Abstract
A 36-year-old male with a history of smoking presented with recurrent right submandibular swelling. Initially diagnosed with pleomorphic adenoma, he underwent total submandibulectomy. After 2 years, he developed a recurrent submandibular mass. Clinical and imaging assessments revealed a 6.2 cm multinodular tumor without invasion of adjacent structures. Surgery under general anesthesia involved tumor resection and lymph node dissection. Pathology confirmed carcinoma ex pleomorphic adenoma, specifically epithelial-myoepithelial carcinoma (EMC). Despite focal necrosis and a tumor size of 10.8 cm, margins were clear, and no lymph node metastasis was detected (pT3N0). Postoperative imaging showed no recurrence or metastasis, and the patient remains disease-free at the 25th month. This case highlights the diagnostic and therapeutic complexities of EMC, a rare malignancy constituting <1% of salivary gland tumors. Multidisciplinary collaboration enabled accurate diagnosis and effective treatment. Early intervention in recurrent pleomorphic adenomas is crucial to mitigate malignant transformation risk. Long-term surveillance is essential due to EMC’s potential for late recurrence.
Introduction
Salivary gland neoplasms represent a heterogeneous group of tumors, accounting for 3%-4% of all head and neck malignancies, with the submandibular gland (SMG) involved in only 8%-22% of cases. 1 Unlike the parotid gland, SMG enlargement is more frequently attributed to nonneoplastic conditions such as sialolithiasis or inflammatory processes. Primary SMG tumors, however, pose unique diagnostic and therapeutic challenges due to their anatomic complexity and histologic diversity. Among these, pleomorphic adenoma (PA)—the most common benign SMG neoplasm—carries a well-documented risk of malignant transformation into carcinoma ex pleomorphic adenoma (CXPA), a rare but aggressive entity constituting 3%-5% of salivary malignancies.2,3
Epithelial-myoepithelial carcinoma (EMC), a rare biphasic tumor first described in 1972, accounts for <1% of salivary gland neoplasms and predominantly arises in the parotid gland. Its occurrence in the SMG is exceedingly rare, with fewer than 50 cases reported globally.1,4 EMC’s indolent clinical presentation often mimics benign lesions, yet its infiltrative growth pattern and propensity for local recurrence demand aggressive management. CXPA, meanwhile, typically manifests in the sixth to seventh decade, with malignant transformation linked to long-standing PA. Younger patients, such as the 36-year-old male presented here, represent atypical demographic outliers, underscoring the need for heightened clinical suspicion in recurrent or rapidly-enlarging SMG masses.
This report details a rare case of SMG-derived EMC arising from CXPA in a young male with a history of recurrent PA. We emphasize the diagnostic pitfalls, therapeutic challenges, and prognostic nuances of this dual pathology, while highlighting the critical role of multidisciplinary collaboration in optimizing outcomes. By contextualizing this case within the existing literature, we aimed to advance the understanding of SMG malignancies and reinforce the importance of lifelong surveillance even in seemingly-low-risk populations.
Case Presentation
A 36-year-old male patient was admitted to our department for the treatment of recurrent right submandibular swelling. He is an active smoker with a 17 pack-year history and reports occasional alcohol consumption.
The patient reported a slowly-enlarging right cervical mass that had been neglected for several years. Consultation with a private otolaryngologist led to a right total submandibulectomy, with histopathologic examination confirming a completely-excised pleomorphic adenoma of the right SMG. Later, the patient presented to our ENT department with a recurrent mass in the submandibular region, which had developed over the preceding months. On clinical examination, there was a 6 cm right submandibular mass, well defined, non-tender, and fixed in the superficial planes, without overlying inflammatory signs (Figure 1a and b). The contralateral submandibular region and bilateral parotid glands were free of palpable abnormalities. There was no palpable cervical lymphadenopathy. Physical examination revealed no labial asymmetry. The floor of the mouth was soft on bidigital palpation, and the saliva from the Wharton’s duct was clear.
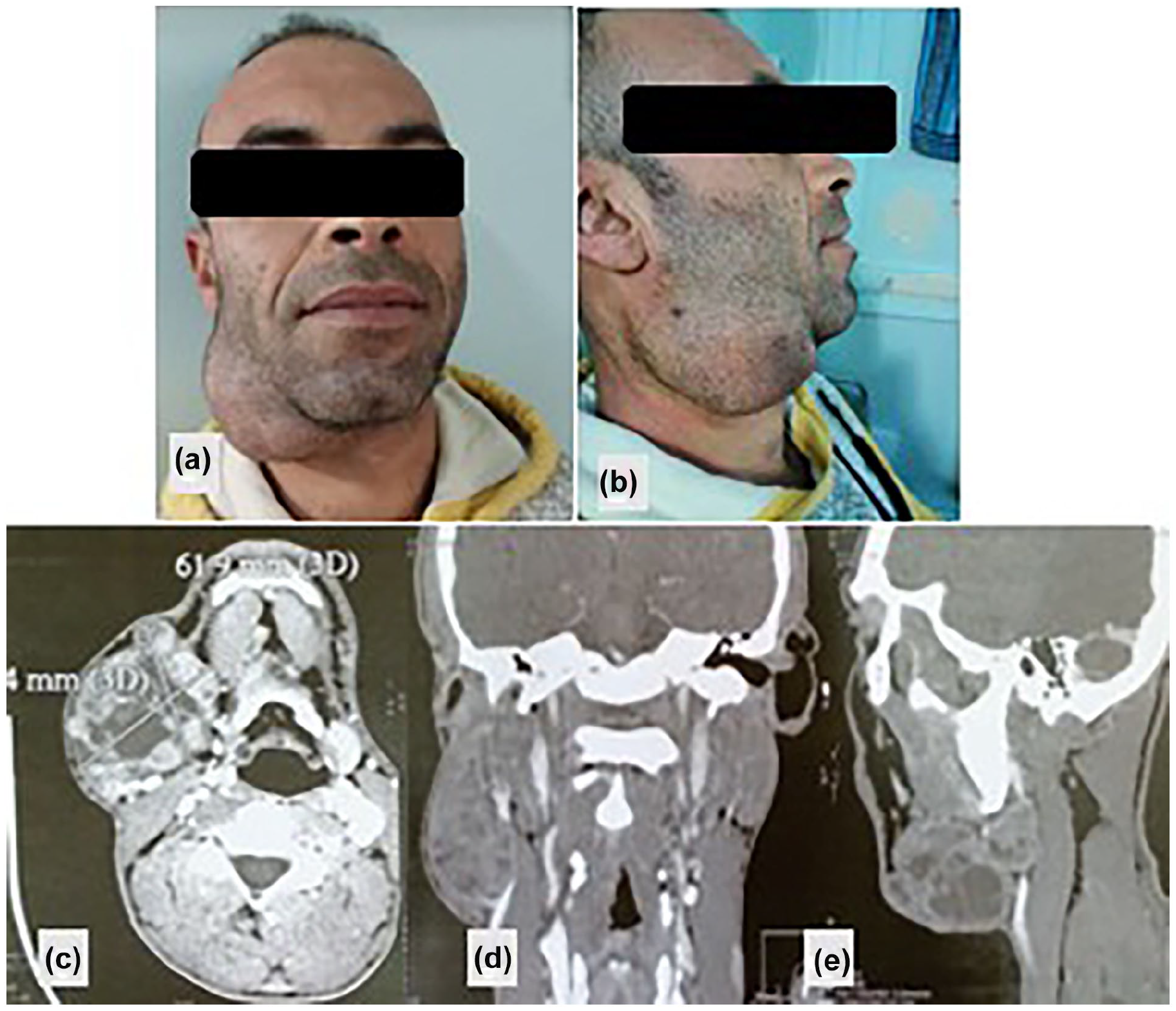
(a and b) Large mass in the right submandibular space adherent to the skin. Cervicofacial CT scan in axial (c), coronal (d), and sagittal (e) reconstruction after contrast injection: demonstrating a multinodular tumor process involving the right submandibular space.
A cervical CT scan performed, which revealed a multinodular tumor mass in the right submandibular space measuring 6.2 × 5.4 × 6 cm. The mass did not infiltrate the floor of the mouth or the mandibular cortex (Figure 1c-e).
The patient underwent surgery under general anesthesia. An elliptical incision was made, incorporating the previous surgical scar and the area of cutaneous adhesion. The submandibular tumor mass was resected, necessitating the sacrifice of the marginal mandibular branch of the facial nerve. During the procedure, the digastric muscle, lingual nerve, and hypoglossal nerve were carefully identified and preserved (Figure 2).
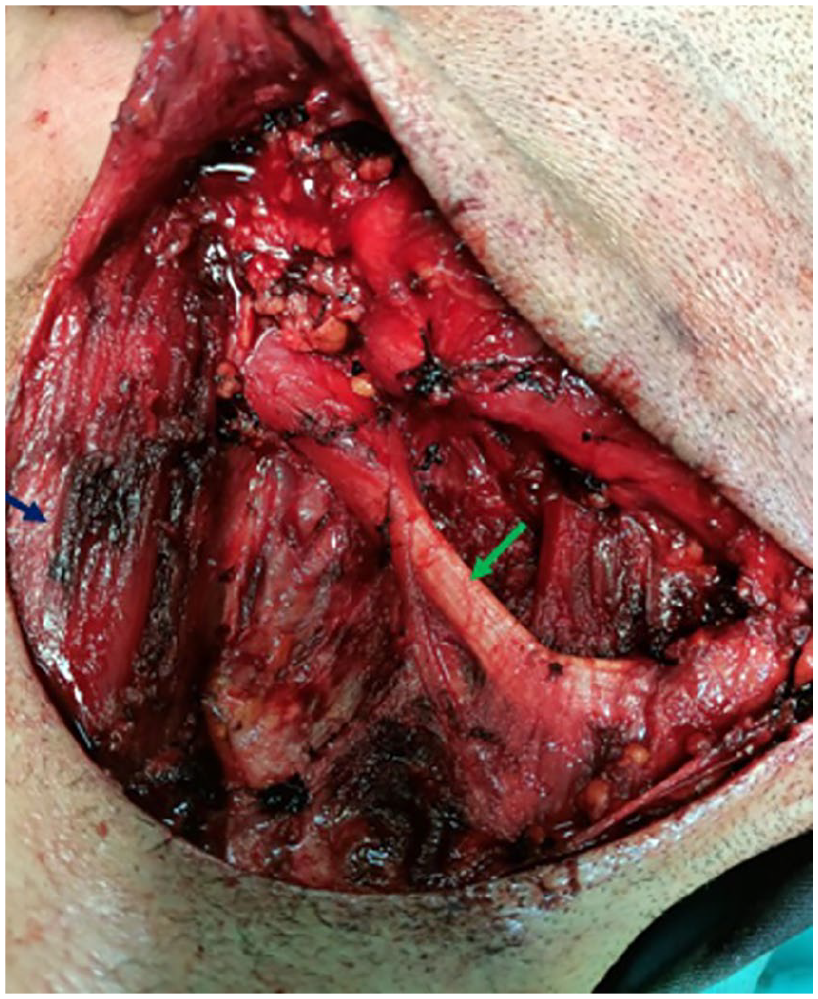
Intraoperative view after the resection of the right submandibular tumor mass: tendon of the digastric muscle (green arrow), sternocleidomastoid muscle (blue arrow).
Subsequently, a lymph node dissection of levels Ib, II, and III was performed. The intraoperative frozen section suggested a diagnosis of CXPA, specifically of the EMC subtype.
The anatomopathological examination with immunohistochemical analysis concluded to a CXPA of the right SMG, which specifically exhibits features of an EMC with a tumor size of 10,8 cm and no evidence of metastasis in the regional lymph nodes (pT3N0) (Figure 3).
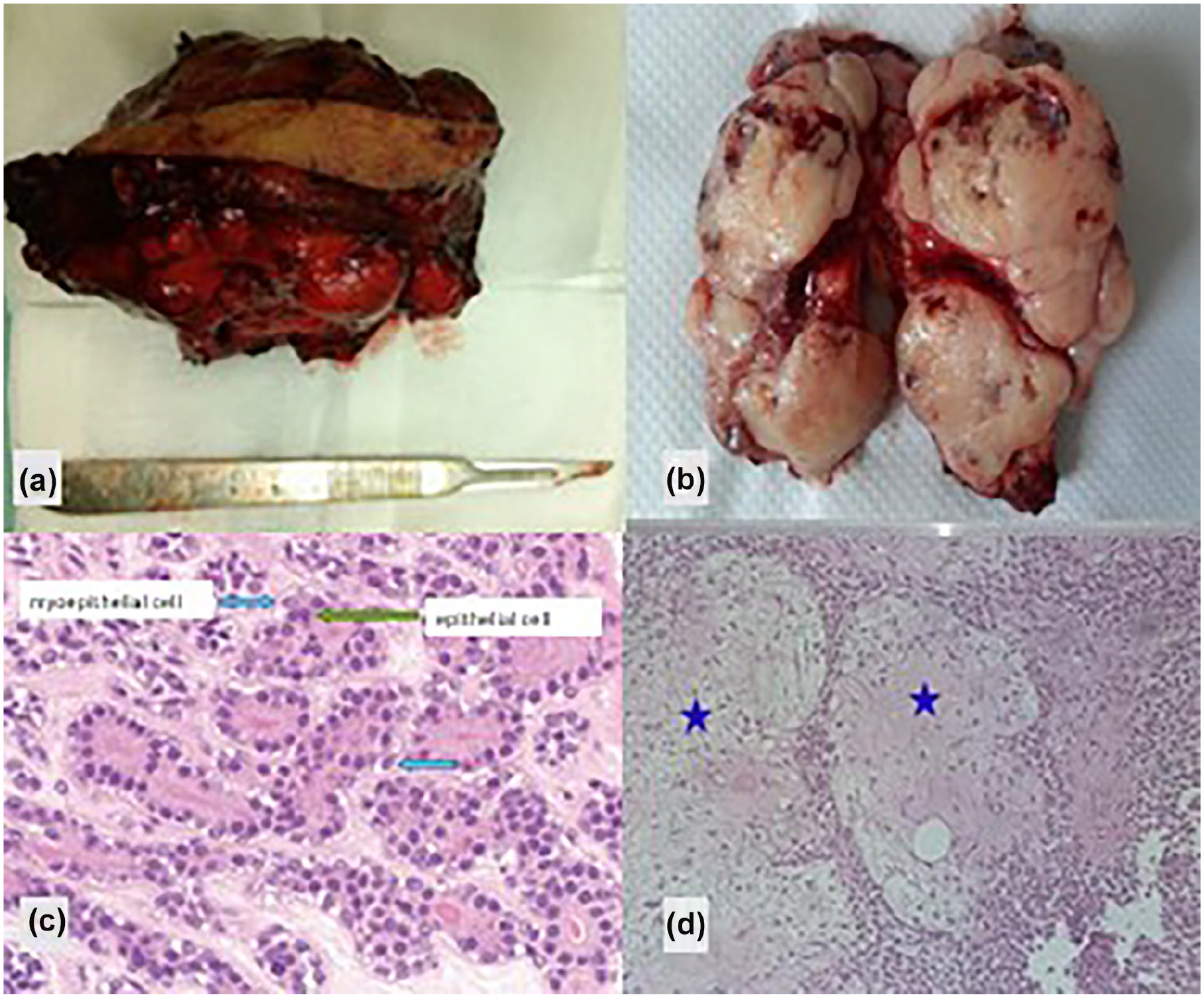
Resected submandibular tumor specimen with the sacrifice of a skin flap (a) : The tumor is multinodular (b), tumor proliferation with a biphasic appearance (c), arranged in tubular structures lined by a luminal base made of epithelial cells with eosinophilic cytoplasms (green arrow) and a basal base, made of myoepithelial cells with clear cytoplasms (blue arrows) (HE × 100). (d) We note the presence, within the tumor, of unequivocal chondro-myxoid foci (blue stars) (HE × 100).
Focal areas of tumor necrosis were present, without tumor involvement at the surgical margins.
A postoperative cervical-thoraco-abdomino-pelvic CT scan performed 2 months after surgery showed no evidence of locoregional recurrence or distant metastasis. At the most recent follow-up, 25 months postoperatively, the patient remains clinically free of disease, indicating good disease control.
Discussion
EMC of the SMG is an exceedingly-rare malignancy, representing <1% of all salivary gland tumors, with most cases originating in the parotid gland. This case of a 36-year-old male with recurrent SMG CXPA transforming into EMC underscores the diagnostic and therapeutic complexities of such tumors, particularly given its atypical demographic presentation and aggressive behavior.2,5
CXPA arises from preexisting pleomorphic adenoma (PA), with a 2–9% risk of malignant transformation over 15 years.2,3 This patient’s untreated, long-standing SMG mass (first noted in 2013 and excised in 2020) exemplifies the critical window for malignant progression. While smoking and alcohol use are not direct etiological factors for EMC, the patient’s 17 pack-year smoking history may have exacerbated genomic instability, potentially accelerating malignant transformation. Notably, CXPA typically manifests in the sixth to seventh decade, making this young patient’s presentation unusual and emphasizing the need for vigilance even in atypical demographics.
EMC’s clinical mimicry of benign lesions often delays diagnosis. In this case, the recurrence of a painless, fixed SMG mass over 11 months raised suspicion for malignancy, although imaging (cervical CT) alone could not confirm it. EMC’s biphasic histology—inner ductal epithelial cells surrounded by clear myoepithelial cells—was confirmed via immunohistochemistry (p63, SMA, CK7 positivity).4,6-8 Despite its deceptively-low-grade cytology, EMC’s infiltrative growth pattern and lack of encapsulation correlate with a 23–50% local recurrence rate, particularly with incomplete excision. The tumor’s rapid growth and fixation to superficial planes necessitated intraoperative frozen section analysis, which guided the extent of resection.
Complete surgical excision with negative margins remains the cornerstone of EMC management.1,5,8 In this case, sacrifice of the marginal mandibular nerve was unavoidable to achieve clear margins, highlighting the delicate balance between oncologic radicality and functional preservation.1,2,4,9 Preservation of the lingual and hypoglossal nerves minimized postoperative morbidity, emphasizing the importance of meticulous dissection in head and neck surgery.1,10,11 Adjuvant radiotherapy, often debated for EMC, was deferred here due to favorable prognostic features (pT3N0, clear margins, no angiolymphatic invasion).
The tumor’s large size (10.8 cm) and focal necrosis contrasted with its indolent immunohistochemical profile. The absence of perineural/vascular invasion and lymph node involvement (pT3N0) aligned with EMC’s typically-favorable prognosis. However, Seethala et al. identify surgical margin status, angiolymphatic invasion, and necrosis as key prognostic determinants, underscoring the need for rigorous histopathologic evaluation. 7
At the 25th month postoperatively, the patient remains disease-free, consistent with EMC’s indolent course when treated aggressively. However, late recurrence (up to 40% of cases) and rare distant metastasis necessitate lifelong surveillance. This case reinforces the importance of serial imaging and clinical monitoring, even in seemingly-low-risk scenarios.
Conclusion
This rare case of salivary myoepithelial carcinoma (EMC) arising from CXPA highlights the complexities in diagnosing and managing salivary gland malignancies in atypical populations. Early intervention in recurrent pleomorphic adenoma is crucial to reduce the risk of malignant transformation. Accurate diagnosis and staging rely on multidisciplinary collaboration, involving radiology, pathology, and surgery. Surgical planning should be tailored to achieve optimal oncologic control while preserving function, and long-term surveillance is essential to detect late recurrence or metastasis. Further research into the molecular drivers of CXPA and EMC could enhance risk stratification and guide adjuvant therapy protocols, particularly for young patients with aggressive disease presentations.
Footnotes
Acknowledgements
None.
Data Availability Statement
Data will be made available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution Faculty of Medicine of Sousse’s Ethics Committee does not require ethics approval for reporting individual cases or case series since we anonymously reported clinical and imaging information concerning our patient’s case.
Statement of Informed Consent
A written informed consent was obtained from the patient for his anonymized information to be published in this article.
