Abstract
For many pathologists, neuropathology is intimidating. Practical approaches for nervous tissue histologic evaluations to meet both routine and advanced study designs can lead to rewarding neuropathology efforts. Cost-effective, high-quality histologic evaluations can occur if animals are exsanguinated quickly, brains removed carefully to maintain structural integrity and avoid dark neuron artifact, immersion-fixed quickly and thoroughly, and trimmed and processed to consistently survey multiple areas. While brightfield examination of H&E-stained sections is generally sufficient for survey evaluations, epifluorescent assessment of neuronal autofluorescence facilitates recognition of neurodegeneration in H&E-stained sections. Fluoro-Jade B or specialized immunohistochemical stains may be required to answer specific questions. Evaluations require that both technical staff and pathologists have a working knowledge of a few easily identified neuroanatomic landmarks and familiarity with use of a detailed brain atlas. At least four coronal sections should be routinely surveyed from young adult rats, with evaluation of comparable areas in other laboratory animal species. This number should be at least doubled if there is reason to suspect morphologic changes in the CNS. This article focuses on technical details of efficient specimen preparation for neuropathologic evaluations involving relatively large numbers of rodents, as well as a practical approach to basic neuroanatomic site identification.
Keywords
Introduction
In this issue, Garman provides details on recognizing specific cell types, both normal and abnormal, as well as some of the artifacts encountered commonly within the CNS (Garman 2011). Some details of CNS tissue collection and interpretation may be found in Fix and Garman (2000) and in Dorman, Brenneman, and Bolon (2002). The current authors previously provided details of proper brain collection for histology, focused on the avoidance and recognition of artifacts, and suggestions for a simplified approach to an understanding of neuroanatomy (Jordan et al. 2007; Jordan et al. forthcoming). The intent of this article is to complement these publications by focusing pathologists and technicians on methods critical for production of the high-quality histologic sections necessary for successful evaluation of CNS tissues in routine toxicology studies, especially studies involving rodents, and to provide additional insights into a simplified approach to neuroanatomy.
Cost-effective, high-quality neuropathologic evaluations require teamwork involving the recording and assessment of detailed clinical signs and gross postmortem observations, careful and timely collection and processing of tissues, and a working knowledge of both neuroanatomy and neuropathology. The process begins well before the necropsy. It is important for the pathologist to have knowledge of the likely target effects (distribution, biochemistry, secondary changes) of the compound so that tissue collections and evaluations can be optimized. Veterinary and animal care staff can provide important clues that nervous system toxicity may exist by noting and recording changes in behavior and other clinical signs. Special techniques such as the functional tests and electrophysiology evaluations outlined by Virginia Moser and Joseph Arezzo (Moser 2011; Arezzo, Litwak, and Zotova 2011) in this issue provide additional knowledge to focus the evaluation. The pathologist and toxicologist can then determine whether routine collection and evaluation procedures are appropriate or if special procedures are warranted.
Tissue Collection and Processing
Since few projects have unlimited resources, the researcher must first determine the appropriate level of detail required for the pathology evaluation. For rapid throughput studies, such as for early toxicity screening, a parasagittal section of brain (longitudinal section lateral to the center line) from each rat, combined with a hemi-coronal section from the contralateral one-half brain, will allow the pathologist to survey many parts of the brain. When the specific CNS targets are unknown, at a minimum commonly targeted sites (e.g., cerebral cortex, hippocampus, cerebellum) should be evaluated. Collection of the hemi-coronal section at the level of the hippocampus (through the widest part of the brain) will help to ensure that amygdaloid nuclei and other common CNS target sites that may be missed in the sagittal section will be sampled. For routine standard toxicology studies with no known CNS liability, these authors recommend histopathologic evaluation of four coronal sections (Figure 1 ) of brain as a routine practice in subsequent studies during the development of that compound. While three sections have been used for many years as the standard in routine rodent toxicology studies, the authors have reviewed studies employing the 3-section trimming paradigm in which the hippocampus, a common primary site of neurotoxicity, was rarely sampled. While some may argue that attention to detail by the technical staff and study pathologist should prevent this deficiency, an alternative 4-section brain trim leaves less opportunity for missing these important portions of the brain. The 4-section trim can be done freehand by trained histotechnicians who recognize the ventral brain landmarks (i.e., a trimming mold is not required), and the four sections can be placed together into a single paraffin block. Therefore, an additional 33% of the brain can be evaluated at almost no additional processing cost. The sections can be most efficiently evaluated by the pathologist if they are arranged in anatomic (rostral-to-caudal) order in a single row on the histologic slide rather than in a 2 × 2 arrangement. An alternative approach would be to provide a combination of a single parasagittal section with four coronal hemisections. Since brain lesions are usually bilateral, even four coronal hemisections would more consistently survey more sites in the brain than are currently evaluated in 3-section trims.
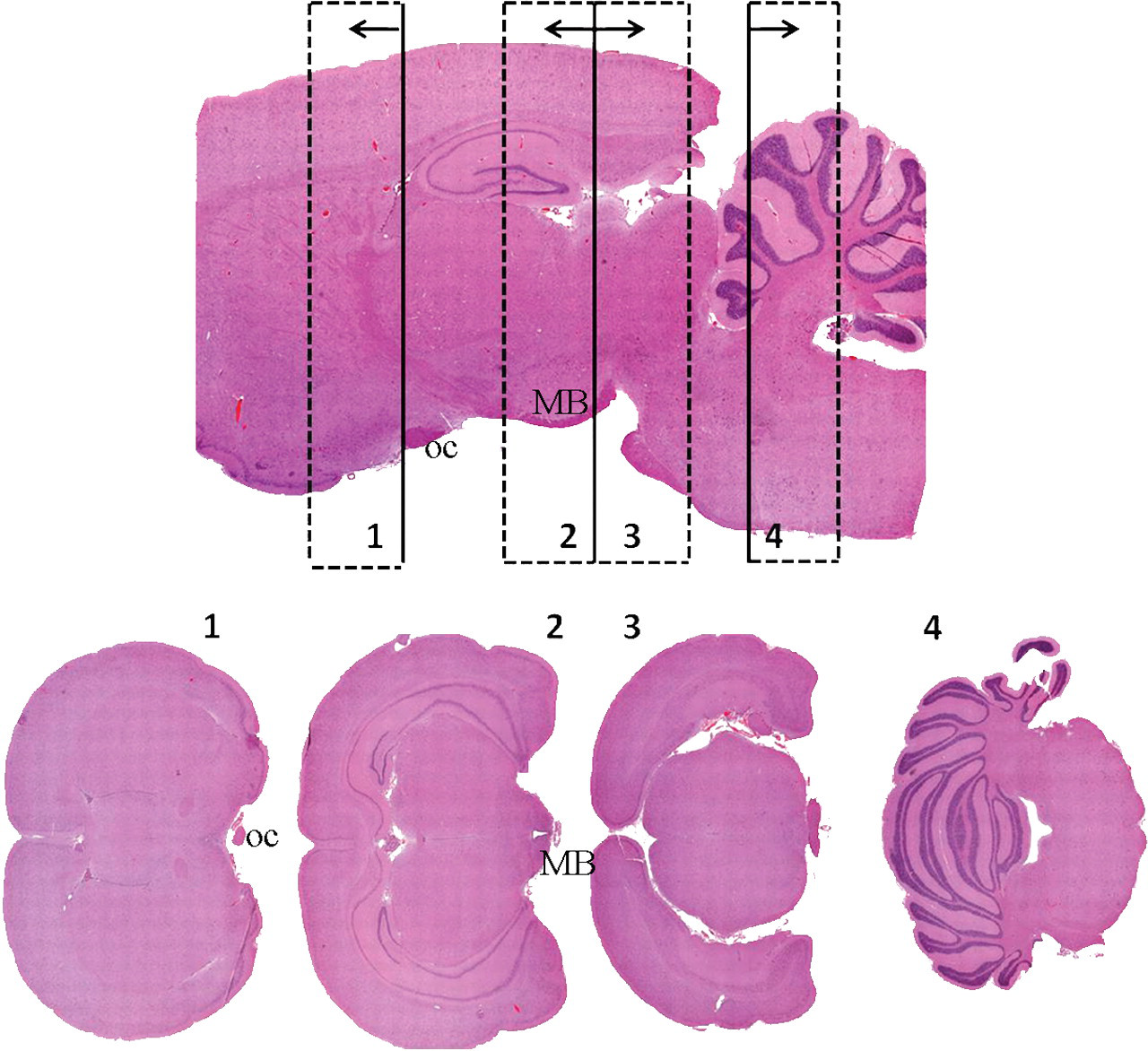
Rat brain, 4-section trimming. Trim levels are indicated on the image of the parasagittal section, and examples of the four resulting coronal histologic sections are depicted. The base of each arrow indicates the embedded surface to become the block face. Note that sections 2 and 3 face each other. Since brain size varies with age, gender, health, etc., ventral landmarks such as the optic chiasm (oc) and mammillary bodies (MB) can be used to help obtain consistent neuroanatomic areas in the histologic sections.
If the compound is known or suspected to be neurotoxic and the specific CNS target sites are uncertain, additional sectioning planes (brain slices) should be evaluated, and perfusion fixation should be considered. Perfusion fixation may increase the ability of the pathologist to detect subtle changes; however, appropriate collection and processing will allow the pathologist to find most lesions detectable by light microscopy following immersion fixation. Slicing the rodent brain at 2 to 3 mm intervals to provide eight to eleven sections from a rat (six to eight sections for a mouse), which can be placed on two to three histology slides, will allow a broad survey of many subanatomic regions in the brain. For most technicians, a brain trimming matrix will be necessary to accurately and consistently trim slices this thin. Tissue-marking dye placed on the surface to be embedded away from the block surface will facilitate consistent orientation of the sections into the paraffin block. Although the authors are unaware of studies to prove this, they expect that small differences in trimming and microtoming would result in the presentation of a broad variety of Bregma levels around the trimming levels to ensure sampling of all but the smallest of subanatomic structures collectively from within each dose group if a sufficient number of animals are evaluated (i.e., ten per sex per group). Primary evaluation and/or peer review of the study by a pathologist with special expertise in neuropathology helps to ensure that small changes are not missed and that background and artifactual changes, which may include vacuoles, pigments, and dark neurons, are not overinterpreted as compound-related changes.
Several important considerations require little, if any, extra effort but can significantly enhance the quality of neuropathologic evaluations in laboratory animals. Examples include rapid and complete exsanguination, careful extraction of the brain from the cranium, gentle manipulation of the brain during and after removal to avoid dark neurons, and attention to histology procedures. The authors have noted improved quality of tissue preservation from rodents perfused with physiologic saline (performed to remove blood from tissues as required for biochemical evaluations) prior to immersion in common aldehyde fixatives, when compared with similarly immersion-fixed tissues from routinely exsanguinated animals. This agrees with Grizzle, Fredenburgh, and Myers (2008), who indicate that proteins, especially those proteins in blood, inactivate fixatives. As will be demonstrated later, both erythrocytes and degenerating neurons are autofluorescent. Since epifluorescence is an excellent tool for finding degenerative neurons, minimizing the number of autofluorescent erythrocytes in a section will make the task of finding autofluorescent degenerative neurons easier. To avoid hypostatic congestion in multiple tissues and to improve the quality of exsanguination, animals should be anesthetized as closely to the time of euthanasia as is practical, and the abdominal vena cava should be severed as quickly as practical while the animal is still in a surgical plane of anesthesia.
The brain should be removed and handled carefully to avoid dark neurons, an artifact that results when pressure is applied to the unfixed brain and that must be differentiated from degenerative neurons (Jortner 2006). A description and excellent examples of dark neurons are presented by Garman in the current issue of this journal (Garman 2011). To avoid dark neurons, instruments used to open the cranium should never impact the brain. The dura mater must be carefully removed prior to any attempts to remove the brain, and cranial nerves should be severed with a sharp blade, not pulled from their attachments. The pituitary may be separated from the brain at the same time with the same sharp instrument. In rats and mice, sharp curved scissors with rounded tips work well to both detach the brain and to scoop it up to be weighed or moved into the fixative. Similarly, brains should not be picked up with forceps or fingers, as either will produce increased pressure and subsequently dark neurons. Attention to careful tissue handling to avoid dark neuron formation should continue through organ weighing and tissue reconciliation steps of the necropsy.
Vacuoles are commonly found as artifacts within the CNS. Common causes include autolysis, excessive concentrations of alcohol, excessive time in otherwise safe concentrations of alcohol, and freezing. Autolysis begins immediately upon death and can result in artifacts resembling neurodegeneration. Li, Ryan, and Ochoa (2003) demonstrated up to 39% increases in absolute liver weights when rats were euthanized by carbon dioxide (C02) and held in the CO2 chambers for twenty-five minutes following cessation of respiration as compared with livers of rats necropsied immediately upon death. Increased centrilobular vacuoles were filled with material consistent with plasma. Similar changes may occur in the brain. The authors have noted vacuolation of thalamic neurons to be one of the earliest markers of a prolonged interval to fixation, perhaps because these nuclei are some of the last to be reached by the formalin following immersion fixation. When brain is a known target, rats can be exsanguinated and brains removed and placed into formalin within two minutes by trained prosectors. Even five to six minutes from euthanasia to formalin immersion will result in very good quality tissue preservation.
During processing, tissues should not be exposed to alcohol concentrations ≥ 70% for prolonged periods. High concentrations of alcohol may lead to artifactual vacuolation, especially of white matter (Wells and Wells 1989; Garman 2011); however, normal alcohol dehydration in histology processors will not produce undue changes. Altered weekend routines during which tissues are maintained for many hours in alcohol prior to commencing the processing sequence are a common source for alcohol-induced vacuolar changes. While pathologists are well aware of the effects of freezing, new technical staff may not always understand the difference between preserving a carcass in the refrigerator and placing it in the freezer. With more tissue being shipped to contract research organizations for processing, the opportunities for tissues being in a truck or on a shipping dock where they can freeze also increases. The importance of avoiding freezing must be regularly shared with those responsible for collecting and shipping tissues.
Pressure artifacts may also be introduced by shipping and handling. Vacuum packing for shipping reduces the chances for accidental drying, but excessive vacuum negative pressure can also significantly alter tissue morphology, often manifesting as pressure artifacts. When shipping or storing, avoid placing the brains within fixative containers that also contain tissue cassettes. Pressure and/or abrasion by the cassettes can remove the surface of the brain and produce indentations in the brain closely resembling forceps marks (Figure 2 ). Handling-induced surface artifacts produced during necropsy can be differentiated from those induced in fixed material during shipping and trimming. Dark neurons and/or free erythrocytes usually accompany those artifacts produced prior to fixation, while neither dark neurons nor free erythrocytes accompany tissue damage that occurs after fixation is complete. Forty-eight or more hours are generally required for sufficiently complete fixation, whether by infusion or immersion, to avoid handling-induced dark neurons. For this same reason, perfusion-fixed brains should remain within the intact (unopened) cranium that is immersed in fixative for an additional forty-eight hours. Even with perfusion fixation, the authors prefer to immerse perfusion-fixed brains in aldehyde fixative for an additional forty-eight hours after removal before trimming for histological evaluation.
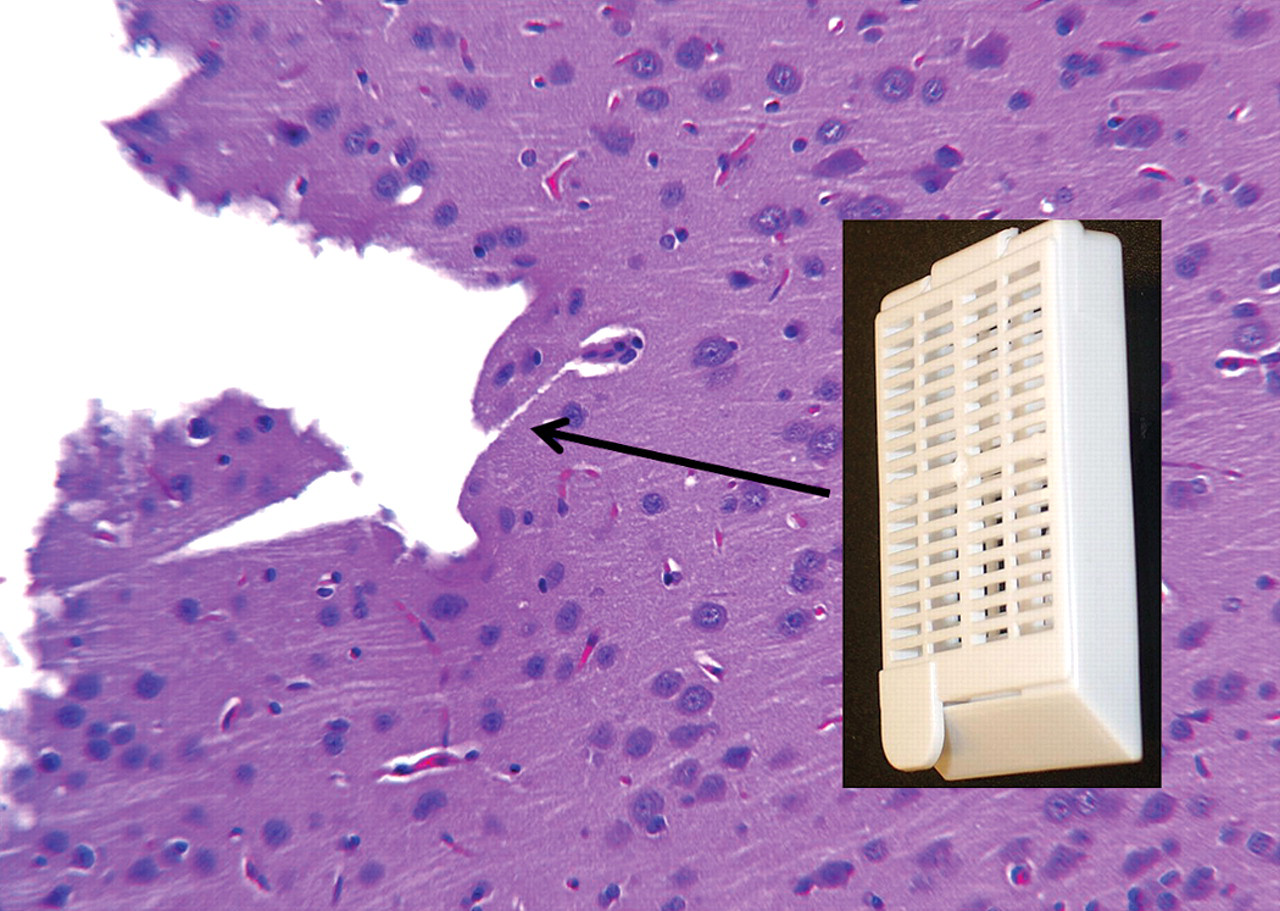
Loss of superficial cortex and indentations with square corners caused by packaging of fixed brain tightly against a tissue cassette. The absence of dark neurons indicates that this was a postfixation artifact.
While brain sections are sometimes microtomed at a thickness of 10 µm or more, most laboratories microtome brains at the same 3 to 6 µm thickness used for other tissues. If brain sections, especially the thinner ones, tend to roll up or shatter on the water after removal from the microtome, the distilled water can be replaced with water containing minerals. Clean tap water is often satisfactory, as is bottled “spring” drinking water. Water bath temperatures of 38–40°C provide a good starting point.
For increased efficiency in the collection of spinal cords, Meikle and Martin (1981) described the following hydraulic extraction method. For rodents, use a 6 or 12 ml syringe filled with tap water and attached to a short blunt needle of diameter that fits snugly into the lumbar spinal canal (eighteen to twenty-two gauge). Completely remove the head by disarticulating at the atlanto-occipital joint or by cleanly severing the spine at one of the cranial cervical intervertebral discs, then cleanly sever the spine at an intervertebral disc in the middle to caudal lumbar region. Insert the needle to the hub in the lumbar canal, then give two to three gentle pushes on the plunger followed by a very firm and rapid push. The spinal cord should pop out onto the working surface intact (Figure 3 ) and with very few histologic artifacts. One disadvantage to this approach is that the spinal nerves and ganglia will no longer be attached to the spinal cord. Note that this method will only work well on animals that have been dead for no more than a very few hours, as autolysis will cause too much tissue softening for this procedure. Because of the differentials in spinal canal diameter, the cord should not be forced rostral to caudal. The cords should not be extracted with formalin because of the potential for a splash hazard. This approach has also been successfully adapted to much larger animals.
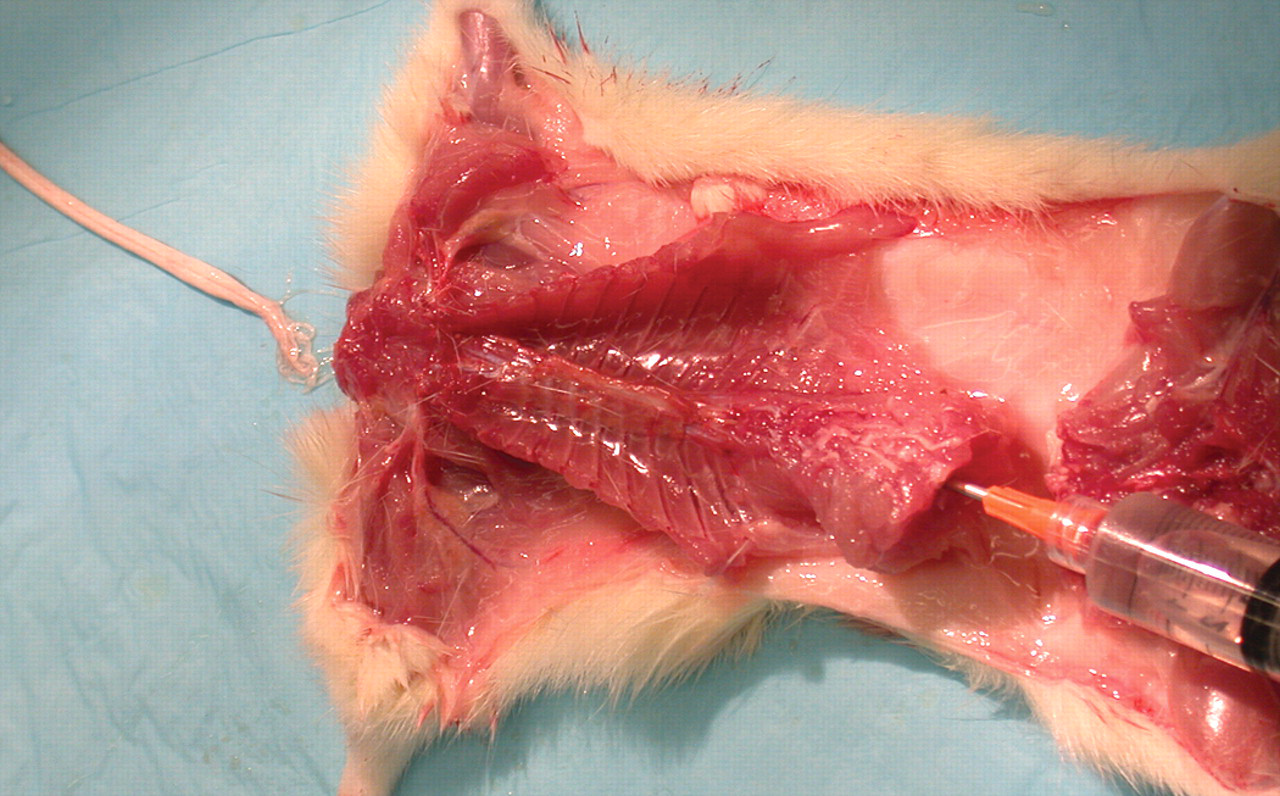
Hydraulic extraction of spinal cord. A rapid push on the plunger of a water-filled syringe attached to a blunt needle inserted into mid–lumbar canal results in rapid extraction of spinal cord. Photo courtesy of Experimur, Chicago, IL.
Special Procedures and Stains
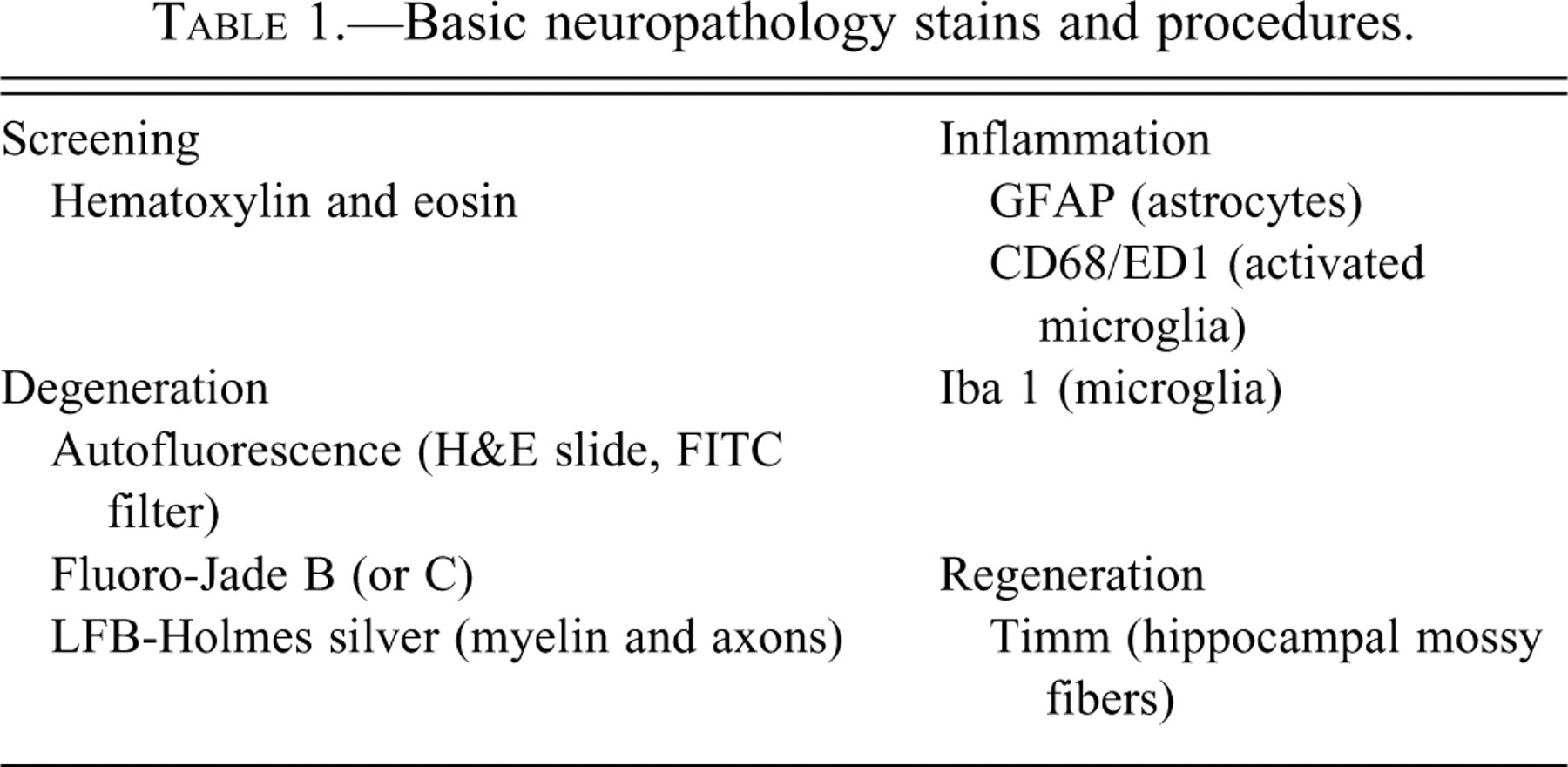
While the goal is to find any abnormality, evaluations of the CNS are generally focused on locating degenerative, inflammatory, and sometimes regenerative changes. While not recommended for routine screening, selected sample stains and procedures to enhance the recognition of these changes, when present, are presented in Table 1 , and examples of some are presented by Garman (2011). Critical assessments can usually be conducted on well-preserved H&E-stained tissues, but special stains may be needed to characterize specific cellular responses or to aid in the identification of no-observed-effect levels for difficult to distinguish changes. A valuable tool for recognition of neuronal degeneration in CNS evaluations is a fluorescent microscope with a fluorescein isothiocyanate (FITC) blue excitation filter (approximately 450 to 490 nm). Many high-quality cubes have narrow band pass filters designed to block emission wavelengths outside of the window characteristic for selected fluorochrome labels, thereby reducing background emissions. However, in the authors' experience, emission filters that allow passage of a somewhat broader band of wavelengths within this portion of the spectrum (e.g., a long pass filter allowing wavelengths ≥ 515 nm, such as the Leica I3 cube [Leica Microsystems, Deerfield, IL]) enhance the sensitivity for discriminating autofluorescent signals that are related to neurodegeneration in H&E-stained slides. As demonstrated in Figures 4 and 5 , autofluorescence is useful for detecting both degenerative neuronal soma and degenerative neuronal processes. Increased sensitivity to detection of neurodegeneration, particularly of dendrites and axons, is provided by the Fluoro Jade B stain (Schmued and Hopkins 2000).
Basic neuropathology stains and procedures.
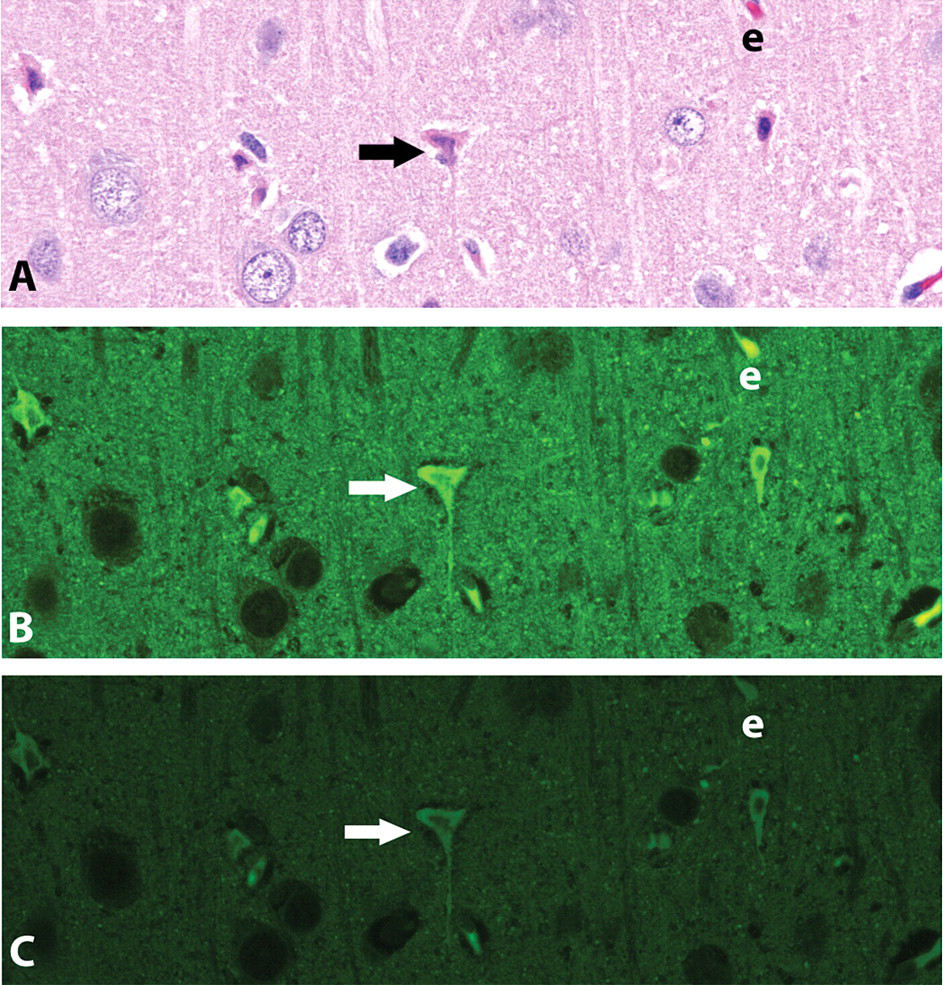
Cingulate cortex of a rat given MK801 (a noncompetitive antagonist of the N-methyl-d-aspartate [NMDA] receptor). A. H&E under brightfield illumination. B. H&E under epifluorescent illumination, Leica I3, a long band pass filter cube. C. H&E under epifluorescent illumination, Leica L5 (narrow band pass) filter cube. Several degenerative and autofluorescent neurons, including the one identified at the arrowhead, are present; an erythrocyte (e) is also autofluorescent. Note that the intensity of autofluorescence is greater in the image obtained using the longer band pass I3 emission filter.
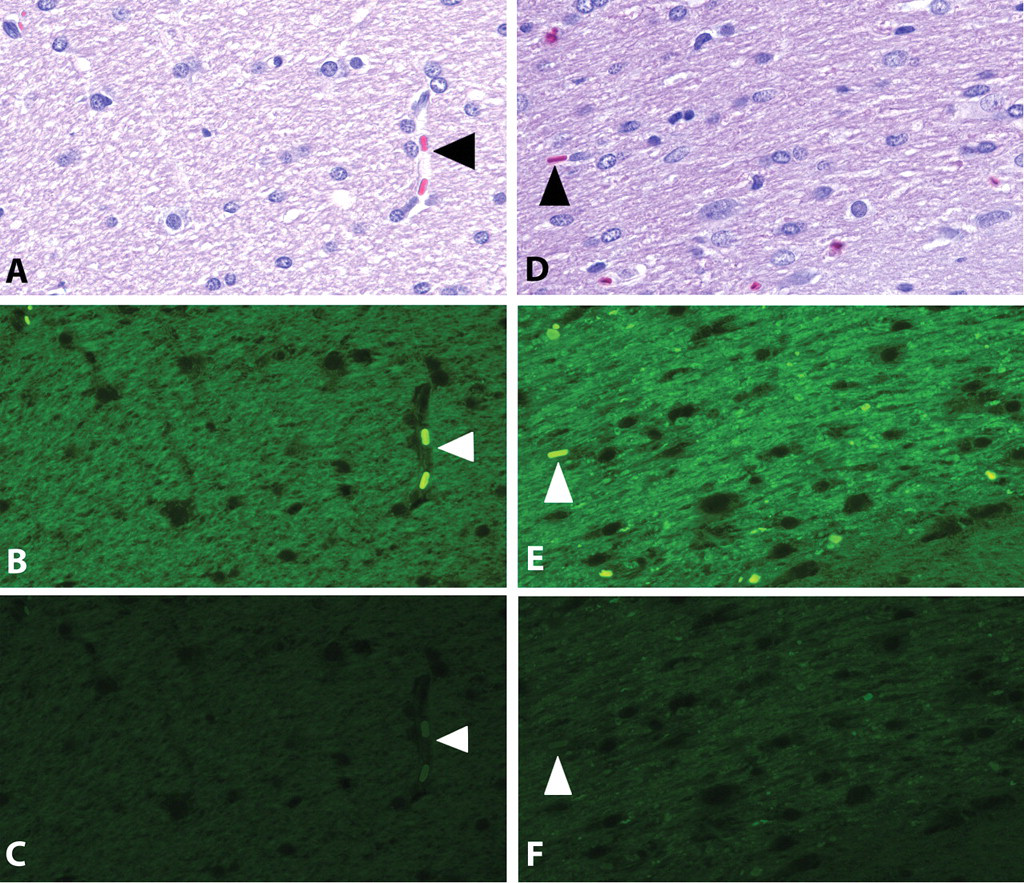
Optic tract of an F344 rat comparing normal structural integrity (A–C) with an atrophic/degenerative response (D–F). A & D: H&E under brightfield illumination. B & E. H&E under epifluorescent illumination, Leica I3 (wide band pass) filter cube. C & F. H&E under epifluorescent illumination, Leica L5 (narrow band pass) filter cube. Arrowheads indicate erythrocytes within capillary lumina. Note that the intensity of autofluorescence is greater in the image obtained using the longer band pass I3 emission filter.
Following are a few practical alternatives to be considered for neuropathology evaluations. If a fluorescent microscope is not available, amino cupric silver stains can be used to identify neurodegeneration, but they require frozen sections and are technically more demanding. Inflammatory reactions in the CNS can be evaluated with multiple histological tools. Immunohistochemical detection of glial fibrillary acidic protein (GFAP) usually labels much of the brain because it recognizes both resting and activated astrocytes, but it is useful in evaluating shifting patterns of astrocyte activity. Antibodies for CD68 and ionized calcium binding adaptor molecule 1 (Iba1) are used frequently for assessing phagocytic and activated microglia (Ito et al. 2001; Kullberg, Aldskogius, and Ulfhake 2001). In special situations (i.e., epileptogenesis), aberrant patterns of hippocampal mossy fiber sprouting can be assessed with the modified Timm’s stain, which allows visualization of zinc-containing glutamatergic synaptic vesicles (Sloviter 1992). However, Timm’s staining cannot be applied to routinely fixed tissues since this method requires addition of sodium sulfide or soldium selenite to the perfusion or immersion fixative to convert soluble synaptic zinc into insoluble complexes that can form the black precipitate in the subsequent silver development staining reaction.
Neuroanatomy Simplified
In addition to being able to detect the unique features of degeneration and inflammation in the brain, a working knowledge of neuroanatomy is important to finding and interpreting changes in the CNS. Recognition of both minimal increases in cellularity (e.g., those caused by glial cell responses to injuries) and decreases in cellularity (e.g., loss of specific neuronal cell populations) as well as other subtle changes can only be appreciated if the investigator recognizes the specific subanatomic site being evaluated and the range of normal features for that location. Even with a good brain atlas, the process of actually matching slides on the microscope with sites depicted in the atlas can be challenging. Since histologic sections rarely match the exact plane of section depicted in an atlas, the pathologist must have a basic working knowledge of neuroanatomy and the ability to envision how the landmarks will be shifted when sections are tilted or skewed from the true coronal planes typically presented in the atlases. A series of atlases authored/coauthored by Paxinos and Watson provides good neuroanatomic references for rodents. These include the fifth edition featuring the rat brain in coronal section (Paxinos and Watson 2005), which is available in both hard copy and as a compact disc. Atlases are also available for the nonhuman primate (Saleem and Logothetis 2007; Paxinos et al. 2009), but the reader is cautioned that there are some differences in the relationships of the neuroanatomic sites between the Rhesus monkey (Macaca mulatta) used for these atlases and the smaller Cynomolgus monkey (Macaca fascicularis) currently used most often for toxicology studies. The few comprehensive atlases of the canine brain that were formerly available are now out of print.
When lesions are encountered in the brain, few, if any, pathologists can readily name all of the structures affected. Most will require the aid of an atlas to properly map the lesions. Familiarity with a few key structures will make identification of lesion location much easier and will help to ensure that changes involving loss of structures are less likely to be overlooked. The following outline, based on less than thirty neuroanatomic features, was prepared as an introduction to using a rat brain atlas (Paxinos and Watson 2005). Although the sizes and relationships vary, attention to the same structures will also prove useful in gaining deeper understanding of neuroanatomy for other species. A hard copy atlas will work, but the ability to quickly migrate caudally and rostrally through the figures makes the task easier with an electronic version. The reader is encouraged to select an individual structure in the outline and flip through the atlas plates to gain an appreciation of the changing shapes of that structure and the relationship of that structure to others in the same plane of section. While this outline is based on coronal sections, it is expected that the three-dimensional vision created in the reader’s mind will readily translate to evaluation in sagittal, horizontal, and the many slightly oblique planes of section commonly encountered in routine toxicology studies.
Bregma refers to the junction of the coronal and sagittal sutures on the skull, and a coronal brain section at this level corresponds to a Bregma level of 0.0 mm. Bregma coordinates represent the number of millimeters rostral (positive numbers) or caudal (negative numbers) to this plane. Ranges given in the outline are based on readily identifiable structures, not necessarily on the full range of each structure.
It is important to note that the olfactory lobe extends rostral and ventral to the cerebral cortex and is demarcated laterally by the rhinal fissure as it joins the cerebral cortex. The rhinal fissure extends the length of the ventrolateral cerebral cortex separating the dorsally located neopallium from the phylogenetically more primitive rhinencephalon. Confusion with the more ventrally located amygdaloid notch can be avoided by noting that the distinctive dense, sometimes serpentine, nuclear layer of the piriform cortex is always ventral to the rhinal fissure. It is recommended that the initial review follow a rostral to caudal pattern. In the most rostral portions of the brain, changes in the profile of the corpus callosum are particularly helpful in neuroanatomical localization. The distinctive shapes of the forceps minor in the rostral corpus callosum and the lateral extension of the corpus callosum into the external capsule are distinctive. Also, the shape of the hippocampus, at both the midline and in lateral and ventral regions, is very useful in locating neuroanatomic sites over nearly one-fourth of the brain, Bregma (B) –1.7 to B –6.7.
Key structures to recognize when using a rat brain atlas (abbreviations correspond to those used in Paxinos and Watson 2005): Bregma 6.6 through 2.2
Shape of olfactory lobe Interface with frontal lobe Formation and depth of rhinal fissure Shape and distance from midline of forceps minor of corpus callosum (fmi) Bregma 2.2 through 1.2
Shape of corpus callosum (fmi) at midline Location of anterior (rostral) commissure (aca) relative to lateral ventricle Bregma 1.2 through –1.6
Shape of midline structures, especially septal nuclei and lateral ventricles Location of anterior (rostral) commissure (aca) relative to bottom of lateral ventricles Shape of aca, B –0.1 through –0.5 Shape of third ventricle, position of fornix, and shape of optic tract, B –0.26 through –1.6 Bregma –1.6 through –4.8
Shape of hippocampus Location of third ventricle Shape of ventral portions of brain and location of optic tract Note relationships of entorhinal and piriform cortices and amygdaloid nuclei to each other and the brain as a whole Bregma –4.5 through –6.3
Shape of midline and ventral hippocampus Appearance and disappearance of mammillary bodies, interpeduncular nuclei, and pontine nuclei Substantia nigra apparent, B –4.5 through –6.3 Medial geniculate nucleus looks like “ears”, B –5.2 through –6.5 Bregma –6.3 through –8.3
Pons is distinctive, B –6.7 through –7.6 Rostral (superior) colliculus is apparent, B –5.6 through –8.0 Caudal (inferior) colliculus is apparent, B –7.6 through –9.8 Compare shape of dorsal midbrain to structures on ventral portion (sections often tangential in this area) Pyramids begin to form at ventral portion of B –8.3, and pineal is often present dorsally Bregma –8.7 through –11.3
Aqueduct transition to 4th ventricle, B –8.7 through –8.8 Presence of cochlear nuclei, B –8.8 through –11.3 Shape and location of pyramid ventrally Rostral cerebellar folia, B –8.6 Use the cerebellar shape to ensure both dorsal and ventral portions of section are properly identified Shape/location of facial nerve and genu, B –10.0 through –10.6 Deep cerebellar nuclei (“roof nuclei,” collectively, the several nuclei within white matter at base of cerebellum) are distinctive, B –10.7 through –11.8 Bregma –11.4 through –13.3
Cerebellum separated from the brainstem Deep cerebellar nuclei present, B –10.7 through –11.8 Few distinguishing characteristics, B –11.96 through –12.8 Consider shape of cerebellum, brain stem, and pyramids Inferior olivary nuclei distinctive in ventral brain stem, B –12.2 through –14.2 (avoid confusing with more rostral and lateral superior olivary and facial nuclei) Hypoglossal nuclei distinct, B –12.8 through –14.6 Bregma –13.7. Rostral end of central canal of the spinal cord
With this basic understanding, the pathologist, when faced with structures in a histologic slide, can quickly create a decision tree to match the site with one or a few plates in the atlas. For instance, if it were of interest to identify the group of neurons at the arrow in Figure 6
, the reader would first recognize that the site is within the medulla oblongata and that the cerebellum is attached. Therefore, the location must be rostral to B –11.4. The absence of deep cerebellar nuclei indicates that the site is rostral to B –10.7, and the absence of the facial nerve means that the site is rostral to B –10.0. The presence of cochlear nucleus (VCA) suggests that it is within the B –8.8 to –11.3 range. Since the caudal end of that range was previously eliminated, the search of the atlas has been narrowed to only a few plates (B –8.8 through the plate rostral to B –10.0). Quick comparison of the histological section to these plates reveals a match at approximately B –9.7 based on the distinctive shapes of the fourth ventricle and the presence and location of the middle cerebellar peduncle (mcp). It is now possible to match locations to reveal that the neuron cluster of interest represents the locus coeruleus (lc), which is just medial to the mesencephalic trigeminal nucleus (Me5).
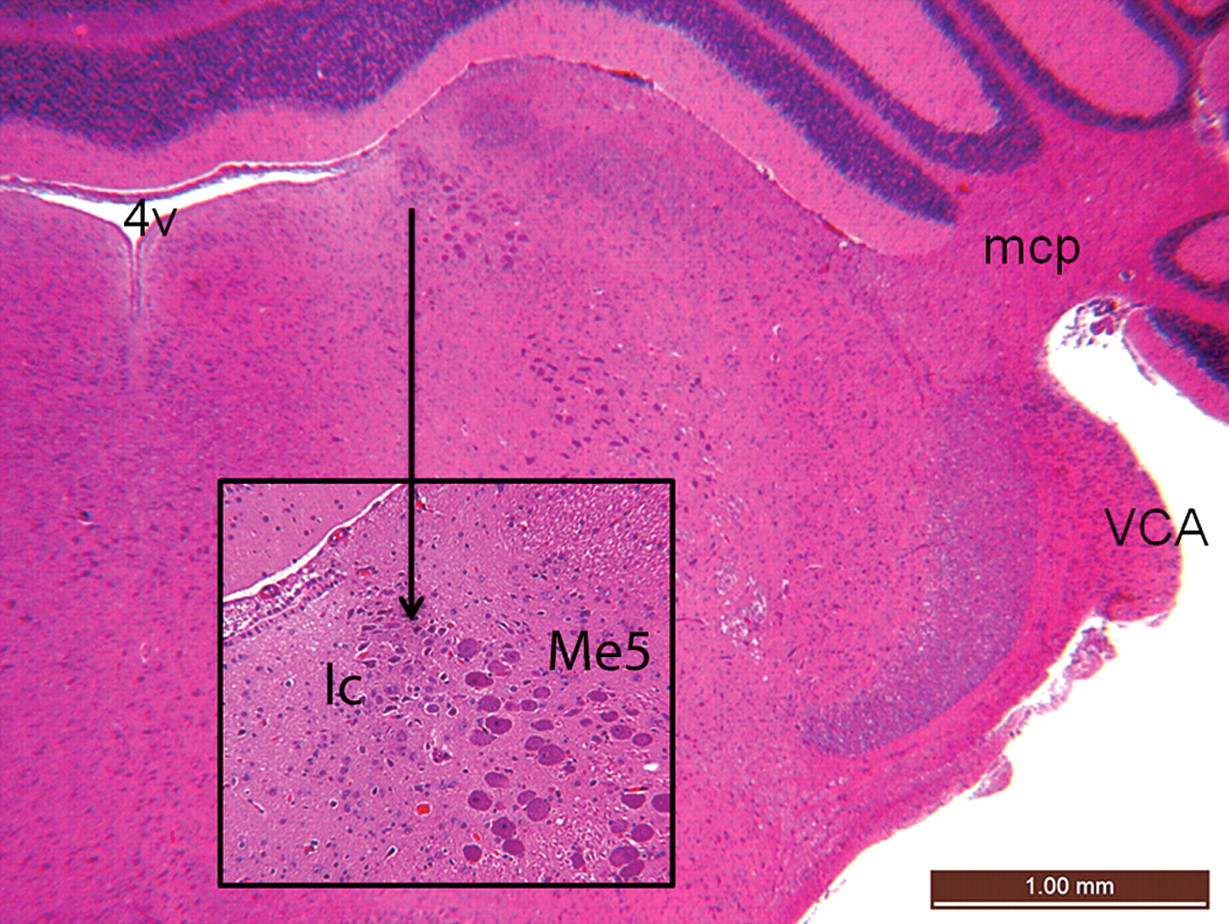
Coronal section of a normal rat brainstem. The inset represents increased magnification of area of interest. Compare to neuroanatomical structures outlined in the text. 4v = fourth ventricle; lc = locus coeruleus; mcp = middle cerebellar peduncle; Me5 = mesencephalic trigeminal nucleus; VCA = ventral cochlear nucleus, anterior (rostral) part. Features are consistent with labeled structures in a recognized atlas of rat brain anatomy (Paxinos and Watson 2005). H&E.
Summary
In summary, cost-effective evaluations of the CNS in toxicology studies begin with knowledge of compound information and clinical signs suggesting specific targeted areas to be evaluated. The brain should be collected following almost complete exsanguination and with minimal external pressure prior to and during fixation. Areas represented within at least four coronal sections provide a practical routine screen of the rodent brain. The pathologist must have the knowledge to recognize not only active inflammatory and degenerative lesions but also loss of neurons without apparent secondary changes. A fluorescent microscope is useful for identification of autofluorescent degenerating neurons either in H&E-stained slides or in Fluoro Jade B–stained sections when increased sensitivity is needed. Other stains are useful for evaluation of inflammatory, degenerative, and regenerative changes. It is hoped that the brief guide to the rodent atlas will facilitate these evaluations.
