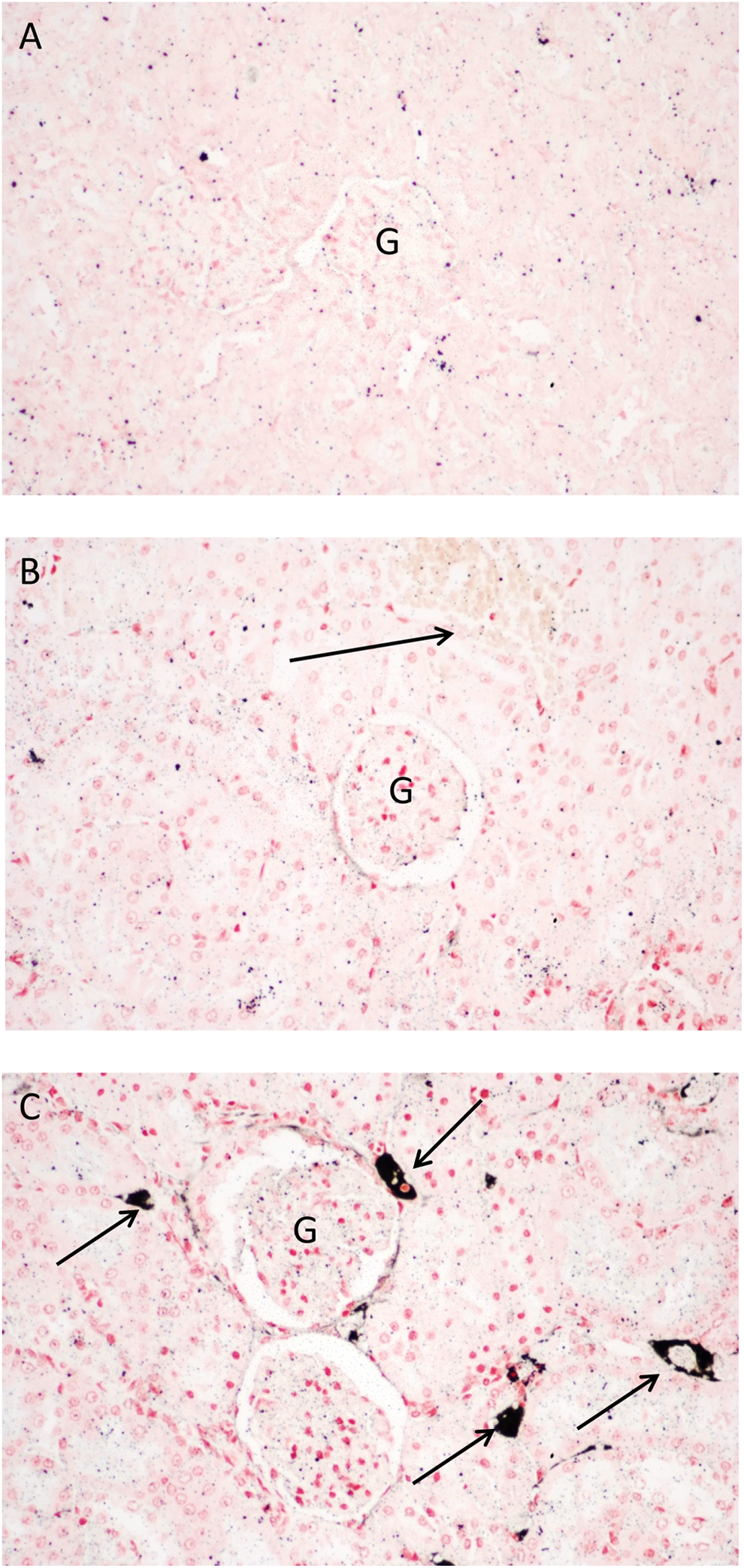
Kidney sections stained using the AMG protocol described here. A, Control kidney. B, Kidney of a male rat following 13 weeks of exposure to silver nanoparticles (average diameter 18-19 nm; 0.6 × 106 particle/cm3, 49 µg/m3). Note lack of AMG reactivity in the perivascular region of this section (arrow). C, Kidney of a male rat following 13 weeks of exposure to silver nanoparticles (average diameter 18-19 nm; 3.0 × 106 particle/cm3, 515 µg/m3). Note intense black AMG reactivity in the perivascular region of the renal cortex (arrows). Sections were counterstained with nuclear fast red and photographed at ×200. AMG indicates autometallography; G, glomeruli.
Intranasal Instillation of Silver Nanoparticles
Intranasal instillation of silver nanoparticles (uncoated, with a nominal size of 25 nm) was accomplished by preparing a suspension of the particles at a concentration of 50 to 62.5 mg/mL in sterile ultrapure water, depending on the body weight of the mice. The suspension was sonicated for 10 seconds and vortexed immediately prior to instillation. Control mice were treated with equivalent volumes of sterile water. The silver nanoparticles suspension was delivered to unanesthetized mice using a P10 pipet and sterile 10 mL pipet tips (ART 10 barrier tips; Thermo Scientific). Dosing volumes ranged from 8 to 10 μL/nostril to give final doses of 10 to 500 mg/kg. Following intranasal administration, silver nanoparticles were localized in multiple organs using the AMG protocol described previously, most notably in spleen, lung, gut-associated lymphoid tissue, liver, and brain. 6,11 In the brain, deposition of silver nanoparticles was most consistently found in the choroid plexus and lateral ventricles following intranasal administration (Figure 3). Despite the presence of silver nanoparticles in the brain, neurobehavioral studies beginning 7 days after 7 consecutive days of dosing male C57BL mice with 50 mg/kg/d of silver nanoparticles, essentially no evidence of neurotoxicity was observed, as evaluated in the Morris water maze and novel object recognition neurobehavioral tests. 11
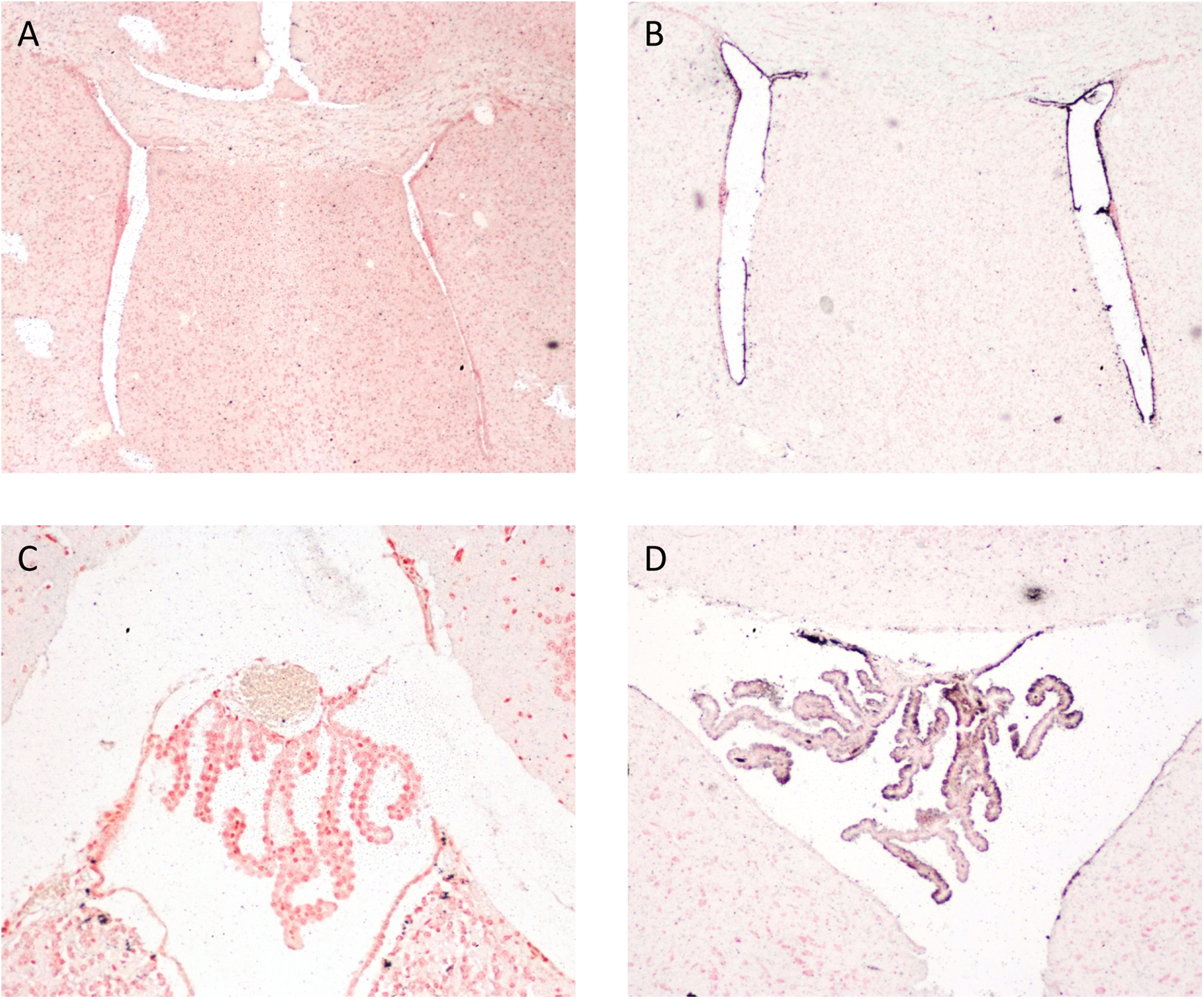
Brain sections from male C57BL mice treated intranasally with 500 mg/kg silver nanoparticles (uncoated, with a nominal size of 25 nm) and euthanized 7 days later. A, Lateral ventricles from a vehicle-treated mouse. B, Lateral ventricles from a silver nanoparticles-treated mouse. Note black AMG reaction product in the cells lining the ventricles (A and B: ×40 magnification). C, Choroid plexus (fourth ventricle) from a vehicle-treated mouse. D, Choroid plexus (fourth ventricle) from a mouse treated with silver nanoparticles. Note black AMG reaction product along the luminal edge of the choroid plexus (×200). Sections were counterstained with nuclear fast red.
Image Capture
Slides were photographed on a Nikon Eclipse E400 microscope, and images were captured using a Nikon D80 digital camera. Images were optimized using the Auto Correct function in Microsoft Office Picture Manager software.
Summary
Although this article has focused on the use of AMG for the detection of silver nanoparticles, modifications of the protocol can be used to detect other metals, such as zinc. For example, using a modified Timm method, Wenzel et al localized the zinc transporter ZnT-3 at both the light and electron microscopic level in mouse and monkey brain. 12,13 The protocol used to localize the transporter, taking advantage of colocalization of the transporter and high levels of zinc in the brain, was similar to that described previously, except that tissues were incubated in 0.4% Na2S prior to incubation in an AMG developer solution. 12,13
In summary, AMG can be a useful technique to localize nanoparticle deposition following systemic administration at the light microscopic level. This technique reduces the need for evaluating metal deposition using transmission electron microscopy, which in turn reduces cost and allows evaluation of larger tissue sections. Methods have also been developed to localize metals within cellular compartments at the electron microscopic level. 13
Footnotes
Author Contributions
Miller, DL and Yu, Il contributed to acquisition, analysis, and interpretation of the data; Genter, MB substantially contributed to conception and design, contributed to acquisition, analysis, and interpretation of the data, and drafted the manuscript. All authors critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded in part by the University of Cincinnati Center for Environmental Genetics (National Institutes of Health [NIH/NIEHS] grant P30-ES006096) and the University of Cincinnati University Research Council; also supported by the Industrial Strategic Technology Development Program (10047829, Development of technologies on testing evaluation method and standardization for nanoproduct and nanomaterial), and funded by the Ministry of Trade, Industry & Energy (MI, Korea).
