Abstract
Radiation exposure from diagnostic medical imaging has increased in Korea. Radiological societies play a key role in radiation safety issues in Korea, including guidelines, accreditation, advocacy, scientific activity, and education. Any medical radiation exposure must be justified, and examinations using ionising radiation must be optimised. Education of referring physicians and radiologists is also important for justification. Medical physicists and radiographers have an important role to play in quality management and optimisation. Regulations are essential to control medical radiation exposure. Therefore, national organisations have made a significant effort to regulate and monitor medical radiation exposure using guidelines, accreditation, and even the law. Medical radiation exposure must be controlled, and this could be achieved by continuous interest from health professionals and organisations.
1. INTRODUCTION
1.1. Healthcare system in Korea
Korea has a public healthcare insurance system with mandatory registration of all healthcare providers and residents providing universal coverage. The public health system mainly relies on privately owned hospitals and is very extensive, covering a large range of medical situations. Nearly all aspects of the healthcare system are regulated by the Korean Government (Chun et al., 2009). Step-by-step expansion of coverage, coupled with demand for a higher quantity and quality of healthcare, have increased investment and resources, and placed heavy financial pressures on the system. Hospitals develop services that are not covered by national health insurance (NHI) in order to increase revenue. Medical services not covered by NHI are also regulated by the Korean Government (Fig. 1).
Overview of healthcare system in Korea. IHA, individual health assessment.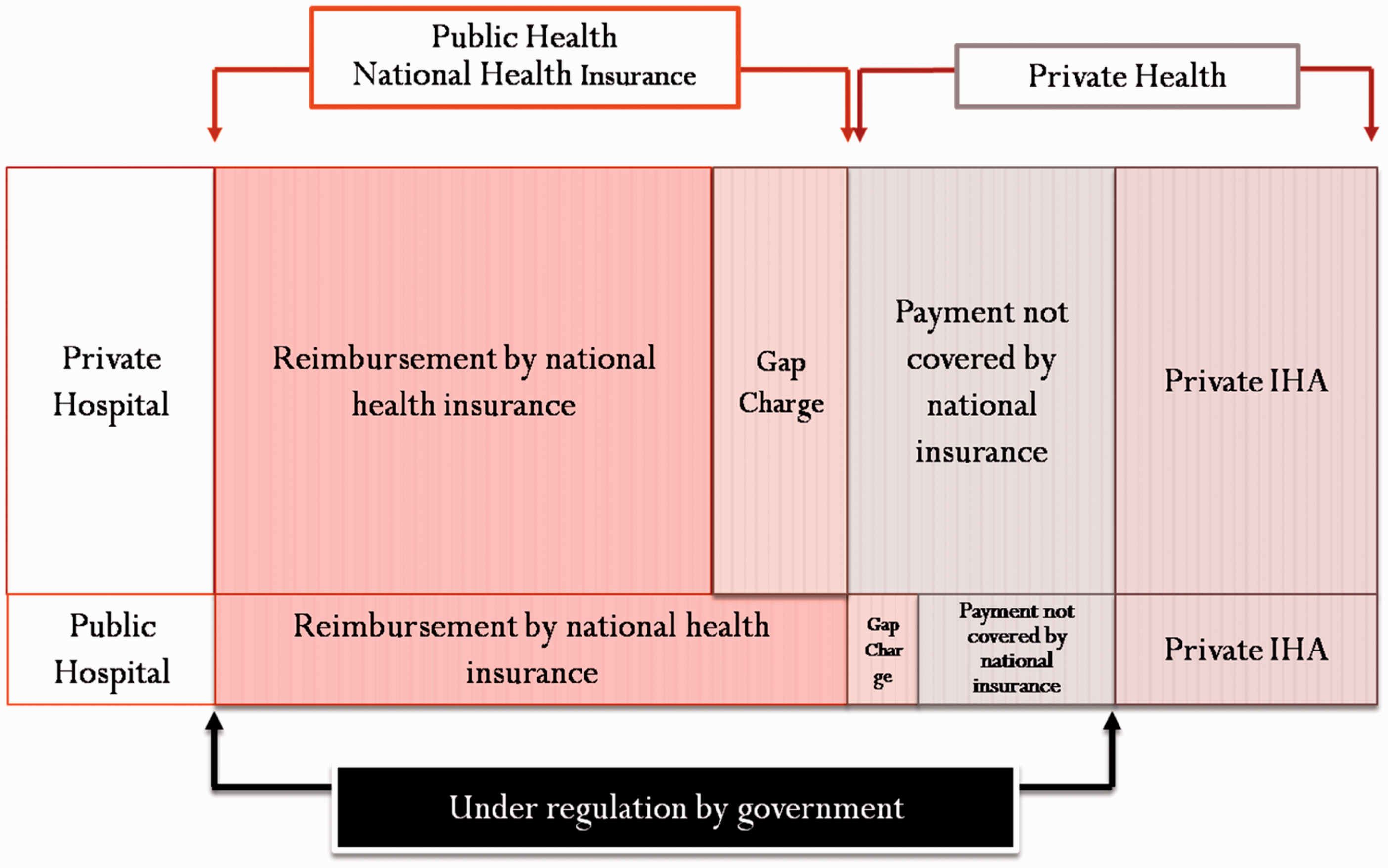
1.2. Current status of radiation exposure in Korea
Medical radiation exposure control system in Korea.

There are two components to the rules under the Medical Services Act: (1) safety control of diagnostic x-ray-generating equipment, and management of equipment and radiological protection facilities, regulated by the Korean Centre for Disease Control; and (2) installation and operation of quality assurance systems for computed tomography (CT), magnetic resonance imaging (MRI), and mammography. These cover the management of equipment, occupational exposure, and radiological protection facilities, but the current regulation is not focussed on patient safety.
Given the size of the population of South Korea, the country has many items of medical imaging equipment, which is indicative of the high number of imaging procedures being undertaken. The number of imaging procedures performed in Korea is comparable, or even higher, compared with the numbers of imaging procedures performed in Organization for Economic Cooperation and Development (OECD) countries. Korea has a very high rate of medical imaging equipment adoption. In 2011, Korea ranked fifth and sixth in the world in terms of the number of MRI and CT units per million population, respectively. However, the number of MRI and CT examinations per 1000 population in Korea seems to be relatively low (OECD, 2013). This is because examinations that are privately funded are not included, yet these represent the majority of examinations in Korea. As these privately funded examinations are highly commercial, quality control is very important in order to prevent abuse.
The annual number of diagnostic radiological examinations in Korea increased rapidly from 2006 to 2011; the total number of diagnostic radiological examinations and CT scans increased by an average of 8% and 14% y−1, respectively. The annual per-caput effective dose in the Korean population increased by 10% from 2007 to 2011. In 2011, the estimated annual per-caput effective dose was 1.4 mSv. The data in this study are probably underestimated because the main data source was NHI data, and did not include out-of-pocket procedures, such as voluntary health screening programmes (Kim, 2013).
Professional organisations are actively collaborating with many national agencies regarding quality and safety. In Korea, national agencies interested in medical radiation exposure include the MOHW; the Ministry of Science, ICT, and Future Planning; and the Ministry of Food and Drug Safety. The National Evidence-based Healthcare Collaborating Agency (NECA) and the Health Insurance Review and Assessment Service (HIRA) exist under MOHW. NECA and HIRA are creating guidelines together with the Korean Society of Radiology (KSR).
2. RADIATION EXPOSURE CONTROL SYSTEM FOR IMAGING STUDIES
2.1. Efforts for justification
Any medical radiation exposure must be justified, and examinations using ionising radiation must be optimised. The first principle is justification of medical exposures. Justification goes beyond optimisation, where awareness, appropriateness, and audit are used for the implementation of justification. Practically, knowledgeable and effective communication with patients (informed consent) and with healthcare professionals (education and training) is needed for awareness. It is vital to raise awareness about safety among health professionals. Communication with patients includes the provision of information about risks and benefits, and gaining informed consent. Appropriate criteria and referral guidelines can be used by physicians to make decisions regarding justification. Lastly, the importance of clinical audits to improve justification is emphasised. The adoption of clinical audits is encouraged through payment systems, accreditation programmes, etc.
There are three stages of radiation exposure control for imaging studies. The first stage is equipment control. In Korea, there are strict regulations regarding radiological protection of patients, implemented as the Radiation Protection Act of the Ministry of Food and Drug Safety in 1995 and the accreditation programme in 2004. The second stage is imaging quality control. In Korea, an accreditation programme exists for phantom test and clinical imaging evaluation; the Medical Act for CT, MRI, and mammography. This accreditation programme is similar to the Mammography Quality Standards Act of the USA.
As of January 2015, Korea has not organised guidelines comparable to the appropriateness criteria of the USA, or iRefer of the UK. Currently, KSR is actively developing clinical diagnostic imaging guidelines adapted from other guidelines. Over the last 2 or 3 y, several guidelines have been published using government funding, and several more guidelines are currently in development. Since 2011, KSR has published many guidelines for radiological imaging and quality management, and contrast management. In 2016, work commenced on the development of comprehensive clinical imaging guidelines. This is a collaborative project between KSR and NECA.
Major efforts for quality and safety made by the Korean Society of Radiology (KSR).

CDC, Centre for Disease Control and Prevention. CT, computed tomography. MRI, magnetic resonance imaging.
2.2. Efforts for optimisation
The as low as reasonably achievable (ALARA) principle is a basic philosophy for the optimisation of radiological protection and safety. First, appropriate equipment, software, technique, and parameters must be used for radiological protection. Dosimetry must be performed and documented to determine typical doses to patients for common procedures for diagnostic radiological procedures. Diagnostic reference levels (DRLs) are used to review whether the optimisation is adequate, or if corrective action is required if the typical doses/activities exceed or fall below the DRL. Quality assurance programmes, including standard operating procedures, equipment quality control and clinical audits, must be established (IAEA, 2014).
After the separation of pharmacy distribution from medical practice in 2000 in order to reduce medical fees, the rate of self-referral increased. Self-referral in diagnostic imaging inevitably leads to overuse. If untrained physicians operate radiological equipment or try to interpret images, the number of poor-quality examinations will increase. On 19 January 2001, the National Assembly of Korea approved various acts, including quality management for specific medical equipment. By this law, quality management of specific medical equipment such as CT, MRI and mammography was launched by MOHW. In collaboration with MOHW, KSR developed guidelines and standards of quality management for mammography, CT and MRI equipment. KSR established the Korean Institute for Accreditation of Medical Imaging (KIAMI) as a relevant professional foundation, and MOHW authorised KIAMI to serve as an organisation for quality inspection. All CT, MRI and mammography equipment must have quality control tests mandated by the Medical Service Act Article 38 [installation and operation of special medical equipment (Do et al., 2007)].
In 2005, 3773 units of medical equipment underwent quality inspection, and 7% failed to meet the required standard. Given that the failure rate was much higher in the pilot study performed before inauguration of quality management, this result shows that the quality of medical imaging performed at certified units is improving, and public health in Korea has benefited from these improvements (Fig. 2).
Failure rates for equipment on quality control tests. CT, computed tomography. Mammo, mammography. MRI, magnetic resonance imaging.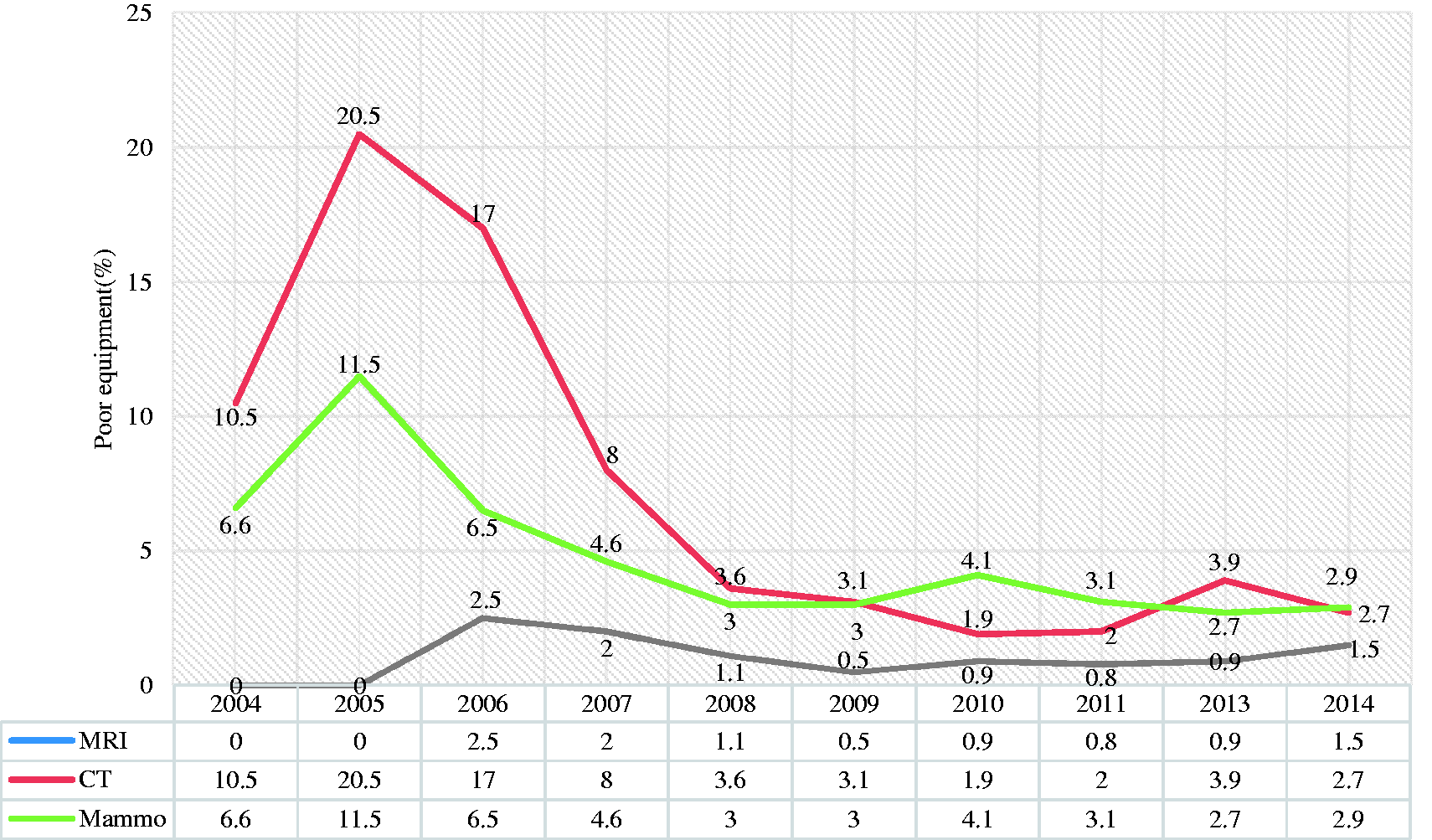
Diagnostic reference levels in Korea.
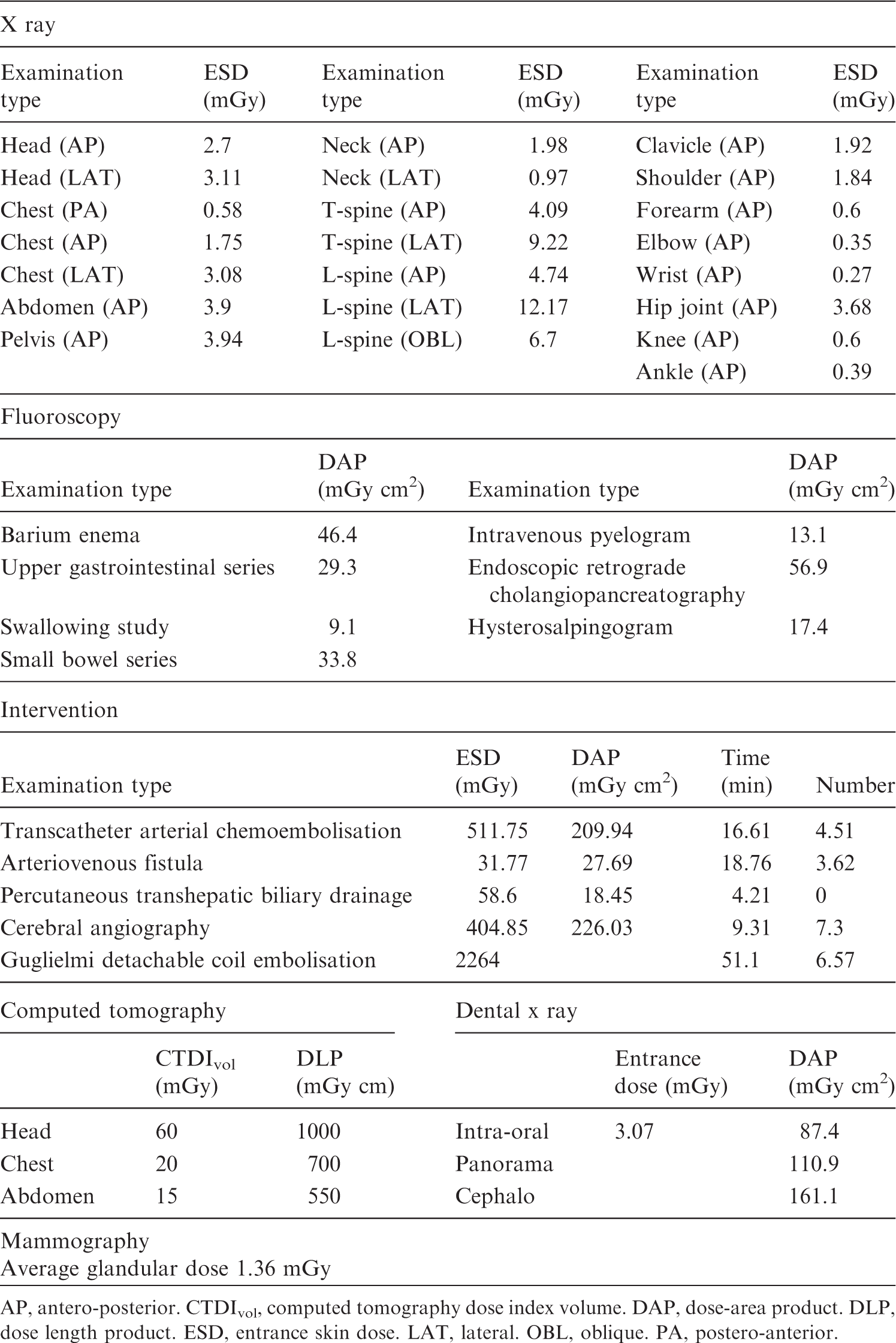
AP, antero-posterior. CTDIvol, computed tomography dose index volume. DAP, dose-area product. DLP, dose length product. ESD, entrance skin dose. LAT, lateral. OBL, oblique. PA, postero-anterior.
3. ISSUES AND CHALLENGES
The first issue and challenge concern diagnostic radiation dose measurement. Dosimetry will have to contribute fundamentally to patient protection, and a clear purpose is required. CT dose index volume and dose length product would be required in the case of CT performance monitoring, and effective dose would be needed for risk assessment purposes. Secondly, there is a need to determine whether it is necessary to measure the radiation dose for every diagnostic radiological examination, or only for relatively high-dose examinations. In some state governments in the USA, dose reporting is required for CT and fluoroscopy. Finally, there is a need to determine the level of enforcement.
Regarding the direction for diagnostic radiation control, there is an issue regarding personal dose tracking in Korea. Some groups believe that it must be necessary for reducing radiation dose. Other groups including radiology professionals do not agree with that idea because it is not applicable for justification and optimisation. Procedure tracking could be a substitute for personal dose tracking. However, there are several issues to consider, such as legal problems with the protection of personal information. HIRA had tried to establish a sharing system for imaging information. There is a need for practical implementation of justification for each diagnostic procedure. The first step affecting patient dose is the clinical decision to expose a patient to radiation, and this is the scope of justification. Education of referring physicians and radiologists is also important for justification.
Although medical radiation is clearly beneficial, there are potential risks, and it is necessary to distinguish between real risks and calculated/theoretical risks. Physicians should work with patient advocacy organisations to communicate the potential radiation risks and health benefits of imaging procedures more effectively (Amis et al., 2007). The dose equivalent should be expressed as the equivalent number of chest radiographs, and the risk of cancers should be expressed as the number of additional cases in the exposed population (BEIR VII, 2006). Patients must be protected from unnecessary and unintended radiation exposure. Understanding, communication, and cooperation of relevant stakeholders will be needed for patient protection. Medical physicists and radiographers play an important role in quality management and optimisation.
4. CONCLUSIONS
Radiation exposure from diagnostic medical imaging has increased in Korea. Each stakeholder plays a unique and complementary role for each patient-centred care system regarding radiological safety. Radiological societies play a key role in radiation safety issues in Korea, including guidelines, accreditation, advocacy, scientific activity, and education. Regulations are essential to control medical radiation exposure. Therefore, national organisations have made a significant effort to regulate and monitor medical radiation exposure using guidelines, accreditation, and even the law.
