Abstract
The objective was to determine the respiratory toxicity of MAP0004, orally inhaled dihydroergotamine (DHE), via inhalation for six months. Forty beagle dogs (twenty females, twenty males) were treated by nose-only inhalation for 182 days. Groups 2 through 5 received MAP0004 (mean doses: 0.045, 0.154, 0.44, 0.825 mg/kg); Group 1 received vehicle only. Groups 1 through 4 received single thirty-minute exposures, whereas Group 5 was exposed twice daily for thirty minutes. Toxicity was assessed from clinical observations, objective evaluations, and clinical and anatomical pathology. Systemic effects were scabbing of ear tips in Groups 3, 4, and 5 and excessive salivation and emesis, observed in Group 5. No changes were observed in the lungs in any dose group. Minimal treatment-related microscopic changes were observed in the respiratory nasal epithelium only in Group 5. No plexiform, vascular media, or fibroproliferative changes in any heart valves were observed in any group. Expected systemic pharmacologic effects were observed only at MAP0004 target doses ≥ 0.224 mg/kg (achieved doses > 0.154 mg/kg), which was more than five times the maximum daily intravenous (IV) human clinical dose of DHE, and more than twenty times the systemic equivalent dose of MAP0004. The no-observed adverse effect level (NOAEL) was the achieved inhaled dose of 0.045 mg/kg, or four times the human clinical dose of MAP0004.
Introduction
Dihydroergotamine mesylate (DHE) has been used for over sixty years for the treatment of migraine headaches (Saper and Silberstein 2006; Silberstein and McCrory 2003). Despite its well-recognized effectiveness (Saper et al. 2006), DHE is associated with side effects, particularly nausea, dizziness, paresthesia, and flushing. In rare instances associated with long-term, high-dose, daily use, ergotism has been observed (MaassenVanDenBrink et al. 1998; Saper and Silberstein 2006). There is a single report of pleuropulmonary changes associated with DHE in which daily dosing of 9 mg occurred over a period of fifteen years. Although the drug is effective, oral, nasal, and subcutaneous administration is associated with highly variable absorption. Consequently, patients may not obtain headache relief or may experience unacceptable side effects (Saper and Silberstein 2006).
MAP0004 is an orally inhaled form of DHE under development for the treatment of acute migraine. The drug is administered using a breath-actuated inhaler (Tempo inhaler), which is easy to use and designed to ensure precise, consistent delivery of drug to the systemic circulation via the lung (Shrewsbury, Cook et al. 2008; Shrewsbury, Kori et al. 2008). MAP0004 achieves a lower peak plasma concentration, but with similar rapidity to peak concentration (Tmax six minutes for IV and twelve minutes for inhaled) and area under the plasma concentration curve (AUC) compared with IV DHE when administered to healthy volunteers (Shrewsbury, Cook et al. 2008). MAP0004 offers the potential for fast, effective, and convenient treatment of acute migraine with a lower risk of adverse events (Shrewsbury, Cook et al. 2008). Although the systemic and nasal inhalation toxicology of DHE is well characterized in humans (Galer et al. 1991; Garcia et al. 2000; Graham et al,. 1984; Lipton 1997; Magoux and Zlotnik 2004; Molkara et al. 2006; Orton and Richardson 1982; Silberstein and McCrory 2003; Tay and Chee 1998) and some systemic data exist in animals (Shrewsbury, Cook et al. 2008), oral inhalation is a novel route of administration, and the effect of chronic exposure from oral inhalation on the cardiopulmonary system is unknown. Thus, the objective of this study was to assess the local respiratory tract and systemic toxicity of MAP0004 in a commercially representative formulation, when administered daily for six months to dogs.
Materials and Methods
Study conduct complied with the Food and Drug Administration Good Laboratory Practice Regulations for the Conduct of Non-clinical Studies and Principles of Good Laboratory Practices. The study was approved by the Institutional Animal Care and Use Committee. The housing and care practices met current Association for Assessment and Accreditation of Laboratory Animal Care International standards and current requirements in the Guide for the Care and Use of Laboratory Animals (1996) and the United States Department of Agriculture through the Animal Welfare Act.
Study Design
Forty beagle dogs, twenty males and twenty females, were assigned to one of five treatment groups. At the start of dosing, animals were about nine or ten months of age and weighed 6.8 to 14.5 kg. The dogs were dosed each day for 182 consecutive days. Animals were exposed to drug in thirty-minute intervals, a time selected to attain a maximum feasible dose but that was still tolerable to the animals. Groups 1 to 4 were exposed to a single daily inhalation exposure, whereas animals in Group 5 were exposed twice daily, once in the morning and again at least four hours later. Dogs in treatment Group 1 received vehicle only; dogs in treatment Groups 2 to 5 received escalating doses of MAP0004 (Table 1
). The total inhaled dose was calculated using the equation:
Treatment groups.
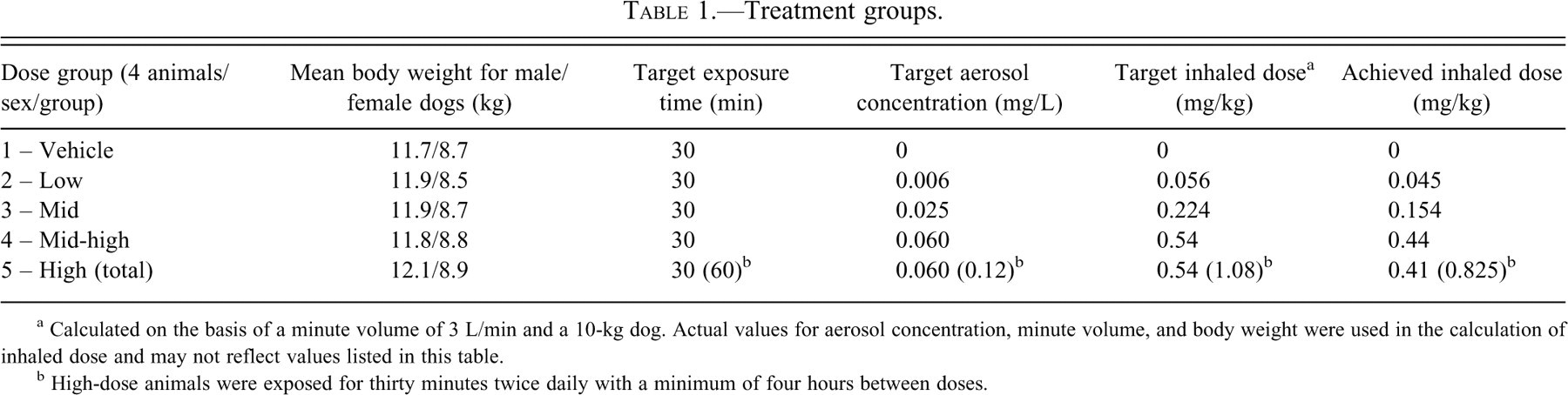
a Calculated on the basis of a minute volume of 3 L/min and a 10-kg dog. Actual values for aerosol concentration, minute volume, and body weight were used in the calculation of inhaled dose and may not reflect values listed in this table.
b High-dose animals were exposed for thirty minutes twice daily with a minimum of four hours between doses.
Study Treatment
MAP0004 was supplied in metered-dose inhaler (MDI) canisters containing a stable concentration (5 mg/mL) of DHE drug particles approximately 0.5 to 4 µm in diameter, suspended in a vehicle consisting of a HFA227/HFA134a propellant blend. The formulation contained no other excipients or additives. The control, vehicle only, consisted of HFA227/HFA134a propellant. Each dose was administered by nasal-only inhalation via an apparatus that consisted of an aerosol generation module and a distribution plenum and could supply up to eight dogs via a delivery tube/mask (Figure 1 ). The generator was calibrated to produce a continuous stream of vehicle only or MAP0004 aerosol to each mask. By adjusting the ratio of airflow to MAP0004 aerosol discharged from MDI units into the plenum, delivered drug concentrations could be adjusted to achieve target inhaled doses ranging from 0.0 mg/kg for the vehicle-only control to approximately 1.08 mg/kg (or 0.54 mg/kg/dose) for the high-dose group (Table 1). The exposure systems were characterized prior to animal exposures using aerosol and chemical analyses. Aerosol particle size distribution (PSD) was measured at the inhalation exposure mask using In-Tox Products (Moriarty, NM) brand cascade impactors consisting of eight stages with effective cut-off diameters ranging from 0.5 to approximately 12 µm. The mean (geometric standard deviation) aerodynamic particle diameter (MMAD) was determined to be 3.6 (1.8) µm, which is appropriate for the canine model.
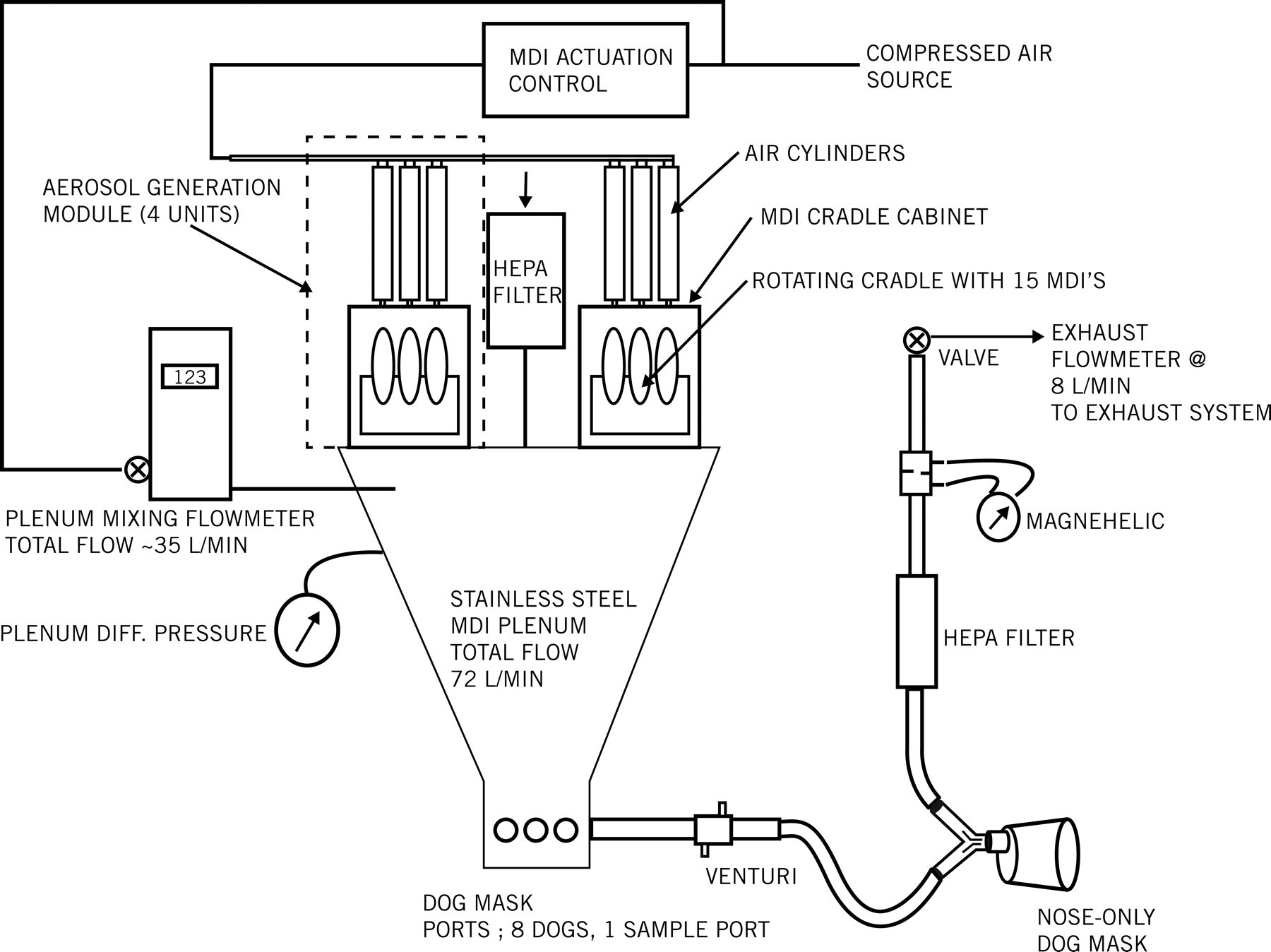
Inhalation dosing apparatus.
Study Evaluations
Potential toxicity was evaluated from clinical observations, physical examinations including body weights and food consumption, ophthalmic examinations, electrocardiogram (ECG) analysis (including interval evaluation), and clinical and anatomical pathology including organ weights. Respiratory function (tidal volume, respiratory rate, and minute volume) was assessed on Day 1 and monthly thereafter. Minute volume was used to calculate the estimated inhaled dose of DHE. Blood samples for toxicokinetic evaluation were obtained from all animals on Days 1 and 28, at Week 13, and at Week 26.
Observations for moribundity and mortality were conducted twice daily, in the morning and in the afternoon, at least six hours apart, seven days per week throughout the study. In addition, cage-side clinical observations for evidence of toxicity were performed at least twice daily. On the days of treatment administration, cage-side clinical observations were performed prior to exposure and at one hour (±15 minutes) post-exposure. Cage-side observations for the high-dose group (Group 5) were conducted at least three times daily, once prior to the first exposure, and again at one hour (±15 minutes) following each exposure. In addition, a clinical observation was recorded on Day 183 prior to scheduled necropsy.
Physical examinations were conducted on all animals assigned to the study by a laboratory veterinarian beginning with Week 4 and then monthly thereafter. The physical examination included manual collection of heart rate and respiratory rate and a recording of rectal temperature. In addition, general appearance and attitude; skin and haircoat; musculoskeletal and extremities; oral cavity and teeth; eyes, ears, and nose; chest and thorax; and abdomen and urogenital area were assessed. Abrasion–tip of ears was recorded when an abrasion was noted at the tips or edges of the ears, and discoloration–tip of tail was recorded when slight hair loss and discoloration were noted. Scabbed area–tip of ears was noted when the abrasions began scabbing over. Individual body weights of study animals were recorded on Day 1 prior to exposure, once weekly thereafter, and on the day before and the day of scheduled necropsy.
Qualitative food consumption was recorded for all animals beginning on Day 1 and daily during the study period, except when fasting was required for scheduled necropsy or clinical pathology blood collection. Food was provided following exposure for overnight consumption, which was assessed the following morning.
Ophthalmic examinations were conducted by a laboratory veterinarian on all animals prior to selection/group assignment (pretest) and again within one week of the final treatment exposure (Week 26).
Electrocardiograms were obtained pre-study and were analyzed by the study physiologist for the presence of significant abnormalities that would exclude a dog from the study. Electrocardiograms were obtained during Week 12 and again during Week 26. Tracings of leads I, II, III, aVR, aVL, aVF, and V10 were collected at a paper speed of 50 mm/second. Additionally, leads I, aVF, and V10 were collected at a paper speed of 10 mm/second. Analysis included morphology of P, QRS, and T waves and measurement of RR, PR, QRS, and QT interval duration. Corrected QT interval durations were calculated using Fridericia’s Formula: QTc = QT/(RR)1/3.
Clinical Laboratory Tests
All dogs were fasted for at least twelve hours prior to scheduled blood sampling for hematology, serum chemistry, and coagulation determinations. Approximately 6 to 8 mL of blood was obtained from each animal from the cephalic or jugular vein pre-test, on Day 29, during Week 12, and prior to scheduled necropsy on Day 183. Urinalysis was performed in conjunction with the clinical laboratory tests except on Day 29, when no urine was collected. Dogs were fasted overnight for at least twelve hours for urine collection, which began the day prior to scheduled blood collection.
Toxicokinetics
On Day 1, Day 28, and during Weeks 13 and 26, whole-blood samples (3.0 mL) were collected at routine intervals after the completion of dosing. Toxicokinetic analysis of plasma concentration time data was performed using WinNonlin to determine relevant toxicokinetic parameters, including observed and fitted peak plasma concentrations (Cmax) and time to peak plasma concentration (Tmax), absorption and elimination rate constants, half-life, clearance, volume of distribution, and area-under-the-plasma concentration time curve (AUC0-inf), and results were dose-normalized. The Cmax data from each dog were evaluated using an appropriate compartmental model, and when the data were not sufficient to be modeled, then noncompartmental analysis was performed.
Necropsy
All dogs were fasted overnight for a minimum of twelve hours and then humanely euthanized on Day 183, the day following their final inhalation exposure, by the administration of a barbiturate overdose, followed by exsanguination. Body weights were recorded prior to the Day 183 necropsy. Necropsy was performed by a board-certified veterinary pathologist. Each necropsy included examination of the external surface of the body; all orifices; the cranial, thoracic, abdominal, and pelvic cavities and their contents; and collection of all tissues and all gross findings. Paired organs were weighed together, and absolute weight, organ-to-body weight ratios, and organ-to-brain weight ratios were reported.
All tissues collected at necropsy were trimmed, processed, embedded in paraffin, sectioned at approximately 5 µm, mounted on glass slides, and stained with hematoxylin and eosin. In addition, any macroscopic lesions identified at necropsy were processed for microscopic examination. A complete histopathological evaluation of the tissues was conducted by a board-certified veterinary pathologist. Microscopic findings were graded semiquantitatively. Minimal (grade 1) represented a change barely exceeding the limits of normal variation; mild (grade 2) represented an easily discernable lesion unlikely to have any biological significance; moderate (grade 3) represented a change affecting a large area of the represented tissue that has the potential to be of some significance; and marked (grade 4) represented a lesion that approached maximal in extent for the lesion in the organ.
Data Analysis
Respiratory measurements were reported as means and standard deviation. All quantitative, clinical pathology, and postmortem data were statistically analyzed (when N ≥ 3). Data were analyzed for effects of MAP0004 by analysis of variance. Bartlett’s test was used to determine homogeneity of data. For homogeneous data across test groups, tests for differences between vehicle and comparison groups were made using Dunnett’s test. For nonhomogeneous data, tests for pairwise differences between vehicle and each of the comparison groups were made using Cochran and Cox’s modified two-sample t test. Statistical significance was reported at the p < .05 level.
Results
Inhaled Aerosol Characteristics
The overall mean achieved aerosol concentrations of MAP0004 indicated close adherence to target values (Table 2 ). Average achieved aerosol concentrations of MAP0004 were 104% to 108% of target levels. Variability of the measurements expressed as relative standard deviation (RSD) was less than 20% for all treatment groups except the low-dose group, in which RSD was 25% in both male and female animals. Achieved inhaled doses, which provide an IV equivalent systemic exposure approximation of DHE delivered from MAP0004, were 60% to 88% of target in male dogs and 73% to 86% in female dogs (Table 3 ). The lower achieved inhaled doses could be attributed to a lower than expected minute volume of 2.26 L/min.
Target and achieved aerosol concentrations of MAP0004.
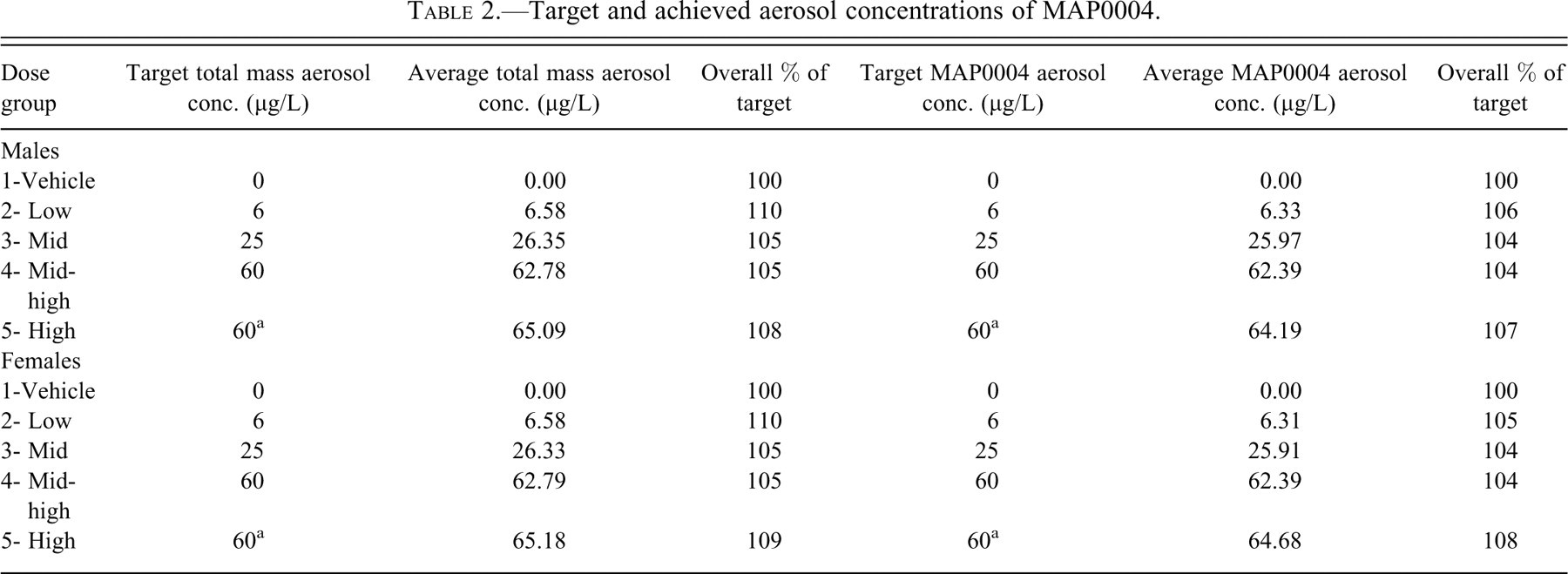
Achieved inhaled doses of DHE delivered from MAP0004.

a Target inhaled dose based on a sixty-minute exposure period (2 × thirty minutes).
Samples for the drug aerosol PSD determination were collected weekly for four weeks and monthly thereafter. Mean (GSD) values of mass median aerodynamic diameter for each group were 3.6 or 3.7 (1.8) µm, and the RSD ranged from 6.9% to 10.1%. Overall, PSD values were stable and in the appropriate range for a drug administered by aerosol inhalation.
Physical Changes
No changes were observed in mean body weight or food consumption. No abnormalities of the eye or surrounding tissues were noted during the study, and ophthalmic examinations were normal for all animals pre-study and at Week 26. No changes in respiratory measurements, hematology, serum chemistry, or urinalysis results were noted during the study. No relevant differences between MAP0004 and vehicle groups were noted for heart rate or ECG tracings, including RR, PR, QRS, QT, and corrected QT intervals. A dose-related increase in the incidence of abrasion and scabbed areas on the tips of the ears and also sporadic discoloration on the tip of the tail was seen in the mid-, mid-high, and high-dose groups.
Toxicokinetic Profile
Toxicokinetic results (Figures 2 and 3 ) showed dose proportionality for MAP0004 for a given dosage level, and no consistent sex-dependent differences were observed. These results indicate that the kinetics of MAP0004 in male and female dogs were linear from Day 1 through Week 26. From Day 1 through Week 26, mean (SD) elimination rate constants in male and female dogs were comparable for the mid-, mid-high, and high-dose exposure levels. Mean (SD) elimination rate constants were 0.00759 (0.00238) min-1 at Day 1 and 0.00584 (0.00158) min-1 at Week 26, and mean (SD) elimination half-life was 103 (29) minutes at Day 1 and 144 (56) at Week 26.
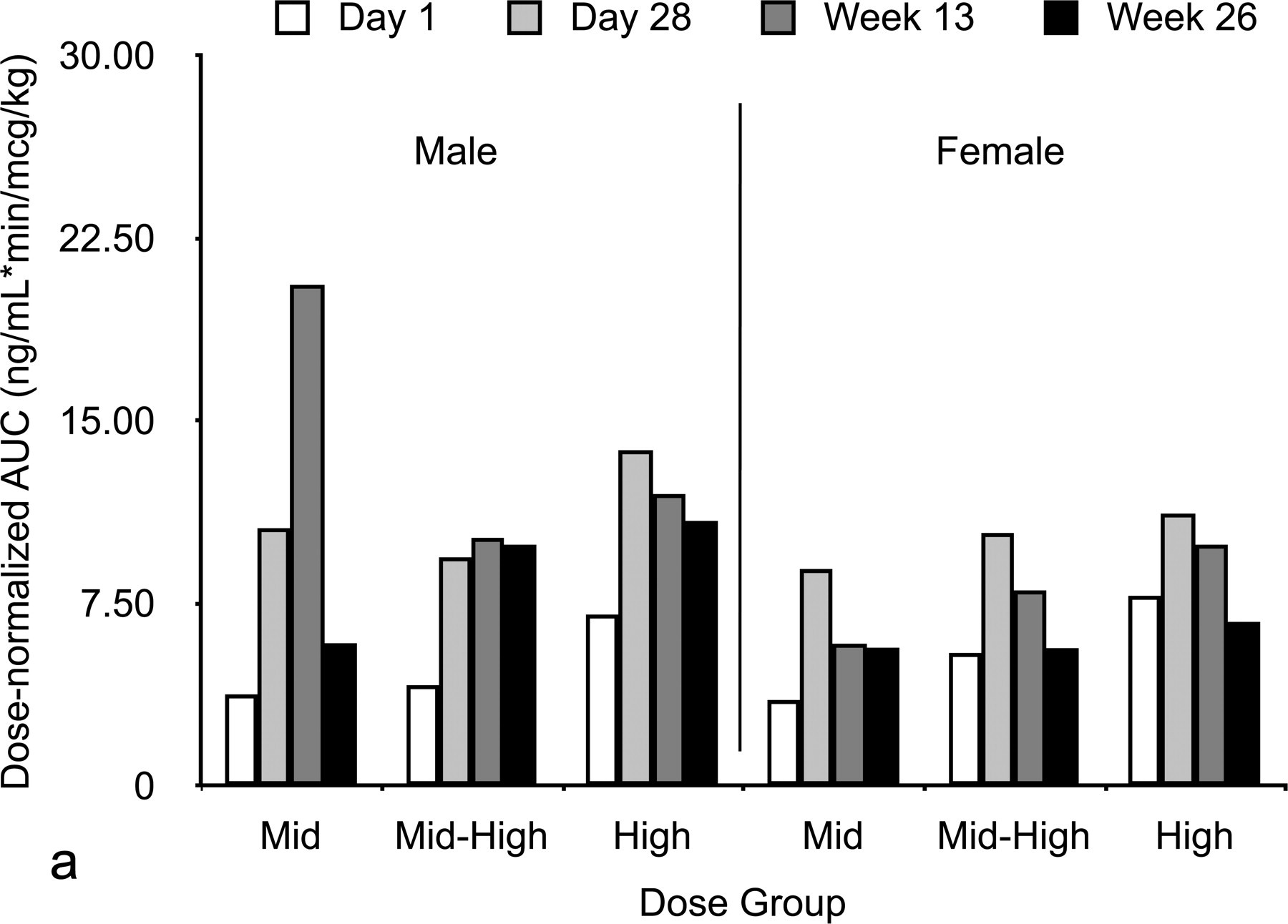
Area under the curve dose-normalized (a) and area-under-the-plasma concentration time curve time 0 to infinity (b) in male and female beagle dogs according to dose group.
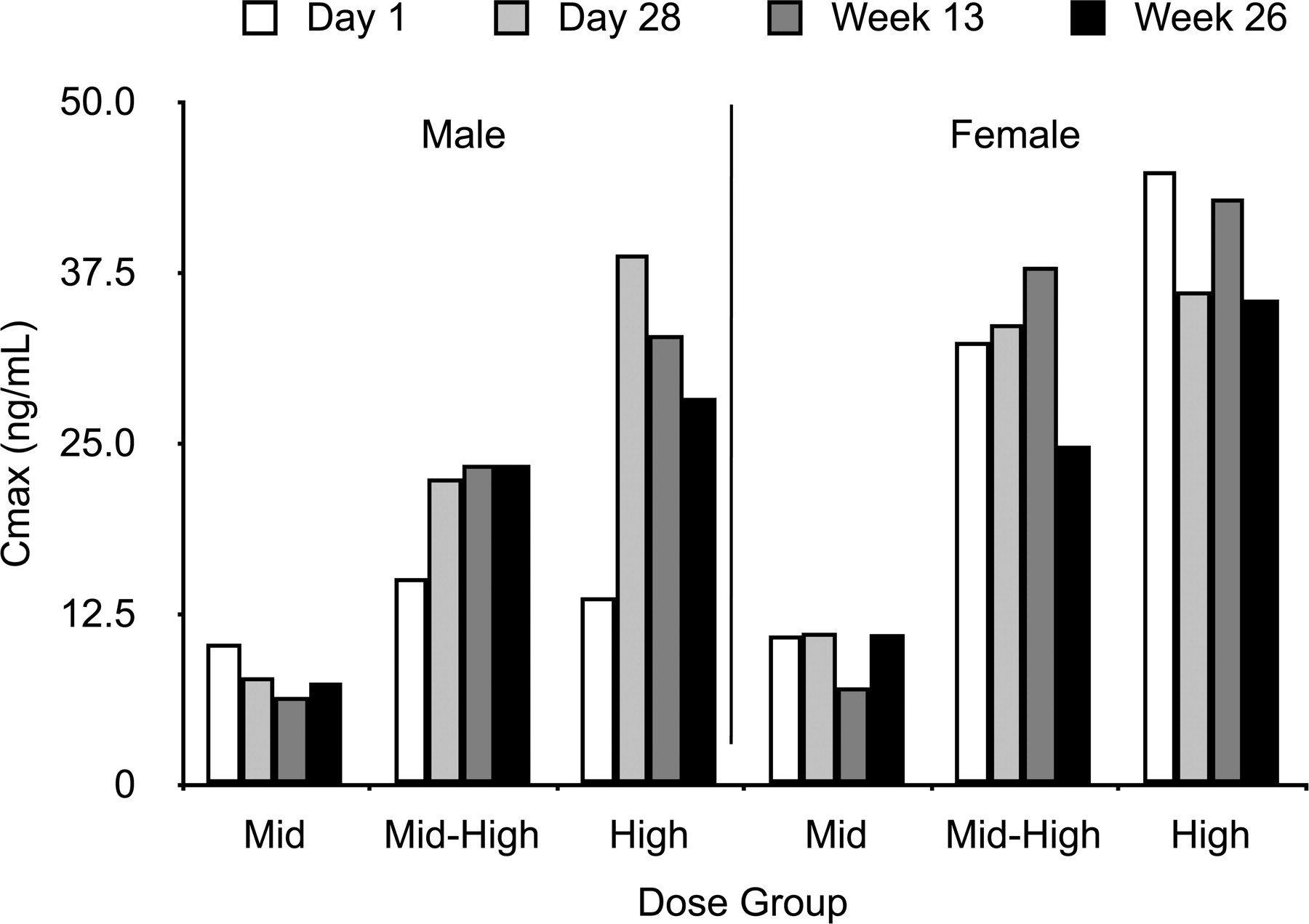
Peak plasma concentration in male and female beagle dogs according to dose group.
At Day 1, mean Cmax and AUC0-inf increased with increasing exposure level for male and female dogs, except for male dogs in the high-dose group, which had a Cmax slightly lower than that observed for the mid-dose group (Figures 2 and 3). Mean dose-normalized AUC increased slightly with dose, but the differences between dosage groups were not sufficient to suggest an effect of dose on these parameters. Starting at twenty-eight days, repeated exposures led to an increase in dose-normalized values of Cmax and AUC0-inf for both male and female dogs when compared to the Day 1 data. As the elimination rates and half-life on Day 1 were comparable to Day 28, this observation suggests there might be a slight reduction in the clearance of MAP0004 following the initial stages of a repeated exposure. However, this trend did not increase in magnitude, and dose-normalized values for Weeks 13 and 26 did not change significantly from those observed on Day 28.
Clinical Abnormalities
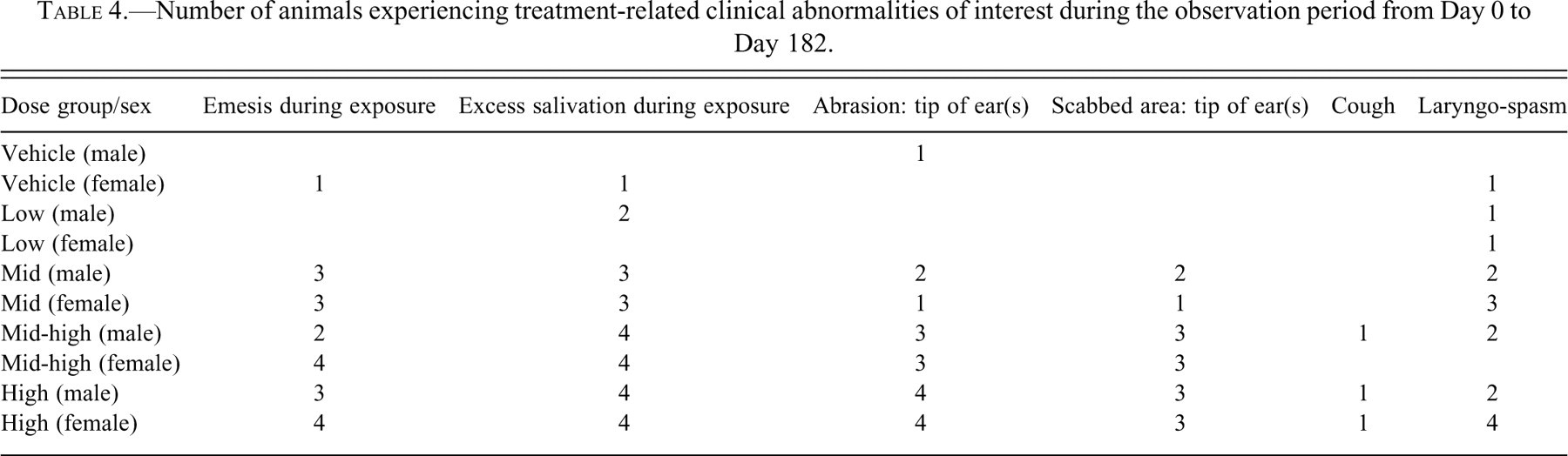
MAP0004-related findings were observed in male and female dogs primarily at the three highest doses (Table 4 ). Emesis and excessive salivation occurred at actual inhaled doses of 0.154 (mid-dose, Group 3), 0.44 (mid-high dose, Group 4), and 0.825 mg/kg/day (high-dose, Group 5) from Day 1 to Day 182 at varying incidences in six to eight animals in each group. Laryngospasm and cough were noted sporadically in a few animals in each dose group but more frequently in the high-dose group, most likely associated with excessive salivation. A dose-related increase in the incidence of abrasion and scabbed areas on the tips of the ears and also sporadic discoloration on the tip of the tail was seen in the mid-, mid-high, and high-dose groups. A variety of other mild clinical abnormalities was recorded for all groups, including the vehicle group, and were considered to be incidental findings unrelated to MAP0004.
Number of animals experiencing treatment-related clinical abnormalities of interest during the observation period from Day 0 to Day 182.
Pathology
No relevant changes in organ weights were noted in any dose group. No evidence of macro- or microscopic changes was observed in the lungs in any dose group. Treatment-related microscopic changes were minor and were noted in the respiratory nasal epithelium and focal areas of the skin of the ears. Histopathological findings revealed that all animals in Groups 3, 4, and 5 had minimal nasal hyperplasia of anterior nasal respiratory epithelium (changes were observed in several anterior locations and barely exceeded the limits of normal variation). No other respiratory pathology findings were observed. In Groups 1 and 2, no respiratory pathology was observed in any tissue.
Discussion
Dihydroergotamine and ergot alkaloids have been used effectively for treatment of acute migraine for over 6 decades (Lipton 1997; Silberstein and McCrory 2003). Despite this long record of use, ergotamine in particular, but also DHE, is associated with safety concerns including ergotism and nausea (Galer et al. 1991; Garcia et al. 2000; Graham et al. 1984; Lipton 1997; Magoux and Zlotnik 2004; Molkara et al. 2006; Orton and Richardson 1982; Silberstein and McCrory 2003; Tay and Chee 1998). Currently, DHE is available for IV, subcutaneous, intramuscular, oral, and nasal administration; however, except for IV use, other routes of administration are associated with variable absorption and unpredictable therapeutic responses (Saper and Silberstein 2006).
MAP0004 is being developed as a novel orally inhaled formulation of DHE for the treatment of acute migraine (Shrewsbury, Cook et al. 2008; Shrewsbury, Kori et al. 2008). A Phase I tolerability and pharmacokinetic study of the cardiovascular effects of DHE given IV or by intrapulmonary administration selected to model inhaled DHE found no unexpected safety concerns (Shrewsbury, Stonerook et al. 2008). No adverse ECG effects were observed and no meaningful differences were observed between IV and intrapulmonary treatment for heart rate and systemic and vascular pressures, which was consistent with findings of an earlier study in dogs (Shrewsbury, Stonerook et al. 2008). In a Phase 2 study (Aurora et al. 2009), orally inhaled MAP0004 was found to be efficacious at doses (1.0 and 2.0 mg nominal doses or 0.5 or 1.0 mg systemic equivalent doses) approximately equal to IV doses of 0.5 mg and 1.0 mg, which were determined in a pharmacokinetic study of healthy volunteers (Shrewbury et al. 2008). However, because of the potential for pulmonary toxicity, chronically inhaled drugs must be carefully evaluated to establish their toxicological profile (Wong 2007). Thus, it was important to investigate and confirm the safety of MAP0004 after oral inhalation in appropriate animal models.
The results from this study showed that the estimated achieved total mass of DHE delivered by MAP0004 was slightly lower than target (Table 3). Overall achieved inhaled doses were lower, ranging from 60% to 88% of target for each of the dose groups; this result was attributed to an actual minute volume of 2.26 L/min versus the expected minute volume of 3.0 L/min that was used to calculate target inhaled doses.
A previous study comparing IV and intrapulmonary administration of DHE in the dog reported a similar Cmax and AUC with both routes of administration, and no differences in cardiovascular toxicity were observed (Shrewsbury, Stonerook et al. 2008).
Plasma concentrations were below the limit of quantitation at Day 1 in the low-dose group and thus are not reported. Toxicokinetic results indicated that MAP0004 dosages were dose proportional, based on similar dose-normalized AUC values. Mean (SD) half-lives for MAP0004 were 103 (29) minutes at Day 1 and 144 (56) at Week 26 for dogs in the mid-, mid-high, and high-dose groups. However, repeated treatment for twenty-eight days led to an increase in dose-normalized AUC for both male (95–181%) and female (43–152%) dogs when compared to the Day 1 data, although elimination rates and half-lives on Day 1 were comparable. This trend did not increase in magnitude, and dose-normalized AUC values for Weeks 13 and 26 were similar to those observed on Day 28. Repeated doses up to twenty-six weeks did not appear to change the elimination rate constant and corresponding half-life values. The increase in AUC suggests that repeated doses between Days 1 and 28 led to a reduction in the clearance of MAP0004. This trend toward reduced clearance leveled off over time, as demonstrated by similar dose-normalized AUC values for Day 28 and Weeks 13 and 26. Thus, although there was a slight indication that clearance was affected by repeated exposure, there was no evidence that this effect led to nonlinear kinetics.
Daily administration of MAP0004 by inhalation at mid and higher target doses (greater than 0.224 mg/kg/day) to beagle dogs resulted in emesis, excessive salivation, and abrasion or scabbing of the tip of the ear. At the highest target dose (1.08 mg/kg/day), emesis and excessive salivation prevented any further dose escalation, and thus defined the maximum feasible dose. Abrasion and/or scabbing of the tip of the ear, observed only in the three highest dose groups, were correlated with dose during monthly physical examinations and were confirmed microscopically by histopathological examination.
Gross necropsy findings attributed to MAP0004 were related to crusts, alopecia, and/or scabbing of the skin of the ears in the mid-, mid-high, and high-dose groups. The skin lesions were thought to be related to the vasoconstrictive properties of DHE. Local necrotizing as a result of peripheral vascular constriction is a known systemic adverse affect, termed “ergotism,” associated with chronic use of ergots. These “ergotism” observations were presumed to be from the chronically high systemic exposure corresponding to plasma levels known to cause peripheral vasoconstriction (Migranal Summary Basis of Approval FDA, NDA20-148; Müller-Schweinitzer 1984). No other physical changes or clinical abnormalities of significance were observed that were attributable to MAP0004.
Treatment-related microscopic changes were minor and were noted only in the anterior nasal respiratory epithelium. All dogs in the high-dose Group 5 were observed to have minimal nasal hyperplasia, compared to generally two or fewer animals in Groups 3 and 4, and no such observations in Groups 1 and 2. Based on this finding, MAP0004 was interpreted to have a minor irritant effect on the nasal epithelium, which manifested itself as increased nasal hyperplasia as a function of dose. The nasal cavity is susceptible to chemically induced injury as a result of exposure to inhaled irritants (Harkema 1990). To achieve the maximum feasible inhalation dosing requires long dosing intervals as in this study, in which dosing time was thirty minutes. To achieve consistent dosing over these intervals, administration with a facemask is required. A consequence of facemask administration is that the dose is delivered to the respiratory tract primarily through the nose. Thus, chronic high-dose exposure to irritants in the nasal passage is a common observation in inhalation studies. It was also observed after chronic nasal administration of DHE in a marketed aqueous spray product (Migranal Summary Basis of Approval FDA, NDA20-148).
Other than the nasal epithelial irritation, no other significant respiratory tract toxicity was observed in dogs exposed at a target dose up to 1.08 mg/kg/day (actual dose 0.825 mg/kg/day) of DHE for six months (equivalent to more than twenty times the maximum safe daily human IV dose). The NOAEL, including nasal irritation, for MAP0004 was found to be at the low dose (target 0.056 mg/kg/day or 0.045 mg/kg/day actual; Table 3). The actual dose corresponds to 1.6 times the maximum daily IV dose and 3.2 times the 1.0 mg systemic equivalent dose of MAP0004. Nasal epithelial irritation is unlikely to be a relevant safety or tolerability factor in humans who will have little, if any, nasal exposure by oral inhalation.
Although the ergot alkaloids are known to be active at the 5-HT2 receptors in the myocardium and have been associated with fibrogenesis in the cardiopulmonary system, DHE is only slightly active at this receptor when present at peak concentrations (5 ng/mL) achieved following inhalation (Cook et al. 2009). In this study, even at the highest chronic doses, no fibrogenic activity was observed. No macro- or microscopic changes were noted in the blood vessels of the lungs or heart in any dose groups. No evidence of plexiform changes in the vascular media, including pulmonary blood vessels, or fibroproliferative changes in any of the heart valves were observed in any group, which is a concern because of the well-known potential toxic effects of ergot alkaloids with chronic administration (Galer et al. 1991; Garcia et al. 2000; Graham et al. 1984; Molkara et al. 2006; Orton and Richardson 1982; Tay and Chee 1998).
Exposure to ergot alkaloids, including DHE, the active ingredient in MAP0004, can produce nausea and/or vomiting and ergotism characterized by peripheral vasoconstriction leading eventually to tissue necrosis in the extremities (Galer et al. 1991). Thus, the observations noted in this study with MAP0004 at higher doses were not unexpected, because they are known pharmacological effects of exposure to ergot alkaloids. The work reported in this study was part of the regulatory requirements for approval of MAP0004. To date, results from clinical studies in healthy volunteers and in migraine patients have not identified any safety issues with administration of a single orally inhaled dose of MAP0004 (Aurora et al. 2009; Shrewsbury, Cook et al. 2008; Shrewsbury, Kori et al. 2008). Further, the incidence of adverse events was remarkably lower with MAP0004 compared with IV DHE in a comparative pharmacokinetic trial, particularly for nausea and emesis, because of a lower Cmax of 5 versus 45 ng/mL with a 1 mg dose of MAP0004 (Shrewsbury, Cook et al. 2008). A study of MAP0004 in healthy adult volunteers with asthma found no adverse pulmonary effects (Shrewsbury, Kori et al. 2008). Ongoing trials are evaluating MAP0004 for the acute treatment of migraine and should provide further support for the safety and tolerability.
MAP0004 is predicted to have a different pharmacological profile from IV DHE because of its markedly different concentration-dependent receptor binding affinity (Cook et al. 2007; Cook and Shrewsbury 2008). In a comparative study of the receptor binding affinity of IV DHE at peak concentration and MAP0004 at a peak concentration equivalent to an inhaled dose of 0.88 mg, IV DHE exhibited broad receptor binding affinity for 5-HT, adrenergic, and dopaminergic receptors that are associated with antimigraine effects, but also with adverse effects of the cardiovascular, central nervous system, and gastrointestinal system. In contrast, MAP0004 exhibited binding affinity for 5-HT1A receptors and 5-HT1B and 5-HT1D receptors, which are primarily associated with its antimigraine efficacy (Cook et al. 2009; Cook et al. 2007; Cook and Shrewsbury 2008). In addition, oral absorption is not expected to contribute to the systemic effects of MAP0004. The oral bioavailability of DHE is less than 1% in healthy subjects (Little et al. 1982). In addition, MAP0004 is administered to humans by oral inhalation rather than nasal inhalation, as done in this study. Oral inhalation results in lung deposition of approximately 30–40% versus approximately 60% with nasal inhalation (Labiris and Dolovich 2003; Suman et al. 1999).
In conclusion, no significant respiratory or cardiopulmonary effects were observed. Daily nose-only exposure to MAP0004 for twenty-six weeks at target inhaled doses of up to 1.08 mg/kg/day (actual dose 0.0825 mg/kg), representing more than twenty times the currently approved daily IV DHE dose, had only limited toxicological effects. Most observations were expected systemic pharmacological effects, including emesis and excessive salivation and abrasions and/or scabbing at the tip of the ears at target inhaled doses of 0.224 mg/kg/day and higher. Minor nasal irritation was observed above the target inhaled dose of 0.056 mg/kg/day. Based on the results and objectives of this study, the NOAEL including nasal irritation of MAP0004 in beagle dogs after twenty-six weeks of exposure was the target inhaled low dose, 0.056 mg/kg (actual dose 0.045 mg/kg), which is 4 times the intended therapeutic dose of MAP0004. Excluding nasal effects, the NOAEL was 0.154 mg/kg which is approximately 14 times the intended therapeutic dose of MAP0004.
Acknowledgment
The authors would like to acknowledge the editorial assistance of Richard S. Perry, PharmD, in the preparation of this manuscript.
Footnotes
This study was supported by MAP Pharmaceuticals, Mountain View, California. Thomas Armer is an employee of MAP Pharmaceuticals, Mountain View, California. MaryEllen Lynch, Ronald Moutvic, and Allen Singer are employees of Battelle Memorial Institute, Columbus, Ohio and have no consulting arrangements, stock ownership, equity interests, or other disclosures that may pose a conflict of interest regarding the publication of this manuscript.

