Abstract
Maleic acid was formulated in 0.7% saline and injected intravitreally in rabbits in order to evaluate ocular safety and tolerability. Maleic acid was formulated within a narrow pH range (2–3), administered in a fixed volume (100 µl), and concentrations ranged from 0.00 to 2.00 mg/eye (0.00 to 12.30 mM vitreous). Ocular evaluations were conducted at 2, 4, and 8 days post injection. Ocular irritation responses were observed at doses from 0.50 mg/eye (3.07 mM vitreous) to 2.00 mg/eye (12.30 mM vitreous) and included conjunctival redness and scleral swelling. Chemosis was observed at 2.00 mg/eye (12.30 mM vitreous). Funduscopic evaluations revealed enlarged retinal blood vessels and optic disk swelling at doses ≥1.50 mg/eye (9.22 mM vitreous), retinal folds and retinal discoloration at 2.00 mg/eye (12.30 mM vitreous). Histopathologic evaluations on days 4 and 8 post injection revealed retinal degeneration at doses ≥1.0 mg/eye (6.15 mM vitreous), conjunctival inflammation at doses ≥1.5 mg/eye (9.22 mM vitreous), and retinal pigment epithelial hypertrophy, optic nerve demyelination, anterior chamber fluid, and conjunctival fibrosis at 2.00 mg/eye (12.30 mM vitreous) maleic acid. The data suggest that maleic acid formulations at ≥1.00 mg/eye (6.15 mM vitreous) were not suitable for intraocular indications.
Introduction
In the pharmaceutical industry today, effective and safe drugs already established in the company pipeline are frequently developed for more than one indication. This approach has been favored when a disease for which the drug was originally developed is associated with an overlapping pathogenesis of another condition. In some disease conditions where angiogenesis is a major underlying pathogenic mechanism such as cancer metastasis, retinopathies associated with diabetes, or age-related macular degeneration, effective therapies can be developed more rapidly using already established drugs that are focused on key targets. One key therapeutic target against angiogenesis is vascular endothelial growth factor (VEGF) (Ferrara et al. 2004; Figueroa et al. 2009; McGimpsey and Chakravarthy 2010). PF-00337210, a VEGF receptor 2 tyrosine kinase inhibitor (VEGFr2TKi) drug, was initially developed for oncology indications but now has expanded use for the treatment of retinal diseases (Marra et al. 2011). However, anticancer drugs developed and formulated for parenteral administration in cancer patients must first be reformulated and tested for the potential safety risks when the route of administration changes from parenteral to local intravitreal injection for patients with retinal disease (Marra et al. 2011).
Changing the route of drug administration to direct ocular delivery required further formulation considerations including drug depot properties, solubility of the drug in the new formulation, and selection of the buffers and counter-ions in order to avoid loss of drug efficacy by free base precipitation (Marra et al. 2011). A significant formulation consideration to be examined for intravitreal injection was the buffering agent, and maleic acid was one such agent that was evaluated for the intravitreal formulation of PF-00337210. Maleic acid was selected for evaluation as a potential buffering agent and/or in situ salt-forming agent because of its hydrophobic properties under acidic conditions, its hydrophilic properties under neutral conditions, and its tolerability for parenteral use in clinical trials (Marra et al. 2011). However, the intravitreal route of maleic acid administration formulated in saline had never been tested, and its ocular safety and tolerability were unknown.
This article reports the ocular toxicity observed in Dutch Belted rabbits given a single intravitreal injection of a counter-ion maleic acid formulation previously used in a parenteral formulation for anticancer drug administration. The objectives of the work included characterization of the ocular safety profile of a parenteral formulation containing a maleic acid counter-ion excipient after a single intravitreal injection with 2, 4, and 8 days of post treatment observations and assessments. The possible mechanistic pathogenesis of maleic acid-induced ocular toxicity was explored using an in vitro biochemical approach.
Materials and Methods
Maleic acid was formulated as an excipient counter-ion for intravitreal injection with 0.7% sodium chloride over a wide range of concentrations and a narrow range of pH and milliosmolality. To determine potential ocular toxicity and tolerability, maleic acid was tested at a fixed intravitreal injection volume (100 µl) in rabbits in toleration studies.
Animals and Husbandry
In all studies, standard procedures and conditions were applied in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. All procedures involving laboratory animals were reviewed and approved by the Pfizer Inc. Institutional Animal Care and Use Committee associated with the facility. The toleration studies were conducted in 26- to 30-week-old female Dutch-Belted rabbits (2–3 kg body weight) supplied by Myrtle’s Rabbitry, Inc. (Thompson Station, TN).
Maleic Acid
Maleic acid (C4H6O5) or cis-2-butenedioic acid
The concentration of vitreal maleic acid in mg/ml was calculated prior to intravitreal dosing by multiplying the concentration of maleic acid in mg/eye by the rabbit vitreal volume (1.4 ml).
Study Design
Maleic acid formulated as an excipient counter-ion for intravitreal injection with 0.7% sodium chloride was tested in 2 rabbits per treatment condition in toleration studies. Maleic acid was given at a fixed intravitreal injection volume of 100 µl and used over a range of concentrations 0.0–2.00 mg/eye (12.30 mM vitreous), approximate pH (~3.0), and milliosmolality (~300 mOsm). The animals were assessed twice daily (morning and afternoon) for mortality, abnormalities, and signs of pain or distress. Body weights were recorded during the pretest period and at termination on day 4 or day 8. In preparation for intravitreal dosing, animals were lightly anesthetized with xylazine/ketamine (10–20 mg/kg Xylazine and 30–50 mg/kg ketamine). Both eyes were irrigated with ophthalmic 0.5% Betadine Solution® (diluted with sterile saline). After 2 minutes, each eye was rinsed with saline, and three drops of 0.5% Proparacaine Hydrochloride were instilled in the eye. Rabbits were given a single 100 µl intravitreal injection of 0.7% NaCl or a maleic acid formulation in an area 3 mm above the corneal limbus in the superior temporal quadrant of the globe of each eye. Ocular signs of toxicity were determined by using a direct and indirect ophthalmoscope on days 2, 4, and 8 of study. Ocular irritation was scored in accordance with the Organization for Economic Cooperation and Development 1987 guidelines. Ophthalmic examination was conducted with a direct slit lamp and ophthalmoscope. Each eye was grossly examined and graded on a scale for the severity of changes using a modified MacDonald-Shadduck Scoring System. Fundus examinations were performed using an indirect ophthalmoscope in eyes with pupils dilated with phenylephrine (AK-Dilate) and cyclopentolate (AK-pentolate, Akorn Inc., Somerset, NJ). Fundus photography was done utilizing a Carl Zeiss Meditec’s FF450 plus fundus camera (Carl Zeiss, Jena, Germany). At study termination on day 4 or 8 post treatment, eyes were removed and fixed in 6% glutaraldehyde solution and processed to slides for histological examination. For each eye, a horizontal section just above the optic disk and at least five step sections taken at 100 micron intervals were examined starting above the optic disk toward the injection site. In addition, one to three 100 micron step sections through the optic nerve were examined. All sections were stained with hematoxylin and eosin. Additional sections to evaluate changes in nerve fibers and myelin were stained using Bielschowsky silver and luxol fast blue, respectively, or were doubled stained with Holmes silver nitrate and luxol fast blue.
An in vitro assay utilizing the antioxidant glutathione and maleic acid was conducted to examine the potential mechanism for the intraocular toxicity. A nonenzymatic reaction between electrophilic carbon double bonds in maleic acid with the nucleophilic thiol group in glutathione forming a covalent bond in the maleic acid-glutathione conjugate, S-(1,2 Dicarboxyethyl) glutathione (DCE-GS), was assayed in triplicate using 18 mM maleic acid and 3.6 mM glutathione. The formation of the S-(1,2 Dicarboxyethyl) glutathione (DCE-GS) was assessed as a function of time by HPLC-UV detection. Briefly, the reaction was run in a 50 mM potassium phosphate buffer at pH 7.4 for 0.5, 1, 2, 4, or 8 hr at 37°C. At the predetermined time points, the reaction was quenched by the addition of 3× by volume of a 50:50 methanol: acetonitrile mixture, and transferred to a –20°C refrigerator for 2 hr. Samples were subsequently dried under a flow of N2, and the residue was redissolved in 10% w/v Na2CO3 solution and derivatized with 2,4-dinitrofluorobenzene (DNFB).
Results
Ocular Concentrations of Maleic Acid and Ocular Irritation Responses
The various concentrations of maleic acid tested in rabbits expressed in mg/eye, mM vitreous, and mg/ml vitreous concentration and ocular irritation responses as assessed by direct ophthalmoscopy are summarized in Table 1. Conjunctival redness, sclera swelling, and chemosis were observed and scored by Draize scoring. Ocular irritation responses observed in rabbits dosed with saline vehicle and ≥0.50 mg/eye (3.07 mM vitreous) maleic acid included a minimal to moderate conjunctival redness and minimal to mild scleral swelling with red brown discoloration at the site of injection. Minimal to moderate chemosis or eyelid swelling, which lasted only 48 hr, was also seen in rabbits given 2.00 mg/eye (12.30 mM vitreous). In general, the ocular irritation responses were dose-related, and the conjunctival, scleral, and eyelid irritation reactions started to subside to minimal severity by day 6 to 8 post treatment (Table 1).
Summary of maleic acid concentrations injected into the eye and ocular irritation responses.
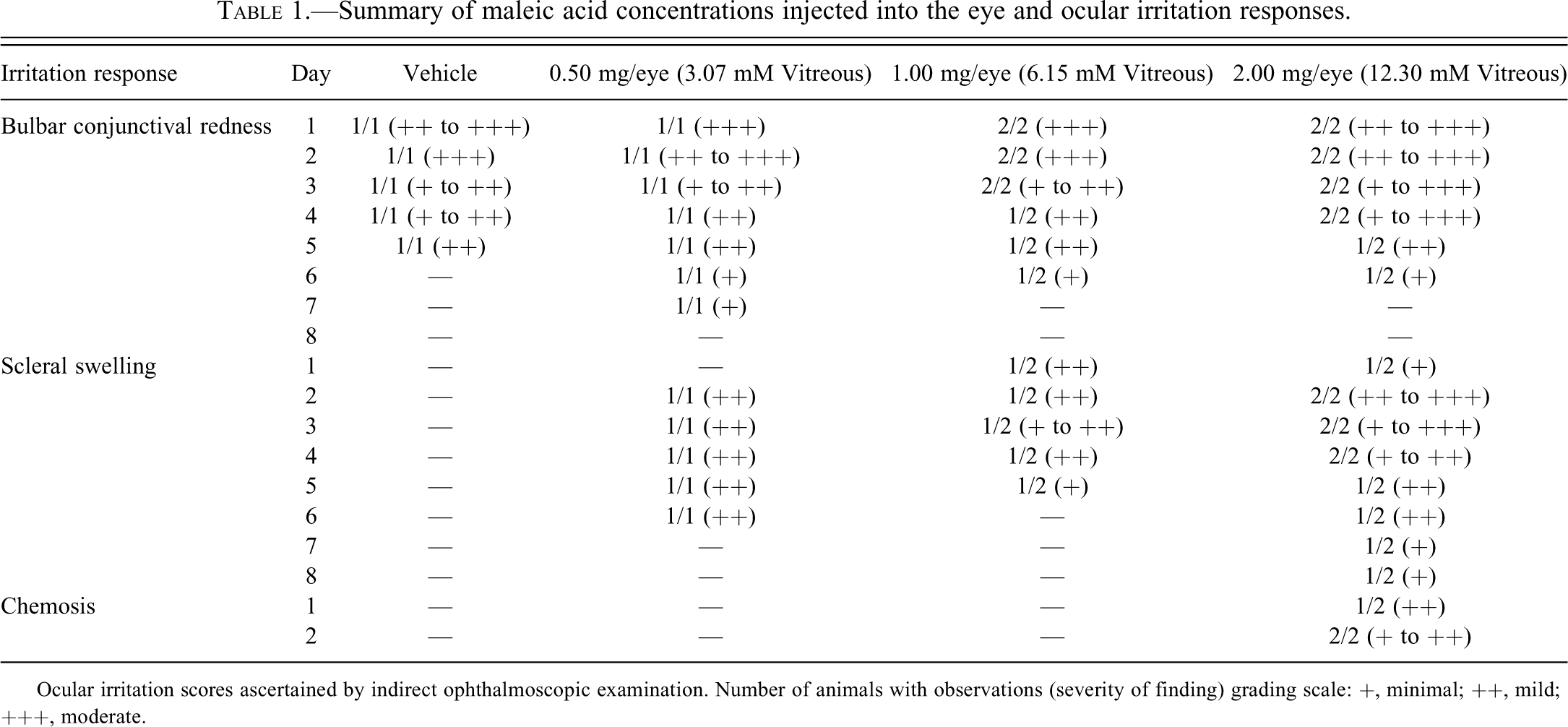
Ocular irritation scores ascertained by indirect ophthalmoscopic examination. Number of animals with observations (severity of finding) grading scale: +, minimal; ++, mild; +++, moderate.
Indirect Ophthalmoscopic Examinations
The spectrum of changes in the retina of rabbits given 2.00 mg/eye (12.30 mM vitreous) maleic acid is illustrated in Figure 1 and listed in Table 2. Fundus photographic images revealed the enlargement and tortuosity of the retinal blood vessels along the inferior temporal quadrants of the medullary rays (Figure 1B, 1C, 1D, and 1E). Blood vessel enlargement was observed in one or both eyes of rabbits injected with ≥1.50 mg/eye (9.22 mM vitreous) maleic acid and persisted through day 8 of the study (Table 2). In more severely affected animals, there was disruption and deterioration of the medullary rays (Figure 1B, 1C, and 1D). Retinal folds and enlargement of tortuous retinal vessels along the inferior temporal aspect of the globe were also observed (Figure 1E). Retinal discoloration (Figure 2F) was observed in the inferior temporal retina of a rabbit given 2.00 mg/eye (12.30 mM vitreous). A minimal to mild swelling of the optic disk or mild papillary edema was also observed at doses ≥1.50 mg/eye (9.22 mM vitreous) (Table 2). In general, retinal changes occurred temporally close to the site of ≥1.50 mg/eye (9.22 mM vitreous) maleic acid injection.
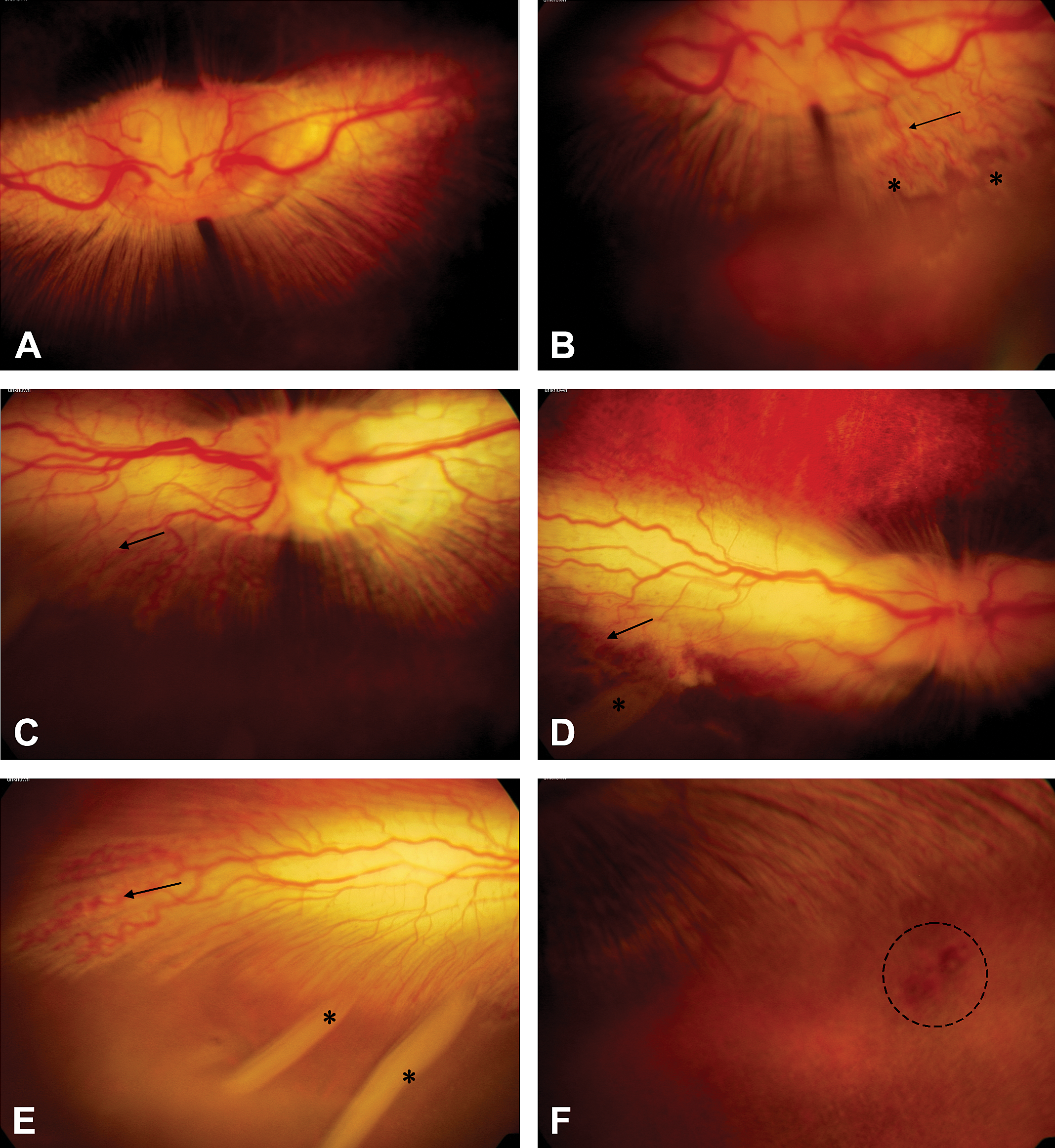
Left eye. Normal rabbit ocular fundus front ophthalmoscopy shown in (A). Note normal retinal vessels, optic disc, and medullary rays, maleic acid predose. (B) Tortuosity of retinal vessels (arrow) along with deterioration of the inferior temporal portion of medullary rays (asterisks) in the same eye 4 days post injection with 2.00 mg/eye (12.30 mM vitreous) maleic acid. (C) Right eye. Rabbit ocular fundus 4 days post injection with 2.00 mg/eye (12.30 mM vitreous) maleic acid. Note the tortuosity of retinal vessels (arrows) in the inferior-temporal portion of the medullary rays. (D) Peripheral aspect of temporal fundus in (C). Note the tortuosity of retinal vessels (arrow) and retinal fold (asterisk). (E) Inferior peripheral aspect of temporal fundus in (D). Note the tortuosity of retinal vessels (arrow) and multiple retina folds (asterisks) in the inferior retina. (F) Left eye. Retinal discoloration (circled area) in the inferior portion of the temporal retina below the medullary rays 8 days post injection with 2.00 mg/eye (12.30 mM vitreous) maleic acid.
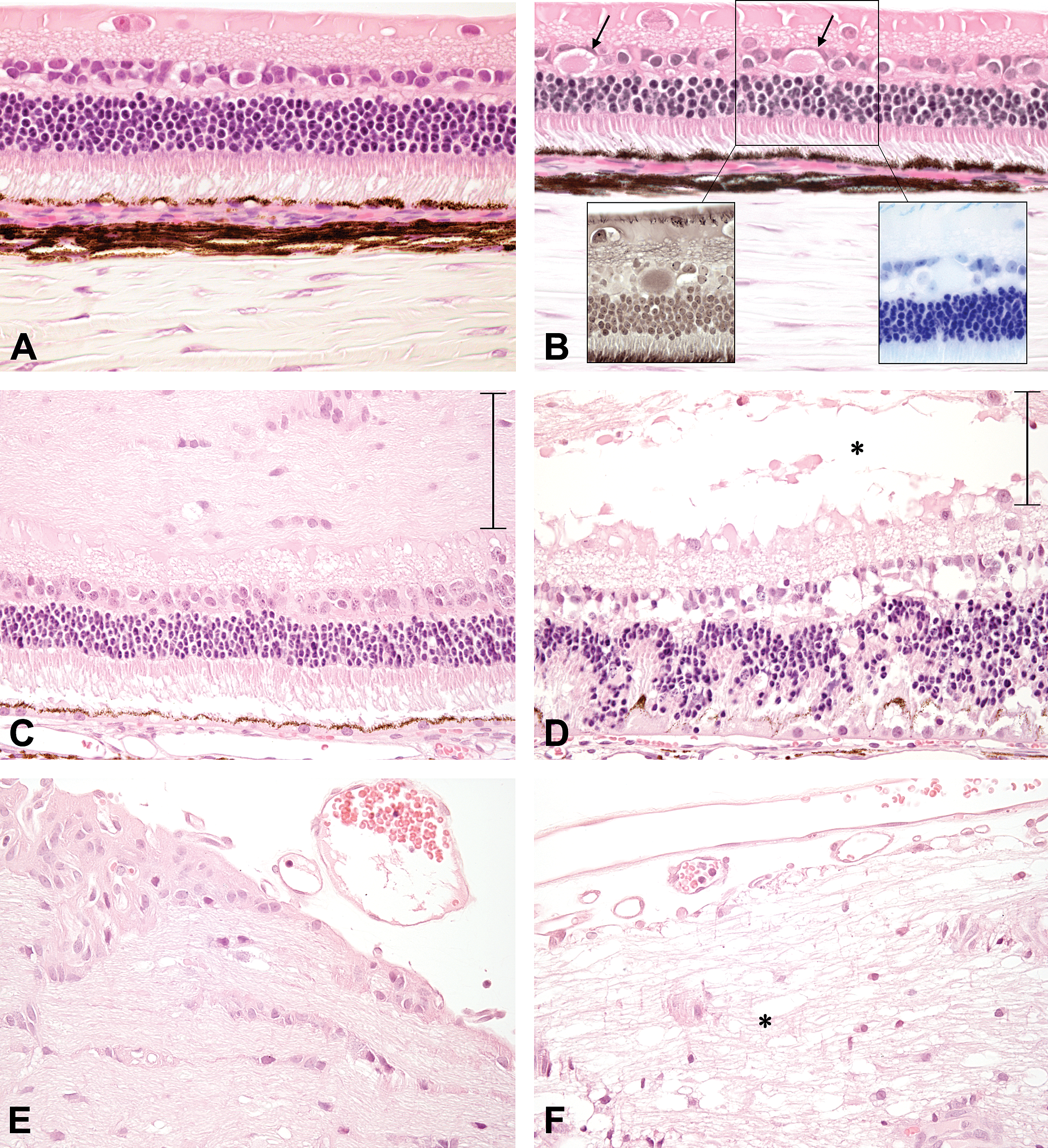
Hematoxylin and eosin staining of the retina from rabbits day 4 post injection. (A) Superior (above the optic nerve) temporal central superior retina from a rabbit given 0.7% NaCl, 600×. (B) Superior temporal central retina from a rabbit given 1.00 mg/eye (6.15 mM vitreous) maleic acid. Note the eosinophilic bodies located within the inner nuclear layer (arrows), 600×. Eosinophilic bodies stained negative for nerve fibers on Bielschowsky silver stain (left inset) and negative for myelin on luxol fast blue (right inset), 600×. (C) Normal medial central retina at the level of the optic nerve in a rabbit given 2.00 mg/eye (12.3 mM vitreous) maleic acid, 400×. Note the thickness of the medullary rays (vertical bar). (D) Demyelination (askerisk) of medullary rays (vertical bar), retinal degeneration and retinal pigmented epithelial hypertrophy in the temporal central retina from the same eye in (C) at the level of the optic nerve, 400×. (E) Normal medial medullary rays at the optic disk from a rabbit given 2.00 mg/eye (12.30 mM vitreous) maleic acid, 400×. (F) Demyelination of temporal medullary rays (asterisk) at the optic disk in the eye listed in (E), 400×.
Summary of maleic acid concentrations injected into the eye, funduscopic observations, and ocular histopathology.
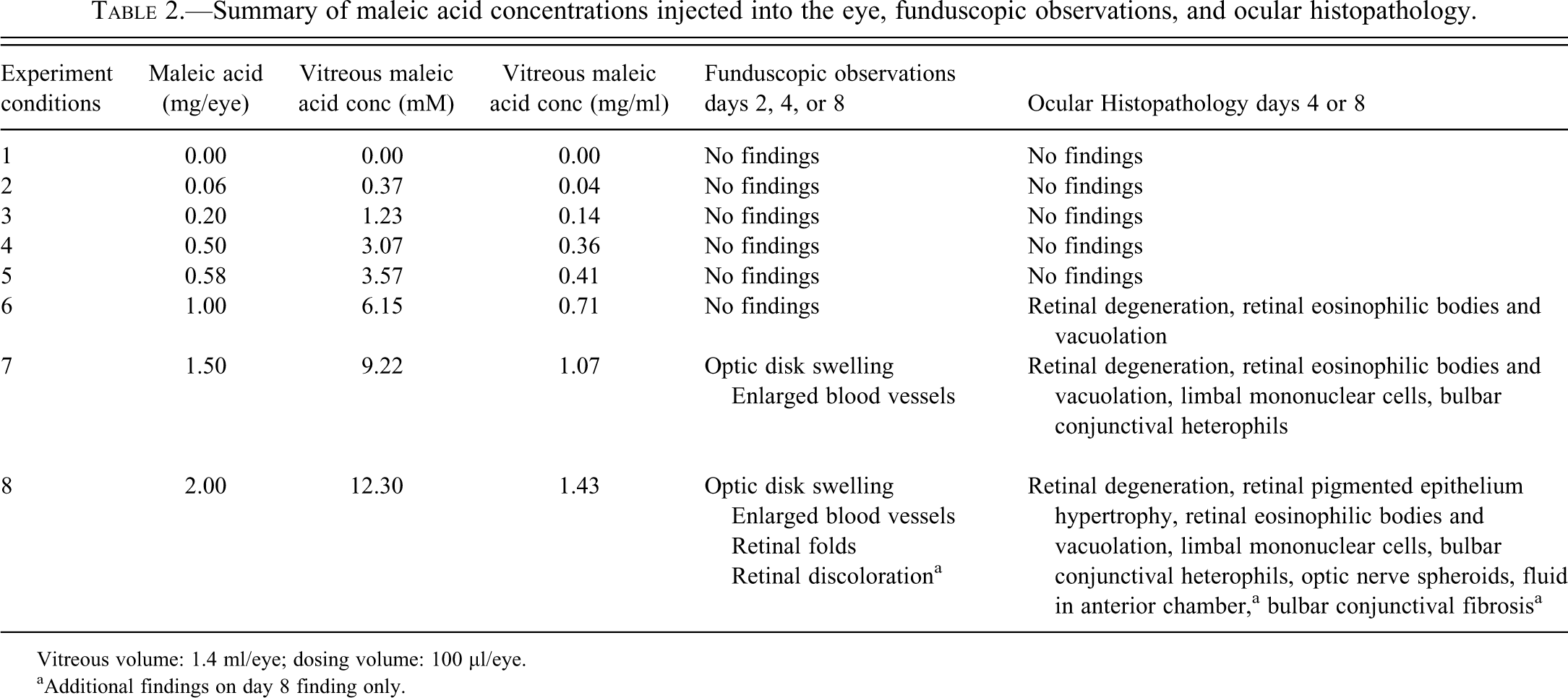
Vitreous volume: 1.4 ml/eye; dosing volume: 100 µl/eye.
aAdditional findings on day 8 finding only.
Histopathologic Evaluations
Microscopic ocular changes are summarized in Table 2 and illustrated in Figures 2 and 3. Changes were observed in the retina at ≥1.00 mg/eye (6.15 mM vitreous), bulbar conjunctiva and limbus at ≥1.50 mg/eye (9.22 mM vitreous), and optic nerve and anterior chamber at 2.00 mg/eye (12.30 mM vitreous) maleic acid on days 4 and 8 post injection. The grading scheme for the inflammatory change in the conjunctiva and degenerative change in the retina and optic nerve ranged from minimal to severe and was based on the percentage of the tissue affected. A grade of minimal corresponded to ≤10%, a grade of mild corresponded to >10% and ≤25%, a grade of moderate corresponded to >25% and ≤50% and a grade of severe corresponded to >50%.
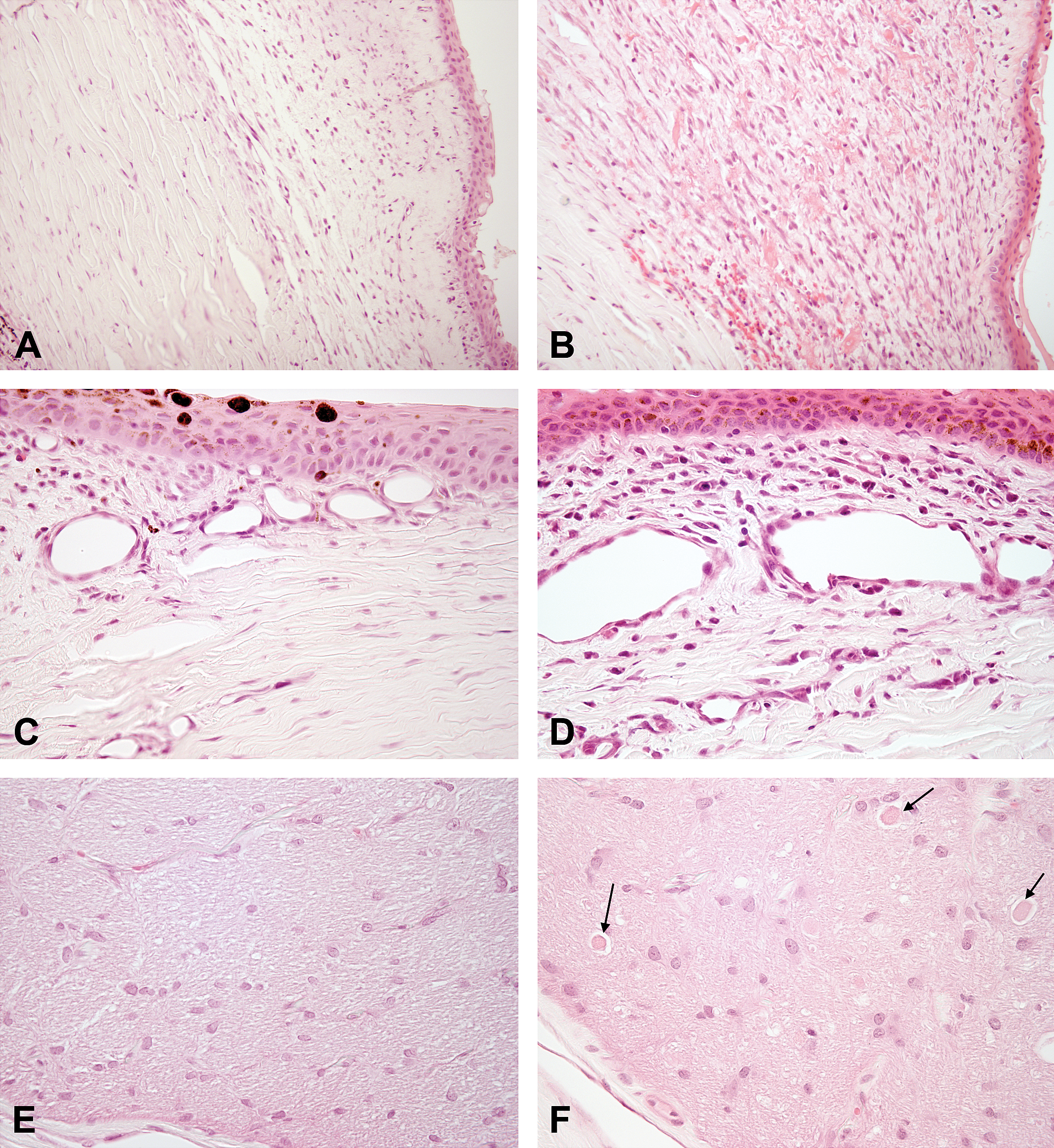
Hematoxylin and eosin staining of the bulbar conjunctiva, limbus, and optic nerve from rabbits 4 days post injection. (A) Bulbar conjunctiva at the injection site from rabbit given 0.7% NaCl. Note the minimal cellular response, 200×. (B) Bulbar conjunctiva at the injection site from a rabbit given 2.00 mg/eye (12.30 mM vitreous) maleic acid. Note the marked swelling of the conjunctiva with edema, hemorrhage, and macrophage infiltration, 200×. (C) Limbus near the injection site of an eye from rabbit given 0.7% NaCl. Note the minimal cellular response, 400×. (D) Limbus near the injection site of an eye given 2.00 mg/eye (12.3 mM vitreous) maleic acid. Note the increased numbers of heterophils and macrophages surrounding limbal vessels, 400×. (E) Normal optic nerve from a rabbit injected with 0.7% NaCl, 400×. (F) Optic nerve with swollen axons (arrows) from rabbit given 2.00 mg/eye (12.30 mM vitreous) maleic acid acid, 400×.
At 4 or 8 days post dose, there were no detectable changes in the retina of rabbits given 0.7% NaCl. Retinal findings in eyes given 0.7% NaCl were similar to predosed eyes (Figure 2A). Similar to 0.7% NaCl, retinal changes were not observed in rabbits given <0.58 mg/eye (3.57 mM vitreous) maleic acid on days 4 or 8 post dose. A dose-related degeneration of the superior (above the optic nerve) temporal central retina was observed in rabbits given ≥1.00 mg/eye (6.15 mM vitreous) maleic acid. Minimal retinal degeneration was observed in rabbits given 1.00 mg/eye (6.15 mM vitreous) and was characterized by the presence of eosinophilic bodies within the inner nuclear layer (INL) (Figure 2B). The nature of these eosinophilic bodies was evaluated for the presence of nerve fibers and myelin. The eosinophilic bodies stained negatively using the Bielschowsky silver (Figure 2B, left inset), on luxol fast blue (LFB) (Figure 2B, right inset), and the Holmes silver nitrate/LFB dual stain (data not shown). A mild to moderate retinal degeneration was observed in rabbits given ≥1.50 mg/eye (9.22 mM vitreous). The moderate degeneration of the temporal peripheral to temporal central retina observed in rabbits given 2.00 mg/eye (12.30 mM vitreous) extended from the site of injection along the medullary rays to the optic nerve (Figure 2D, 2F). The moderate temporal central retinal degeneration was characterized by vacuolation and edema or demyelination of the medullary rays, loss of nuclei in the INL, vacuolation of the inner plexiform layer (IPL), disorganization and loss of nuclei in the ONL, and loss of photoreceptors (Figure 2D). Demyelination of the medullary rays was confirmed on LFB (data not shown). The presence of hypertrophy (cobblestone appearance) of the retinal pigment epithelium (RPE) was consistent with an adaptive response of the RPE to photoreceptor injury and loss (Figure 2D). These changes were restricted to the temporal side of the globe injected with maleic acid because the retina on the medial side was within normal limits (Figure 2C, 2E). There were minimal focal changes in the superior temporal peripheral retina near the ora serrata that included degeneration and scar formation that was consistent with localized needle trauma during the intravitreal injection procedure (data not shown). In general, all changes described in the retina were not reversible by day 8 post injection and correlated with funduscopic observations.
Histopathologic changes on day 4 post dose in the temporal bulbar conjunctiva, limbus, and optic nerve in rabbits given 0.7% NaCl or maleic acid are illustrated in Figure 3 and listed in Table 2. There was a minimal inflammatory response in the bulbar conjunctiva of a rabbit given 0.7% NaCl that consisted of few heterophils and macrophages in conjunctival stroma (Figure 3A). A dose-related mild to moderate inflammatory response in the bulbar conjunctiva was observed in rabbits given ≥1.50 mg/eye (9.22 mM vitreous) maleic acid. The moderate inflammatory response in bulbar conjunctiva was characterized by edema and hemorrhage in the stroma accompanied by moderate numbers of heterophils and macrophages (Figure 3B). A minimal mononuclear cell infiltrate was observed in the limbus of rabbits given 0.7% NaCl (Figure 3C). In contrast, mild limbal heterophil and macrophage infiltrates were observed around vessels near the corneal surface epithelium of rabbits given 2.00 mg/eye (12.30 mM vitreous) (Figure 3D). Histopathologic changes in the limbus and bulbar conjunctiva correlated with the ocular irritation responses. Minimal swelling of axons typically surrounded by a clear space (spheroids) was observed within the optic nerves of rabbits given 2.00 mg/eye (12.30 mM vitreous) (Figure 3F). No adverse ocular changes were evident at 0.58 mg/eye (3.57 mM vitreous) (Table 2), and this concentration was considered to be the no observed adverse effect level (NOAEL).
Rate of Maleic Acid Glutathione Conjugate Formation
The in vitro formation of the covalent bond between the second carbon atom in the carbon-carbon double bond of maleic acid with the sulfur atom in glutathione produced a glutathione-maleic acid conjugate, S-(1,2-Decarboxyethyl) glutathione (DCE-GS) (Figure 4). The reduction of the electrophilic carbon-carbon double bond to a carbon single bond and the removal of hydrogen atom from the thiol group in glutathione and subsequent nucleophilic attack of the sulfur group in glutathione on the second carbon in maleic acid, resulted in a covalent bond between the sulfur and carbon atoms (Figure 4). The concentration of DCE-GS ranged from 0 to 0.70 mM after 8 hr of incubation (Figure 4). Nonenzymatic formation of DCE-GS after the addition of 18.3 mM maleic acid to 3.6 mM glutathione under physiological conditions (pH 7.4, 37°C) occurred at a rate of 0.08 mM/hr in a linear pattern (Figure 4).
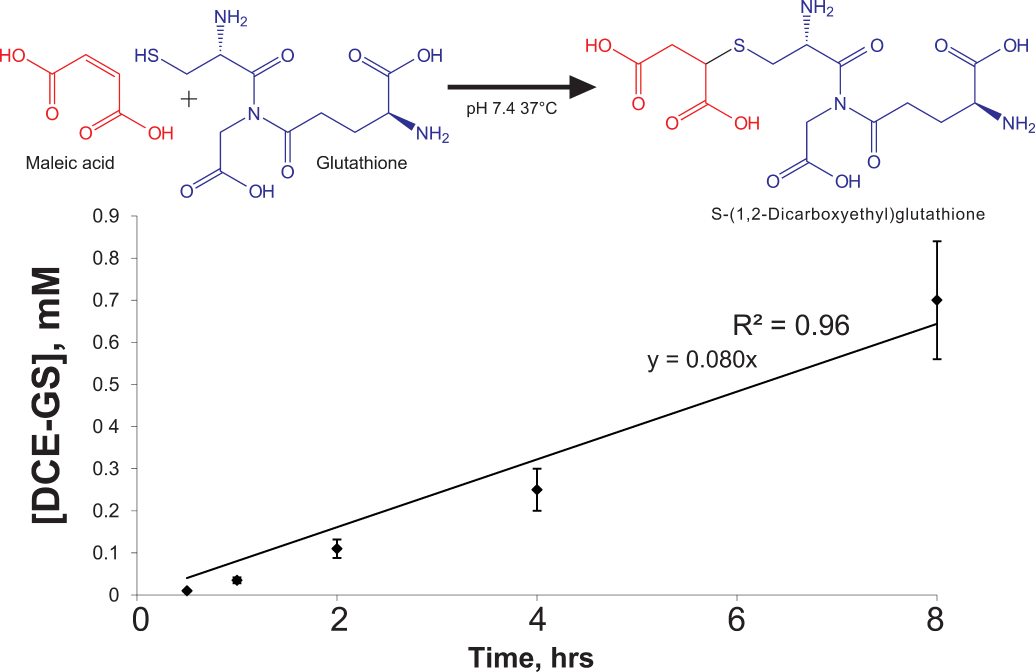
In vitro, nonenzymatic reaction of maleic acid with GSH. The complete reaction mixture consisted of 1 mL volume 50 mM potassium phosphate buffer at pH 7.4 containing 18 mmol and 3.6 mmol of each substrate, maleate and GSH resp. Samples were incubated at 37°C in the dark. DCE-GS was quantitated against a reference standard curve using an HPLC-UV system, following derivatization of standards and samples with DNFB as described in METHODS. DCE-GS is produced at approximately 80 µM/hr. Data are mean ±
Discussion
Maleic acid was not well tolerated intraocularly in rabbits at concentrations ≥1.00 mg/eye (6.15 mM vitreous) because of a dose-related minimal to moderate localized degeneration within the peripheral, central and inferior temporal retina. Clinical signs of conjunctival redness and scleral swelling at ≥0.50 mg/eye (3.07 mM vitreous) maleic acid, papillary edema and enlarged tortuous retinal vessels at ≥1.50 mg/eye (9.22 mM vitreous) and chemosis, retinal discoloration, and retinal folds at 2.00 mg/eye (12.30 mM vitreous) maleic acid were evident. The no observed adverse effect level (NOAEL) in rabbits given maleic acid by intravitreal injection was 0.58 mg/eye (3.57 mM/ vitreous) because the conjunctival redness and swelling was minimal to mild and eventually subsided 8 days after intravitreal injection, and retinal degeneration was not observed.
The mechanism of maleic acid–induced retinal degeneration is unknown. The retinal changes observed in the INL and ONL at 1.00 mg/eye (6.15 mM vitreous) dose level suggested that cells in the INL are affected before the outer segments of the retina in maleic acid mediated toxicity. Administration of maleic acid by intravitreal injection resulted in the presence of eosinophilic bodies in the INL. The mammalian retina has radially aligned groups of neurons ensheathed by a common Müller cell (Reichenbach et al. 1993). Radially aligned groups of neurons are organized into columnar units comprised of Müller cells, bipolar cells, amacrine cells and cell bodies of rods or into extracolumnar units comprised of ganglion cells, another population of amacrine cells horizontal cells, and cell bodies of cones (Reichenbach et al. 1993). In the rabbit retina, one Müller cell forms a central framework where approximately 11 rod photoreceptors, two bipolar cells, and one amacrine cell are organized into the columnar unit (Reichenbach et al. 1993). Cells most likely affected initially by intravitreal maleic acid administration are the cells organized in the columnar units. Thus, the eosinophilic cell bodies in the INL may represent altered function of cells (Müller, bipolar, or amacrine) in the INL that could ultimately lead to irreversible photoreceptor injury. The origin and nature of the eosinophilic bodies remain uncertain as they were negative using a Bielschowsky silver stain for nerve fibers, LFB stain for myelin (Figure 2B insets), or with Holmes silver nitrate/LFB dual stain for nerve fibers and myelin, respectively (data not shown).
Intravitreal administration of maleic acid with subsequent formation of eosinophilic bodies in the INL may initially disrupt the functions of the Müller cells and lead to retinal injury. Müller cells are the predominant glial cell of the vertebrate retina and constitute the only astrocytic population in the non-vascularized (merangiotic) rabbit retina (Reichenbach et al. 1993). Müller cells form a seal between retinal tissue and the vitreous body by spanning the neural retina with their end feet along the inner limiting membrane and their microvilli along the outer limiting membrane (Wolburg and Berg 1987). The functions of Müller cells are diverse and similar to those of astrocytes, oligodendrocytes, and ependymal cells located in other central nervous system regions in that they express numerous voltage-gated channels and neurotransmitter receptors, recognize a variety of neuronal signals, and trigger cell depolarization and intracellular calcium (Ca++) waves (Newman and Reichenbach 1996). Müller cells regulate neuronal activity by altering the extracellular concentrations of neuroactive substances via potassium (K+) spatial buffering currents and possessing high-affinity carriers for glutamate and gamma-amino-butryic-acid (GABA) (Newman and Reichenbach 1996). Disturbance of K+ conductance in Müller cells has been implicated in retinal degeneration and retinal detachment in rabbit models (Francke et al. 2005). An important role in pH regulation is suggested by the expression of Na+HCO3 − cotransporter and carbonic anhydrase on Müller cells (Newman and Reichenbach 1996). Retinal neurons are nourished by the glycogen stores of Müller cells (Newman and Reichenbach 1996). Protection of retinal neurons from oxidative stress is likely mediated in part by Müller cells because of the presence of high levels of reduced glutathione (GSH) in rabbit Müller cells and other inner retinal cells (Pow and Crook 1995; Huster et al. 2000).
The reduced form of the tripeptide glutathione (GSH) is one of several low-molecular-weight antioxidants located within the eye to protect the organ against reactive oxygen species (ROS) produced under conditions of oxidative stress (Rose et al. 1998). GSH is composed of glycine, cysteine, and glutamic acid and synthesized in two steps by γ-glutamylcysteine synthetase (γ-GCL) followed by glutathione synthetase, with each step requiring energy from adenosine triphosphate (ATP) with subsequent hydrolysis to adenosine diphosphate (ADP) and inorganic phosphate (Lehman-McKeeman 2008). Within the rabbit eye the vitreous humor, retinal astrocytes within the myelinated inner nerve fiber layer, Müller cells and horizontal cells in the INL, and RPE contained abundant detectable levels of GSH, while outer photoreceptor segments were GSH-deficient (Pow and Crook 1995; Rose et al. 1997). Cytoprotective oxidation of reduced GSH with subsequent formation of glutathione conjugates can occur through enzymatic activity of glutathione transferases (GSTs) or through nonenzymatic reactions (Morgan and Friedman 1938a, 1938b; Lehman-McKeeman 2008). Substrates of insufficient electrophilic character for direct conjugation to GSH must first be biotransformed by the enzymatic activity of GST to an electrophilic metabolite prior to conjugation to GSH. After the formation of the electrophilic metabolite, conjugation takes place by deprotonation of GSH to GS− by an active site tyrosine or serine in GST and leads to the formation of a covalent bond between GS− and the free radical, nitrenium ions or carbonium ions. Substrates that are sufficiently electrophilic due to the presence of double bonds or a strained ring system can be directly conjugated to GSH in the absence of GSTs (Lehman-McKeeman 2008). The presence of the carbon double bond along with two carboxy groups in maleic acid was shown to be sufficiently electrophilic for direct nonenzymatic conjugation to the thiol group in GSH (Morgan and Friedman 1938a). Thus, it is likely that depletion of GSH stores from the vitreous humor and retina in the rabbit eyes following intravitreal injection of ≥1.00 mg/eye (6.15 mM vitreous) maleic acid with the formation of glutathione-maleic acid conjugates either by enzymatic or nonenzymatic reactions led to overproduction of free radicals with subsequent injury of the highly sensitive GSH deficient photoreceptors.
The ability to detect the toxic effects of maleic acid on the retina may be a function of time and may depend on the level of glutathione depletion because the normal metabolic supply of glutatione in ocular tissues can be depleted between 4 to 8 hr (Gukasyan et al. 2002). Funduscopic retinal changes (papillary edema) in rabbits given 2.00 mg/eye (12.30 mM vitreous) maleic acid by intravitreal injection were first observed 2 days after intravitreal injection. The retinal folds correlated with the histopathology findings of retinal degeneration and RPE hypertrophy on day 4 post injection. The deterioration of the medullary rays and tortuosity of blood vessels observed on the fundus correlated with histopathologic findings of demyelination of medullary arrays with degeneration of underlying retina, RPE hypertrophy, and congested blood vessels, respectively. The red discoloration of the retina observed by funduscopy correlated with histopathologic findings of retinal degeneration and RPE hypertrophy on day 8. The relatively low levels (0.5–0.8 mM) of glutathione in the rabbit vitreous humor (Rose et al. 1997) were likely depleted nonenzymatically within 8 hr post-maleic acid treatment given the nonenzymatic conversion of 0.08 mM /hour for maleic acid conjugation. When photoreceptors are under conditions of stress (i.e., glutathione depletion), the rate of renewal of the outer segments in rods and cones cannot keep up with oxidizing chemical reactions (Winkler 2008). Therefore, intracellular glutathione concentrations (5–10 mM) (Lehman-McKeeman 2008) within Müller cells, horizontal cells, and RPE may have been depleted to a level where the exquisitely sensitive photoreceptors are stressed to a point that the renewal of outer segments in rods and cones photoreceptors was not capable of overcoming oxidizing toxic effects of maleic acid or endogenous ROS.
The tortuosity of retinal vessels and optic nerve changes observed in rabbits following maleic acid treatment may be related to the retinal vascular structure in this species. The rabbit meroangiotic retina consists of a horizontal band of vessels that emanate medially and laterally from each side of the optic disk and branch in a side branch manner (Samuelson 2007). There are four capillary areas located within the horizontal band; superficial net capillaries, deep capillaries of the nerve fiber layer, peripheral capillaries, and peripapillary capillaries (Samuelson 2007). The horizontal band and capillaries are confluent with the myelinated nerve fiber layer (medullary rays). Blood vessel changes were restricted to the same side as the injection site and involved the deep capillaries of the nerve fiber layer and peripheral capillaries along inferior temporal retina. The changes were likely restricted to these areas because the force of gravity could create an increased intravitreal maleic acid concentration gradient along the more inferior aspect (at and below the equator) of the eye. Tortuous blood vessels within the rabbit retina may also be explained in part by the toxic effect of maleic acid on the medullary rays (demyelination) because the vasculature is intimately associated with the nerve fiber layer of the medullary rays.
The demyelination of the medullary rays may also explain the changes observed in the optic nerves. Because the afferent tracts of the optic nerve originate in the nerve fiber layer of the retina, the demyelination change observed in the medullary rays at 2.00 mg/eye (12.30 mM vitreous) maleic acid dose level was probably extensive enough to be observed in the axonal tracts of the optic nerve as swollen axons (spheroid formation). Axonal spheroid formation as a result of an accumulation of degenerative organelles was consistent with a pattern of distal axonopathy and could be best explained by a disturbance of optic nerve antero-grade axonal transport incited by direct damage of the myelinated INL by maleic acid.
Leakage of maleic acid out on to the ocular conjunctival limbal surface after the intravitreal injection likely incited local inflammatory changes in the conjunctiva and limbus. Because maleic acid was reported to induce local contact irritation when applied to rabbit corneas (Grant 1986), it is feasible that leakage/back flush of maleic acid on to fibroblasts within the conjunctiva and limbus likely resulted in local inflammatory changes.
In this work, we described the ocular toxicity induced by intravitreal maleic acid administration in rabbits. The ocular effects were considered dose-related, but the mechanism(s) involved are still uncertain. Given the importance of antioxidants in protecting the metabolically active photoreceptors, the toxic effects of maleic acid may be due to compromise of a number of antioxidant defense systems, including glutathione and/or outer segment renewal. These results underscore the importance of assessing new formulations of excipients for potential ocular toxicities that were previously shown to be safe in intravenous formulations.
Footnotes
Acknowledgments
Special thanks to individuals who directly contributed to this work: Winston Evering for review of the manuscript and helpful discussions; Carlos Esparanza, Jessica Frey, and Anthony Wong for histology assistance; and Constance Benedict for graphic arts assistance.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article: The work was supported by funding by Pfizer Inc.
