Abstract
Myocardial mononuclear cell infiltrate is a spontaneous cardiac finding commonly identified in laboratory cynomolgus monkeys. The infiltrates are predominantly composed of macrophages with lesser lymphocytes and are not typically associated with histologically detectable cardiomyocyte degeneration. These infiltrates are of concern because they confound interpretation of test article–related histopathology findings in nonclinical safety toxicology studies. The interpretation of safety studies would be simplified by a biomarker that could identify myocardial infiltrates prior to animal placement on study. We hypothesized that monkeys with myocardial mononuclear cell infiltrates could be identified before necropsy using an ultrasensitive immunoassay for cardiac troponin I (cTnI). Serum cTnI concentrations in monkeys with myocardial infiltrates were not higher than those in monkeys without infiltrates at any of the sampling times before and on the day of necropsy. Increased serum cTnI levels are not suitable for screening monkeys with myocardial mononuclear cell infiltrates before placement in the study.
Keywords
The cynomolgus monkeys used in nonclinical drug safety assessments often have spontaneous cardiac findings of unknown cause and progression (Chamanza et al. 2006; Chamanza et al. 2010; Vidal et al. 2010; Zabka et al. 2009). In particular, myocardial mononuclear cell infiltrates, composed of macrophages with lesser lymphocytes, are commonly identified (Chamanza et al. 2010). Recently, during nonclinical safety toxicology studies sponsored by Hoffmann-La Roche, a test article–independent increase in the incidence and severity of these infiltrates in monkeys was identified. The infiltrates consisted of multifocal to coalescing myocardial infiltrates composed of macrophages and lymphocytes, predominantly in the ventricular subendocardium and subepicardium and without associated cardiomyocyte injury. In previous safety studies, there were rare instances when infiltrates were associated with a cluster of protozoa that were consistent with amastigotes of Trypanosoma spp. These cardiac findings are of concern because they confound the interpretation of test article–related histopathology findings in nonclinical safety toxicology studies. Thus, a biomarker that would identify myocardial mononuclear cell infiltrates in monkeys before they are included in a safety study would simplify the interpretation of cardiac histopathology findings.
Cardiac troponin (cTn) I is a sensitive biomarker of cardiac damage in laboratory animals (O’Brien et al. 2006) and humans (Thygesen et al. 2007). The recent development of an ultrasensitive cTnI assay made it possible to identify baseline serum cTnI concentrations, which in cynomolgus monkeys are in the 1- to 5-pg/mL range (Schultze et al. 2008; and unpublished data). This ultrasensitive assay detects increases in serum cTnI concentrations that previously were undetectable with less sensitive assays, thereby allowing for the identification of small changes of serum cTnI concentrations from baseline levels.
We hypothesized that the presence of myocardial infiltrates would be associated with sublethal or lethal cardiomyocyte injury, resulting in elevated serum cTnI concentrations on the day of necropsy and perhaps even prior to necropsy. This hypothesis was tested retrospectively on serum samples from a nonclinical safety toxicology study in which myocardial infiltrates were identified in 5 of 48 monkeys. We conclude, however, that serum cTnI is not increased at any time in monkeys with myocardial mononuclear cell infiltrates and is therefore not a marker of this finding.
Methods
Monkeys and Treatment
Cynomolgus monkeys (Macaca fascicularis) of Mauritius origin, aged 3 to 5 years (Covance Research Products, Inc., Alice, TX), were acclimated 3 weeks before study initiation. During treatment, monkeys were housed individually in stainless-steel cages in a controlled environment (temperature, 22°C ± 2°C; humidity, 50 ± 19%; light-dark cycle, about 12 hr) and were provided Purina Certified Primate Diet 5048 biscuits twice daily and assorted fresh fruit or vegetables. Reverse osmosis–filtered water was provided ad libitum by means of an automatic watering system. A vehicle or a proprietary test article given at 3 doses was administered subcutaneously each week for 52 weeks, with a recovery period of 12 weeks for a subset of animals. The proprietary test article is not known to cause immunologic effects.
All experiments were conducted in accordance with the guidance of the Covance-Madison Animal Care and Use Committee.
Clinical Chemistry
Serum cTnI concentrations were evaluated retrospectively from serum samples taken twice before the study, at week 52, and on the day of necropsy for all animals. In animals diagnosed with grades 2 or higher mononuclear cell infiltrates, serum cTnI concentrations were also measured from samples taken at weeks 12 and 26. Blood samples (1 to 1.5 mL) obtained from the saphenous vein were collected into serum separator tubes (without anticoagulant) and allowed to clot for at least 30 min at room temperature. After cold centrifugation (15 min at 3,000 RPM), the samples were transferred to individual, labeled, amber screw-cap vials and stored at –70°C until analysis. Serum samples were assayed together, within 3 to 18 months of sample collection. In addition, routine clinical pathology parameters including leukocyte total and differential counts, fibrinogen, and aspartate aminotransferase were performed on all samples.
All serum samples (50 µL each) were run in duplicate, and serum cTnI concentrations were measured using an ultrasensitive cTnI immunoassay (Singulex, Alameda, CA), according to the manufacturer’s recommendations. Final serum cTnI concentrations were reported as individual values and represent the mean of the duplicate measurements. Individual and group mean data were evaluated and reported.
Histopathologic Analysis
Standard sections of the heart, consisting of 2 parasagittal sections encompassing the right and left ventricles and atria, as well as the interventricular septum, were evaluated. Samples were routinely processed into 5-µm-thick paraffin-embedded sections placed onto glass slides and stained with hematoxylin and eosin. Slides were read unblinded by a board-certified pathologist followed by a formal peer review process, which entailed review of all slides by a second pathologist and agreement on diagnosis and severity grades.
The severity of mononuclear cell infiltrates was graded subjectively from 0 to 5 (most severe) such that 0 = no infiltrate; 1 = less than 5-min aggregates of mononuclear cell infiltrates; 2 = more than 6 small, multifocal, noncoalescing aggregates of mononuclear cell infiltrates; 3 = multiple, at times coalescing, small to medium-sized aggregates of mononuclear cell infiltrates with a broad distribution throughout the myocardium; 4 = multifocal coalescing mononuclear cell infiltrates at times effacing the architecture of the myocardium; and 5 = architecture of the heart severely effaced by the mononuclear cell infiltrates. Of note, grades 4 and 5 were not identified in monkeys with myocardial mononuclear cell infiltrates in this and other studies. Grading of the infiltrates was based on common pathology practice and previous experience.
Results
Grade 2 or 3 mononuclear cell infiltrates occurred in 5 of 48 monkeys, including 2 of 12 vehicle-treated animals (Figure 1). The infiltrates accounted for up to about 2% of the surface area of the 3 sections of the heart evaluated for each animal. This amount of infiltrate is greater than we had observed in previous studies and that which is reported in the literature (Chamanza et al. 2006; Vidal et al. 2010). The myocardial mononuclear infiltrates were not treatment related, and there were no test article–related cardiac findings in this study. There were no time-dependent changes in cTnI concentrations, and all individuals were within reference range for cynomolgus monkeys (unpublished data and data from Singulex). Organisms consistent with amastigotes of Trypanosoma spp. were absent in the monkeys from this study but had been observed, although rarely, in association with this finding in previous toxicology studies in monkeys.
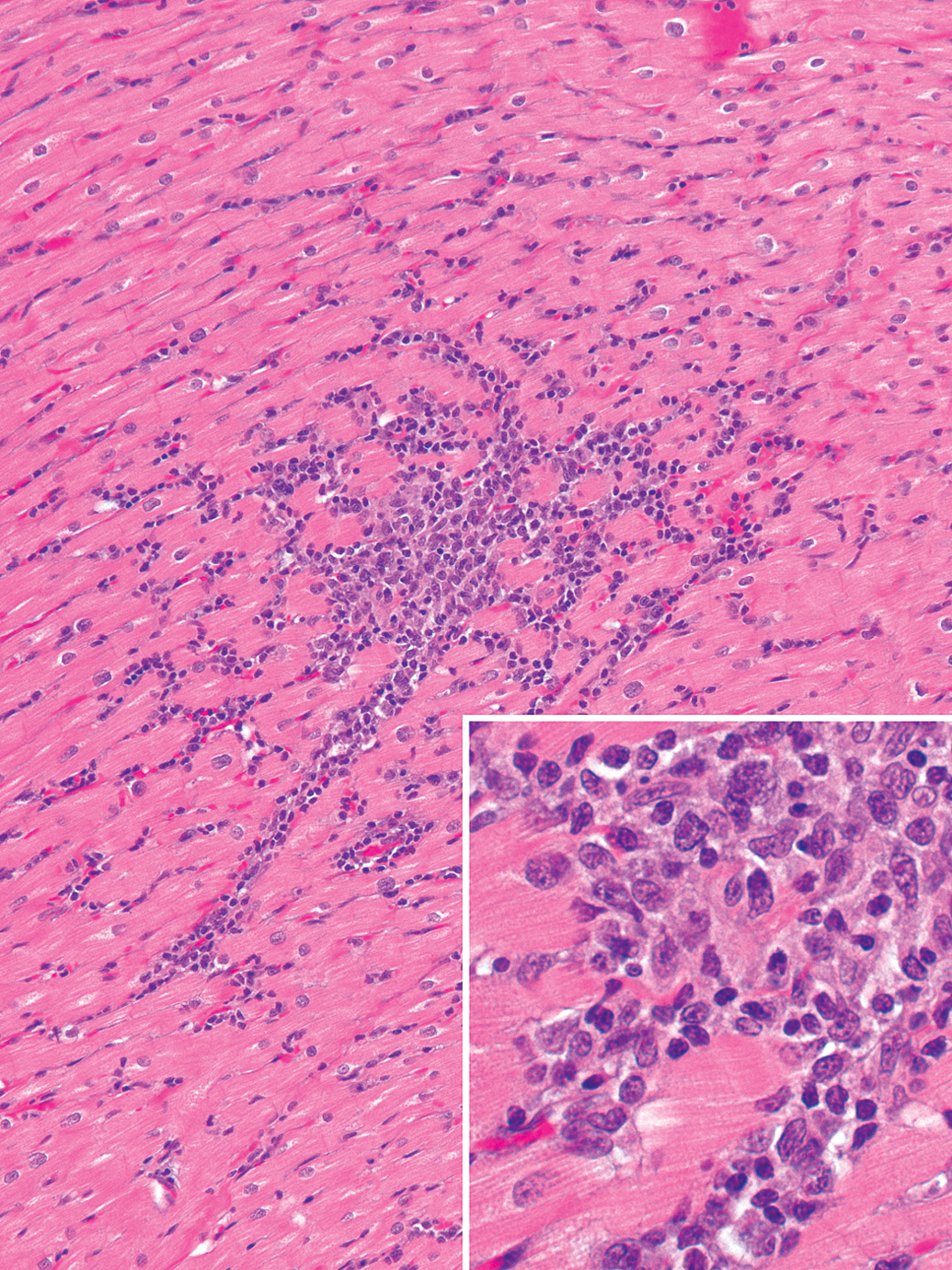
Spontaneous myocardial mononuclear cell infiltrates in the heart of a cynomolgus monkey. The infiltrate is focal and predominantly composed of macrophages and fewer lymphocytes (inset) and is not associated with cardiomyocyte degeneration (hematoxylin and eosin).
Serum cTnI concentrations in monkeys with grade 2 or 3 mononuclear cell infiltrates were not elevated above baseline values at any of the times evaluated. The average (±standard deviation) of the serum cTnI concentration was 1.9 ± 1.1 pg/mL (range, 0.4 to 4.2 pg/mL) in the 5 monkeys with grade 2 or 3 mononuclear cell infiltrates (Figure 2). These concentrations were similar to serum cTnI concentrations in monkeys without or with only grade 1 mononuclear cell infiltrates (1.9 ± 2.3 pg/mL; range, 0.3 to 13 pg/mL, with one outlier at 23.1 pg/mL). Serum cTnI concentrations in monkeys receiving the vehicle or test article were similar. All other clinical pathology parameters in the monkeys with mononuclear cell infiltrates were unremarkable (data not shown).
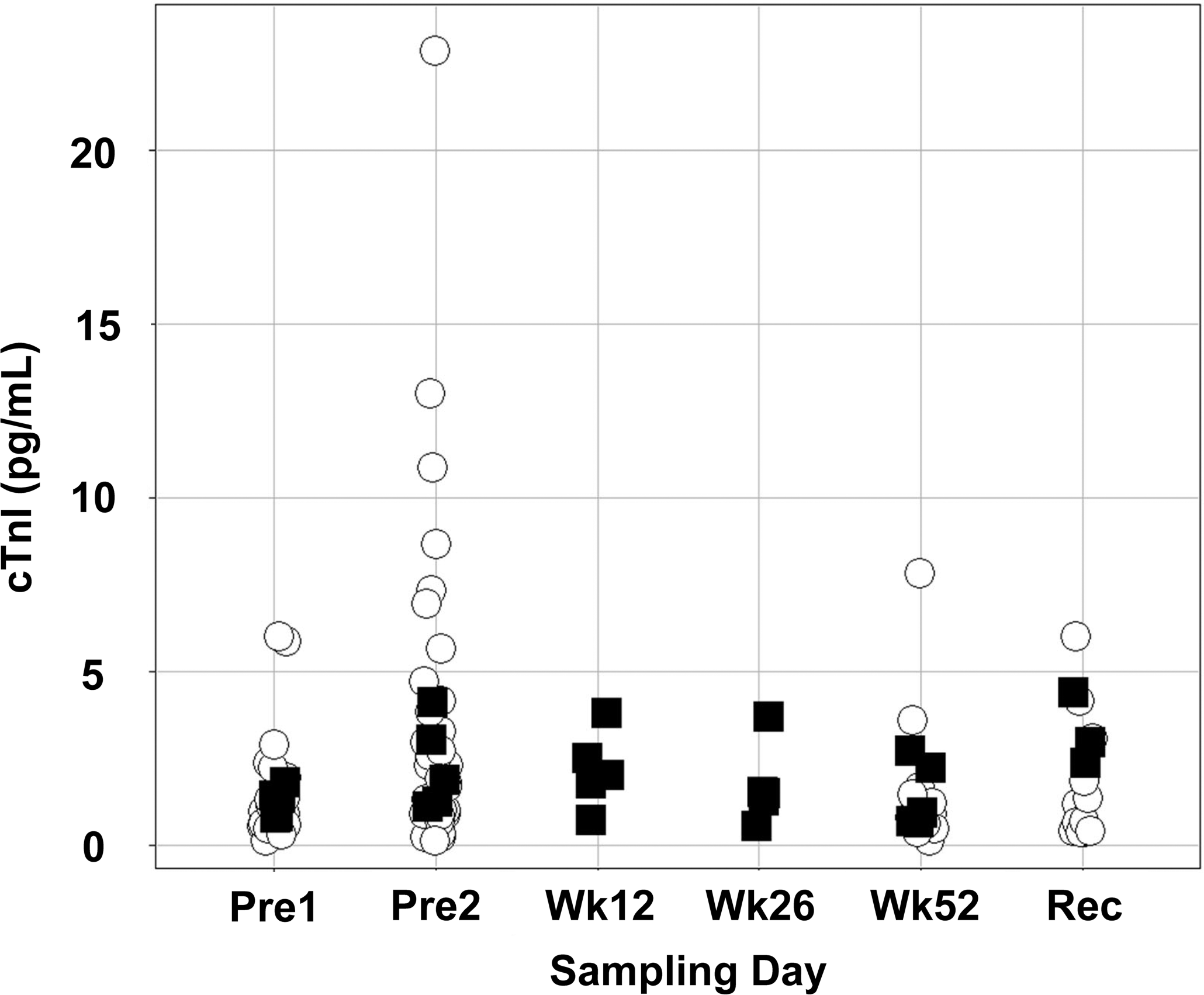
Serum cardiac troponin I (cTnI) concentrations were no higher in cynomolgus monkeys with grade 2 or 3 (on a scale of 0 to 5) myocardial mononuclear cell infiltrates (▪) than they were in those with grades 0 or 1 (^). The sera were evaluated for cTnI concentrations at 2 predose time points (Pre1 and Pre2) and at necropsy on week 52 (Wk52) or after a 12-week recovery phase (Rec) for all animals, and on study weeks 12 (Wk12), 26 (Wk26), and 52 for animals with grades 2 or 3 myocardial mononuclear cell infiltrates.
Discussion
The goal of this study was to determine whether serum cTnI concentrations could identify monkeys with spontaneous myocardial mononuclear cell infiltrates. Serum cTnI concentrations were not elevated in any of the monkeys with grade 2 or 3 mononuclear cell infiltrates in the myocardium at any of the time points assessed.
Serum cTnI is a sensitive marker of myocardial injury in all species, including the cynomolgus monkey (Apple et al. 2008; Minomo et al. 2009). At the onset of myocardial necrosis in the monkey, cTnI is rapidly released into the blood in an amount and duration that depends on the size of the affected tissue (Minomo et al. 2009). Persistent high concentrations of serum cTnI are the result of continued leaching of cTnI from the remains of necrotic cells (Hickman et al. 2010). In minor cardiac damage, serum cTnI increases of smaller magnitude can be detected briefly. This narrow window of time for detecting serum cTnI changes is the direct result of its rapid clearance in species commonly used in toxicology studies (Dunn et al. 2011) and in humans (Gerhardt et al. 1991). Therefore, the identification of serum cTnI increases requires knowledge of the kinetic clearance rate of cTnI and the size and nature of the anticipated cardiac damage.
Serum cTnI concentrations also may increase before histologic changes occur in viral myocarditis (Lim et al. 2005), during myocardial ischemia before necrosis in humans (Hickman et al. 2010), or even in the absence of histopathologic findings in rats receiving rosiglitazone (Mikaelian et al. 2011). The identification of small increases in serum cTnI in these conditions requires a high-density sampling strategy that accounts for the rapid clearance of cTnI (Dunn et al. 2011). Routine toxicology studies, however, including the one reported here, are not amenable to high sampling densities because multiple blood draws would interfere with the evaluation of other safety parameters and because repeated handling may increase the stress on the monkeys. Therefore, the identification of cardiac effects with small increases in serum cTnI concentrations would require dedicated studies reflecting the fast clearance of serum cTnI, the kinetics of the test article, and the time of maximum effect of the test article on the heart.
The absence of increased serum cTnI concentrations and the lack of histologic evidence of cardiomyocyte degeneration differentiate the condition described here from the findings in models of myocarditis, in which increases in serum cTn are associated with cardiomyocyte damage (Bachmaier et al. 1995; Lim et al. 2005; Smith et al. 1997). As reported here, normal serum concentrations of cTnI in monkeys with myocardial mononuclear cell infiltrates imply that cardiomyocyte damage, not cellular infiltration, is responsible for the serum cTnI increases seen in animal models of myocarditis.
Limitations of the Study
An inherent limitation of this study was the poorly characterized effect of long-term frozen storage on the stability and structural integrity of cardiac troponins, specifically when using an ultrasensitive assay (Agarwal et al. 2011; Basit et al. 2007). Assay validation studies performed by Singulex have indicated that cTnI is stable when stored at –80°C for up to 6 months; however, stability testing beyond 6 months was not performed (unpublished manufacturer data, Singulex). The samples collected at the time of necropsy in this study were analyzed within 6 months following collection, which is within the period of stability of cTnI. Basit et al. (2007) identified that cTnT concentrations remain stable for up to 12 months when stored at –70°C, with marginal decreases in analyte concentration following 24 months of frozen storage. Therefore, some cTnI degradation may have occurred for the samples collected predose and on weeks 12 and 26. However, possible cTnI degradation would not invalidate the major finding of this study, which was that monkeys with myocardial mononuclear cell infiltrates had normal serum cTnI concentrations.
Conclusions
In conclusion, serum cTnI concentrations were not increased in a retrospective assessment of samples from cynomolgus monkeys with grade 2 or 3 myocardial mononuclear cell infiltrate in the absence of microscopically detectable cardiomyocyte damage. Therefore, we conclude that serum cTnI is not a marker for myocardial mononuclear cell infiltrates and thus cannot be used to screen monkeys for this spontaneous cardiac finding that can confound histopathologic interpretations in nonclinical safety studies.
Footnotes
Acknowledgment
The authors thank Ms. Rachael Fernandes for her outstanding technical support.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Hoffmann-La Roche, Inc.
Abbreviations
