Abstract
Inflammatory myofibroblastic tumor (IMT) is an uncommon mesenchymal neoplasm rarely encountered in the pediatric larynx, with the subglottis representing an atypical subsite. This report details a diagnostically instructive case of a 10-year-old girl presenting with 18 months of progressive dyspnea and exertional stridor, notably without hoarseness. Fibroendoscopic laryngoscopy identified a well-circumscribed right subglottic mass, confirmed on computed tomography scan to cause partial airway narrowing. The lesion’s firm intraoperative consistency contrasted with common benign mimics. Histopathology revealed spindle cell proliferation with mixed inflammation (histiocytes, giant cells), while immunohistochemistry demonstrated focal anaplastic lymphoma kinase (ALK) and smooth muscle actin (SMA) positivity, CD68+ histiocytes, and CD1A negativity—collectively confirming IMT and excluding Langerhans cell histiocytosis. Complete endoscopic excision was achieved. This case underscores critical learning points: (1) IMT must be considered in children with progressive stridor/dyspnea, even lacking hoarseness, particularly with subglottic localization, and firm consistency; (2) Subglottic IMTs primarily manifest airway obstruction rather than voice changes typical of glottic lesions; (3) Focal ALK expression, present here, supports clonality but does not preclude a favorable outcome with complete resection; and (4) Immunohistochemistry (ALK/SMA/CD68/CD1A) is indispensable for accurate diagnosis and avoiding misdirected therapy. Despite excellent early postoperative results consistent with the generally favorable prognosis of pediatric IMT, vigilance for recurrence—highest within the first 2 years—remains essential. This report reinforces complete conservative resection as the cornerstone of management for localized disease, reserving adjuvant therapies for complex or recurrent scenarios. Multidisciplinary collaboration optimizes outcomes while preserving laryngeal integrity in children.
Keywords
Introduction
Inflammatory myofibroblastic tumor (IMT) is a rare mesenchymal neoplasm, accounting for minority of all soft tissue tumors. 1 IMT demonstrates site-dependent variations in age and gender distribution, most frequently arising in the lungs, abdominal cavity, and retroperitoneum. 2 Other common sites include the gastrointestinal tract, pelvis, mediastinum, genitourinary tract, and head and neck.3,4 Laryngeal IMT is a rare entity most frequently arising in the true vocal cords. 5 Its exact prevalence is difficult to determine due to its rarity, histological heterogeneity, and historical misclassification as a reactive or an inflammatory process. Laryngeal IMT often present with hoarseness or dysphonia. Although head and neck IMTs primarily occur in adults, laryngeal IMTs span a broad age spectrum and exhibit no consistent sex predilection. 6 Histologically, it exhibits spindle-cell proliferations with variable myxoid, hypercellular, or hypocellular fibrous patterns, accompanied by inflammatory infiltrates (plasma cells, lymphocytes), and approximately 50% to 60% demonstrate anaplastic lymphoma kinase (ALK) immunoreactivity. 7 Systemic inflammatory symptoms (e.g., fever, weight loss) occur in few cases and typically resolve postresection. 8 Although categorized as intermediate malignancy with low metastatic risk, local recurrence is possible, particularly in ALK-negative cases; however, prognosis remains favorable, especially in pediatric patients, even with unresectable, or ALK-negative tumors. 9
Case Presentation
A 10-year-old girl presented with a history of dyspnea persisting for approximately 18 months, accompanied by exertional stridor. There was no hoarseness, choking episodes, dysphagia, hemoptysis, or systemic symptoms such as fever or weight loss. Her past medical history included hypothyroidism. On examination, fibroendoscopic laryngoscopy revealed a rounded, well-circumscribed lesion located in the right subglottic region (Figure 1; Supplemental Video 1). To further characterize the lesion, a noncontrast computed tomography scan was performed, which demonstrated an 8 mm × 13 mm mass in the right subglottic area causing partial airway narrowing (Figure 2). The patient underwent a direct laryngoscopy under general anesthesia for lesion removal. Following complete excision, the lesion was described as firm, measuring approximately 5 mm × 8 mm. Histopathology revealed spindle cell proliferation with dense mixed inflammatory infiltration, histocytes, and occasional giant cells. Immunostains showed focal positivity for ALK and smooth muscle actin (SMA). The histiocytes stained positive for CD68, while the lesion was negative for CD1A (Figure 3). These findings are consistent with a diagnosis of IMT.
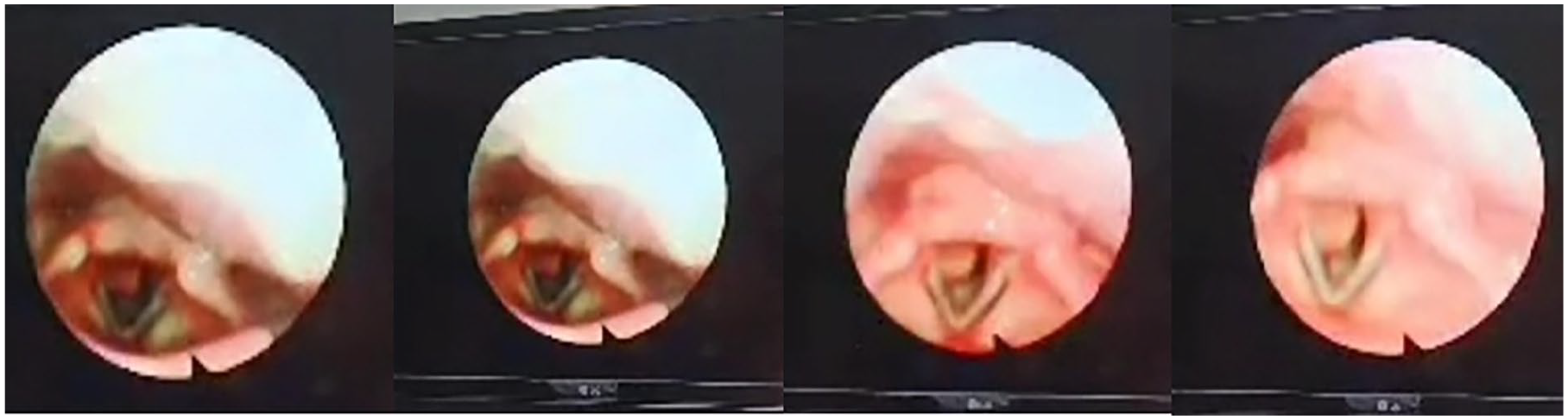
Rounded lesion in the right subglottic region.
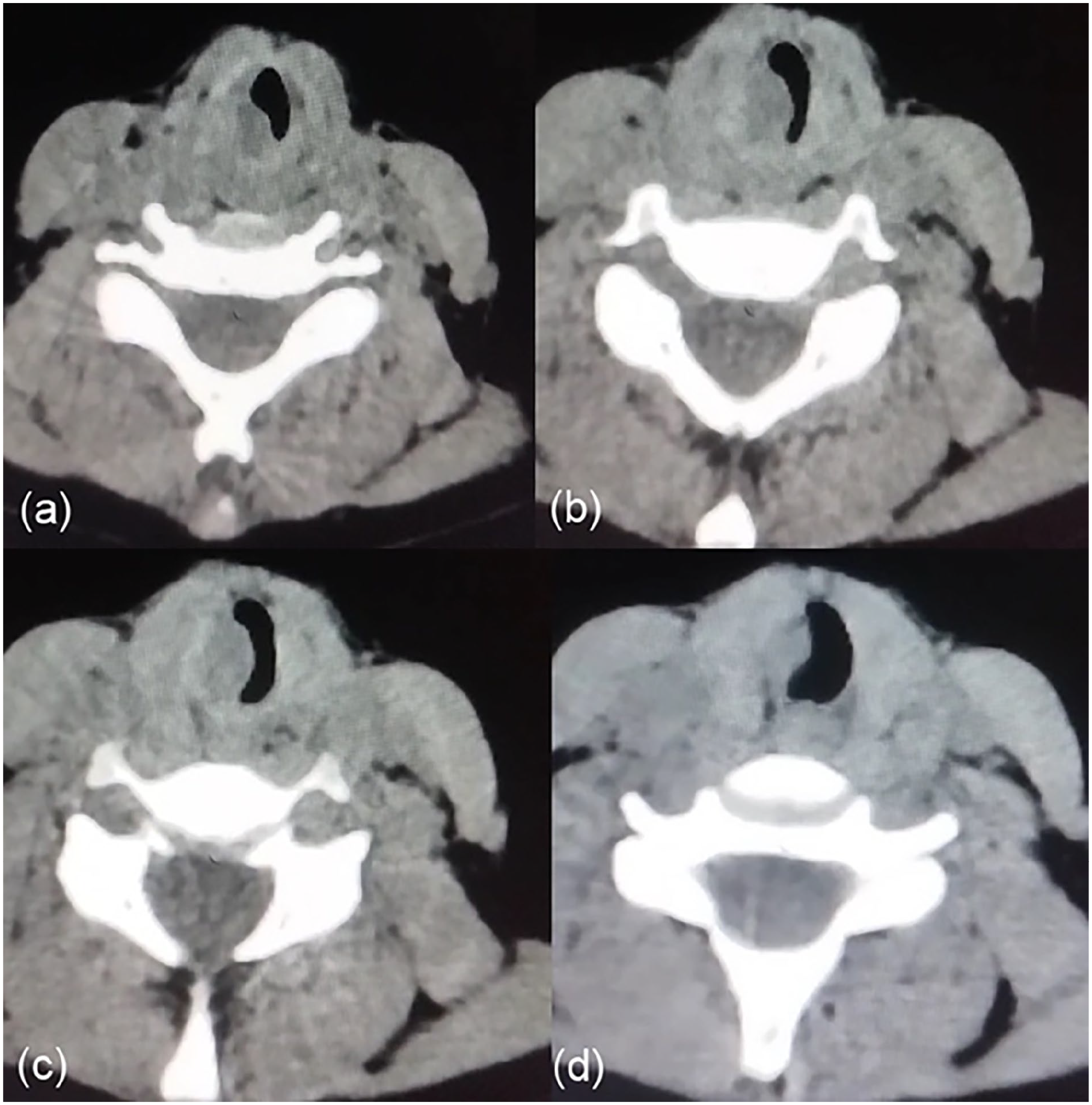
Noncontrast CT scan with sequential descending images demonstrated an 8 mm × 13 mm lesion in the right subglottic area causing narrowing of the airway. CT, computed tomography.
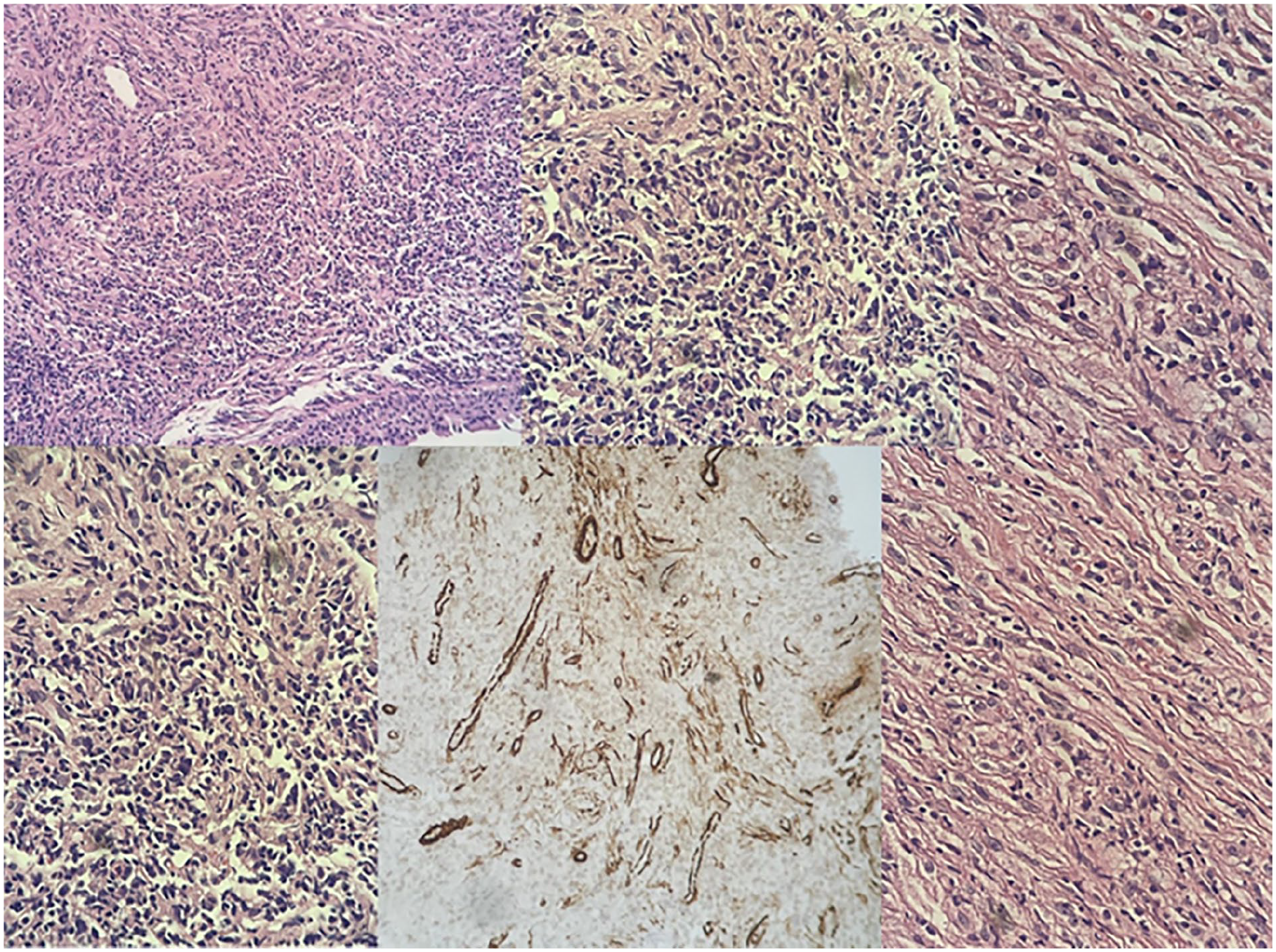
Histopathology revealed spindle cell proliferation with dense mixed inflammatory infiltration, histocytes and occasional giant cells. Immunostains for ALK and SMA markers were focally positive. CD68 marker stains histocytes. CD1A marker in negative. ALK, anaplastic lymphoma kinase; SMA, smooth muscle actin.
Discussion
This pediatric subglottic IMT highlights a rare presentation in an uncommon laryngeal subsite, emphasizing diagnostic and therapeutic nuances. This pediatric subglottic IMT illustrates key clinicopathological features and management considerations for this rare entity. The presentation with prolonged dyspnea and exertional stridor—without hoarseness—reflects the subglottic location’s tendency to cause airway obstruction rather than vocal dysfunction, contrasting with glottic IMTs where hoarseness predominates. The 18-month symptom duration aligns with the typically indolent growth of IMTs, which may delay diagnosis and risk progressive airway compromise.
Histopathological findings of spindle cell proliferation with mixed inflammation (histiocytes, giant cells), focal ALK/SMA positivity, and CD68+ histiocytes are diagnostic hallmarks of IMT. 4 The focal ALK expression supports a clonal neoplastic process, consistent with reports that 50% to 60% of IMTs harbor ALK rearrangements. 4 The CD68 positivity and CD1A negativity effectively exclude Langerhans cell histiocytosis, a critical differential for pediatric subglottic masses. 3 Complete surgical excision remains the cornerstone of management to minimize recurrence risk, estimated at 8% to 18% for laryngeal IMTs. 5 The endoscopic approach used here is favored for well-circumscribed lesions, offering reduced morbidity and faster recovery compared to open techniques while achieving clear margins. 6 The firm intraoperative consistency noted in this case contrasts with the friability of papillomas or granulomas, providing a useful clinical clue to IMT. Several features underscore this case’s instructive value: the subglottic location represents a less common site for laryngeal IMTs compared to the vocal cords, accounting for only ~22% of cases. 5 The absence of systemic symptoms like fever or weight loss aligns with most laryngeal IMT presentations, though such features occasionally occur in extrapulmonary sites. 2 The excellent early postoperative outcome reinforces that pediatric IMTs generally have favorable prognoses with complete resection, even when ALK expression is focal rather than diffuse. Long-term vigilance remains essential given recurrence risks, particularly within the first 2 years. 6 While adjuvant therapies like corticosteroids or ALK inhibitors are reserved for unresectable or recurrent disease, their role is less defined in completely resected pediatric cases like this one. 4 This case reinforces that IMT should be considered in children with progressive stridor or dyspnea, even without hoarseness, particularly when lesions demonstrate firm consistency and subglottic localization. The subglottic location poses unique airway management challenges, as circumferential growth may necessitate complex reconstruction if resection compromises structural integrity—a concern less prominent in supraglottic IMTs. While systemic symptoms were absent here, elevated inflammatory markers (e.g., CRP/ESR) reported in 15% to 30% of extrapulmonary IMTs could serve as postoperative surveillance tools, particularly in ALK-negative cases with higher recurrence risk. 8 The focal ALK expression warrants discussion of reflex molecular testing, as ALK fusions (detected in 50%-60% of IMTs) may predict responsiveness to targeted agents like crizotinib if recurrence occurs, potentially avoiding cytotoxic chemotherapy. 9 This contrasts with pulmonary inflammatory pseudotumors, which historically lacked targetable drivers and showed progression from organizing pneumonia to fibrous histiocytoma patterns. 8
Analysis of 31 laryngeal IMTs revealed recurrences in 9 cases (29%), predominantly within 2 to 12 months postresection—shorter than the 24-month window noted in extrapulmonary IMTs. Our patient’s focal ALK expression, though prognostically favorable, warrants vigilance during this high-risk period. Recurrence correlated strongly with incomplete excision (e.g., endoscopic debulking without clear margins) or steroid monotherapy, reinforcing complete resection as imperative. 5 Unlike pulmonary IMTs, laryngeal IMTs show inconsistent steroid sensitivity. Case 1 in Alhumaid’s series received postoperative corticosteroids for granuloma prevention, yet steroids failed entirely in a steroid-refractory subglottic IMT (Case 2). This heterogeneity suggests steroids may aid inflammation control postresection but lack efficacy as primary therapy. In pediatric subglottic cases like ours, steroids remain adjunctive—reserved for residual inflammation or granuloma mitigation. 5 Three reported laryngeal IMTs (9.7%) followed direct trauma (intubation, thyroglossal cyst excision, or airbag injury). While absent in our case, this association implies mucosal injury could initiate dysregulated myofibroblast proliferation in susceptible individuals. Subglottic tissue—vulnerable to mechanical stress—may thus be predisposed, particularly in children with smaller airways. 5 Alhumaid stresses that pediatric subglottic IMTs are frequently misdiagnosed as recurrent respiratory papillomatosis or foreign-body reactions. Our case’s firm consistency (intraoperatively) and CD1A negativity provided critical differentiation from Langerhans histiocytosis—a common subglottic mimic in children. Immunohistochemistry (ALK/SMA/CD68) remains essential to avoid unnecessary chemotherapy or radiation. 5
Finally, multidisciplinary planning is critical for pediatric subglottic lesions; otolaryngologists must balance radical excision against laryngeal development preservation, while oncologists should reserve therapies like methotrexate/vinblastine (ORR 54% in advanced IMTs) for unresectable scenarios given this case’s excellent surgical outcome. 9
Conclusion
This pediatric subglottic IMT underscores the diagnostic importance of considering rare mesenchymal tumors in children with progressive stridor, even without hoarseness. The atypical location and firm endoscopic consistency provided critical clinical clues, while ALK/SMA immunopositivity and CD1A negativity confirmed the diagnosis and excluded mimics like Langerhans histiocytosis. Complete endoscopic excision achieved a favorable outcome, aligning with the generally excellent prognosis of pediatric IMTs despite focal ALK expression. However, the risk of recurrence—particularly within 24 months—necessitates vigilant long-term surveillance. This case reinforces that conservative resection suffices for localized disease, reserving adjuvant therapies for complex scenarios. Multidisciplinary collaboration remains essential to balance curative intent with laryngeal preservation in pediatric airways.
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases. Written informed consent was obtained from the patient’s legal guardian for publication of this case report and accompanying images.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statements
The data that support the findings of this study are available from the author or corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
