Abstract
The purpose of this study was to compare the toxicity of three marketed corticosteroid receptor agonists (mometasone furoate, budesonide, or flunisolide) to the stomach of female CD-1 mice following oral administration via the diet for up to 52 weeks, with a 16-week recovery period (budesonide and flunisolide). A range of tissues was examined by light microscopy, accompanied by clinical pathology measurements to assess anticipated corticosteroid effects as a surrogate marker of systemic drug exposure. Microscopic changes seen in the stomach with each corticosteroid included pyloric hyalinization. This previously unreported finding was investigated using histochemical and immunohistochemical techniques and was found to consist of hyalinized collagen, in association with increased immunohistochemical signal for transglutaminase-2 and osteopontin. The significance of the osteopontin finding is unclear; however, the ability of transglutaminase-2 to facilitate the formation of degradation resistant protein bonds implies this protein may be involved in the pathogenesis of this change. Furthermore, published evidence that transglutaminase-2 may be induced by a corticosteroid agonist raises the possibility that pyloric stomach hyalinization may be a class effect of corticosteroids via the action of this enzyme.
Introduction
Hyaline change/hyalinization, from the Greek hualinos (glass), is a descriptive term used to denote a glassy, amorphous eosinophilic tissue appearance with hematoxylin and eosin (H&E) staining. This tinctorial change may be produced by a variety of alterations and does not represent a particular pattern of accumulation (Kumar 2005). Hyalinization is therefore seen in association with various conditions, including amyloidosis (Abbas 2005), hypertension (Hall and Gomez-Sanchez 1986), and scar formation (Ungar, Laufer, and Diengott 1956).
Hyalinization within the pyloric stomach of CD-1 mice was noted in a 2-year inhalation carcinogenicity study of a novel corticosteroid agonist undergoing development within GlaxoSmithKline (GSK). Although the finding was noted in a small number of control animals, there was a dose-related increase in incidence and severity in the high dose in both sexes and in the intermediate-dose females. Similar effects were not seen in the corresponding rat study. To our knowledge, this finding has not been previously reported for this compound class, and an investigative study was therefore undertaken to assess whether pyloric hyalinization was associated with treatment with other marketed corticosteroid agonists and to investigate the nature of this change. The investigative study, conducted in female CD-1 mice, consisted of oral (dietary) dosing of the corticosteroid agonists mometasone furoate, budesonide, and flunisolide to separate groups of animals, with treatment periods of 13, 27, 36 (budesonide and flunisolide), or 52 weeks. Following treatment for 36 weeks (budesonide and flunisolide only), a 16-week recovery period was included. In addition to light microscopic examination of a range of potential target organs and clinical pathology measurements, histochemistry, immunohistochemistry (IHC), and electron microscopy (EM) were used to characterize changes in the pyloric stomach.
Materials and Methods
Animals and Study Design
Female CD-1 mice (Crl:CD-1[ICR]) were obtained from Charles River Laboratories Ltd. (Kent, UK) and were housed in groups of four animals/cage. On the first day of dosing (day 1), all animals were 7 to 8 weeks old. Animal assignment, target, and estimated achieved doses and dosing duration are shown in Table 1 . Mometasone furoate, budesonide, or flunisolide was administered in diet (ground Harlan Teklad 2014 Global Rodent Maintenance Diet) at concentrations predicted to achieve the target doses shown in Table 1. Diet concentrations were adjusted weekly/fortnightly as appropriate throughout the study. Controls (throughout the study) and treated animals (during the pretreatment period) were offered diet alone. Drinking water (filtered domestic water supplied by Three Valleys Water plc and periodically analyzed by Severn Trent Laboratories, Bridgend) was available ad libitum. All treated and control groups consisted of twelve females. Doses were selected on the basis of findings of studies of the oral tolerance of the test materials, conducted outside GSK using different strains of mice and various oral administration methods (Nasonex™ Nasal Spray [Mometasone furoate monohydrate] NDA 20-762 Review and Evaluation of Pharmacology and Toxicology Data, Original Review #1 1997; Rhinocort Respules™ [Budesonide Nebulizing Suspension] NDA 20-929 Review and Evaluation of Pharmacology/Toxicology Data Review #2 1998; Bronalide™ Inhaler System [Flunisolide] NDA 18-340 Summary Basis of Approval 1984). Females only were examined as the pyloric hyalinization (though present in both sexes) was previously noted to be more apparent in females.
Group assignment, estimated achieved doses, and treatment duration.
a36 weeks treatment and 16 weeks off-dose.
All in vivo studies were ethically reviewed within GSK and carried out in accordance with the Animal (Scientific Procedures) Act 1986.
Observations
Body weights and food consumption were recorded daily in the pretreatment period and daily/twice weekly (as appropriate) throughout the study and on the day of necropsy. Clinical observations were conducted daily and a full clinical examination was performed once during the pretreatment period and in treatment weeks 12, 26, and 52.
Termination
At the end of the treatment period, all animals were killed by exsanguination after inhalation of a rising concentration of carbon dioxide.
Histology
Tissues collected included stomach, adrenals, liver and gall bladder, mandibular and mesenteric lymph nodes, skin, spleen, thymus or thymic area, and any macroscopic abnormalities. All tissues were fixed with 10% neutral buffered formalin, processed to 3- to 7- µm sections and stained with H&E. Additional stains used on selected stomach sections included chloranilic acid and von Kossa for calcium/calcium salts (all groups dosed for 13 weeks with concurrent controls), Congo Red for amyloid, Masson’s trichrome for collagen, and Alcian blue (pH 2.5) and periodic acid-Schiff (PAS) for mucosubstances (all groups dosed for 27 weeks with concurrent controls). Sections were examined by light microscopy (data was peer-reviewed).
Immunohistochemistry
IHC was carried out on formalin fixed and paraffin embedded sections. A standard indirect peroxidase method (avidin/biotin complex/horseradish peroxidase-diaminobenzidine) was used. Primary antibodies included transglutaminase-2/tissue transglutaminase (Genetex), laminin 1&2 and smooth muscle actin (Abcam), tissue nonspecific alkaline phosphatase (TNAP, Santa Cruz Biotech, Santa Cruz, CA), intestinal alkaline phosphatase and fibronectin (ABR), and osteopontin (R&D systems).
Antigen retrieval techniques were necessary for all antibodies except osteopontin and included microwave heating for 15 minutes in Vector unmasking solution (Vector Laboratories, Burlingame, CA) (transglutaminase-2), proteinase K treatment for 10 minutes at room temperature (fibronectin and laminin 1&2), and 10% formic acid treatment for 10 minutes at room temperature (TNAP and intestinal alkaline phosphatase).
Isotype control antibodies appropriate to each primary antibody were included in all IHC runs. Isotype controls included rabbit immunoglobulin fraction-normal and rabbit serum-normal (Dako Cytomation) and goat F(ab) fragments (Serotec).
Stomach sections from control animals were used as positive tissue controls for the majority of antibodies used, including transglutaminase-2 (endothelial cells; Verderio, Johnson, and Griffin 2004). A region of mouse soft tissue mineralization was used as a positive control for osteopontin, and mouse lung tissue was used for TNAP (type II pneumonocytes; Edelson, Shannon, and Mason 1988).
IHC was performed on all groups dosed for 27 weeks and the concurrent controls. All IHC markers were scored on a 5-point scale encompassing the area stained and stain intensity, where 1 = minimal positive staining, 5 = maximum positive staining, and 2–4 = arbitrary ordinal classes between 1 and 5.
Electron Microscopy
Animals selected for EM included two 13-week controls, four treated animals from the week 13 cohort (one animal given mometasone furoate, one animal given budesonide, and two animals given flunisolide, all at 500 µg/kg/day) and four animals from the week 27 cohort (one animal given budesonide, one animal given mometasone furoate, and two animals given flunisolide, all at 200 µg/kg/day). Samples of residual pyloric stomach fixed in phosphate buffered 4% formaldehyde/1% glutaraldehyde were processed for EM. This involved postfixation in 1% Millonig’s buffered osmium tetroxide and processing into Agar 100 resin. Toluidine blue–stained survey sections (1 µm thickness) were prepared and examined by light microscopy to locate the areas of interest. Ultrathin sections (60–90 nm) were then prepared, stained with uranyl acetate and lead citrate, and examined using a Hitachi H7500 transmission electron microscope, operated at 60 kV.
Hematology and Clinical Chemistry
At termination, 0.4 mL blood was collected from six animals/group (or three animals/group at week 36 and recovery) into tubes containing EDTA potassium. Blood samples were analyzed for total blood counts using an Advia 120 automated hematology analyzer (Bayer Diagnostics, Newbury, UK).
Clinical Chemistry
At termination, 0.6 mL blood was collected from six animals/group (or three animals/group at week 36 and recovery) into tubes containing lithium heparin and plasma collected following centrifugation. Plasma samples were analyzed for a range of parameters that included total protein and lipids using an Advia 1650 automated clinical chemistry analyzer (Bayer Diagnostics) and, where sample volume allowed, corticosterone concentration using radioimmunoassay (Diagnostic Product Corporation, CA, USA).
Adrenal Weights
The adrenals (paired and post fixation) were weighed from each animal.
Data Handling and Statistical Analysis
Data for body weights and organ weights are expressed in terms of times concurrent control group mean values. Statistical analysis of hematology/clinical chemistry data was performed on data sets of nominally n = 6 animals (occasionally conducted on smaller group sizes when intercurrent deaths occurred, or in the recovery groups) using a decision tree within Provantis Tables and Statistics Software (Instem Life Sciences Ltd, Staffordshire, UK).
Results
Histology
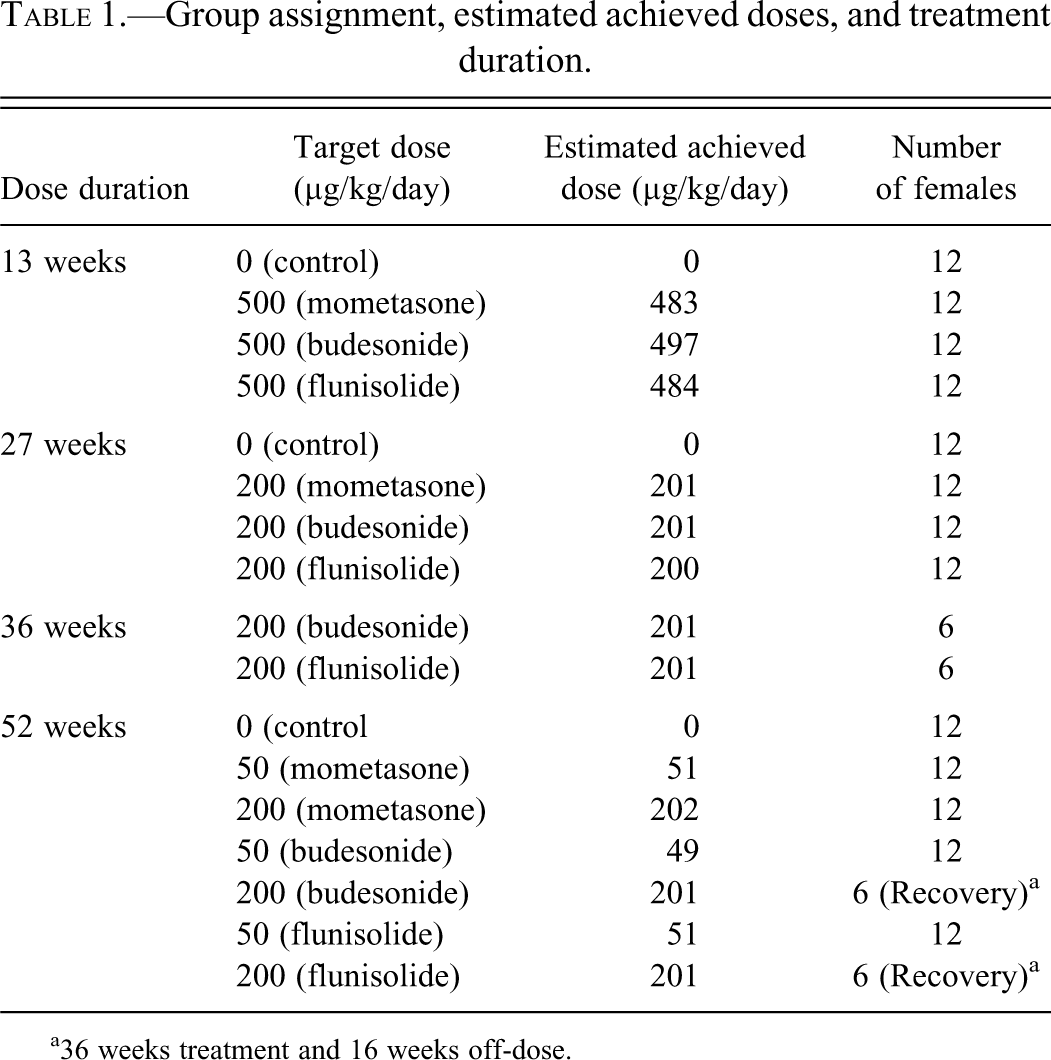
Hyalinization of the pyloric stomach (shown in Figure 1 ) was noted in the majority of treated animals, with each of the three corticosteroid agonists investigated, at all time points, and at all doses, with the exception of the low dose (50 µg/kg/day) group given flunisolide for 52 weeks in which pyloric stomach hyalinization was recorded in approximately half the animals. Hyalinization expanded the lamina propria of the pyloric region of the stomach, was restricted exclusively to this area, and ranged in severity from minimal to moderate based on subjective scoring of the extent of the finding within the section. In addition, hyalinization was accompanied by a range of findings that were common to each of the corticosteroid agonists. These findings included the presence of multinucleate giant cells in the pyloric lamina propria, hyperplasia of the pyloric region, prominent vasculature within the pyloric lamina propria, increased vacuolation of the pyloric epithelium, pyloric crypt dilation, focal edema within the lamina propria, inflammatory cell infiltration, and focal necrosis within the pyloric region. Increased numbers of spindle cells with Anitschkow-type nuclei were also noted in the affected areas. Anitschkow-type nuclei characteristically have condensation of nuclear chromatin into a central bar running the long axis of the nucleus, and irregular indentations of the nuclear membrane. This phenotype, originally identified in the heart, is associated with inflammatory change (Zak 1947). The greatest incidence and severity of these changes were noted in animals given corticosteroid at target doses of 500 µg/kg/day for 13 weeks, and they were much reduced at lower doses with longer duration of dosing. As such, only giant cells, prominent vasculature, and minimal dilation of the pyloric crypts were noted in a minority of animals at the 52-week time point.
(a) Moderate hyalinization (indicated by an arrow) within the pyloric stomach of a female CD-1 mouse given flunisolide at a target dose of 200 µg/kg/day for 52 weeks (H&E ×10). (b) Congo red stain demonstrating pale birefringence consistent with collagen bundles but negative for amyloid (Congo Red and polarized light ×10). (c) Congo Red positive control—Amyloid plaques in mouse cerebrum showing typical “apple-green” birefringence under polarized light.
Recovery animals consisted of groups (n = 6) given either budesonide or flunisolide for 36 weeks followed by a 16-week recovery period. In these animals, pyloric stomach hyalinization without any accompanying findings was noted in one animal from the group given budesonide and two animals from the group given flunisolide. Recovery was not assessed for mometasone. The H&E appearance of the hyalinized areas was the same at all time points, and in recovery animals, and significant change in incidence and severity was noted only in recovery groups, where reduced incidence and minimal severity indicated partial reversal of the finding.
Test-article–related microscopic observations were also recorded in the adrenals, mandibular and mesenteric lymph nodes, skin, spleen, and thymus (week 13); the stomach, adrenals, and spleen (week 27), stomach, adrenals, mandibular and mesenteric lymph nodes, and spleen (week 36); and stomach, adrenal, mesenteric lymph nodes, and skin (week 52). These findings included lymphoid depletion (mandibular and mesenteric lymph nodes, spleen, and thymus), atrophy of the zona fasciculata (adrenal glands), and within the skin, a higher proportion of hair follicles in telogen or catagen stages of the hair cycle (various time points). These are expected changes for corticosteroid receptor agonists, and were not apparent in animals at the end of the recovery period.
Immunohistochemistry and Histochemistry
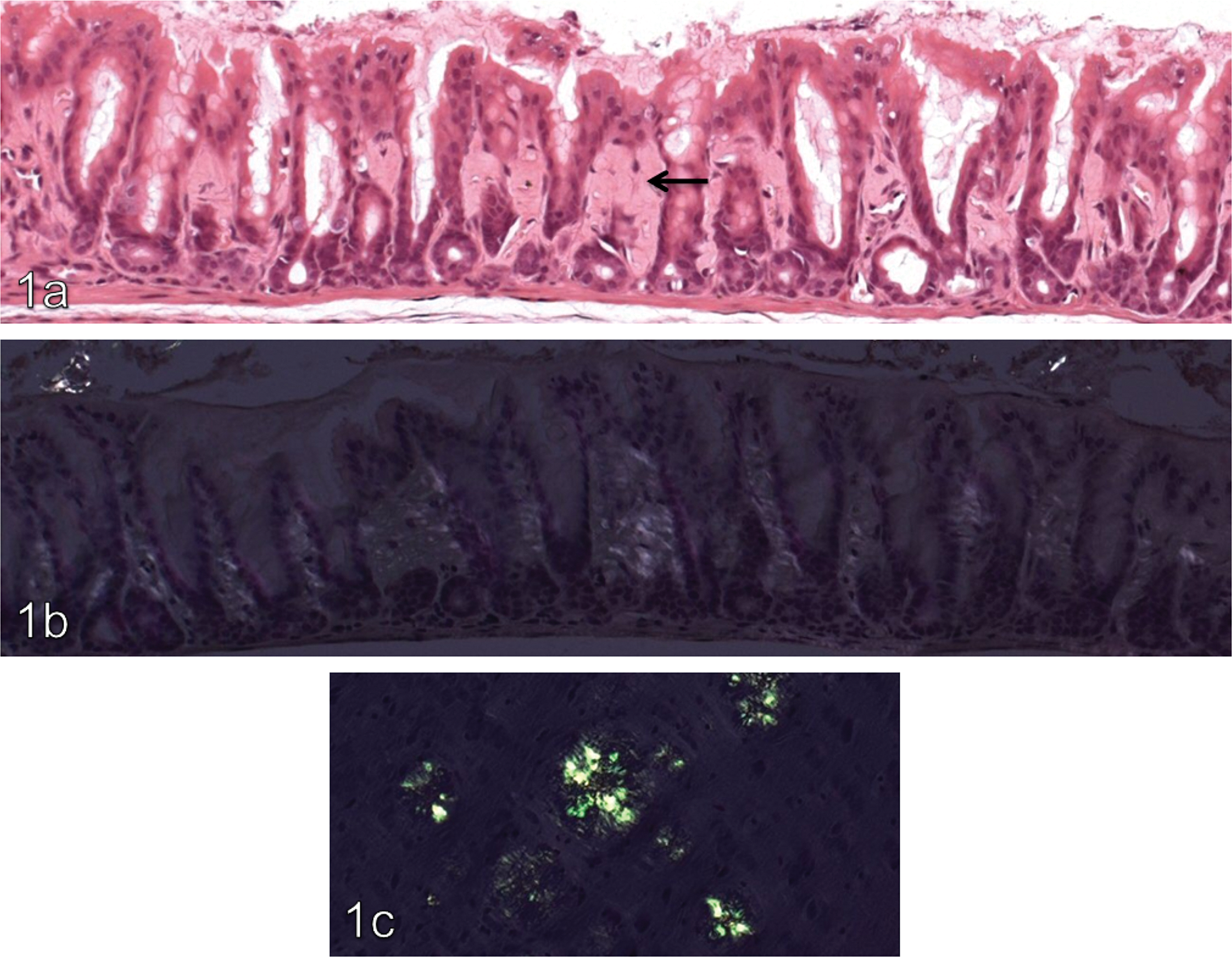
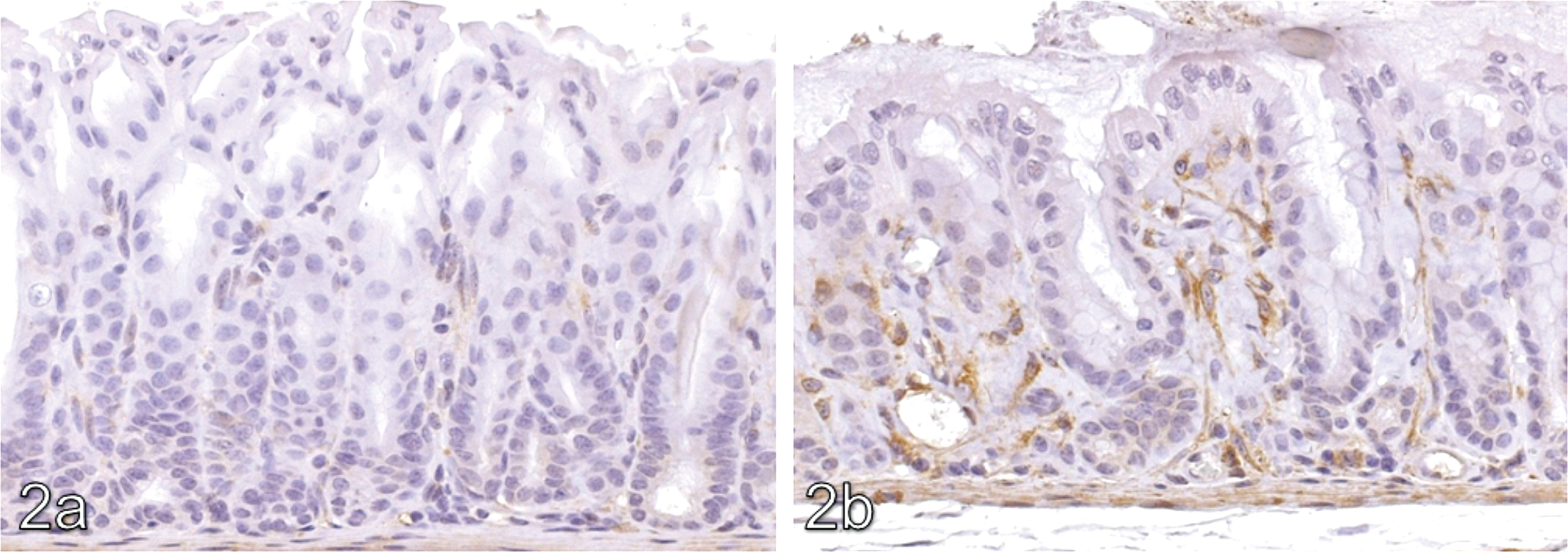
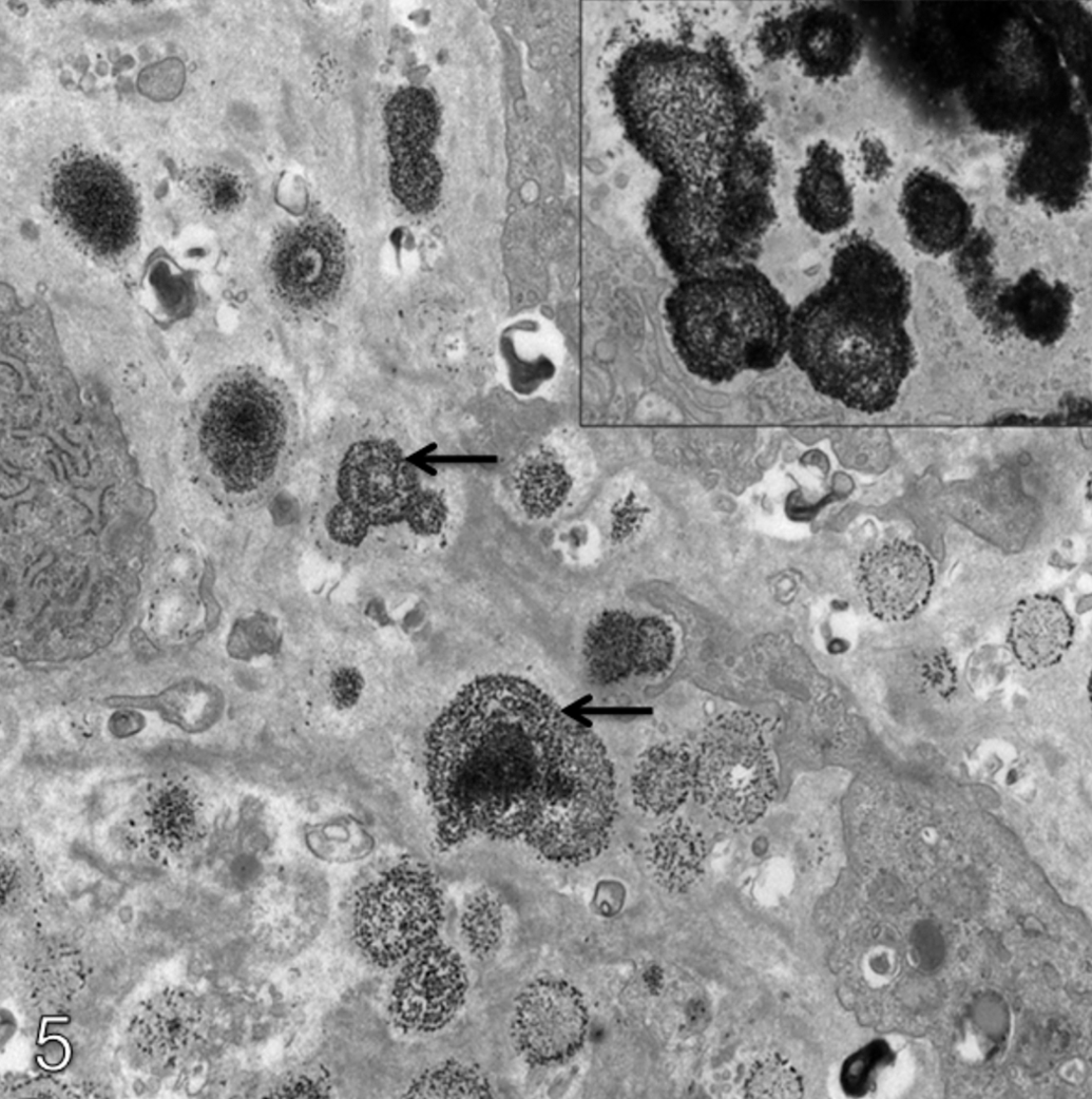
The lamina propria of the pyloric stomach in treated animals, including areas of hyalinization, showed increased immunohistochemical positivity against transglutaminase-2 compared with concurrent controls. Typical results for transglutaminase-2 are shown in Figure 2 . In addition, there was a focal increase in osteopontin signal (illustrated in Figure 3 ) in non-hyalinized regions of the lamina propria of treated animals compared with the same anatomical region in controls. In the control group focal staining with osteopontin within the lamina propria was noted at a grade 1 level only, whilst in treated animals the intensity of this focal staining was increased to grade 3 to 4 (mometasone), grade 2 to 4 (budesonide), and grade 1 to 3 (flunisolide). TNAP signal was considered equivocal because of a broad signal range in controls (graded up to 3). Fibronectin signal was not present in areas of hyalinization, but was increased in the unhyalinized areas of the lamina propria of treated animals compared with controls.
Transglutaminase IHC ×20. (a) Minimal positivity in the pyloric lamina propria of a 27-week control animal. (b) Moderate transglutaminase-2 positivity in the pyloric lamina propria of a CD-1 mouse given a target dose of 200 µg/kg/day budesonide for 27 weeks.
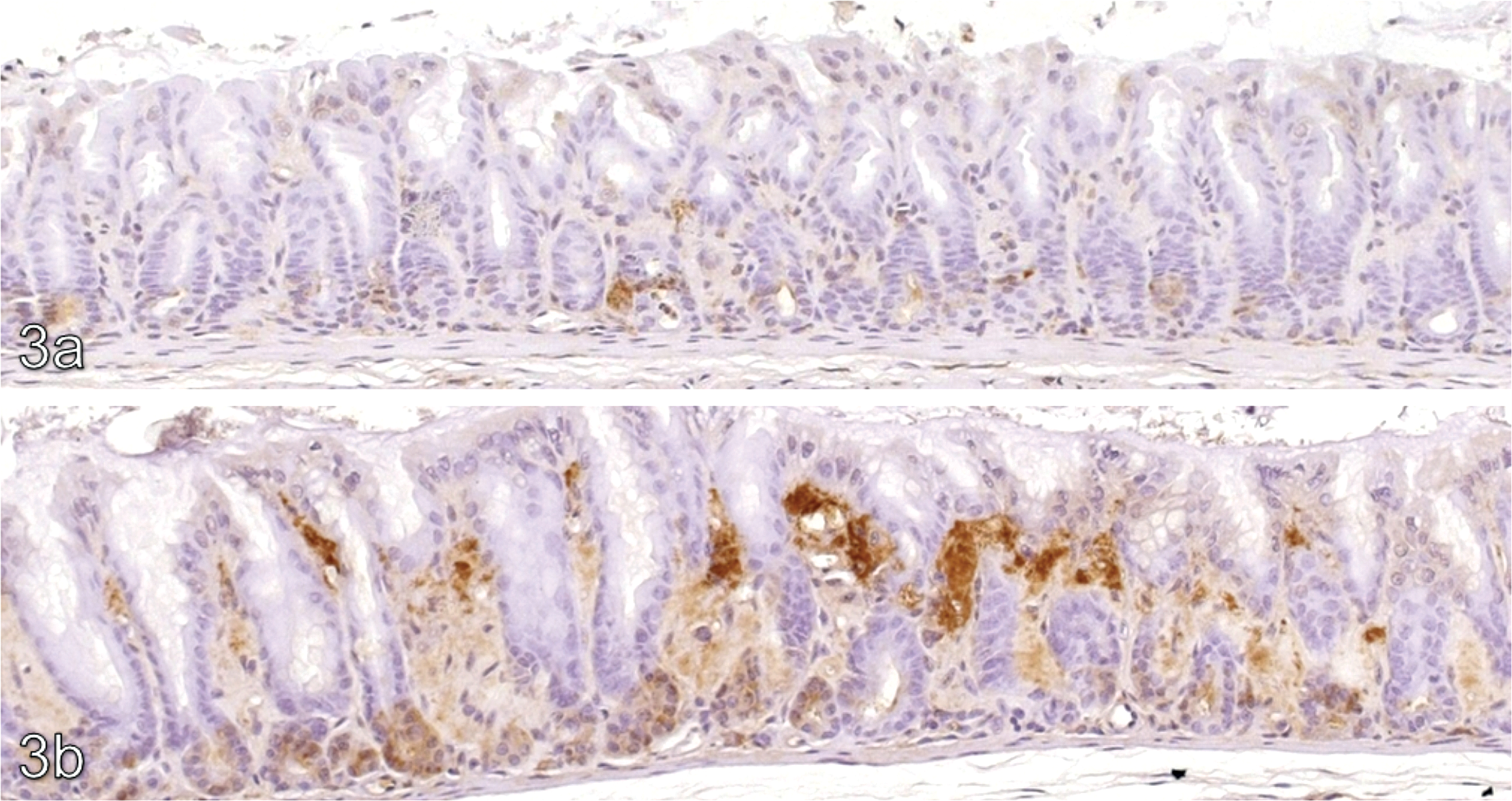
Osteopontin IHC x10. (a) Minimal positivity in a 27-week control. (b) Marked osteopontin positivity within the pyloric lamina propria of a female CD-1 mouse given budesonide at a target dose of 200 µg/kg/day for 27 weeks.
Laminin 1&2 was positive within the lamina propria (graded 2), but grade 1 signal only was noted in areas of hyalinization, whilst intestinal alkaline phosphatase signal was uniform in both lamina propria and areas of hyalinization (grade 2). Average IHC scores for each of these antibodies are shown in Table 2 .
Average IHC scores noted in pyloric lamina propria and areas of hyalinization in controls and animals given 200 µg/kg/day mometasone, budesonide, or flunisolide for 27 weeks.
Scores are on a 5-point scale, with 1 = minimum positive staining, 5 = maximum positive staining, and 2–4 ordinal classes between 1 and 5. A dash (—) indicates no areas of hyalinization present in the group. LP, lamina propria; HY, areas of hyalinization.
aFocal staining in the lamina propria.
Marked (grade 4) smooth muscle actin positivity was noted within the Anitschkow-type cells seen in areas of hyalinization.
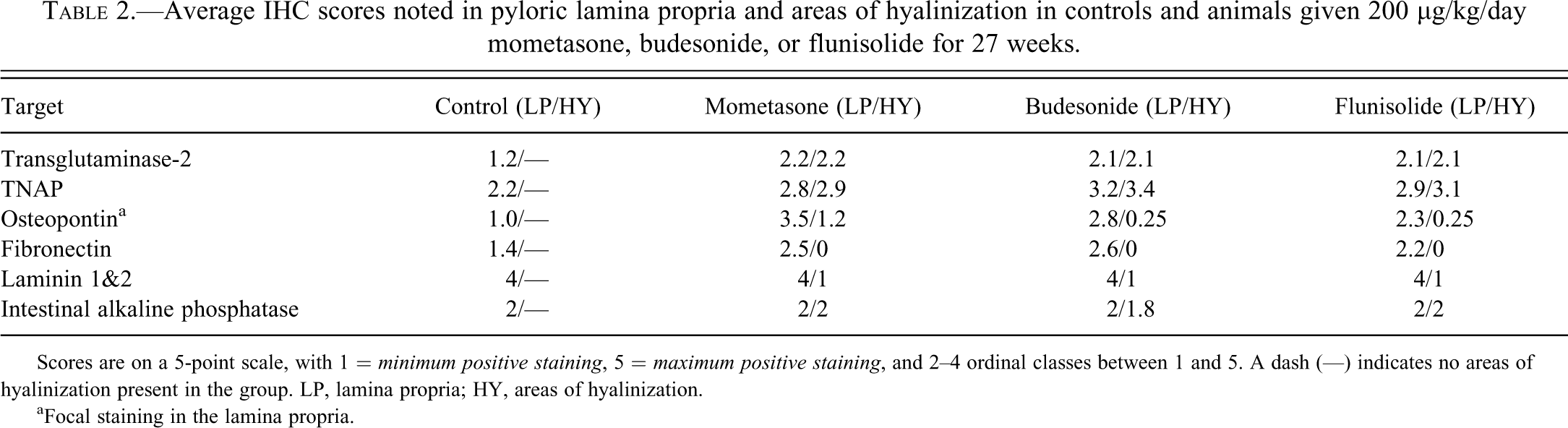
Histochemical staining of the pyloric lamina propria was positive with Masson’s trichrome (indicating collagenous connective tissue in the areas of hyalinization, as shown in Figure 4 ). PAS and alcian blue (pH 2.5) staining of mucous producing cells of the pyloric mucosa was positive in both control and treated animals, and there was no difference in staining pattern or intensity between control and treated animals. Congo Red histochemistry was negative in all sections, and is illustrated in Figure 1.
Masson’s trichrome ×10 (a) 27-week control. (b) Increased collagen present within the lamina propria of the pyloric stomach of a female CD-1 mouse given flunisolide at a target dose of 200 µg/kg/day for 27 weeks.
Electron Microscopy
Administration of mometasone furoate, budesonide, or flunisolide at 500 µg/kg/day for 13 weeks or at 200 µg/kg/day for 27 weeks resulted in ultrastructural changes in the pyloric region of the stomach, which were usually more pronounced in the high dose animals. The 36-week and 52-week groups were not assessed.
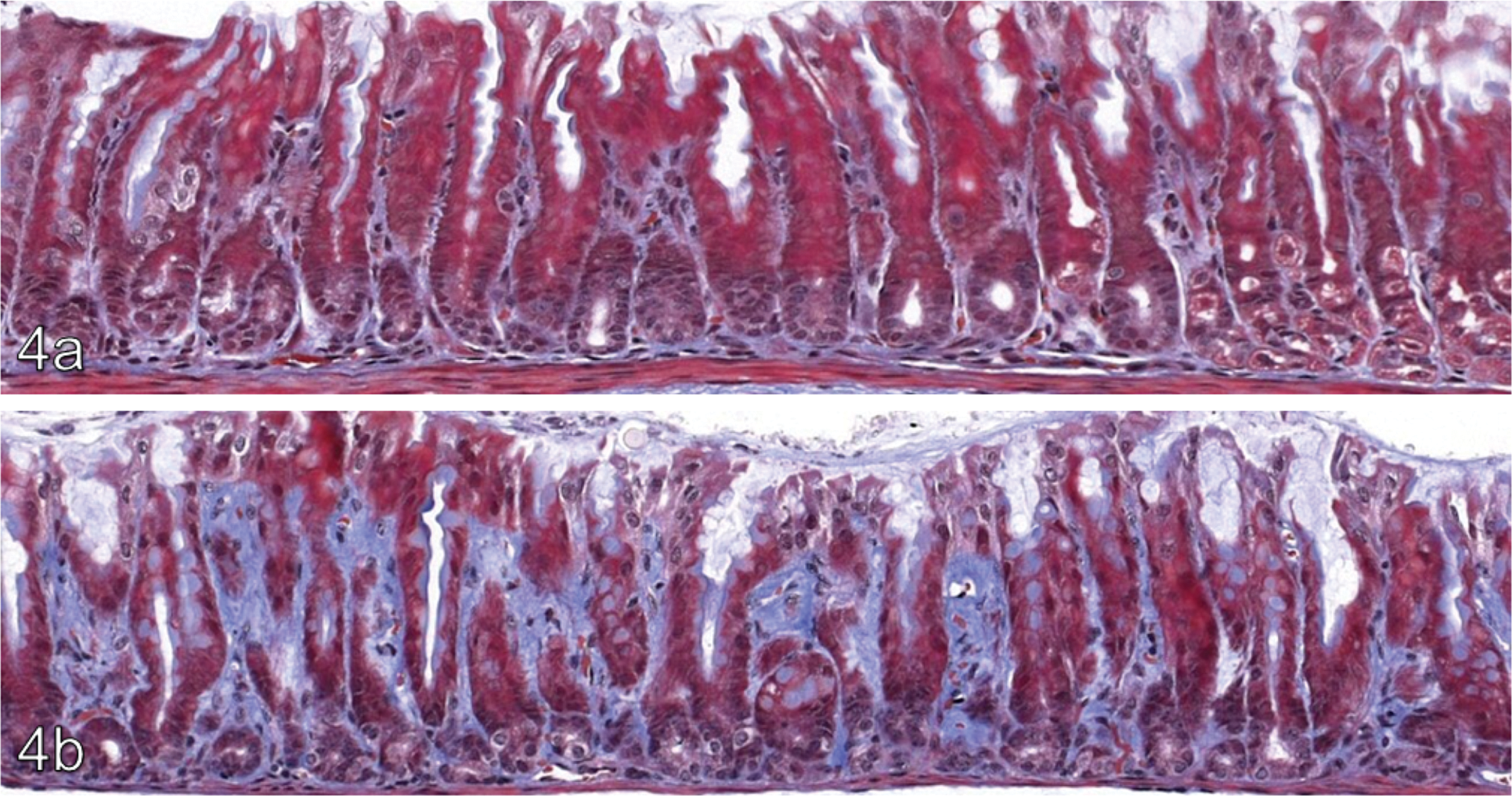
The lamina propria of the pyloric mucosa was thickened due to an increase in extracellular matrix (ECM). Focally, the ultrastructural appearance of the ECM was altered, containing groups of small rounded electron lucent areas and electron dense granular material. A proportion of these areas contained small amounts of the electron dense granular material or electron dense lamellar structures. The electron dense granular material, which was occasionally arranged into laminated target-shaped spherical bodies, is shown in Figure 5 and had an ultrastructural appearance consistent with mineralization.
Electron dense granular material (arrows) consistent with mineralization, arranged in spherical bodies in a female CD-1 mouse given mometasone at a target dose of 500 μg/kg/day for 13 weeks. Inset shows a cluster of electron dense bodies seen in the same animal (main and inset x1,500).
Focally, multinucleate cells were present within the lamina propria. These cells contained a few lysosomal bodies and on rare occasions electron dense material considered to be mineral. An increase in the number and size of myofibroblasts within the lamina propria was also noted (IHC staining shown in Figure 6 ), as was increased dilation of the capillaries within the lamina propria. The surface epithelial cells of the pyloric mucosa contained increased amounts of lipid, particularly in the animals treated with mometasone furoate or flunisolide. The lipid was seen free in the cytoplasm and also in the matrix of a few mitochondria. On occasion mucous cells containing intramitochondrial lipid were also seen. Dilation of the intercellular spaces between the surface epithelial cells (particularly in animals given mometasone furoate or flunisolide) was also noted.
Smooth muscle actin IHC x10. Myofibroblasts in the lamina propria of the pyloric stomach, identified by smooth muscle actin IHC. Compared with a 27-week control animal (a), myofibroblasts appear larger and more numerous in treated animals, as illustrated by a section from a CD-1 mouse given budesonide at a target dose of 200 µg/kg/day for 27 weeks (b).
In-Life Findings and Intercurrent Deaths
Food Consumption
Inclusion of the steroids did not affect the palatability of the diet, and food consumption for all treated groups was comparable with control throughout the study.
Body Weight
Group mean body weight loss and/or reduced body weight gain was noted in animals given 200 µg/kg/day (budesonide or flunisolide) or 500 µg/kg/day (all compounds), generally for the first month of the study. Thereafter, weight gain was generally comparable with that noted in control animals. Body weight loss was greatest in animals given 500 µg/kg/day and several animals given either mometasone or flunisolide were killed within the first month of the study due to progressive weight loss (20–30%) and where possible, were replaced. There was no obvious effect in animals given 50 µg/kg/day.
Clinical Observations
Hair loss (neck and/or thorax) was apparent in animals receiving mometasone, budesonide, or flunisolide (200 µg/kg/day) from week 22. This finding was partially/fully resolved within 2 weeks from cessation of treatment (budesonide and flunisolide).
Adrenal Weights
Compared with controls, each steroid was associated with a reduction in absolute adrenal weight following treatment at 500 µg/kg/day for 13 weeks (–20 to –50%), and 200 µg/kg/day for 27 weeks (–20 to –50%). Full (budesonide) or partial (flunisolide) recovery was apparent following the off-dose period. Adrenal weights were not affected in animals given 50 µg/kg/day.
Clinical Pathology—Hematology and Clinical Chemistry
Clinical pathology data (summarized in Table 3 ) were consistent with systemic corticosteroid exposure, particularly in groups given the highest doses. Corticosterone data at weeks 36, 52, and in recovery animals were not reported because of analytical difficulties.
Summary of group mean hematology/clinical chemistry data.
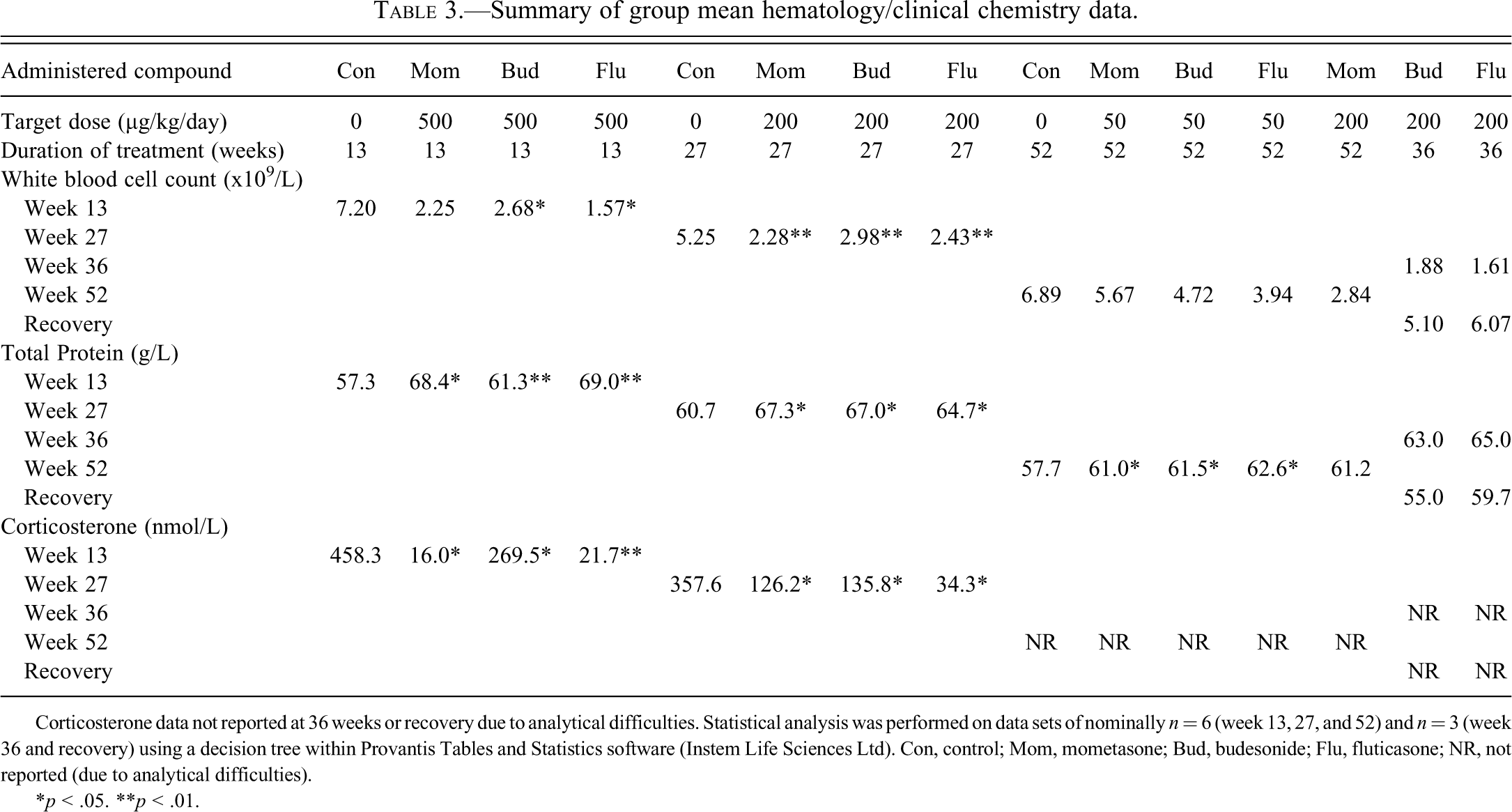
Corticosterone data not reported at 36 weeks or recovery due to analytical difficulties. Statistical analysis was performed on data sets of nominally n = 6 (week 13, 27, and 52) and n = 3 (week 36 and recovery) using a decision tree within Provantis Tables and Statistics software (Instem Life Sciences Ltd). Con, control; Mom, mometasone; Bud, budesonide; Flu, fluticasone; NR, not reported (due to analytical difficulties).
*p < .05.
**p < .01.
Discussion
The purpose of this study was to compare the toxicity of three marketed corticosteroid receptor agonists (mometasone furoate, budesonide, and flunisolide) to selected potential target tissues in female CD-1 mice following oral administration via the diet for up to 12 months. Findings consistent with corticosteroid dosing were noted in a number of organs and by various clinical pathology parameters. In addition, a novel finding (hyalinization) was noted within the pyloric stomach.
Hyalinization within the lamina propria of the pyloric stomach was noted at all time points, with all compounds and all doses examined at each time point. The term hyaline is descriptive, denoting a homogenous, glassy, pink appearance with H&E staining. Multiple processes may produce hyalinization within the extracellular domain, resulting from a number of distinct mechanisms. A well-characterized example is the hyalinization of the walls of arterioles caused by extravasation and deposition of plasma/basement membrane protein as a consequence of hypertension (Kumar 2005). In addition, deposition of the pathogenic proteinaceous substance amyloid will result in a hyaline appearance in routine tissue sections (Abbas 2005). Hyalinization has not been reported within the pyloric stomach in association with corticosteroid dosing, and as such a range of histochemical, immunohistochemical, and electron microscopic investigations were performed in an attempt to further characterize the nature of this change.
Amyloidosis was considered a possible cause of pyloric hyalinization given the identical histological appearance on H&E stained sections, the fact that amyloidosis is a common finding in aged CD-1 mice (Engelhardt, Gries, and Long 1993, HogenEsch, Gruys, and Higuchi 1996), and the fact that long-term immunosuppressive therapy has been associated with an increased incidence of systemic (AA type) amyloidosis (HogenEsch et al. 1993; Kanoh, Yogo, and Ohnaka 1990; Zilko and Dawkins 1975). Hyalinization was not noted in any other organs in this study including the adrenal, which is frequently involved in systemic amyloidosis (Engelhardt, Gries, and Long 1993; Frith and Chandra 1991). A localized gastrointestinal form of amyloidosis that can involve the stomach (AApoAII type) has been described in aged C57BL/Ka mice; however, the incidence of this particular form of amyloidosis was decreased by immunosuppressive therapy that included corticosteroid (HogenEsch et al. 1993). Histochemical staining of stomach sections from all animals from the 27-week cohort was carried out using a Congo Red technique for amyloid (Elghetany, Saleem, and Barr 1989), and in addition EM examination of sections from animals from the 13-week and 27-week cohorts for each corticosteroid dosed group was performed. Sections were negative by Congo red technique, and the typical ultrastructure of amyloid (approximately 7–10 nm diameter, rigid, nonbranching fibrils of indeterminate length) was not revealed by EM examination, indicating amyloidosis is not responsible for the pyloric hyalinization noted in this study (Doi et al. 2007; HogenEsch, Gruys, and Higuchi 1996). By EM, areas of hyalinization instead appeared consistent with increased ECM and contained rounded electron lucent areas within which were found lamellar or granular electron dense material (the latter suggestive of mineralization), and separate areas of granular electron dense material also considered likely to represent mineralization.
Areas of hyalinization were then assessed for the presence of normal ECM/basement membrane components including laminin, fibronectin, and collagen. Both laminin and fibronectin results were unremarkable in areas of hyalinization, though interestingly fibronectin signal was increased in the surrounding lamina propria of all corticosteroid treated groups compared with concurrent controls. This positive effect of corticosteroid on fibroblast fibronectin production has been reported previously in the skin of the hairless mouse (Schwartz et al. 1994). Masson’s trichrome stained the lamina propria uniformly blue in treated animals, indicating the presence of significant amounts of collagen within areas of hyalinization and the surrounding lamina propria. Collagen may appear hyalinized in scar tissue, due to decreased ECM degradation. One mechanism by which this can occur is by increased activity of transglutaminase-2/tissue transglutaminase, a multifunctional enzyme that has a role to play in protein transamidation and structural adhesion, and displays GTPase activity (Verderio, Johnson, and Griffin [2004] and references therein). Through these mechanisms transglutaminase-2 is involved in organization of the ECM, and in the skin has been shown to induce irreversible collagen cross-links that are resistant to enzymatic degradation (Collighan, Cortez, and Griffin 2002; Lorand and Graham 2003; Priglinger et al. 2003). There is also evidence that elevated transglutaminase-2 activity is associated with the pathogenesis of hypertrophic scarring, a condition in which there is abnormal turnover of ECM and in which affected tissue often appears hyalinized (Collighan, Cortez, and Griffin 2002). Immunohistochemical staining for transglutaminase-2 was conducted on the 27-week cohort (all compounds), and within control animal sections a positive signal was seen at grade 1 level within the lamina propria in the majority, whilst in each of the treated groups increased signal intensity, particularly in areas of hyalinization, was noted (majority grade 2, and several 3). This finding was not considered related to the prominent vasculature noted in a number of hyalinized areas, as the transglutaminase IHC score was unaffected by the wide variation in the incidence of prominent vasculature across the three corticosteroid groups.
In addition to its ability to increase the resistance of collagen to proteolytic and mechanical damage, transglutaminase-2 activity may also enhance the rate of ECM production (Verderio, Johnson, and Griffin 2004), and therefore shift the balance of ECM turnover in favor of deposition. This raises the possibility that increased transglutaminase-2 activity within the pyloric lamina propria is the cause of the hyalinization noted in this study. Transglutaminase-2 release is also greatly increased in situations of tissue damage and cellular stress, which poses the question of whether transglutaminase activity causes hyalinization, or is a reaction to it. This question is central to research on the role of transglutaminase in aberrant wound healing, and current evidence, though incomplete, favors a causal role in, rather than an effect of, abnormal healing (Verderio, Johnson, and Griffin 2004). Furthermore, there is in vitro evidence that treatment with a corticosteroid agonist (dexamethasone) can cause induction of transglutaminase-2 (Johnson, Scholfield, and Griffin 1998). This suggests that increased transglutaminase-2 activity as a result of chronic exposure of the stomach to corticosteroids might result in the pyloric hyalinization seen with the compounds investigated, and may also explain why this finding has not been noted with other drug classes. Why the finding should be restricted to the pyloric stomach is unclear but may be linked to accumulation of drug substance in this region resulting in particularly high local exposure.
The possibility that areas of mineralization might develop within areas of hyalinization was investigated using a number of histochemical techniques to detect the presence of mineral (von Kossa and chloranilic acid) and with immunohistochemical markers, which if positive can be associated with mineralization (osteopontin, TNAP, and intestinal alkaline phosphatase). Differences between control and treated animals considered to be test-article–related were noted with osteopontin. Osteopontin is known to immunolocalize to regions of mineralization (Gericke et al. 2005), and as such, the increased signal in treated animals may be linked to the areas noted by EM examination (though the two could not be directly colocated). Increased TNAP signal was considered equivocal due to a broad range of signal intensity in controls.
The nature of the EM changes were difficult to characterize, as both histochemical techniques used to detect the presence of mineral (von Kossa and chloranilic acid) were negative in all animals examined. It is known that excess cortisol can result in a tissue environment that attracts and binds calcium (Capen 2001), so it is possible that, although beyond the sensitivity of the histochemical techniques used, the EM evidence of mineralization indicates a direct effect of the corticosteroids.
Cells within the lamina propria seen to have Anitschkow-type nuclei (Nakatsuji et al. 1997), which were seen in association with areas of hyalinization across all compounds, were suspected to be myofibroblasts, consistent with evidence that areas of hyalinization are composed of collagen (as myofibroblasts are involved in ECM production and collagenous tissue contraction; Nakatsuji et al. 1997). This was confirmed by positive immunohistochemical signal for α-smooth muscle actin in all 27-week cohort-treated animals. Subsequent EM examination also confirmed the presence of increased numbers and size of myofibroblasts within the lamina propria. The presence of these cells in areas of hyalinization is not considered to be a consequence of the prominent vasculature also noted in a number of animals in the affected areas, as the myofibroblasts occurred independently of blood vessels and were also seen in affected mice where prominent vasculature was not a feature of the areas of hyalinization.
Other treatment-related findings within the stomach were restricted to the pyloric region and included a range of inflammatory changes such as multinucleate giant cells, prominent vasculature, edema, and neutrophilic inflammatory cell infiltrates all within the lamina propria. The greatest range of inflammatory pathology was seen in the 13-week cohort, reflecting the higher dose (500 µg/kg/day) received by these animals. These changes are likely due to the inhibitory effect of corticosteroids on the synthesis of prostaglandins considered to be protective to the gastric mucosa, particularly prostaglandin E2 (Nobuhara, Ueki, and Takeuchi 1985). These findings also raised the possibility that pyloric hyalinization could arise as a scar at the site of previous injury, and this was investigated by a review of short term (<13 week) studies of an unrelated corticosteroid that caused identical pyloric hyalinization in the CD-1 mouse (unpublished data). Gastric inflammatory/ degenerative changes were not noted during this review, indicating that pyloric hyalinization is not preceded by significant damage to the stomach wall.
Epithelial hyperplasia was noted within the pylorus in the majority of animals from the 13-week cohort given 500 µg/kg/day, and in animals given 200 µg/kg/day for 27 weeks, but was not present in animals from the 36- or 52-week cohorts treated with the same dose. This hyperplasia is likely to be related to the inflammatory and degenerative changes also seen in this region of the stomach in a number of mice from both these cohorts.
Increased vacuolation of the surface epithelium of the pyloric stomach was noted in a minority of animals from the 13-week (all compounds), 27-week (mometasone and budesonide), and 36-week cohorts (budesonide and flunisolide). Although the incidence of this finding was low, this region is a common site of artifactual damage during tissue handling/fixation such that the true incidence might be higher than indicated. Alcian blue and PAS histochemistry was used to assess whether any shift in the type of mucins produced by the surface epithelium had occurred in treated animals (27-week cohort—all compounds), which could account for this finding. With both techniques the staining characteristics were identical between control and treated animals, and as EM examination revealed lipid droplets in these cells, the increased vacuolation is considered due to lipid accumulation in the affected cells. This was considered to be an adaptive change, as there was no evidence of epithelial degeneration by light microscopy or EM.
In the absence of any toxicokinetic data in this study, the measurement of clinical pathology parameters and histological assessment of a range of corticosteroid sensitive organs were included as a surrogate marker of systemic drug exposure. Positive findings in these tissues/parameters included lymphoid atrophy of the mandibular and mesenteric lymph nodes, spleen, and thymus (Compton and Cidlowski 1986); and a reduction in circulating total leucocytes, predominantly reflecting lower lymphocyte counts and often accompanied by reductions in monocytes, eosinophils, neutrophils, and large unstained cells (data not shown). The molecular mechanisms involved in these changes are not fully understood, though loss of cytokine support and induction of apoptosis are known to be important factors (Chrousos 1995; Schwimmer and Parker 2001; Weber and Iwata 2009; Libretto 1995). Cortical atrophy within the adrenal glands of treated animals was noted at all time points whilst controls were unaffected, as were recovery animals (the finding is assumed to have resolved). This change was generally associated with a reduction in organ weight and a marked lowering in plasma corticosterone, reflecting feedback on the hypothalamo-pituitary-adrenal axis following exogenous corticosteroid administration (Loose et al. 1980; Schäcke, Wolf-Dietrich, and Asadullah 2002; Peters et al. 2007).
A higher proportion of hair follicles in treated animals from various time points were noted to be in telogen or catagen stages of the hair cycle compared with controls or recovery animals, which were predominantly in the anagen stage. This reflects a corticosteroid block on initiation of the anagen stage of the hair cycle (Stenn et al. 1993).
Other findings in this study, each reported to be a consequence of corticosteroid administration, included transient body weight loss and/or reduced body weight gain (De Vos et al. 1995; Libretto 1995) and increases in plasma total protein concentrations reflecting increased protein synthesis (Libretto 1995; Schäcke, Wolf-Dietrich, and Asadullah 2002).
Pyloric hyalinization is considered to represent a novel class effect of corticosteroid agonists in the CD-1 mouse, given the high incidence of this change in animals treated with three different corticosteroid agonists. It is restricted to the lamina propria of the pyloric stomach; is not related to amyloid accumulation; and in its early stages is associated with, though not preceded by, local inflammatory change. EM and histochemical investigation characterized the finding as an expansion of collagen-rich ECM, with focal areas suggestive of mineralization. The increased numbers of myofibroblasts in the affected area are the likely source of the expanded ECM, and the hyaline appearance is considered likely to be related to the activity of the corticosteroid responsive enzyme transglutaminase-2.
Pyloric hyalinization is considered likely to be a local effect; however, a contribution from systemic exposure cannot be ruled out. A metabolite effect is unlikely given that the same change was seen with multiple corticosteroids. The main differential for pyloric hyalinization is amyloid accumulation, given the high historical incidence of amyloidosis in the CD-1 mouse, and that the two findings are indistinguishable by H&E staining. Histochemistry or electron microscopy may be used to differentiate amyloidosis from pyloric hyalinization.
The significance of this finding in terms of human risk assessment was considered minimal. The finding is nonadverse, has only been identified in the CD-1 mouse, did not progress in terms of incidence and severity with increased duration of dosing, and showed clear signs of reversibility following a 16-week off-dose period.
Footnotes
This work was funded by GlaxoSmithKline R&D.
Abbreviations
Acknowledgment
The authors thank Mr. John Bowles for help with the figures.
