Abstract
Palatal perforation, a diagnostically complex condition, arises from diverse etiologies including neoplasms, infections, or inflammatory disorders. Patients often experience dysphagia, dysarthria, and social distress due to nasal regurgitation. We report a 22 year-old cachectic woman with isolated palatal perforation secondary to diffuse large B-cell lymphoma (DLBCL). She presented with a 2-month history of a painless, progressive midline ulcer unresponsive to antibiotics, culminating in a 2 cm perforation. Biopsies revealed necrotic tissue, fungal elements, and atypical lymphocytes; immunohistochemistry confirmed DLBCL (CD20+, CD3−). Imaging and labs excluded systemic involvement, infections (HIV, syphilis), and autoimmune conditions. Despite lacking classic B-symptoms, elevated lactate dehydrogenase (392 U/L) and iron-deficiency anemia suggested malignancy. This case highlights the diagnostic challenge of DLBCL mimicking benign lesions and underscores the need for multidisciplinary evaluation integrating histopathology and immunohistochemistry. Early diagnosis is critical, as R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone) chemotherapy can improve outcomes. Clinicians should consider lymphoma in persistent palatal lesions, even without systemic signs. This report emphasizes the role of timely, targeted therapy in otolaryngologic manifestations of systemic malignancies.
Keywords
Introduction
Palatal perforation is a diagnostic challenge arising from chronic necrosis of the soft or hard palate, creating an abnormal communication between the oral and nasal cavities. 1 Prolonged palatal ulcers may progress to perforation, a rare occurrence in adults, with etiologies spanning neoplasms (eg, lymphoma, squamous cell carcinoma), inflammatory conditions like granulomatosis with polyangiitis, infections (syphilis, tuberculosis, fungal osteomyelitis), or ischemia from cocaine use.2,3 Less common causes include autoimmune disorders and radiation therapy. 4 Clinically, patients may experience oral dysfunction, including dysphagia, dysarthria, regurgitation of fluids, and nasal regurgitation, which profoundly impact quality of life. 5 Additionally, chronic nasal speech and recurrent infections due to food or liquid entering the nasal cavity can lead to social embarrassment and psychological distress. 6 In cases of suspected malignancy, immunohistochemistry, and molecular studies are critical. For instance, diffuse large B-cell lymphoma (DLBCL), the most common non-Hodgkin lymphoma (NHL) subtype, may present atypically in the palate, mimicking benign ulcers or infections, thereby delaying diagnosis. 7 We present a case of a young, cachectic patient with isolated palatal perforation ultimately attributed to DLBCL, highlighting the diagnostic complexity. Isolated extranodal DLBCL of the palate is extremely rare and may lack systemic symptoms like B-symptoms, further complicating early detection. 8
Case Presentation
A 22 year-old Syrian woman presented to the ENT clinic with a 2-month history of a painless, progressively enlarging palatal ulcer resulting in a midline perforation (2 cm diameter) involving the anterior soft palate and extending into the hard palate, accompanied by nasal regurgitation, halitosis, and a nasal escape. Despite multiple empiric antibiotic courses, the lesion persisted.
Her medical history was unremarkable, with no allergies, substance use (tobacco, alcohol, or illicit drugs), or prior trauma/surgery. Physical examination revealed cachexia (BMI 12.45 kg/m2), which she attributed to lifelong constitutional thinness, and intranasal findings were normal. Intraoral examination confirmed the perforation (Figure 1) without lymphadenopathy. Three sequential biopsies demonstrated necrotic tissue with neutrophilic infiltrates, fungal elements, and finally atypical lymphocytic infiltrates with necrosis, consistent with NHL (Figure 2). Immunohistochemistry confirmed DLBCL (CD20+, CD3−; Figure 3).
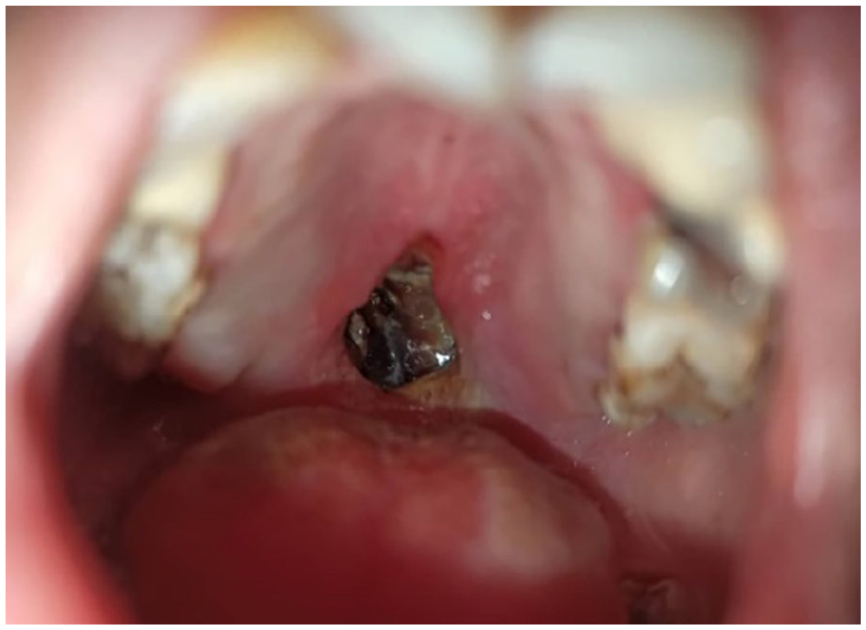
Midline 2 cm palatal perforation involving the anterior soft palate and extending into the hard palate.
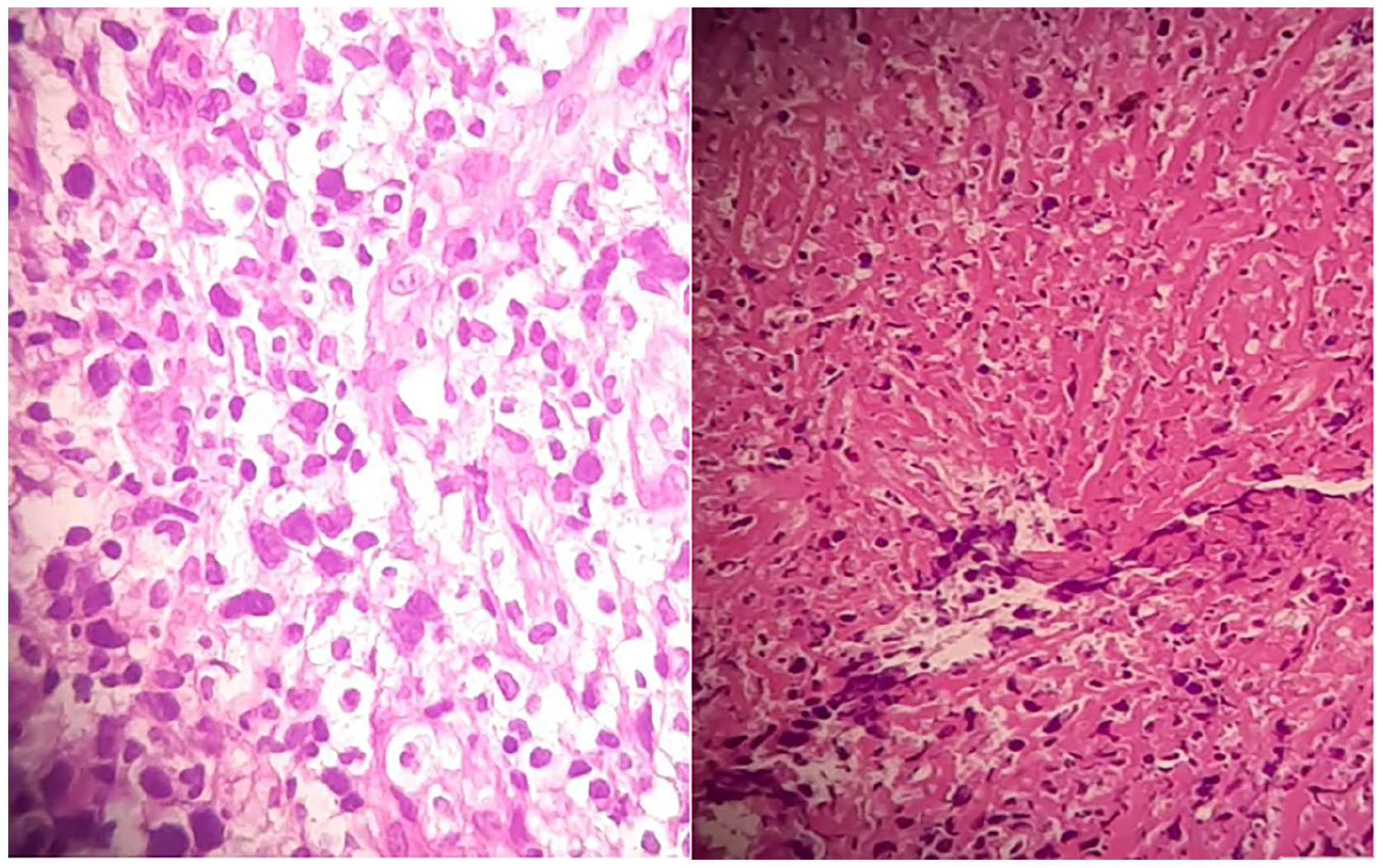
Histopathology (H&E stain) demonstrating atypical lymphocytic infiltrate with geographic necrosis, consistent with DLBCL, a subtype of non-Hodgkin lymphoma. DLBCL, diffuse large B-cell lymphoma.
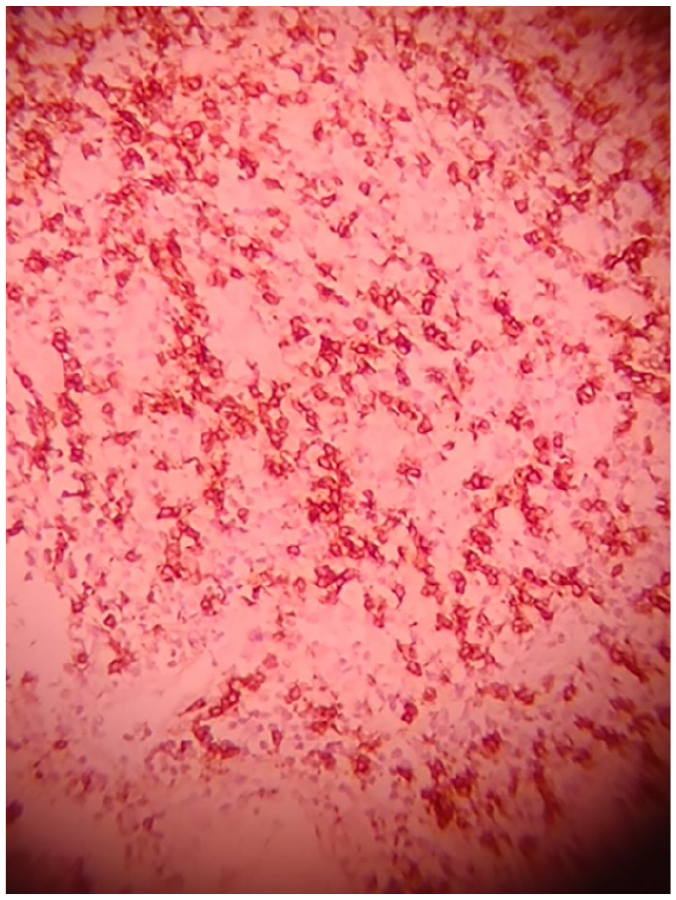
Immunohistochemistry (20× magnification) showing strong CD20 positivity (brown staining) in neoplastic lymphocytes, confirming B-cell origin. CD3 (not shown) was negative, excluding T-cell lineage.
Contrast-enhanced Computed tomography (CT) imaging of the neck, chest, abdomen, and pelvis revealed nonspecific findings. Laboratory studies showed elevated lactate dehydrogenase (392 U/L), iron-deficiency anemia (recurrent low hemoglobin, magnesium, potassium), and normal renal/hepatic function (urea 23 mg/dL, creatinine 0.8 mg/dL, AST 16 U/L, ALT 12 U/L, ALP 68 U/L). Infectious and autoimmune workups were negative (HIV-negative, normal anti-tTG IgA). Gastrointestinal evaluation, including duodenal biopsy, ruled out malabsorption. Following hematologic confirmation of DLBCL, she was transferred to oncology for chemotherapy [eg, R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone) regimen], with palatal rehabilitation deferred until disease control.
Discussion
Isolated palatal perforation is a rare clinical entity with diverse etiologies, including trauma, chronic infections, autoimmune disorders, and granulomatous diseases.1-4 In our patient, no history of significant trauma or infection was identified, prompting further investigation. Lymphadenopathy, a hallmark of lymphoma, 9 is uncommon in oral presentations; however, lymphoma must remain a key differential diagnosis. Although rare, oral lymphoma can manifest as palatal masses, ulcerations, or perforations, often mimicking benign lesions. 5 While most cases are part of disseminated disease, primary extranodal lymphoma confined to the oral cavity or jaws is exceptionally rare. 5 Large B-cell lymphoma of the oral cavity typically presents with painless swelling, nonhealing ulcers, and systemic B-symptoms (fever, night sweats, weight loss).5-10 Submucosal lesions at the hard-soft palate junction are particularly suggestive.5-10 However, nonspecific symptoms can delay diagnosis, necessitating a high clinical suspicion. 10 In our case, histopathology revealed chronic inflammation and ulceration, 11 but atypical lymphoid infiltrates prompted immunophenotyping, confirming DLBCL.
Extranodal lymphomas of the head and neck are predominantly B-cell derived, yet natural killer/T-cell lymphoma, particularly the extranodal subtype (ENKTL), is the most frequently reported palatal variant. 12 ENKTL, historically termed “midline lethal granuloma,” is strongly associated with Epstein–Barr virus (EBV).13-15 Our patient’s diagnosis of DLBCL—rather than ENKTL—highlights the importance of immunohistochemical differentiation (eg, CD20+ for DLBCL vs EBV markers for ENKTL) to guide management. 7 Roche et al 9 reported a 28 year-old male smoker with a 7 month history of a painful, exophytic palatal ulcer, B-symptoms, and hypermetabolic FDG uptake Standardized Uptake Value (SUVmax >42.8), consistent with aggressive DLBCL (Ki-67: 90%). In contrast, our 22 year-old cachectic female (BMI 12.45 kg/m2) presented with a painless 2 month perforation, no B-symptoms, and unremarkable imaging. Initial biopsies suggested fungal infection, delaying diagnosis until immunohistochemistry confirmed DLBCL (CD20+, CD3−). Both cases received R-CHOP chemotherapy: Roche et al 9 achieved remission, while ours awaits long-term follow-up. These contrasting presentations—exophytic versus perforative lesions, systemic versus localized symptoms—illustrate DLBCL’s clinical diversity. While Roche et al 9 emphasize PET–CT’s role in staging aggressive disease, our case underscores the diagnostic pitfall of mimicking infections in immunocompetent patients.
DLBCL management requires multidisciplinary collaboration among hematologists, oncologists, and surgeons.16,12 First-line therapy involves R-CHOP. 17 Early-stage disease may require 3 cycles of CHOP ± radiotherapy, whereas refractory cases warrant bone marrow transplantation.17,18 Advanced stages often incorporate agents such as methotrexate, etoposide, or cytarabine. 18 Surgery plays a minimal role, and vigilant follow-up is critical, as >50% of patients relapse. 18
Conclusion
Palatal perforations pose diagnostic challenges, particularly when linked to malignancies like lymphoma. Absence of classic symptoms (eg, B-symptoms) and mimicry of infections demand histopathology and immunophenotyping for definitive diagnosis, as seen in our DLBCL case. Multidisciplinary collaboration (otolaryngology, oncology, pathology) is vital for timely intervention. While R-CHOP chemotherapy remains primary, surgical measures may address perforations. Diagnostic delays, exemplified by our patient’s initial misdiagnosis, underscore the need for clinical vigilance. Standardized protocols and expanded case reporting are crucial to optimize management and outcomes in this rare yet clinically significant presentation.
Footnotes
Acknowledgements
The authors are grateful for their colleges in Al-Mouwasat hospital in the ENT–HNS department.
Ethical Considerations
Our institution does not require ethics approval for reporting individual cases.
Consent for Publication
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
