Abstract
Cytoplasmic vacuolation of sensory neurons has been reported to occur within the dorsal root ganglia in studies investigating various neuropathic conditions including the effects of neurotoxic chemicals. In this study, we investigated this lesion in adult (98–119 days old) male Long-Evans rats, after multiple exposures to two organophosphates (tri-ortho-tolyl phosphate [TOTP] and chlorpyrifos) and the modifying effects of concurrent corticosterone. Tri-ortho-tolyl phosphate was administered by gavage (75, 150, or 300 mg/kg) every other day between days 14 and 28 and between days 49 and 63, chlorpyrifos (60 mg/kg) was administered subcutaneously on days 7 and 42, and corticosterone was provided in the drinking water throughout the study at a concentration of 400 µg/mL. Although relatively uncommon, there was an increase in frequency of cytoplasmic vacuoles seen in treatment groups having multiple exposures to TOTP. They were characterized as peripherally located, single-limiting membrane-bound structures in the neuronal perikarya. There was no associated cell death, even when vacuoles were large. This is the initial report of an association of this change following exposure to neurotoxic organophosphates.
Introduction
Cytoplasmic vacuolation of sensory neurons is a striking finding that has been reported to occur in the dorsal root ganglia following exposure to several toxic agents (Beiswanger et al. 1993; Cavaletti et al. 2007; Jaatinen et al. 1994; Melli et al. 2008), and in diabetes mellitus (Kamiya et al. 2006), traumatic axonal injury (Groves et al. 1997), and aging (Groves and Scaravilli 2005). They also have been seen at very low incidences in normal animals (Groves and Scaravilli 2005). In this report, we demonstrated that cytoplasmic vacuoles may also develop within neurons of the dorsal root ganglia as a result of organophosphate-induced delayed neurotoxicity (OPIDN).
Organophosphate-induced delayed neurotoxicity usually develops several days to weeks after exposure to certain organophosphate esters and is associated with post-dosing neurotoxic esterase inhibition (Ehrich and Jortner 2002). In hen and rat models, lesions consist of bilateral axonopathy progressing to Wallerian-like myelinated fiber degeneration (Jortner et al. 1989; Jortner et al. 2005; Veronesi 1984), with distal regions of long myelinated fibers profoundly affected. This pattern has been noted in rats with experimentally induced OPIDN (Jortner et al. 2005; Veronesi 1984). In these studies, lesions were prominent in fibers of the sural nerve and distal gracile fasciculus neurites originating from dorsal root ganglia neurons. In this study, we evaluated vacuoles within the soma of rat dorsal root ganglion neurons associated with exposure to tri-ortho-tolyl phosphate (TOTP), the organophosphate classically used for induction of OPIDN (Cavanagh 1954) and chlorpyrifos, an organophosphate that has been considered to possibly play a role in human neurologic illness (Richardson 1995).
Materials and Methods
Neuropathological analysis was performed on dorsal root ganglion sections from a previous study (Jortner et al. 2005). These sections were from adult male Long-Evans rats treated with different combinations of two organophosphates (TOTP and chlorpyrifos), as well as corticosterone. Animals were maintained in the Virginia Tech vivarium under controlled conditions with twelve-hour light/dark cycles. Pelleted feed (Harlan Tek-lab 2018 pellet) and tap water were provided ad libitum. Doses were selected on the basis of inducing a range of neurologic effects, and dosing schedules were designed to minimize the incidence of death owing to acute organophosphate toxicity. Routes of administration were chosen based on classical experimental designs of previous studies (Ehrich et al. 1995; Richardson 1995). All protocols were in compliance with Virginia Tech’s Animal Welfare Assurance and approved by Virginia Tech’s Institution of Animal Care and Use Committee. Briefly, chlorpyrifos (in a 1 mL/kg corn oil vehicle) was administered as a single dose (60 mg/kg) on day 7 and day 42 by s.c. injection. Tri-ortho-tolyl phosphate (75, 150, or 300 mg/kg), in a corn oil vehicle, was administered every other day by oral gavage during two treatment courses between days 7 and 28 and days 42 to 63 (totaling fourteen doses). Corticosterone was administered continuously from day 0 through day 90 in the drinking water (400 μg/mL w/v) as a model for chronic stress. The estimated corticosterone dose for each animal was 13.51 + 2.08 mg/day (mean + SD, n = 12) (Jortner et al. 2005).
Animals were sacrificed on days 28, 63, and 90 (the latter followed a four-week period free of organophosphate exposure). Cohorts were used for biochemical study, including brain neurotoxic esterase activity, as reported in Ehrich et al. (2004) and Jortner et al. (2005). Three to six animals from each group were sacrificed on day 28, 63, or 90 for neuropathologic study (Jortner et al. 2005). Animals were deeply anesthetized with intraperitoneal sodium pentobarbital, followed by transcardial perfusion-fixation via a perfusion pump (85–90 mL/min) using 4% paraformaldehyde and 0.2% glutaraldehyde in 0.1 M phosphate buffer (pH = 7.4) at room temperature; 2% osmium tetroxide in 0.1 M phosphate buffer was used as a post-fixation solution. Lumbar dorsal root ganglia were dissected from the perfused rats, and longitudinal sections were made. Sections were post-fixed in osmium tetroxide, embedded in epoxy resin, sectioned at a thickness of 1 μm, and stained with toluidine blue and safranin (as a counter-stain for collagen) for analysis with light microscopy.
For the present study, ganglia from rats sacrificed on day 90 (when the neuropathic changes were best developed); (Jortner et al. 2005) were evaluated. The rats remaining at this time point had either been dosed with 300 mg/kg TOTP (with or without chlorpyrifos and/or corticosterone) or were controls. Using light microscopy, total neuron counts were made for one section of one dorsal root ganglion from each of the rats. Neurons were counted either if the nucleus was identifiable or if a distinguishable satellite cell rim could be seen surrounding the cell. Cells exhibiting cytoplasmic vacuoles were also counted, and calculations were made to determine percentage of total cells as well as percentage of animals that had these vacuoles located within neuronal cell bodies.
Tissue blocks yielding sections exhibiting different stages of vacuole development were selected and prepared for transmission electron microscopy. These blocks were sectioned at 80-90 nm thickness, and the sections were picked up on copper grids and then stained with uranyl acetate and lead citrate. These were then examined with a Zeiss 10C transmission electron microscope.
No statistical analyses were performed for this descriptive study.
Results
In groups treated with TOTP (alone or in combination with chlorpyrifos, corticosterone, or both), cytoplasmic vacuoles were seen within the soma of dorsal root ganglia neurons using light microscopy (Figure 1 and Table 1). Light microscopy did not reveal any abnormalities in the dorsal root ganglion neurons from the control group (Figure 1E). In what was considered early development, the vacuoles were solitary, small, and peripherally located in the cell body (Figures 1A and 1B). In cells exhibiting later stages of development, the vacuoles increased in size and occupied more of the cellular volume (Figure 1C). In the most advanced stages of development, they were quite large and occupied the majority of the visible cellular volume (Figure 1D). Despite this finding, there was no associated cell death. Most of the affected neurons appeared to be of the large, light variety of dorsal root ganglia neurons (Rambourg et al. 1982).
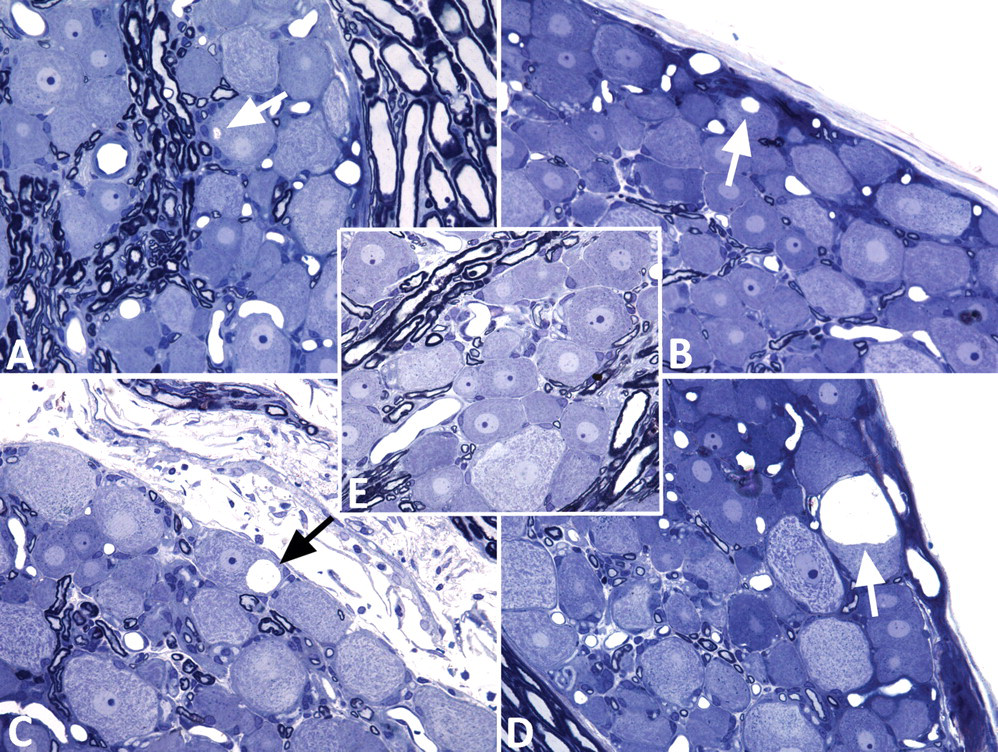
Light micrographs of dorsal root ganglia stained with toluidine blue and safranin. Progressive enlargement of neuronal somal cytoplasmic vacuoles is indicated by arrows in Figures 1A, 1B, 1C, and 1D. This enlargement was not seen in a ganglion from a control rat (Figure 1E). (A) tri-ortho-tolyl phosphate (TOTP) 300 mg/kg + chlorpyrifos 60 mg/kg; (B) TOTP 300 mg/kg + corticosterone (13.51 ± 2.08 mg/day); (C) TOTP 300 mg/kg + chlorpyrifos 60 mg/kg; (D) TOTP 300 mg/kg + chlorpyrifos 60 mg/kg.
Morphometric data for day 90.
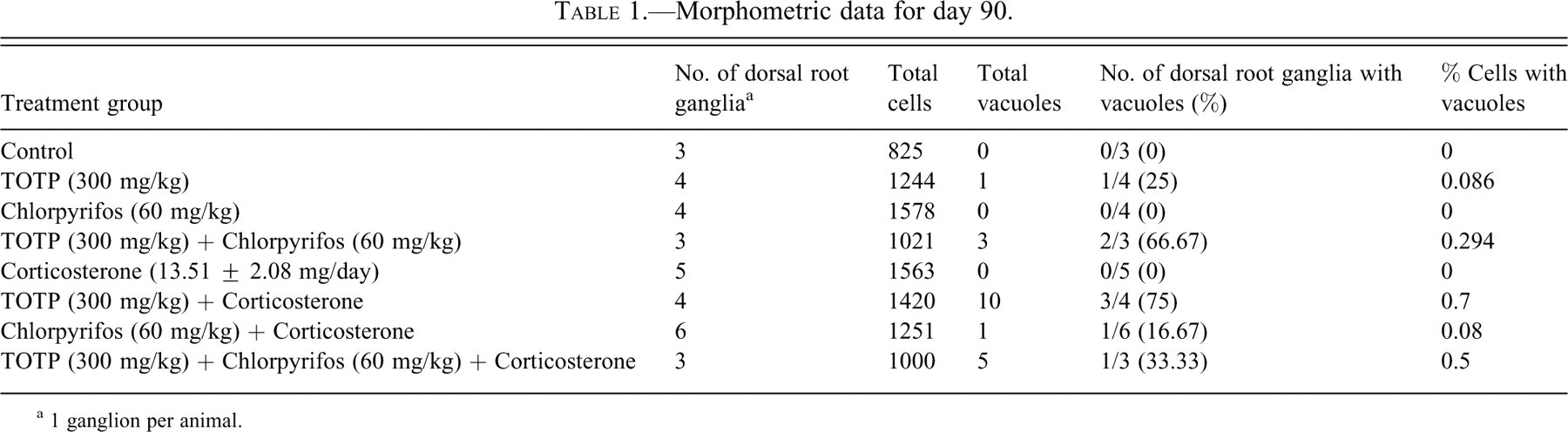
a 1 ganglion per animal.
Transmission electron microscopy did not reveal any abnormalities in the neurons of control rats (Figure 2A). Affected cells in the animal groups exposed to TOTP were confirmed to have vacuoles in the neuronal cytoplasm, and not within the satellite cells (Figures 2B and 2C). Even in instances where the vacuoles were located close to the neuronal cellular membrane, it was observed that profiles of the vacuoles were entirely surrounded by the neuronal cytoplasm (Figure 2C). Vacuoles had a single-limiting membrane that did not contain embedded ribosomes (Figure 2D). Analysis at different points of development showed that this membrane was present when the vacuoles were relatively small, and that it continued to be maintained throughout the later stages. Fine cellular debris was consistently observed within the vacuole lumen, and adjacent cytoplasm protruded into the large vacuoles (Figure 2D).
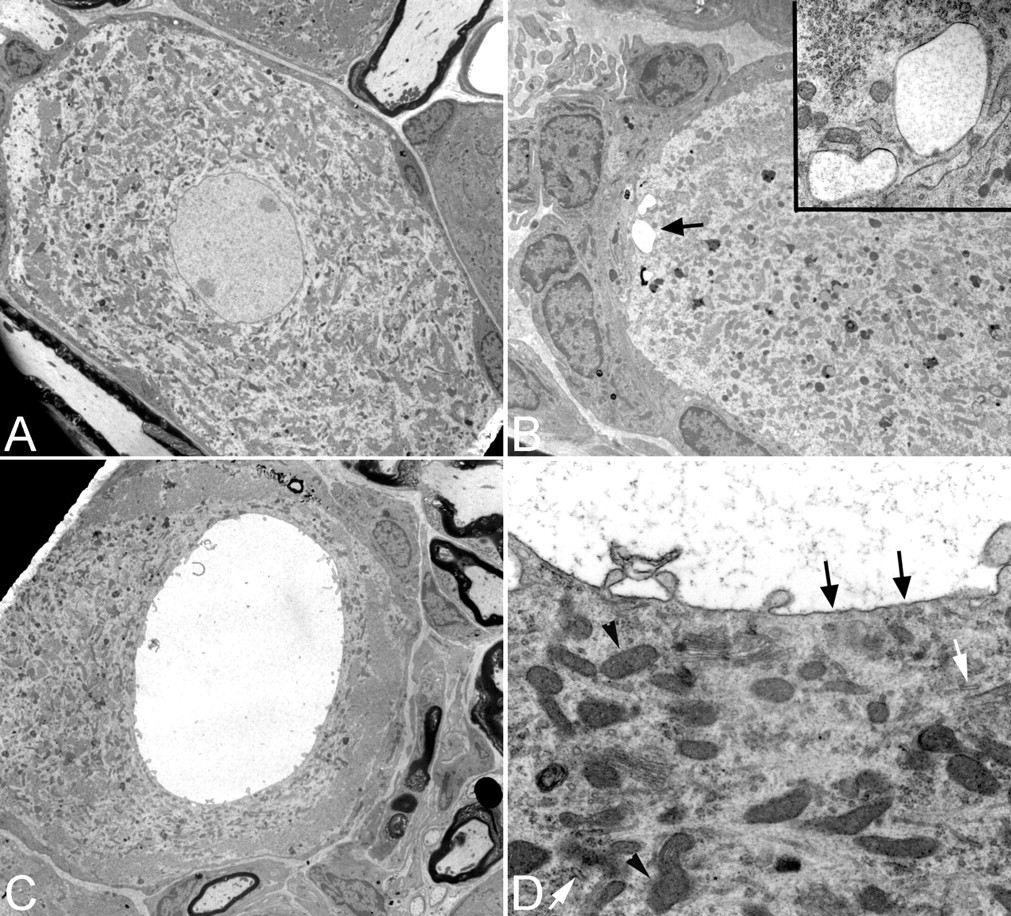
Electron micrographs of large neurons from the lumbar dorsal root ganglia taken from a control animal (Figure 2A) and from an animal exposed to tri-ortho-tolyl phosphate (300 mg/kg) and chlorpyrifos (60 mg/kg) (Figures 2B–2D). (A) Large, light neuron from a control rat (original magnification = 1600×); (B) small vacuoles in the periphery of a neuron (arrow) (original magnification = 3150×), seen at greater magnification in the inset (magnification = 31,500×); (C) large cytoplasmic vacuole (original magnification = 2000×); (D) membranous lining (arrows) of a vacuole, with surface protrusions. Note lumen containing fine cellular debris. (original magnification = 20,000X). Arrow heads denote normal mitochondria, white arrows denote rough endoplasmic reticulum.
Examination of organelle morphology in these vacuolated dorsal root ganglia neurons revealed very few abnormalities. There was no detectable swelling of the endoplasmic reticulum, Golgi apparatus, or mitochondria associated with the vacuoles (Figure 2D). The location and arrangement of organelles were not noticeably altered when the vacuoles were small. The location of organelles did not appear to be affected until the vacuoles became large, which resulted in displacement of the cytoplasm (as noted previously with light microscopy). Affected cells did not exhibit recognizable signs of death (as also noted with light microscopy), regardless of the severity or stage of development of the vacuoles.
Except for one rat exposed only to chlorpyrifos and corticosterone (see below), morphometric analysis showed that vacuolated neurons were restricted to animals receiving TOTP, either alone or in combination with chlorpyrifos, corticosterone, or both chlorpyrifos and corticosterone (Table 1). These vacuoles were relatively uncommon. They were not noted in groups exposed to chlorpyrifos alone, or corticosterone alone. There was one vacuole in the group exposed to chlorpyrifos and corticosterone without TOTP (Table 1).
Discussion
Several studies have reported the occurrence of cytoplasmic vacuolation of sensory neurons within the dorsal root ganglia under a variety of neuropathologic and toxicologic conditions. Chemotherapeutic agents, such as paclitaxel, cisplatin (Melli et al. 2008), and bortezomib (Cavaletti et al. 2007) have been shown to induce numerous small neuronal, cytoplasmic vacuoles that originate from damaged mitochondria. Similarly, after exposures to toxicants like 3-acetylpyramide (Beiswanger et al. 1993) and acrylamide (Groves and Scaravilli 2005; Lehning et al. 1998; Schaumberg and Berger 1993), sensory neurons have been reported to develop many small vacuoles as a result of inhibition of NAD/NADP-dependent pathways and decreased Na/K-ATPase activity, respectively. These vacuoles differ from the large vacuoles we report. Such large, solitary vacuoles have been reported to occur in the sensory neurons of control animals, albeit at very low incidences (~1 vacuolated neuron/10,000 neurons counted) (Groves et al. 1997). However, when axonal transport has been chemically or physically disrupted, the ratio of vacuolated neurons to nonvacuolated neurons has been shown to increase beyond that seen in controls (Groves and Scaravilli 2005). These include studies of peripheral axonal transection (Groves et al. 1997), ligation (Groves et al. 1997), and compression (Tang et al. 2009) injuries. In addition, chemical neurotoxicity with capsaicin (Hiura and Ishizuka 1989), ethanol (Jaatinen et al. 1994), and pronase (Groves and Scaravilli 2005) has also elicited these large dorsal root ganglia neuronal vacuoles. At least some of these compounds have been shown to impair axonal transport (Kawakami et al. 1993; Malatova and Cizkova 2001). Disrupted axonal transport has been reported in hens with OPIDN (Moretto et al. 1987). In the current study, we have shown that chronic TOTP exposure, giving rise to peripheral distal axonopathy and fiber degeneration (Jortner et al. 2005), may induce cytoplasmic vacuolization of dorsal root ganglion neurons, which is morphologically consistent with that seen in disease states associated with impaired axonal transport.
Although there was an increase in frequency of vacuolar lesions in TOTP-exposed groups compared with controls, it is notable that the frequency was still relatively low. The reported incidence for vacuolization of sensory neurons varies among different studies depending on the chemical or pathological condition being investigated, but previously reported morphometric data have shown that frequencies of vacuolated cells can range from relatively high (>10% of total cells, as in the case of NMDA antagonist administration [Auer and Coulter 1994]), to relatively low (1% to 2% of total cells, as in the case of nerve transection [Groves et al. 1997]). The data from our current study revealed that the frequency of vacuolated neurons/neurons counted was much lower (between 0.086% and 0.7%) (Table 1) than reports of other studies that made similar vacuolar frequency calculations under different pathological conditions (Groves and Scaravilli 2005). This low incidence is similar to that reported by Groves et al. (1997) under conditions of nerve-crush injuries (where vacuole frequencies were as low as 0.09%). Groves and colleagues concluded that the difference in vacuole frequencies between transected and crushed nerves was based on differences in axonal regenerative capabilities. In nerve crush, the axon is locally destroyed and leads to distal Wallerian degeneration. However, Schwann cells and their basal lamina are preserved, thus maintaining the original axonal path of connectivity (Groves et al. 1997). Subsequent proliferation of Schwann cells within this basal lamina–bound tube (Band of Büngner) allows for faster and more efficient regeneration of the injured axon when compared with axons of nerves with damaged and displaced Schwann cell sheaths (as in the case of nerve transection). Previous work has shown that following OPIDN exposure, there is efficient regeneration of injured peripheral axons similar to that seen in nerve-crush injuries (Jortner et al. 1989). Also, regenerating peripheral nerve fibers were noted in TOTP-exposed animals in a neuropathology report of the present study (Jortner et al. 2005). Thus, it is not surprising that neurons in our rats with OPIDN and in those with nerve-crush injuries have similarly low frequencies of vacuoles, because neurons in both cases can regenerate their axons. We did find one vacuole in an animal exposed to chlorpyrifos and corticosterone without TOTP. However, this finding was likely attributed to the low-frequency occurrence of these vacuoles that can be found in control rats (Groves et al. 1997) or aging rats (Groves and Scaravilli 2005) rather than a result of chemically induced axonopathy.
Our previous report of components of this study indicates that chlorpyrifos and corticosterone did not induce axonal degeneration at the doses used in this experiment, nor did they appear to interact with TOTP in enhancing axonal degeneration (Jortner et al. 2005). This result is consistent with our current findings that show that neither chlorpyrifos nor corticosterone induces vacuole formation when administered alone. The fact that TOTP induced vacuole formation whereas chlorpyrifos did not is most likely because chlorpyrifos was administered in doses too low to induce OPIDN. This statement is supported by observations that chlorpyrifos, at levels administered in this study, did not inhibit hippocampal neurotoxic esterase (Jortner et al. 2005). Although chlorpyrifos is able to induce OPIDN, Richardson (1995) noted increased doses of chlorpyrifos will often cause mortality owing to acute cholinergic effects before OPIDN develops. Although the findings of the present study suggest an increased incidence of vacuoles where chlorpyrifos and corticosterone were administered with TOTP (TOTP alone = 0.086%; TOTP + chlorpyrifos = 0.294%; TOTP + corticosterone = 0.7%, TOTP + chlorpyrifos + corticosterone = 0.5%), the small sample size used in this descriptive study makes us hesitant to draw any definitive conclusions in this regard.
Although the intracytoplasmic neuronal vacuoles were morphologically distinct, we were unable to find evidence of their origin. Suggested origins vary among reported studies. However, it is most commonly thought that they arise from swollen intracellular organelles, including mitochondria (Auer and Coulter 1994; Cavaletti et al. 2007), endoplasmic reticulum (Auer and Coulter 1994; Groves and Scaravilli 2005), and the Golgi apparatus (Kamiya et al. 2006). The ultrastructural analysis in this study showed that there is a single, limiting membrane encompassing the vacuoles, which has been previously noted by Jones and Cavanagh (1984) as a sign that the vacuoles are fluid filled and potentially originate from cellular organelles. However, a definitive origin for the vacuoles was not determined. We observed no evidence of a prior double membrane or the presence of cristae, making it unlikely that mitochondria are the source. The rough endoplasmic reticulum is also unlikely to be the source because of the lack of ribosomes embedded in the vacuole membrane. We found no evidence to eliminate the smooth endoplasmic reticulum or the Golgi apparatus as the source, thus they remain potential candidates for the origin.
The most intriguing aspect of these striking vacuoles is that they did not appear to coincide with or relate to cell death and/or degeneration. Ultrastructural analysis of affected neurons did not reveal any morphological signs of apoptosis or other forms of cell death, regardless of how severely the cells were vacuolated. This was a particularly surprising observation in cells wherein the vacuoles sometimes occupied the majority of the observed cell volume.
This was the initial observation, indicating that long-term exposure to the organophosphate TOTP may induce vacuoles within dorsal root ganglion neuronal cell bodies. This is likely related to the peripheral nerve myelinated fiber degeneration that was elicited by this toxicant, as noted in an earlier report of this study (Jortner et al. 2005).
Footnotes
Acknowledgments
This study was funded in part by DAMD17-99-1-94-89, US Army Medical Research and Material Command. Support for T. Rogers-Cotrone was provided by the Institute of Critical Technology and Applied Science of Virginia Tech.
