Abstract
Bordering the ventricular cerebrospinal fluid (CSF) are epithelial cells of choroid plexus (CP), ependyma and circumventricular organs (CVOs) that contain homeostatic transporters for mediating secretion/reabsorption. The distributional pathway (“nexus”) of CP-CSF-ependyma-brain furnishes peptides, hormones, and micronutrients to periventricular regions. In disease/toxicity, this nexus becomes a conduit for infectious and xenobiotic agents. The sleeping sickness trypanosome (a protozoan) disrupts CP and downstream CSF-brain. Piperamide is anti-trypanosomic but distorts CP epithelial ultrastructure by engendering hydropic vacuoles; this reflects phospholipidosis and altered lysosomal metabolism. CP swelling by vacuolation may occlude CSF flow. Toxic drug tools delineate injuries to choroidal compartments: cyclophosphamide (vasculature), methylcellulose (interstitium), and piperazine (epithelium). Structurally perturbed CP allows solutes to penetrate the ventricles. There, CSF-borne pathogens and xenobiotics may permeate the ependyma to harm neurogenic stem cell niches. Amoscanate, an anti-helmintic, potently injures rodent ependyma. Ependymal/brain regions near CP are vulnerable to CSF-borne toxicants; this proximity factor links regional barrier breakdown to nearby periventricular pathology. Diverse diseases (e.g., African sleeping sickness, multiple sclerosis) take early root in choroidal, circumventricular, or perivascular loci. Toxicokinetics informs on pathogen, anti-parasitic agent, and auto-antibody distribution along the CSF nexus. CVOs are susceptible to plasma-borne toxicants/pathogens. Countering the physico-chemical and pathogenic insults to the homeostasis-mediating ventricle-bordering cells sustains brain health and fluid balance.
Keywords
Introduction
Integrated Topics and Objectives
Neurons are exquisitely and adversely sensitive to toxicants and pathogens. Normally the impermeable blood-brain barrier (BBB) and blood-cerebrospinal fluid (CSF) barrier (BCSFB) act together to protect the neuronal networks from potentially injurious agents in blood. Historically, the BBB has received more pharmacologic, toxicologic, and pathologic attention than the BCSFB, or choroid plexus (CP). In a disease or toxicity context, this is ironic because BCSFB is more vulnerable (Levine 1987) than cerebral capillaries to many foreign invaders. Often the CP or an ependymal circumventricular organ (CVO) is the first site of central penetration by a deleterious substance or pathogen (Siso, Jeffrey, and Gonzales 2010). CP is also of unique interest to toxicologists and pathologists because its main secretion, the cerebrospinal fluid, rapidly and widely disseminates throughout the central nervous system (CNS) the various substances that have penetrated a breached BCSFB. Thereafter, the toxicants, pathogens, and cells that permeate the CSF are readily accessible via bulk flow circulation to neurons and stem cells over wide expanses of brain. Accordingly, the two aims of this review are to: (a) characterize the intricate physiology of the CP, CSF, and ependyma and (b) assess how harmful xenobiotics and microbes (e.g., viruses, protozoans) damage the CP and ependyma, thereby destabilizing the brain interior (Krinke et al. 1983; Levine, Sowinski, and Nochlin 1982; Roth and Krinke 1994). Regions injured include the delicate neurogenic niches and body-regulatory centers near ventricular CSF.
Central Nervous System Toxicology/Pathology: Differential Effects of Barrier Phenomena
Toxicants and pathogens vary greatly in ability to penetrate particular regions of the central nervous system. Consequently, knowledge of permeation patterns across diverse transport interfaces is essential to understand specific disruptions of brain and CSF. BBB refers to the widespread impermeable capillaries in the CNS. However, BBB is sometimes used inappropriately to include the BCSFB and other central sites of impermeability. Therefore, more precise classification of CNS transport interfaces is needed. Care should be taken to delineate properties/reactions of the membrane boundaries that separate blood, CSF, and brain. Bidirectional secretory and reabsorptive phenomena should also be considered. This is true in a physiologic-pharmacologic sense and in relation to toxicologic-pathologic effects on barrier disruption.
Transport specialists concur that the BBB resides primarily in the tight junctions of brain microvessels or capillaries. BCSFB most commonly (and in this review) is used to refer to tight junctions in CP epithelium (but should be more inclusively defined if meant also to include CNS-inward flux across arachnoid membrane into subarachnoid space). The lateral ventricular CSF-brain interface, or ependyma, is permeable to macromolecules, and therefore does not usually act as a barrier. Certain regions of the ependymal wall contain specialized CVOs adjacent to CSF. The CVOs, like CP, have permeable capillaries and are thus open windows to the systemic circulation. Thus, each CVO does not have a BBB.
Because BBB is endothelial and BCSFB epithelial, there are consequently barrier differences in tight junction (impermeability) properties and in types of transporters expressed at the respective barrier cell membranes. These are important distinctions in view of how toxicants and pathogens differentially interact with BBB and BCSFB. Cerebral capillaries and choroid plexuses extensively mediate homeostatic mechanisms to protect the neuronal microenvironment. When xenobiotic agents and viruses disrupt BBB and BCSFB, they not only breach barriers but also may disrupt homeostatic transporter actions that benefit neurons.
Mechanisms for Keeping a Pristine Environment for Neurons
To function optimally, the brain requires a clean environment for the neural parenchyma. Neurons need a stable extracellular or interstitial fluid (ISF) of specialized composition to maintain an extracellular environment that promotes efficient transmission of impulses along axons and through synapses. ISF that typically bathes neurons is relatively low in concentrations of plasma proteins, cytokines, catabolite waste products, erythrocytes, leukocytes, immune cells, pathogens, drug metabolites, and toxicants. Three mechanisms mainly accomplish CNS microenvironmental cleansing: (1) an impermeable BBB and BCSFB that restrict diffusion of plasma water-soluble molecules into CNS; (2) reabsorptive organic solute transporters at the CNS-facing side of barrier cells for actively extruding into blood certain organic acids (anions) and bases (cations) in ISF or CSF; and (3) the sink action of CSF to remove from the brain various metabolites and impurities for excretion, by bulk flow, from the ventricles to downstream venous/lymphatic exit sites.
Fundamental differences exist in the workings of BBB (cerebral capillaries) versus BCSFB (choroid plexus). The latter purifies the CSF interior and periventricular regions with homeostatic systems to keep brain and body fluids toxicant free and in physiologic balance. Attenuation of CP transport capacity can profoundly impair cerebral metabolism and the fluid environment of neurons (Spector and Johanson 2010a). Accordingly, we analyze BCSFB transporters, selective CP permeability, the CSF and peptide secretions that choroid epithelial cells generate, as well as fluid flow through the ventricular system. CSF streaming down the neuraxis from lateral to fourth ventricles reaches many regions. The largest portion of ventricular CSF is eventually convected to the subarachnoid space for bulk fluid clearance across arachnoid villi and lymphatic drainage channels that follow the olfactory nerve and discharge their contents into the nose. However, a small but significant portion of CSF-borne ions and organic solutes diffuses across or is taken up by border cells at the ventricular margins.
Compartmental Aspects of the Physiologic Nexus
Modeling features of the specific distributional pathway, or “nexus,” connecting the CP, CSF, ependyma, and brain are shown in Figure 1 . In a transport physiology sense, a nexus can be viewed as a series of connected compartments within an organ, through which substances passively move or are actively transferred, to effect an action or function at a downstream target. A nexus example is the facilitated diffusion of plasma glucose via carrier proteins across brain endothelium into the interstitial fluid, and then diffusion to nearby neurons for carrier-mediated uptake of the sugar into axoplasm. Another nexus pathway is the diffusion of plasma-borne angiotensin across permeable capillaries of the subfornical organ (a CVO) into the interstitial space, through which this peptide diffuses to neurons. Yet another nexus, and the main theme of this review, is the movement of a micronutrient (or hormone or xenobiotic) across CP into ventricular CSF for bulk flow to target cells in the ependyma and underlying periventricular brain (Figure 1). Nexuses are convenient transport models to delineate precise point(s) along distributional pathways interrupted by pathogens or toxicants.
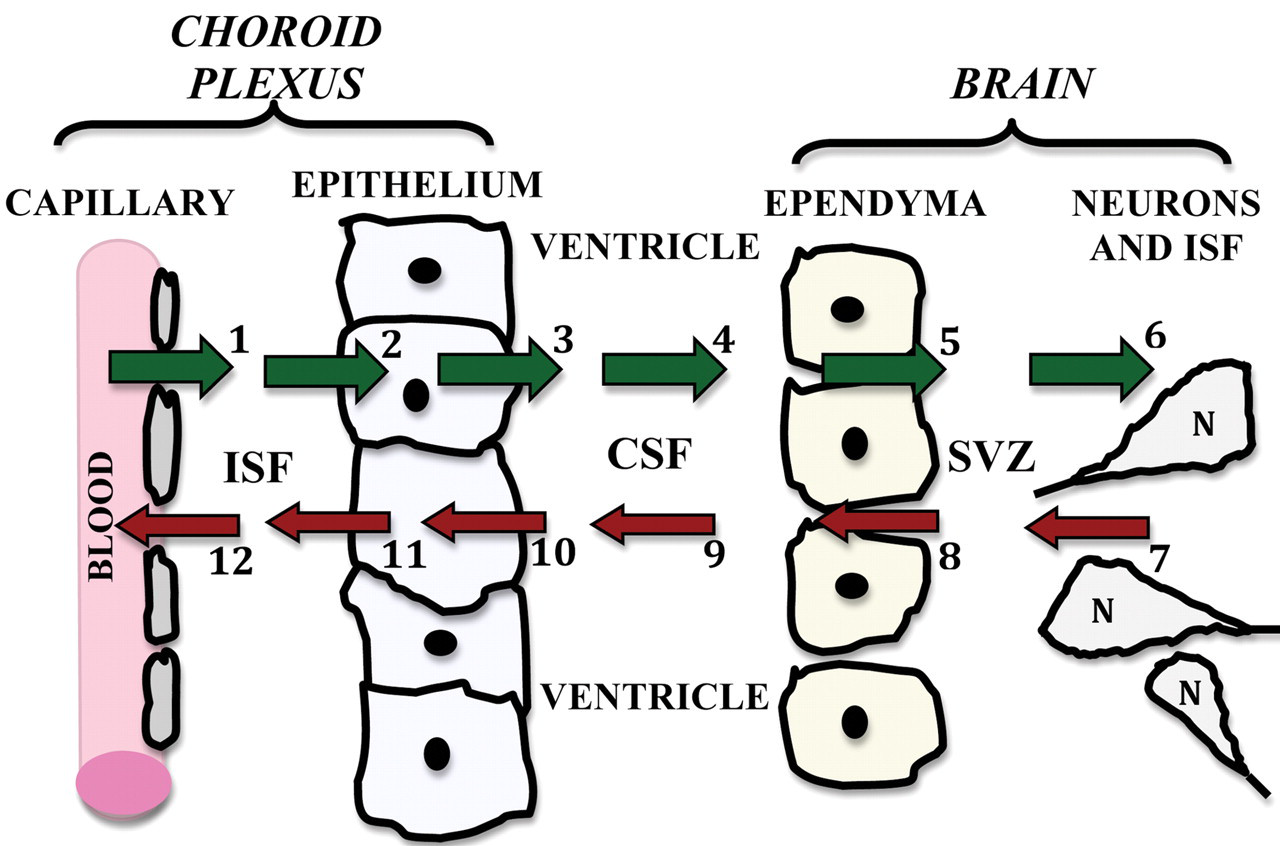
Central role of the choroid plexus (CP)-cerebrospinal fluid (CSF) nexus in exchanging materials with brain. Ions, water, and organic molecules passively filter out of CP capillaries (arrow 1) into interstitial fluid (ISF). This is the first step in the distributional nexus to the brain (green arrows). Solutes diffuse through ISF up to the basolateral membrane of the CP epithelium. Active mechanisms in membranes transfer solutes sequentially across the basolateral (arrow 2) and apical membranes (arrow 3) into ventricular CSF. As CSF flows (arrow 4) from ventricles into the cisterna magna, a small fraction of the CSF-borne substances diffuse across ependyma (arrow 5) into periventricular brain or are taken up by specialized ependymal cells of circumventricular organs (CVOs) in the ventricular walls. Ependyma-penetrating substances diffuse through brain ISF (arrow 6) for transport into neurons (N); blood-brain barrier (BBB)/capillaries, not depicted, are interspersed among neurons. Material inflow to CNS interior thus sequentially involves CP, CSF, ependyma/CVOs, and brain. Viewed as a distributional nexus, this CNS-inward pathway conveys solutes across the blood-CSF barrier (BCSFB) eventually to targets in the caudate nucleus, hippocampus, dentate gyrus, subventricular zone (SVZ), and hypothalamus. In the opposite direction, there is a reverse nexus (red arrows) for catabolites released by neurons/glia into the brain ISF. Accordingly, cerebral catabolites such as homovanillic acid diffuse through ISF (arrow 7), and down a transependymal concentration gradient into the CSF (arrow 8). By bulk flow of CSF (arrow 9), catabolites are convected to the subarachnoid space (not depicted) or to the CP for active removal from the ventricles across the apical surface (arrow 10) followed by extrusion across the basolateral membrane (arrow 11). Therefore, some endogenous or drug metabolites end up being cleared passively into the blood via microvessels (arrow 12) and venules draining the CP (Johanson et al. 2000). Overall CSF is simultaneously a source (arrows 1 to 6) and a sink (arrows 7 to 12) for distributing molecules, depending on the prevailing concentration gradients between ventricular CSF and brain ISF. As such, the CSF and bordering cells constitute a nexus for mediating trophic (CSF to brain) and excretory (brain to CSF) fluxes.
As the medium for distributing materials within ventricles and to ependyma and subjacent brain, the CSF plays a primary role in modulating subventricular and hypothalamic regions. Nexus is a concept applicable to CSF distributional phenomena from uterine life to senescence. It operates in several states: Ontogenetic: For fetal brain development, the neurogenic niches in the subventricular zone (SVZ) depend on growth factors and neurotrophins secreted and transported by CP-CSF for promoting stem cell conversion to neurons (Owen-Lynch et al. 2003). A steady supply of choroidally derived peptides (Chodobski and Szmydynger-Chodobska 2001), nucleosides (Redzic et al. 2005), and vitamins (Spector and Johanson 2006, 2007b) furnished to CSF is critical for feeding the periventricular regions (the domain harboring neurogenic stem cells) that drive normal brain development. Containing sparse capillary networks, the early fetal cortex evidently needs nutritional supplies delivered via CP-CSF as well as plasma. Substantial glycogen deposition in prenatal CP epithelium points to a BCSFB source of substrate for energy needs by developing brain (Kappers 1958). Endocrine: As a hormonal signal relay station, the lateral ventricle CP epithelium takes up blood-borne hormones/peptides (Dietrich et al. 2007) by basolateral endocytosis or carriers (arrow 2, Figure 1) and releases them apically into CSF (arrow 3, Figure 1). By CSF bulk flow, these plasma-derived hormones are carried to the third ventricle region where they readily permeate the arcuate nucleus (Rodriguez, Blazquez, and Guerra 2010) and bind receptors in specific hypothalamic nuclei. In this manner, endocrine feedback loops modulate the hypothalamic-pituitary axis for regulated secretion of hormones into blood. Moreover, the co-localized fluid-balancing peptides arginine vasopressin and basic fibroblast growth factor, regulated in CP epithelium and hypothalamic paraventricular nucleus, mediate osmolality homeostasis in CSF and plasma (Gonzalez et al. 2010). Pharmacologic: Therapeutic agents destined for CNS targets must circumvent the neurovascular and epithelial barrier systems that thwart agent penetration into brain. BBB has been widely manipulated to enhance drug access to neuronal targets, albeit with limited success. An alternative strategy is to pharmacologically exploit the BCSFB by altering CP permeability and transporter capability. Modification of BCSFB can alter CP function per se or expedite drug delivery to brain regions close to CSF (Johanson, Duncan, et al. 2005). Immunologic: Normally there is highly regulated traffic of immune cells and molecules across BCSFB to monitor and adjust CSF immune status. In the relapse state of certain autoimmune diseases, leukocyte movement across CP into the ventricles and adjacent periventricular regions is augmented to the brain’s detriment (Prendergast and Anderton 2009). Finer control of immunoglobulin and immune cell access to CSF may prevent exacerbation of CNS immunopathology. Geriatric/therapeutic: With advancing age, there is progressive deterioration of CP secretory ability and CSF turnover rate (Silverberg et al. 2001). This reduces the excretory, lymph-like functions of CSF and impairs brain metabolism. As a result, neurogenic niches in the SVZ and dentate gyrus are disrupted. Consequently, the altered stem cells and hippocampal neurons near CSF can hasten the decline of cognition. Therapeutic strategies to reduce aging damage to CP and ependyma would likely preclude disruption of subventricular neurogenic and CSF homeostaticphenomena in aging (Johanson et al. 2004). Toxicologic/pathologic: CP, ependyma, and CVOs are vulnerable, to a variable degree, to attacks by pathogens and toxicants. Some blood-borne viruses and parasitic agents have a predilection for binding, damaging, and penetrating the BCSFB. Differential abilities by xenobiotics (and pathogens) to disturb BBB endothelial cells versus CP epithelial cells are manifest by the specific neuropathology inflicted downstream of the initial insult to the barrier locus.
Toxicology and Pathology Models of Damage to CSF-Bordering Cells
The CSF fills four ventricles and the mesencephalic aqueduct of Sylvius in the brain interior. Far from being static sacs of fluid, the ventricular CSF exerts many dynamic actions, biophysical as well as biochemical, on the brain parenchyma. Basically two types of epithelial cells form the border that encompasses the CSF: choroidal and ependymal. Thus the internal (ventricular) surface of the brain is comprised mainly of a single layer of choroid plexus epithelium or ependyma (D. E. Smith, Johanson, and Keep 2004). CP epithelial cells do not generally contact the brain; rather, they extend into the ventricular CSF. On the other hand, the ependyma intimately and extensively adjoin the neural tissue. CP has its own interstitium, but the ependymal cells share an underlying interstitial space with cerebral parenchyma (Figures 1 and 7 ).
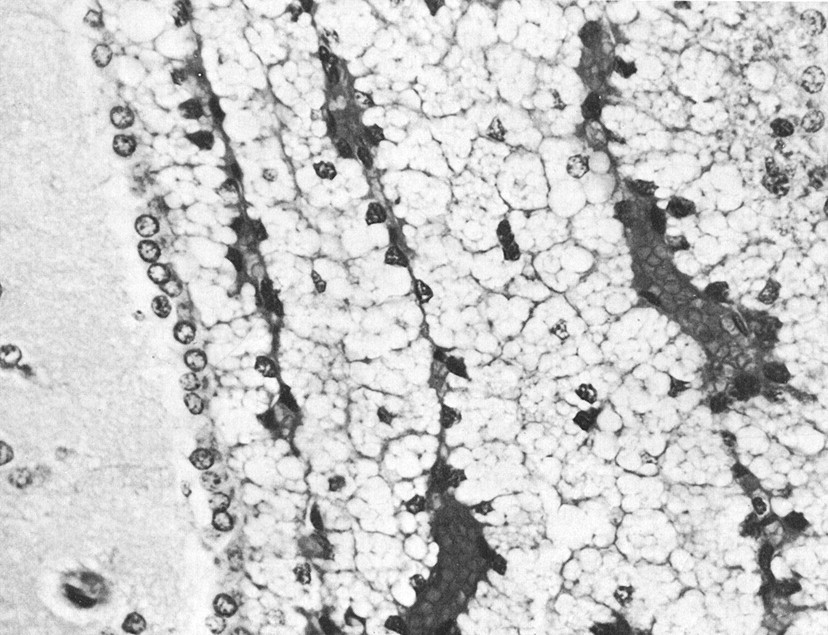
Vacuole formation in the choroid plexus (CP) epithelium of rats treated with a tertiary amine. The cytoplasm of most epithelial cells is filled with vacuoles that give the tissue the look of hydropic pallor. This intense vacuolar response was typical of rats injected with piperamide (500 or 750 mg/kg SC) and then necropsied one day later. Vacuoles displace the nuclei to a basal position. Ependyma and the adjacent periventricular brain appear normal (the slight separation between the ependyma and the brain tissue is an artifact). Congested CP fills the lateral ventricle. Experimental data are summarized in Table 4 and interpreted by Levine (1994). Hematoxylin and eosin, ×450. Due to unavoidable circumstances the above figures were not originals but were scanned from reprints.
CP is readily dissectable as a tissue tuft for investigations. Ependyma is not as easily separated intact as a monolayer from underlying brain. Such architectural or topographical differences in CP versus ependyma help to explain fundamental differences in experimental models evolving in the field. In vitro, CP is popular due to straightforward tissue excision from the ventricles (Crossgrove, Li, and Zheng 2005; Q. R. Smith and Johanson 1985; Sanderson, Khan, and Thomas 2007). Alternatively, the in vivo systemic modeling emphasized in this review uses microscopy and regional tissue staining to delineate how structures in the CP-CSF-ependyma-brain nexus (Figure 1) respond to systemically administered xenobiotics.
Information abounds on CP and ependyma pathology, especially for tumors (Krinke et al. 2000; Netsky and Shuangshoti 1975). Spontaneous rodent tumors in the brain are less differentiated than their human counterparts. Morphologic characteristics of CNS tumors in rats and mice, including CP papillomas and ependymomas, have been thoroughly treated (Krinke et al. 2000). Microbial and immune cell permeation of CP have also been widely documented (Engelhardt and Sorokin 2009; Petito 2004). Toxicants of CP fall into two major categories: inorganic agents and organic agents. For example, Zheng and colleagues extensively recapitulated the toxic effects of the elements Pb, Fe, Cd, Cu, Hg, and Mn on CP-CSF functions (Aschner, Vrana, and Zheng 1999; Zheng 2001).
Toxico-Pathologic Considerations for Choroid Plexus-Cerebrospinal Fluid
A sparsely studied distributional pathway is the transport route from CP to CSF and then to periventricular regions (Figure 1). This paucity of attention is surprising in view of the critical guardian and multiprovider roles of CP-CSF for interior brain: the hippocampus (Johanson et al. 2000), neurogenic niches (Miyan, Nabiyouni, and Zendah 2003), and hypothalamic nuclei (Rodriguez, Blasquez, and Guerra 2010). In addition to CSF-mediated delivery of micronutrients, growth factors, and neurotrophins to periventricular regions (Johanson et al. 2008), there are toxico-pathologic issues associated with conveyance of microbes (Levine 1987) and metals (Zheng 2001) along this nexus. Better control of the spread of pathogens, toxic drug metabolites, and cancer cells (Glantz and Johanson 2008) requires additional insight for therapeutically manipulating CSF distribution (Johanson, Duncan, et al. 2005) (Figure 1).
Due to the CP’s primary role in sustaining CSF dynamics, it is important to uncover mechanisms by which pathogens and toxicants alter BCSFB integrity. Upon breaching of the CP barrier (the first line of defense for the CNS interior) by CSF-permeating noxious agents, certain additional injuries are caused downstream in the nexus at the ependymal boundary (the second protective interface for deep brain regions abutting CSF-filled cavities). Simultaneous disruption of CP, ependyma, and CVOs renders the brain (and peripheral organs under control of circumventricular brain centers) vulnerable to invading agents. Multiple functions can be destabilized. Therefore, ependymal/subependymal lesions have wide-ranging effects. Ependymal wall lesions are diffuse or circumscribed. Phenomenologically, regionalized lesions in the CNS interior can be related to progressive damage along specific loci in the CP-CSF-ependyma-brain nexus (Krinke et al. 1983; Levine, Sowinski, and Nochlin 1982).
Pathogen-disrupted CP initiates the development of some brain diseases. Phenotypically, the glycoconjugate expression of the choroid epithelial and ventricular ependymal cells are such that particular viruses are bound and incorporated intracellularly (Ormerod and Raseroka 1988). Similarly, Trypanosoma brucei, the protozoan agent that causes African sleeping sickness, begins CNS offensive actions by penetrating and disorganizing CP (Philip et al. 1994). Trypanosome injection into rodents or goats induces neuropathology akin to the human African trypanosomiasis syndrome (Darsaud et al. 2003; Moulton 1986; Poltera et al. 1980). Thus, trypanosome administration to mammals provokes localized (choroidal) inflammatory reactions that chronically worsen and disrupt the BCSFB (Van Marck et al. 1981). Following infection of CP by Trypanosoma brucei, there ensue multiple sequelae that progressively injure the CSF-brain nexus and finally the BBB (Sanderson et al. 2008). If left pharmacologically unchecked, this intra-CNS pathology cascade that begins with CP invasion by trypanosomes can be fatal (Moulton 1986).
Another pathogen with a penchant for CP is human immunodeficiency virus (HIV, the cause of acquired immunodeficiency syndrome [AIDS]). Dendritic cells and monocytes in CP interstitium are infected with HIV in AIDS patients. The ratio of CP to brain infection with HIV is generally greater than twofold. CP infection precedes HIV-induced encephalitis. Even in asymptomatic AIDS, the CP involvement with infection precedes that of brain (Petito et al. 1999). Replication-competent HIV in CP is similar to, but not identical with, that in brain (Burkala et al. 2005). Petito and colleagues (1999) postulated that CP is the site for hematogenous spread and a reservoir for productive HIV infection during clinical latency. Collectively, these histopathologic observations on CP relative to brain (regional and temporal) implicate CP-CSF nexus participation in disseminating HIV into CNS. Moreover, molecular analyses of viral DNA in CP (Burkala et al. 2005) corroborate early participation of the BCSFB pathway for later distribution to brain (putatively by the CSF nexus). Unfortunately, CP may provide an environment promoting evolution of drug-resistant HIV strains with CNS tropism (Chen, Wood, and Petito 2000). Involvement of CP in initial stages of HIV infection suggest pharmacological targeting of virus trapped in CP interstitium (i.e., not yet having accessed CSF). Remedial agents in blood would then not have to pass through the restrictive BCSFB. Rather, the drugs would readily traverse leaky capillaries to reach the HIV-laden immune cells in the CP interstitial space.
Neuropathologic Principles for Analyzing Choroid Plexus and Ependyma
Pathologists regard CP in the domain of neuropathology rather than general pathology (Levine 1987). This is understandable because CP, an invaginating fold of pia within the ventricles, is physically inside the CNS. However, due to absent tight junction barriers between endothelial cells in the plexus capillary network, the CP functions more like a renal-type secretory organ rather than a neural center (Spector and Johanson 1989). Therefore, a useful pathophysiologic concept is the comparison between CP epithelium and the renal proximal tubules (PT), both of which share many functional and ultrastructural similarities. However, CP acts like a reverse kidney in that the primary choroidal function is fluid secretion (Johanson et al. 2008) rather than reabsorption. This reflects Na+ pump localization apically in CP (Parmelee and Johanson 1989) but basolaterally in PT. Still, the similar structural adaptations and transporter protein expression, CP versus PT, prompt comparisons of cross-reactions in the respective basement membrane-epithelial systems (Johanson, Stopa, and McMillan 2010). Many toxicologic effects on CP mimic damage to PT or other nephron regions (Levine 1987).
Great permeability of the choroidal capillaries promotes interendothelial penetration of macromolecules (arrow 1, Figure 1). Consequently, the choroid ISF compartment (Q. R. Smith, Pershing, and Johanson 1981), unlike brain interstitium, is readily accessible to plasma proteins, cytokines, and immunoglobulins. This makes CP parenchyma more vulnerable to systemic disease (Levine 1987) than is the BBB-protected brain. Plasma-borne auto-antibodies and immune complexes (June et al. 1979) wreak havoc with CP basement membrane and epithelium (Peress, Roxburgh, and Gelfand 1981). This weakens the BCSFB and places the brain at risk for CSF-transmitted infections. Immune cell and leukocyte movement through CP displays transfer dynamics different from BBB due to capillary and barrier cell (epithelial vs. endothelial) variations (Engelhardt and Sorokin 2009). Paracellular permeability in BCSFB is greater than at BBB (Thomas and Segal 1998). Thus, distinctive properties of the CP explain the differential barrier penetration of pathogens, xenobiotics, and immune cells from blood to CSF compared with their transfer from blood to brain.
Regional differences in structure and function occur in CP tissues of lateral, third, and fourth ventricles. Ventricular CP variations include epithelial cell pH, K+/Na+, organic anion transport capability, water content, and blood flow (Harbut and Johanson 1986; Murphy and Johanson 1990; Pappenheimer, Heisey, and Jordan 1961; Szmydynger-Chodobska, Chodobski, and Johanson 1994). Inflammatory responses also vary in third ventricle CP versus the structure of the CP in other regions (Levine, Saltzman, and Ginsberg 2008). Given the regional differences in CP baseline physiology and structure, one expects variations in how a particular CP responds to pathogens and toxic agents. Neuropathologic differences across CP regions have implications for functional variations. Similar principles apply to comparison of the heterogeneous ependyma that display unique profiles of cilia and junctional apparatus. Particular toxicologic effects in ventricular regions can often be ascribed to peculiar features of each type of CSF-bordering cell.
Surface epithelium is a term used interchangeably with CSF-bordering cells. Normally, and especially in disease states, there is sloughing of choroidal and ependymal cells into CSF. Surface epithelium was found in 5–10% of human patients with CNS inflammation, neoplasms, compression, and seizures (Wessmann et al. 2010). However, there were no significant associations between specific disease types and the incidence of surface epithelial cells in CSF. Further methodological refinements in analyzing surface epithelial cytology in CSF may yield data to distinguish specific CNS disorders.
Complementary Circulatory Systems in the Brain
Understanding differences in molecular trafficking across BCSFB versus BBB, with patho-toxicologic implications, depends on precise knowledge of circulatory inflow and vascular ultrastructures. CNS uses two circulations to convey trophic/metabolic materials into and out of brain. Such a dual arrangement of the vascular and CSF circulatory systems sets up a complex steady state of nutrient influx and catabolite efflux for neuronal networks (Johanson 2008). Blood supplies the brain intrahemispherically from the inside via the cerebral capillaries. Concurrently, from the outside of the hemispheres, CSF provides supportive materials to neurons from fluid cavities surrounding both the internal (ventricular) and external (subarachnoid) surfaces of the brain. Both the vascular and CSF circulations have secretory (influx) as well as reabsorptive (efflux) components of solute distribution.
Why does the brain need CSF throughput in addition to the primary vascular perfusion? Complex circulatory physiology, linking CSF hydrodynamics to cerebral hemodynamics, relates to the specialized anatomy and needs of the CNS. In a drainage capacity, the CSF uniquely acts as a quasi-lymphatic system (Johanson 2008). Moreover, by being a shock absorber, CSF dampens vascular pulsations (Zou et al. 2008) that would otherwise physically damage the fragile brain capillaries (Stopa et al. 2008). Third, CSF is proximate to periventricular neurogenic niches. Consequently, CSF constituents support stem cells and neurons via specialized choroidal secretions in fetal life (Redzic et al. 2005) when brain capillary density is low. Later in old age when cortical microvessel functions dwindle (Miller et al. 2008; Silverberg et al. 2010), CP secretions may become important to support cerebral angiogenesis as well as neurogenesis. Working in tandem, the CSF and blood provide complementary circulatory support to maintain cerebral metabolism and structural integrity.
Parallel arterial input of materials to CNS is mediated by two systems that are foremost in transferring substances: the BBB (microvascular endothelium) and BCSFB (choroidal epithelium). Each major transport interface features a distinctive array of ions/molecules exchanged between blood and CNS regions (Johanson et al. 2008). A prominent feature of CP is its great secretory abilities. In this case, the nexus mediates material flow from the CP origin to putative targets in CNS (Johanson, Miller, et al. 2010). Compartments of the CSF nexus, diagrammed in Figure 1, are analyzed in the following in the sequence of the physiological flow.
Distributional Nexus: Choroid Plexus-CSF-Ependyma-Brain
Substances secreted by CP tissues into the ventricles traverse a series of boundaries and compartments to reach potential downstream receptors at neuronal and glial membranes. For perspective on the CSF-brain interior, some distinguishing features of the tissue compartments and systemic properties of the nexus are addressed in the following. Fine histological features of epithelial cells and organelles are presented further along in the review.
Choroid Plexus as a Mini-Organ
CP is a secretory engine that actively synthesizes and transports a plethora of inorganic and organic solutes destined for brain (Johanson 2008). Collectively, the plexus tissues are a small fraction (~0.002) of total brain mass. Industrious activity of the epithelium, however, necessitates a choroidal blood flow rate that is 5- to 10-fold greater than mean cerebral blood flow (CBF) (Kadel, Heistad, and Faraci 1990; Szmydynger-Chodobska, Chodobski, and Johanson 1994). Brisk vascular perfusion of CP supports the energy and substrate demands of a high degree of epithelial metabolism. As the main generator of CSF, the CP epithelia in the four ventricles transfer most (up to 75%) of the water molecules diffusing from plasma into CNS. Prolific water movement through CP epithelial aquaporin 1 channels (Oshio et al. 2003) into the ventricles produces the CSF medium to convey solutes into and out of brain. Continual fluid production by CP is the driving force behind steady CSF streaming along the neuraxis.
Ventricular Cerebrospinal Fluid
Understanding how the CP epithelial and ependymal workings impact the brain is predicated on knowledge of CSF dynamics. Cells that border the ventricular system affect, and are affected by, the CSF volume, flow, pressure, and composition. Dynamic interplay between CP and ependyma, with CSF as the intermediary, reveals the multiple homeostatic actions of cells lining the brain’s internal surface. Upon disruption by pathogens and toxicants, the consequent CSF dyshomeostasis impairs brain metabolism. This is presumably due to compromised transport and barrier mechanisms. Salient features of CSF as a circulating medium are discussed in the following relative to toxico-pathology.
Regular turnover of CSF in the ventricular system allows optimal neuronal functioning. Human CSF is renewed three to four times daily (Silverberg et al. 2002). Flow of CSF is from lateral ventricles to the third and then through the narrow mesencephalic aqueduct into the fourth ventricle. CSF advances toward drainage sites (Grzybowski et al. 2006) by bulk flow (arrows 4 and 9, Figure 1) driven by a hydrostatic pressure gradient between CSF and venous blood (Johnston, Boulton, and Flessner 2000; Pollay 2010). CSF bulk flow distributes endogenous materials widely throughout the CNS. The term 3rd circulation has been coined for CSF flow in regard to its major role in central fluid dynamics. Several minutes after intracerebroventricular (ICV) injection, radiolabeled test tracers are swept by CSF convection to regions distant from lateral ventricles (Ghersi-Egea et al. 1996). Substances both beneficial (vitamins) and harmful (cytokines) rapidly distribute throughout the CNS after gaining access to ventricles and downstream (subarachnoid) regions (Figure 1).
Lacking true lymphatic capillaries for drainage, the CNS needs the CSF to continually flow in order to excrete potentially harmful cerebral catabolites and to remove peptides that leak into the brain. Concentration gradients, from brain ISF to ventricular CSF, promote net diffusion of cerebral metabolites and excess proteins into the ventricles (arrow 8, Figure 1). Such CSF sink action (Parandoosh and Johanson 1982) rids the brain of metabolic products and toxic agents/pathogens that access CNS. Overwhelming the clearance or excretory capacity of CSF by diseases/toxicants may injure the brain and protective CP/ependymal cells.
Ependyma/Subependymal Regions
Ependymal and periventricular regions quickly take up toxic as well as trophic substances transported into CSF by CP (arrows 1–5, Figure 1). The permeable CSF-brain interface in the lateral ventricles permits extensive, dynamic diffusion of endogenous/xenobiotic agents into (arrow 5, Figure 1) and out of (arrow 8, Figure 1) the brain. Ependymal destruction by toxicants and diseases further distorts the delicate biochemical balance between CSF and ISF (Johanson 2008). This imbalance neurochemically destabilizes the periventricular regions. That a steady supply of micronutrients and trophic factors is critical for ependymal well-being is exemplified by the finding that malnutrition leads to deformation and metabolic disruption of ependyma, at least early in development (S. P. Sharma and Manocha 1977).
Brain Regions Adjacent to Ventricular CSF
Major neuronal networks (cholinergic, serotonergic, etc.) lie in subependymal, neurogenic regions proximate to CSF (Miyan, Nabiyouni, and Zendah 2003). Subventricular neurons are accessible to endogenous and exogenous materials that diffuse from CSF across the ependyma (Figure 1). Modulated regions include the hippocampus (engaged in memory), the SVZ and dentate gyrus (neurogenesis), hypothalamus (endocrine and autonomic regulation), periaqueductal gray (pain regulation), and the pons-medulla (respiratory and cardiovascular control centers). These homeostatically sensitive areas exhibit compensatory responses to alterations in CSF osmolality, [K+], [Na+], [Cl−], [Ca++], pCO2, pO2, and pH. Therefore, periventricular regions need to be carefully protected against debilitation by toxic/pathogenic insults from foreign agents in ventricular fluid.
Choroid Plexus-Ependyma Disruption: Implications for Brain
Brain health relies upon sound barrier/homeostatic mechanisms in CP and cerebral capillaries for protection against blood-borne toxicants. Impermeable tight junctions (Brightman and Reese 1969) and reabsorptive solute transporters at BCSFB interfaces (Spector and Johanson 2010b) normally protect the CNS against attack from foreign agents (Zheng 2001). Advanced aging, infectious agents, and toxic materials, especially with chronic exposure, can all undermine the integrity of the transport interfaces (Behl et al. 2009; Shi and Zheng 2007). Compromised barriers render the CNS vulnerable to injury. The BCSFB in CP, as a regulated gateway for molecular and cellular traffic into the brain (Johanson 2008), will be highlighted in the following.
Weakening of BCSFB augments penetration of harmful plasma-borne materials into CSF and ultimately into the brain. Collapse of CP-CSF results from many stresses on CNS: arterial hypertension (Murphy and Johanson 1985), transient forebrain ischemia (Palm et al. 1995), hyperthermia (H. S. Sharma, Duncan, and Johanson 2006), traumatic brain injury (H. S. Sharma, Zimmermann-Meinzingen, and Johanson 2010; Szmydynger-Chodobska et al. 2009), and advanced aging (Preston 2001). Upon BCSFB damage in pathophysiologic states, the increased paracellular permeability allows accession of plasma proteins and other markers to CSF. Commonly, the elevated protein concentration in CSF leads to ventriculomegaly and periventricular edema (H. S. Sharma and Johanson 2007). As a consequence, cognitive/behavioral abilities in animals are compromised when CSF-bordering hippocampal and hypothalamic regions are injured by physical stressors (H. S. Sharma, Duncan, and Johanson 2006). However, there are relatively few systematic analyses of agent stress on the CP-CSF-ependyma-SVZ-brain nexus (Figure 1).
Normally the CNS transport interfaces ward off mild to moderate threats by invading bacteria, viruses, and xenobiotics. Vigorous challenges by virulent pathogens and toxic drugs (Levine 1987), though, reduce the homeostatic reserve of transport interfaces. This leads to damage of CSF-contacting neural regions. It is not feasible to predict exactly the untoward responses by CP to potent noxious materials. A particular toxicant can interfere with tight junctions or with the nutritional/trophic transporters at the BCSFB (Spector and Johanson 2010b) that vectorially direct solutes to neuronal targets via CSF. Extreme interference with CP function causes CSF dyshomeostasis that harms brain.
Ventricular CSF-Bordering Cells: Histology and Physiology
Before reviewing effects of organic agents on CSF-mediated distribution, we describe normal ultrastructure and function of ventricle-contacting cells. Three types of cells demarcate ventricular CSF: choroid epithelial, ependymal, and circumventricular (Table 1). Within these major groupings are subpopulations of the parenchymal CP, ependyma, and CVOs. Cellular heterogeneity and phenotype importantly bear on evaluations of specific endocrine, pharmacologic and toxicologic responses. Cells contacting the CSF are part of a dual classification of neuroepithelia: those suspended in CSF (CP) or those comprising the ventricular wall (ependyma and CVOs).
Characteristics of epithelial cells bordering the ventricular cerebrospinal fluid (CSF) system.

a LV, 3V, and 4V refer to the lateral, third, and fourth ventricles, respectively.
b LV ependyma have intercellular gap junctions, whereas some 3V ependymal cells have tight junctions.
Choroid Plexus Epithelial Cells
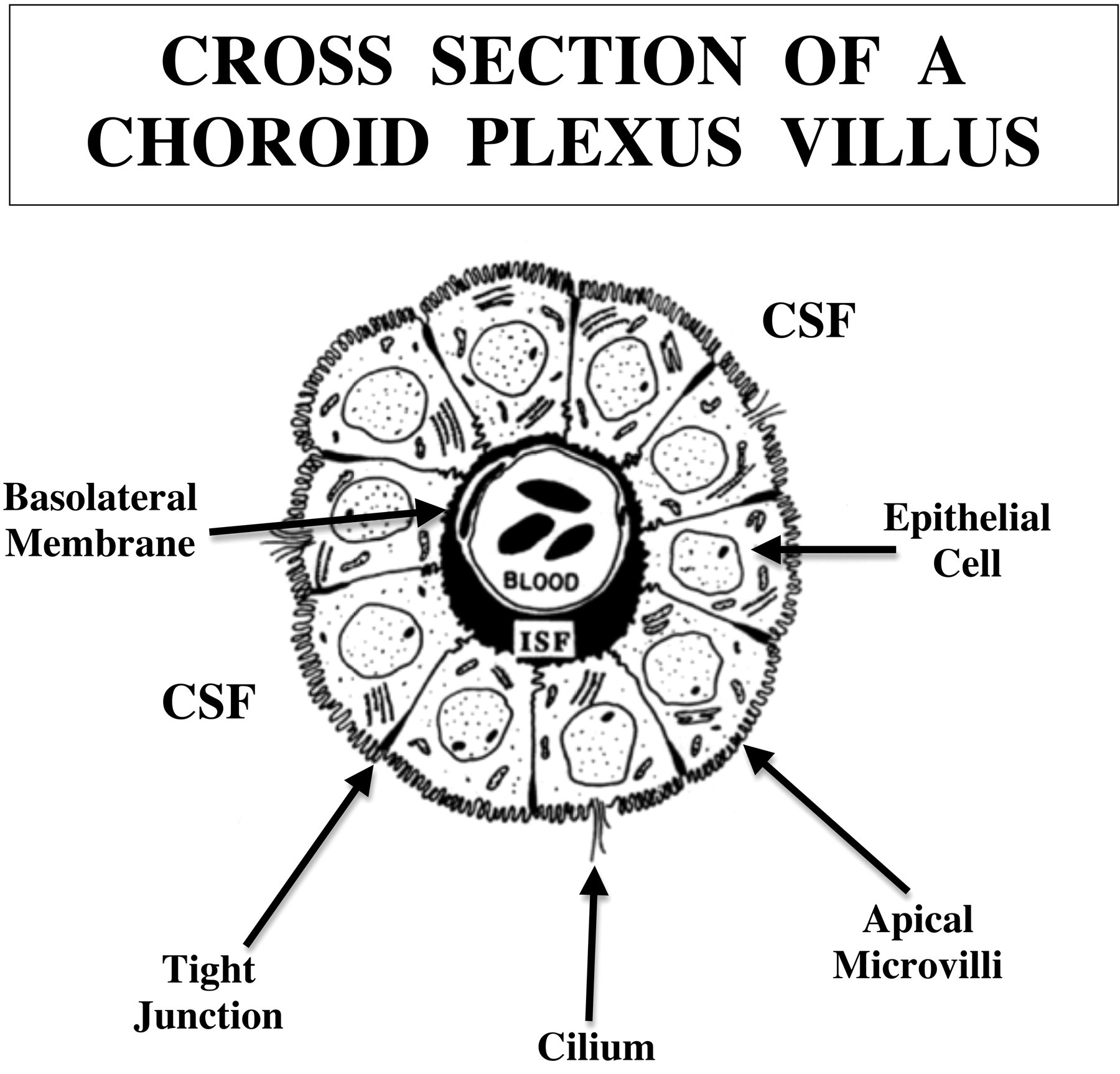
Anatomically, the base of the choroid epithelial fronds is anchored to the brain at fixed points in the lateral, third, and fourth ventricles. Ramifying outward from where the stalk attaches to the ventricular wall, the tufts of CP are suspended within CSF. Physically this arrangement provides enormous epithelial surface area for translocation of nutrients and drugs between plasma and CSF. However, this same large transport area is potentially available for untoward leakage of plasma proteins and harmful agents into CSF. The basic parenchymal element in CP is cuboidal epithelium. An ultrastructural image of a typical CP epithelial cell is portrayed in Figure 2. Large surface areas promote extensive transport across the basolateral (plasma-facing) and apical (CSF-facing) membranes. Because the BCSFB is equipped for heavy-duty transport, severe pathologic damage to CP markedly distorts CSF homeostasis and ventricular configuration.
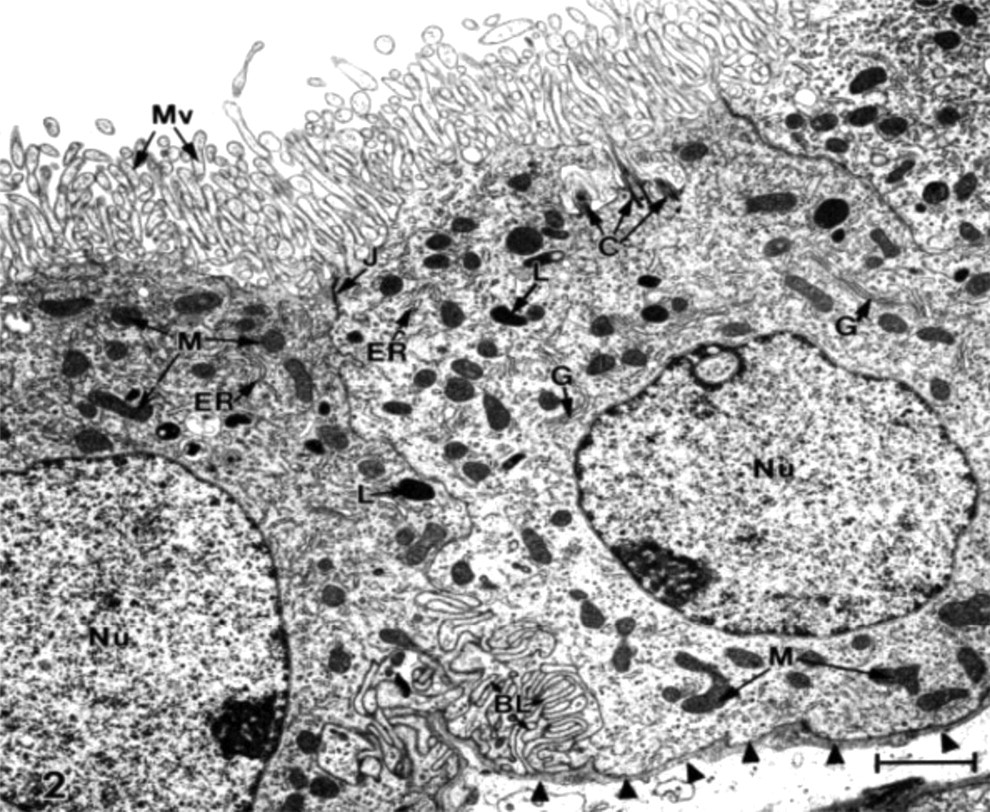
Ultrastructure of typical choroid plexus (CP) epithelium.
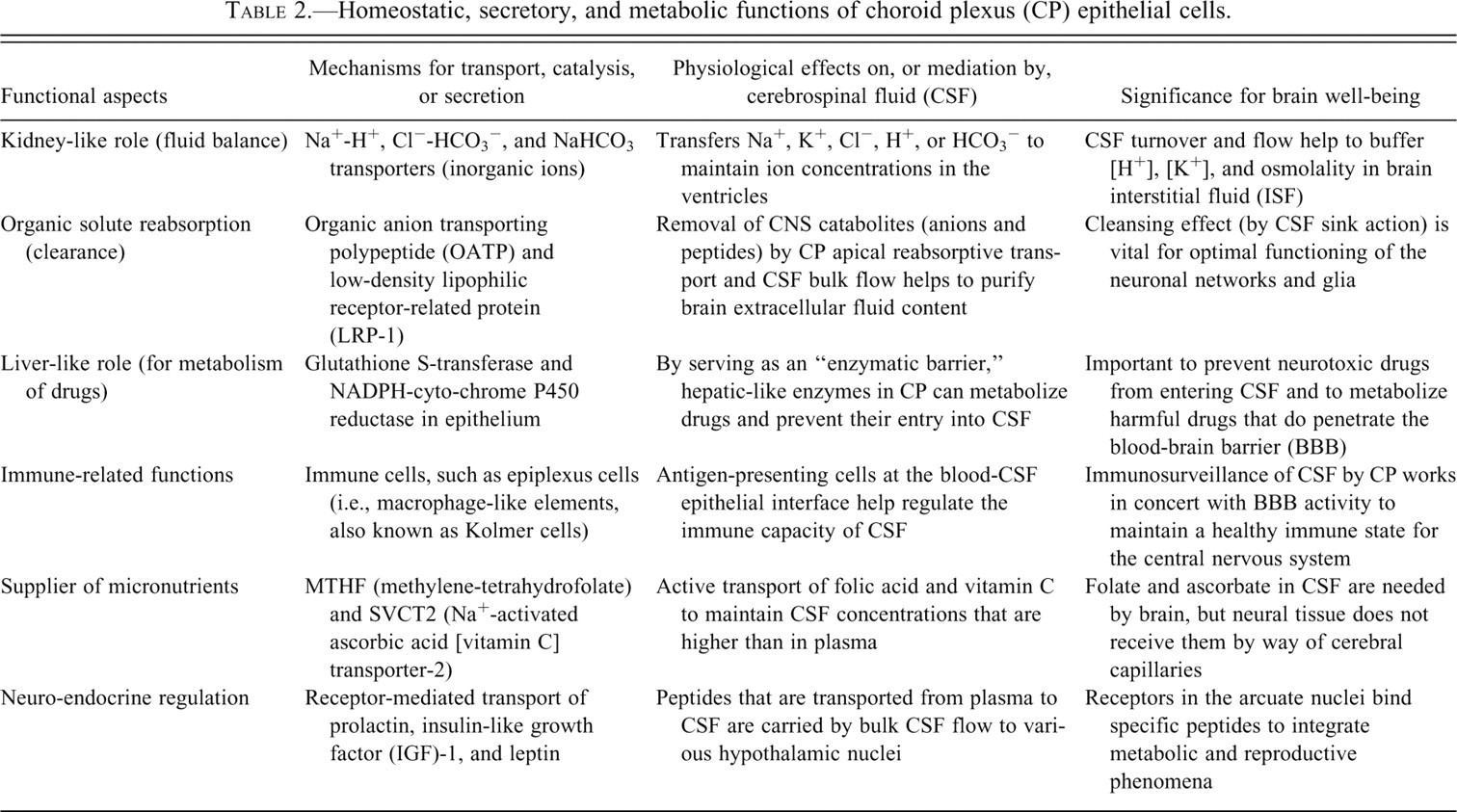
As a multipurpose organ, the CP carries out diverse tasks: secretory, synthetic, and reabsorptive (Table 2 ). Best known among the secretory functions is CSF formation. CSF forms at the uniform rate of ~0.4 ml/min/g CP in mammals. To initiate CSF formation, the basolateral membrane takes up Na+, K+, Cl−, and HCO3 − via several ion co-transporters and exchangers (Johanson et al. 2008; P. D. Brown et al. 2004). Ions and water taken up from the interstitium by epithelial basolateral membrane (arrow 2, Figure 1) then move through cytoplasm to the opposite (apical) side. There the apical membrane extrusion mechanisms (arrow 3, Figure 1) include ion channels, aquaporin 1, and co-transporters. Apical membrane Na+ pumping (Q. R. Smith and Johanson 1980), by setting up the appropriate transmembrane ion gradients, actively propels CSF production.
Homeostatic, secretory, and metabolic functions of choroid plexus (CP) epithelial cells.
Drugs that induce renal natriuresis or diuresis consistently reduce CSF turnover into the ventricles. CSF formation is inhibited 10–30% by sympathetic, cholinergic, and serotonergic neurotransmitters (Nilsson, Lindvall-Axelsson, and Owman 1992). Neuropeptides such as arginine vasopressin, atrial natriuretic peptide, and angiotensin II also slow down CSF production by 15–20% (Johanson et al. 2008). Upon secretion, the nascent CSF mixes with brain extracellular fluid as it flows down the neuraxis. CSF flow decreases in aging. In neurodegenerative disease (Silverberg et al. 2001) or drug poisonings, the CSF turnover rate (i.e., formation rate / volume can be cut in half. Adverse consequences include reduced clearance, and hence accumulation, of proteins and cerebral metabolites. ISF retention of uncleared waste products impairs brain metabolism and behavior. Normally the CSF with its debris is efficiently reabsorbed into lymph and/or venous blood at multiple sites in arachnoid tissue (Grzybowski et al. 2006; Johnston et al. 2004).
In addition to forming CSF, the CP homeostatically protects the brain (Table 2). Acting like a kidney (Spector and Johanson 1989), the CP helps to keep the chemical composition of CNS extracellular fluid in a stable state. In true kidney-like fashion, the CP actively sets the ionic composition, pH, and osmolality of extracellular CSF. Low permeability imposed by tight junctions at the BCSFB allows ion and molecular gradients to be established secondary to active transport. For example, Ca++ and K+ in CSF are held at concentrations lower than plasma, while Mg++ and Cl− are kept at higher levels. CSF titers of numerous growth factors, fluid-regulating peptides, and proteins are maintained at 0.01 to 0.001 of their respective concentrations in plasma. Regulated CSF ions benefit the neurons that require finely maintained extracellular concentrations.
Second, a plethora of reabsorptive solute transporters in the apical membrane actively removes potentially toxic organic acids and peptides from CSF (arrow 10, Figure 1). Organic anion transporting polypeptides (OATPs) are part of an increasingly appreciated gene superfamily (Spector and Johanson 2010b). OATP was initially immunolocalized to the CP apical membrane (Angeletti et al. 1997), a strategic location for clearing metabolites from CSF (Ohtsuki et al. 2003). In BCSFB, the OATPs mediate reabsorptive (excretory) transport of a wide spectrum of amphipathic organic solutes in CSF: steroid conjugates, neurotransmitter anion catabolites (homovanillic acid, a major metabolite of catecholamine neurotransmitters), bile salts, anionic oligopeptides, drugs, and xenobiotics (Hagenbuch and Meier 2003). CSF levels of certain drugs are kept at subtherapeutic low levels by OATP and the P-glycoprotein transporter, the potential roles of which in CP need evaluation for therapeutic agents such as suramin and eflornithine, agents that are used against human African trypanosomiasis (Sanderson et al. 2008; Sanderson, Khan, and Thomas 2007).
Another CP reabsorptive transporter of organic solutes, with a wide spectrum of substrate affinity, is the family of low density lipophilic receptor-related proteins (LRP). Located apically, the LRP-1 and LRP-2 clearance transporters keep peptides and peptide fragments in the CSF from building to toxic levels (arrow 10, Figure 1). As a promiscuous transporter, LRP-1 can clear from CSF/brain as many as forty types of molecules including APOE4, amyloid precursor protein, and amyloid oligopeptide fragments (Crossgrove, Li, and Zheng 2005). LRP-2 (or megalin) has also been implicated in removing amyloid from CSF (Alvira-Botero and Carro Forthcoming). Other apically located organic solute transporters, including PEPT2 (proton-coupled oligopeptide transporter 2), prevent CSF accumulation of compounds (e.g., 5-aminolevulinic acid [ALA], a heme precursor) to toxic levels (Hu et al. 2007; D. E. Smith, Johanson, and Keep 2004). Reabsorptive transport from CSF is essential because ALA leaks into CSF from blood and needs to be quickly cleared from the ventricles (arrow 10, Figure 1) to avoid toxicity (Terr and Weiner 1983).
Third, hepatic-like metabolism in BCSFB epithelium minimizes toxic drug accumulation in the CSF and CNS. This liver-type role affords brain an extra line of defense against toxic drugs and compounds (Strazielle, Khuth, and Ghersi-Egea 2004). Xenobiotic molecules induce drug-metabolizing enzymes in liver and CP. In vitro, the expression of UDP-glucuronosyltransferase (UGT1A6 isoform) is upregulated in rat CP epithelium when tissue is challenged with 3-methycholanthrene (3-MC) or paraquat (Gradinaru et al. 2009), leading to a two- to threefold increase in the choroidal 1-naphthol glucuronidation activity. UGT1A6 mRNA expression (polymerase chain reaction [PCR] analysis) was also augmented more than twofold after incubation with these two drugs. Drug-metabolizing enzymes in the BCSFB transform xenobiotics that might harm the brain if left unmetabolized.
Fourth, an immune-type function is subserved by CP through the activity of antigen-presenting cells in the epithelial lining (Nathanson and Chun 1989), thereby permitting immune surveillance of CSF. Normally just a few immune cells migrate from plasma into ventricles to monitor CSF immunologic status. Under pathologic or toxicologic stress, however, the upregulation of CP chemokines, integrins, selectins, and matrix metalloproteinases renders the BCSFB more penetrable (Prendergast and Anderton 2009). Consequently, many inflammatory cells invade CSF and provoke CNS autoimmune disease. Since CP epithelium engages in diverse tasks to defend and cleanse CSF, the BCSFB itself needs protection against toxicant and foreign molecules (Johanson, Silverberg, et al. 2005). High levels of antioxidant glutathione in CP (Cooper and Kristal 1997) comprise a biochemical defense against oxidant molecules transported or metabolized by epithelial cells. Overall, the CP in healthy young adult humans has sufficient barrier, metabolic, enzymatic, and reabsorptive capabilities to keep CSF composition sufficiently pure for optimal neuronal performance.
Finally, over and above its homeostatic role in stabilizing neurochemical composition and immune health of CSF-CNS, the CP is the chief supply route for transferring plasma-borne materials into the ventricles. As much as 70–80% of water, ion, vitamin, and peptide transfer into CNS occurs across choroidal epithelium (arrows 2 and 3, Figure 1) (Johanson et al. 2008). Vitamins B (folate) and C are actively transported into CNS preponderantly via the blood-CSF interface (Spector and Johanson 2006). There is also significant neuroendocrine peptide distribution mediated by hormonal transport mechanisms at the BCSFB interface (Kozlowski 1986). Prolactin, insulin-like growth factor (IGF)-1, and leptin use CP as a hormonal signal relay station in being transported from plasma to ventricles (Dietrich et al. 2008; Redzic et al. 2005). Then these peptides are convected by CSF bulk flow to hypothalamic regions such as the arcuate nuclei (Rodriguez, Blazquez, and Guerra 2010). In this manner, the CP-CSF participates in hormonal signal transfer within neuroendocrine feedback loops for modulating feeding/satiety and reproductive behaviors.
Translocation of plasma ions, micronutrients, and water into CNS is mainly via CP. Serious injury of BCSFB by microbes or xenobiotics alters brain homeostasis. When the CSF milieu is substantially changed, brain regions close to ventricles suffer damage to neurogenesis and cognition. Many pathogens, toxicants, and neurological disorders damage the plexus. Following stroke and trauma, not only is CSF delivery of trophic factors to neurons compromised, but also the CSF removal of brain catabolites is diminished (Johanson et al. 2000). Molecules in ventricular CSF, even large proteins, have access by diffusion (down concentration gradients) to periventricular brain regions (arrow 5, Figure 1). In the reverse direction, catabolically generated macromolecules in the hemispheres diffuse from interstitium into the ependyma into the ventricular CSF (arrow 8, Figure 1). Proper steady-state partitioning of molecules between CP-CSF and brain requires intact ependyma.
Ependymal Cells with Gap Junctions Versus Tight Junctions
Structurally and functionally, the ependyma lining the cerebral ventricles and central canal of the spinal cord is heterogeneous. Ontogenetically, there is progression of ependyma as a multiple layer in the fetus to a single layer in adults. Various cell types comprise the fetal ependymal wall (Davson and Segal 1996). These include the tanycyte and ependymal astrocyte as well as the true ependymal cells that persist into adulthood. All three cell types have cilia and microvilli at their CSF-facing apical surfaces. However, structural differentiation occurs at the basal (brain-facing) sides. Tanycytes and radial glia send long dense fibers to the CSF. The tanycyte’s other end projects basally to termini in certain hypothalamic nuclei, which receive via axonal flow those neuropeptides (“endocrine signals”) taken up from the CSF (Rodriguez et al. 2005). The ependymal astrocyte is a columnar cell with branching peripheral processes. After birth, both tanycytes and ependymal astrocytes dwindle in number so that the ventricular lining in adults is mainly ependyma proper. Even in adulthood the ependyma depend on growth factors such as vascular endothelial growth factor (VEGF) and transforming growth factor beta (TGFβ), both secreted by CP, to maintain stable cellular structure and fluid balance with surrounding ISF (Maharaj et al. 2008). Diminished growth factor support for endothelial and ependymal cells leads to vascular permeability changes and periventricular edema.
Ependymal cells proper are not all equal. Heterogeneity among ependyma consists of cells having cilia or not, and in possessing leaky gap junctions or the tighter zonulae occludentes (not as prevalent). Most lateral ventricle ependymal cells have intercellular gap junctions just under the apical surface. Gap junctions are highly permeable. Large proteins, the size of ferritin (mw ~445,000), readily diffuse through the paracellular space to cross the ependyma and thus easily move between CSF and brain. Accordingly, the ependymal interface between ventricular fluid and underlying brain is not regarded as a CSF-brain barrier. Being highly permeable, the ependyma is thus unlike the restrictive BCSFB and BBB, which impede paracellular diffusion.
Although the lateral ventricle ependymal system is homogeneous, the ependyma lining the third and fourth ventricles are heterogeneous. In specific regions of third ventricle (3V), there are tight junctions between specialized ependymal cells. Given the proximity of 3V to hypothalamus, the structurally modified ependyma here reflects specialized (regulated) functional relationships between 3V CSF and the adjacent paraventricular nucleus. In regions such as the arcuate nucleus (Rodriguez, Blazquez, and Guerra 2010), the nearby CSF has “access” to hypothalamus in that hormones exchange via diffusion across the ependymal interface. Brain midline CVOs (Gross 1992) in 3V and 4V are especially heterogeneous.
More than being a structural boundary between ventricular CSF and brain, the ependyma is a dynamic interface mediating the movement of cilia, CSF, and migrating neuroblasts. On the ependymal apical surface is a network of cilia that beat in coordination to facilitate CSF circulation. Each cilium is a subcellular organelle emanating from the cell’s interior. Planar cell polarity signaling may be controlled by cilia (Fischer and Pontoglio 2009). Cadherin genes (Celsr2 and Celsr3) regulate planar cell polarity. Celsr genes that are mutated or absent (knocked out) compromise the development and planar organization of ependymal cilia. This leads to defective CSF dynamics and hydrocephalus. Many ciliopathies render CP and ependyma dysfunctional, thereby distorting the volume and composition of CSF secretion (Banizs et al. 2005). Migration of neuroblasts from the lateral ventricle wall to the olfactory lobe relies upon ciliary-guided flow to forebrain sites for insertion into circuits as interneurons (Sawamoto et al. 2006). Proper development and operation of the brain depend on normally functioning cilia in CP and ependyma.
Currently surging research on neurogenesis in the SVZ is generating additional insights on ependyma. As a delicate interstitial microenvironment for stem cells (Spector and Johanson 2007a), the neurogenic niches of dentate gyrus and SVZ require compositional stability. This assures finely regulated gliogenesis and neurogenesis, even in adulthood. Repair mechanisms are essential to the integrity of the ependymal lining disrupted by diseases (hydrocephalus) and disorders (trauma). Ependyma are also damaged in advanced aging. One restorative mechanism is SVZ-mediated repair of the ependymal wall by astrocyte insertion in regions where ependyma are detached. When new astrocytes incorporate in gaps between ependymal cells, they take on antigenic and morphologic characteristics of neighboring ependymal cells (Luo et al. 2008). This constitutes evidence for non-neuronal repair as well as the more established neuronal reconstitution carried out by SVZ elements.
In addition to dynamic interaction between ependyma and SVZ for repair mechanisms, there is homeostatic interplay between CSF, ependyma, and the periventricular brain in fluid balance. Even though the CSF-brain interface permits macromolecule diffusion between ependymal cells, implying unregulated water and ion paracellular movements, there is still great plasticity of expression of ion and water channels in ependyma. Responding to elevated CSF pressure or a change in CSF Na+ concentration, the ependyma upregulates aquaporin 4 water pores and epithelial Na+ channels (H. W. Wang et al. 2010). This may reflect regulation of ependymal cells per se, or controlled fluid transfer into brain across ependymal regions with tight junctions. Whatever the explanation for induced channel expression, the findings point to sensitivity of the ependymal interface to CSF changes.
Circumventricular Organs in Ependymal Wall
Nested within the third and fourth ventricular walls are specialized regions that house ependymal mini-organs, the CVOs (Vio et al. 2008) (Figure 3 ). Overall their primary functions can be sensory, secretory, or both (see Table 3 ). CVOs bridge endocrine and autonomic phenomena (Uschakov et al. 2009), particularly the homeostatic adjustment of fluid composition, volume, and osmolality (Henry, Grob, and Mouginot 2009). CVOs are sites of action or release of fluid-regulating peptidergic neurotransmitters, cytokines, and hormones (Feher et al. 2010; Sotthibundhu, Phansuwan-Pujito, and Govitrapong 2010). Anatomically, CP is not a CVO because the preponderance of choroidal tissue does not reside in the ventricular wall. Instead, the CP dangles within the ventricular spaces away from the ependymal wall.
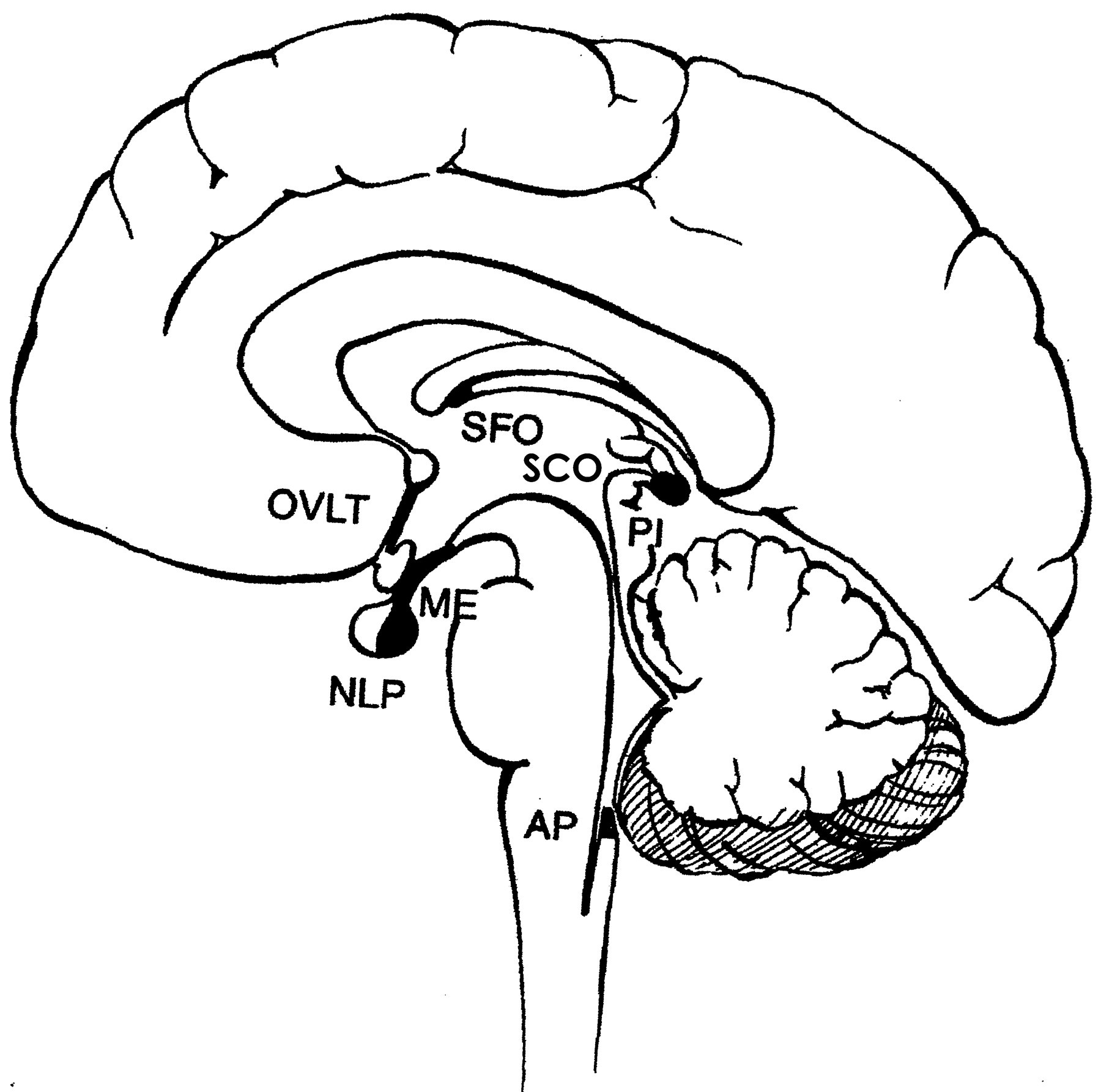
Localization and characteristics of the seven circumventricular organs (CVOs) in the human brain. Occurring mainly as tiny “mini-organs” around the ventricular system margin, the CVOs cluster mainly around the third ventricle. Anatomically the most dorsal CVO is the subfornical organ (SFO). Going clockwise around the third ventricle are the subcommissural organ (SCO), pineal gland (PI), neural lobe of the pituitary gland (NLP), median eminence (ME), and organum vasculosum of the lamina terminalis (OVLT). Further down the neuraxis, more distally in the cerebrospinal fluid (CSF) system, lies the area postrema (AP) near the fourth ventricle. The CVOs are among few sites in the central nervous system (CNS) lacking a blood-brain barrier (BBB). Therefore the parenchymal neurons and ependyma in CVOs sense concentrations of compounds in blood and make homeostatic adjustments to restore fluid balance in brain and periphery. Significantly, the diffusing blood-borne factors or peptide signals are not restricted by the BBB in CVOs. Due to unavoidable circumstances the above figures were not originals but were scanned from reprints.
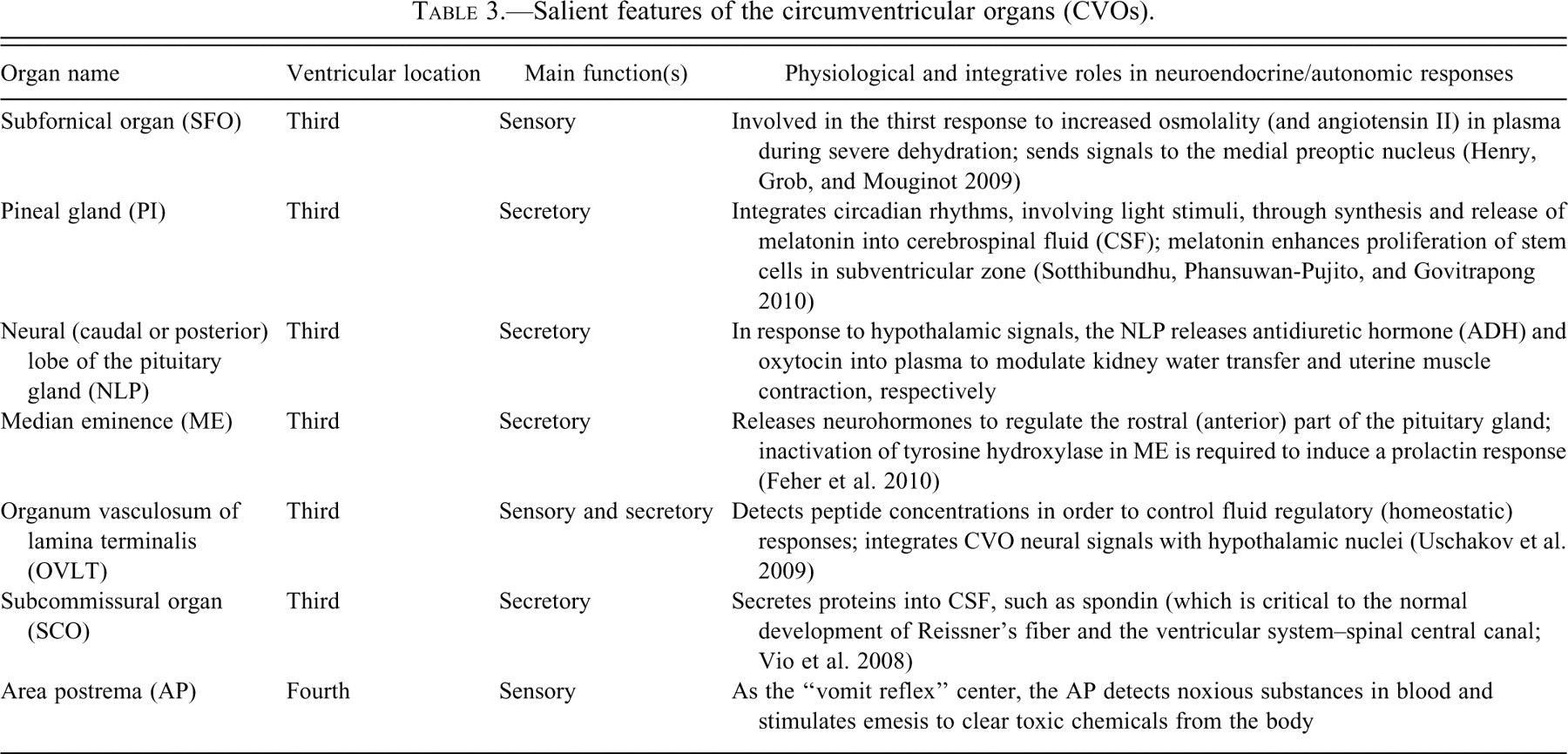
Salient features of the circumventricular organs (CVOs).
Lateral ventricle walls do not contain CVO structures. However, the midline neuraxis running through the third and fourth ventricles has many CVOs. Each one lacks a BBB. The third ventricle harbors the subfornical organ (SFO), subcommissural organ (SCO), and organum vasculosum of the lamina terminalis (OVLT) and connects to narrow recesses leading to the median eminence (ME), neural lobe of the hypophysis (pituitary gland [NLP]), and the pineal gland (PI). Protruding into the fourth ventricle is a vasculo-epithelial pouch called the area postrema (AP). CVOs receive peptide signals from CSF as well as plasma. Similar to CP, a prominent and distinguishing feature of SFO, SCO, OVLT, and AP is the permeable capillary network allowing peptide signals to diffuse from plasma to sensory receptors in the CVO interior. Peptide signaling is transduced by neurons to electrical impulses for neurotransmission out of CVOs toward central regions that integrate reflex responses to altered plasma chemistry. The SFO, for example, receives enhanced angiotensin II signaling from plasma for integrating the thirst response to dehydration by stimulating drinking. Each CVO domain is encompassed by an epithelial ring (with tight junctions) that physically and functionally separates the mini-organ’s “business” from surrounding brain. Consequently, the CVO’s extracellular milieu bears a closer resemblance to plasma than to brain interstitial fluid. CVOs are “open physiologic windows” into the brain. They allow CNS reception of plasma peptide signaling molecules that would otherwise be centrally excluded by the “closed door” of the BBB.
CVOs can be categorized into two groups according to their location in the ventricles: those at the confluence of the ventricular cavities (SFO, SCO, and AP) and those lining the ventricular recesses (NLP, OVLT, and PI). Vascular architecture, topographically, varies from one CVO to another (Duvernoy and Risold 2007), but the cardinal feature of each CVO is a leaky BBB allowing intimate barrier-free contact between blood and neural tissue. Open capillaries in CVOs means that endothelial transporters are not needed to actively move substrate (e.g., peptides) into CNS.
CVOs are functionally like CP in having highly permeable capillaries open to nearby ISF. Contrariwise, due to BBB restriction on diffusion, the brain ISF is not in open passive communication with blood. However the SFO, SCO, OVLT, and AP are unlike the CP in that the former CVOs are engaged in transducing plasma hormonal signals to electrical messages. On the other hand, CP basolateral receptors take hormones from blood (e.g., leptin, IGF-2, prolactin) for transepithelial conveyance into CSF. By endocrine-like CSF bulk flow, these CP-secreted signaling peptides are conveyed to CNS targets (Kozlowski 1986; Scott et al. 1977). Generally speaking, the CVOs and CP take up plasma-borne peptides and relay the “hormonal information,” by neurons or CSF, to peptide-receptor bearing cells in hypothalamus and other central regions. Endocrine systems throughout the body can consequently be modulated by compensatory neural and hormonal output from CNS. Such feedback loops, involving intermediate transport/signaling steps via CP and CVOs, constitute homeostatic machinery for restoring distorted body fluids to a stable, specialized composition. Hence, toxic/pathological injuries to CP, hypothalamus, and CVOs, including periventricular lesions, affect not only the local brain environment but also the activity of endocrine systems throughout the body.
Periventricular Lesions and the Choroid Plexus-CSF Nexus
Periventricular lesions are commonly downstream sequelae of the uncontrolled passage of substances through a compromised BCSFB. Materials that are inappropriately transferred or that leak across the CP include infectious agents, blood components (cells and proteins), and tumor cells. Representative lesions for each group exemplify types of damage inflicted on the ventricular wall and underlying brain. For example, the protozoan Toxoplasma gondii frequently infects the CNS of immunocompromised AIDS patients. Acting as a staging platform, CP receives and activates Toxoplasma, which in turn engenders choroid plexus inflammation (Falangola and Petito 1993) that promotes CSF dissemination to periventricular sites where infectious agent and leukocytes accumulate (Stahl and Turek 1988). Ependyma and CP in all four ventricles become diffusely necrotic (Bourgouin et al. 1992). Another infection of CP-CSF is by ovine lentivirus, which invades periventricular sites as part of a more generalized encephalomyelitis (Brodie, Bickle, and DeMartini 1995); the CSF lentiviral markers reflect CNS pathology. Various blood cells have greatly restricted access to CSF except in pathophysiologic conditions associated with autoimmune disease or hemorrhage into CP-ependymal zones. Augmented leukocyte (T-cell) penetration across the CP into CSF occurs in relapsing multiple sclerosis (MS) in humans or experimental allergic encephalitis (EAE) models of MS, leading to formation of hallmark periventricular lesions and ependymal/subventricular destabilization. Consequently, neurogenesis is inhibited by T cell release of granzyme B (T. Wang 2010). Recent findings in an EAE model point to T lymphocyte antigen specificity in defining the topography of periventricular lesions (Berger et al. 1997). Moreover, extensive erythrocyte permeation across BCSFB distorts CSF composition and interferes with ependyma/SVZ functions. Thus, in neonatal brain stressed by hypoxia/ischemia, the choroidal/intraventricular and severe subependymal hemorrhaging perturb the interstitial fluid environment in periventricular white matter (Bernert et al. 1988). Tumors (e.g., high-grade B-cell lymphoma) may form directly in the CP (Cecchi et al. 2008) and migrate across BCSFB to become entrenched in the periventricular brain. Metastatic CNS lymphoma, having entered CSF via the CP, can disseminate to periventricular sites (Kobayashi et al. 2009). Primary intracerebral malignant lymphomas, albeit rarely, fulminate in CP and putatively escape the BCSFB to permeate subependymal sites along the lateral, third, and fourth ventricular walls (Shibata 1989). Specific pharmacologic targeting of CP/BCSFB may be used to thwart penetration of cancer cells (as well as viruses and potentially harmful leukocytes) into the CSF-ependyma-SVZ nexus. Studies of toxicant-induced disruption to the nexus, as described in the following, furnish useful basic data for formulating prophylactic strategies against infection or measures to counter toxicant exposure.
Organic Agents Used in Choroido-Ependymal Toxicology Studies
Drugs from different classes have been tested for toxicity to CSF-bordering cells and underlying the brain. Tertiary amines were extensively analyzed for deleterious effects on BCSFB and the CSF-brain interface. A series of compounds analyzed includes molecules with one, two, or three tertiary amines (Levine 1977). All contain a cyclic structure, either saturated (piperazine and piperidine) or unsaturated (naphthyridine and pyridine). Agents used in ventricular cell-CSF investigations, including piperidine/piperazine derivatives and amoscanate, are compiled in Table 4 . Most of these drugs are anti-parasitic. Their CSF toxicity is fairly consistent among mammals. Organic agents listed in Table 4 were used experimentally to inform on the extent and location of CP-ependyma injuries. Adult rats were commonly used for in vivo experimentation. A typical approach involves microscopy and tissue staining, and time course analysis (days to weeks), to assess development and reversibility of drug-induced lesions in CP, ependyma, and brain.
Organic agents used for in vivo toxicology studies of choroid plexus (CP), ependyma, and periventricular brain.
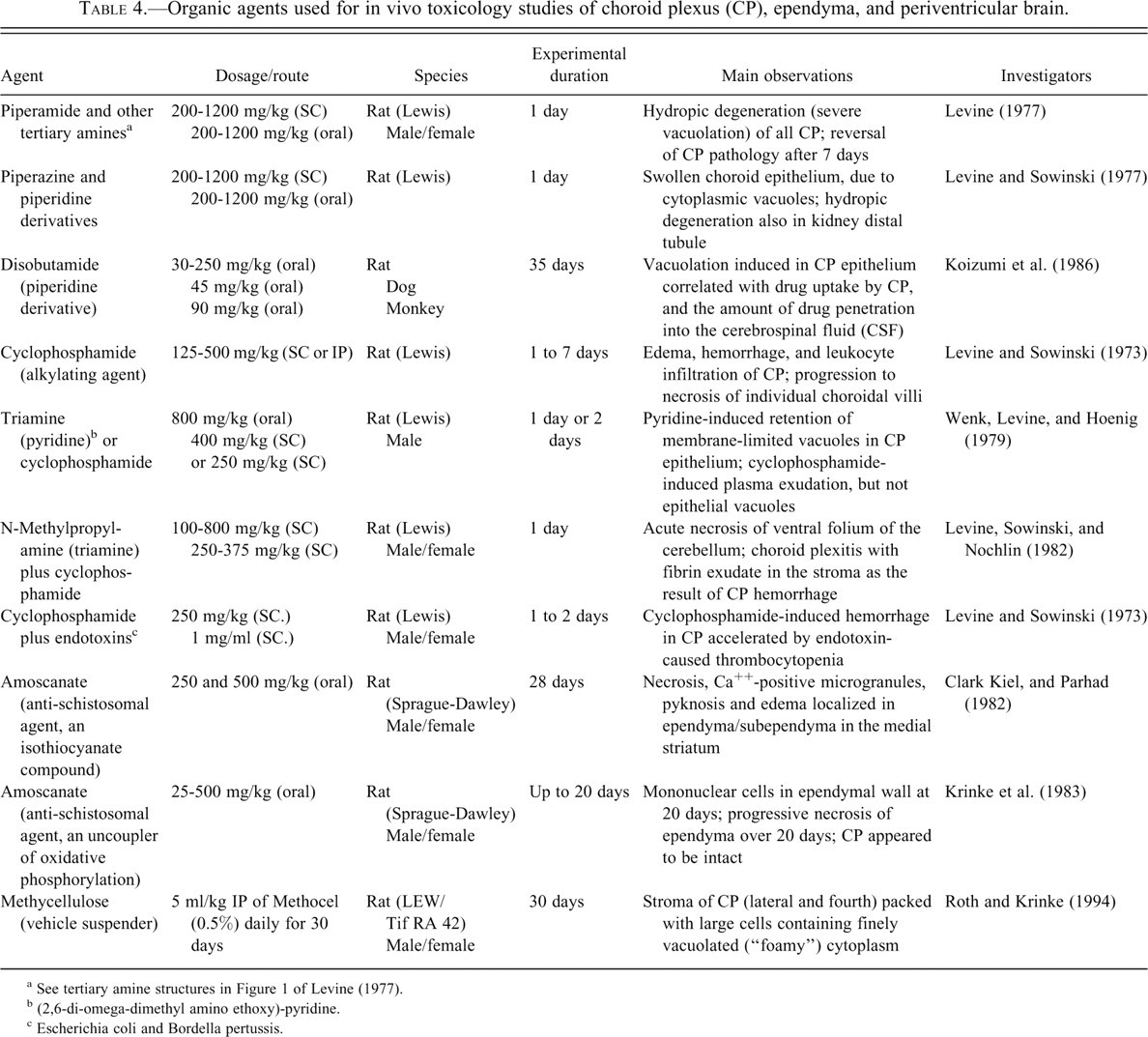
a See tertiary amine structures in Figure 1 of Levine (1977).
b (2,6-di-omega-dimethyl amino ethoxy)-pyridine.
c Escherichia coli and Bordella pertussis.
Vulnerability of the CSF-Brain Interior to Toxicants and Pathogens
A gateway duty has been ascribed to BCSFB (Johanson et al. 2008). Thus, a significant CNS port of entry in CP is offered to plasma molecules, pathogens, and immune cells. Ventricular CSF nourishes and protects brain by an array of mechanisms controlled at the CP epithelial zone and ependymal interface. Due to dissimilar structures and molecular expressions of CP versus ependyma, various toxicants and pathogens exert differential effects on CSF-bordering epithelium.
Upon penetration of a damaged BCSFB, a given substance flows to the ependyma and regions adjacent to CSF (arrows 2–5, Figure 1). Injury to periventricular brain sites (arrow 6, Figure 1), causing dyshomeostasis (Aksamit, Parnell, and Johnson 1999), is often but not always curtailed by enzymatic defense systems at CSF interfaces that degrade penetrating toxicants. In this manner the Pi isoform of glutathione S-transferase that is expressed in ependymal, arachnoid, and pial cells enzymatically buffers the brain against toxicant penetration from CSF (Carder et al. 1990).
Many toxicology studies of CNS have observed drug-induced formation of vacuoles within various neural cell populations. Regionality of induced vacuoles in brain has been a topic of interest (Wells and Krinke 2008). Vacuolation has clinical importance because impaired drug elimination from neurons and epithelial cells, which promotes vacuole generation, may lead to toxicity and even death. With cell swelling and organelle disruption, the protective properties of epithelial cells of BCSFB may become seriously compromised. Therefore, it is important to understand the mechanisms of vacuole formation in CP and ependyma and to prevent vacuole formation and proliferation or counter any untoward effects pharmacologically.
Injuries to Choroid Plexus
Delineating the effects of toxic agents involves consideration of CP compartmental anatomy. Each CP villus has an inner vascular core consisting of a rich network of blood vessels (Figure 4 ). Nearly 20% of CP mass is vasculature and blood cells. These elements impart a red cast to the tissue (Johanson, Reed, and Woodbury 1974). Lying in the intermediate zone of the CP is the interstitial space. It has a highly developed extracellular matrix that modulates the location and functions of both vascular and epithelial cells. The interstitium comprises about 15% of CP mass (Q. R. Smith, Pershing, and Johanson 1981). Notably the interstitium of the CP, like that of brain, lacks true lymphatic capillaries. On the outside perimeter of the choroidal villi is a single layer of epithelium. This epithelial ring interfaces with the interstitium on the basal side and contacts the CSF via a lush microvillous surface at the apical pole. Severe pathologic changes to the CP may include damage to all three compartments: blood vessels, interstitium, and epithelium. Certain drugs destroy one specific compartment, or a single toxicant can perturb all three CP regions (Levine 1987). It is also useful to identify drug combinations that intensify damage to multiple compartments within CP.
Cross-sectional area of a choroid plexus (CP) villus. Reconstructed from micrographs, this schematic portrays three major compartments of the CP: an inner vascular core (20% of tissue volume), the intermediate interstitial zone containing a loose stroma and interstitial fluid (ISF) zone (15% of tissue volume), and an outer circumferential ring of epithelial cells (65% of tissue volume). Solutes in blood readily diffuse out of CP capillaries into and through the ISF up to the epithelial basal membrane, which restricts diffusion (Johanson and Woodbury 1978). To cross the blood-cerebrospinal fluid barrier (BCSFB), a substance must be actively transported across the epithelium (transcellular route) and/or diffuse passively between cells (paracellular route). Diffusion of water-soluble organic solutes is a small proportion of the total molecular flux across CP. Active transporters, facilitated diffusion mechanisms, ion channels, and aquaporins (regulated water pores) conduct transcellular trafficking through the basolateral and apical membranes in the BCSFB. Toxic agents or pathogens can damage one, two, or all three compartments of the villus.
Vascular impairment
Cyclophosphamide (CY) at high doses injures the CP vasculature, especially in the fourth ventricle (Levine and Sowinski 1973). Although the plexus vessels bear the brunt of attack, other compartments of CP eventually become injured. CY is an experimental tool for rupturing the BCSFB and promoting penetration of water-soluble drugs into the ventricles. In rats, a destabilized CP vasculature leads to hemorrhage, fibrin plugs in capillaries, focal leukocyte infiltration, and exudation from impaired vessels (Figure 5 ). Edema fluid contains phagocytes and erythrocytes. In severe cases, CY-induced hemorrhagic infarction spreads over large expanses of CP in one or more ventricles. Substantial passage of fluid out of choroidal capillaries compresses the epithelium (Levine and Sowinski 1973). Destabilization of CP initiated by the grossly ruptured vascular elements and adjacent interstitium is depicted in Figure 5. Due to additive pathologic effects and BCSFB breaching, the administration of CY followed by a tertiary amine (acting by a different mechanism) lessens the amine dose that damages periventricular brain (Levine, Sowinski, and Nochlin 1982).
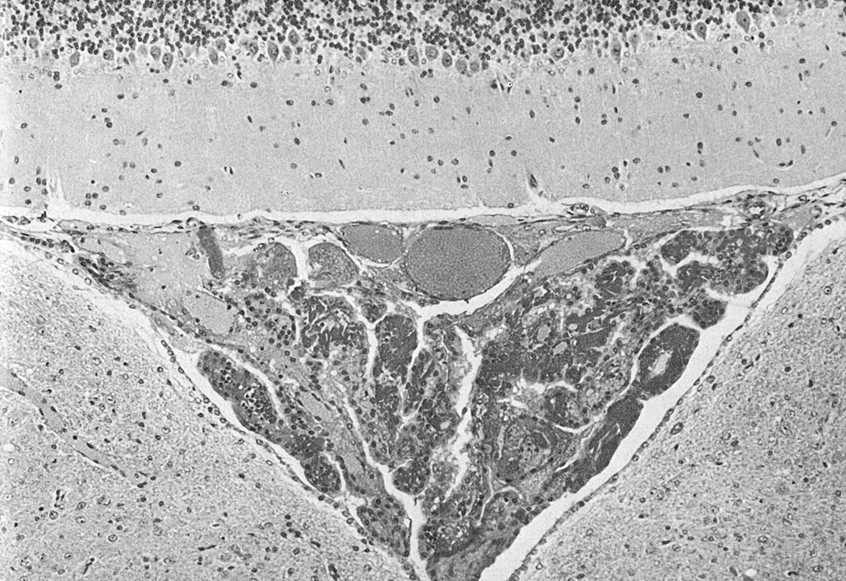
Adverse effects of cylcophosphamide (CY) on the fourth ventricle choroid plexus (CP). Two days after CY administration to adult rats, there is severe choroid plexus inflammation (“plexitis”). The CP (shaped like an upside-down triangle) fills the entire fourth ventricle and is disrupted by hemorrhage, necrosis, and exudates of plasma/fibrin in the stroma. Neuronal/glial cells in the cerebellum (top) and medulla (bottom) appear normal. Experimental conditions are summarized in Table 4 and described by Levine, Sowinski, and Nochlin (1982). Stain: hematoxylin and eosin, ×170. Due to unavoidable circumstances the above figures were not originals but were scanned from reprints.
Interstitial impairment
The interstitium of CP is accessible to cells of the peripheral immune system, including macrophages. Methylcellulose, a vehicle for suspending drugs given orally, has extensive uses in toxicology assessments. Methylcellulose dissolved in water and injected intraperitoneally (IP) into rats for 30 days (see Table 4) substantially modified the interstitium between blood vessels and epithelium of CP (Roth and Krinke 1994). Following methylcellulose treatment, numerous foam-like cells appeared within the interstitium (Figure 6 ). The foamy cells stained with RCA-1, reflecting a scavenger characteristic. These large cells in the stroma contained central nuclei surrounded by finely vacuolated, “foamy” cytoplasm. This phenomenon in CP (but not in small intestine or lungs) may be the result of uptake of methycellulose by peritoneal macrophages that then migrate to CP and infiltrate the stroma (Roth and Krinke 1994).
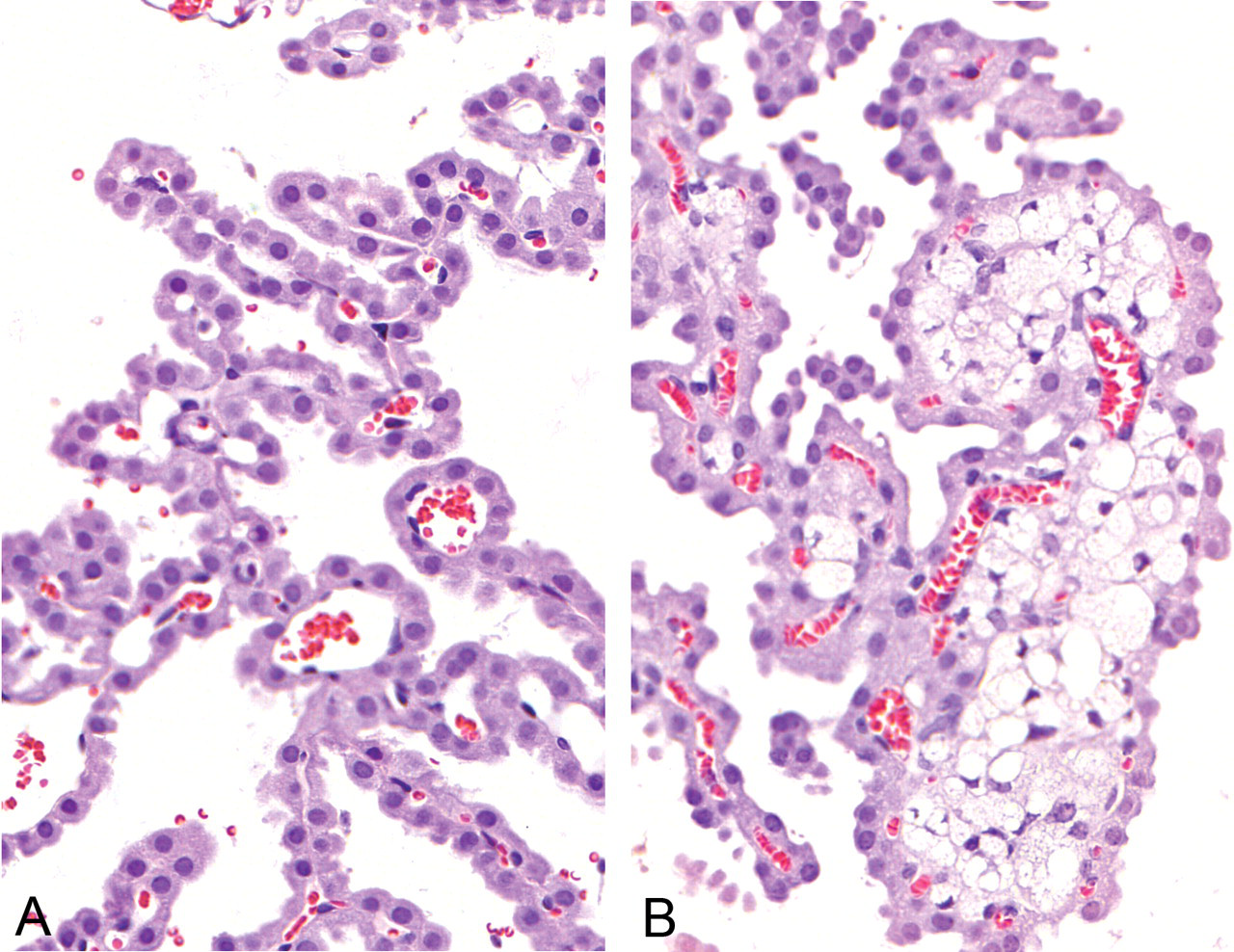
Foam cells in the stroma of the choroid plexus (CP) of rats injected with methylcellulose (0.5% Methocel, 5 ml/kg IP daily for 30 days). The “foamy” cells (enlarged, light-colored cells in the right panel) may be macrophages that ingested methylcellulose in the peritoneum and then migrated to the CP, where they penetrated the interstitial space and caused swelling of tissue. Control CP (left panel), devoid of foam cells, displays a normally appearing epithelial cell lining and vascular elements in each villus core. Experimental data are summarized in Table 4 and described by Roth and Krinke (1994). Hematoxylin and eosin, ×400.
Swelling of CP results from interstitial space distension (Figure 6). Fluid engorgement prompts consideration of two unfavorable outcomes: edema-associated restriction to diffusion of solutes between blood and CSF and a possible “mass effect” of enlarged tissue in the ventricles that occludes CSF flow. Due to macrophage distortion (enlargement) of CP interstitium, a caveat is offered about using methylcellulose vehicle to study immune cell trafficking into the CP interstitium (Chinnery, Ruitenberg, and McMenamin 2010; Petito and Adkins 2005) and through BCSFB (e.g., MS and EAE models). Toxicology studies of material exchange between CP and CSF should address the integrity of the stromal-epithelial relationship.
Epithelial impairment
Tertiary amines cause primary damage to CP epithelial cells. A relatively selective lesion of the epithelium is caused by piperamide, a trypanocidal agent that produces marked CP vacuolation in rodents (Benitz and Kramer 1968). “Hydropic degeneration” describes the extensive epithelial vacuolation after treatment with triamine or tertiary agents: piperidine, piperazine, disobutamide, and pyridine (Table 4). Tilorone, a tertiary-amine potent activator of hypoxia-inducible factor-1, at repeated high doses causes degenerative vacuolation in CP and kidney (Levine and Sowinski 1977). Membrane-bound vacuoles that appear in CP do not always form in ependyma. Enhanced vacuole formation in CP of lateral, third, and fourth ventricles relates to lysosomal degradation of invading xenobiotic (tertiary amine).
Lysosomes are small, spherical organelles containing thin, parallel, concentric lamellae. They break up cellular debris or foreign agents for excretion. As a result, vacuoles are generated. Characteristics of tertiary amine-induced vacuoles (Levine 1977; Wenk, Levine, and Hoenig 1979) are summarized in Table 5 . Extensive tertiary-amine elicited vacuoles compress epithelium and alter CP tissue topography (Figure 7). Severe vacuolar swelling of CP epithelium may fill ventricles with enlarged CPs, thereby creating obstructive hydrocephalus (Benitz and Kramer 1968).
Characteristics of vacuole formation in mammalian choroid plexus (CP) epithelium.

BCSFB, blood-CSF barrier; ICV, intracerebroventicular; PO, oral; SC, subcutaneous.
Elucidating vacuole phenomena enhances understanding of BCSFB toxicokinetics (i.e., the rate and extent of toxicant transportation into and out of the CSF) and toxicogenomics (e.g., phospholipidosis induction in lysosomes by cationic amphiphilic drugs [CAD]). Intense vacuole formation usually reflects excessive accumulation of xenobiotic material that was not properly digested within lysosomes, thereby leading to an iatrogenic lysosomal storage disease. Phospholipidosis in lysosomal disorders can be iatrogenic (drug side effects) or genetic (mutated enzymes or membrane-bound proteins). Lamellated inclusions characteristic of “phospholipidosis” are also related to epithelial vacuolation. Various CADs induce generalized lipidosis of rat CP epithelium, but to different degrees (Frisch and Lullmann-Rauch 1979); such CAD agents have a hydrophobic component and a hydrophilic side chain. For instance, chloroquine produces large cytoplasmic vacuoles. Vacuole ultrastructure after chloroquine treatment suggested storage of water-soluble materials in addition to non-water-soluble polar lipids. Quinacrine, 4,4’-diethylaminoethoxyhexestrol, chlorphentermine, iprindole, 1-chloro-amitriptyline, and clomipramine caused formation of lamellated or crystalloid inclusions as observed in drug-induced lipidosis.
Drug-lipid complexes in lysosomes, especially those affecting the CP, deserve more pharmacologic attention. Pegylated (polyethylene glycol [PEG]-coupled) molecules help to reduce immunogenicity and prolong bioavailabilty of biopharmaceutical drugs. However, they are typically removed from the system by phagocytic cells (e.g., macrophages) or secretory cells (e.g., CP epithelium in the brain, proximal tubule epithelium in the kidney), a process that leads over time to vacuole formation. This finding may have major implications for developing pharmaceutical agents. For instance, certolizumab (CIMZIA®) was not approved by the European Medicines Agency for treatment of Crohn’s disease based on its look of efficacy; however, this agent is used to treat other disorders despite producing vacuolation of CP in safety studies. Two key questions arise in such situations (Wagner et al. 2008): “How does CP epithelium metabolize pegylated compounds?” and “Are there sufficient BCSFB disruptions by PEG-induced phospholipidosis to alter CSF composition and dynamics?”
A relationship exists between drug concentrations in CP, the extent of induced vacuole formation, and drug penetration into the CSF. Testing of a new candidate anti-diabetic drug in rats daily for 4 or 16 weeks revealed extensive vacuolation and swelling of the CP (Figure 8 ), but not ependyma (which also differed from CP in the expression and distribution of various cellular elements) (Table 6 ). The candidate agent may have been more accessible to CP (plasma side of tissue) relative to the CSF, by which the ependyma is exposed. Nevertheless, cumulative evidence demonstrates that several types of drugs augment epithelial vacuolation and thereby distort CP structure and function. Future studies of lysosomal storage diseases, for example, Niemann-Pick disease, should include CP-CSF analyses (Elleder, Jirasek, and Smid 1975). This enables evaluating possible phospholipidosis-inducing effects on the BCSFB, material transport into CSF-brain, and ventricular fluid dynamics.
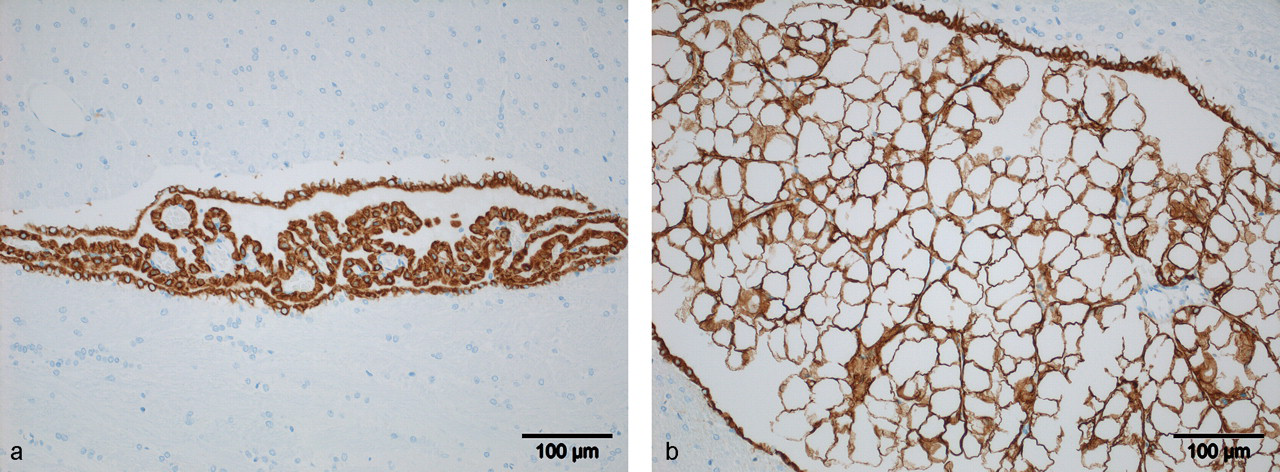
Vacuole formation in the choroid plexus (CP) epithelium after treatment with a novel anti-diabetic drug. 8a shows the CP (labeled by immunohistochemistry to demonstrate pan-cytokeratin) in a control lateral ventricle from a ~150-g female rat given vehicle. 8b portrays a swollen, hydropic CP, replete with large intra-epithelial vacuoles, in a rat receiving a high dose (1000 mg/kg/day) of an anti-diabetic compound for 16 weeks. Lesions seen in all 10 high-dose rats were as severe as those seen in the right panel. The vacuoles likely contain complexes of drug and lipid. Ependymal cells on the edge of ventricle also stained for pan-cytokeratin, while blood vessels did not stain. Magnification is 20× for both images. Pictures provided by J. Funk (unpublished data).
Immunostaining of choroid plexus (CP) epithelial and ependymal cells after treatment of rats with a candidate anti-diabetic agent.a
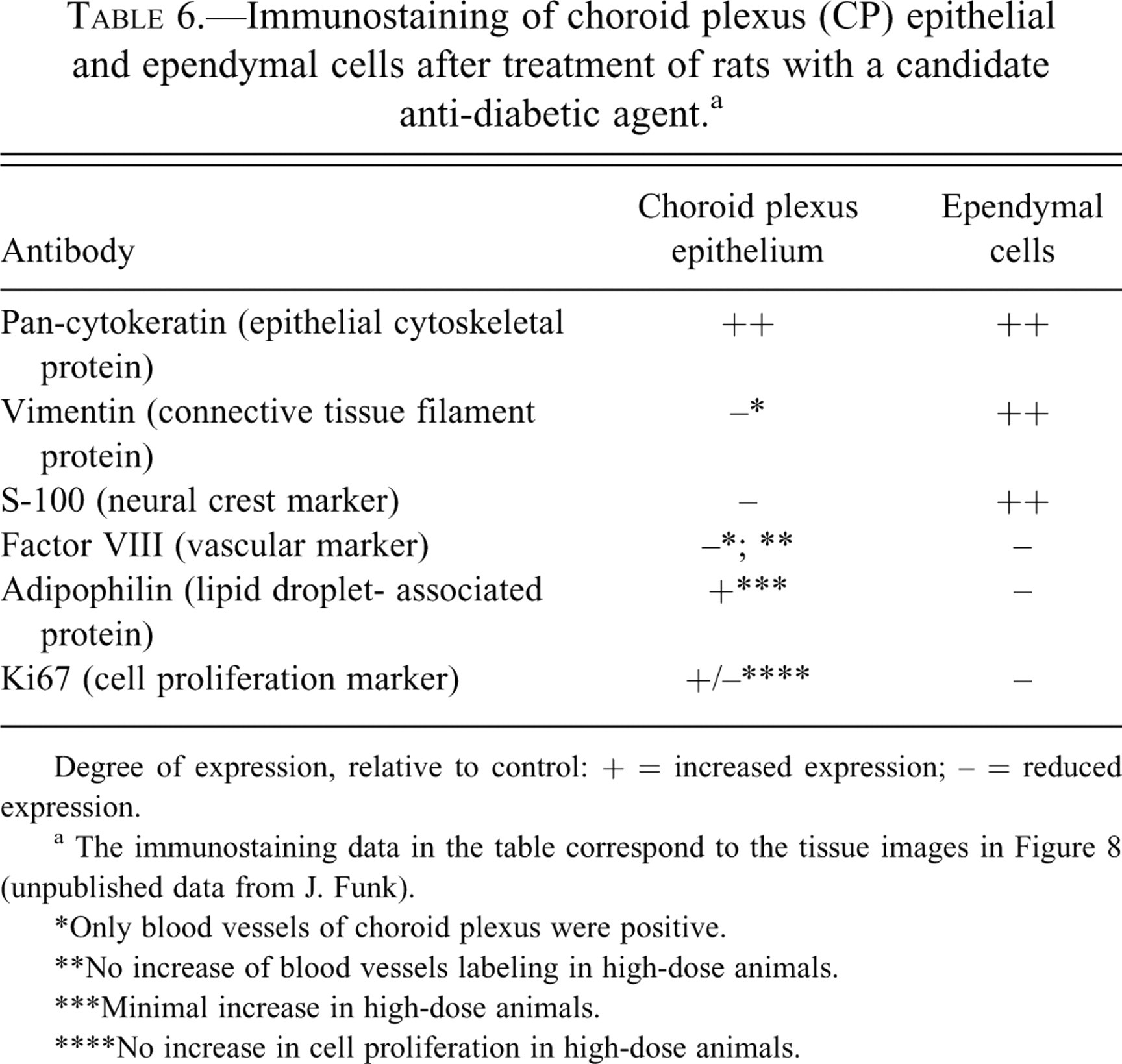
Degree of expression, relative to control: + = increased expression; – = reduced expression.
a The immunostaining data in the table correspond to the tissue images in Figure 8 (unpublished data from J. Funk).
*Only blood vessels of choroid plexus were positive.
**No increase of blood vessels labeling in high-dose animals.
***Minimal increase in high-dose animals.
****No increase in cell proliferation in high-dose animals.
To date, many CP vacuole assessments have dealt with tertiary amine effects. However, other major classes of compounds/disorders predispose toward vacuole formation at the BCSFB and in various epithelia. Amphiphilic CAD agents—which are well-known inducers of vacuoles, phospholipidosis, and lysosomal storage disorders—include antidepressants, antiarrythmics, antimalarials, and cholesterol-lowering agents. Toxicogenomic studies of CAD-induced vacuole formation and lysosomal expression (e.g., in HepG2 liver-derived cells) used Affymetrix Microarray Analysis® to reveal several mechanisms that putatively result in phospholipidosis: inhibition of lysosomal phospholipase, inhibition of lysosomal enzyme transport, enhanced phospholipid biosynthesis, and augmented cholesterol production (Sawada, Takami, and Asahi 2005).
Important practical goals are to identify specific gene markers for faulty lipid storage and to detect drugs with phospholipidosis-inducing potential by in vitro screening (Kasahara et al. 2006). In addition to assessing physicochemical attributes of compounds (alkalinity and lipophilicity) that induce phospholipidosis, new models are capable of factoring the physiologic volume of distribution (Hanumegowda et al. 2010). Collectively these new approaches will likely accelerate toxicologic assessments and facilitate the discovery of new agents to treat lysosomal storage problems encountered with brain and CP.
Modeling of vacuole formation, in the context of unwanted drug side effects, should account for pharmacokinetic parameters of the induction agent. This can be exemplified with data for disobutamide distribution in the CP and CSF of different species. Disobutamide, a bis tertiary amine that was originally investigated for its anti-arrhythmic properties, contains 1-piperidinebutanamide; it is a highly alkaline (basic) molecule (pKa1 = 8.6; pKa2 = 10.2) and thus is strongly partitioned into lysosomes due to their acidic pH (4.5). Within lysosomes, this drug reliably induces clear vacuoles, so it is a great aid in investigating intracellular drug storage (Ruben et al. 1989). Interestingly, disobutamide in vivo causes vacuolation in CP of rats and monkeys, but not in dogs (Table 5). However, this CAD is a weak penetrator of canine CP, attaining an in vivo concentration only 0.01% of that attained in rat CP (Koizumi et al. 1986). Still, this drug accesses the dog CP epithelium sufficiently to induce vacuoles, either in vitro (isolated CP in artificial CSF) or when given by ICV injection to living animals (Table 5). Thus, when comparing vacuole induction among species, it is critical to set the experimental conditions to assure that test agents adequately permeate the epithelium. Therefore, a combination of in vitro and in vivo preparations in three species was required to demonstrate the relationship among disobutamide uptake by CP, induction of vacuoles in the choroidal epithelium, and in vivo penetration into CSF across BCSFB (Koizumi et al. 1986). Experimentation reveals the need to explore species differences in the etiology, incidence, and effect of vacuole induction development of CADs or other lysosomal-altering agents (Halliwell 1997).
Disease-induced phospholipidosis also occurs in the CP. Typically in Reyes disease, there is vacuolation of CP epithelium and possibly associated brain edema (R. E. Brown and Madge 1972). Are these two hydropic alterations serially linked by CSF nexus phenomena? Hyperammonemia plus aspirin or viral infection causes vacuolation in the CP of ferrets, a common animal model of Reyes disease (Rarey, Davis, and Deshmukh 1987). Tertiary amines, which also induce vacuoles, are derived from ammonia. All in all, the vacuolation phenomena in CP suggest the increased passage of pathogenic molecules into CSF, and then from ventricles into the brain. Strengthening of this postulate comes from similar observations in several other models of BCSFB breakdown: protein penetration into CSF, ventriculomegaly, ependymal damage, and periventricular edema (H. S. Sharma, Duncan, and Johanson 2006; Murphy and Johanson 1985).
Injuries to Ependyma
Even though CP epithelium and the ependyma have a common embryologic origin, there are important differences in function, structure, and protein expression between these two cell types. This affects how each cell type responds to a given drug. The data for amoscanate, an anti-schistosomal agent (Table 4), exemplify differences in toxicologic effects on CSF-bordering cells. In several mammalian species, a single curative oral dose is typically 10 to 60 mg/kg (Bueding, Batzinger, and Petterson 1976). Adsorbable amoscanate given to an adult induced a lesion in the ependyma but not in CP by three days after administration of consecutive high oral doses (125 or 500 mg/kg) (Clark, Kiel, and Parhad 1982; Krinke et al. 1983). Amoscanate caused necrosis by 28 days to the lateral walls of both telencephalic ventricles; interestingly, the lesion (Figure 11 ) was confined to the caudate/putamen surface and generally spared the brain, spinal cord, and peripheral nerves. Significantly, minute lesions occasionally occurred close to CSF (i.e., in the fornix or septum), thereby providing a clue about the pharmacokinetics of amoscanate distribution. Taken together, the neuropathology pointed to CSF (rather than plasma in periventricular capillaries) as the direct source of drug that damages regions proximate to the ventricles. Amoscanate likely penetrated the BCSFB and then flowed within the CSF, inflicting damage to sensitive cells at the ventricular border (arrows 2–5, Figure 1). Increasing the number of consecutive doses resulted in erosion of the ventricular lining, glial reaction, and granule deposits (Krinke et al. 1983). Amoscanate also elicited massive ultrastructural damage in ependymal cells (Figure 12 ).
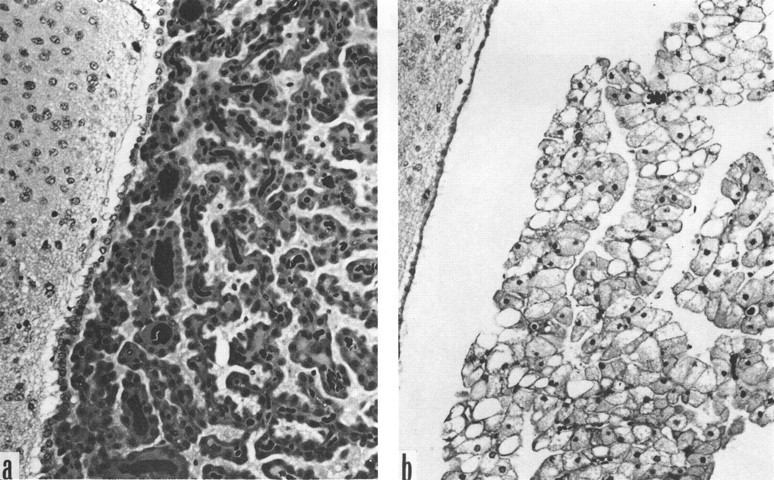
Disobutamide induction of vacuoles in rat choroid plexus (CP) epithelium. The CP villi 5-week-old female rats given vehicle (a) or disobutamide (250 mg/kg orally for 13 weeks; b). Epithelial cells in the CP, but not the ependyma, are swollen with large, clear cytoplasmic vacuoles following disobutamide treatment (see Table 4 and Koizumi et al. 1986). Ependyma and cerebral parenchyma, on left side of each image, appear normal. Magnification is 50× for both images. Due to unavoidable circumstances the above figures were not originals but were scanned from reprints.
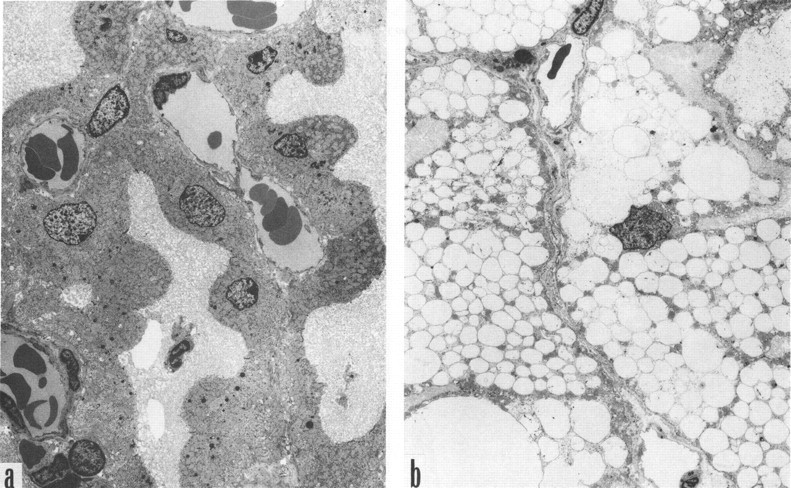
Ultrastructure of choroid plexus (CP) in rats treated with disobutamide. The CP villi in 5-week-old male rats following treatment with disobutamide (250 mg/kg orally for 13 weeks). Untreated animals (left panel) have healthy lysosomes and lush apical membrane microvilli. Disobutamide-induced vacuolation (right panel) in the epithelium is extensive, displacing nuclei and organelles; in addition, apical microvilli are not as evident after drug treatment. Magnification is 1,000× for both images. Images are from Koizumi et al. (1986). Due to unavoidable circumstances the above figures were not originals but were scanned from reprints.
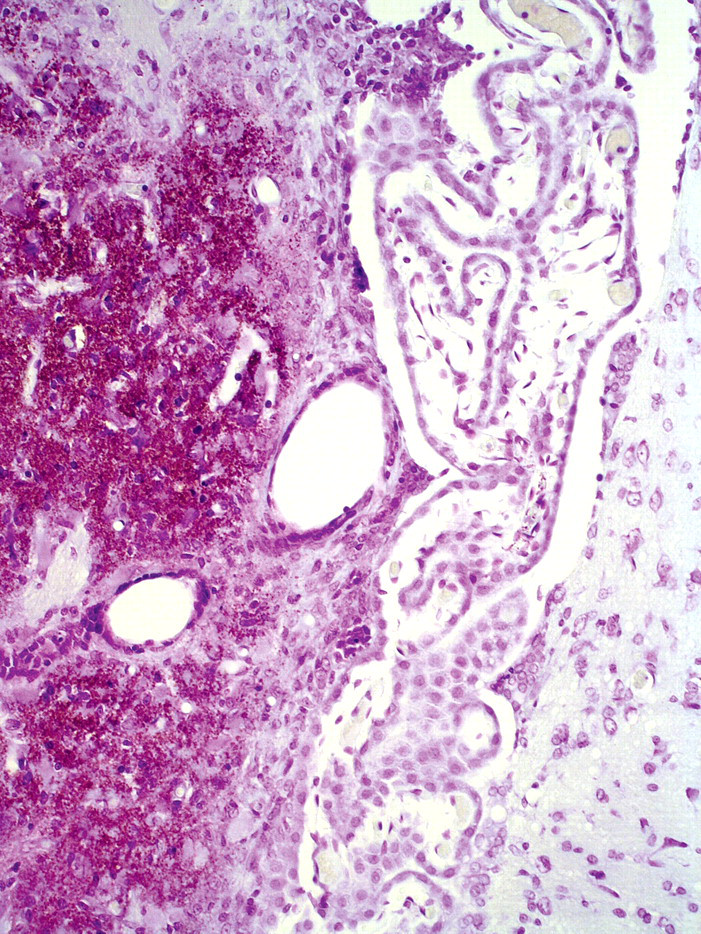
Destruction of ependyma and periventricular brain by amoscanate. Sprague-Dawley rats treated with amoscanate (500 mg/kg/day suspended in oil, given orally for 10 days). In the medial caudate/putamen, there was deposition of microgranules. Choroid plexus was intact, but the ependyma covering the caudate/putamen was severely damaged. Magnification is 20×. From Krinke et al. (1983). Cresyl violet.
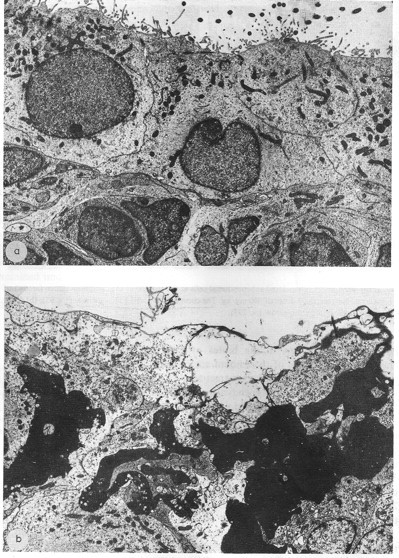
Ultrastructural view of ependyma damaged by amoscanate.
Another study of high-dose amoscanate in rats (250 or 500 mg/kg for 28 days) yielded similar findings of lateral ventricle wall damage, particularly the neuropil of medial striatum (Clark, Kiel, and Parhad 1982). All intoxicated animals, but no controls, developed discrete necrotic lesions adjacent to the lateral wall of lateral ventricle, commonly extending 1 mm into the striatum. Immediately after initial oral dosing, rats displayed less exploratory activity. Ependymal cells overlying necrotic brain lesions often survived but were pyknotic. Extracellular spaces in ependymal/subependymal regions were edematous and spongiform (Clark, Kiel, and Parhad 1982). Less damage was inflicted on the medial wall of the lateral ventricle than on the lateral counterpart. Chronic treatment with high doses of amoscanate does not injure other parts of the cerebral hemispheres, cerebellum, brainstem, or CP in the third and fourth ventricles (Clark, Kiel, and Parhad 1982; Krinke et al. 1983). Similarly, walls of third and fourth ventricles remained intact.
Highly specific adverse responses of the ependyma (certain regions) to amoscanate have been reproducible. Interesting issues arise about the differential toxicity of this drug along points in the distributional nexus: CP to CSF, ependyma, SVZ, and ultimately the brain (Figure 1). Amoscanate, even in a high-dose regimen, evidently does not harm CP (as examined by microscopy). A CP capable of normal transport function actively concentrates certain endogenous substances and drugs in CSF relative to plasma (J. H. Wang and Takemori 1972; Spector and Johanson 2006). One possibility is that certain membrane transporters of organic solutes by CP (Table 2) actively pump amoscanate, and other drugs, into CSF (arrow 3, Figure 1). This notion needs evidence from CSF concentration data for various therapeutic agents. Exposure of ependyma to amoscanate in the CSF likely explains the sequential toxicity involving ependyma, subependyma, and (at high doses and after long exposure times) eventually the underlying brain. Pharmacokinetically, the ependyma overlying caudate/striatal regions may have been exposed to higher CSF concentrations of amoscanate than ependyma in other regions. An alternative but not mutually exclusive explanation for ependymal toxicity is the specific responsivity (phenotype) of individual cells to a particular drug. Ependymal heterogeneity, discussed previously, means that some cells may be more vulnerable than others to xenobiotic agents. Ependyma adjacent to the caudate-putamen is particularly sensitive to some foreign agents in CSF. Further research will be necessary to ascertain whether specific ventricular regions are less able to protect caudate/putamen and striatum against invasion by cancer cells, immune cells, and pathogens circulating in the CSF.
Injuries to Circumventricular Organs
Systematic toxicologic/pathologic analyses of CVOs are rare. A recent review, however, emphasizes pathogen entry into the CNS through CVO portals (Siso, Jeffrey, and Gonzalez 2010). Certain trypanosomes, for example, invade brain by way of CVO “open windows” (permeable capillaries). Toxicants also permeate via the CVOs. A compelling example of specific CVO disruption is by the excitotoxicant domoic acid. As follow-up to an episode of mussel poisoning in Canada, experiments in mice tested the effects of high-dose domoic acid (7 mg/kg). Domoate-induced neuropathology occurred within 30 minutes and was confined to the CNS, mainly to three CVOs: SFO, OVLT, and AP (Bruni et al. 1991). Particularly intriguing about domoic acid overload was hypodipsia or decreased drinking, likely due to damage of the SFO that normally mediates the thirst response.
Medicinals also toxically stress the CVOs. A prominent example is adriamycin (doxorubicin), a cytotoxic antibiotic used to treat malignant tumors. This agent extensively penetrates the CVOs and damages circumventricular neurons (to cause axon terminal degeneration), glia (disrupted nucleus and nucleolus), and extracellular space (edema). Doses in mice comparable to those used in chemotherapy in human patients injured the NLP, ME, and AP. Adverse effects were evident after 30 days (Bigotte and Olsson 1983). Morphologic alterations in all three CVOs were pronounced.
Enhanced appreciation of the loci, characteristics, and functions of CVOs prompts consideration of the SFO, SCO, OVLT, and AP as potential pathologic regions of interest. A primary toxicologic consideration about permeable CVOs is that toxicants do not have to overcome the BBB to access and damage the organ. Rather, the leaky capillaries in CVOs enable toxic compounds to reach the inner epithelial parenchyma and neuronal endings. Ironically, the parenchyma of CVOs (unlike other brain regions protected by BBB) may be more vulnerable to certain drugs/metabolites in plasma than in CSF.
Can drugs, toxicants, and cells in plasma access the CSF by moving through CVO structures? Probably, but likely only to a limited extent. Even though CVO capillaries lack tight junctions between the endothelial cells, the epithelial cells in these mini-organs have occluding tight junctions. This constitutes a diffusion barrier between the CVO and CSF. Accordingly, the CVO perivascular space is segregated by tight junctions from the CSF-milieu of adjacent neuropil (Krisch, Leonhardt, and Buchheim 1978). Still, there is some macromolecular and cellular trafficking into, and likely through, the CVOs into nearby brain and CSF. In EAE models, the CD45+ leukocytes penetrated SFO, ME, OVLT, and AP (Schulz and Engelhardt 2005). Enriched EAE inflammatory cell infiltrates were found outside but near each CVO, intimating immune cell penetration into CNS/CSF by transit across these four CVOs.
Although considerable evidence indicates that CVOs are extensively modulated and affected by plasma-borne solutes and cells, nonetheless there is minimal insight regarding how CSF-borne hormones, pathogens, toxicants, and cells interact with CVOs from the apical-side of each mini-organ. Tanycyte fibers contacting CSF pick up ventricular signaling molecules and convey these neuroendocrine hormones into hypothalamic portal capillaries (Rodriguez et al. 2005). Putative CVO involvement with tanycyte and hypothalamic homeostatic actions, especially as modified by CSF-convected toxicants, deserves more investigation.
Perspectives on Cerebrospinal Fluid Toxicology and Pathology
Breakdown of CP by xenobiotic agents and pathogens compromises CSF homeostasis with profound implications for many CNS functions. Regulated secretions by CP represent a lifeline of micronutritional and trophic support for neurons. In addition, the reabsorption of brain catabolites and drug metabolites by choroid epithelial cells pivotally maintains CSF purity. Moreover, fine regulation of immune and neuroendocrine phenomena at the BCSFB bears not only on brain well-being but also that of peripheral organs. Therefore, maintaining integrity of the blood-CSF transport interface is vital for health.
Throughout life, the CP is challenged by blood-borne toxicants, pathogens (viruses and protozoans), and various xenobiotics. Usually the BCSFB remains intact as it successfully wards off threatening agents and protects the CNS. Unfortunately, some stressors, given at high “doses” or for long periods, overwhelm CP homeostatic capacities so that foreign agents can invade CSF. Because the ependyma is readily permeable in many ventricular locations, a CSF-borne toxicant has easy access to periventricular brain regions. Such accession challenges neurogenic niches requiring a stable, clean environment to generate new neurons for supporting cognitive function. Maintaining CP defense systems is therefore of prime significance. A sound BCSFB thwarts penetration of harmful molecules into CSF-brain.
Insight is needed on mechanisms underlying CP vulnerability to certain viruses (HIV) and parasites (e.g., schistosomes and trypanosomes). Undoubtedly one vulnerability factor is the great permeability of the CP capillary system and high blood flow. This assures extensive delivery of materials to interstitium and the epithelial cell basolateral membrane. Another potential susceptibility factor, quite neglected in research, is the wide variety of epithelial cell phenotypes revealed by differential lectin staining of glycoconjugates (McMillan et al. 2003). Accordingly, one choroid epithelial cell may express particular glycosylated proteins on its surface while neighboring epithelium does not; the implication is that some cells at the BCSFB may bind a particular virus or protein, whereas neighboring cells may not. Since binding is integral to endocytotic and transcytotic phenomena, it is pertinent to obtain more information on CP cell receptor affinities.
Certain cells of the immune system migrate through CP (especially in autoimmune relapse states), perhaps less so across brain capillaries. Here the epithelial cells of the BCSFB differ markedly in structure and function from the endothelial cells of the BBB. These unique barrier phenotypes, including the expression of proteins (structural and enzymatic), likely explain most differences in translocational phenomena. Diseases such as MS and pathogen infections worsen as proteins and particles migrate from CP-CSF into periventricular regions. This likely contributes to lesion spread into the brain. Elucidating CSF-brain immunopathology relies on new approaches for tracking aberrant immune cell and immunoglobulin permeation (arrows 1–3, Figure 1) at the BCSFB.
Agents for treating parasitic diseases, such as anti-trypano-somic drugs, can be harsh on CP structure and physiology. New antiparasitic agents are being sought that are less likely to perturb the BCSFB. Vacuole formation in CP, provoked by various types of drugs, is vexing due to potential deleterious effects on CSF dynamics as well as CP epithelial phospholipid metabolism. Preservation of lysosomal/Golgi functions in CP is central to efficient scavenging/reabsorptive actions to keep CSF clean. Procuring information about lysosomal system integrity at the BCSFB will facilitate development of new strategies and treatments for pharmacotherapy and detoxification of central disorders. This includes minimizing CSF impurities in advanced aging and early neurodegeneration (Johanson et al. 2004).
The brain, a highly protected and specialized organ, is exquisitely sensitive to changes in CSF composition. There is a continuing need for systematic research to delineate and boost protective mechanisms in CP. Therapeutic strategies should be developed to stabilize the blood-CSF and CSF-brain interfaces upon, or before, exposure to toxic and pathogenic agents.
