Abstract
This manuscript is intended to provide a best practice approach to accurately and consistently assess toxicant-induced bone marrow effects of test articles. In nonclinical toxicity studies, complete blood count data in conjunction with the histological examination of the bone marrow are recommended as the foundation for assessing the effect of test articles on the hematopoietic system. This approach alone can be used successfully in many studies. However, in some situations it may be necessary to further characterize effects on the different hematopoietic lineages, either by cytological or flow cytometric evaluation of the bone marrow. Both modalities can be used successfully, and which one is selected will depend on the expertise, preference of the facility, and the nature of the change in the bone marrow. Other specialized techniques such as clonogenic assays or electron microscopy are used rarely to further characterize hematotoxicity. The indications and techniques to successfully employ histological, cytological, or flow cytometric evaluation as well as clonogenic assays and electron microscopy are reviewed.
Summary Of Major Recommendations
Regulatory Guidance
Few detailed recommendations are available from regulatory agencies on the assessment of the hematopoietic system. Nonclinical toxicity evaluation of test articles must usually include evaluation of a rodent and a nonrodent species. Therefore, pathologists must be familiar with the normal microarchitecture and cytological features of the bone marrow of multiple species, including but not limited to the mice, rats, dogs, and nonhuman primates. Concurrent controls should be evaluated together with the appropriate test article–treated groups. Successful evaluation of the hematopoietic system will depend on examination of good-quality samples by an experienced individual correlating the findings in the hematologic, histological, toxicokinetic, and in-life data as well as cytological and or flow cytometric data, if available.
Histological Evaluation
Histological evaluation alone will provide an assessment of overall hematopoietic cellularity, can identify subtle focal or multifocal lesions not easily identified by smear evaluation (i.e., necrosis or inflammation), and may allow for a general estimation of the proportion and maturation of granulocytic and erythroid cells, as well as megakaryocyte cellularity. Histological evaluation may provide data not obtained by cytologic evaluation, such as changes to the structural organization of the hematopoietic environment, endosteum, bone, interstitium, adipose tissue, and vasculature.
Cytological Evaluation
The need for cytological evaluation of bone marrow smears should be decided on a case-by-case basis. If the evaluation of the combined study data indicates that characterization of the hematopoietic system was, or will be, inadequate using hematology and histopathology, then cytologic evaluation of the bone marrow may be considered. Noteworthy situations that may warrant cytological examination of bone marrow smears include: a need to differentiate early hematopoietic precursors, the desire to investigate incompletely characterized increases or decreases in hematopoietic cellularity of the bone marrow relative to the peripheral blood cell numbers, and the requirement to determine whether histological changes in bone marrow cellularity reflect alterations in lymphoid versus erythroid cells. Cytological assessment may also be considered if there are uncharacterized atypical or abnormal blood cells in circulation, or abnormalities in erythrocyte indices (mean corpuscular volume [MCV], mean corpuscular hemoglobin concentration [MCHC], etc.) suggestive of abnormal erythropoiesis. Cytologic evaluation may not be necessary when decreased bone marrow cellularity is caused by decreases in food consumption or when peripheral blood lymphocyte counts alone are decreased. Cytological examination also may not be necessary when there are appropriate (responsive) increases in peripheral blood cell counts and/or bone marrow cellularity, or if there are granulocyte or platelet dysfunction disorders.
Flow Cytometric Evaluation
Flow cytometry can be used in addition to or instead of cytological examination to further characterize changes in the bone marrow. Indications for flow cytometric examination include criteria similar to those stated above for cytological assessment. However, if the changes present in the bone marrow need detailed morphological assessment, then cytological examination would be the preferred technique.
Hematopoiesis
The bone marrow is the major hematopoietic organ and a primary lymphoid organ responsible for the production of erythrocytes, granulocytes, lymphocytes, monocytes, and platelets (Travlos 2006a). Since the bone marrow is a site of intense cell multiplication and maturation, it can be influenced by drugs that affect hematopoietic cell types specifically or cellular proliferation/differentiation nonspecifically. Overall, hematotoxicity that is preclinically identified predicts toxicity in human clinical trials, with a 91% concordance (Olson et al. 2000). The successful production of blood cells by the bone marrow depends on a complex interplay between hematopoietic stem cells, committed progenitor cells, and the bone marrow microenvironment, which includes adventitial reticular cells, adipocytes, macrophages, osteoblasts, and extracellular matrix material (Gasper 2000; Meyer and Harvey 1998; Weiss and Geduldig 1991). A basic understanding of this process, which leads to hematopoietic cellular differentiation and release of these cells into the circulation, is important to accurately interpret the changes in bone marrow and blood that may be seen in nonclinical toxicity studies.
Regulatory Guidelines for Bone Marrow Evaluation
Regulatory guidelines from agencies responsible for the investigation of the safety and efficacy of human health products—the United States Food and Drug Administration (FDA); the European Medicines Agency (EMA); and Japan’s Ministry of Health, Labor, and Welfare (MHLW)—and for pesticides and toxic substances evaluation (U. S. Environmental Protection Agency [EPA]) contain few detailed recommendations regarding bone marrow cytological and histological evaluation in nonclinical toxicity studies. None of the guidance documents gives specific recommendations on bone marrow harvesting techniques and slide preparation or bone marrow cytological or flow cytometric evaluation. They also do not provide any indication on the exact decision criteria for when bone marrow cytological or flow cytometric assessment should be undertaken. The recommendations of the different agencies are briefly discussed below.
The Redbook from the Center for Food Safety and Applied Nutrition (CFSAN) of the FDA (FDA November 2003) recommends obtaining blood samples for hematological evaluation from a minimum of ten rodents of each sex per group at least three times during a subchronic study. Bone marrow histological sections from the sternum should be prepared from all animals. All tissues from the animals in the control and high-dose groups should be examined initially, and if there are treatment-related effects, then the next lower dose level tested should be examined until a no observable-effect level (NOEL) is established. Bone marrow cytology slide preparation from each animal is recommended, but these slides would need to be examined microscopically only if effects on the hematopoietic system were noted.
The Center for Drug Evaluation and Research (CDER) and the Center for Biologics Evaluation and Research (CBER) of the FDA have issued guidelines for immunotoxicity studies for human pharmaceuticals (Food and Drug Administration April 2006). The EMA adopted similar guidelines for immunotoxicity testing during the same period (EMA May 2006). They underline the importance of hematological evaluation and bone marrow histological evaluation, and they mention that cytological evaluation would be appropriate if either unexplained alterations in peripheral blood cell lines or histopathological findings are observed.
The EMA also refers to bone marrow cytological evaluation in a Note for Guidance on Repeated Dose Toxicity (European Medicines Agency July 2000). During these studies, bone marrow from the sternebrae, femur, or vertebrae should be studied histologically in high-dose and control groups from rodents, and in all animals in nonrodent studies. Hematological parameters should be monitored as well. Bone marrow cellularity should be evaluated terminally, but the guidance does not recommend a specific method. Microscopic evaluation of bone marrow cytology is not directly requested, but the guidance refers to a previous publication (Weingand et al. 1996) stating that bone marrow cytology is not routinely necessary as a screening test in nonclinical toxicity studies unless appropriately indicated by a test article effect on the erythron, leukon, or thrombon.
In a Guideline for Toxicity Studies of Drugs (Yakuji Nippo 1999), the Japanese MHLW recommends hematological examination on all animals during repeated-dose studies, but also for practical consideration, it accepts limited evaluation of some animals in each group. Bone marrow is indicated as a tissue to evaluate histologically, but the guideline does not mention bone marrow smear evaluation.
The Center for Veterinary Medicine (CVM) guidance on Target Animal Safety for Veterinary Pharmaceutical Products (FDA May 2007) recommends conducting hematology testing at several points during repeated-dose studies and lists bone marrow sections and marrow smears as analyses to consider. The EPA lists bone marrow as a tissue to evaluate histologically, whereas bone marrow cytological evaluation and reticulocyte counts may be warranted if there is a test chemical effect on the hematopoietic system (EPA 1988).
Bone marrow cytological evaluation is not specifically addressed in the EMA, FDA, EPA, or Japanese MHLW guidance documents on carcinogenic potential (EPA August 1998; EMA July 2002; FDA July 1997; Yakuji Nippo 1999). The EMA, EPA, and the Japanese MHLW guidance documents list bone marrow as a tissue to evaluate histologically.
Recommendations for Performing Histological Evaluation of the Bone Marrow
Examination of standard hematologic parameters and bone marrow histology in nonclinical toxicity studies should be used as the first step in evaluating the effect of a test article on the hematopoietic system, and it is often sufficient to provide an accurate assessment. Evaluation and interpretation of hematology and histology within a study requires consideration of in-life findings, toxicokinetic data, concurrent control data, and the dose-response relationship. Bone marrow should be examined judiciously since the histomorphology may reflect changes resulting from moribund condition, body weight loss, or decreased body weight gain, which could be mistakenly concluded to be test article–related.
More comprehensive evaluation of test article–related effects on hematopoiesis, including cytologic evaluation of bone marrow smears and/or flow cytometry, is indicated in instances in which further characterization of an effect is needed. Bone marrow smears can be made at the time of necropsy, which allows for possible future analysis by cytological evaluation. Additional techniques, including clonogenic hematopoietic progenitor cell assays and electron microscopy, can be applied to elucidate mechanistic information of test articles that have demonstrated hematopoietic effects.
Histological evaluation alone will provide an assessment of overall hematopoietic cellularity, can identify subtle focal or multifocal lesions not easily identified by smear or flow cytometric evaluation (i.e., necrosis or inflammation), and may allow for a general estimation of the proportion of granulocytic to erythroid cells, iron stores (particularly with the use of special stains for iron), megakaryocyte cellularity, and gross megakaryocyte morphology. Histology can also identify changes to the structural organization of the hematopoietic environment, endosteum, bone, interstitium, adipose tissue, and vasculature. The histological changes identified in the bone marrow are interpreted with the complete blood count data to make an assessment of the effect of the test article on the bone marrow. The quality of the histological evaluation depends on the quality of the sample evaluated; the techniques for properly collecting samples will be addressed below. Additionally, wide animal-to-animal variation and sampling site- and age-related differences (Cline and Maronpot 1985; Weiss 1986) necessitate the review of potential treatment-related effects against appropriate concurrent controls. In rodents, the results of the histological examination of the spleen are important in the overall assessment of the hematopoietic system because the spleen is often a site of prominent hematopoiesis in these species. The spleen less commonly shows these reactive alterations in adult dogs and nonhuman primates.
Recommendations Regarding When to Perform Bone Marrow Cytological Examination
Whether or not to perform cytological evaluation of bone marrow smears should be decided on a case-by-case basis by the pathologists (clinical pathologist and anatomic pathologist) in consultation with the study director after assessing the hematologic data, histopathologic findings, in-life observations, and toxicokinetic data. Dose response, tolerability, severity, and reversibility of the combined study findings, as well as the stage of development of the drug, should also be taken into account. If the evaluation of the combined data indicates that characterization of the hematopoietic system was inadequate using the standard assays, (i.e., histological evaluation and complete blood count), then cytologic evaluation of the bone marrow may be considered. In addition, the impact on or usefulness of the bone marrow smear results in the clinical safety plan should be considered. When cytologic examination of bone marrow smears is necessary, the examination may be conducted at any stage in the development of a drug; however, routine cytological examination of bone marrow smears is not recommended for carcinogenicity studies. In cases in which examination of bone marrow smears was conducted in an earlier study, it may not be necessary to evaluate in later studies, but the need for this assessment should be decided on a case-by-case basis. Below is a noninclusive list of situations that may warrant cytological examination of bone marrow smears if evaluation of other study data was deemed insufficient to adequately characterize findings in the hematopoietic system.
Differentiation of Early Hematopoietic Precursors
Histology alone is insufficient to differentiate between all types of immature hematopoietic precursors. Therefore, if differentiation or identification of early stages of cellular development is deemed necessary by the pathologists in consultation with the study director, then evaluation of bone marrow smears may be considered. Examples of situations in which detailed evaluation of early precursors may be considered include atypical morphology of leukocytes that is unrelated to inflammation, sepsis, antigenic stimulation, known pharmacologic effect of the drug, and/or drug-induced phospholipidosis (vacuolation), and cases of suspected drug-induced hematopoietic neoplasia that are not sufficiently classified by evaluation of blood smears, hematology data, and/or histopathology.
Additional Characterization of Effects on Hematopoietic Cell Lines
Significant decreases in single or multiple blood cell lines may warrant cytological examination of the bone marrow if routine evaluation of hematology and histology data was insufficient to characterize the effect. Examples include significant decreases in blood neutrophil counts in a nonclinical toxicity study, with no evidence of an underlying response to acute tissue inflammation or known antiproliferative effect of the test article; marked, persistent, and/or consistent decreases in peripheral eosinophil counts with no evidence of physiological stress or glucocorticoid-like effects; significant decreases in platelet counts with no evidence of an underlying use, sequestration, or destruction of platelets; or marked, persistent, and/or consistent decreases in monocyte counts. Significant increases in blood cells may also warrant cytological examination of the bone marrow if routine evaluation of hematology and histology data was insufficient to characterize the effect. For example, significant increase in the numbers of red blood cells (absolute erythrocytosis) that is not caused by dehydration or hemoconcentration, pharmacology (i.e., recombinant erythropoietin, hormones, cytokines), or known modulation of erythropoietin production by the test article; marked increases in neutrophils that are not caused by inflammation or necrosis, excitement/stress response (i.e., catecholamine/glucocorticoid-related), or administration of pro-inflammatory cytokines; and persistent or marked increases in platelet counts that are not caused by increased muscular activity, blood loss, iron deficiency, inflammation, and/or administration of growth factors or cytokines.
Determining Whether Changes in Bone Marrow Cellularity Result from Changes in the Erythroid versus Lymphoid Population
Although it is possible to differentiate some granulocytic cells from erythroid cells on histological samples, differentiation of the erythroid series from the lymphoid cells is more challenging. This differentiation is of increased relevance in rodents since these species have greater numbers of lymphoid cells than do dogs or primates. Therefore, if differentiation of erythroid versus lymphoid precursors is deemed necessary, then bone marrow cytology should be considered.
Changes in Erythrocyte Indices or Red Cell Morphology
Changes in red cell indices such as MCV and MCHC may suggest abnormal erythropoiesis or neoplasia and may be accompanied by large or megalocytic erythroid cells in the blood. If detection/confirmation and detailed characterization of abnormal erythropoiesis (e.g., as suggested by megalocytes or megaloblastic changes) or other red cell developmental morphologic abnormalities (such as siderocytes or basophilic stippling) is deemed necessary, then bone marrow cytological assessment may be appropriate, since compared to the peripheral blood, large numbers of these abnormal erythrocytes may be found in the bone marrow.
Atypical Changes in Leukocyte Morphology in the Peripheral Blood That Are Unrelated to Inflammation (i.e., “toxic” changes), Antigenic Stimulation (i.e., lymphoid reactivity), or Known Effects of the Drug (i.e., vacuolation caused by phospholipidosis)
Atypical leukocytes observed in peripheral blood may be indicative of abnormal leukocyte maturation, neoplasia, and/or immune modulation. Atypical cells may be characterized by immaturity, altered size, abnormal cytoplasmic vacuolation, altered nuclei/cytoplasmic ratios, asynchrony of the nucleus and cytoplasm, altered chromatin, alterations in stain affinity, the presence of atypical granules, and/or other cytological alterations. In these situations, bone marrow cytological evaluation may help characterize the extent of the test article effect in hematopoietic precursors.
Conditions/Situations in Which Cytologic Evaluation of the Bone Marrow May Not Be Necessary
As indicated above, cytologic evaluation of the bone marrow should be considered on a case-by-case basis after evaluation of other study data, including peripheral blood data and histopathologic findings. If the available study data indicate that the hematopoietic system was adequately evaluated, then cytologic evaluation of bone marrow smears may not be necessary. Below is a noninclusive list of additional situations in which cytologic evaluation of the bone marrow may not be necessary.
Changes in Bone Marrow Cellularity Occur Concurrently with Decrements in Food Intake
Decreased food intake may have a major impact on the hematopoietic system, particularly in rats, and these effects should not be confused with direct test article–related findings. Body weight and food consumption data should be evaluated in conjunction with hematological parameters and bone marrow histopathology. Several published studies have shown that the effect of decreased food consumption on the hematopietic system is proportional to the degree of food restriction and the length of the study. For example, 25% to 75% food restriction studies in rats (Wistar or Sprague Dawley) demonstrated marked changes in hematopoietic cell production. In studies lasting from two weeks to two years, important decreases in leukocytes (neutrophils, lymphocytes, and monocytes), decreases in reticulocytes and platelets, and reduction of erythroid precursors in bone marrow were observed, along with decreased body weight (Ogawa et al. 1985; Seki et al. 1997). In rats subject to significant food restriction (75%), necrosis and bone marrow degeneration were observed after only two weeks (Levin et al. 1993). In mice, decreased protein intake was associated with bone marrow cellular depletion and a halt of the precursor cell cycle (Borelli et al. 2009). Effects of food restriction on the hematopoietic system have been documented less often in other species, but similar effects of lower magnitude have been observed in dogs (Hill et al. 2005; Lawler et al. 2007).
Changes in Peripheral Blood Lymphocyte Numbers
Bone marrow lymphocyte density does not correlate well with peripheral lymphocyte counts (Yoffey and Courtice 1970), and the number of lymphocytes in peripheral blood may be influenced by rates of production, recirculation and use, or destruction of lymphocytes. Recirculating lymphocytes spend more time in specific organs than in peripheral blood, hence, increases or decreases in peripheral lymphocyte numbers do not necessarily reflect altered lymphopoiesis. Therefore, bone marrow cytological evaluation will likely not add value when assessing most changes in peripheral lymphocyte counts.
Responsive or Regenerative Changes in the Bone Marrow or Blood
In these situations, evaluation of hematologic and histopathologic data should be sufficient for assessment of the hematopoietic system. For example, increased cellularity of the bone marrow is often observed as an appropriate response to increases in cell turnover. Decreased numbers of red blood cells in the blood may be accompanied by histologically recognizable increases in the proportion or number of erythroid cells in the bone marrow (i.e., erythroid hyperplasia). Increases in the number or proportion of myeloid cells (i.e., granulocytic hyperplasia) is often associated with inflammation. Increases in the number (i.e., megakaryocytic hyperplasia) or changes in the morphology of megakaryocytes can develop in association with peripheral consumption of platelets and inflammation.
Platelet or Granulocyte Dysfunction
Platelet or granulocyte dysfunction are rarely associated with morphologic changes, so cytological assessment of the bone marrow would be of little value (Andreasen and Roth 2000; Boudreaux 2000). Platelet dysfunction is typically characterized by increased bleeding time with concurrent normal platelet counts, and normal coagulation times. Platelet aggregation testing and/or immunological evaluation of surface adhesion molecules are possible methods to further characterize platelet functional deficiencies. Acquired drug-related granulocyte functional defects may be caused by decreased adherence, chemotaxis, phagocytosis, or degranulation of leukocytes. Typically animals have persistent and progressive peripheral neutrophilia with hypersegmented neutrophils, decreased neutrophilic tissue infiltration, and recurrent pyogenic infections. The bone marrow granulocytic compartment is usually increased, and bone marrow histopathological assessment together with the hematology evaluation should be adequate to evaluate the bone marrow. Evaluation of neutrophil surface markers and/or granule content may also be useful.
Recommendations on When to Use Flow Cytometry to Evaluate the Bone Marrow
Similar to cytologic evaluation of the bone marrow, flow cytometry can be used to provide a more detailed assessment of drug-induced effects on hematopoiesis when compared to evaluation of hematologic data and histological examination. Flow cytometric evaluation of the bone marrow should be decided on a case-by-case basis by the pathologists in consultation with the study director and should be used if there is insufficient characterization of the effects of a test article on hematopoiesis after evaluating the hematologic data, histopathologic findings, in-life observations, and toxicokinetic data. The stage of development of the drug, technical capabilities or expertise of the laboratory, and understanding the known or expected hematopoietic effects of the test article should also be considered. Flow cytometry, when appropriately executed, may be used in place of manual differential counts of bone marrow hematopoietic cells and thus can be applied to many of the situations outlined above for cytological assessment. However, flow cytometry cannot assess cellular morphologic changes, and thus bone marrow smears or cytospin preparations of bone marrow cell suspensions should always be collected in case morphologic evaluation is necessary. If it is determined that flow cytometry will be used for bone marrow assessment, this decision must be made prior to the end of the in-life portion of the study, since cells cannot be preserved long term. Therefore, this analysis cannot be made retrospectively as can be done with cytological smears. Flow cytometry is efficient at classifying the hematopoietic cells into the major categories and in some cases, depending on the method used, can further classify the cells into the proliferating and nonproliferating pools. However, the standard flow cytometric techniques cannot provide complete classification of all the different cell types as can cytological assessment. Therefore, flow cytometry can be used to assess major changes in neutrophilic granulocytic, lymphocytic and erythrocytic lineages; this type of assessment may provide the desired level of additional characterization needed for many studies. However, more extensive assessment of these lineages, their development orderliness, as well as detailed characterization of the eosinophilic, monocytic, basophilic, and megakaryocytic cell populations are not easily accomplished using the standard flow cytometric techniques that are reviewed below, although tools can be developed to address these issues. In addition, if detailed cellular morphologic examination of the bone marrow is critical (for example, in evaluating megaloblastic changes in the erythroid lineage), it should be done by cytological examination because flow cytometry does not permit this type of assessment.
There are some significant advantages of flow cytometry over a microscopic differential cell count, including the speed with which data can be acquired and the precision of the technique, based on the much larger number of cells that are evaluated (10,000–35,000) by this method compared to the manual method, which evaluates notably fewer cells (300–500). Furthermore, flow cytometric methods are not dependent on the training of the microscopist or the quality of the cytological slide preparations for accurate cell classification. Not only can the relative percentage of major cell populations be evaluated, but in rodent species, the absolute counts of the different cell populations can also be assessed if the entire marrow of a long bone is processed and a total nucleated cell count is determined. The choice to use flow cytometry versus microscopic examination will depend on the type and amount of technical expertise, the instrumentation availability, the historical experience at a given facility, as well as the nature of the change in the bone marrow.
Recommendations Regarding When to Employ Specialized Bone Marrow Techniques
Although it is not often necessary to employ additional specialized techniques as part of the routine evaluation of bone marrow in nonclinical safety studies, clonogenic assays and electron microscopy may be useful in certain instances to further characterize the mechanism of bone marrow toxicity. These evaluations may be part of a separate investigative study or might (if planned for prospectively) be done in conjunction with a nonclinical toxicity study. It is recommended that clonogenic assays be used when an in vitro system is needed to better understand the effect of the test article on the ability of different lineages of hematopoietic cells to proliferate and differentiate. Electron microscopy (usually the transmission rather than the scanning modality) should be considered as a tool to answer specific questions about a morphologic change that cannot be resolved at the light microscopic level to better understand the pathogenesis of the hematotoxicity. Specifically, electron microscopy may be indicated to investigate test article–induced cellular inclusions or effects on subcellular structures or organelles (cell–cell junctions, nucleus, endoplasmic reticulum, lysosomes, etc.); abnormal extracellular matrix deposition; test article–induced injury to hematopoietic, stromal, vascular, osteogenic, or adventitial cells; and/or changes in the bone marrow microenvironment.
Recommendations on How to Collect Samples and to Perform Histological Evaluation of Bone Marrow
Bone Marrow Collection and Processing
Collection
The goal of sample collection is to obtain samples of good quality that reflect the physiological state of the bone marrow. Timely collection of bone marrow samples for histology is important to provide the best quality specimens. In our collective experience, samples taken within twenty minutes of death provide good quality with minimal post mortem artifact; however, shorter times are best, particularly if cytologic smears are needed (see cytology section below). For large laboratory animal species (e.g., dogs, nonhuman primates, pigs), any bone that contains red marrow can be evaluated (e.g., rib, sternum, vertebrae, proximal humerus/femur, and ilium). In these species, the central (diaphyseal) marrow cavities of long bones may be almost entirely replaced by fat, so these sites should be avoided. For small laboratory animal species (e.g., rat, mouse, guinea pig), samples of bone marrow for histological examination may be acquired from any one of several bones with hematopoietically active marrow (e.g., sternum, rib, humerus, and femur). Regardless of age, the distal tibia of the rat has a demonstrable lack of active hematopoiesis, suggesting that this site is unacceptable for histological bone marrow evaluations (Cline and Maronpot 1985). The sternum and distal femur are sites that have been routinely used for bone marrow histology in small laboratory animals.
When a saw is used to obtain samples of bone marrow at necropsy, the heat generated by the sawing process may damage the tissue adjacent to the cut and can result in sampling-attributable artifact. Therefore, tissue sections should be taken at a distance from the saw cut. Attached muscle should be trimmed from the periosteal surface and, particularly in large samples, the bone should be opened at one or both ends to allow direct access of fixative into the marrow cavity. More detailed discussions regarding ante- or postmortem sample collection methods in large and small animal species are available in other references (Bono et al. 2000; Cline and Maronpot 1985; Feldman and Seely 1988; Fero 2005; Goodwin and Jerome 1987; Grindem 1989; Klein et al. 1991; Tyler and Cowell 1989).
When necessary, bone marrow core biopsies or aspirates for histological or cytological evaluation may be collected from a live animal when interim samples are necessary to understand the kinetics of a hematologic response. For live animal collection, bone marrow core biopsies or aspirates in the dog and nonhuman primate are best obtained from the iliac crest because hematopoietic cellularity at this site is most consistent and varies less with age than at other sites (Kushida et al. 2002; Penny and Carlisle 1970).
Tissue Fixation
Adequate tissue fixation is essential for the production of high-quality sections of bone marrow (i.e., preservation of morphological detail) and may be achieved using one of several fixative solutions. Because of its use as a routine general purpose fixative and ability to provide adequate fixation, 10% neutral buffered formalin is recommended for use in routine nonclinical toxicity studies (Bain et al. 2001; Burns and Bretschneider 1990; Hedrich and Bullock 2004; Kiernan 1990; Weiss 1987). At least a 10:1 volume-to-volume ratio of fixative to sample should be used, and samples should be allowed to fix for at least twenty-four hours prior to processing. Careful consideration must be given in advance to alternative methods of fixation or to cryopreservation of fresh frozen tissue if specialized evaluations are necessary, such as immunohistochemistry or molecular methods.
Decalcification
Paraffin-embedded, decalcified sections of bone marrow are recommended for routine use in nonclinical toxicity studies because of the practicality, familiarity, and ease of use, and because well-prepared tissues are of adequate diagnostic quality for safety evaluation. There are several types of chemical decalcifying agents (such as organic acids, mineral acids, chelators) and physical decalcification techniques (such as immersion, sonication, microwave) for decalcification of bone marrow specimens. Decalcification by tissue immersion in a chelating agent, such as ethylene diamine tetraacetic acid (EDTA) or a weak organic acid, is recommended. The main differences between these two options is that the chelating approach will require more exchanges into fresh fluid and will require a longer period for complete decalcification, but chelation may also provide better preservation of fine morphological details. The decalcification process may result in considerable shrinkage artifact if improperly performed. Weak organic acids (e.g., formic or acetic) may lead to loss of cytological detail and may impair metachromatic staining. Stronger mineral acids (e.g., hydrochloric and nitric), commonly used in the rapid-type decalcification methods, may also be used but can result in unacceptable sections if improperly performed. Acid and chelation decalcification techniques may remove iron from the tissue, although less stainable iron is lost with EDTA. Either EDTA or weak acid decalcification can also be used for enzyme- and immunostaining procedures. Additional details of decalcification procedures are reviewed elsewhere (Bain et al. 2001; Callis and Sterchi 1998; Kiernan 1990; Weiss 1987).
Embedding and Sectioning
Bone marrow embedded in paraffin provides adequate resolution and detail for most routine histological evaluations and thus is recommended. An obvious advantage of paraffin embedding is that this method can be applied in essentially any diagnostic histology laboratory using automated processors. Routine, approximately 5-µm sections are recommended. Alternatively, embedding in plastic may be considered in situations in which increased resolution of cellular detail and reduced shrinkage artifact are needed. This method also eliminates the need for a decalcification step. The reader is directed to the list of references for additional information on embedding of tissues (Bain et al. 2001; Burns and Bretschneider 1990)
Staining
Hematoxylin and eosin (H&E) staining is adequate and recommended for evaluation of the bone marrow in routine nonclinical toxicity studies. Should hematology and other study data indicate the need for a more detailed examination of bone marrow histology, Romanowsky (e.g., Giemsa) and Perl’s iron stain (Prussian Blue reaction for iron) are commonly used. The Romanowsky stains can be useful in the identification of erythroid precursors, plasma cells, and mast cells, and differentiation of neutrophil and eosinophil granules (Bain et al. 2001). Acid-decalcified tissues have some loss of basophilic staining with the Giemsa stain, whereas the use of chelating-type decalcifying agents (i.e., EDTA) improves Giemsa staining. Perl’s iron stain is used for the assessment of tissue iron stores. Additional information regarding embedding, sectioning, and staining (including histochemical and immunohistochemical) techniques can be found elsewhere (Bain et al. 2001; Beckstead et al. 1981; Bennett et al. 1976; Burns and Bretschneider 1990; Kiernan 1990; Moosavi et al. 1981; Weiss 1987).
Histological Evaluation of the Bone Marrow
It is essential that the pathologist has a good working knowledge of bone marrow microanatomy in the test species and performs a systematic examination of all bone marrow components to achieve a thorough evaluation. It is also important that bone marrow from animals in the treated groups is compared to concurrent controls. Initially, the entire section should be scanned at low magnification (i.e., 40×) for a general evaluation of section and staining adequacy. At this point, bone marrow sections are evaluated for adequate size, composition (e.g., composed primarily of cortical bone with associated marrow), and evidence of crush or other artifactual change. At this magnification, an overall estimation of bone marrow cellularity (comparing the approximate proportion of hematopoietic to adipose tissue), including the distribution and numbers of megakaryocytes, should be performed. Bone abnormalities and focal marrow lesions (e.g., metastatic infiltrates, granulomas) may also be apparent at this magnification.
Following the initial low-magnification review, hematopoietic and stromal components, hemosiderin, and lesions observed on low magnification should be evaluated at higher magnifications (100×–400×). Trabecular bone thickness should be examined along with the presence and numbers of osteoblasts, osteoclasts, and Howship’s lacunae, particularly if the bone marrow section is the only bone tissue to be evaluated in a study.
Overall hematopoietic cellularity should be evaluated, and a general estimate of the proportions of granulocytic to erythroid cells (or myeloid to erythroid-lymphoid cells in rodents) in control and test article–treated animals should be assessed. This step should be followed by an examination of the individual hematopoietic tissue cell lineages. In paraffin–embedded, H&E–stained sections, some stages of the erythroid and granulocytic cells, adipocytes, macrophages, and megakaryocytes can be readily identified. However, hematopoietic stem cells, very immature granulocytic and erythroid cells, mast cells, lymphocytes, monocytes, and stromal cells may not be easily identified because of low cell numbers or indistinctive morphology. Generally, detailed evaluation of individual cells is better performed by light microscopic examination of bone marrow cytology preparations.
The granulocytic and erythroid series should be evaluated for cell morphology, relative proportions of immature and mature precursors, synchrony of maturation, and evidence of dysplasia or neoplasia. Abnormalities in the relative proportions of the specific granulocytic lineages (neutrophilic, eosinophilic, basophilic) should be evaluated. Megakaryocytes should be examined for overall cellularity, location, presence of clusters, and morphology, including size and nuclear lobularity.
Because of small size and difficulty differentiating from erythroid precursors, unequivocal identification of individual lymphoid cells in decalcified, H&E–stained, paraffin-embedded sections of bone marrow is not readily accomplished. Despite the difficulty in visualizing individual lymphoid cells, aggregates of lymphocytes are straightforward to detect and have been reported in marrow sections of healthy dogs (Weiss 1986), in nonhuman primates with retroviral infections (Lowenstine 2003), and in wild-caught nonhuman primates (Ito et al. 1992). Lymphoid cell aggregates or lymphoid follicles are not usually found in normal rodents (Frith et al. 2000; Geldof et al. 1983).
For further detailed discussion of histological bone marrow examination, the reader is directed to other reviews on bone marrow evaluation (Andrews 1998; Bain et al. 2001; Buckley 1995; Elmore 2006; Travlos 2006b; Valli et al. 2002; Wickramasinghe et al. 1992).
Reporting of Histopathology Findings
The occurrence of background or incidental findings may be recorded in the individual animal data if desired, but in general, such data are not included in the pathology narrative unless the pathologist is unsure as to its relationship to test article administration. Samples of inadequate quality for evaluation should be recorded as such in the individual animal data together with a brief indication as to why it was nondiagnostic (for example, autolysis, not collected, etc.). The pathology narrative should indicate whether evaluation of the bone marrow at any given dose(s) was not possible because of too many nondiagnostic samples.
Test article–related changes should be recorded and graded in the individual animal data. As recommended in previous publications (Elmore 2006; Haley et al. 2005), whenever possible, treatment-related changes should be recorded in the individual animal data in descriptive or semiquantitative terms (e.g., decreased granulocytic cellularity, decreased hematopoietic cellularity, increased CD20 immunoreactivity) instead of clinical, interpretive, or diagnostic terms (e.g., “erythroid hyperplasia,” “granulocytic hyperplasia,” “aplasia”). Because multiple hematopoietic cell lines coexist in the bone marrow, it may be difficult to differentiate a true decrease in the absolute numbers of a particular lineage from a change in the proportion relative to the overall cellularity. An example of such a situation is when there is an increase in the proportion of granulocytic cells and the pathologist must try to determine whether it results from an increase in the numbers of granulocytic cells, a decrease in the numbers of erythroid cells, or both. A descriptive approach to recording the data would be encouraged in this situation. Interpretive terms such as atrophy, hyperplasia, hypoplasia, and so on may be reserved for the pathologist’s interpretation of the bone marrow findings within the context of the study in the narrative section of the report. The pathology narrative should summarize, describe, and interpret treatment-related changes and should be in context of other observations, including hematological data, in-life findings, and, if available, cytological or flow cytometric bone marrow assessment, in concurrent controls and treated animals.
Recommendations on How to Collect Samples and Perform Cytological Evaluation of Bone Marrow
Bone Marrow Harvesting
Bone marrow is typically collected at necropsy, following anesthesia and euthanasia. Bone marrow cells should ideally be the first sample collected at necropsy to optimize preservation of cytological detail. Because the marrow will quickly begin to clot and bone marrow cells will start to undergo autolysis, cytomorphology will be optimal if bone marrow specimens are collected within five minutes after euthanasia (Smith et al. 1994; Valli et al. 2002). However, a laboratory may want to determine the time after death, which may vary by species and which will provide adequate quality of bone marrow smears to perform cytological evaluation. The femur and sternum are acceptable sites for bone marrow collection from most small animal (rodent) species. The bone should be removed and, if femur is used, it should be opened longitudinally to expose the marrow cavity. It is important to visualize the marrow to ensure collection of active marrow, which is red, rather than yellow, less-active (fatty) sites. In older animals, especially nonrodent species, active marrow may not be present in the mid-shaft or distal femur. In larger animals (dog, cat, primate, rabbit, swine), the rib or sternum is a preferred site for bone marrow cytology because of the more uniform activity of the marrow at these sites and the ease of collection. The rib or sternum should be removed from the carcass and opened along the long axis to expose the marrow cavity. Once open, the bone is then squeezed with forceps, pliers, or a similar instrument to extrude some marrow from the exposed surface. Slides should be made immediately. Other collection sites may be successfully used depending on the experience and skill of the necropsy team. Although proximal femur may be used in large animals, one needs to be aware of the potential artifacts created by the harvesting technique, such as heat from a bone saw, and the longer time required to access this site to obtain active marrow.
Because of the rapid loss of cytological integrity after death, collection of bone marrow for cytology from animals that have died on study (found dead) is without value. Bone marrow cytology smears obtained from moribund animals should be examined judiciously since the cytology may reflect moribund-related changes, which could be mistakenly concluded to be test article related rather than a consequence of physiological failure.
Bone marrow collection from live animals is rarely required and is a consideration only for large animals, such as dogs. A number of references may be consulted for information regarding collection techniques (Freeman 2000; Harvey 2001; Smith et al. 1994).
Slide Preparation Techniques and Staining
There are various techniques that, with practice, result in acceptable and representative bone marrow smears. The choice of technique may depend on the experience of the individuals who are collecting the samples. With practice, any of these techniques will produce good-quality slides. With any of the following techniques, excess diluent should be avoided, since doing so will cause background staining and will prolong the drying process. Prolonged drying from excess diluent or unduly thick samples will result in cell condensation, which compromises subsequent cell identification. Excessive cell lysis may be caused by too little diluent or when samples have started to clot. Acceptable performance of the collection method in the species of interest should be demonstrated by the individuals who will collect the samples prior to use in a study.
Paint Brush Technique
Clean, natural-bristle (sable) brushes are used to obtain the bone marrow sample and apply it to the slide. The brush is dipped in 5% bovine serum albumin (BSA), allogenic serum, or fetal calf serum mixed 2:1 with 7.5% EDTA to slightly wet the bristles and serve as a diluent. Small (e.g., 1 mL) aliquots of a diluent can be frozen in individual vials and thawed as needed for studies. The brush bristles are blotted to remove extra diluent and create a narrow tip. The tip then is used to obtain some marrow from the exposed cavity, which is then applied to a clean glass microscope slide in 2–4 wavy lines (Valli et al. 2002; Figure 1 ).
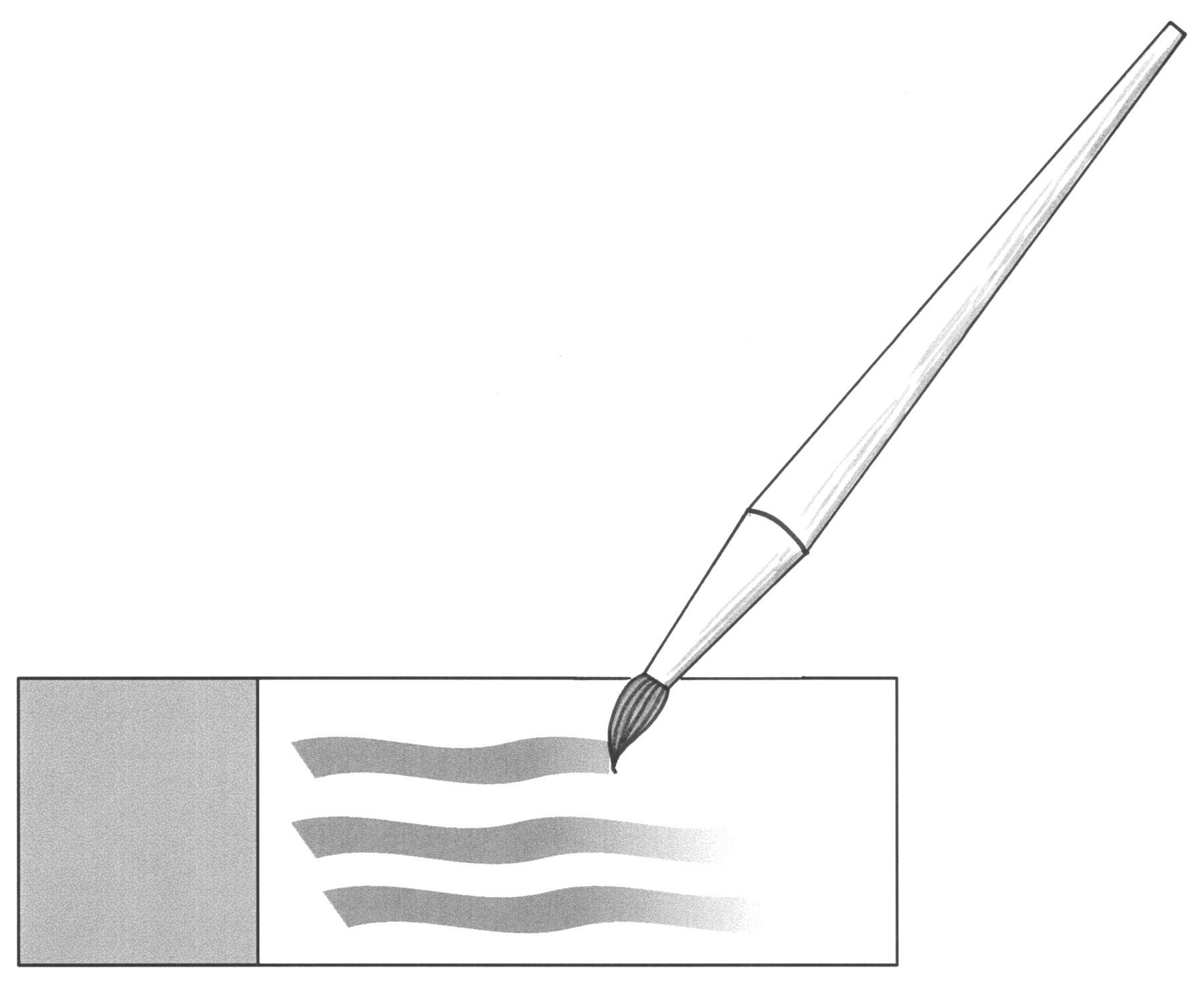
Bone marrow smear preparation: paint brush technique.
To a limited degree, the size of the animal should dictate the size of the brush needed. There is no uniform size numbering system for brushes among vendors, so the size will need to be confirmed prior to conducting a study. It is necessary to use a very small brush to access the marrow cavity of a mouse, which limits the amount of bone marrow applied to a slide. A larger brush should be used for other species. To prevent cross-contamination between animals, separate brushes should be used for each animal, or brushes must be rinsed thoroughly with distilled water to lyse residual cells, and then meticulously dried prior to use on another animal during necropsy.
Push Slide Technique
This technique is similar to that used to make cytology or hematology slides. First, one drop of diluent (as described above) is placed at one extremity of a glass slide. Marrow is collected as described above or with a small forceps and gently mixed with the diluent on the slide. The suspension is then spread in an even film using a second glass slide (held at an angle) as a spreader. The spreader slide is slid forward the length of the slide to produce a smear (Provencher Bolliger 2004; Valli et al. 2002; Figures 2 and 3 ).
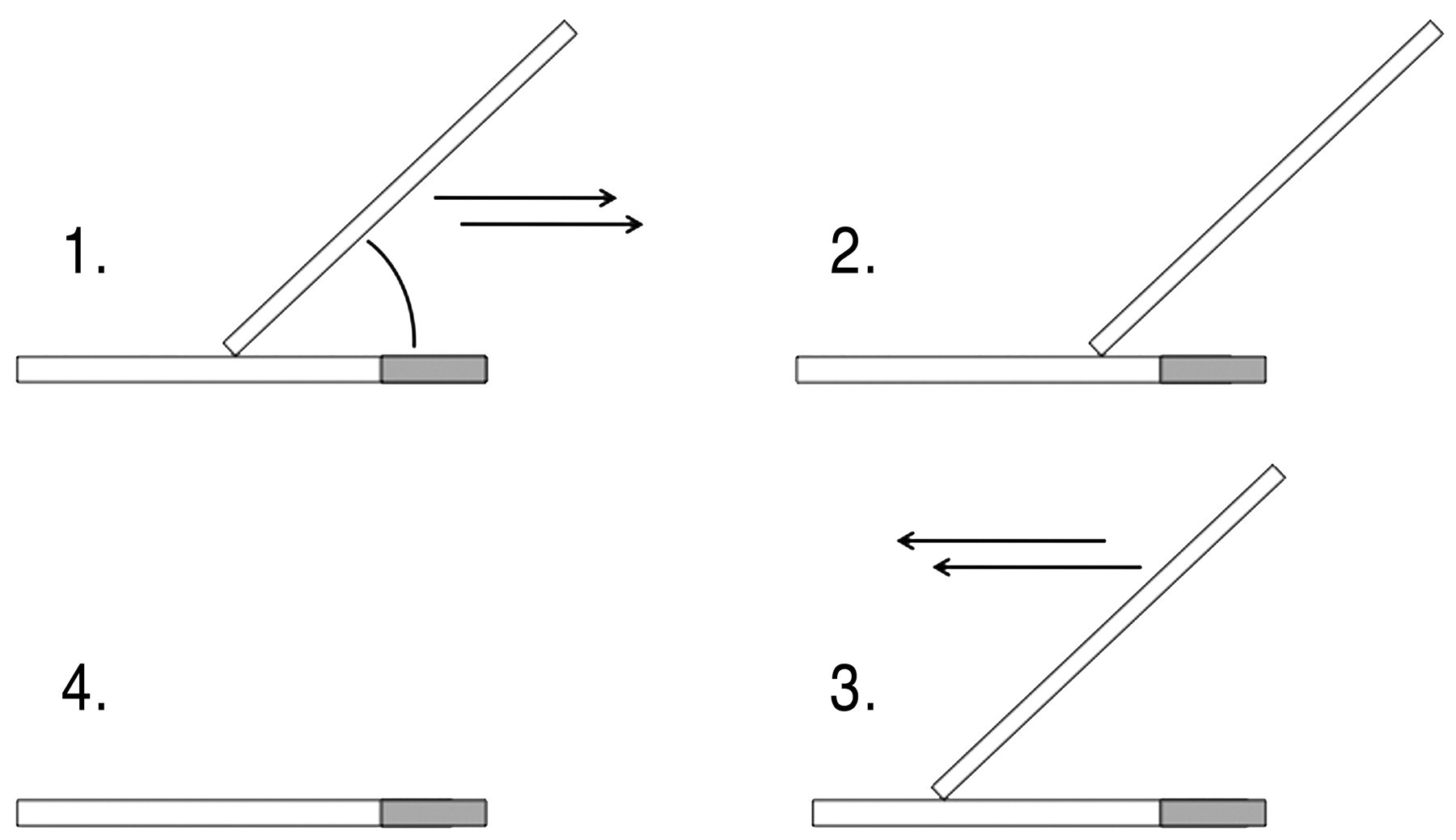
Bone marrow smear preparation: push slide technique.
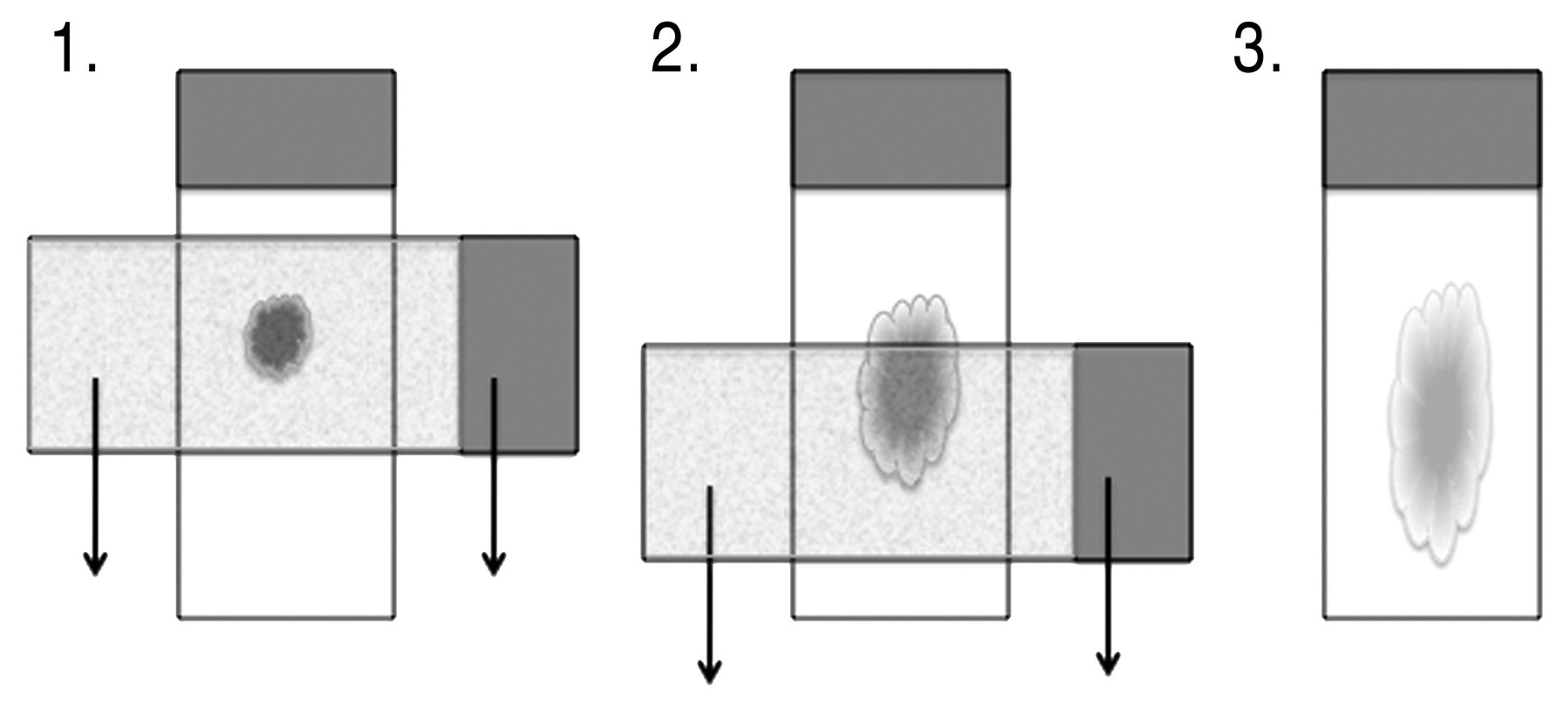
Bone marrow smear preparation: squash or pull technique.
Squash or Pull Prep Technique
Bone marrow is collected as described for the push technique and a drop is placed toward the end of the slide. The suspension is spread by placing a second microscope slide over the sample perpendicular to the slide with the sample and pulling the two slides apart. The weight of the top slide should be the only pressure exerted on the sample.
Cytocentrifuge Technique
In mice and young rats, marrow samples may be collected by flushing the marrow from the femur or (less commonly) the humerus. The femur is removed from the carcass, and the ends are removed by transecting between the lesser trochanter and the neck of the femoral head (proximal end) and just proximal to the intercondyloid fossa of the distal epiphysis (distal end). A needle attached to a syringe filled with appropriate buffer is used to flush the contents of the marrow cavity into a collection tube. After resuspension, the sample is cytocentrifuged for cytological examination, and the hematopoietic cells can be quantified on a hematology analyzer or flow cytometer (Provencher Bolliger 2004; Valli et al. 2002).
Ideally, at least two slides should be made from each animal with any of the techniques described above. To avoid formalin exposure, which will adversely affect staining, the slides should be removed from the necropsy or histology area as soon as possible after preparation and allowed to air-dry for several hours (or overnight) in an area not exposed to formalin fumes or dust prior to staining to avoid artifacts caused by moisture retention.
Staining
It is not necessary to fix the slides in methanol prior to routine staining. If there will be a long delay between collection and staining (weeks to months), slides may be fixed in absolute methanol for 1 to 2 minutes and then air-dried, although timely staining of the slides, generally within a few days, will allow for the best preservation of cytomorphology. Various Romanowsky stains can be used for routine evaluation of bone marrow cytology. Typical stains include modified Wright’s-Giemsa (such as that used in an automated hematology stainer), Giemsa, and May-Grunwald Giemsa. A good-quality brand of stain is recommended for better consistency between lot numbers. The staining time usually needs to be increased from that used for peripheral blood and depends on the thickness and cellularity of the preparation (Freeman 2000). Acceptable performance of the staining method should be demonstrated with specimens from the species of interest prior to use in a study.
Cytological Evaluation
It is critical that the person cytologically evaluating the bone marrow smears is an experienced evaluator. Bone marrow smears should not be examined in a blinded fashion, as it is necessary to consider the variability and establish a baseline for control animals in a specific study before it is possible to critically evaluate smears from treated animals. Samples are usually collected on all animals in the study. All groups may be evaluated, or one may choose to evaluate animals in the control and high-dose groups and then proceed down to mid- and low-dose groups until a NOEL is determined. Bone marrow cytology slides must be shown to be of adequate quality (i.e., adequate area(s) with uniform staining of cells arranged in a monolayer) in order to confidently interpret that the specimens reflect the physiological state of the animal and not extensive artifactual alterations that might have been induced by sample collection or slide preparation techniques. Slides that are of inadequate quality should not be evaluated.
The bone marrow cytological preparation should be scanned at low magnification (40×–100×) to determine areas of appropriate cell density and adequate staining to perform a cytological evaluation. Detailed cytological evaluation of hematopoietic cells should be done under oil immersion magnification (500×–1,000×) to assess the cytomorphology of the hematopoietic cells present and to assess adequacy of differentiation in each lineage (Ryan 2001). All the major lineages should be evaluated. Megakaryocytes are not enumerated, but their morphology can be assessed. The cytological bone marrow assessment may be done by either a qualitative assessment, similar to the histological evaluation, or by a quantitative method. The decision of whether qualitative or quantitative assessment must be performed for a particular study should be made by the study clinical pathologist in consultation with the study director and other scientists who are knowledgeable of the activity, toxicologic effects, and target of the test article. These methods are outlined below. All of these methods can be used successfully to further characterize the changes noted histologically, but they give different levels of detail of this assessment.
A qualitative assessment of cell maturation and morphology is key and is the foundation of the quantitative assessment. For some studies, the qualitative assessment may be used as the only method of evaluation, but it should also accompany each type of quantitative evaluation. Qualitative assessment alone in lieu of a quantitative assessment can be useful to further characterize the histological change. In addition to abnormalities in morphology, the relative proportions and maturation of the different cell lineages can be assessed. Qualitative assessment can also be used to obtain valuable information when there is an adverse effect on overall cellularity, or when one or several cell lines is present in very low numbers, or when cytological changes preclude definitive cell classification.
For all quantitative approaches, it is recommended that 300 to 500 nucleated cells be evaluated (Ryan 2001). Enumeration of 500 cells per slide may be preferable when there are few animals in each treatment group. In rodent studies in which group sizes are typically larger (ten/sex/group), enumeration of 300 to < 500 cells may be sufficient. Bare nuclei or lysed cells should not be included in the numerical evaluation.
If the definitive quantitative approach is chosen, the nucleated cells should be categorized into granulocytic, erythroid, and lymphoid lineages. Identification of all maturation stages for neutrophilic and erythrocytic precursors is suggested to provide a basis for determining whether there is a shift to less-mature (left) or more-mature (right) stages. A myeloid (granulocytic)-to-erythroid (M:E) ratio can be calculated from the quantitative differential count. It is not usually necessary to identify developmental stages for eosinophilic, basophilic, lymphoid, or other cell types. A modified quantitative approach is to do a partial differential, which categorizes the cells into the major categories (lymphoid, erythroid, and granulocytic) but does not enumerate each specific maturation stage for the erythroid or granulocytic cell type.
In addition, a further differentiation may be used to classify cells into proliferative and maturation (nonproliferative) phases of development. For granulocytic cells, myeloblasts, promyelocytes, and myelocytes are categorized into the proliferative population, whereas the metamyelocytes, band cells, and segmented cells are considered part of the maturation phase. Erythroid cells also may be categorized into proliferative phase (rubriblasts, prorubricytes, and basophilic rubricytes) versus maturation phase (polychromatophilic rubricytes and metarubricytes). Because partial differential evaluations group several developmental stages together, this approach is not as complete as a full differential but is often adequate to characterize the major cytological findings in the bone marrow. The proportions of proliferating versus nonproliferating cells can be used to calculate an erythroid maturation index, myeloid maturation index, and overall maturation index. Maturation indices are sometimes used for semiquantifying left or right shifts in hematopoietic cell maturation.
Reporting of Cytological Data
A report of bone marrow cytology should include a statement of specimen adequacy, quantitative and qualitative data that were generated during the evaluation, and an interpretation by a qualified and experienced clinical pathologist that correlates the bone marrow cytology findings with all of the study data. As with histology, samples of inadequate quality for evaluation should be recorded as such in the individual animal data together with a brief indication as to why the evaluation was nondiagnostic (for example, excessive “smudge” cells, not collected). The clinical pathology narrative should indicate whether evaluation of the bone marrow at any given dose(s) was not possible because of too many nondiagnostic samples. Use of diagnostic interpretive terminology (hypocellularity, hyperplasia, etc.) should be avoided or used very judiciously. Instead, morphological descriptions of changes, such as increases or decreases relative to control, should be favored over the use of diagnostic interpretive terminology. Often the spectrum of changes in nonclinical toxicity studies may not fall into a well-described or established clinical category and the use of clinical interpretive terminology could be misleading. Statistics sometimes may be used to assist in identifying effects. The clinical pathology narrative should summarize, describe and interpret treatment-related changes in the context of other findings including hematology data, histopathology changes, and in-life findings.
Recommendations of Flow Cytometric Techniques to Use for Bone Marrow Examination
There are several published techniques for flow cytometry examination of bone marrow (Criswell, Bleavins, Zielinski, and Zandee 1998; Criswell, Bleavins, Zielinski, Zandee, and Walsh 1998; Criswell et al. 2000; Kakiuchi et al. 2004; Kurata et al. 2007; Saad et al. 2000; Schomaker et al. 2002; Shah et al. 1988; Weiss et al. 2000; Zhao et al. 2005). These techniques, as well as advantages and disadvantages of the different methods, will be briefly described below. The flow cytometric technique selected will depend on the technical expertise, equipment, and experience available at a given facility. No matter which flow cytometric technique is used, bone marrow cytological preparations should be made (either bone marrow smears or cytocentrifuge preparations) to potentially evaluate morphological changes, since flow cytometry does not allow for detailed morphological assessment.
Size (forward-angle light scatter) versus complexity (side-angle light scatter) has been used to type dog bone marrow hematopoietic cells, which resulted in the following subcategories: immature and mature erythroid cells, immature granulocytic cells, metamyelocytes, band/segmented neutrophils, and megakaryocytes (Weiss et al. 2000). The advantage of this method is that the cells do not go through an antibody-labeling process, making the procedure simpler. The disadvantage of this technique is the potential change in the location of the cells in the scatter plot resulting from the disease/toxicological state, which may make accurate gating difficult. In addition, this method cannot easily distinguish between erythroid and lymphoid cells, a critical determination when evaluating rodent bone marrow.
Another method combines the use of cell size and complexity with the use of fluorochromes and allows for accurate classification of bone marrow cell populations (Criswell, Bleavins, Zielinski, and Zandee 1998). The following subpopulations can be delineated: non-nucleated red blood cells, maturing erythroid/lymphoid, proliferating erythroid, maturing granulocytic, proliferating granulocytic, and megakaryocytes. The lymphocytes can then be further distinguished from the erythroid population using a combination of T cell- and B cell–specific antibodies. This method is accurate and reproducible in normal rats, as well as in rats undergoing different treatments with hematological modifiers including phenylhydrazine, phlebotomy, cyclophosphamide, and hematopoietic growth factors GM-CSF, EPO, and SCF (Criswell, Bleavins, Zielinski, Zandee, and Walsh 1998; Criswell et al. 2000) and provides comparable results to those obtained with the manual method. The advantage of this method is that it can also be used in dogs and nonhuman primates, although the reproducible separation of the different subpopulations can sometimes be challenging in the latter species. The disadvantage of this technique is that it is somewhat dependent on the flow cytometer platform used.
Cell-specific surface markers for the different lineages have also been used to enumerate the different subpopulations in the bone marrow of rats (Kakiuchi et al. 2004; Kurata et al. 2007; Schomaker et al. 2002; Zhao et al. 2003) and humans (Shah et al. 1988). Most of these methods have employed monoclonal antibodies specific for different bone marrow populations, often CD71 or CD45 alone (Schomaker et al. 2002; Weiss 2004) or in combination (Kakiuchi et al. 2004; Saad et al. 2000) to differentiate the erythroid lineage versus the granulocytic/lymphoid lineage, respectively. To distinguish the granulocytic from the lymphoid subpopulations, antibodies to CD45 must be combined with antibodies to other cell markers or combined with the evaluation of side scatter (Saad et al. 2000). The advantage of this cell surface antigen detection approach is that it allows relatively easy separation of the different bone marrow populations. A disadvantage of this approach is that it needs to be validated for each species of interest and modulation of surface antigen expression may lead to misclassification of cells.
Recommendations of Techniques Used for Clonogenic Assays and Electron Microscopy of Bone Marrow
There are several bone marrow clonogenic techniques that have been successfully used. Which technique to use will depend on the technical expertise and experience available at a given facility. The most common historical approach was to quantify multilineage- or single lineage–committed hematopoietic progenitors (i.e., colony-forming units) using viscous or semisolid matrices and culture supplements that promote proliferation and differentiation of the hematopoietic cells (Erickson-Miller et al. 1997; McMullin et al. 1998). Other approaches have employed the intracellular ATP (iATP) concentration, which varies proportionately to the proliferation rate. This assay has been used for cell populations with varying stages of maturity, but measurement of iATP can be affected by circumstances other than cell proliferation, such as apoptosis, necrosis, cytotoxicity, contamination with fibroblasts (Fan and Wood 2007; Slater 2001). A similar approach uses purchased stem cells, usually of human, baboon, canine, or mouse origin, with the addition of cytokines and growth factors to support cell development. Rather than measurement of iATP, it employs a single incubation with a lanthanide-conjugated primary antibody and subsequent time-resolved fluorescence spectroscopy so that the myeloid, erythroid, and megakaryocytic lineages can be objectively quantified (Greenwalt et al. 2001).
In vivo bone marrow cytological and histological specimens or in vitro bone marrow cell culture samples can be evaluated by electron microscopy (Alvi et al. 2001; Burns and Yook 1988). Successful electron microscopic evaluation of bone marrow requires timely preplanning to allow proactive rapid sample collection and fixation to achieve optimal tissue preservation. Transmission electron microscopy (TEM) is employed more frequently than scanning electron microscopy (Chamberlain et al. 1975; Deldar et al. 1988; Kitajima et al. 2007; Okamatsu et al. 2007; Rouselle and Wicks 2008; Samsom et al. 1972). For routine TEM, tissues are typically fixed with an aldehyde fixative, such as glutaraldehyde or Trump’s fixative, postfixed in osmium tetroxide, and embedded in hard plastic media such as epoxy resins (Burns and Yook 1988). Methacrylates, such as glycol methacrylate, are not recommended for routine TEM because of a variety of limitations (Dykstra and Reuss 2003; Hayat 2000). Thick sections, also called semithin sections, are usually stained with toluidine blue and are used to evaluate the suitability of the sample and select an area for TEM evaluation (Hayat 2000). Thin sections, also called ultrathin sections, are the sections used for TEM evaluation and can be stained with a variety of techniques (Dykstra and Reuss 2003; Hayat 2000). Uranyl acetate and lead citrate are used routinely (Deldar et al. 1988).
Assessment of the hematopoietic system is an important component of nonclinical toxicity testing. The complete blood count data and histologic evaluation of bone marrow are the foundation of this assessment. If there is insufficient characterization of the effect of the test article on hematopoiesis using these tools, a more detailed assessment can be done by cytologic and flow cytometric evaluation. Specialized techniques such as clonigenic assays and electron microscopy may also be helpful in certain situations. Following the guidelines outlined in this manuscript on how to use these different tools effectively will ensure accurate assessment of the hematopoietic system.
Footnotes
The views expressed in this article are those of the authors and do not necessarily represent the policies, positions, or opinions of their respective organizations. The recommendations in this article are endorsed by the Society of Toxicologic Pathology, American Society for Veterinary Clinical Pathology, The American College of Veterinary Pathologists, British Society of Toxicological Pathologists, and the European Society of Toxicologic Pathology.
This is an opinion article submitted to the Regulatory Forum and does not constitute an official position of the Society of Toxicologic Pathology or the journal Toxicologic Pathology. The Regulatory Forum is designed to stimulate broad discussion of topics relevant to regulatory issues in toxicologic pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to
