Abstract
The authors performed a pathological examination of a 5-year-old female laboratory Japanese monkey who developed cortical blindness and epileptic seizures. Generalized, tonic-clonic seizures started to occur during behavioral training to get the animal to enter a carrying cage for future psychological experiments. Blindness was suspected because of a lack of approaching behavior toward foods such as fruits. Although the monkey was extensively treated with anticonvulsants, the clinical signs did not improve. An increased serum creatine phosphokinase (CPK) level and bilateral occipital brain atrophy were detected. Histopathologically, a severe degree of cerebromalacia was detected bilaterally in the occipital lobe, and necrosis and gliosis were seen mainly in the temporal lobe. Focal inflammation was found in the meninges. No other changes were observed in other nervous tissues or organs, and no signs of a parasitic or viral infection were found in the systemic organs. Spontaneously occurring lesions in the central nervous system have been rarely reported in laboratory monkeys. In the present case, the cause of cerebromalacia could not be confirmed, but the relationship between symptoms such as abnormal vision and the presence of brain lesions was distinct. The authors believe that this case is a valuable historical control case for the laboratory Japanese macaque.
Introduction
In Japan, the Japanese macaque, Macaca fuscata, is preferred for nonhuman primate experiments, particularly in the field of neuroscience, because of its high level of intelligence, gentle nature, and relatively homogeneous genetic background (Isa et al. 2009). Additionally, there is abundant data on the social behavior of wild Japanese monkeys (Isa et al. 2009). Although the large amounts of anatomical and neurophysiological data gathered from studies of the Japanese macaque have greatly contributed to the understanding of higher brain function, historical control data regarding pathological lesions have not been widely available. In this article, we describe a case of cerebromalacia in a female Japanese monkey with spontaneously occurring cortical blindness and epileptic seizures, and we discuss the relationship between the symptoms and brain lesions.
Materials and Methods
Animal Information and Clinical Signs
Our laboratory at Kansai Medical University received a 4-year-old female Japanese macaque (Macaca fuscata) from the National Bioresource Project (NBRP) “Nihonzaru” of the Ministry of Education, Culture, Sports, Science and Technology, Japan (http://www.nbrp.jp/index.jsp) (Isa et al. 2009). Before arriving at our laboratory, a standard medical examination was performed by the breeder. The monkey was found to be in good health, with normal body weight and food consumption, and several routine hematological and serum biochemical parameters were within the normal range. The results from fecal parasite detection, the tuberculin reaction, and serological tests for Shiga bacillus, Salmonella, and herpes B virus were negative. The monkey was raised in our laboratory for about 1 year without any experimental procedures, such as surgery or treatment, but she did receive behavioral training to enter a carrying cage for future psychological experiments. The monkey was fed a PS diet (Oriental Yeast Co., Ltd, Kyoto, Japan) and had free access to water. She was singly housed in a stainless steel cage in a controlled environment of 22 ± 2°C, 60 ± 10% humidity, and a 12-hr light-dark cycle. Adequate consideration was taken to avoid unnecessary anxiety or pain to the animal. Animal care and procedures were conducted in accordance with the ethical guidelines of Kansai Medical University.
At the age of 5 years, the monkey suddenly developed neurological symptoms, including epileptic seizures. First, recurrent epileptic seizures occurred mainly on the left side of the face, the left arm, and the left foot. In the absence of a seizure, the animal ingested food and water normally. Second, blindness was suspected because she had no reaction or approaching behavior toward foods such as fruits. As treatment for the seizures, she received intramuscular injections of diazepam (1.5 to 2.5 mg, Cercine injection®, Takeda, Osaka, Japan) and phenobarbital (20 to 30 mg, Phenobal®, Daiichi-Sankyo, Tokyo, Japan) for 2 weeks. Despite extensive treatment with these anticonvulsants, their effect was transient and severe seizures recurred.
A poor prognosis based on severe clinical signs prompted us to euthanize the monkey at 1.5 months after the onset of clinical abnormalities. Routine biochemical and hematological analyses of serum were conducted before the animal was euthanized. The animal was deeply anesthetized with pentobarbital sodium and then euthanized by exsanguination from the cervical aorta. A complete necropsy was performed, during which principal organs were weighed.
Pathological Examination
Specimens of all systemic organs were fixed in 10% buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E) for routine histopathological examination. We examined tissues of the central and peripheral nervous systems, including the bilateral cerebrum (frontal, parietal, and temporal lobes and thalamus), bilateral cerebellum, medulla oblongata, spinal cord (C1, C3, C7, T1, T3, T5, T7, T9, T10, L1, L3, L5, L7, and S1), sciatic nerve, and bilateral eyes and optic nerves. According to the reference by Wu, Dubach, and Robertson (2000), coronal sections of cerebrum were made anterior (+) and posterior (–) to the anterior commissure (ac) at ac +3 mm, 0 mm, –4 mm, –10 mm, –15 mm, –19 mm, –25 mm, and –30 mm. Two veterinary and human pathologists certified by the Japanese College of Veterinary Pathology and the Japanese Society of Pathology (K.Y. and A.T., respectively) performed the histopathological and morphometrical evaluations. To indicate the localization of neuronal damage, necrosis, demyelination, glial reaction, and inflammation, the damaged area was circled by a red line on each brain section. Additional serial sections of brain tissue were stained with periodic acid-Schiff stain (PAS), phosphotungstic acid hematoxylin stain (PTAH), polyclonal antibody to glial fibrillary acidic protein (1:500 dilution; GFAP, Dako, Glostrup, Denmark) as an astroglial immunomarker, or with monoclonal antibody to CD68 (1:50 dilution; clone KP-1, Dako) as a microglial immunomarker. A labeled streptavidin-biotin staining kit (Dako) and antigen retrieval by pressure-cooker heating (Pascal, Dako) were used for immunostaining.
Results
The blood chemical analysis revealed increased amounts of serum creatine phosphokinase (CPK: 2,050 IU/L), aspartate amino transferase (AST: 87 IU/L), lactate dehydrogenase (LDH: 642 IU/L), and potassium (K: 5.5 mEq/L), compared with the control ranges reported for Japanese monkeys (CPK: 215 ± 279 IU/L, AST: 27 ±10 IU/L, LDH: 232 ± 88 IU/L, K: 3.9 ± 0.4 mEq/L; see Historical Control Data of Japanese Monkeys n.d., http://www.nips.ac.jp/tech/ipr/koike/NihonMonky_data_data.pdf). In contrast, the serum albumin level (ALB: 2.9 g/dl), the albumin-to-globulin ratio (A/G: 0.9), and the total cholesterol level (T-CHO: 66 mg/dl) were lower than the published control ranges (ALB: 4.2 ± 0.5 g/dl, A/G: 1.6 ± 0.4, T-CHO: 143 ± 27 mg/dl). A hematological examination revealed no abnormal changes in blood counts compared with historical control levels in Japanese monkeys.
Bilateral occipital brain atrophy (Figures 1A and 1B) and thymic involution were macroscopically detected. The remaining cerebrum showed edema at the surface. Histopathologically, a severe degree of cerebromalacia was detected in both occipital lobes (Figures 2 and 3). Disorganization of brain tissue due to necrosis, gliosis, and vascular proliferation was seen in the same area. Proliferation of GFAP-positive astroglia and infiltration of CD68-positive microglia was confirmed (Figure 4). In the temporal, parietal, and frontal lobes, focal demyelination, gliosis, microglial infiltration, and focal inflammation of mononuclear cells in the meninges were also seen (Figure 3). Neuronal lesions were more extensive on the right side of the frontal, parietal, and temporal lobes of the cerebral cortex, compared with the left side (Figure 2). No vascular lesions, such as thrombosis or degeneration, were observed in the brain, even in tissues stained with PAS or PTAH. Neither parasites nor viral inclusions were found in the nervous system. Moreover, no other neuronal damage was detected in other nervous tissues, including the retina and optic nerve (Figure 5 ).
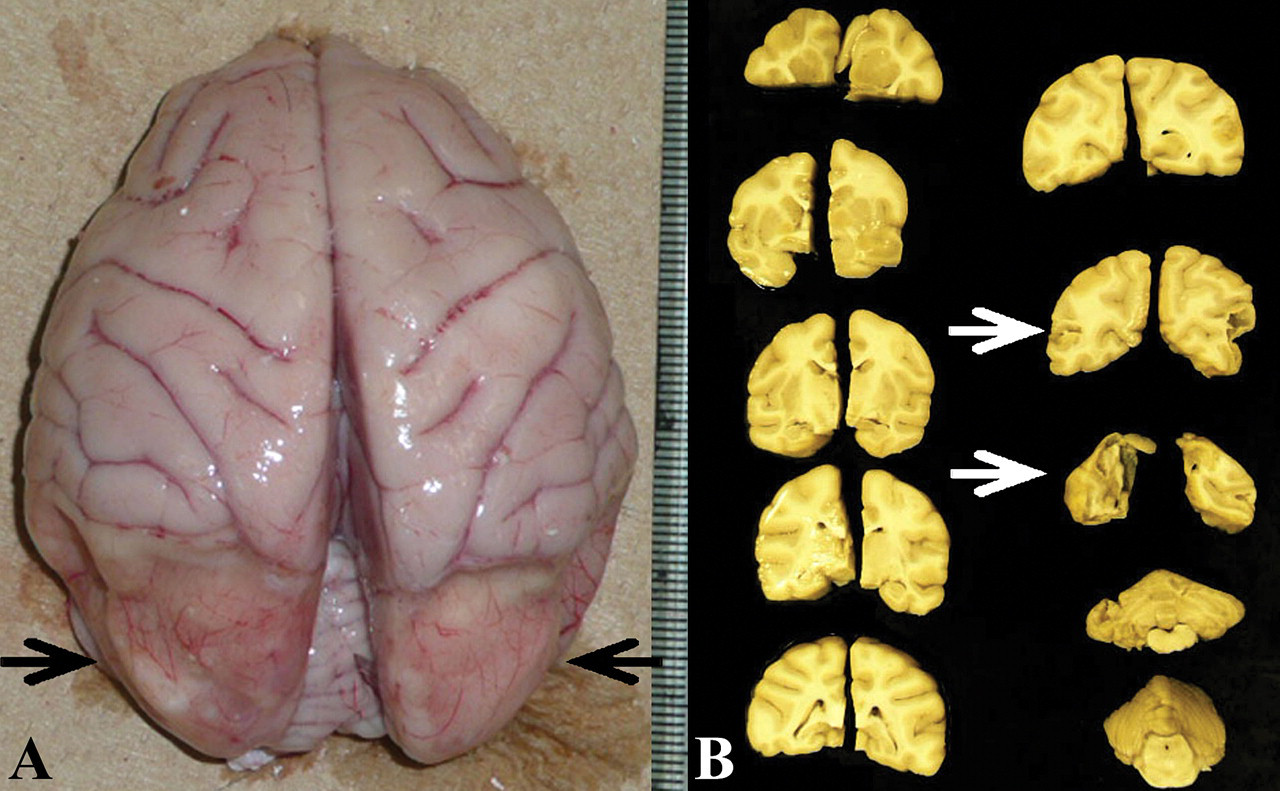
Macroscopic brain lesions. A: The impression (atrophy) can be seen in the bilateral occipital lobe (arrows). The remaining cerebrum is edematous. B: Macroscopic lesions of formalin-fixed brain. The extensive damage is indicated by arrows.
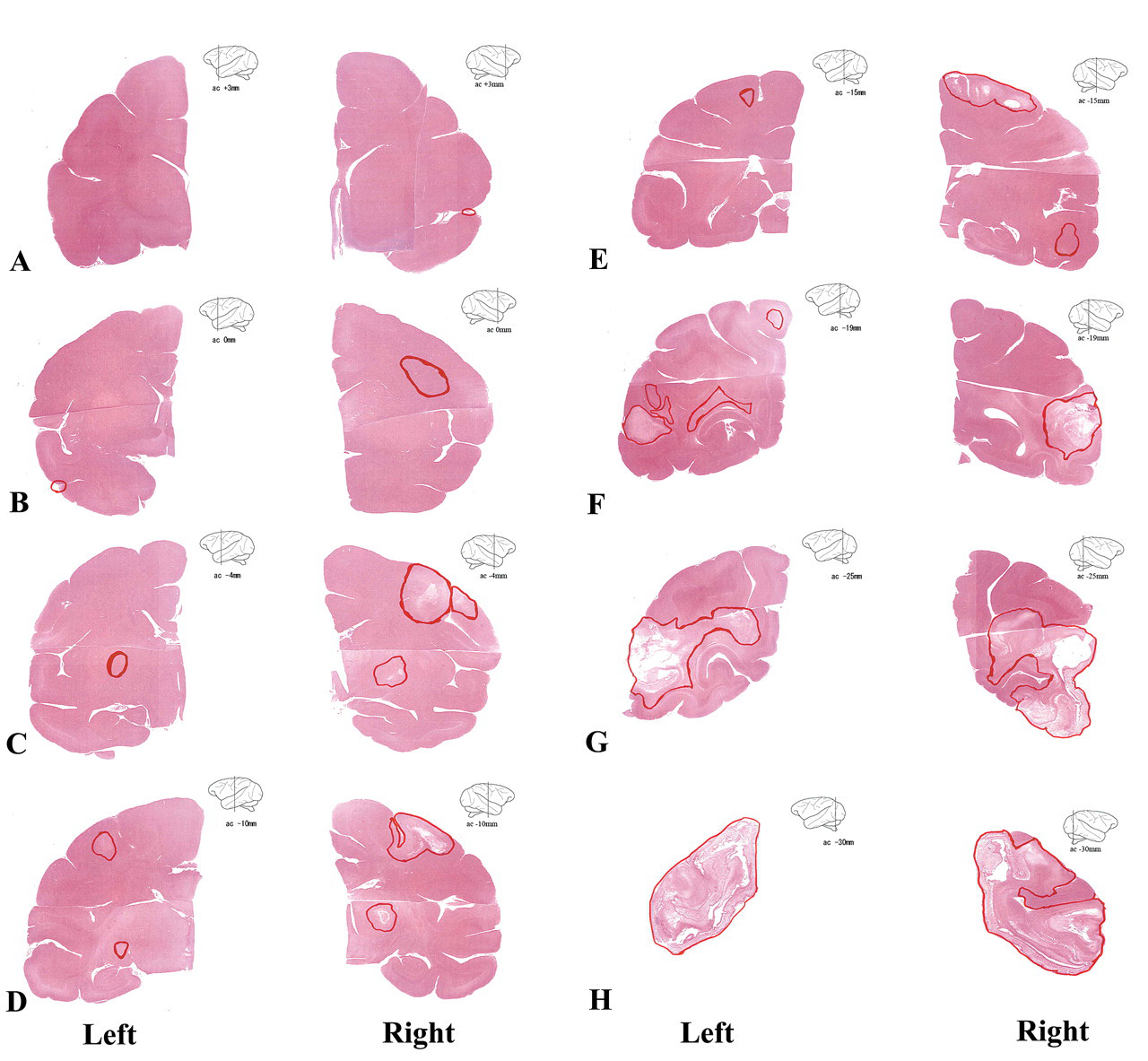
Mapping of brain lesions. Coronal sections of brain anterior (+) and posterior (–) to the anterior commissure (ac). (A) ac +3 mm, (B) 0 mm, (C) –4 mm, (D) –10 mm, (E) –15 mm, (F) –19 mm, (G) –25 mm, and (H) –30 mm. Multiple foci of cerebromalacia, demyelination, necrosis, gliosis, and/or inflammation are located in the red circle areas. H&E.
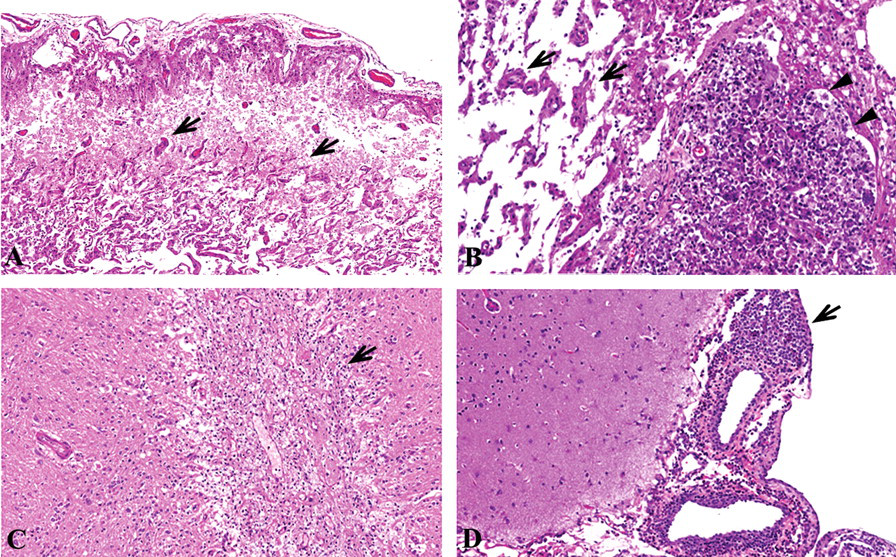
Histopathological features of the damage in the cerebrum. H&E. A: Cerebromalacia in the occipital lobe. Disorganization, gliosis (arrows), and vascular proliferation can be seen. ×100. B: Cerebromalacia in the occipital lobe. Glial shrub (arrows), focal infiltration of macrophage and lymphocytes (arrowheads) can be seen. ×200. C: Gliosis in temporal lobe (arrow). Infiltration of microglia also can be seen. ×100. D: Mononuclear cell infiltration in meninges of temporal lobe (arrow). ×100.
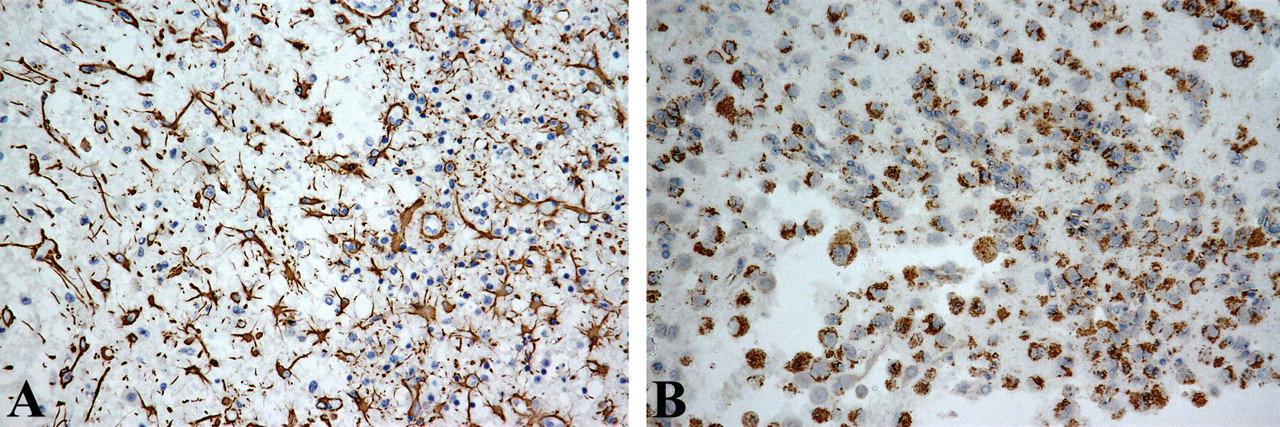
Immunohistochemical staining of the brain. The proliferation of GFAP-positive astroglia (A) and infiltration of CD68-positive microglia (B) can be seen. ×200.
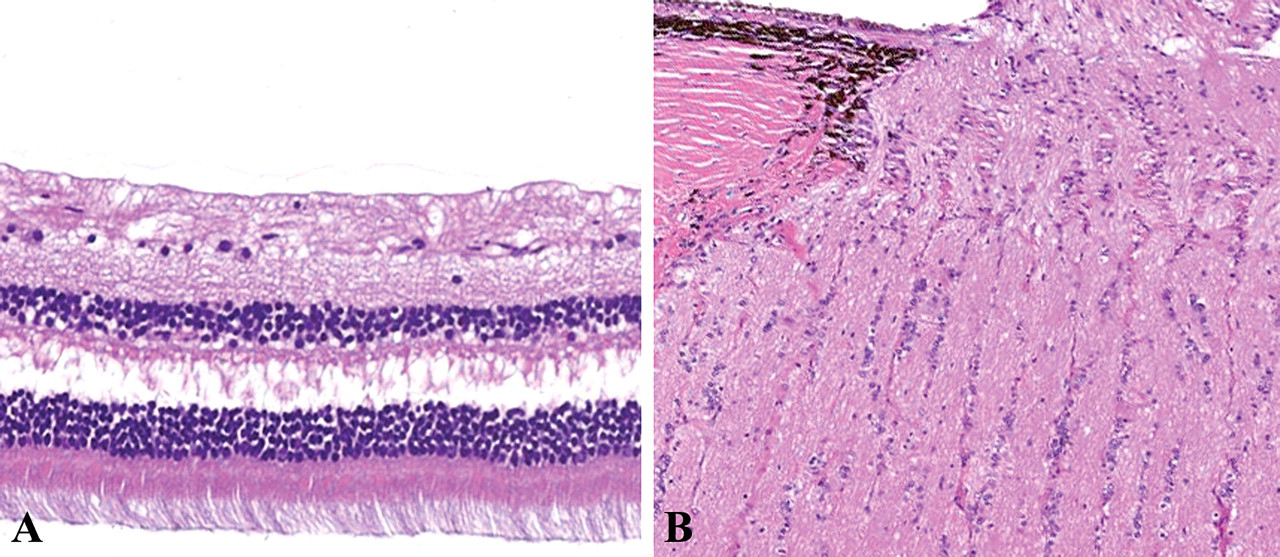
Retina (A) and optic nerve (B). H&E. No abnormal change can be seen. ×200.
In other organs, we observed thymus involution; mononuclear cell infiltration in the heart, tongue, and salivary gland; and inflammation in the mesentery and mesenteric lymph nodes, suggesting spontaneously occurring or age-related lesions (Lowenstine 2003; Ito et al. 1992). There were no findings to indicate parasitic, fungal, or viral infections in the systemic organs.
Discussion
This case was characterized by extensive neural damage and reactive changes in the bilateral occipital lobes of the cerebrum. The neural damage, suggestive of subacute phase lesions with glial reactivity, was related to clinical seizures. The lesions were accompanied by increased numbers of GFAP-positive astroglia and CD68-positive microglia, suggesting that the course of the brain damage matched the duration of clinical signs.
The relationship between clinical neurological signs and brain injury has traditionally presented diagnostic difficulties for the neuropathologist. In this case, the early clinical sign was muscular tremors contained mainly to the left side of the face, the left arm, and the left foot; these tremors were similar to Jacksonian convulsions, which are characterized by seizures involving one or several parts of the body on the same side (Lende and Popp 1976). Jacksonian seizures indicate focal lesions on the contralateral side of the brain, usually in the area of the motor cortex. In our case, this neurological sign may be related to the neuronal lesions that were detected extensively on the right side of the frontal, parietal, and temporal lobes of the cerebral cortex (Figure 2).
A severe degree of cerebromalacia was observed bilaterally in the occipital lobes of our monkey. The occipital lobe contains the primary visual cortex. Damage to the occipital cortex results in the total or partial loss of vision in a normal-appearing eye; this is referred to as cortical blindness or cerebral blindness (Flanagan, Kline, and Cure 2009). Our Japanese monkey exhibited the clinical signs of blindness, but she had no abnormal lesions in the retinal or optic nerves (Figure 5). Therefore, the cerebromalacia of the occipital lobe must be critical to the blindness in this case, which should be diagnosed as cortical blindness.
In humans, many etiologies of cortical blindness have been reported, such as bilateral occipital infarction in the vascular territory of the posterior cerebral arteries, trauma to the occipital lobe, and infectious diseases (Flanagan, Kline, and Cure 2009). In our monkey, anomalies and anastomosis of the posterior cerebral arteries could not be examined by angiography. However, no vascular lesions, such as thrombosis or degeneration, were observed in the brain, even in tissues stained with PAS or PTAH. Though infiltration of mononuclear cells into the brain was apparent, neither viral inclusions nor parasites were found in the nervous system.
In human cases with ischemic brain injury, a mononuclear-cell reaction could be seen 5 days after the ischemic episode (Barcikowska-Litwin et al. 1987). This information suggests that the brain lesions in our monkey were not related to acute viral infection but, instead, constituted a subacute reaction to ischemic necrosis. Brain damage might have occurred at the early time point when seizures began.
Several changes in serum chemistry values were noted, including increased levels of CPK, AST, LDH, and K and decreased levels of ALB and T-CHO. The increased CPK levels may reflect the marked damage to the skeletal muscle and/or brain, consistent with the observed seizures and neuronal necrosis. The other changes might reflect the deteriorating condition of the animal.
Naturally occurring lesions in the central nervous system have rarely been reported in laboratory monkeys. The following neuronal diseases have been reported in wild and laboratory nonhuman primates: inflammatory diseases such as xanthogranuloma (Lee et al. 1996); infection with rabies, toxoplasma, and bacteria (Schmidt 1978); aging changes such as pigmentation and calcium deposition (Ito et al. 1992); vascular aging changes and the formation of senile plaques and neurofibrillary tangles (Kodama et al. 2010; Lowenstine 2003; Yoshimura et al. 1978); congenital malformations such as ependymal cyst (Bergin, Campbell, and Agnew 2008), arterial duplication, and hydrocephalus (Schmidt 1978); and ischemic changes due to vascular thrombosis (Harber et al. 1996; Sheffield, Squire, and Strandberg 1981).
Although there was a report of an infant lion-tailed macaque with cerebral palsy characterized by abnormal MRI signal intensity within the parietal-occipital lobes, the animal did not exhibit any abnormalities in visual function (Fitch et al. 1988). It was suggested that ischemic infarction or hemorrhage near the time of birth caused the neuroanatomic deficits in this animal. In some colonies of Japanese monkeys, myeloencephalitis with Streptococcus infection (Lair et al. 1996), polymicrogyria (Schmidlin et al. 2009), neuropathy due to coyotillo intoxication (Joiner et al. 1975), multifocal malacia due to Baylisascaris larva migrans (Sato et al. 2005), and cerebellar cortical atrophy (Sanford and Mehren 1979) have been reported.
In our case, the cause of neuronal lesions could not be confirmed, but the relationship between the symptoms, including abnormal vision, and the brain lesions was clear. This case is valuable as a reference to historical control data for laboratory Japanese monkeys.
Footnotes
Acknowledgments
The authors thank Ms. T. Akamatsu for her excellent technical assistance and Ms. A. Shudo for manuscript preparation.
The authors declare to have no competing financial interests. This research was supported in part by a Grant-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science (JSPS) (22591954).
