Abstract
Agent solubility is a problem for aspiration of agents into lungs for chemopreventive efficacy evaluation, since many agents have to be dissolved in solvents. These solvents may be toxic to the lung epithelium. A study was conducted in female A/J mice to determine toxicity of different solvent concentrations by using saline, dimethyl sulfoxide (DMSO), ethanol, polyethylene glycol 400 (PEG-400), and labrasol for 1, 5, and 28 days via aspiration route. Toxicity was determined by measuring changes in body weight and bronchoalveolar lavage fluid (BALF). No significant difference was observed in body weight, differential cell counts, lactate dehydrogenase (LDH) and total protein in all solvent groups compared to saline by 28 days except 50% ethanol. However, myeloperoxidase (MPO) and macrophage inflammatory protein-2 (MIP-2) showed significant increase in 2% and 10% DMSO, 10% ethanol, 0.1% and 2% PEG-400, and 1% labrasol by longer dosing. All solvents except for 10% ethanol and 2% PEG-400 are suitable for agent aspiration.
Introduction
The strategy of delivering a drug directly to its target tissue is aimed at maximizing efficacy while limiting toxicity. Delivery of agents to the respiratory tract as aerosols can be accomplished by nose-only inhalation route. 1 Our previous study demonstrated a significant inhibition of benzo[a]pyrene-induced mouse lung tumors by delivering a chemopreventive agent, Oltipraz, to the respiratory tract directly employing nose-only inhalation. 2 However, aerosolization of agents to obtain appropriate particle size for inhalation route requires large amounts of drug, which is a potential problem for drug development. An alternative method is to use oropharyngeal aspiration for delivering a drug directly to the respiratory tract. The aspiration method has been used in administering virus, bacteria, or soluble antigens directly to the respiratory tract in animal models and is a very noninvasive method compared to intratracheal instillation. 3,4 Our previous data comparing inhalation and aspiration methods for administrating an agent (9, 10 Phenanthrene diamine) to lung airway epithelia indicated that aspiration can be an equally effective route of exposure while reducing the need for inhalation equipment and amount of chemopreventive agent required (unpublished).
However, agent solubility is a major obstacle in employing the aspiration route, as many of the agents have poor water-solubility and need to be dissolved in organic solvents. There are reports of solvents altering the intestinal membrane barrier functions and causing damage to the intestinal epithelium. 5,6 Therefore, nonaqueous solvents may create potential toxicity to the normal lung epithelium, which could produce adverse effects on the efficacy of potential chemopreventive agents. In order to address this issue, a feasibility study was conducted to determine the appropriate nontoxic concentration of different solvents that can be used to dissolve chemopreventive agents for aspiration into the lungs.
A variety of solvents has been used to improve the solubility of poorly water-soluble drugs and many of them are well documented 7 for administration in animals. Dimethyl sulfoxide (DMSO) has been reported to be inert to other compounds 8 and has a high solubilization capacity and a very low toxicity. 7 Dimethyl sulfoxide has been used as a vehicle in preclinical studies via various administration routes such as subcutaneous, 9 intravenous, 10 and intraperitoneal 11 injections. Yoon and colleagues 12 reported inhibitory effects of DMSO administered at a dose of 1 mL/kg on acetaminophen-induced hepatotoxicity in rats. There are also reports of DMSO as a vehicle for aerosol formulations 13,14 ; however, these studies were performed only for a short period (i.e., single exposure lasting few hours) and did not address any toxicity issues associated with DMSO.
Ethanol is another common solubilizing vehicle and is frequently used as a co-solvent with propylene glycol 13 or Cremophor EL 14 in aqueous or organic solvent formulations of water-insoluble compounds. It is also a popular solvent for aerosol formulation with concentrations ranging from 2% to 100% as a mixture with normal saline in various animal models such as mice, 15 rats, 13 guinea pigs, 16 pigs, 17 and dogs. 18 Bund and colleagues 17 showed that low dose of ethanol (0.12 mg/kg/min) by inhalation did not affect the pulmonary hemodynamics or ventilation functions in pigs. However, as most of the ethanol-based aerosol formulations were tested for relatively short period (i.e., 30 minutes to 4 hours), no information on the toxicological effects of ethanol is available either with multiple or long-term exposures. Malik and colleagues 19 investigated toxicity in mice continuously exposed to ethanol vapor. However, administration of vapor is quite different from a local delivery to the lungs and it is difficult to calculate the exact dose that was actually deposited in the lungs. More recently, Montharu and colleagues 20 evaluated lung tolerance of ethanol in a rat model of intratracheal administration in a 4-day toxicity study. They demonstrated that 10% ethanol was well tolerated in the lung by performing histopathology and biochemical analyses of bronchoalveolar lavage fluid (BALF).
Polyethylene glycol (PEG)-400 is another chemical, soluble in both water and nonpolar solvents, with high hydrogen-bond-forming capacity, solubilizes hydrophobic compounds, 21 and is known to be unreactive toward other chemicals. 22 It has been extensively used in various cosolvent systems for parenteral administration, 23 oral microemulsion formulation, 24 and micronization or crystallization for inhalation. 25 Although 2-week aerosol inhalation study on PEG 3350 was reported in rats, 26 little information exists on the toxicological effects of PEG-400 following inhalation or aspiration in animal models.
Labrasol is a nonionic surfactant composed of PEG esters and glyceride, with medium length acyl chains. Nonionic surfactants are known to be less toxic than ionic surfactants to biological membrane. 27 Labrasol is obtained from coconut oil and shows very low toxicity in rats with an LD50 of 22 g/kg. Labrasol was reported to enhance the intestinal absorption of hydrophobic, poorly absorbable drugs such as gentamycin, 28 insulin, 29 and vancomycin 30 in rats. Labrasol also has been widely employed as a co-emulsifying reagent for topical application 31 and oral administration 32 of poorly water-soluble compounds. Labrasol showed almost no intestinal membrane damage at a concentration of up to 0.1% (v/v) 33 or in a 4-week toxicity study of a vehicle blend containing 40% labrasol by oral route in rats. 34 Again, there are no reports of studies focused on the toxicological effects of labrasol administration via inhalation or aspiration. As is evident from the literature on these various solvents, most of the studies were performed only for shorter periods without addressing the long-term effects. In this study, 4 solvents were tested as potential vehicles for chemopreventive agents administered via aspiration, which includes dimethyl sulfoxide (DMSO), ethanol, PEG-400, and labrasol. The toxic response was determined at 3 time points (1, 5, and 28 days after aspiration) per solvent in BALF by measuring endpoints on cell damage (cell counts, total protein, lactate dehydrogenase [LDH]) and inflammation (myeloperoxidase [MPO], macrophage inflammatory protein-2 [MIP-2]). Cursory lung pathology of animals in the high-dose group was also performed.
Materials and Methods
Animals
Eight-week-old, viral antigen- and specific pathogen-free Strain A/J female mice were purchased from the Jackson Laboratory (Bar Harbor, ME) and quarantined for up to 2 weeks in a temperature-controlled, pathogen-free animal facility before use. All study animals were provided with fresh deionized, chlorinated water, and pelletized, semipurified AIN-76A diet (casein 20%,
Chemicals
Labrasol were obtained from the NCI Chemical Repository (Rockville, MD). Ethanol (200 proof with purity >99.5%), DMSO (>99.7% in purity), PEG-400, and trypan blue solution were purchased from Sigma-Aldrich (St Louis, MO). All other chemicals obtained from commercial sources were of reagent grade.
Animal Experiments
Animals were weighed and randomized based on body weights to different groups and treated by aspiration with saline (control group) or the 4 test solvents in saline at 2 different concentrations (2% and 10% for DMSO, 10% and 50% for ethanol, 0.1% and 2% for PEG-400, and 0.02% and 1% for labrasol). Three different dosing periods were tested; once a day for a single day (3 animals/dose group), for 5 consecutive days (4 animals/dose group), or for 5 days/week for 4 weeks (4 animals/dose group). After dosing, animals were observed for morbidity/mortality and sacrificed on days 2, 8, and 29 for 1-, 5-, and 28-day dosing groups, respectively. Animal body weight was measured immediately prior to the first administration and before sacrifice or once a week for 28-day dosing group.
Aspiration
Mice were anesthetized with isoflurane vapor via an anesthesia machine. Upon verification of loss of muscular tension, the mice were secured on restraint board by hooking the upper and lower incisors with thin rubber bands, thereby holding the mouth open. Blunt forceps were used to gently pull the tongue out to one side of the mouth to prevent swallowing. A pipette tip filled with saline or test solvents was placed in the distal part of oropharynx. The test solvent was gently released and aspirated into the lower respiratory tract by natural reflex. The volume of the dosing solution did not exceed 0.2 mL/100 g (~50µL/mouse) of the body weight.
Determination of Pulmonary Cytotoxicity and Inflammation
On days 2, 8, or 29 after dosing, all animals were sacrificed and BALF was collected for evaluating toxicity and inflammation. Lung tissues were collected at days 8 and 29 for pathological examination. The cell pellet obtained by centrifuging BALF fluid at 1000 g for 10 minutes at 4°C was used for estimating total and differential cell scoring and the supernatant for analysis of total protein, LDH, MPO, and MIP-2.
Total Protein Measurement
A total of 10 µL of lavage fluid supernatant was incubated with 200 µL of BCA (Bicinchoninic Acid) reagent (Pierce Biotechnology, Rockford, IL) for 30 minutes at 37°C. After incubation, the absorbance (OD) at 550 nm was read within 30 minutes on an UV-Vis Spectrophotometer (SpectraMax 340). The protein concentration was calculated according to the OD value of the standard curve with bovine serum albumin (BSA) as a standard and expressed as mg/L.
Measurement of LDH
The level of LDH in the lavage fluid was measured using CytoScan LDH-Cytotoxicity Assay Kit (G-Biosciences, Maryland Heights, MO). Briefly, 50 µL of lavage fluid supernatant was incubated with 50 µL of LDH reconstitute substrate mix (provided in the kit) in a 96-well plate at 37°C for 30 minutes. After incubation, 50 µL of 0.1 mol/L H2SO4 was added to the mixture and mixed well to stop the reaction. The absorbance (OD) at 492 nm was read within 30 minutes on an UV-Vis Spectrophotometer (SpectraMax 340). The LDH level in the lavage fluid was calculated based on the standard curve, normalized with protein concentration and expressed as ng/µg protein.
Total Cell Count in BALF
Cells collected from the lavage fluid were suspended in 1 mL of F-12 media supplemented with 10% FBS. Cell viability was determined by Trypan Blue exclusion by mixing equal volume of cell suspension and trypan blue solution using a hemocytometer and a light microscope. The number of total cells was measured by Coulter Counter (Coulter Electronic, Hialeah, FL) and expressed as the number of viable cells/mL.
Differential Cell Scoring
Approximately 5 × 10 4 cells were used for cell smear in slides, fixed in methanol for 10 minutes, air-dried, and stained with Diff-Quik Stain Kit (Fisher Scientific, Pittsburgh, PA). Macrophages, neutrophils, and lymphocytes were identified and scored under a light microscope. A total of 500 cells per animal were counted by random field counting. The number of each type of cells was expressed as a percentage of total cells.
Measurement of MPO
The level of MPO was measured in the lavage fluid using Myeloperoxidase EIA Kit (Oxis International Inc, Beverly Hills, CA) based on the manufactures' instruction. The MPO level in the lavage fluid was calculated based on the standard curve and expressed as ng/mL.
Measurement of MIP-2
The level of MIP-2, inflammation-specific cytokine, was measured in the lavage fluid using Mouse MIP-2 (IL-8) ELISA Kit (ARP American Research Products, Inc, Belmont, MA) based on the manufactures' instruction. The MIP-2 level in the lavage fluid was calculated based on the standard curve and expressed as pg/mL.
Pathological Analysis
Lung tissues were collected for each high dose at days 8 and 29 for pathological examination. The lungs were inflated with 10% neutral buffered formalin, with the fixing solution introduced via a cannula passed down the endotracheal tube at a pressure of 22 cm H2O, and then fixed in the same for 24 hours. Sections (5 μm) of paraffin-embedded tissues were stained with hematoxylin and eosin (H&E) and then examined microscopically.
Statistical Analysis
Statistical comparisons were made by Student t-test using GraphPad Prism software (GraphPad Software, Inc, La Jolla, CA). Results were considered significant using a P < .05 and were expressed as mean ± SD.
Results
Body Weight Changes
There was no significant body weight loss in all solvent exposure groups compared to saline control group after 1- and 5-day exposure (data not shown). There was a slight reduction of body weights in all dosing groups including saline control by the end of the 28-day exposure period (Figure 1 ). A statistically significant body weight reduction (16%-18% loss from the first week of dosing) was observed at the end of the exposure period in 10% ethanol and 2% PEG-400-treated groups, where as in the other exposure groups, the body weight loss was less than 10%. None of the solvents caused any animal death except for 50% ethanol, which killed all animals after the first dosing.
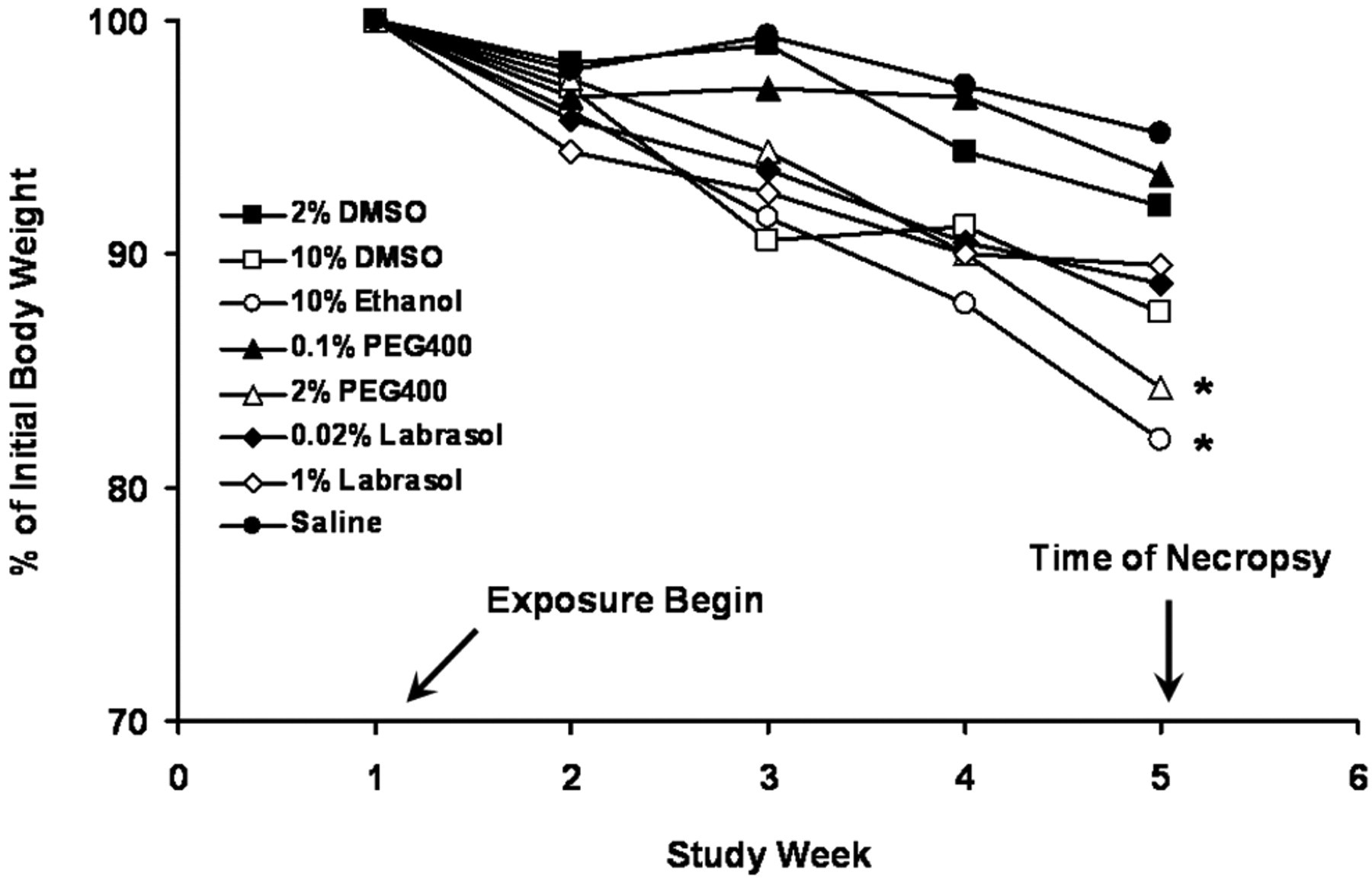
Data on animal body weight in 28-day solvent toxicity study (*P < .05 vs initial body weight).
Effect on LDH
As shown in Figure 2 , there was no significant change in the concentration of LDH in BALF in all solvent exposure groups compared to that of saline control group (P > .05) at all time points. The level of LDH after 1-day exposure was higher than those measured at later time points (i.e., 5- and 28-day exposure) in all solvent exposure groups including saline group except for 0.1% PEG-400 and 1% labrasol. In 0.1% PEG-400 exposure group, the LDH level remained the same during the entire exposure period. Highest LDH concentration was observed at 5-day exposure in 1% labrasol group.
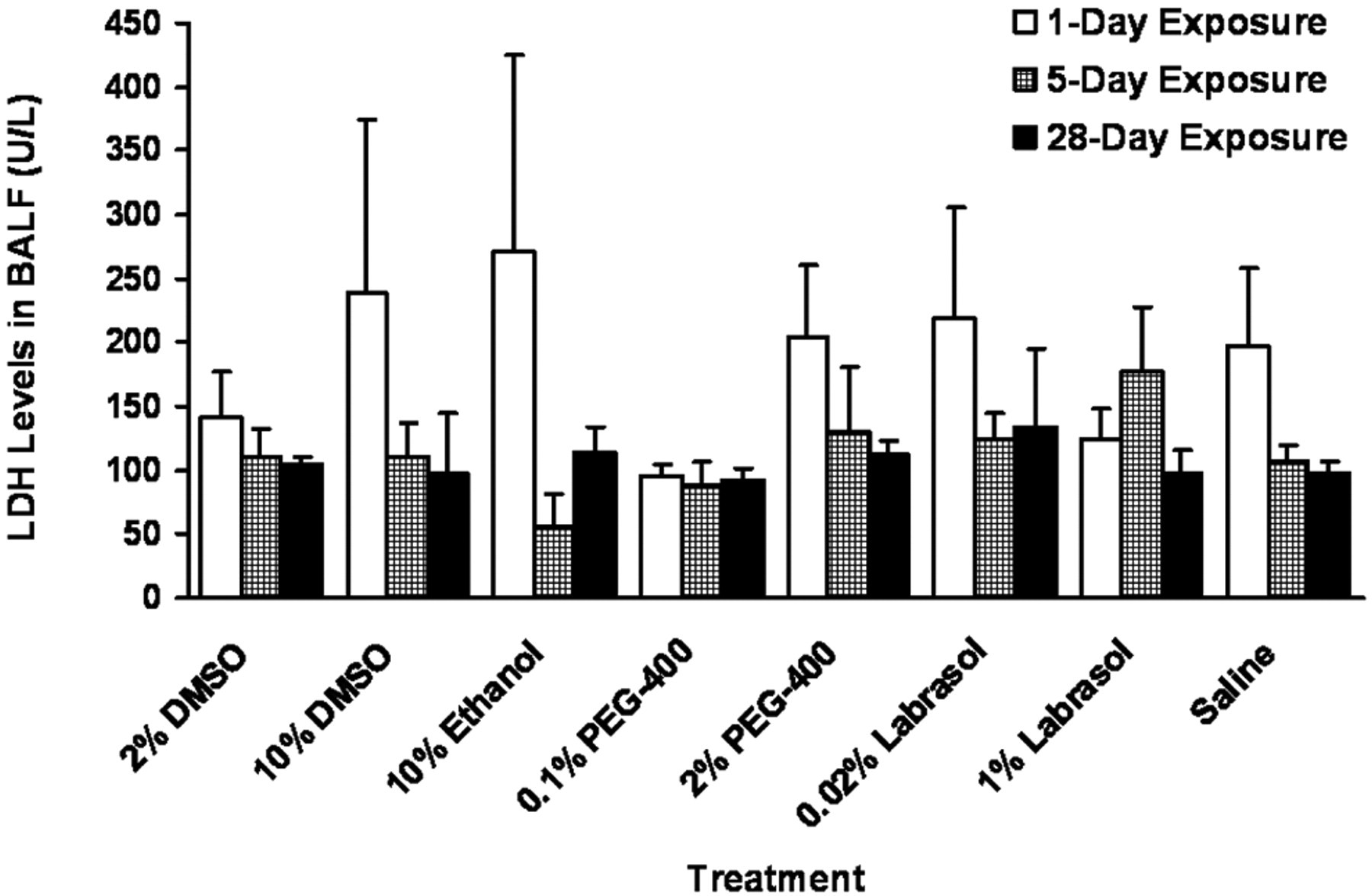
Results on lactate dehydrogenase (LDH) levels in bronchoalveolar lavage fluid (BALF) at various time points postdosing.
Effect on Total Protein
There was no statistically significant change in total protein concentration in BALF in all solvent exposure groups except for 1% labrasol which showed 16-fold increase (3200 mg/L as albumin) in total protein concentration compared to that of saline-treated group (200 mg/L as albumin) after 1-day exposure (Figure 3 ). However, the concentration of total protein in 1% labrasol-treated group decreased to the level of saline by 5 days of exposure and remained the same until the end of the exposure period (28 days). Similar to the LDH level, the total protein concentrations in all solvent exposure groups were highest after 1 day exposure and decreased with time.
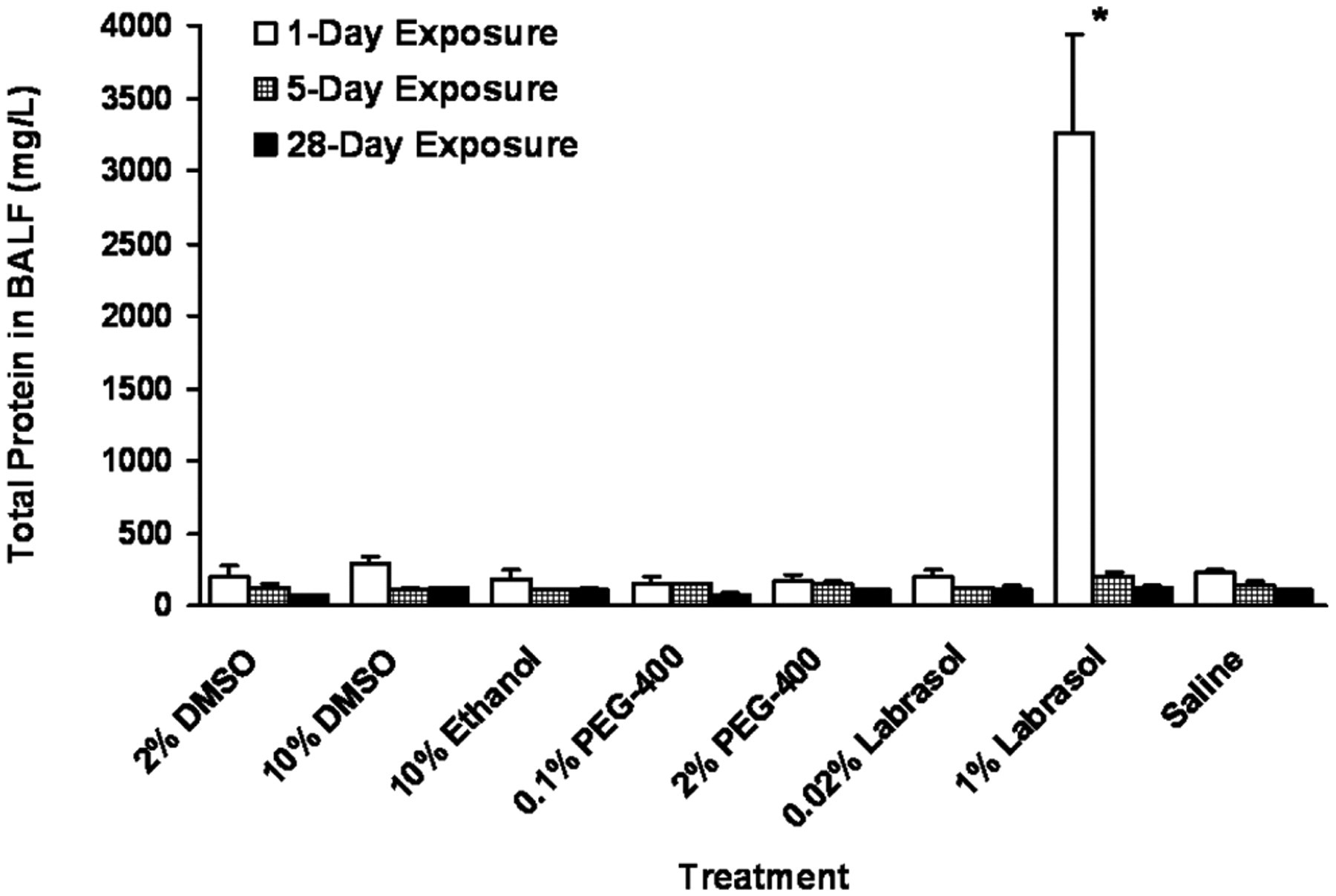
Total protein in bronchoalveolar lavage fluid (BALF) at various time points postdosing (*P < .05 vs saline control).
Effect on Total Cells and Different Cell Types
There was no significant change in the total cell counts in BALF by any solvent treatment during the entire exposure period except for 1% labrasol (Figure 4A) that showed a temporary increase at 1-day exposure compared to saline control. The majority of cells in BALF were macrophages followed by neutrophils (featuring a multilobed nucleus; arrows in Figure 4C) and lymphocytes as shown in Figure 4C. No statistically significant increase was observed in the percentage of macrophages or lymphocytes (data not shown) or neutrophils in all solvent groups compared to that of saline control group at all time points (Figure 4B). However, with respect to neutrophils, there was 3-fold decrease in 10% DMSO and 10% ethanol groups after 1 day dosing compared to the saline control group. Labrasol-treated group at a dose of 1% showed high neutrophil counts after 1 day (50%) and 28 days (22%) of exposure, however, none of these values was statistically significant compared to saline control group (P > .05).
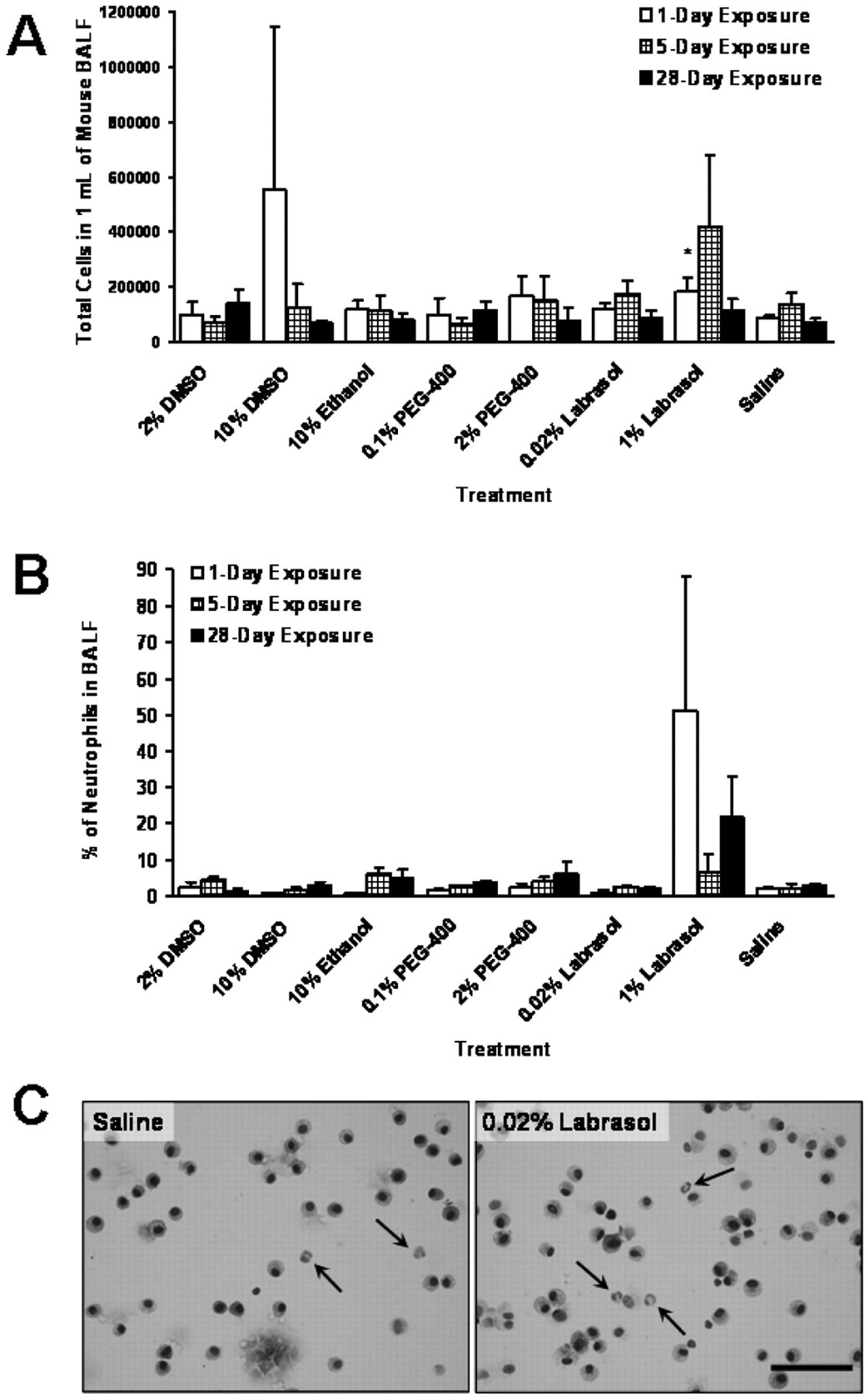
Total cells (A), neutrophil cell counts (B) and representative micrographic images of differential cell counts (C, neutrophils represented by arrows; bar = 100 μm) in BALF after solvent exposure at various time points (*P < .05 vs saline control).
Effect on MPO
The MPO concentration in saline-treated group was highest (17.4 ng/mL) after 1-day exposure and lowest (3.5 ng/mL) at 5-day exposure among the 3 time points tested (Figure 5 ). Similar pattern of MPO concentration changes was found in 10% DMSO, 0.1% PEG-400, and 0.02% and 1% labrasol exposure groups. However, 1% labrasol group showed significantly increased MPO concentration at 1- and 28-day exposure compared to saline control group. The concentration of MPO also increased significantly after 5-day exposure with 2% DMSO, 10% ethanol, and 2% PEG-400 and after 28-day exposure with 2% PEG-400.
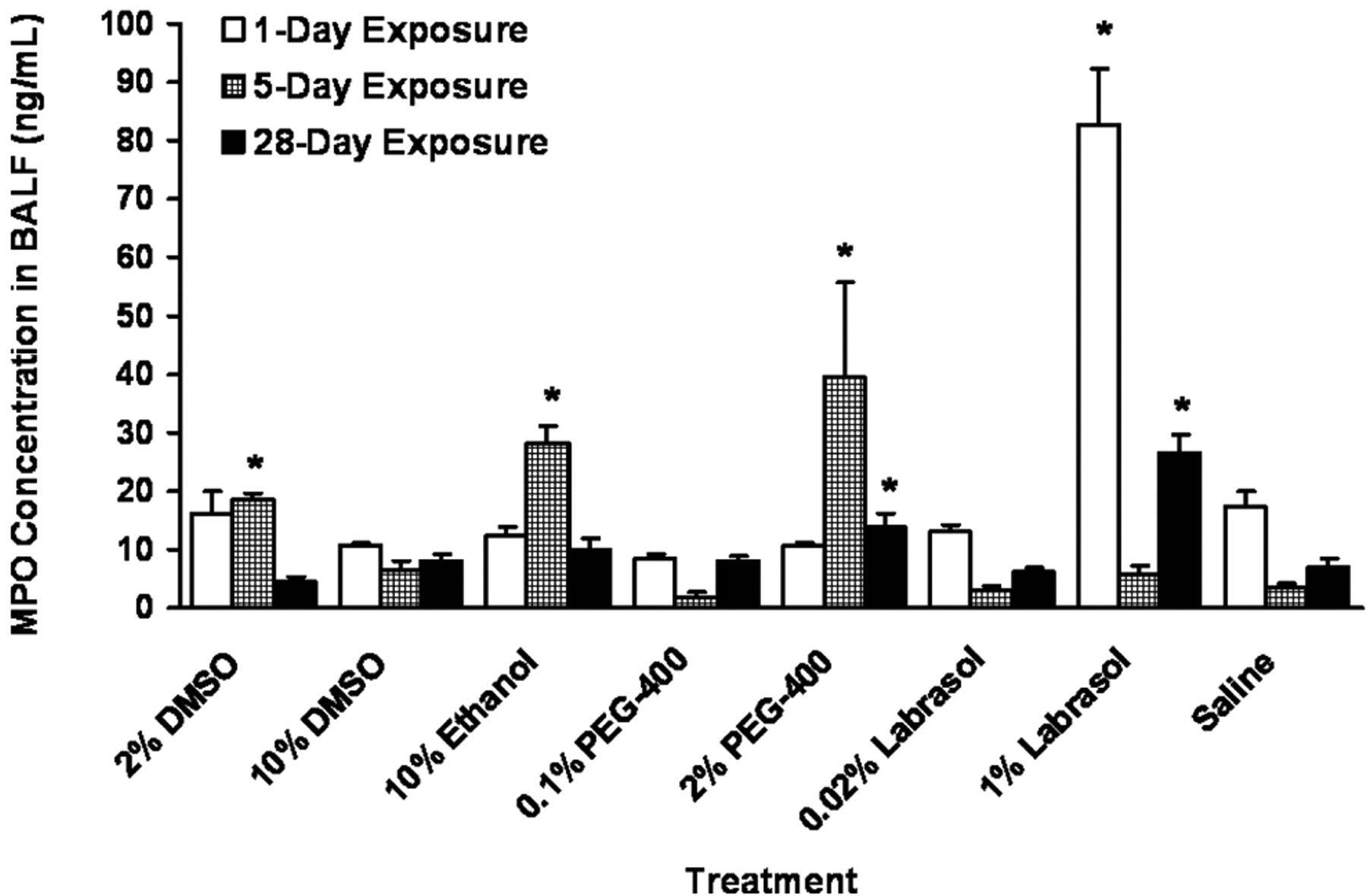
Myeloperoxidase (MPO) concentration in bronchoalveolar lavage fluid (BALF) at various time points postdosing with solvents (*P < .05 vs saline control).
Effect on MIP-2
The level of MIP-2 in BALF from saline-treated mice was approximately 4 to 5 pg/mL at shorter exposures (1 and 5 days of exposure), which decreased significantly after the longer period of 28-day exposure to 0.5 pg/mL (Figure 6 ). Most of the solvent exposure groups showed similar pattern of MIP-2 concentration changes over the 28-day exposure period. The exceptions were the significant increases observed in MIP-2 concentration after 5-day exposure with 10% DMSO, 5- and 28-day exposure with 10% ethanol and 0.1% PEG-400 compared to the saline control group.
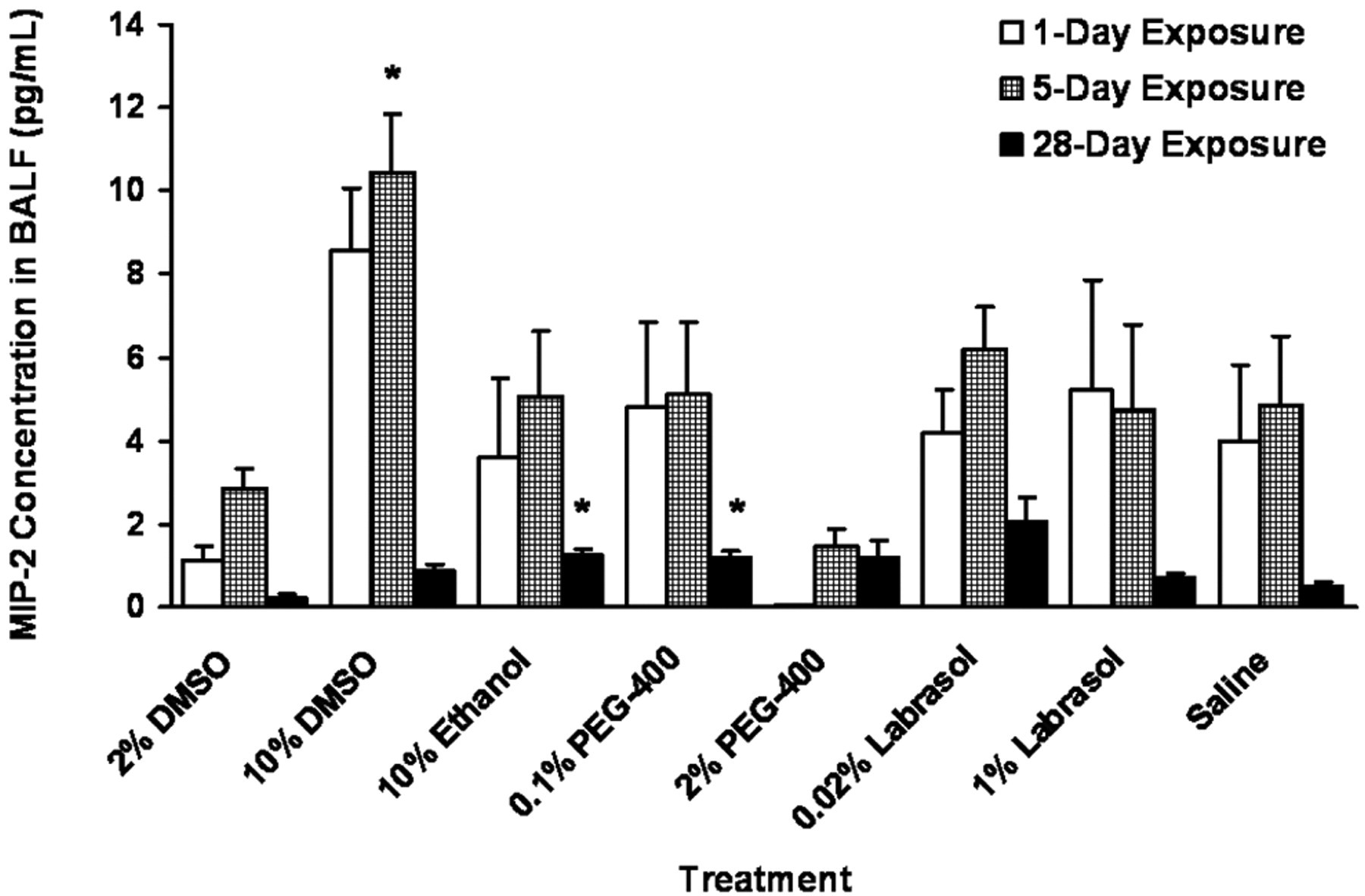
Macrophage inflammatory protein-2 (MIP-2) levels in bronchoalveolar lavage fluid (BALF) after dosing with solvents (*P < .05 vs saline control).
Pathological Evaluation
Pathological analysis of lung tissues from 5- and 28-day dosing groups is shown in Table 1 . The lung sections from 5-day exposure to various solvents showed that there was mild pulmonary congestion in the lungs of all animals, except the one from 10% DMSO, which demonstrated moderate congestion. A few neutrophils were found in lung sections of 2% DMSO and 1% labrasol groups. A few foci of neutrophils infiltrating were also found in section of 10% DMSO. There were no other pathological findings in any of the other sections. In the lung sections from 28-day exposure, a mild congestion was found in the lungs of all animals except the ones from 2% DMSO, 10% DMSO, and 2% PEG-400, all of which showed moderate congestion in their lungs. The infiltrating of lymphocytes, macrophages, and neutrophils were found in 2% DMSO, 10% ethanol, 0.1% PEG-400, and 2% PEG-400 exposure groups. A few lymphocytes and neutrophils were also noticed in the section from saline control. There were no other pathological findings in these sections. As shown in Figure 7 , leukocytic (neutrophil and/or lymphocyte) infiltration was found in peribronchial or perivascular regions. Lungs in all treatment groups including saline control showed mild-to-moderate pulmonary congestion featuring dilated capillaries in alveolar walls (arrow in the insert of Figure 7) and transudate, an extravascular fluid that replaced the air, accumulation in the alveolar lumen (asterisks in the insert of Figure 7) after 5- and 28-day exposure.
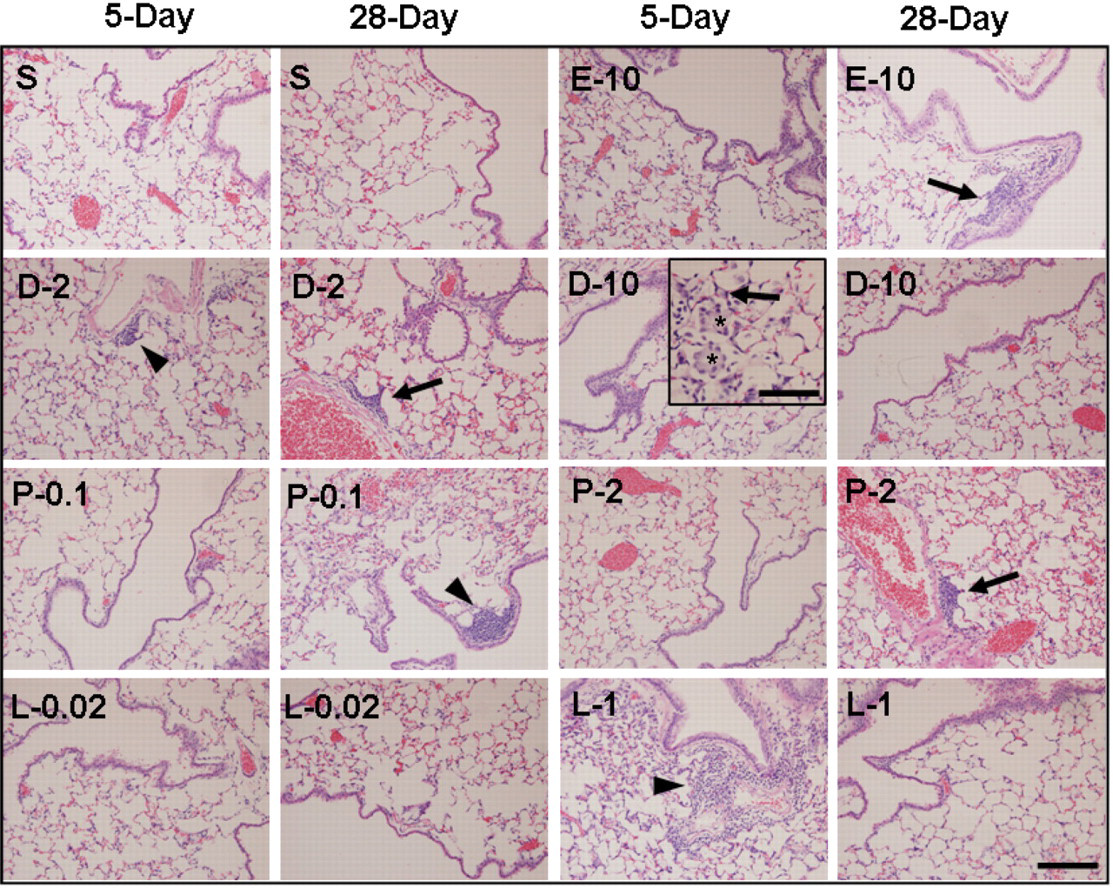
Representative micrographic images of hematoxylin and eosin (H&E)-stained lungs for pathological evaluation (S, saline; E-10, ethanol 10%; D-2, DMSO 2%; D-10, DMSO 10%; P-0.1, PEG400 0.1%; P-2, PEG400 2%; L-0.02, labrasol 0.02%; L-1, labrasol 1%; neutrophil (arrowheads) or lymphocyte (arrows) infiltration; dilated capillaries (arrow in the insert, bar = 100 μm); alveolar lumen filled with transudate (asterisks in the insert); bar = 200 μm.
Pathological Observation of Lung Tissues From Solvent Toxicity Study
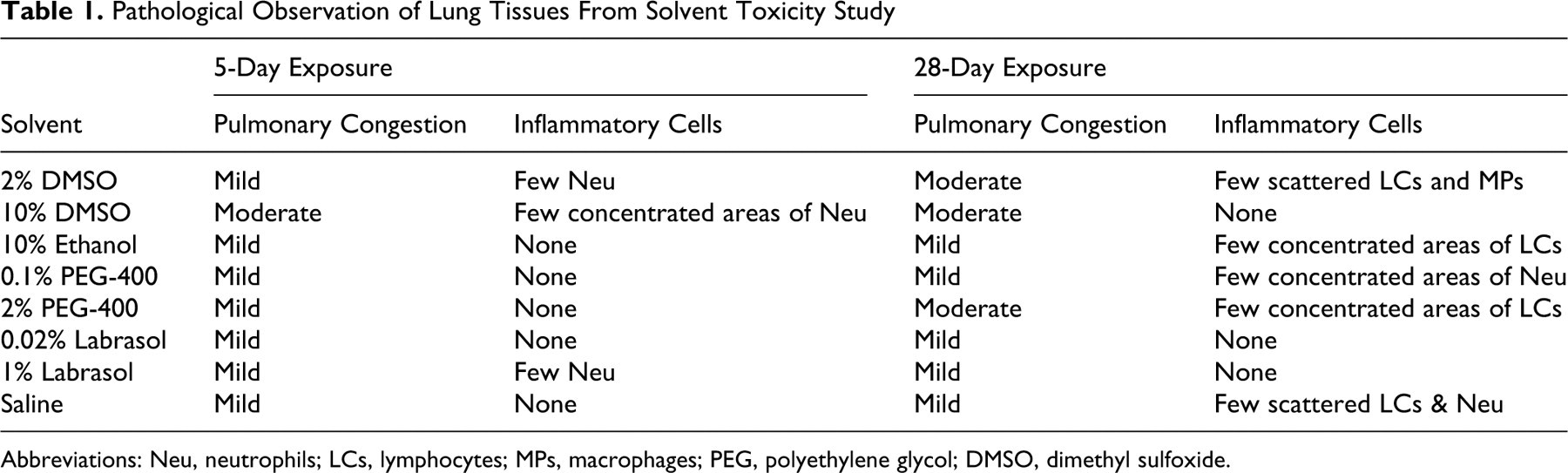
Abbreviations: Neu, neutrophils; LCs, lymphocytes; MPs, macrophages; PEG, polyethylene glycol; DMSO, dimethyl sulfoxide.
Discussion
This study is the first study designed to determine the pulmonary toxicity that may be associated with different solvents by administering them directly to the mouse lung via aspiration. Aspiration seems to be an easier and equally efficient method compared to inhalation of aerosol or intratracheally by microspray to investigate solvent-associated toxicity using controlled, local delivery of solvents to the lungs at exact doses. All solvent-exposure groups showed a similar pattern in body weight changes compared to saline group at all 3 time points (data shown for only 28-day exposure). The body weight loss of mice in saline-treated group was less than 5% of the initial average body weight (17.6 g) and the average body weight in this group at the time of necropsy was not significantly different (16.8 g) compared to the initial body weight (P = .6706). There are some reports of isoflurane enhancing toxicity of chemicals. 35,36 However, animals in these studies were exposed to more severe regimen of isoflurane compared to what we have used in our present study. For example, Rice and colleagues 35 showed that when animals were exposed to up to 0.5% of isoflurane 4 hours/day, 5 days/week for 9 weeks, the body weights of test groups (0.02%-0.5% of isoflurane) varied within 10% of that in air-only exposed group. In another study, when animals were exposed to 0.4% isoflurane 4 hours/day, 5 days/week for 78 weeks, there was a 5% to 8% of body weight loss compared to air-alone group, 36 however, without any gross toxic effects. Recently, Perucho and colleagues 37 demonstrated that isoflurane exposure only affected animal groups with an increased susceptibility to environmental or genetic disease by increasing mortality and apoptosis, but not in the normal healthy mice. In contrast to all these studies, the animals in our study were exposed to 0.5% isoflurane less than 5 minutes per day, 5 days/week for 6 weeks. Although we do not have a sham exposure group in our study, it is difficult to conclude that our relatively mild exposure regimen of isoflurane in the current study would cause or enhance any toxic effect in mice treated with saline alone. Moreover, since the levels of all biomarkers in solvent treatment groups were compared to that in the vehicle (saline) control group, any transient effect of isoflurane will be evident in all groups. In addition, the body weight drop observed in our study seemed to be temporary as we have observed recently (data not shown) that the animals regained their body weight after 8 to 9 weeks of aspiration in an ongoing long-term study (a 16-week chemoprevention bioassay using the same mouse model). With regard to 10% ethanol and 2% PEG-400-treated groups, a significant body weight reduction was observed after 28-day exposure, which indicates that the exposures to these solvents at these doses are less tolerated compared to the other solvents.
Measuring LDH has been applied as a valid method to evaluate membrane damage leading to cytoplasmic component leakage and an increase in LDH level indicates a potential cellular lysis. 2,38 None of the solvents in the current study seemed to induce a cellular lysis in the lung as there was no significant difference in the concentration of LDH in BALF during the entire exposure period. In an in vitro model of rat intestinal mucosa using everted gut sac, Cornaire and colleagues 39 showed that 0.5% labrasol caused a 2-fold increase in LDH release over the control. In contrast, in our study, 1% of labrasol produced nearly identical level of LDH to saline control group, implying that labrasol could be more tolerated in vivo than in vitro environment.
Evaluating total protein level has been routinely used as a toxicity index indicating tissue damage. 40 Total protein concentration measured in the supernatant of BALF after 1 day of exposure showed a temporary increase (16-fold) compared to saline control by 1% labrasol, presumably due to the disruption of the alveolar air spaces and/or bronchial airways. No other solvents caused any significant changes in the total protein concentration at all time points. The current finding on the effect of 1% labrasol on protein concentration is in accordance with the previous report by Lin and colleagues, 33 who demonstrated a significant increase in protein level by 0.1% labrasol using an in vitro diffusion chamber method.
No statistically significant increase in the neutrophils was observed in all solvent groups compared to saline control group during the entire exposure period, indicating that there was no solvent-associated early inflammation in the lung. Increased macrophage numbers implies potential late-stage pulmonary inflammation. 41,42 No solvents tested in this study induced any significant changes in macrophage or lymphocyte numbers at all time points (data not shown).
Change in the level of MPO, a measure of neutrophilic inflammation, was observed by various solvents tested in this study. PEG-400 at 2% caused a significant increase in MPO concentration after 5- and 28-day exposure compared to those of saline control group, indicating a potential neutrophil sequestration upon repeated exposure. Pathological examination of H&E-stained lung tissue section also showed that 2% PEG-400 caused moderate congestion and lymphocyte infiltration following 28 days of exposure. A temporary increase in MPO concentration was also found in the groups exposed to 1% labrasol after 1 day, and 2% DMSO and 10% ethanol after 5 days. Increased level of MPO at early stage of exposure (ie, 1- or 5-day exposure) seemed to decrease to the level of saline control group after longer period of exposure (i.e., 28-day exposure) in most solvent exposure groups tested in this study. However, significantly high level of MPO compared to saline control group persisted until after 28 days of exposure in 2% PEG-400 and 1% labrasol-exposure groups, implying that these solvents could cause acute and subacute inflammation.
The observation in MIP-2 concentration change by the solvents was well correlated with the pathological examination in the respective lung tissues. The level of MIP-2 in most solvent exposure groups was similar to that of saline-treated group at all time points. However, DMSO at 10% showed a temporary increase in MIP-2 concentration in BALF. This pro-inflammatory cytokine was also found to be significantly increased after 28-day treatment with 10% ethanol and 0.1% PEG-400 compared to saline control group, indicating potential early inflammation events by these solvents.
The effects of individual solvent on pulmonary toxicity and inflammation can be summarized as follows; 2% DMSO caused increase of MPO levels after 5 days of exposure and showed infiltration of neutrophils, macrophages, and lymphocytes by the pathological examination; 10% DMSO increased MIP-2 concentration at 5 days and neutrophil infiltration along with moderate congestion; 10% ethanol showed a significant increase in MPO concentration at 5 days and MIP-2 level at 28 days and caused lymphocyte infiltration; 0.1% PEG-400 also increased the concentration MIP-2 at 28 days along with neutrophil infiltration; 2% PEG-400 caused a significant increase in MPO level upon repeated exposure (i.e., 5 and 28-day time points) and induced lymphocyte infiltration; 1% labrasol increased total protein at 1 day and MPO level after 1- and 28-day exposure and also showed a certain level of neutrophil infiltration. In this study, the saline group including some of the solvent groups showed very mild case of pulmonary congestion. Even with the appropriate height (22 cm) of the fixative solution reservoir above the animals, it is still possible that there can be some artifacts in inflation or fixing of some of the lung samples, which can result in mild congestion. To our knowledge, this is the first study for testing the toxicity of different solvents to the normal lung epithelium by aspiration. The data from this study indicated that 0.02% labrasol (equivalent to 0.4 μL labrasol/kg body weight) aspiration resulted in low pulmonary cytotoxicity and inflammation and thus would be suitable to be used as a vehicle for subsequent aspiration studies in a rodent model (e.g., chemoprevention study).
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: (Contract/grant no.: N01-CN-53301) from the National Cancer Institute, Bethesda, Maryland 20892, USA.
