Abstract
This article outlines the changes and underlying rationale for modifications to the histopathological evaluation of the nervous system during toxicology and carcinogenesis studies conducted by the National Toxicology Program (NTP). In the past, routine evaluation of the nervous system was mostly limited to three sections of brain, and occasionally the spinal cord and peripheral nerves. Factors such as the increasing occurrence of human neurological diseases and associated economical cost burden, the role of unidentified environmental stressors in neurodegenerative disorders, multiple therapeutic drug-induced neuropathies noted in human clinical trials, and the exponential use of environmental chemicals with unknown neurotoxic potential necessitate a more extensive evaluation of the nervous system. The NTP has modified its protocol to include examination of key anatomic subsites related to neurodegenerative diseases such as Parkinson’s disease. Modifications include four additional sections of the brain. Increasing the number of brain sections permits examination of a greater number of specific anatomic subsites with unique vulnerability. In addition, the spinal cord, peripheral nerves, trigeminal ganglion, and intestinal autonomic ganglia will be evaluated as needed. It is expected that this modified approach will increase the sensitivity of detecting neurotoxicants and neurocarcinogens important in human neurologic and neurodegenerative disorders.
Since its inception in 1978, one of the major goals of the National Toxicology Program (NTP) has been to identify carcinogens (based on carcinogenicity for laboratory animals under conditions of study as adopted by NTP since 1983) based on 2-year rodent bioassays. The standard three sections of the brain examined in such routine studies were taken at the levels of (1) the basal ganglia, including the cerebrum at the frontal cortical level; (2) the thalamus at the mid-infundibular level; and (3) a mid-cerebellar section that included the cerebellum and medulla. For studies in which clinical neurological signs were noted, or studies in which agents were suspected to be neurotoxic, sections of the spinal cord and the sciatic nerve were also examined. In addition to the identification of carcinogens, the NTP has recently started a number of initiatives to assess adverse, nonneoplastic toxicological effects. Since environmental factors that affect nonneoplastic conditions such as immune-mediated diseases (Virgolini et al. 2005) and reproductive disorders (Davey et al. 2008) are increasingly common, initiatives within the NTP have included enhanced immunopathology as well as revised criteria for histopathological evaluation in reproductive and developmental toxicity studies. Similarly, increasing attention to the role of environmental factors in neurological disorders such as Parkinson’s disease (Cory-Slechta et al. 2005) and autism (Cavagnaro 2007) also emphasizes the need for improved toxicity endpoints in the evaluation of the nervous system.
Since the field of neuroscience continues to evolve rapidly, it is important that the linked fields of neuropathology and neurochemistry be effectively poised to screen any potential toxicants affecting neurochemical pathways (Bolon, Funk, and Sills 2010; Jett 2010; Mittal and Ganesh 2010). It is also critical that the neuropathology evaluation examines regions that are linked to subclinical changes (sensory, memory, learning deficits) that may not be obvious with routine clinical observations in initial screening studies. For these reasons, adopting a more precise, systematic approach to histopathological assessment of the nervous system is especially prudent even at the preliminary stages of toxicity screening studies. Although the complexity of the nervous system warrants a coordinated approach with multiple toxicity endpoints that include neurobehavioral, neurophysiological, and neuropathology endpoints, the focus of this article specifically addresses the modifications to the histopathological evaluation of the nervous system in routine NTP studies.
Modified Protocol and Specific Rationale
The modified protocol entails preparation of seven sections of the rat and mouse brain (Figure 1). In addition, three sections of the spinal cord (cervical, thoracic, and lumbar); peripheral nerves, namely, sciatic and tibial in rat, and the sciatic only in mice; cranial nerve (trigeminal nerve components with ganglion); and autonomic plexuses from routine sections of colon (Table 1) will be evaluated as warranted. Because of the time and detailed technique required to dissect the tibial nerve in mice in large-scale toxicological studies, the tibial nerve will be retained in situ and evaluated in mice only if lesions are noted in the rat/mouse sciatic nerve or in the rat tibial nerve. The additional sections of brain have been selected to increase the probability of detecting potential neurotoxicant effects at a greater number of specific sites (Table 2 ). For example, the olfactory bulb is particularly vulnerable to inhaled toxicants such as manganese (Dorman et al. 2006), uranium (Monleau et al. 2005), cobalt (Persson, Henriksson, and Tjalve 2003), and naphthalene (NTP 2000). Hence, compounds with a predilection for the olfactory bulb can be evaluated for toxicological significance following short- and long-term exposures that is especially relevant in environmental/occupational settings (Aschner, Erikson, and Dorman 2005). In addition, the olfactory bulb is also a site for continual neuronal death and neuronal replacement in adult mammals since cells are continuously generated in the subventricular zone and continue to migrate rostrally (Grabiec, Turlejski, and Djavadian 2009) to the olfactory bulb during life.
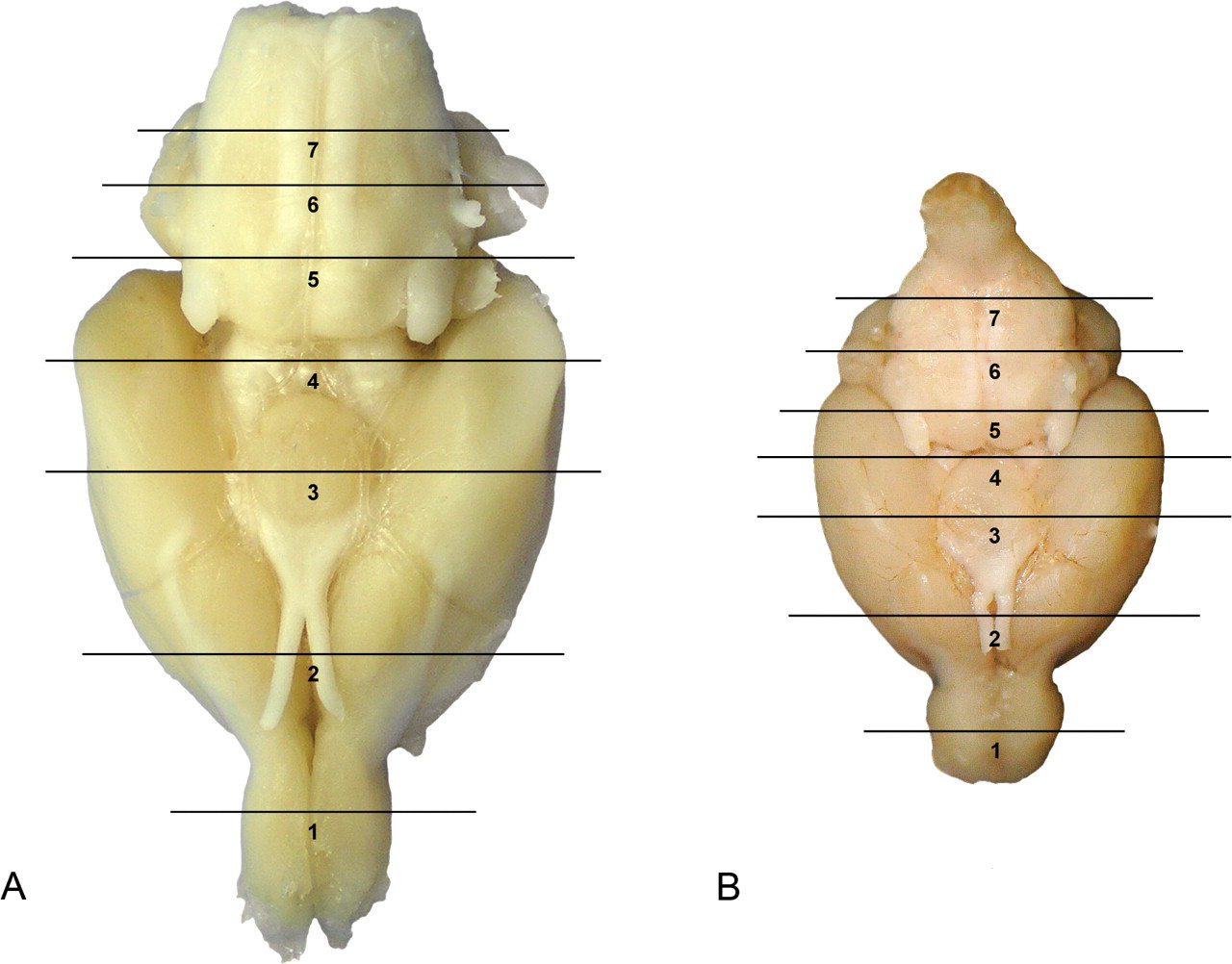
Ventral brain sectioning for the rat (a) and for the mouse (b). Numbers 1 to 7 indicate the level of sections. Levels 2, 3, and 6 comprise the traditional three brain sections. Sections added to the modified protocol include levels 1, 4, 5, and 7. (c) Rat: coronal sections corresponding to levels depicted in Figure 1a and Table 2. Levels 2, 3, and 6 comprise the traditional three brain sections. Sections added to the modified protocol include levels 1, 4, 5, and 7.
Neuropathology Evaluation for National Toxicology Program Rodent Studies a

a Standard procedures for neuropathology evaluations include immersion fixation with formalin and preparation of hematoxylin and eosin–stained slides.
b Modified sampling of the brain increased from 3 to 7 sections.
c Sampling based on the potential of a chemical to target the nervous system.
d Tibial nerve in the mouse will be processed and evaluated based on neuropathology findings in the mouse/rat sciatic nerve or rat tibial nerve.
Anatomic Regions and Some Corresponding Functional Significance a
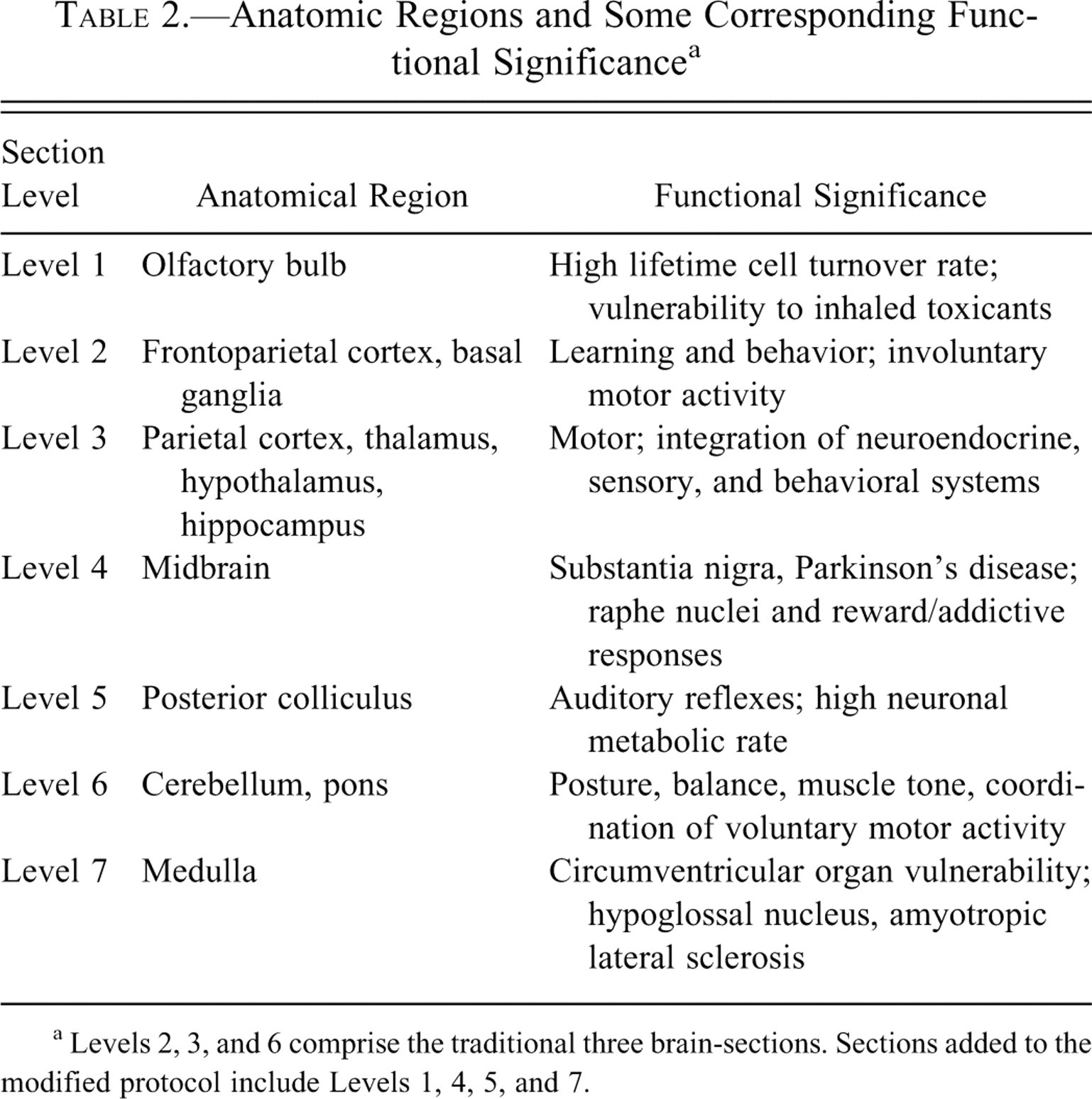
a Levels 2, 3, and 6 comprise the traditional three brain-sections. Sections added to the modified protocol include Levels 1, 4, 5, and 7.
The additional section through the anterior colliculus contains the substantia nigra, a site important to Parkinson’s disease in humans (Takahashi and Wakabayashi 2001). The NTP standard three brain sections do not contain the substantia nigra. With the increasing need to delineate the potential role of environmental factors associated with Parkinson’s disease in humans (Bronstein et al. 2009), it is important to examine this site during routine screening of chemicals, drugs, and biological agents.
The section through the posterior colliculus was specifically added because it is one of the most metabolically active areas in the brain. The posterior colliculus is an auditory reflex center associated with continuous auditory processing (Faingold, Gehlbach, and Caspary 1991; Webster 1995), with the largest microvascular blood volume (Cremer, Cunningham, and Seville 1983; Scremin 1985), highest rate of blood flow (Sakurada et al. 1978), and the highest rate of glucose metabolism (Sokoloff 1981; Sokoloff et al. 1977), rendering it particularly vulnerable to toxicants.
Finally, the section through the medulla was included to permit examination of the hypoglossal nucleus as well as cranial nerve nuclei linked to amytropic lateral sclerosis in humans. In addition, the area postrema, a circumventricular organ in the brain (lacking a complete blood brain barrier) that constitutes a site susceptible to blood-borne toxicants, will be evaluated in this section.
Although additional sectioning of the brain in routine toxicity studies is designed to improve the sensitivity of detecting nonneoplastic effects of toxicants on the nervous system, such an approach to brain sampling will also enhance the ability to detect brain tumors in 2-year carcinogenicity studies. Detection of brain tumors (gross and microscopic) can be difficult prefixation and postfixation for many reasons. For example, the lissencephalic nature of the rodent brain often obscures the primary subcortical location of many tumors. For example, in chronic inhalation studies of ethylene oxide in F344/N rats (Garman, Snellings, and Maronpot 1985, 1986), 23 primary brain tumors were detected by additional sectioning of the brain. Of these 23 tumors, only 2 were detected grossly and only 3 animals had clinical signs. In a review of more than 500 NTP studies, only one agent (glycidol) induced nonmeningeal brain tumors with the NTP classification of “clear evidence” of carcinogenic activity (male and female rats), while 9 other studies in rats and/or mice had slightly increased incidences of brain tumors considered as “equivocal evidence” of carcinogenic activity (Sills et al. 1999). The implementation of additional brain sectioning in the modified NTP protocol will enhance the ability to detect microscopic brain tumors.
In addition to the brain, evaluation of the spinal cord, peripheral nerves, and autonomic nervous system will be conducted based on the potential of a chemical or its metabolite to target the nervous system. Although changes in motor activity are robust indicators of agents that affect the nervous system and form an important component of a more complete assessment of the nervous system, other subtle and specific clinical signs that reflect impaired nervous system function must also be evaluated. For example, vitamin B6 toxicity demonstrates differential vulnerability within the spinal cord sensory pathways (Hoover and Carlton 1981; Krinke et al. 1981). Specifically, in the dog, afferents from the glabrous skin of the central pad conducting centrally via the dorsal columns are susceptible to vitamin B6 toxicity, while muscle and hair receptor afferents ascending in the dorsal spinocerebellar and spinocervical tracts are vitamin B6 toxicity resistant. Another example that demonstrates differential susceptibility is exposure to dibromoacetic acid in rats, in which the lesions are more severe in the lumbar region compared with the cervical region (Moser et al. 2004). In addition, extensive arborization of nerves from the brain and spinal cord innervating skeletal muscle and visceral organs of other systems underscore the need for examination of the peripheral and autonomic components of the nervous system. Numerous toxicants affect peripheral nerves, and a review of the literature indicates that limited preclinical screening procedures have resulted in significant subsequent neurotoxicity in humans. Examples include sensorimotor axonal peripheral neuropathy observed with 2′, 3′ dideoxycytidine, an AIDS therapeutic agent (Dalakas, Semino-Mora, and Leon-Monzon 2001), as well as taxane-induced neuropathy (Hilkens et al. 1996). Particularly, taxanes (anticancer agents) produce temporal and differential vulnerability between various components of the axon and cell body (Argyriou et al. 2008). Since animal studies confirm particular vulnerability of the distal portions of the peripheral nerves (Spencer 2000, p. 38), systematic evaluation of peripheral nerves should include both proximal and distal nerves. Hence, the modified NTP protocol includes sections of the proximally located sciatic nerve, the distally located tibial nerve, and the cranial nerve—the trigeminal components and its associated ganglion. Finally, the autonomic nervous system is vital for normal function of visceral organs of the cardiovascular, respiratory, gastrointestinal, and genitourinary systems. For example, although acrylamide is well known for its major peripheral neuropathic effects, other toxic effects of acrylamide on intestinal and urinary bladder function are associated with neuropathological changes within the somatosensory, somatomotor, and autonomic ganglia (LoPachin, Ross, and Lehning 2002), indicating that lesions in the autonomic nervous system have a direct effect on physiological processes in other systems. For this reason, specific evaluation of the autonomic ganglia will be included within existing sections of the colon.
Supporting Rationale
A number of factors support NTP’s position to modify the protocol for histopathologic evaluation of the nervous system in routine toxicity screening of chemicals, drugs, and biological agents. Although the majority of the chemicals investigated by the NTP in the past three decades include those with relevance to occupational and environmental exposures, the significance of effective screening and identification of potential neurotoxins and neurocarcinogens today is even greater because of the increasing prevalence of neurological diseases (Steenland et al. 2009; McCormack et al. 2002; Cavagnaro 2007) and the remarkable cost burden to the economy (Table 3 ). For example, a longitudinal study over a 20-year period (McCormack et al. 2002) by the California Department of Developmental Services documents doubling of mental retardation, a two-thirds increase in epilepsy, a three-quarter increase in cerebral palsy, and an unprecedented 12-fold increase in autism between 1987 and 2007 (Cavagnaro 2007). The rise in the incidence of autism dramatically exceeded the simultaneous increase in population growth in the longitudinal study by Cavagnaro (2009).
Estimated Prevalence of Neurological Disorders and Associated Costs to the U.S. Economy
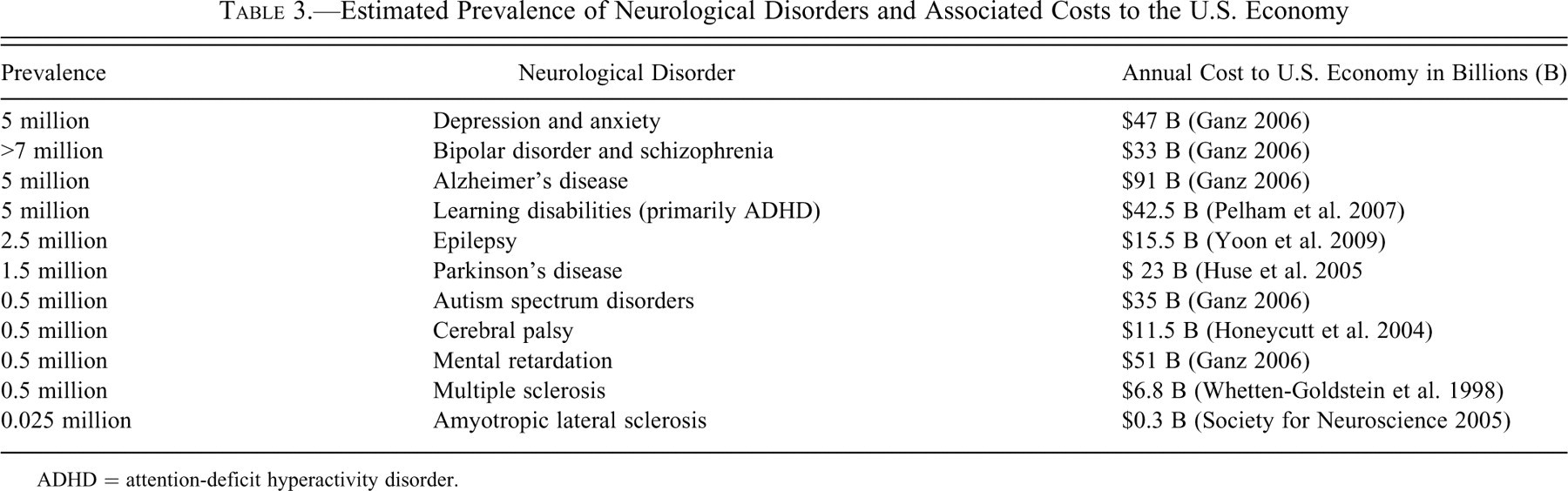
ADHD = attention-deficit hyperactivity disorder.
The role of unidentified environmental stressors in neurodegenerative disorders has been repeatedly implicated in several reports (Altevogt, Hanson, and Leshner 2008; Bronstein et al. 2009; Mayeux 2004). Less than 5% of disease incidence is due to a single gene mutation alone (Willett 2002). An expert committee at the U.S. National Research Council (NRC) concluded that 25% of developmental disabilities arise through interactions between environmental factors and individual genetic susceptibility (U.S. NRC 2000). For example, experimental studies show that transgenic mice (Richfield et al. 2002) have an enhanced vulnerability to pesticide exposures resulting in a Parkinson’s disease-like phenotype (Cory-Slechta et al. 2005), supporting the concept of the interaction between environmental factors and genetic aberrations in neurodegenerative disorders.
Neuropathological evaluation plays an important role in screening for environmental etiologies of neurodegenerative disorders, and histopathological criteria exist for a number of human neurological disorders including Alzheimer’s disease (Perl 2010), epilepsy (Blumcke 2009; Prayson 2010), Parkinson’s (Dickson et al. 2009; Tanner et al. 2011), cerebral palsy (Folkerth 2005), some forms of mental retardation (Armstrong 2002; von Deimling et al. 1995), multiple sclerosis (Stadelmann 2007), and amyotropic lateral sclerosis (Kato 2008). Similarly, neuropathological findings following chemical exposure have been reported in animals (Kato 2008; Woodruff-Pak 2008; Hattori and Sato 2007), and several animal models for chemically induced neurodegenerative diseases exist. Examples include rotenone, paraquat, maneb (Cicchetti, Drouin-Ouellet, and Gross 2009), MPTP (Smeyne and Jackson-Lewis 2005), and 6-hydroxy-dopamine (da Conceicao et al. 2010) for Parkinson’s disease; trimethyltin (Ishida et al. 1997) and pilocarpine (Curia et al. 2008) for epilepsy; cuprizone (Torkildsen et al. 2008) for multiple sclerosis; and 3-nitropropionic acid for Huntington’s disease (Ramaswamy, McBride, and Kordower 2007).
As a public health interagency government program, the NTP is committed to identifying chemicals that target the nervous system. This is critical because of the estimated 84,000 estimated chemicals in the United States registered for commercial use with the U.S. Environmental Protection Agency (2010), 3,000 to 4,000 are high production volume (HPV) chemicals (i.e., production exceeding 1 million pounds per annum). Sixty-seven percent of these HPV chemicals have never been tested for effects on the nervous system (Roe, Pease, and Silbergeld 1997). A reasonable attempt to identify the number of existing neurotoxic chemicals, based on a published report (Grandjean and Landrigan 2006), reveals that at least 1,000 chemicals are known to be neurotoxic in laboratory experimental studies and that 201 agents are known to be neurotoxic in humans (excluding drugs, food additives, microbial toxins, snake venoms, and biogenic toxins). Hence, the exponential increase in chemicals in the environment and lack of data on their neurotoxic effects underscore the importance of improving histopathological evaluation of the nervous system in routine NTP studies.
Furthermore, because assessment via structure-activity relationships (SAR) constitutes an important component of preliminary screening for toxicity, it is important to note that chemicals affecting the nervous system generally do not conform to the SAR paradigm for toxicity prediction (Levine and Butcher 1990). Several examples exist in the literature, including acrylamide, diethylbenzene, and acetylpyridine. Specifically, although acrylamide monomer is toxic, polymers are not (Spencer 2000); 1,2-diethylbenzene is toxic while 1,3-diethylbenzene is not (Gagnaire et al. 1991; Sabri et al. 2007); and 3-acetylpyridine is toxic (Anderson and Flumerfelt 1980) while 2-acetylpyridine and 4-acetylpyridine are not. These examples demonstrate that even small changes in the position of the chemical groups within the chemical structure of an agent or drug can result in adverse changes in the nervous system. Moreover, even derivatives of the same compound can damage different locations with different effects. For example, MPTP destroys cells in the ventral tegmental area and substantia nigra (dopaminergic system), while 2-NH2-MPTP destroys cells in the dorsal raphe (serotonergic system). Thus, because predictive criteria with SAR is limited for evaluating neurotoxicants, it is prudent to expand other endpoints, such as histopathology, toward a more comprehensive and systematic manner to increase the sensitivity of capturing neurotoxicants within toxicity/carcinogenicity screening programs.
The nervous system is unusually complex, and unlike most organs, the brain and spinal cord encompass hundreds of distinct cell populations, most of which have been delineated to be functionally significant via lesion pathway studies (Carlson et al. 2000; Colman et al. 2005). In fact, distinct cell populations within anatomic subsites interact to form specific functional neuronal circuits within the brain and spinal cord (Paxinos 1995). Lesions identified in one anatomic subsite may also be manifested at other sites within a specific pathway or circuit (Garman 2003). For example, lesions resulting from exposure to carbonyl sulfide (COS) were initially identified in the posterior colliculus (not a site within the standard three-section approach). However, follow-up studies by the NTP clearly demonstrated that COS also affects the anterior olivary nucleus, nucleus of the lateral lemniscus, medial geniculate nucleus, and the cortex, all anatomic subsites that link to the central auditory pathways (Sills et al. 2004; Morgan et al. 2004). Another example is p-chloramphetamine, which destroys cells in the raphe nuclei, a small distinct serotonergic cell population within the midbrain. Injury at this site can have far-reaching consequences via serotonergic circuits that project throughout the brain via numerous pathways (Mamounas et al. 1991). Hence, minimal pathologic lesions confined to small anatomic subsites warrant a more detailed evaluation since it is likely to be amplified via expansive neuronal circuitry in the brain (Sills et al. 2004). Additional sectioning of the brain will allow for an increased number of anatomic subsites to be routinely evaluated during toxicity/carcinogenicity screening of compounds.
Conclusion
Identification of neurotoxicants by comprehensive neuropathology evaluations is a vital component of routine toxicity/carcinogenicity screening programs. The focus of this article is limited to the rationale for modification of the histopathological endpoints alone for assessing neurotoxicity/neurocarcinogenicity in routinely conducted studies within the NTP. A thorough, truly comprehensive histopathology evaluation of the brain should ideally involve serial sectioning to ensure that all anatomic subsites are examined (Garman, personal communication). Although this approach is impractical for routine screening studies, at the minimum it is still important to include areas of unique susceptibility as outlined above and summarized in Table 2. Although the modified protocol will involve additional time and resources, the advantage of adopting a more extensive approach to histopathological evaluation of the nervous system is prudent in the current environment of increasing occurrence of neurological disorders (Steenland et al. 2009: McCormack et al. 2002; Cavagnaro 2007), unidentified environmental stressors associated with neurodegenerative disorders (Altevogt, Hanson, and Leshner 2008; Bronstein et al. 2009; Mayeux 2004), and exponential increase in the use of chemicals having unknown effects on the nervous system (Grandjean and Landrigan 2006).
Footnotes
Acknowledgments
The authors thank fellow NTP colleagues Drs. Jean Harry, Joseph Roycroft, and Raj Chhabra, and Dr. James P. Morrison of PAI, Charles River, for their invaluable input during the development of this modified protocol, and Drs. Gordon Flake and Mark Cesta for their detailed internal review of this article. Also, the authors are deeply grateful for the comments, opinions, and advice from many toxicologic pathologists with experience and interest in neuropathology who have all generously contributed throughout the process of establishing a modified NTP approach for evaluating the nervous system. We thank Mr. David Sabio (digital imaging), Ms. Stephanie Holmgren (library information services), Ms. Beth Mahler and the National Institute of Environmental Health Sciences histopathology core laboratory, and Mr. Shean Chiou (software) for their technical expertise and assistance during the preparation of this article. This research was supported (in part) by the Intramural Research Program of the National Institutes of Health, National Institute of Environmental Health Sciences.
