Abstract
During baseline evaluation prior to a preclinical safety study, a 10-month-old male pure-bred Beagle dog was found to have marked thrombocytopenia (6 × 103 platelets [PLT]/µL) associated with a mean platelet volume (MPV) of 17.9 fL. Tests for Rickettsia rickettsii, Ehrlichia canis, and Borrelia burgdorferi were negative. Buccal bleeding time was normal. Over 3 months, PLT were 4 to 141 × 103 PLT/µL, and MPV was 11.4 to 25.1 fL; however, PLT were <50 × 103 PLT/µL and MPV was >16 fL during most of this period. Antinuclear antibody (ANA) and anti-PLT antibody tests were negative. Genotyping for the presence of a beta 1-tubulin mutation demonstrated the normal wild-type gene. Treatment with prednisone resulted in normal values after only 3 days. Ultrastructure of enlarged PLT was consistent with that of immature PLT, characterized by reduced numbers of peripheral microtubules and the presence of rough endoplasmic reticulum, free ribosomes, Golgi apparatus, and a prominent canalicular system. PLT ultrastructure and glucocorticoid responsiveness supported a diagnosis of immune-mediated thrombocytopenia that was masked by the cyclic nature of PLT decreases and lack of clinical signs. Inclusion of such a dog in a preclinical safety study could result in misinterpretation of clinical pathology findings.
Case Presentation
During baseline evaluation (hematology, coagulation, and clinical chemistry measurements) for inclusion in a preclinical safety study at Abbott Laboratories, marked thrombocytopenia (6 × 103 platelets [PLT]/µL) associated with a mean platelet volume (MPV) of 17.9 fL (June 8, 2009) was noted in an intact male pure-bred Beagle dog, aged 10 months, from a commercial breeder of laboratory Beagle dogs in the United States. No clinical signs were observed. The dog’s history included an intestinal infection at the age of 4 months, which was treated with erythromycin and metronidazole for 5 consecutive days. The dogs’ vaccination status included a nasal Bordetella-parainfluenzavirus-adenovirus vaccine, repeated canine parvovirus modified live vaccine, and a Bordetella vaccine injection as a puppy. Later repeated vaccinations included canine papillomavirus 1 and 2, distemper-adenovirus-parainfluencavirus-parvovirus-leptospirosis, and one against rabies. The vaccination protocol at the breeder’s facility was completed at the age of 4 months. The dog had regular treatments with pyrantel pamoate anthelmintics and ivermectin at the breeder’s facility. At 5 months of age, PLT and MPV were normal at 704 × 103 PLT/µL and MPV of 9.8 fL. At 7 months of age, a respiratory infection was treated with azithromycin for 5 consecutive days. The dog was delivered to Abbott Laboratories at 9 months of age (April 30, 2009). Tap water was available ad libitum through an automatic watering system. Standard food ration (Harlan 2025C Tekland Global 25% Protein Dog Diet, produced by Harlan Tekland, Madison, WI) was provided each day. Environmental conditions (temperature at 71 ± 6°F and humidity between 30% and 70%) were documented daily. The dog was observed at least once daily for general condition and signs of petechial hemorrhages. No clinical signs indicating a bleeding disorder were observed during a 7-month observation period. There was no evidence of prolonged bleeding after venipuncture. Body weight measured upon arrival was 9.3 kg. In the following 7 months (quarterly measurement), the dogs’ weight was between 9.00 and 10.3 kg.
In the first month of the observation period, blood was collected approximately once weekly; in the second, third, and fourth month of the observation period, blood was collected approximately twice weekly, except for daily blood collection for 3 days after the glucocorticoid treatment initiation (Table 1 ). Blood samples (approximately 2 to 8 mL depending on the proposed analysis) were collected from the Vena jugularis or Vena cephalica using a 21-gauge needle (Kendall Monoject, Tyco Healthcare, Mansfield, MA), and the blood was transferred to the corresponding test tubes.
Selected Hematology Data and Glucocorticoid Treatment Data
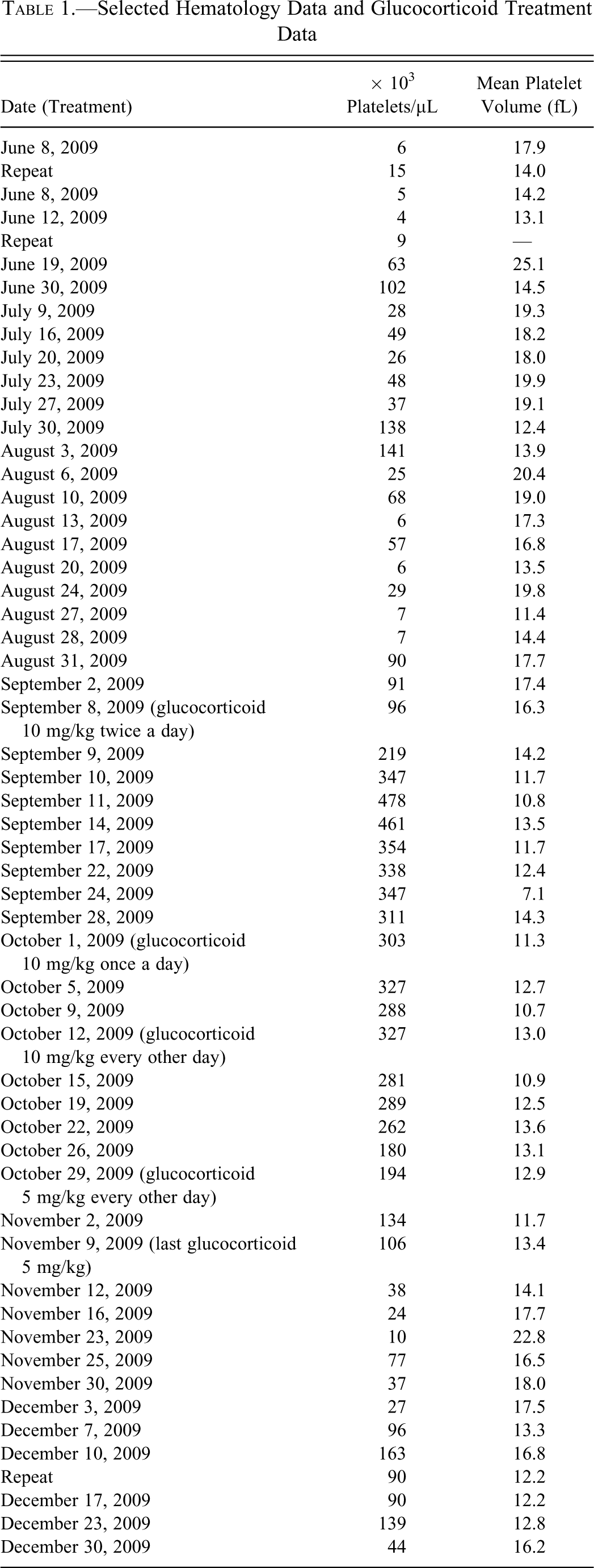
For hematology evaluation, blood samples were collected in 2-mL sample tubes (Becton Dickinson, Franklin Lakes, NJ) using K2 EDTA anticoagulant, stored at room temperature (RT), and evaluated on the same day. For coagulation parameters, blood was collected in 2-mL sample tubes (Monoject, Sarstedt AG & Co., Newton, NC) with 3.8% sodium citrate as anticoagulant and stored at RT until evaluation on the day of sampling. Clinical chemistry samples were collected in 3-mL sample tubes (Sarstedt AG & Co.) with no additive, centrifuged at 1500 rpm for 10 min, stored at RT, and analyzed the same day. Hematology parameters were analyzed at each time point blood was collected (Table 1) and were evaluated with a Siemens ADVIA 2120 Analyzer (Siemens Healthcare Diagnostics, Tarrytown, NY) with Multispecies software and Abbott Cell Dyn 3700 (Abbott Diagnostics, Abbott Park, IL) to confirm the PLT. Complete blood (cell) counts were performed. Peripheral blood smears were prepared, air dried, and stained using a Wescor automated slide stainer Model-Aerspray 7120 (Wescor Inc., Logan, UT) using a modified Romanowsky stain. Blood smears were examined for clumping and size to verify the PLT and MPV. Hematology parameters (except PLT and MPV) revealed no significant differences from the physiological range (Abbott Laboratories reference range) throughout the 7-month observation period. There was one observance of mild transient monocytosis and occasional mild increases in white blood cells and neutrophils, which were attributed to pododermatitis and dog bite injury (second and third month of the observation period). Over the first 3 months, PLT were 4 to 141 × 103 PLT/µL and MPV was 11.4 to 25.1 fL (Abbott Laboratories reference range 170-403 × 103 PLT/µL, MPV 9.7-15.8 fL), indicating rare periods with PLT counts slightly higher than 100 × 103 PLT/µL. However, PLT were <50 × 103 PLT/µL and MPV was >16 fL during most of this period (Table 1). Decreased PLT were mostly associated with increased MPV values. Artifactual clumping was never observed in blood smears from repeated blood collections, and enlarged thrombocytes were confirmed in blood smears (Figure 1 ). Glucocorticoid (prednisone, Deltasone, West-Ward Pharmaceuticals, Eastown, NJ) treatment was started in the fourth month of the observation period (September 8, 2009) with 10 mg/kg orally twice a day for 21 days, then reduced to 10 mg/kg orally once a day for 14 days, to 10 mg/kg orally every other day for 14 days, and finally to 5 mg/g orally every other day for 14 days. The PLT increased to normal values of 347 × 103 PLT/µL after only 3 days of glucocorticoid treatment. PLT levels dropped after discontinuation of the glucocorticoid in the sixth month of the observation period (Table 1). PLT and MPV levels over a period of 3 months are shown in Figure 2 .
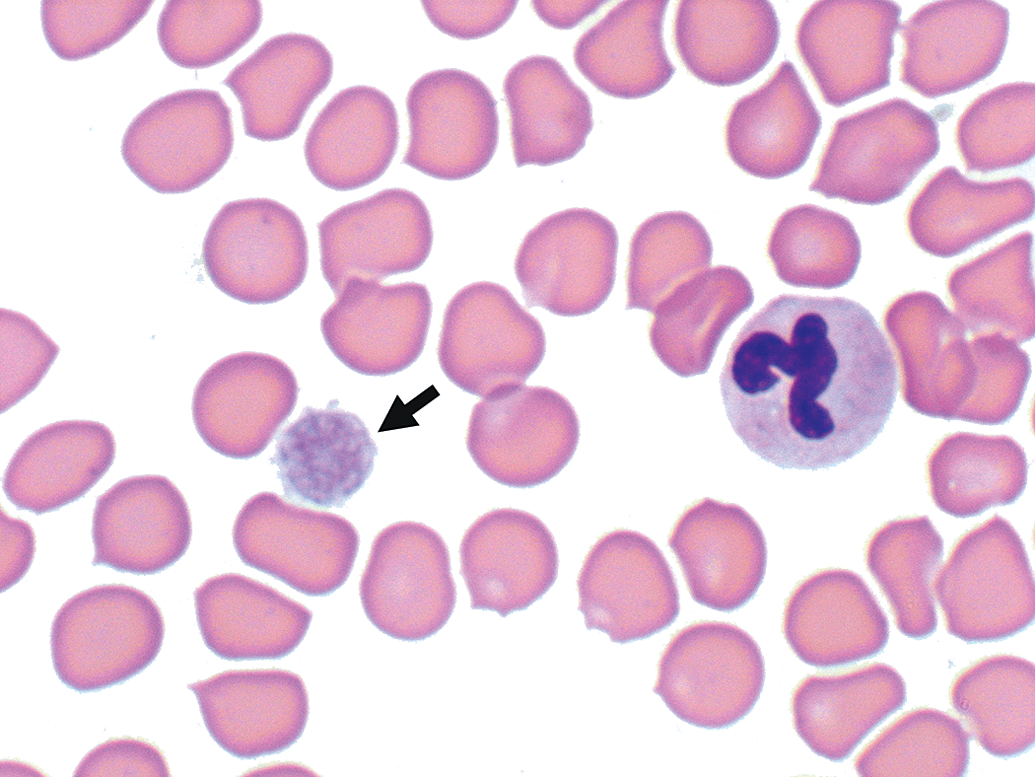
Blood smear. Image at 1000× magnification of large-diameter platelet (arrow).
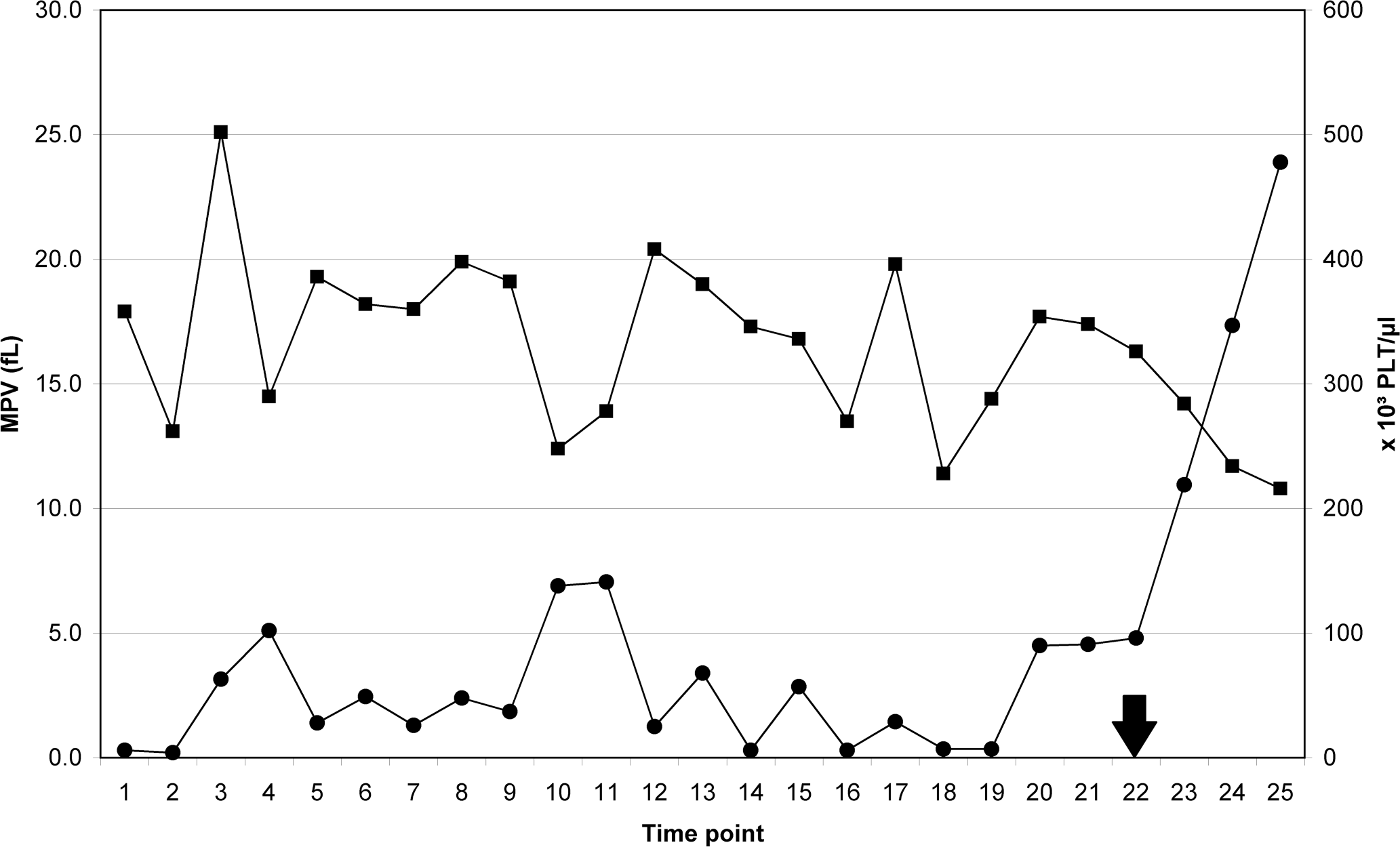
Platelets (PLT) and mean platelet volume (MPV) over 3 months. • = PLT, ▪ = MPV; arrow indicates the start of glucocorticoid treatment.
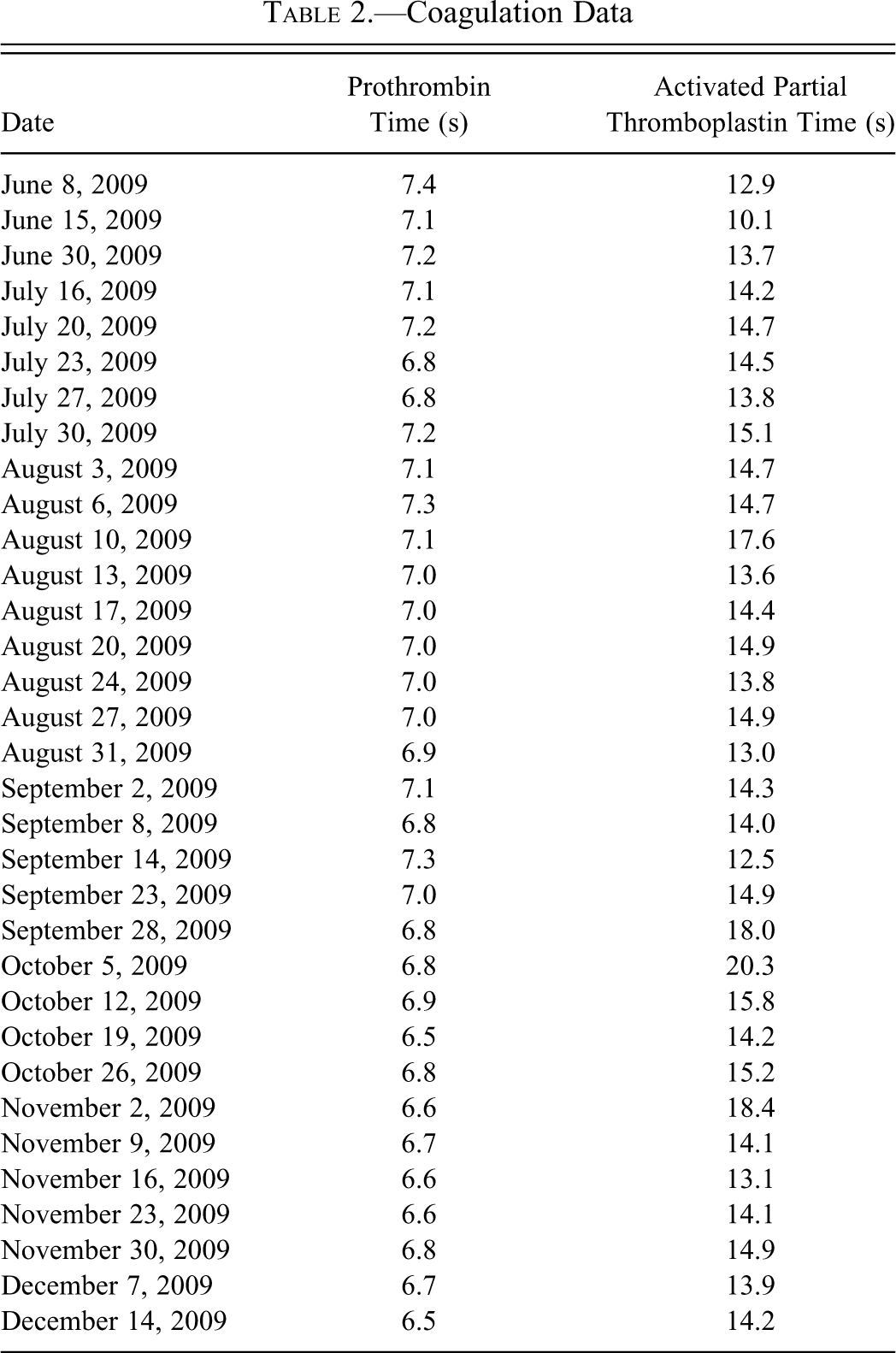
Coagulation profiles were evaluated with an AMAX Destiny Analyser (Trinity Biotech, Wicklow, Ireland). Coagulation parameters were not evaluated at each time point blood was collected but were evaluated on a regular basis (Table 2 ). Activated partial thromboplastin time (APTT [seconds]) and prothrombin time (PT [seconds]) revealed no differences from the physiological range (Abbott Laboratories reference range) through the 7-month observation period despite a mild increase in APTT at four time points (17.6 sec, 18.4 sec, 20.3 sec, and 18.4 sec; Abbott Laboratories reference range APTT 12.4– 16.4 sec). Corresponding PT values were within or minimally below the reference range (Abbott Laboratories reference range PT 7.0-8.4 sec). These mild increases in APTT were considered to be the result of activation of the coagulation cascade during blood collection (Dameron et al. 1992) rather than a real change in coagulation time. A buccal mucosal bleeding time test was carried out using a Surgicutt Junior Bleeding Time Device (Jorgensen Laboratories, Loveland, CO), following the supplier’s recommended procedure. Buccal bleeding time was normal (2 min 36 sec). (Normal buccal bleeding time for dogs is less than 4 min; http://www.jorvet.com/manuals.shtml; Côté 2007).
Coagulation Data
Serum biochemical profiles were evaluated with an Abbott Aeroset (Abbott Diagnostics, Abbott Park, IL). Clinical chemistry parameters were not analyzed at each time point blood was collected but evaluated on a regular basis. Results for clinical chemistry parameters did not show any significant differences from the physiological range (Abbott Laboratories reference range) through the 7-month observation period. Tests for Rickettsia rickettsii, Ehrlichia canis, and Borrelia burgdorferi performed in the first month of the observation period at Antech Diagnostics (Oak Brook, IL) were negative. Corresponding PLT was 9 × 103 PLT/µL. The antinuclear antibody (ANA) test was performed twice in the first month of the observation period via an indirect fluorescent antibody method (Antech Diagnostics, Kit Immuno Concepts, Sacramento, CA) and was negative. Corresponding PLT were 28 and 49 × 103 PLT/µL, respectively.
Clinical physical examination by a veterinarian, which included inspection of the mucosal membranes, palpation of lymph nodes, and cardiac as well as lung auscultation, was performed on a regular basis and revealed no abnormalities. During the second and third month of the observation period, the dog was diagnosed with pododermatitis and a dog bite injury. For a short duration, he was treated topically with a combination of antimicrobial and anti-inflammatory compounds.
Abdominal radiographs (left and right lateral, ventrodorsal) were performed in the second month of the observation period using an Innovet Select, model E7242X (Summit Industries, Chicago, IL), and abdominal ultrasound of the kidneys, liver, and spleen was performed using a LOGIQ Book (GE Medical Systems, Milwaukee, WI). No abnormalities were detected.
Bone marrow aspirate and biopsy were taken from the iliac bone using routine methods and with the dog under anesthesia and analgesia (McGuire 1991). A Jamshidi needle was used for the bone marrow aspiration. Three repeated bone marrow aspiration and biopsies were acellular and therefore nondiagnostic.
Phenotyping via gene sequencing was performed in the third month of the observation period to determine whether the affected dog had a G745A single nucleotide polymorphism (snp) in the beta 1-tubulin gene according to routine test methods as described below. Five milliliters of whole blood (EDTA) were obtained from the affected dog and from one normal control dog (not age matched since this was not considered critical for this kind of test). DNA was isolated manually using Flexigene DNA Kit (Qiagen, Valencia, CA) reagents, following the manufacturer’s protocol. The Genbank sequence (XM-543075) for the canine beta 1-tubulin gene was used with Vector NTI software (version 10.3.0, Invitrogen, Carlsbad, CA) to design sequencing primers bordering the G745A snp. Primers were commercially prepared by Integrated DNA Technologies (Coralville, IA). Fifty nanograms of DNA was added to 25 µL PCR mastermix (1× PCR High Fidelity Buffer [Invitrogen], 2 mM MgSO4 [Invitrogen], 0.2 mM dNTP [Sigma, St. Louis, MO], and 0.03 mM Platinum High Fidelity Taq [Invitrogen]). DNA sequencing was performed on the 3130xl Genetic Analyzer (ABI, Carlsbad, CA) following the manufacturer’s protocol and analyzed with Sequencher software (version 4.5, Gene Codes Corporation, Ann Arbor, MI). Corresponding PLT for the affected dog was 6 × 103 PLT/µL. Gene sequencing showed the affected dog and the control dog to be homozygous wild type (GG) for G745A in the beta 1-tubulin gene.
In the first month of the observation period, PLT from a normal control dog and from the affected dog (4 × 103 PLT/µL and 102 × 103 PLT/µL, respectively) were prepared by centrifuging whole blood (EDTA) at 3500 rpm for 20 min to obtain a buffy coat, which was fixed with 3% glutaraldehyde. Epoxy blocks were prepared for light and transmission electron microscopy using standard laboratory procedures. Epoxy blocks were trimmed, and semi-thin sections were cut at 1 µm with a Leica Ultracut ultra microtome (Leica Microsystems, Bannockburn, IL), stained with toluidine-blue, and scanned using a ScanScope CS (Aperio Technologies Inc, Vista, CA). Ultra-thin sections (90-100 nm) were cut on a Leica Ultracut ultra microtome, stained with 2% methanolic uranyl acetate and Reynolds’ lead citrate, and examined with a JEOL JEM-1400 electron microscope (JEOL, Peabody, MA). By light microscopy of the semi-thin sections, there was only a thin layer of PLT in the buffy coat of the thrombocytopenic dog (4 × 103 PLT/µL), and the PLT were very large and rounded (Figure 3 ). These PLT were up to two times larger than in normal control dogs, and almost none of the PLT had a normal discoid shape. Discoid shape was indicated depending on the random section direction through the PLT appearing flat when sectioned transversally. In the sample collected when the PLT were 102 × 103 PLT/µL, nearly all PLT had a normal size and discoid shape, but also PLT with increased diameter were present.
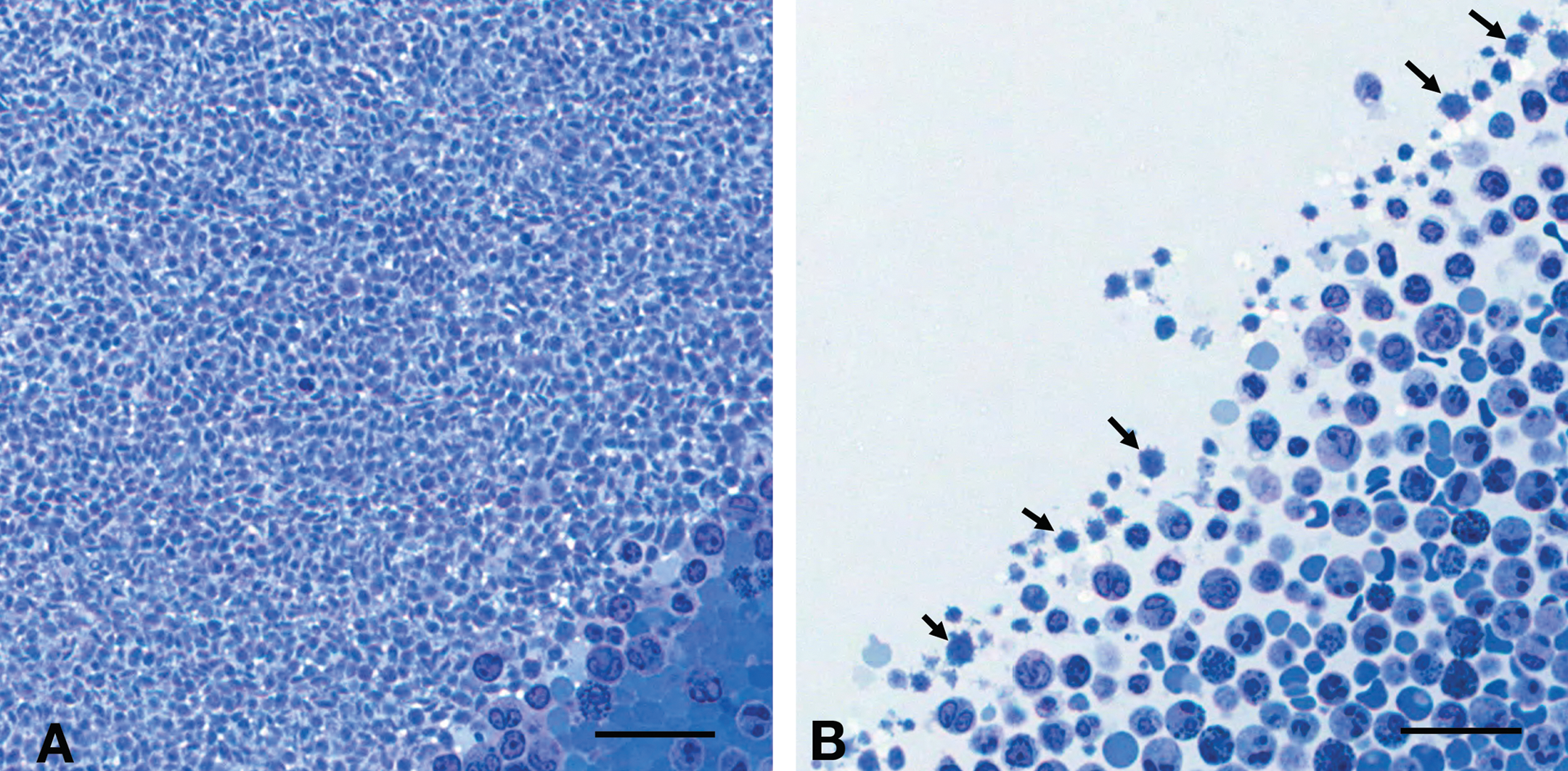
Buffy coat, semi-thin section, stained with toluidine blue. (A) Normal control dog. (B) Thrombocytopenic dog. A very thin to absent layer of platelets (arrows). Almost all platelets have a large diameter and rounded shape; almost none of the platelets show a normal discoid shape. A, B: scale bar = 25 µm.
Transmission electron microscopy of the ultra-thin sections of the buffy coat from the thrombocytopenic dog at 4 × 103 PLT/µL revealed large PLT with a rounded shape, presence of rough endoplasmic reticulum, free ribosomes, and remnants of the Golgi apparatus (Figure 4 ). In addition, there was a prominent canalicular system, which is normally inconspicuous in mature PLT (Zucker-Franklin, 1990). This complement of organelles is characteristic of immature PLT. There was no indication of discoid shape at random section direction of the PLT at 4 × 103 PLT/µL (Figure 5 ). The ultrastructure of the majority of PLT taken when PLT was 102 × 103 PLT/µL was normal, but approximately one third of the PLT had increased diameter and rounded shape (Figure 5).
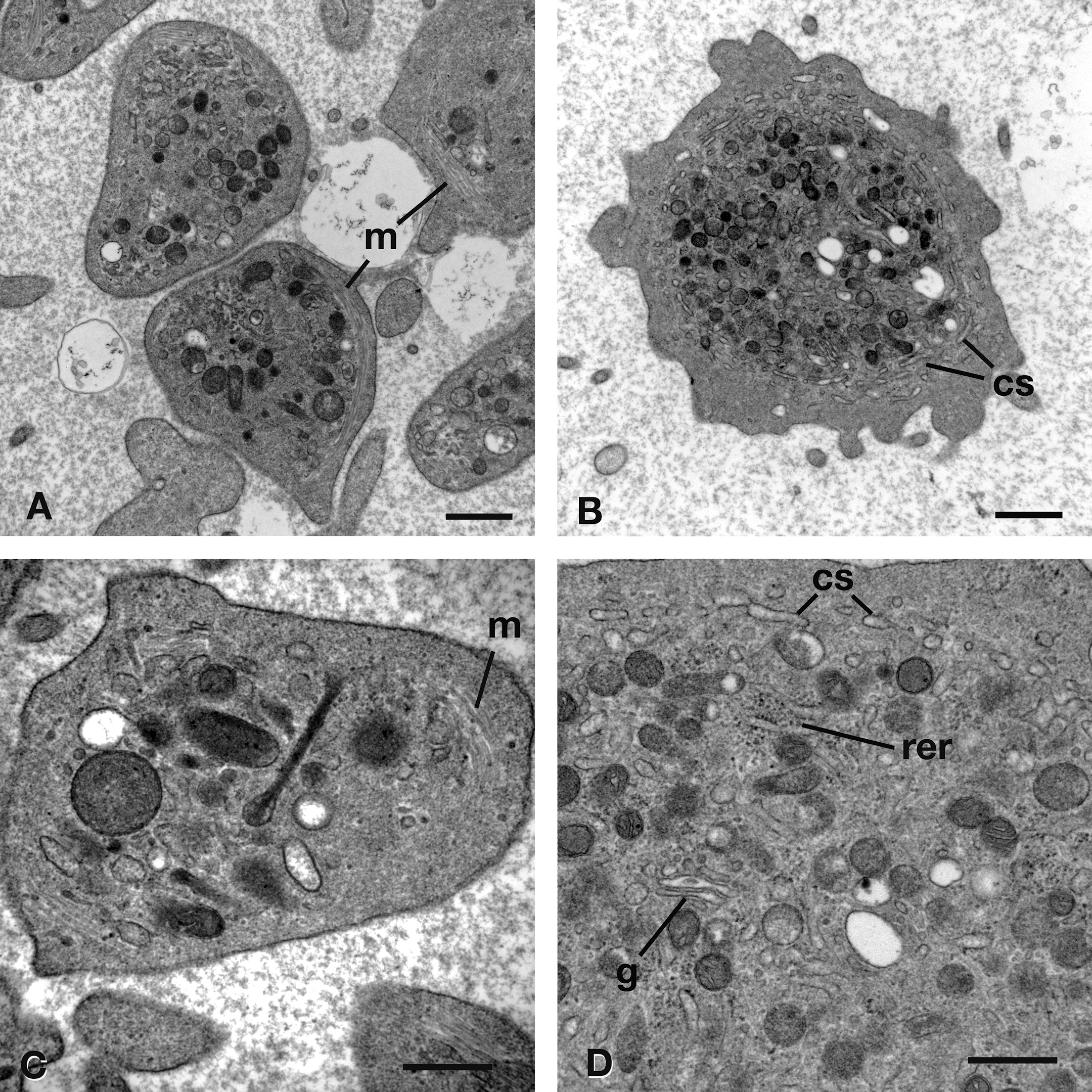
Ultrastructure of platelets (PLT) from normal control (A, C) and thrombocytopenic (B, D) dog. (A, C) PLT from the normal control dog have a prominent circumferential band of microtubules (m). (B, D) PLT from the thrombocytopenic dog (4 × 103 PLT/µL) lack circumferential microtubules and contain rough endoplasmic reticulum (rer), remnants of Golgi apparatus (g), and a prominent canalicular system (cs). A, B: scale bar = 1 µm; C, D: scale bar = 500 nm.
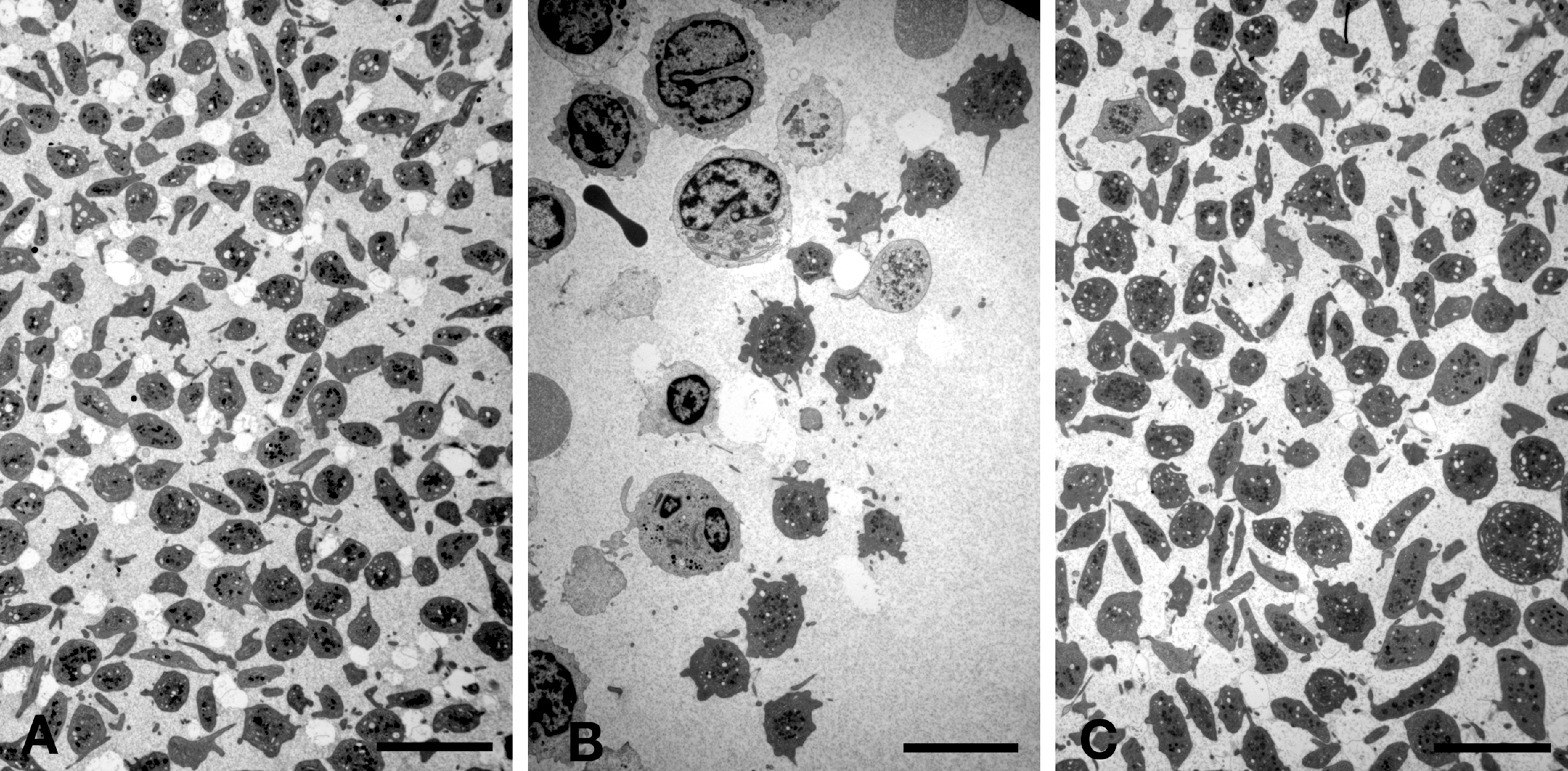
Buffy coat, ultra-thin section, transmission electron microscope images from normal control (A) and thrombocytopenic dog at 4 × 103 platelets (PLT)/µL (B) and at 102 × 103 PLT/µL (C). (A) PLT from the normal control dog have equal diameter and normal discoid shape. (B) PLT from the thrombocytopenic dog at 4 × 103 PLT/µL are very large and rounded in shape. Only few PLT are present. (C) The majority of PLT from the thrombocytopenic dog at 102 × 103 PLT/µL have normal size and discoid shape. Approximately one third of the PLT have increased diameter and rounded shape. A-C: scale bar = 5 µm.
The glucocorticoid responsiveness and the PLT drop after discontinuation of the glucocorticoid (Table 1) supported a diagnosis of immune-mediated thrombocytopenia. Therefore, an anti-PLT antibody test was performed with a PLT surface–associated immunoglobulin G (PSAIgG) assay to detect IgG bound to the surface of dog PLT (Kansas State Veterinary Diagnostic Lab, Manhattan, KS). After a 2-week washout period of the glucocorticoid on November 23, 2009, blood samples of the affected dog with corresponding 10 × 103 PLT/µL and of three age- and sex-matched control dogs were tested for anti-PLT antibodies and were all negative.
Discussion
Thrombocytopenia (Abbott Laboratories reference <170 × 103 PLT/µL) is one of the most commonly recognized hematologic disorders seen in veterinary medicine (Zimmerman 2000). It is essential to understand the common pathogenesis of thrombocytopenias, particularly in the case of preclinical safety studies, where it is important that background findings do not confound test item–related changes. Several mechanisms were considered for the macrothrombocytopenia described here.
Physical examination findings in dogs with marked thrombocytopenia <25 × 103 PLT/µL may be petechia; ecchymoses; ocular hemorrhage; bleeding in the gastrointestinal tract, manifesting as melena; and pale mucous membranes (Côté 2007; Jackson and Kruth 1985), but these were not observed in this case. In addition, most of the reported causes for thrombocytopenia were ruled out for the dog described here. Some of the mechanisms that were considered include hypersplenism with abnormal PLT distribution, suppression of bone marrow PLT production, direct drug-induced effects, or increased peripheral blood PLT consumption after bone marrow release (Weiss 2000; Zimmerman 2000; Russel and Grindem 2000). Hypersplenism was ruled out in this dog since abdominal radiographs and abdominal ultrasound were normal.
Infections with several agents, including R. ricketsii (Scott 2000; Côté 2007; Grindem et al. 1999; Pantonawitz 2003), E. canis (Scott 2000; Côté 2007; Grindem et al. 1999; Pantonawitz 2003; Bulla et al. 2004), and B. burgdorferi (Pantonawitz 2003) may produce thrombocytopenia in dogs due to many different mechanisms including bone marrow suppression, increased PLT consumption, or immune-mediated pathogenesis (Weiss 2000; Scott 2000; Russel and Grindem 2000). However, there was no evidence of infection with any of these agents in this dog.
Peripheral blood PLT consumption may occur with severe hemorrhage, vasculitis, or disseminated intravascular coagulopathy, or it may be the result of accelerated PLT destruction via infectious diseases and both nonimmune mechanisms (directly caused by medications) or immune-mediated mechanisms (Weiss 2000; Zimmerman 2000; Scott 2000; Russel and Grindem 2000; Handagama and Feldmann 1986). There was no evidence for any of these mechanisms, with the exception of immune-mediated thrombocytopenia.
Immune-mediated thrombocytopenia can be either primary, idiopathic (Scott 2000; Putsche and Kohn 2008; Wilkerson et al. 2001), or autoimmune in etiology (Scott 2000), or it can be secondary due to neoplasia (Scott 2000), infections (Scott 2000; Côté 2007; Grindem et al. 1999; Pantonawitz 2003; Bulla et al. 2004; Wilkerson et al. 2001; Jackson and Kruth 1985), or following vaccinations (Dodds 2001; Scott 2000; Jackson and Kruth 1985) and treatment with antibiotics (Scott 2000; Handagama and Feldmann 1986). Thrombocytopenia may occur as a consequence of treatment with macrolid antibiotics (erythromycin), aminoglycoside antibiotics (streptomycin), penicillins, and chloramphenicol (Handagama and Feldmann 1986; Zimmerman 2000) as well as sulfonamides (Handagama and Feldmann 1986; Jackson and Kruth 1985). Possible mechanisms are a direct bone marrow suppression, an increased PLT destruction, or induction of immune-mediated thrombocytopenia via several pathways (Handagama and Feldmann 1986; Scott 2000; Aster and Bougie 2007). Antibiotics most often induce drug-dependent PLT-reactive antibodies, in which case PLT numbers increase after discontinuation of the drug (Handagama and Feldmann 1986). In rare cases, drugs induce autoantibodies that are capable of destroying PLT in the absence of the sensitizing agent (Aster and Bougie 2007; Aster 2000). These compounds seem to provoke an immune response directed at an unadulterated antigen on the PLT’s surface membrane (Zimmerman 2000). Another rare immune-mediated mechanism is induction of hapten-dependent antibodies, where the hapten links covalently to a membrane protein and induces a drug-specific immune response (Aster and Bougie 2007). The principal antibody class associated with immune-mediated thrombocytopenia in the dog is IgG (Lewis and Meyers 1996a; Jackson and Kruth 1985). The PLT surface–associated antibody is often directed against PLT glycoproteins such as the fibrinogen receptor antigen gpIIb/IIIa (Lewis and Meyers 1996a, 1996b). This dog had normal PLT at the age of 5 months after completion of his vaccination protocol and after treatment with erythromycin and metronidazole (aged 4 months); therefore, the vaccinations and treatment with these antibiotics were not considered as a cause of the thrombocytopenia. But thereafter (aged 7 months), he was treated with azithromycin for 5 consecutive days, and his PLT were unknown until the baseline evaluation at Abbott Laboratories at the age of 10 months. Adhikari et al. (1996) reported about the occurrence of thrombocytopenia in humans treated with azithromycin for 5 consecutive days. A 50% reduction in PLT was observed in 12.29% of the patients. It cannot be ruled out that the treatment with azithromycin was involved in the development of the thrombocytopenia in this case. The ANA test is a diagnostic test to detect systemic lupus erythematosus (SLE), an autoimmune disorder. SLE may be associated with immune-mediated thrombocytopenia (Scott 2000), but the negative ANA does not rule out an immune-mediated thrombocytopenia.
The PSAIgG, which is a specific test to detect PLT surface–associated IgG, was negative in this case; however, PSAIgG levels are not always increased in dogs with primary and secondary immune-mediated thrombocytopenia (Wilkerson et al. 2001).
Immune-mediated thrombocytopenia may be successfully treated with glucocorticoids (prednisone or prednisolone; Zimmerman 2000; Scott 2000; Putsche and Kohn 2008; Jackson and Kruth 1985), which was also successful in this case.
In humans, several inherited giant PLT disorders associated with thrombocytopenia are well characterized (Mhawech and Saleem 2000). In the dog, thrombocytopenia occurs as an inherited disorder in the Cavalier King Charles Spaniel, caused by a mutation of the gene for beta 1-tubulin in PLT microtubules (Davis et al. 2008). Idiopathic and asymptomatic macrothrombocytopenia exhibits an autosomal recessive trait (Pedersen et al. 2002; Singh and Lamb 2005), in which affected dogs show thrombocytopenia (<100 × 103 PLT/µL) and giant PLT but no bleeding diathesis (Davis et al. 2008; Pedersen et al. 2002; Singh and Lamb 2005; Cowan et al. 2004). The beta 1-tubulin gene DNA sequence at nonsynonymous snp position G745A from this dog was homozygous wild type; therefore, the described polymorphism resulting in microtubule dysfunction (reduction of marginal band of microtubules/reduced release of PLT from megakaryocytes; Davis et al. 2008) was not likely to be the cause of macrothrombocytopenia.
The ultrastructure of normal PLT is well characterized. Mature PLT are discoid in shape and contain granules, mitochondria, a dense tubular system, an open canalicular system, and circumferential (peripheral) microtubules (Tablin 2000; Kuter et al. 1997; Maldonado, Pintado, and Pierre 1974). In contrast, immature or “young” PLT may often be bigger in size and rounded in shape and additionally contain remnants of the Golgi apparatus, rough endoplasmic reticulum, free ribosomes, and reduced numbers of peripheral microtubules (Kuter et al. 1997; Maldonado, Pintado, and Pierre 1974), as described in the PLT from the thrombocytopenic dog at 4 × 103 PLT/µL.
In conclusion, PLT ultrastructure, glucocorticoid responsiveness, and the PLT drop after discontinuation of the glucocorticoid supported a diagnosis of immune-mediated thrombocytopenia despite negative ANA and negative PLT surface–associated IgG (Wilkerson et al. 2001).
The immune-mediated thrombocytopenia was masked by the cyclic nature of PLT decreases and lack of bleeding disorders and clinical signs, and therefore, inclusion of such a dog in a preclinical safety study could have resulted in misinterpretation of clinical pathology findings. Application of baseline evaluation prior to the preclinical safety study allowed the detection of the unexpected thrombocytopenia that otherwise would have been missed and could have been confound with test item–related changes.
Ethical Statement
The dog was housed and treated in compliance with the regulations of the United States, and animal welfare was in compliance with the U.S. Department of Agriculture’s Animal Welfare Act (9 CFR parts 1, 2, and 3). The Guide for the Care and Use of Laboratory Animals, Institute of Laboratory Animal Resources, National Academy Press, Washington, DC, was followed. Abbott Laboratories laboratory animal programs are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Footnotes
Acknowledgments
The authors would like to thank David Cugier for the technical preparation of the materials for transmission electron microscopy and Diann Weddle for providing the blood smear image.
