Abstract
The objective of this study was to investigate acute lung toxicity caused by Asian sand dust. Simulated Asian sand dust collected from the Tennger desert in China (CJ-2 particles) and Asian sand dust collected from the atmosphere in Japan (Tottori particles) were used. Saline suspensions of 50, 200, 800, and 3,000 µg Asian sand dust were intratracheally instilled to ICR mice. Localized accumulation of the dust particles was observed in the bronchioles and the alveoli of the lung tissues; acute inflammatory changes characterized by infiltration of macrophages and neutrophils were observed around the particles. Degenerated alveolar walls and bronchial epithelial cells, as well as a weakened positive immunolabeling for laminin, were observed to be associated with particle attachment. Positive immunolabelings for interleukin-6, tumor necrosis factor–α inducible nitric oxide synthase, and dimeric copper- and zinc-containing superoxide dismutase were observed mainly in the inflammatory cells in the lesions; these findings were not observed in the controls or in areas lacking lesions. These results suggest that Asian sand dust particles caused damage to the lung tissue through a direct physical effect. In addition, secondary released cytokines and oxidative stress generated in the lesion may be involved in the development of the acute lung toxicity.
Introduction
Asian sand dust aerosol (known as kosa aerosol) originates from the arid and semiarid areas of China and is transported from East Asia to countries in the Pacific region, including South Korea (Chung and Yoon 1996), Japan (Mori et al. 1999), and the United States (Uematsu et al. 1983; Tratt et al. 2001). Since 2000, the frequency and scale of dust events giving rise to Asian sand dust aerosols have increased rapidly in East Asia (Mori et al. 2003). This seems to be because of environmental changes, such as desertification and climate changes in central and northwestern China (Ichinose et al. 2005). Epidemiological studies in humans have shown that ambient Asian sand dust particles are associated with an increase in pulmonary and cardiovascular morbidity and mortality in Korea (Kwon et al. 2002; Lee et al. 2007) and Taiwan (Chen et al. 2004).
Chemical analyses of Asian sand dust in China and Japan have been carried out by many researchers. In industrialized areas of eastern China, sulfur oxides and nitrogen oxides are produced, and these gases are adsorbed onto mineral particles in Asian sand dust (Mori et al. 2003). Fungal spores and walls (β-glucan) and the lipopolysaccharides of gram-negative bacteria from the atmosphere are also adsorbed onto particles of Asian sand dust (Ichinose et al. 2005), resulting in an increase in concern about possible adverse respiratory effects in eastern Asia. The degree of respiratory inflammation caused by Asian sand dust has been shown to be dependent on the amount of adsorbed toxic material, including air pollutants (Ichinose et al. 2005).
Previous studies (Nishikawa et al. 2000) have determined that the major mineralogical component of Asian sand dust is silica (SiO2), which is derived mainly from feldspar and quartz. Excessive exposure to quartz (crystalline silica) has been linked to pulmonary diseases, such as silicosis, tuberculosis, chronic bronchitis, chronic obstructive pulmonary disease, and lung cancer (Rimal et al. 2005). There have been many studies on the pathogenesis of pulmonary toxicity induced by crystalline silica, several of which (Castranova et al. 2002; Rao et al. 2004; Rimal et al. 2005) have evaluated the role of cytokines and oxidative stress markers, including tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), dimeric copper- and zinc-containing superoxide dismutase (Cu/Zn SOD), and inducible nitric oxide synthase (iNOS).
Although the pulmonary toxicity of crystalline silica is well known, there is relatively little information about the toxicity of Asian sand dust. Data regarding the toxicity of Asian sand dust in experimental animals are limited, and the precise mechanisms of inflammation caused by Asian sand dust are not clearly understood. Asian sand dust itself, free from ambient pollutants, may exhibit cytotoxicity to the alveolar epithelial cells in vitro (Kim et al. 2003). However, to the best of our knowledge, there have been no in vivo pathological studies focusing on lung toxicity induced by Asian sand dust particles free from chemical and biological substances. The investigation of lung inflammation after intratracheal instillation of Asian sand dust could help in clarifying the effects of atmospheric Asian sand dust on the respiratory system.
The purpose of this study was to describe the acute lung toxicity and pulmonary inflammation mechanism caused by the intratracheal instillation of Asian sand dust in mice.
Materials and Methods
Animals
A total of 63 male ICR mice (5 or 6 weeks old) were obtained from CLEA Japan Inc. (Tokyo, Japan). Animals were fed a CE-2 diet purchased from CLEA Japan, and water was given ad libitum. The mouse cages were placed in a conventional room, where the temperature was maintained at about 25°C and humidity at 55% to 70%. All animal experiments were performed according to the Tottori University guidelines for animal welfare (http://www.tottori-u.ac.jp/kouhou/kisokusyuu/reiki_honbun/u0950581001.html).
Preparation of Particle Samples
Asian sand dust particles from two sources were used in this study: CJ-2 particles and Tottori particles. The CJ-2 particles were simulated Asian mineral dust obtained from General Science Corporation (Tokyo, Japan). These particles were collected from the surface soil in the southwestern part of the Tengger desert in north-central China, where dust storms occur frequently (Nishikawa et al. 2000). The mean particle diameter was approximately 0.03 mm ± 0.01 mm, according to the manufacturer’s data sheet. The Tottori particles were Asian sand dust particles collected from the atmosphere in Tottori prefecture, Japan, by a deposit gauge during dust storm events in March 2008.
Particles from both sources were hot-air sterilized at 300°C for 1 hour to remove toxic substances (microbiological substances as well as chemicals including nitrogen oxide and sulfur oxide) adhering to the Asian sand dust. The sterilization temperature was determined in accordance with previous studies (Ichinose, Yoshida, Sadakane, et al. 2008; Ichinosse, Yoshida, Hiyoshi, et al. 2008; Yanagisawa et al. 2007). It has been reported that neither the chemical composition nor the shape of mineral particles changed, even when heated to 700°C (Iwatsuki et al. 1994, 1998).
Elemental Analysis of Particle Samples
The elemental composition of the CJ-2 particles was described in the manufacturer’s data sheet. The mineralogical content of the Tottori particles was determined by inductively coupled plasma atomic emission spectroscopy (ICPS-1000 IV, Shimazu, Kyoto, Japan). Inorganic ions of the elements in the Tottori particles were analyzed by ion chromatography (DX-320, Dionex, CA).
Preparation of Suspensions
The sterilized samples of the two different particles were each suspended in 0.05 mL of sterilized saline solution for instillation. The suspensions were deflocculated via ultrasonic disintegration for 3 minutes. Four doses (50, 200, 800, and 3,000 µg) of the particle samples (CJ-2 particles and Tottori particles) were chosen to determine dose effects on lung toxicity in mice. The maximum deposition of particles in the lungs of a single mouse was calculated using tidal volume and breathing rate (Ichinose, Yoshida, Sadakane, et al. 2008; Ichinosse, Yoshida, Hiyoshi, et al. 2008). The maximum deposition per week in the Japanese national air quality standard for suspended particulate matter (0.1 mg/m3) was approximately 30 µg. The instillation doses (50, 200, 800, and 3,000 µg) in the present study represent 1.7, 6.6, 26.4, and 99 times that amount, respectively. Control mice received 0.05 mL of a saline solution.
Morphology of the Asian Sand Dust Particles in the Suspensions
Morphological examination of the Asian sand dust particles in the suspensions was performed with a scanning electron microscope (Model X-650, Hitachi, Tokyo, Japan).
Study Protocol
The ICR mice were divided into nine groups according to treatment: saline only (n = 9), 50 µg of CJ-2 particles (n = 11), 200 µg of CJ-2 particles (n = 11), 800 µg of CJ-2 particles (n = 8), 3,000 µg of CJ-2 particles (n = 8), 50 µg of Tottori particles (n = 4), 200 µg of Tottori particles (n = 4), 800 µg of Tottori particles (n = 4), and 3,000 µg of Tottori particles (n = 4).
The mice were anesthetized by an intraperitoneal injection of sodium pentobarbital (5 mg/100 g body weight). The suspensions were agitated immediately before intratracheal instillation. Of the suspension, 0.05 mL was instilled with an intratracheal cannula to each mouse. Each intratracheal instillation procedure took 3 seconds.
Twenty-four hours after a single exposure of sand dust particles, the mice were killed by exsanguination under deep anesthesia induced by intraperitoneal injection of sodium pentobarbital.
Pathological Examination
Four mice from each group were used for pathological examination and immunohistochemistry. The lung lobes were separated, and longitudinal sections from each lobe were made. These longitudinal sections were placed into embedding cassettes and fixed by immersion in 10% neutral-buffered formalin. In the lungs of the mice treated with saline alone and with 800 µg of CJ-2 particles, the other half of the longitudinal sections was used for electron microscopy. The formalin-fixed lung tissues were routinely processed and embedded in paraffin for histopathological and immunohistochemical examination. Sections approximately 3-µm thick were cut and stained with hematoxylin and eosin. Histopathological changes were graded for the bronchial region (bronchial score) and the alveolar region (alveolar score). The bronchial score was defined according to the degree of bronchiolitis, bronchitis, and degeneration of mucosal membrane (epithelium) in the bronchi and bronchioli, as follows: 0 = no histopathological change; 1 = minimal reaction; 2 = mild reaction, observable at high magnification; 3 = moderate reaction, observable at low magnification; and 4 = severe reaction. The alveolar score was defined according to the degree of alveolitis, intra-alveolar hemorrhage, edema and fibrin exudates in the alveoli, as follows: 0 = no histopathological change; 1 = minimal reaction; 2 = a small, localized lesion; 3 = multiple small, localized lesions involving more than one area; and 4 = as diffuse severe lesions. The pathological examination was performed by two pathologists in a blind manner.
Immunohistochemistry
Paraffin-embedded sections of the lungs of mice treated with saline alone (n = 4) and with 3,000 µg of CJ-2 particles (n = 4) were used for immunohistochemical detection of laminin, TNF-α, IL-6, Cu/Zn SOD, and iNOS. For antigen retrieval, the sections were placed in 0.05% proteinase K for 30 minutes (for detection of laminin) or placed in citrate buffer solution (pH = 5.4; for detection of TNF-α, IL-6, Cu/Zn SOD, and iNOS) and microwaved. Endogeneous peroxidase activity was quenched with 3% H2O2 at room temperature for 30 minutes. The slides were then blocked with 10% normal goat serum (laminin, TNF-α, Cu/Zn SOD and iNOS) or 10% bovine serum albumin (IL-6) for 5 minutes with microwave treatment (laminin, TNF-α, IL-6, and Cu/Zn SOD) or for 30 minutes at room temperature (iNOS). Thereafter, the sections were reacted overnight at 4°C with primary antibodies (anti-laminin; Dako, Glostrup, Denmark, 1:1,000 dilution; anti–TNF-α, Monosan, Uden, the Netherlands, 1:15 dilution; anti–IL-6, Santa Cruz Biotechnology, Santa Cruz, CA, 1:200 dilution; anti-Cu/Zn SOD, Stressgen Bioreagents, Victoria, Canada, 1:200 dilution; and anti-iNOS, Wako Pure Chemical Industries, Osaka, Japan, 1:2,000 dilution) or with an equivalent amount of phosphate-buffered saline as a negative control. Immunoreaction complexes were detected using the avidin-biotin complex method with a labeled streptoavidin biotin kit (Dako). The positive reactions resulted in brown staining with 3, 3′- diaminobenzidine tetrahydrochloride, and the sections were counterstained with hematoxylin.
Electron Microscopy
Half of the longitudinal sections of each lobe of the lungs treated with saline alone (control, n = 4) and with 800 µg of CJ-2 particles (n = 4) were used for electron microscopy. Cubes of 1 to 2 mm3 were made from each section. The small blocks were fixed in glutaraldehyde for 3 hours at 4°C, rinsed in 0.1 M phosphate buffer (pH = 7.4), postfixed for 1 hour in 1% osmium tetroxide, dehydrated in alcohol, and embedded in epoxy resin. Semi-thin (1 µm) sections were stained with 1% toluidine blue to locate interesting areas for electron microscopic examination. Ultrathin sections stained with uranyl acetate and lead citrate were then examined under a TEM-100CX electron microscope (Japan Electron Optical Laboratory, Tokyo, Japan).
Analysis of Bronchoalveolar Lavage Fluid (BALF)
Lungs from several animals in each group of control and CJ-2–treated mice were used for examination of BALF: saline alone (5 of 9 mice), 50 µg CJ-2 (7 of 11 mice), 200 µg CJ-2 (7 of 11 mice), 800 µg CJ-2 (4 of 8 mice) and 3,000 µg CJ-2 (4 of 8 mice). After euthanasia, the trachea was cannulated and the lungs were lavaged with three injections of 1.2 mL of 37°C saline. The lavage fluid was harvested by gentle aspiration. The average volume retrieved was 86% (3.10 mL) of the amount instilled (3.60 mL). The three lavage fluids were combined, preserved in a plastic tube, cooled to 4°C, and then centrifuged at 3,000 rpm for 10 minutes. The supernatants were kept at –80°C until analysis for total protein. Total protein was analyzed with a BCA Protein Assay Kit (Thermo Fisher Scientific, IL) according to the manufacturer’s protocol. The cell pellets were used for measuring the total number of cells, cell viability, and differential cell count. Cell viability was assessed by Trypan blue dye exclusion using a hemocytometer chamber. Differential cell counts were assessed on cytologic preparations. The slides were stained with Diff-Quick (International Regents Corp., Kobe, Japan). A total of 600 cells were counted using light microscopy.
Statistical Analysis
All data were expressed as the mean ± standard error. Statistical significance was determined by a Student’s t-test for two-group comparisons. For all comparisons, p values less than 5% (p < .05) were considered statistically significant.
Results
Morphology of Asian Sand Dust Particles in the Suspensions
As shown in Figure 1, particles from both sources (CJ-2 particles, Tottori particles) were pleomorphic and varied in size. The particle sizes in suspension, of the CJ-2 particles and the Tottori particles, ranged from 1 to 30 µm and 1 to 25 µm, respectively.

Scanning electron microscopy images of CJ-2 particles (A) and Tottori particles (B). Both sources of particles showed pleomorphism and varied sizes. Bar = 2 µm.
Content of the Particles
Table 1 shows the concentration of the elements in the sand dust particles. The primary element was Si, making up 28.0% and 9.9% of the CJ-2 and Tottori particles, respectively. The CJ-2 particles also contained 5.9% Al, 5.3% Ca, 3.0% Fe, 1.7% K, and 1.6% Mg. The Tottori particles contained 1.9% Al, 3.3% Ca, 2.8% Fe, 0.4% K, and 0.5% Mg.
Concentrations of the elements in the sand dust particles.
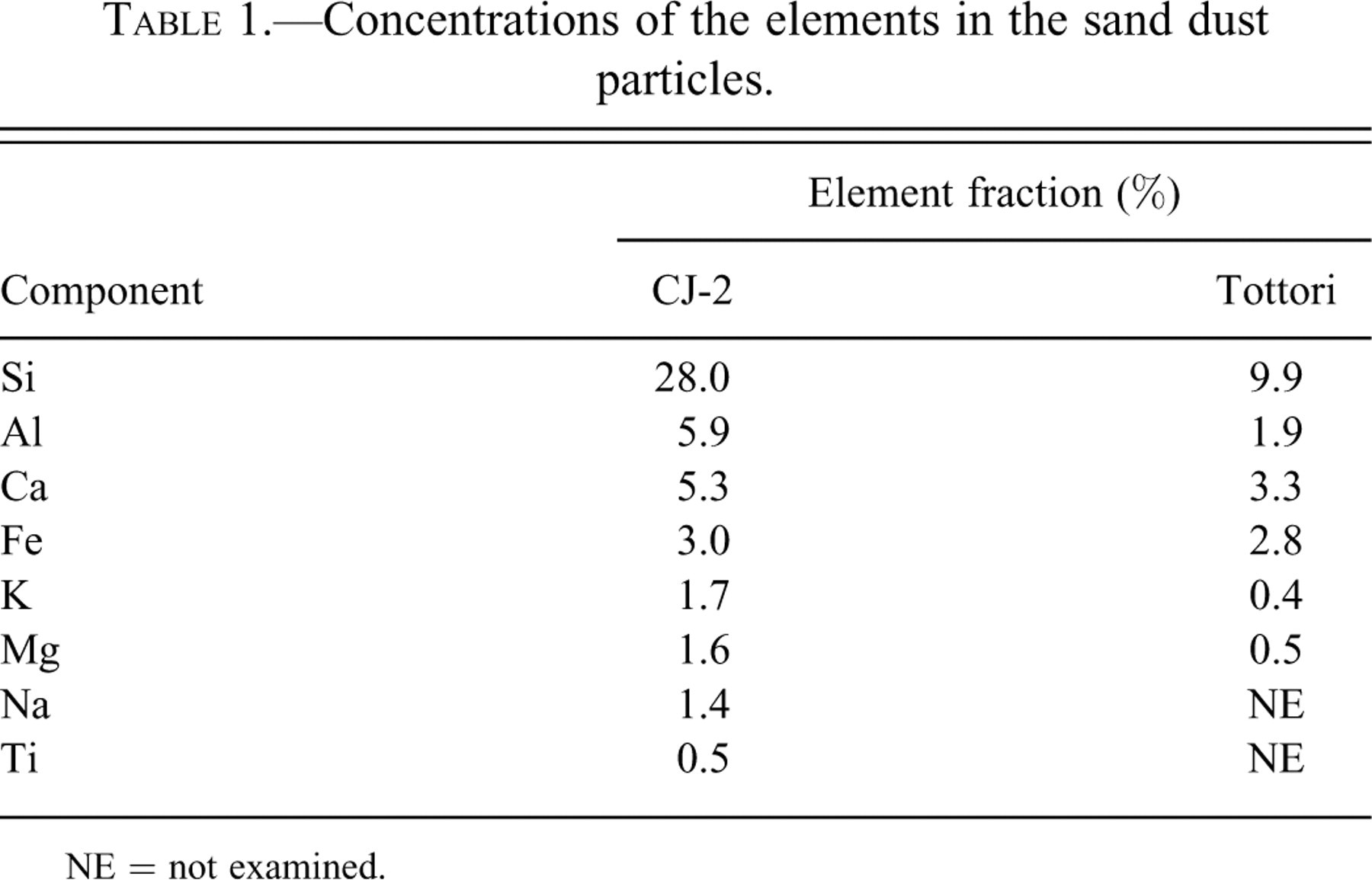
NE = not examined.
Histopathology of the Lungs
Histological sections from control animals instilled with normal saline showed normal bronchiolar and alveolar architectures with occasional mild hemorrhage. The mild hemorrhage observed in the controls may be associated with euthanasia because similar findings were observed in the lungs without instillation. In histological sections from animals instilled with sand dust particles, localized accumulation of the dust particles in the bronchioles and alveoli was observed; acute inflammatory changes characterized by infiltration of macrophages and neutrophils were observed around the particles (Figure 2). The intensity of the inflammation tended to increase with the amount of particles instilled (Figure 3). As compared with the controls, the lungs exposed to Asian sand dust particles showed increased numbers of neutrophils and macrophages (Figure 4A, 4C) in the areas around these particles, extending into the alveoli and the bronchoalveolar junction (Figure 4A). Intra-alveolar hemorrhage and the exudation of serum protein into the alveolar spaces (Figure 4A) were also noted. Large particles were observed within the bronchiolar and alveolar spaces. Small particles were occasionally seen in the cytoplasm of alveolar macrophages (Figure 4C). In the groups instilled with 800 and 3,000 µg of particles, some lesions of bronchial epithelial cell degeneration (Figure 4B) were observed in association with particle attachment, and some lesions of alveolar epithelial cell degeneration (Figure 4C) were observed in association with particles and/or inflammatory infiltration. Obstruction of the terminal bronchioles by the instilled particles and inflammatory cells was also observed. Alveolar epithelial cell degeneration was observed regardless of the obstruction of the airways. There was no difference in either the incidence or the severity of the histological changes between the lungs treated with different sources of particles.
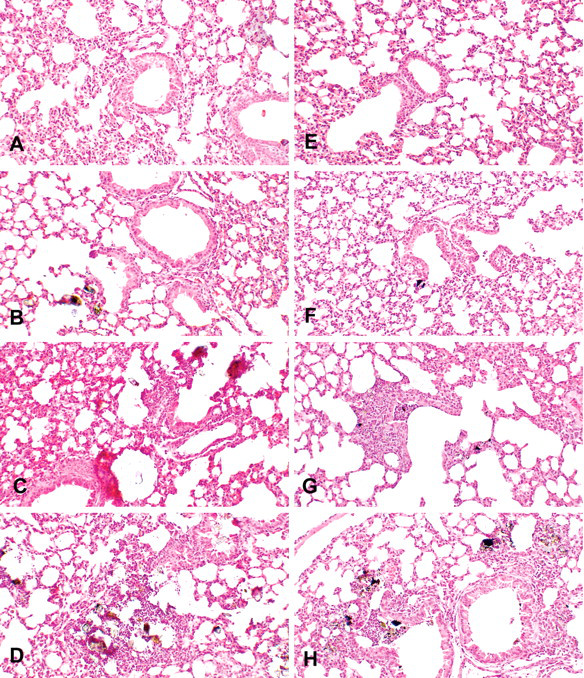
Histological examination of the lungs treated with CJ-2 and Tottori particles showed acute inflammation. Representative lung sections taken from mice instilled with CJ-2 particles—(A) 50 µg/mouse, (B) 200 µg/mouse, (C) 800 µg/mouse, and (D) 3,000 µg/mouse—and from mice instilled with Tottori particles—(E) 50 µg/mouse, (F) 200 µg/mouse, (G) 800 µg/mouse, (H) 3,000 µµg/mouse. Hematoxylin and eosin 100×.
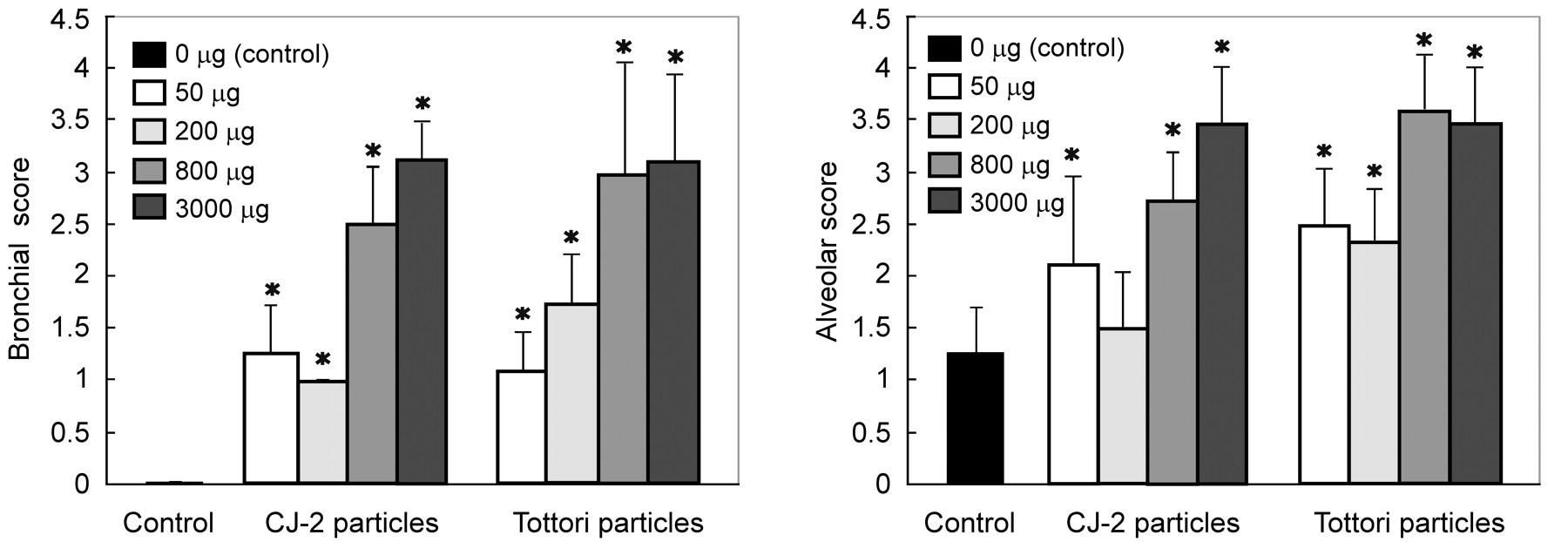
Bronchial and alveolar scores were derived according to the intensity of changes in the alveoli, bronchioles, and bronchi, as observed with a light microscope. *Significant difference (p < .05) from the control group.
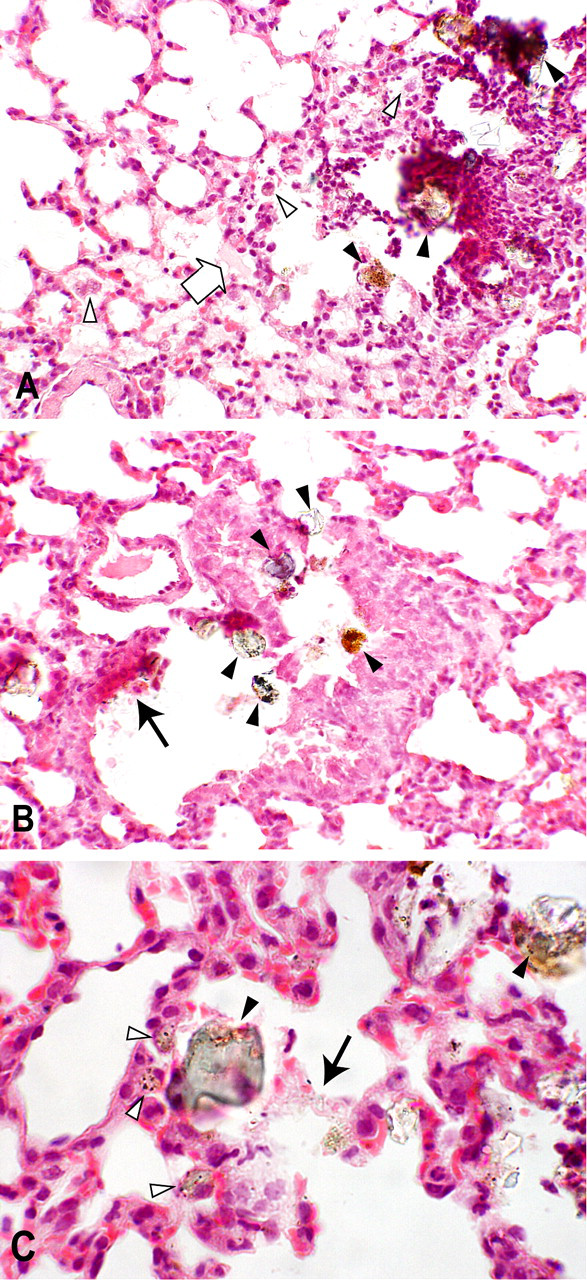
Histological changes of the lungs treated with 3,000 µg of CJ-2 particles. (A) Infiltration of neutrophils (upper right) into the alveoli and the bronchoalveolar junction, an increased number of macrophages (white arrowheads), and exudation of the serum protein into the alveolar space (white arrow). (B) Degenerated bronchiolar epithelial cells (black arrow). 200×. (C) Degenerated alveolar epithelial cells (black arrow). Large particles are shown by black arrowheads. Small particles are shown by white arrowheads. Hematoxylin and eosin. (A, B) 200×. (C) 400×.
Immunohistochemistry of the Lungs
Laminin
In the control lung tissue, positive immunolabeling for laminin was expressed as a thin, continuous line in the basement membranes of the bronchioles and blood vessels, around the bronchial glands, and along the alveolar septa (Figure 5A). Weak, barely positive staining was observed in a discontinuous pattern along the basement membranes of the bronchioles and alveoli of mice exposed to the particles. The basement membranes of the alveoli containing clumps of sand particles and inflammatory cells showed interruptions, with a patchy distribution of immunolabelings (Figure 5B).
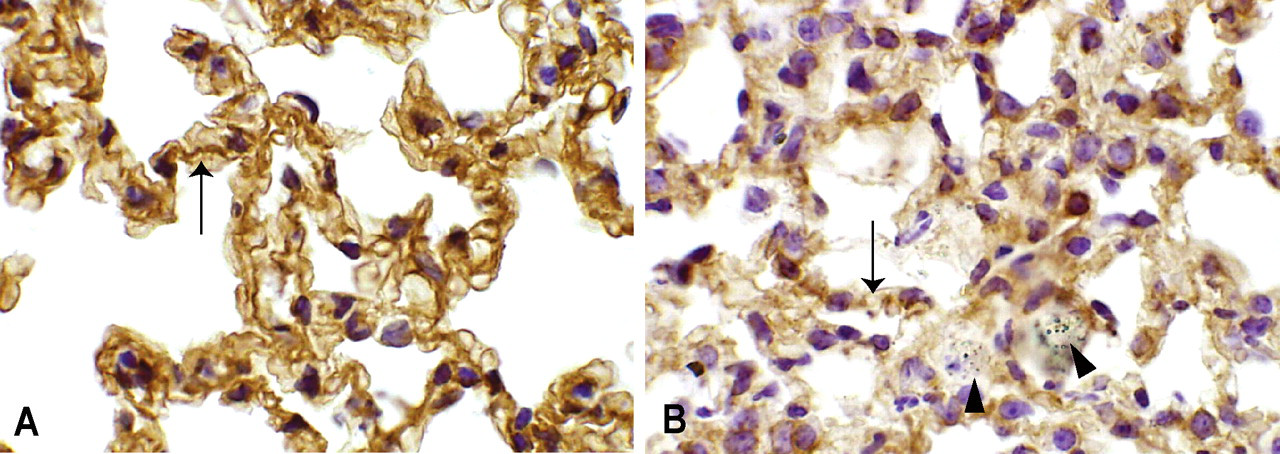
Laminin immunohistochemstry of the lungs of mice treated with (A) saline solution and (B) 3,000 µg of CJ-2 particles. (A) Positive immunolabeling for laminin (arrow). (B) Weakly positive discontinuous immunolabeling for laminin (arrow) around the particles (arrowheads). 570×.
TNF-α
The cytoplasm of the inflammatory cells, including macrophages and neutrophils (Figure 6B), and fibroblasts around the bronchioles and peribronchiolar blood vessels in the inflammatory lesions showed positive immunolabelings for TNF-α in the mice treated with CJ-2 particles. Type 2 alveolar epithelial cells in the inflammatory lesions also showed positive immunolabelings for TNF-α.
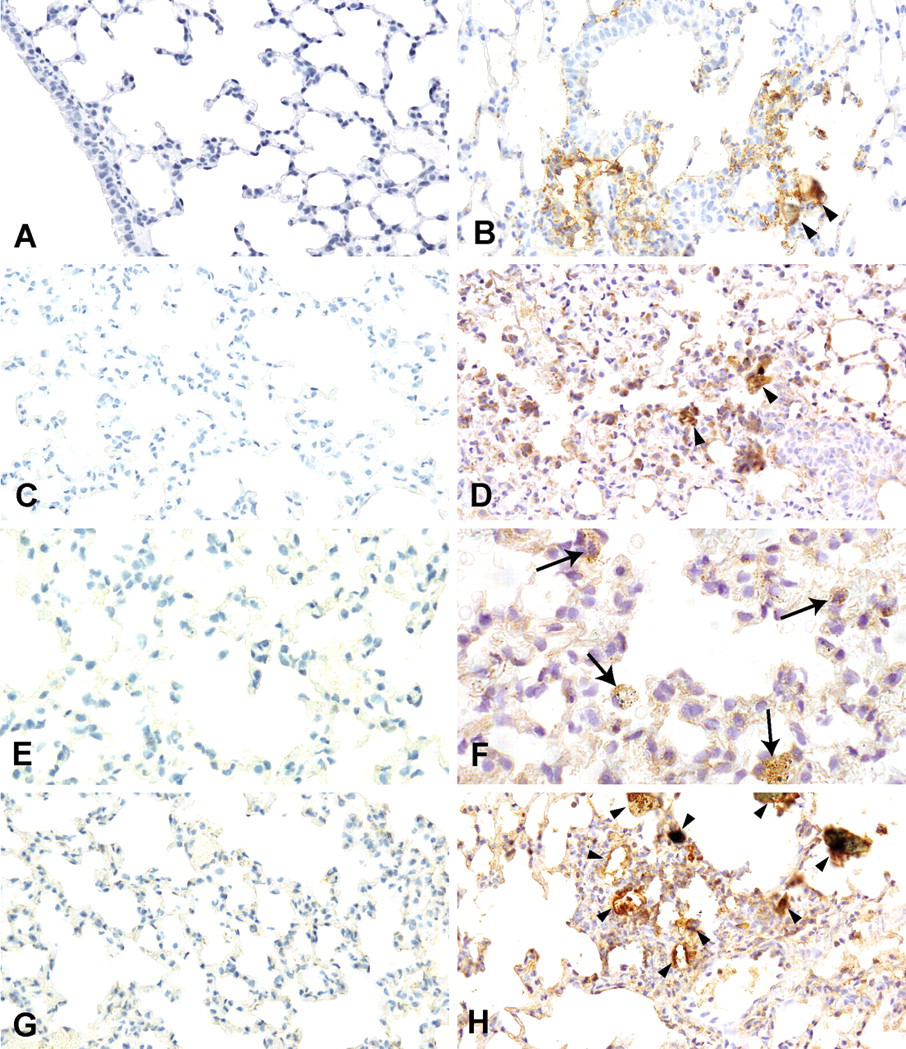
Immunohistochemichal localization of (A) tumor necrosis factor–α (TNF-α), (C) interleukin-6 (IL-6), (E) dimeric copper- and zinc-containing superoxide dismutase (Cu/Zn SOD), and (G) inducible nitric oxide synthase (iNOS) in the lungs of mice treated with saline (control group). Immunohistochemichal localization of (B) TNF-α, (D) IL-6, (F) Cu/Zn SOD, and (H) iNOS in the lungs of mice treated with 3,000 µg of CJ-2 particles. (F) Macrophages, containing black particles in the inflammatory lesions, showed positive immunolabeling for Cu/Zn SOD with a granular staining pattern (arrows). Arrowheads indicate particles. (A-D, G, H) 400×. (E, F) 600×.
IL-6
The cytoplasm of the inflammatory cells (Figure 6D), type 1 alveolar epithelial cells, and type 2 alveolar epithelial cells in the inflammatory lesions showed positive immunolabelings for IL-6 in the mice treated with CJ-2 particles.
Cu/Zn SOD
The cytoplasm of the macrophages (Figure 6F), type 1 alveolar epithelial cells, type 2 alveolar epithelial cells, bronchial epithelial cells, and endothelial cells in the inflammatory lesions showed positive immunolabelings for Cu/Zn SOD in the mice treated with CJ-2 particles.
iNOS
The cytoplasm of the inflammatory cells in the inflammatory lesions of the mice treated with CJ-2 particles showed positive immunolabelings for iNOS (Figure 6H).
Electron Microscopy
The alveolar epithelial cells of the control group treated with saline alone showed ruffling on the surface and an increased number of vesicles of various sizes. The alveolar epithelial cells of the animals treated with Asian sand dust particles had features indicating considerable damage to the lung tissues: edematous degeneration with marked cellular vacuolar changes leading to desquamation (Figure 7). Edematous changes and dissociation of the basement membranes were occasionally associated with particle attachment (Figure 7A). Instilled particles, ranging from 30 to 500 nm in size, were observed in the alveolar lumen, the phagolysosomes of macrophages, and the lamellar bodies of type 2 alveolar epithelial cells, as well as on the surfaces of the alveolar epithelial cells. Some mitochondria showed swelling and disruption of cristae, resulting in the formation of concentric structures known as myelin figures (Figure 8).
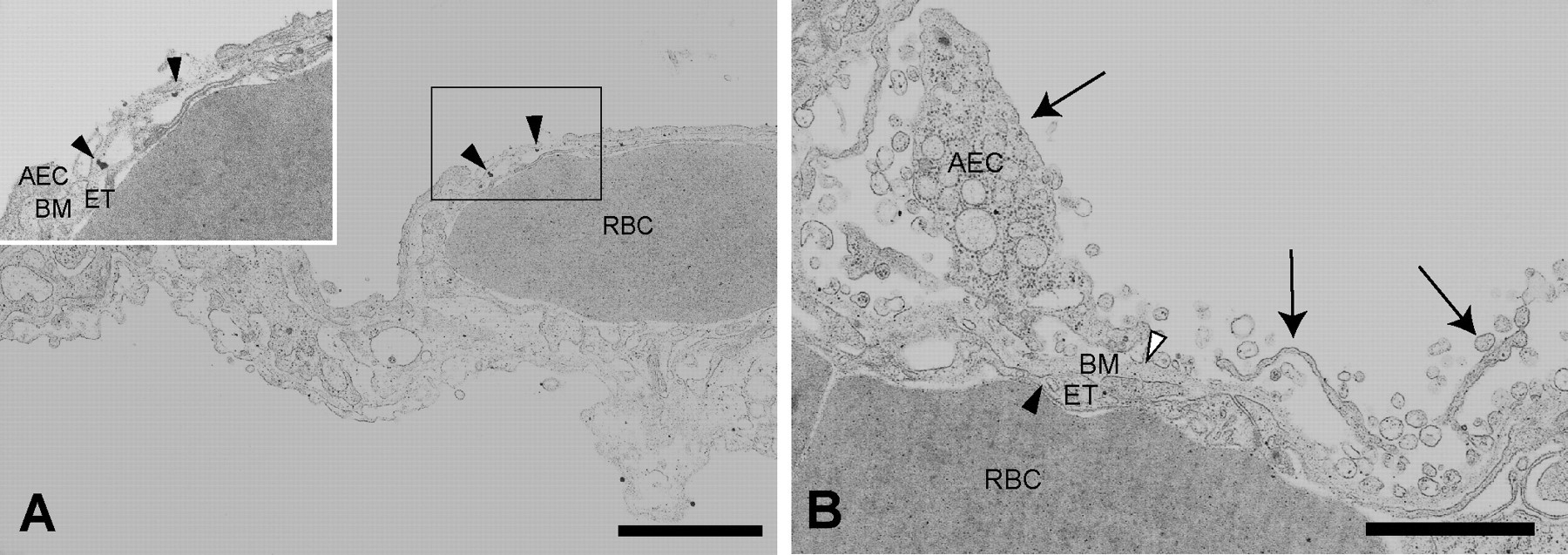
Transmission electron microscopic image of the lungs treated with 800 µg of CJ-2 particles. (A) Alveolar epithelial cells showing marked edematous cytoplasm. Inset: Dissociation of degenerative alveolar epithelial cells and the basement membrane associated with the attachment of CJ-2 particles (arrowheads). (B) Desquamation of degenerative alveolar epithelial cells with vacuolar appearance (arrows). AEC = alveolar epithelial cell, BM = basement membrane, ET = endothelial cell, RBC = red blood cell. Bars = 2 µm.
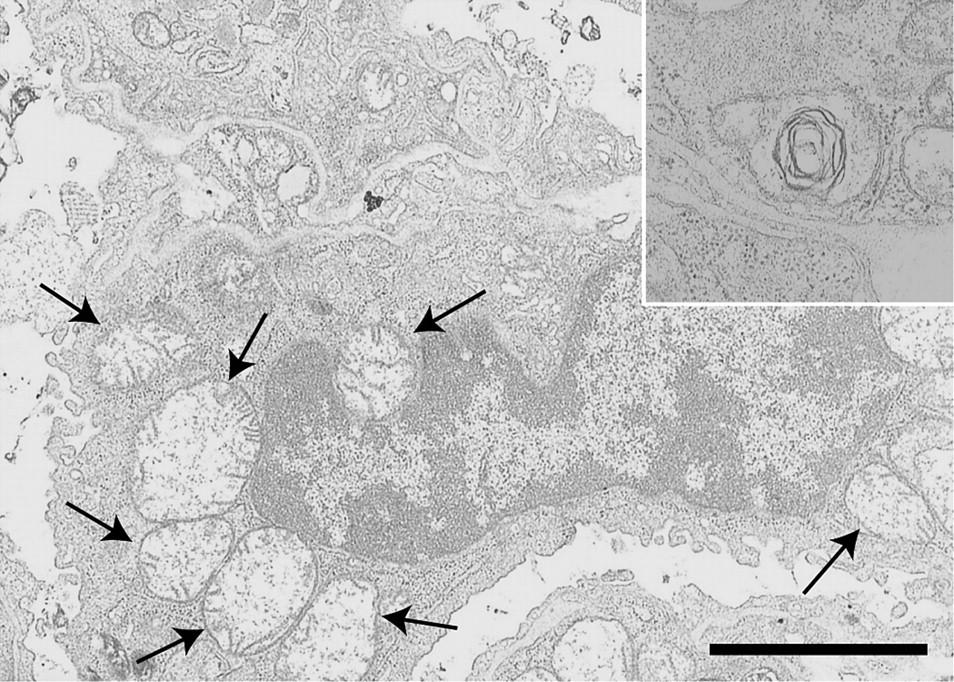
Transmission electron microscopic image of the lungs treated with 800 µg of CJ-2 particles. Morphological changes of mitochondria in the alveolar macrophage. Note the swollen mitochondria with the disappearance of cristae (arrows) and formation of myelin figures (inset). Bar = 3 µm.
Cytology and Total Protein Analysis of BALF
Table 2 shows the cellular and biochemical parameters of BALF in the groups of mice treated with CJ-2 particles. Total cell numbers significantly decreased at the 800-µg level compared with the control. The percentage of neutrophils and the total protein concentration increased in a dose-dependent manner; at the 800- and 3,000-µg levels, this increase was significant compared with that of the control. Cell viability was significantly decreased at the 800- and 3,000-µg levels compared with the control.
Cellular and biochemical parameters of bronchoalveolar lavage fluid.
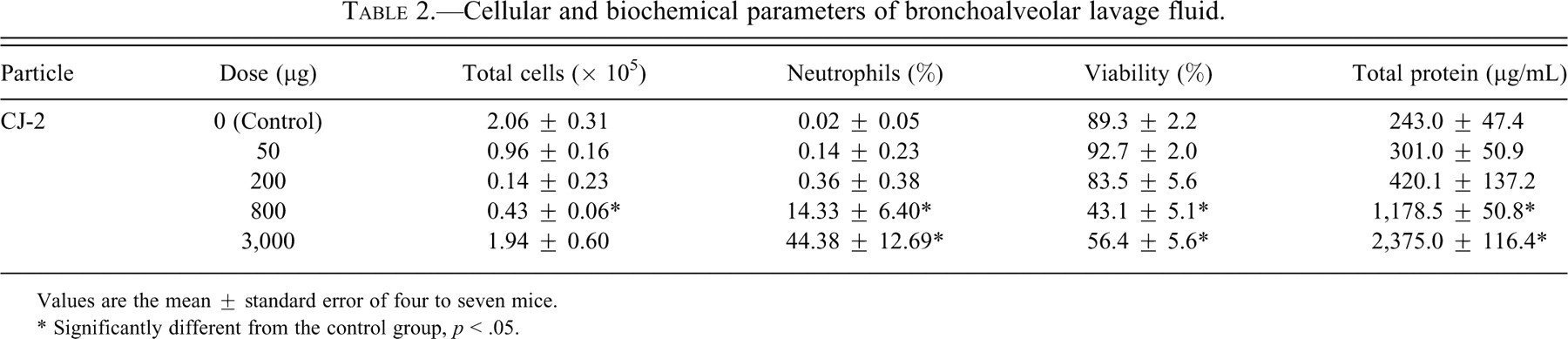
Values are the mean ± standard error of four to seven mice.
* Significantly different from the control group, p < .05.
Discussion
The present study describes acute pulmonary toxicity induced in mice by the intratracheal instillation of the mineral component of Asian sand dust particles, free of chemical and biological substances. Possible mechanisms for the damage (inflammatory changes) of lung tissues are also discussed.
Inhalation studies are the ideal experimental approach for assessing the effect of ambient particles. However, intratracheal instillation is an easier method than inhalation and has been proposed as a reliable route for assessing the pulmonary toxicity of particles in rodents (Warheit et al. 2005; Yokohira et al. 2008). In addition, similar histopathological results have been previously observed for both methods (Warheit et al. 2005). Therefore, we chose to use intratracheal administration in these experiments to evaluate the pulmonary toxicity of inhaled Asian sand dust.
In this study, mineral components of Asian sand dust particles caused acute inflammatory changes in the respiratory system as observed 24 hours after exposure; the primary inflammatory cells observed were alveolar macrophages and neutrophils in both BALF and lung tissues. Histological sections of the lungs from animals intratracheally instilled with both CJ-2 particles and Tottori particles showed bronchitis and alveolitis characterized by infiltration of macrophages and neutrophils around the particles. Cellular degeneration of the alveolar walls was also observed in the lung tissues examined by electron microscopy. These pathological findings such as bronchitis and alveolitis were consistent with previous reports on mice exposed to 200 to 800 µg of Asian sand dust (Hiyoshi et al. 2005; Ichinose et al. 2005; Ichinose, Yoshida, Hiyoshi, et al. 2008). In this study, obvious and severe histopathological changes were observed in the groups instilled with 800 and 3,000 µg of dust particles. The intensity of the inflammation in the lungs, percentage of neutrophils, and total protein concentration in the BALF increased in a dose-dependent manner. Histological and BALF examination of the lungs of mice exposed to particles showed that the mineral component of Asian sand dust, free from chemical and biological substances, can cause acute pulmonary toxicity at 24 hours after instillation when the treatment doses are relatively high.
An increase in the number of inflammatory cells was apparent in the histological sections, whereas BALF analysis showed no increase in total cell number. The observed difference in the number of inflammatory cells between the histology and the BALF analysis may be explained by the pattern of inflammatory changes in the tissues; the inflammatory lesions were multifocal, making it difficult to harvest all of the inflammatory cells from the lesions, and thus, their numbers in BALF were underestimated in the exposed mice. Obstruction of the terminal bronchioles by the instilled particles, which was actually observed in the histology, may also affect the recovery of the inflammatory cells in BALF.
In this study, we used both CJ-2 particles and Tottori particles to examine the acute lung toxicity caused by Asian sand dust particles. A previous study reported that the CJ-2 particles were made as a reference material for Asian sand dust aerosol (Nishikawa et al. 2000). CJ-2 particles and Asian sand dust particles collected in Japan and China have been shown to be quite similar in terms of size and composition, with comparable concentrations of elements and minerals (Nishikawa et al. 2000). Therefore, the Tottori particles were also expected to induce similar pathological changes. There was no difference in either the quality or the quantity of histological changes between the lungs treated with CJ-2 particles and those treated with Tottori particles. The similar histopathological changes observed in this study can be explained by the similar characteristics of these particles.
Quartz (crystalline silica) is known to cause cytotoxic and genotoxic effects in pneumocytes (Schins et al. 2002). Amorphous and crystalline silica (Murphy et al. 1998) as well as Asian sand dust (Ichinose et al. 2005; Ichinose, Yoshida, Sadakane, et al. 2008; Ichinose, Yoshida, Hiyoshi, et al. 2008; Lei et al. 2004; Yanagisawa et al. 2007) have been reported to cause inflammatory responses with the release of inflammatory cytokines in the lungs of experimental animals. The Asian sand dust used in the present study has a mineral composition that includes montmorillonite, vermiculite, mica, gypsum, chlorite, kaolinite, calcite, feldspar, and quartz (Quan et al. 1996). The major inorganic chemical element in the Asian sand dust tested in this study was Si, which was derived from quartz (crystalline silica) and feldspar (Nishikawa et al. 2000). A previous report suggested that the concentration of SiO2 contributed to a majority of the inflammatory responses induced by Asian sand dust (Ichinose, Yoshida, Sadakane, et al. 2008); Si may be responsible for the inflammatory changes.
Proinflammatory cytokines (IL-6, TNF-α) were shown to be present in the inflammatory lesions in mice lungs treated with Asian sand dust particles in this study. IL-6 is a pleiotrophic cytokine with multiple activities (Goldsby et al. 2003). TNF-α is a key cytokine in acute inflammation and is related to tissue injury. TNF-α is responsible for the induction of chemokines in the lungs in response to crystalline silica, and the expression of chemokines is modulated by the presence of TNF-α (Pryhuber et al. 2003). The results obtained in this study suggest that these cytokines may be involved in the mechanism of the acute lung toxicity induced by Asian sand dust particles.
iNOS was also present in the inflammatory lesions in mice lungs treated with Asian sand dust particles in this study. Nitric oxide (NO) and reactive oxygen species (ROS) production may be involved in silica-induced pulmonary inflammation and damage (Blackford et al. 1994; Castranova et al. 2002; Huffman et al. 1998; Porter et al. 2006). A previous silica inhalation study determined that the sites of iNOS activation and NO-mediated damage were associated with lesions in the lungs (Porter et al. 2002). In this study, lung tissue samples taken 24 hours after exposure contained Cu/Zn SOD. Silica has been reported to cause oxidative stress as a result of the generation of ROS (Rimal et al. 2005; Vallyathan et al. 1988). SODs are thought to be one of the first lines of antioxidant defense and are highly efficient at protecting cells and tissues against oxidative stress by catalyzing the dismutation of superoxide radicals to hydrogen peroxide (Liu et al. 2000). The positive Cu/Zn SOD immunoreactivity demonstrated in the treated lungs in this study may indicate a pulmonary response to oxidant stress caused by Asian sand dust.
Electron microscopy in this study demonstrated mitochondrial structural damage such as myelin figures. These are regarded as the result of the disassociation of lipoproteins, which facilitates water uptake and intercalation between lamellar membrane stacks (Li et al. 2003). Damage to the mitochondria may also contribute to the generation of oxidative stress as an intracellular source of ROS (González-Flecha 2004; Li et al. 2003). These findings indicate that oxidative stress may be involved in the development of the acute lung toxicity induced by Asian sand dust particles.
A weakening in the positive immunolabeling for laminin around the particles was demonstrated in the inflammatory pulmonary lesions in this study. Laminin plays a central role in the stability of the basement membranes, as well as in the control of cellular interactions. Because laminin is present in all pulmonary basement membranes, it can be used as a marker for normal alveolar structures (Aumailley and Smyth 1998; Gil and Martinez-Hernandez 1984). In our electron microscopy observations, the dissociation of the basement membrane was occasionally found to be associated with particle attachment. The histological changes demonstrated in this study may be, in part, a direct effect of the Asian sand dust particles. As suggested in a previous study, the alveolar epithelial cells may have been damaged by the physical stimulus of the attachment of ultrafine carbon particles to the basement membrane (Shimada et al. 2006). Pulmonary fibrosis may result from a disturbance of the equilibrium between the synthesis and degradation of the pulmonary extracellular matrix (ECM; Dunsmore and Rannels 1996). Basement membranes at the air-blood barrier contain ECM including laminin (Hernandez 1988). This study showed a weakened positive immunolabeling for laminin in the inflammatory lesions, suggesting that accelerated degradation of the ECM may have been induced in the acute inflammatory lesions. The ECM changes observed in the early stages may develop into fibrosis in the chronic stages. Further work is necessary to define the chronic effects of Asian sand dust.
Instillation via a cannula introduced into the trachea is a straightforward method; however, it may disturb the surface lining ultrastructure of the airway or the alveoli including mucus, surfactant, macrophages, and foreign material such as nanoparticles (Mühlfeld et al. 2007). We chose to immerse the lung tissues in fixative after making longitudinal sections to prevent particle relocation; however, fixation of lung samples by immersion in fixative is not a preferred technique because the lung tissue is collapsed and artifactual changes may occur (Mühlfeld et al. 2007). Intratracheal instillation, which puts pressure on the lung tissues, has the potential to injure the bronchioles and alveolar walls. In addition, the instillation method may cause exposed particles to localize and accumulate in the lung tissues. In the groups instilled with 800 and 3,000 µg of particles, some lesions of alveolar epithelial cell degeneration were observed in association with particles and/or inflammatory cell infiltration. In contrast, electron microscopy showed diffuse degenerative changes in the alveolar epithelial cells of mice treated with 800 µg in association with/without particles. The suggestive findings of autolysis and degeneration of the alveolar epithelial cells were not observed in the control animals by light and electron microscopy. These findings suggest that mild degeneration of the alveolar epithelial cells occurred at least at the electron microscopic level. Degeneration of alveolar epithelial cells may be exacerbated in association with particles and/or inflammatory cell infiltration. This degeneration may also result, in part, from the very focal nature of the distribution of the instilled particles. In addition, the focal accumulation of particles, as observed by light microscopy, might delay the action of the fixative during immersion, with this delayed fixation resulting in the degeneration of the alveolar and bronchiolar epithelial cells.
In conclusion, this study demonstrated that the mineralogical components of Asian sand dust particles, free from chemical and biological pollutants, caused acute inflammatory changes in the lung tissue and BALF in vivo. Both the direct effects of the Asian sand dust particles and secondary released cytokines may be involved in the development of the acute lung toxicity; oxidative stress generated in the lesion may be responsible for the lung tissue damage.
Footnotes
Acknowledgments
This work was supported in part by the Global COE Program (Global Center of Excellence for Dryland Science), MEXT, Japan, and Research Fellowships of the Japan Society for the Promotion of Science for Young Scientists. The authors thank Ms. E. Kawahara at Tottori University for her excellent assistance in electron microscopy and Ms. Y. Yamamoto for her technical assistance.
