Abstract
Inflammatory myofibroblastic tumor (IMT) is an uncommon neoplasm that predominantly affects children and young adults. It can occur in soft tissues throughout the body, most commonly in the lungs, but it is rare in the larynx. A 24-year-old woman presented to the respiratory department of our hospital with breathing difficulties. Following a cervical computed tomography scan that identified a laryngeal mass, she was referred to our department for further management. The patient underwent transoral endoscopic plasma resection for laryngeal IMT under general anesthesia. The postoperative pathological examination confirmed the diagnosis of IMT. The patient had a favorable postoperative outcome, with no recurrence observed during follow-up.
Introduction
Inflammatory myofibroblastic tumor (IMT) is a distinctive neoplasm composed of myofibroblasts and fibroblastic spindle cells, accompanied by an inflammatory infiltrate consisting of plasma cells, eosinophils, and/or lymphocytes. This tumor has a low metastasis potential.1,2 It can arise at various anatomical sites. However, laryngeal involvement is rarely reported.3,4 IMT is prone to misdiagnosis in clinical practice. Currently, the literature indicates that an external cervical approach is predominantly used for complete resection of laryngeal IMT, whereas the transoral approach is less frequently employed. In this article, we present a case of laryngeal IMT treated with transoral endoscopic plasma resection, aiming to provide new insights into future therapeutic strategies for the management of laryngeal IMT.
Case Report
The patient was a 24-year-old woman who developed persistent hoarseness 6 months prior to presentation. Two months before admission, she began experiencing breathing difficulties, which were more pronounced when lying supine at night. She occasionally awoke with a sensation of suffocation and reported episodes of choking on water. She subsequently sought evaluation at the Respiratory Department of our hospital. Neck computed tomography (CT) revealed a nodular hyperdense lesion on the right vocal cord extending into the glottic cavity. The lesion measured approximately 1.2 × 1.0 × 1.4 cm and resulted in significant luminal stenosis (Figure 1A and B). Consultation with the Department of Ear, Nose, and Throat was recommended. Physical examination revealed a red, broad-based mass in the right subglottic region. The lesion involved the first tracheal ring, the anterior margin of the vocal process, and the posterior commissure (Figure 1C and D). Further inquiry into the patient’s medical history revealed that she had undergone surgery under general anesthesia for thyroid nodules 2 years earlier. Based on her medical history and the current examination findings, we initially suspected that the mass on the right vocal cord might represent a contact granuloma, possibly secondary to prior endotracheal intubation during general anesthesia.
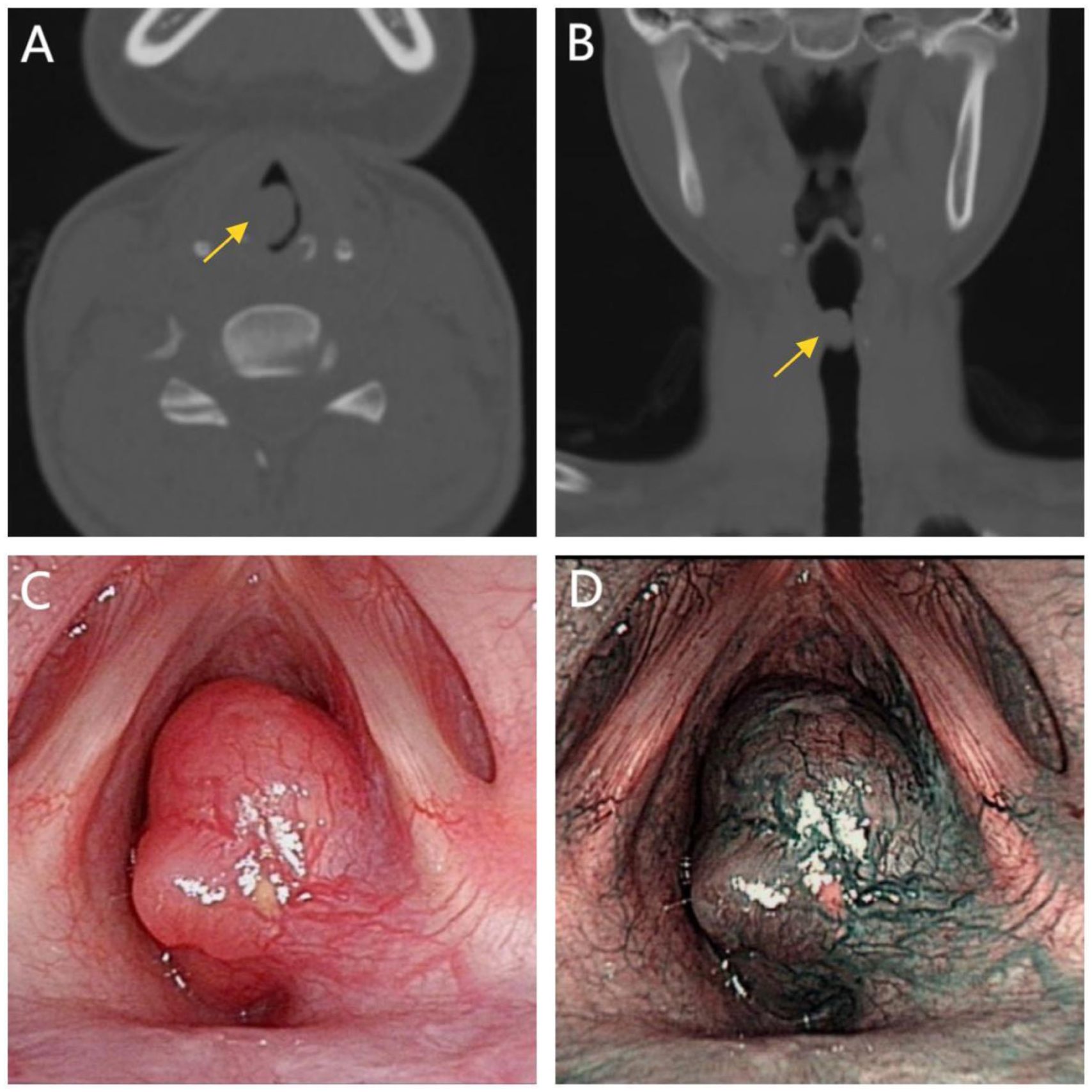
(A) Sagittal CT scan of the neck, with the mass indicated by an arrow. (B) Coronal CT scan of the neck, with the mass indicated by an arrow. (C, D) Laryngoscopy revealed a pale pink mass under the right subglottic area. (C) Illustrates the morphology of the mass under white-light mode, while (D) depicts the lesion under narrow-band imaging mode, demonstrating no abnormal vascular patterns. CT, computed tomography.
We administered a triamcinolone acetonide injection along the tumor margin under laryngoscopic guidance. As the mass was non-adherent to the underlying tissue and its margins were well-demarcated, we proceeded with complete resection using a 403 plasma knife in alternating cutting and coagulation modes. Throughout the procedure, the clear surgical field greatly facilitated identification of the tumor margins and significantly reduced the operative time. Postoperative histopathological examination revealed mixed infiltration of myofibroblasts and inflammatory cells (Figure 2A and B). Immunohistochemical analysis showed the following results: BCL-2 (weakly positive), CD34 (negative), EMA (negative), Desmin (negative), Ki-67 (positive, approximately 10%), MyoD1 (negative), PR (negative), Vimentin (positive), S-100 (negative), ALK (positive), SMA (positive), Calponin (partially positive), and CD10 (positive) (Figure 2C-F). Based on these findings, the patient was diagnosed with laryngeal IMT. As the tumor was completely resected and no evidence of metastasis was observed, the patient did not receive postoperative radiotherapy or chemotherapy. At postoperative day 1 and at 1- and 2-month follow-up visits, the right vocal cord appeared smooth, with no evidence of recurrence, and the patient’s voice had recovered satisfactorily (Figure 3).
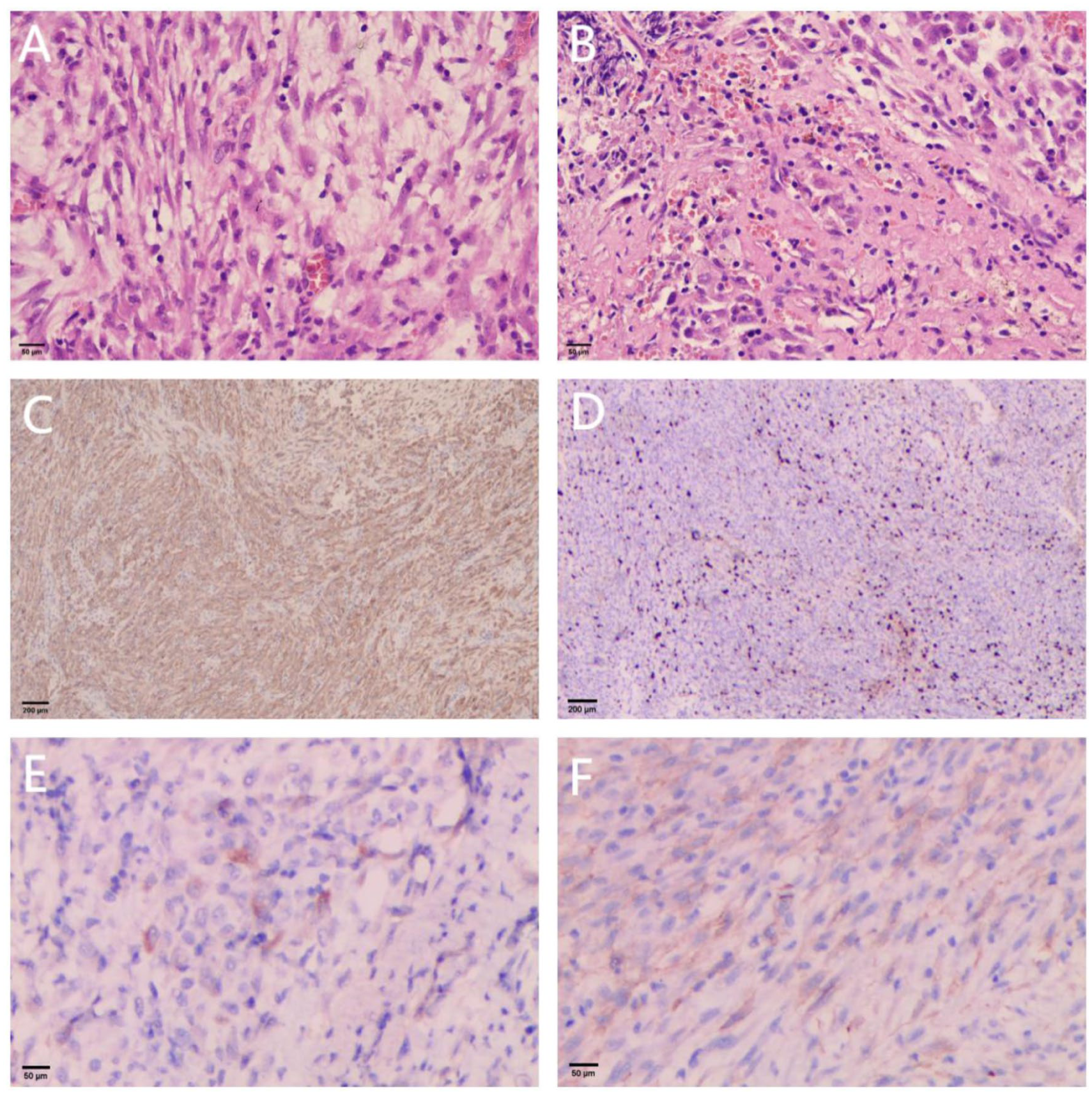
(A, B) HE staining. (A) Shows myofibroblasts with vesicular nuclei, small nucleoli, and eosinophilic cytoplasm; (B) shows stroma with extensive inflammatory cell infiltration. (C-F) Immunohistochemistry staining. (C) Shows ALK (positive); (D) shows Ki67 (positive, approximately 10%); (E) shows Desmin (negative); and (F) shows SMA (positive).
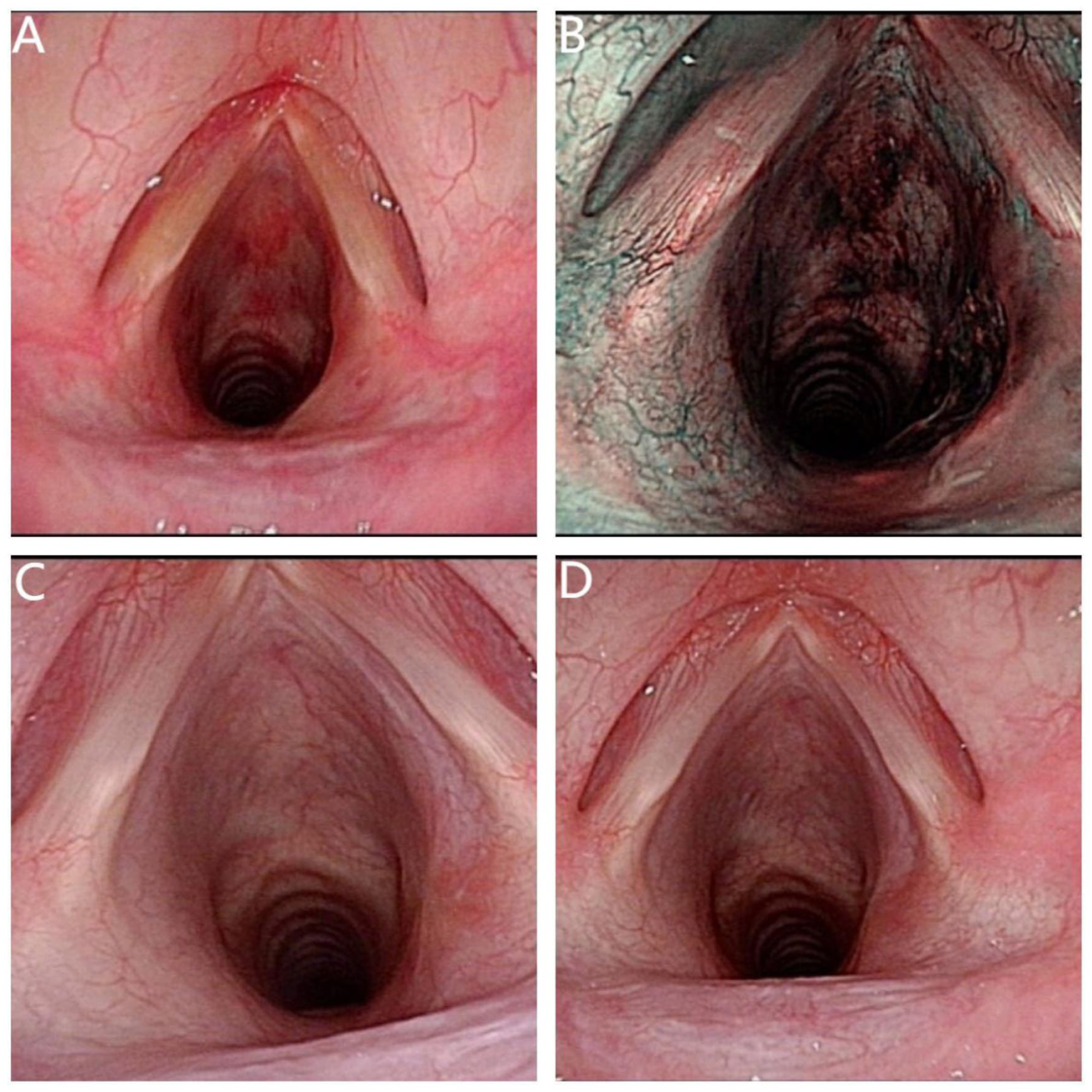
(A, B) Laryngoscopic examination performed on the postoperative day 1. (A) Shows the lesion under white-light mode; (B) shows the lesion under narrow-band imaging mode. (C): Laryngoscopic findings at 1 month postoperatively. (D) Laryngoscopic findings at 1 month postoperatively.
Discussion
IMT is a borderline neoplasm with intermediate biological behavior between benign and malignant. It originates from mesenchymal tissue, is characterized by local invasiveness, and carries a low risk of metastasis. IMT can arise in soft tissues throughout the body, most commonly in the lungs, and is rare in the larynx. 5 The vocal cord is the most frequently involved site, followed by the subglottic region, whereas supraglottic involvement is less common. 6 The etiology and pathogenesis of IMT remain unclear. Current hypotheses suggest that its development may be associated with chronic inflammation, trauma, autoimmune diseases, surgery, viral infections, and abnormal wound healing. 7
IMT lacks specificity in both its clinical presentation and imaging characteristics. Laryngeal IMT typically presents initially with hoarseness. On laryngoscopic examination, the lesion often appears as a polypoid or nodular mass and may be accompanied by inflammatory changes or ulceration. Imaging findings vary widely, ranging from well-circumscribed soft tissue masses to ill-defined infiltrative lesions. This variability is likely related to the relative proportions of inflammatory and fibrotic components within the tumor. In most cases, the lesion appears as a homogeneous, isodense mass. Necrosis and calcification may occasionally be observed. On contrast-enhanced imaging, the tumor generally demonstrates homogeneous enhancement. In more aggressive cases, infiltration of adjacent tissues and varying degrees of bone destruction may be present. This clinical and radiological heterogeneity makes the diagnosis of laryngeal IMT challenging and increases the risk of misdiagnosis.
IMT is primarily diagnosed based on pathological examination. Histologically, it is characterized by a proliferative mixture of myofibroblasts and inflammatory cells, including lymphocytes, plasma cells, eosinophils, and macrophages. According to the cellular and stromal features, IMT can be classified into 3 basic histological patterns: (1) myxoid/vascular (mucous vascular) type, (2) compact spindle cell type, and (3) hypocellular fibrous (oligofibrous) type. 8 Immunohistochemical analysis plays a significant role in the diagnosis, as it helps differentiate IMT from other spindle cell and solid tumors.
The optimal treatment for laryngeal IMT is complete surgical resection. Surgical approaches include endoscopic resection, laryngofissure, partial laryngectomy, and total laryngectomy. Endoscopic resection can be performed using either a CO2 laser or a plasma knife to achieve complete tumor removal. For patients with unresectable lesions, corticosteroid therapy and radiotherapy or chemotherapy may provide benefit.7,9 Despite treatment, disease recurrence can occur following either conservative or surgical management. 10 Recurrent cases may be managed with repeat surgical excision or laryngofissure, and the prognosis is generally favorable. For patients with advanced, unresectable IMT harboring ALK translocations, crizotinib represents a targeted therapeutic option. 11
Conclusion
Laryngeal IMT is a rare borderline neoplasm with an unclear etiology. Its clinical presentation and imaging findings are nonspecific, making diagnosis challenging. Histopathological examination, supplemented by routine immunohistochemistry, remains the gold standard for definitive diagnosis. Radical surgical resection is currently regarded as the most effective treatment, and postoperative prognosis is generally favorable. However, the potential for recurrence and, rarely, metastasis underscores the need for careful long-term follow-up.
Footnotes
Author Note
All authors agree that the receipt of letters, reprint requests, and proofs will be sent by Wanjie Luo.
Ethical Considerations
The research obtained written informed consent from the patient and was approved by the Ethics Committee of the Zigong Fourth People’s Hospital.
Consent for Publication
All authors have approved the content of the manuscript and have agreed to its publication.
Author Contributions
Fengxia Fan is the corresponding author, and she contributed to the conception of the study. Wanjie Luo is the first author who contributed to the writing of this manuscript. Xuemei Pen, Shuang Ma, Tao Xu, and Shenghao Yang contributed to the collection of all images and organizing the information. All authors read and approved the final manuscript. Wanjie Luo, Xuemei Pen, Shuang Ma, Tao Xu, and Shenghao Yang contributed equally to this work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data are included in this paper.
