Abstract
Formulation of nonclinical evaluations is a challenge, with the fundamental need to achieve multiples of the clinical exposure complicated by differences in species and routes of administration-specific tolerances, depending on concentrations, volumes, dosing regimen, duration of each administration, and study duration. Current practice to approach these differences is based on individual experience and scattered literature with no comprehensive data source (the most notable exception being our 2006 publication on this same subject). Lack of formulation tolerance data results in excessive animal use, unplanned delays in the evaluation and development of drugs, and vehicle-dependent results. A consulting firm, a chemical company, and 4 contract research organizations conducted a rigorous data mining operation of vehicle data from studies dating from 1991 to 2015, enhancing the data from this author’s 2006 publication (3 of the six 2015 contributors were also 2006 contributors). Additional data were found in the published literature. The results identified 108 single-component vehicles (and 305 combination formulations) used in more than 1,040 studies across multiple species (dog, primate, rat, mouse, rabbit, guinea pig, minipig, pig, chick embryo, and cat) by multiple routes for a wide range of study durations. The tabulated data include maximum tolerated use levels by species, route, duration of study, dose-limiting toxicity where reported, review of the available literature on each vehicle, guidance on syringe selection, volume and pH limits by route with basic guidance on nonclinical formulation development, and guidance on factors to be considered in nonclinical route selection.
Introduction
From the point of initiation of development of a new drug or other test material, continuing on until the therapeutic enters serious clinical trials, perhaps the weakest link in investigations of both pharmacodynamic and toxicodynamic evaluations are formulations. In evaluating the nonclinical safety of potential new drugs, it is required that the material of interest be formulated in a manner that allows adequate administration of the test substance, with little or no effects on test animals attributable to the vehicles used in the delivery of the actual test material. The formulation must be chemically and physically suitable for the intended route of administration, optimize the stability of the active ingredient, and preferably (in most cases) maximize the systemic bioavailability of the drug. Because historically the process of vehicle selection has been mostly one of custom or personal choice, a range of vehicles have been most commonly used in formulation. Regrettably, information as to their suitability, utility, and the volume of nonclinical use limitations and species-specific considerations are not generally reflected in the literature or taught in any formal manner. Although recent years have seen the appearance of training courses on preformulation and articles on the development of early formulations (ie, Li and Zhao 1 ) proliferate, these concentrate on physical–chemical features. Certainly, such information on vehicle suitability for use in animals is not attainable in any organized source or on the Internet. This article, an expanded and updated version of the earlier work by Gad et al, 2 was undertaken to rectify the deficiency in information for those seeking to select suitable vehicles for nonclinical dosing of animals.
General Preclinical Formulation Principles
Dosing formulations for preclinical studies should be selected with consideration of a number of desirable characteristics. 3 –6 A compilation of practical route-specific considerations to be taken into account when choosing vehicles and implementing their use in different species is presented in Table 1. Dose–volume limits provided by animal use and care guidances are presented in Table 2. 3,4 In Table 3, information on the selection of appropriate needles for use for different routes in different species is presented. In Table 4, an index of vehicles/excipients covered in this publication, their synonyms, basic characteristics, and the species and routes in which the vehicles were evaluated is provided. Tables 5 to 113 present data on acceptable vehicle usage by individual vehicles, whereas tabular data as to tolerated mixture formulations have been added to this article in the final table (Table 114).
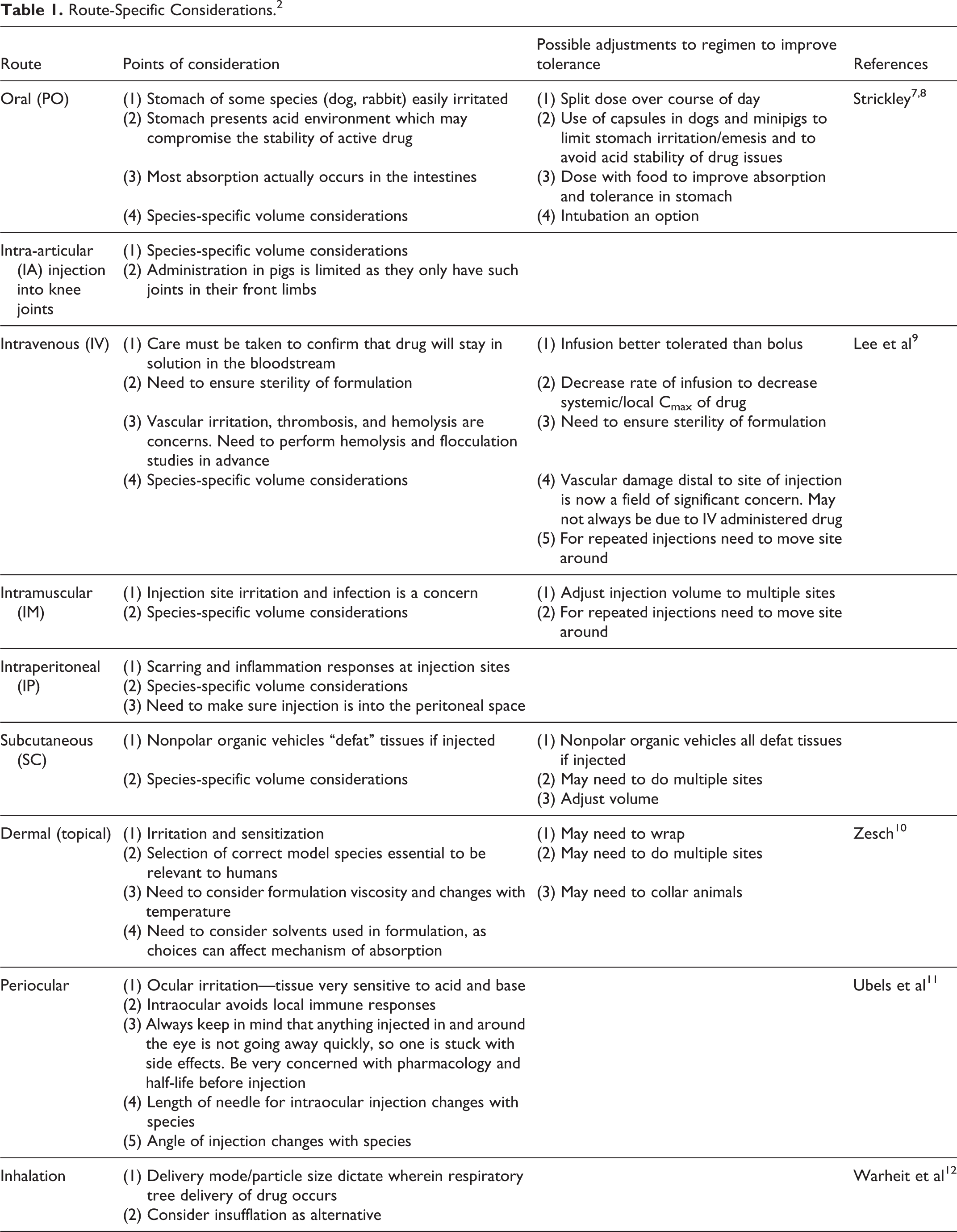
Route-Specific Considerations. 2
Volume Guidelines for Administration of Compounds by Route of Administration to Laboratory Animals. 3
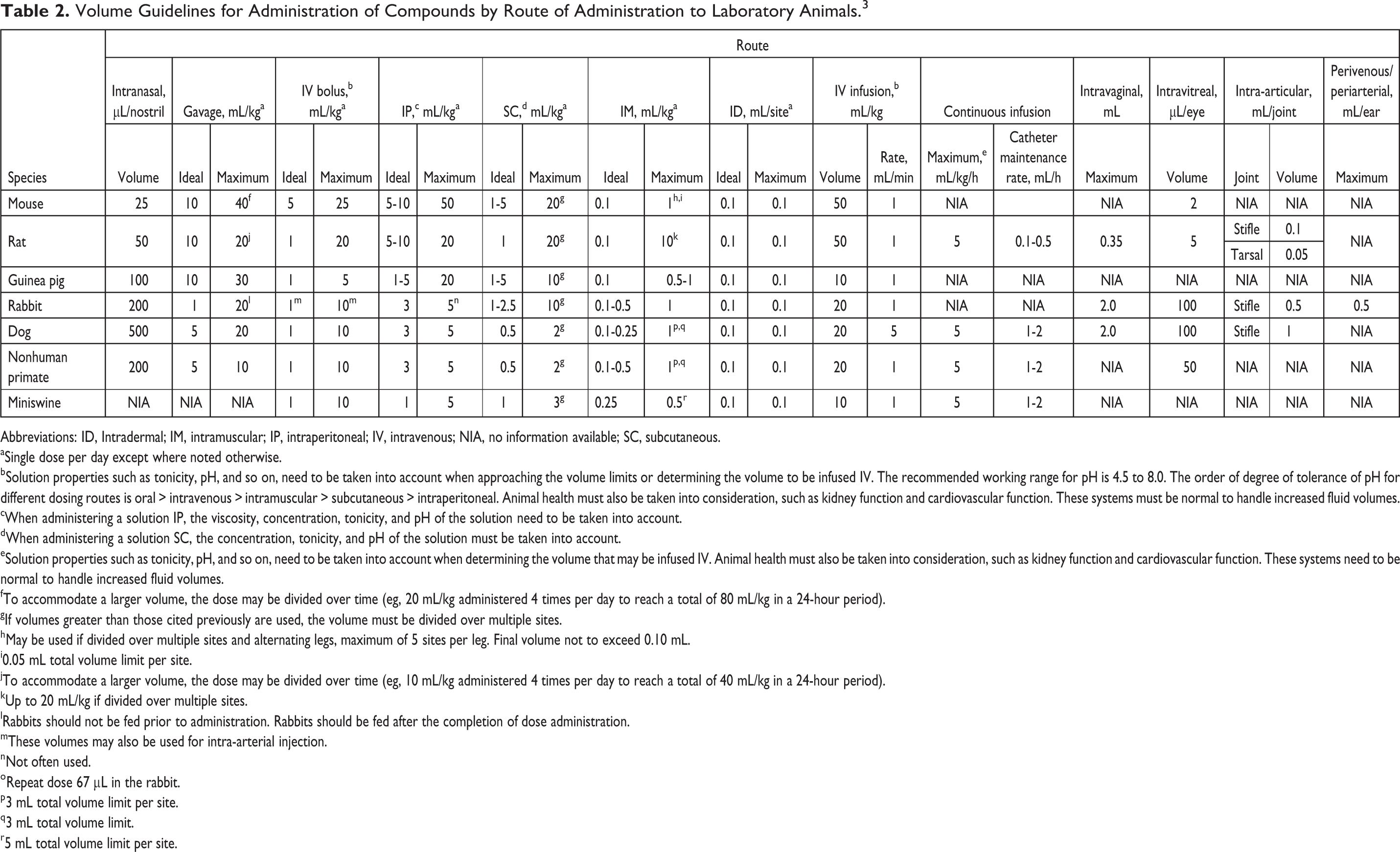
Abbreviations: ID, Intradermal; IM, intramuscular; IP, intraperitoneal; IV, intravenous; NIA, no information available; SC, subcutaneous.
aSingle dose per day except where noted otherwise.
bSolution properties such as tonicity, pH, and so on, need to be taken into account when approaching the volume limits or determining the volume to be infused IV. The recommended working range for pH is 4.5 to 8.0. The order of degree of tolerance of pH for different dosing routes is oral > intravenous > intramuscular > subcutaneous > intraperitoneal. Animal health must also be taken into consideration, such as kidney function and cardiovascular function. These systems must be normal to handle increased fluid volumes.
cWhen administering a solution IP, the viscosity, concentration, tonicity, and pH of the solution need to be taken into account.
dWhen administering a solution SC, the concentration, tonicity, and pH of the solution must be taken into account.
eSolution properties such as tonicity, pH, and so on, need to be taken into account when determining the volume that may be infused IV. Animal health must also be taken into consideration, such as kidney function and cardiovascular function. These systems need to be normal to handle increased fluid volumes.
fTo accommodate a larger volume, the dose may be divided over time (eg, 20 mL/kg administered 4 times per day to reach a total of 80 mL/kg in a 24-hour period).
gIf volumes greater than those cited previously are used, the volume must be divided over multiple sites.
hMay be used if divided over multiple sites and alternating legs, maximum of 5 sites per leg. Final volume not to exceed 0.10 mL.
i0.05 mL total volume limit per site.
jTo accommodate a larger volume, the dose may be divided over time (eg, 10 mL/kg administered 4 times per day to reach a total of 40 mL/kg in a 24-hour period).
kUp to 20 mL/kg if divided over multiple sites.
lRabbits should not be fed prior to administration. Rabbits should be fed after the completion of dose administration.
mThese volumes may also be used for intra-arterial injection.
nNot often used.
oRepeat dose 67 μL in the rabbit.
p3 mL total volume limit per site.
q3 mL total volume limit.
r5 mL total volume limit per site.
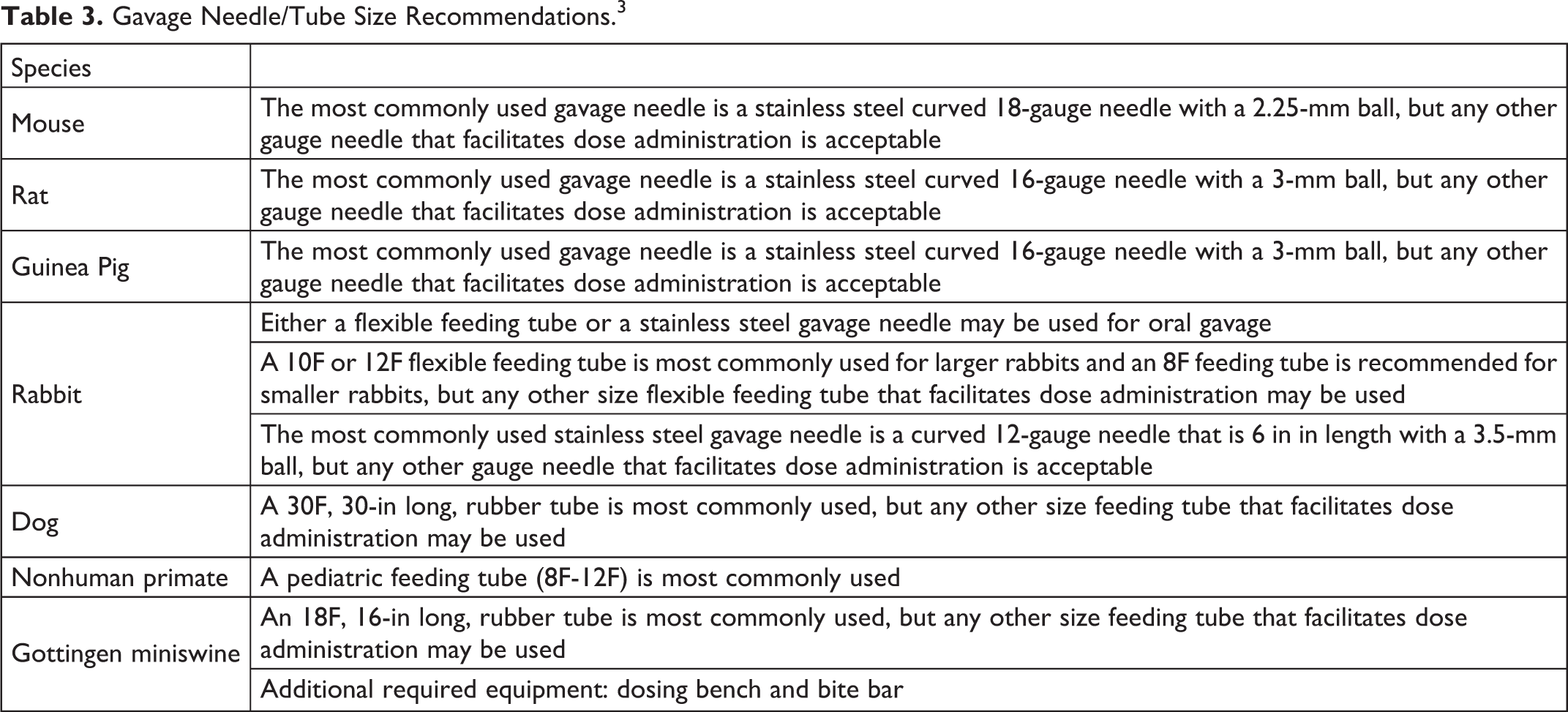
Gavage Needle/Tube Size Recommendations. 3
Index of Vehicles and Excipients—Codex and Details.
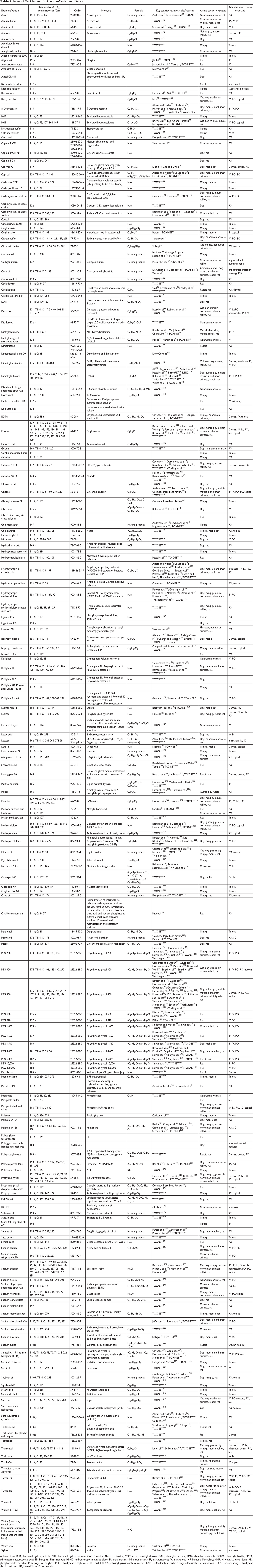
Abbreviations: BHA, butylated hydroxyanisole; BHT, butylated hydroxytoluene; CAS, Chemical Abstracts Service; CMC, carboxymethylcellulose; DAM, diacetylmonoxime; DMA, dimethyl acetamide; DMSO, dimethylsulfoxide; EDTA, ethylenediaminetetraacetic acid; EP, European Pharmacopeia; HPMC, hydroxypropyl methylcellulose; IA, intra-articular; IM, intramuscular; IP, intraperitoneal; IV, intravenous; NF, National Formulary; NMP, N-Methyl-2-pyrrolidone; PBS, phosphate-buffered saline; PEG, polyethylene glycol; PET, polyethylene terephthalate; PO, oral; PVP VA, polyvinylpyrrolidone/vinyl acetate; RAMEB, Randomly methylated β-cyclodextrin; SC, subcutaneous; TPGS, D-α-tocopheryl polyethylene glycol succinate; USP, United States Pharmacopeia.
Acacia.
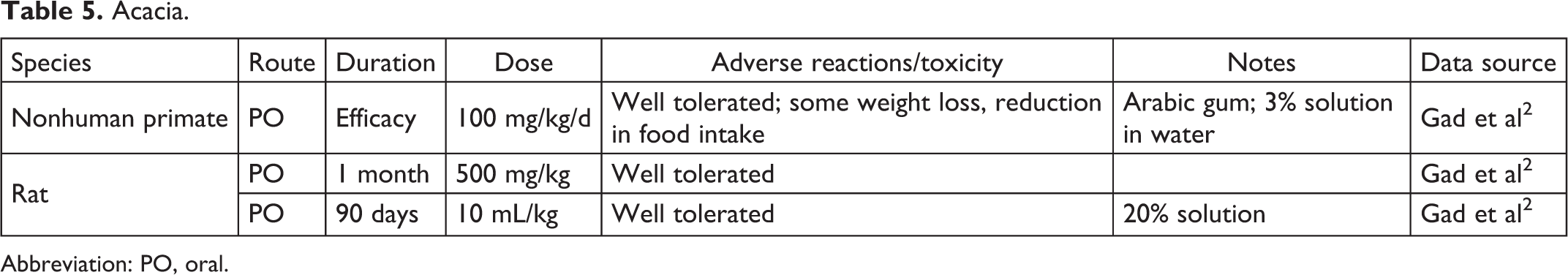
Abbreviation: PO, oral.
Acetic Acid.
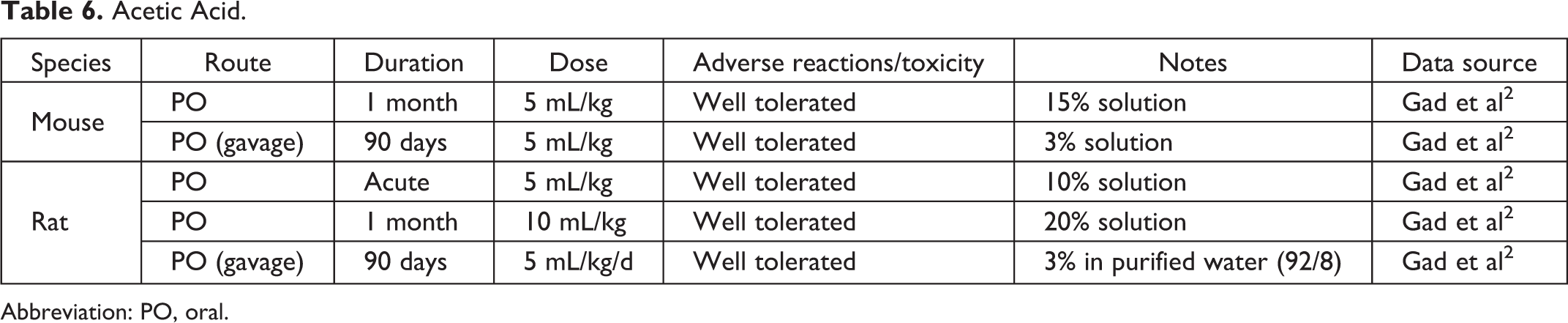
Abbreviation: PO, oral.
Acetone.
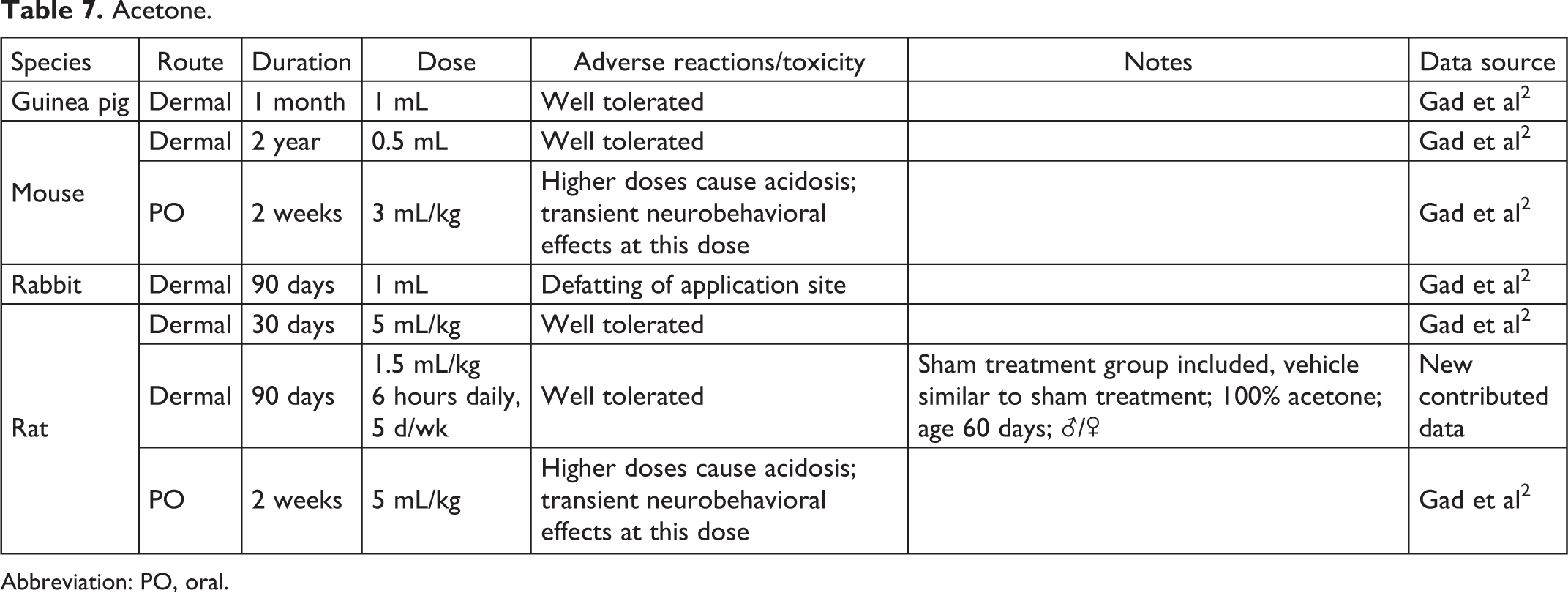
Abbreviation: PO, oral.
Acetylmethylamide.

Abbreviation: ADME, absorption, distribution, metabolism, excretion; PO, oral.
Alginic Acid.

Abbreviation: IP, intraperitoneal.
Anecortave Acetate.

Abbreviation: SC, subcutaneous.
Avicel CL-611.
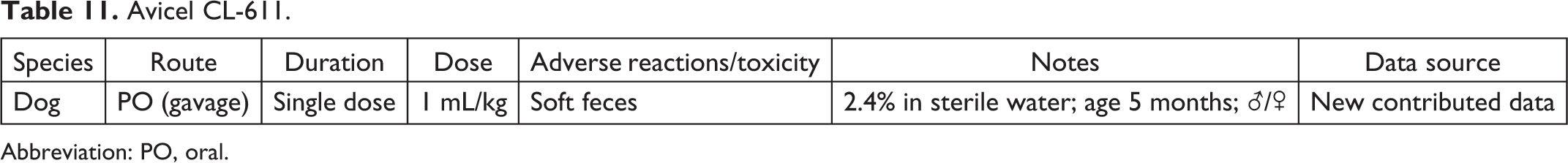
Abbreviation: PO, oral.
Balanced Salt Saline.

Abbreviations: GLP, good laboratory practice; q14d, every 14 days; SD, single dose.
Basal Salt Solution.

Abbreviations: GLP, good laboratory practice; SD, single dose.
Benzoic Acid.
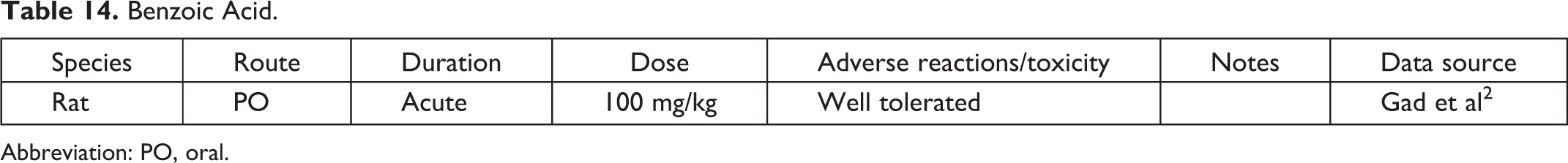
Abbreviation: PO, oral.
Beta-Cyclodextrin.
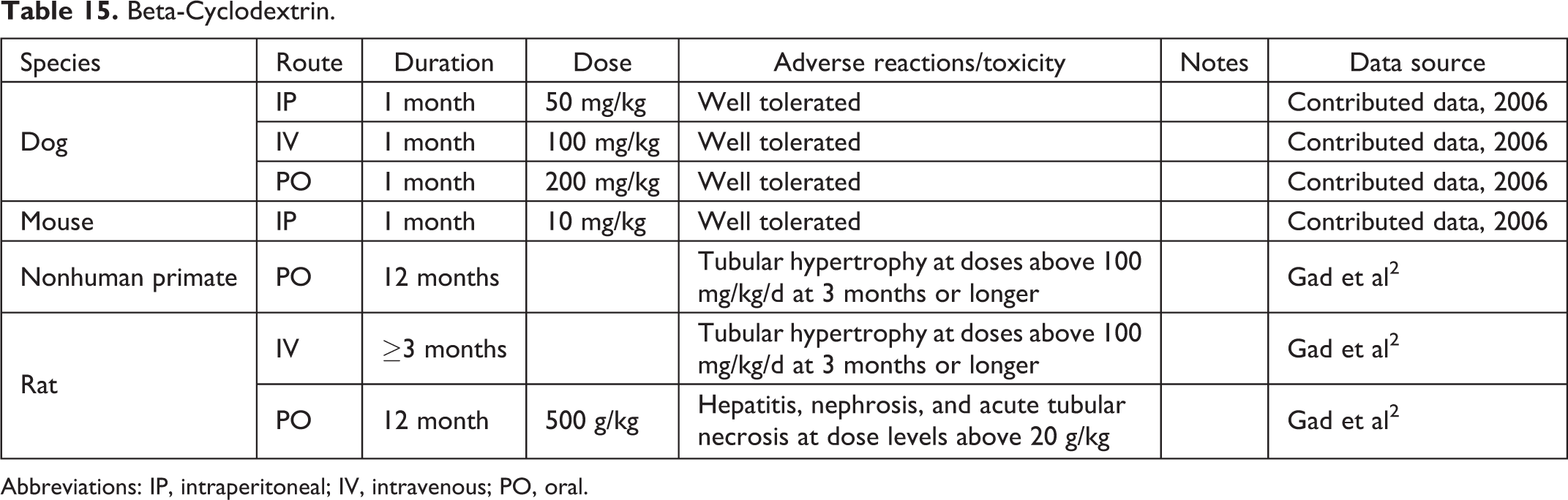
Abbreviations: IP, intraperitoneal; IV, intravenous; PO, oral.
Bicarbonate Buffer, pH 9.5.
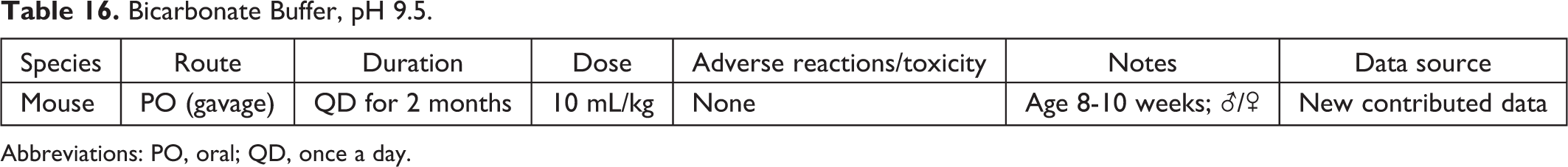
Abbreviations: PO, oral; QD, once a day.
Calcium Chloride.

Abbreviations: IV, intravenous; SC, subcutaneous.
Canola Oil.
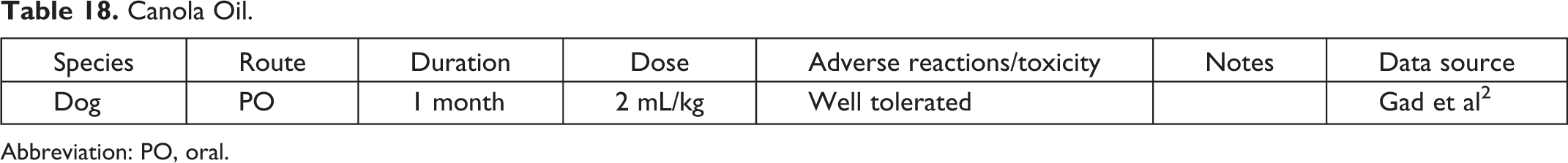
Abbreviation: PO, oral.
Capryol 90.
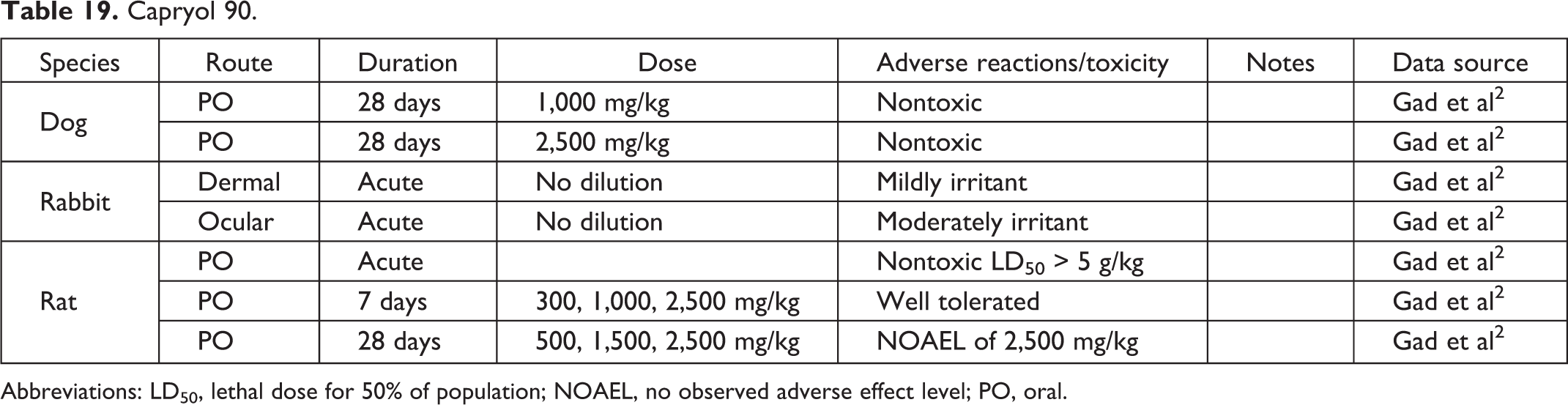
Abbreviations: LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PO, oral.
Captisol.
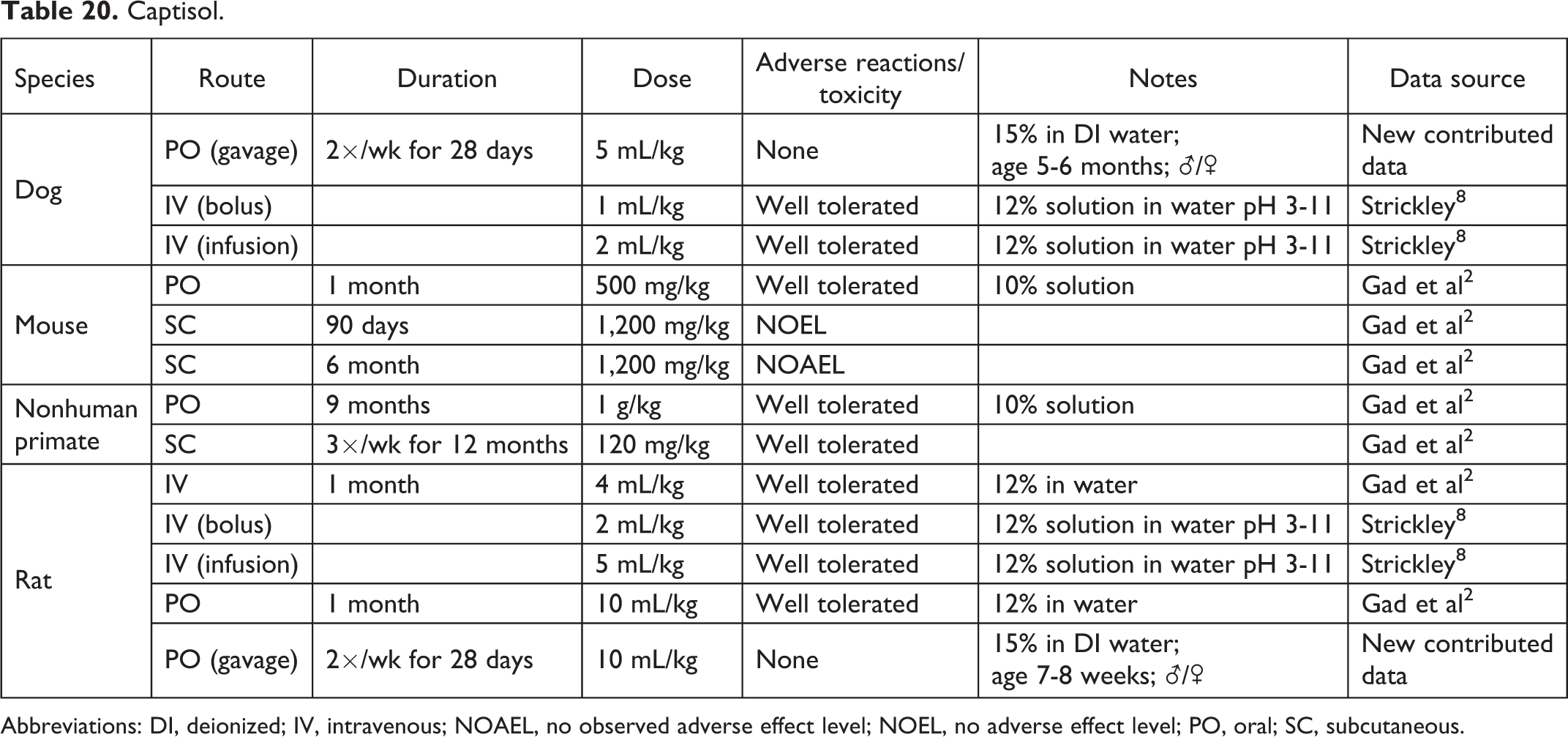
Abbreviations: DI, deionized; IV, intravenous; NOAEL, no observed adverse effect level; NOEL, no adverse effect level; PO, oral; SC, subcutaneous.
Carboxymethylcellulose.
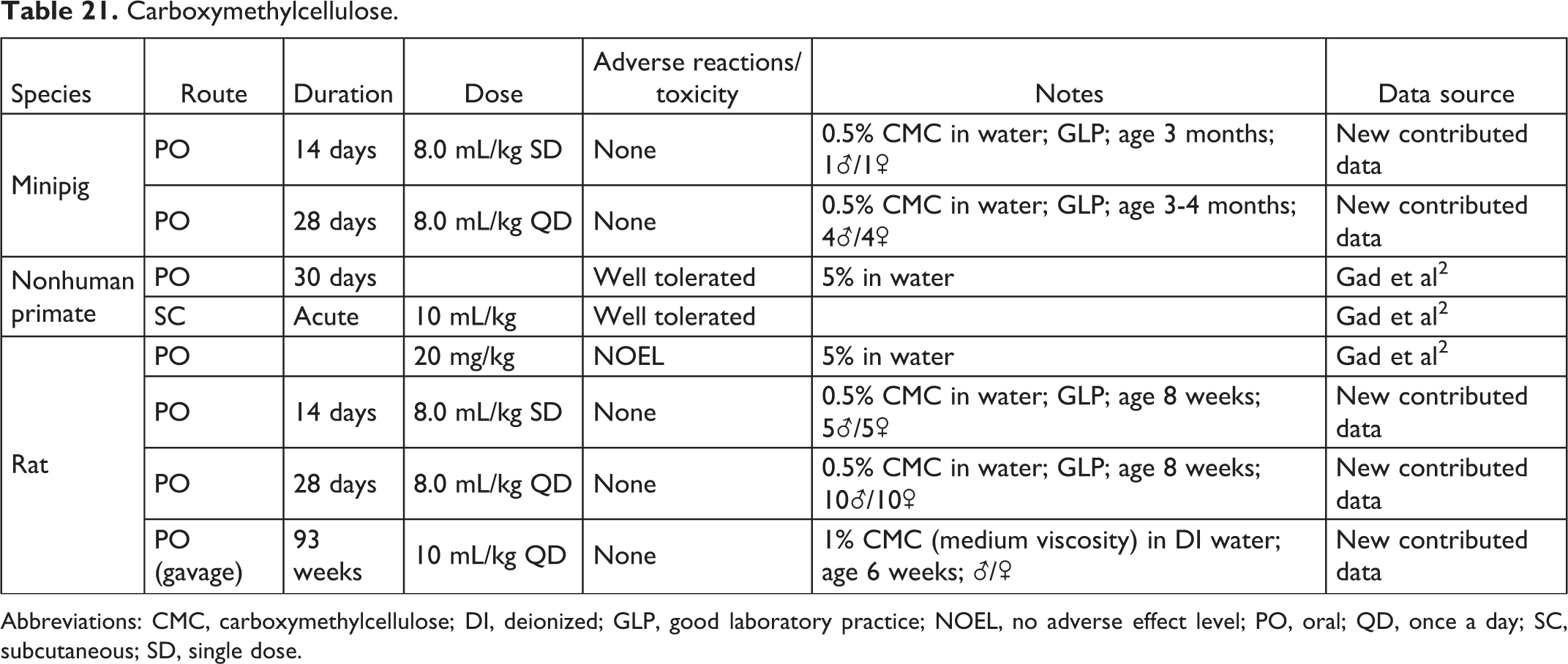
Abbreviations: CMC, carboxymethylcellulose; DI, deionized; GLP, good laboratory practice; NOEL, no adverse effect level; PO, oral; QD, once a day; SC, subcutaneous; SD, single dose.
Carboxymethylcellulose Calcium.
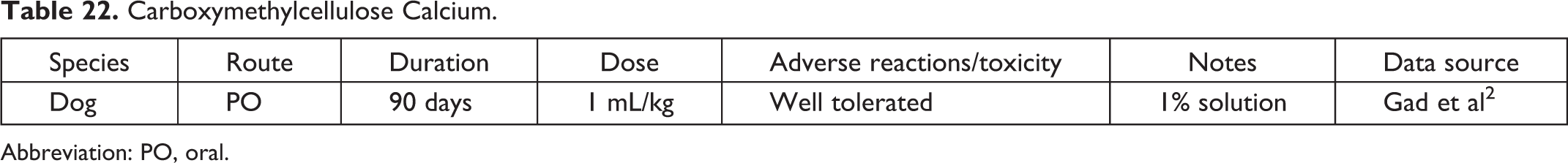
Abbreviation: PO, oral.
Carboxymethylcellulose Sodium.

Abbreviation: PO, oral.
Cetyl Alcohol.

Abbreviation: IP, intraperitoneal.
Citrate Buffer.
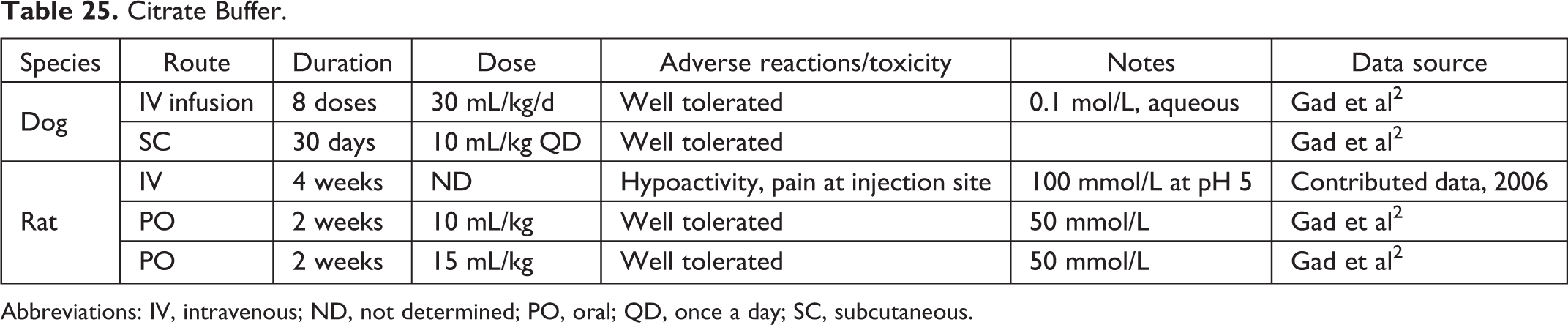
Abbreviations: IV, intravenous; ND, not determined; PO, oral; QD, once a day; SC, subcutaneous.
Citric Acid Buffer.
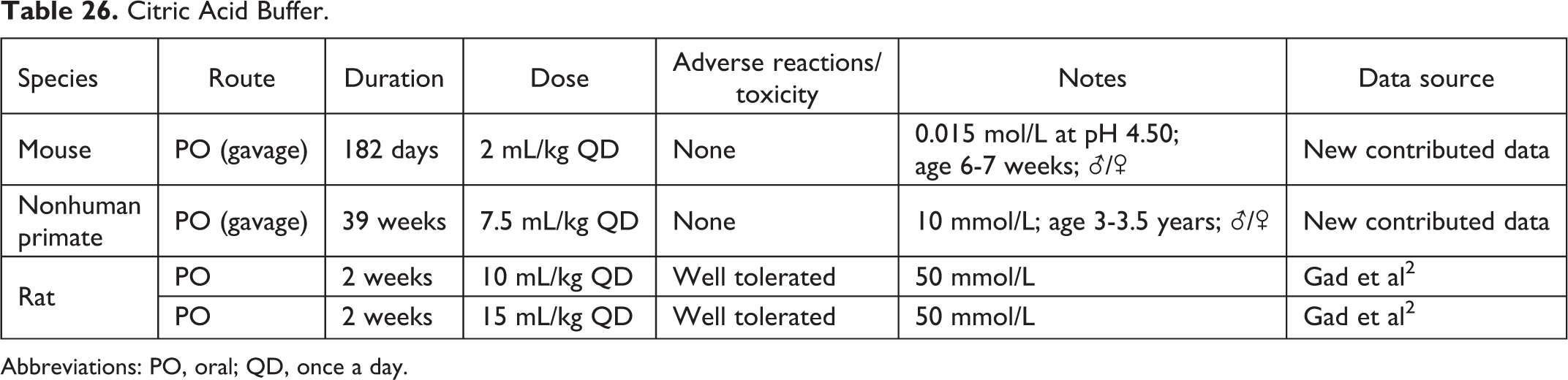
Abbreviations: PO, oral; QD, once a day.
Collagen Matrix.

Corn Oil.
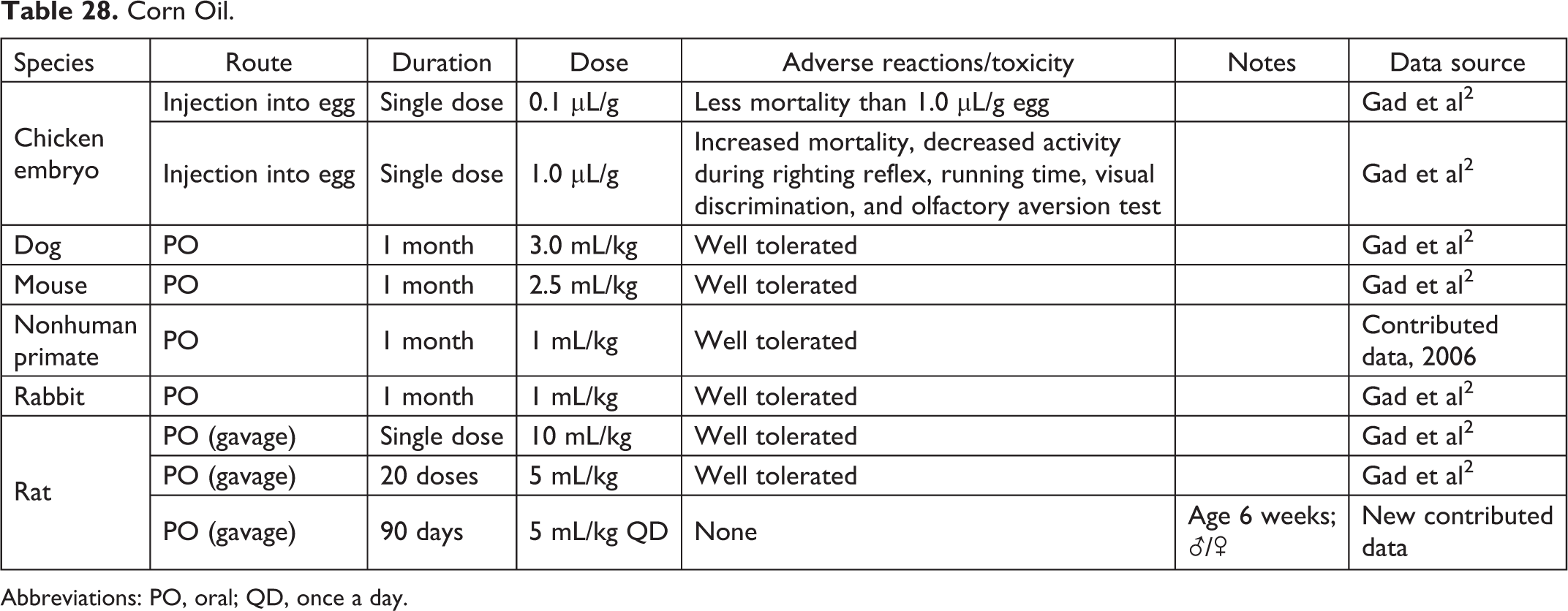
Abbreviations: PO, oral; QD, once a day.
Cottonseed Oil.

Abbreviation: SC, subcutaneous.
Cyclohexane.
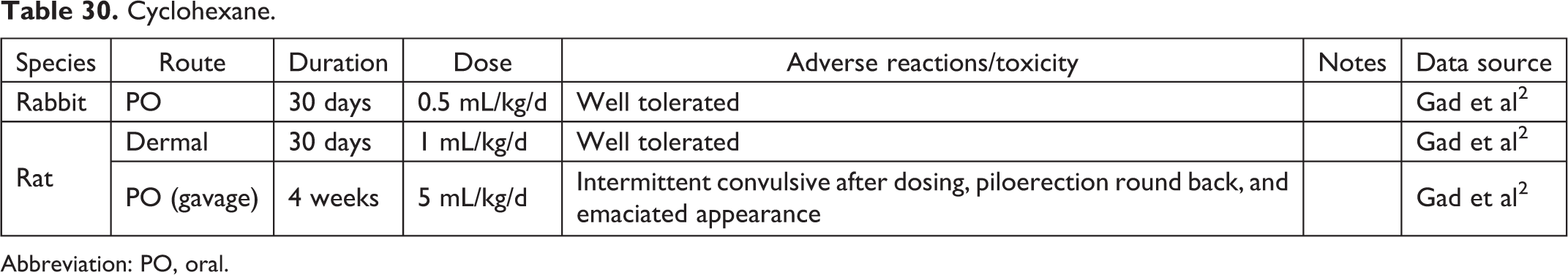
Abbreviation: PO, oral.
Dextrose.
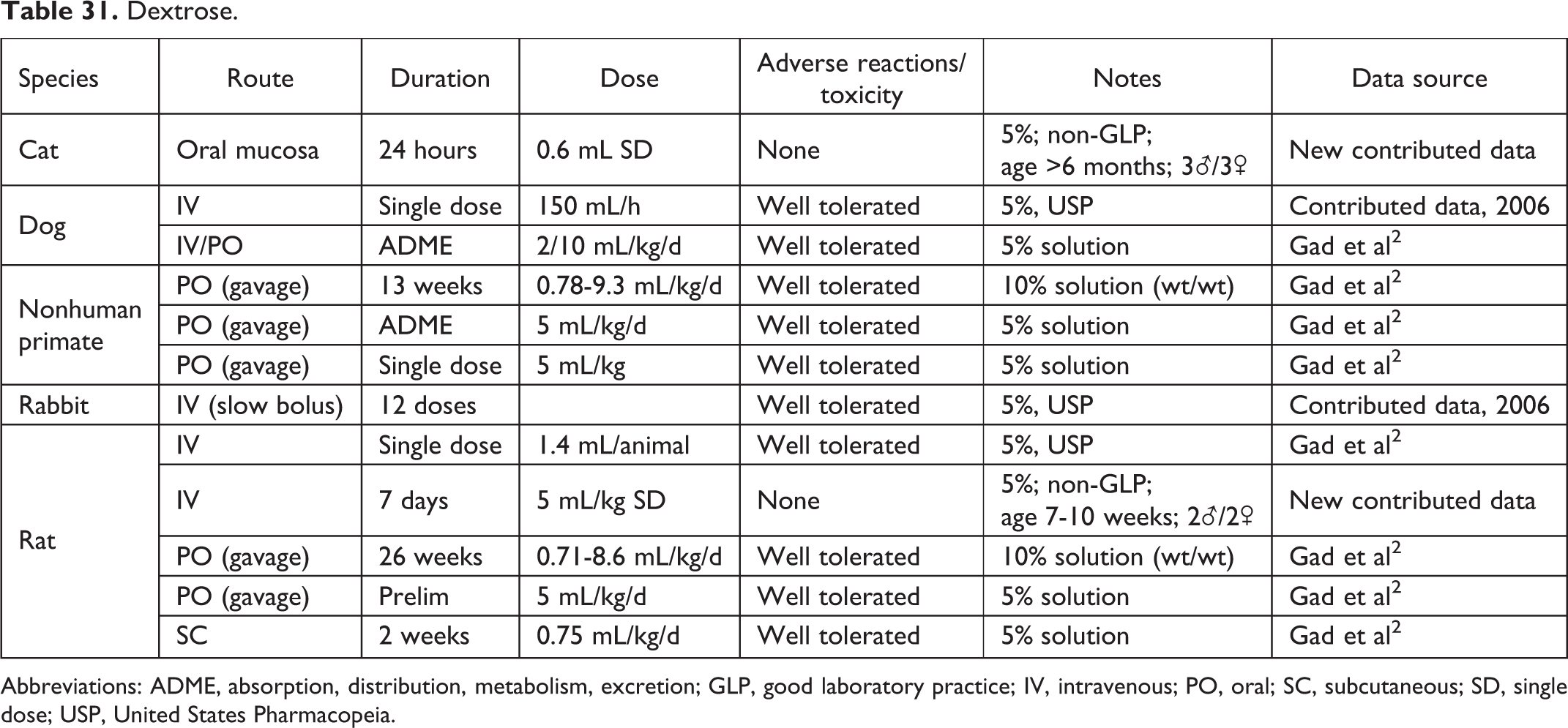
Abbreviations: ADME, absorption, distribution, metabolism, excretion; GLP, good laboratory practice; IV, intravenous; PO, oral; SC, subcutaneous; SD, single dose; USP, United States Pharmacopeia.
Dichlorvos.
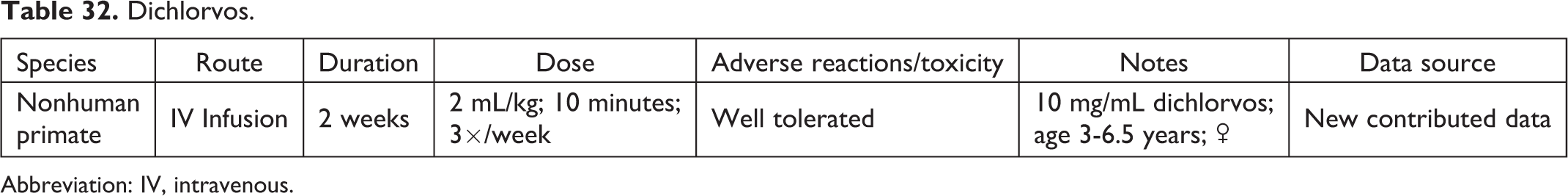
Abbreviation: IV, intravenous.
Diethylacetamide.
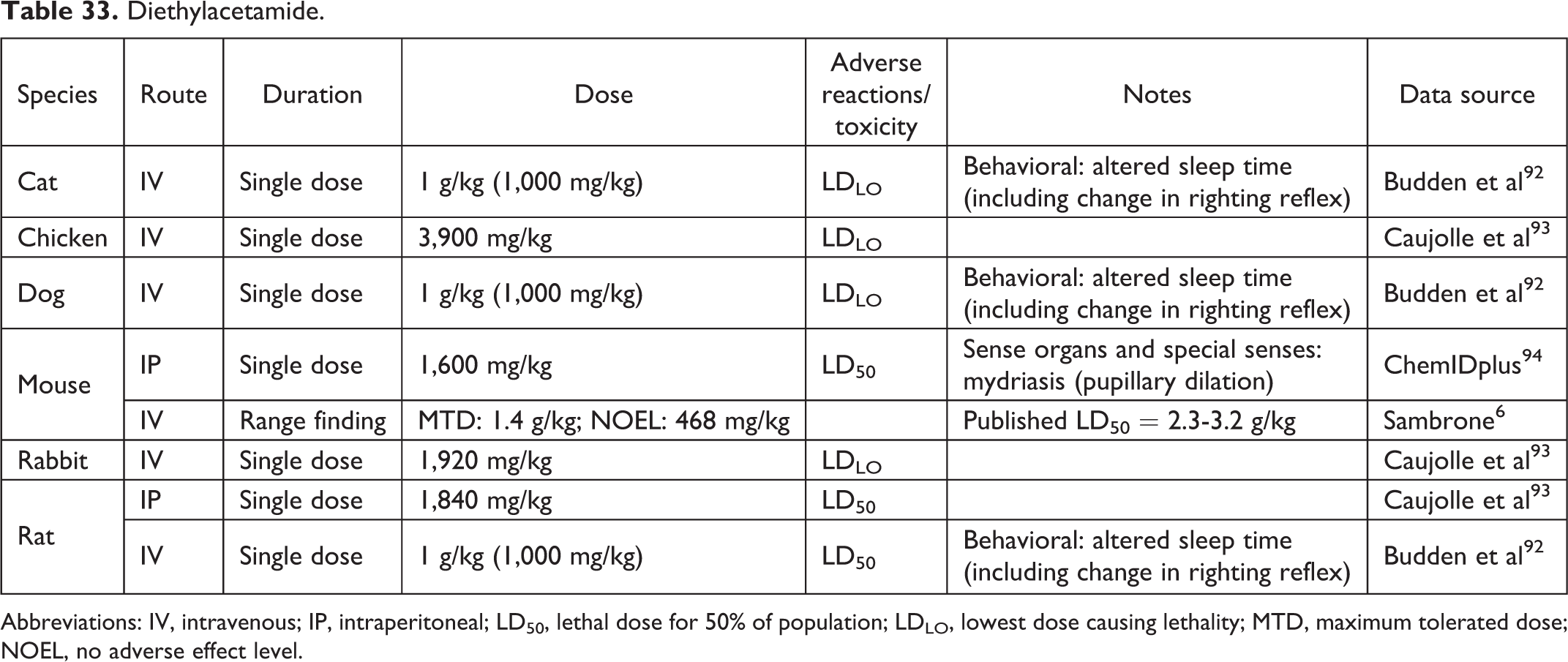
Abbreviations: IV, intravenous; IP, intraperitoneal; LD50, lethal dose for 50% of population; LDLO, lowest dose causing lethality; MTD, maximum tolerated dose; NOEL, no adverse effect level.
Diethyleneglycol-Monoethylether.
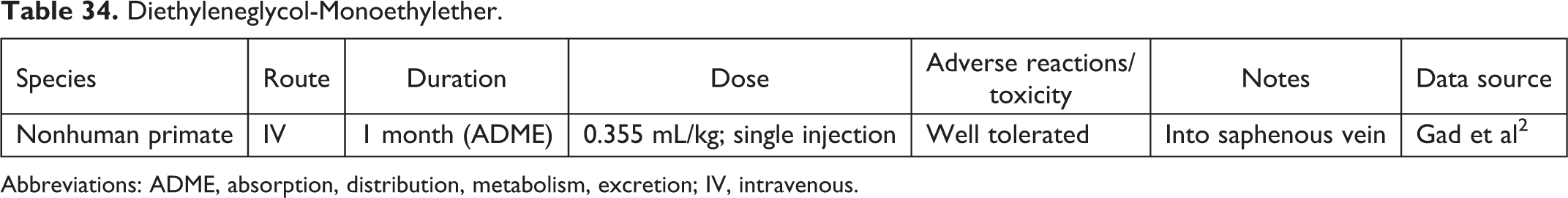
Abbreviations: ADME, absorption, distribution, metabolism, excretion; IV, intravenous.
Dimethylacetamide.
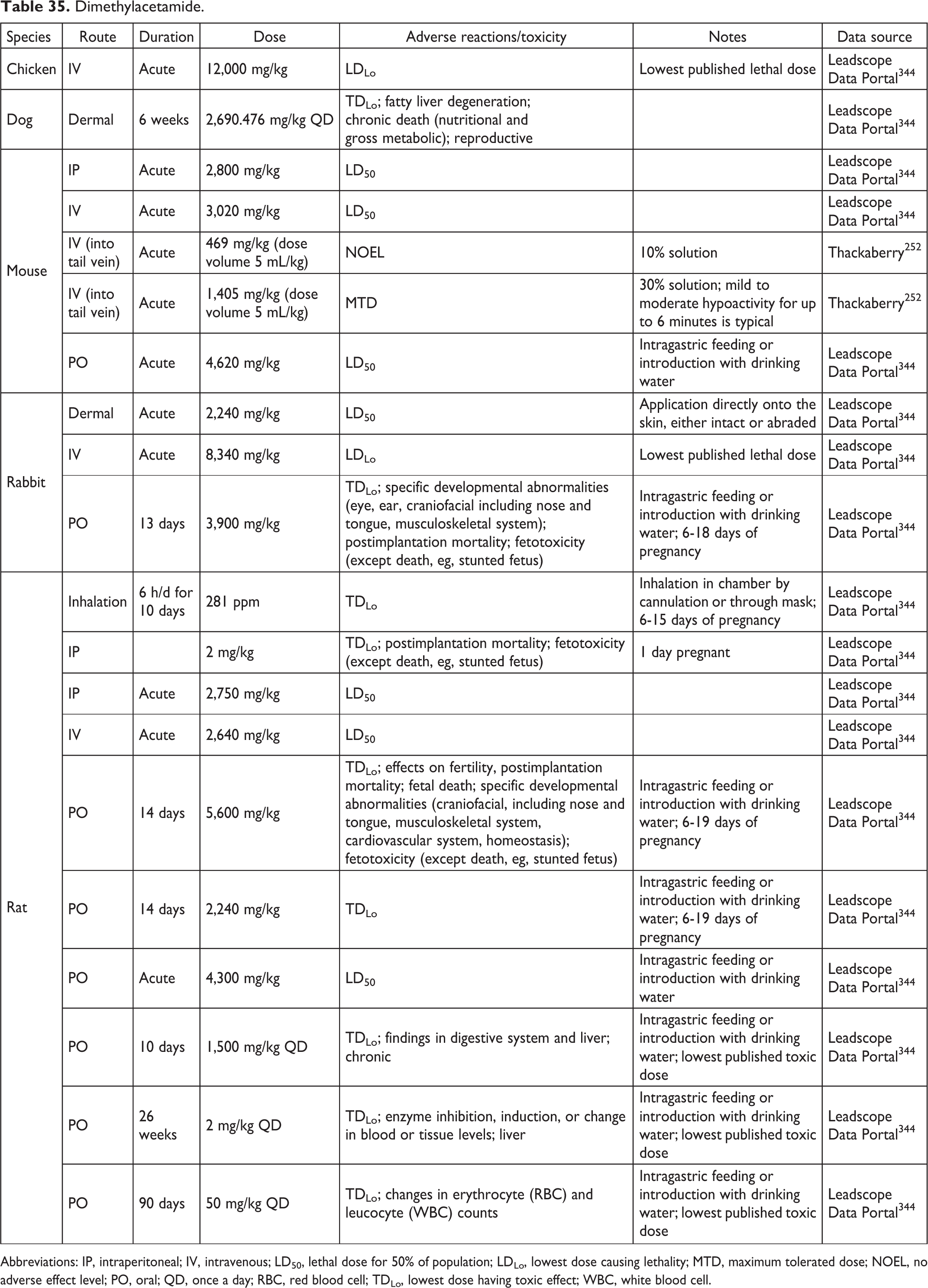
Abbreviations: IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; LDLo, lowest dose causing lethality; MTD, maximum tolerated dose; NOEL, no adverse effect level; PO, oral; QD, once a day; RBC, red blood cell; TDLo, lowest dose having toxic effect; WBC, white blood cell.
Dimethylsulfoxide.
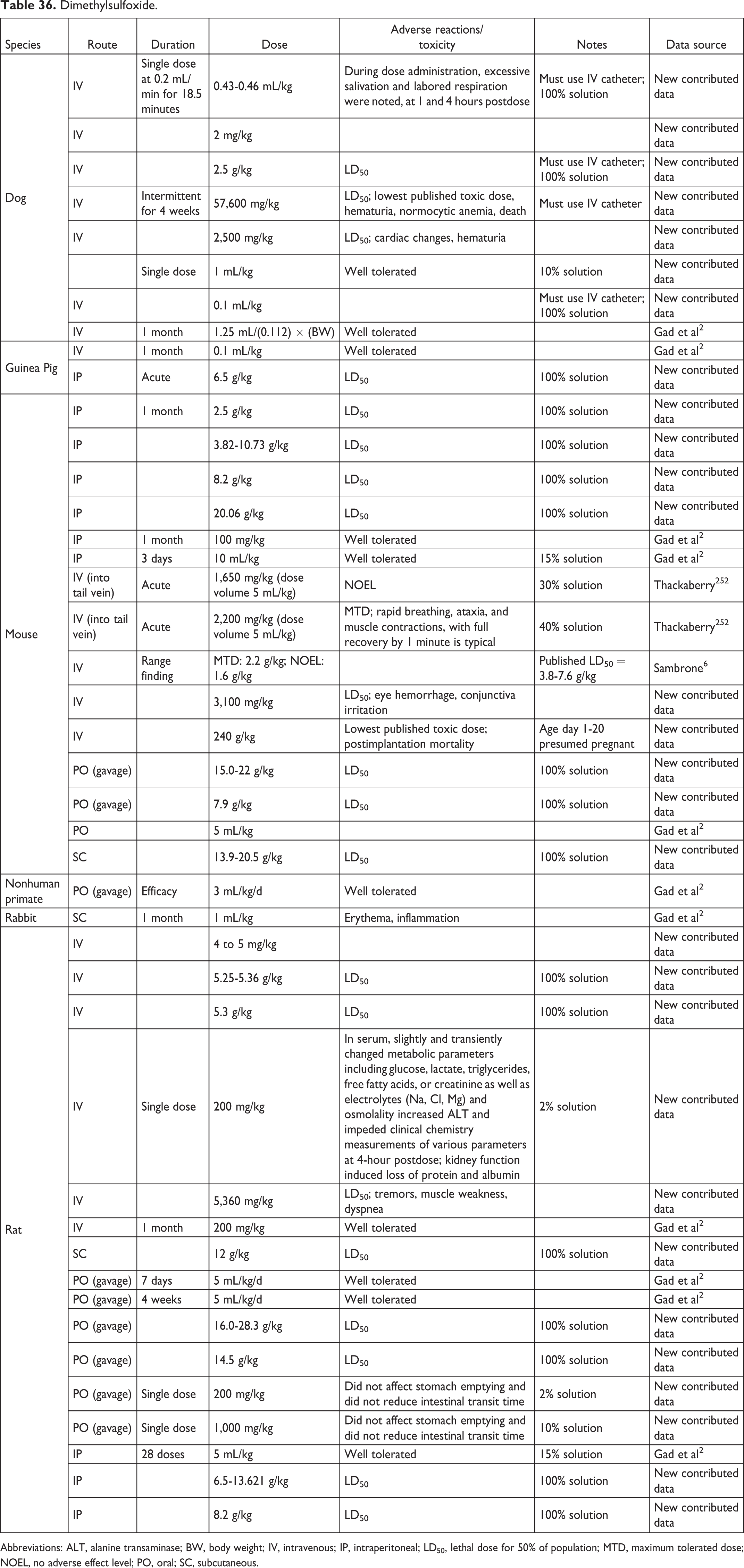
Abbreviations: ALT, alanine transaminase; BW, body weight; IV, intravenous; IP, intraperitoneal; LD50, lethal dose for 50% of population; MTD, maximum tolerated dose; NOEL, no adverse effect level; PO, oral; SC, subcutaneous.
Dulbecco Modified Phosphate-Buffered Saline.
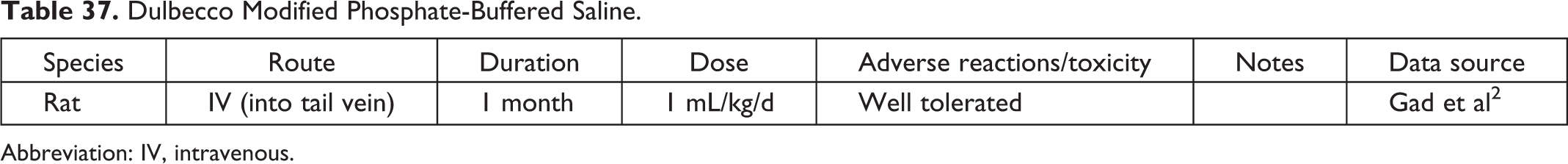
Abbreviation: IV, intravenous.
Dulbecco Phosphate-Buffered Saline.
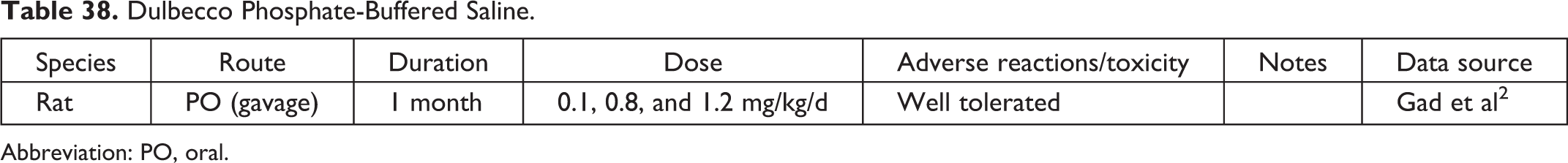
Abbreviation: PO, oral.
Ethanol.
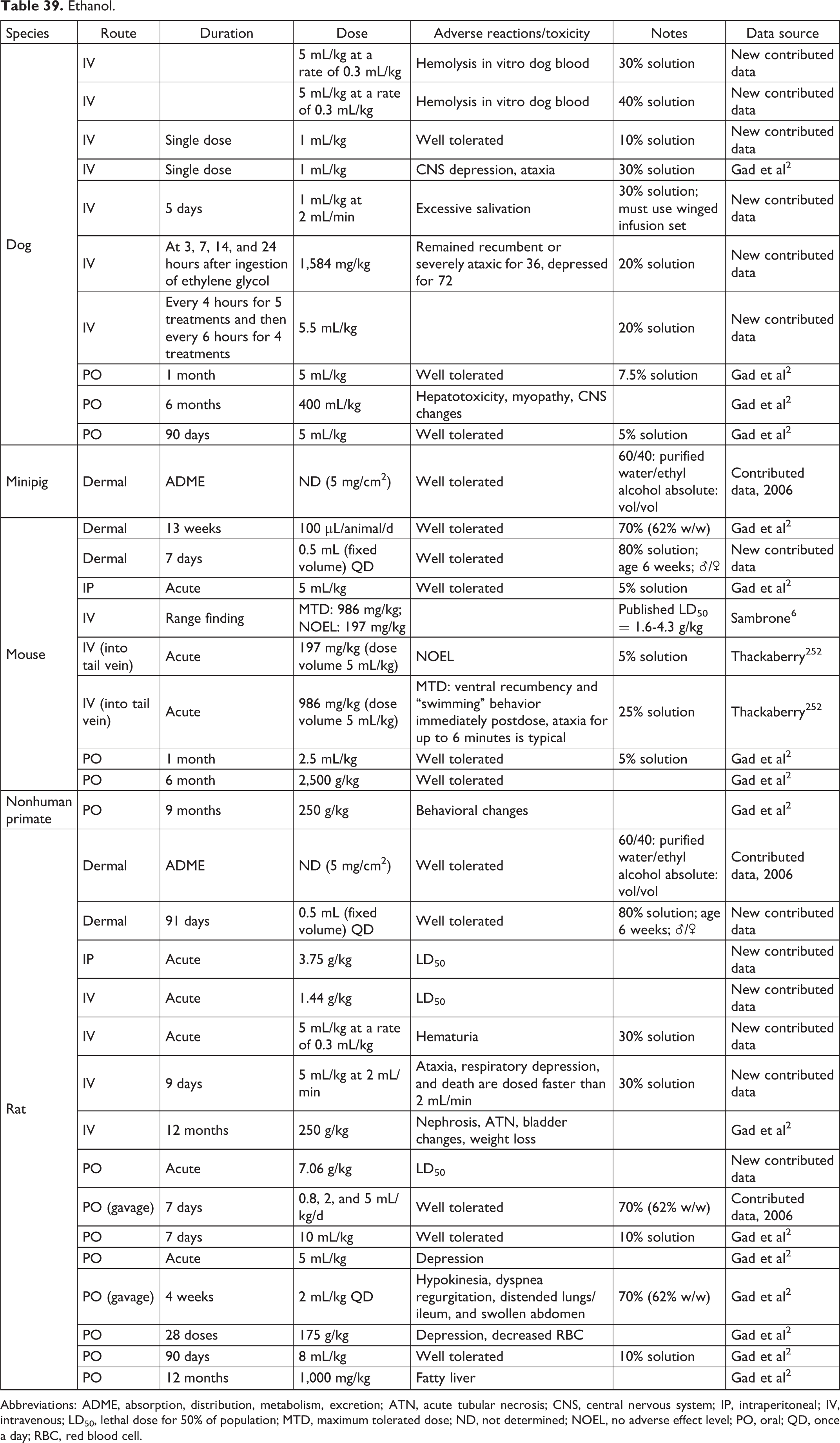
Abbreviations: ADME, absorption, distribution, metabolism, excretion; ATN, acute tubular necrosis; CNS, central nervous system; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; MTD, maximum tolerated dose; ND, not determined; NOEL, no adverse effect level; PO, oral; QD, once a day; RBC, red blood cell.
Gelatin Capsules.
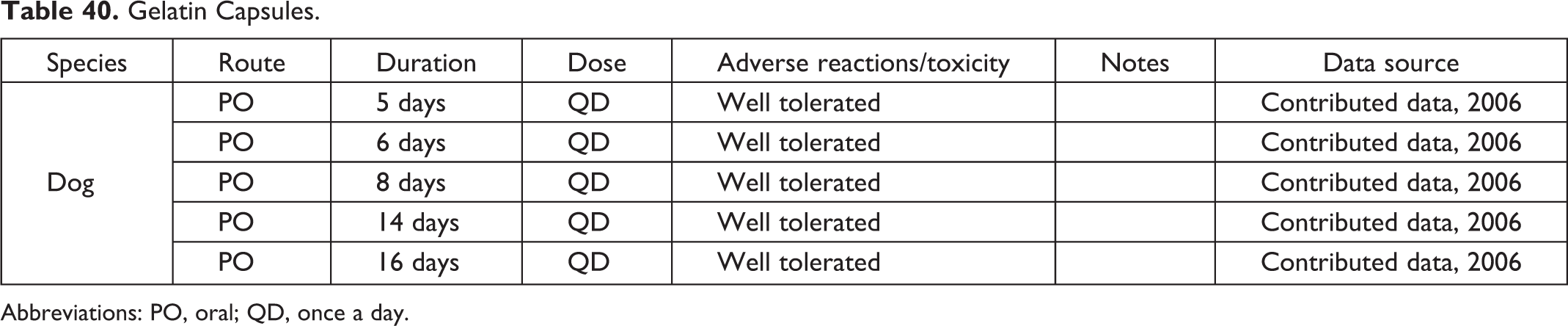
Abbreviations: PO, oral; QD, once a day.
Gelatin Phosphate Buffer.

Abbreviations: GLP, good laboratory practice; q14d, every 14 days.
Gelucire 44/14.
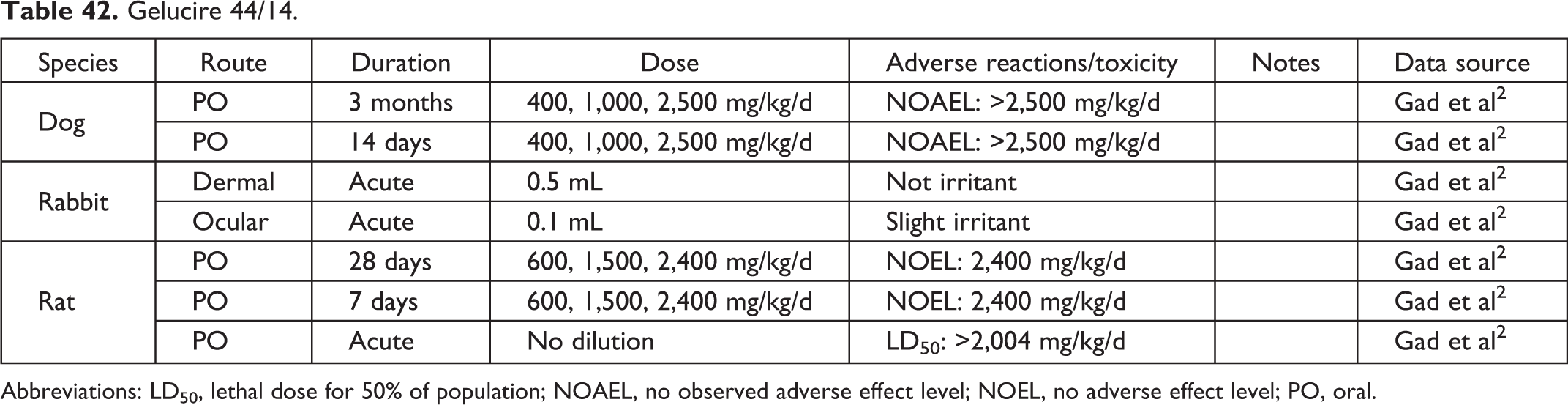
Abbreviations: LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; NOEL, no adverse effect level; PO, oral.
Gelucire 50/13.
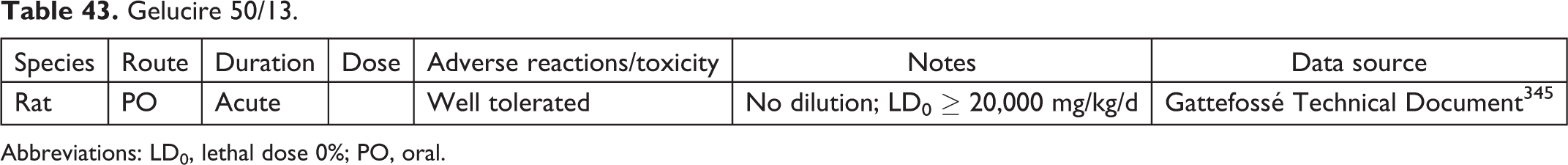
Abbreviations: LD0, lethal dose 0%; PO, oral.
Gluconic Acid.
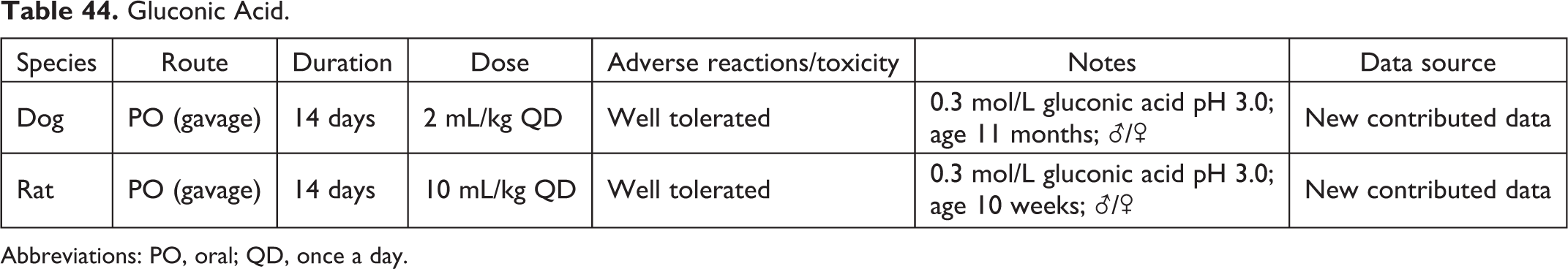
Abbreviations: PO, oral; QD, once a day.
Glycerol.
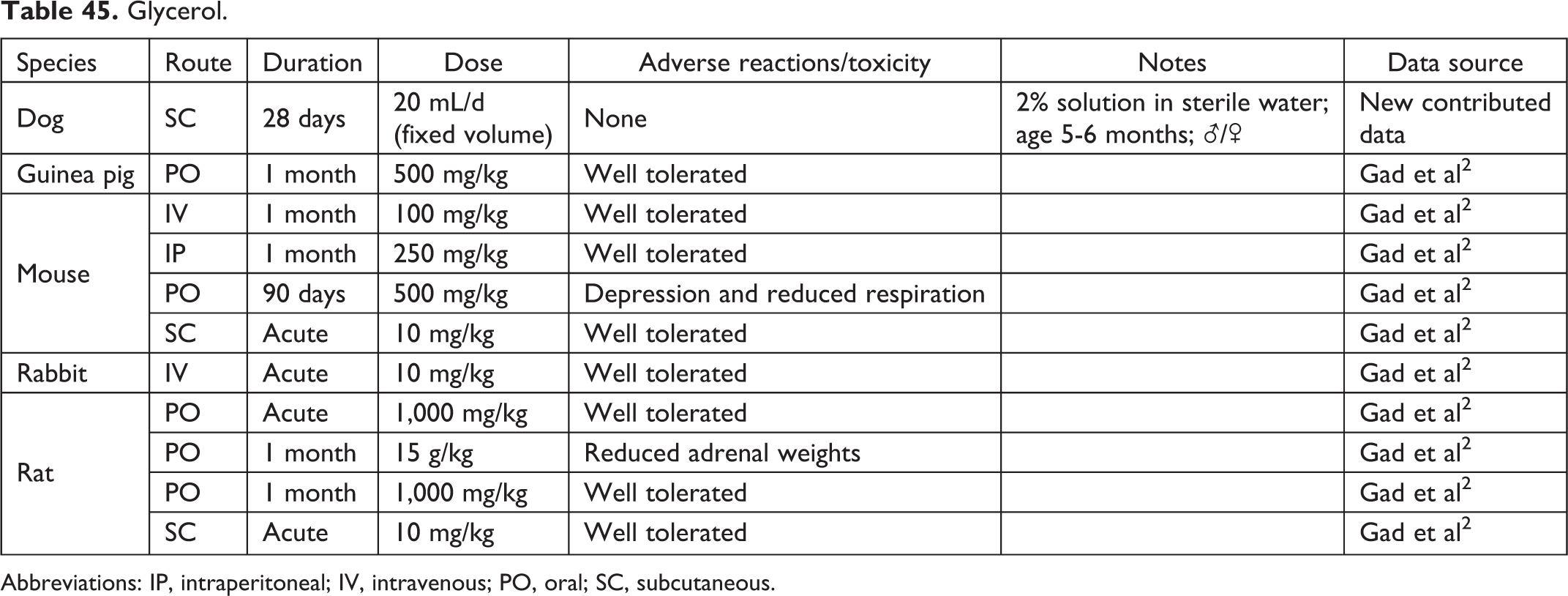
Abbreviations: IP, intraperitoneal; IV, intravenous; PO, oral; SC, subcutaneous.
Glycofurol.

Abbreviation: IV, intravenous.
Gum Tragacanth.
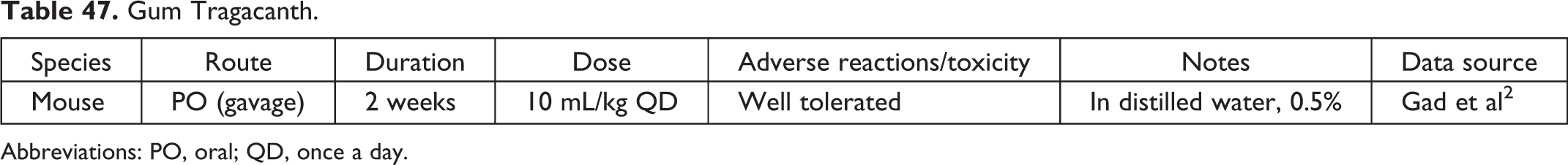
Abbreviations: PO, oral; QD, once a day.
Gum Xanthane.

Abbreviation: PO, oral.
Hydrochloric Acid.
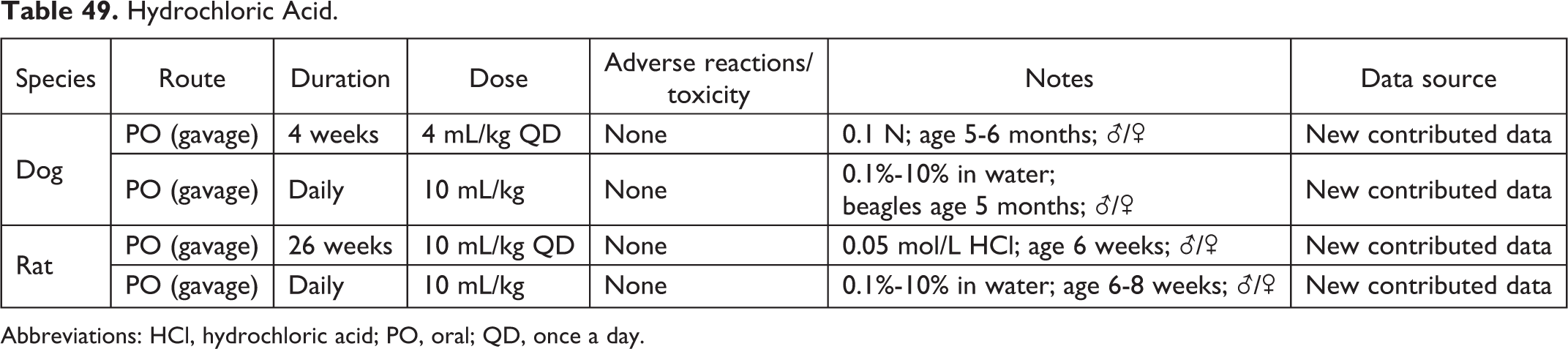
Abbreviations: HCl, hydrochloric acid; PO, oral; QD, once a day.
Hydroxyethyl Cellulose.

Abbreviation: PO, oral.
Hydroxypropyl-β-Cyclodextrin.a
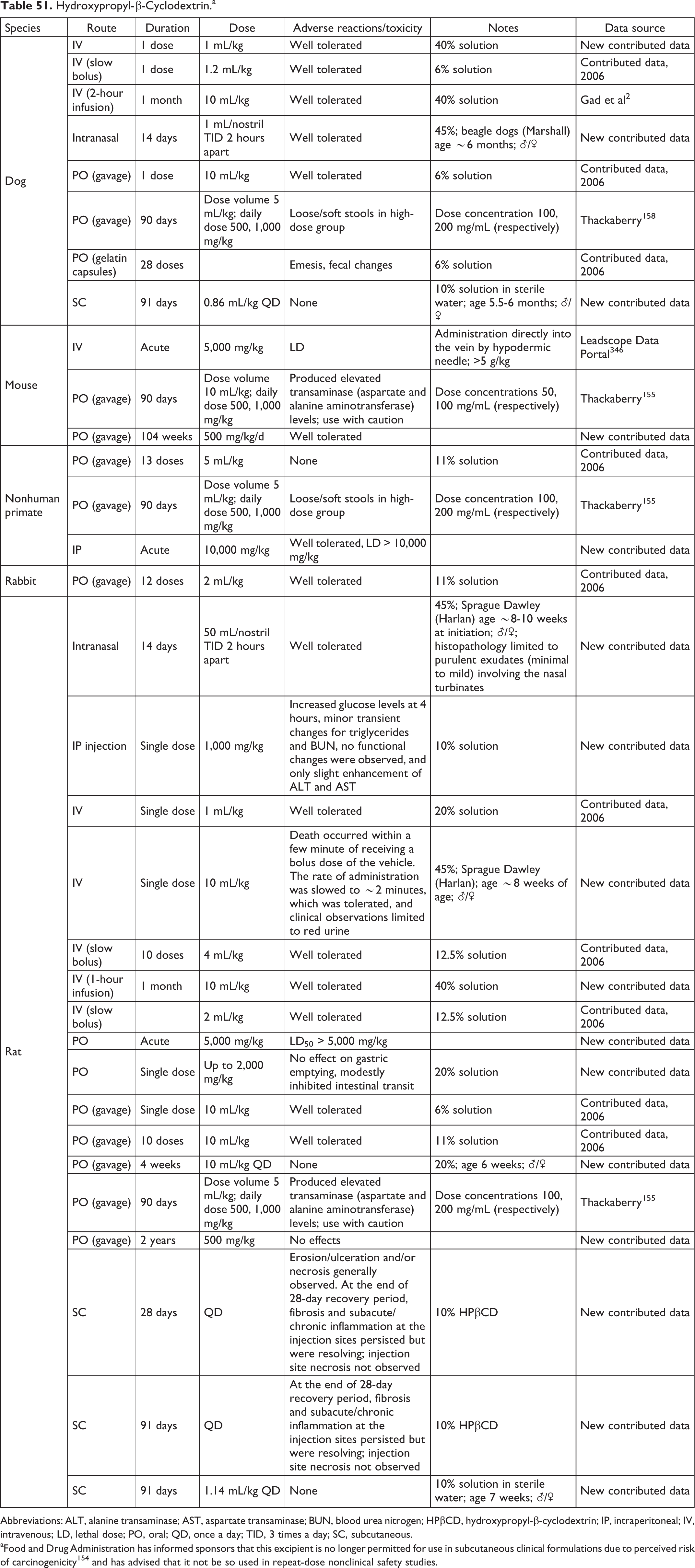
Abbreviations: ALT, alanine transaminase; AST, aspartate transaminase; BUN, blood urea nitrogen; HPβCD, hydroxypropyl-β-cyclodextrin; IP, intraperitoneal; IV, intravenous; LD, lethal dose; PO, oral; QD, once a day; TID, 3 times a day; SC, subcutaneous.
aFood and Drug Administration has informed sponsors that this excipient is no longer permitted for use in subcutaneous clinical formulations due to perceived risk of carcinogenicity 154 and has advised that it not be so used in repeat-dose nonclinical safety studies.
Hydroxypropyl Cellulose.

Abbreviation: PO, oral.
Hydroxypropyl Methylcellulose.
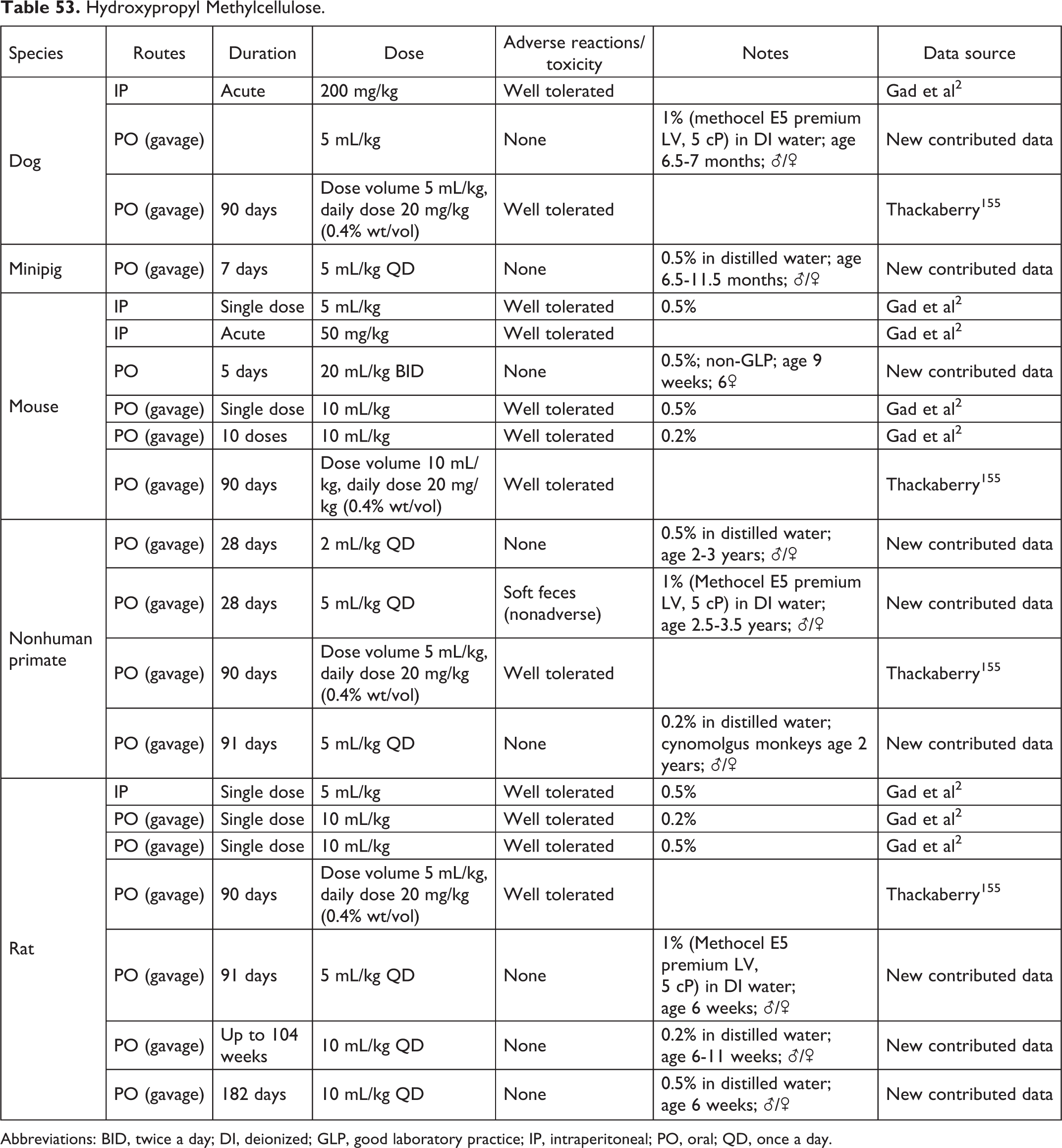
Abbreviations: BID, twice a day; DI, deionized; GLP, good laboratory practice; IP, intraperitoneal; PO, oral; QD, once a day.
Hypotonic Phosphate-Buffered Saline.

Abbreviations: GLP, good laboratory practice; IV, intravenous; SD, single dose.
Isopropyl Alcohol.

Isopropyl Myristate.

Kolliphor EL.
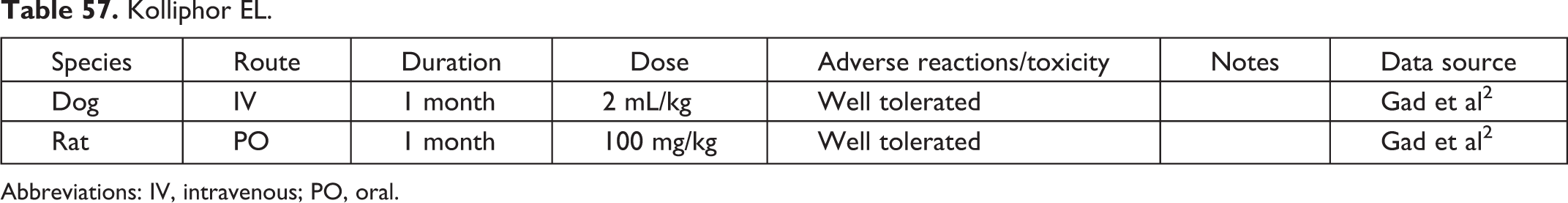
Abbreviations: IV, intravenous; PO, oral.
Kolliphor ELP.

Kolliphor RH40.
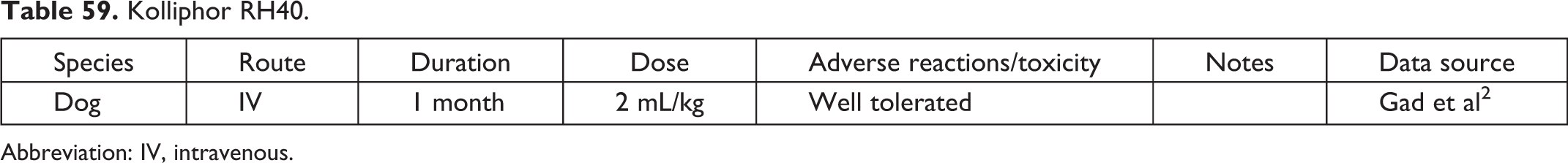
Abbreviation: IV, intravenous.
Labrafil M1944.
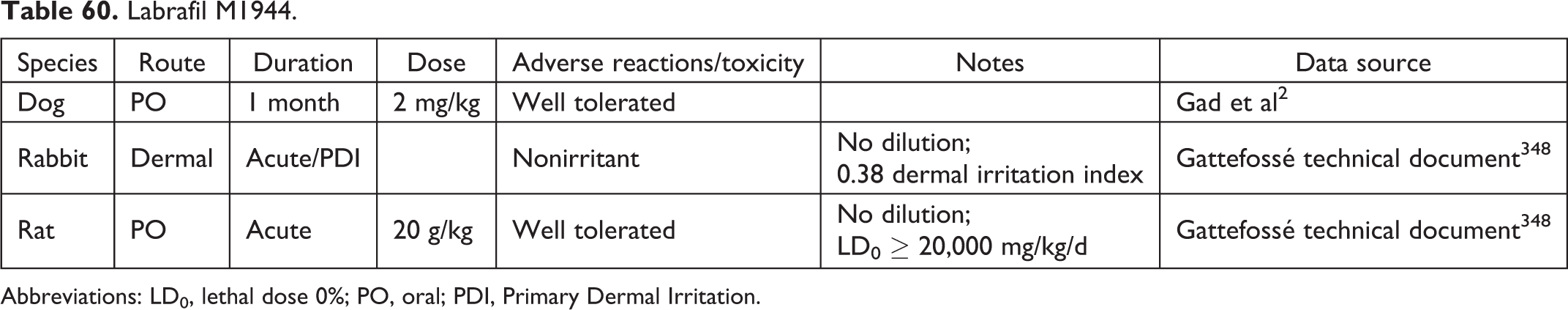
Abbreviations: LD0, lethal dose 0%; PO, oral; PDI, Primary Dermal Irritation.
Labrasol.
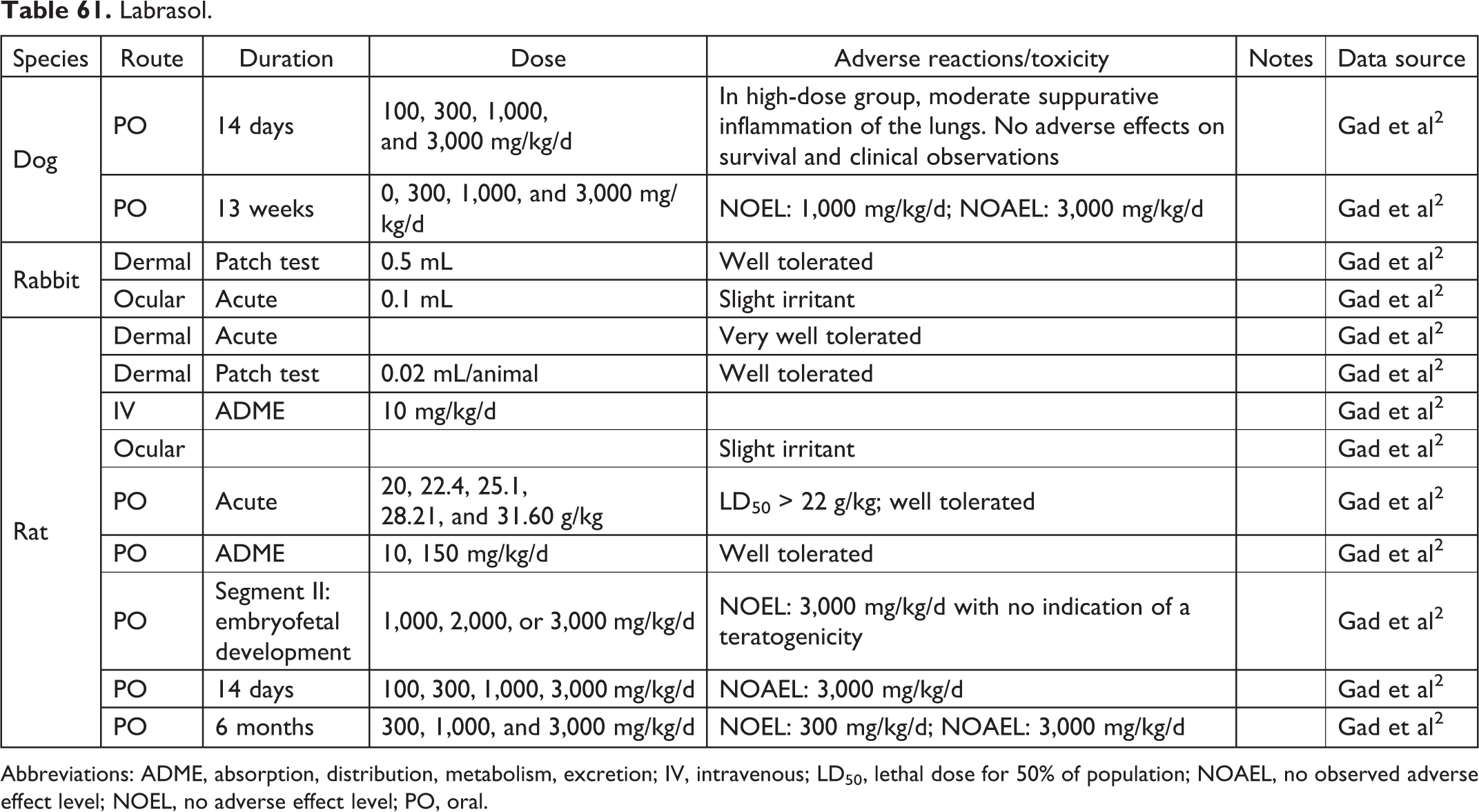
Abbreviations: ADME, absorption, distribution, metabolism, excretion; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; NOEL, no adverse effect level; PO, oral.
Lactose.

Lanolin.

Lauroglycol 90.
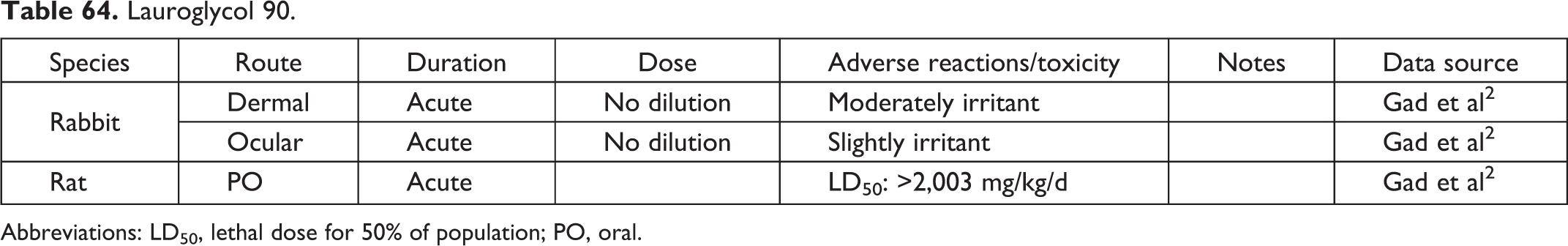
Abbreviations: LD50, lethal dose for 50% of population; PO, oral.
Maltitol Solution.

Abbreviation: IP, intraperitoneal.
Maltol.
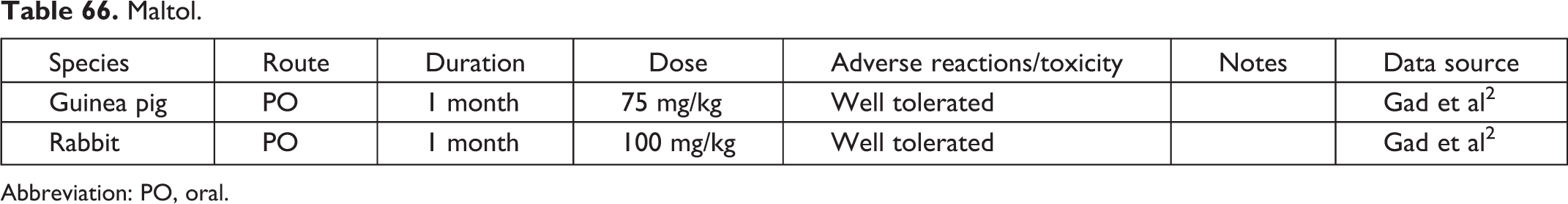
Abbreviation: PO, oral.
Mannitol.
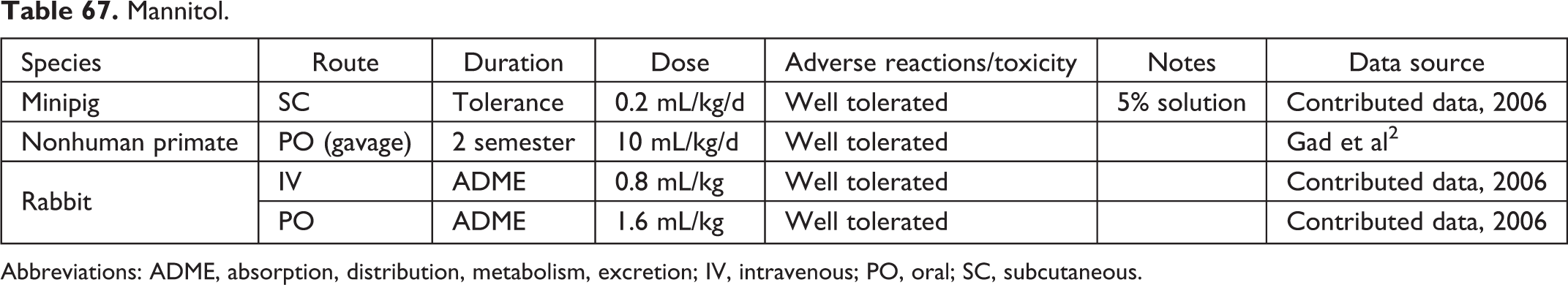
Abbreviations: ADME, absorption, distribution, metabolism, excretion; IV, intravenous; PO, oral; SC, subcutaneous.
Methylcellulose.
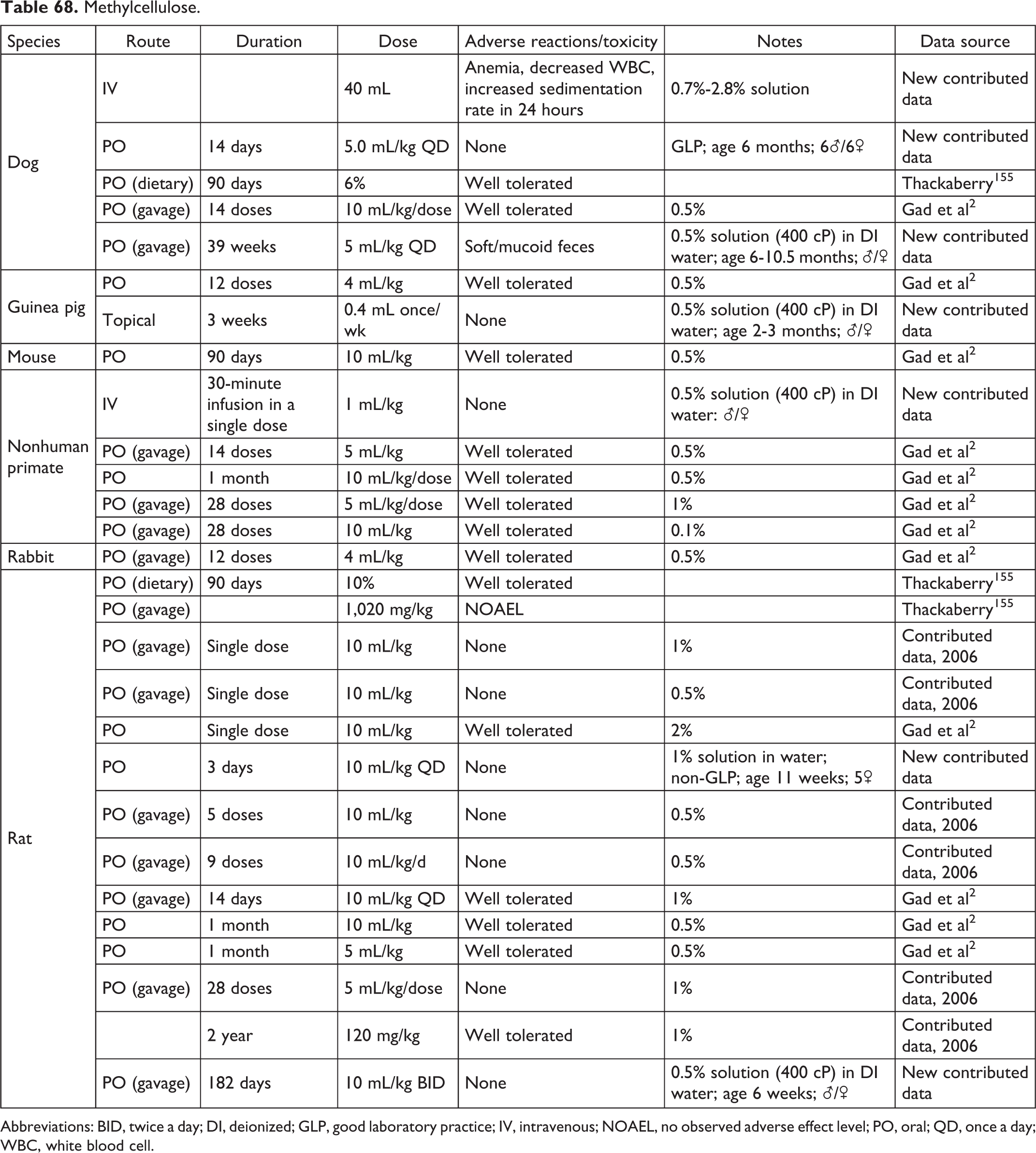
Abbreviations: BID, twice a day; DI, deionized; GLP, good laboratory practice; IV, intravenous; NOAEL, no observed adverse effect level; PO, oral; QD, once a day; WBC, white blood cell.
Methylpyrrolidone.
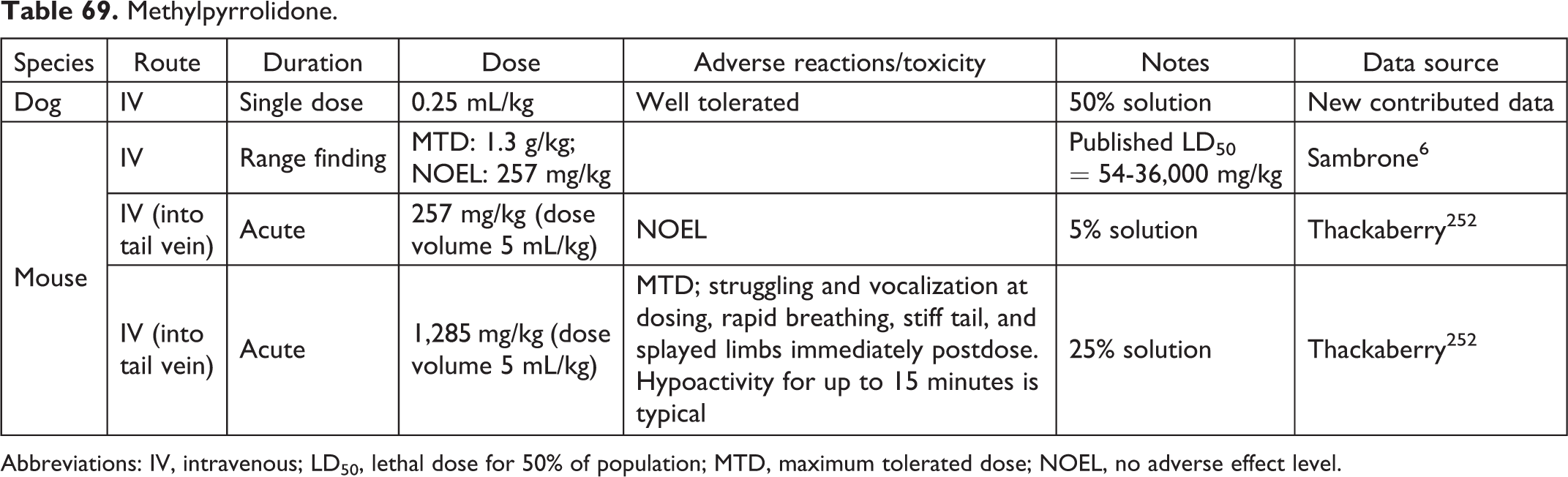
Abbreviations: IV, intravenous; LD50, lethal dose for 50% of population; MTD, maximum tolerated dose; NOEL, no adverse effect level.
Mineral Oil.
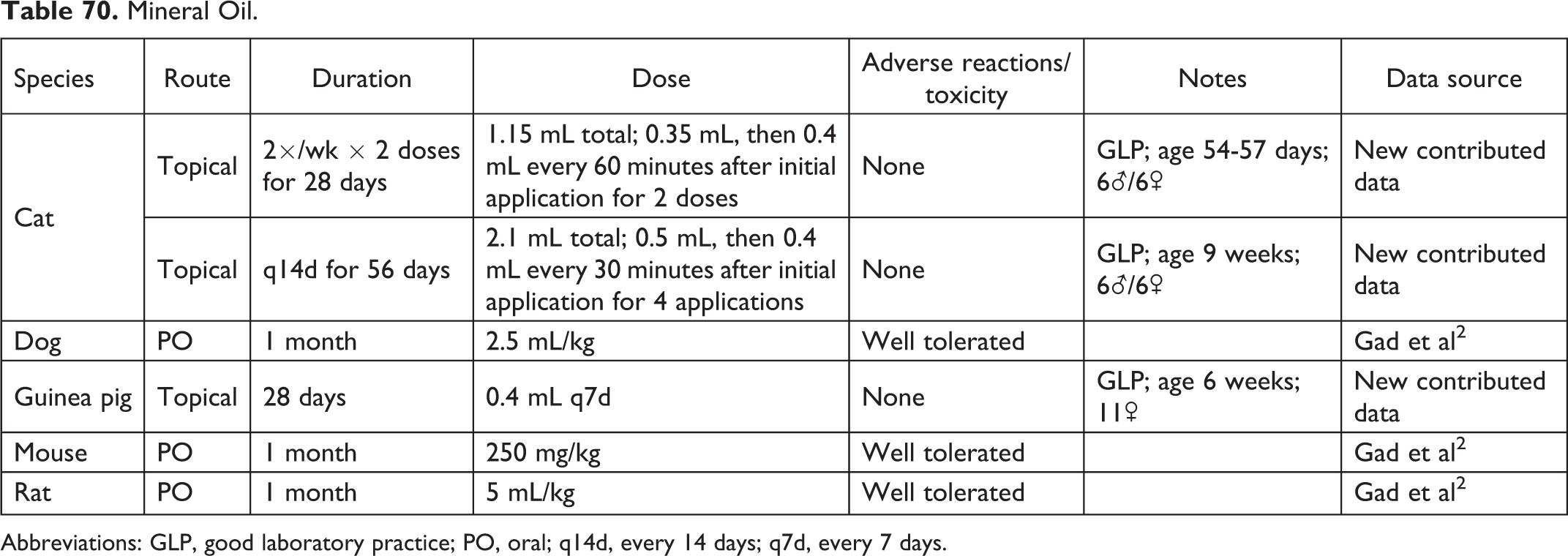
Abbreviations: GLP, good laboratory practice; PO, oral; q14d, every 14 days; q7d, every 7 days.
Olive Oil.

Abbreviations: PO, oral; QD, once a day.
Peanut Oil.
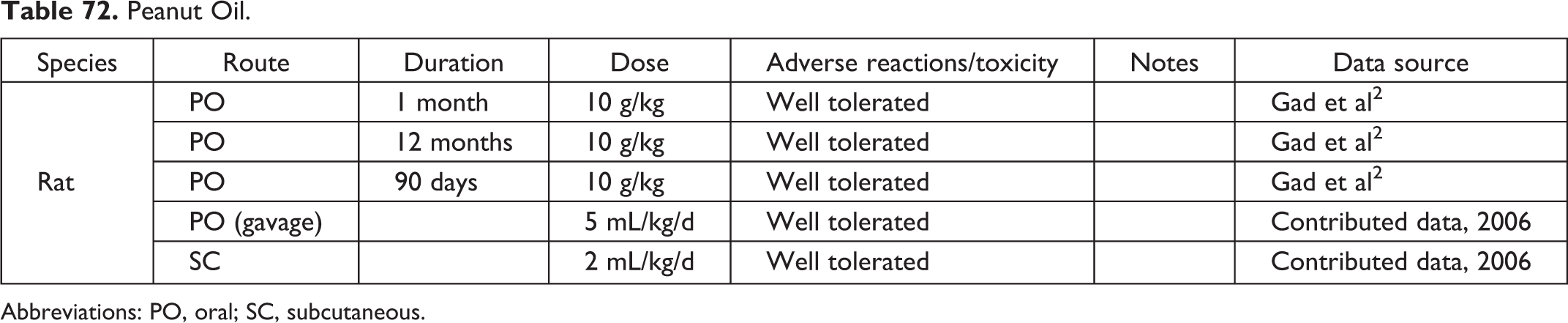
Abbreviations: PO, oral; SC, subcutaneous.
PEG 200.
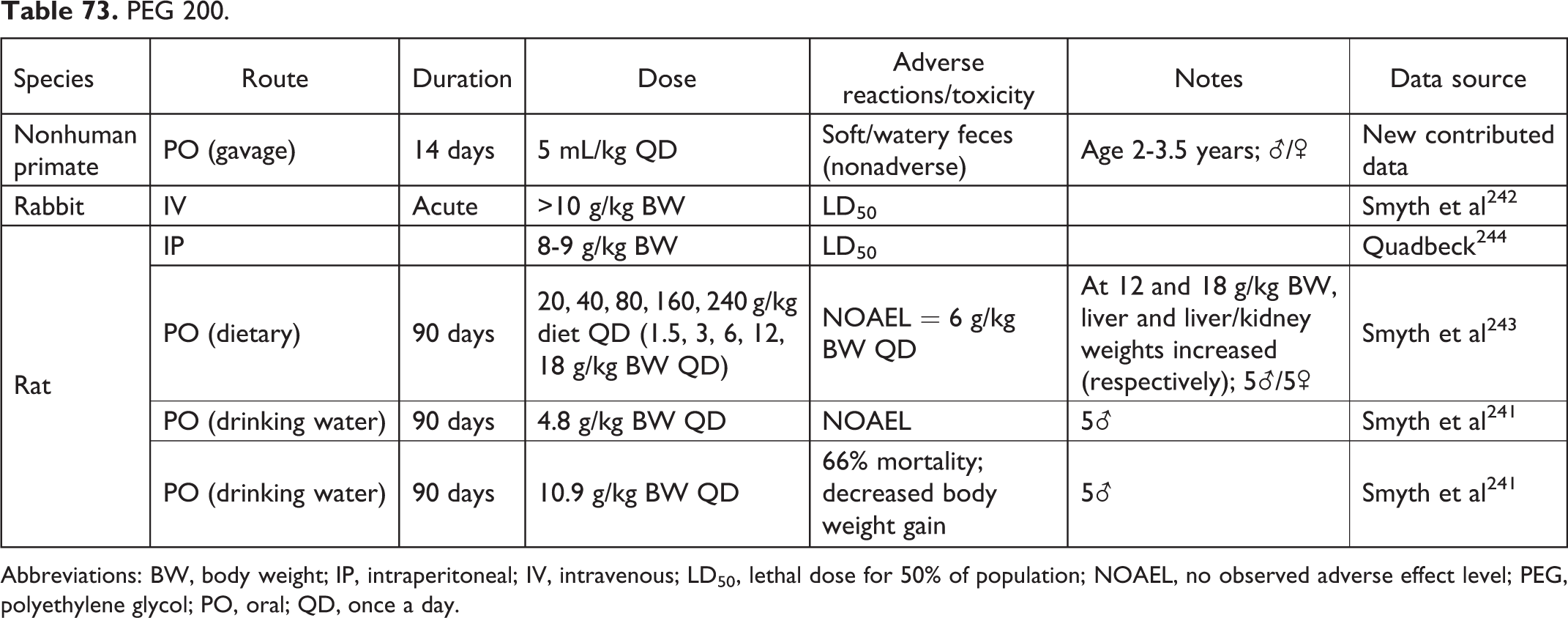
Abbreviations: BW, body weight; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
PEG 300.
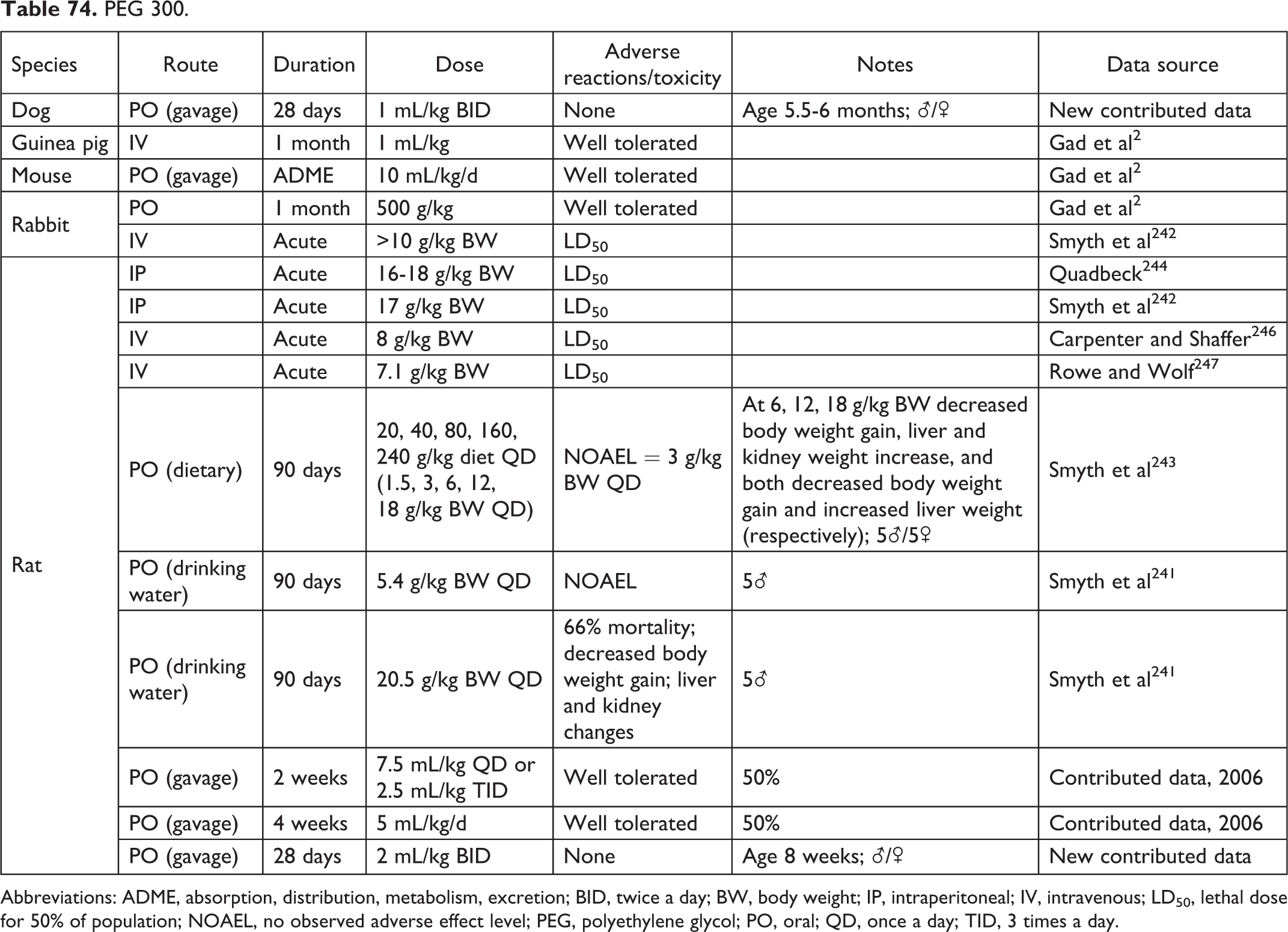
Abbreviations: ADME, absorption, distribution, metabolism, excretion; BID, twice a day; BW, body weight; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day; TID, 3 times a day.
PEG 400.
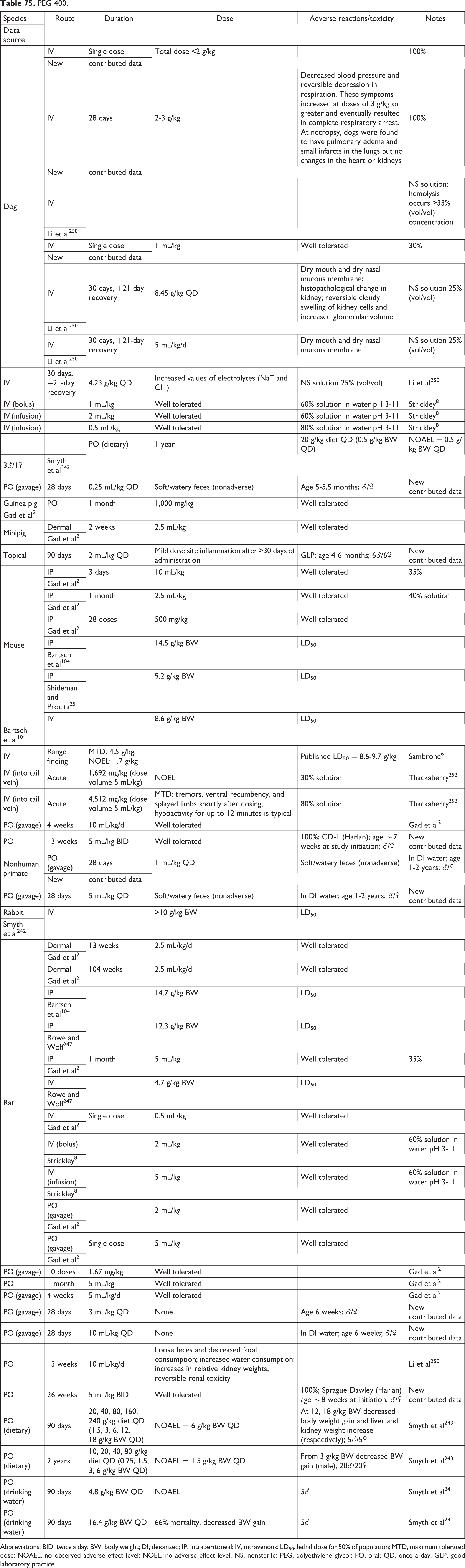
Abbreviations: BID, twice a day; BW, body weight; DI, deionized; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; MTD, maximum tolerated dose; NOAEL, no observed adverse effect level; NOEL, no adverse effect level; NS, nonsterile; PEG, polyethylene glycol; PO, oral; QD, once a day; GLP, good laboratory practice.
PEG 600.
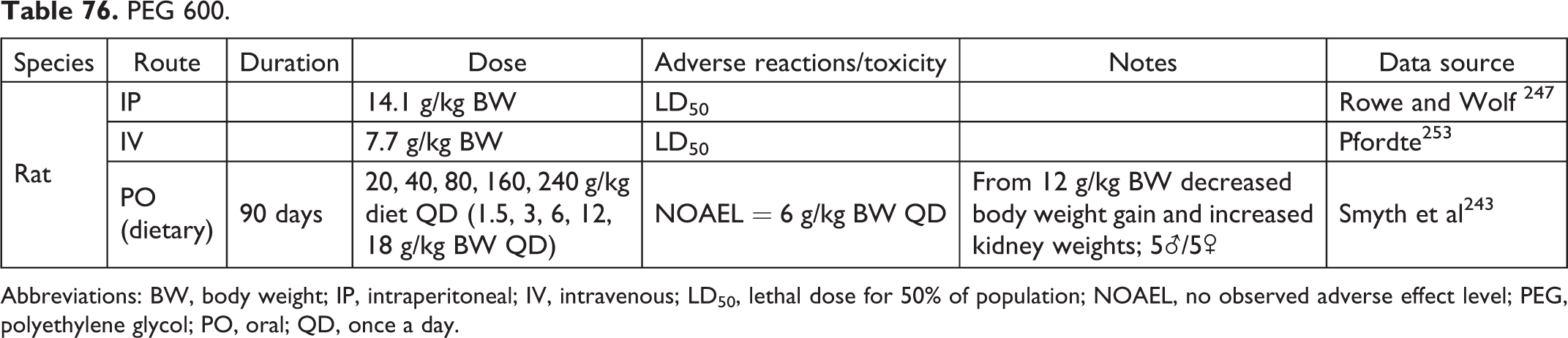
Abbreviations: BW, body weight; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
PEG 810.

Abbreviations: BW, body weight; IV, intravenous; LD50, lethal dose for 50% of population; PEG, polyethylene glycol; SC, subcutaneous.
PEG 1,000.
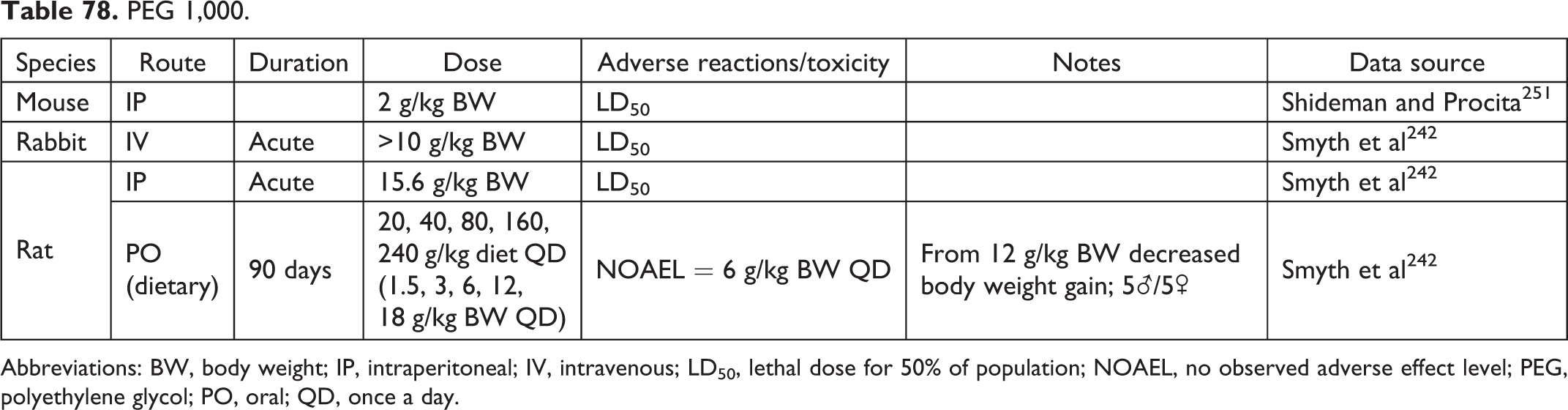
Abbreviations: BW, body weight; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
PEG 1,500.
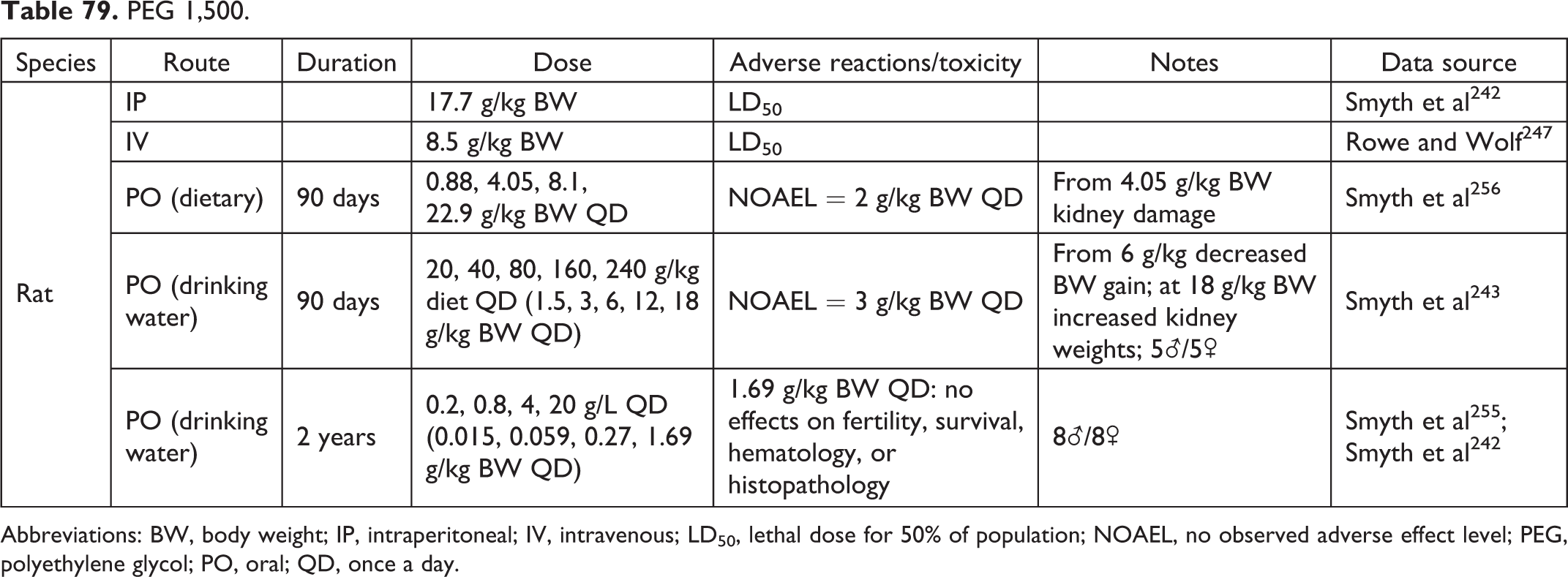
Abbreviations: BW, body weight; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
PEG 1,540.
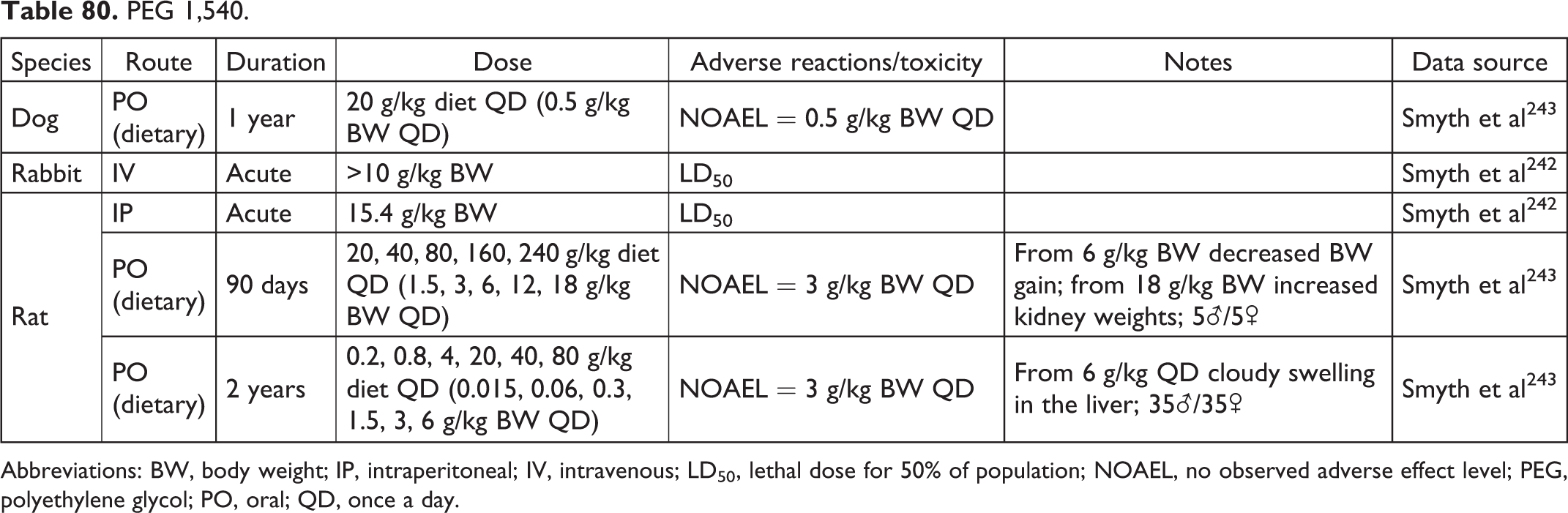
Abbreviations: BW, body weight; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
PEG 4,000.
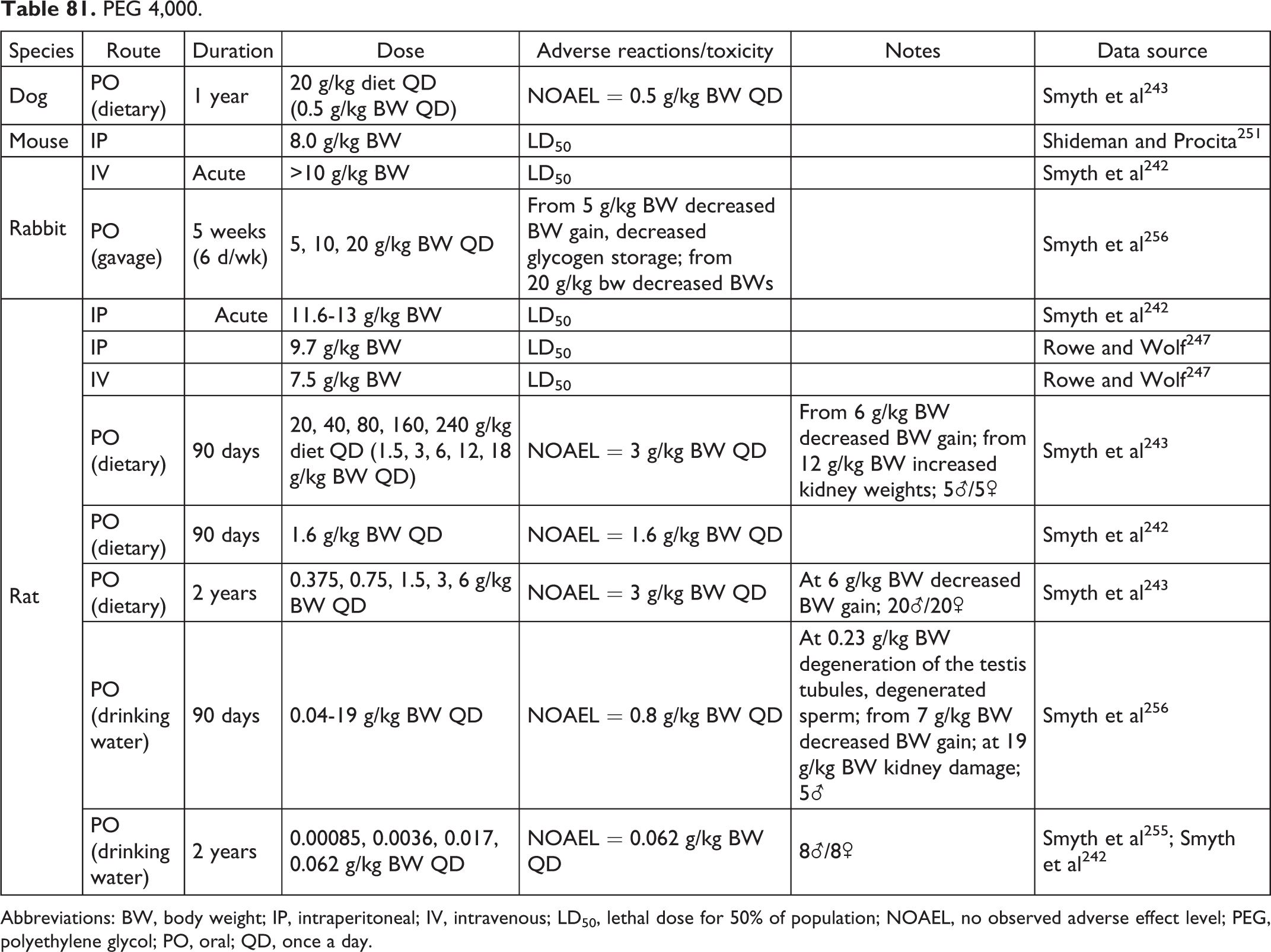
Abbreviations: BW, body weight; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
PEG 6,000.
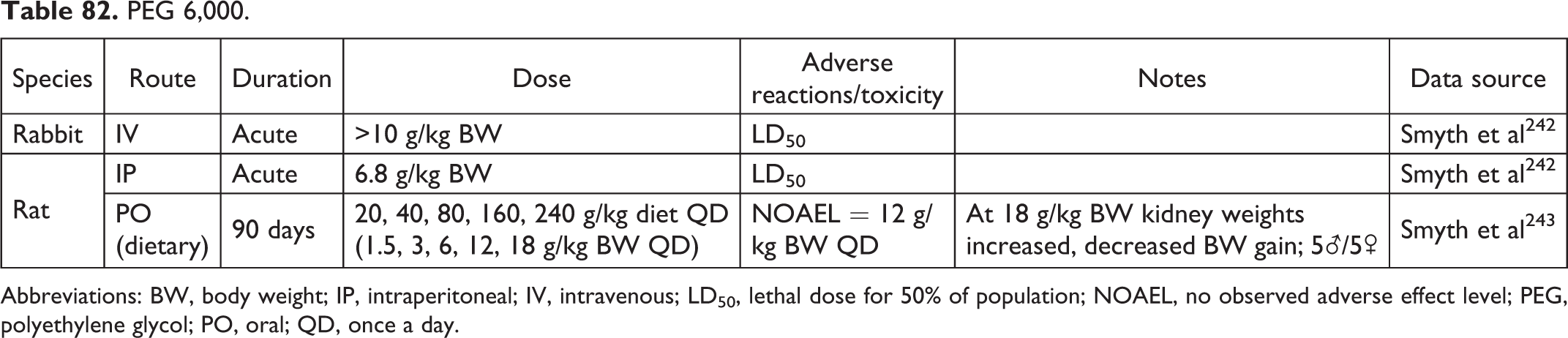
Abbreviations: BW, body weight; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
PEG 10,000.

Abbreviations: BW, body weight; IP, intraperitoneal; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
PEG 4,000,000.

Abbreviations: BW, body weight; IV, intravenous; LD50, lethal dose for 50% of population; NOAEL, no observed adverse effect level; PEG, polyethylene glycol; PO, oral; QD, once a day.
Petrolatum.

Phosphate-Buffered Saline.
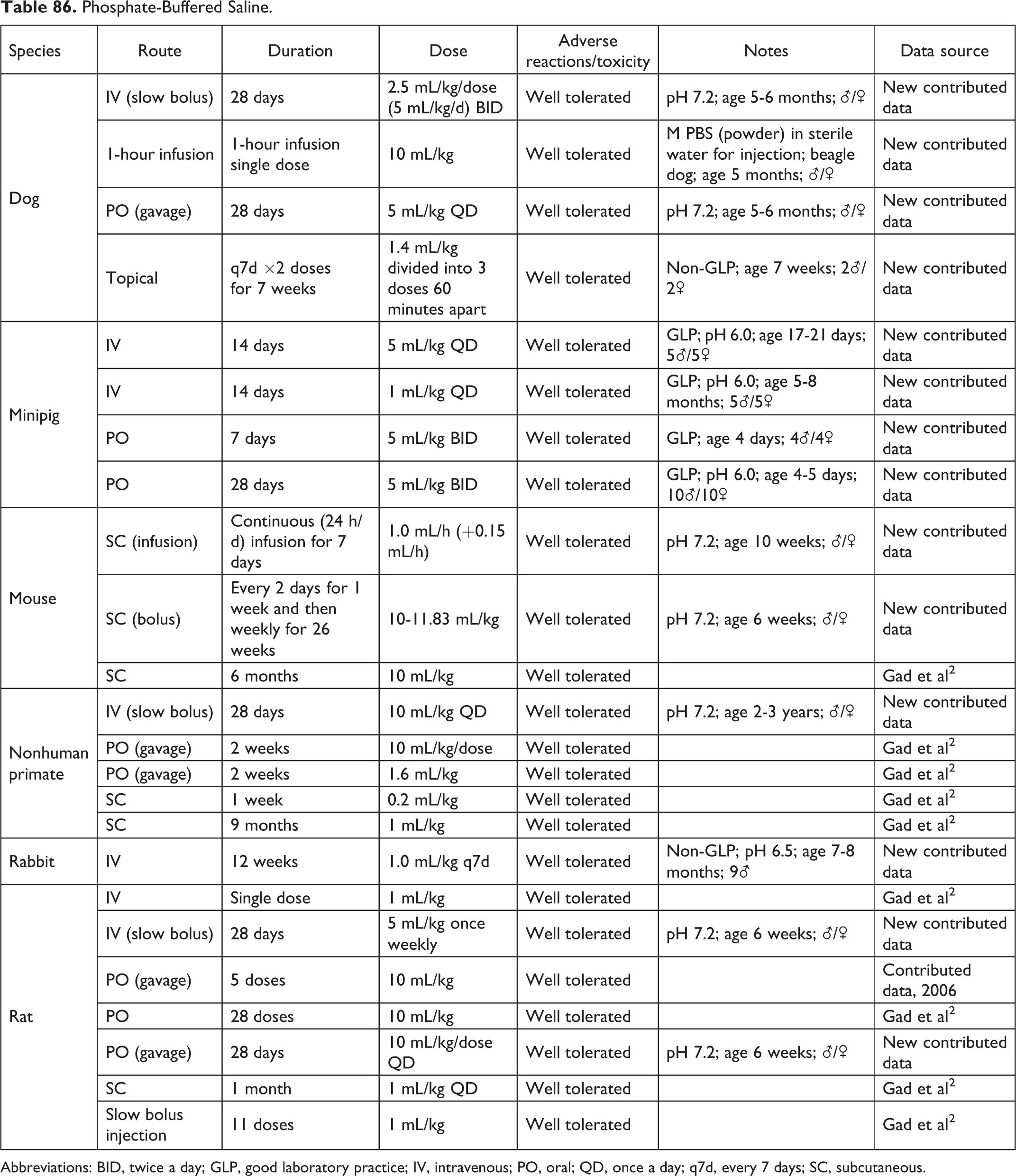
Abbreviations: BID, twice a day; GLP, good laboratory practice; IV, intravenous; PO, oral; QD, once a day; q7d, every 7 days; SC, subcutaneous.
Poloxamer 188.

Abbreviations: DI, deionized; PO, oral; SC, subcutaneous.
Poly(glycolide-co-dl-lactide) Microspheres.

Abbreviations: GLP, good laboratory practice; SD, single dose.
Polyglycerol Oleate.

Abbreviations: LD50, lethal dose for 50% of population; PO, oral.
Polyvinylpyrrolidone.

Abbreviations: IM, intramuscular; PO, oral.
Propylene Glycol.
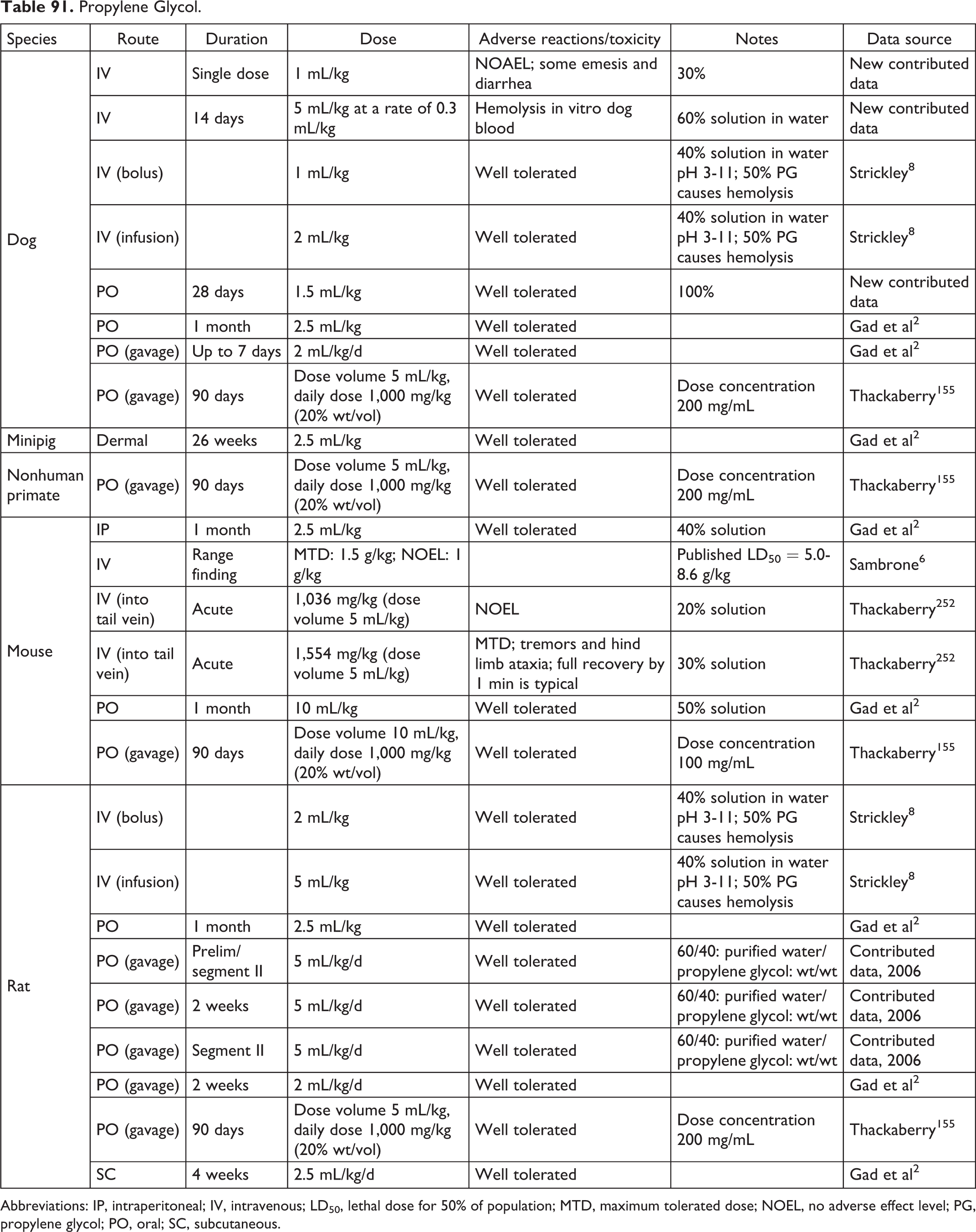
Abbreviations: IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; MTD, maximum tolerated dose; NOEL, no adverse effect level; PG, propylene glycol; PO, oral; SC, subcutaneous.
Randomly Methylated β-Cyclodextrins.
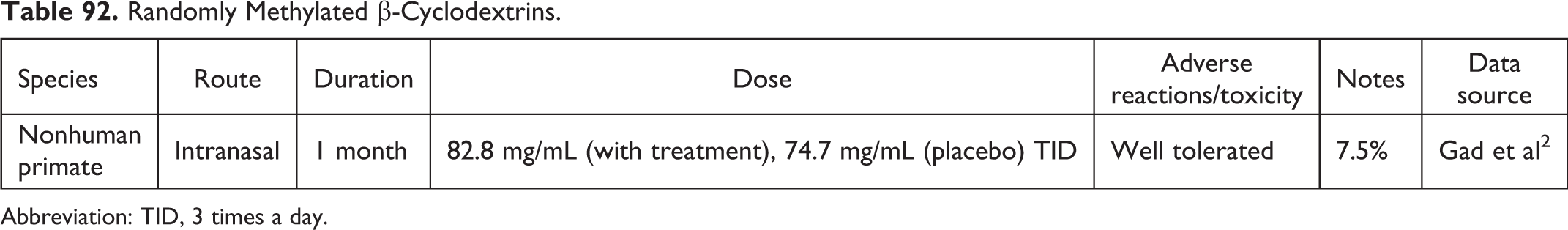
Abbreviation: TID, 3 times a day.
Safflower Oil.

Abbreviation: SC, subcutaneous.
Saline (pH Adjusted).
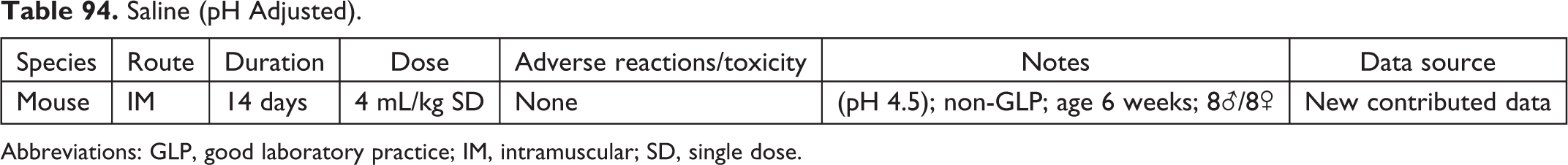
Abbreviations: GLP, good laboratory practice; IM, intramuscular; SD, single dose.
Sesame Oil.
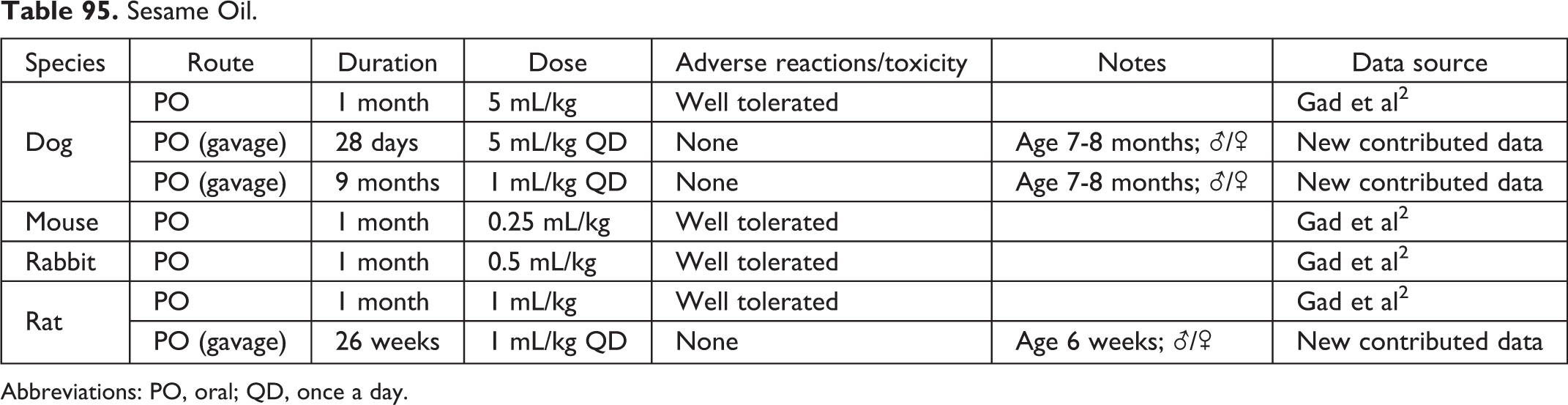
Abbreviations: PO, oral; QD, once a day.
Sodium Acetate Trihydrate Buffer.
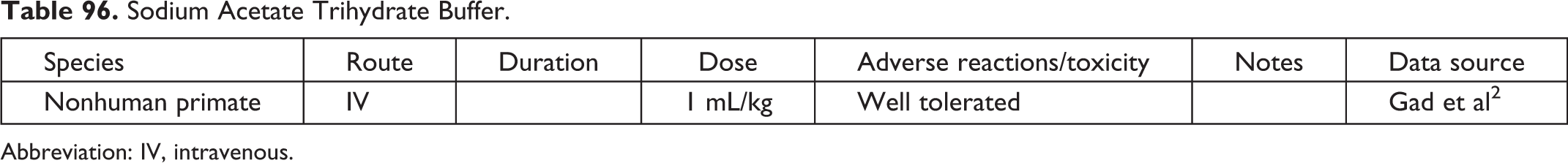
Abbreviation: IV, intravenous.
Sodium Chloride.
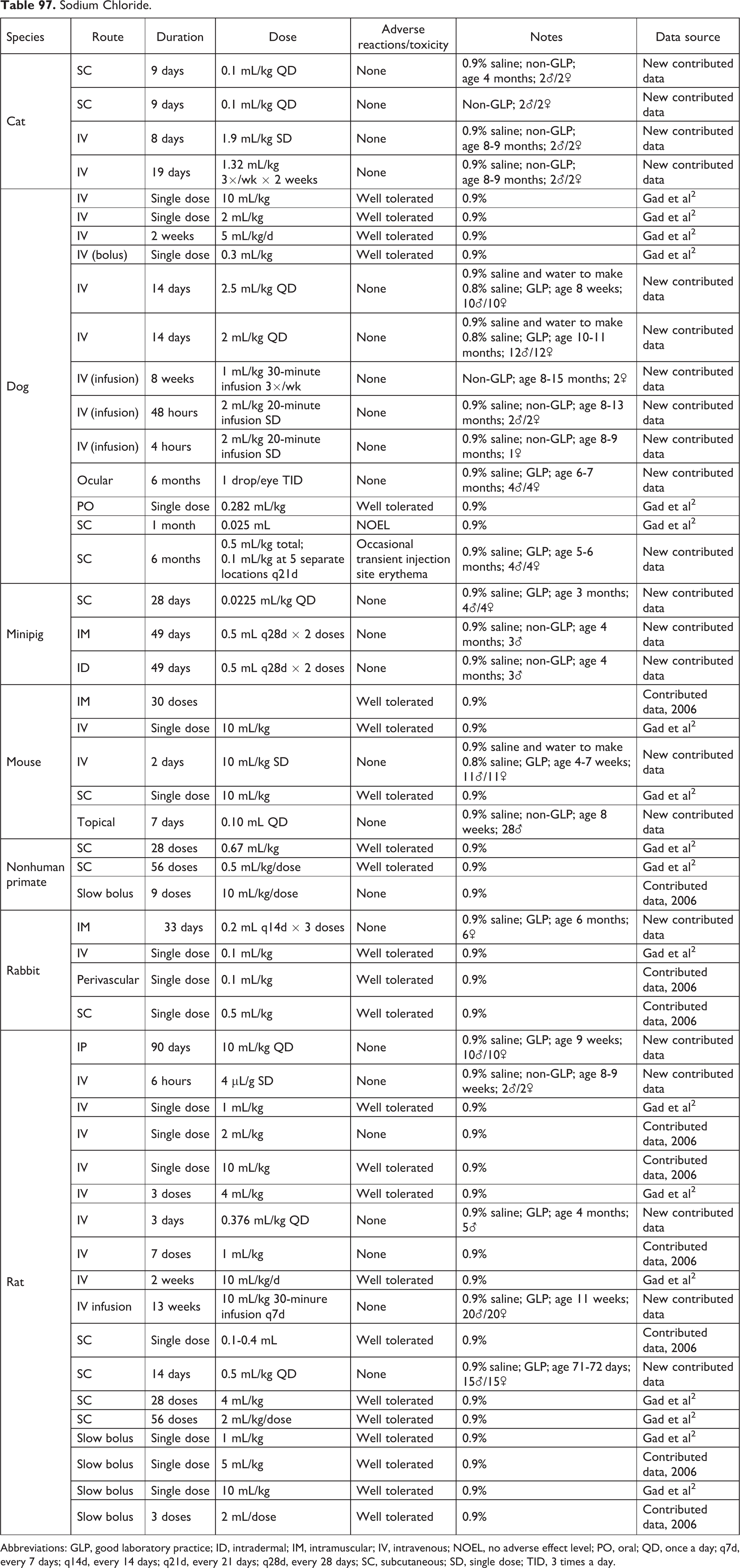
Abbreviations: GLP, good laboratory practice; ID, intradermal; IM, intramuscular; IV, intravenous; NOEL, no adverse effect level; PO, oral; QD, once a day; q7d, every 7 days; q14d, every 14 days; q21d, every 21 days; q28d, every 28 days; SC, subcutaneous; SD, single dose; TID, 3 times a day.
Sodium Dihydrogen Phosphate Dihydrate.
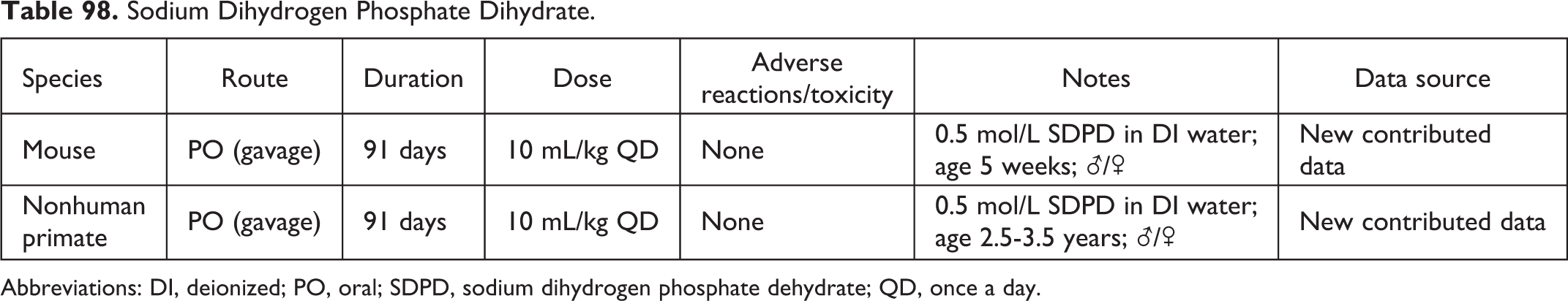
Abbreviations: DI, deionized; PO, oral; SDPD, sodium dihydrogen phosphate dehydrate; QD, once a day.
Sodium Metabisulfite.
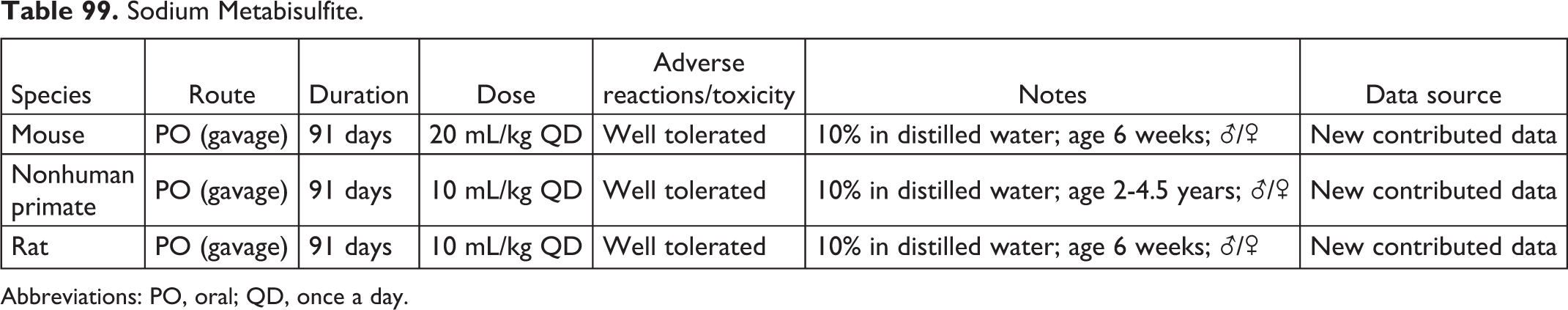
Abbreviations: PO, oral; QD, once a day.
Sodium Phosphate Buffer.
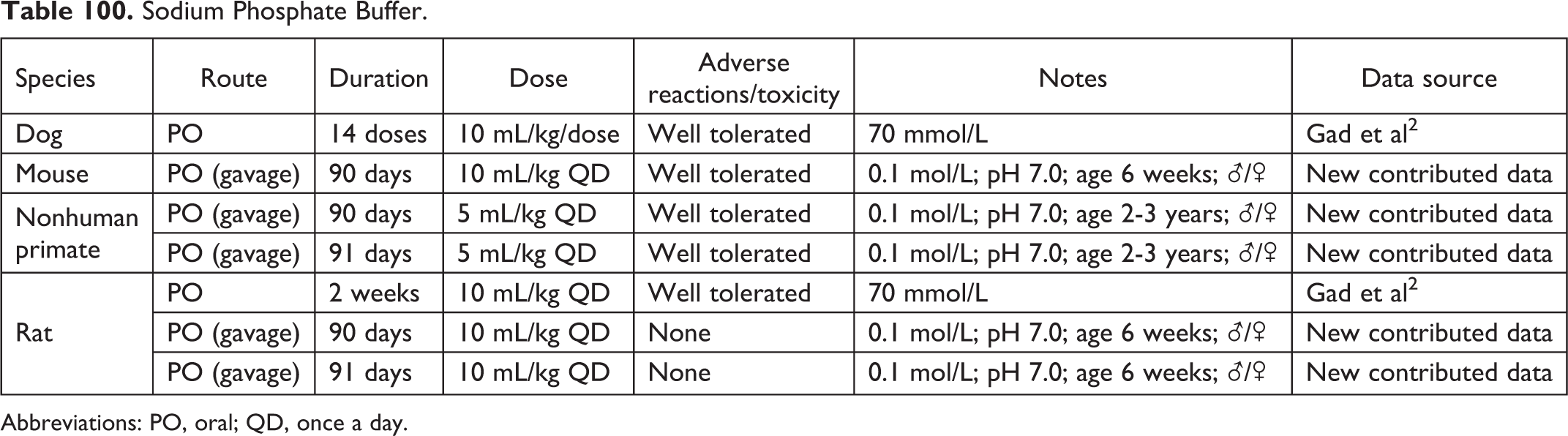
Abbreviations: PO, oral; QD, once a day.
Sodium Sulfite.

Abbreviations: DI, deionized; QID, 4 times a day.
Solutol HS15.
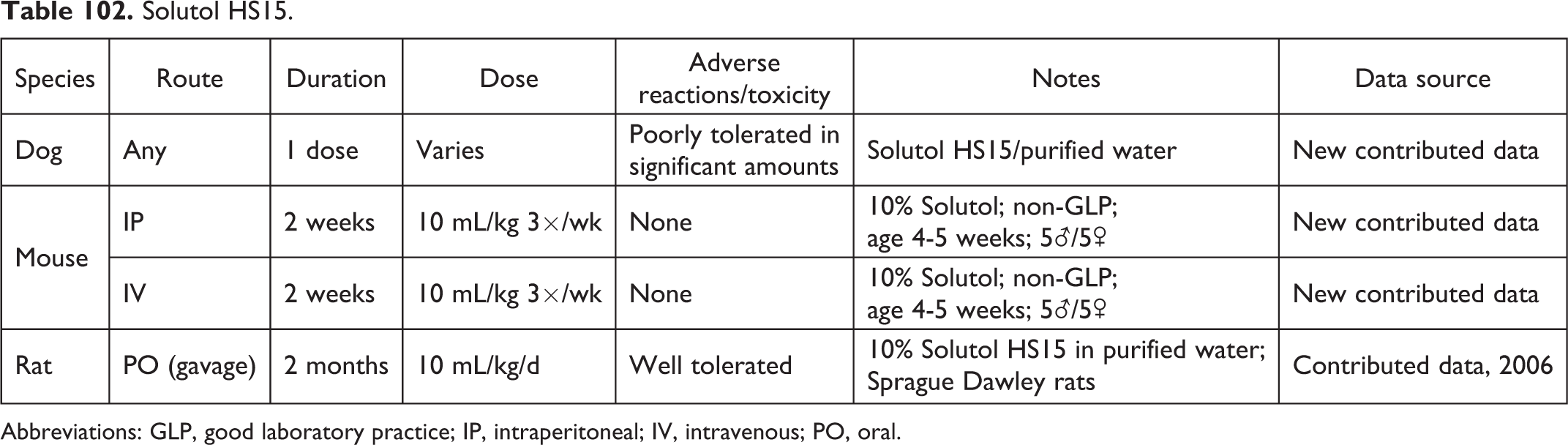
Abbreviations: GLP, good laboratory practice; IP, intraperitoneal; IV, intravenous; PO, oral.
Soybean Oil.
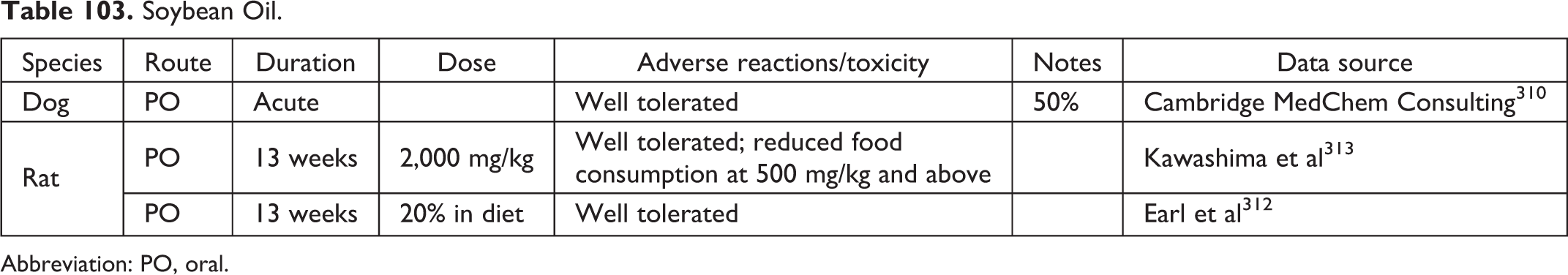
Abbreviation: PO, oral.
Sulfobutylether-β-cyclodextrin.
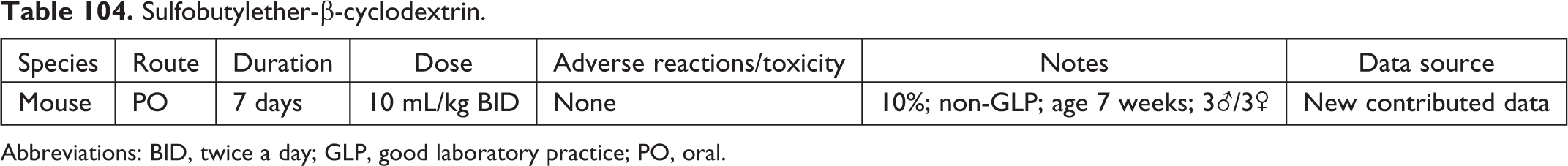
Abbreviations: BID, twice a day; GLP, good laboratory practice; PO, oral.
Tartaric Acid.
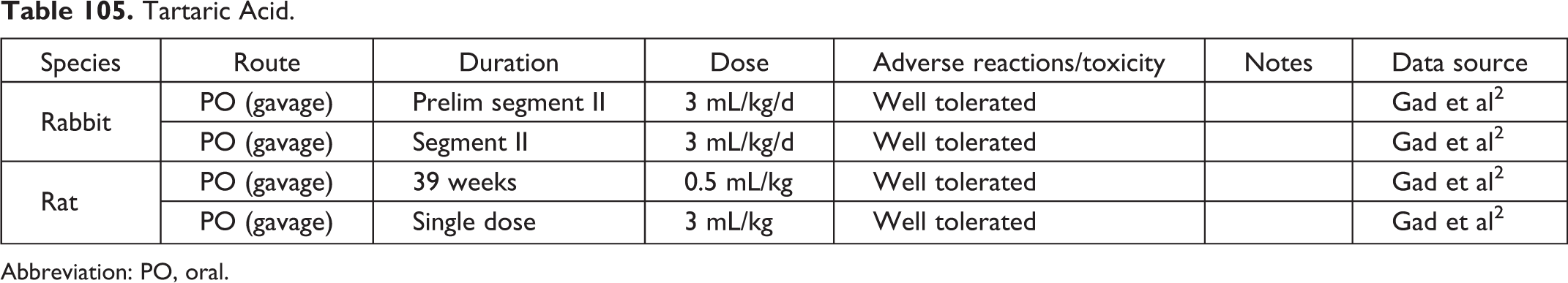
Abbreviation: PO, oral.
Terbinafine HCl Placebo Nail Lacquer.

Transcutol.
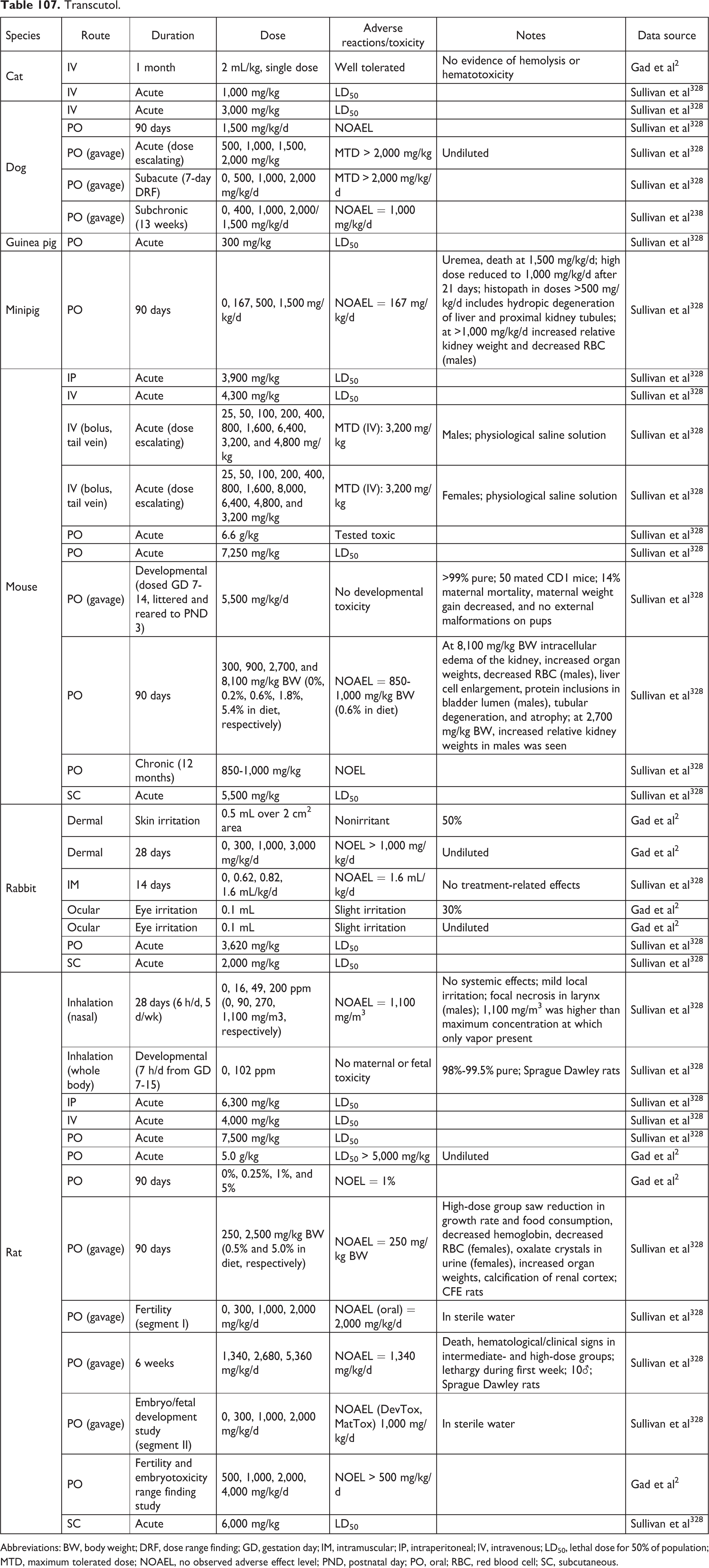
Abbreviations: BW, body weight; DRF, dose range finding; GD, gestation day; IM, intramuscular; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; MTD, maximum tolerated dose; NOAEL, no observed adverse effect level; PND, postnatal day; PO, oral; RBC, red blood cell; SC, subcutaneous.
Trisodium Citrate Dihydrate.
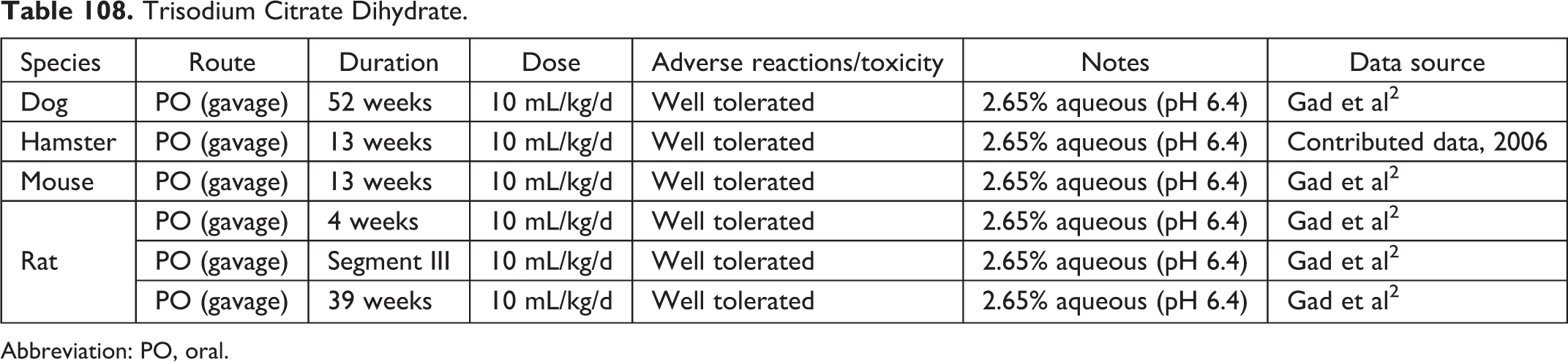
Abbreviation: PO, oral.
Tween 20.
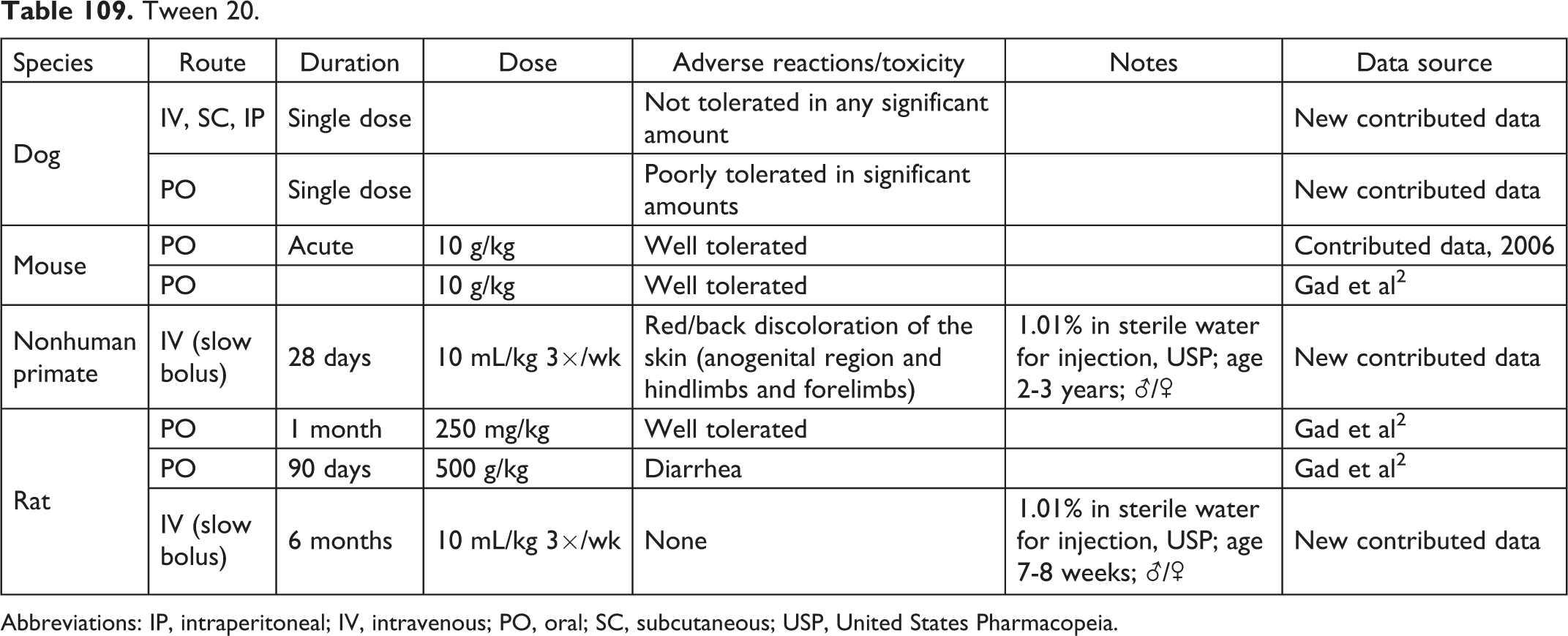
Abbreviations: IP, intraperitoneal; IV, intravenous; PO, oral; SC, subcutaneous; USP, United States Pharmacopeia.
Tween 80.
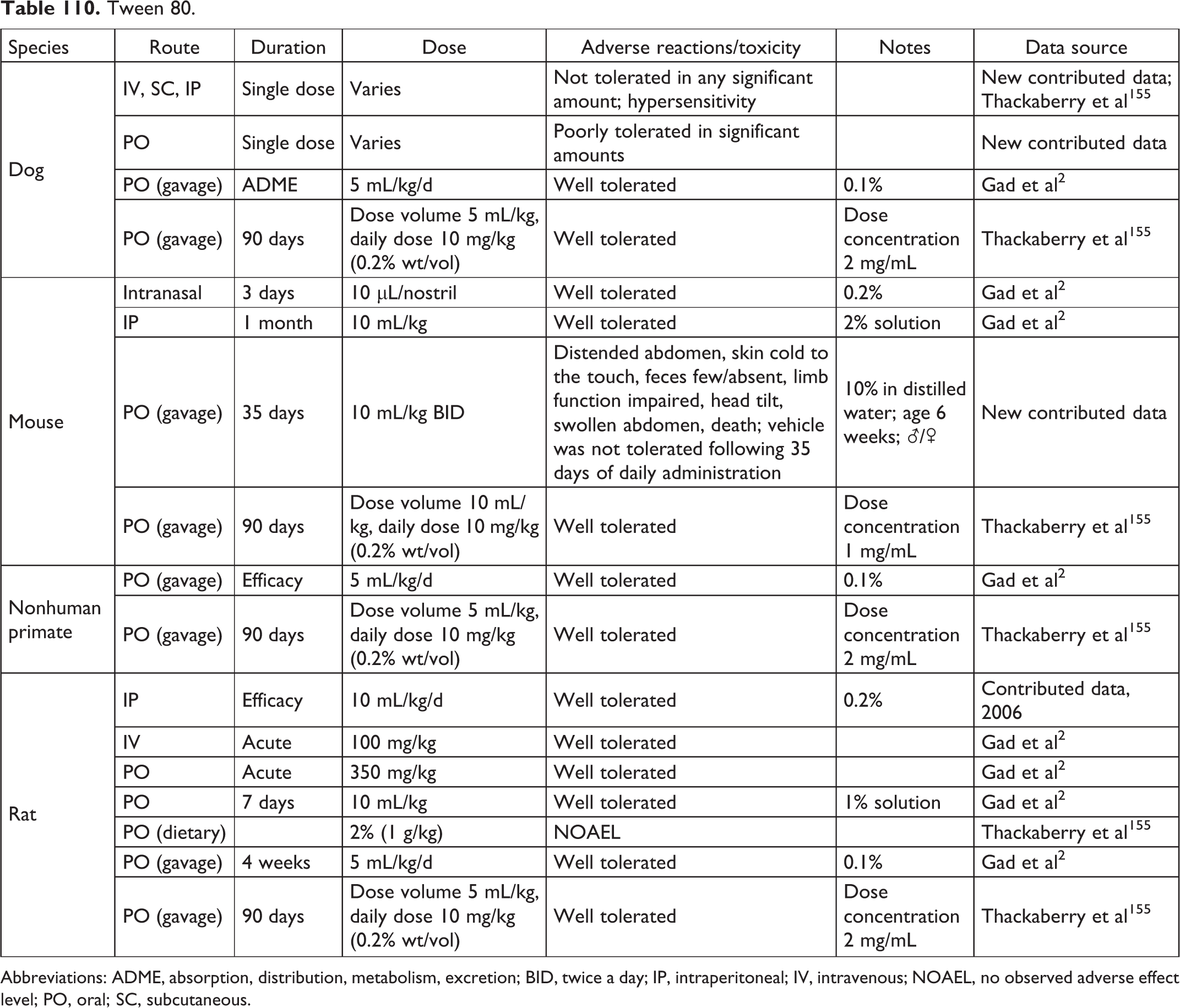
Abbreviations: ADME, absorption, distribution, metabolism, excretion; BID, twice a day; IP, intraperitoneal; IV, intravenous; NOAEL, no observed adverse effect level; PO, oral; SC, subcutaneous.
Vitamin E TPGS.
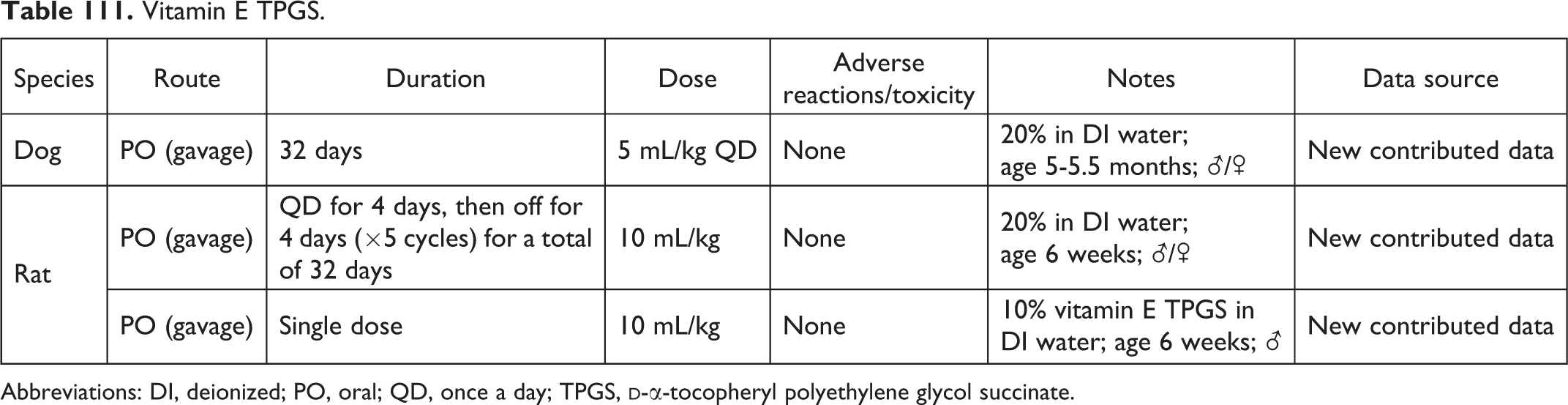
Abbreviations: DI, deionized; PO, oral; QD, once a day; TPGS,
Water.
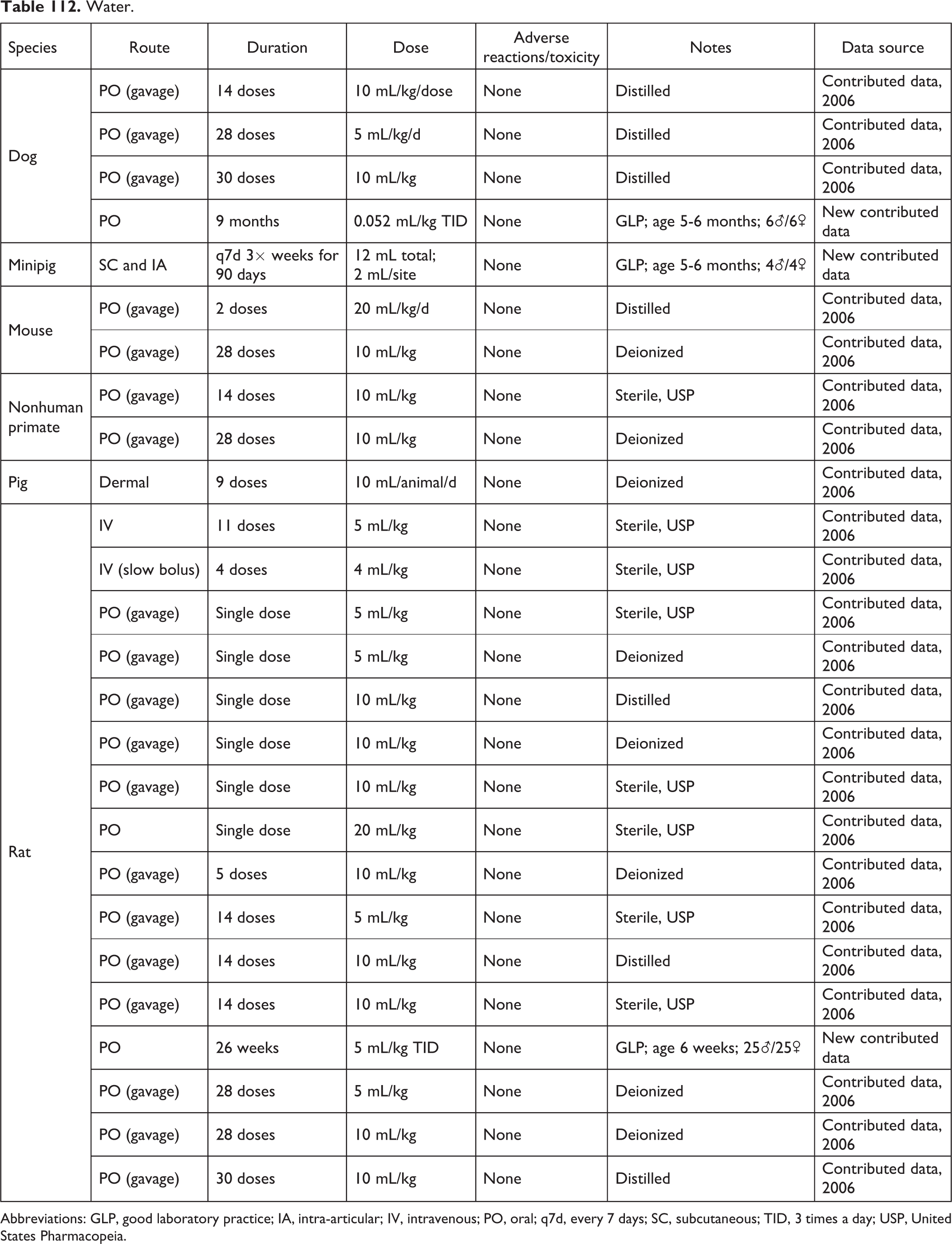
Abbreviations: GLP, good laboratory practice; IA, intra-articular; IV, intravenous; PO, oral; q7d, every 7 days; SC, subcutaneous; TID, 3 times a day; USP, United States Pharmacopeia.
Xylitol.

Combination Formulations.

Abbreviations: ADME, absorption, distribution, metabolism, excretion; AST, aspartate transaminase; BHA, butylated hydroxyanisole; BID, twice a day; BHT, butylated hydroxytoluene; CMC, carboxymethylcellulose; DAM, diacetylmonoxime; DI, deionized; DMA, dimethyl acetamide; DMSO, dimethylsulfoxide; EDTA, ethylenediaminetetraacetic acid; EOD, every other day; EP, European Pharmacopeia; EtOH, ethanol; GLP, good laboratory practice; HF, hydrofluoric acid; HPβCD, hydroxypropyl-β-cyclodextrin; HPMC, hydroxypropyl methylcellulose; IA, intra-articular; IM, intramuscular; IP, intraperitoneal; IV, intravenous; LD50, lethal dose for 50% of population; MTD, maximum tolerated dose; NF, National Formulary; NMP,
Fundamental physicochemical considerations in the development of formulations should be observed. Preparation of the formulation should not involve heating of the test material to a point that alters its chemical or physical characteristics or so as to harm test animals receiving the formulation. If the material is a solid and is to be assessed for dermal effects and/or absorption, its shape and particle size should be preserved. Multicomponent test materials (mixtures) should be formulated so that the administered form accurately represents the original mixture (ie, components should not be selectively suspended or taken into solution). Formulations should preserve the chemical stability and identity of the test material. The formulation should minimize the total required test volumes and should consider effects on systemic pH and osmolarity. Formulations should use just enough solvent or vehicle to achieve the intended goal, unless there is reason to further dilute the active ingredient. The formulation should be as easy as possible to accurately administer. Highly viscous solutions or suspensions should be avoided in parenteral and intratracheal administrations.
A particular concern is oily solutions in rabbits—rabbits are particularly susceptible to respiratory infections, and withdrawal of a gavage tube after oral dosing leaves a film along the esophagus which can be aspirated into the lungs. The pH of dosing formulations should be between 5 and 9, if possible (although volume influences the limits of this range). Acids or bases should not be used to dilute or solubilize the test material (for both humane reasons and to avoid pH partitioning or stability issues in either the gut or the renal tubule). If a parenteral route is to be used, final solutions should be as nearly isotonic as possible, and if low or high pH solutions are used, injection volume should be minimized (it is possible to overwhelm a body’s ability to maintain a stable systemic pH). Do not assume a solution will remain in solution upon injection into the bloodstream. It is usually a good idea to verify that the drug stays in solution upon injection by placing some drops into the plasma. Formulations for use by parenteral routes should be as endotoxin free as possible and sterile if for other than a few administrations. Particularly, if the test material (or formulation component) is biologically derived or produced, it should be evaluated for acceptable endotoxin content before actual formulation preparation to preclude problems. If use is to be more than a single injection, steps (such as 0.2 μm filtration) should be taken to ensure suitable sterility. 3,4
Methods
Six separate organizations (BASi, Calvert, Gad Consulting Services [GCS], MPI, Exxon, and Sinclair Labs) undertook a review of their files to identify and collect data on control vehicles they had used in studies over approximately 25 years (1991-2015). Each in vivo study considered was conducted under good laboratory practices and were general toxicity studies with end points evaluated per International Conference on Harmonisation (ICH) or Organization for Economic Cooperation and Development (OECD) guidances. Every such study conducted during this period had its vehicle control group evaluated. If the vehicle was other than water, the highest no observable adverse effect level for the vehicle formulation used was determined and this was added to the database (note that this is different from merely providing literature values of lowest toxic dose levels). Extracted in this manner by the participating organizations, the maximum nontoxic volume is reported by species, route, and duration. The nature of any dose-limiting toxicity for a vehicle or vehicle combination was also identified. These data build on data published from the previous collection (Gad et al 2 ), which had also included the former CIT Laboratories. In addition, this effort was supplemented by capturing some published literature. The total number of vehicles disclosed was large, with more than 108 different entities being reported.
The data were then assembled, quality assured, and organized in the tables presented, with materials listed in alphabetic order. These data tables are indexed in Table 4, which also provides basic physical–chemical data, synonyms, Chemical Abstracts Service (CAS) number, and general toxicity references on these vehicles.
The actual acceptable vehicle usage data are presented in Tables 5 to 113 with a single vehicle per table and arranged in alphabetical order by common name. New to this effort are the vehicle formulation combinations. A final table (Table 114) presents data on combination formulations that have been found useful and for which tolerable levels were reported. Most of these combination formulations are from contributed data—both from the initial collection (as yet unpublished) and from the more recently contributed data.
In Tables 5 to 114, for data sources, “Gad et al 2 ” refers to single-vehicle data published in 2006, “Contributed data, 2006” refers to as yet unpublished contributed data from the initial (2006) collection effort, and finally “New contributed data” refers to the more recent collection effort.
Formulation of drugs and, in a less sophisticated manner, all test materials for evaluation in intact animal systems (whether done for efficacy/pharmacology testing or for toxicology) is a field of expertise of its own. Although there are a few books on the formulation of human drugs (Racz 350 , for example), no such volume is known to the authors to exist for data on tolerable levels of preclinical vehicles and formulations.
Although the development of pharmaceutical formulations for marketed clinical products is done in a rigorous manner, what is used for nonclinical testing (and early clinical formulation) is much more pragmatic. The reader is referred to Racz, 350 Yalkowsky, 351 and Weiner and Kotkoskie 352 for more detail as to the principles of vehicle and formulation component selection.
Recognizing that there were both general rules for selection of routes of administration in nonclinical sections as well as approaches to modify the administration by these routes, the first 2 authors have considered and added guidance on these points based on their combined 70 years’ experience in the field (see Tables 1 –3).
Discussion
Although some of the vehicle options and choices presented here may seem unusual, it should be noted that they represent actual uses ranging from the discovery phase of drug development through early nonclinical safety assessment, and in many cases, all the way through to use in marketed products. The reader is invited to inspect the Food and Drug Administration’s (FDA) Inactive Ingredient List (IIG) at www.fda.gov/cder/drug/iig/inact.pdf for an idea of formulation components (excipients) used in clinical formulations. 353
For all formulations, the ability to accurately administer an aliquot of what has been prepared, with each aliquot being of uniform content, is a primary requirement. With few exceptions (ie, capsule fills), this means achieving a solution or (second choice) stable suspension or emulsion.
Although the first choice for any systemic route is always a modification of an aqueous-based vehicle, the physiochemical characteristics of the test material dictate which available options are suitable, 350,351 especially in recent years where proteins and “rocks, gums, and tars” have become more common as potential drugs with high target receptor specificity but very poor physicochemical characteristics. One starts with either a polar or nonpolar solvent, depending on which one achieves adequate dissolution of the test material and works from there (usually by trying a combination of the two with the addition of a surfactant). Solubility enhancers such as the various cyclodextrins are also increasingly used, most commonly as accompanying primary components (with the drug moiety) along with an aqueous or other polar vehicle or solvent.
Recently, research has expanded the range of routes of use of some solvents such as Transcutol (Lyon, France). Because it is not just which chemical molecule(s) is administered but also at what concentration, by what route, and at what rate, Tables 1 to 3 were derived to present considerations and potential uses of these technique factors. A means of synthesis of considerations of tolerance, improvement in target site delivery, and bioavailability and considerations of formulation stability is badly needed and being explored.
To make the database presented here as accessible as possible and to broaden its content, GCS will again setup an online version with free access (see www.gadconsulting.com), which provides an online mechanism to submit new data and will maintain and update the electronic site for 5 years after publication of this article (until 2020). This portion of the Web site will be updated.
Footnotes
Author Contributions
S. Gad contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. L. Eagle contributed to design and drafted the manuscript. C. Spainhour contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. C. Shoemake, D. R. Stackhouse Pallman, A. Stricker-Krongrad, P. Downing, R. Seals, K. Polhamus, and J. Daly contributed to acquisition, analysis, and interpretation, and drafted the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work ensuring integrity and accuracy.
Authors’ Note
Gad Consulting Services maintains files and copies of all the source data and references and will endeavor to answer all reasonable queries and provide access to researchers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
