Abstract
Based upon recent epidemiological studies of ocular exposure, the Main Commission of the International Commission on Radiological Protection (ICRP) in ICRP Publication 118 states that the threshold dose for radiation-induced cataracts is now considered to be approximately 0.5 Gy for both acute and fractionated exposures. Consequently, a reduction was also recommended for the occupational annual equivalent dose to the lens of the eye from 150 mSv to 20 mSv, averaged over defined periods of 5 years. To support ocular dose assessment and optimisation, Committee 2 included Annex F within ICRP Publication 116. Annex F provides dose coefficients – absorbed dose per particle fluence – for photon, electron, and neutron irradiation of the eye and lens of the eye using two dosimetric models. The first approach uses the reference adult male and female voxel phantoms of ICRP Publication 110. The second approach uses the stylised eye model of Behrens et al., which itself is based on ocular dimensional data given in Charles and Brown. This article will review the data and models of Annex F with particular emphasis on how these models treat tissue regions thought to be associated with stem cells at risk.
1. Introduction
Radiation exposure to the eye and lens of the eye may occur within many occupational and medical settings (Ainsbury et al., 2009). Ocular exposures can be a routine facet of radiological protection within any facility where spatially broad fields of gamma rays, neutrons, or charged particles may arise. Occupational exposures to the lens of the eye may occur in nuclear power plants, in research accelerator laboratories, or in industrial irradiation facilities. Exposures of the eye of medical patients may result during computed tomography (CT) examinations of the head or during neurovascular interventional procedures (Stein et al., 2010). These same medical radiation sources may also result in ocular exposures to physicians, nurses, and radiological technologists. Low-energy photon fields are produced through machine scatter and leakage to varying degrees across all forms of diagnostic imaging – radiography, diagnostic fluoroscopy, CT, and nuclear medicine. For the latter, the patients themselves serve as an additional radiation source. Scatter fields of higher energy photons may be experienced within areas of external beam radiation therapy. Some of the highest occupational ocular exposures in medicine involve physicians, radiologists, and cardiologists performing fluoroscopically guided interventions and, more recently, CT angiography (Vañó et al., 2010). Unique exposures to the lens of the eye from high linear energy transfer (LET) radiations occur during space exploration activities. Aircraft crew are also subject to neutron and charged-particle radiation fields following cosmic ray interactions with the aircraft hull. In these scenarios, the lens of the eye, owing to its superficial location within the body, may receive absorbed doses that can potentially exceed the threshold for tissue reactions (Rafnsson et al., 2005).
The primary radiobiological concern during ocular irradiation is the induction of one or more radiogenic cataracts. This paper will cover the International Commission on Radiological Protection (ICRP) recommendations for dose thresholds and dose limits for ocular exposure, review dosimetric models for assessment of absorbed dose to the eye and lens of the eye using Monte Carlo radiation transport methods, and conclude with a review of the most recent reference dose conversion coefficients for photon, electron, and neutron ocular exposures as given in Publication 116 (ICRP, 2010).
2. Icrp Recommendations for Dose Thresholds and Dose Limits for Ocular Exposures
The most recent recommendations by the Main Commission of ICRP were published in 2007 in Publication 103 (ICRP, 2007). In this document, the Commission recommended an annual dose limit for planned exposure situations set at 20 mSv effective dose, with an additional annual limit on the equivalent dose to the lens of the eye of 150 mSv. It is noted that the lens of the eye is thought to be subject to tissue reactions alone (formerly deterministic effects), and thus the lens of the eye is not a constituent tissue of the effective dose, even in its remainder term. For members of the general public, the annual limits on effective dose and equivalent dose to the lens of the eye are 1 mSv and 15 mSv year−1, respectively. The Commission, however, explicitly noted that: ‘this limit [to the lens of the eye] is currently being reviewed by an ICRP Task Group’, inferring that the dose limit could be subject to change given current reviews of the radiobiology and radiation epidemiology literature.
While the parameter values used in the calculation of effective dose – specifically wR and wT – were updated in Publication 103 (ICRP, 2007) over those given previously in Publication 60 (ICRP, 1991), the numeral limits for annual effective dose and annual equivalent dose to the lens of the eye remained unchanged from the 1990 Recommendations. In both cases, the annual limit for equivalent dose to the lens of the eye is based, in part, on assumed values of the threshold estimate of acute absorbed dose for 1% incidence of morbidity after whole-body gamma ray exposure. For cataract formation (visual impairment), the assumed dose threshold was thought to be approximately 1.5 Gy with a time-to-effect of several years (see Table A.3.4 of Publication 103). The primary references for this dose threshold were Publication 41 (ICRP, 1984) and Edwards and Lloyd (1996).
In 2011, the Main Commission of ICRP issued its Statement on Tissue Reactions as Part 1 of Publication 118 (ICRP, 2012). Part 2 of that report included a comprehensive review of early and late effects of radiation in normal tissues and organs including the lens of the eye. A wide variety of human epidemiological studies were reviewed that supported or questioned a lower (or even zero) threshold model for radiation induction of cataracts. Studies that supported a lower threshold covered ocular exposures to patients undergoing diagnostic procedures or radiotherapy, astronauts, atomic bomb survivors, residents in highly contaminated buildings, nuclear power plant workers, first responders to the Chernobyl accident, and workers in various medical exposure fields. Studies that questioned a lowering of the dose threshold for radiogenic cataracts included additional reviews of ocular exposures in diagnostic medicine, radiotherapy, and nuclear power plants.
Following this review, the Commission concluded that recent epidemiological evidence suggests that for some tissue reactions, particularly those with very late manifestation, threshold doses are lower than previously assumed. For the lens of the eye, the Commission, in their 2011 Statement (ICRP, 2012), indicates that the threshold in absorbed dose for cataract induction is now considered to be not higher than 0.5 Gy, which is lower by more than a factor of 3 from that stated in Publication 60 and Publication 103 (ICRP, 1991, 2007). Resultantly, for occupational exposures in planned exposure situations, the Commission now recommends an equivalent dose limit to the lens of the eye of 20 mSv year−1, averaged over defined periods of 5 years, with no single year exceeding 50 mSv. This nominal dose limit is thus a factor of 7.5 lower than that given previously in its 1990 and 2007 Recommendations (ICRP, 1991, 2007). The 2011 Statement (ICRP, 2012) did not specifically address changes in the annual dose limits for ocular exposures to members of the general public.
3. Use of Operational Quantities for Assessing Dose TO THE LENS OF THE EYE
The revised ICRP annual limit of 20 mSv on the radiation exposure to the lens of the eye is based on the radiation protection quantity, equivalent dose. This quantity is not directly measurable within the exposed individual, and thus cannot be used directly for radiation monitoring. ICRP thus adopts various operational quantities as defined by the International Commission on Radiation Units and Measurements (ICRU) for demonstration of compliance with regulations on occupational exposures and for dose optimisation. Three operational quantities are formally defined: the ambient dose equivalent H*(d); the directional dose equivalent H'(d,Ω); and the personal dose equivalent Hp(d) (ICRU, 1993). The depth parameter d is given as 0.07 mm, 3 mm, or 10 mm (in tissue-equivalent material) for monitoring exposures of the skin, lens of the eye, or effective dose, respectively. Both the ambient and directional dose equivalent are applied to area monitoring, while the personal dose equivalent is reserved for individual monitoring of radiation workers (via personal dosimeters). While the quantity H*(10) is formally assigned for area monitoring in the control of effective dose, all three quantities – H*(3), H'(3,Ω), and Hp(3) – have been used or proposed for control and monitoring of dose to the lens of the eye in relation to the ICRP dose limits. Very few physical dosimeters, however, have been constructed and/or calibrated to assess these operational quantities properly for d = 3 mm. In both Publication 103 (ICRP, 2007) and Publication 116 (ICRP, 2010), the Commission notes that dosimeters designed to measure H'(0.07,Ω) and Hp(0.07) for skin dose monitoring may also be applied for the purpose of monitoring exposure of the lens of the eye. With increased interest in dose assessment for the lens of the eye, given the Commission's downward revision of its annual dose limit, many investigators are putting new emphasis on devices that measure dose equivalent at the nominal 3-mm depth in tissue.
4. Use of Dose Coefficients for Assessing Dose TO THE LENS OF THE EYE
An alternative approach to dose assessment for the lens of the eye is through the use of numerical computation of particle fluence coupled with reference external fluence-to-dose conversion coefficients. A dose conversion coefficient for external radiation fields is defined as the tissue-equivalent dose per particle fluence (for example, in units of pGy cm–2). Values of the dose conversion coefficient for the lens of the eye as a function of particle energy may then be compared to values of H*(3)/Φ, H'(3,Ω)/Φ, and Hp(3)/Φ in determining the appropriateness of the operational quantities in monitoring whether or not annual limits have been met or exceeded. Dose conversion coefficients for the lens of the eye have recently been compiled in Publication 116 using geometric models of the eye and lens of the eye, and are reviewed briefly below (ICRP, 2010).
5. Dosimetric Models for Dosimetry of the lens of the eye
Current reference values for dose conversion coefficients for the lens of the eye adopted by the Commission in Publication 116 (ICRP, 2010) were developed using two geometric models of the eye. The first is the voxelised representation of the head, eye, and lens of the eye within the Publication 110 reference voxel phantoms of the reference adult male and reference adult female (ICRP, 2009). The second is a geometric stylised eye model taken from the studies by Behrens et al. (2009). This stylised eye model was used to supplement dose conversion coefficients for the lens of the eye derived from the Publication 110 phantoms (ICRP, 2009), where the lower voxel resolution within the later did not permit adequate spatial resolution of energy deposition within the lens of the eye, particularly at low-incident particle energies. The Behrens et al. (2009) model was, in turn, based upon an early and seminal study by Charles and Brown (1975) of ocular dimensions and the location of cells at risk.
5.1. Geometric model of Charles and Brown (1975)
Charles and Brown (1975) sought to provide guidance on improved dosimeter design for monitoring dose to the lens of the eye. Their main thesis was that the radiosensitive cells responsible for radiogenic cataract induction were not distributed uniformly across the whole lens of the eye and, thus, their specific location should be quantified and used as the basis for improved dosimeter design. Figs 1 and 2 show the relevant structures of the eye and lens of the eye reviewed in this study.
The eye in its sagittal cross-section. AC, anterior chamber; CB, ciliary body; ON, optic nerve; Cj, conjunctiva; SL, suspensory ligament of lens; Ch, choroid; V, vitreous body; C, cornea; F, fovea; I, iris; L, lens; R, retina; S, sclera. Source, Fig. 1 of Charles and Brown (1975). Transverse section of the lens. Source: Fig. 2 of Charles and Brown (1975).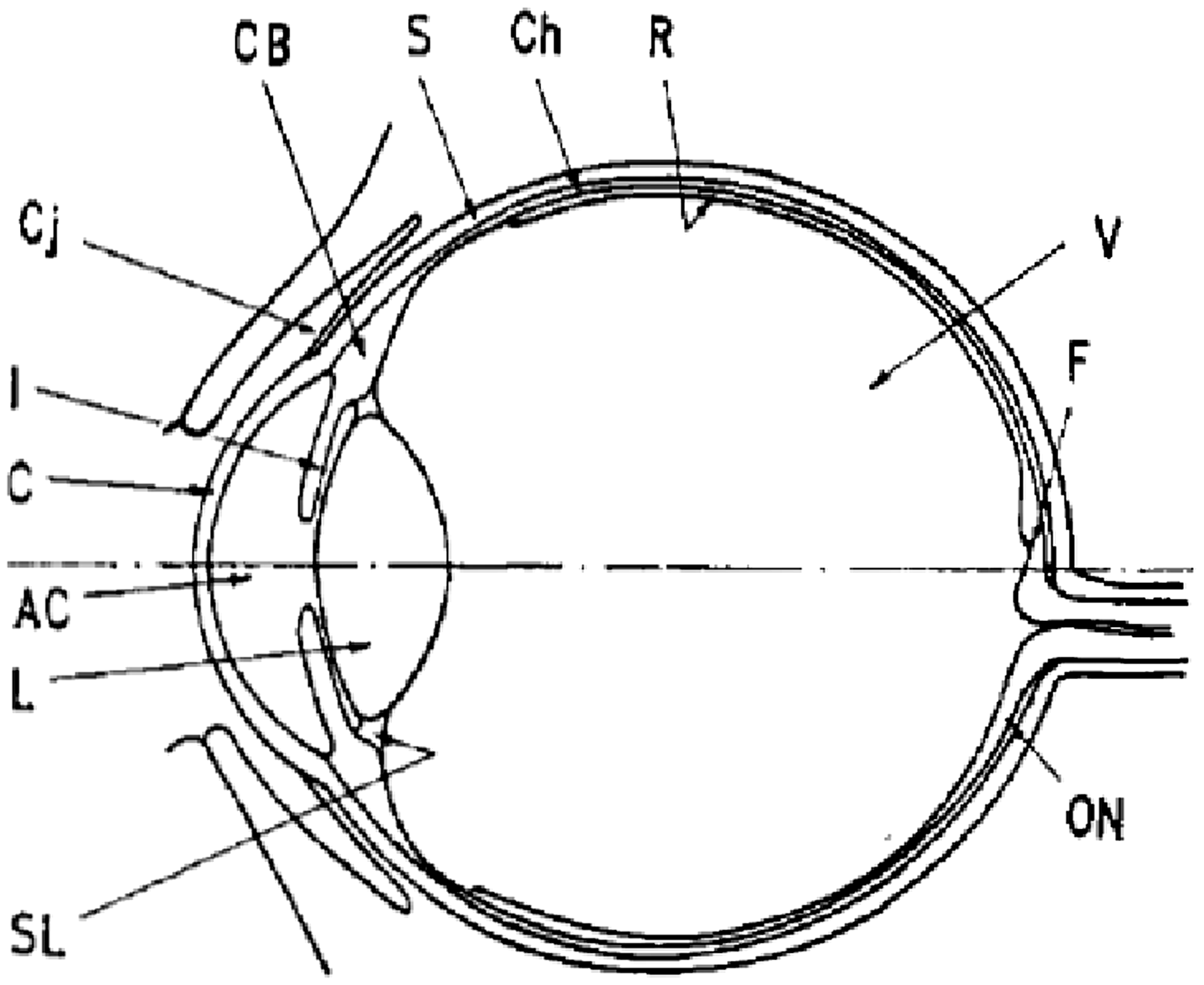
5.2. Location of cells at risk
The authors noted that Goldman and Liechti (1938) were perhaps the first to demonstrate that radiation damage to the equatorial region of the lens was a prerequisite to radiogenic cataract induction. The lens is a transparent elastic structure that is biconvex in shape, with the rounded junction of the two surfaces referred to as the ‘equator'. Goldman and Liechti used narrow x-ray beams to irradiate selective portions of rabbit lens, and found that cataract formation occurred only within the equatorial region of the irradiated lens. In a subsequent study by Alter and Leinfelder (1953), quadrantal exposure of the rabbit lens with x rays produced partial cataracts only in the exposed section, and no cataracts were produced when only the central region of the lens was irradiated. Von Sallman et al. (1955) further demonstrated that the central area of the lens was relatively radio-resistant to deuterons irradiation, whereas the equatorial region of the lens was highly radiosensitive with regard to lens opacities. These and other experiments reviewed by Charles and Brown provided evidence that the damaged cells of the germinative epithelium in the equatorial region of the lens were the precursors for radiation-induced cataract formation.
5.3. Data of cells at risk
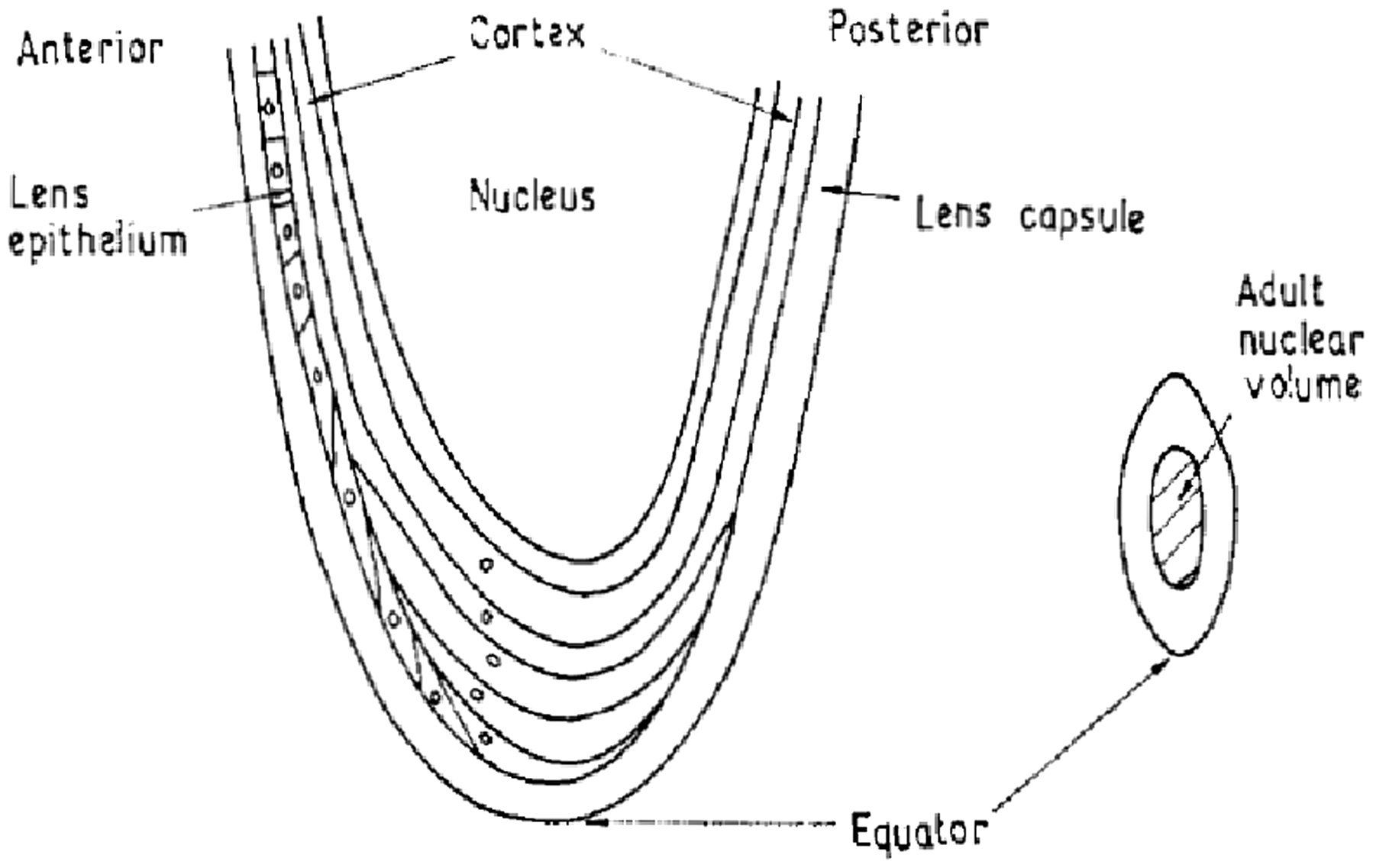
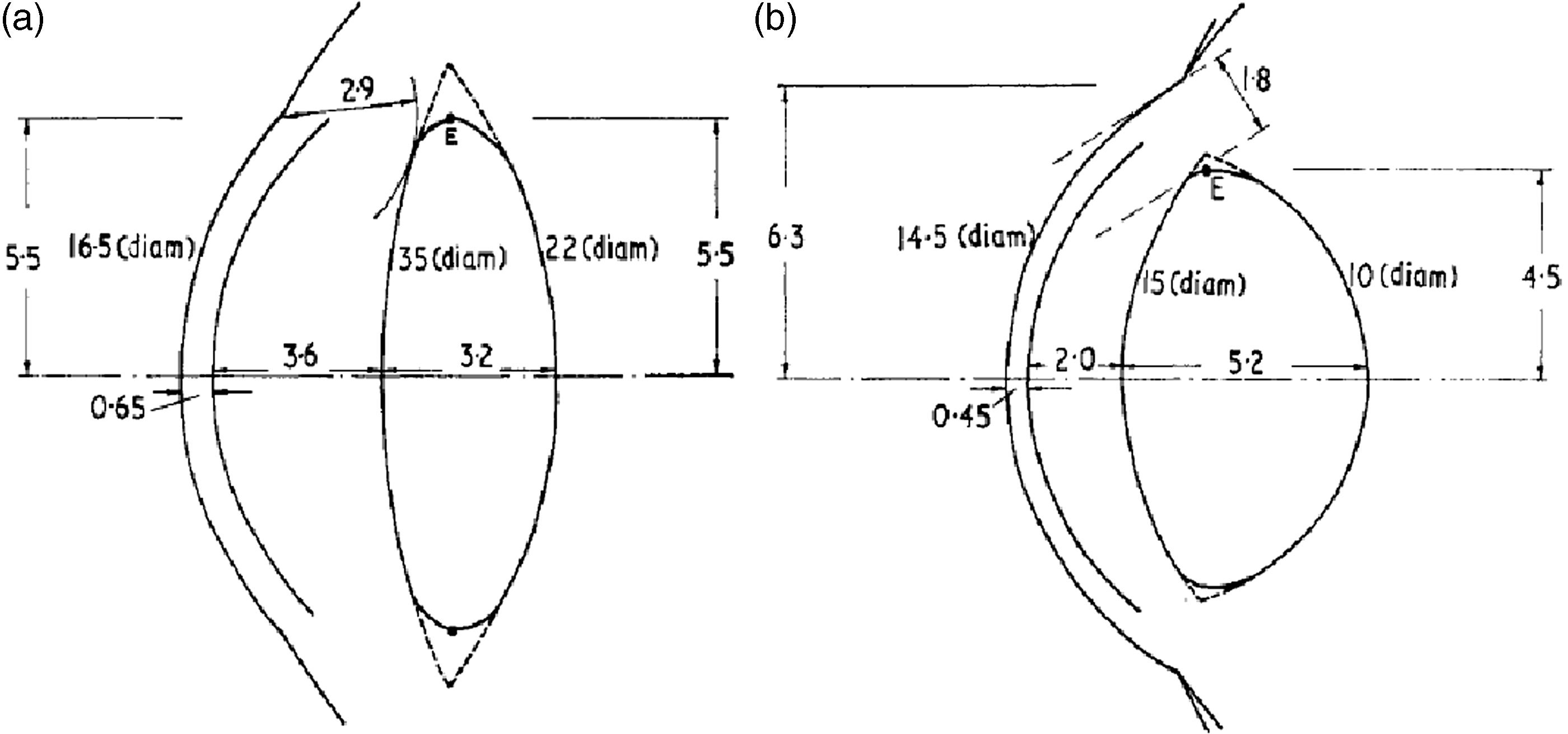
The question of the depth of the cells at risk was further addressed in Charles and Brown (1975) through the development of geometric models of the normal (emmetropic) eye and their extremes as given in the myopic (shorted-sighted) eye and the hypermetropic (long-sighted) eye. Dimensional data were gathered on nine parameters: (1) the radius of curvature of the anterior surface of the cornea; (2) the radius of curvature of the anterior surface of the lens; (3) the radius of curvature of the posterior surface of the lens; (4) the radius of curvature of the anterior surface of the sclera; (5) the depth of the anterior chamber along the optical axis; (6) the thickness of the cornea along the optical axis; (7) the thickness of the lens along the optical axis; (8) the diameter of the cornea; and (9) the equatorial diameter of the lens. Figs 3 and 4 show examples of the acquired data gathered from prior studies by Lowe and Clark (1973a,b) and Brown (1973, 1974a,b). Fig. 5 displays the final ‘reference’ eye model constructed by Charles and Brown. The extremes in ocular dimension – represented by the young myope and the older hypermetrope – are shown in Fig. 6. Data from Fig. 5 were used extensively in the chapter on eye anatomy given in Publication 89 (ICRP, 2002).
Radii of curvature of the lens as a function of subject age. Source: Fig. 4 from Charles and Brown (1975). Anterior chamber depth as a function of subject age. Source: Fig. 4 from Charles and Brown (1975). Mean ocular dimensions for the unaccommodated eye. The indicated tolerances represent the limits found in normal adults aged 20–65 years. Source: Fig. 8 from Charles and Brown (1975). Mean ocular dimensions for the deep lying lens of the young myope (a) and for the shallow lying lens of the older hypermetrope (b). Source: Figs 9 and 10 from Charles and Brown (1975).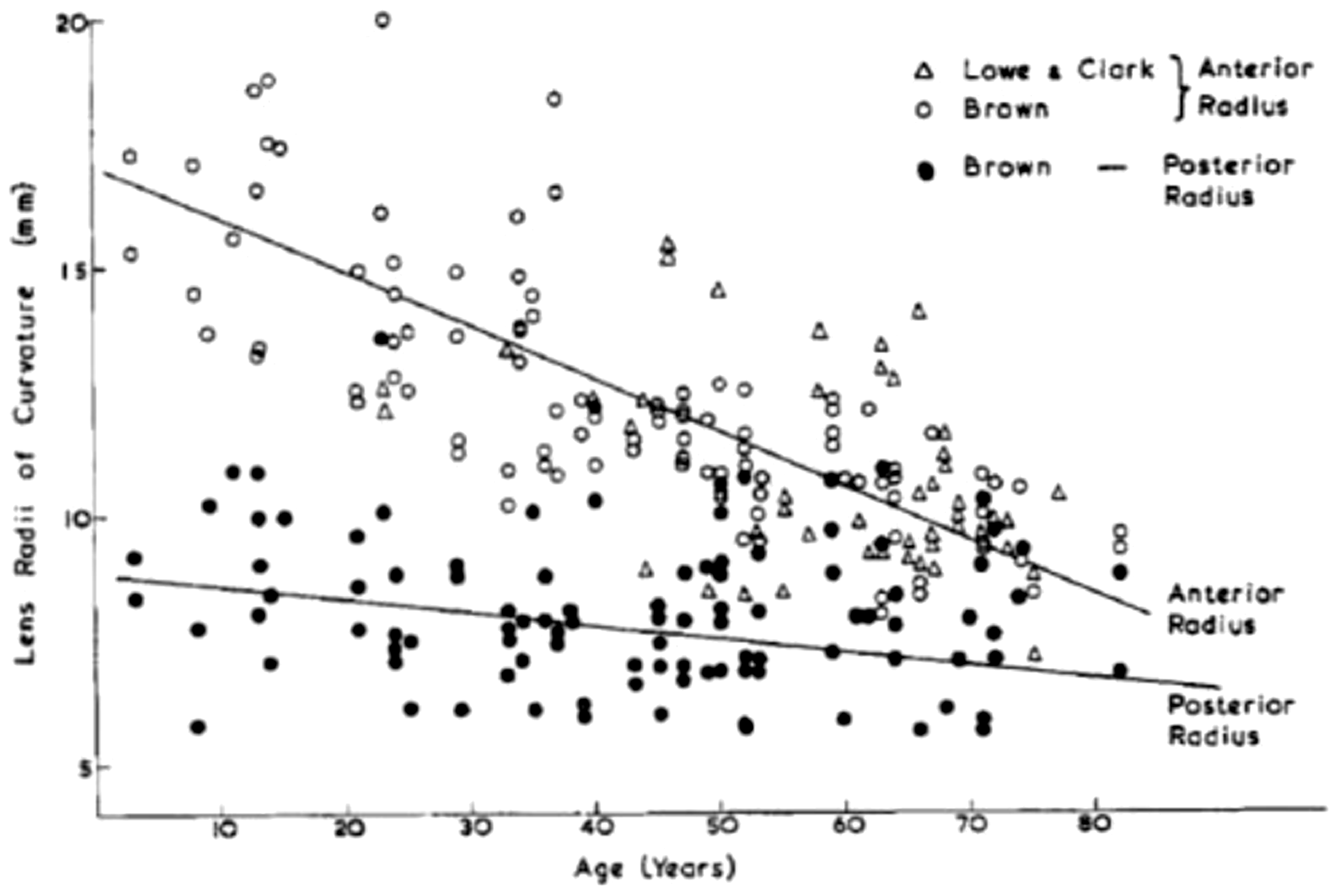
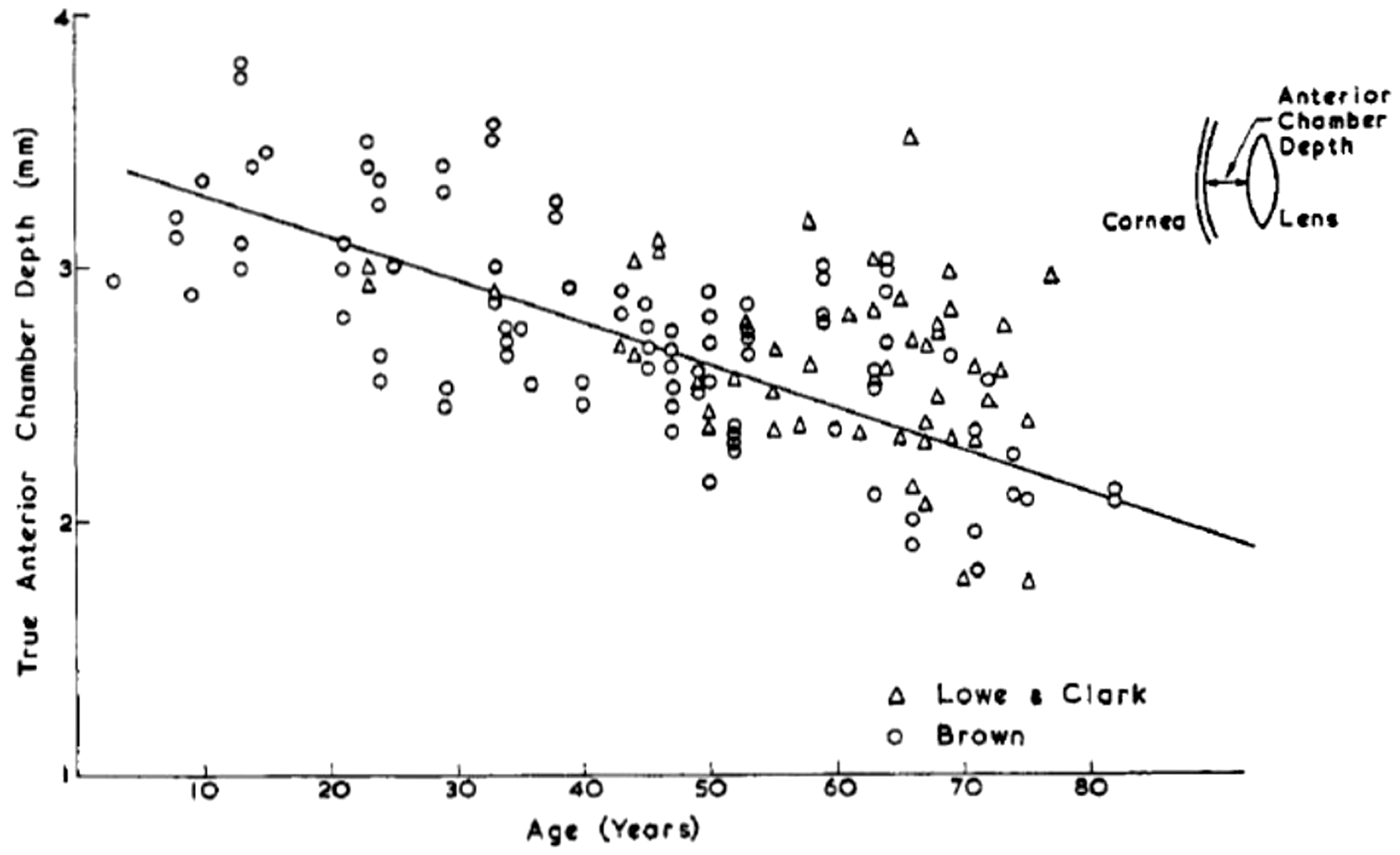
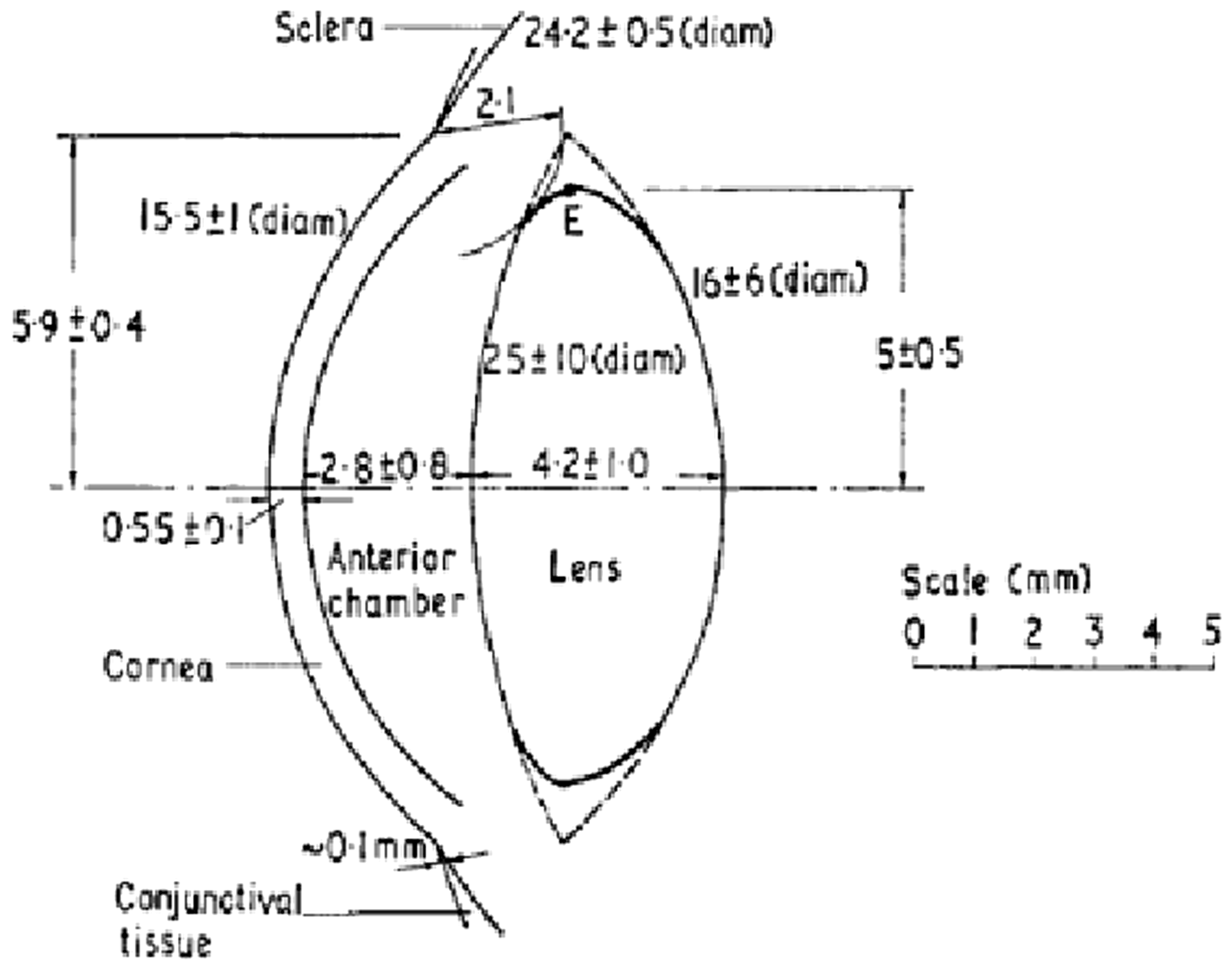
In their review, given the dimension models of the eye shown above, Charles and Brown (1975) came to the following conclusions as relevant to radiological protection of the eye. First, because the curvature of the anterior cornea is always greater than that of the anterior surface of the lens, the equatorial portion of the lens – wherein lie the cells at risk – is positioned at a depth shallower to the eye surface than at any other region of the lens. Second, the depth of the lens equator varies very little over the extremes of eye geometry in the normal adult population. The depth of the equator as measured to the sclera–cornea junction is 2.6 ± 0.6 mm, and the depth measured in the anterior–posterior (AP) direction is 3.1 ± 0.3 mm. The lower and upper limits correspond to older and younger age groups, respectively. Third, the minimum depth of any part of the lens is indicated by drawing an arc from the cornea–sclera junction, which just touches the lens. This corresponds to a value of 2.3 ± 0.4 mm and is associated with a lens region just anterior to the equator.
In their recommendations for dosimeter design, the authors note that a planar dosimeter that measures tissue dose at a depth of approximately 3 mm should give ‘a reasonable overall measure of the mean equatorial dose for a variety of eye and irradiation geometries likely to be met during the lifetime's exposure experience at radiation protection levels’. Furthermore, an ideal dosimeter would integrate the tissue dose between depths of 2.5 and 3.5 mm as this 1-mm thickness covers the approximate extent of the equatorial regions of the lens over which the cells at risk lie.
5.4. Geometric eye model of Behrens et al. (2009) for electron dosimetry
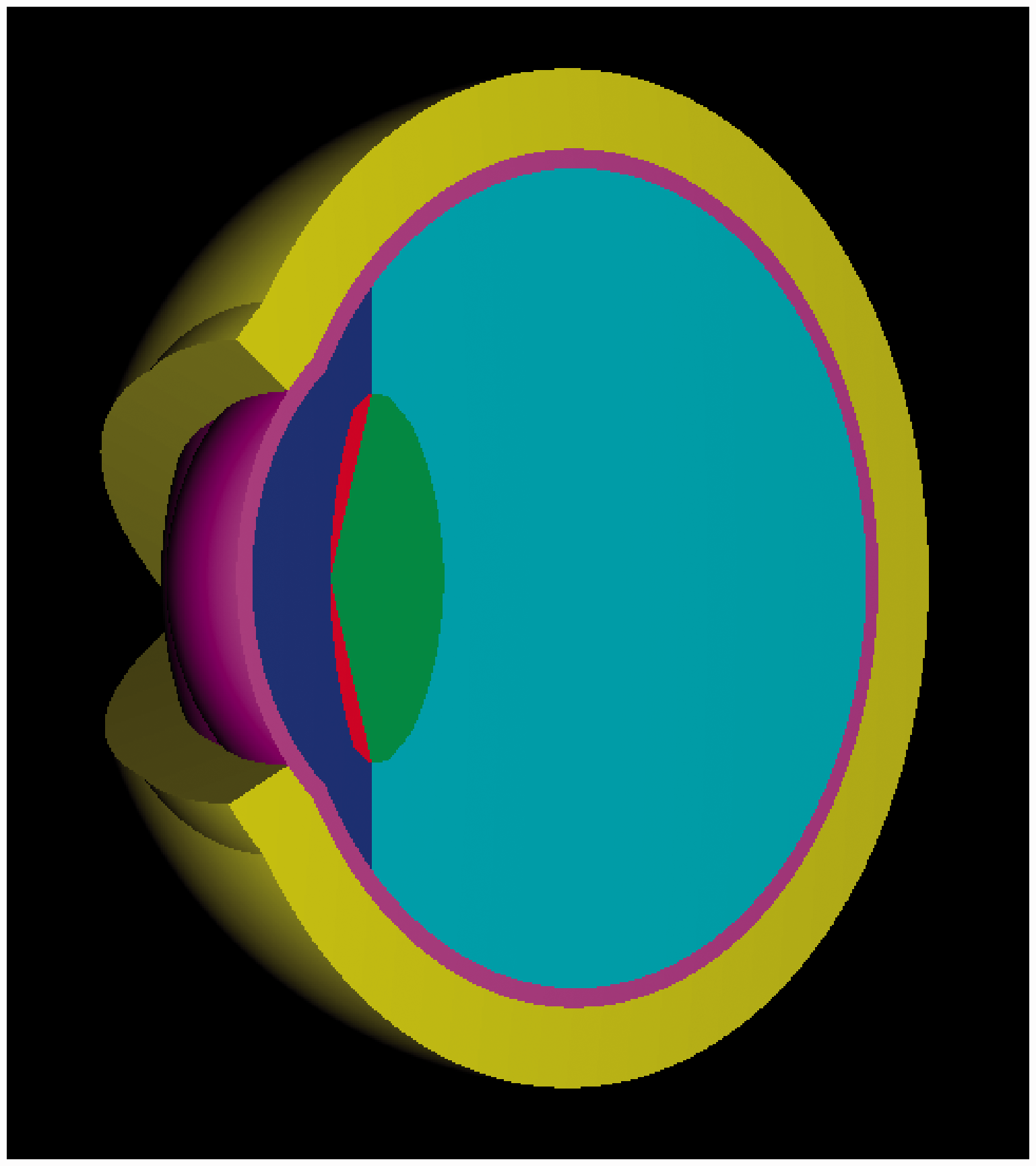
In a study by Behrens et al. (2009), a new three-dimensional (3D) geometric, stylised model of the eye was developed for the purpose of computing dose conversion coefficients for externally incident mono-energetic electrons using Monte Carlo radiation transport methods. The model was constructed, in large part, based upon dimensional data taken from Charles and Brown (1975). Figs. 7 and 8 show the Behrens et al. eye model in its 2D schematic and 3D pictorial views, respectively. The model includes an analysis of the elemental composition and mass density of each of the coloured regions of Fig. 8. Mass densities (units of g cm–3) were assigned as 1.09 for the eye lid, 1.06 for the lens (both sensitive and insensitive regions), 1.00 for the aqueous and vitreous humour, and 1.076 for the cornea. Data sources included Publication 23 (ICRP, 1975) and Report 56 (ICRU, 1997).
Eye geometry used in the study by Behrens et al. (2009). All dimensions are in millimetres and M denotes the x-position of the centre of the eye sphere. Source: Fig. 1 of Behrens et al. (2009). Three-dimensional view of the eye geometric model of Behrens et al. The following colour code is used: yellow: skin of eye lid; red: sensitive region of lens of the eye; green: insensitive region of lens of the eye; dark blue: aqueous humour; light blue: vitreous humour; magenta: cornea. Source: Fig. 2 of Behrens et al. (2009).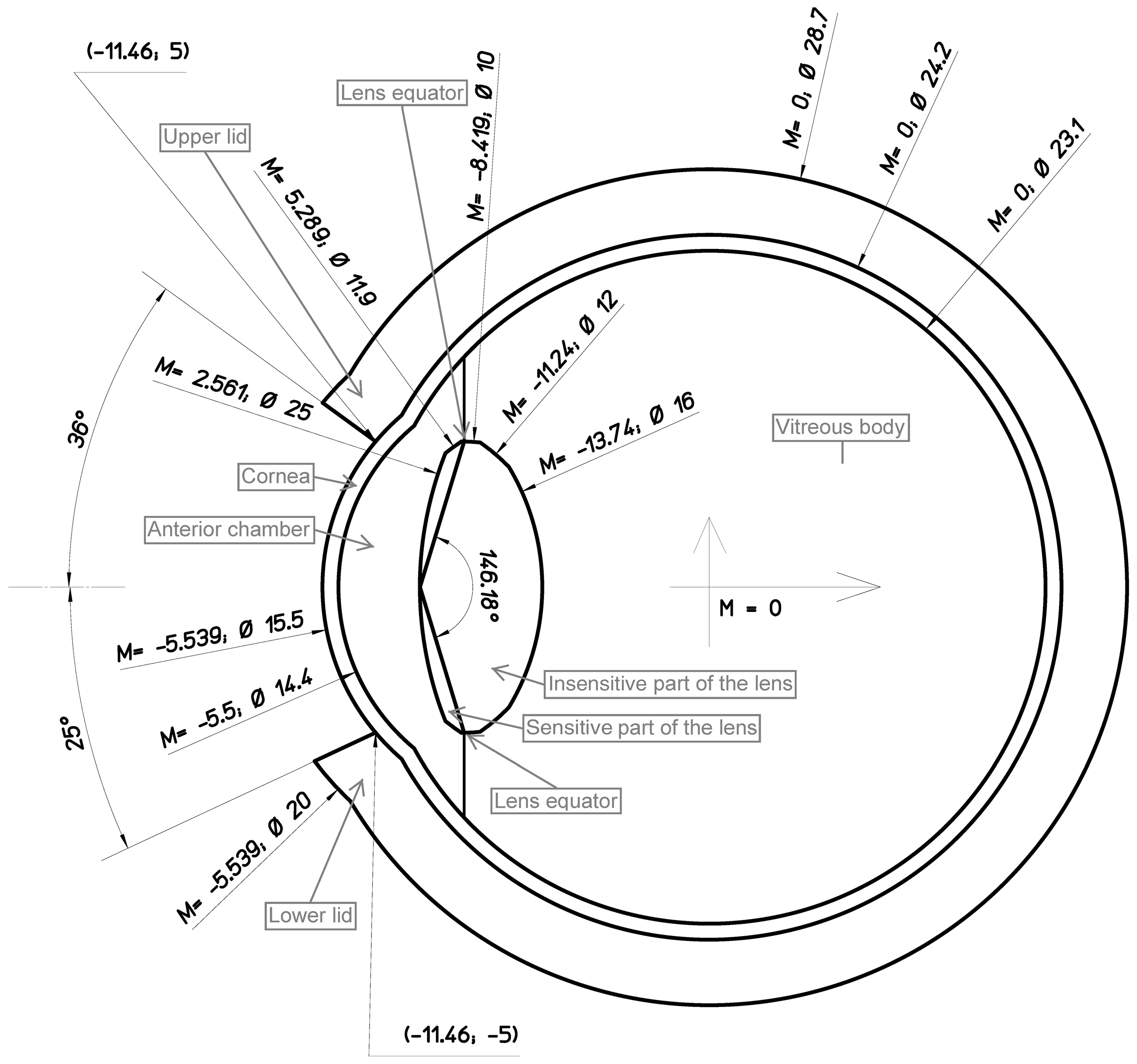
The model shown in Figs. 7 and 8 results in a tissue-equivalent depth of the sensitive volume of the lens between 2.80 mm and 3.82 mm (mean 3.36 mm) and a sensitive volume thickness of between 0 mm and 0.595 mm (mean 0.495 mm). The mass of the entire lens and its sensitive volume are given at 229.1 mg and 39.2 mg, respectively. The authors note that the mean depth of 3.36 mm is quite similar to the nominal value of 3 mm in the definition of the operational quantities, directional dose equivalent H'(3,Ω) and personal dose equivalent Hp(3) for ocular exposure monitoring and regulatory compliance.
In Behrens et al. (2009), the eye model was used to assess dose conversion coefficients for mono-energetic electrons normally incident upon the eye and at three off-axis polar angles – 15°, 30°, and 45° – and for three directions – side, bottom, and top of the eye. The range of electron energies considered were 100 keV to 12 MeV. All Monte Carlo simulations were performed using EGSnrc Version 4. In this study, the geometry of the surrounding head and tissue structures were not considered in that: (1) electrons at angles of incidence less than 45° were considered as most relevant to occupational and medical exposures; and (2) electrons of energies below several MeV have small tissue ranges and thus would not reach the lens of the eye at larger incident angles. Virtually all beta particles of relevance to radiological protection have endpoint energies below approximately 3.5 MeV.
5.5. Geometric eye model of Behrens and Dietze (2011) for photon dosimetry
In 2011, the original stylised eye model used for Monte Carlo radiation transport simulation of external electron ocular irradiation was extended to the study of photon exposures of the eye and lens of the eye. In this study, Behrens and Dietze (2011) used the geometric model of Behrens et al. (2009) described above, but considered the influence of scatter and attenuation of incident photons striking the exposed individual and their influence on ocular tissue dose. Accordingly, a left and right stylised eye model was inserted within a larger gender-averaged whole-body anatomic model as defined by Kramer et al. (1982) (described in detail within Appendix A of their paper). The resulting model is shown in Fig. 9. Mono-energetic photons of energies ranging from 5 keV to 10 MeV were considered under standard ICRP exposure geometries: AP; posterior–anterior (PA); left lateral; average of left and right lateral (LAT); and rotational (ROT) incidence. All Monte Carlo simulations were performed using EGSnrc Version 4 with full secondary charged particle transport. The dose conversion coefficients to the sensitive volume of the lens and the entire lens volume were valuated.
View of the geometry used in the Monte Carlo simulations studies of Behrens and Dietze for external photon irradiation of the eye and lens of the eye. Source: Fig. 1 of Behrens and Dietze (2011).
5.6. Geometric eye model of Manger et al. (2012) for neutron dosimetry
In 2012, the previous studies of electron and photon eye dosimetry were further extended to neutron ocular exposures. In a study by Manger et al. (2012), the stylised eye model of Behrens et al. (2009) was again adopted and inserted into a larger model of the head and neck. In this study, the latter model selected was that of the modified Oak Ridge National Laboratory stylised phantom as reported by Han et al. (2006). Dose conversion coefficients to the whole lens of the eye and to its sensitive layer were reported for discrete mono-energetic neutron energies between 10–9 and 20 MeV for AP, RLAT (right lateral), and ROT orientations. All simulations were performed using MCNPX Version 2.6.0.
5.7. Eye models in the ICRP reference phantoms
Publication 116 (ICRP, 2010) represents the very first application of the Publication 110 reference phantoms – both the adult male and the adult female – in the calculation of reference dose conversion coefficients within ICRP Committee 2. These ICRP reference phantoms are consistent with the information given in Publication 89 (ICRP, 2002) on the reference anatomical parameters for both male and female adults and were constructed through modification of the voxel models ‘Golem' and ‘Laura' (Zankl and Wittmann, 2001; Zankl et al., 2005) of two individuals whose body height and mass closely resembled the reference data. Each phantom is represented as a 3D array of cuboid voxels, arranged in columns, rows, and slices. Each entry in the array identifies the organ or tissue to which the corresponding voxel belongs. The male reference computational phantom consists of 1.95 million tissue voxels (excluding voxels representing the surrounding vacuum), each with a slice thickness (corresponding to the voxel height) of 8.0 mm and an in-plane resolution (i.e. voxel width and depth) of 2.137 mm, corresponding to a voxel volume of 36.54 mm3. The female reference computational phantom consists of 3.89 million tissue voxels, each with a slice thickness of 4.84 mm and an in-plane resolution of 1.775 mm, corresponding to a voxel volume of 15.25 mm3. The number of individually segmented structures is 136 in each phantom, and 53 different tissue compositions have been assigned to them.
In the Publication 110 reference male phantom, the right/left eye and lens of the eye are approximately 7.3 g and approximately 0.19 g, respectively (ICRP, 2009). At a mass density of 1.05 g cm–3, these tissue regions correspond to 190 voxels for each male eye, but only approximately 5 voxels for each male lens. Correspondingly, in the Publication 110 reference female phantom, the right/left eye and lens of the eye have masses of approximately 7.3 g and approximately 0.20 g, respectively, which correspond to voxel counts of approximately 456 voxels for each eye and 12 voxels for each lens. Transaxial views of both models are shown in Fig. 10. For moderately to highly penetrating external radiations, reasonable estimates of dose to the lens of the eye can be expected from Monte Carlo radiation transport within the Publication 110 reference phantoms. For low-energy penetrating radiations, the coarse nature of the voxel geometry in these phantoms will not fully capture rapidly changing dose gradients for external ocular irradiations. Furthermore, it is not possible to model the sensitive volume of the lens of the eye at these voxel dimensions. It was for these reasons that reference dose conversion coefficients in Publication 116 (ICRP, 2010) were supplemented with values determined using the various stylised eye models presented above.
Transaxial views through the Publication 110 reference computational phantoms [male (a) and female (b)] showing the voxelised representation of the eye and lens of the eye.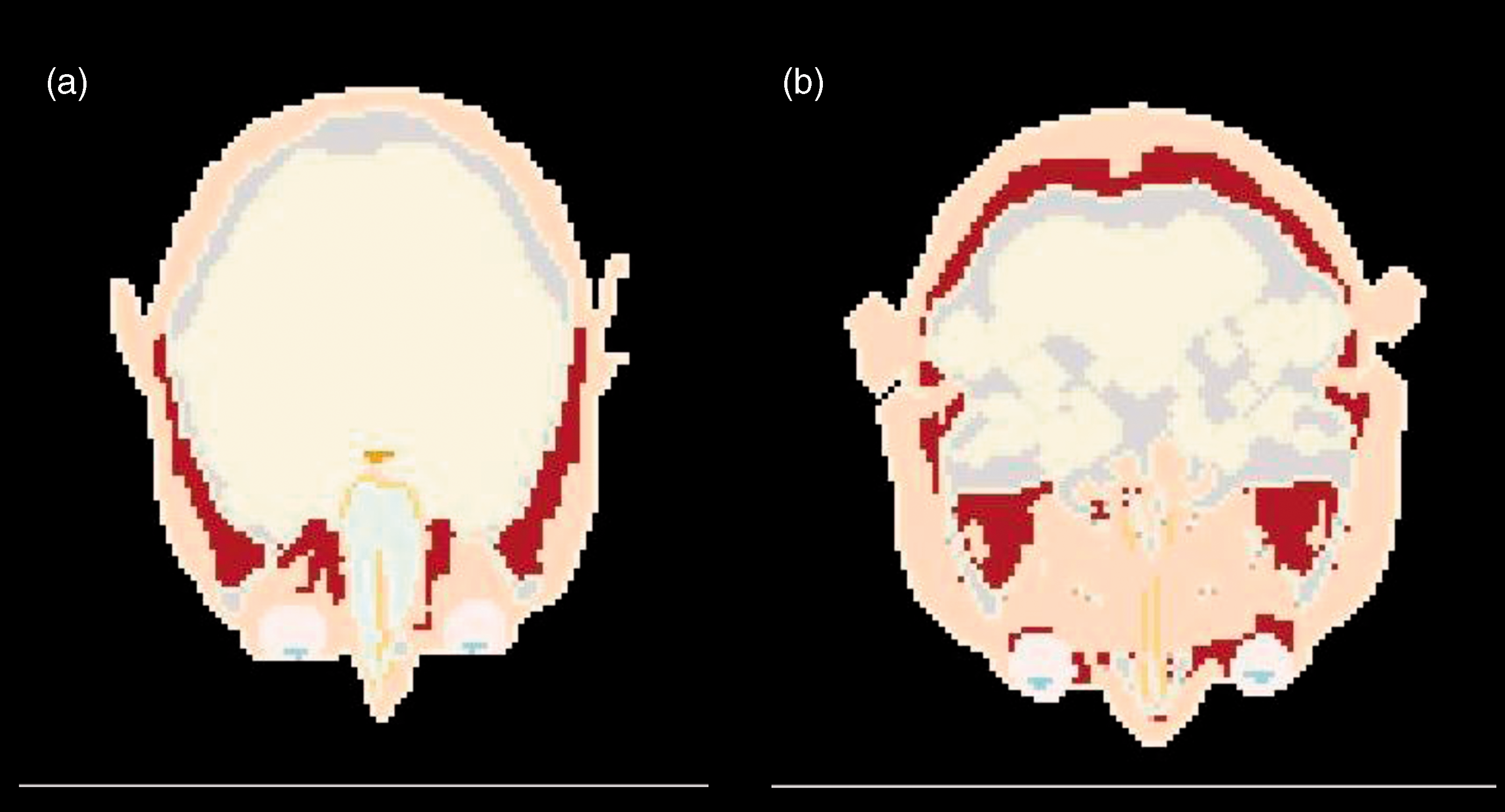
6. Dose Coefficients from Annex F of Publication 116
Annex F of Publication 116 (ICRP, 2010) presents the Commission's recommended reference dose conversion coefficients for ocular exposures given in terms of absorbed dose to the lens of the eye per particle fluence (pGy cm2). These reference conversion coefficients were defined either based upon values using the stylised eye model or from the voxelised model of the lens of the eye within the Publication 110 reference computational phantoms (ICRP, 2009). Reference dose conversion factors for the lens of the eye are given for external photons (10 keV to 10 GeV), electrons (10 keV to 10 GeV) and neutrons (1 meV to 10 GeV) in tabular form within Annex F of Publication 116. A more extensive listing of these conversion coefficients is given in the electronic annex of Publication 116 for photons, electrons, neutrons, positrons, protons, muons, pions, and He ions.
6.1. Photon irradiation of the eye
Data for external photon irradiation of the lens of the eye are shown in Fig. 11 for the AP, LAT, and ROT irradiation geometries. Data points show separately the dose conversion coefficients for the reference phantoms (gender averaged), the total lens within the stylised eye model and the sensitive volume of the lens of the eye within the stylised eye model. Solid curves give the final values of ICRP reference conversion coefficients for external photons as a function of photon energy. The graphs within Fig. 11 indicate that, with the exception of photon energies below approximately 20 keV for the AP geometry and 50 keV for other geometries, there is good agreement across the various eye models – voxel and stylised. Above 2 MeV, and in particular for AP irradiation, the data for the stylised model have a tendency to be less than those from the Publication 110 reference phantoms (ICRP, 2009). It can also be seen that absorbed doses in the sensitive region of the lens of the eye are reasonably represented by the reference values at photon energies up to 1 MeV.
Absorbed doses for the lens of the eye per photon fluence (pGy cm2) for anterior–posterior (AP), average of left and right lateral (LAT), and rotational (ROT) irradiation as calculated with the male and female reference computational phantoms and with the stylised eye model of Behrens and Dietze (2011). The solid curves show the Publication 116 reference values. Source: Fig. F.3 of ICRP (2010).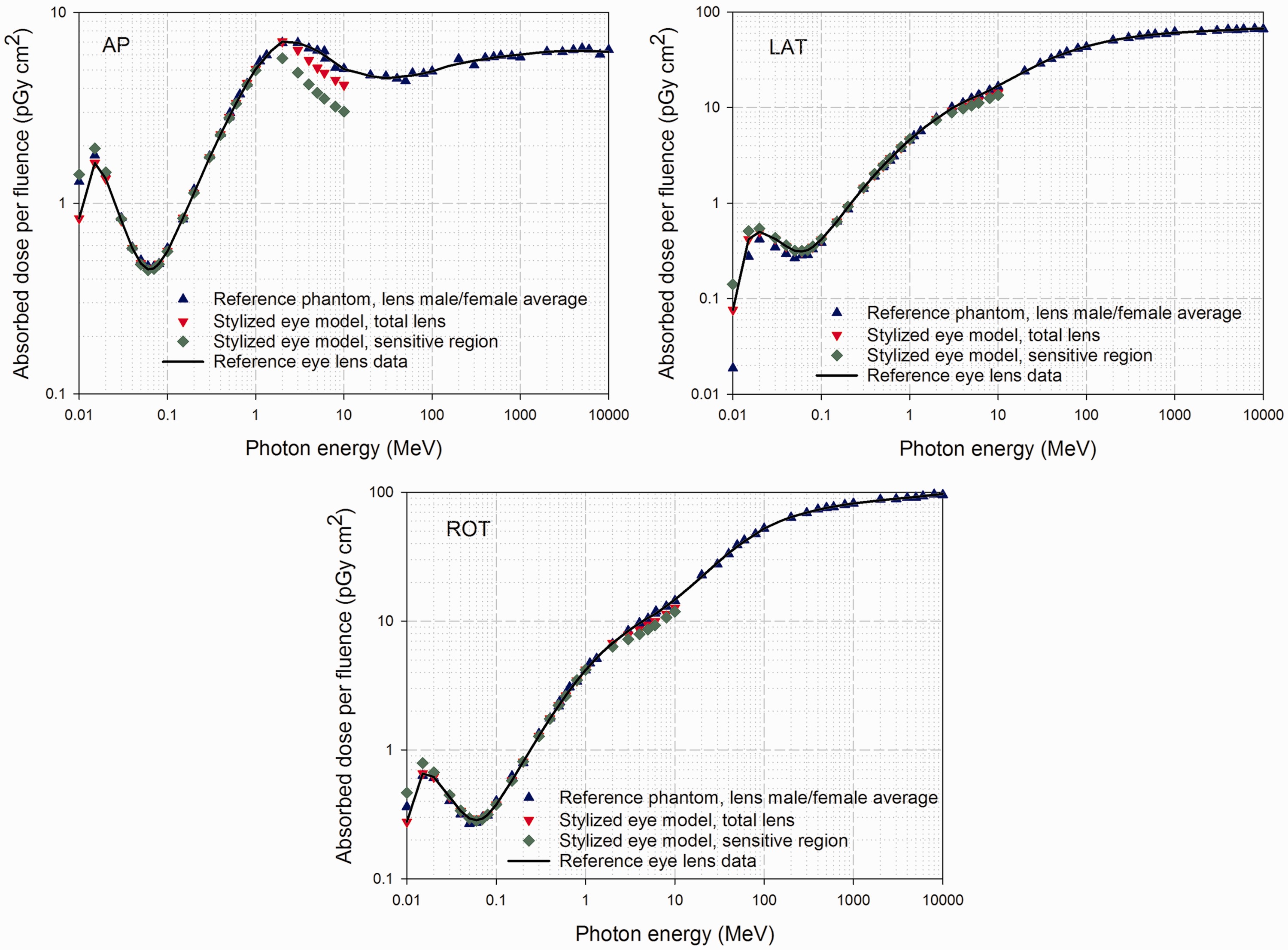
Following a conservative approach, the data from the stylised model was used in Publication 116 (ICRP, 2010) as reference dose conversion coefficients for AP, PA, LAT, and ROT geometries for energies up to 2 MeV. For all other situations, the reference conversion coefficients for the lens of the eye were evaluated as their gender-averaged value from the Publication 110 male and female computational phantoms (ICRP, 2009).
6.2. Electron irradiation of the eye
Fig. 12 shows a similar comparison of model results for external electron irradiation of the eye and lens of the eye under AP irradiation. Below 1 MeV, the lens dose for the stylised eye model is significantly lower than that given by the lower resolution eye model of the reference computational phantoms. This is due to the fact that at least one voxel in the reference phantom is in direct contact with the phantom's exterior (vacuum in the Monte Carlo simulations), which is not equivalent to that in the real ocular anatomy whereby the externally incident electrons would have to first traverse both the cornea and anterior chamber of the eye prior to reaching the lens. At energies between 1 and 10 MeV, however, there is good agreement between the model results.
Absorbed dose for the lens of the eye per electron fluence (pGy cm2) for anterior–posterior (AP), as calculated with the male and female reference computational phantoms and the stylised eye model of Behrens et al. (2009). The solid curves show the Publication 116 reference values. Source: Fig. F.4 of ICRP (2010).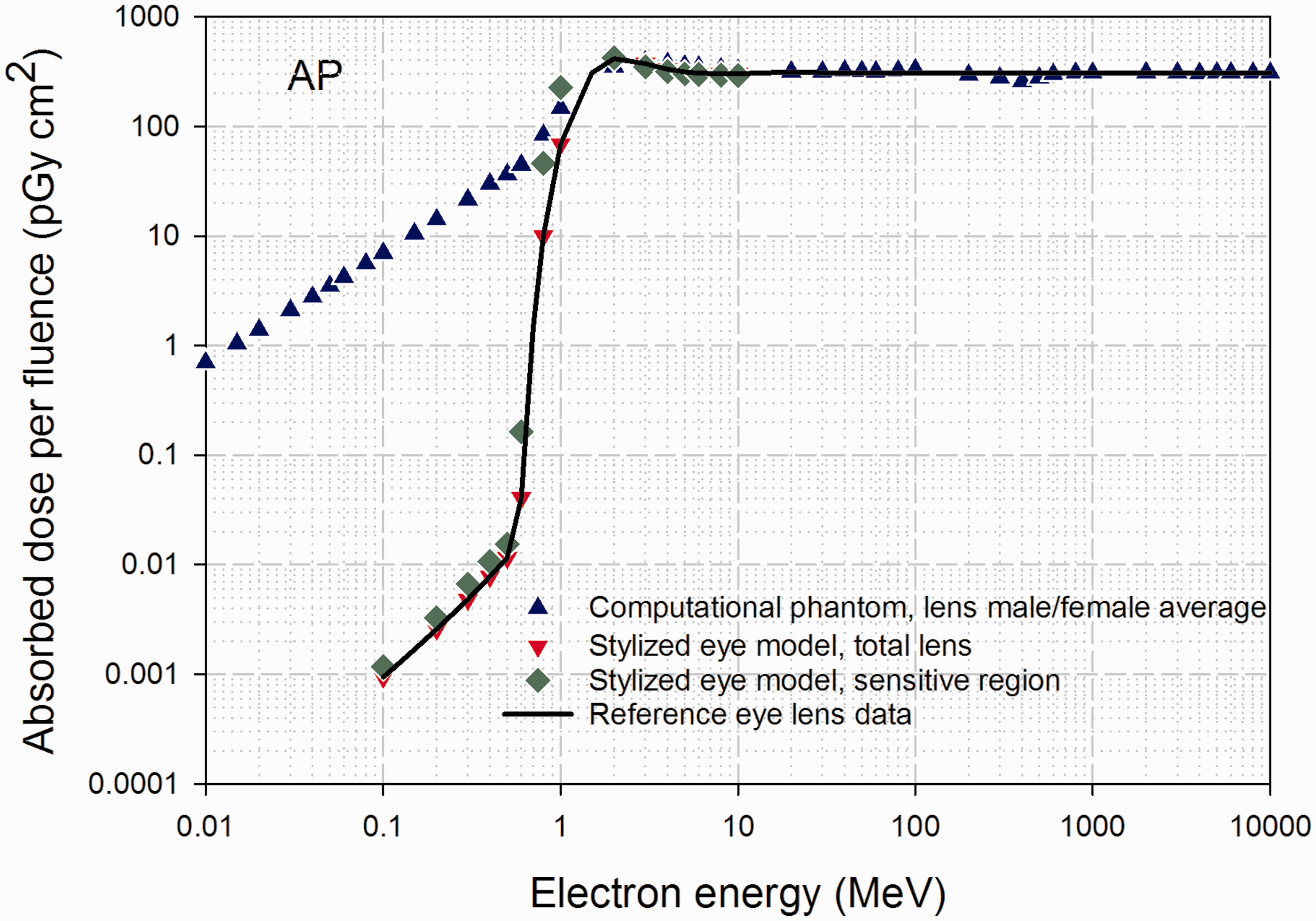
Among the irradiation geometries considered in Publication 116 (ICRP, 2010), the only significant dose to the lens of the eye for electrons of energies below 1 MeV are those normally incident under AP irradiation. Data for AP irradiation can be considered as conservative upper estimates for the other geometries below 1 MeV electron energy. For isotropic (ISO) irradiation and energies above 1 MeV, and for PA irradiations at all energies, no significant differences between the dose conversion coefficients from voxel and stylised models were evident. For ISO geometry and electrons with energies lower than 1 MeV, the same dose conversion coefficients as for AP geometry were adopted. For all other situations, reference data were evaluated as their gender-averaged values from the reference computational phantoms.
6.3. Neutron irradiation of the eye
Fig. 13 shows a further comparison of model results for external neutron irradiation of the eye and lens of the eye under the AP, LAT, and ROT irradiation geometries. Data points show separately the dose conversion coefficients for the reference phantoms (gender averaged), the total lens within the stylised eye model, and the sensitive volume of the lens of the eye within the stylised eye model. Solid curves give the final values of ICRP reference conversion coefficients for external neutrons as a function of neutron energy. Manger et al. (2012) concluded that the dose to the sensitive region of the lens of the eye was similar to that of the total lens over all neutron energies and exposure geometries, with the greatest differences being 13% at the lowest incident neutron energy. Regardless of the orientation of the exposure, no significant difference was found in the absorbed dose in the sensitive volume and total volume of the lens of the eye.
Absorbed doses for the lens of the eye per neutron fluence (pGy cm2) for anterior–posterior (AP), average of left and right lateral (LAT), and rotational (ROT) irradiation as calculated with the male and female reference computational phantoms and with the stylised eye model of Manger et al. (2012). The solid curves show the Publication 116 reference values. Source: Fig. F.5 of ICRP (2010).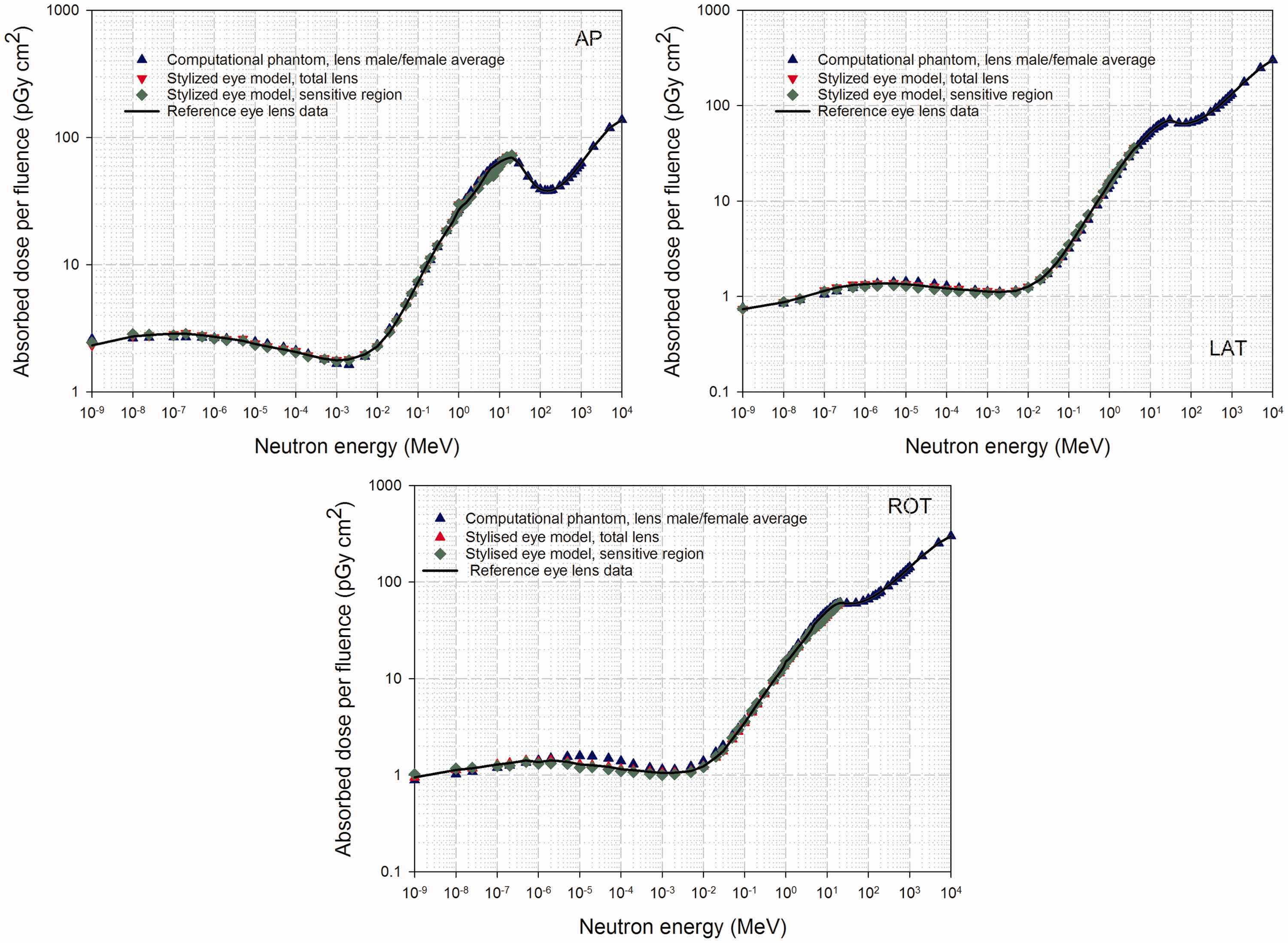
Reference values for dose conversion coefficients for externally incident neutrons are given in Publication 116 (ICRP, 2010) as taken from the stylised model of Manger et al. (2012) for AP, LAT and ROT geometries and energies up to 4 MeV. All other reference conversion coefficients were assigned as their gender-averaged values seen in the male and female reference computational phantoms.
6.4. Relationship to the operational quantities for the lens of the eye
An important analysis presented in Chapter 5 of Publication 116 (ICRP, 2010) is the comparison of the new reference dose conversion coefficients for external radiation exposure with the various operational quantities used for either area or individual monitoring of workers exposed to these fields. Such comparisons are shown in Fig. 14 for photon irradiation of the lens of the eye under various exposure situations (AP, PA, LAT, ROT, and ISO). In Fig. 14, it is clear that the ambient dose equivalent evaluated at a tissue-equivalent depth of 3 mm, H*(3), is a reasonably good estimator of photon dose to the lens of the eye under AP irradiation at energies between 100 keV to 1 MeV and is a very conservative estimator of that same dose at energies below 100 keV, and above 1 MeV. H*(3) is an even more conservative estimator of dose to the lens of the eye for other irradiation geometries across the energy range considered.
Ratios of equivalent dose to the lens of the eye (Publication 116) for various exposure situations (AP, anterior–posterior; PA, posterior–anterior (PA); LAT, average of left and right lateral; ROT, rotational; ISO, isotropic) to the ambient dose equivalent at 3-mm depth H*(3) from Publication 74 for externally incident photons. Source: Fig. 5.8 of ICRP (2010).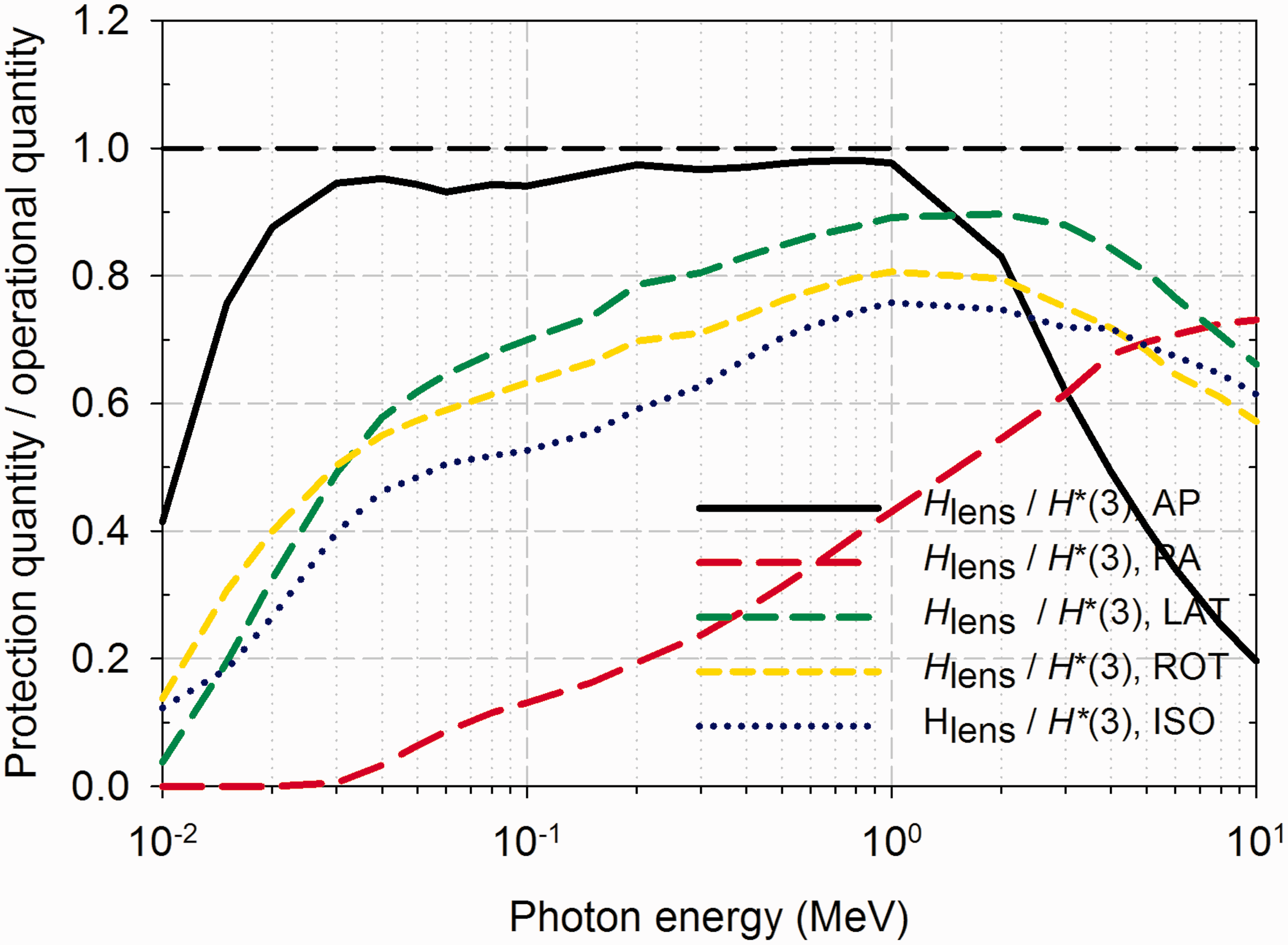
A comparison of dose to the lens of the eye for externally incident mono-energetic electrons and the operational quantities H*(3) and H'(3) are shown in Fig. 15. At electron energies of 3 MeV and above under AP irradiation, the ambient dose equivalent evaluated at 3-mm tissue-equivalent depth provides a very good estimate of the dose to the lens of the eye and is a conservative estimate at lower energies. For both PA and ISO irradiation geometries, H*(3) is shown to be a conservative estimator of electron dose to the lens of the eye at energies up to 100 MeV, above which the ambient dose equivalent underestimates the value of dose to the lens of the eye. The authors of Publication 116 (ICRP, 2010) note that precise data for the personal dose equivalent Hp(3) on a head phantom are not available at present. Furthermore, ICRU is currently evaluating the suitability of all existing operational quantities, their calibration procedures, and appropriate calibration phantoms.
Ratios of equivalent dose to the lens of the eye [Publication 116 (ICRP, 2010)] for various exposure situations (AP, anterior–posterior; PA, posterior–anterior (PA); LAT, average of left and right lateral; ROT, rotational; ISO, isotropic) to the directional dose equivalent H'(3) for electrons from Publication 74 and to the ambient dose equivalent H*(3) based on data from Ferrari and Pelliccioni (1994). Source: Fig. 5.9 of ICRP (2010).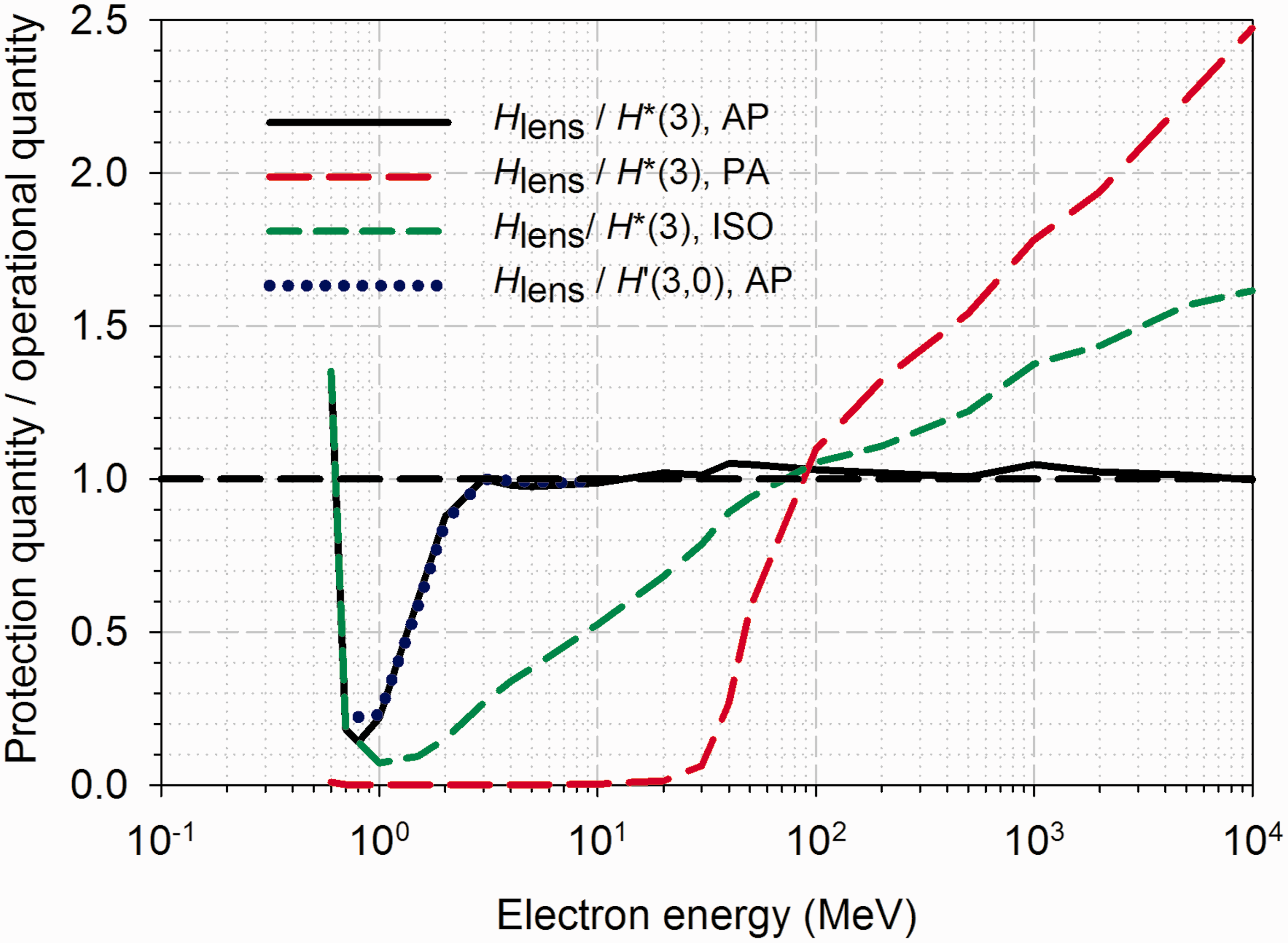
7. Summary
In the past decade, new and significant information has arisen regarding our understanding of the radiation risk of cataract formation. As a result, in Publication 118 (ICRP, 2012), ICRP revised downwards the presumed threshold for tissue reactions of the lens of the eye at 1% morbidity incidence from 1.5 Gy to a current value of no more than 0.5 Gy, a reduction by a factor of 3. Accordingly, the annual limit recommended by ICRP for exposures of the lens of the eye has similarly been revised downwards from 150 mSv to 20 mSv, a reduction of a factor of 7.5. Occupational monitoring of eye exposures to patients, physicians, and radiation workers has thus become of increased importance. To assist in computational assessments of dose to the lens of the eye in exposed persons, Annex F of Publication 116 (ICRP, 2010) provides newly approved reference data on = conversion coefficients for the lens of the eye – absorbed dose to the lens of the eye per particle fluence (pGy cm2) – for external fields of photons, electrons, and neutrons. These reference conversion coefficients were determined using two data sets – one based on Monte Carlo simulations with the Publication 110 reference computational phantoms (ICRP, 2009) and another based upon stylised models of the eye in which the more radiosensitive layer of the lens is included as a target region. Given a priori knowledge of the particle fields at the level of the eye exposure, estimates of lens dose can be assessed using these reference coefficients. Regarding dosimetric monitoring of eye exposures, further work is needed in the development of meaningful operational quantities and their associated calibration phantoms for lens dosimetry, particularly in light of these new and lower annual limits.
