Abstract
Clinical and nonclinical studies have implicated glucagon-like peptide-1 (GLP-1) receptor agonist therapy as a risk factor for acute pancreatitis in patients with type 2 diabetes. Therefore, it is critical to understand the effect that dulaglutide, an approved GLP-1 receptor agonist, has on the exocrine pancreas. Dulaglutide 8.15 mg/kg (approximately 500 times the maximum recommended human dose based on plasma exposure) was administered twice weekly for 12 months to cynomolgus monkeys. Serum amylase and lipase activities were measured and 6 sections of each pancreas were examined microscopically. Ductal epithelial cell proliferation was estimated using Ki67 labeling. Dulaglutide administration did not alter serum amylase or lipase activities measured at the end of treatment compared to control values. An extensive histologic evaluation of the pancreas revealed no changes in the acinar or endocrine portions and no evidence of pancreatitis, necrosis, or pancreatic intraepithelial neoplasia. An increase in goblet cells noted in 4 of the 19 treated monkeys was considered an effect of dulaglutide but was not associated with dilation, blockage, or accumulation of mucin in the pancreatic duct. There was no difference in cell proliferation in ductal epithelium between control and dulaglutide-treated monkeys. These data reveal that chronic dosing of nondiabetic primates with dulaglutide does not induce inflammatory or preneoplastic changes in exocrine pancreas.
Keywords
Introduction
Glucagon-like peptide-1 (GLP-1) receptor agonists exert several physiological actions that make this class of drugs attractive for patients with type 2 diabetes. Beneficial effects include increasing insulin biosynthesis and glucose-dependent insulin secretion, suppressing glucagon secretion, slowing gastric emptying, and reducing food intake (Asmar and Holst 2010; Baggio and Drucker 2007).
Dulaglutide is a long-acting GLP-1 receptor agonist approved for use as a once-weekly subcutaneous injection for the treatment of type 2 diabetes (Dungan et al. 2014; Nauck et al. 2014; Umpierrez et al. 2014; Weinstock et al. 2015; Wysham et al. 2014; Barrington et al. 2011a; Glaesner et al. 2010; Umpierrez et al. 2011). The molecule consists of two identical, disulfide-linked chains, each containing an N terminal GLP-1 analog sequence covalently linked to a modified human immunoglobulin G4 (IgG4) Fc fragment by a small peptide linker (Barrington et al. 2011b). Unlike native GLP-1, dulaglutide is less susceptible to degradation by dipeptidyl peptidase-4 (DPP-4) and has an increased molecular weight that slows absorption and minimizes renal clearance. This results in a prolonged half-life of approximately 5 days. Dulaglutide exhibits GLP-1-mediated effects, including potentiation of glucose-dependent insulin secretion, inhibition of glucagon secretion, delay in gastric emptying, and weight loss (Barrington et al. 2011a; Barrington et al. 2011b; Glaesner et al. 2010; Geiger et al. 2012; Grunberger et al. 2012; Umpierrez et al. 2011).
Postmarketing surveillance and clinical trials data have revealed a potential association between use of GLP-1 receptor agonists and pancreatitis and/or pancreatic cancer (Denker and Dimarco 2006; Ahmad and Swann 2008; Franks, Lee, and George 2012; Elashoff et al. 2011). However, observational and claims-based surveillance studies suggest that while the development of pancreatitis is closely associated with having type 2 diabetes, the risk is not increased with the use of GLP-1 receptor agonists (Dore et al. 2009, 2011; Girman et al. 2010; Garg, Chen, and Pendergrass 2010; Wenten et al. 2012). Rodent studies using the GLP-1 receptor agonists exenatide or liraglutide have also yielded conflicting results ranging from exacerbation of pancreatitis, induction of pancreatic acinar inflammation, and proliferation of pancreatic ducts to attenuation of chemically induced pancreatitis and elevated expression of anti-inflammatory cytokines and/or an absence of changes in pancreatic exocrine structure (Gier et al. 2012; Nachnani et al. 2010; Tatarkiewicz et al. 2010; Koehler et al. 2009; Rouse et al. 2014). Limited studies of GLP-1 receptor agonists in nonhuman primates (NHPs) have not identified effects on the exocrine pancreas. One study of liraglutide administered to NHP at supraphysiological doses for 87 weeks did not report any pancreatic structural changes (Nyborg et al. 2012). An additional report described a 52-week NHP study of liraglutide and 13- and 52-week NHP studies of semaglutide (a structurally different GLP-1 receptor agonist) and did not report any histopathologic alterations of the exocrine pancreas (Gotfredsen et al. 2014).
In a routine chronic (9-month) toxicity study of dulaglutide in cynomolgus monkeys, no effects on the exocrine pancreas were noted; however, this study was conducted prior to clinical reports of a potential association of GLP-1 receptor agonists and pancreatitis. To more fully examine the potential effects of dulaglutide on the exocrine pancreas of the NHP, exocrine pancreatic evaluations were incorporated into a study designed to determine if dulaglutide had proliferative effects on thyroid C-cells. Briefly, 19 cynomolgus monkeys were administered 8.15 mg/kg dulaglutide by subcutaneous injection twice weekly for 12 months. This dose represents approximately 500 times the maximum recommended human dose based on plasma exposure (de la Peña et al. 2014). Because pancreatic end points were incorporated into the study near the end of the dosing phase, serum amylase and lipase activities were only measured at the end of the treatment period. An extensive histologic and immunohistochemical examination was conducted on multiple regions of the pancreas and included review by both veterinary and human pathologists to determine if dulaglutide altered exocrine pancreatic structure. This evaluation included a focused examination of pancreatic ducts for the presence of proliferative or dysplastic lesions comparable to human pancreatic intraepithelial neoplasia (PanIN) 1 to 3. Proliferation of duct luminal cells was evaluated using immunohistochemical labeling with the cell proliferation marker Ki67. This study represents a comprehensive morphologic evaluation of the monkey exocrine pancreas following chronic dosing with a GLP-1 receptor agonist and is unique with respect to the number of monkeys evaluated, the number of pancreatic sections examined, and prospective evaluation by a pathology working group consisting of expert veterinary and human pathologists.
Methods
Experimental Animals
All studies were performed in accordance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare. Male cynomolgus monkeys (Macaca fascicularis; origin China) aged 2 to 3 years were obtained from Covance Research Products Inc. (Alice, TX) and housed in climate-controlled rooms maintained at a 12-hr light/dark cycle, fed a standard primate diet (#2055C; Harlan Laboratories, Inc., Indianapolis, IN), and given free access to water. Monkeys were individually housed during the study, except when commingled for environmental-enrichment purposes. The monkeys in the study were negative for B-virus, Simian retrovirus (SRV), Simian immunodeficiency virus (SIV), Simian T-cell leukemia virus (STLV1), and measles.
Treatment
Monkeys received vehicle (10 mM citrate buffer [pH 6.5 ± 0.2], 4.6% mannitol [w/v], and 0.02% [w/v] polysorbate 80) or 8.15 mg/kg dulaglutide twice weekly by subcutaneous injection for 12 months (n = 20 monkeys per treatment group through day 35 of the treatment period). The dose of dulaglutide was selected because it was the maximally tolerated dose established in prior monkey repeat-dose toxicity studies. This dose produced systemic exposures in monkeys approximately 500 times the maximum recommended human dose based on plasma exposure. Dosing was suspended in 1 monkey after receiving the first 2 dulaglutide doses due to low food consumption and body weight loss. After 4 missed doses, this animal regained the lost body weight but was unable to maintain that weight once dosing resumed. On day 36, this animal was removed from study due to a failure to accommodate to GLP-1-mediated pharmacology, leaving 19 monkeys in the dulaglutide group. Solutions were prepared once weekly and stored at 2°C to 8°C. The dose volume was 1 ml/kg. Animals were checked twice daily (morning and evening) for mortality, abnormalities, and signs of pain or distress. Throughout the dosing phase, food consumption was assessed qualitatively and body weight was recorded at least twice weekly. At the end of the study, monkeys were weighed, euthanized with an intravenous injection of sodium pentobarbital (50 mg/kg), exsanguinated, and a necropsy was performed.
Measurement of Plasma Dulaglutide Levels
Blood was collected approximately 72 hr post-dose on days 88, 179, 270, and 365 and was processed to plasma to assess dulaglutide levels. Dulaglutide plasma concentrations were determined using a validated enzyme-linked immunosorbent assay (ELISA). Briefly, microtiter plates were coated with mouse antihuman IgG (Fc) antibody (Southern Biotech, Birmingham, Alabama). Dulaglutide standards, controls, and samples were prepared in monkey plasma. Following preparation, the samples were incubated on the coated plates for 1 hr at room temperature. The dulaglutide complex on the plate was bound with a mouse IgG2a κ anti-GLP-1 antibody (Pierce, Rockford, IL) and then detected using a goat anti-mouse IgG2a-horseradish peroxidase (IgG2a-HPR) (Bethyl, Montgomery, TX) with tetramethylbenzidine (TMB) substrate. The standard curve ranged from 1.00 to 160 ng/ml with 2.00 and 160 ng/ml being the lower and upper limits of quantitation, respectively.
Measurement of Serum Amylase and Lipase
Blood was collected from the femoral vein of fasted monkeys on the day of sacrifice and centrifuged, and the serum was separated for analysis. Serum amylase and lipase activities were measured according to the manufacturer’s protocol using an automated clinical chemistry analyzer (Modular Analytics, Roche Diagnostics, F. Hoffmann-La Roche AG, Konzern Hauptsitz, Grenzacherstrasse 124, CH-4070 Basel, Schweiz).
Histologic Processing of the Pancreas
For all animals surviving to scheduled necropsy, the entire pancreas was resected at necropsy, assessed for any macroscopic abnormalities, and collected for systematic histologic evaluation. Pancreata were not weighed because an approximate 3-cm portion of the duodenum was collected with the pancreas at necropsy to aid in tissue orientation during trimming and embedding. Pancreata were fixed in 10% neutral-buffered formalin at room temperature for at least 15 min and then trimmed to obtain 3 transverse blocks of the head, 2 transverse blocks of the body, and 1 transverse block of the tail. Blocks were marked with tissue marking dye (distal surfaces of the sections of head and tail, opposing surfaces of the sections of body), trimmed further as needed, fixed for an additional 48 to 72 hr, dehydrated, and embedded in paraffin (marked surface down) using standard histologic procedures. One section (4–5 µm) was cut from each block, stained with hematoxylin and eosin, and examined with a light microscope. One additional section from each of the head blocks (3/pancreas) was immunolabeled with the cell proliferation marker Ki67. A block from the head region was selected for Ki67 assessments as data from a rodent study with a GLP-1 receptor agonist indicated increased Ki67 labeling in the head, but not tail, regions of the pancreas (Gier et al. 2012). Automated immunohistochemical labeling was performed on a Ventana Discovery XT immunostainer (Roche Diagnostics) using a rabbit monoclonal antibody to Ki67 (SP6; Spring Bioscience, Pleasanton, CA) diluted 1:200 for 60 min and a biotin-conjugated donkey anti-rabbit secondary antibody (Jackson ImmunoResearch Laboratories, Inc., West Grove, PA) diluted 1:1,000 for 32 min. Two negative controls, using buffer or negative control rabbit immunoglobulin fraction (Dako North America, Inc., Carpinteria, CA) diluted 1:500 instead of the primary antibody, were included in each analysis.
Histologic Evaluation of the Pancreas
Hematoxylin and eosin–stained (H&S) sections were initially evaluated by a veterinary (study) pathologist who reviewed all sections of the pancreas and entered semiquantitative histologic observations into a computerized data collection system (Path/Tox System Pristima; Xybion Medical Systems Corporation, Morris Plains, New Jersey) using a standard toxicologic pathology 0- to 5-point evaluation scheme (Crissman et al. 2004). Following the initial evaluation, a peer review of the microscopic observations was performed by a second veterinary pathologist. Both the initial evaluation and peer review were conducted with the knowledge of treatment group (Crissman et al. 2004). Subsequently, a pathology working group of veterinary (TR, PWS, SDS, and JLV) and human medical (GK) pathologists reviewed all of the slides from the head of the pancreas and any additional slides in which the study and peer review pathologist identified histologic findings. All pathologists evaluated both the endocrine and exocrine pancreas for qualitative abnormalities. Pancreatic ducts were carefully evaluated for morphologic features consistent with human PanIN based on established criteria (Hruban et al. 2001). In addition, a subset of the pathology working group (TR, PWS, and GK) performed a review of the slides blinded to treatment group to identify and grade increased numbers of goblet cells within interlobular ducts. Diagnostic criteria used to assign a severity grade to the finding of increased goblet cells are listed in Table 1.
Diagnostic criteria for grading goblet cell increases in interlobular ducts.

Evaluation of Pancreas Sections Labeled for Ki67
Evaluation of Ki67 labeling in duct luminal epithelial cells in sections from the head of the pancreas was initiated by examining the 3 slides at low magnification to identify the section that contained the duct with the thickest wall that had a tunica muscularis and was completely surrounded by pancreatic lobules, and this was designated the primary section. Large ducts external to the pancreatic lobules (papilla/ampulla or bile duct) that were present in some sections were not evaluated. In the primary section, the thickest walled duct was evaluated at 400× magnification. Ki67 labeling was estimated by counting Ki67-labeled cells in a total of 1,000 duct luminal epithelial cells. Within the transverse section of the duct selected for counting, cell counting was initiated at the top (12 o’clock position) of each duct and proceeded in a clockwise direction until 1,000 cells were counted. Ki67 labeling of pancreatic cell types other than duct luminal or ductal glandular epithelium was not evaluated. The mean percentage of Ki67-labeled duct luminal epithelial cells was calculated for each monkey.
Statistical Analysis
One-way analysis of variance (ANOVA) and the Dunnett adjusted t-test at a 5.0%, two-tailed probability level was conducted on body weight, amylase, and lipase where appropriate. Findings were significant with p ≤ .05.
Results
Dulaglutide Plasma Concentrations
Plasma concentrations of dulaglutide were determined after 3, 6, 9, and 12 months of twice weekly administration of 8.15 mg/kg dulaglutide. Plasma concentrations were measured approximately 72 hr post-dose on days 88, 179, 270, and 365. At each collection interval, all monkeys administered dulaglutide 8.15 mg/kg had measurable plasma dulaglutide levels, and the mean plasma levels were similar (less than 2-fold difference) over the 12-month study duration (Table 2). These plasma exposures represented approximately 500 times the maximum recommended human dose based on plasma exposure (de la Peña et al. 2014).
Mean plasma concentration of dulaglutide in male monkeys given subcutaneous injections of 8.15 mg/kg dulaglutide twice weekly for 12 months.

Note: Plasma concentrations were measured approximately 72 hr post-dose. n = 19 monkeys/measurement. SD = standard deviation.
Animal Growth, Clinical Observations, and Serum Amylase and Lipase Measurements
Monkeys administered dulaglutide 8.15 mg/kg had decreased food consumption compared to controls, most notably during the first 4 weeks of the study, resulting in secondary body weight loss over the same period. Veterinary examinations were performed on several monkeys receiving dulaglutide during the first week of study due to the severity of decreases in food consumption and body weight. Four monkeys required veterinary intervention in the form of lactated Ringer’s solution and/or oral corn syrup for treatment of dehydration in the days following the first dose of dulaglutide. During the early portion of the dosing phase, dulaglutide-treated monkeys were reported with few or no feces correlating with decreased food and dietary supplement consumption, and several individual monkeys were noted clinically as thin. The severity of the GLP-1 receptor agonist–mediated effects on appetite and resultant decreases in body weight diminished after the first month of dosing and apart from 3 monkeys (1 that was removed from study on day 36 due to a failure to accommodate, 1 that was not dosed on day 22 due to body weight loss, and 1 that was not dosed on day 75 due to decreased food consumption and dehydration), all monkeys were deemed to be of acceptable health by the veterinary staff to remain on study and receive all intended dulaglutide doses. Lower mean body weights were observed for dulaglutide 8.15 mg/kg-treated monkeys compared to controls throughout the 12-month treatment period (p ≤ .05; Figure 1A). In addition, control monkeys gained significantly more weight over the course of the study compared to monkeys given dulaglutide (mean weight change: 1.5 vs. 0.7 kg, respectively; p ≤ .05). Although not significant, monkeys treated with dulaglutide for 12 months had numerically lower levels of serum amylase and lipase activity compared to controls (p = .24, serum amylase; p = .07, serum lipase; Figure 1B and C).
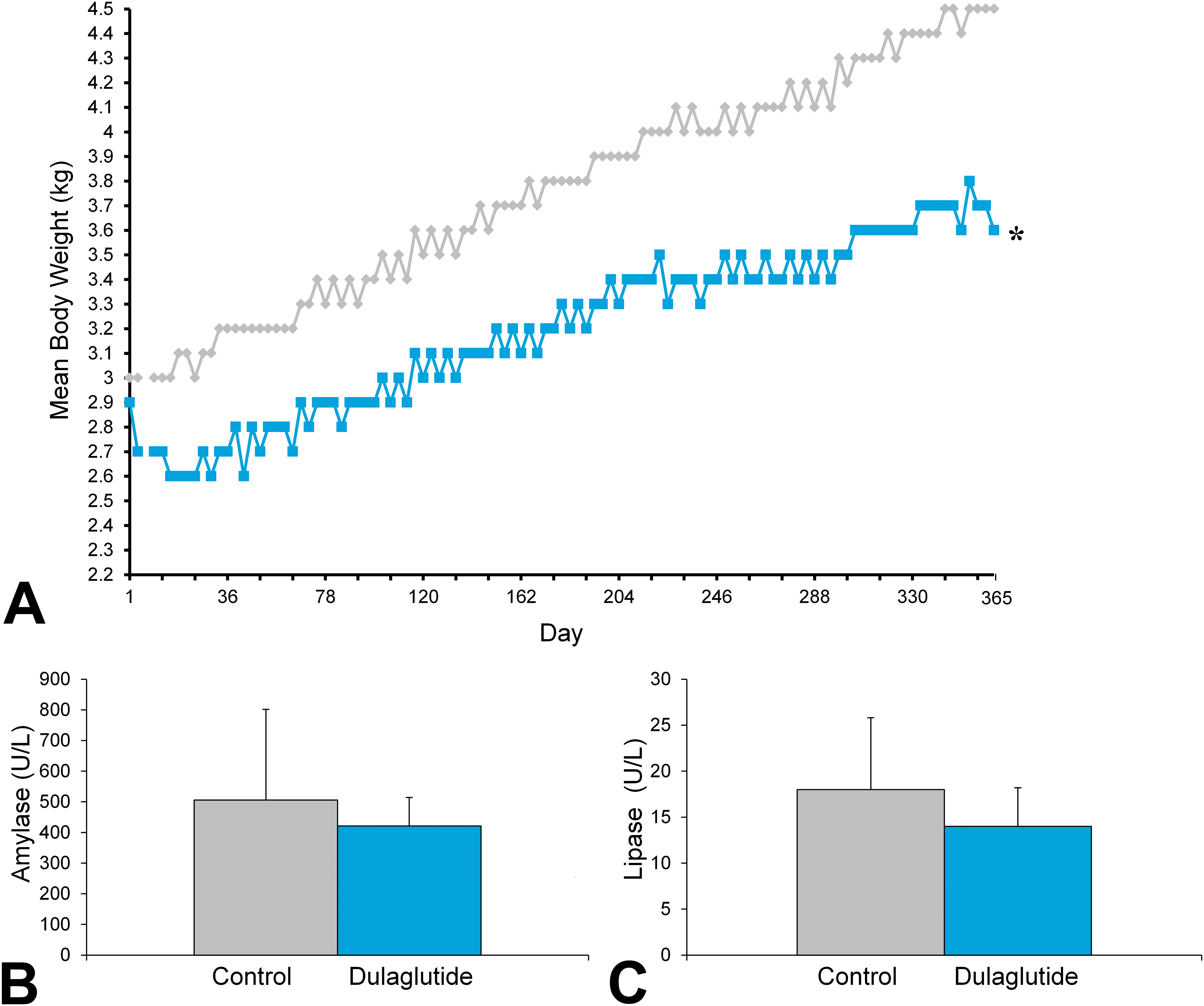
Animal growth and serum amylase and lipase measurements. (A) Mean body weight was lower in monkeys treated with dulaglutide for 12 months. *p ≤ .05 versus control. Diamonds = control monkeys; squares = dulaglutide-treated monkeys. (B) No effect of dulaglutide on serum amylase or (C) serum lipase was observed following 12 months of treatment (p = .24 vs. control and p = .07 vs. control, respectively). Gray bars = control monkeys; blue bars = dulaglutide-treated monkeys.
Pancreatic Duct Morphology in Control Monkeys
To date, the literature has not provided a detailed description of normal pancreatic duct morphology in cynomolgus monkeys. Thus, an initial review of the pancreas focused on evaluating normal variations in the morphology of the pancreatic ducts. In control monkeys, transverse sections of pancreas typically contained a central large (main) pancreatic duct, a few interlobular and intralobular ducts, and many intercalated ducts. Main pancreatic ducts were lined by a variably folded simple to pseudostratified columnar epithelium that varied markedly in height among animals and less markedly between regions (head, body, or tail) of the same pancreas. Some main ducts had abrupt variations in epithelial height within the same microscopic field (Figure 2). Epithelial height gradually decreased in interlobular ducts, becoming cuboidal in intralobular ducts. The amount of mucin in the apical cytoplasm of duct luminal epithelium also varied widely among animals. When present, mucin-containing epithelial cells were generally irregularly distributed among non-mucin-containing cells. In addition, some animals also had intraepithelial goblet cells, usually occurring as rare scattered individual cells in the main and interlobular ducts. In 6 of the 20 control monkeys, goblet cell numbers were considered minimally increased (Table 3) compared to other control animals.
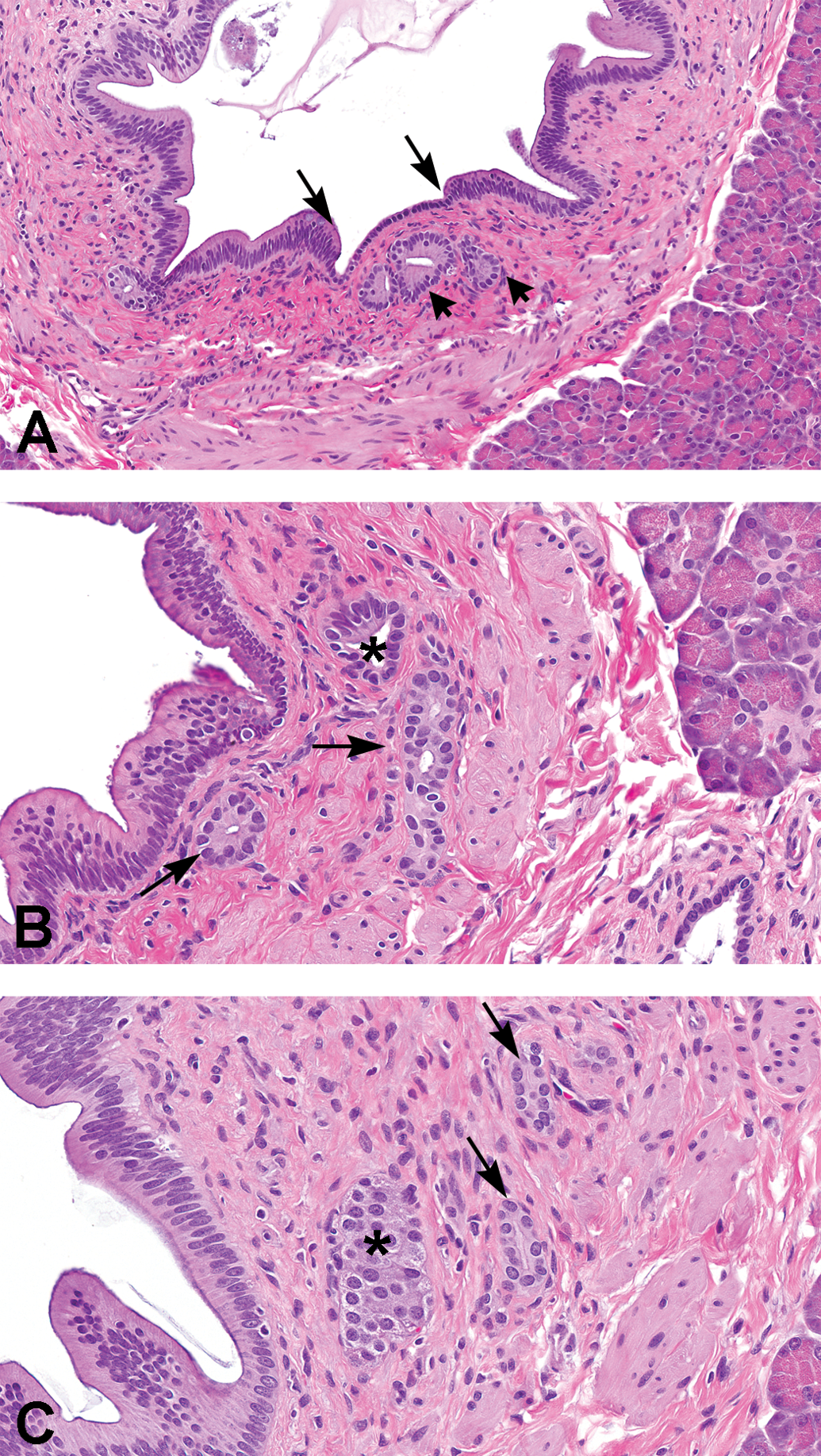
(A–C) Control cynomolgus monkeys. (A) Head of pancreas. Note the marked variation in epithelial height within the same large duct profile (long arrows). Short arrows indicate pancreatic duct glands. (B) Head of pancreas. Arrows indicate duct glands in propria of main duct. Duct gland epithelial cells have larger nuclei and paler cytoplasm than duct lining epithelium. Asterisk indicates a structure with features intermediate between those of a duct gland and an interlobular duct. (C) Body of pancreas. Pancreatic duct glands (arrows) and an islet (*) in propria of main duct.
Incidence of microscopic alterations in the pancreas of monkeys administered 8.15 mg/kg dulaglutide or vehicle control twice weekly for 12 months.
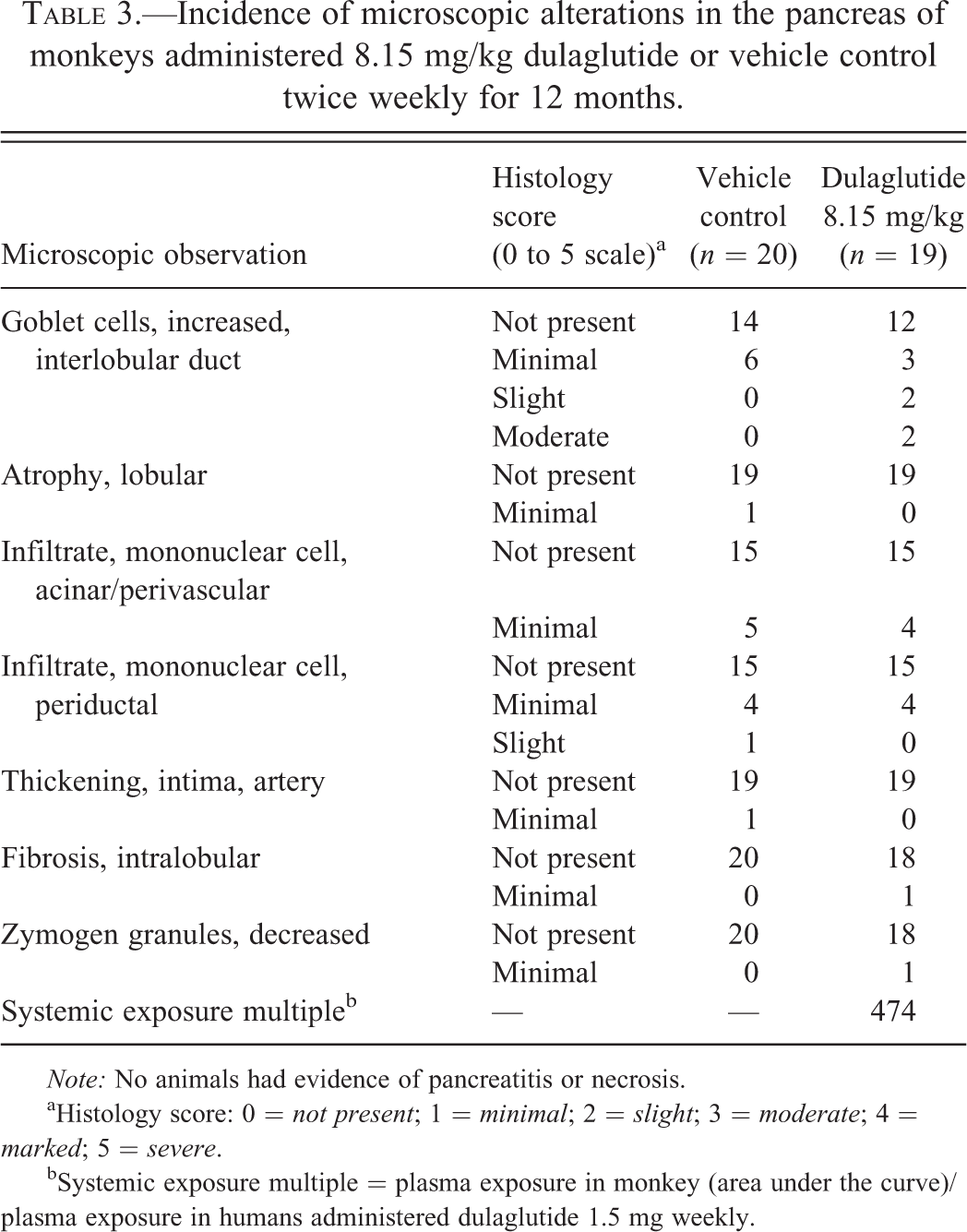
Note: No animals had evidence of pancreatitis or necrosis.
aHistology score: 0 = not present; 1 = minimal; 2 = slight; 3 = moderate; 4 = marked; 5 = severe.
bSystemic exposure multiple = plasma exposure in monkey (area under the curve)/plasma exposure in humans administered dulaglutide 1.5 mg weekly.
Small tubular structures consistent with pancreatic duct glands (Strobel et al. 2010) were identified in the propria and tunica muscularis of the main and interlobular ducts in all regions of the pancreas (Figure 2). Again, the frequency of such structures varied among individual monkeys. Structures consistent with pancreatic duct glands comprised epithelial cells that were larger and had more cytoplasmic mucin than the adjacent duct epithelium. Putative duct glands were also smaller in caliber than would be expected for the next generation of a given duct, however, some tubular structures were of uncertain identity (Figure 2B) because they had characteristics intermediate between those of a duct gland and an interlobular or intralobular duct.
Pancreatic Duct Morphology in Dulaglutide-treated Monkeys
The only morphologic change attributed to dulaglutide treatment was a slight or moderate increase in goblet cells in interlobular ducts in 4 of the 19 monkeys treated with dulaglutide (Figure 3 and Table 3). Slight or moderate increases in interlobular duct goblet cells were not identified in control animals. The histologic appearance of the individual goblet cells was similar to that of the controls, in that they were well differentiated and contained abundant pale-staining cytoplasm consistent with mucin. The apparent increase in numbers of goblet cells in these monkeys was not associated with duct dilation, blockage, or accumulation of mucin and thus was considered nonadverse. No other changes in morphology of either large or small caliber ducts were noted in dulaglutide-treated monkeys. Neither control nor dulaglutide-treated monkeys had morphologic changes consistent with PanIN.
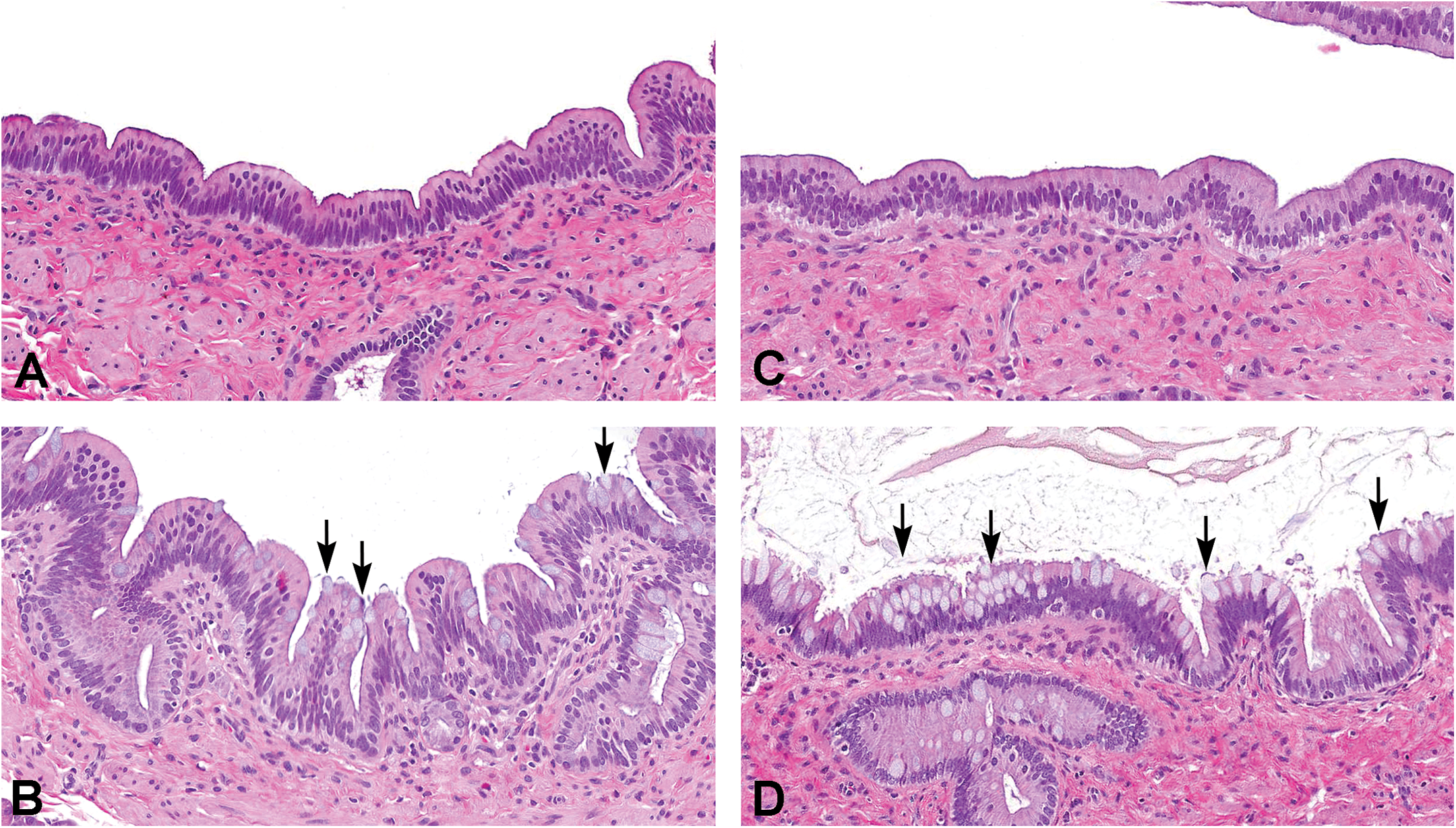
Pancreatic ducts of control (A, B) and dulaglutide-treated (C, D) monkeys. In most control (A) and dulaglutide-treated (C) monkeys, interlobular duct goblet cells were either not observed or occurred as rare scattered individual cells. (B) Six of the twenty control monkeys had minimal focal areas in which goblet cells (arrows) were observed in interlobular ducts. (D) Four of nineteen monkeys had slight to moderate numbers of goblet cells in interlobular ducts (arrows). Refer to Table 1 for criteria for grading increases in interlobular duct goblet cells.
Morphologic Observations in Acinar Pancreas and Interstitium
There was no evidence of pancreatitis in either control or dulaglutide-treated monkeys (Table 3). Mononuclear cell infiltrates are common in the cynomolgus monkey in a variety of tissues (Chamanza et al. 2010) and were noted as small foci in either periductal, acinar, or perivascular locations. The incidence and severity of these infiltrates were similar in control and dulaglutide-treated monkeys. Observations that were limited to a single monkey included minimal arterial intimal thickening (control), minimal intralobular fibrosis (dulaglutide treated), and a minimal focal decrease in zymogen granules (dulaglutide treated). Based on the minimal magnitude and occurrence in a single monkey, these observations were not attributed to dulaglutide treatment but were considered background changes that can occur in cynomolgus monkeys. There were no other alterations noted in overall acinar structure.
Assessment of Duct Cell Proliferation
Relative frequencies of Ki67-labeled duct luminal and glandular epithelial cells in the head of the pancreas were assessed to provide an estimate of duct epithelial cell growth fraction. Notable interanimal variability in the relative frequency of Ki67-labeled duct luminal and glandular epithelial cells occurred in both control and dulaglutide-treated monkeys. Importantly, there was no difference in duct epithelial cell proliferation between control and dulaglutide-treated groups based on either an inspection of group means or in terms of individual animal values (Figure 4).
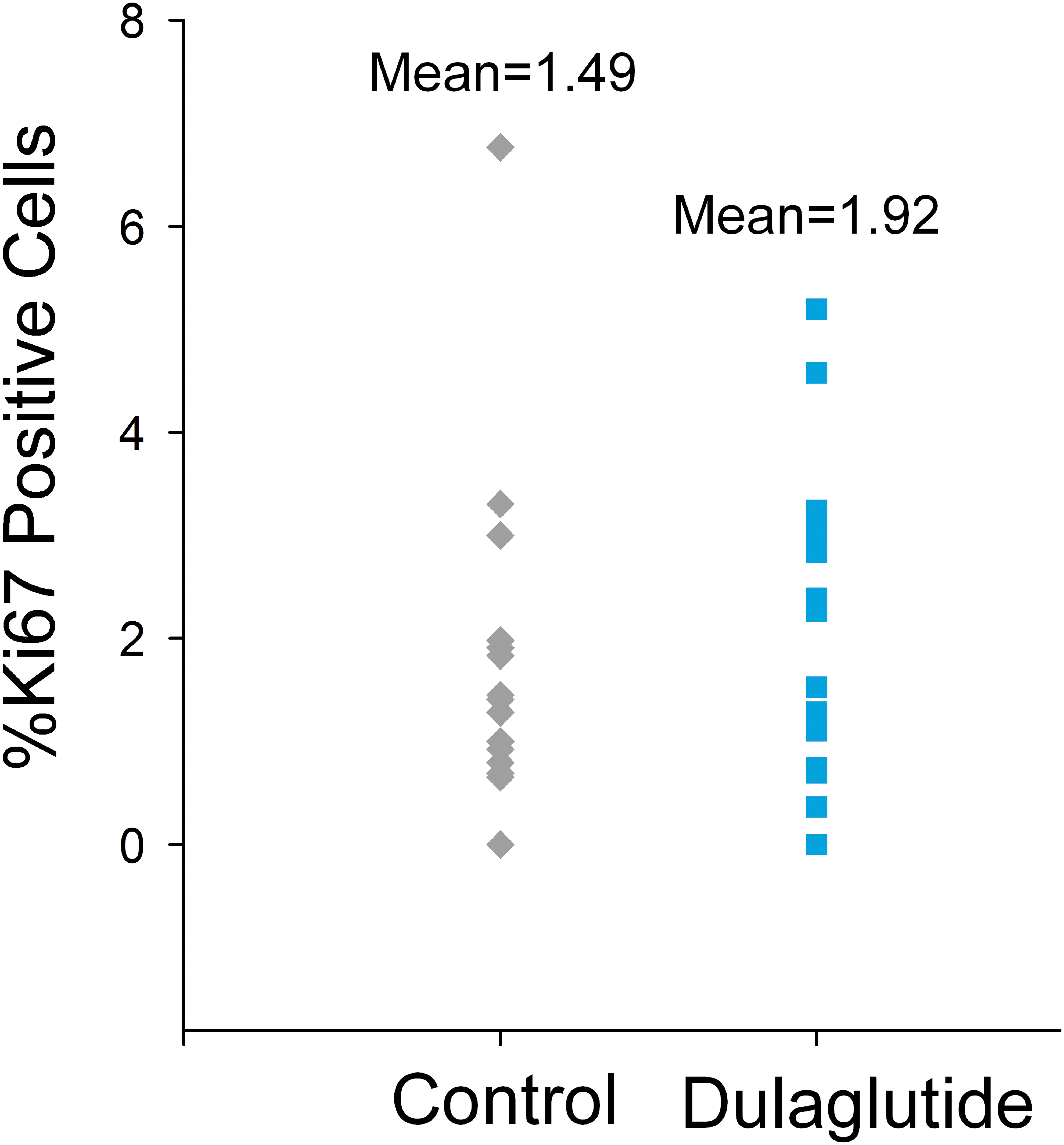
Pancreatic duct luminal epithelial cell proliferation (Percentage Ki67 positive cells) was similar in controls (n = 20) and monkeys administered dulaglutide (n = 19) for 12 months. Each symbol represents an individual monkey. Ki67 labeling of pancreatic duct glandular epithelial cells was also similar in both groups (data not shown).
Discussion
Marketed GLP-1 receptor agonists have been reported to be associated with a potential increased risk of pancreatitis in diabetic patients, however, clinical reports and analyses have yielded conflicting results, with some studies suggesting an association of these agents with acute and/or chronic pancreatitis, while other studies have failed to find any association (Butler et al. 2013a; Dore et al. 2011; Dore et al. 2009; Elashoff et al. 2011; Garg, Chen, and Pendergrass 2010). To better understand the potential effects of GLP-1 receptor agonists on pancreatic inflammation, studies in a variety of animal models have been conducted but have yielded conflicting results. Pancreatic inflammation in nondiabetic rats treated with exenatide was reported by Nachnani et al. (2010); however, pancreatic inflammation was not detected in nondiabetic rats treated with the same dose of exenatide in a subsequent study (Gier et al. 2012). Studies in diabetic rodent models also have provided conflicting results. Studies of Zucker diabetic fatty (ZDF) rats (an animal model of type 2 diabetes) treated with liraglutide (Vrang et al. 2012) or exenatide (Tatarkiewicz et al. 2010) did not detect an increase in pancreatic inflammation. In mice administered a high-fat diet, exenatide was reported to exacerbate pancreatic acinar cell injury (Zhang et al. 2013; Rouse et al. 2014). In chemically induced pancreatitis models in rodents, there was either no effect or an apparent attenuation of pancreatitis with GLP-1 receptor agonist treatment (Koehler et al. 2009; Tatarkiewicz et al. 2010). Standard toxicology studies in rodents, monkeys, or dogs using liraglutide (Nyborg et al. 2012), exenatide (Tatarkiewicz et al. 2010), and sitagliptin, a DPP-4 inhibitor (Forest et al. 2013), did not identify pancreatitis as an effect of treatment.
In this 12-month study, monkeys administered dulaglutide at nearly 500 times the systemic exposure level observed in humans given once-weekly dulaglutide 1.5 mg did not show any clinical signs suggestive of pancreatitis (vomiting) nor did they have elevated levels of amylase and lipase at the end of the treatment period. Because the study was not originally designed to study exocrine pancreatic injury, pre-study and sequential measurements of amylase and lipase were not conducted. There was no evidence that dulaglutide induced inflammatory or necrotizing lesions of the pancreas based on a thorough histologic examination of multiple regions of the pancreas. Concerns have been raised that the absence of pancreatitis in standard toxicity studies of GLP-1 receptor agonists could be, in part, due to the fact standard nonclinical toxicology protocols collect a single section of the pancreas, and the studies do not include determination of amylase and lipase (Gale 2012). The current study provides assessment of pancreatic enzymes and extensive histologic analyses focused on the exocrine pancreas in a large group (n = 19–20) of NHPs administered a supraphysiological dose of a GLP-1 receptor agonist for a prolonged period of time. The study design did not provide for morphologic assessments at multiple intervals during the dosing phase. While it is plausible that minor transient effects may have occurred during the study, the absence of notable changes in exocrine architecture, residual fibrosis, or other histologic alterations suggests that any such event was unlikely. The absence of findings indicative of pancreatitis in this monkey study is consistent with the weight of evidence from previously published studies of other GLP-1 receptor agonists in monkeys, suggesting that these agents do not induce pancreatitis in NHP models (Gotfredsen et al. 2014; Nyborg et al. 2012).
In addition to a potential role in pancreatitis, the role of incretin therapies in inducing proliferation of pancreatic ducts or promoting pancreatic cancer is still debated (Butler et al. 2013b; Nauck 2013). In a rodent model of type 2 diabetes (human islet amyloid polypeptide transgenic rat), sitagliptin was associated with increased ductal cell turnover and metaplasia. Gier et al. (2012) reported proliferation of epithelial cells in the main pancreatic duct and pancreatic duct glands in nondiabetic rats as well as an increased formation of murine PanIN lesions in a genetically modified mouse model of pancreatic cancer administered exenatide. In a human autopsy series, use of incretin-based therapies was associated with an increase in whole pancreas cell proliferation and presence of PanIN (Butler et al. 2013a); however, a variety of methodological issues, such as differences between the studied groups with respect to age, gender, disease duration, and treatments, limit the ability to interpret this study (Drucker 2013; Kahn 2013; Bonner-Weir, In’t Veld, and Weir 2014).
In contrast to studies suggesting a relationship between incretin therapy and proliferation of the exocrine pancreas, toxicology studies using liraglutide (Nyborg et al. 2012; Gotfredsen et al. 2014), exenatide (Tatarkiewicz et al. 2010), lixisenatide (European Public Assessment Report, Lyxumia [EPAR] 2012), albiglutide (Mirabile, Kambara, and Maier 2013), semaglutide (Gotfredsen et al. 2014), and sitagliptin (Engel et al. 2010) did not detect proliferative lesions in the pancreas due to treatment nor were pancreatic tumors increased in rodent carcinogenicity studies. These studies included evaluation of pancreatic histology in mice, rats, dogs, and monkeys in toxicology studies that utilized relatively high doses of these agents. No changes in the pancreas histology were noted; however, many of these studies did not include an analysis of all regions of the exocrine pancreas, nor did they assess pancreatic duct cell proliferation. In the 52-week primate study of liraglutide, quantitative histology was conducted to determine if changes in the mass of pancreatic cell types could be detected (Gotfredsen et al. 2014). In this study, no changes were found in ductal cell volume, however, the number of monkeys per group was relatively small (n = 4/sex/group). This study used robust stereologic-based methods to measure mass of both exocrine and endocrine pancreas, however, it did not appear to include qualitative evaluation of various portions of the pancreatic duct for focal hyperplastic changes. In a meta-analysis of both approved and unapproved GLP-1-based therapeutics encompassing 15,480 healthy nondiabetic rodents and 2,475 healthy nondiabetic monkeys, the Food and Drug Administration (FDA) did not identify any definitive adverse effects of incretins on the pancreas (Egan 2014). Furthermore, the FDA reported that toxicology studies of exenatide (Tatarkiewicz et al. 2010), liraglutide (Vrang et al. 2012), and sitagliptin (Forest et al. 2013) in ZDF rats did not demonstrate definitive drug-related adverse effects on exocrine pancreas histology or proliferation (Egan et al. 2014).
To better understand whether dulaglutide causes proliferative effects on the exocrine pancreas, the current study included an expanded histologic sectioning paradigm designed to systematically survey the ductal tree and incorporated the use of a cell-proliferation marker, Ki67, to assess for effects on cell proliferation of the duct epithelium. The study also included evaluations by a pathology working group of veterinary and human pathologists to identify PanIN or PanIN-like lesions. The study did not include immunohistochemical or quantitative evaluation of the endocrine pancreas, however, the qualitative histologic evaluation did assess all components of the pancreas.
In the current study, which is the first study to include a large number of healthy, control monkeys with systematic sampling of the exocrine pancreas, there was no evidence of proliferative or dysplastic lesions consistent with PanIN following dulaglutide administration for 12 months at a supraphysiological dose (Hruban et al. 2001). While spontaneous occurrence of PanIN-like lesions would not be expected in relatively young monkeys, it was important to carefully evaluate the potential of a GLP-1 receptor agonist to induce either proliferation of pancreatic ducts or the formation of focal hyperplastic lesions such as PanIN. The relatively large group size and histologic sampling scheme of this study allowed for more extensive characterization of the normal variability in duct structure that occurs in cynomolgus monkeys. Specifically, there was greater variability in the height and organization of the pancreatic ductal epithelium in both the control and dulaglutide-treated monkeys than is typically appreciated in routine toxicity studies that examine a single section of pancreas. It should be noted that PanIN-like lesions induced by a novel Glial cell line–derived neurotrophic factor (GDNF) protein construct have been described in monkeys (Ohshima-Hosoyama et al. 2012), confirming that PanIN lesions can be induced in cynomolgus monkeys. The current study indicates that PanIN-like changes are not induced by dulaglutide treatment of monkeys.
In 4 of the 19 dulaglutide-treated monkeys, there was an increase in goblet cells in the interlobular ducts. Although the role of goblet cells within pancreatic duct epithelium is not clearly described in the literature, goblet cells secrete mucus as a protective barrier for mucosal epithelia. In the current study, the increase in goblet cells was not associated with inflammatory or proliferative changes, ductal dilation, or blockage. The goblet cells were well differentiated and showed no features consistent with intraductal pancreatic mucinous neoplasia (Hruban et al. 2001). Based on the lack of correlative findings suggesting ductal injury or proliferation, the biologic relevance of this small increase in goblet cells appears to be limited. It is uncertain if the goblet cell change in these four monkeys was a direct effect of dulaglutide or secondary to the nutritional or metabolic effects known to be induced by dulaglutide treatment. The potential for direct, receptor-mediated effects of dulaglutide on the ductal epithelium appears unlikely due to the lack of GLP-1 receptor expression in pancreatic duct epithelial cells using a well-characterized monoclonal antibody (Pyke et al. 2014). It is less clear if the effects of dulaglutide on food consumption, body weight, or general metabolic status could have contributed to the goblet cell alterations in this subset of monkeys. Bonnet monkeys provided mildly protein-deficient diets were observed to have a variety of structural changes in the pancreas, including ductular hyperplasia and goblet cell metaplasia, which supports the concept that nutritional factors can modify pancreatic duct morphology in the primate (Sandhyamani, Vijayakumari, and Nair 1999). In this protein-deficient model other structural changes such as acinar atrophy and islet cell hyperplasia were noted, changes that were not observed in the present study of dulaglutide.
The evaluations in the current study were primarily focused on the exocrine pancreas, and detailed study of the structure and function of the endocrine pancreas was not included. In the human autopsy series of patients administered incretin-based therapies (Butler et al. 2013a), an expansion of pancreatic α-cell mass and discrete α-cell lesions in a patient administered exenatide was observed, however, methodological concerns and reanalyses of these specimens have called these interpretations into question (Bonner-Weir, In’t Veld, and Weir 2014). Although the current study did not quantify α- or β-cell mass, no changes in the histologic appearance of islet cells were noted. Furthermore, the discrete focal proliferations of α cells described by Butler et al. would have been detected with the methods employed in this study (Butler et al. 2013b). The lack of changes based on qualitative histologic evaluation in the current study is consistent with lack of an effect on endocrine cell mass and proliferation rate in monkeys treated with liraglutide (Gotfredsen et al. 2014).
While there was no histological evidence of pancreatitis or preneoplastic lesions after administration of dulaglutide, the monkeys used in this study were young, healthy animals that did not have type 2 diabetes. It has been documented that humans with type 2 diabetes have a 1.5- to 3-fold greater risk of developing pancreatitis and a 2.1-fold greater risk of developing pancreatic cancer compared to individuals without type 2 diabetes (Garg, Chen, and Pendergrass 2010; Girman et al. 2010; Noel et al. 2009; Everhart and Wright 1995). Thus, it cannot be ruled out that monkeys with type 2 diabetes may have responded differently to dulaglutide. Furthermore, studies in primates may not always translate into similar findings in humans. Despite these limitations, it is important to note that the rodent data suggesting a proliferative effect on pancreatic ducts were also derived from young, nondiabetic animals (Gier et al. 2012). As such, the lack of findings in these nondiabetic monkeys provides evidence that the rodent findings are not predictive of similar effects in monkeys or humans.
Although the physiological mechanism is not well understood, one of the beneficial effects of GLP-1 receptor agonists in patients with type 2 diabetes is sustained weight loss (Buse et al. 2009; Drucker et al. 2008; Vilsboll et al. 2012). In line with this, monkeys administered dulaglutide for 12 months gained significantly less weight compared to controls. As observed with other GLP-1 receptor agonists, dulaglutide treatment caused reductions in food consumption resulting in secondary reductions in body weight relative to control monkeys. While the monkeys in this study were not obese, the observation that dulaglutide impacted weight gain is consistent with the known effects of GLP-1 receptor agonists.
In summary, this study represents one of the most thorough studies of the exocrine pancreas of the NHP following chronic dosing with a GLP-1 receptor agonist based on the relatively large group size, number of histologic sections examined, and prospective evaluation by a panel that included veterinary and human pathologists. Our data are in agreement with other monkey studies using GLP-1 receptor agonists and indicate that chronic dosing of nondiabetic primates with dulaglutide does not induce inflammatory or preneoplastic changes in the exocrine pancreas in monkeys.
Footnotes
Acknowledgments
The authors of this article would like to acknowledge Whitney Sealls (Eli Lilly and Company) for writing and editorial assistance and Jin Xue (Covance) for performing the Ki67 immunohistochemistry, and Steve Van Adestine (Covance) for digital image preparation.
Author Contribution
Authors contributed to conception or design (JV, RB, SS); data acquisition, analysis, or interpretation (JV, RB, JB, JM, SS, TR, TP, TJR, PS, GK); drafting the manuscript (JV, RB); and critically revising the manuscript (JB, JM, SS, TR, TP, TJR, PS, GK). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. JLV and TJR are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. SDS performed the initial microscopic evaluation of the pancreas, counted positive cells in Ki67-labeled sections of pancreas, and reviewed/edited the manuscript. TJR, PWS, and GK participated in the histopathological analysis of the pancreas sections including the Ki67-stained ducts, discussions on data analysis and interpretation, and editing of the research abstract and manuscript. All authors provided final manuscript approval.
Author Note
Some information presented in this study was presented in abstract form at the National Institute of Diabetes and Digestive and Kidney Diseases workshop on Pancreatitis-Diabetes-Pancreatic Cancer held in Bethesda, Maryland, on June 12–13, 2013. Paul W. Snyder is now employed by Experimental Pathology Laboratories, Inc., Sterling, VA.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JLV, RAB, JLB, and JAM are employees of Eli Lilly and Co. SDS, TP, and TR are/were employees of Covance Inc., which has a contractual relationship with Eli Lilly and Co. TJR, PWS, and GK are consultants for Eli Lilly and Co.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Eli Lilly and Company.
