Abstract
While many areas of radiation protection have formed so-called ‘platforms’ in Europe which provide strategic research agendas for their areas of interest, this did not happen for a long while for medical exposure, which is the application of ionising radiation that causes the greatest man-made exposure, at least in first world countries. Finally, in 2015, a European medical radiation protection strategic research agenda was set up, and a corresponding platform was launched in 2016. This was named ‘EURAMED’ – the European Alliance for Medical Radiation Protection Research. In its strategic research agenda, EURAMED defined its vision for medical radiation protection and the corresponding research needed. Five major topics were identified, ranging from measurements of medical application-related parameters such as exposures and image quality and radiation biology aspects relevant for medical applications to individual optimisation strategies, to optimal use of techniques and harmonisation of practises, and finally to justification of the use of ionising radiation in medicine, all based on sufficient infrastructures for quality assurance. The ultimate goal is to reduce radiation exposure and risk individually for patients and staff by interdisciplinary research between clinicians, physicists, and engineers. Therefore, it is essential that the results are translated into clinical practice.
Keywords
1. BACKGROUND
Radiation protection research in Europe has been dominated strategically by the platforms MELODI, EURADOS, NERIS, and ALLIANCE for nearly a decade following publication of the report of the High Level Expert Group on Low Dose Ionising Radiation (HLEG, 2009). However, medical applications of ionising radiation represent the greatest man-made source of ionising radiation exposure to European inhabitants. Therefore, following an initiative from MELODI, EURADOS, and five European medical associations – European Association of Nuclear Medicine (EANM), European Federation of Organisations for Medical Physics (EFOMP), European Federation of Radiographer Societies (EFRS), European Society of Radiology (ESR), and European Society for Radiotherapy and Oncology (ESTRO) – as well as the European-Commission-funded project OPERRA, the first European strategic research agenda (SRA) for medical radiation protection was elaborated. While this SRA was approved by the boards of the five medical associations mentioned above, preparations were made for establishing a platform like the other four (MELODI, EURADOS, NERIS, and ALLIANCE) for medical radiation protection research in Europe in collaboration with those other platforms that are interested in single aspects of such research. This platform, named the ‘European Alliance for Medical Radiation Protection Research’ (EURAMED), was officially launched in October 2016 during Radiation Protection Week in Oxford, UK. At that time, it was driven as a joint initiative of the European Institute for Biomedical Imaging Research. In October 2017, EURAMED became a legal entity in its own right. From 2018, EURAMED will be open for membership applications. Meanwhile, EURAMED is collaborating with the other platforms to constitute a roadmap for radiation protection research in Europe, and is also setting up its own roadmap focusing specifically on medical radiation protection research. This paper will describe the vision for medical radiation protection (research) based on the SRA, the first ideas for the specific roadmap, and the mission and vision statements of EURAMED.
2. MISSION AND VISION OF EURAMED
EURAMED was launched based on defined mission and vision statements taken from its statutes (EURAMED, 2017).
2.1. The vision statement
EURAMED’s vision is ‘to lead the European research activities in medical radiation protection, and to assume an umbrella function for the harmonisation of practice to advance the European radiation protection safety culture in medicine.’
2.2. The mission statement
EURAMED’s mission is:
‘to jointly improve medical care through sustainable research efforts in medical radiation protection; to serve as a platform for medical radiation protection research, linking researchers and clinicians, adopting a harmonised approach to lobbying at European level to impact the European research funding landscape; and to develop an aligned approach and response to European research calls.’
3. STRATEGIC RESEARCH AGENDA AND ROADMAP APPROACH
As described above, EURAMED (and its predecessor group) has developed an SRA, and is currently preparing a roadmap to describe the way to achieve significant benefits in terms of radiation protection for exposures in the medical context.
3.1. The strategic research agenda
The SRA is published by EANM, EFOMP, EFRS, ESR, and ESTRO under the title ‘Common strategic research agenda for radiation protection in medicine’ (EANM, EFOMP, EFRS, ESR, ESTRO, 2017). Many of the following statements are taken from this document.
EANM, EFOMP, EFRS, ESR, ESTRO, (2017) highlights the main messages related to research in the field of medical radiation protection. These are to:
‘overcome the fragmentation of medical radiation protection research in Europe; identify research areas of joint interest in the field of medical radiation protection; improve the use of ionising radiation in medicine; collect stakeholder feedback and seek consensus; and emphasise the importance of clinical translation and evaluation of research results.’
EANM, EFOMP, EFRS, ESR, ESTRO, (2017) states that ‘the research topics considered necessary and most urgent for effective medical care and efficient in terms of radiation protection are summarised in five main themes:
Measurement and quantification in the field of medical applications of ionising radiation Normal tissue reactions, radiation-induced morbidity and long-term health problems Optimisation of radiation exposure and harmonisation of practices Justification of the use of ionising radiation in medical practice Infrastructures for quality assurance’
The content of each topic will be summarised below.
3.1.1. Measurement and quantification in the field of medical applications of ionising radiation
In this topic, radiation protection research requires previous research on measurement and quantification to characterise exposures, look for individual exposures of patients, determine the uncertainties related to exposure characterisation, and develop quality metrics for image quality and therapy description. Regarding exposure determination, it is necessary to investigate which exposure characteristics can be meaningful in medical applications of ionising radiation, particularly due to the very pronounced localisation of exposures resulting in variations of absorbed doses of orders of magnitude, even in single organs (Schlattl et al., 2007). There is obviously a large common basis of interest in this respect with EURADOS. Further details can be found in the SRA.
3.1.2. Normal tissue reactions, radiation-induced morbidity, and long-term health problems
This topic reflects the biological research laying foundations for effective radiation protection in medical applications of ionising radiation. It is therefore strongly linked with the interests of MELODI. The necessary research includes the following aspects: exposure-associated cancer risk – dose, dose distribution, and dose-rate dependence; non-cancer effects in various tissues and radiobiology-based effect models for individual morbidity endpoints; individual patient-related radiation sensitivity and early biomarkers of response and morbidity; and radiobiological mechanisms of radiation-induced side effects and protective strategies.
3.1.3. Optimisation of radiation exposure and harmonisation of practices
The goal of optimising radiation exposure in medical application is one of the overarching goals of radiation protection research in the medical field. It needs to consider how to transfer the results into clinical practice, and how to harmonise this throughout Europe. This is particularly important because exposure conditions due to medical applications vary widely between different European countries, as shown in Fig. 1 (European Commission, 2014), and even within single member states. Research should therefore focus on: patient-tailored diagnosis and treatment; full exploitation and improvement of technology and techniques; clinical and dose-structured reporting; and protection of staff, patients, carers, and the general public.
Per caput effective doses for different countries. The relative contributions of plain radiography, fluoroscopy, computed tomography, and interventional radiology are shown. BE, Belgium; LU, Luxembourg; HU, Hungary; IS, Iceland; DE, Germany; EE, Estonia; NO, Norway; FR, France; CH, Switzerland; PT, Portugal; IT, Italy; ES, Spain; UA, Ukraine; CY, Cyprus; CZ, Czech Republic; EL, Greece; PL, Poland; LT, Lithuania; ME, Montenegro; LV, Latvia; DK, Denmark; AT, Austria; IE, Ireland; RS, Serbia; SE, Sweden; SK, Slovakia; MK, Macedonia; HR, Croatia; MT, Malta; SI, Slovenia; NL, Netherlands; FI, Finland; BG, Bulgaria; UK, United Kingdom; RO, Romania; MD, Moldova. For EL, data for the contributions of fluoroscopy and interventional radiology were not available. Source: European Commission (2014).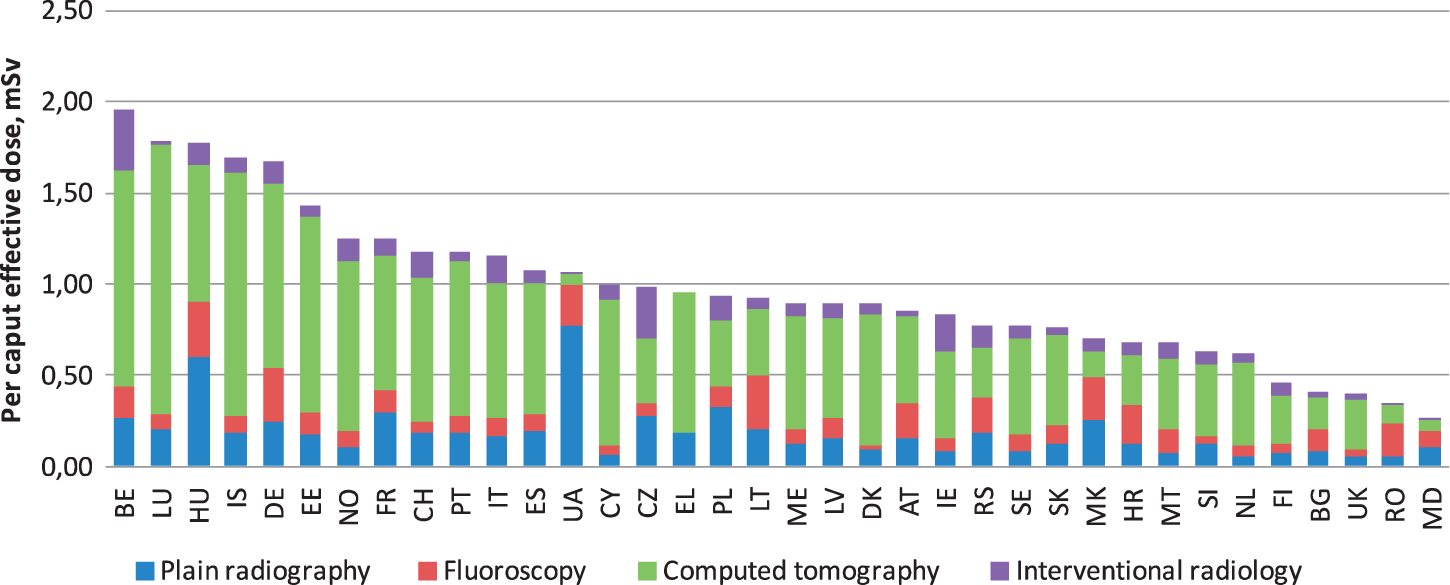
3.1.4. Justification of the use of ionising radiation in medical practice
In the field of medical radiation protection, the principle of justification is one of the key pillars, as underlined in the European Basic Safety Standards Directive (Council of the Europan Commission, 2014). The main aspect is to weight the benefits against the risks of a procedure. In the future, a stronger focus will have to be placed on the patient’s right to influence the decision. Research in this area should include benefit/risk assessment and communication, and improvement of use of evidence-based guidelines. The former is obviously related to the interests of social societies and humanities research.
3.1.5. Infrastructures for quality assurance
EANM, EFOMP, EFRS, ESR, ESTRO, (2017) stated that ‘it is important to perform investigations on tissue reactions, optimisation procedures as well as risk and benefit evaluations, to rely on optimal, quality assured data, which are gathered under defined conditions and which are necessary for various reasons including legal questions pertaining or specific to the research to be performed. In addition, the clinical system of medical applications of ionising radiation has to be standardised (Bolch et al., 2009) and evaluated concerning its effectiveness in radiation protection’. Therefore, research should focus on: data coding, collection, and management; comprehensive medical database/imaging biobanks; developing key performance indicators for quality and safety; audit systems; and education and training metrics. These questions are obviously related to the approaches of the other four platforms.
3.2. Roadmap
The roadmap documents highlight the importance of harmonisation of practices, and how this can be fostered by medical radiation protection research as well as the need to develop methods for individualisation of applications of ionising radiation in medical diagnosis and therapy. For diagnostic procedures, the focus should be on those that might cause higher local or effective doses than those in standard static projectional radiography (e.g. of the thorax). The EURAMED internal roadmap is focusing on the need to follow the full chain of optimising radiation procedures for optimal radiation protection of patients and staff in medicine. This means that all necessary research to optimise a procedure has to be done in order to generate the greatest benefit for the population in terms of radiation protection. Due to financing constraints, this means that there is always a need for exemplary procedures which need to be focused on. Optimisation has to be based on quantitative metrices for exposure, but also for quantitative metrices for the outcome, as on biological aspects, on individual optimisation for single patients, on harmonised best practice use of the medical equipment available, and justification. Only such a global approach seems to guarantee the best possible results.
4. Approaches
The approach as defined by the EURAMED internal roadmap to look for exemplary procedures and optimise them along the chain that has been described within the five major topics of the EURAMED SRA should be applied to all types of medical applications of ionising radiation. This means that optimisation should be based on projects from the fields of diagnostic and interventional radiology, as well as from nuclear medical diagnostics and therapy, and radiation therapy. It is of great importance to cover research on modern technologies (e.g. ion or proton therapy, or new interventional procedures) and hybrid imaging systems (e.g. positron emission tomography/computed tomography or positron emission tomography/magnetic resonance imaging). It is also of great importance to evaluate which cases require the most modern (and expensive) technologies, and how the patients can be protected as well as possible if such technologies are not available. Methods need to be developed to decide the degree of individualisation that has to be performed in each case, and how this can be done in a standardised way. Approaches for this already exist in the European FP7 projects MADEIRA and ANDANTE, and in the current HORIZON 2020 MEDIRAD project.
