Abstract
Limited information has been published on the use of cardiac troponin I (cTnI) as a biomarker of cardiac injury in monkeys. The purpose of these studies was to characterize the cTnI response seen in cynomolgus macaques during routine dosing and blood collection procedures typically used in preclinical safety studies and to better understand the pathogenesis of this response. We measured cTnI using two different methods, the Siemens Immulite cTnI assay and the more sensitive Siemens Troponin I-Ultra assay. We were able to demonstrate that after oral, subcutaneous, or intravenous dosing of common vehicles, as well as serial chair restraint for venipuncture blood collection, that minimal to mild transient increases in cTnI could be detected in monkeys with both assays. cTnI values typically peaked at 2, 3, 4, or 6 hr after sham dosing and returned to baseline at 22 or 24 hr. In addition, marked increases in heart rate (HR) and blood pressure (BP) occurred in monkeys during the restraint procedures, which likely initiated the cTnI release in these animals. Monkeys that were very well acclimated to the chairing procedures and had vascular access ports for blood sampling did not have marked increases in HRs and BP or increases in cTnI.
Cardiac troponin I (cTnI) is being used more commonly in preclinical safety assessment studies. There are multiple publications, primarily utilizing rats, which demonstrate the cTnI response to acute cardiac injury and a limited number of studies that show the response to chronic injury (Herman et al. 1998; Koh, Nakamura, and Takahashi 2004; O’Brien et al. 2006; Reagan et al. 2013; Schultze et al. 2009; York et al. 2007; Zhang et al. 2006). The results of these and other studies demonstrated how to effectively utilize this biomarker to detect cardiac injury in preclinical safety studies. Based on an extensive review of the historical literature as well as inclusion of the authors’ experiences utilizing this biomarker, a submission was made to the Federal Drug Administration (FDA) for the qualification of cTnI. In 2012, cTnI was qualified by the FDA as a marker of cardiac necrosis in rats and dogs for use in preclinical studies (Woodcock 2012). At the time the qualification document was submitted in late 2008, there were limited published studies demonstrating the use of this biomarker in nonhuman primates (NHP) studies, although more recently, additional publications have become available (Dunn et al. 2012; Minomo et al. 2009; Schultze et al. 2015; Takeuchi et al. 2008; Zabka, Irwin, and Albassam 2009). Based on the paucity of published information on the use of this biomarker in NHP at the time of submission, cTnI was not officially qualified for use in NHP.
To use this biomarker effectively in all species, including the NHP, several analytical factors should be considered to ensure accurate assessment of cardiac injury. Critical analytical factors include the demonstration that the assay cross-reacts in the species of interest, has an acceptable dynamic range, shows good reproducibility, and shows excellent sensitivity and specificity. Most automated assays that are utilized to measure cTnI in the preclinical setting are assays developed to measure cTnI in humans. The sensitivity of these assays has continued to evolve, and many of the manufacturers are producing second- and third-generation assays that are highly sensitive and show good precision around the lower limits of quantification. Many of these assays do effectively measure cTnI in the commonly used preclinical toxicity species, but some assays are more effectively utilized than others. A publication by Apple et al. (2008) reviewed several of the commercial assays approved by the FDA for use in humans and demonstrated that both the Siemens Immulite cTnI and the Siemens Troponin I-Ultra assays could be effectively utilized in the more common preclinical species, including in cynomolgus and rhesus macaques.
Preanalytical variables including site of collection, quality of the samples, and timing of the sampling, as well as methods of restraint and blood sample collection are also critical factors for accurate assessment of a cardiac troponin response, especially in NHP studies. A recent publication using the Singulex Errena ultrasensitive cTnI assay showed that restraint associated with sham oral and intravenous dosing in cynomolgus macaques induced a transient elevation in cTnI (Schultze et al. 2015). The purpose of this publication is to further characterize the effect of restraint in cynomolgus macaques on the cTnI results using the Siemens Immulite cTnI and the Siemens Troponin I-Ultra assays. We also wanted to assess the underlying physiological state of these animals undergoing restraint by measuring heart rate (HR) and blood pressure (BP) to better understand conditions that may lead to cTnI increases.
Materials and Methods
Experimental Animals
All monkeys used throughout the studies were cynomolgus macaques (Macaca fasicularis) from Charles River Primates (Houston, TX), all Mauritius in origin. Monkeys were fed a standard diet of pelleted food (Certified Primate Diet 5K91; LabDiet, St. Louis, MO) provided twice daily and supplemented with vegetable and/or fruit. Municipal drinking water, purified by reverse osmosis, was provided ad libitum, and there was a 12 hr light (6:00 am to 6:00 pm) and 12 hr dark cycle. All experiments were conducted in accordance with the current guideline for animal welfare (National Research Council Guide for the Care and Use of Laboratory Animals). All study protocols were reviewed and approved by Pfizer Institutional Animal Care and Use Committee. All animals had been previously acclimated to handling and chair restraint before the start of the experiments. Typically, when animals are brought into the facility, they go through at least a 1-month habituation period, before they are used in a preclinical safety study, which includes socialization and gradual introduction to handling, chair restraint, and sham procedures using positive reinforcement.
Experimental Procedures
Four separate studies were conducted, which assessed the impact of chair restraint and vehicle administration on cTnI concentrations (Table 1).
Study Designs That Were Conducted to Assess the Impact of Chair Restraint and Vehicle Administration on Cardiac Troponin I Concentrations.
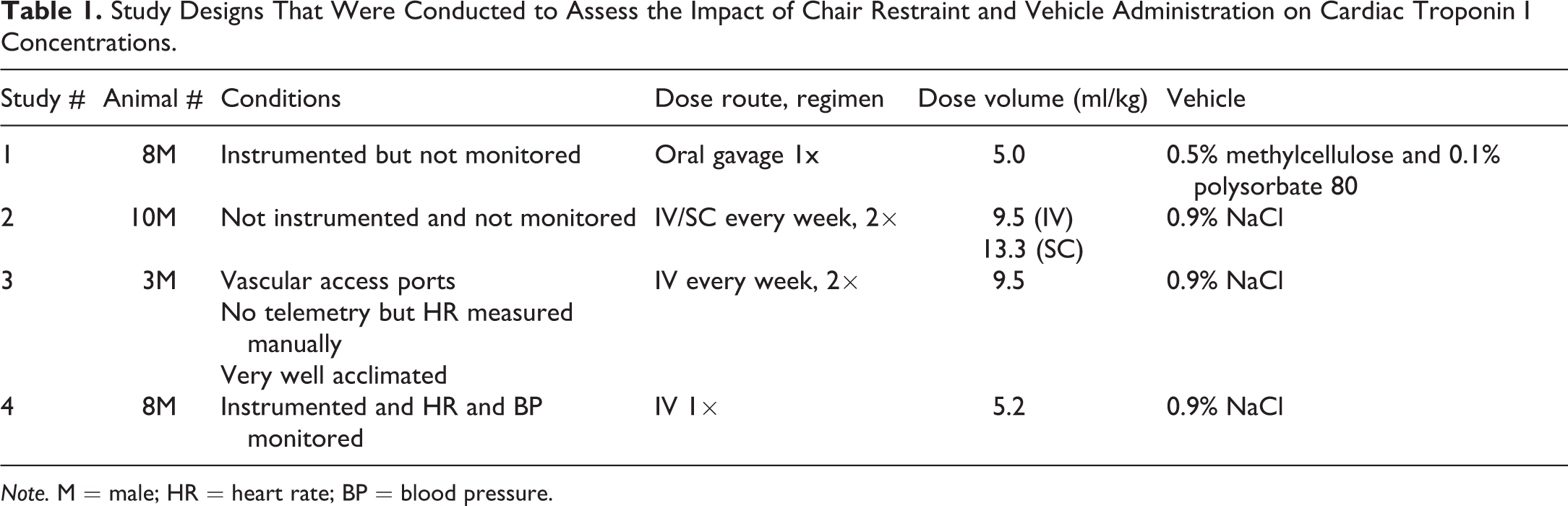
Note. M = male; HR = heart rate; BP = blood pressure.
In the first study, 8 male monkeys (4.9–7.6 kg) instrumented with telemetry transmitters (D70-PCT; Data Sciences International, St. Paul, MN) were subjected to chair restraint and oral gavage procedures. For the telemetry surgery, a pressure catheter was placed in the femoral artery and advanced to the descending aorta, and a biopolar intracardiac lead was advanced through the jugular vein into the trabeculae of the right ventricle for electrocardiogram collection (D70-PCT; Data Sciences International). At least a 2-week period was allowed postsurgery before animals were used on the study. A complete blood count and clinical biochemistry panel were done before the study started and showed no abnormalities. Animals received a single oral dose (5 ml/kg) of 0.5% methylcellulose and 0.1% polysorbate 80, and serial blood samples were at predose (0) and 0.5, 1, 2, 4, 6, and 24 hr postdose (HPD) for troponin analysis using the Siemens Immulite cTn I assay. The monkeys remained in the chair between the 0- and 2-HPD sampling time points but were returned to their cages after the 2, 4, 6, and 24 hr collections with chair restraint. Although these animals had telemetry transmitters, cardiovascular parameters were not assessed during the blood collection procedures in this study.
In the second study, 10 male monkeys (4.2–6.7 kg) were dosed intravenously (9.5 ml/kg) or subcutaneously (13.3 ml/kg) with sodium chloride (0.9%) once weekly for 2 weeks (2 total doses). These monkeys were not telemeterized. Animals were restrained in chairs, and blood samples were taken predose (0), 2, 6, and 24 HPD for troponin analysis using the Siemens Immulite cTn I assay. The monkeys remained in their chairs between the 0 and 2 hr sampling time points but were returned to their cages after the 2, 6, and 24 hr collections.
For the third study, 3 male monkeys (6.6–8.0 kg) were evaluated. These monkeys were colony animals surgically implanted with subscapular venous access ports that were very well acclimated to handling (restraint, dosing, and bleeding). Blood samples were drawn predose (0) and 2, 4, 6, and 24 HPD for troponin analysis using the Siemens Immulite cTnI assay. The monkeys remained in their chairs between the 0 and 2hr sampling time points but were returned to their cages after the 2, 4, 6, and 24hr collections with chair restraint. HRs were obtained manually predose and during each blood collection chairing session (pulse rates were determined for 15 s in triplicate at each time point). A dose of sodium chloride (0.9%) was given intravenously (9.5 ml/kg) on days 1 and 8.
The fourth study used 8 male monkeys (5.7–8.4 kg) that had been previously instrumented with telemetry transmitters (as described for the first study). A complete blood count and clinical biochemistry panel were done before the study started and showed no abnormalities. Approximately 1 hr of baseline telemetry data was collected in their cages in order to establish resting HR and BP values (Ponemah Physiology Platform, P3 Plus, Version 4.8, Data Sciences International) before the animals were restrained. The telemetry receivers followed the monkeys from their cages to the restraint chairs, and HR and BP were continuously monitored during each restraint session and when animals were returned to their cages between restraint sessions. For the initial dosing session, each animal was placed in the restraint chair for approximately 20 min and then received an intravenous bolus of 0.9% sodium chloride (5.2 ml/kg). A blood sample was taken 4 days prior to dosing, and predose (0), 5 min, and 3, 6, and 22 HPD for analysis of troponin using the Siemens Troponin I-Ultra assay as well as aspartate aminotransferase (AST), alanine aminotransferase (ALT), and creatine kinase (CK). Animals were returned to the telemetry cages after collection of the 5-min blood samples (for a total restraint time of 30 min at the time of dosing). Restraint sessions for subsequent blood sampling were shorter, with each chairing session lasting approximately 10 min. One-minute mean HR and mean BP values were derived from the BP signals. Individual mean BP and HR values were calculated as a 60-min average in the telemetry cage for baseline, a 30-min average during the first restraint, and a 3-min average at the time of the 3-, 6-, and 22-HPD restraint procedures.
Historical review of our database included the review of the Siemens Troponin I-Ultra assay results from 485 monkeys (273 males and 212 females) from multiple studies over a 4-year period of time. Age range for these monkeys was 2 to 6 years.
Laboratory Testing
Whole blood was collected by a femoral venipuncture from monkeys and allowed to clot before separation by centrifugation at approximately 173 relative centrifugal force for 10 min. The serum was aliquoted and analyzed for cTnI concentrations, AST, ALT, and/or CK activities immediately or frozen at ∼−70°C until tested. AST, ALT, and CK assays were all run on the ADVIA 1650 analyzer (Siemens Healthcare Diagnostics, Tarrytown, NY) using standard techniques.
Siemens Immulite cTn I assay, previously DPC, is an enzyme-labeled chemiluminescent immunoassay using a murine monoclonal antibody attached to solid phase (bead) and a polyclonal antibody conjugated to alkaline phosphatase (Siemens Healthcare Diagnostics). Both antibodies bound to the cTnI in the sample, unbound sample and enzyme conjugate were removed and then a chemiluminescence substrate was added resulting in the complex emitting light which was directly proportional to the amount of cTnI in the sample. This assay was validated for use in cynomolgus macaques and run on the Immulite 1000 immunoanalyzer following the manufacturer’s instructions. The lower limit of detection for this assay was 0.2 ng/ml. This assay is a first-generation cTnI assay and is not as sensitive of an assay compared with many other assays that are now available.
Siemens Troponin I-Ultra assay, previously Bayer, is a chemiluminometric 3-site sandwich immunoassay which is a second-generation assay for cTnI (Siemens Healthcare Diagnostics). The assay includes a polyclonal goat anti-troponin I antibody labeled with acridinium ester as well as 2 biotinylated mouse monoclonal anti-troponin antibodies. The anti-troponin antibodies bound to the cTnI in the sample, and then the biotin in the immune complexes bound to the streptavidin-labeled magnetic particle. The complex emitted light and was directly proportional to the amount of cTnI in the sample. The assay was run on the ADVIA Centaur following the manufacturer’s instructions (Siemens Healthcare Diagnostics). It was fully validated for use in cynomolgus macaques, and the lower limit of quantitation of 0.03 ng/ml was utilized.
Results
Four separate studies were conducted, which assessed the impact of chair restraint and vehicle administration on cTnI levels. Animals in three studies (studies 1, 2, and 4) received the standard facility training for chair restraint, whereas animals in one study (study 3) received greater training and were much more acclimated to chair restraint than the other 3 cohorts.
In study 1, 8 cynomolgus macaques were dosed by oral gavage with vehicle (0.5% methylcellulose and 0.1% polysorbate 80). Serial blood samples were taken for cTnI analysis using the Siemen’s Immulite cTnI assay, just before dosing (predose), as well as 0.5, 1, 2, 4, 6, and 24 HPD. Animals were continuously chair restrained from the time of the predose sample collection to the 2-HPD sample collection and briefly chaired restrained for subsequent blood collection. The prestudy samples were consistently below the lower limit of detection of the assay, <0.2 ng/ml (Table 2). Most (5/8) monkeys had detectable cTnI responses (0.2–1.0 ng/ml) 1 to 6 HPD with the peak response occurring 4 to 6 HPD and returned to <0.2 ng/ml at 24 HPD.
Serial Cardiac Troponin I Results (Siemens Immulite cTnI Assay) in Monkeys Dosed by Oral Gavage, Study 1.
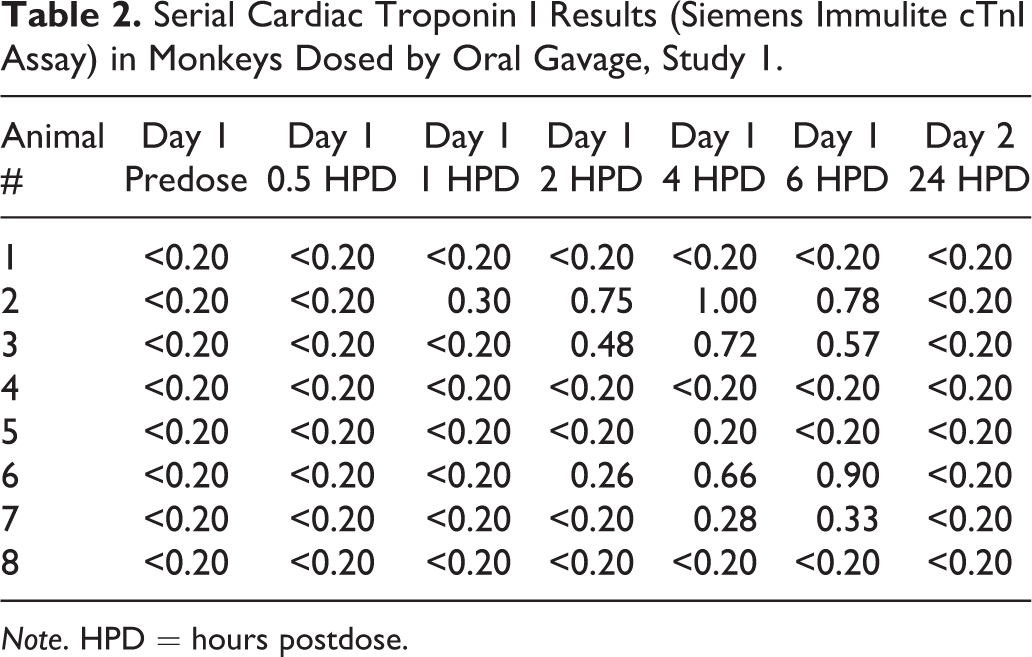
Note. HPD = hours postdose.
In study 2, 10 cynomolgus macaques were dosed with 0.9% sodium chloride intravenously (n = 5) or subcutaneously (n = 5; Table 3). Serial blood samples were taken for cTnI analysis using the Siemen’s Immulite cTn I assay, just before dosing (predose) as well as 2, 6, and 24 HPD. This was done on day 1 and again a week later on day 8. Animals were continuously chair restrained from the time of the predose sample collection to the 2-HPD sample collection and briefly chair restrained for subsequent blood collection. The predose samples were consistently below the limit of detection of the assay, <0.2 ng/ml, on both days 1 and 8. Most (4/5) monkeys dosed intravenously had detectable cTnI responses (0.28–1.9 ng/ml) at 2 and/or 6 HPD with the peak response most often occurring at 2 HPD and returned to <0.2 ng/ml at 24 HPD. Monkeys dosed subcutaneously (3/5) also had cTnI (0.21–0.48 ng/ml) increases at 2 and/or 6 HPD, with the peak response occurring most often at 2 HPD and returned to <0.2 ng/ml at 24 HPD.
Serial Cardiac Troponin I Results (Siemens Immulite cTnI Assay) from Monkeys Dosed with Saline Intravenously (n = 5) or Subcutaneously (n = 5), Study 2.
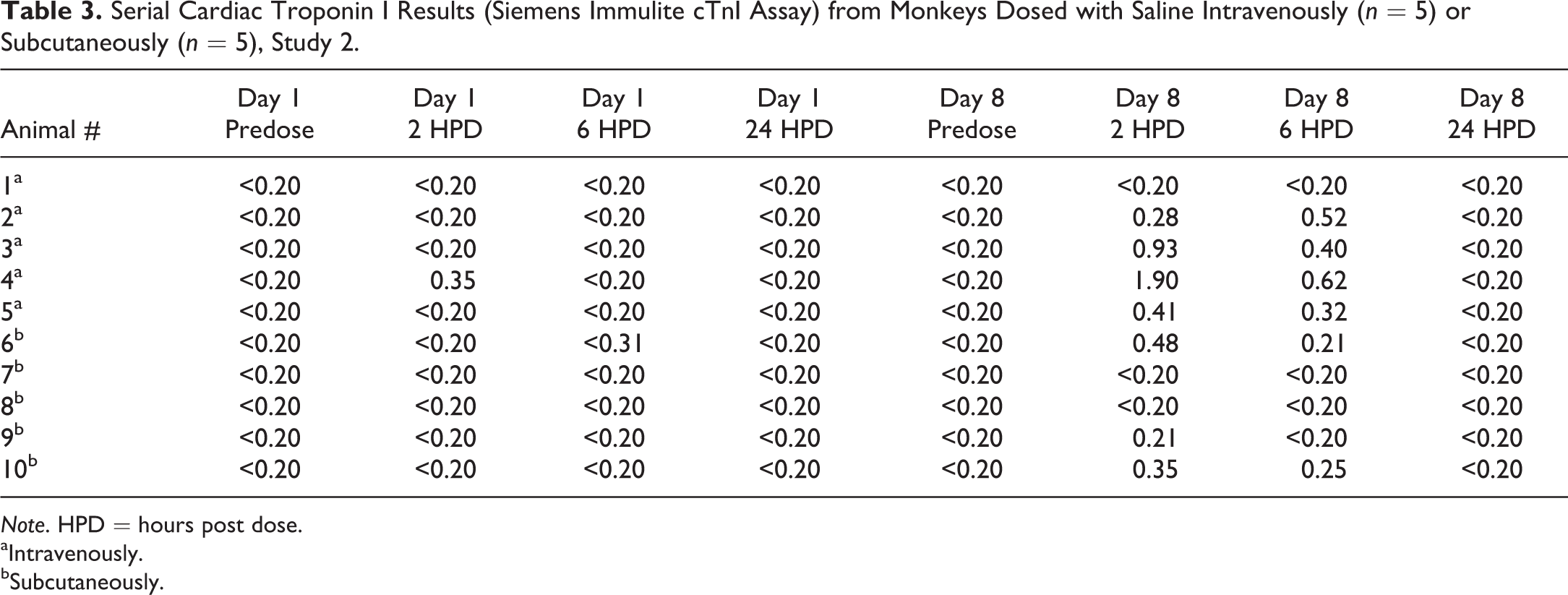
Note. HPD = hours post dose.
aIntravenously.
bSubcutaneously.
In study 3, 3 cynomolgus macaques that had vascular access ports and were very highly conditioned to the chairing, and blood sampling process received 0.9% sodium chloride intravenously on day 1 and again on day 8. Samples were taken from the vascular access ports, while chaired, before dosing (predose) as well as 2, 6, and 24 HPD (Table 4) for cTnI analysis using the Siemen’s Immulite cTnI assay. Animals were continuously chair restrained from the time of the predose sample collection to the 2-HPD sample collection and briefly chaired restrained for subsequent blood collection. A prestudy sample was also taken prior to day 1. cTnI values in these monkeys were all <0.2 ng/ml throughout the sampling period. HRs in these animals were highest on the prestudy day, HR ranges of 136 to 211 beats per minute (BPM), and lower at initiation of the dosing phase (HR ranged from 123 to 177 BPM on day 1 and 124 to 163 BPM on day 8).
Serial Cardiac Troponin I Results (Siemens Immulite cTnI Assay) and Heart Rates from 3 Monkeys with Vascular Access Ports, Study 3.
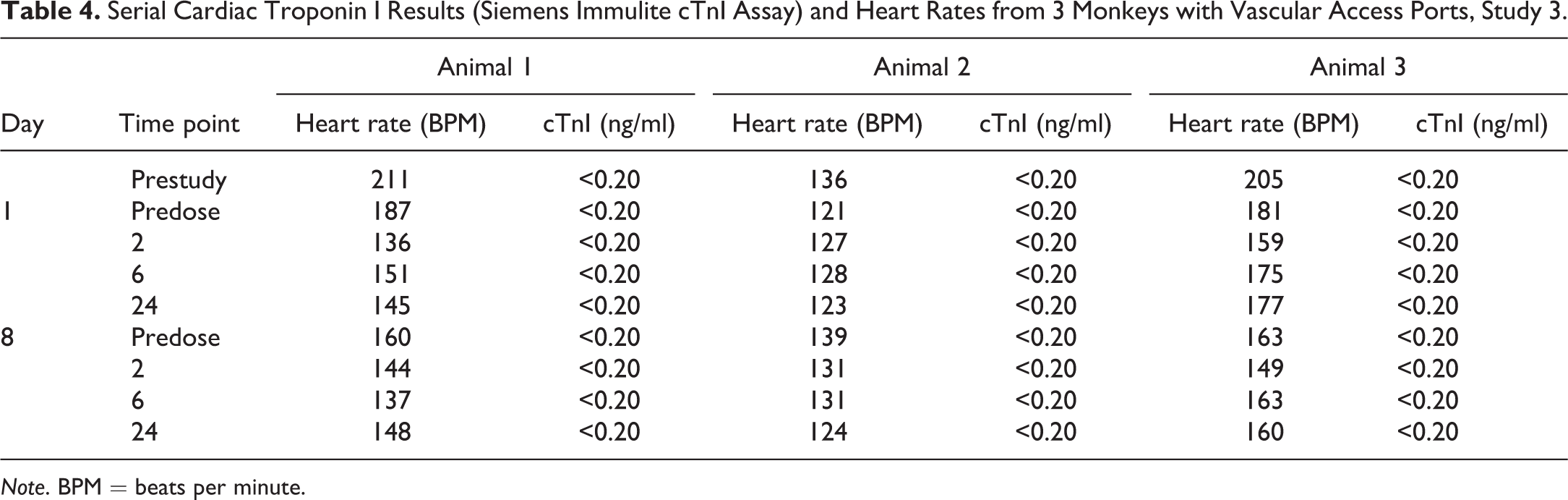
Note. BPM = beats per minute.
In study 4, cynomolgus macaques received 0.9% saline intravenously during a 30-min chairing session on day 1; and cTn I concentrations (Figure 1), CK (Figure 2), AST (Figure 3), and ALT activities were serially measured. cTnI was measured using the Siemens Troponin I-Ultra assay. The cTnI was below the limits of quantitation (0.03 ng/ml) in all monkeys on day -4 and day 1 at time 0- and 5-min postdose. Most monkeys, 7/8, had a detectable cTnI response at 3 and 6 HPD, and these values ranged from 0.03 to 0.43 ng/ml, which indicates the variability that can be seen between monkeys, even though they were handled similarly. The cTnI concentration was below the limit of quantitation in all animals at 22 HPD (day 2). CK values were quite variable throughout the sampling period, including a significant elevation in 1 monkey (animal 2) at time 0. The greatest consistent increases (16× to 44× baseline) were at 6 HPD, where 5/8 monkeys had increases relative to the baseline values. Values in these animals ranged from 1,265 to 5,087 U/L at 6 HPD compared with values of 78 to 180 U/L in these same animals on day -4. The AST values were also quite variable, but there were increases (2× to 2.5×) of AST values in 4/8 monkeys at 6 HPD, and they remained either elevated or returned to baseline at 22 HPD. AST values in these 4 animals ranged from 57 to 106 U/L compared with values of 29 to 53 U/L on day -4. ALT values remained fairly steady overtime (data not shown).
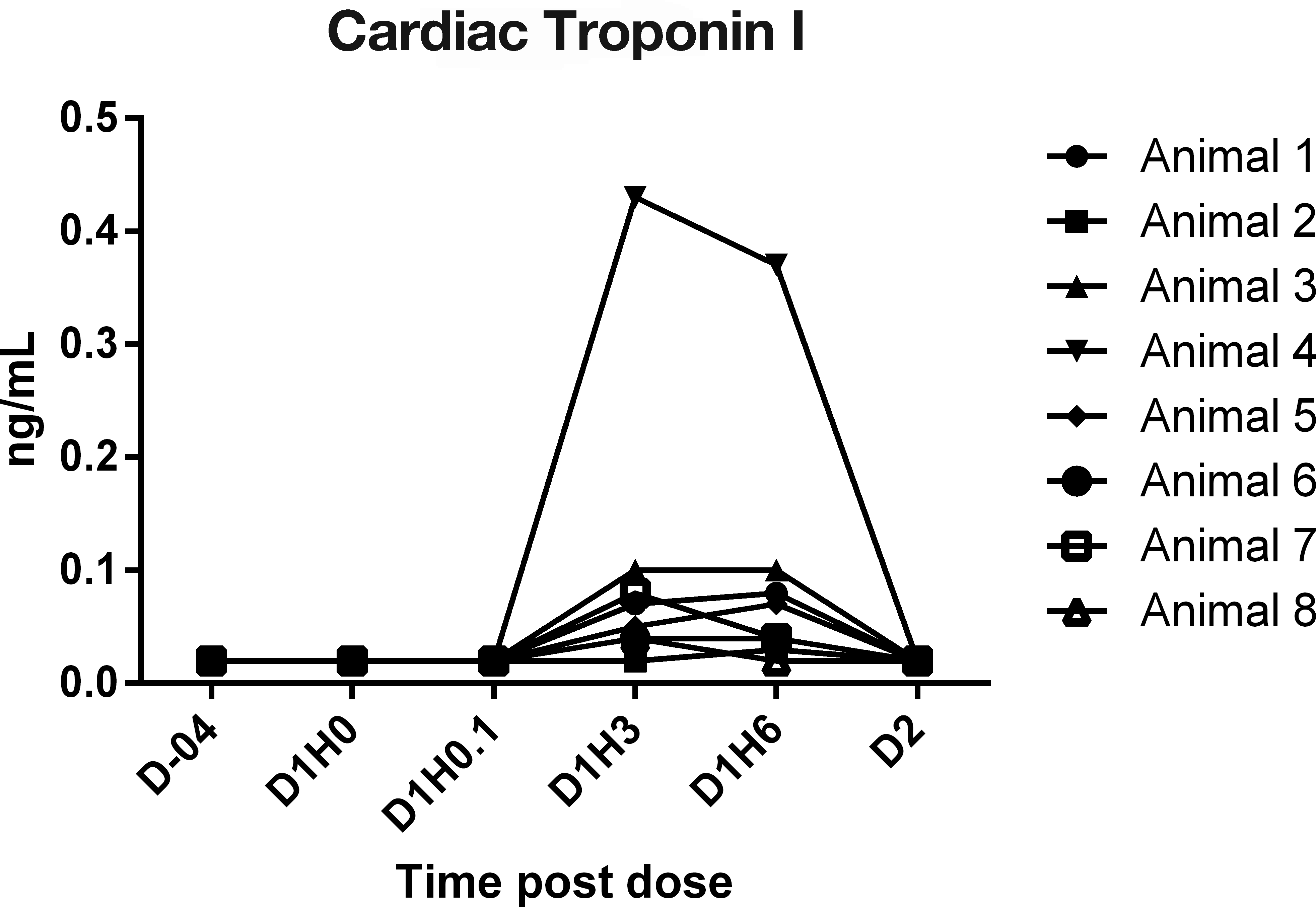
Serial cardiac troponin I concentrations (Siemens Troponin I-Ultra Assay) in chaired monkeys, study 4. Note. D = day; H = hr.
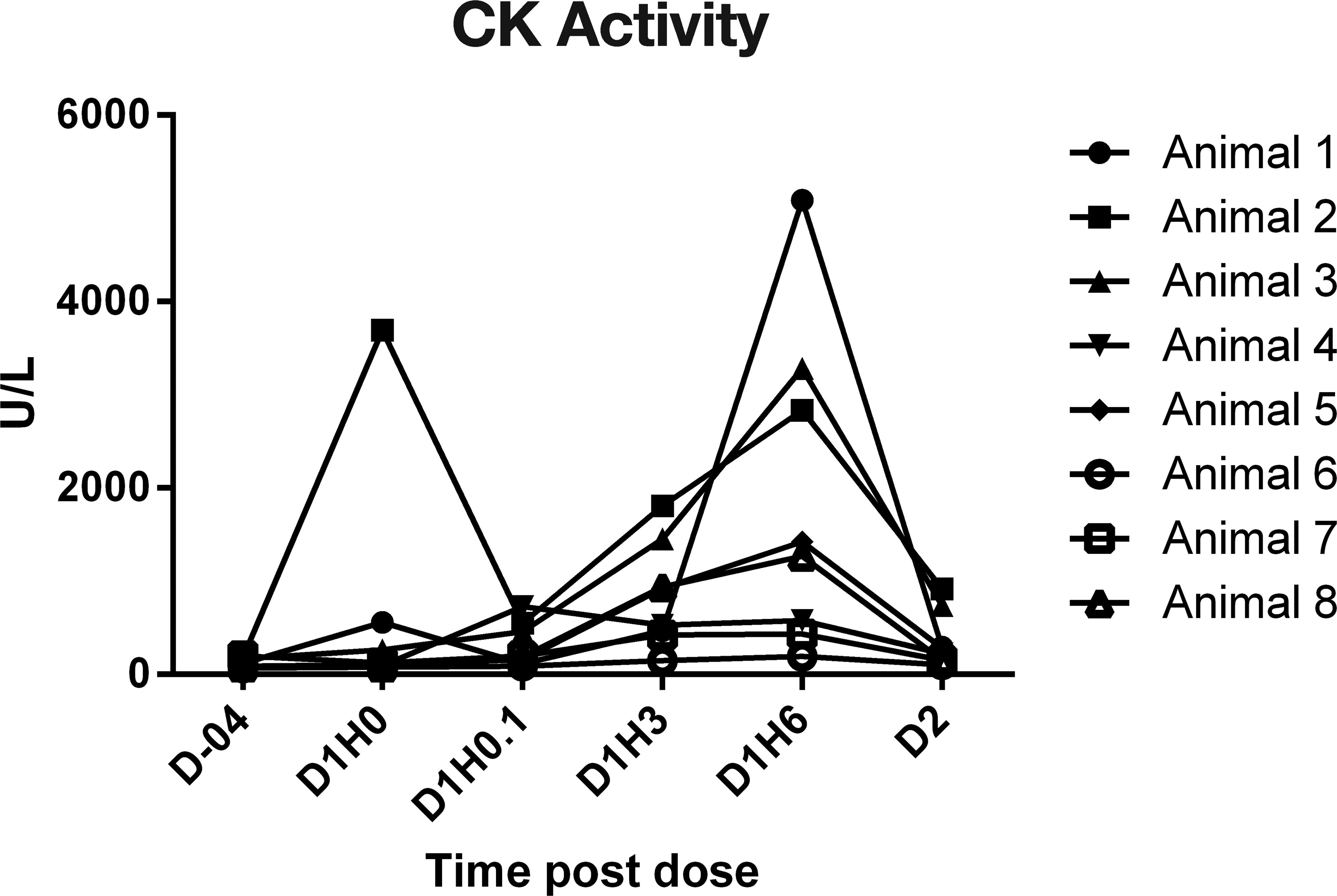
Serial creatine kinase activity results in chaired monkeys, study 4. Note. D = day; H = hour.
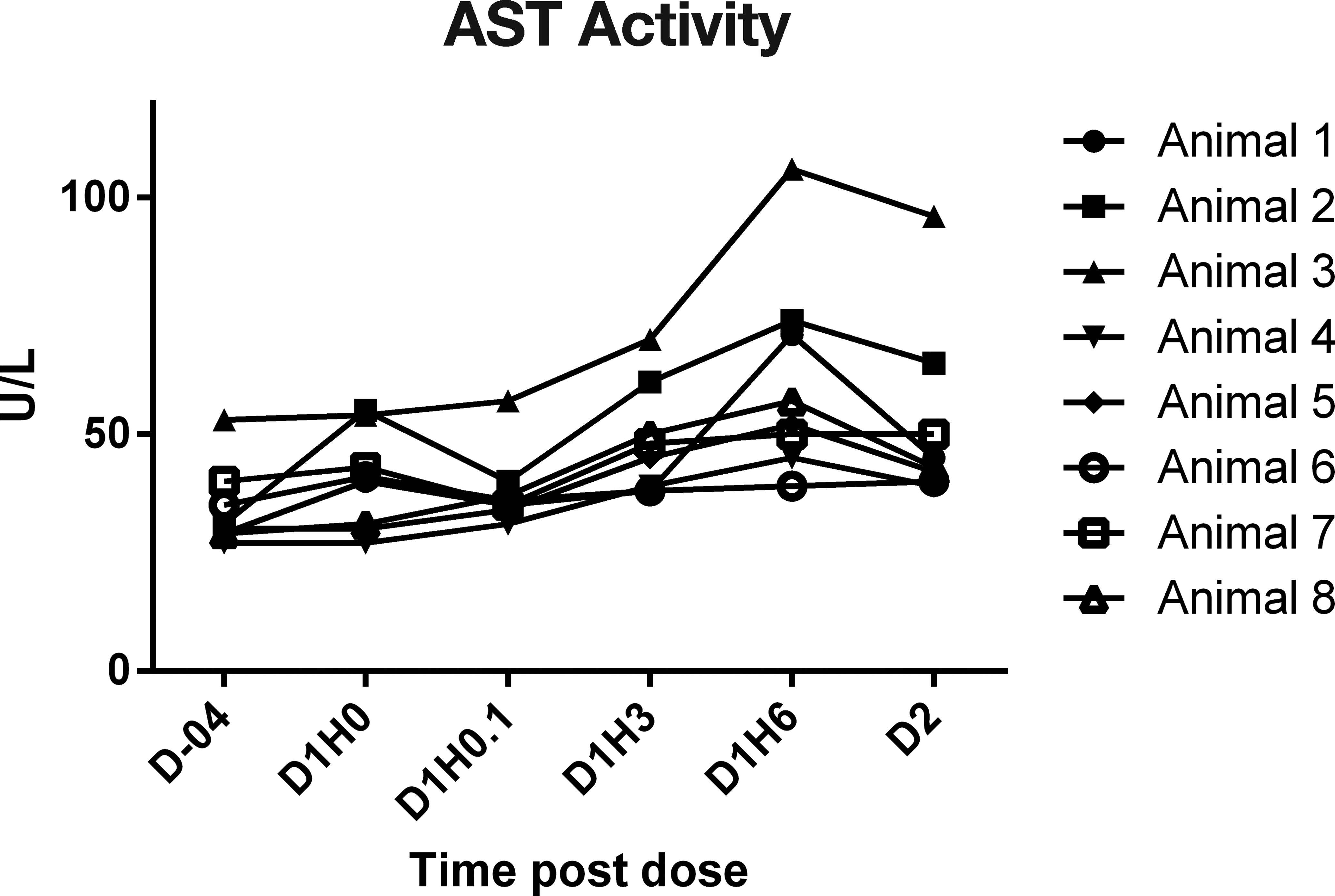
Serial aspartate aminotransferase activity results in chaired monkeys, study 4. Note. D = day; H = hour.
The HR and mean BP of each monkey were measured by telemetry (Table 5) in their home cages 1 hr prior to chair restraint, and the mean (±SD) values were 116 ± 22 BPM (range of 78–152 BPM) and 81 ± 6 mmHg (range of 72–91 mmHg), respectively. Chair restraint and IV administration of saline increased HR and BP to 233 ± 11 BPM and 124 ± 11 mmHg, respectively (during the 30-min chair session). Subsequent chairing sessions at 3, 6, and 22 HPD produced similar increases in HR to 232 ± 16, 231 ± 14, 228 ± 23 BPM and mean BP to 123 ± 13, 124 ± 12, 124 ± 14 mmHg, respectively. The overall ranges of HR values were 188 to 254 BPM at 3, 6, and 22 HPD. Figures 4 and 5 demonstrate the increased HR and mean BP during 4 periods of restraint for a representative animal (number 5). Note the higher the HR and BP values each time the animal is restrained.
Individual Heart Rate and Mean Blood Pressure Data from Monkeys Dosed with Saline Intravenously (n = 8), Study 4.
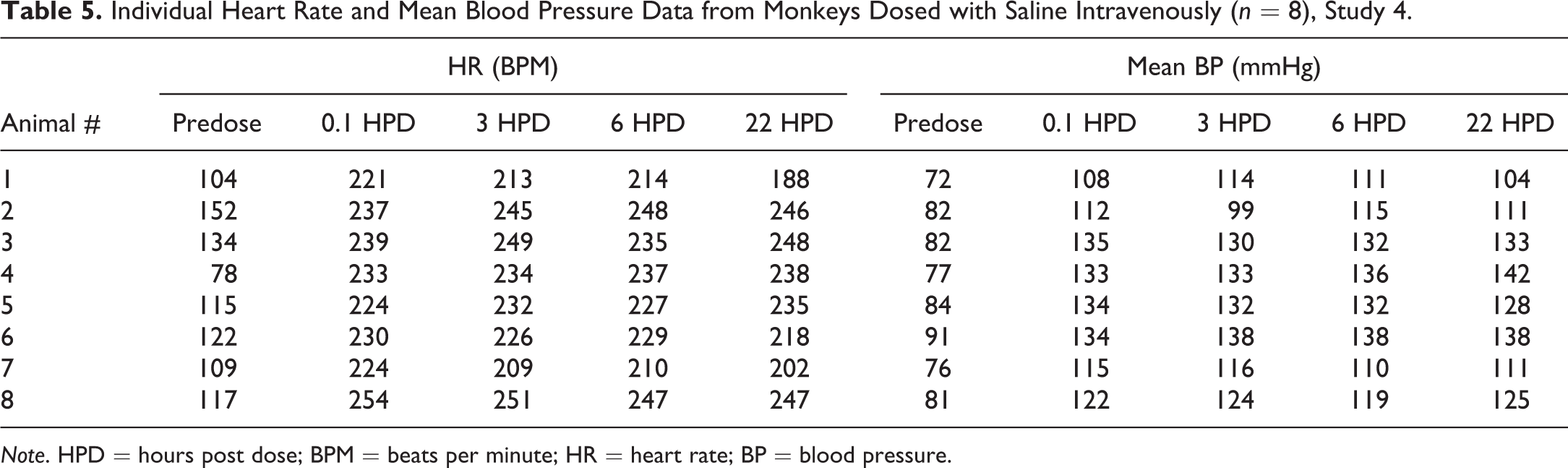
Note. HPD = hours post dose; BPM = beats per minute; HR = heart rate; BP = blood pressure.
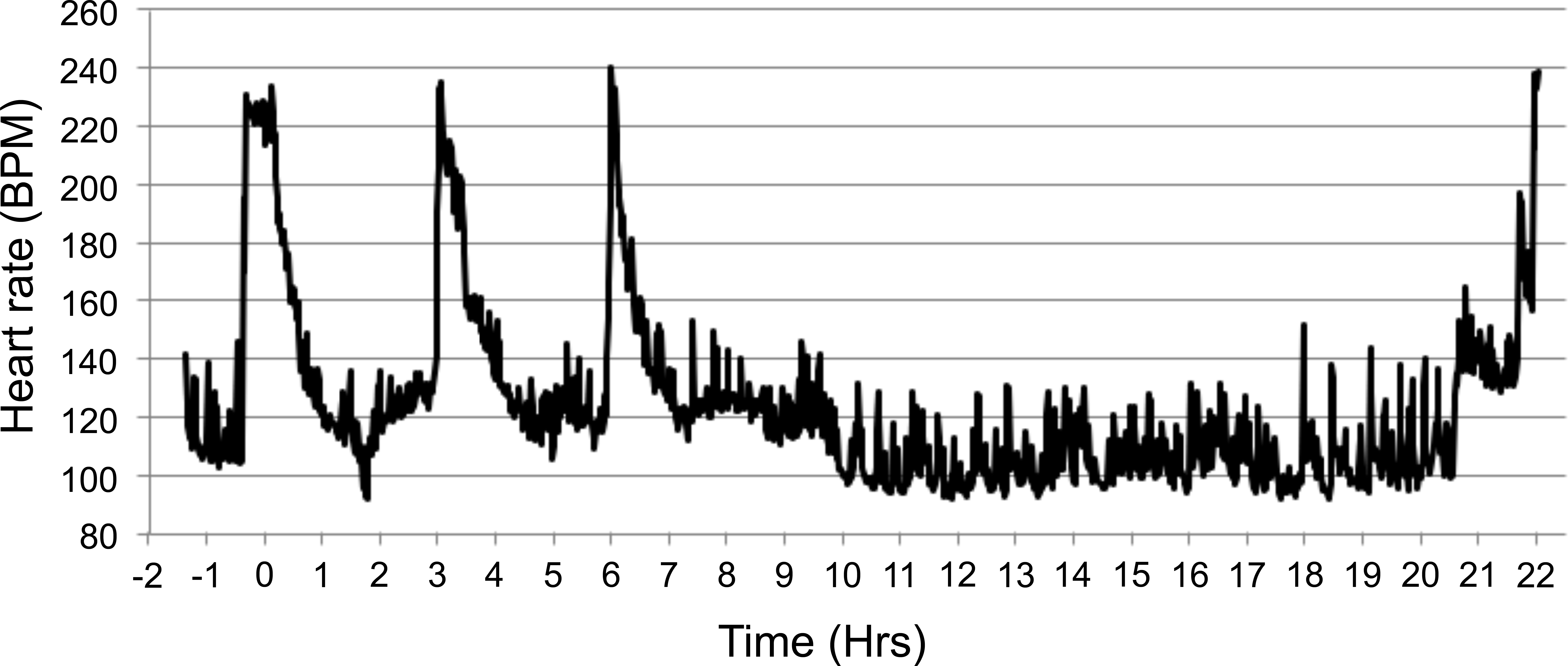
Effect of intravenous administration of saline and repeated chairing on heart rate in a single representative animal (monkey 5). 0 hr = time of dosing. Subsequent chair sessions were at 3, 6, and 22 hr. Lights were turned off at 8.5 hr and on at 20.5 hr, study 4. Note. BPM = beats per minute; Hrs = hours.
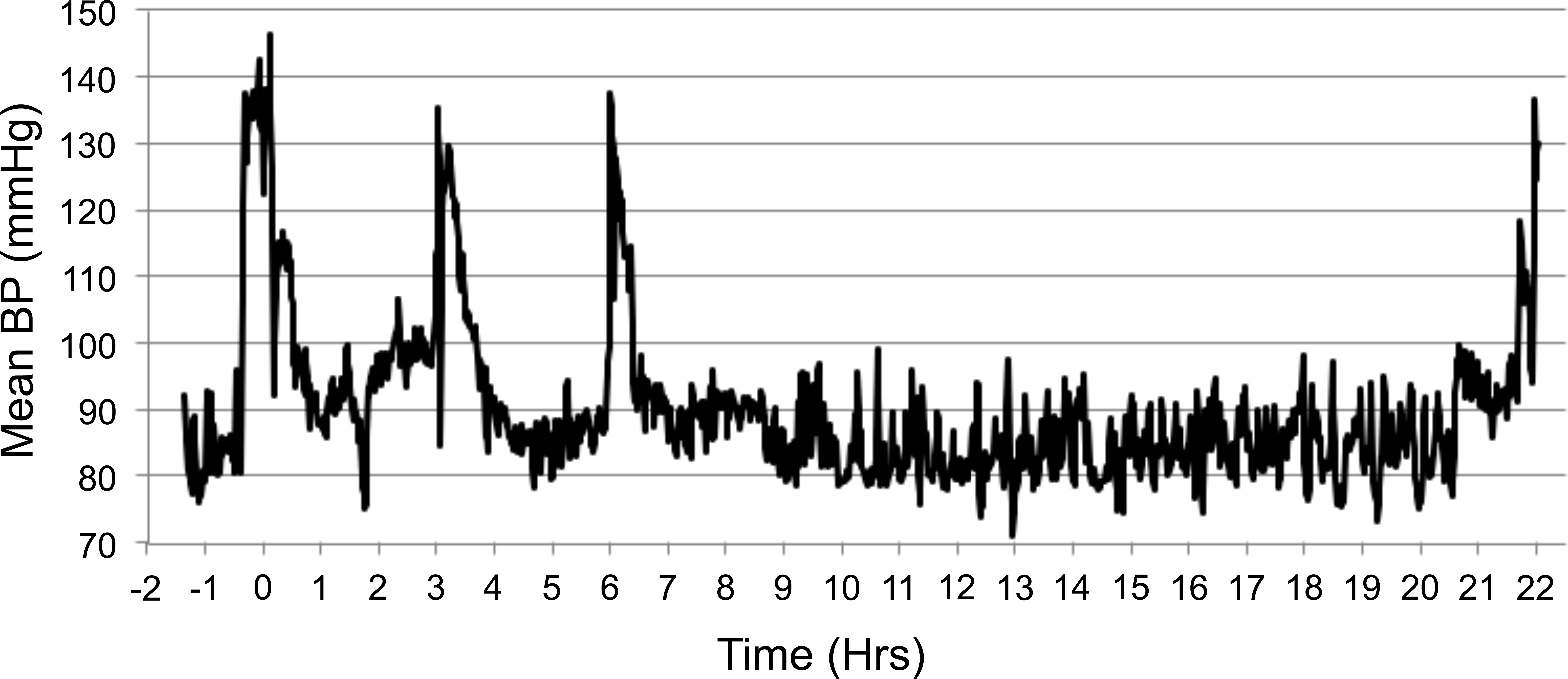
Effect of intravenous administration of saline and repeated chairing on mean BP in a single animal (monkey 5). 0 hr = time of dosing. Subsequent chair sessions were at 3, 6, and 22 hr. Lights were turned off at 8.5 hr and on at 20.5 hr, study 4. Note. BP = blood pressure; Hrs = hours.
A historical review of our database of Siemens Troponin I-Ultra assay results was also performed. We evaluated the first baseline cTnI sample result from 485 monkeys (273 males and 212 females) from studies over a 4-year period. These samples were taken from fasted animals that were chaired, and typically no other blood sampling or other procedures were done around the time of this assessment. We found that 97.5% of the samples have values <0.03 ng/ml. Only 12 of samples had cTnI values ≥0.03 ng/ml, with a range of values 0.03 to 0.14 ng/ml. This represented only 2.5% of all the samples with a similar percentage of males and females with values ≥0.03 ng/ml.
Discussion
Cardiac troponin measurement in cynomolgus macaques can be effectively done in preclinical toxicity studies. Careful attention to experimental design is necessary, including methods for animal restraint and blood sampling in order to minimize the variability of cTnI results. Doing so will allow accurate determination, if a change in cTnI is related to the blood collection technique versus a test article–related effect. We have shown with a first-generation Siemens Immulite cTnI assay that normal cynomolgus macaques can have minimal increases in cTnI associated with routine handling that could occur during the conduct of a preclinical safety study. This was demonstrated after intravenous and subcutaneous dosing of saline or oral gavage of 0.5% methylcellulose and 0.1% polysorbate 80 followed by chair restraint for serial blood sampling by venipuncture. All of these animals had previously gone through an acclimation period, including chair restraint, to get used to handling. Typically, the cTnI response peaked at 2, 4, or 6 HPD after administration of commonly used vehicles, and then all returned to baseline at 24 HPD. HRs were not measured in these animals, but it was hypothesized that the restraint produced markedly elevated HRs, which possibly led to very small areas of localized ischemia in the heart with subsequent cardiac injury and release of cTnI. An alternate hypothesis, which is discussed below, is cytoplasmic cTnI is released into the circulation without cardiomyocyte necrosis.
We then repeated a similar experiment in animals that had vascular access ports which were very well conditioned and accustomed to being handled. After receiving an intravenous injection of saline, serial samples were taken for analysis of cTnI as was done in the previous experiment but using the vascular access ports. None of these animals showed increases in cTnI. HRs (manually determined) were not typically as high (HR ranged from 123 to 177 BPM) at 2, 6, or 24 HPD in the monkeys with vascular access ports compared with 188 to 254 BPM at 3, 6, and 22 HPD in the subsequent experiment, where the HRs were measured by telemetry. Therefore, the highly trained animals with vascular access ports were less excited during the blood sampling procedures, as evidenced by the lower HR values, and did not have increases in troponin likely because they lacked localized ischemic areas in the heart, although histologic evaluation of the heart was not performed on these or any other animals used in these studies. We cannot totally rule out that there would have been cTnI elevations if we would have assessed more animals with vascular access ports, but we repeated these finding on two separate occasions in these animals (days 1 and 8).
The monkeys that were used in the telemetry experiment were acclimated to handling but were not as well acclimated as the animals with vascular access ports. These animals were telemeterized to get a more thorough evaluation of the cardiovascular response, and the cTnI was measured by a second-generation troponin assay, Siemens Troponin I-Ultra assay, a more sensitive assay than the Siemens Immulite cTnI assay. This experiment again demonstrated that elevations occurred in cTnI associated with the handling of these animals. HR and mean BP in these animals increased 2× and 1.5×, respectively, compared with their baseline values, and typically reached these magnitudes during each restraint period. It was hypothesized that these repeated changes in the cardiovascular system were associated with local ischemic events in the hearts, with subsequent cardiac injury and release of cTnI. Because it takes some time for the troponin to be released from a cardiac myocyte, this elevation was not seen 5 min after the dose of saline but was first noticed at the 3 hr time point and peaked at 3 or 6 HPD. Note also due to the small magnitude of the injury in the heart that likely occurred, the cTnI elevations were not sustained (below lower limit of quantitation at 22 HPD). This probably is due to minimal damage that was induced in the heart as well as the limited half-life of troponin which was determined to be 1.85 hr in the dog and 0.8 hr in the rat (Dunn et al. 2011). Similar half-life in monkeys is likely but has not been published. We have consistently only seen minor, fairly transient cTnI responses associated with a stress of multiple restraints as demonstrated above. Furthermore, a review of our historical database showed that after only a single restraint most of the time (97.5%) values were below the limit of quantitation of the Siemens Troponin I-Ultra assay. Only rarely (2.5%) were values above the limit of quantitation of this assay, and the greatest magnitude of this response was 0.14 ng/ml after a single restraint, which was less than the highest concentration (0.43 ng/ml) seen in the animals with serial periods of restraint. This suggests that the duration of restraint and conduct of multiple restraints can contribute to the magnitude of cardiac injury and/or they capture a delayed cTnI response. Therefore, when planning for sampling in a study, it is best to schedule the bleeding of animals to evaluate a cTnI response away from times in the study where multiple bleeds are necessary for other procedures. For example, assessing the cTnI response after multiple samples are taken for toxicokinetic analysis may not be ideal. However, with a larger insult and/or ongoing cardiomyocyte necrosis, increases in cTnI can be much greater in magnitude than those seen associated with chair restraint and can be sustained past 24 HPD. This was recently demonstrated in a study where monkeys were administered isoproterenol, and vasopressin and cTnI concentrations peaked at 4 to 6 hr after a single dose and remained increased 3 to 4 days after dosing (Minomo et al. 2009). At the highest dose of isoproterenol used, peak mean cTnI values were ∼10 ng/ml, utilizing an ELISA. Myocardial necrosis was confirmed histologically in these animals. Lower doses of isoproterenol may lead to shorter periods of time of increased cTnI.
In addition to the changes in cTnI concentrations in the telemeterized monkeys, there were increases in CK and AST activities, with no increase in ALT. These changes were likely due to the muscle injury but based on the lack of specificity of these analytes, it cannot be determined if this was due to cardiac or skeletal muscle injury. Based on the cTnI responses, there was likely cardiac injury but concurrent skeletal muscle injury associated with restraint was also likely. Skeletal muscle injury is further supported in the animal with an elevation in CK values on day 1 prior to dosing, without concurrent increase in cTnI concentrations.
Schultze et al. (2015) have performed similar experiments to those discussed above. They looked at cTnI concentrations in resting NHP as well as those that had been shamed dosed by oral gavage or intravenous saline injection. The main differences between the studies by Schultze et al. and our studies were the following: the assay they used for measuring cardiac troponin was the Singulex Erenna cTnI assay which is reported to have a detection limit of <0.001 ng/ml, which is much lower than the assays used above; the assays we used are more typical of what are used routinely in a preclinical safety assessment study. They also were able to collect blood samples with minimal stress using the Dilab automated blood sample collection system with no restraint of the animals. This type of collection is not typically used routinely, and the chair restraint that was used in our studies is more typical of what is done in a preclinical toxicology study. We also had multiple collections which may be done to capture the kinetics of a cTnI response during a study where they only had a single dose/restraint period. They showed that baseline values of cTnI in resting cynomolgus monkeys were in the range of 0.2 to 9.6 pg/ml, with a mean value of 2.5 pg/ml (0.0025 ng/ml) which is less than the detection limit of either of the Siemens’ assays used in the current experiments. They further showed as we did that there were cTnI responses in some animals that were sham dosed. Of the 10 animals that were sham oral dosed, 2 had values of ∼ 60 or 215 pg/ml, and 4 of the 9 animals that were dosed intravenously had increases in cTnI with values ≤50 pg/ml. We saw similar values with the Siemens Troponin I-Ultra Assay but based on the lack of standardization of detection antibodies and calibrator material, it is difficult to compare the magnitude of the values of 1 analyzer directly to another. Although the Singlex Erenna and Siemens Troponin I-Ultra assays are both calibrated to the National Institute of Standards and Technology (NIST) cardiac troponin standard, they do use different detection antibodies.
We further documented the marked increase in HR and mean BP that occur in monkeys that are chair restrained for procedures such as dosing or blood collection. Although none of these animals went to necropsy to microscopically assess the heart, as we stated above, we postulated that the marked increases in HR and mean BP led to small localized ischemic areas in the heart, which caused cardiac injury and subsequent release of cTnI. Endogenous release of catecholamines in rabbits has been shown to increase BP and HR and precedes myocardial necrosis (Downing and Chen 1985). Pacing the hearts of dogs is known to induce cardiac troponin release and people with supraventricular tachycardia also have increases in cardiac troponin (Ben Yedder, Roux, and Paredes 2011; Chow et al. 2010; O’Brien et al. 2006). In people with supraventricular tachycardia, the duration of the arrhythmia did not correlate with the troponin concentration, but the maximal HR correlated with magnitude of the cardiac troponin increase (Ben Yedder, Roux, and Paredes 2011; Chow et al. 2010). Tachycardia, due to the shortening of diastole, can lead to a reduction in oxygen supply to the myocardium which could lead to myocardial necrosis. An alternative explanation for increase in cardiac troponin is that it is released into the circulation without cardiac necrosis. Not all cardiac troponin is bound in the contractile apparatus of the cardiac myocyte and some (5–8%) is free in the cytosol, and it is postulated that it could be released associated with processes other than myonecrosis (Herman et al. 2006; White 2011; Zhang et al. 2008). It is further suggested that this would result in a transient increase in cardiac troponin with a rapid rise and fall within 24 hr similar to what we saw in our studies. Other potential mechanisms that have been suggested that can lead to the release of troponin without necrosis include apoptosis, normal myocyte turnover, cellular release of proteolytic troponin degradation products, increased cellular wall permeability, or formation and release of membranous blebs (White 2011). This is further supported by the very low fluctuating levels of cTnI that can be detected in the circulation of normal animals using the newer ultrasensitive assays such as the Singulex Errena cTnI (Schultze et al. 2015, 2009). Although these mechanisms for release of cardiac troponin from intact cells cannot be totally ruled out, minimal myocellular necrosis leading to transient elevation of cardiac troponin also cannot be ruled out.
cTnI can be used successfully to assess for cardiac toxicity in preclinical safety studies utilizing NHP. To detect an increase in cTnI, it is critical that sampling is done at the appropriate time(s) postdose to be able to document the increase in cardiac troponin. The appropriate time course is going to be dependent on the type of toxicant used, the dose level, the frequency of dosing, and the mechanism for cardiac injury. For a toxicant that induces cardiac necrosis very rapidly, a peak response occurs early, and if higher doses of a toxicant are used this response can continue for a prolonged period of time due to the ongoing necrosis. It is critical to evaluate concurrent control animals as well because as has been shown above, chair restraint itself may lead to release of troponin, which can complicate the assessment of this biomarker. Assessing the magnitude and the time course of the response in the concurrent controls compared with those animals that receive the test article, as well as relative to baseline concentrations, should lead to a correct interpretation of the cardiac troponin response and allow for accurate cardiac risk assessment in preclinical safety studies.
Footnotes
Acknowledgments
The authors like to thank Kristen Borg, Carrie Michon, Mary Ellen Christina, Deborah Burt, and Dingzhou Li as well as other laboratory and technical staff for their assistance with these studies and/or preparation of the manuscript.
Author Contribution
Authors contributed to conception or design (WR, RB, PH, SL, MS, DB, JS); data acquisition, analysis, or interpretation (WR, RB, PH, SS, SL, MS, DB, JS); drafting the manuscript (WR, JS); and critically revising the manuscript (WR, RB, PH, SS, SL, MS, DB, JS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
