Abstract
Granulomatosis with polyangiitis (GPA), formerly known as Wegener’s granulomatosis, is a small vessel vasculitis characterized by necrotizing granulomas affecting various organs. This case report presents a 79-year-old male with GPA manifesting as pyoderma gangrenosum-like cutaneous lesions involving the bilateral parotid glands. The patient presented with painful, progressively-enlarging wounds that were affecting his quality of life. The initial workup was inconclusive, and the patient was briefly lost to follow-up. Upon return, the lesions had progressed to salivary fistulas, and the patient exhibited facial nerve weakness. Chest radiographs revealed worsening lung densities. A punch biopsy indicated granulomatous inflammation with necrosis, and autoimmune lab work showed elevated antinuclear antibody, erythrocyte sedimentation rate, C-reactive protein, and cytoplasmic antineutrophilic cytoplasmic antibodies levels. The patient was diagnosed with GPA and started on Rituximab therapy. The patient responded well to therapy and had resolution of ulcerations and facial nerve weakness. This case is a rare presentation of GPA affecting bilateral parotid glands with involvement of the facial nerve. It underscores the importance of considering GPA in the differential diagnosis of necrotic skin lesions and highlights the challenges in diagnosing GPA due to nonspecific histologic findings and wide variation of clinical presentation.
Introduction
Granulomatosis with polyangiitis (GPA), formerly known as Wegener’s granulomatosis, is a small vessel vasculitis characterized by necrotizing granulomas affecting different organs throughout the body.1-3 GPA primarily affects the upper respiratory tract, lung, and kidneys with the kidneys subject to necrotizing glomerulonephritis. Head and neck manifestations of GPA are commonly seen affecting the nose, paranasal sinuses, middle and inner ear, and the subglottis. 4 The etiology of the disease is largely unknown, but it is believed to be an autoimmune disease with a poorly understood environmental trigger. As an autoimmune disease, it is associated with cytoplasmic antineutrophilic cytoplasmic antibodies (c-ANCA). Among the many ways GPA affects patients, cutaneous manifestations also occur and present as palpable purpura, papules, and nodules, and necrotizing ulcers similar to pyoderma gangrenosum. 5 In this study, we present a case of a patient with pyoderma gangrenosum-like cutaneous lesions involving bilateral parotid glands.
Case Presentation
The authors present a 79-year-old male, who presented to the otolaryngology clinic with draining wounds involving bilateral parotid glands that developed 6 weeks prior and have been progressively getting larger. The wounds were painful and impacting the patient’s ability to eat and sleep. The initial workup confirmed that the patient was negative for HIV, and he was scheduled for a fine needle aspiration (FNA) and biopsy with a presumed diagnosis of Warthin’s tumor. FNA did not give a diagnosis as the fine needle failed to provide enough tissue to fully evaluate the skin pathology. The patient was briefly lost to follow-up but had two hospital visits during that time. The patient’s skin ulcers had progressed to salivary fistulas involving the bilateral parotid glands (Figure 1). In addition to worsening of the skin ulcerations, the patient’s facial nerve also became involved on the left side only. There was a slight, but noticeable weakness of the upper branch documented as a House-Brackmann 3 out of 6 weakness. The patient also had chest radiographs obtained during his hospital visits that showed lung densities that were progressively worsening.
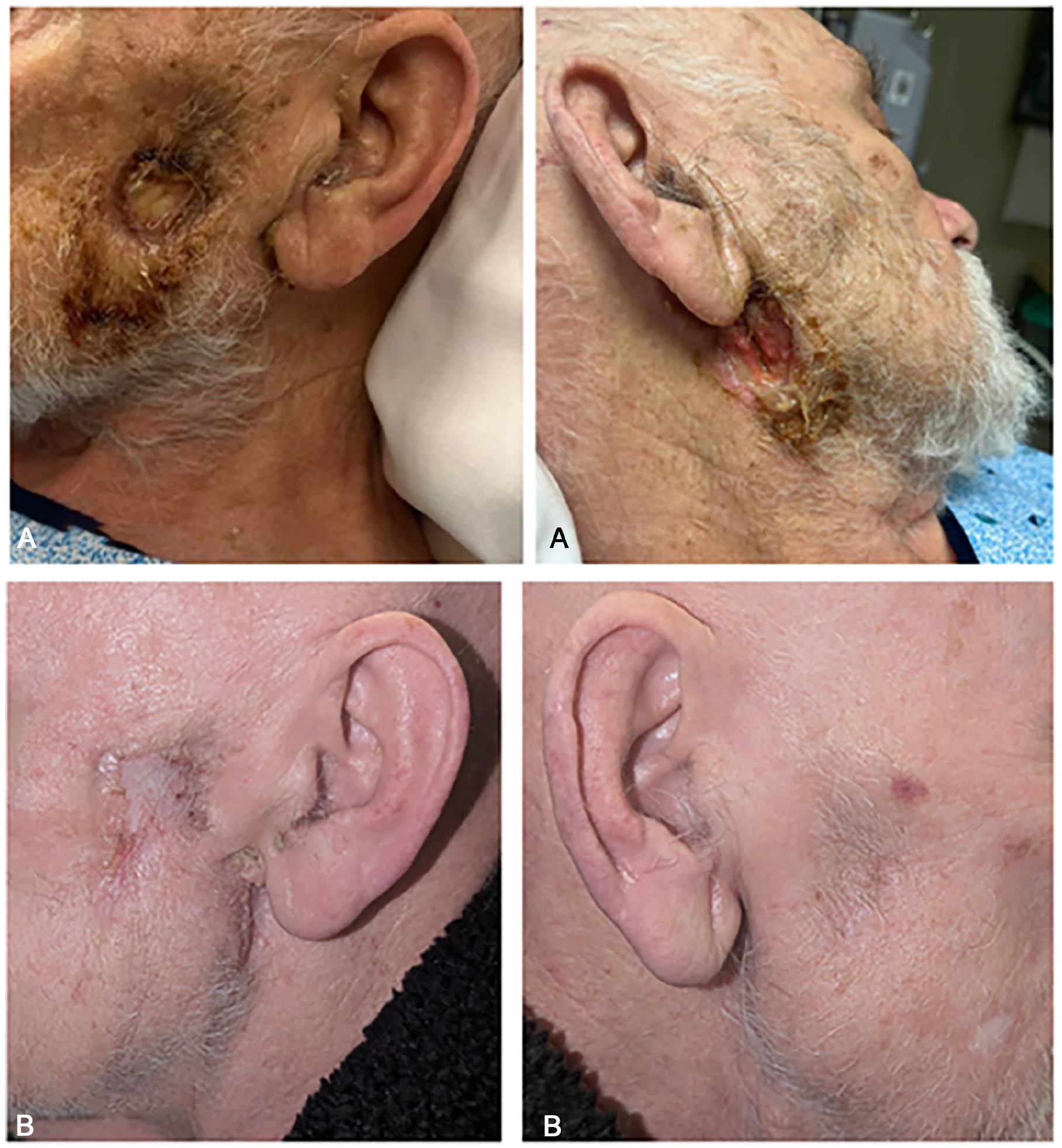
(A) Left and right bilateral parotid gland necrotic ulcerative skin lesions seen in clinic prior to treatment. (B) Left and right bilateral parotid gland necrotic ulcerative skin lesions seen in clinic following the initiation of treatment.
Due to the ulcerative skin lesions resembling pyoderma gangrenosum, a punch biopsy was performed on the necrotic tissue. There was evidence of granulomatous inflammation with necrosis, but these were largely non-specific results (Figure 2). Based on the pathology specimen, the concern for pyoderma gangrenosum was noted, but clinical history suggested there was another cause. There was no evidence of necrotizing vasculitis, but there was a concern for GPA which can present with cutaneous pyoderma gangrenosum-like ulcerations. The pathologist reviewing the patient’s case also reviewed specimens from the patient’s recent prostate procedure, which demonstrated acute, chronic, and granulomatous inflammation with necrosis. This too is also a non-specific finding in the prostate, but since the lungs and skin were also involved by an acute and granulomatous inflammatory process with necrosis, GPA was favored among the differential diagnoses.
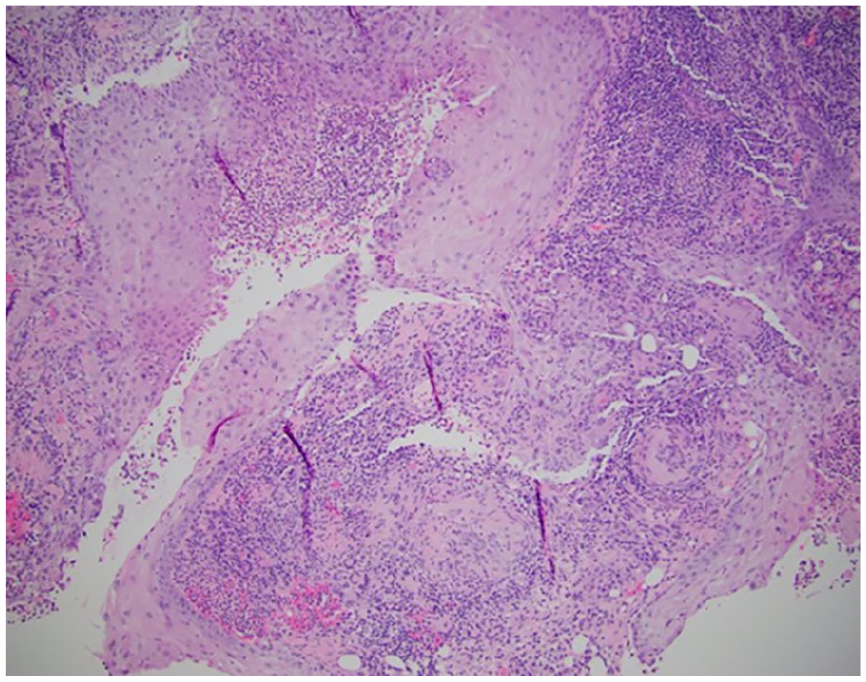
Soft tissue mass of the parotid gland showing small fragments of tissue with acute and granulomatous inflammation with necrosis.
Autoimmune lab work was ordered for the patient, which revealed an elevated antinuclear antibody, erythrocyte sedimentation rate, C-reactive protein, and c-ANCA with a 1:320 ratio (normal <1:20). Myeloperoxidase antibodies and Proteinase 3 (PR3) levels were not found to be elevated. Renal workup also revealed significant proteinuria, red blood cells were present in abundance, and there was an elevated protein-to-creatinine ratio. Based on the totality of results (Table 1), the patient was diagnosed with GPA and referred to rheumatology for further evaluation. The patient was started on induction therapy with Rituximab and subsequently placed on maintenance therapy with Rituximab. The patient responded well to treatment. The ulcerations (Figure 1) and facial nerve weakness resolved returning to a House-Brackmann 1.
Patient’s Critical Laboratory Values That Aided in the Diagnosis and Treatment of GPA. This Table Also Provides Lab Value Trends After Initiation of Therapy.
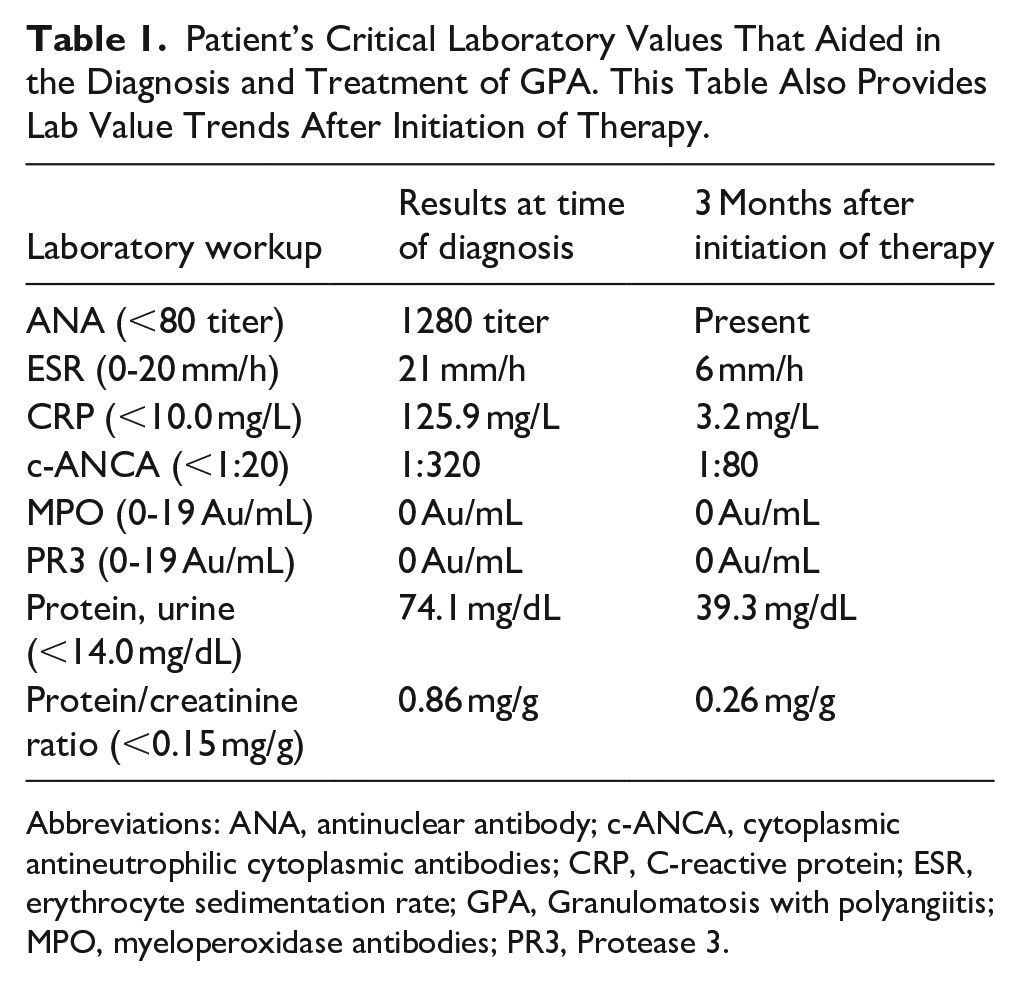
Abbreviations: ANA, antinuclear antibody; c-ANCA, cytoplasmic antineutrophilic cytoplasmic antibodies; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; GPA, Granulomatosis with polyangiitis; MPO, myeloperoxidase antibodies; PR3, Protease 3.
Discussion
GPA is an uncommon multisystem disease characterized by necrosis, granulomatous disease, and small vessel vasculitis. While it is seen in all racial groups, it predominantly affects Caucasians of both sexes and the mean age at diagnosis is 40. 6 The characteristic features of this disease are granulomas of the upper airway, necrotizing small vessel vasculitis, and glomerulonephritis. 1 Otolaryngologic features are typically the initial clinical manifestations seen in patients with the nasal cavity being the most involved followed by otologic manifestations. 4 As in the case of our patient, facial nerve palsy can be seen in 7% to 10% of patients.4,6 Cutaneous involvement can occur in 50% of patients with a wide variance of clinical presentation. 7 Our patient presented with progressively worsening pyoderma granulosum-like necrotic skin lesions. Given the proximity of the skin lesions to the parotid glands, as the lesions progressed, they fistulized with the parotid gland tissue leading to salivary drainage from the skin whenever the patient ate.
The differential diagnosis is broad given the multisystem involvement and includes rheumatoid vasculitis, pyoderma gangrenosum, lymphoma, tuberculosis, infectious etiology, and sarcoidosis. 6 The patient presented also had a broad differential, his history of prior prostate abscess, fevers, and parotid gland lesions was originally assumed to be infectious. When the patient failed to respond to antibiotics, the differential had to be broadened. The differential diagnosis began to narrow after chest radiographs revealed pulmonary lesions and renal function studies showed increasing creatinine levels and overall diminished renal function.
Diagnosis of GPA can be a challenging proposition as there is no specific criteria. The American College of Rheumatology recommends that 2 out of the 4 criteria must be present to diagnose GPA: nasal or oral inflammation, abnormal chest radiograph, urinary sediment with RBCs or casts, or granulomatous inflammation on biopsy. 5 Examples of histologic features seen in GPA include the presence of histologic features such as positive serologic testing like c-ANCA and PR3 are highly specific for GPA in its active phase, but are not essential or required to make the diagnosis. 6 ANCA activity is an important test for patients with GPA. ANCA serves to help with diagnosis and to act as a surrogate marker for disease activity. For this patient, his clinical response to maintenance therapy is evident with the resolution of his cutaneous lesions. However, rheumatology has followed the patient’s serologic response with ANCA panels every 4 months, which has shown a dramatic decline in c-ANCA.
Histologically, GPA presents a challenge for diagnosis relying on biopsies. Biopsies of head and neck specimens typically result in nonspecific findings consistent with necrotizing granulomatous inflammation.3,4,6 In the case of this patient, diagnosis was delayed due to insufficient tissue sampling. However, once adequate tissue sampling was obtained through a punch biopsy, there was clear evidence that the patient also displayed similar nonspecific histologic findings (Figure 2). The diagnosis was determined based on the entirety of the clinical picture presented by the patient: multisystem involvement, positive serologic markers, lab workup, and radiographic evidence.
Treatment of GPA focuses on addressing the autoimmunologic processes driving the granulomatous multisystem inflammation. Therapy for this disease has been induction with glucocorticoids and cyclophosphamide or methotrexate, but the advent of monoclonal antibodies and biologics have introduced effective alternatives such as Rituximab. 5 The patient presented was started on induction therapy with Rituximab and a steroid, before being transitioned to maintenance therapy.
Conclusion
GPA can be a fatal condition if not treated in a timely manner. Without treatment, mortality is due to necrotizing glomerulonephritis. 7 The patient’s condition was initially challenging to diagnose due to nonspecific histologic findings and insufficient tissue sampling. However, a comprehensive evaluation, including autoimmune lab work and radiographic evidence, led to the diagnosis of GPA. This case underscores the importance of considering GPA in the differential diagnosis of necrotic skin lesions and is an example of a rare presentation of GPA manifesting as cutaneous pyoderma gangrenosum-like lesions involving the bilateral parotid glands.
Footnotes
Ethical Considerations
This case report was conducted in accordance with Lehigh Valley Health Network IRB procedures and protocols.
Consent to Participate
Written informed consent was obtained as part of this case report.
Author Contributions
Spencer H. Short: primary author, literature review, and editor. Michael McGlone: secondary author and literature review. Jason F. Ohlstein: principal investigator.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
There are no data associated with this case report.
Trial Registration
Trial registration was not required for this case report.
