Abstract
The concept of lifetime radiation risk of stochastic detrimental health outcomes is important in contemporary radiation protection, being used either to calculate detriment-weighted effective dose or to express risks following radiation accidents or medical uses of radiation. The conventionally applied time-integrated risks of radiation exposure are computed using average values of current population and health statistical data that need to be projected far into the future. By definition, the lifetime attributable risk (AR) is an approximation to more general lifetime risk quantities and is only valid for exposures under 1 Gy. The more general quantities, such as excess lifetime risk (ELR) and risk of exposure-induced cancer, are free of dose range constraints, but rely on assumptions concerning the unknown total radiation effect on demographic and health statistical data, and are more computationally complex than AR. Consideration of highly uncertain competing risks for other radiation-attributed outcomes are required in appropriate assessments of time-integrated risks of specific outcomes following high-dose (>1 Gy) exposures, causing non-linear dose responses in the resulting ELR estimate.
Being based on the current population and health statistical data, the conventionally applied time-integrated risks of radiation exposure are: (i) not well suited for projections many years into the future because of the large uncertainties in future secular trends in the population-specific disease rates; and (ii) not optimal for application to atypical groups of exposed persons not well represented by the general population. Specifically, medical patients are atypical in this respect because their prospective risks depend strongly on the original diagnosis, the treatment modality, general cure rates, individual radiation sensitivity, and genetic predisposition. Another situation challenging the application of conventional risk quantities is a projection of occupational radiation risks associated with space flight, both due to higher radiation doses and astronauts’ generally excellent health condition due to pre-selection, training, and intensive medical screening.
An alternative quantity, named ‘radiation-attributed decrease of survival’ (RADS), known in past general statistical literature as ‘cumulative risk’, is recommended here for applications in space and medicine to represent the cumulative radiation risk conditional on survival until a certain age. RADS is only based on the radiation-attributed hazard rendering an insensitivity to competing risks or projections of current population statistics far into the future. Therefore, RADS is highly suitable for assessing semi-personalised radiation risks after radiation exposures from space missions or medical applications of radiation.
1. Conventional risk metrics
The current system of radiation protection (ICRP, 2007) is set to reduce detrimental stochastic effects due to radiation exposure to a practically achievable minimum. Correspondingly, risks associated with the harmful effects need to be quantified and used prospectively for assessment of radiation detriment due to stochastic effects, of which malignant neoplasms represent the major health concern.
For the purposes of expressing cumulative risk of detrimental effects of radiation exposure, the concept of time-integrated risk based on representation of cumulative failures (disease occurrences) within a certain period or lifetime has been in use for decades (e.g. Vaeth and Pierce, 1990; Kellerer et al., 2001). This concept is generally based on survival statistics methodology and requires, for the risk assessment, models of radiation risk per se and detailed statistical information on the population of interest, including demographic data (life tables) and health statistics (incidence rates of various diseases).
The goal of the present work is to briefly review metrics used to express prospective risks of radiation exposure for radiation protection of populations and of special or atypical groups, such as medical patients and space crew members, not well represented by the general population data.
1.1. Time-integrated or lifetime attributable risk
A risk metric widely and commonly applied to express future detrimental effects of radiation exposure (e.g. risk of stochastic effects outcomes such as malignant neoplasms) stems from survival statistics methodology (e.g. Selvin, 1996; Kalbfleisch and Prentice, 2002; Kleinbaum and Klein, 2012) and is termed the ‘time-integrated or lifetime attributable risk’ (AR) (Vaeth and Pierce, 1990; Thomas et al., 1992; Kellerer et al., 2001):
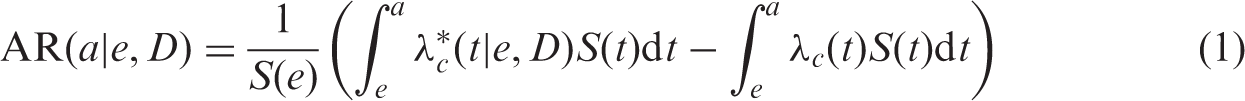
1.2. Risk of exposure-induced cancer
Eq. (1) assumes that radiation exposure does not affect the population’s survival chances and, therefore, the survival function for the general population can be used to express radiation risk for the exposed population. Generally, this assumption has some weaknesses, and exposure to higher doses of radiation (>1 Gy) may result in detrimental effects which can reduce the population survival chances. To account for the effect of reduced survival, the concept of risk of exposure-induced cancer (REIC) was introduced (UNSCEAR, 1994, 2000), replacing the survival function for the general population, 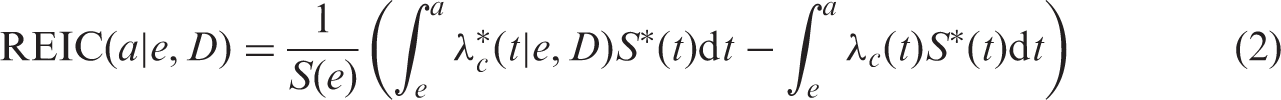
REIC assumes that the baseline incidence rates of the outcome of interest are the same in the exposed and the non-exposed populations. This assumption is valid only at low-dose exposures, where the radiation-attributed excess rate is small in comparison with the baseline rate and other competing hazards. Radiation exposure creates additional hazards and, correspondingly, reduces the number of competing outcomes in the population. Qualitatively, this effect is illustrated in Fig. 1, which shows that an attributed hazard results in attributed disease cases in the population, thus reducing the number of disease cases due to spontaneous cancer or other competing causes.
An illustration of cumulative risks in non-exposed (left) and exposed (right) populations. Additional hazard due to the radiation exposure reduces the cumulative risks of spontaneous cancers and of other – non-cancer-related – competing diseases or mortality causes.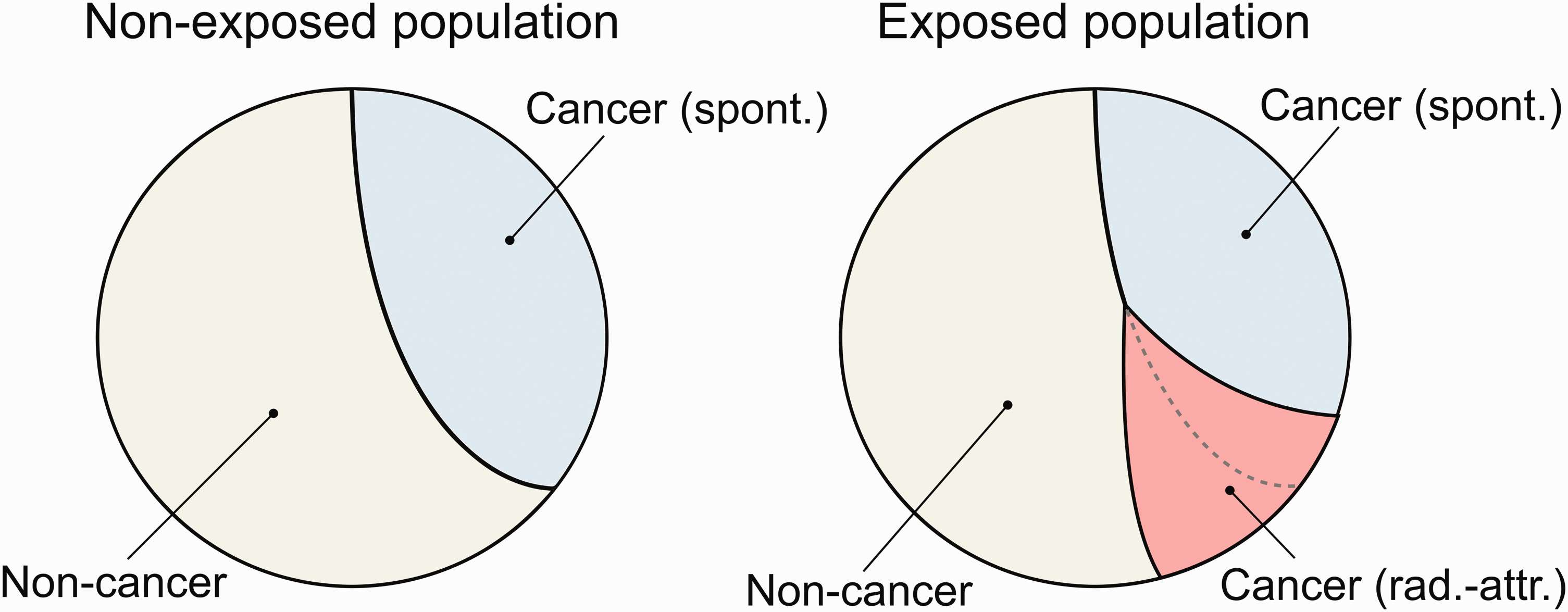
1.3. Excess time-integrated or lifetime risk
Another quantity for time-integrated risk, excess risk (ER), is robust against effects of competing risks and can better model the observations by expressing risk as the difference between the time-integrated risks of the disease in matching exposed and non-exposed populations:
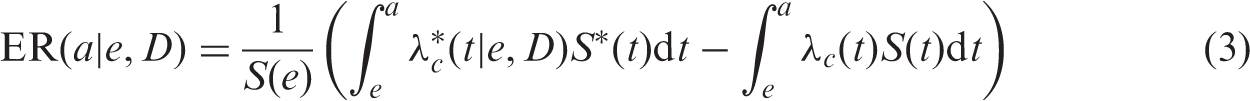
With the upper integration limit in Eq. (3) set to infinity, this quantity is conventionally termed the ‘excess lifetime risk’ (ELR).
ER can be regarded as a general quantity which correctly addresses integral risks of radiation exposure when comparing two identical exposed and non-exposed populations. However, even this definition, as discussed by Thomas et al. (1992), is not free from conceptual difficulties connected with radiation exposures at high doses (>1 Gy), because the total survival in the exposed population becomes significantly reduced in comparison with that in the non-exposed population, and the resulting ER [Eq. (3)] is decremented during integration to the full lifetime of the non-exposed population.
1.4. Limitations of the conventional risk metrics
AR is a low-dose approximation which works well for rare diseases, such as cancer, if the effect of radiation on the disease rate and, consequently, survival chances, can be neglected. When radiation-attributed risk is increased (by an exposure to higher dose) or the outcome of interest is a more prevalent cause, such as cardiovascular disease, the effects of attributed competing risks become essential and the simple approximation is no longer valid. Due to this, AR is not well suited for medical and occupational exposures related to doses >1 Gy, and may lead to significant overestimations of AR or paradoxes. For example, a radiation treatment plan which maximises survival chances of a patient (i.e. the ‘best’ treatment plan) will always be associated with the highest AR of the second primary cancer or other late effects.
Both REIC and ER consider survival in the exposed population; however, while the former expresses the radiation effect via excess rate in the exposed population alone, the latter derives the radiation effect as the difference of the outcome incidence rates in the non-exposed and exposed populations. REIC assumes the incidence rates of spontaneous disease (baselines) to be the same in the exposed and non-exposed populations. This assumption is not necessarily always valid, especially at high-dose exposures. ER is methodologically straightforward and appropriately represents a fraction of the exposed population which will develop the additional outcomes of interest in comparison with the identical non-exposed population. On the other hand, ER decrements when it is integrated to times exceeding the lifetime of the exposed population; REIC is free from this feature.
These risk metrics have common properties which lead to certain difficulties for prospective risk estimations. Computation of the risks [Eqs (1–3)] involves survival functions and disease incidence rates which are generally taken from demographic and health statistical data registered in the target population for a given year. These data are neither representative for generations nor are they properly suited for long-term risk projections and integrations during decades into the future.
Another shortcoming associated with Eqs (1–3) is due to use of the general population statistics for their computation. Estimations of radiation risks become less credible for persons whose survival chances are not represented by those of the general population (e.g. medical patients having a disease and undergoing radiation therapy, or occupationally exposed people who are pre-selected and undergo periodic medical checks or screening, such as astronauts or nuclear workers).
2. Attributable fractions and radiation-attributed decrease of survival
2.1. Survival chances and excess risk
The survival function can be factorised using incidence rates for specific outcomes, including diseases of interest:

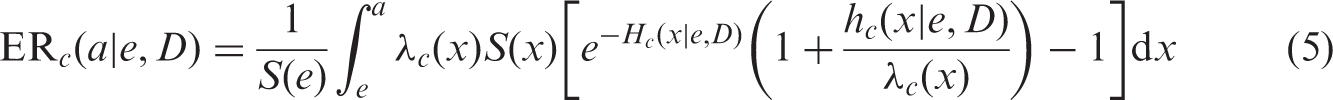
An analogue of Eq. (5) was presented previously in the UNSCEAR 2006 Report (UNSCEAR, 2006, Annex A, Appendix B), where it was noticed that this equation explicitly shows that the time-integrated radiation risk is inherently non-linear function of dose, even for the radiation risk models with linear dose-response effects. Apparently, for low-dose exposures, which result in small excess incidence rates, Eq. (5) converges to the conventional definition of AR (Eq. 1), which can therefore be regarded as a linear low-dose approximation.
2.2. Attributable and baseline survival fractions
The general formula for ER (Eq. 5) is computationally complex and is not free from the inherent reduction of ER at ages close to the lifetime of the general population. Alternatively, detrimental effects of radiation exposure can be expressed via reduction of survival chances due to radiation-attributed or spontaneous diseases. For this purpose, the baseline and attributable survival reduction fractions are recommended for application (see details in Ulanowski et al., 2019):
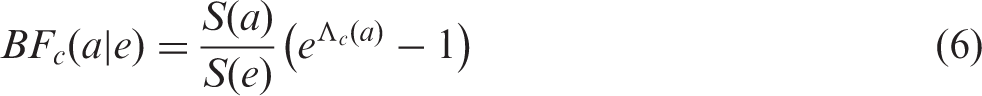
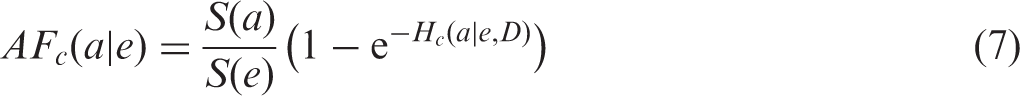
These quantities [Eqs (6, 7)] represent fractions of the population being alive and exposed at age
Both ER [Eqs (3, 5)] and the survival fractions [Eqs (6, 7)] represent the detrimental effect of spontaneous or attributed disease as a fraction of the population alive at age
2.3. Radiation-attributed decrease of survival
The attributable survival fraction [Eq. (7)] is computationally simpler than the formula for ER [Eq. (5)]; however, calculation of the former metric still requires the values of the survival function which can be influenced by many competing causes, especially at ages above 50 years. The additional radiation-attributed hazard reduces the survival curve, so the ratio of the attributable fraction (AF) [Eq. (7)] and the general survival function at age 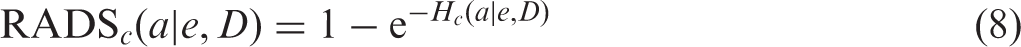
This represents the net effect of radiation-attributed hazard and was known in statistical literature as ‘cumulative risk’ (Esteve et al., 1994).
An illustration of the differences between the conventional risk metrics, AF, and RADS is given in Fig. 2, where the time-integrated estimates are shown for outcomes of all solid cancers for males and females after whole-body exposure at 20 years of age with a colon dose of 1 Gy. The details of the risk model and data used are given in the next section. For AF and RADS, the model uncertainty bands for the 95% confidence level are shown as shaded areas.
Comparison of various risk metrics for the incidence of all solid cancers following whole-body exposure at 20 years of age with a colon dose of 1 Gy for females (left) and males (right).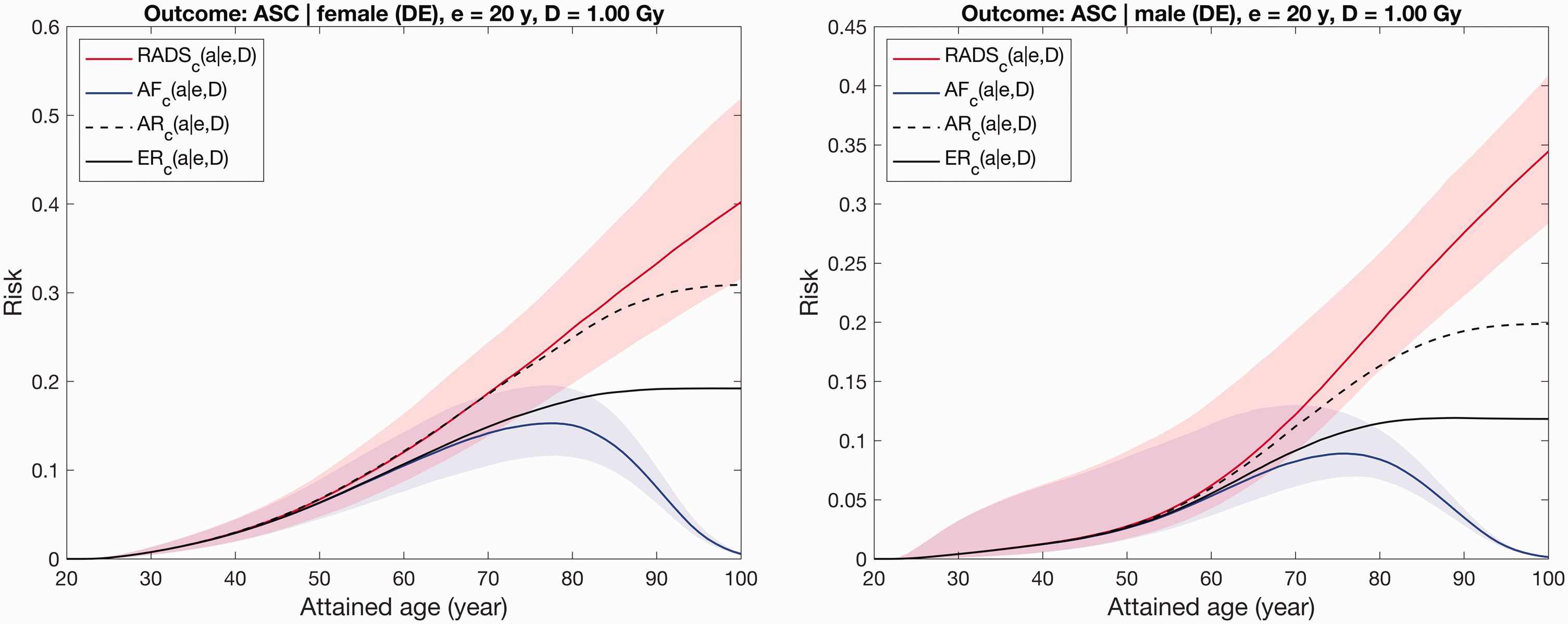
As seen from Fig. 2, all risk metrics are close to each other during ages below 50 years. At 50 years, when the other competing risks begin to reduce the general population’s survival curve, the risk estimates diverge. AR significantly overestimates in comparison with ER; this demonstrates an effect of radiation-attributed incidence on the survival function. AF follows the behaviour of the survival function and reduces at older ages exceeding 80 years. RADS demonstrates insensitivity to the competing outcomes and reduction of survival; the age dependence of RADS is defined by that of the applied models for excess incidence rate, so RADS reflects model-extrapolated expectations.
2.4. Effect of competing radiation-attributed risks
In the preceding subsections, Eqs (5–8) present situations when the only outcome of interest is radiation-attributed, and the other competing failure causes are due to spontaneous diseases or other mortality causes not attributed to radiation. This assumption seems reasonable at low-dose exposures when competing attributable excess rates are small, and only the outcome of interest affects the survival curve. For exposure to high doses, such an assumption loses plausibility as, besides the outcome considered, other competing radiation-attributed outcomes affect the survival and incidence of the considered outcome. Accounting for the effect of the competing radiation-attributed outcomes results in the following:
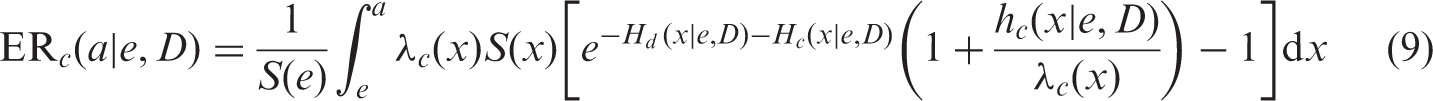
Correspondingly, RADS is also affected by the competing radiation-attributed risks and can be written as follows:
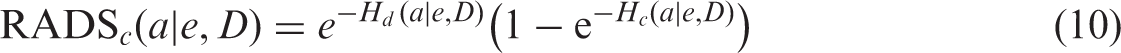
It follows from Eq. (10) that the competing radiation-attributed risks reduce RADS for the considered outcome of interest. That is, forecasting radiation risks for persons exposed to high doses (e.g. medical patients, astronauts) requires a coherent consideration of all possible radiation-attributed risks.
3. Examples and discussion
The sample calculations below were performed for several outcomes, ranging from the combined all solid cancer incidence to rare thyroid cancer incidence, and different radiation organ doses from 0.1 to 3 Gy, for both sexes. For the selected outcomes, all the risk metrics described above were used and their values are shown together for different times following radiation exposure.
Risk computation and uncertainty modelling methodology, used to produce the tables below, is described in Walsh et al. (2019) and Ulanowski et al. (2020). The risk model for all solid cancers is as given by Grant et al. (2017), without adjustment for smoking status. The risk model for thyroid cancer was taken from Jacob et al. (2014a,b). The female breast cancer risk model was based on the pooled study of Preston et al. (2002) and was applied as described in Ulanowski et al. (2020). The calculations were performed using the German-specific population statistics for 2013–2015 (Statistisches Bundesamt, 2016) and cancer incidence data for 2013–2014 from the German national cancer register (RKI-GEKID, 2017).
Time-integrated point estimates for different risk metrics for all solid cancer incidence following radiation exposure with dose of 3 Gy (colon) of a 10-year-old female.
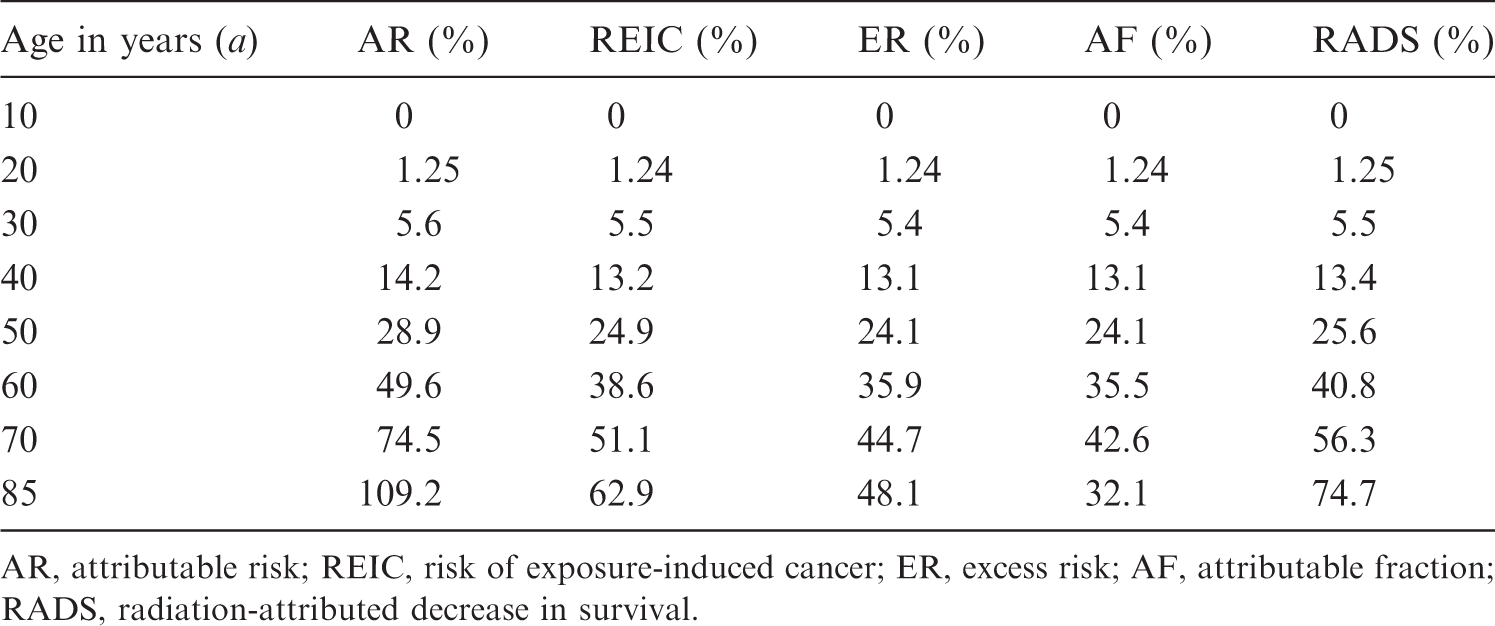
AR, attributable risk; REIC, risk of exposure-induced cancer; ER, excess risk; AF, attributable fraction; RADS, radiation-attributed decrease in survival.
Time-integrated point estimates for different risk metrics for female breast cancer incidence following radiation exposure with breast dose of 0.1 Gy (colon) of a 20-year-old female.
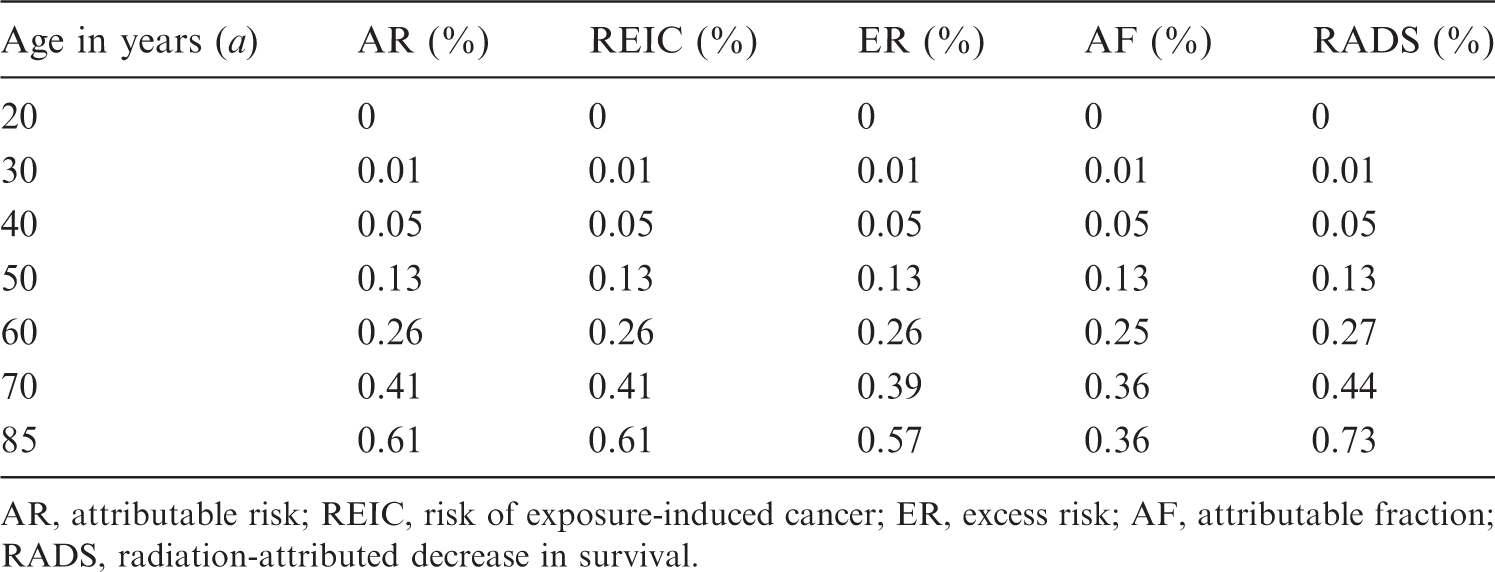
AR, attributable risk; REIC, risk of exposure-induced cancer; ER, excess risk; AF, attributable fraction; RADS, radiation-attributed decrease in survival.
Time-integrated point estimates for different risk metrics for thyroid cancer incidence following radiation exposure with a thyroid dose of 0.1 Gy of a 10-year-old female.
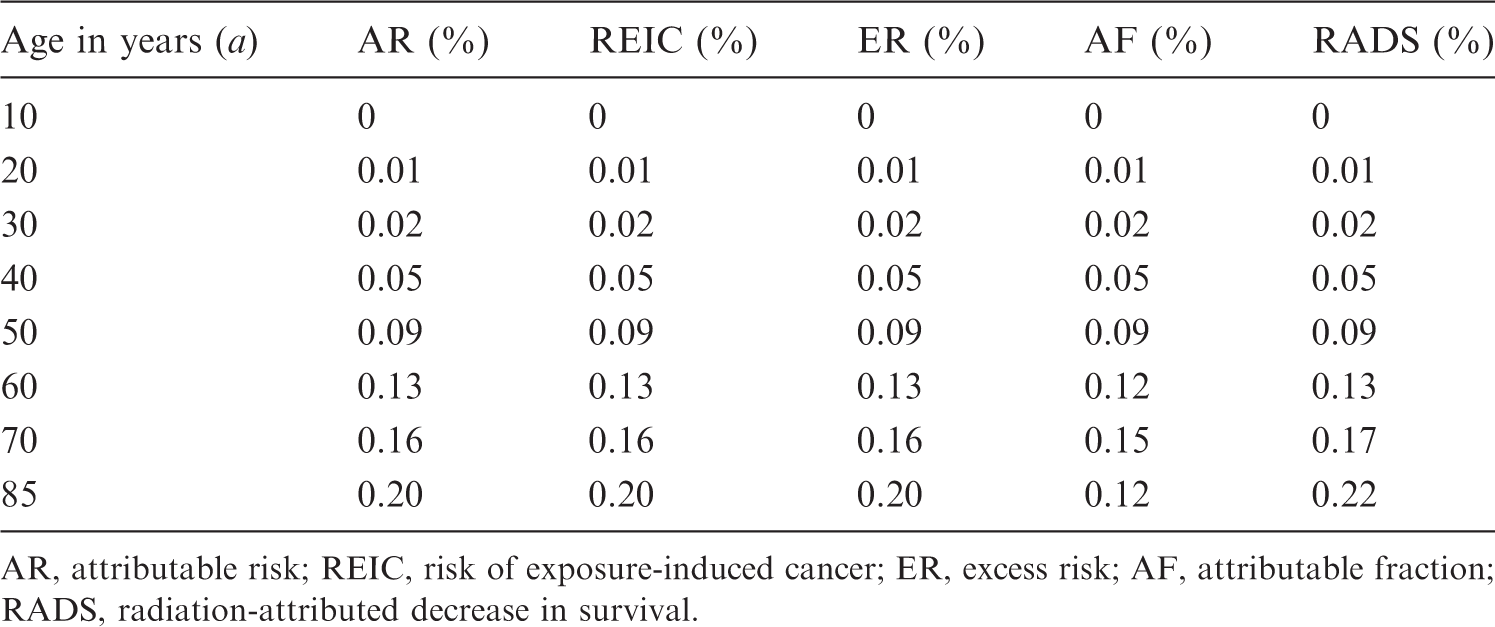
AR, attributable risk; REIC, risk of exposure-induced cancer; ER, excess risk; AF, attributable fraction; RADS, radiation-attributed decrease in survival.
The results of sample calculations indicate that, of the conventional risk metrics, only ER accounts for the effect of survival reduction due to attributed excess rates or competing outcomes. AR, being a linear approximation, is prone to strong overestimation of radiation risk which, in the case of high-dose exposure, may result in implausible values. Both REIC and ER account for the effect of radiation exposure on survival function, but they define the excess cases differently (the former metric implicitly assumes baseline incidence in the exposed population to be the same as in the non-exposed population, while the latter metric considers the radiation effect between the exposed and non-exposed matching populations). RADS, in this sense, is the most robust quantity as it, by definition, does not depend on survival changes, thus being better suited for risk projections for exposures at high doses or for populations or groups different from the general population.
4. Conclusions
Exposure to ionising radiation is known to increase risks of harmful health effects, of which malignant neoplasms are the main stochastic detrimental effects. In many situations of unavoidable radiation exposure, either occupational or medical, risks of additional future health effects are estimated and compared with spontaneous incidence observed in the contemporary population. Correspondingly, the conventional techniques of radiation risk assessment are based on the application of contemporary demographic and health data, and are representative for an average member of the current general population. However, medical patients treated with radiation are unlikely to be similar to the average, mostly healthy, member of the general population; people exposed occupationally, such as astronauts, are often selected based on their health status, they undergo periodical medical checks and screenings, and as such they are not well represented by the average member of the general population. Use of current, cross-sectional, population statistics for projection of lifetime radiation risks also brings significant uncertainties to the risk estimates due to unknown future changes of health and vital statistics. This paper reviews the conventional metrics used to express future radiation risks, demonstrates their limitations and difficulties with their use, and suggests an alternative quantity to express the risk, which is insensitive to competing risks and robust against unknown future changes in the population’s health and demographic data.
For risk projections, where future survival and health statistics are unknown, RADS is suggested and is complementary to conventional lifetime risk quantities. RADS represents the risk of radiation detriment alone and has the following advantages:
independence from current and unknown future temporal trends in population survival functions known at the time of estimation – only the estimated radiation-attributed incidence rate (hazard) is required for this quantity; aids in avoiding paradoxical situations in radiation therapy, because the same radiation dose applied for patients with cancer diagnosed at different stages will result in the same radiation risk of the second primary cancer, regardless of the differences in relative survival; and a higher degree of suitability for application in risk assessments for exposed but highly atypical populations (e.g. astronauts), where baseline rates and survival functions pertaining to the general population would be poor approximations (due to distinctly different levels of lifestyle factors such as smoking and fitness, pre-selection, and different levels of medical surveillance or cancer screening).
The suggested quantity, RADS, is better than conventional risk metrics suited for projections of personalised risks; however, individual variability or predisposition may bring significant uncertainty to such projections. RADS is conditional on an adopted radiation risk model, which is typically derived from an epidemiological cohort with a certain applicability domain; therefore, plausible risk projections using RADS are anticipated either within the applicability domain or by using radiation risk models with well-justified highly significant parameters.
RADS is not suggested as a replacement for conventional quantities used to communicate radiation risks for the purposes of radiation protection of populations exposed to low doses, such as AR or ER. RADS has its own niche – communicating radiation-attributable risks for atypical groups or individuals and application for radiation doses >1 Gy (e.g. interventional and therapeutic radiation exposures).
