Abstract
BILN 2061 is a potent, reversible inhibitor of hepatitis C virus NS3/NS4A serine protease. Early clinical proof of principle with the drug was offset by the results of subsequent safety studies in Rhesus monkeys revealing cardiotoxicity that featured myocardial vacuolation corresponding to mitochondrial swelling. Here we describe an investigation into the nature, onset, and reversibility of the lesion, and an assessment of potentially predictive biomarkers for the change. Rhesus monkeys were orally administered 1,000 mg/kg/day BILN 2061 and either necropsied after one, three, fourteen, or twenty-eight doses or afforded a ten-week recovery period. The results of electrocardiographic and plasma troponin I and T measurements were unaffected by BILN 2061, but cardiac myocytic vacuolation, correlated with mitochondrial swelling, was observed after three or more doses. Echocardiographic traces obtained after twenty-eight consecutive days of dosing revealed two animals with diminished left ventricular cardiac ejection fraction. One animal was immediately necropsied and exhibited marked cardiotoxicity. The other was afforded a ten-week treatment-free period during which the left ventricular ejection fraction returned to normal. All recovery animal hearts were microscopically and ultrastructurally normal. High-dose BILN 2061 cardiotoxicity in Rhesus monkeys appeared early in the treatment regimen and exhibited reversibility. A reliable biomarker has yet to be identified.
Introduction
Hepatitis C virus (HCV), a hepatotropic RNA virus, is a member of the Flavivirus family and may cause acute, asymptomatic hepatitis, but fulminant hepatitis is rare. It is estimated that over 3% (170 million) of the world’s population is currently infected with HCV (Alter 1996) and that about 70% of these acute cases have the potential to progress to a chronic state featuring variable degrees of hepatic inflammation and fibrosis. Chronic HCV infection is a risk factor for cirrhosis, liver failure, and sometimes hepatocellular carcinoma and is therefore one of the most important liver diseases worldwide and the most common hepatic cause of serious morbidity leading to death or candidacy for liver transplantation (Hoofnagle 2002).
Medical treatment options for chronic HCV infections are limited. Early studies of interferon α (IFN) in chronic HCV infection revealed prompt declines in serum and hepatic HCV RNA titers in some patients and long-term responses with a sustained loss of serum HCV and clinical resolution of the chronic infection (Hoofnagle 1986). Experience with IFN monotherapy demonstrated efficacy in only a small fraction of patients (9–20%) with chronic HCV infection (DiBiscegilie and Hoofnagle 2002).
The antiviral ribavirin as a monotherapy for HCV infection is ineffective, but ribavirin and IFN as a combination therapy increased the overall sustained virologic response (SVR) rate to approximately 40% for all HCV types (DiBiscegilie and Hoofnagle 2002). Use of the combination was associated with a high incidence of adverse drug reactions. Subsequent research led to the development of pegylated interferon (pegIFN), which features a prolonged plasma half-life, thereby permitting once-weekly administrations. Combination therapy with pegIFN and ribavirin has increased SVR rates to about 55% (Glue et al. 2000; Lindsay et al. 2001). Although the exact mechanism of action and nature of the synergism between the two molecules has not been clearly elucidated (Lau 2002), the current “gold standard” for the management of chronic HVC infection is still the combination of a parenterally administered pegylated interferon with ribavirin. Despite these advances, a large unmet medical need exists for many patients with chronic HCV infection, and extensive research efforts are ongoing to find more effective and better-tolerated treatment modalities.
Inhibition of viral enzymes critical to replication of HCV represents a viable treatment mode for the management of chronic HCV infection. HCV NS3/NS4A serine protease, a virally encoded enzyme essential for viral replication, is a likely target. Hepatitis C virus is a 9.6 kbase single positive-strand mRNA virus and contains a long, open reading frame that encodes several structural and nonstructural viral proteins. The HCV serine protease leads to proteolysis of the HCV translated polyprotein into HCV-specific structural and nonstructural enzymes such as helicase and RNA-dependent RNA polymerase (also required for viral replication). Treatment of chronic HCV infection with viral-specific inhibitors that target these essential enzymes, in combination with pegIFN with or without ribavirin, may enhance SVR and facilitate long-term resolution of chronic hepatitis.
BILN 2061 (Boehringer Ingelheim) is a novel, orally active, reversible inhibitor of HCV NS3/NS4A serine protease. It is a macrocyclic mimic of an HCV polyprotein peptide-cleavage product and acts through competition for NS3/NS4A serine protease binding sites, and it is the first HCV viral-specific inhibitor to enter clinical trials (Lamarre et al 2003). The drug exhibits potent and specific inhibition of serine protease in enzymatic and cell-based assays with an EC50 in the 1-nM range. The compound was submitted to a wide range of other serine proteases (including host serine proteases) and was not active for any of the proteases tested. When administered to either naïve or previously treatment-failure patients infected by genotype-1 HCV, doses as low as 25 mg bid for two days induced rapid and dose-related reductions in viral load (>2 log10) (DeFrancesco and Migliaccio 2005).
The early clinical proof of principle with BILN 2061 was tempered by the results of subsequent safety studies conducted in Rhesus monkeys that revealed an unusual and specific cardiotoxicity, myocardial vacuolation observable with light microscopy, within four weeks of beginning a daily dosing regimen. In one study, a monkey administered 1,000 mg/kg/day for twenty-eight days died of congestive heart failure. In our experience, most Rhesus monkeys administered 500 or 1,000 mg/kg/day for twenty-eight days exhibit myocardial vacuolation. Myocytic degeneration was rarely observed, and when observed, it was of minimal severity. Ultrastructural examination of heart tissue exhibiting myocardial vacuolation revealed the vacuoles to be swollen mitochondria (unpublished results).
This communication describes the conduct, results, and interpretation of a four-week study in Rhesus monkeys conducted to: (1) characterize the time course for the onset of myocardial vacuolation and mitochondrial swelling induced by the administration of 1,000 mg/kg/day of BILN 2061; (2) determine the histopathological progression of the lesion over the four-week dosing period; (3) determine the potential for reversibility of the lesion during a ten-week recovery period; and (4) assess the potential utility of electro- and echocardiography and measurement of serum troponin I and T and brain natriuretic peptide (BNP) as predictive biomarkers for cardiotoxicity.
Materials and Methods
Sexually mature male and female rhesus monkeys (Macaca mulatta, four to six years old, purchased from an AALAC-accredited source) were used for the study. Forty-four animals entered the two-week pretest phase, during which they were acclimated to laboratory conditions, handling, and oral gavage dosing with drug vehicle. Thirty-eight of the animals were randomized to control (four/sex) and treatment (fifteen/sex) groups. Animals weighed 4 to 7 kg at the initiation of dosing.
BILN 2061 was formulated in an aqueous vehicle composed of 5.6% tris, 1.1% sodium carbonate, 27.8% propylene glycol, 22.2% vitamin E TPGS (D-α-tocopheryl polyethylene glycol 1000 succinate), and 43.5% water. Vehicle or BILN 2061 (1,000 mg/kg) was administered in daily gavage volume doses of 10 mL/kg for twenty-eight consecutive days. Routine antemortem criteria included daily assessments of clinical signs, food consumption, and body weight maintenance.
Serum concentrations of troponins I and T were determined using an electrochemiluminescent immunoassay (using the ORIGEN analyzer) that used a two-site combination of antibodies. The troponin assays were validated for use in nonhuman primates and conducted in house at a contract laboratory (Analytics, Inc.), and standard operating procedures (SOPs) for the analytic methods are in place at that lab. Troponin blood samples were collected from animals at multiple time points throughout the study including the pretest phase; drug days 2, 4, 8, 15, 22; at terminal sacrifice; and at the end of the recovery phase. Heart tissue samples were taken at necropsy, and plasma samples were taken at similar time points as those taken for troponin analysis to measure brain natriuretic peptide (BNP) tissue gene expression and plasma levels. For the heart tissue, BNP messenger ribonucleic acid (mRNA) levels were studied using the Affymetrix GeneChip analyzer and TaqMan real-time polymerase chain reaction (PCR) probes. Plasma BNP peptide levels were measured with an enzyme-linked immunosorbant assay (ELISA).
Electrocardiographic (ECG) and echocardiographic examinations were conducted during the pretest weeks, one day prior to the scheduled sacrifice, and on recovery animals at the conclusion of the recovery phase. Echocardiograms were performed in conjunction with the ECGs in unconscious animals. Heart rates and PQ/QRS/QT durations were quantified from ten-lead ECG tracings. Echocardiographic evaluations were performed using a 10-MHz vector linear probe. Two-D images and M-mode evaluations were acquired using the parasternal long access (2-D) and parasternal short access (2-D and M-mode) at the level of the papillary muscles. Left ventricular ejection fraction (LVEF), fractional shortening (FS), left ventricular end-systolic diameter (LVDs), left ventricular end-diastolic diameter (LVDd), aortic blood flow (ABF), left ventricular end-systolic volume (LVVs), and left ventricular end-diastolic volume (LVVd) were measured or calculated.
During the dosing phase, groups of three dosed animals per sex were sacrificed and subjected to necropsy twenty-four hours after the administration of one, three, fourteen, or twenty-eight daily doses. One male and one female control were sacrificed twenty-four hours after vehicle administration on days 1, 3, and 14. The remaining monkeys (one per sex controls and three per sex dosed animals) were observed for an additional ten-week, treatment-free recovery period. At the conclusion of the recovery period, all remaining animals were sacrificed and subjected to necropsy and tissue collection.
Prior to sacrifice, each animal was anesthetized with sodium pentobarbital and then exsanguinated. Complete necropsies were performed, and macroscopic observational data were collected. Comprehensive sets of necropsy wet tissues were collected from each monkey and preserved in 10% neutral buffered formalin. Sections of heart and soleus muscle were also collected and fixed for electron microscopy.
The formalin-fixed heart and skeletal muscle samples were routinely processed, sectioned at 3 (heart) or 5 (skeletal muscle) µm, and stained with hematoxylin and eosin. Special stains performed on the myocardial tissue included periodic acid-Schiff reaction and Nile blue (for lipofuscin accumulation), Masson’s trichrome (for fibrous connective tissue accumulation), and Von Kossa stain (for presence of calcification).
All aspects of this study were conducted in accordance with our Institutional Animal Care and Use Committee (IACUC) and governmental guiding principles in the use of animals in research, ensuring the welfare of our study animals at all times.
Results
All animals survived until scheduled sacrifice, and there were no BILN 2061–associated clinical signs. The oral administration of 1,000 mg/kg/day for up to twenty-eight consecutive days was without effect on food consumption or body weights. Troponin analyses revealed normal concentrations of both troponin I and T in the pretest, terminal sacrifice, and recovery-phase blood samples. Brain natriuretic peptide gene expression (mRNA) was elevated in animals with mild, moderate, and marked severities of cardiotoxicity, but the elevations were most prominent in the two animals exhibiting reduced ejection fraction and/or marked cardiotoxicity. The expression of BNP in the recovery phase high-dose animals was comparable to the controls, which was consistent with recovery from the lesion. Plasma BNP peptide levels were measured by ELISA. However, the results were inconclusive because of the large individual variability and limited applicability of the reagent to monkey samples.
Microscopic examination of H&E–stained skeletal muscle revealed no evidence of BILN 2061–induced changes, whereas light microscopy of heart tissues revealed unequivocally time-dependent increases in incidence and severity of myocardial vacuolation (Figure 1 ). Control animals had normal heart histology (Figure 2A ), which was indistinguishable from tissues collected from monkeys sacrificed twenty-four hours after a single dose of BILN 2061. Myocardial vacuolation, characterized by the presence of myofibers containing clear-staining sarcoplasmic vacuoles, usually in clusters in cross-section and linearly arranged in longitudinal sections (Figure 2C), was observed in animals dosed for three or more days. Myocytes containing abundant vacuolation appeared swollen in moderate to marked cases of vacuolation.
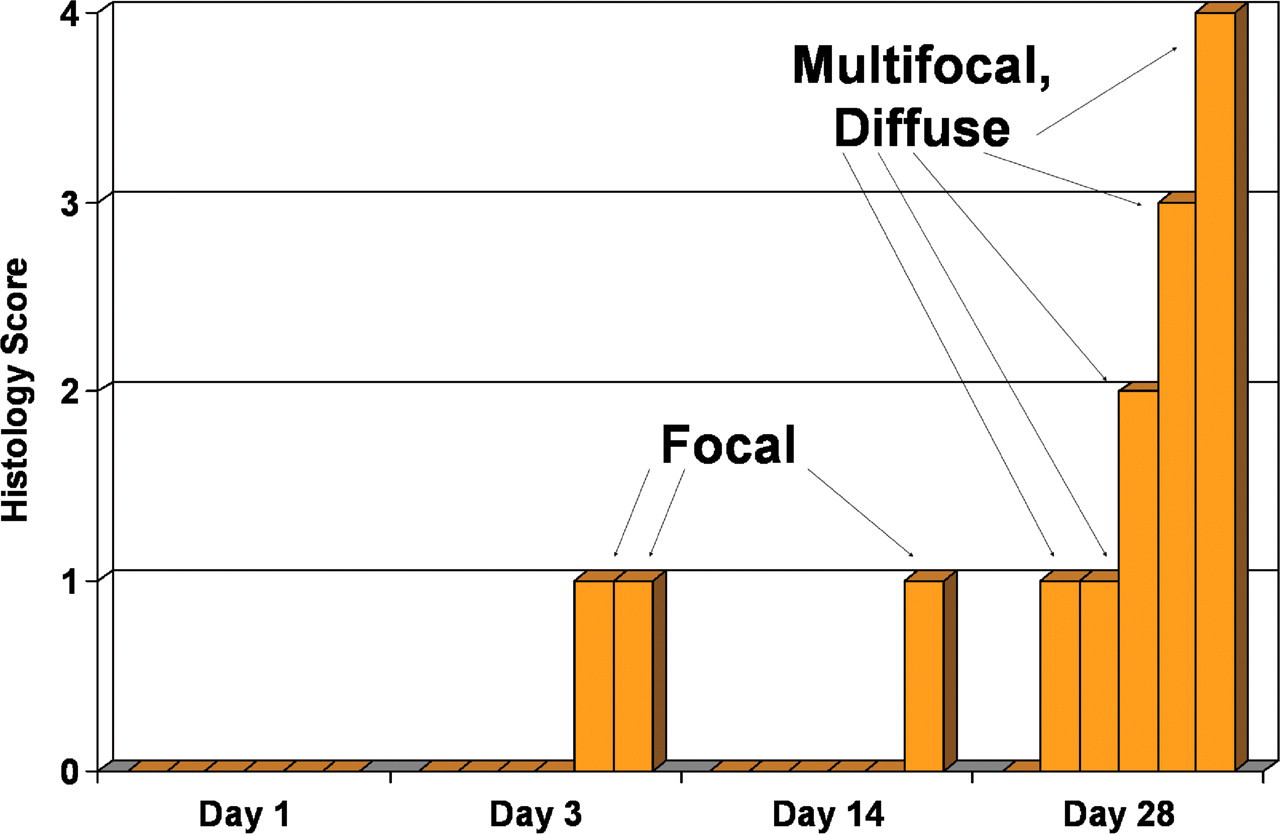
Graphical representation of the number of animals affected with light microscopic cardiac myocyte vacuolation and the severity of the change. Six orange boxes/bars at each time point represent six individual animals. Each time point represents the duration of dosing with 1,000 mg/kg/day of BILN 2061. The histology scores are 1 = minimal, 2 = mild, 3 = moderate, and 4 = severe. Day 1: no animals affected. Day 3: two animals affected with minimal focal vacuolation. Day 14: one animal affected with minimal focal vacuolation. Day 28: five animals affected with minimal/mild/moderate/severe multifocal to diffuse vacuolation.
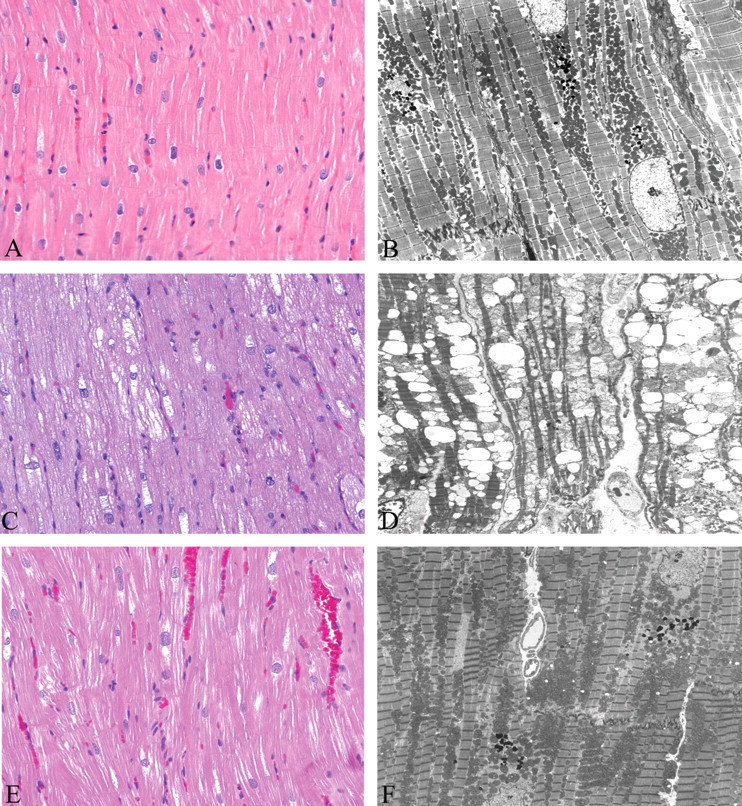
Light micrographs (A, C, E) and electron ultramicrographs (B, D, F) of heart tissue from a control animal (A, B), a BILN 2061–treated animal (C, D), and a BILN 2061–treated animal with a ten-week recovery period (E, F). Light microscopy: (A) Control animal from the drug phase of the study. Normal myocytes with homogeneous even staining of the sarcoplasm. 200×. (C) Treated animal from the drug phase of the study that received 1,000 mg/kg/day BILN 2061 for twenty-eight days. Multifocal myocytes with sarcoplasmic vacuolation. (E) Recovery-phase animal that received 1,000 mg/kg/day BILN 2061 for twenty-eight days (echocardiogram showed decreased ejection fraction) followed by ten weeks with no treatment. Normal myocytes with homogeneous, even staining of the sarcoplasm. Electron microscopy: (B) Control animal from the drug phase of the study. Normal myofibrils and normal mitochondria. (D) Treated animal from the drug phase of the study that received 1,000 mg/kg/day BILN 2061 for twenty-eight days. Mitochondria are swollen and often increased in size with moderate to marked electron lucency of the mitochondrial matrix. Cristae were shortened and/or reduced in number and displaced to the margins of the inner membrane. Myofibrils are displaced by the swollen mitochondria. (F) Recovery-phase animal that received 1,000 mg/kg/day BILN 2061 for twenty-eight days (echocardiogram showed decreased ejection fraction) followed by ten weeks with no treatment (echocardiogram returned to normal after recovery). Normal myofibrils with no evidence of mitochondrial swelling.
After three and fourteen days of dosing, minimal focal vacuolation was observed in two of six (33%) and one of six (17%) animals, respectively. After twenty-eight days of treatment, five of six (83%) animals were affected, with minimal (two of six), mild (one of six), moderate (one of six), and severe (one of six) multifocal to diffuse vacuolation. No degeneration/necrosis or inflammatory cell infiltrates were present in association with the vacuolated myocytes (Figure 2C). Special staining techniques revealed no evidence of collagen or lipofuscin deposition, or mineralization, in either control or dosed monkeys.
Electron microscopy was performed on heart tissue from all control and dosed animals (Figures 2B, D, F). The myocardial vacuolation observed with light microscopy correlated with swollen and cleared myocardial mitochondria (Figure 2D) and the severity and distribution of the ultrastructural change correlated with the histopathological lesion grading. Affected mitochondria were often increased in size. In the more minimal manifestations of mitochondrial injury, there was some lucency of the mitochondrial matrix, but the cristae appeared intact. In more severe changes, there was moderate to marked electron lucency of the mitochondrial matrix, and cristae were shortened and/or reduced in number and displaced to the margins of the inner membrane (Figure 2D). Occasionally, adjacent affected mitochondria appeared to have fused together. The inner mitochondrial membrane was focally disrupted in a few foci. Myofibrils were often displaced by the swollen mitochondria. No calcium precipitates were evident. Normal-appearing mitochondria were sometimes present adjacent to affected organelles in the same affected myocyte. In dosed animals, there was no evidence of increased lysosome formation, electron-dense aggregates, autophagy, or necrosis in any of the micrographs examined.
All animals afforded the ten-week recovery period were free of evidence of drug-related cardiotoxicity. Myocytes from dosed and control recovery group animals appeared normal by light microscopy, and all examined cells had normal complements of normal-appearing mitochondria (Figures 2E and 2F, respectively). Special stains revealed no evidence of collagen or lipofuscin deposition, fibrosis, or calcification, and there was no evidence of the presence of permanent ultrastructural cell injury (Figure 2F).
Electrocardiographic measurements were consistently within normal limits for all animals during all phases of the study. Of the echocardiographic parameters evaluated, in the pretest phase, LVEF data from all animals yielded a baseline range of 63–84% (Figure 3 ). Echocardiographic examinations of animals dosed one, three, or fourteen days revealed no functional cardiac abnormalities, although several of these animals exhibited myocardial vacuolation and mitochondrial swelling by light and electron microscopy. Examinations of animals dosed for twenty-eight consecutive days, however, revealed two with reduced cardiac LVEF (decreased to 55–56%). One of these animals, sacrificed on day 29, exhibited severe myocardial vacuolation with swollen mitochondria (Figures 2C, D), and the other, presumed to also have severe heart lesions after twenty-eight days of dosing, was placed in the ten-week recovery group. During the recovery phase, this animal’s reduced LVEF progressively improved, and by week 10 of recovery it was within the normal range (68%). Light and electron microscopic examinations of the post-recovery heart tissue from this monkey revealed no evidence of treatment-associated structural changes (Figures 2E, 2F).
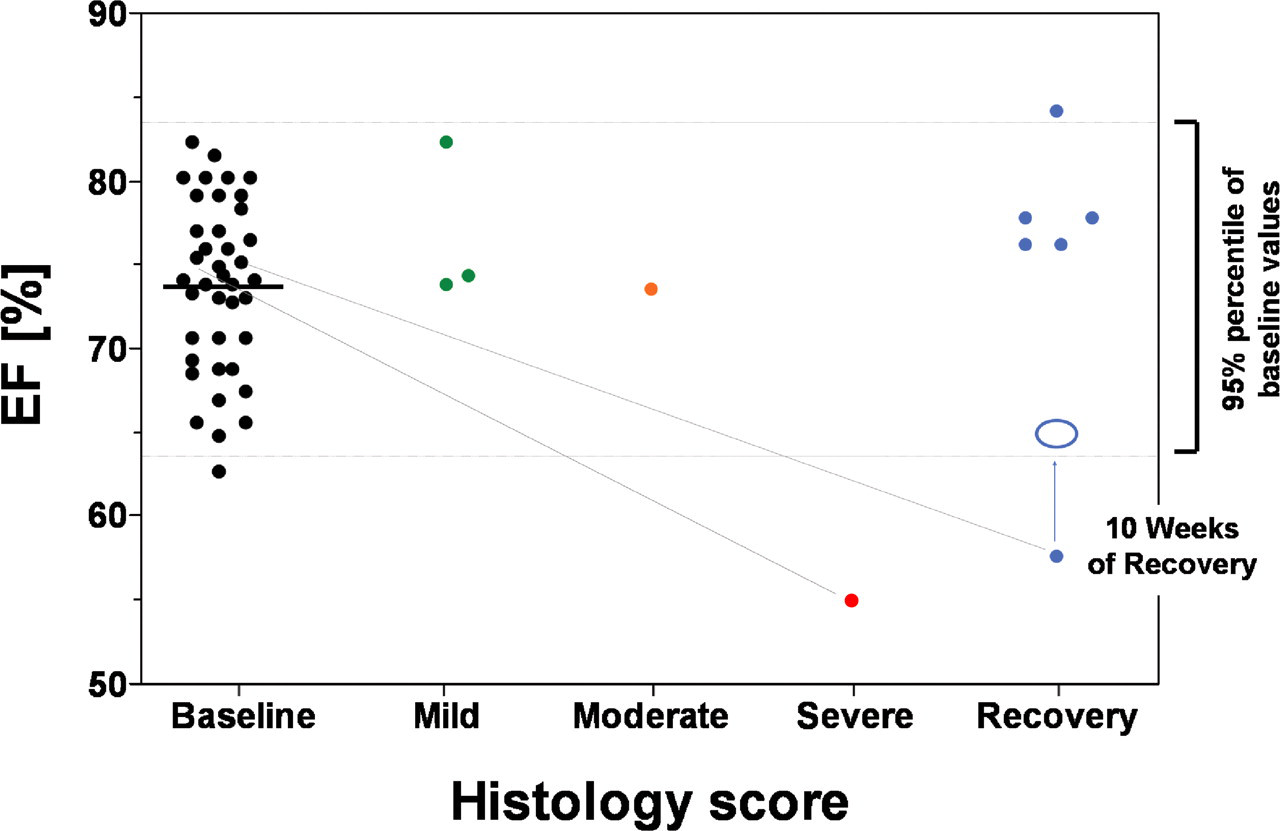
Graphical representation of cardiac myocyte vacuolation grading data (histology score) and ejection fraction data (EF). The black dots represent the normal ejection fractions for all study animals in the pretest phases of the study (range 63–84% EF). The green, orange, and red dots represent drug-phase animals after twenty-eight days of drug exposure with minimal/mild, moderate, and marked myocardial vacuolation, respectively. Note the animal with the severe lesion (red dot) and another animal placed in the recovery group (blue dot) have ejection fractions below the normal range (55–56%). The blue dots collectively represent the six drug-treated recovery animals after ten weeks in the recovery phase. The blue circle represents the animal in which the ejection fraction returned to normal (68%) after the ten-week recovery period. The hearts of all animals in the recovery group were histopathologically normal.
Discussion
Mitochondria, cytosolic organelles key in cellular ATP generation and calcium homeostasis, are composed of an outer and inner membrane, cristae, and an inner matrix. The outer membrane is similar to other cellular membranes with abundant cholesterol and ion permeability. The inner membrane is densely packed with proteins and contains almost no cholesterol (Scheffler 1999). The inner membrane is rich in cardiolipin, essential for the optimal function of the electron transport chain. The cristae are protein-rich, disc-shaped structures that are continuous with the inner membrane via tube-like structures. The inner membranes are impermeable to ions, which is critical in the maintenance of the electrochemical proton gradient that supplies energy for ATP generation. The inner matrix is composed of proteins (enzymes) and abundant calcium ions in an aqueous medium. Unlike skeletal muscle mitochondria, the myocardial organelle is incapable of anaerobic metabolism to support contraction, thus, myocardial myocytes contain more mitochondria than do skeletal muscles.
Mitochondria are intimately associated with the cytoskeleton and are anchored within the cell by microtubules and desmin, an intermediate filament. The mitochondria may relocate along the microtubules in the cell via an ATP-dependent mechanism. It is postulated that the location of mitochondria within a cell dictates the energetics level exhibited (Scheffler 1999). The lifespan of mitochondria varies among tissue types and even between individual cells within a tissue and depends on nuclear signaling and cellular energy demands (ATP). Mitochondrial degradation is initiated by ATP-dependent proteases located in the mitochondrial matrix, then cellular lysosomes engulf the degraded mitochondria by the process of autophagia.
Ultrastructural mitochondrial changes in response to injury have been described (Kumar et al. 2005). Early and reversible injury generally features organelle swelling, margination of the cristae, and rarefaction of the matrix secondary to an influx of aqueous fluid, and phospholipid-rich densities may form in the matrix. Affected adjacent swollen mitochondria may fuse. As the injury persists and progresses, calcium precipitates form in the matrix, and mitochondria may rupture. The ultimate fate of the cell containing injured mitochondria depends on the proportion of organelles that survive the intracellular insult. If sufficient intact mitochondria remain, the cell can retain its viability and, if the insult is removed, the mitochondria can repopulate the cell through mitochondrial division. If the insult progresses to irreversible injury, the myocyte undergoes necrosis (apoptosis or oncosis).
The cardiotoxic effects observed in the current study ranged from a relatively minimal mitochondrial swelling, with cristae appearing to be intact, to a more marked change, where there is nearly complete loss of the inner mitochondrial ultrastructure. The mitochondrial injury was apparent after as few as three daily doses, and both incidence and severity increased with continued administrations. The most severe effects were observed in animals that had been dosed for twenty-eight consecutive days.
Although drug-induced cardiotoxicity with mitochondrial swelling has been reported with other xenobiotics (e.g., anthracyclines), ultrastructural alterations beyond swollen mitochondria (e.g., dilated smooth endoplasmic reticulum, lipid vacuoles, lipofuscin accumulation) are also usually present. In contrast, BILN 2061–induced cardiotoxicity is unique in that the only ultrastructural abnormality present is the mitochondrial swelling. The rest of the cellular architecture is normal.
In our experience, outright cardiac myocyte degeneration in Rhesus monkeys exposed to high doses of BILN 2061 is rare. Historically, the degeneration was observed by light microscopy in approximately 10% of monkeys dosed with 500 to 1,000 mg/kg/day of BILN 2061 for twenty-eight days and was characterized by the presence of scattered individual hypereosinophilic and shrunken myofibers in the absence of any inflammatory cell infiltration.
Although echocardiographic changes were observed in two monkeys that exhibited severe cardiotoxicity, the potential utility of the changes as a biomarker for this type of myocardial injury is doubtful, because less-severe yet unequivocal cardiotoxicity was without effect on echocardiographic measures. The results of electrocardiography were without predictive utility for even the most severe cases of toxicity. The failure of troponin analysis to predict the presence of the lesion was not surprising, since myocyte degeneration and necrosis were never observed as prominent features of this drug–induced cardiotoxicity. Although the BNP gene expression in the heart tissue of animals with cardiotoxicity was increased over control animals, the plasma BNP peptide analysis data were inconclusive.
The soleus muscle was collected in this study to serve as a possible surrogate tissue to detect BILN 2061–associated injury in skeletal muscle. This muscle was chosen based on the fact that it is composed of an abundance of slow twitch type 1 muscle fibers. Both cardiac myocytes and soleus muscle type 1 fibers have abundant mitochondria in the sarcoplasm, and both metabolize predominantly fatty acids for the production of ATP. Based on these similarities, it was hoped that the soleus muscle would manifest a mitochondrial toxicity similar to that observed in affected cardiac myocytes; however, changes in this tissue typical of BILN 2061 cardiac myocyte toxicity were not observed. The reason for the soleus muscle’s resistance to toxicity is uncertain. One possible reason for the resistance of the soleus muscle type 1 fibers to BILN 2061 toxicity may be related to tissue drug distribution differences and/or low tissue concentration of the molecule in the skeletal muscle tissue.
Early clinical proof of principle studies with BILN 2061 failed to reveal evidence of cardiotoxicity in humans. Subsequent observation of high-dose cardiotoxicity in monkeys motivated an effort to identify reliable biomarkers for the early detection of this type of induced myocardial change to ensure safety during further clinical studies. To date, these efforts have been unsuccessful, and preclinical assessments of two BILN 2061 analogues revealed similar cardiotoxicity. Consequently, Boehringer Ingelheim has suspended its product development program with macrocyclic analogues of BILN 2061.
Footnotes
Acknowledgment
The authors would like to thank John Mennear for his expert review of this manuscript in preparation for submission for publication.
