Abstract
Minipigs are an emerging nonrodent alternative for ocular toxicology owing to anatomical similarities in the minipig eyes when compared to humans. Ocular structures and components from Göttingen minipigs were characterized and compared to species commonly used in toxicology. Ocular reference data from Göttingen minipig including intraocular pressure, vitreous electrolyte and thiol concentration, and electroretinography (ERG) data are essential to model characterization and data interpretation during drug safety assessments. Intravitreal positive control agents including gentamicin, indocyanine green, and glycine were used to demonstrate ERG alterations caused by retinal cell toxicity, light transmission obstruction, or neurotransmission interferences, respectively. Electrolyte concentrations of the aqueous and vitreous humors from Göttingen minipigs were similar to other species including humans. The reference data presented herein supports the use of the Göttingen minipig as an alternate nonrodent species in ocular toxicology.
Introduction
Refinement and characterization of animal models for ocular toxicology are crucial in drug development with implications on testing strategies and study designs. Rabbits and nonhuman primates (NHP) have been extensively used partly because of their relevant ocular dimensions, and in the case of NHPs, the physiological similarities with the human eye. Although rabbit eyes are larger and facilitate evaluation of potential changes, their anatomy and cellular structures present a number of differences from that of humans as discussed in the current article. On the other hand, the use of primates in biomedical research raises ethical concerns. 1,2
Minipigs represent another alternative nonrodent species for preclinical ocular toxicological assessments based on anatomical and physiological similarities to humans. 3 Ocular structure differences between species are well characterized in the literature. Table 1 summarizes anatomical characteristics of ocular structures across standard preclinical species as presented by Weir and Collins, 4 which may bare implications for species selection. As an animal model, minipigs possess potential advantages as an alternative nonrodent species compared to NHPs including animal availability, reduced biosecurity, and reduced maintenance costs. In addition, the use of the minipig in preclinical research is confronted with fewer ethical challenges compared to NHPs.
Species Comparisons of Ocular Characteristics.

a Corneal structures.
The scientific community has actively developed applications using the minipig as an animal model for safety assessment in ophthalmic therapies, medical devices, surgical procedures, and pathogenesis of ocular diseases. For example, a retinal hypoxia model was successfully induced in the minipig using microsphere embolization. Electroretinogram (ERG) evaluation in minipigs showed a reduction in the amplitudes of the scotopic and photopic b-wave, photopic a-wave, and oscillatory potentials following embolization. 5 Minipigs have also been used as a suitable model for glaucoma research. 6
Noteworthy temperature and pH differences were noted for vitreous and aqueous humor measurements in rabbits, NHPs, and minipigs when comparing different locations within the ocular globe. Interestingly, ocular pH appeared to be lower in minipigs when compared to NHPs and rabbits. 7
The Göttingen minipig is an available strain and one of the most commonly used porcine models in preclinical research as outlined by the RETHINK project. 2,8 Although minipigs are a commonly used preclinical model and included in the Organisation for Economic Co-operation and Development (OECD) alternative species guideline No. 409, there are nonetheless some challenges in their use. 2 There is a lack of robust historical data, and their rapid growth can limit handling and place stress on an otherwise limited and costly drug supply.
The objective of the current article was to evaluate the scientific literature and to present data for electroretinography, ocular electrolytes, and vitreous humor thiol content in the Göttingen minipig and other common preclinical species. Technical considerations associated with these assessments are also presented.
Materials and Methods
Statement on Use and Care of Animals
For all investigations, care and use of animal procedures were conducted in accordance with principles outlined in the current Guide to the Care and Use of Experimental Animals published by the Canadian Council on Animal Care and the Guide for the Care and Use of Laboratory Animals published by the Institute of Laboratory Animal Resources. Citoxlab North America’s facility is Association for Assessment and Accredidation for Laboratory Animal Care (AAALAC) accredited, and the procedures were reviewed and approved by the Institutional Animal Care and Use Committee prior to conduct. All procedures were conducted per Standard Operating Procedures in place at the time of conduct.
Ocular Parameters and Intraocular Pressure Measurements
Following euthanasia, the eyes were enucleated, trimmed, and weighed. Aqueous and vitreous humors were collected with a standard sterile needle and syringe and weighed in a pretared tube. Lense was also dissected and weighed. Following the removal of vitreous humor, the remaining tissue was weighed, and vitreous humor weighed was calculated as follows:
Vitreous weight = ocular globe weight − (aqueous humor + lens + remaining tissue weight). (1)
Intraocular pressure (mm Hg) was measured in vivo with a Mentor Tono-Pen XL tonometer. After calibration and bilateral instillation of Alcaine 0.5% to the corneal surface, at least 3 measurements were collected and averaged. Readings >10% of the mean were not used.
Indirect Ophthalmoscopy (Funduscopic)
Prior to fundus imaging, animals were anesthetized with zolazepam (minipigs and dogs) or ketamine and xylazine (primates, rats, and rabbits), positioned, and both eyes received 0.5% tropicamide as a mydriatic. Fundus images were captured using Kowa RC-2 fundus camera with photographic film.
Electrolyte and Thiol Content
Aqueous humor and vitreous humor were collected from 13 Gottingen minipigs (n = 26 eyes), 10 cynomolgus monkeys (n = 20 eyes), 9 beagle dogs (n = 18 eyes), 7 New Zealand White rabbits (n = 14 eyes), and 25 Sprague-Dawley rats (vitreous only). Intra-animal age and body weight ranges were comparable. Prior to collection, animals were deeply anesthetized, and all eyes received 0.5% proparacaine topically, followed by a 10-mL flush with physiological saline. Vitreous humor (targeted 0.2 mL) was then collected using a standard sterile 5-mL syringe with a 25-G needle, and aqueous humor (targeted 0.2 mL) was collected using a standard sterile 1-mL syringe with a 30-G needle. Following collection, the eyes were treated topically with 0.3% Ciloxan antibiotic and a bland ophthalmic lubricant was applied bilaterally to protect the corneas during recovery. Aqueous and vitreous humors were transferred into plain tubes and placed on wet ice pending analysis. Due to their viscosity, vitreous samples were diluted with sterile water (1:2) or as needed. Electrolytes were quantified in aqueous and vitreous humors by a Cobas 6000 module c501 (Roche Diagnostics, Ltd.) set to plasma/serum mode. 9 All parameters were expressed in mmol/L. Samples that were contaminated with blood were not considered for the analysis.
Vitreous humor from minipigs was also collected for thiol analysis following the same protocol as described earlier. Immediately after collection, a 10-µL volume in triplicate was mixed with a working solution of Measure-IT (Molecular Probes Inc, Eugene, Oregon). The plate was protected from light (dark) pending analysis. Samples were mixed with the working solution immediately after collection and then analyzed within 20 minutes of collection. Fluorescence quantification was performed using a Spectra Max plate reader (Molecular Devices, LLC) set at excitation/emission 494/517 nm and analyzed by SoftMax Pro GXP (Molecular Devices, LLC).
Electroretinograms
Electroretinograms were recorded using the EPIC-4000 (LKC Technologies, West Midlands, United Kingdom) from all animals while under isoflurane anesthesia following a dark adaptation period of at least 30 minutes prior to evaluation. Three different test items were administered as unilateral intravitreal injections into the posterior chamber at a dose volume of 100 µL. Gentamicin (5 mg/eye, n = 3), indocyanine green (ICG; 0.25 mg/eye, n = 4), and glycine (5 mg/eye, n = 3) were used as positive controls inducing retinal toxicity, light transmission interferences, and neurotransmission inhibition, respectively. The contralateral eye served as an untreated control.
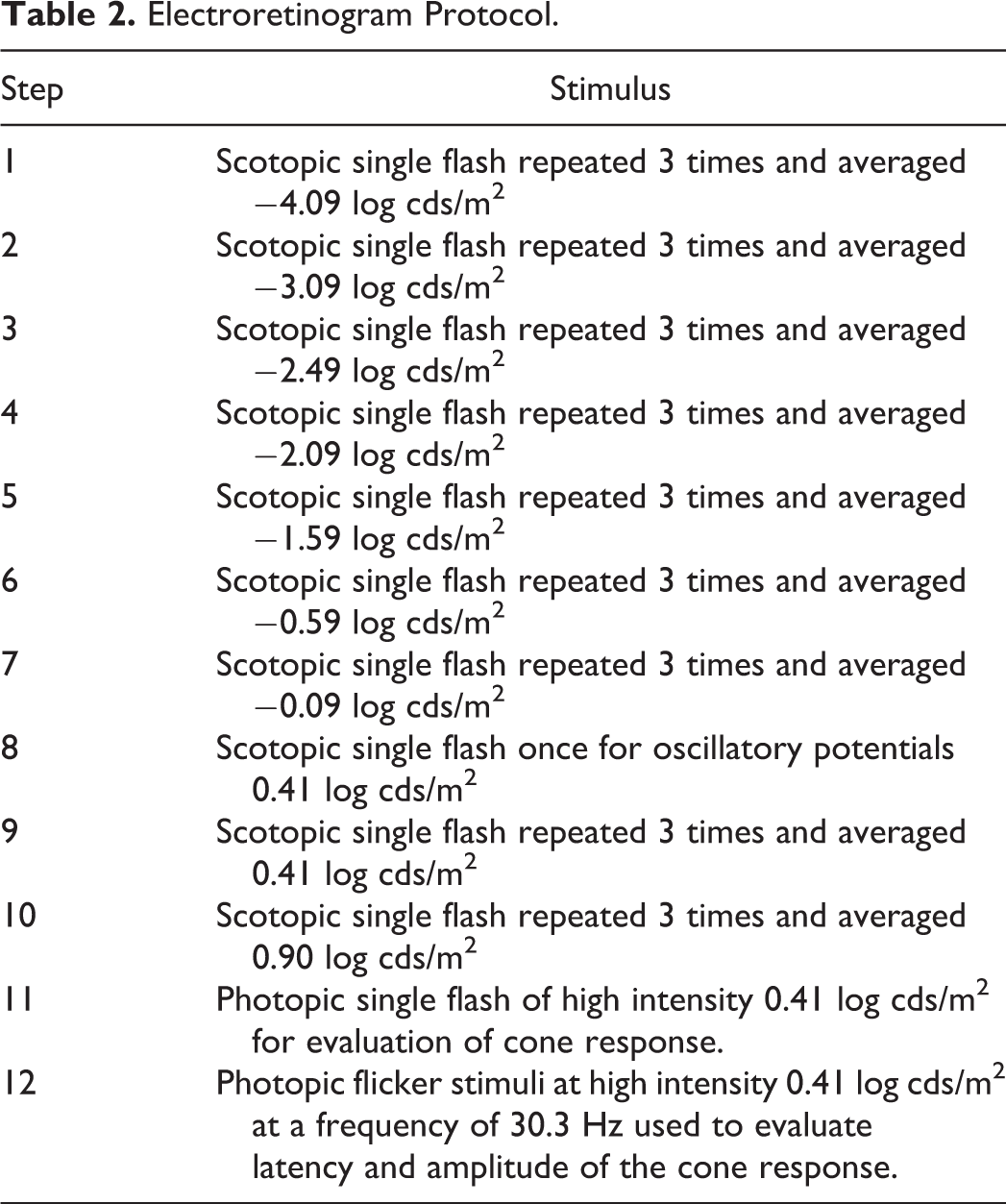
A mydriatic agent (Mydriacyl) was administered to both eyes at least 30 minutes before ERG monitoring. A topical ophthalmic anesthetic (Alcaine) was applied to both corneas. The animals were instrumented with corneal electrodes (ERG-Jet), which were prewetted with 0.5% methylcellulose to ensure corneal hydration and electrical conduction. The ground electrode was placed approximately at Cz (mid-point on top of the cranium), and reference electrodes were placed under each lower eyelid. The animal’s head was then centered into the Ganzfeld dome stimulator. The ERG recording commenced following test item administration at 0.5 to 1 hour postdose for gentamycin; 2.5 to 3.5 hours postdose for ICG, and 1.5 to 2 hours postdose for glycine. The ERG protocol included the steps shown in Table 2.
A prestimulus baseline of 20 milliseconds was recorded with a total sweep length of 256 milliseconds. No prestimulus baseline was included for the photopic flicker stimuli (step 12).
Steps 1 to 7, 9, and 10 were used to evaluate scotopic luminance response and oscillatory potentials. A minimum of 10 seconds was allowed between each flash for steps 1 to 7, 9, and 10 to allow the retina to recover its sensitivity. Following step 10, animals were subjected to light adaptation, using the Ganzfeld background light, for at least 5 minutes.
Electroretinogram Analysis
The ERG waveforms were transformed with the Fast Fourier method to determine the frequency components of waveform data, which were then used to qualitatively assess the presence of electromagnetic interferences (60 Hz and harmonics). The ERG waveforms were averaged by the system for each light intensity from −4.09 log cds/m2 to 0.90 log cds/m2 (except for step 8 which is a single flash). Analysis of ERGs included measurement of amplitudes and latency of a-waves and b-waves for the ERG waveform average of step 4 for low intensity and step 9 for high intensity, respectively. The maximal b-wave amplitude at the saturation point of steps 1 to 10 was determined (Vmax). Retinal sensitivity (Log
Electroretinogram Protocol.
Statistical Analysis
Numerical values were expressed as mean ± standard error of the mean. Statistical significance was determined using an analysis of variance test followed by a Bonferroni correction for multiple comparisons. For each test item, data were obtained from a minimum of at least 3 animals. Charts and figures were prepared with Microsoft Excel and Power Point.
Results
Ocular Parameters and Electrolyte Characterization
Table 3 summarizes the ocular structure weights, tonometry, and thiol concentration in the vitreous humor of 7 minipigs (n = 14 eyes) of approximately equal age and body weight. Gender combined values are presented.
Ocular Parameters in Minipigs.a
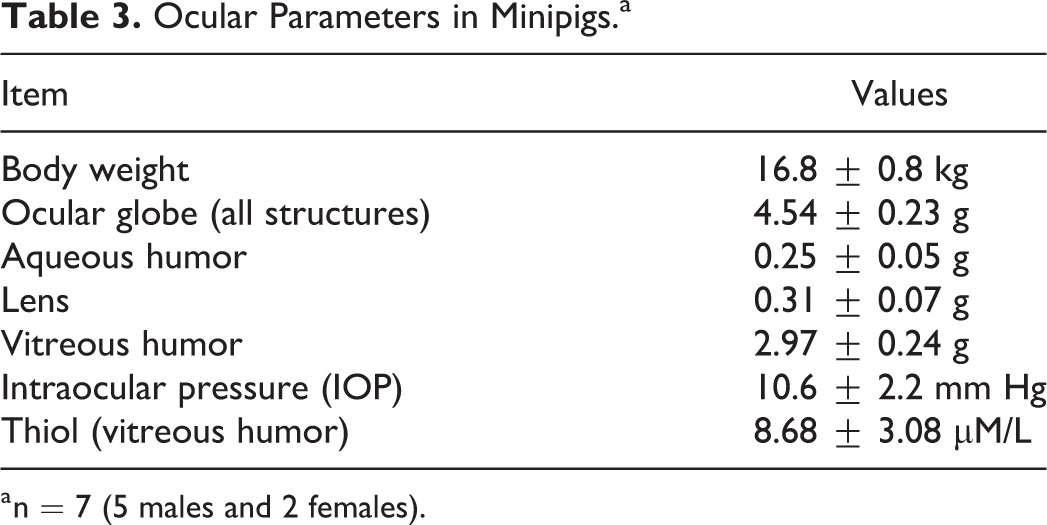
a n = 7 (5 males and 2 females).
Figure 1 shows representative pictures of fundus from different species. In addition to the differences described in Table 1 (presence of macula lutea, fovea centralis, and tapetum lucidum), minipig eyes have a slightly horizontal oval optic disc and blood vessels arising from its center, while primates have 4 pairs of venules and arterioles extending from an oval optic disc. Dogs normally have 3 to 4 retinal veins and several small arteries that emerge from the central area of the optic disc that is located at the tapetal–nontapetal junction. In rats, all veins and arteries radiate from the optic disc (center to periphery). Finally, rabbits differ the most with respect to the optic disc structure (merangiotic), which is horizontal with a deeply cupped appearance and with myelin rays extending nasally and temporally from the optic disc.
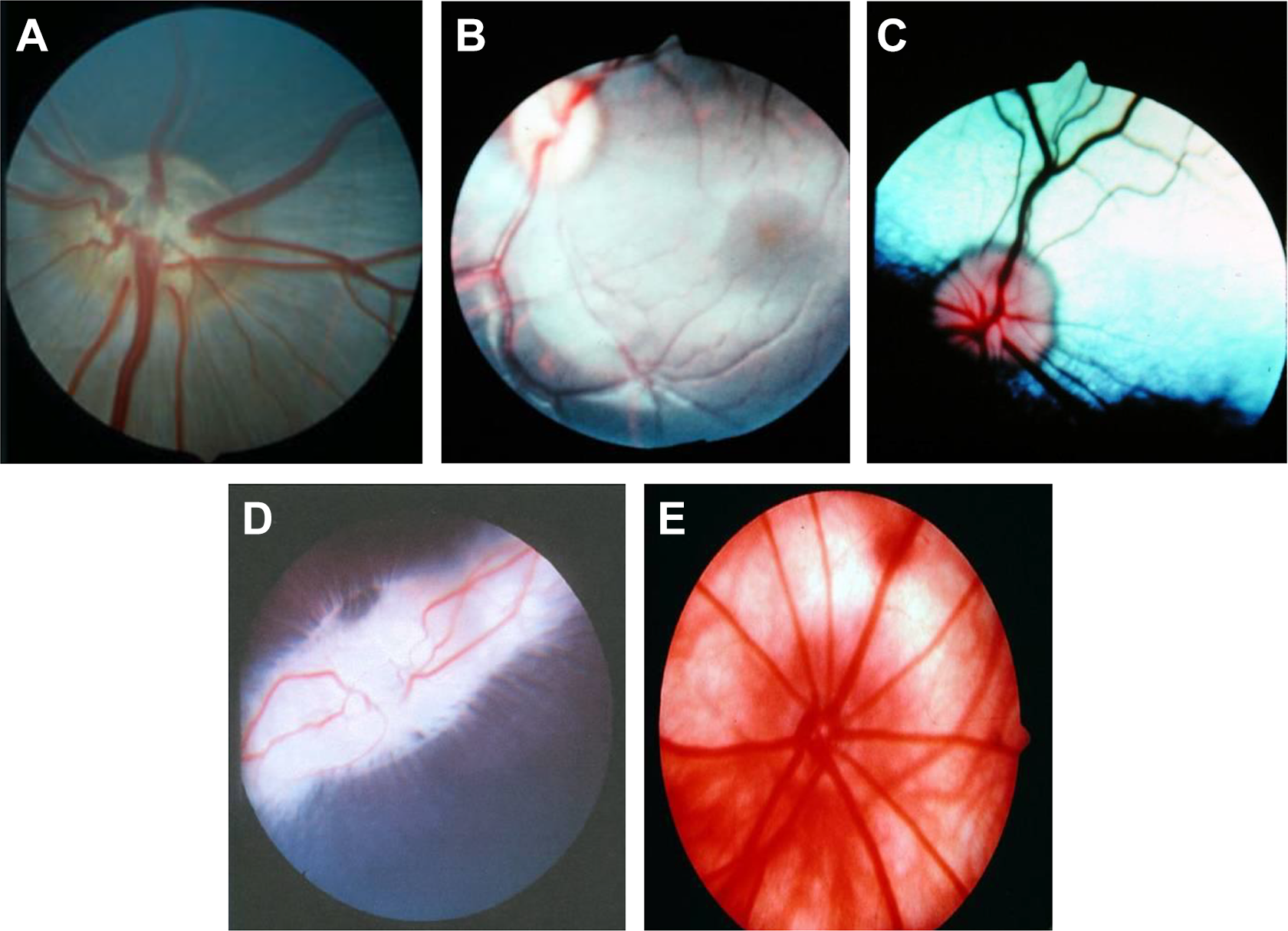
Representative fundus images from different species. A, Minipig. B, Primate (Rhesus). C, Dog. D, Rabbit. E, Rat.
As shown in Table 4, when comparing across species, the rank order of electrolyte concentrations in both aqueous and vitreous humors was sodium, chloride, potassium, calcium, and phosphorus, except in rats where potassium was greater than calcium and phosphorus. Sodium and chloride concentrations were generally higher in aqueous and/or vitreous samples across species when compared to calcium, potassium, and phosphorus. For each species except rats, electrolyte concentrations were generally comparable between their respective aqueous and vitreous humors, with the exception of phosphorus, where the aqueous–electrolyte concentration was slightly higher than vitreous humor levels. Aqueous and/or vitreous humor concentrations of sodium, potassium, and chloride were generally similar to species-specific serum levels; however, phosphorus and calcium concentrations were slightly below serum levels, and vitreous potassium concentrations in rats were 2.7-fold higher than serum potassium concentrations. This difference is likely due to terminal sample collection, as potassium levels have been reported as 5.07 ± 0.09 mmol/L following in vivo collection, 14 which is comparable to levels in other species reported here.
Electrolyte Concentrations of the Common Preclinical Species and Humans in Aqueous and Vitreous Humors.
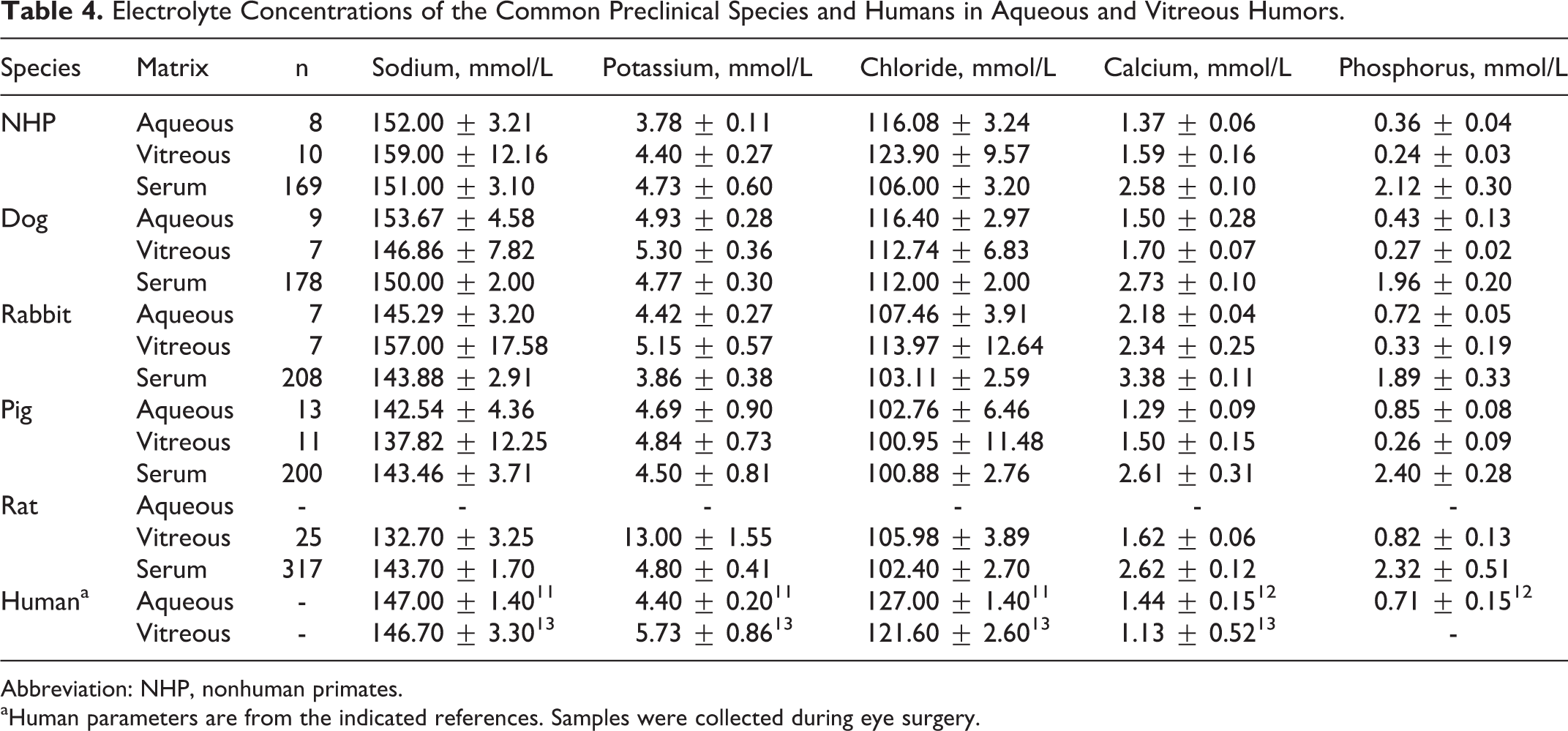
Abbreviation: NHP, nonhuman primates.
aHuman parameters are from the indicated references. Samples were collected during eye surgery.
Electroretinogram
Electroretinography was evaluated in isoflurane anesthetized minipigs treated with test items known to alter retinal function. Standard ERG waveforms from untreated eyes are presented in Figure 2. An increase in scotopic a-wave amplitudes in eyes receiving 5 mg Gentamicin (−0.59 to 0.90 log cds/m2) and eyes receiving 5 mg Glycine (0.41 to 0.90 log cds/m2) was observed when compared to control eyes (Figure 3A). Similarly, scotopic b-wave amplitudes were increased for glycine-treated eyes (−2.09 to 0.90 log cds/m2), whereas decreased amplitudes were observed in eyes given gentamicin (−1.59 to 0.90 log cds/m2; Figure 3B). At lower light intensities (2.09 to −0.90 log cds/m2), b-wave amplitude was mildly decreased after administration of 0.25 mg ICG, but at greater light intensities (≥0.41 log cds/m2), the amplitudes were generally within the control range (Figure 3B). A decrease in photopic b-wave amplitudes was noted for the glycine- and gentamicin-treated eyes. There were no treatment-related effects on scotopic or photopic a- or b-wave implicit time and latency (Figures 4A and B). As expected, marked reductions in oscillatory potentials (∼4-fold) and flicker response (∼3-fold) were noted in glycine- and gentamicin-treated eyes as well (Figure 5A).
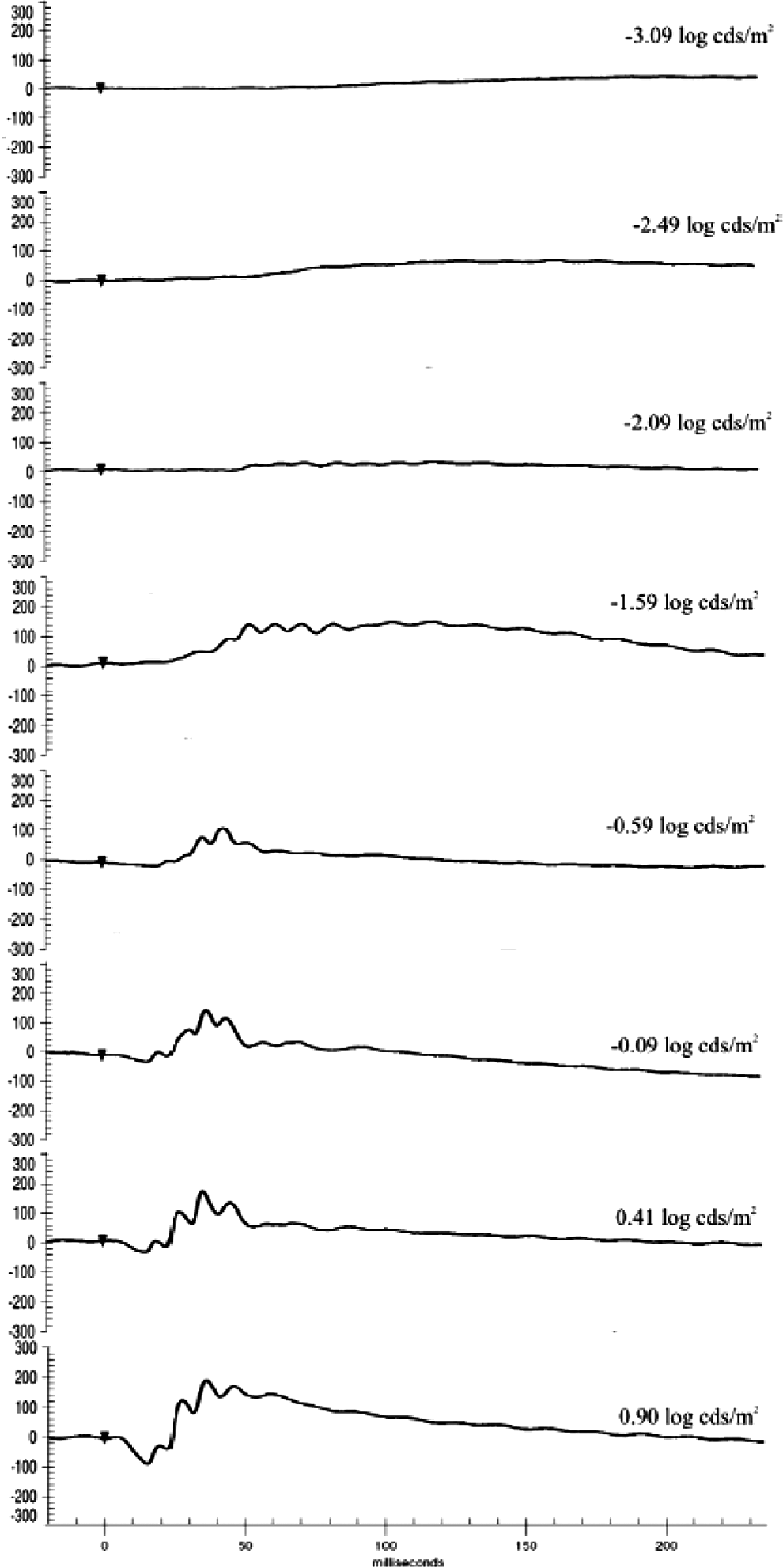
Standard scotopic waveforms in Göttingen minipigs.
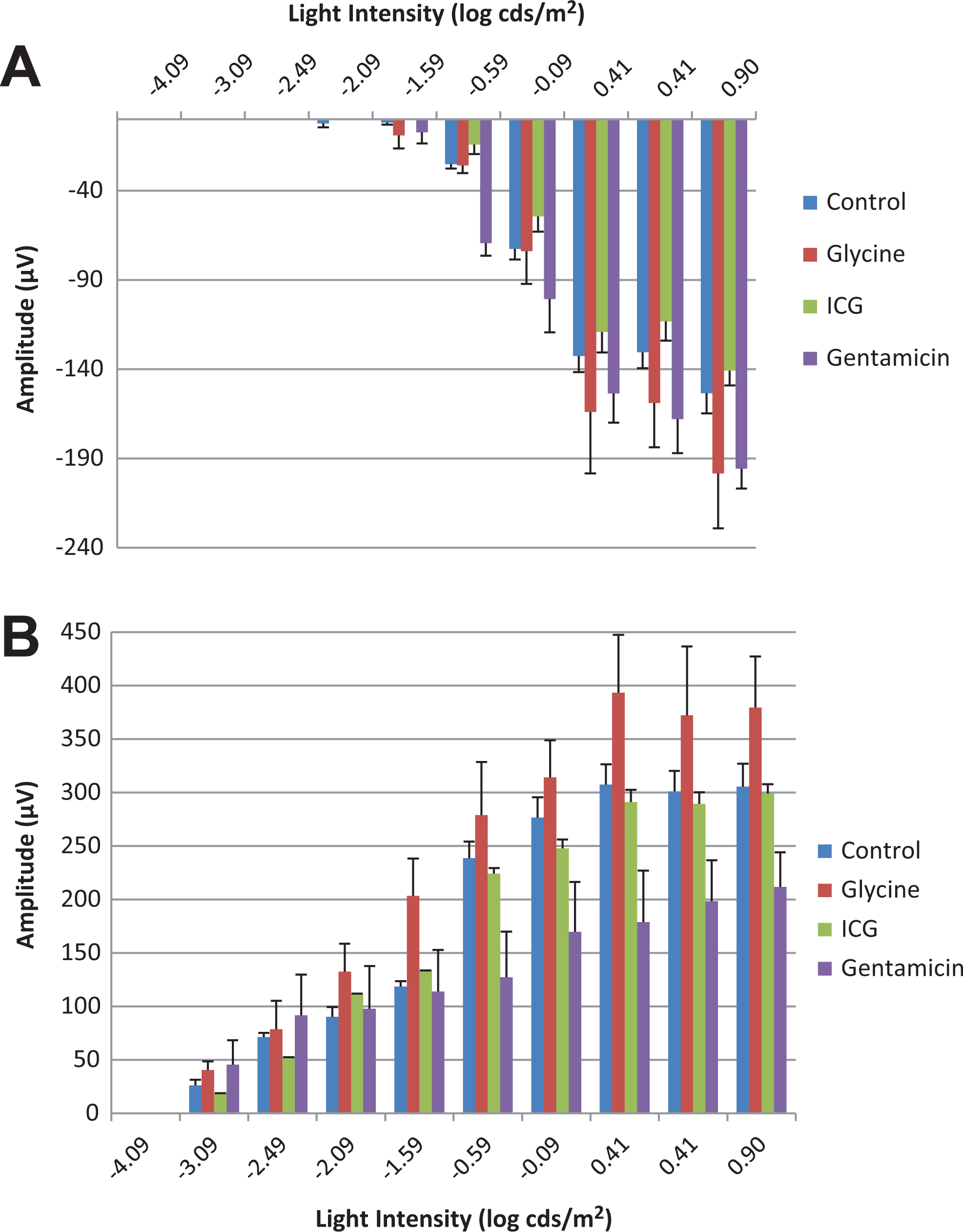
A, Scotopic a-wave amplitude of Göttingen minipigs treated with positive control agents. B, Scotopic b-wave amplitude of Göttingen minipigs treated with positive control agents.
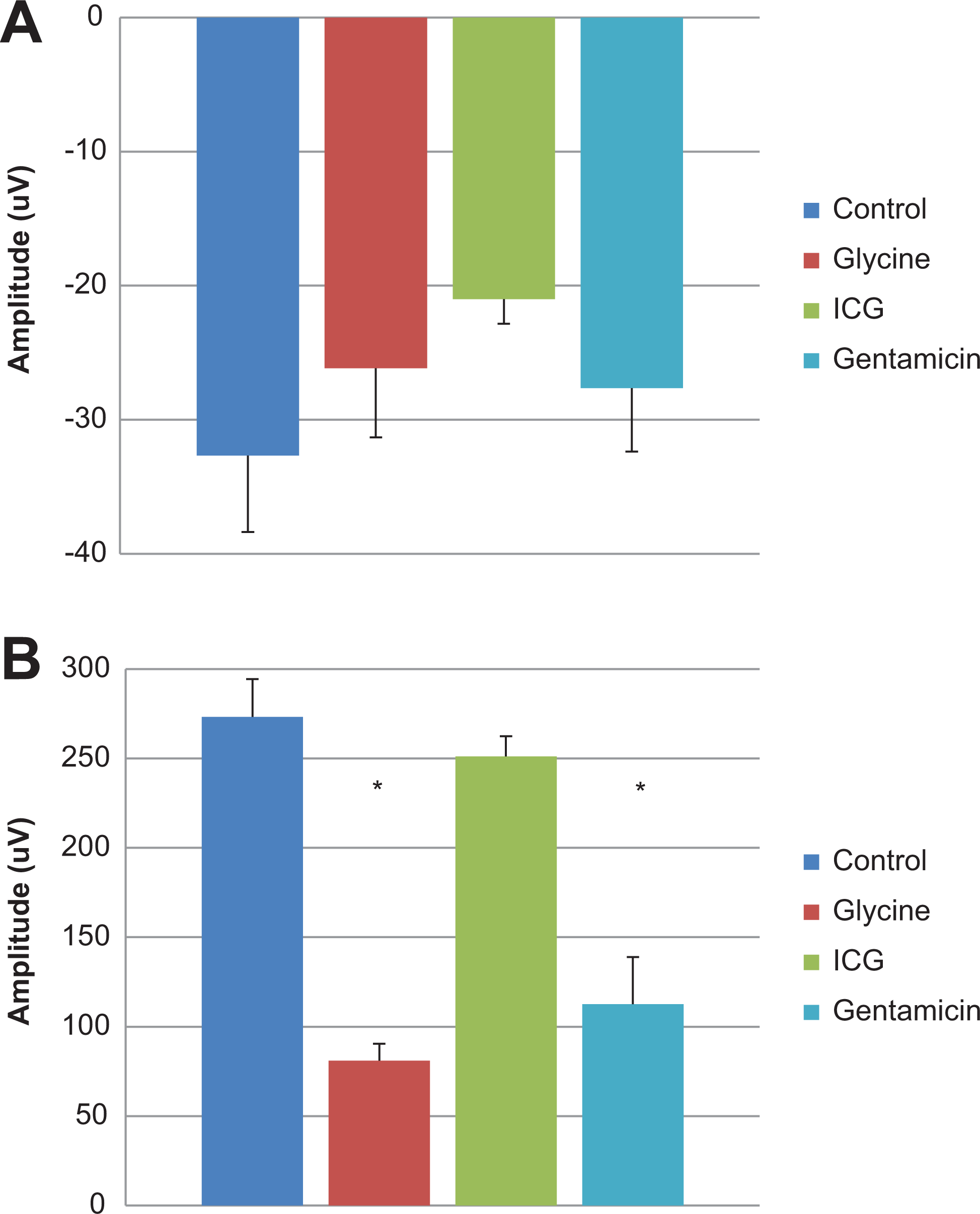
A, Photopic a-wave amplitude of Göttingen minipigs at 0.41 log cds/m2 treated with positive control agents. B, Photopic b-wave amplitude of Göttingen minipigs at 0.41 log cds/m2 treated with positive control agents.
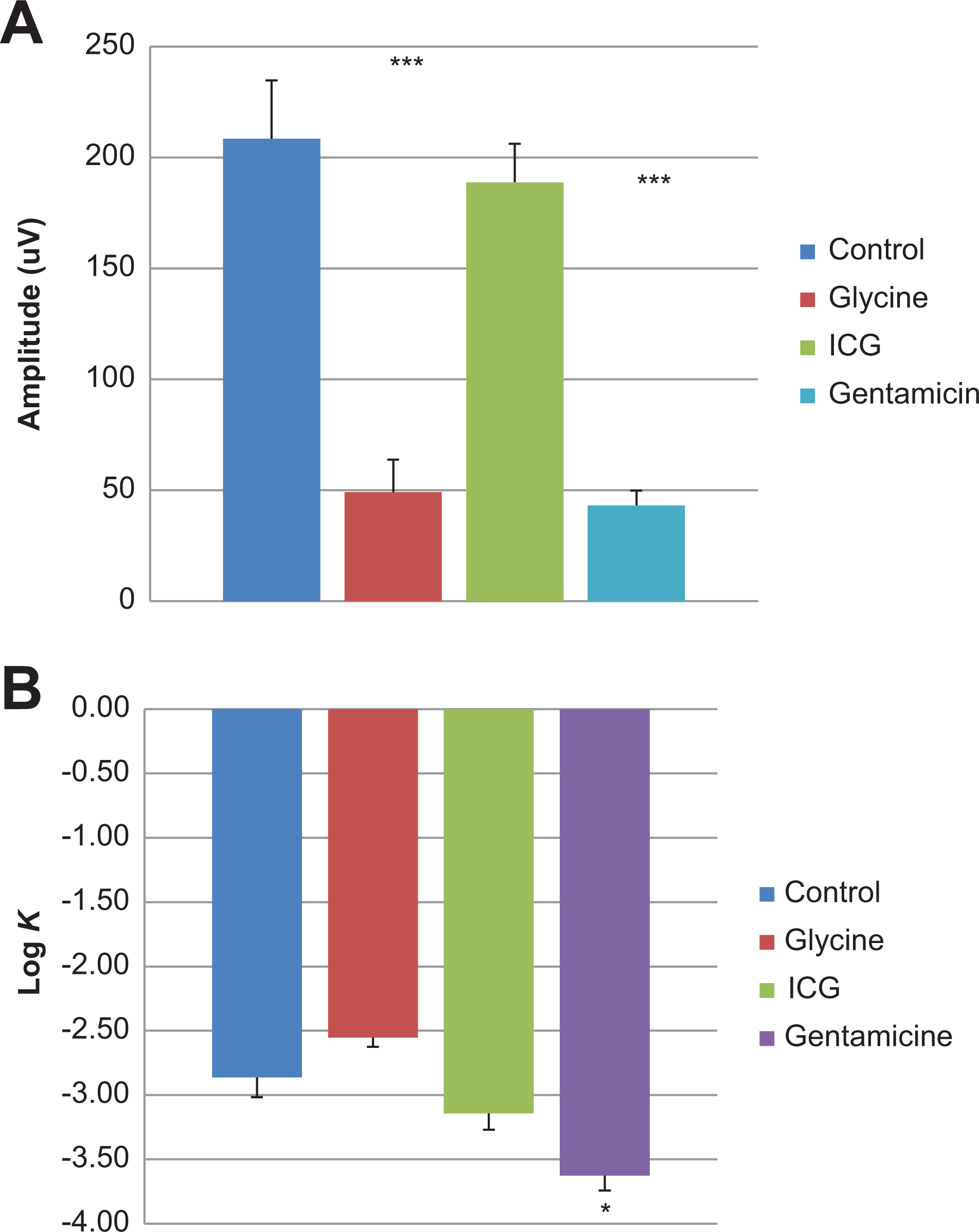
A, Oscillatory potentials of Göttingen minipigs at 0.41 log cds/m2 treated with positive control agents. B, Retinal sensitivity of Göttingen minipigs at 0.41 log cds/m2 treated with positive control agents.
Log
Discussion
This study provides a reference for electrolyte levels in the eye humors of common preclinical species, including minipigs in comparison to previously published human values. Sodium, potassium, chloride, 15 and calcium 16 concentrations in Göttingen minipigs were comparable to rabbit values in vitreous and aqueous humors. Moreover, similar values in the vitreous were observed in published porcine parameters, with the exception of calcium, 17 where calcium concentrations were 4-fold higher than the values observed in the present work. No phosphorus concentrations or thiol concentrations were found reported in the literature for porcine aqueous or vitreous humors.
Eyes are considered to be under high oxidative stress because they are exposed to direct light, oxygen tension (mainly in the retina), and high metabolic activity. 18 High free thiol content has been observed in the retina, lens, and choroid. However, thiol content was observed in the vitreous humor, which was expected since this enzyme is mitochondrial in origin and vitreous humor is essentially noncellular. 18 Thus, free thiol results suggest the presence of antioxidant in the vitreous humor, possibly contributing to protect ocular structures against free radical damage in the minipig. Electrolytes may interact with drugs that are delivered intravitreally or intracamerally and could potentially affect chemical properties and behavior.
Literature is scarce on ERG 19 in the minipig, and the results presented herein illustrate ERG changes following intravitreal administration in minipigs with expected effects noted for b-wave and oscillatory potentials following glycine or gentamicin administration. On the other hand, the effects of ICG administration were mild in comparison and observed at lower light intensity, which was expected for a positive control interfering with light transmission.
Glycine is a recognized predominant physiological inhibitory neurotransmitter in the retina. Clinically, transient visual disturbances have been reported in some individuals given glycine intravenously, 20 which were followed by prolonged visual evoked potentials. Moreover, in patients undergoing surgery where glycine was used as an irrigation fluid showed absence of oscillatory potentials. 21
Gentamicin is an antibiotic from the aminoglycoside group known to elicit retinal toxicity. A reduction in scotopic b-waves has been shown in rabbits following intravitreal or in vitro gentamicin treatment, 22 which was dose dependent and reversible. This selective reduction may be explained by accumulation of glutamate in the retina, as gentamicin impairs glutamate transport. Hence, the ERG evaluation methodology presented here was considered suitable for the assessment of retinal toxicity in preclinical toxicology studies using minipigs across a range of mechanisms of action.
Overall, the results suggest the minipig is a relevant nonrodent species for drug safety evaluation involving ocular end points. While the selection of the most appropriate preclinical model in drug safety assessment relies on complex considerations, the increasing availability of minipig data supports its use when scientifically justified.
Footnotes
Author Contributions
Luis Fernando Negro Silva contributed to conception, acquisition, analysis, interpretation, and drafted the manuscript; Christian Li contributed to conception and critically revised the manuscript; Paula Juliana Brizuela de Seadi Pereira drafted the manuscript; Wendy Tang contributed to acquisition; Michelle Dubuc-Mageau contributed to acquisition; Roy Forster critically revised the manuscript; Robert Tavcar critically revised the manuscript; Andy Makin critically revised manuscript; Simon Authier contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
