Abstract
Ewing’s sarcoma (ES), the second most common primary bone cancer in adolescents, predominantly affects males with a peak incidence at 15 years of age. While extraskeletal presentations are rare, tongue involvement is uncommon, with few cases reported globally. ES typically manifests as a firm swelling, often asymptomatic or accompanied by mild discomfort, and is managed primarily with chemotherapy or surgery. In this report, we present a rare case of a 17-year-old female with extraskeletal ES presenting as progressive swelling on the left side of the tongue. The patient responded impressively to chemotherapy, then was given radiation therapy with complete resolution of the disease. This case highlights the diagnostic and therapeutic challenges of managing such an unusual presentation, contributing valuable insights to the literature, and guiding future care for similar cases.
Introduction
Ewing’s sarcoma (ES), a highly aggressive malignancy, is the second most common primary bone cancer in adolescents and young adults, peaking around 15 years of age. Representing <5% of all soft tissue sarcomas 1 exhibits a male predominance with a 3:2 male-to-female ratio. 2 The ES family of tumors includes classical bone sarcomas, extraskeletal ESs, and malignant primitive neuroectodermal tumors. 3 While skeletal forms are more prevalent, affecting the long bones of the limbs (58%), extraskeletal manifestations are rare, typically found in the lower limbs, chest wall, paravertebral tissues, and retroperitoneum, with only 1% to 4% of cases occurring in the head and neck region. 4
While its precise origin remains uncertain, it is hypothesized to arise from neuroectodermal cells. Other potential origins include primitive mesenchymal cells in the bone marrow and immature reticular cells. Genetic translocations, particularly between chromosomes (11;22) (q24;q12) and (21;22) (q22;q12), are thought to play a significant role in its development. 5
This case concerns a 17-year-old female diagnosed with ES, who developed progressive swelling on the left side of her tongue. The case highlights the clinical, histological, and immunohistological findings and the challenges encountered during diagnosis and treatment.
Case Report
A 17-year-old female with no significant past medical or surgical history presented with a 1-month history of progressive posterior tongue swelling. On physical examination, a large, irregular, exophytic mass was noted on the posterior tongue, exhibiting areas of ulceration, necrosis, and surrounding erythema features clinically suggestive of a malignant neoplasm (Figure 1).
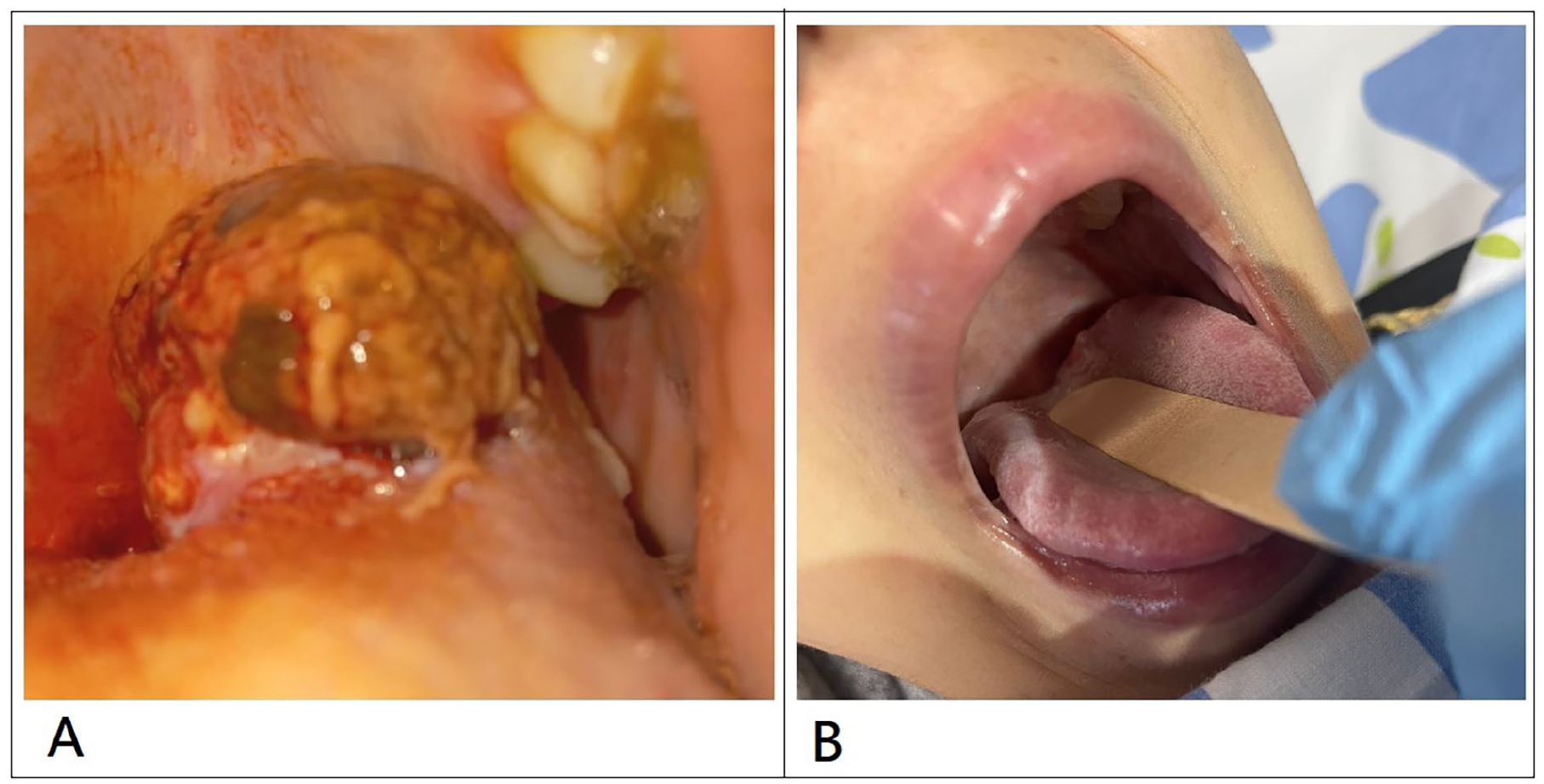
Ewing sarcoma of the tongue before and after treatment. (A) Ulcerated tongue mass prior to treatment. (B) Complete clinical resolution post-treatment.
A contrast-enhanced neck computed tomography (CT) revealed a 3-cm isodense mass at the left base of the tongue, abutting the left lateral wall of the hard palate within the oropharynx and causing narrowing of the oropharyngeal inlet. Subsequent magnetic resonance imaging (MRI) of the neck demonstrated a 4.3 × 2.5 cm lesion involving the left anterior two-thirds of the tongue. The mass appeared iso-hypointense on T1-weighted images, hyperintense on T2/STIR sequences, and showed diffusion restriction on diffusion-weighted imaging with near-homogeneous post-contrast enhancement. There was evidence of invasion into the base of the tongue with mild extension into the left vallecula and anterior tonsillar pillar. Multiple subcentimeter-sized cervical lymph nodes were noted, some exhibiting contrast enhancement and diffusion restriction (Figure 2).
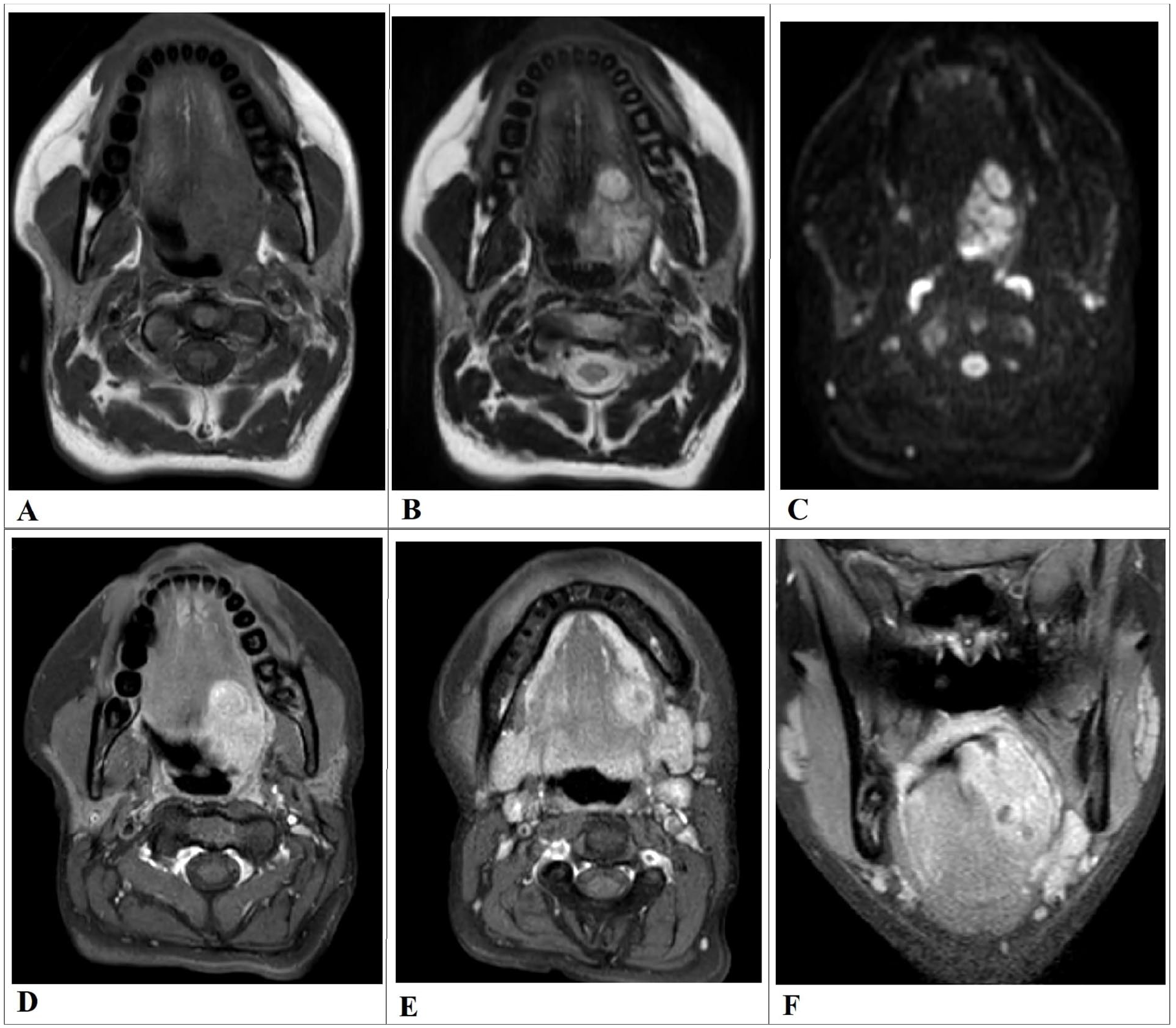
Multisequence MRI of the neck demonstrates a left-sided exophytic polypoid mass arising from the base of the tongue. (A) The lesion appears iso-hypo-intense on T1-weighted imaging and heterogeneously hyperintense on T2-weighted imaging (B), DWI reveals restricted diffusion (C), suggestive of high cellularity; the ADC map is not shown. Following intravenous contrast administration, the mass exhibits vivid enhancement on contrast-enhanced T1-weighted imaging (D). Mildly enlarged, enhancing upper cervical lymph nodes are seen (E), raising suspicion for nodal metastasis. In addition, coronal post-contrast T1-weighted MRI showing a well-defined, heterogeneously enhancing mass centered in the posterior tongue, with extension into the base of tongue and possible involvement of the floor of mouth (F). ADC, apparent diffusion coefficient; DWI, diffusion-weighted imaging; MRI, magnetic resonance imaging.
An excisional biopsy of the left tongue mass was performed. Histopathological examination revealed a small round blue cell tumor with strong membranous CD99 and nuclear FLI-1 positivity on immunohistochemistry, and negative for CK-pan, SOX10, Desmin, CD34, synaptophysin, chromogranin, GFAP, CK7, CK20, actin, S100, and P63. These findings were consistent with ES of the tongue (Figure 3).
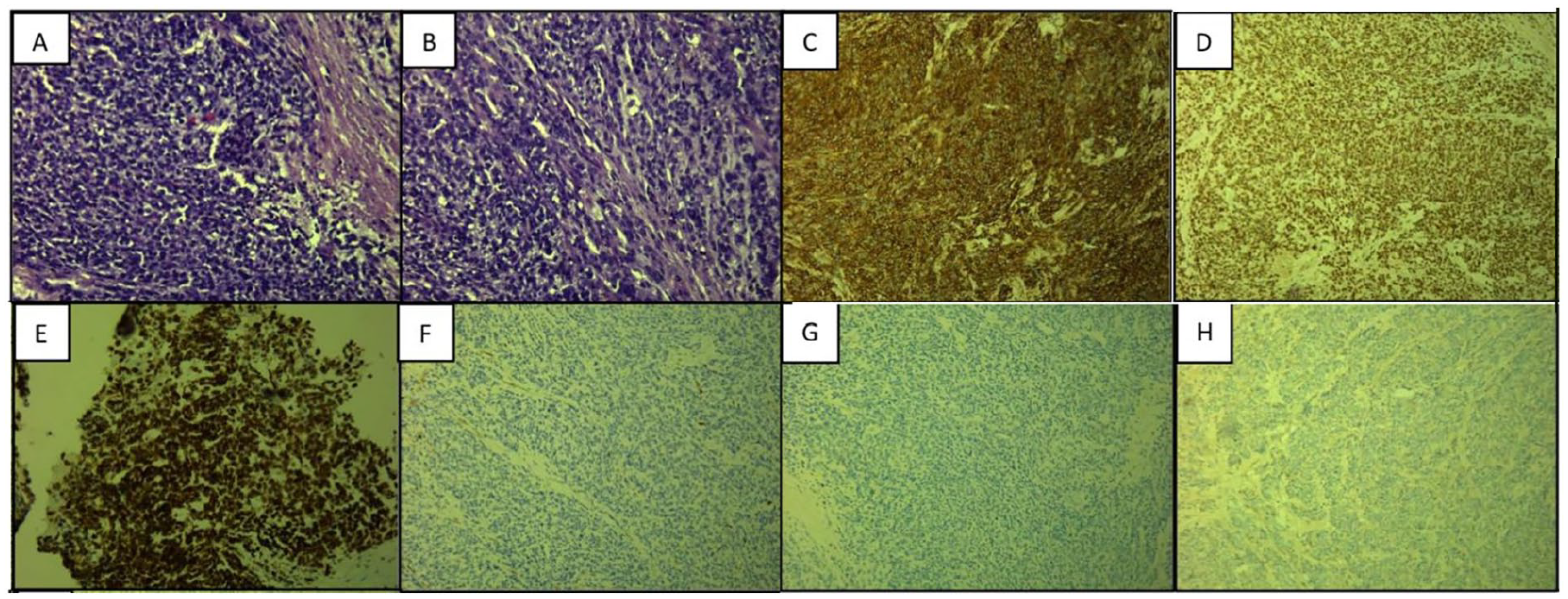
Representative histopathological and immunohistochemical features of an undifferentiated small round-cell tumor showing classic morphology (A, B), CD99 and FLI1 positivity (C, D), WT1 focal expression (E), and absence of Desmin, S100, and CK-pan staining (F–H).
Comprehensive staging investigations included bilateral bone marrow biopsies, which revealed no evidence of malignancy. A whole-body PET-CT scan demonstrated a hypermetabolic mass at the left paracentral tongue base, measuring 4 × 3.2 cm with a maximum standardized uptake value of 14.3 (Figure 4). The patient was initiated on chemotherapy according to the AEWS0031 protocol, which consists of 14 total cycles alternating between vincristine, doxorubicin, and cyclophosphamide (VDC) and ifosfamide with etoposide (IE). After 4 cycles, a follow-up MRI revealed a marked treatment response with clinical complete resolution of the lesion.
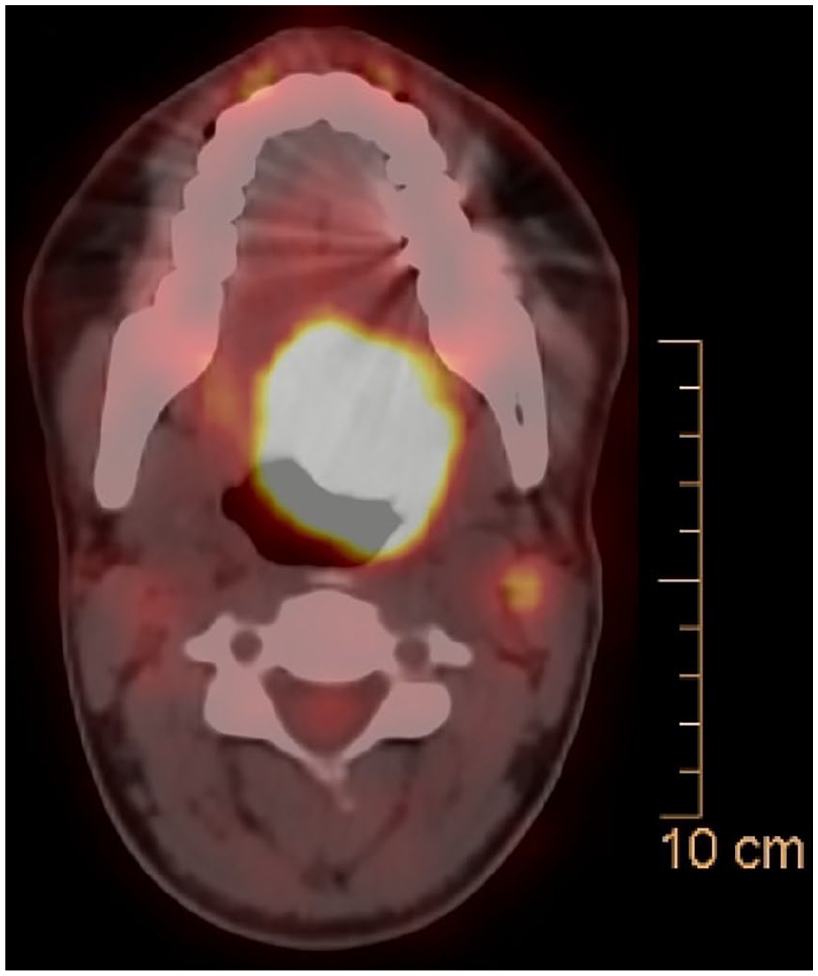
PET-CT scan reveals an intensely hypermetabolic mass localized to the left base of the tongue, consistent with a metabolically active tumor. Hypermetabolic activity is also noted in the left upper cervical lymph nodes, supporting metastatic nodal involvement. CT, computed tomography.
A multidisciplinary tumor board reviewed the case and concluded that surgical resection would result in significant morbidity and impact quality of life. Therefore, the decision was made to continue with non-surgical management. The patient received an additional 4 cycles of chemotherapy with good tolerance and continued response, followed by definitive radiotherapy (50.4 Gy in 28 fractions). The AEWS0031 protocol includes 14 cycles of alternating VDC and IE chemotherapy and has shown improved outcomes in localized ES. In our case, although an early treatment response was seen after 4 cycles, the patient completed the full regimen to ensure durable remission. Post-radiotherapy, MRI demonstrated complete resolution of the disease. The patient is currently under regular surveillance at the oncology clinic, with the last PET scan after 6 months showing no evidence of recurrence (Figure 5).
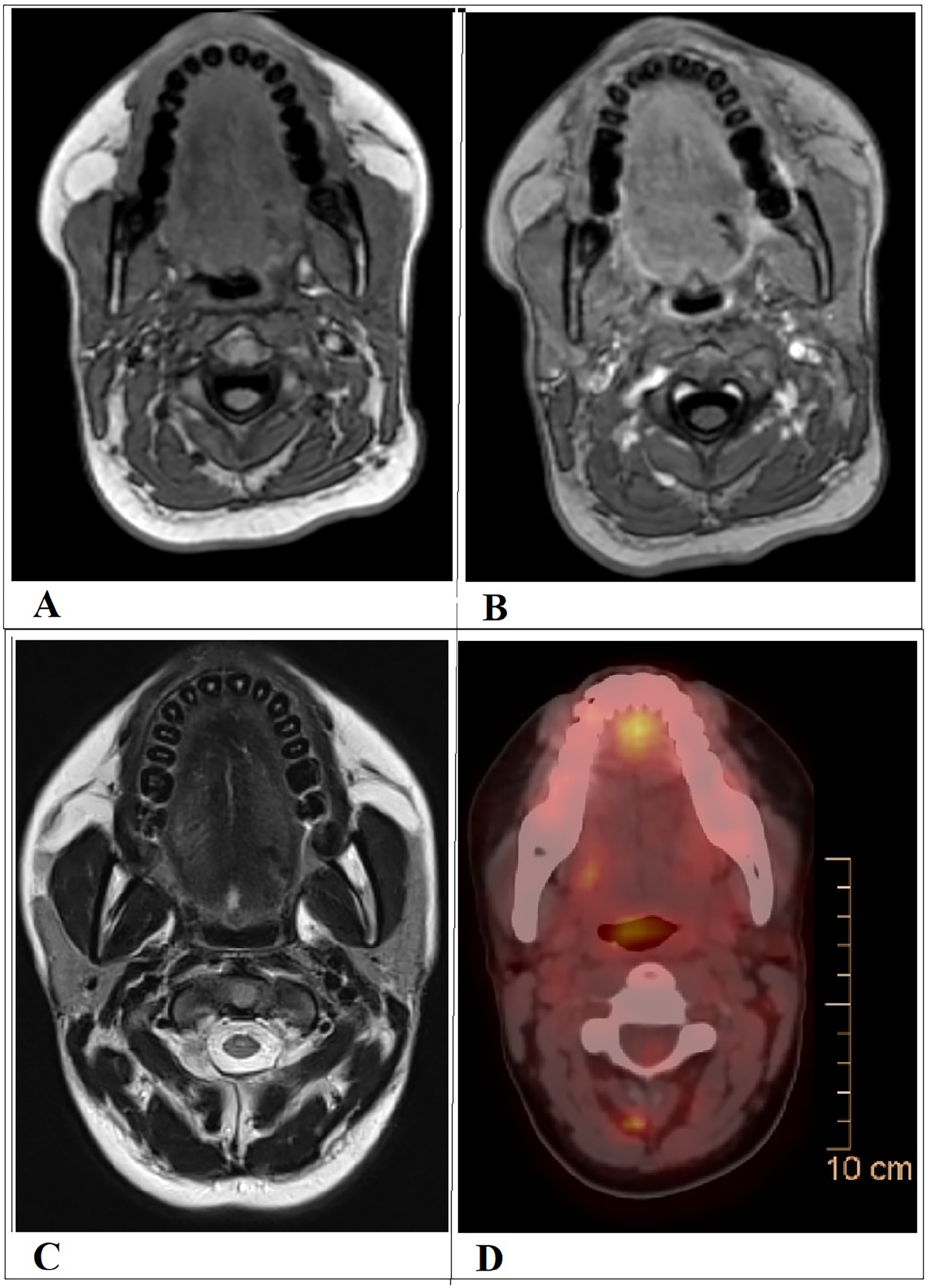
Follow-up multisequence MRI demonstrates complete resolution of the previously noted left tongue base mass. A hypointense fibrotic band is seen at the prior tumor site across T1-weighted (A), post-contrast T1-weighted sequences (B), and T2-weighted (C), indicating post-treatment fibrosis. PET-CT (D) confirms the absence of residual hypermetabolic activity, consistent with a complete metabolic response. CT, computed tomography; MRI, magnetic resonance imaging.
Discussion
Here, we report a young female patient with a rare extraosseous localization of ES. She achieved a successful recovery following treatment with chemotherapy.
ES, a rare and aggressive small round blue cell tumor, was first identified by Dr James Ewing in 1921. 6 It primarily affects the pelvis and long bones, with only 3% of cases occurring in the oral and maxillofacial region. Of these, the mandible is most commonly involved (68%), followed by the maxilla (28%) and soft tissues (2.8%). Extraosseous forms of ES account for 1.5% to 4% of pediatric sarcomas and 4% of soft tissue tumors. They show a male predominance and are typically present between ages 5 and 25, 7 as demonstrated in the patient’s age.
The clinical presentation of ES varies depending on tumor location. 7 In our patient, a firm, well-defined mass with painful ulcerated regions was observed, covered by whitish mucosa. Such findings underscore the diagnostic challenge posed by ES, given its nonspecific radiological and histological features, which overlap with other small round-cell tumors. 8
Genetic analysis, including EWSR1-FLI1 translocation, a characteristic finding in 85% to 90% of ES cases, further solidified the diagnosis.7,9 However, molecular testing for EWSR1 rearrangement was not performed in this case, as it was not available in our institution; the diagnosis of ES was supported by the classical histopathological features and the characteristic immunohistochemical profile, including diffuse membranous CD99 and nuclear FLI-1 positivity. These findings, combined with the tumor’s anatomical location, patient age, and robust clinical response to Ewing-specific chemotherapy (AEWS0031 protocol), provide strong support for the diagnosis. While molecular confirmation can further refine subclassification in round-cell sarcomas, it may not be essential when histologic and immunophenotypic features are definitive and the therapeutic response is consistent with classical ES.
Radiologically, ES often appears as a heterogeneous mass on CT and demonstrates distinct MRI characteristics, including isointense signals on T1-weighted images and hyperintense signals on T2-weighted images. However, these imaging modalities are primarily valuable for assessing tumor extent, resectability, and metastatic spread rather than confirming the diagnosis. 3 In this case, imaging played a role in staging and evaluation of the spread.
Historically, the prognosis of ES was poor, with treatments limited to surgery and radiotherapy, survival rate of roughly 20% after 3 years. 9 The introduction of neoadjuvant chemotherapy, coupled with advancements in diagnostic techniques and multimodal therapy, has revolutionized patient outcomes due to its high chemosensitivity. Modern approaches integrating surgical excision, chemotherapy, and radiotherapy have significantly improved survival rates.7,10 Depending on whether adults or children are involved, overall survival with this technique presently ranges between 59% and 81% after 3 years, and the disease-free survival rate is roughly 60% after 5 years. 9 Through multidisciplinary techniques and multi-institutional trials, improvements in ES outcomes have been made. 11 This case highlights the importance of a multidisciplinary approach in managing complex oncological cases, ensuring thorough investigation and tailored treatment strategies.
While numerous studies have examined factors influencing prognosis, such as tumor size, stage, and patient age, most focus on osseous ES. 11 This case highlights the importance of comprehensive imaging and histopathological evaluation. It underscores the potential for favorable outcomes in patients with rare presentations of ES when treated with a multidisciplinary approach.
Conclusion
This case highlights the rarity of ES arising in the tongue and underscores the value of a multidisciplinary approach in achieving curative outcomes while minimizing functional impairment. Early diagnosis, the appropriate use of imaging and histopathological tools, and a tailored treatment plan were pivotal to the successful management of this patient.
Footnotes
Acknowledgements
The authors are grateful to the doctors in the radiology and pathology departments at Augusta Victoria Hospital for their assistance in collecting patient data.
Ethical Considerations
Ethical approval to report this case was obtained from the Research Ethics Committee at Al Quds University.
Consent for Publication
Written informed consent for the publication of this case report, including all clinical details and accompanying images, was obtained from the patient’s legally authorized representative (parent/legal guardian), as the patient is a minor (17 years old). A copy of the written consent is available for review by the Editor-in-Chief of this journal upon request.
Author Contributions
Elias Edward Lahham: led the writing and editing of the main manuscript, coordinated contributions from co-authors, and finalized content for submission. Jamal Al-Sa’ed and Mayar Karaki: contributed extensively to the drafting of the main manuscript and assisted in literature review and clinical discussion. Mahmoud Ramahi: provided expert input on radiological imaging, interpreted scans, and supported final editing of the manuscript. Mohammad Fatayer: conducted pathology review, including histological slide evaluation, and contributed to the discussion of pathological findings. Khadra Salami and Hani Saleh: reviewed the patient’s oncology records and chemotherapy regimen, and contributed expertise in pediatric oncology for case analysis. Jalal Qawasmeh: oversaw the radiation oncology aspects of the case, including development and review of the radiation treatment plan.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used to support the findings of this study are included in the article.
