Abstract
N-phenylanthranilic acid is a chloride channel blocker that causes renal papillary necrosis in rats. Studies were conducted in two strains of male rats to evaluate novel biomarkers of nephrotoxicity. Han-Wistar rats were given daily oral doses of 50, 350, or up to 700 mg/kg/day of NPAA, and Sprague-Dawley rats were given 50 or 400 mg/kg/day of NPAA. Rats were euthanized on days 8 and 15. The candidate kidney injury biomarkers renal papillary antigen-1 (RPA-1, for collecting duct injury), clusterin (for general kidney injury), α-glutathione-S-transferase (a proximal tubular marker), and µ-glutathione-S-transferase (a distal tubular marker) were measured in urine by enzyme immunoassay. Characteristic degeneration and necrosis of the collecting duct and renal papilla were observed in Han-Wistar rats at the high dose on day 8 and at the mid and high doses on day 15, and in Sprague-Dawley rats given the high dose on days 8 and 15. Increases in urinary RPA-1, and to a lesser extent urine clusterin, were generally associated with the presence of collecting duct injury and were more sensitive than BUN and serum creatinine. On the other hand, decreases in α-glutathione-S-transferase without proximal tubule lesions in both strains and decreases in µ-glutathione-S-transferase in Sprague-Dawley rats only were not associated with morphological proximal or distal tubule abnormalities, so both were of less utility. It was concluded that RPA-1 is a new biomarker with utility in the detection of collecting duct injury in papillary necrosis in male rats.
Keywords
Introduction
N-phenylanthranilic acid (NPAA) is a biphenyl analogue of fenamic acid and chloride channel blocker that causes renal papillary necrosis (RPN) in rats after repeated oral dosing for fourteen days (Williams et al. 2003). Animal models of RPN have used agents such as 2-bromethanamine and nonsteroidal anti-inflammatory drugs including cyclooxygenase inhibitors (Bach and Bridges 1985a; Bach and Bridges 1985b; Bach et al. 1991; Bach and Nguyen 1998; Kahn et al. 1998), but NPAA provides a better model of chronic RPN free of gastrointestinal toxicity (Hardy and Bach 1984; Thanh et al. 2001; Williams 2003). Further, RPN induced by NPAA after repeated dosing is considered more representative of drug-induced RPN than the very acute effect resulting from administration of the widely used 2-bromoethanamine.
Identification of improved preclinical biomarkers of nephrotoxicity is currently an unmet safety biomarker need. Conventional markers employed in regulatory toxicology studies (blood urea nitrogen, serum creatinine, urine cytology, and semiqualitative dipstick evaluation) lack both sensitivity and specificity, particularly for detection of RPN (Bach and Nguyen 1998). Renal papillary necrosis is a nephrotoxicity with few biomarker changes except in advanced disease, yet it is important because of its prevalence, as it underlies analgesic nephropathy in humans (Burry 1978). Although advanced imaging techniques are revealing undiagnosed RPN in the clinic (Lang et al. 2004), there is a strong need for biomarkers to diagnose RPN at early stages of the disease.
Several urinary proteins have been proposed as sensitive biomarkers of renal injury, some with the potential to characterize the localization of kidney lesions. Among these candidate biomarkers, four were selected for evaluation by the Health and Environmental Sciences Institute (HESI) Biomarkers Nephrotoxicity Working Group: renal papillary antigen-1 (RPA-1), clusterin, α-glutathione-S-transferase (α-GST), and µ-glutathione-S-transferase (µ-GST). Renal papillary antigen-1 is specifically expressed by collecting duct cells (Price et al. 2010) and is a renal papillary antigen released in the urine in the event of collecting duct injury (Hildebrand et al. 1999). Renal papillary antigen-1 corresponds to the antigen recognized by the monoclonal antibody PapX 5C10 produced after immunization of mice with material prepared from rat papilla homogenate (Falkenberg et al. 1996). The identity of RPA-1 and its biological role remain unknown, but it is considered as a leakage marker of collecting duct injury in the rat and, as such, a potential biomarker for the detection of renal papillary necrosis in the rat. Clusterin has been shown to be upregulated and released in the urine when the kidney is damaged (Rosenberg and Silkensen 1995). Clusterin has no specific renal location and has been suggested to play an anti-apoptotic role or to be involved in cell protection, cell aggregation, and cell attachment during tissue remodeling and repair (Rosenberg and Silkensen 1995). α-Glutathione-S-transferase is localized in the proximal tubule in both the rat and the human, whereas in the distal tubule, µ-GST (GSTYb1) is the isoform found in rats and Π-GST is the isoform found in humans (Rozell et al. 1993). The increased presence of GSTs in the urine after nephrotoxic injury is attributed to leakage from the tubular epithelial cells into the tubular lumen secondary to cell damage (Harrison et al. 1989).
The objective of this work, conducted as part of the HESI committee on biomarkers of nephrotoxicity program, was to evaluate whether the novel candidate urinary biomarkers RPA-1, clusterin, α-GST, and µ-GST report renal injury with better sensitivity and diagnostic utility than conventional clinical pathology parameters in rats given repeated oral doses of NPAA. A similar study was previously conducted in the same context with the proximal tubular toxicant cisplatin (Gautier et al. 2010). The rat is the most extensively studied species in investigations of nephrotoxicity and is a standard rodent species used in preclinical safety assessment. Two strains of rat commonly used in the pharmaceutical industry (Han-Wistar and Sprague-Dawley) were used in this study to assess the robustness of these candidate novel biomarkers.
Materials and Methods
Animals and Husbandry
Male Wistar Hannover rats (substrain Han-Wistar rat: AZHsdBrlHan: Wist) were supplied at eight to ten weeks of age by the AstraZeneca Rodent Breeding Unit (Alderley Park, Cheshire, UK) and weighed 220–270 g at the start of treatment. Rats were acclimatized for one week and were housed five per cage in solid-bottom cages except during urine collection periods, when they were housed individually in metabowls. Rats were kept on a standard twelve-hour light/dark cycle and given RM1 (E) SQC pelleted diet (Special Diets Services Ltd., England) ad libitum. Filtered tap water was provided ad libitum from the site drinking system.
Male Sprague-Dawley virus-antibody–free rats [Crl: CDTM IGS] at ten to twelve weeks of age and weighing 250–450 g at initiation of dosing were obtained by GlaxoSmithKline from Charles River Laboratories, Inc., Raleigh, NC. Following an acclimation period of at least two days, rats were housed individually in clear plastic cages with Alpha-dri bedding (Shepherd Specialty Papers, Inc., Kalamazoo MI) and kept in an environment with a temperature of 64°F–79°F, 30–70% relative humidity, and a twelve-hour light/dark cycle. Rats were given 5002 Certified Rodent Diet (PMI Nutrition International, Brentwood, MO) ad libitum. Filtered tap water was available ad libitum from an automated drinking watering system.
Both studies were conducted in compliance with the principles specified in national and international regulatory authority test guidelines on the toxicity testing of new drugs. All animal husbandry and experimental procedures were in accordance with the Guide for the Care and Use of Laboratory Animals (NIH Publication, 25, No. 28, August 16, 1996), or UK Home Office Guidelines (Animals [Scientific Procedures] Act 1986) in accordance with Standard Operating Procedures of Safety Assessment and Laboratory Animal Sciences in effect at GlaxoSmithKline and AstraZeneca on the day a procedure was performed, and were approved by the Institutional Animal Care and Use Committee of the GlaxoSmithKline (Sprague-Dawley study), or an Internal Ethics Committee and the UK Home Office guidelines (Wistar study).
Study Design and Procedures
The dosage regimens and experimental procedures are summarized in Table 1. In both studies, NPAA (Batch number 11310MA, Sigma-Aldrich, St. Louis, MO) in 1.25% carboxymethylcellulose or the same volume of vehicle (10 mL/kg) was administered once daily by oral gavage for seven or fourteen consecutive days. The first day of dosing was designated as day 1.
Dosage regimens, numbers of animals, and timing of key procedures by strain.
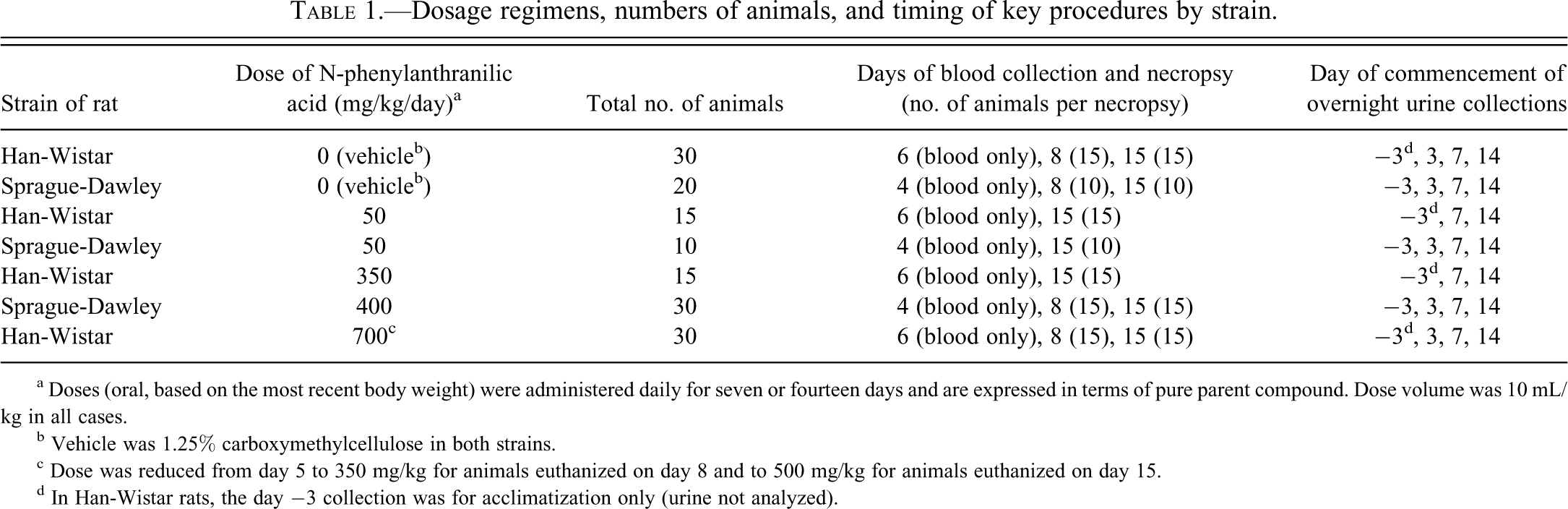
a Doses (oral, based on the most recent body weight) were administered daily for seven or fourteen days and are expressed in terms of pure parent compound. Dose volume was 10 mL/kg in all cases.
b Vehicle was 1.25% carboxymethylcellulose in both strains.
c Dose was reduced from day 5 to 350 mg/kg for animals euthanized on day 8 and to 500 mg/kg for animals euthanized on day 15.
d In Han-Wistar rats, the day −3 collection was for acclimatization only (urine not analyzed).
Doses were selected based on dose range-finding studies in each strain, as toxicokinetic data were not available. For Han-Wistar rats, a dose of 700 mg/kg/day given orally for fourteen days was found to be the maximal tolerated dose. In the Han-Wistar biomarker study, the high dose of 700 mg/kg/day was reduced because of excessive toxicity to 350 mg/kg/day on day 5 for the rats euthanized on day 8, and to 500 mg/kg/day for the rats euthanized on day 15. Blood chemistry showed small but statistically significant increases in plasma urea concentrations in treated rats on days 8 and 15. No other clinical chemistry changes, including plasma creatinine or transaminase values, were associated with the administration of NPAA.
Dose selection for Sprague-Dawley rats was based on dose-range-finding studies in which oral doses ≥800 mg/kg/day of NPAA for up to seven days in Sprague-Dawley rats were associated with systemic manifestations including morbidity, gastric ulceration, and renal papillary necrosis, whereas seven daily doses of 400 mg/kg/day produced renal papillary necrosis in the absence of systemic toxicities. Based on these data, Sprague-Dawley rats were given 0, 50, or 400 mg/kg/day of NPAA for the biomarker study based on tolerance rather than directly matching the doses used for Han-Wistar rats.
Animals were examined clinically and weighed daily. Food and water consumption were also measured (data not shown). Han-Wistar rats were allowed food ad libitum during all overnight urine collections and prior to the scheduled necropsies on days 8 and 15. Sprague-Dawley rats were fasted overnight during urine collections and before the scheduled necropsies on days 8 and 15. An interim urine collection was also performed on days 3/4 and 7/8 in both strains. For urine collections, all animals were individually placed in metabolism cages or bowls, where they had free access to drinking water. Urine was collected over ice over seven hours (Han-Wistar rats only, results not shown) and during an overnight period of sixteen or seventeen hours for Sprague-Dawley and Han-Wistar rats, respectively. At the end of the collection period, urine volume was recorded, then centrifuged at 500 × g for ten minutes within two hours from sample collection for cytologic examination of urinary sediment (Han-Wistar rats only) and urinalysis. Urine samples for the novel biomarker assays were prepared by addition of stabilizing buffer (Argutus Medical, Dublin, Ireland) in the ratio of one volume buffer to four volumes urine, and immediately frozen in cryo vials (2 mL, Nalge Nunc Int., Denmark) at −70°C until analyzed by enzyme immunoassay (EIA).
Euthanasia was by exsanguination under anaesthesia induced by halothane (ICI, UK) in Han-Wistar rats or isoflurane (Baxter Healthcare, Wayne, PA) in Sprague-Dawley rats. The kidneys were collected, weighed, and prepared for histopathological examination in both strains as described below. The livers were removed and weighed, but not processed in Han-Wistar rats. Blood samples for clinical pathology parameters were collected as heparinized plasma (Han-Wistar) or serum (Sprague-Dawley) for interim blood collections from a lateral tail vein and for terminal collections at necropsy from the inferior vena cava or abdominal aorta.
Histopathological Examination
Kidneys were examined at necropsy for macroscopic abnormalities (findings not shown) and placed in 10% neutral phosphate-buffered formalin. Because papillary necrosis can be apex limited, step sectioning of both kidneys was performed for both strains. Up to six sections were produced and examined for each rat in both studies, and histopathology scoring was performed on the section with the most severe manifestation. Kidneys were sectioned longitudinally for Sprague-Dawley rats and transversely for Han-Wistar rats. Kidneys were fixed for twenty-four to forty-eight hours prior to sectioning, embedding in paraffin, and collection of multiple 4-µm step sections through the papillary tip. Intervening step sections were used for histochemical staining (PAS-Alcian Blue, Han-Wistar rats only) and immunohistochemistry. Sections were stained with hematoxylin and eosin (H&E) and periodic acid–Schiff (PAS)-Alcian Blue for light microscopic examination.
The histopathology examination was conducted according to published best practices (Burkhardt et al. 2010; Crissman et al. 2004). A board-certified veterinary pathologist performed an initial assessment with full knowledge of the treatment groups. After this initial assessment, the pathologist either conducted a “targeted masked evaluation” blinded to treatment group to ensure consistency of diagnoses and grading (Sprague-Dawley rats) or performed quality control by cross-checking slides chosen at random to ensure that gradings had not drifted (Han Wistar rats). In the study with Sprague-Dawley rats, a peer review was conducted by a second board-certified veterinary pathologist. After individual animal pathology reports were produced, an ILSI-HESI pathology working group reviewed representative slides from all studies conducted in order to harmonize nomenclature and severity grading, for creation of a diagnostic lexicon, and to peer review results of studies conducted with all of the nephrotoxicants evaluated by the HESI Committee on Biomarkers of Nephrotoxicity. Histopathology data were assessed, and similar terminologies were combined to remove redundancies. At all stages of the histopathological evaluation, the pathologists had no knowledge of the clinical pathology or biomarker data to avoid any bias in the analysis.
Immunohistochemistry Methods
Immunohistochemistry was performed to confirm the segment of injury. All kidneys from Han-Wistar rats were stained for RPA-1 antigen expression in collecting ducts (RPA-1 mouse monoclonal PapX 5C10 antibody, IgG1 from Prof F. Falkenberg, CIRES) on a Ventana Discovery autostainer (Ventana Medical Systems, Tuscon, AZ), using a standard CC1 pre-treatment, three hours primary incubation, biotinylated rabbit anti-mouse at a dilution of 1:200 (Dako E0464) and DABmap kit (VMS -760124). Immunostaining of the loops of Henle was also performed (PAP1.3 mouse monoclonal 1C11 antibody, IgG from Prof F. Falkenberg, CIRES) using a LabVision Autostainer 720, with the antibody incubated for sixty minutes at room temperature at a dilution of 1:50, then labeled polymer mouse EnVision (K4007 Dako) for thirty minutes, and visualized with 3′3-diaminobenzidine (DAB). Staining intensity in the papilla was quantified on a 0–4+ scale by the pathologist.
For Sprague-Dawley rats, a serial immunohistochemistry procedure was performed on selected kidneys to co-label tissue sections with α-GST (Rozell et al. 1993) and calbindin-D28 (Taylor 1982; Vekaria et al. 2006) for visualization of proximal and distal convoluted tubules, respectively. Similarly, co-labeling with Tamm-Horsfall protein (Vekaria et al. 2006) and aquaporin 2 (Vekaria et al. 2006) was performed for visualization of medullary thick ascending limb and collecting duct, respectively. These experiments were performed on the Ventana Discovery System autostainer, and all reagents, unless specified otherwise, were obtained from Ventana Medical Systems, Inc. (Tucson, AZ). Briefly, sections were deparaffinized and rehydrated. Nonspecific staining was blocked with 3% hydrogen peroxide and protein block (DAKO, Carpinteria, CA). For co-labeling, serial sections were incubated with either polyclonal rabbit anti-aquaporin 2 (Sigma Aldrich, St Louis, MO) at a concentration of 5 µg/mL or mouse monoclonal anti-calbindin-D28 at a dilution of 1:500 (Abcam Ltd, Cambridge, MA) for one hour. Anti-aquaporin 2 or anti-calbindin-D28 antibodies were labeled with goat anti-rabbit IgG biotinylated (1:200, Vector Labs, Burlingame, CA) or horse anti-mouse IgG biotinylated (1:100, Vector Labs, Burlingame, CA), respectively, followed by streptavidin-horseradish peroxidase. Immunoreactivity was visualized with DAB. For double immunostaining, sections stained with Aquaporin-2 were co-labeled with polyclonal sheep Tamm-Horsfall protein antibody (Chemicon, Temecula, CA) at a concentration of 3 µL/mL, and sections stained with calbindin-D28 were co-labeled with rabbit anti-αGST (1:500, CalBiochem, San Diego, CA) for one hour. Anti-Tamm-Horsfall protein and anti-αGST antibodies were labeled with anti-sheep IgG (Jackson ImmunoResearch, West Grove, PA) or anti-rabbit IgG (Vector Labs, Buringame, CA), respectively. Secondary antibodies were detected with streptavidin-AP or Fast Red chromagen. Sections were counterstained with hematoxylin, dehydrated, cleared, and coverslipped.
Traditional Clinical Chemistry Parameters
The blood parameters BUN, creatinine, and total protein and the urine parameters volume, pH, specific gravity/osmolarity, glucose, total protein, creatinine, GGT, and NAG were measured in both strains. In the study with Han-Wistar rats, urine parameters were determined using a Hitachi chemistry analyzer (Maidenhead, UK) or a Clinitek 500 urinalysis instrument (Bayer, Newbury, UK). In the study with Sprague-Dawley rats, assays were performed on an Advia 1650 (Siemens, Tarrytown, NY). In both strains, creatinine in both blood and urine was determined using the Jaffe (alkaline picrate) method, and urinary total protein was measured with the pyrogallol red method.
Enzyme Immunoassays for Exploratory Biomarkers of Nephrotoxicity
Urinary α-GST, GSTYb1 (µ-GST), clusterin, and RPA-1 were measured in rat urine with quantitative solid phase enzyme immunoassays (EIA) developed by Argutus Medical Ltd (Dublin, Ireland) as described by Gautier et al. (2010). The assays were performed on the Triturus automated ELISA instrument (Grifols, UK) for Han-Wistar rats and manually using a microplate reader Spectra Max 340 spectrophotometer (Molecular Devices Corporation, Sunnyvale, CA) for Sprague-Dawley rats. The assay range was 0–100 µg/L for α-GST, µ-GST, and RPA-1 and 0–5 µg/L for clusterin. Commercially available control materials with assigned ranges (Argutus Medical Ltd, Dublin, Ireland) were used to monitor EIA performance daily. Analytical validation of the EIAs included assessment of linearity, accuracy, repeatability (intra-assay precision), and intermediate precision (intralaboratory precision). All assays performed very well with respect to linearity (r 2 ≥ 0.98) and accuracy (91–106%). Estimated repeatability and intermediate precision coefficients of variation were generally <10%. It was concluded that the EIAs for these biomarkers are fit for use in biological qualification studies.
Expression of Urinary Biomarker Data
Preliminary analyses were conducted with urinary biomarker data expressed both as total excretion and normalized to urinary creatinine; the conclusions drawn were similar using both methods. It was first checked that urinary creatinine amounts (concentration × volume) were similar across all dose groups and thus unaffected by treatment. Normalization to creatinine is the most consistent method of data presentation used in the literature. Therefore, all of the urinary biomarker data shown in the tables are expressed as urinary biomarker concentration relative to urinary creatinine concentration. In the text, the term “level” is used to describe the biomarker values expressed in this way. Fold differences in the normalized values were calculated relative to the time-matched control mean value for each strain.
Statistical Analysis
All data are presented as mean ± standard deviation. For each biomarker and sampling day, differences between treated versus time-matched control animals were evaluated by one-way analysis of variance using SAS software (version 9.1, SAS, Inc., Cary, NC). Where applicable, p values were adjusted for multiple testing via the Dunnett test. p < .05 was considered to be statistically significant. Organ weights were subject to analysis of variance followed by trend testing using SAS.
Results
Clinical Observations
In Han-Wistar rats, the dose of 700 mg/kg/day of NPAA was not well tolerated and was associated with body weight loss and clinical observations of decreased activity, altered skin tone, hunched posture, piloerection, ataxia, and collapse in some animals from day 2, when food and water intake were reduced (data not shown). Two animals were euthanized early because of toxicity. On day 5, the high dose was reduced to 350 mg/kg/day in animals sacrificed on day 8 and to 500 mg/kg/day in animals sacrificed on day 15. There was evidence of recovery from day 5 onward, following the dose reduction. Altered skin tone was still present in animals terminated on day 8, but there was recovery by day 15. Red solutes were observed in urine in three animals at the end of the two-week dosing period. There were no compound-related adverse clinical signs in animals dosed at 50 or 350 mg/kg/day. No change in urine volume was observed for any group. In the Han-Wistar strain, group mean water consumption was increased throughout the dosing period in the rats initially given 700 mg/kg/day and in the rats given 350 mg/kg/day, with values reaching approximately 1.5- to twofold of those in control animals. There was no effect on water consumption in animals given 50 mg/kg/day.
In Sprague-Dawley rats, the doses of 400 and 50 mg/kg/day were well tolerated. There were no significant clinical observations.
Organ Weight Changes
In Han-Wistar rats, significant increases in absolute paired kidney weights (13.8% at 350 mg/kg/day and 21.2% at 700/500 mg/kg/day, p < .001) and relative to body weight (10.5% at 350 mg/kg/day and 20.3% at 700/500 mg/kg/day, p < .001) were observed on day 15. Liver weights were unchanged in NPAA-treated Han-Wistar rats relative to controls.
In Sprague-Dawley rats, mean paired kidney weights were increased approximately 17% (absolute and relative kidney to body weight) for rats given 400 mg/kg/day of NPAA on day 8, primarily because of an increase in relative kidney weight in a single rat. Mean paired kidney weights for test article–treated Sprague-Dawley rats did not differ from concurrent controls on day 15.
Macroscopic Observations
Macroscopic findings were limited to bilateral renal enlargement in a single Sprague-Dawley rat given 400 mg/kg/day of NPAA on day 8. This finding was associated with a 2.5-fold increase in % kidney (relative to body) weight compared to concurrent controls.
Histopathological Examination
The main treatment- and dose-related changes in H&E kidney sections in Han-Wistar rats were degenerative and regenerative changes affecting collecting ducts in the papilla and outer medulla/inner cortical regions (Table 2). Minimal to mild papillary necrosis (tubular cell degeneration/necrosis of papilla collecting duct, Figure 1A) was observed on day 8 at 700/350 mg/kg/day, and on day 15 with a dose-responsive increase in incidence and severity in rats given ≥50 mg/kg/day of NPAA. Minimal to moderate focal degeneration/necrosis of corticomedullary collecting ducts was observed on day 8 in 11/13 rats given 700/350 mg/kg/day of NPAA, and on day 15 in 1/10 rats at 50 mg/kg/day, 10/15 rats at 350 mg/kg/day, and 13/15 rats at 700/500 mg/kg/day. Medullary collecting duct regenerative basophilia was associated in some cases. Cortical tubular dilation, cortical tubular hyaline casts, and/or chronic cortical interstitial inflammation (nephritis with mononuclear cell infiltration) were present on day 8 in rats given 700/350 mg/kg/day, and on day 15 at an increased incidence and severity relative to control in rats given
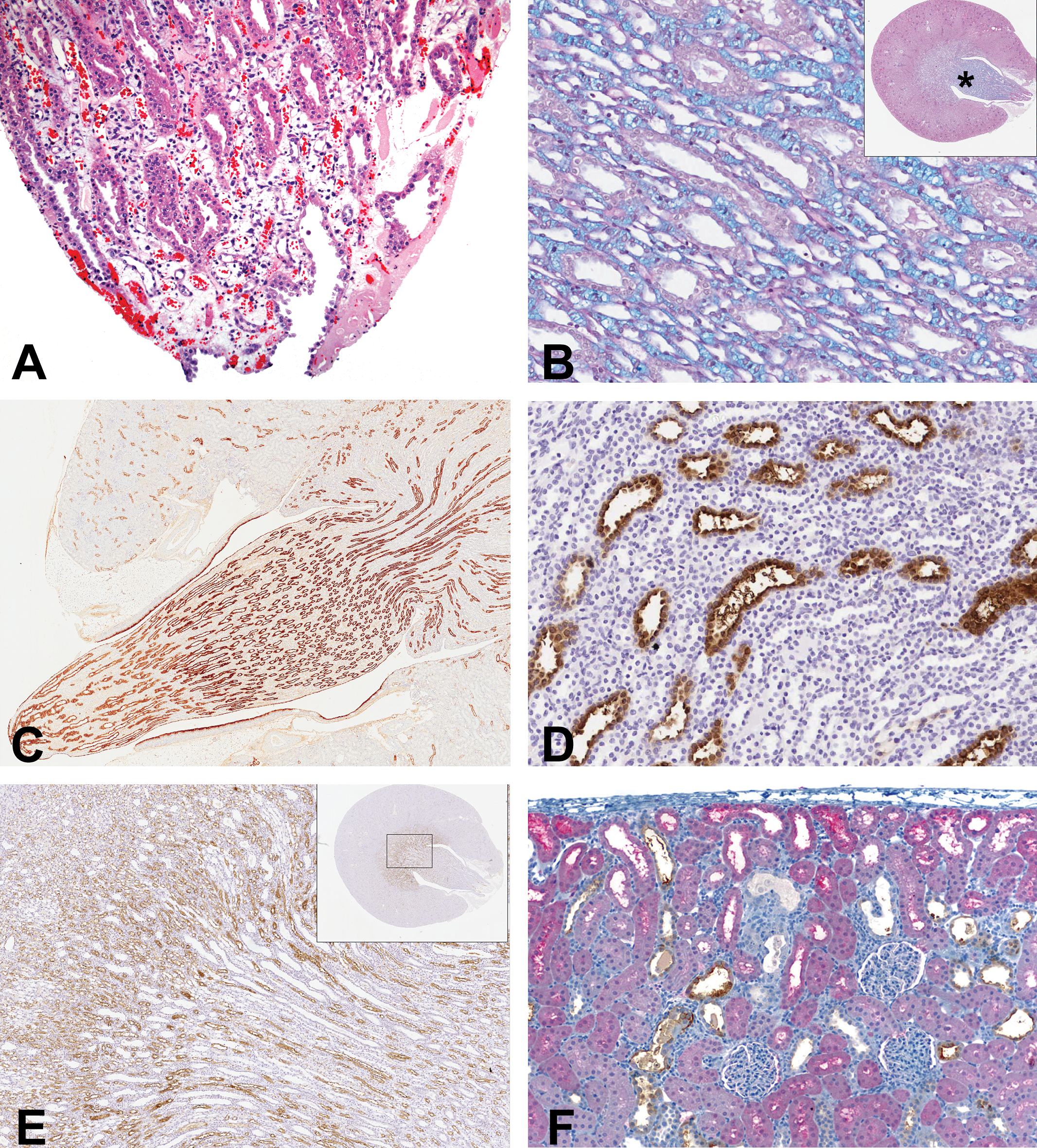
Renal histopathology following NPAA administration. Minimal papillary necrosis of tip with necrosis, hemorrhage, and inflammation of collecting ducts in a Han-Wistar rat given 700 mg/kg/day of NPAA for seven days (A). Hematoxylin and eosin, original magnification ×5. Collecting ducts and loops of Henle (both pink) separated by interstitial acid glycosaminoglycan matrix (blue) in a Han-Wistar rat treated with 700 mg/kg/day of NPAA for seven days (B). Periodic acid-Schiff Alcian Blue, field from mid-papilla indicated by asterisk in upper right thumbnail, original magnification ×20. Immunohistochemistry for RPA-1 in collecting ducts of cortex, outer medulla, and papilla in kidney from control Han-Wistar rat (C). Original magnification ×4. Immunohistochemistry for RPA-1 in collecting ducts of cortex, outer medulla, and papilla in kidney from a Han-Wistar rat treated with 700 mg/kg/day of NPAA for seven days with positive staining of hypertrophic collecting duct and epithelial blebbing into the lumen in mid-papilla (D, ×20). Immunohistochemistry for loop of Henle marker PAP1.3 in Han-Wistar rats treated with 700 mg/kg/day of NPAA for fourteen days showing staining of thin limb in inner medulla and papilla and thick ascending limb in outer medulla. Note that collecting ducts are dilated and unstained (E, ×5). Immunohistochemistry for α-GST in proximal tubules (red chromagen) and calbindin D28 in distal tubule (brown chromagen) in a Sprague-Dawley rat given 800 mg/kg/day for seven days in a dose range-finding study demonstrating morphologically normal segments of proximal tubules labeled with α-GST in the presence of degenerate profiles of calbindin D28–positive distal tubules and collecting ducts (unstained; F, ×10).
Incidence of histopathological observations in kidney of Han-Wistar rats.
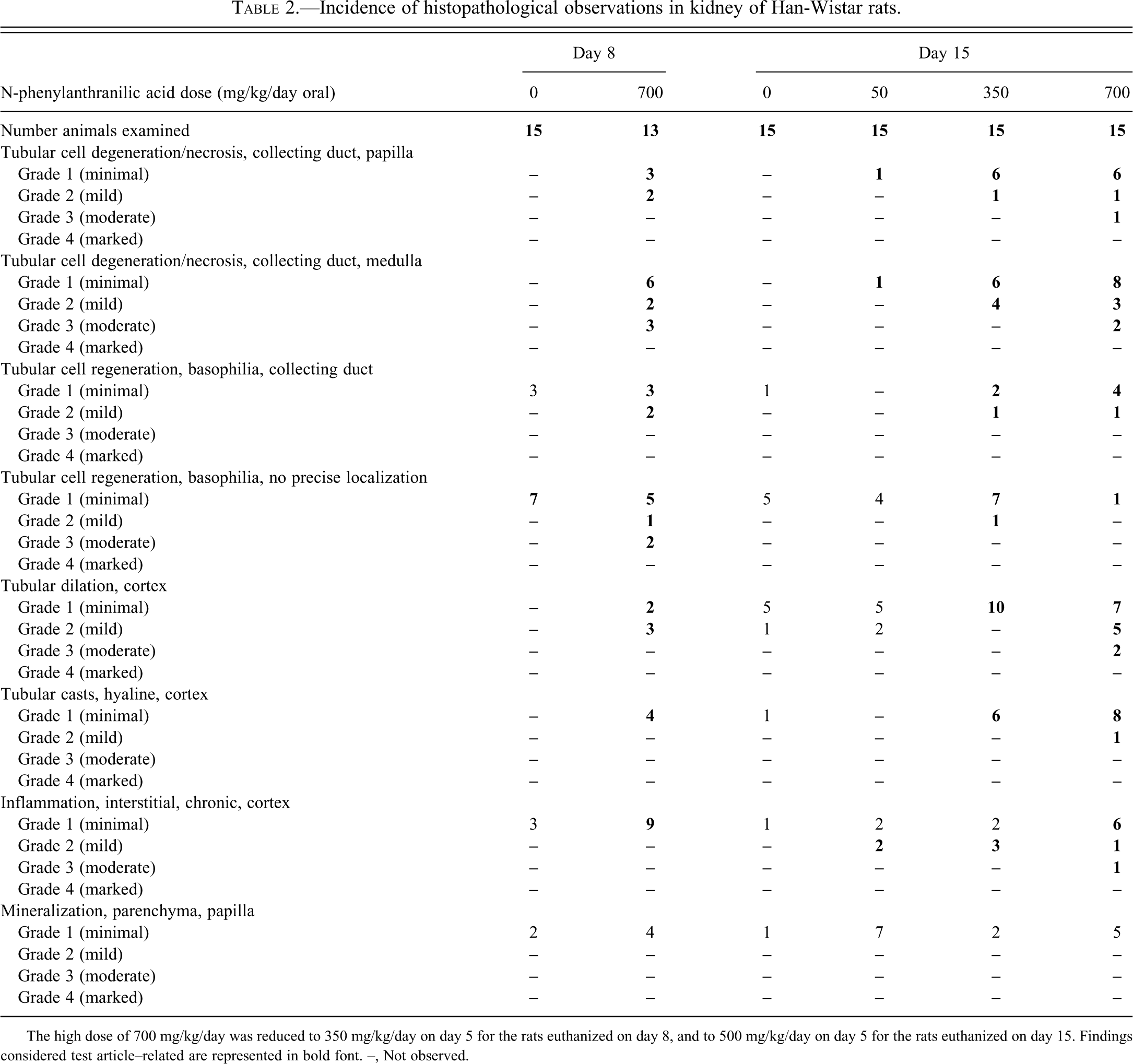
The high dose of 700 mg/kg/day was reduced to 350 mg/kg/day on day 5 for the rats euthanized on day 8, and to 500 mg/kg/day on day 5 for the rats euthanized on day 15. Findings considered test article–related are represented in bold font. –, Not observed.
Periodic acid-Schiff Alcian Blue–stained sections in Han-Wistar rats demonstrated an increase in papillary matrix (glycosaminoglycans, or GAGs) which appeared as blue material between collecting ducts, loops of Henle and papillary GAG-producing interstitial cells (Figure 1B). This increase was most pronounced toward the papilla tip. Image analysis of the papilla showed no significant increase in the ratio of matrix to papilla area, but the absolute area stained by Alcian Blue was increased. Transverse sections of control and treated kidney in Han-Wistar rats were immunostained for the collecting tubule marker RPA-1 using the mouse monoclonal antibody clone PapX 5C10 (Falkenberg et al. 1996), which specifically stains cortical, outer-medullary and inner-medullary/papillary collecting ducts (Figure 1C). Some but not all of the dilated tubules in the cortex were shown to be of collecting duct origin. Renal papillary antigen-1 staining of collecting ducts in all three regions was increased following NPAA treatment at 350 and 750/500 mg/kg/day on day 8 (Figure 1D) and day 15, both for the corticomedullary region, where degenerative and regenerative pathology was evident on H&E staining, and in the papilla, where collecting tubule hypertrophy was observed. This increase combined with the increased area of Alcian blue–positive matrix explains the lack of significant change in the ratio of the two main components of the papilla. Immunohistochemistry for a loop of Henle marker using the PAP1.3 mouse monoclonal antibody clone 1C11 showed no treatment-related effects of NPAA in the papilla or in the thick ascending limbs (Figure 1E).
In Sprague-Dawley rats, minimal to moderate papillary necrosis was identified in the majority of rats given 400 mg/kg/day on day 8, and in all rats given 400 mg/kg/day on day 15 (Table 3). Papillary necrosis was associated with diffuse or segmental dilation of cortical tubular segments in rats given 400 mg/kg/day on days 8 and 15. Minimal to mild cortical tubular degeneration/necrosis was identified in rats given 400 mg/kg/day on days 8 and 15. Tubular cell regeneration was present with a dose-responsive increase in incidence and severity in rats given 400 mg/kg/day on days 8 and 15. Immunohistochemical staining with nephron segment–specific markers α−GST (proximal tubule), calbindin D28 (distal tubule), Tamm-Horsfall protein (medullary thick ascending limb of the loop of Henle), and aquaporin 2 (collecting duct) localized the cortical tubular injury to the distal convoluted tubule, and the cortical tubular dilation to the distal convoluted tubule and collecting duct (Figure 1F). Correlated microscopic findings for Sprague-Dawley rats given 400 mg/kg/day of NPAA with bilateral renal enlargement on day 8 included papillary necrosis with acute inflammation and transitional epithelial hyperplasia, diffuse collecting duct hypertrophy and dilation, cortical tubular degeneration, dilation, and/or basophilia and cast formation, consistent with papillary necrosis with obstructive nephropathy and associated acute pyelonephritis. Morphologic findings for this rat were associated with azotemia and increases in urinary clusterin and µ-GST compared to concurrent control values.
Incidence of histopathological observations in kidney of Sprague-Dawley rats.
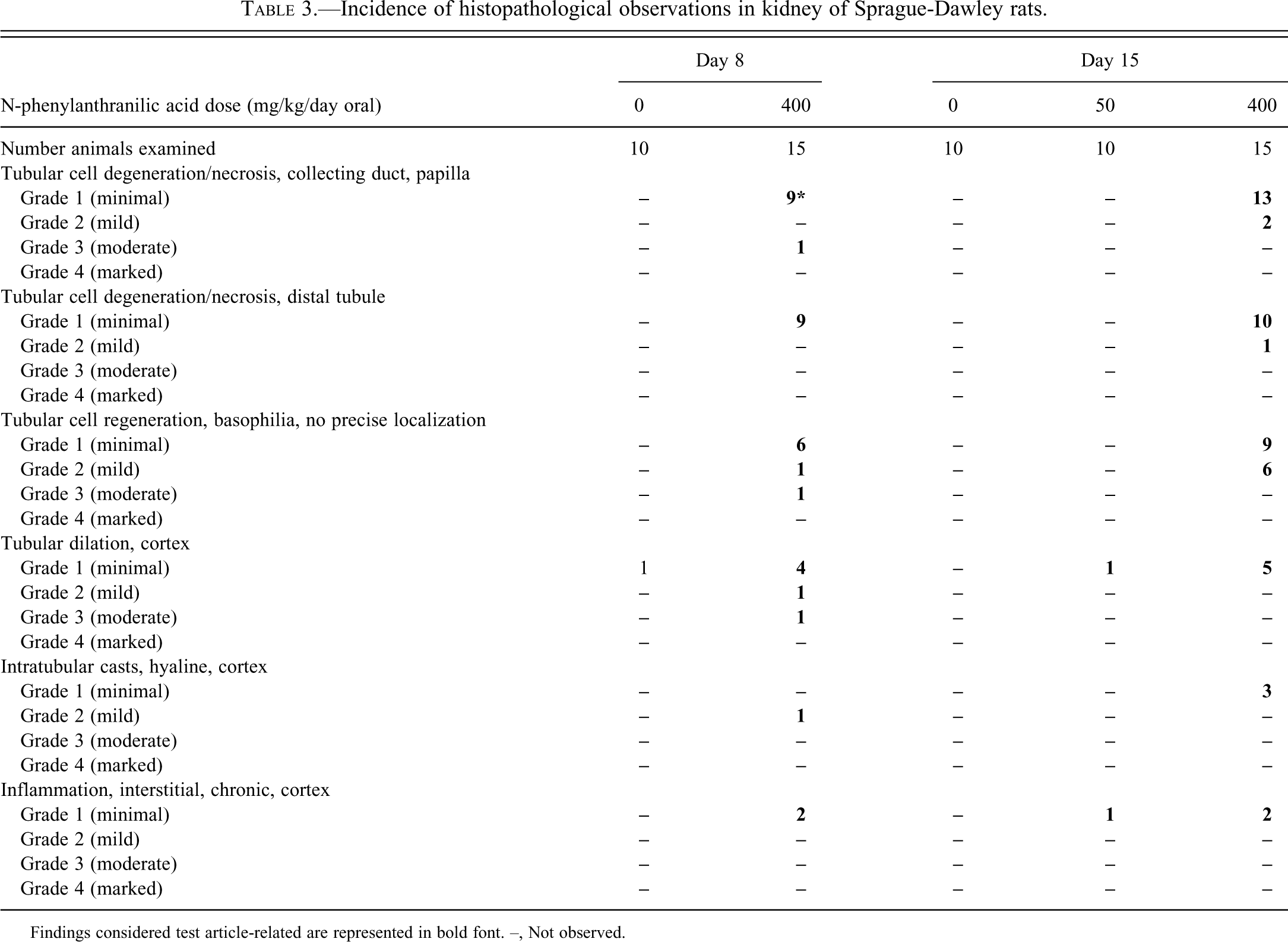
Findings considered test article-related are represented in bold font. –, Not observed.
Changes in Traditional Clinical Biochemical Parameters
In Han-Wistar rats, there was a minimal increase in BUN at the high dose on day 15; however, plasma creatinine, urinary glucose (data not shown), and total protein were not significantly increased (Table 4). Urinary NAG showed dose- and treatment-related increases at 700 mg/kg/day on days 4 and 8 (to 3.8-fold) and days 15 (to 5.1-fold), but the pathology severity scores were not proportionate to the magnitude of the NAG elevations (data not shown). Urinary GGT showed modest increases at the high dose on day 4 (2.2-fold) and day 8 (to 1.5-fold) with return to control range on day 15.
Traditional blood and urine clinical chemistry parameters in Han-Wistar rats.
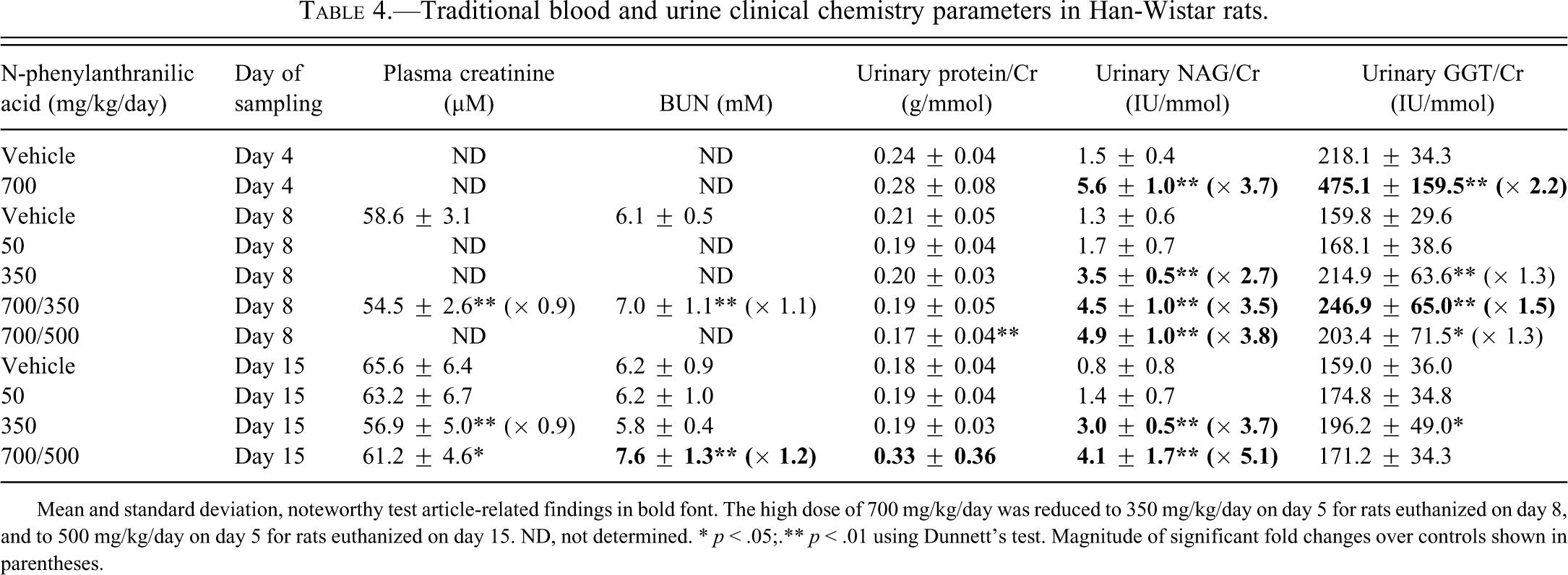
Mean and standard deviation, noteworthy test article-related findings in bold font. The high dose of 700 mg/kg/day was reduced to 350 mg/kg/day on day 5 for rats euthanized on day 8, and to 500 mg/kg/day on day 5 for rats euthanized on day 15. ND, not determined. * p < .05;.** p < .01 using Dunnett’s test. Magnitude of significant fold changes over controls shown in parentheses.
No noteworthy changes were observed in the mean values of traditional clinical pathology parameters in Sprague-Dawley rats (Table 5).
Traditional blood and urine clinical chemistry parameters in Sprague-Dawley rats.
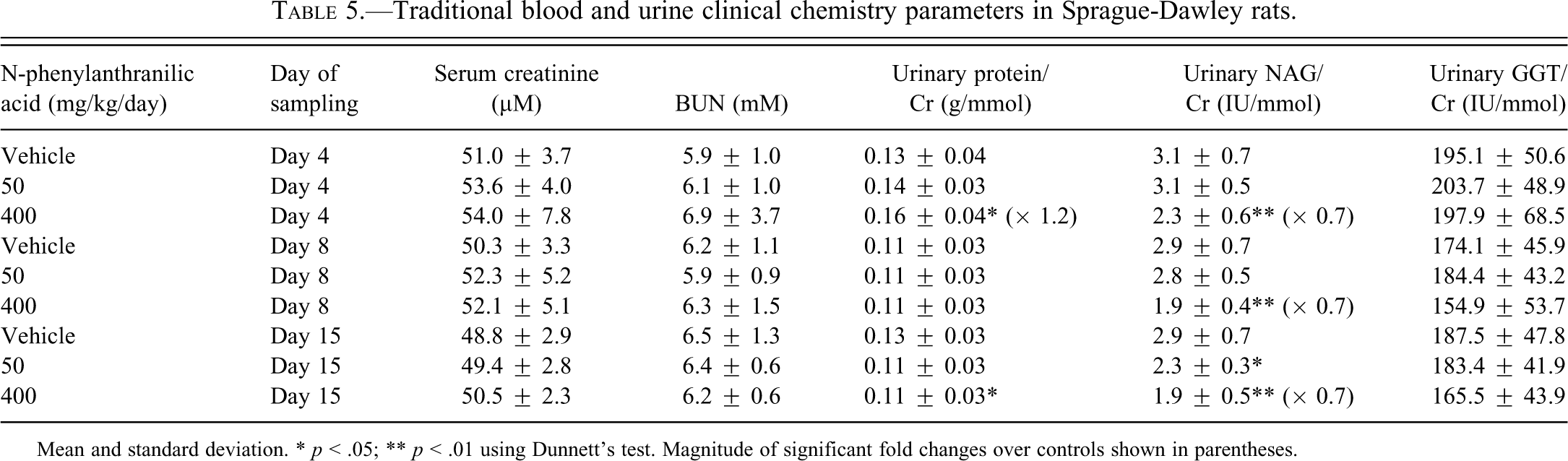
Mean and standard deviation. * p < .05; ** p < .01 using Dunnett’s test. Magnitude of significant fold changes over controls shown in parentheses.
Changes in Novel Urinary Biomarkers
Mean urinary RPA-1 values in Han-Wistar rats given 700 mg/kg/day were increased twenty-six–fold on day 4 and about tenfold for days 8 and 15, compared to time-matched controls (Table 6). A tenfold increase was also seen at 350 mg/kg/day on both days 8 and 15. On day 15, the magnitude of the RPA-1 increase was similar between the high- and mid-dose groups despite the greater severity of collecting duct necrosis/degeneration in the high-dose group on day 15 (Figure 2). Urinary clusterin was increased at 700 mg/kg/day at all collection points during the dose period, and at 350 mg/kg/day on days 8 and 15. The greatest magnitude clusterin increase occurred in the high-dose group on day 15 and was generally proportionate to the pathology grade (Figure 3). Mean urinary α-GST values for rats given 700 mg/kg/day were decreased at all collection points and at 350 mg/kg/day on days 8 and 15 (Figure 4). There were no remarkable changes in urinary µ-GST levels with the exception of two rats given 400 mg/kg/day of NPAA on day 15 with mild (grade 2) collecting duct degeneration/necrosis (data not shown).
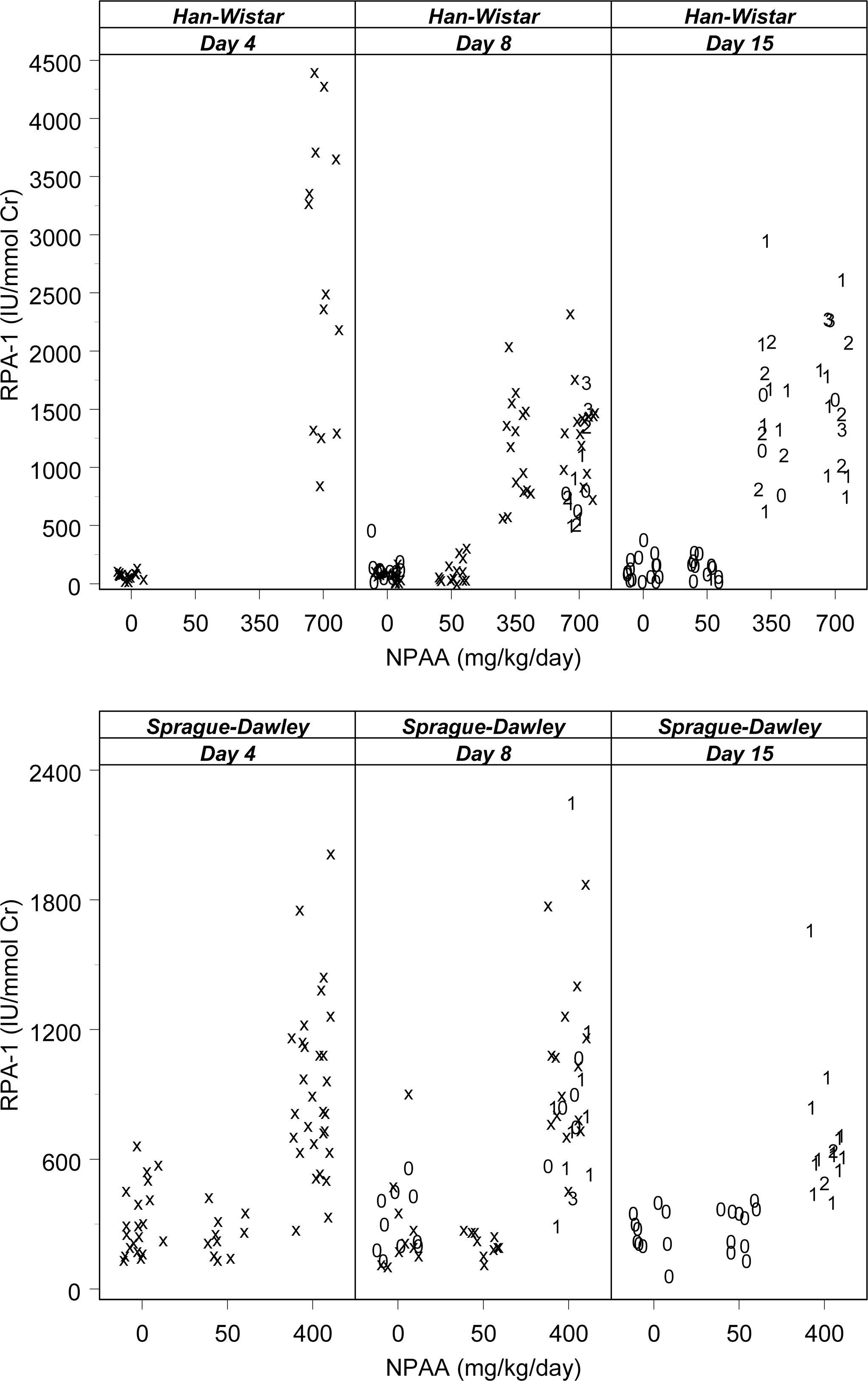
Overview of renal papillary antigen-1 (IU/mmol urine creatinine) versus N-phenylanthranilic acid dose levels (mg/kg/day) by rat strain and study day with individual histopathology scores. Numeric symbols indicate histopathology grades for collecting duct degeneration/necrosis in both papilla and medulla (grade 0 = absent, grade 1 = minimal, grade 2 = mild, grade 3 = moderate, grade 4 = severe). Interim samples without contemporaneous histopathology are indicated by “x.” The high dose of 700 mg/kg/day in Han-Wistar rats was reduced to 350 mg/kg/day on day 5 for the rats euthanized on day 8, and to 500 mg/kg/day on day 5 for the rats euthanized on day 15.
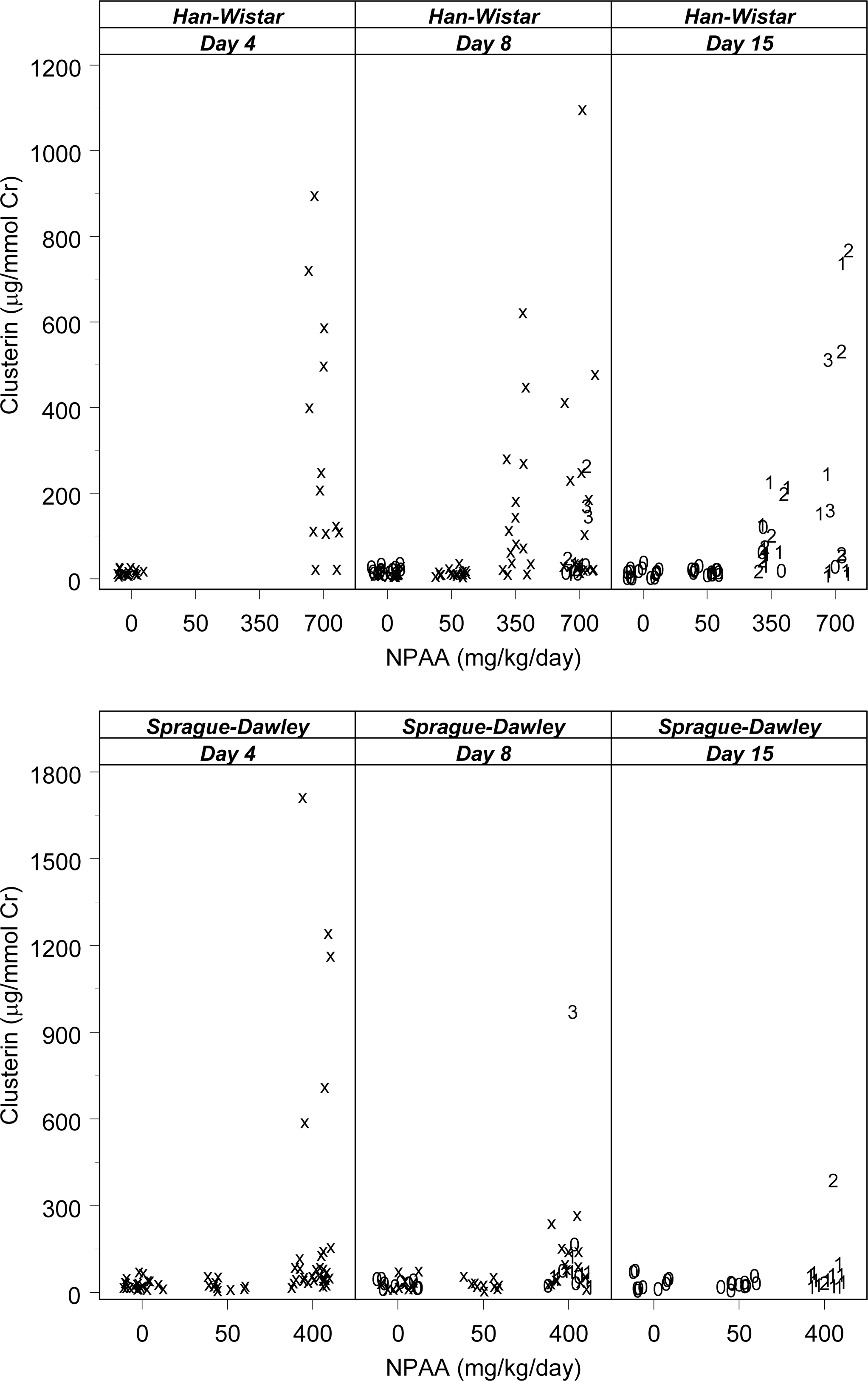
Overview of urinary clusterin (µg/mmol urine creatinine) versus N-phenylanthranilic acid dose levels (mg/kg/day) by rat strain and study day with individual histopathology scores. Numeric symbols indicate histopathology grades for collecting duct degeneration/necrosis in both papilla and medulla (as in Figure 2). Interim samples without contemporaneous histopathology are indicated by “x.” The high dose of 700 mg/kg/day in Han-Wistar rats was reduced to 350 mg/kg/day on day 5 for the rats euthanized on day 8, and to 500 mg/kg/day on day 5 for the rats euthanized on day 15.
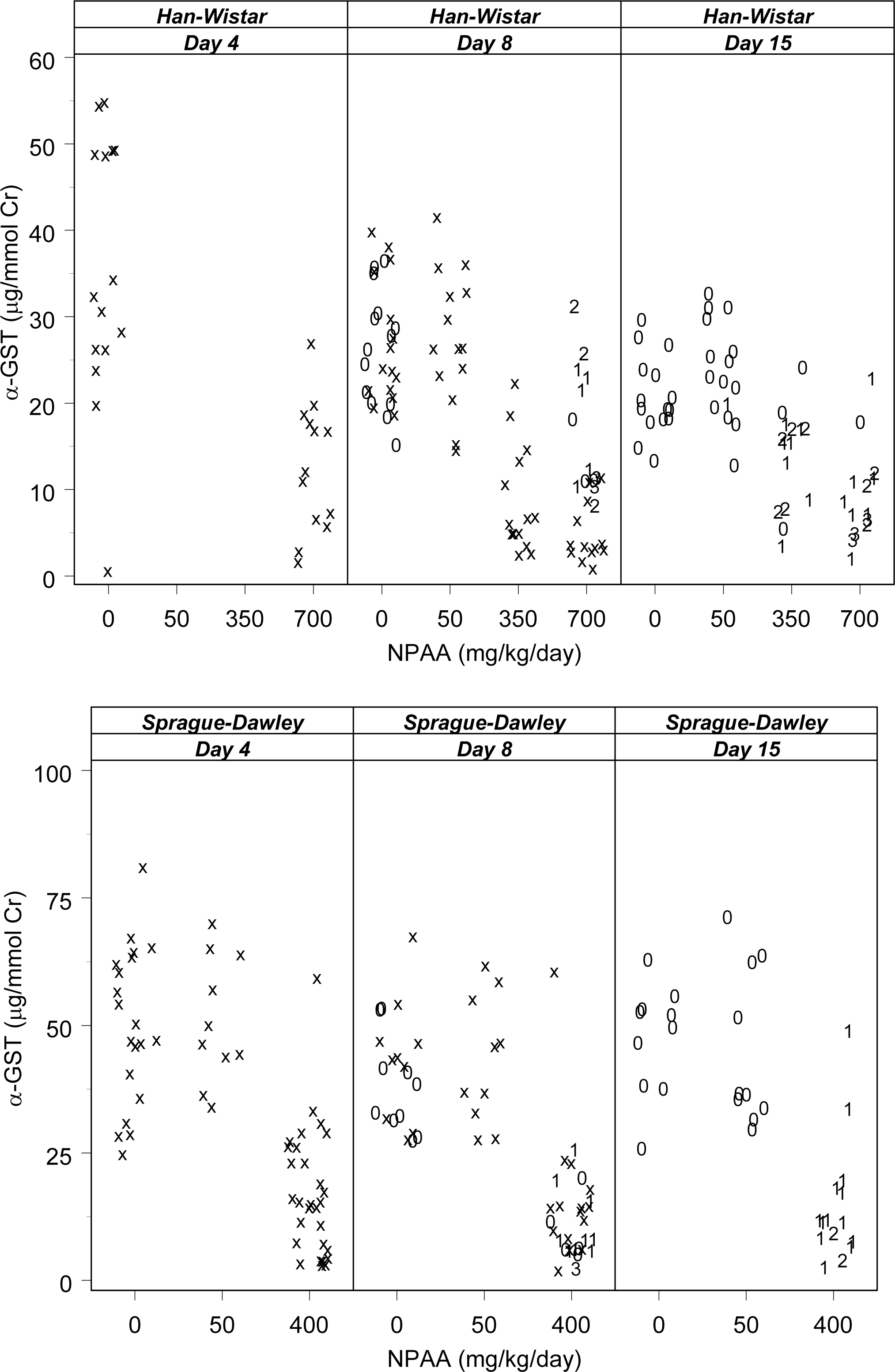
Overview of urinary α-glutathione-S-transferase (µg/mmol urine creatinine) versus N-phenylanthranilic acid dose levels (mg/kg/day) by rat strain and study day with individual histopathology scores. Numeric symbols indicate histopathology grades for collecting duct degeneration/necrosis in both papilla and medulla (as in Figure 2). Interim samples without contemporaneous histopathology are indicated by “x.” The high dose of 700 mg/kg/day in Han-Wistar rats was reduced to 350 mg/kg/day on day 5 for the rats euthanized on day 8, and to 500 mg/kg/day on day 5 for the rats euthanized on day 15. A single outlier value of 3,024.0 µg/mmol urine creatinine on day 4 in one Sprague-Dawley rat treated at 400 mg/kg/day of N-phenylanthranilic acid, which would skew the scale of the vertical axis, has been deleted.
Novel urinary biomarkers of nephrotoxicity in Han-Wistar rats.
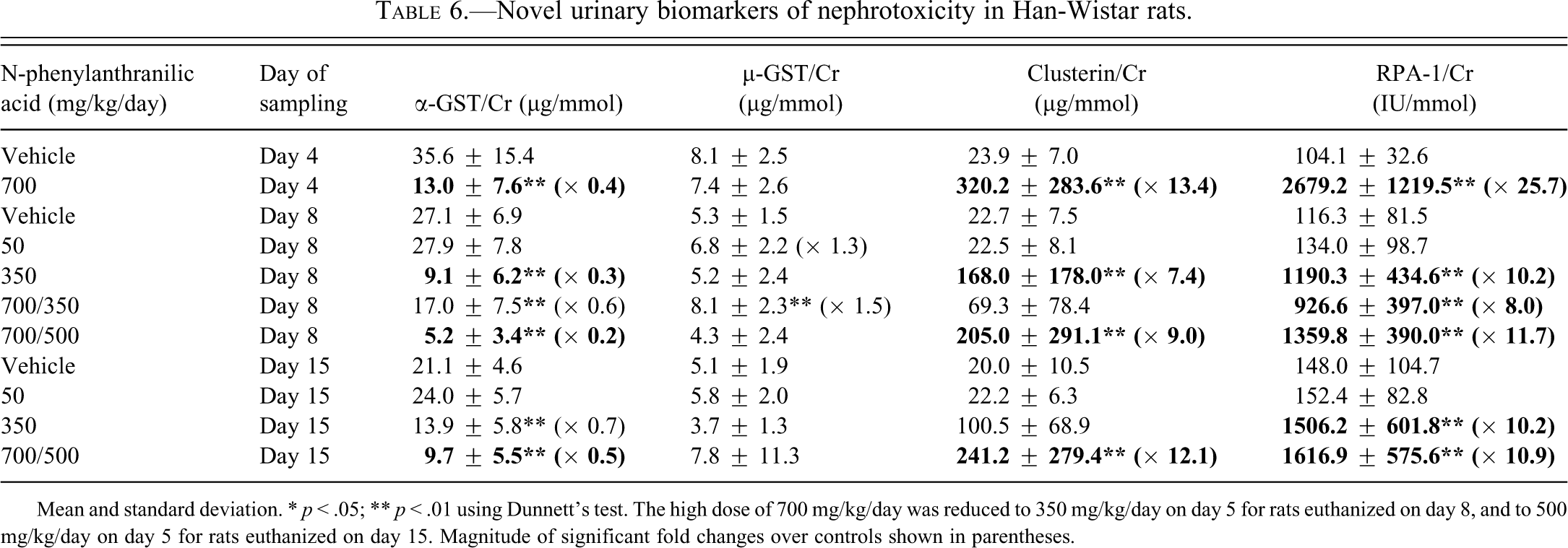
Mean and standard deviation. * p < .05; ** p < .01 using Dunnett’s test. The high dose of 700 mg/kg/day was reduced to 350 mg/kg/day on day 5 for rats euthanized on day 8, and to 500 mg/kg/day on day 5 for rats euthanized on day 15. Magnitude of significant fold changes over controls shown in parentheses.
In Sprague-Dawley rats, mean urine RPA-1 values for rats given 400 mg/kg/day were increased up to 3.1-fold on days 4, 8, and 15 compared to the time-matched controls (Table 7). Of note, five animals presented increases in RPA-1 in the absence of collecting duct degeneration/necrosis in the high dose group on day 8 (Figure 2). Urinary clusterin was increased at 400 mg/kg/day on days 4 and 8 (5.9- and 2.9-fold, respectively). Mean urinary α-GST and µ-GST values were decreased to 70% and 60%, respectively, for rats given 400 mg/kg/day on days 4, 8, and 15 compared to control values.
Novel urinary biomarkers of nephrotoxicity in Sprague-Dawley rats.

Mean and standard deviation. * p < .05; ** p < .01 using Dunnett’s test. Day 4 outlier values of 3,024.0 µg α-GST/mmol urine creatinine and 1,138.2 µg µ-GST/mmol urine creatinine for one Sprague-Dawley rat given 400 mg/kg/day of N-phenylanthranilic acid were excluded from calculation. Magnitude of significant fold changes over controls shown in parentheses.
Discussion
The objective of this study was to evaluate the performance of a panel of urinary biomarkers as indicators of renal injury in comparison with traditional clinical pathology parameters in two rat strains rat commonly used in the pharmaceutical development. Only male rats were used, and responses in females may differ. Nephrotoxic injury was induced by administration of NPAA, a compound known to cause RPN. Urine and blood samples were collected on days 4 (urine only), 8, and 15, and histopathological evaluation of the kidney was done on days 8 and 15. The response of the exploratory urinary biomarkers α-GST, µ-GST, clusterin, and RPA-1 was compared to that of the traditional clinical pathology parameters in blood and urine and related to the observed renal histopathology. There were some minor differences in the protocols between the two participating laboratories, for example, feeding status during urine collection, anesthetic used prior to exsanguination at necropsy, technical platform used for measurement of standard clinical pathology parameters, and processing of kidneys for histopathological examination. However, such differences in practice between laboratories are common in the conduct of animal toxicity studies and provide real world context. As in the previously published study conducted in the context of HESI using Han-Wistar and Sprague-Dawley rats treated with cisplatin (Gautier et al. 2010), these differences in study protocols were not considered to compromise the primary objective of determining whether the novel biomarkers performed better than traditional clinical pathology parameters as indicators of nephrotoxic injury.
Nephrotoxic injury was seen in both strains at all doses of NPAA above 50 mg/kg/day and was characterized by degeneration and necrosis of the collecting duct in the papilla, and severity ranged from minimal to moderate. The nature and incidence of renal injury was very similar between the two strains, although the severity of the collecting duct changes caused by NPAA was much greater in Han-Wistar rats initially given a higher dose. Indeed, degeneration and necrosis of collecting ducts were observed in both the papilla and the medulla in Han-Wistar rats, but only in the papilla in Sprague-Dawley rats. Further, the diagnosis of medullary tubular regeneration/basophilia was unique to Han-Wistar rats and was considered secondary to the severity of injury seen in medullary collecting ducts early in the treatment of this strain with NPAA. These differences are believed to be attributable both to an intrinsic strain difference in sensitivity to NPAA and to the fact that the top dose at the start of treatment in the Han-Wistar rat was not tolerated (700 mg/kg/day vs 400 mg/kg in the Sprague-Dawley rat) and had to be reduced during the course of the study. No toxicokinetic data were collected to assess whether adsorption, distribution, metabolism, and excretion characteristics of NPAA differed between the strains.
The prenecrotic changes observed in the papilla (increased interstitial matrix, hypertrophy of collecting ducts) were consistent with the increase in kidney weight, and the observed pathology primarily affected collecting ducts of the outer medulla in Han-Wistar rats, as confirmed by RPA-1 immunohistochemistry. Injury to the collecting ducts in the papilla and medulla is followed by injury to cortical tubules (collecting ducts and/or distal tubules). Indeed, distal tubular degeneration in Sprague-Dawley rats occurred in association with collecting duct degeneration or necrosis; similarly, cortical tubular dilation was identified in a large proportion of high-dose Han-Wistar rats. In the case of Sprague-Dawley rats, the overall severity of the injury was much less, and injury to the distal tubules preceded cortical collecting duct injury. There was no evidence of proximal tubular injury in either strain, and tubular injury was largely confined to degeneration/necrosis of cortical and medullary collecting ducts with or without regeneration. The localization of injury to distal tubules and not to proximal tubules was confirmed by immunohistochemistry using antibodies directed against calbindin D28 and α-GST, respectively.
Conventional plasma and urinary biomarkers failed to detect renal papillary necrosis or preceding pathologies, except urinary NAG, which was clearly increased in Han-Wistar rats given 700 mg/kg/day on days 4, 8, and 15, and at 350 mg/kg/day on days 8 and 15. This finding is in accordance with literature showing that urinary NAG increases detect RPN following administration of toxins such as 2-bromoethanamine (Stonard et al. 1987). Of note, urinary NAG was slightly but significantly decreased in Sprague-Dawley rats given the high dose of NPAA on days 4, 8, and 15. Although NAG is a lysosomal enzyme that has highest activity in the S3 segment of the proximal tubule (Le Hir et al. 1979) and therefore is not specific for papillary damage, increases in urinary NAG following NPAA administration might be a secondary effect of tubular obstruction and back pressure causing tubular dilatation, which was more prevalent and of greater severity in the Han Wistar rats.
Urinary RPA-1 was increased on days 4, 8, and 15 at the high dose in Han-Wistar rats and Sprague-Dawley rats, and on days 8 and 15 at the mid-dose of 350 mg/kg/day in Han-Wistar rats. This finding confirms results previously reported in the literature in Han-Wistar rats (Hildebrand et al. 1999; Price et al. 2010). The magnitude of increase in urinary RPA-1 after NPAA treatment was greater in Han-Wistar than in Sprague-Dawley rats, consistent with the greater severity of pathology observed in the Han-Wistar rats. Of note, some individual treated animals in both strains had increases in RPA-1 in the absence of collecting duct degeneration/necrosis, suggesting potential prodromal utility for RPA-1 in the rat. However, the current NPAA studies were not designed to address prodromal properties, and further investigation specifically addressing this point is required before any conclusion can be drawn. Concerning the organ/tissue specificity of RPA-1 for renal injury, there is limited information at the present time. However, there is a high likelihood that the presence of RPA-1 in urine will prove specific for renal injury because, to our knowledge, the protein to which RPA-1 antibody is directed is not found in any other tissue except the urothelium of the urinary tract and epididymis (Betton et al. 2007). These tissues were not examined and so could provide another source of biomarker shedding.
Urinary clusterin (a biomarker of general kidney damage) increased in both strains, with more pronounced changes in Han-Wistar rats. As for RPA-1, urinary clusterin was increased early (day 4) at a time preceding the onset of papillary necrosis by NPAA determined in earlier studies. However, as the earliest time point for necropsy was day 8, it is not possible to determine with certainty whether this biomarker response preceded the pathological changes. Clusterin is known to be up-regulated throughout the nephron in response to renal injury. For example, urinary clusterin was increased following collecting duct injury induced by lithium in rats (Dieterle et al. 2010) and in niferacetam-induced RPN in dogs (Tsuchiya et al. 2005). However, urinary clusterin can also be increased when proximal tubules are injured (Dieterle et al. 2010; Gautier et al. 2010), and clusterin has been qualified by the U.S. Food and Drug Administration (FDA) and the Europeam Medicines Agency (EMA) as a biomarker of general renal inury after review of exploratory biomarker data submissions by Critical Path Institute PSTC and HESI (Critical Path Institute 2008 and 2010; HESI 2010).
The levels of α-GST in urine were consistently decreased in the presence of collecting duct degeneration/necrosis caused by NPAA in both rat strains. The significance of this decrease in the absence of an effect on proximal tubules is unknown and may be metabolic or analytical in origin, rather than indicative of pathology. A decrease in the levels of urinary µ-GST (considered a distal tubular injury biomarker) was also observed in parallel with the decreases in α-GST in Sprague-Dawley rats given 400 mg/kg/day of NPAA. However, a decrease in urinary µ-GST is inconsistent with the observation of distal tubular lesions in Sprague-Dawley rats. In Han-Wistar rats, there was no consistent pattern of change in urinary µ-GST either with respect to dose or direction of change. These data preclude any conclusion about the utility of urinary µ-GST as a biomarker of injury to the distal tubule or collecting duct.
In conclusion, urinary RPA-1 and clusterin are sensitive biomarkers that can detect collecting duct injury induced by NPAA in Han-Wistar and Sprague-Dawley rats in the absence of increases in BUN and serum creatinine. Of note, a meta-analysis was performed with data from the NPAA and the other HESI studies conducted with cisplatin and gentamicin. Results showed that RPA-1 had better sensitivity and specificity characteristics than all other traditional clinical pathology parameters tested (BUN, serum creatinine, urinary total protein, and NAG) for the diagnosis of collecting duct injury in the rat (Harpur et al. 2011). These data were used to qualify RPA-1 for voluntary use in detecting acute drug-induced kidney injury, particularly in the collecting ducts in rats, by FDA and EMA (HESI 2010). Further work remains to be done to further explore potential prodromal utility and reversibility of RPA-1 changes. Another important objective will be to translate the use of RPA-1 from preclinical to clinical studies; however, no human equivalent has yet been identified for RPA-1, and preliminary studies have shown that the monoclonal antibody used to recognize rat RPA-1 does not detect a similar antigen in the human kidney. Therefore, alternative biomarkers for human applications need to be evaluated. Immunoassays have recently been developed for detecting collecting duct damage using antibodies directed against aquaporin-2 and 3C7 antigens (Sourial et al. 2010). These assays have the potential advantage over RPA-1 in that they recognize both rat and human antigens; consequently, the same assay could be used both preclinically and clinically.
Footnotes
Acknowledgments
We thank the key supporting staff of AstraZeneca (Dai Davies, Sarah Gould, Sally Price, and Alison Bigley) and GlaxoSmithKline (Rosanna Mirabile, Mark Watkins, Tammy Lambert, and Rosemary McGeary) for their scientific and technical input, and the HESI Nephrotoxicity Biomarkers Working Group under the leadership of Dr. E. Harpur. We also thank Biotrin (now Argutus Medical, Dublin, Ireland) for the provision of immunoassay kits.
The International Life Sciences Institute (ILSI) Health and Environmental Sciences Institute (HESI) Committee on Biomarkers of Nephrotoxicity is a consortium of pharmaceutical companies and government and academic scientists, whose mission is to advance development of biomarkers of target organ toxicity that bridge from the preclinical to the clinical stages of drug development. Participants in the consortium are AstraZeneca Pharmaceuticals, Bayer Schering, Argutus (formerly Biotrin), Bristol-Myers Squibb, GlaxoSmithKline, Pfizer, sanofi-aventis, and the University of Arizona. HESI is an international, nonprofit organization that stimulates and supports scientific research that contributes to the collaborative identification and resolution of health and environmental issues of concern to the public, scientific community, government agencies, and industry.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
