Abstract
Asian sand dust (ASD) events are associated with an increase in pulmonary morbidity and mortality. The number of ASD events has increased rapidly in the east Asian region since 2000. To study the chronic lung toxicity of ASD, saline suspensions of low doses (200 and 400 µg) and high doses (800 and 3,000 µg) of ASD were intratracheally instilled into ICR mice. Animals were sacrificed at 24 hr, 1 week, or 1, 2, or 3 months after instillation. Histopathological examination revealed that ASD induced acute inflammation at 24 hr after instillation. The acute inflammation was transient and subsided at 1 week and 1 month after instillation. At 2 and 3 months after instillation, focal infiltration of lymphocytes with accumulation of epithelioid macrophages, which is a suggestive finding of transformation to granuloma, and granuloma formation were occasionally observed. Aggregation of macrophages containing particles was observed in the pulmonary lymph nodes at 3 months after instillation in high-dose groups. Prolonged inflammatory foci (granuloma) and presence of ASD particles in pulmonary lymph nodes would have a chance to induce immunological modulation leading to adverse health effects in the exposed animals.
Introduction
Asian sand dust (ASD; known as kosa aerosol) originating from the arid deserts of Mongolia and China causes severe air pollution annually in the Asia-Pacific area, including China, Korea, and Japan (Yeo et al. 2010). Epidemiological studies of ASD in humans have shown that ambient ASD particles are associated with an increase in pulmonary (Bell, Levy, and Lin 2008; Chan et al. 2008; Cheng et al. 2008; Chiu et al. 2008; Lai and Cheng 2008) and cardiovascular problems (Bell, Levy, and Lin 2008; Chan et al. 2008; Yang, Cheng, and Chen 2009) and an increase in daily mortality in Korea (Hwang et al. 2005; Kwon et al. 2002; Lee, Son, and Cho 2007) and Taiwan (Chen et al. 2004). The frequency and scale of dust events giving rise to ASD aerosols have increased rapidly in the east Asian region since 2000 (Mori et al. 2003). Because of recent environmental changes such as desertification, humans and animals are at increased risk of frequent exposure to ASD and resultant adverse health effects of ASD on the respiratory system.
Previous studies on the acute pulmonary toxicity caused by intratracheally instilled ASD demonstrated that the mineralogical components of ASD particles, free from chemical and biological pollutants, caused acute inflammatory changes in lung tissues (Naota et al. 2010). It has also been reported that ASD particles enhanced eosinophilic lung inflammation induced by ovalbumin (Hiyoshi et al. 2005; Ichinose Yoshida, Hiyoshi, et al. 2008; Ichinose, Yoshida, Sadakane, et al. 2008).
Previous study (Nishikawa, Quan, and Morita 2000) has determined that the major mineralogical component of ASD is crystalline silica (SiO2). Occupational exposure to crystalline silica is associated with silicosis, lung cancer, pulmonary tuberculosis, and chronic obstructive pulmonary disease (Calvert et al. 2003). In addition, silicosis patients often develop autoimmune diseases (Kumagai et al. 2010; Parks, Conrad, and Cooper 1999). It has been reported that experimental chronic exposure to crystalline silica caused granuloma formation (Huang et al. 2001; Kumar 1989; Langley et al. 2004, 2010; Saffiotti and Stinson 1988) and/or fibrosis (Porter et al. 2004) in rats. In addition, granulomatous inflammation, which consists of epithelioid macrophages containing crystalline silica particles, in lung-associated lymph nodes has been reported in rats exposed to crystalline silica (Friedetzky et al. 1998; Huang et al. 2001; Porter et al. 2002).
Although the chronic pulmonary toxicity of crystalline silica is well known, there has been no in vivo pathological study focusing on chronic lung toxicity induced by ASD particles. The purpose of this study was to describe the chronic lung toxicity caused by the intratracheal instillation of ASD in mice. In addition, the pathogenesis of chronic pulmonary toxicity induced by ASD was discussed.
Materials and Methods
Animals
A total of 361 male ICR mice (5 or 6 weeks old) were obtained from CLEA Japan Inc. (Tokyo, Japan). The animals were fed a CE-2 diet purchased from CLEA Japan and water was given ad libitum. The mouse cages were placed in a conventional room, where the temperature was maintained at about 25°C and the humidity was maintained at 55 to 70%. All animal experiments were performed according to the Tottori University guidelines for animal welfare (http://www.tottori-u.ac.jp/kouhou/kisokusyuu/reiki_honbun/u0950581001.html). Body weight changes were recorded weekly to assess general health status.
Preparation of Particle Samples
CJ-2 particles, simulated ASD, were used as the ASD particles in this study. The CJ-2 particles were obtained from General Science Corporation (Tokyo, Japan). They were collected from the surface soil in the southwest part of the Tengger desert in north-central China, where dust storms occur frequently (Nishikawa, Quan, and Morita 2000). The mean particle diameter was approximately 0.03 mm ± 0.01 mm, according to the manufacturer’s data sheet. The CJ-2 particles contain 28.0% Si, 5.9% Al, 5.3% Ca, 3.0% Fe, 1.7% K, and 1.6% Mg, according to the manufacturer’s data sheet. They were hot-air sterilized at 300°C for 1 hr to remove toxic substances (microbiological substances as well as chemicals including nitrogen oxide and sulfur oxide) adhering to them. The sterilization temperature was determined in accordance with previous studies (Ichinose, Yoshida, Hiyoshi, et al. 2008; Ichinose, Yoshida, Sadakane, et al. 2008; Yanagisawa et al. 2007). It has been reported that neither the chemical composition nor the shape of mineral particles changed even when heated to 700°C (Iwatsuki, Kanda, and Fukazawa 1994; Iwatsuki, Kyotani, and Katsubar 1998).
Preparation of Suspensions
The sterilized particles were suspended in 0.05 mL of sterilized saline solution for instillation. The suspensions were deflocculated via ultrasonic disintegration for 3 min. Four doses (low doses: 200 and 400 µg; and high doses: 800 and 3,000 µg) of the particles were chosen to determine dose effects on lung toxicity in mice. The maximum deposition of particles in the lungs of a single mouse was calculated using tidal volume and breathing rate (Ichinose, Yoshida, Hiyoshi, et al. 2008; Ichinose, Yoshida, Sadakane, et al. 2008). The maximum deposition per week in the Japanese national air quality standard for suspended particulate matter (0.1 mg/m3) is approximately 30 µg. The instillation doses (200, 400, 800, and 3,000 µg) in the present study represent 6.6, 13.2, 26.4, and 99 times that amount, respectively (Naota et al. 2010). Control mice received 0.05 mL of a saline solution.
Study Protocol
A total of 361 mice were randomly divided into five control and ten exposure groups (Table 1).
Number of animals used in this study.
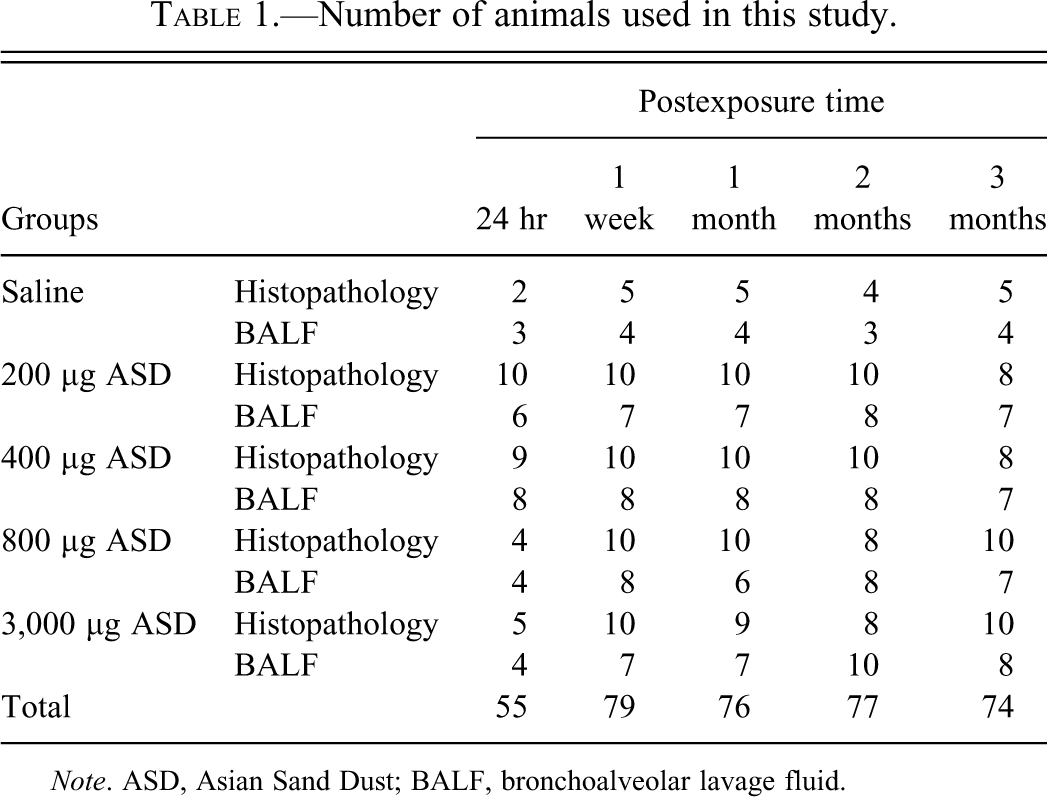
Note. ASD, Asian Sand Dust; BALF, bronchoalveolar lavage fluid.
The mice were anesthetized by an intraperitoneal injection of sodium pentobarbital (5 mg/100 g body weight). The suspensions were agitated immediately before intratracheal instillation, and 0.05 mL of the applicable suspension was instilled with an intratracheal cannula to each mouse. Each intratracheal instillation procedures took 3 sec.
The animals in each group were euthanized by exsanguination under deep anesthesia induced by intraperitoneal injection of sodium pentobarbital at 24 hr, 1 week, or 1, 2, and 3 months after instillation.
Pathological Examination
Two to five mice from each control group and four to ten mice from each group of particle-treated mice were used for pathological examination (Table 1) and immunohistochemistry. On dissection, the trachea was immediately exposed and 0.5 mL of 10% neutral-buffered formalin was instilled gently with the use of syringe via a tracheal cannula at low pressure; formalin injection was carried out within 15 min after death. The whole lungs were removed and fixed by immersion in 10% neutral-buffered formalin for 2 days. Then, the lung lobes were separated, and transverse sections from each lobe were made. These transverse sections and pulmonary lymph nodes were placed into embedding cassettes and fixed by immersion in 10% neutral-buffered formalin for 3 days. Formalin-fixed tissues of lungs and pulmonary lymph nodes were routinely processed, and embedded in paraffin for histopathological and immunohistochemical examination. Sections of approximately 3-µm thick were cut and stained with hematoxylin and eosin. The pathological examination was performed by two pathologists. The area of each granuloma was measured using polygon area measurement function of a microscope digital camera DP21 (Olympus, Tokyo, Japan). The number of granuloma was counted in the lung of each mouse.
Immunohistochemistry
Paraffin-embedded sections of the lungs of mice treated with saline alone and with 3,000 µg of the particles were used for immunohistochemical detection of α-smooth muscle actin, CD3, CD20, and tumor necrosis factor-α (TNF-α). For antigen retrieval, the sections were placed in citrate buffer solution (pH = 5.4) and microwaved. Endogenous peroxidase activity was quenched with 3% H2O2 at room temperature for 30 min. The slides were then blocked with 10% normal goat serum for 5 min with microwave treatment. Thereafter, the sections were reacted with primary antibodies over night at 4°C (anti-α-smooth muscle actin, Dako, Glostrup, Denmark, 1:80 dilution; anti-CD3, Dako, Glostrup, Denmark, 1:80 dilution; anti-CD20, Thermo Fisher Scientific, Fromont, CA, 1:400 dilution; and anti-TNF-α, Monosan, Uden, the Netherlands, 1:20 dilution). The primary antibodies were replaced with phosphate-buffered saline in negative controls. After the reaction with the primary antibodies, the sections were incubated with a peroxidase-labeled polymer conjugated to secondary antimouse and rabbit antibodies (EnVision + kit/HRP (DAB), Dako, Glostrup, Denmark) for 30 min at room temperature. The positive reactions resulted in brown staining with the 3,3′-diaminobenzidine tetrahydrochloride (DAB) as a chromogen, and the sections were counterstained with hematoxylin.
Analysis of Bronchoalveolar Lavage Fluid (BALF)
Three or four mice from each control group and four to ten mice from each group of particle-treated mice were used for examination of BALF (Table 1). After euthanasia, the trachea was cannulated, and the lungs were lavaged with three injections of 1.2 mL of 37°C saline. The lavage fluid was harvested by gentle aspiration. The average volume retrieved was 94.1% (3.38 mL) of the amount instilled (3.60 mL). The three lavage fluids were combined, preserved in a plastic tube, cooled to 4°C, and then centrifuged at 3,000 rpm for 10 min. The supernatants were kept at −80°C until analysis for total protein. Total protein was analyzed with a BCA Protein Assay Kit (Thermo Fisher Scientific, Rockford, IL) according to the manufacturer’s protocol. The cell pellets were used for measuring the total number of cells, cell viability, and differential cell count. Cell viability was assessed by trypan blue dye exclusion using a hemocytometer chamber. Differential cell counts were assessed on cytologic preparations. The slides were stained with Diff-Quick (International Regents Corp., Kobe, Japan). A total of 600 cells were counted using light microscopy.
Statistical Analysis
All data were expressed as the mean ± standard deviation. Statistical analysis of the cellular and biochemical parameters of BALF was determined by a Student’s t test for two-group comparisons. Statistical analysis of the histopathological changes was conducted using Fisher’s protected least significant difference test in analysis of variance (Statview, Abacus Concepts, Inc., Berkeley, CA). For all comparisons, p values less than 5% (p < .05) were considered statistically significant.
Results
Gross Findings of the Lungs
At 3 months after instillation of 3,000 µg ASD particles, the surface of the lungs was rough. In addition, multiple tiny whitish foci were observed in the parenchyma and surface (data not shown). No significant changes were observed in other groups.
Histopathology of the Lungs
Histological sections from control animals instilled with saline showed normal bronchiolar and alveolar architectures at all time points after treatment (Figures 2A, 3A, 4A, 5A, and 6A).
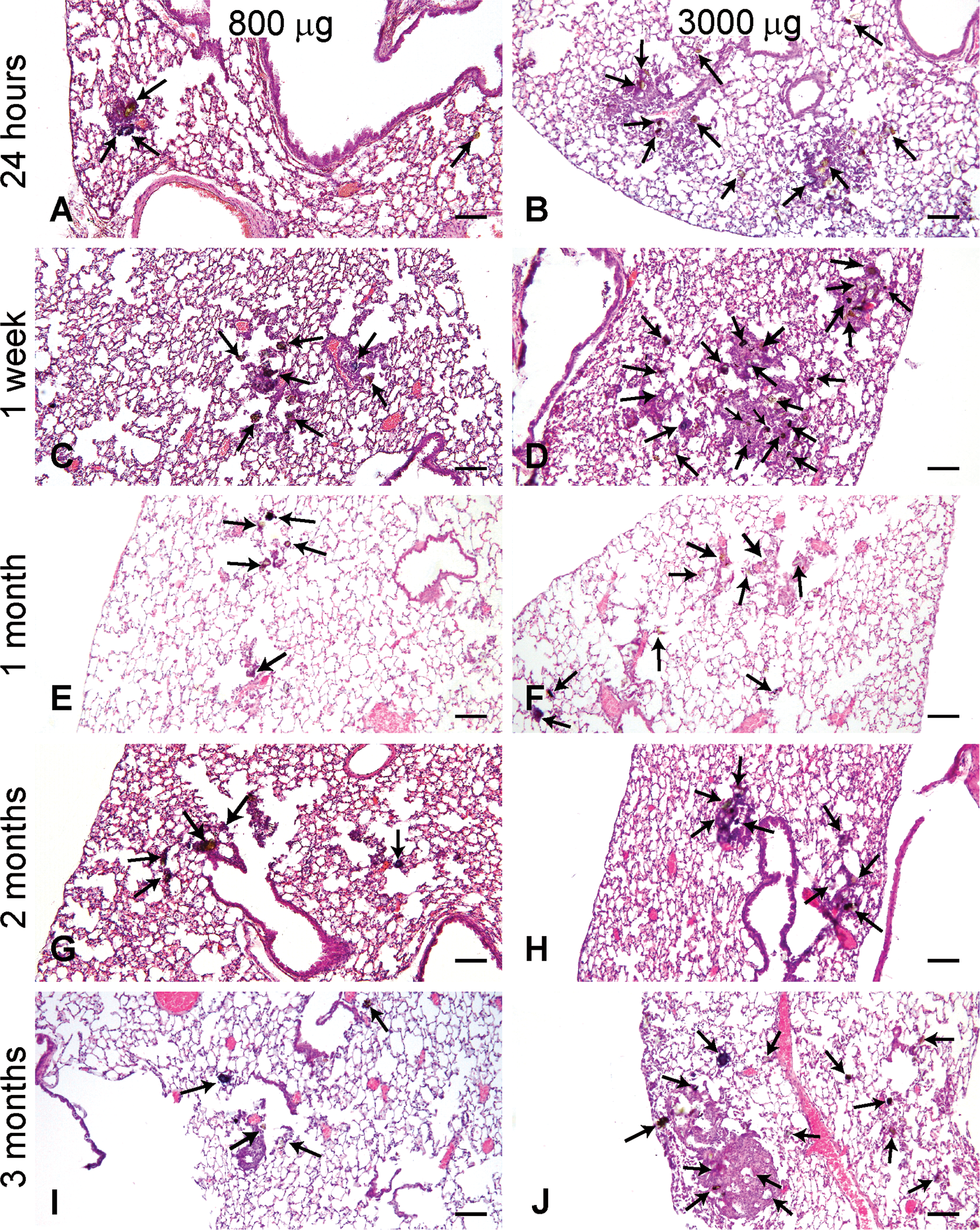
Histological changes of the lungs treated with Asian sand dust (ASD) particles. Representative lung sections taken from mice instilled with 800 µg ASD at (A) 24 hr, (C) 1 week, (E) 1 month, (G) 2 months, and (I) 3 months, and 3,000 µg ASD at (B) 24 hr, (D) 1 week, (F) 1 month, (H) 2 months, and (J) 3 months after instillation. Multifocal accumulation of the particles (arrows) was predominant around the bronchioles, and infiltration of inflammatory cells occurred around the particles. Hematoxylin and eosin. Bars = 80 µm. ×40.
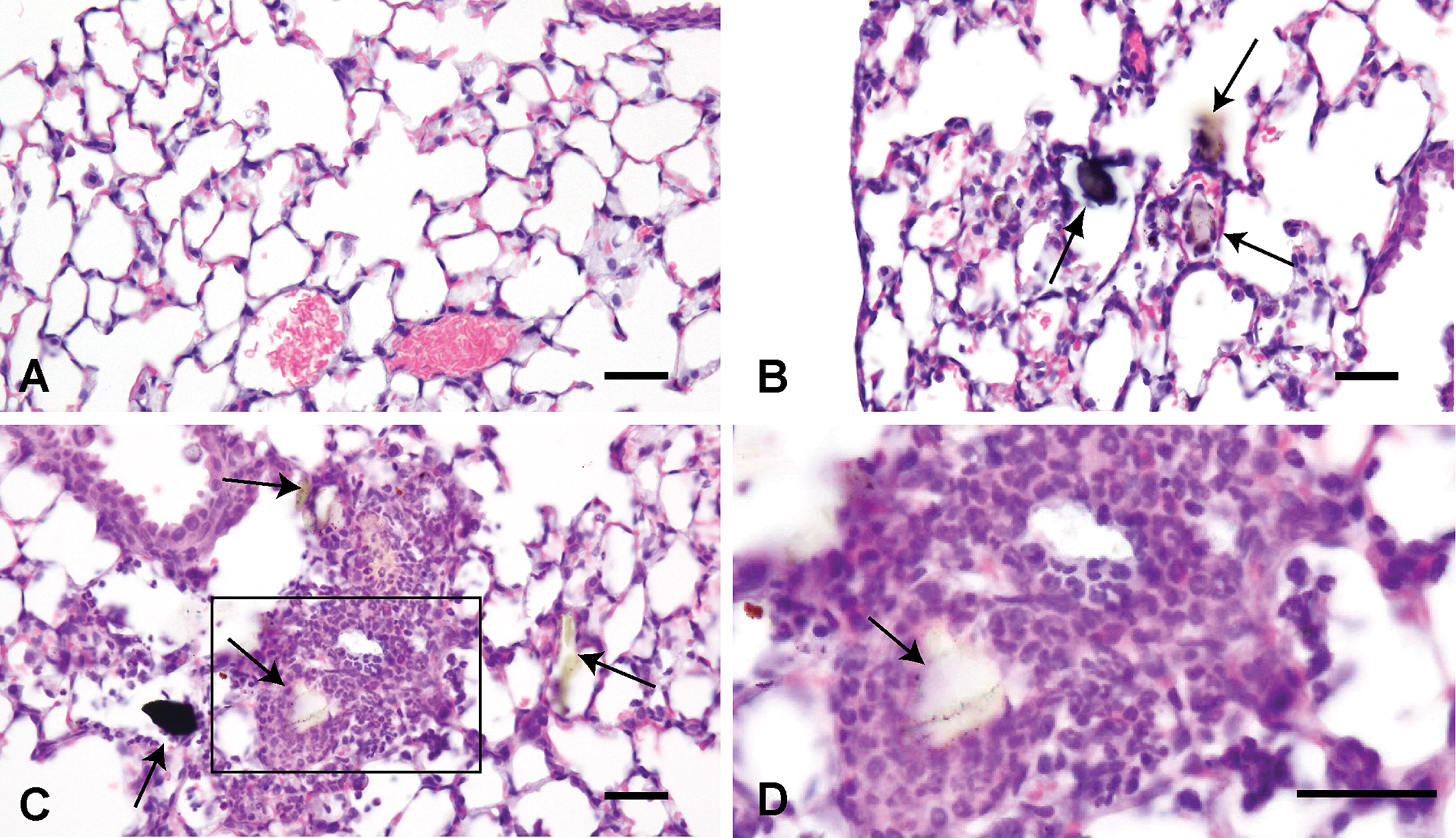
Histological changes of the lungs treated with saline solution (A) and 3,000 µg Asian sand dust (ASD) particles (B, C, D) at 24 hr after instillation. B: Thickening of the alveolar wall. C: Infiltration of inflammatory cells into the alveoli and bronchoalveolar junction. D: High magnification of the oblong box in C. Many neutrophils and some macrophages around the particle. B, C, D: Large particles are indicated by arrows. Hematoxylin and eosin. Bars = 30 µm. ×400 (A–C), ×945. (D).
Histological changes of the lungs treated with saline solution (A) and 3,000 µg (B–D) Asian sand dust (ASD) particles at 1 week after instillation. C: High magnification of the oblong box in B. The inflammatory foci are primarily composed of macrophages (white arrowhead), spindle-shaped cells (black arrowheads), and a small number of neutrophils. Hematoxylin and eosin (A, B, C). D: Spindle-shaped to elongated cells in the inflammatory lesion show positive immunolabelings for α-smooth muscle actin. α-smooth muscle actin immunohistochemistry. B, C, D: Large particles are indicated by black arrows. Bars = 100 µm. ×100. (A, B), 30 µm. ×400. (C, D).
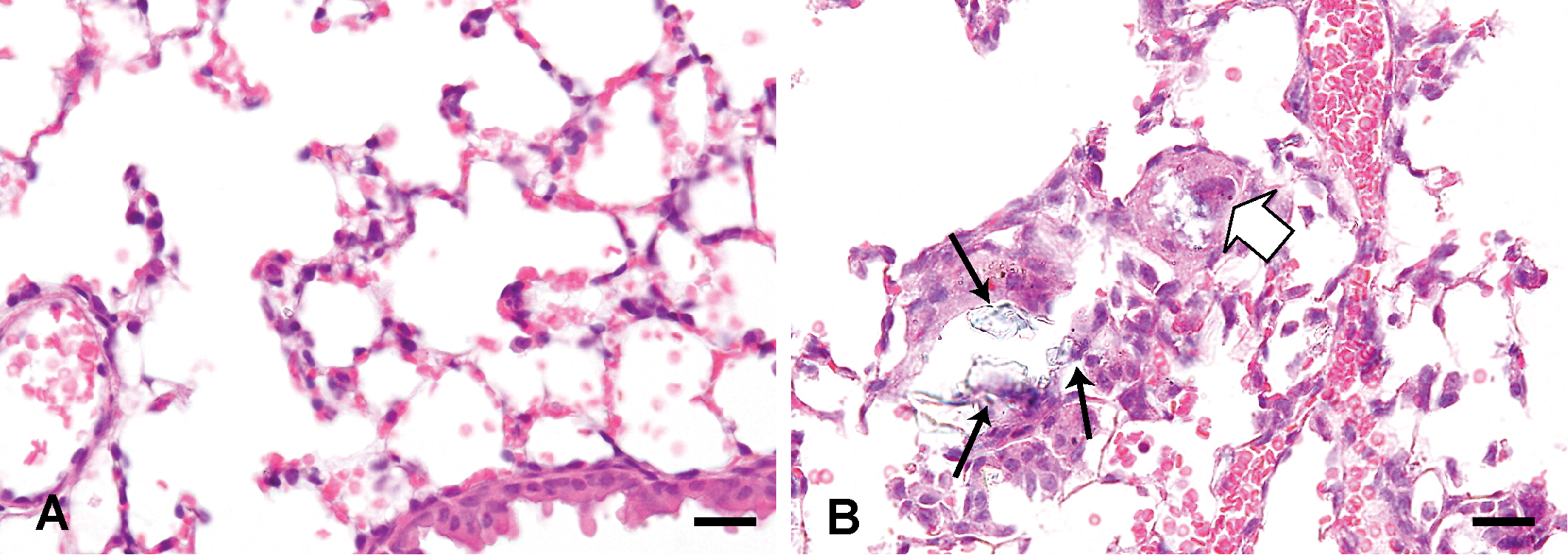
Histological changes of the lungs treated with saline solution (A) and 3,000 µg Asian sand dust (ASD) particles (B) at 1 month after instillation. B: A multinucleated giant cell (white solid arrow) and large particles (black arrows). Hematoxylin and eosin. Bars = 20 µm. ×400. (A, B).
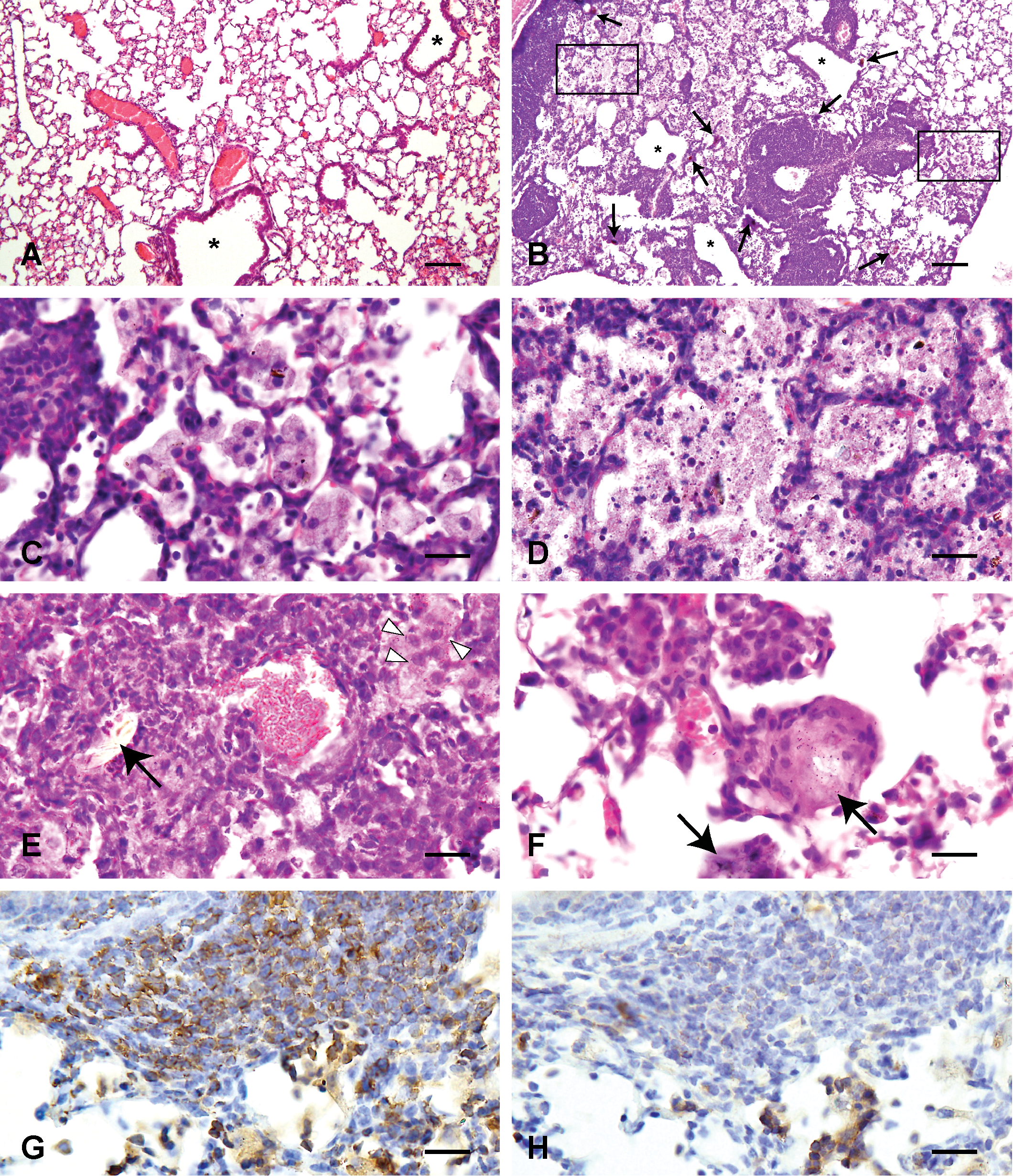
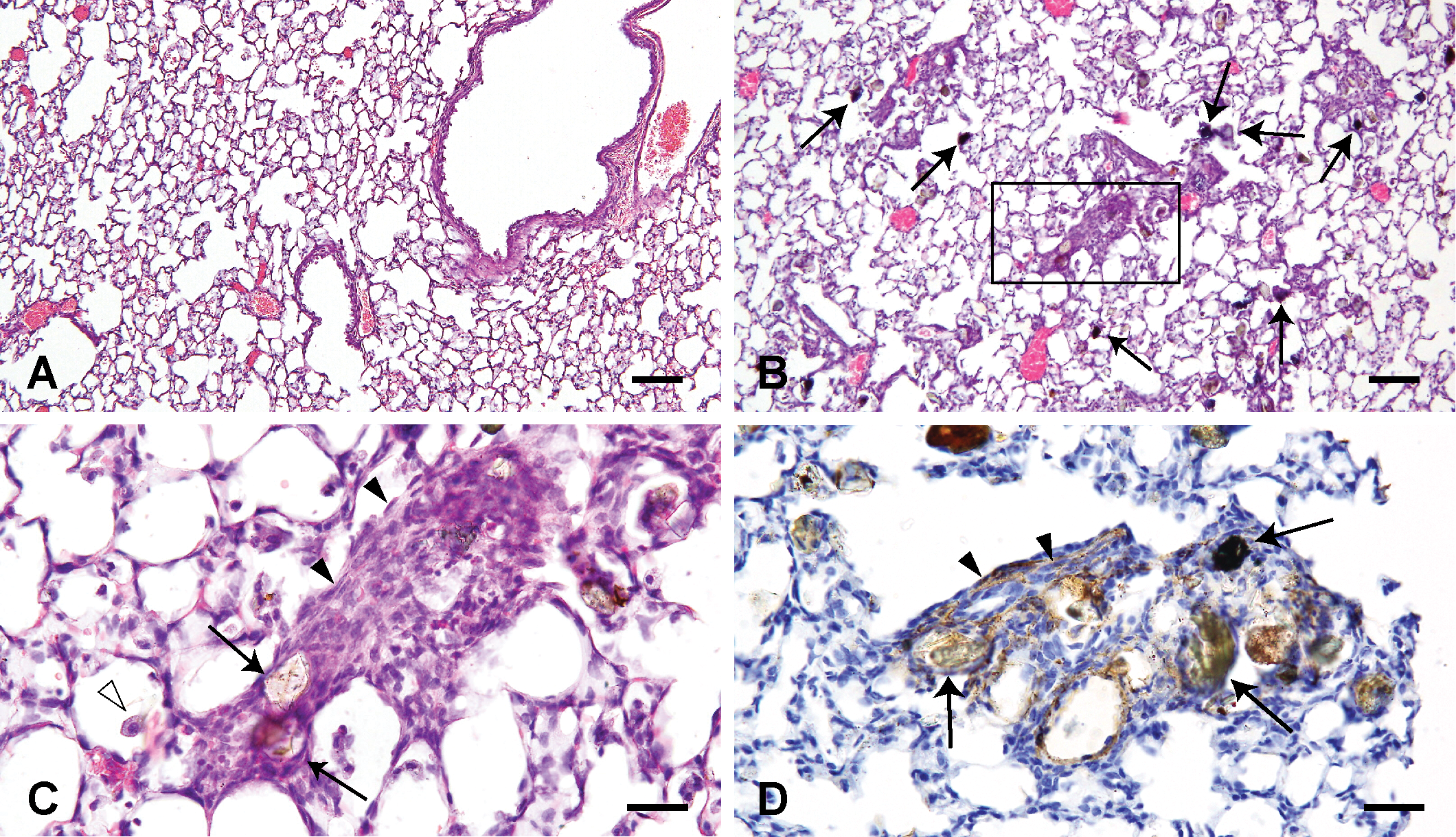
Histological changes of the lungs treated with saline solution (A) and 3,000 µg Asian sand dust (ASD) particles (B–H) at 2 months after instillation. B: Infiltration of lymphocytes around blood vessels and bronchioles (asterisks). C: High magnification of the right oblong box in B. Infiltration of large foamy macrophages. D: High magnification of the left oblong box in B. Necrotic cells with scattered fine particles. E: Neutrophils and focal accumulation of epithelioid macrophages (white arrowheads). F: A granuloma. Large particles are indicated by black arrows (B, D, E, F). Hematoxylin and eosin (A–F). G: CD3-positive T lymphocytes in the lesion. CD3 immunohistochemistry. H: The same area as in G. CD20-positive B lymphocytes in the lesion. CD20 immunohistochemistry. Bars = 100 µm, ×100. (A, B), 20 µm. ×400. (C–H).
In histological sections from animals instilled with the ASD particles, the particles were observed in the lungs at all time points and at both doses. The ASD particles observed in the lung tissues were varied in color, size, and form. The number of coarse particles observable at low magnification (40×) in the lungs of a mouse was consistent throughout the time of observation. Fine particles were observed throughout the lungs. Multifocal accumulation of the particles, predominantly around the bronchioles, was observed at all time points after treatment (Figure 1). Inflammatory changes were observed around the particles (Figure 1). The intensity and frequency of the inflammation tended to increase with the amount of the particles instilled (Figure 1). Colocalization of bronchial epithelial cell changes and particles was observed; desquamation and focal degeneration of bronchial epithelial cells were observed in association with particle attachment at all time points after treatment in high-dose groups. At 1 week after instillation, the size of the inflammatory foci was similar to that observed in lungs at 24 hr after instillation (Figure 1A–D) in high-dose groups. The inflammatory foci observed in lungs at 1 month seemed to become smaller in size than those observed at 1 week after instillation (Figure 1C, D, E, and F) in high-dose groups.
Representative lung lesions at each time point are shown in Figures 2 through 6. At 24 hr after instillation, thickening of the alveolar wall (Figure 2B) and acute inflammatory changes, such as infiltration of many neutrophils and some macrophages into the alveoli and bronchoalveolar junction, were observed (Figure 2C and D).
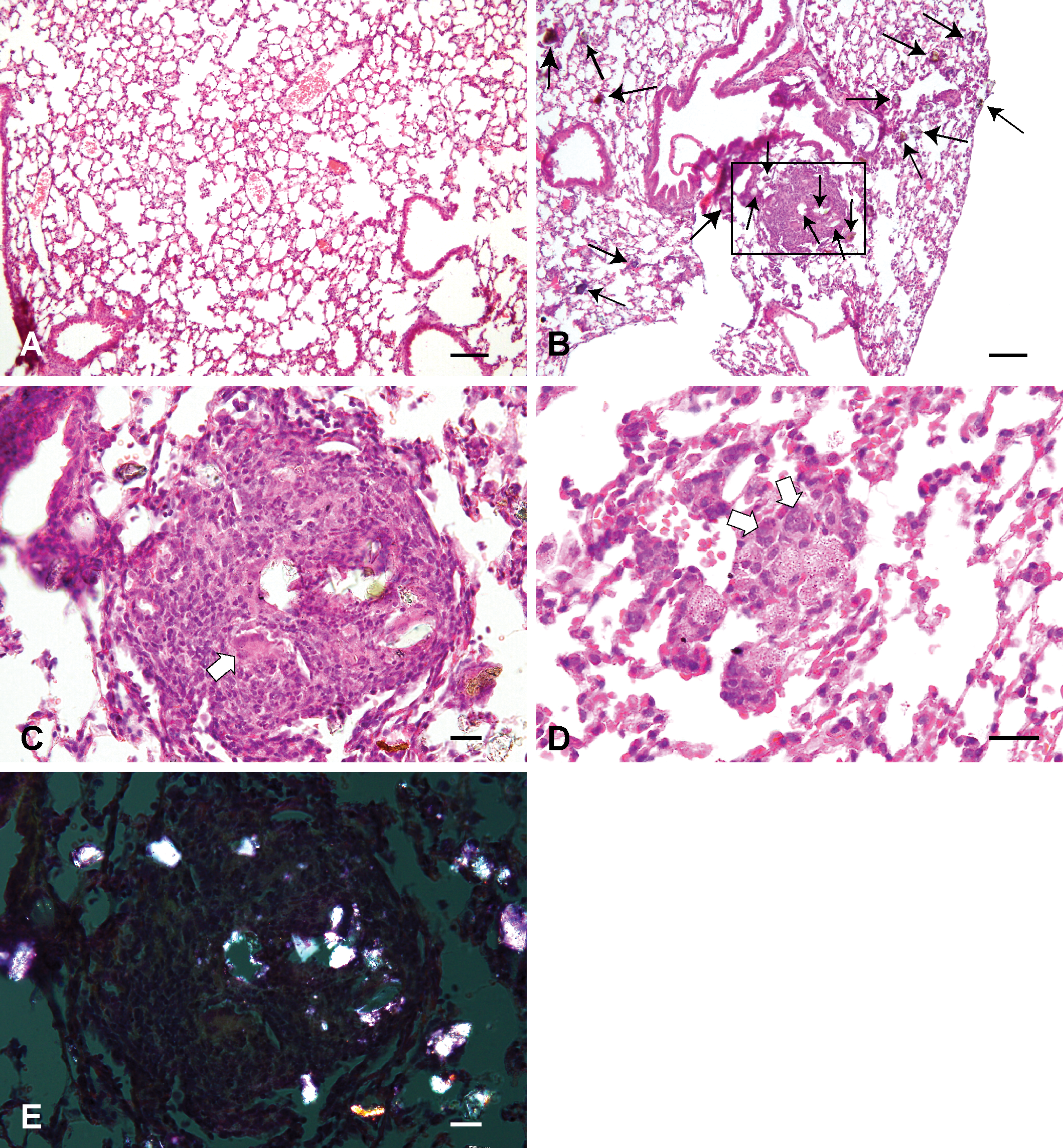
Histological changes of the lungs treated with saline solution (A) and 3,000 µg Asian sand dust (ASD) particles (B–E) at 3 months after instillation. B: Formation of granulomas. The inflammatory reactions around the colored particles (short arrows) were weaker than the ones observed around crystal-like transparent particles (long arrows). C: High magnification of the oblong box in B. The granulomas were composed of epithelioid cells, macrophages, multinucleated giant cells (white solid arrows), neutrophils, and crystal-like transparent particles. D: Focal aggregation of macrophages and multinucleated giant cells without large particles. E: The same area as in C. The particles in the granuloma are visualized as birefringent and white crystals in the polarized light. Hematoxylin and eosin. Bars = 100 µm. 100×. (A, B), 20 µm. 400×. (C–E).
At 1 week after instillation, the inflammatory foci were primarily composed of spindle shaped to elongated cells, macrophages and a small number of neutrophils (Figure 3C) in high-dose groups. Inflammatory foci with a small number of macrophages were also observed in all particle-treated groups.
At 1 month after instillation, the inflammatory foci were composed of macrophages and occasional multinucleated giant cells (Figure 4B). In addition to the inflammatory responses noted in the lungs of the 1 month group, the following changes were noted in the lungs at 2 months after instillation. Infiltration of lymphocytes around blood vessels and bronchioles with large particles were observed in 37.5% of mice treated with 3,000 µg particles (Figure 5B, Table 2). Large foamy macrophages (Figure 5C), necrotic cells with scattered fine particles (Figure 5D), neutrophils (Figure 5E), and focal accumulation of epithelioid macrophages (Figure 5E) were occasionally observed in the lesions. Small granulomas were observed in 10% of mice treated with 400 µg of particles and in 37.5 % of mice treated with 3,000 µg particles (Figure 5F, Table 2).
Quantitative analysis of the histopathological changes at 2 and 3 months after instillation.
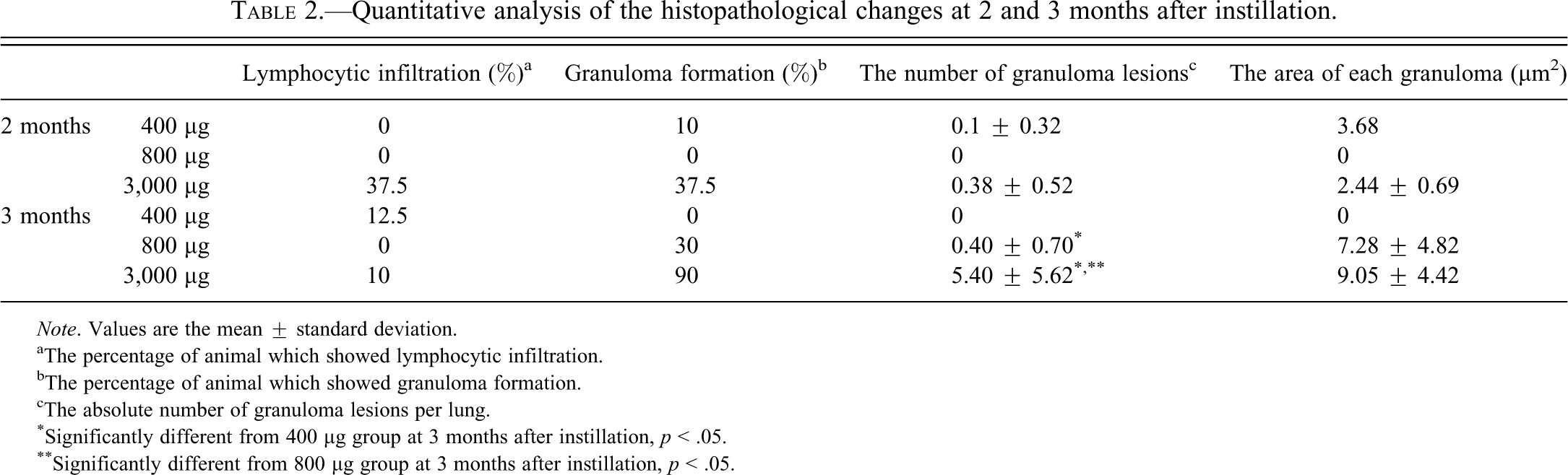
Note. Values are the mean ± standard deviation.
aThe percentage of animal which showed lymphocytic infiltration.
bThe percentage of animal which showed granuloma formation.
cThe absolute number of granuloma lesions per lung.
*Significantly different from 400 µg group at 3 months after instillation, p < .05.
**Significantly different from 800 µg group at 3 months after instillation, p < .05.
At 3 months after instillation, granulomas of various sizes and infiltration of lymphocytes were observed in high-dose groups. The number of granuloma lesions and the area of each granuloma tended to increase along with the amount of particles instilled (Table 2); granuloma formation was observed in 30% of mice treated with 800 µg particles and in 90% of mice treated with 3,000 µg particles (Table 2). Obvious inflammatory reactions associated with the crystal-like transparent particles were seen at all time points. On the other hand, the inflammatory reactions around colored particles were weaker than those observed around the crystal-like transparent particles (Figure 6B).
The granulomas were composed of epithelioid cells, macrophages, multinucleated giant cells, neutrophils, and large ASD particles (20–30 µm) frequently associated with crystal-like transparent particles (Figure 6C). Focal aggregations of macrophages were present without large particles at 2 and 3 months (Figure 6D) after instillation. Crystal-like transparent particles observed frequently in granulomas were visualized as birefringent and white crystals in the polarized light (Figure 6E).
Lymphocytic infiltration was observed in 12.5% of mice treated with 400 µg of particles and in 10% of mice treated with 3,000 µg of particles; the finding was the same histopathological changes as observed at 2 months after instillation (Table 2).
Histopathology of the Pulmonary Lymph Nodes
Histological sections of the pulmonary lymph nodes from control animals instilled with normal saline and low-dose groups showed no significant changes at any points after treatment (data not shown).
At 24 hr and 1 week after instillation, mild infiltration of neutrophils was observed. At 1 month after instillation, macrophages containing fine particles were present in the marginal sinus (Figure 7A and B). At 2 months after instillation, macrophages containing fine particles were frequently observed in the entire area (Figure 7D and E). At 3 months after instillation, focal aggregations of macrophages containing fine particles were seen in one of the seven mice treated with 800 µg particles and two of the seven mice treated with 3,000 µg particles (Figure 7G and H). ASD particles were visualized as birefringent and white crystals in polarized light (Figure 7C, F, and I).
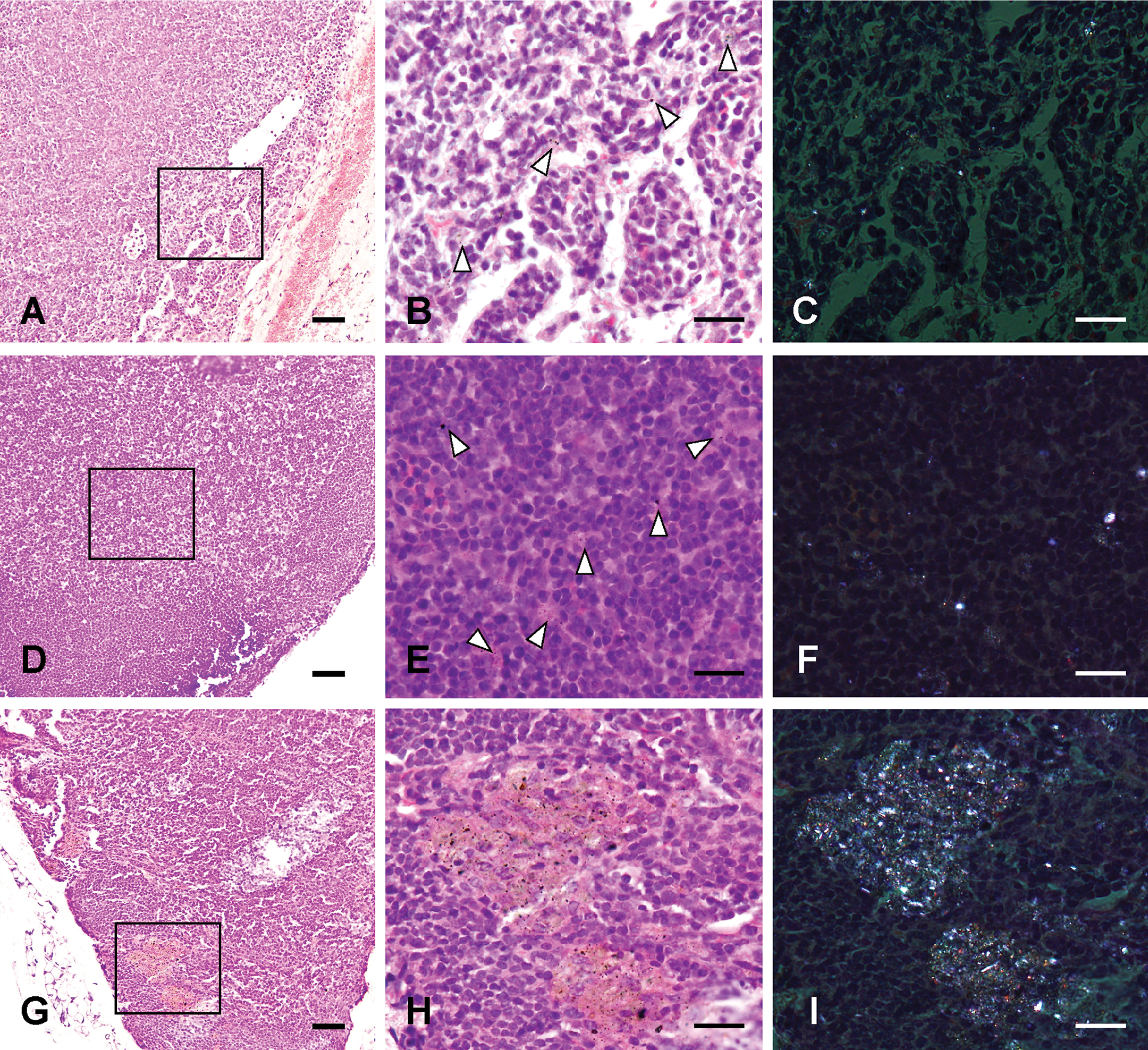
Histological changes of the pulmonary lymph nodes treated with 3,000 µg Asian sand dust (ASD) particles. Representative pulmonary lymph nodes sections taken from mice at 1 month (A–C), 2 months (D–F), and 3 months (G–I) after instillation. B: High magnification of the oblong box in A. Macrophages containing fine particles (white arrowheads) in the marginal sinus. C: The same area as in B. E: High magnification of the oblong box in D. Macrophages containing fine particles were frequently observed in the entire area. F: The same area as in E. G, H: Focal aggregations of macrophages containing fine particles (black solid arrows). H: High magnification of the oblong box in E. I: The same area as in H. C, F, I: Particles are visualized as birefringent and white crystals in polarized light. Hematoxylin and eosin. Bars = 50 µm. ×200. (A, D, G), 20 µm. ×400. (B, C, E, F, H, I).
Immunohistochemistry of the Lungs
α-Smooth muscle actin
Inflammatory foci composed of spindle shaped to elongated cells at 1 week after ASD instillation showed positive immunolabelings for α-smooth muscle actin (Figure 3D). The positive immunolabelings were not observed in the inflammatory foci of the other time points.
CD3 and CD20
Many lymphocytes around blood vessels and bronchioles at 2 months after ASD instillation showed positive immunolabelings for CD3 (T lymphocyte marker; Figure 5G). Some scattered lymphocytes in lesions at 2 months after ASD instillation showed positive immunolabelings for CD20 (B lymphocyte marker; Figure 5H).
TNF-α
The cytoplasm of the inflammatory cells, including macrophages and neutrophils (Figure 8A), and fibroblasts around the bronchioles and peribronchiolar blood vessels in the inflammatory lesions showed positive immunolabelings for TNF-α at 24 hr after ASD instillation. Inflammatory lesions were negative for TNF-α at 1 week (Figure 8B) and 1 month (Figure 8C) after ASD instillation. At 2 and 3 months after ASD instillation, foamy macrophages and granulomatous lesions (Figure 8D and E) showed positive immunolabelings for TNF-α.
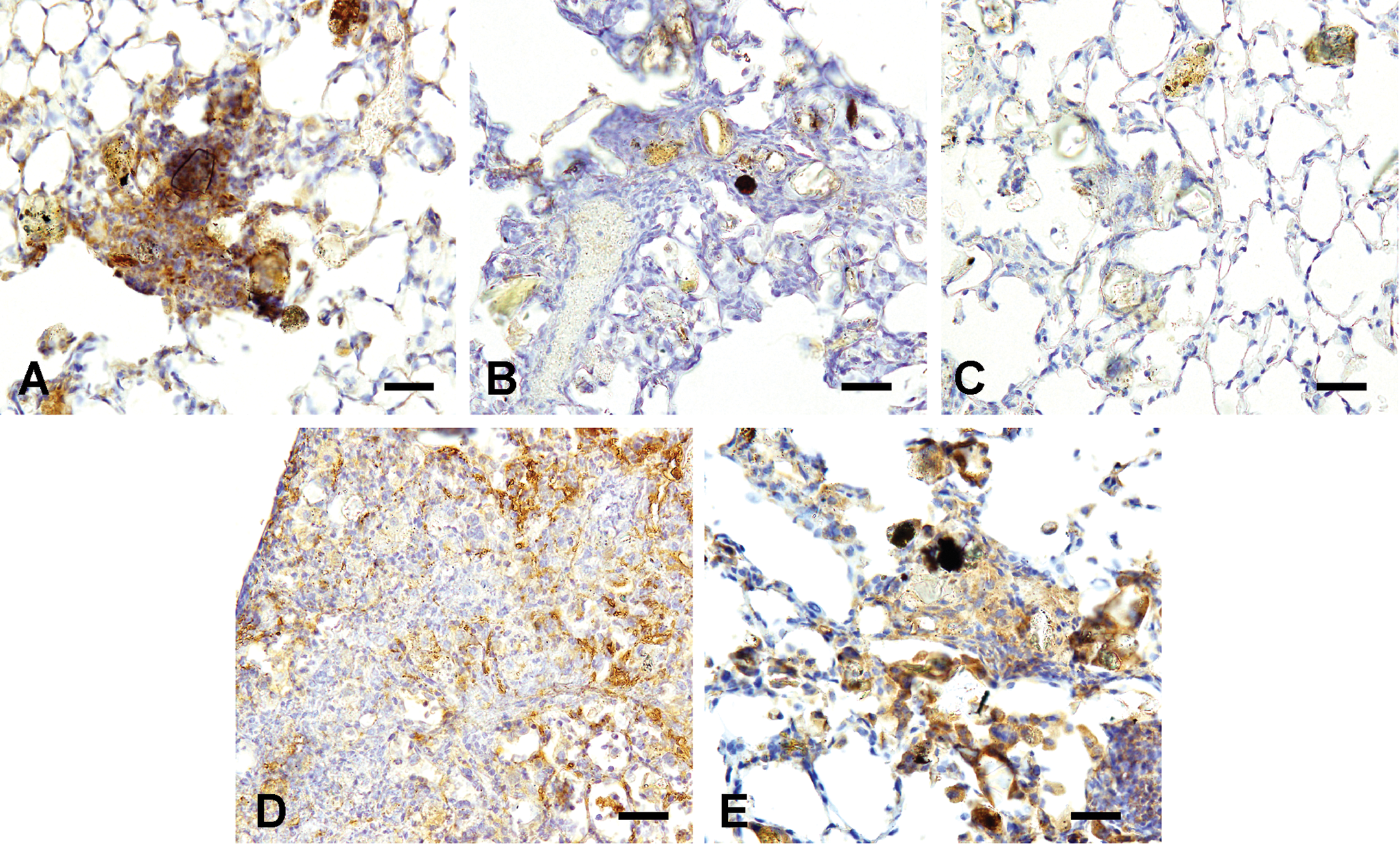
Tumor necrosis factor-α (TNF-α) immunohistochemistry of the lungs treated with 3,000 µg of Asian sand dust (ASD) particles at 24 hr (A), 1 week (B), 1 month (C), 2 months (D), and 3 months (E) after instillation. A: Inflammatory lesions showed positive immunolabelings for TNF-α. B, C: Inflammatory lesions showed negative immunolabeling for TNF-α. D, E: Positive immunolabelings for TNF-α in granulomatous lesions. Bars = 30 µm. ×400.
Cytology and Total Protein Analysis of BALF
Table 3 shows the cellular and biochemical parameters of BALF in the groups of mice treated with ASD particles. Total cell numbers significantly decreased in treated groups at 24 hr after instillation compared with the controls. At 1 month after instillation, total cell numbers significantly increased in treated groups compared with the controls. The percentage of neutrophils tended to increase with the amount of the particles instilled at 24 hr after instillation. At 1 week after instillation, the percentage of neutrophils decreased to the control level. The percentage of multinucleated giant cells tended to increase with the amount of particles instilled at 1, 2, and 3 months after instillation. Cell viability did not show significant changes at any time points after instillation compared with the controls. The total protein concentration significantly increased at the 3,000 µg level at 2 months after instillation compared with the controls.
Cellular and biochemical parameters of bronchoalveolar lavage fluid.
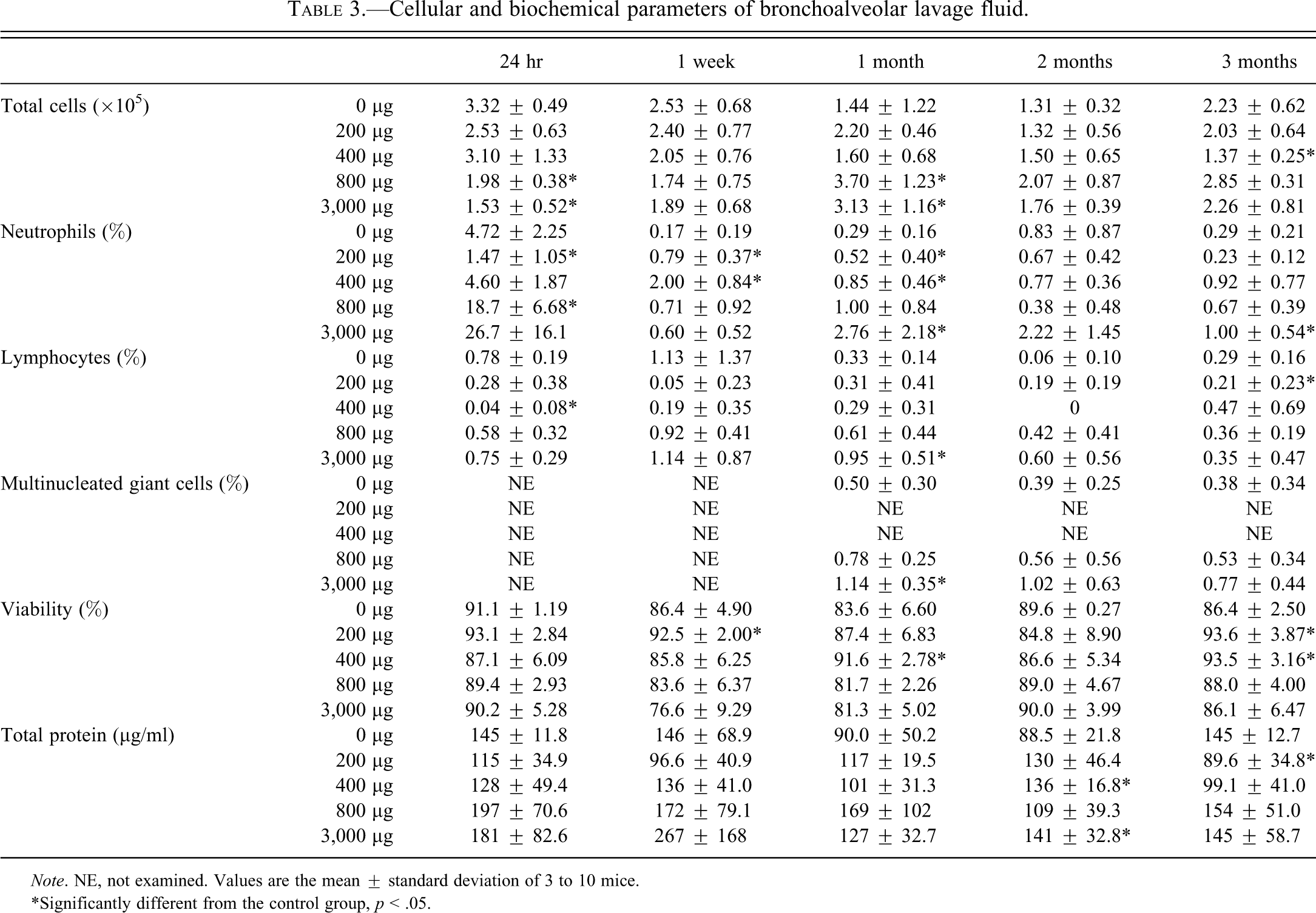
Note. NE, not examined. Values are the mean ± standard deviation of 3 to 10 mice.
*Significantly different from the control group, p < .05.
Discussion
The present study describes chronic pulmonary toxicity induced in mice by the intratracheal instillation of the mineral component of ASD particles, free from chemical and biological substances.
CJ-2 particles were used as ASD in this study. A previous study reported that CJ-2 particles were made as a reference material for ASD aerosol (Nisikawa et al. 2000). CJ-2 particles and ASD particles collected in Japan and China have been shown to be quite similar in terms of size and composition, with comparable concentrations of elements and minerals (Nisikawa et al. 2000). Indeed, similar histopathological changes were obtained in the lungs treated with CJ-2 and ASD collected in Tottori, Japan, in our previous study (Naota et al. 2010).
Inhalation studies are the ideal experimental approach for assessing the effect of ambient particles. However, intratracheal instillation is an easier and less expensive method than inhalation and has been proposed as a reliable route for assessing the pulmonary toxicity of particles in rodents (Warheit et al. 2005; Yokohira et al. 2008). Similar histopathological results have been previously observed for both methods (Warheit et al. 2005). In addition, the deposited dose in lung was more precisely determined in instillation compared with inhalation. Therefore, we chose to use intratracheal administration in these experiments in order to evaluate dose-dependent changes caused by inhaled ASD.
The time courses of pulmonary inflammatory response in mouse lungs exposed to ASD were unique at each time point. The acute inflammatory changes were transient and subsided at 1 week after instillation. These histopathological changes are consistent with the results of analysis of BALF. After instillation, the lung lesions seemed to diminish. However, late exacerbation of inflammation, which is characterized by infiltration of lymphocytes and granuloma formation, was observed at 2 and 3 months after instillation. The late exacerbation of inflammation was obvious in high-dose groups and observed less frequently in low-dose groups. However, it is expected that delayed response may occur in the low-dose groups when observed at more advanced time points.
Regarding granuloma formation, two pathogenesis have been recognized: foreign body granuloma and immune-mediated granuloma. Foreign body granuloma is incited by relatively inert foreign bodies. Typically, foreign body granulomas are formed when materials such as talc, sutures, or other fibers are large enough to preclude phagocytosis by single macrophages and do not incite either an inflammatory or an immune response (Ramzi, Vinay, and Tucker 1999). Epithelioid cells and giant cells are apposed to the surface and encompass the foreign body (Ramzi, Vinay, and Tucker 1999). On the other hand, immune-mediated granuloma is caused by insoluble particles that are capable of induction of a cell-mediated immune response. In these responses, macrophages engulf the foreign materials, process and present some kind of antigen of foreign materials to appropriate T lymphocytes (Ramzi, Vinay, and Tucker 1999). As a result, T lymphocytes are activated.
In this study, the crystal-like transparent particles in granulomas showed birefringence and were white in color under polarized light; this finding is a characteristic of crystalline silica. The finding of granuloma formation associated with crystal-like transparent particles suggests that crystal-like transparent particles (crystalline silica) may induce an inflammatory reaction and may be involved in granuloma formation. In addition, focal infiltration of lymphocytes with aggregation of epithelioid cells was observed at 2 and 3 months after instillation, which is suggestive of transformation of the observed inflammatory changes into granulomas (Figure 9). Similar findings have been reported in a previous inhalation study of crystalline silica in rats (Langley et al. 2004); granulomatous changes were detected in lungs at 17 weeks after inhalation subsequent to the infiltration of large numbers of lymphocytes and neutrophils into the lung at 10 weeks after inhalation. Furthermore, in the present study, granulomas were first observed following translocation of macrophages containing particles in pulmonary lymph nodes at 1 month after instillation (Figure 9). Translocation of macrophages containing particles (crystalline silica) into pulmonary lymph nodes may indicate association with activation of the immune system by instilled ASD particles. These findings suggest that the granulomas formed by instillation of ASD particles in this study should be classified as immune-mediated granulomas.
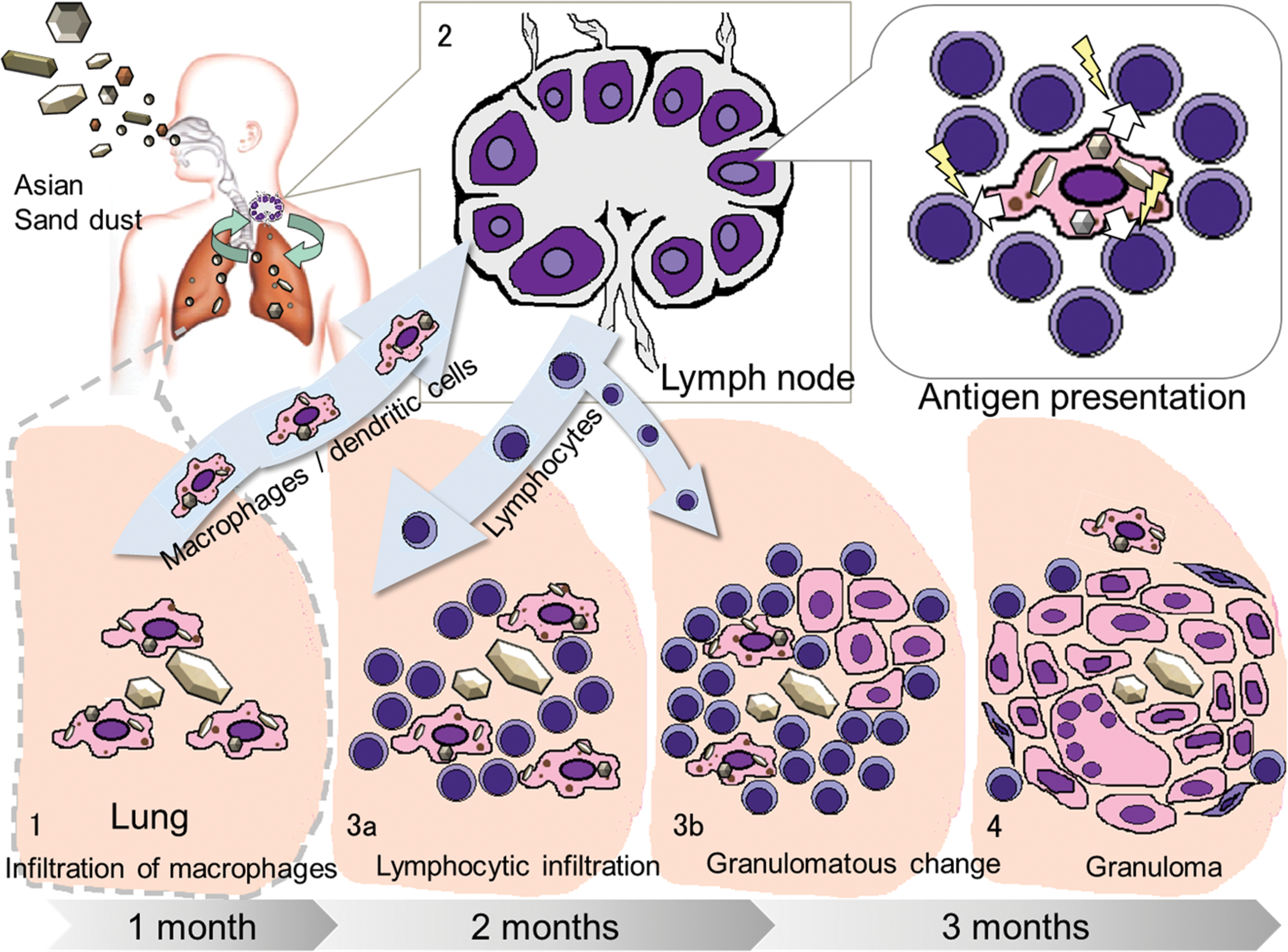
Schematic diagram of a hypothesis for the granuloma formation after Asian sand dust (ASD) instillation. Inflammatory lesions may develop from (1) to (4) as described below: (1) small inflammatory foci in lungs at 1 month, (2) translocation of macrophages containing fine particles into the pulmonary lymph nodes at 1 month, (3) focal infiltration of lymphocytes with accumulation of epithelioid macrophages at 2 and 3 months, and (4) granuloma formation at 2 and 3 months after instillation.
A previous study (Nishikawa, Quan, and Morita 2000) has determined that the major mineralogical component of ASD is silica (SiO2), which is derived mainly from feldspar and quartz (crystalline silica). Occupational exposure to crystalline silica leads to silicosis, which is characterized by a progressive granulomatous and fibrogenic response in the lung (Ishihara et al. 2001). Another well-known health outcome associated with silica exposure is an increase in the incidence of autoimmune disorders (Maeda et al. 2010; Parks, Conrad, and Cooper 1999) including rheumatoid arthritis (Rosenman, Moore-Fuller, and Reilly 1999), scleroderma (Martin et al. 1999), systemic lupus erythematosus (Parks and Cooper 2006), and antineutrophilic cytoplasmic antibody–related nephritis (Tervaert, Stegeman, and Kallenberg 1998). Experimental exposure of crystalline silica is known to cause granuloma formation (Huang et al. 2001; Kumar 1989; Langley et al. 2004, 2010; Saffiotti and Stinson 1988) and/or fibrosis (Porter et al. 2004) in the lungs of rats. In a previous chronic experiment, granulomatous inflammation consisting of epithelioid macrophages containing crystalline silica particles in the lung-associated lymph nodes was demonstrated in rats at 6 weeks (Friedetzky et al. 1998), 76 days (Porter et al. 2004), and 5 months (Huang et al. 2001) and in mice at 1 week after inhalation of crystalline silica. Friedetzky et al. (1998) also demonstrated that germinal centers and T-cell regions disappeared in thoracic lymph nodes at 6 weeks after inhalation of silica. Friedetzky et al. (1998) concluded that marked disorganization of thoracic lymph node structures such as granuloma formation and disappearance of B and T lymphocytes may contribute to the immune abnormalities in silicosis patients. Activation of T lymphocytes has been reported in silicosis patients (Otsuki et al. 2011; Wu et al. 2006). Activation of T lymphocytes in silicosis patients may result from recurrent exposure to crystalline silica or retention of crystalline silica in the body (Kumagai et al. 2010; Wu et al. 2006). Immunohistochemistry with lymphocytic markers showed that infiltrated lymphocytes observed in the lesions at 2 month after instillation consisted of a mixed population of many CD3-positive T lymphocytes and some CD20-positive B lymphocytes. Kumar (1989) has also reported infiltration of mixed population of T and B lymphocytes in the lungs of mice treated with crystalline silica. Further investigation is required as to the association of T lymphocytes infiltration in the development of granulomatous lesions.
Previous study has suggested that crystalline silica particles, which were transported into pulmonary lymph nodes, obtain the opportunity to react with circulating immunocompetent cells (Kumagai et al. 2010). The findings of granuloma formation in lungs and aggregation of macrophages containing particles in pulmonary lymph nodes in the present study are similar to the findings induced by crystalline silica in experimental animals. Translocation of particles containing macrophages and retention of the particles in pulmonary lymph nodes suggest that ASD particles may continuously stimulate the immune system. Attention has to be paid on the possible immunological modulation by ASD in exposed humans.
In this study, positive immunolabelings for α-smooth muscle actin were observed in inflammatory foci at 1 week after instillation. Myofibroblasts, which are characterized by de novo expression of α-smooth muscle actin, become activated to migrate into damaged tissue and to synthesize extracellular matrix components (Hinz 2007) by cytokines locally released from inflammatory and resident cells (Werner and Grose 2003). When normal healing occurs, resolution of myofibroblast apoptosis would terminate progression of fibrosis (Hinz 2007). In this study, in accordance with the diminution of lung lesions at 1 month after instillation, inflammatory foci showing positive immunolabelings for α-smooth muscle actin were not observed at 1 month after instillation or later. Although findings of apoptosis of myofibroblasts were not observed at the examined time points, apoptosis was expected to have occurred during 1 week to 1 month after instillation.
TNF-α induces the recruitment of inflammatory cells. In addition, TNF-α has been associated with proliferation of fibroblasts and secretion of extracellular matrix components in silicosis and coal workers’ pneumoconiosis (Vanhée et al. 1995). In the present study, an increased number of α-smooth muscle actin-positive fibroblasts was observed at 1 week after instillation following observation of acute inflammatory foci, which showed positive immunolabelings for TNF-α at 24 hr after instillation. Experimental silicosis in rats showed predominant granulomatous nodules (Saffiotti and Stinson 1988) rather than fibrotic nodules. Lung lesions in animals are generally less old (immature), and they rarely become as densely fibrotic as those seen in humans (Green, Vallyathan, and Hahn 2007). Although deposition of collagen fibers was not detected in the sections stained with Masson’s trichrome stain throughout the time course of the experiment (data not shown), positive immunolabelings for TNF-α in granulomatous lesions at 2 and 3 months after instillation in the present study suggest potential fibrosis, which is observed in silicosis patients (Green, Vallyathan, and Hahn 2007), in the ASD-exposed animals.
In conclusion, this study demonstrated that mineralogical components of ASD particles, free from chemical and biological pollutants, caused granulomas with multinucleated giant cells in lung tissues at 3 months after intratracheal instillation. Thus, cautions regarding ASD are required from the viewpoint of public health, especially for humans and animals who live in the area with severe ASD events. Intratracheal instillation of a high dose of ASD was the exposure method used in this study; this method is different from atmospheric exposure of humans to ASD in the natural environment. Further investigation on the pulmonary toxicity of ASD in a similar natural environment should be conducted using an inhalation method with low concentrations of ASD particles.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
Abbreviations
Acknowledgments
This work was supported in part by the Global COE Program (Global Center of Excellence for Dryland Science), MEXT, Japan and Research Fellowships of the Japan Society for the Promotion of Science for Young Scientists. The authors thank K. Rattanapinyopituk and Ms. Y. Yamamoto for their technical assistance.
